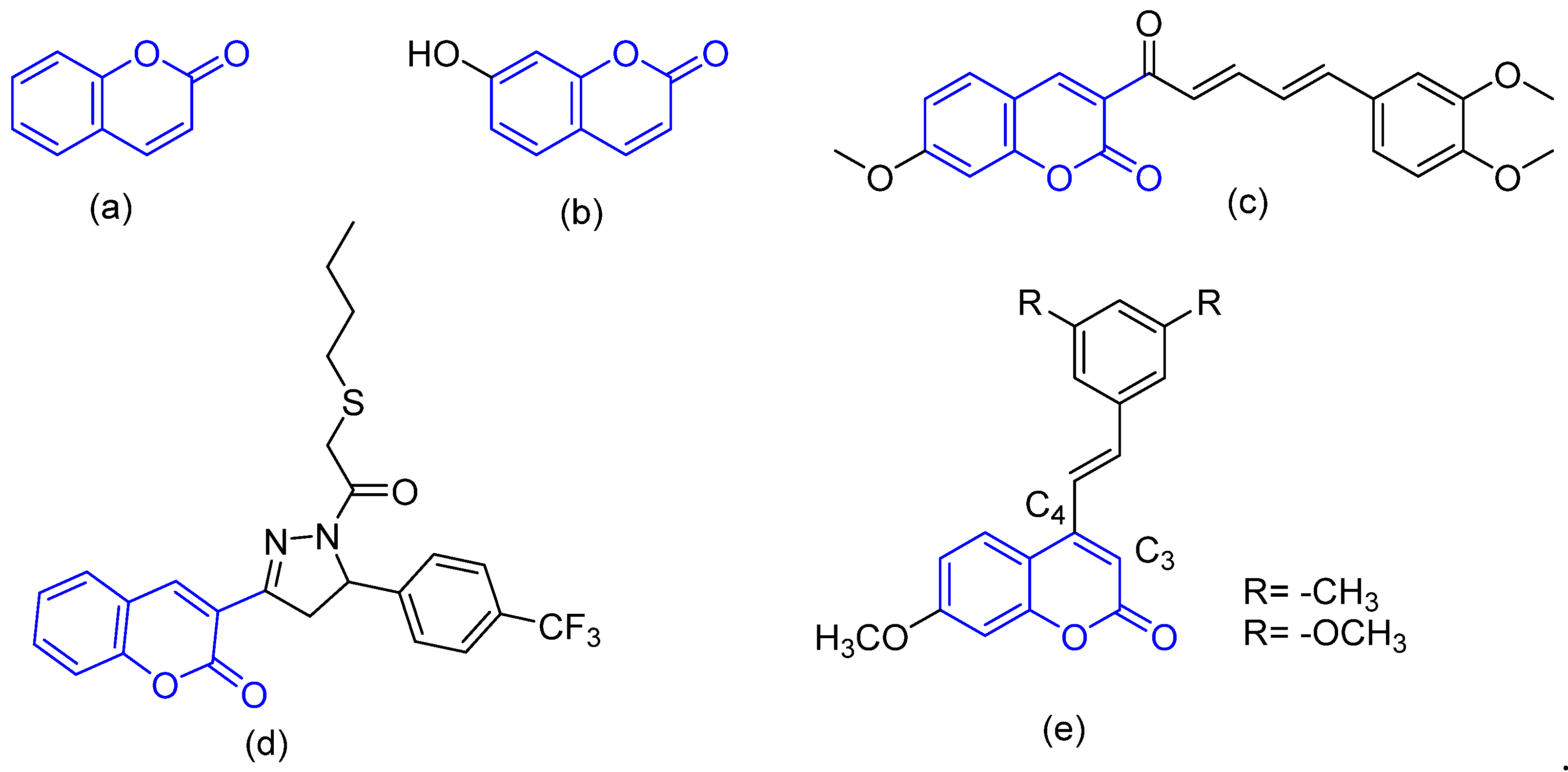
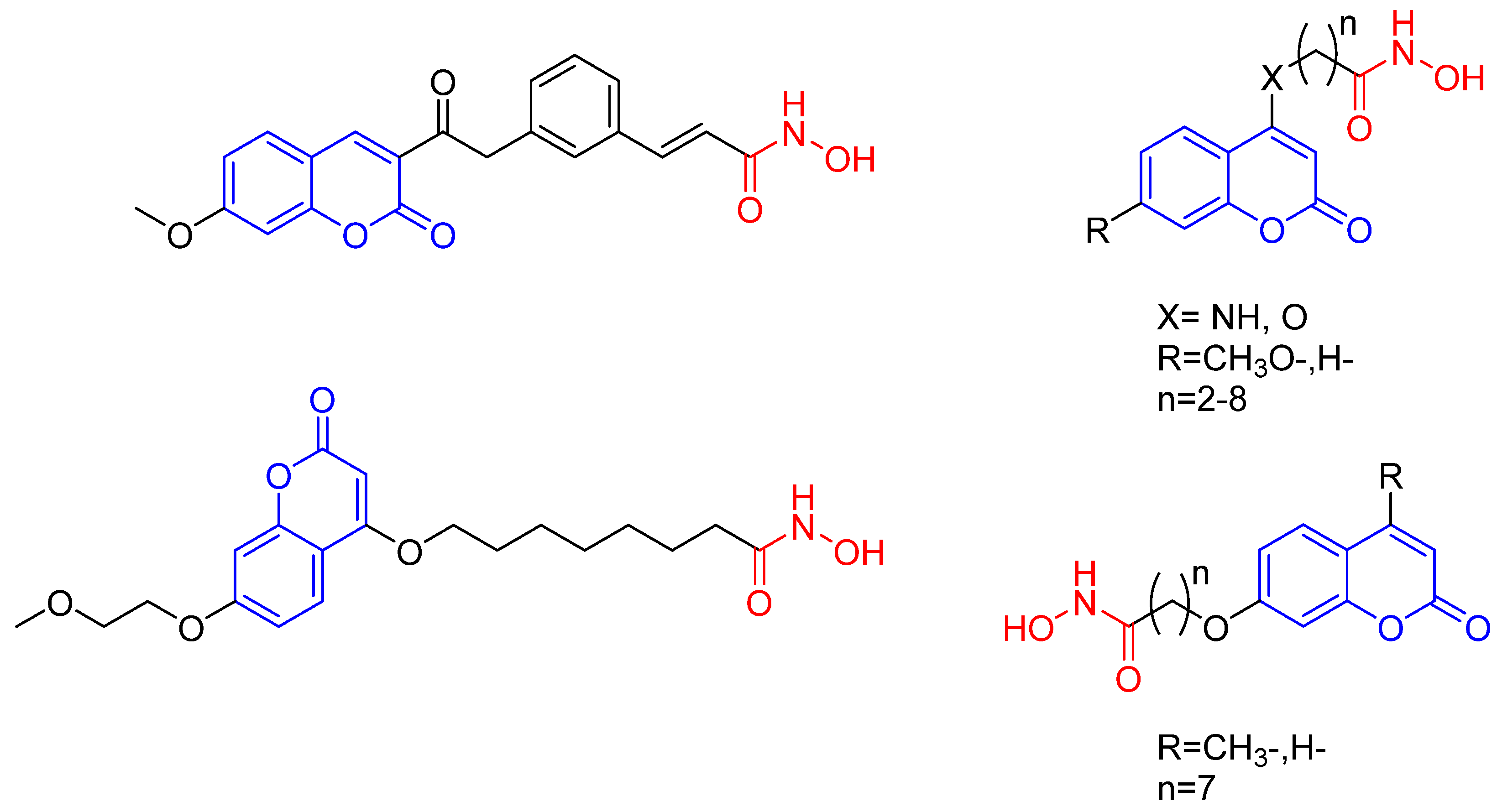
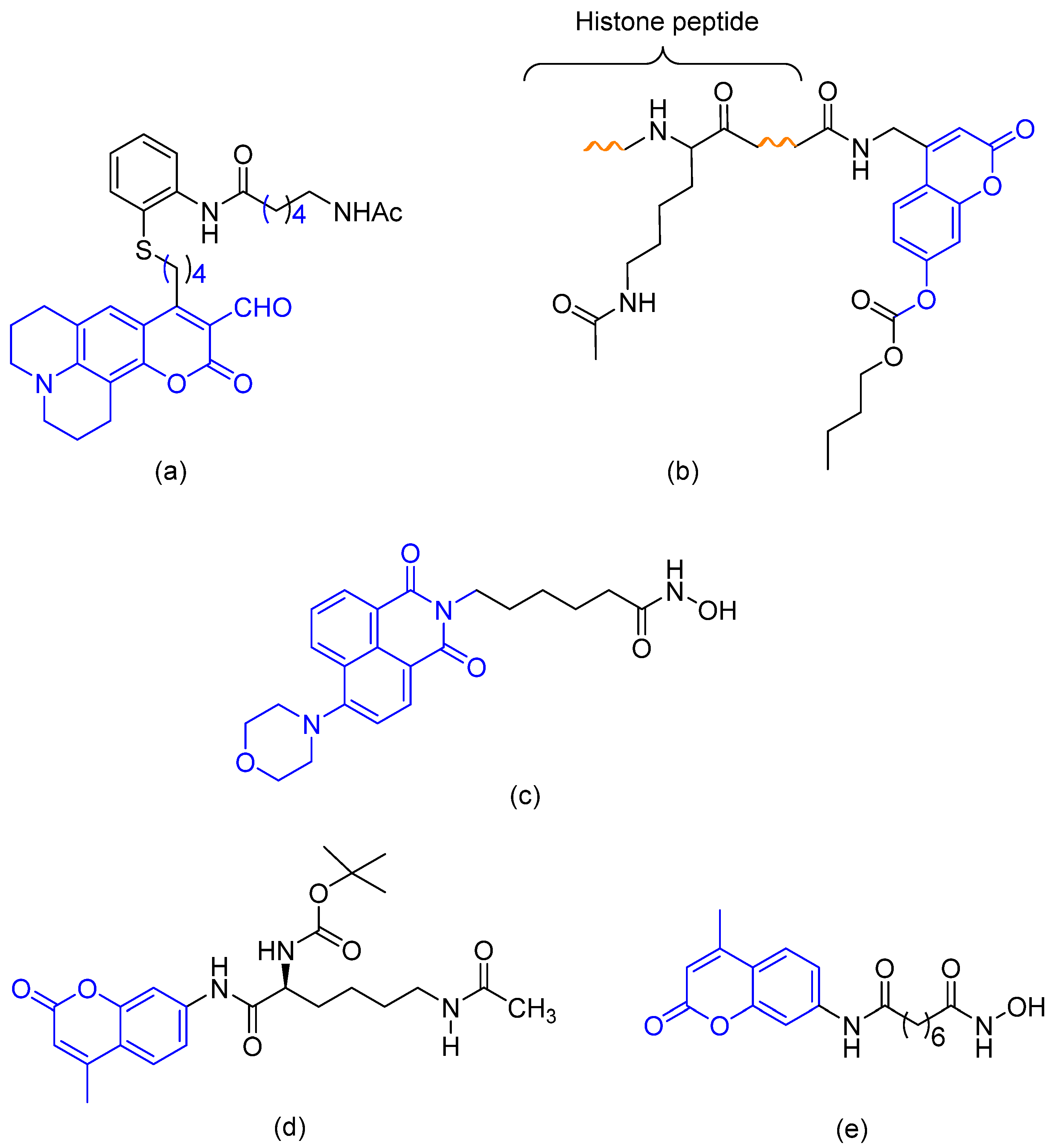
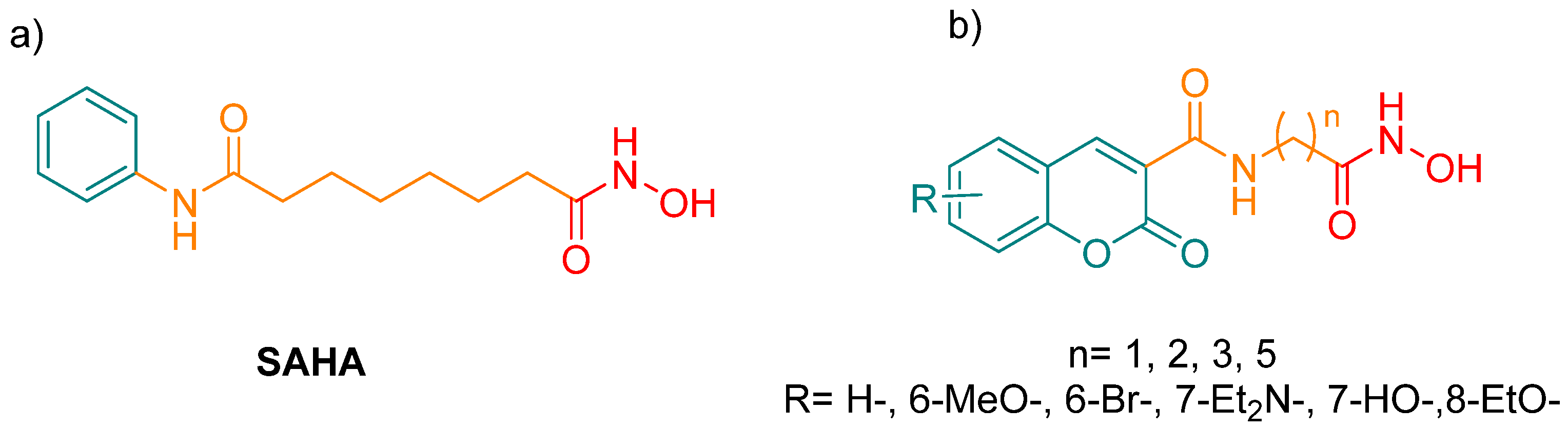
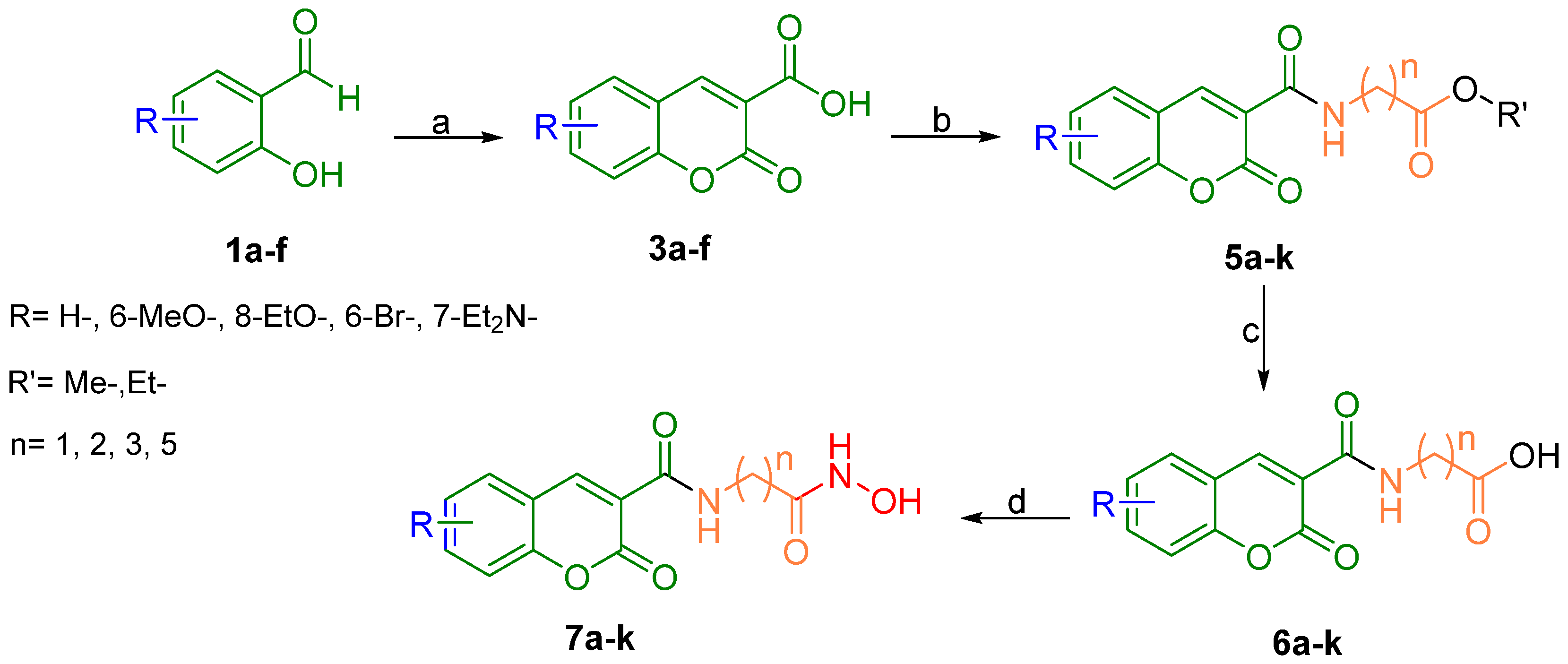
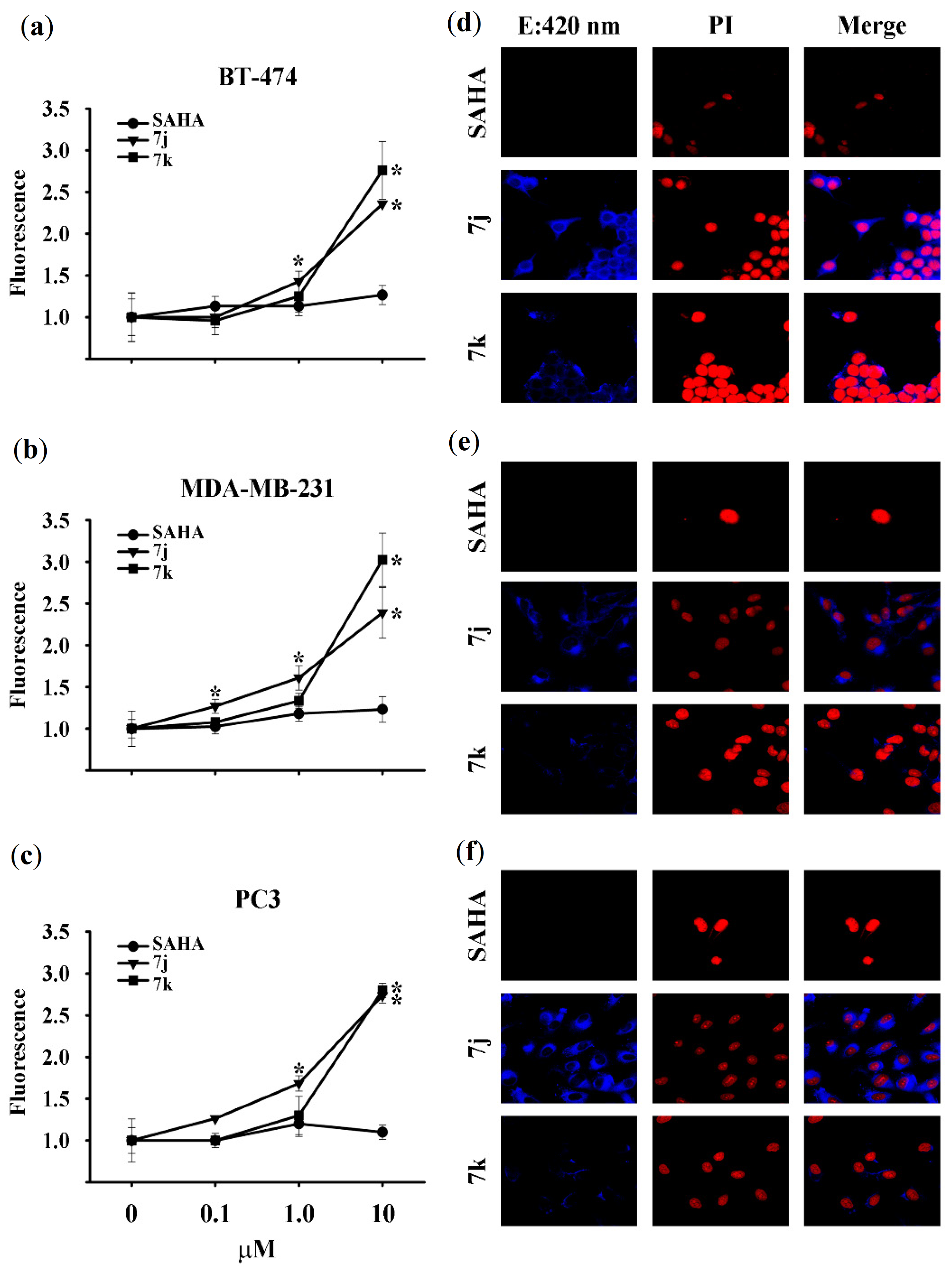
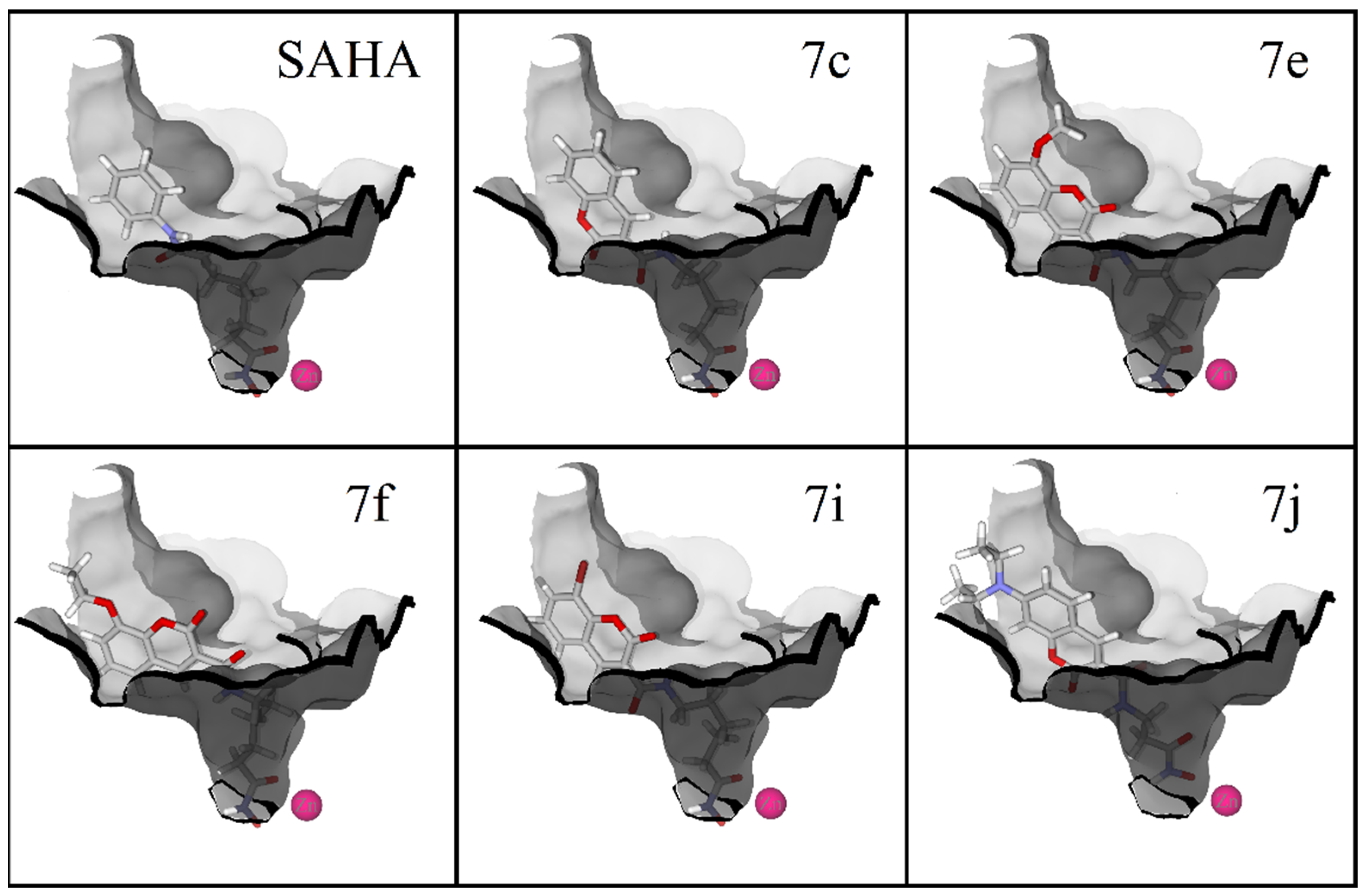
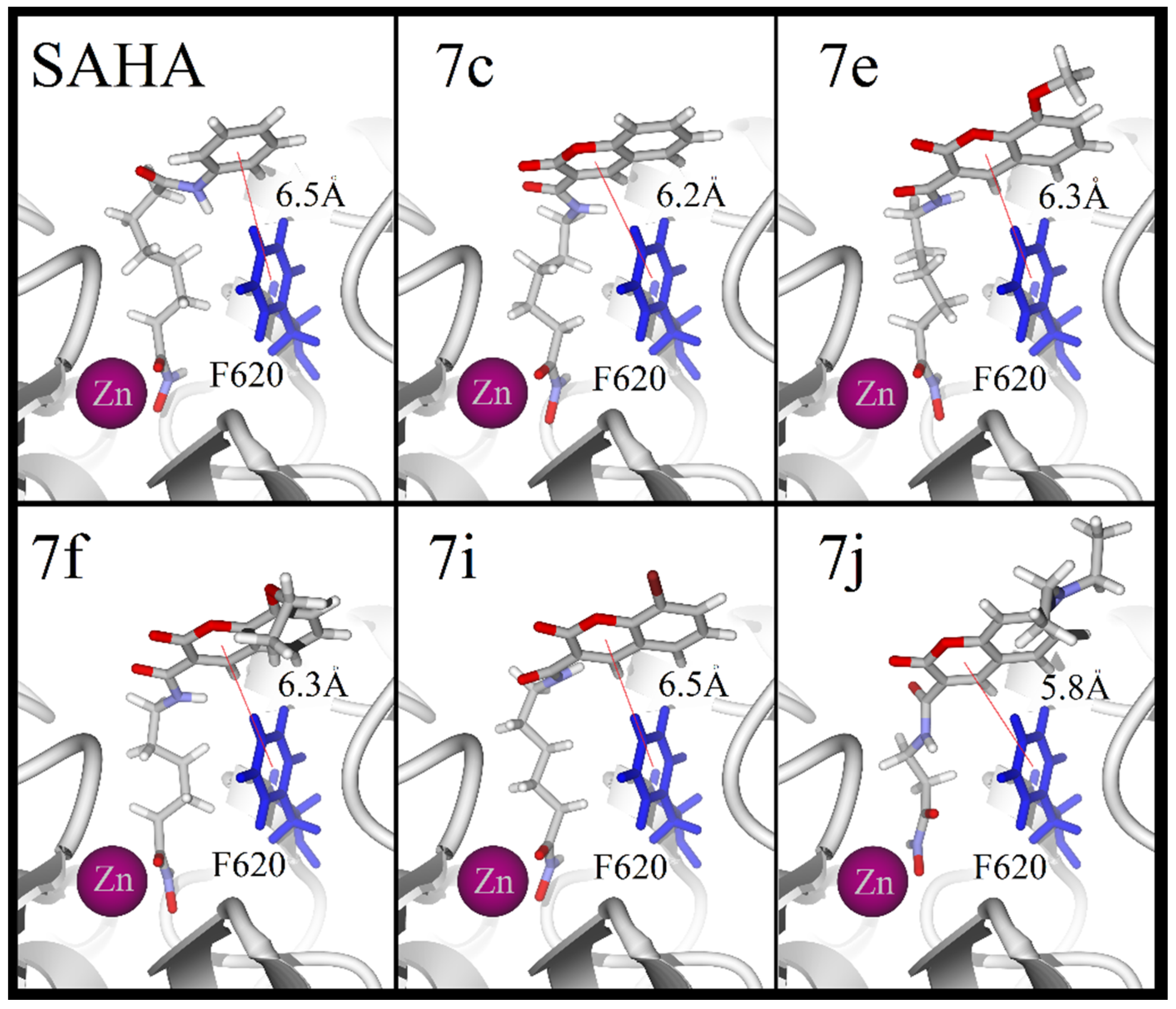
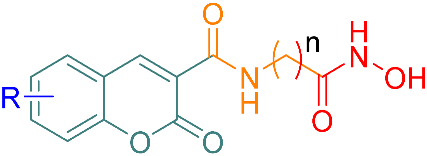
3.1. Chemistry
All solvents and reagents were commercially available and used without further purification. Melting points were determined on a RY-1 MP apparatus. Absorption spectra were recorded on a Perkin-Elmer Lambda 50 apparatus (Perkin-Elmer Inc., Waltham, MA, USA), and Emission spectra on a FL-7000 FL spectrometer (Hitachi Inc., Chiyoda, Tokio, Japon). ESI-HRMS spectra were obtained on a Maxis Impact ESI-QTOF-MS spectrometer (Bruker Corporation 40 Manning Road Billerica, MA 01821, USA) and a Bruker Daltonics mass spectrometer (Bruker Corporation 40 Manning Road Billerica, MA 01821, USA). 1H-NMR and 13C-NMR spectra were acquired in CDCl3 or DMSO-d6 solutions with the following NMR spectrometers: a Bruker Avance III HD NMR spectrometer [Bruker Corporation 40 Manning Road Billerica, MA 01821, USA] with a Bruker Ascent 400 MHz magnet or a Bruker Avance III HD with a Bruker Ultra Shield 500 MHz HD magnet. Spectra were recorded at 25 °C with TMS and solvent signals allotted as internal standards. Chemical shifts are expressed in ppm (δ) and J-values in hertz.
