Conifers Phytochemicals: A Valuable Forest with Therapeutic Potential
Abstract
:1. Introduction
| Subclasss | Main Examples of Each Class | Conifer spp. | References |
|---|---|---|---|
| Terpenes | |||
| Monoterpenes | β-myrcene, α-pinene, limonene, α-terpinene, thujone, camphene, β-pinene, thujole, Δ-3-carene, phellandrene | Pinus roxburghii, P. pinea, P. wallichiana, P. pinaster, P.sylvestris, P. gerardiana, P. nigra, P. radiata, Thuja occidentalis, Abies alba, Picea abies, Metasequoia glyptostroboides | [12,15,16,17] |
| Sesquiterpenes | Laurenobiolide, farnesene, inulicin, vernodalin, 3H-benzofuaran-2-one, 4-methyl-3-methoxy-3H-benzofuaran-2-one, 4,9(α)-dihydroxynardosin-6-en, delta-cadinene, alpha-humulene, beta-cedrene, trans-caryophyllene, cubenol | P. mariana, Juniperus foetidissima, A. alba, P. abies, M. glyptostroboides, J. phoenicea, P. roxburghii | [8,12,18,19,20] |
| Diterpenes | Paclitaxel, 10-deacetylbaccatin III, tasumatrol B, taxodal, sandaracopimaric acid, taxodione, xanthoperol, andrographolide, gibberellin A8, 7α-hydroxysandaracopimaric acid, gibberellin A12, gibberellin A12 aldehyde, gibberellin A15, 14α-hydroxyisopimaric acid, 12-hydroxydehydroabietic acid, gibberellin A19, gibberellin A9, carnosol, lathyrol, E-communic acid, 15-hydroxy-8(17),13(E)-labdadiene-19-carboxilic acid, holophyllin A, holophyllin D, sugiol, ferruginol | Taxus brevifolia, T. baccata, T. globosa, T. distichum, P. mariana, J. taxifolia, M. glyptostroboides, A. holophylla, J. excelsa, J. communis, J. excelsa, J. communis | [18,19,21,22,23,24,25,26,27] |
| Nitrogen Compounds | |||
| Alkaloids | Vellosimine, 1,6-dehydropinidine, cis-pinidine, 1,6-dehydropinidinone, epipinidinone, cis-pinidinol, trans-pinidine, euphococcinine, α-pipecoline 1, (−)-pinidine | P. mariana, P. abies, P. sabiniana, P. torreyana, | [13,18,28] |
| Lignans | Lariciıesinol, taxiresinol, 3’-demethylisolariciresino1-9’-hydroxyisopropylethe, isolariciresinol, deoxypodophyllotoxin, (−)-secoisolariciresinol, 3, 3-demethylisolariciresinol, isotaxiresinol 2, α-conidendrin, (+)-pinoresinol, (−)-matairesinol, arctiin, dibenzylbutyrolactol, (−)-wikstromol, (−)-traxillagenin, (−)-arctigenin, traxillaside, 4′-deme-thyltraxillagenin, [(2R,3R)-2-(4′’-hydroxy-3′’-methoxybenzyl)-3-(4′-hydroxy-3′,5′dimethoxybenzyl)-butyrolactone] | T. baccata, J. taxifolia, J. sabina, J. virginiana, J. virginiana, P. roxburghii, Cedrus deodara, T. nucifera | [29,30,31,32,33,34,35] |
| Polyphenols: Flavonoids | |||
| Flavanonols | Taxifolin, cedeodarin | C. deodara, L. simbraca, P. roxburghii, P. mariana, P. abies, A. pindrow, A. excelsa; P. pinea, P. halepensis, P. pinaster, P. gerardiana | [33,36,37,38,39,40] |
| Flavones | Pilosanol B, luteolin, apigenin, apigenin 6-C-b-glucopyranoside | P. mariana, A. excelsa, P. abies, P. sylvestris, P. menziesii, P. menziesii, J. communis, A. angustifolia, L. deciduas | [18,38,39,41,42,43] |
| Biflavones | Bilobetin, cupressuflavone II-7-O-methyl-robustaflavone | T. wallichiana, C. macrocarpa, A. angustifolia | [43,44,45,46] |
| Flavonols | Quercetin, dihydroquercetin, rutin, kaempferol, dihydrokaempferol | J. communis, J. oxycedrus, P. gerardiana, P. roxburghii, P. wallichiana, A. angustifolia, P. abies, L. deciduas, P. sylvestris, P. menziesii, M. glyptostroboides, J. excelsa, P. mariana, J. foetidissima | [18,33,41,42,43,47,48,49,50] |