3.1.1. General Procedure for the Preparation of Coumarin-3-Carboxylic Acids (3a–f)
In a round-bottom flask, a solution of the corresponding salicylaldehyde (1 mmol) and Meldrum’s acid (1.2 mmol) was stirred in water at reflux for 5–6 h. The precipitate was obtained by filtration. The purification was carried out by recrystallization with MeOH and cold water [
42,
43].
2-oxo-2H-chromene-3-carboxylic acid (3a), white solid (82% yield); mp = 183–184 °C; IR (KBr): νmax/cm−1 = 1746, 1685, 1614, 1569; UV-Vis (MeOH): λmax/nm = 296; 1H-NMR (500 MHz, CDCl3): δ 12.26 (s, 1H, H-3a), 8.96 (s, 1H, H-4), 7.80 (dd, J = 14, 7.7 Hz, 2H, H-5,8), 7.51 (t, J = 8.9 Hz, 2H, H-6,7); 13C-NMR (125 MHz, CDCl3): δ 164.10, 162.42, 154.59, 151.52, 135.79, 130.52, 126.28, 118.48, 117.23, 114.90; HRMS (ESI) m/z calcd. for C10H6O4: [M + H]+ 191.0339, found 191.0342.
6-methoxy-2-oxo-2H-chromene-3-carboxylic acid (3b), yellowish solid (90% yield); mp = 202–205 °C; IR (KBr): νmax/cm−1 = 1761, 1607, 1622, 1574; UV-Vis (MeOH): λmax/nm = 295; 1H-NMR (500 MHz, CDCl3): δ 12.37 (s, 1H, H-3a), 8.91 (s, 1H, H-4), 7.43 (d, J= 9.1 Hz, 1H, H-8), 7.36 (dd, J = 9.1, 2.8 Hz, 1H, H-7), 7.12 (d, J = 2.8 Hz, 1H, H-5), 3.91 (s, 2H, H-6a); 13C-NMR (125 MHz, CDCl3): δ 164.22, 162.55, 157.25, 151.22, 149.22, 124.44, 118.90, 118.34, 115.00, 111.01, 56.05; HRMS (ESI) m/z calcd. for C11H8O5: [M + H]+ 221.0444, found 221.0446.
6-bromo-2-oxo-2H-chromene-3-carboxylic acid (3c), white solid (85% yield); mp = 195–197 °C; IR (KBr): νmax/cm−1 = 1737, 1716, 1690, 1609; UV-Vis (MeOH): λmax/nm = 291; 1H-NMR (500 MHz, CDCl3): δ 12.06 (s, 1H, H-3a), 8.87 (s, 1H, H-4), 7.90 (d, J = 2.1 Hz, 1H, H-8), 7.87 (dd, J = 8.8, 2.2 Hz, 1H, H-7), 7.40 (d, J = 8.8 Hz, 1H, H-5); 13C-NMR (125 MHz, CDCl3): δ 163.41, 161.82, 153.33, 150.09, 138.42, 132.45, 119.82, 118.97, 116.01; HRMS (ESI) m/z calcd. for C10H5BrO4: [M + H]+ 268.9444, found 270.9428.
8-ethoxy-2-oxo-2H-chromene-3-carboxylic acid (3d), greenish solid (80% yield); mp = 186–188 °C; IR (KBr): νmax/cm−1 = 1756, 1674; UV-Vis (MeOH): λmax/nm = 309; 1H-NMR (500 MHz, CDCl3): δ 12.32 (s, 1H, H-3a), 8.91 (s, 1H, H-4), 7.38 (t, J = 7.9 Hz, 1H, H-6), 7.30 (d, J = 7.9 Hz, 2H, H-5,7), 4.24 (q, J = 7.0 Hz, 2H, H-8a), 1.54 (t, J = 7.0 Hz, 3H, H-8b); 13C-NMR (125 MHz, CDCl3): δ 163.89, 162.53, 151.78, 146.81, 144.31, 126.16, 121.23, 119.19, 118.17, 114.90, 65.32, 14.64; HRMS (ESI) m/z calcd. for C12H10O5: [M + H]+ 235.0601, found 235.0604.
7-(diethylamino)-2-oxo-2H-chromene-3-carboxylic acid (3e), white solid (85% yield); mp = 195–197 °C; IR (KBr): νmax/cm−1 = 1737, 1716, 1690, 1609; UV-Vis (MeOH): λmax/nm= 291; 1H-NMR (500 MHz, CDCl3): δ 12.06 (s, 1H, H-3a), 8.87 (s, 1H, H-4), 7.90 (d, J = 2.1 Hz, 1H, H-8), 7.87 (dd, J = 8.8, 2.2 Hz, 1H, H-7), 7.40 (d, J= 8.8 Hz, 1H, H-5); 13C-NMR (125 MHz, CDCl3): δ 163.41, 161.82, 153.33, 150.09, 138.42, 132.45, 119.82, 118.97, 116.01; HRMS (ESI) m/z calcd. for C10H5BrO4: [M + H]+ 268.9444, found 270.9428.
7-hydroxy-2-oxo-2H-chromene-3-carboxylic acid (3f), brownish solid (90% yield); mp= 263–264 °C; IR (KBr): νmax/cm−1 = 3126, 1712, 1683, 1619; UV-Vis (MeOH): λmax/nm = 351; 1H-NMR (500 MHz, DMSO-d6): δ 11.87 (s, 1H, H-7a), 8.64 (s, 1H, H-4), 7.70 (d, J = 8.6 Hz, 1H, H-5), 6.82 (dd, J = 8.6, 2.1 Hz, 1H, H-6), 6.70 (d, J = 2.0 Hz, 1H, H-8); 13C-NMR (125 MHz, DMSO-d6): δ 164.66, 164.39, 158.12, 157.42, 149.87, 132.44, 114.46, 112.83, 111.06, 102.24; HRMS (ESI) m/z calcd. for C10H6O5: [M + H]+ 207.0288, found 207.0291.
3.1.2. General Procedure for the Preparation of Coumarin-3-Carboxamides (5a–k)
In a dry round-bottom flask purged with N2, the corresponding coumarin (1 mmol), 4-(dimethylamino)pyridine (DMAP) (5% mol), and carbonyldiimidazole (CDI) (1.1 mmol) were dissolved in DMF. After 30 min of stirring at room temperature, hydrochloride amine salt was added and stirring continued overnight. Water was added when the reaction was completed. The resulting precipitate was obtained by filtration and purified by recrystallization with MeOH and cold water.
Methyl (2-oxo-2H-chromene-3-carbonyl)glycinate (5a), white solid (84% yield); mp = 181–183 °C; IR (KBr): νmax/cm−1 = 3328, 3054, 1749, 1710, 1655; UV-Vis (MeOH): λmax/nm= 300; 1H-NMR (500 MHz, CDCl3): δ 9.25 (s, 1H, H-3a), 8.91 (s, 1H, H-4), 7.77–7.56 (m, 2H, H-5,7), 7.50–7.31 (m, 2H, H-6,8), 4.26 (d, J = 5.6 Hz, 2H, H-3b), 3.80 (s, 3H, H-3c); 13C-NMR (125 MHz, CDCl3): δ 169.73, 161.91, 161.24, 154.55, 148.77, 134.30, 129.90, 125.35, 118.52, 117.96, 116.72, 52.42, 41.69; HRMS (ESI) m/z calcd. for C13H11NO5: [M + H]+ 262.0710, found 262.0718.
Ethyl 3-(2-oxo-2H-chromene-3-carboxamido)propanoate (5b), white solid (60% yield); mp = 128–130 °C; IR (KBr): νmax/cm−1 = 3348, 3055, 1726, 1706, 1647, 1612; UV-Vis (MeOH): λmax/nm = 297; 1H-NMR (500 MHz, CDCl3): δ 9.14 (s, 1H, H-3a), 8.90 (s, 1H, H-4), 7.81-7.56 (m, 2H, H-6,8), 7.39 (m, 2H, H-5,7), 4.20 (q, J = 7.1 Hz, 2H, H-3d), 3.76 (q, J = 6.3 Hz, 2H, H-3b), 2.66 (t, J = 6.4 Hz, 2H, H-3c), 1.29 (t, J = 7.1 Hz, 3H, H-3e); 13C-NMR (125 MHz, CDCl3): δ 171.77, 161.59, 161.26, 154.47, 148.36, 134.06, 129.80, 125.27, 118.61, 118.39, 116.65, 60.84, 35.44, 34.21, 14.20; HRMS (ESI) m/z calcd. for C15H15NO5: [M + H]+ 290.1023, found 290.1029.
Methyl 6-(2-oxo-2H-chromene-3-carboxamido)hexanoate (5c), white solid (85% yield); mp = 88–90 °C; IR (KBr): νmax/cm−1 = 3321, 3054, 1742, 1721, 1706, 1607; UV-Vis (MeOH): λmax/nm = 298; 1H-NMR (500 MHz, CDCl3): δ 8.91 (s, 1H, H-4), 8.83 (s, 1H, H-3a), 7.73–7.63 (m, 2H, H-6,8), 7.39 (m, 2H, H-5,7), 3.67 (s, 3H, H-3g), 3.47 (dd, J = 13.1, 6.9 Hz, 2H, H-3b), 2.34 (t, J = 7.5 Hz, 2H, H-3f), 1.68 (tt, J = 14.8, 7.4 Hz, 4H, H-3c,3e), 1.49–1.36 (m, 2H, H-3d); 13C-NMR (125 MHz, CDCl3): δ 174.02, 161.47, 154.39, 148.26, 133.98, 129.79, 125.28, 118.59, 116.61, 51.51, 39.66, 33.91, 29.09, 26.52, 24.58; HRMS (ESI) m/z calcd. for C17H19NO5: [M + H]+ 318.1336, found 318.1345.
Ethyl 4-(6-methoxy-2-oxo-2H-chromene-3-carboxamido)butanoate (5d), yellowish solid (81% yield); mp = 131–132 °C; IR (KBr): νmax/cm−1 = 3325, 2981, 2948, 1734, 1698; UV-Vis (MeOH): λmax/nm= 297; 1H-NMR (500 MHz, CDCl3): δ 8.92 (s, 1H, H-3a), 8.86 (s, 1H, H-4), 7.34 (d, J = 9.1 Hz, 1H, H-8), 7.24 (dd, J = 9.1, 2.9 Hz, 1H, H-7), 7.08 (d, J = 2.9 Hz, 1H, H-5), 4.15 (q, J = 7.1 Hz, 2H, H-3e), 3.88 (s, 3H, H-6a), 3.51 (dd, J = 13.2, 6.8 Hz, 2H, H-3b), 2.41 (t, J = 7.4 Hz, 2H, H-3d), 1.98 (p, J = 7.2 Hz, 2H, H-3c), 1.26 (t, J = 7.1 Hz, 3H, H-3f); 13C-NMR (125 MHz, CDCl3): δ 172.95, 161.64, 156.61, 149.01, 148.14, 122.57, 118.98, 118.54, 117.73, 110.67, 60.48, 55.92, 39.12, 31.72, 24.79, 14.22; HRMS (ESI) m/z calcd. for C17H19NO6: [M + H]+ 334.1285, found 334.1304.
Methyl 6-(6-methoxy-2-oxo-2H-chromene-3-carboxamido)hexanoate (5e), pale yellowish solid (90% yield); mp = 101–102 °C; IR (KBr): νmax/cm−1 = 3310, 2939, 2859, 1745, 1717, 1697, 1658; UV-Vis (MeOH): λmax/nm = 297; 1H-NMR (500 MHz, CDCl3): δ 8.88 (s, 1H, H-3a), 8.86(s, 1H, H-4), 7.33 (d, J = 9.1 Hz, 1H, H-8), 7.24 (dd, J = 9.1, 2.9 Hz, 1H, H-7), 7.08 (d, J = 2.9 Hz, 1H, H-5), 3.88 (s, 3H, H-6a), 3.67 (s, 3H, H-3g), 3.46 (dd, J = 13.1, 6.9 Hz, 2H, H-3b), 2.34 (t, J = 7.5 Hz, 2H, H-3f), 1.67 (tt, J = 15.1, 7.5 Hz, 4H, H-3c,3e), 1.52–1.36 (m, 2H, H-3d); 13C-NMR (125 MHz, CDCl3): δ 173.99, 161.55, 156.59, 148.97, 148.02, 122.49, 119.00, 118.63, 117.69, 110.65, 55.91, 51.50, 39.64, 33.90, 29.09, 26.52, 24.58; HRMS (ESI) m/z calcd. for C18H21NO6: [M + H]+ 348.1442, found 348.1480.
Methyl 6-(8-ethoxy-2-oxo-2H-chromene-3-carboxamido)hexanoate (5f), white solid (68% yield); mp= 98–99 °C; IR (KBr): νmax/cm−1 = 3343, 1718, 1655, 1613; UV-Vis (MeOH): λmax/nm = 311; 1H-NMR (500 MHz, CDCl3): δ 8.88 (s, 1H, H-4), 8.86 (s, 1H, H-3a), 7.27 (d, J = 7.6 Hz, 1H, H-6), 7.25 (t, J = 3.9 Hz, 1H, H-7), 7.18 (dd, J = 7.7, 1.4 Hz, 1H, H-5), 4.22 (q, J = 7.0 Hz, 2H, H-8a), 3.67 (s, 3H, H-3g), 3.46 (dd, J = 13.2, 6.9 Hz, 2H, H-3b), 2.34 (t, J = 7.5 Hz, 2H, H-3f), 1.67 (tt, J = 14.7, 7.5 Hz, 4H, H-3c, 3e), 1.52 (t, J = 7.0 Hz, 3H, H-8b), 1.47–1.39 (m, 2H, H-3d); 13C-NMR (125 MHz, CDCl3): δ 173.96, 161.52, 161.17, 148.46, 146.41, 144.31, 125.07, 120.84, 119.44, 118.62, 116.73, 65.16, 51.45, 39.65, 33.91, 29.09, 26.52, 24.59, 14.68; HRMS (ESI) m/z calcd. for C19H23NO6: [M + H]+ 362.1598, found 362.1620.
Methyl (6-bromo-2-oxo-2H-chromene-3-carbonyl)glycinate (5g), white solid (46% yield); mp = 160–162 °C; IR (KBr): νmax/cm−1 = 3337, 3048, 1740, 1724, 1655; UV-Vis (MeOH): λmax/nm= 290; 1H-NMR (500 MHz, CDCl3): δ 9.18 (s, 1H, H-3a), 8.82 (s, 1H, H-4), 7.83 (d, J = 2.3 Hz, 1H, H-5), 7.76 (dd, J = 8.8, 2.3 Hz, 1H, H-7), 7.31 (d, J = 8.8 Hz, 1H, H-8), 4.25 (d, J = 5.6 Hz, 2H, H-3b), 3.80 (s, 3H, H-3c); 13C-NMR (125 MHz, CDCl3): δ 169.63, 161.39, 160.59, 153.32, 147.40, 137.00, 131.92, 119.96, 119.00, 118.45, 118.01, 52.48, 41.73; HRMS (ESI) m/z calcd. for C13H10NO5: [M + H]+ 339.9742, found 339.9802.
Ethyl 3-(6-bromo-2-oxo-2H-chromene-3-carboxamido)propanoate (5h), white solid (58% yield); mp = 148–149 °C; IR (KBr): νmax/cm−1 = 3383, 3053, 1734, 1717, 1657; UV-Vis (MeOH): λmax/nm = 292; 1H-NMR (500 MHz, CDCl3): δ 9.09 (s, 1H, H-3a), 8.82 (s, 1H, H-4), 7.83 (d, J = 2.0 Hz, 1H, H-5), 7.74 (dd, J = 8.8, 2.1 Hz, 1H, H-7), 7.29 (d, J = 8.8 Hz, 1H, H-8), 4.20 (q, J = 7.1 Hz, 2H, H-3d), 3.75 (q, J = 6.2 Hz, 2H, H-3b), 2.66 (t, J = 6.3 Hz, 2H, H-3c), 1.29 (t, J = 7.1 Hz, 3H, H-3e); 13C-NMR (125 MHz, CDCl3): δ 171.76, 161.06, 160.58, 153.22, 147.00, 136.74, 131.84, 120.07, 119.43, 118.36, 117.90, 60.87, 35.49, 34.11, 14.19; HRMS (ESI) m/z calcd. for C15H14BrNO5: [M + H]+ 368.0128, found 368.0132.
Methyl 6-(6-bromo-2-oxo-2H-chromene-3-carboxamido)hexanoate (5i), white solid (77% yield); mp = 147–149 °C; IR(KBr): νmax/cm−1 = 3336, 3051, 1734, 1715, 1658; UV-Vis (MeOH) λmax/nm = 292; 1H-NMR (500 MHz, CDCl3): δ 8.84 (s, 1H, H-4), 8.75 (s, 1H, H-3a), 7.84 (d, J = 2.2 Hz, 1H, H-5), 7.74 (dd, J = 8.8, 2.3 Hz, 1H, H-7), 7.30 (d, J = 8.8 Hz, 1H, H-8), 3.67 (s, 3H, H-3g), 3.46 (dd, J = 13.1, 7.0 Hz, 2H, H-3b), 2.34 (t, J = 7.5 Hz, 2H, H-3f), 1.67 (tt, J = 15.0, 7.5 Hz, 4H, H-3c,3e), 1.50–1.37 (m, 2H, H-3d); 13C-NMR (125 MHz, CDCl3): δ 173.98, 160.91, 160.84, 153.16, 146.89, 136.67, 131.83, 120.14, 119.56, 118.33, 117.92, 51.52, 39.75, 33.89, 29.05, 26.50, 24.57; HRMS (ESI) m/z calcd. for C17H18BrNO5: [M + H]+ 396.0441, found 396.0448.
Ethyl 3-(7-(diethylamino)-2-oxo-2H-chromene-3-carboxamido)propanoate (5j), yellow solid (40% yield); mp = 134–136 °C; IR (KBr): νmax/cm−1 = 3444, 3320, 2940, 1739, 1702; UV-Vis (MeOH): λmax/nm = 419; 1H-NMR (500 MHz, CDCl3): δ 9.08 (s, 1H, H-3a), 8.69 (s, 1H, H-4), 7.42 (d, J = 9.0 Hz, 1H, H-5), 6.64 (dd, J = 9.0, 2.1 Hz, 1H, H-6), 6.49 (d, J = 1.8 Hz, 1H, H-8), 4.19 (q, J = 7.1 Hz, 4H, H-7a), 3.73 (q, J = 6.3 Hz, 2H, H-3d), 3.45 (q, 2H, H-3b), 2.64 (t, J = 6.5 Hz, 2H, H-3c), 1.29 (t, J = 7.2 Hz, 3H, H-3e), 1.24 (t, J = 7.2 Hz, 6H, H-7b); 13C-NMR (125 MHz, CDCl3): δ 171.94, 163.25, 162.61, 157.67, 152.55, 148.09, 131.13, 110.21, 109.91, 108.35, 96.59, 60.72, 45.07, 35.25, 34.49, 14.20, 12.42. HRMS (ESI) m/z calcd. for C19H24N2O5: [M + H]+ 361.1758, found 361.1780.
Methyl 6-(7-(diethylamino)-2-oxo-2H-chromene-3-carboxamido)hexanoate (5k), yellow solid (61% yield); mp = 90–91 °C; IR (KBr): νmax/cm−1 = 3367, 2978, 1735, 1688, 1650; UV-Vis (MeOH): λmax/nm = 418; 1H-NMR (500 MHz, CDCl3): δ 8.80 (s, 1H, H-3a), 8.70 (s, 1H, H-4), 7.43 (d, J = 8.9 Hz, 1H, H-5), 6.64 (dd, J = 8.9, 2.3 Hz, 1H, H-6), 6.50 (d, J = 2.0 Hz, 1H, H-8), 3.67 (s, 3H, H-3g), 3.49–3.38 (m, 6H, H-7a,3b), 2.33 (t, J = 7.5 Hz, 2H, H-3f), 1.66 (qd, J = 15.0, 7.4 Hz, 4H, H-3c,3e), 1.51–1.36 (m, 2H, H-3d), 1.24 (t, J = 7.1 Hz, 6H, H-7b); 13C-NMR (125 MHz, CDCl3): δ 174.08, 163.08, 162.80, 157.60, 152.47, 148.01, 131.08, 110.43, 109.91, 108.40, 96.56, 51.49, 45.06, 39.42, 33.97, 29.28, 26.58, 24.66. HRMS (ESI) m/z calcd. for C21H28N2O5: [M + H]+ 389.2071, found 389.2102.
3.1.3. General Procedure for the Preparation of N-(2-Oxo-2H-Chromene-3-Carboxamide) Acids (6a–k)
In a round-bottom flask, the corresponding ester (1 mmol) was added and dissolved in 10 mL of THF:H
2O (1:2). The mixture was stirred at room temperature before adding LiOH (7 mmol) and stirring was continued overnight. Upon completion of the reaction, HCl was slowly added until a precipitate appeared, which was filtered and purified by recrystallization with MeOH and cold water to afford the desired product. Diethylamine derivatives were synthesized by using the Mravljak, J. et al. methodology [
59].
(2-oxo-2H-chromene-3-carbonyl)glycine (6a), white solid (93% yield); mp = 246–247 °C; IR (KBr): νmax/cm−1 = 3315, 2967, 2686, 1759, 1712, 1638; UV-Vis (MeOH): λmax/nm = 297; 1H-NMR (400 MHz, DMSO-d6): δ 12.78 (s, 1H, H-3c), 8.99 (s, 1H, H-3a), 8.86 (s, 1H, H-4), 7.95 (d, J = 7.4 Hz, 1H, H-8), 7.71 (t, J = 7.5 Hz, 1H, H-7), 7.46 (d, J = 8.1 Hz, 1H, H-5), 7.39 (t, J = 7.0 Hz, 1H, H-6), 4.00 (s, 2H, H-3b); 13C-NMR (100 MHz, DMSO-d6): δ 171.27, 161.63, 160.84, 154.46, 148.58, 134.77, 130.89, 125.64, 118.89, 118.66, 116.64, 41.97; HRMS (ESI) m/z calcd. for C12H9NO5: [M + H]+ 248.0553, found 248.0561.
3-(2-oxo-2H-chromene-3-carboxamido)propanoic acid (6b), white solid (70% yield); mp = 199–200 °C; IR (KBr): νmax/cm−1 = 3323, 3068, 1725, 1706, 1635; UV-Vis (MeOH): λmax/nm = 297; 1H-NMR (400 MHz, DMSO-d6): δ 12.34 (s, 1H, H-3d), 8.85 (s, 1H, H-3a), 8.82 (s, 1H, H-4), 7.93 (d, J = 7.4 Hz, 1H, H-8), 7.69 (t, J = 7.4 Hz, 1H, H-7), 7.44 (d, J = 8.2 Hz, 1H, H-5), 7.38 (t, J = 7.1 Hz, 1H, H-4), 3.47 (d, J = 5.0 Hz, 2H, H-3b), 2.46 (d, J = 7.7 Hz, 2H, H-3c); 13C-NMR (100 MHz, DMSO-d6): δ 173.58, 161.46, 160.86, 154.35, 148.14, 134.58, 130.76, 125.59, 119.08, 118.92, 116.59, 35.55, 34.13; HRMS (ESI) m/z calcd. for C13H11NO5: [M + H]+ 262.0710, found 262.0719.
6-(2-oxo-2H-chromene-3-carboxamido)hexanoic acid (6c), white solid (75% yield); mp = 145–146 °C; IR (KBr): νmax/cm−1 = 3357, 1710, 1694, 1527; UV-Vis (MeOH): λmax/nm = 299; 1H-NMR (500 MHz, DMSO-d6): δ 12.01 (s, 1H, H-3g), 8.85 (s, 1H, H-4), 8.69 (t, J = 5.6 Hz, 1H, H-3a), 7.99 (d, J = 7.4 Hz, 1H, H-8), 7.88–7.65 (m, 1H, H-7), 7.51 (d, J = 8.3 Hz, 1H, H-5), 7.44 (t, J = 7.4 Hz, 1H, H-6), 3.39–3.29 (m, 2H, H-3b), 2.22 (t, J = 7.3 Hz, 2H, H-3f), 1.62–1.47 (m, 4H, H-3c,3e), 1.36–1.27 (m, 2H, H-3d); 13C-NMR (125 MHz, DMSO-d6): δ 174.86, 161.48, 160.85, 154.31, 147.72, 134.45, 130.67, 125.57, 119.62, 118.96, 116.58, 39.42, 34.04, 29.15, 26.43, 24.64; HRMS (ESI) m/z calcd. for C16H17NO5: [M + H]+ 304.1179, found 304.1184.
4-(6-methoxy-2-oxo-2H-chromene-3-carboxamido)butanoic acid (6d), light yellow solid (81% yield); mp = 200–203 °C; IR (KBr): νmax/cm−1 = 3311, 1700, 1531; UV-Vis (MeOH) λmax/nm = 298; 1H-NMR (500 MHz, DMSO-d6): δ 12.12 (s, 1H, H-3e), 8.78 (s, 1H, H-4), 8.74 (t, J = 5.8 Hz, 1H, H-3a), 7.52 (d, J = 2.9 Hz, 1H, H-5), 7.43 (d, J = 9.1 Hz, 1H, H-8), 7.32 (dd, J = 9.1, 3.0 Hz, 1H, H-7), 3.82 (s, 3H, H-6a), 3.36 (dd, J = 13.1, 6.7 Hz, 2H, H-3b), 2.30 (t, J = 7.4 Hz, 2H, H-3d), 1.78 (p, J = 7.2 Hz, 2H, H-3c); 13C-NMR (125 MHz, DMSO-d6): δ 174.54, 161.66, 160.88, 156.40, 148.78, 147.54, 122.29, 119.64, 119.35, 117.65, 112.24, 56.27, 39.00, 31.59, 24.99; HRMS (ESI) m/z calcd. for C15H15NO6: [M + H]+ 306.0972, found 306.0974.
6-(6-methoxy-2-oxo-2H-chromene-3-carboxamido)hexanoic acid (6e), light yellow solid (81% yield); mp = 142–144 °C; IR (KBr): νmax/cm−1 = 3322, 1721, 1702, 1575; UV-Vis (MeOH): λmax/nm = 298; 1H-NMR (500 MHz, DMSO-d6): δ 8.79 (s, 1H, H-4), 8.72 (s, 1H, H-3a), 7.52 (s, 1H, H-5), 7.42 (d, J = 9.2 Hz, 1H, H-8), 7.38-7.27 (m, 1H, H-7), 3.82 (s, 3H, H-6a), 3.32 (dd, J = 13.0, 6.7 Hz, 2H, H-3b), 2.23 (t, J = 7.3 Hz, 2H, H-3f), 1.61–1.49 (m, 4H, H-3c,3e), 1.40–1.30 (m, 2H, H-3d); 13C-NMR (125 MHz, DMSO-d6): δ 174.85, 161.45, 160.98, 156.41, 148.77, 147.61, 122.32, 119.52, 119.36, 117.66, 112.23, 56.26, 39.42, 34.04, 29.14, 26.44, 24.64; HRMS (ESI) m/z calcd. for C17H19NO6: [M + H]+ 334.1285, found 334.1288.
6-(8-ethoxy-2-oxo-2H-chromene-3-carboxamido)hexanoic acid (6f), white solid (83% yield); mp = 169–170 °C; IR (KBr): νmax/cm−1 = 3337, 1716, 1605, 1542; UV-Vis (MeOH): λmax/nm = 312; 1H-NMR (500 MHz, DMSO-d6): δ 12.02 (s, 1H, H-3g), 8.80 (s, 1H, H-4), 8.69 (t, J = 5.7 Hz, 1H, H-3a), 7.49 (d, J = 7.1 Hz, 1H, H-7), 7.40 (d, J = 7.6 Hz, 1H, H-5), 7.34 (t, J = 7.9 Hz, 1H, H-6), 4.20 (q, J = 6.9 Hz, 2H, H-8a), 3.32 (dd, J = 13.2, 6.7 Hz, 2H, H-3b), 2.22 (t, J = 7.3 Hz, 2H, H-3f), 1.59–1.49 (m, 4H, H-3c,3e), 1.42 (t, J = 7.0 Hz, 3H, H-8b), 1.38–1.29 (m, 2H, H-3d); 13C-NMR (125 MHz, DMSO-d6): δ 174.84, 161.45, 160.63, 147.96, 145.97, 143.74, 125.49, 121.55, 119.60, 117.31, 65.03, 39.43, 34.05, 29.15, 26.44, 24.64, 15.01; HRMS (ESI) m/z calcd. for C18H21NO6: [M + H]+ 348.1442, found 348.1452.
(6-bromo-2-oxo-2H-chromene-3-carbonyl)glycine (6g), white solid (83% yield); mp = 269–270 °C; IR (KBr): νmax/cm−1 = 3341, 1726, 1648, 1560; UV-Vis (MeOH) λmax/nm = 291; 1H-NMR (500 MHz, DMSO-d6): δ 12.84 (s, 1H, H-3c), 9.02 (t, J = 5.5 Hz, 1H, H-3a), 8.87 (s, 1H, H-4), 8.27 (d, J = 2.3 Hz, 1H, H-5), 7.90 (dd, J = 8.8, 2.3 Hz, 1H, H-7), 7.49 (d, J = 8.9 Hz, 1H, H-8), 4.07 (d, J = 5.5 Hz, 2H, H-3b). 13C-NMR (125 MHz, DMSO-d6): δ 171.16, 161.32, 160.33, 153.48, 147.25, 136.87, 132.70, 120.75, 119.79, 118.89, 117.14, 42.00. HRMS (ESI) m/z calcd. for C12H8NO5: [M + H]+ 325.9659, found 325.9668.
3-(6-bromo-2-oxo-2H-chromene-3-carboxamido)propanoic acid (6h), white solid (50% yield); mp = 251–252 °C; IR (KBr): νmax/cm−1 = 3380, 1705, 1657, 1533; UV-Vis (MeOH): λmax/nm = 292; 1H-NMR (500 MHz, DMSO-d6): δ 12.39 (s, 1H, H-3d), 8.88 (t, J = 5.8 Hz, 1H, H-3a), 8.84 (s, 1H, H-4), 8.25 (d, J = 2.2 Hz, 1H, H-5), 7.88 (dd, J = 8.8, 2.3 Hz, 1H, H-7), 7.47 (d, J = 8.9 Hz, 1H, H-8), 3.54 (q, J = 6.4 Hz, 2H, H-3b), 2.54 (t, J = 6.7 Hz, 2H, H-3c); 13C-NMR (125 MHz, DMSO-d6): δ 173.50, 161.16, 160.35, 153.38, 146.79, 136.70, 132.59, 120.80, 120.25, 118.85, 117.10, 35.62, 34.11; HRMS (ESI) m/z calcd. for C13H10NO5: [M + H]+ 339.9815, found 339.9817.
6-(6-bromo-2-oxo-2H-chromene-3-carboxamido)hexanoic acid (6i), white solid (93% yield); mp = 182–183 °C; IR (KBr): νmax/cm−1 = 3347, 1715, 1657, 1562; UV-Vis (MeOH): λmax/nm= 293; 1H-NMR (500 MHz, DMSO-d6): δ 12.01 (s, 1H, H-3g), 8.80 (s, 1H, H-4), 8.66 (t, J = 5.7 Hz, 1H, H-3a), 8.25 (d, J = 2.2 Hz, 1H, H-5), 7.88 (dd, J = 8.8, 2.3 Hz, 1H, H-7), 7.47 (d, J = 8.8 Hz, 1H, H-8), 3.31 (dd, J = 13.1, 6.7 Hz, 2H, H-3b), 2.22 (t, J = 7.4 Hz, 2H, H-3f), 1.59–1.47 (m, 4H, H-3c,3e), 1.37–1.28 (m, 2H, H-3d); 13C-NMR (125 MHz, DMSO-d6): δ 174.84, 161.18, 160.32, 153.33, 146.37, 136.59, 132.51, 120.83, 120.78, 118.85, 117.08, 39.47, 34.05, 29.11, 26.42, 24.64; HRMS (ESI) m/z calcd. for C16H16NO5: [M + H]+ 382.0212, found 382.0289.
3-(7-(diethylamino)-2-oxo-2H-chromene-3-carboxamido)propanoic acid (6j), yellow crystals (75% yield); mp = 202–205 °C; 1H-NMR (DMSO-d6, 500 MHz): δ 8.84 (t, J = 5.1 Hz, 1H), 8.65 (s, 1H), 7.66 (d, J = 8.9 Hz, 1H), 6.78 (d, J = 8.6 Hz, 1H), 6.59 (s, 1H), 3.54–3.50 (m, 2H), 3.47 (dd, J = 13.7, 6.8 Hz, 4H), 2.49 (t, J = 6.2 Hz, 2H), 1.14 (t, J = 6.9 Hz, 6H).
6-(7-(diethylamino)-2-oxo-2H-chromene-3-carboxamido)hexanoic acid (6k), yellow crystals (73% yield); mp = 150–152 °C; 1H-NMR (DMSO-d6, 500 MHz): δ 8.67–8.62 (m, 2H), 7.68 (d, J = 9.0 Hz, 1H), 6.80 (dd, J = 9.0, 2.2 Hz, 1H), 6.62 (d, J = 2.0 Hz, 1H), 3.48 (dd, J = 14.0, 7.0 Hz, 4H), 3.31–3.27 (m, 2H), 2.21 (t, J = 7.3 Hz, 2H), 1.51 (dq, J = 14.8, 7.4 Hz, 4H), 1.31 (dd, J = 15.2, 8.1 Hz, 2H), 1.14 (t, J = 7.0 Hz, 6H).
3.1.4. General Procedure for the Preparation of the Title Compounds (7a–k)
In a dry round-bottom flask purged with N2, the corresponding carboxylic acid (1 mmol), DMAP (5% mol) and CDI (1.1 mmol) were dissolved in DMF. After 30 min of stirring at room temperature, hydroxylamine hydrochloride was added and stirring continued overnight. Upon completion of the reaction, sodium bicarbonate was added. The resulting precipitate was obtained by filtration and purified by recrystallization with MeOH and cold water.
N-(2-(hydroxyamino)-2-oxoethyl)-2-oxo-2H-chromene-3-carboxamide (7a), white solid (50% yield); mp = 226–227 °C; IR (KBr): νmax/cm−1 = 3304, 3240, 3043, 1709, 1668; UV-Vis (MeOH): λmax/nm = 297; Em (MeOH): λmax/nm = 406; 1H-NMR (400 MHz, DMSO-d6): δ 10.68 (s, 1H), 9.04 (s, 1H), 8.90 (s, 1H), 8.00 (s, 1H), 7.76 (s, 1H), 7.61–7.35 (m, 2H), 3.92 (s, 2H); 13C-NMR (100 MHz, DMSO-d6): δ 165.66, 161.61, 160.84, 154.38, 148.16, 134.71, 130.84, 125.64, 118.92, 118.88, 116.62, 41.09; HRMS (ESI) m/z calcd. for C12H10N2O5: [M + Na]+ 285.0488, found 285.0505.
N-(3-(hydroxyamino)-3-oxopropyl)-2-oxo-2H-chromene-3-carboxamide (7b), white solid (45% yield); mp = 170–173 °C; IR (KBr): νmax/cm−1 = 3469, 3302, 3194, 1708, 1648; UV-Vis (MeOH) λmax/nm = 303.12; Em (MeOH): λmax/nm = 405; 1H-NMR (500 MHz, DMSO-d6): δ 8.88 (s, 1H), 8.87 (s,1H), 7.99 (d, J = 6.6 Hz, 1H), 7.76 (dd, J = 11.5, 4.2 Hz, 1H), 7.51 (d, J = 8.3 Hz, 1H), 7.45 (t, J = 7.2 Hz, 1H), 3.54 (dd, J = 12.7, 6.5 Hz, 2H), 2.29 (t, J = 6.7 Hz, 2H); 13C-NMR (125 MHz, DMSO-d6): δ 167.78, 161.45, 160.78, 154.36, 148.09, 134.59, 130.76, 125.60, 119.11, 118.91, 116.60, 116.56, 36.12, 32.39. HRMS (ESI) m/z calcd. for C13H12N2O5: [M + H]+ 277.0819, found 277.0860.
N-(6-(hydroxyamino)-6-oxohexyl)-2-oxo-2H-chromene-3-carboxamide (7c), white solid (50% yield); mp = 110–112 °C; IR (KBr): νmax/cm−1 = 3336, 3207, 1706, 1537; UV-Vis (MeOH): λmax/nm = 329.96; Em (MeOH): λmax/nm = 405; 1H-NMR (500 MHz, DMSO-d6): δ 8.84 (s, 1H), 8.68 (s, 1H), 7.97 (d, J = 6.3 Hz, 1H), 7.74 (s, 1H), 7.50 (d, J = 7.4 Hz, 1H), 7.44 (s, 2H), 3.31 (d, J = 3.4 Hz, 3H), 1.96 (s, 2H), 1.53 (s, 5H), 1.29 (s, 2H); 13C-NMR (125 MHz, DMSO-d6): δ 169.47, 161.48, 160.85, 154.30, 147.71, 134.46, 130.66, 125.58, 119.59, 118.94, 116.57, 39.49, 32.66, 29.17, 26.50, 25.31; HRMS (ESI) m/z calcd. for C16H18N2O5: [M + Na]+ 341.1114, found 341.1019.
N-(4-(hydroxyamino)-4-oxobutyl)-6-methoxy-2-oxo-2H-chromene-3-carboxamide (7d), light green solid (30% yield); mp = 154–156 °C; IR (KBr): νmax/cm−1 = 3531, 3241, 1706, 1554; UV-Vis (MeOH): λmax/nm = 363; Em (MeOH): λmax/nm = 458; 1H-NMR (500 MHz, DMSO-d6): δ 8.80 (s, 1H), 8.75 (s, 1H), 7.54 (d, J = 2.2 Hz, 1H), 7.44 (d, J = 9.1 Hz, 1H), 7.32 (dd, J = 9.1, 2.5 Hz, 1H), 3.82 (s, 3H), 3.32 (dd, J = 13.1, 6.6 Hz, 2H), 2.03 (t, J = 7.5 Hz, 2H), 1.82–1.72 (m, 2H); 13C-NMR (125 MHz, DMSO-d6): δ 169.09, 161.64, 160.89, 156.40, 148.79, 147.62, 122.35, 119.63, 119.37, 117.70, 112.25, 56.28, 39.22, 30.32, 25.72; HRMS (ESI) m/z calcd. for C15H16N2O6: [M + H]+ 321.1081, found 321.1132.
N-(6-(hydroxyamino)-6-oxohexyl)-6-methoxy-2-oxo-2H-chromene-3-carboxamide (7e), light green solid (87% yield); mp = 75–77 °C; IR (KBr): νmax/cm−1 = 3318, 2935, 1702, 1575; UV-Vis (MeOH): λmax/nm = 299; Em (MeOH): λmax/nm = 458; 1H-NMR (500 MHz, DMSO-d6): δ 10.36 (s, 1H), 8.81 (s, 1H), 8.72 (s, 1H), 7.54 (s, 1H), 7.44 (d, J = 9.0 Hz, 1H), 7.33 (d, J = 8.9 Hz, 1H), 3.82 (s, 3H), 3.31 (s, 2H), 2.21 (t, J = 7.2 Hz, 1H), 1.97 (d, J = 7.2 Hz, 1H), 1.53 (d, J = 6.3 Hz, 4H), 1.37–1.27 (m, 2H); 13C-NMR (125 MHz, DMSO-d6): δ 169.52, 161.48, 161.01, 156.43, 148.80, 147.65, 122.36, 119.60, 119.40, 117.70, 112.28, 56.30, 39.48, 32.65, 29.18, 26.50, 25.30; HRMS (ESI) m/z calcd. for C18H20N2O5: [M + Na]+ 371.1219 found 371.1194.
8-ethoxy-N-(6-(hydroxyamino)-6-oxohexyl)-2-oxo-2H-chromene-3-carboxamide (7f), light green solid (48% yield); mp = 167–169 °C; IR (KBr): νmax/cm−1 = 3335, 1933, 1716, 1539; UV-Vis (MeOH): λmax/nm= 311; Em (MeOH) λmax/nm= 486; 1H-NMR (500 MHz, DMSO-d6): δ 8.81 (s, 3H), 8.69 (s, 3H), 7.50 (d, J = 7.6 Hz, 3H), 7.41 (d, J = 8.0 Hz, 3H), 7.34 (t, J = 7.9 Hz, 3H), 4.20 (q, J = 6.8 Hz, 6H), 1.53 (s, 12H), 1.42 (t, J = 6.9 Hz, 11H), 1.32 (dd, J = 17.8, 7.6 Hz, 7H); 13C-NMR (125 MHz, DMSO-d6): δ 169.51, 161.47, 160.65, 147.99, 145.97, 143.72, 125.53, 121.55, 119.61, 117.32, 65.02, 34.06, 32.64, 29.19, 26.50, 25.30, 15.02; HRMS (ESI) m/z calcd. for C18H22N2O6: [M + H]+ 362.3820, found 362.3843.
6-bromo-N-(2-(hydroxyamino)-2-oxoethyl)-2-oxo-2H-chromene-3-carboxamide (7g), white solid (46% yield); mp = 202–205 °C; IR (KBr) νmax/cm−1 = 3417, 3272, 1712, 1537; UV-Vis (MeOH): λmax/nm = 294; Em (MeOH): λmax/nm = 424; 1H-NMR (500 MHz, DMSO-d6): δ 9.01 (d, J = 4.6 Hz, 1H), 8.86 (s, 1H), 8.28 (s, 1H), 7.90 (d, J = 8.8 Hz, 1H), 7.50 (d, J = 8.9 Hz, 1H), 3.92 (d, J = 5.2 Hz, 2H); 13C-NMR (125 MHz, DMSO-d6): δ 165.55, 161.34, 160.34, 153.43, 146.83, 136.83, 132.67, 120.78, 120.13, 118.91, 117.14, 41.13; HRMS (ESI) m/z calcd. for C12H9BrN2O5: [M + H]+ 339.9695, found 339.9711.
6-bromo-N-(3-(hydroxyamino)-3-oxopropyl)-2-oxo-2H-chromene-3-carboxamide (7h), white solid (45% yield); mp = 185–187 °C; IR (KBr): νmax/cm−1 = 3354, 3040, 2854, 1708, 1538; UV-Vis (MeOH): λmax/nm = 295; Em (MeOH): λmax/nm = 415; 1H-NMR (500 MHz, DMSO-d6): δ 8.86 (d, J = 5.4 Hz, 1H), 8.83 (s, 1H), 8.26 (s, 2H), 7.89 (dd, J = 8.8, 1.8 Hz, 1H), 7.48 (d, J = 8.8 Hz, 1H), 3.53 (dd, J = 12.6, 6.4 Hz, 3H), 2.28 (t, J = 6.7 Hz, 2H; 13C-NMR (125 MHz, DMSO-d6): δ 161.16, 160.30, 153.40, 146.78, 146.58, 136.72, 136.54, 132.57, 120.82, 118.86, 117.06, 36.19, 32.31; MS (ESI) m/z calcd. for C13H11BrN2O6: [M + H]+ 356.1440, found 356.1252.
6-bromo-N-(6-(hydroxyamino)-6-oxohexyl)-2-oxo-2H-chromene-3-carboxamide (7i), white solid (65% yield); mp = 202–205 °C; IR (KBr): νmax/cm−1 = 3344, 2934, 1724, 1559; UV-Vis (MeOH): λmax/nm = 283; Em (MeOH): λmax/nm = 424; 1H-NMR (500 MHz, DMSO-d6): δ 8.80 (d, J = 4.2 Hz, 2H), 8.67 (t, J = 5.7 Hz, 2H), 8.25 (dd, J = 6.4, 2.2 Hz, 2H), 7.88 (dd, J = 8.8, 2.3 Hz, 2H), 7.47 (dd, J = 8.6, 5.5 Hz, 2H), 3.30 (dd, J = 12.6, 6.4 Hz, 5H), 1.96 (t, J = 7.3 Hz, 4H), 1.52 (dt, J = 14.9, 7.4 Hz, 8H), 1.29 (dd, J = 14.7, 7.7 Hz, 4H); 13C-NMR (125 MHz, DMSO-d6): δ 169.41, 162.78, 161.19, 160.36, 153.32, 146.40, 136.61, 132.51, 120.79, 118.87, 117.09, 39.52, 36.25, 29.16, 26.48, 25.32; HRMS (ESI) m/z calcd. for C16H17BrN2O5: [M + H]+ 397.0321, found 397.1750.
7-(diethylamino)-N-(3-(hydroxyamino)-3-oxopropyl)-2-oxo-2H-chromene-3-carboxamide (7j), yellow solid (82% yield); mp = 126–128 °C; IR (KBr): νmax/cm−1 = 3231, 1699, 1616; UV-Vis (MeOH): λmax/nm = 419; Em (MeOH): λmax/nm = 468; 1H-NMR (500 MHz, DMSO-d6): δ 8.78 (t, J = 5.7 Hz, 1H), 8.65 (s, 2H), 7.67 (d, J = 9.0 Hz, 1H), 6.78 (d, J = 9.0 Hz, 1H), 6.60 (d, J = 1.3 Hz, 1H), 3.47 (q, J = 6.8 Hz, 4H), 2.25 (t, J = 6.6 Hz, 2H), 2.01 (t, J = 7.5 Hz, 2H), 1.13 (t, J = 6.9 Hz, 6H); 13C-NMR (125 MHz, DMSO-d6): δ 169.13, 167.85, 162.60, 162.05, 157.70, 152.87, 148.16, 132.02, 110.57, 108.12, 108.10, 96.34, 44.79, 12.77; HRMS (ESI) m/z calcd. for C17H21N3O5: [M + H]+ 348.1554, found 348.1570.
7-(diethylamino)-N-(6-(hydroxyamino)-6-oxohexyl)-2-oxo-2H-chromene-3-carboxamide (7k), yellow solid (82% yield); mp = 173–174 °C; IR (KBr): νmax/cm−1 = 3122, 1700, 1618; UV-Vis (MeOH): λmax/nm = 418; Em (MeOH): λmax/nm = 470; 1H-NMR (500 MHz, DMSO-d6): δ 8.78 (t, J = 5.7 Hz, 1H), 8.65 (s, 2H), 7.67 (d, J = 9.0 Hz, 1H), 6.78 (d, J = 9.0 Hz, 1H), 6.60 (d, J = 1.3 Hz, 1H), 3.47 (q, J = 6.8 Hz, 4H), 2.25 (t, J = 6.6 Hz, 2H), 2.01 (t, J = 7.5 Hz, 2H), 1.13 (t, J = 6.9 Hz, 6H); 13C-NMR (125 MHz, DMSO-d6): δ 169.13, 167.85, 162.60, 162.05, 157.70, 152.87, 148.16, 132.02, 110.57, 108.12, 108.10, 96.34, 44.79, 35.87, 32.68, 12.77. HRMS (ESI) m/z calcd. for C20H27N3O5: [M + H]+ 389.1951, found 389.1969.