| Flavan-3-ols | Monomers: (−)-epicatechin, (−)-epicatechin-3-gallate, (+)-catechin, sennidin A, (−)-epigallocatechin, | P. pinaster, P. pinea, P. halepensis, P. roxburghii, P. wallichiana, P. gerardiana, J. foetidissima, A. angustifolia, P. abies, L. deciduas, P. sylvestris, J. communis, P. menziesii, J. oxycedrus, M. glyptostroboides, J. excelsa | [18,33,39,40,41,43,47,48,49,50] |
| Polymers: Procyanidin B1, B2, procyanidin A2, | P. halepensis, P. pinea, P. pinaster | [40] | |
| Phenolic acids | |||
| Benzoic acids | p-hydroxybenzoic acid, 2,5-dihydroxobenzoic acid, gallic acid, 4-hydroxybenzoic acid, protocatechuic acid, ellagic acid | P. abies, L. deciduas, P. sylvestris, P. menziesii, P. kesiya, J. communis, A. excelsa, P. roxburghii, P. wallichiana, P. gerardiana, L. deciduas, J. communis | [33,38,41] |
| Hydroxycinnamic acid | Caffeic acid, t-cinnamic Acid, p-coumaric acid, vanillic acid, ferulic acid, salicylic acid, sinapic acid, syringic acid, chlorogenic acid, 5-caffeoylquinic acid, caffeic acid 4-O-glucoside | P. abies, L. deciduas, P. sylvestris, T. baccata, P. mariana, P. pinaster, P. kesiya, L. deciduas, J. communis, P. menziesii, M. glyptostroboides | [18,21,39,41,42,49] |
| Stilbenes | trans-resveratrol, resveratrol, trans-pinosylvin, cis-stilbenes, pinosylvin, dihydro-monomethyl, trans-stilbenes, trans-piceatannol, trans-piceid, trans-isorhapontin, trans-isorhapontigenin, phenanthrenes, astringin, trans-astringin | P. mariana, P. abies, J. communis, P. pinaster, P. sylvestris, P. strobes, P. roxburghii, P. wallichiana, P. gerardiana, P. merkusii | [8,18,39,51,52,53,54,55,56,57] |
2. Conifers Phytochemicals Components
2.1. Terpenes
2.2. Alkaloids
2.3. Polyphenols
2.3.1. Flavonoids
2.3.2. Lignans
2.3.3. Stilbenes
2.3.4. Tannins
3. Traditional Medicinal Uses
4. Conifers Extracts Rich in Phytochemical with Putative Health Effects
4.1. Oxidative Stress
4.2. Synergism between ROS and other Diseases
4.2.1. Antioxidant Activity
4.2.2. Anti-Inflammatory Activity
4.2.3. Anticancer Activity
| Conifer spp. | Part Used | Compounds | Nature of Extract | Radical Scavenging Assay | Dose/Concentration | Main Effects | References |
|---|---|---|---|---|---|---|---|
| Aurocaria cookii | Leaves | Phenolic compounds | Methanol, chloroform, petroleum ether | DPPH | 1000 μg/mL | Methanol extract shows the best antioxidant activity with 63% inhibition, higher than the other two compounds | [147] |
| A. excelsa | Needle | Flavanoids | Methanol | DPPH | 50–72.5 μg/mL | Methanol/water extract shows antioxidant activity | [38] |
| C. deodara | Heart wood | Tannins, flavonoids, and phenolic compounds | Water/alcohol | DPPH, superoxide radical-scavenging activity, ABTS | DPPH-IC50 (μg/mL): 61.89 (water extract), 75.79 (alcohol extract) superoxide radical-scavenging activity— IC50 (μg/mL): 87.76 (water extract), 121.55 (alcohol extract). ABTS-IC50 (μg/mL): 115.29 (water extract), 122.42 (alcohol extract). | DPPH radical-scavenging activity and the reducing power of C. deodara were potent in water and alcohol extract | [148] |
| C. japonica | All parts | Phenolic compounds | Methanol | ORAC, SOD | 4.09–7.64 TE/mg 3.63–4.06μg/mL | The methanol extracts from each part of C. japonica except for pollen showed strong activities in the bioactivity assays. | [149] |
| J. communis | Berry | Flavanoids (quercetin rutin, apigenin) chlorogenic acid | Alcohol/Water | DPPH | EC50 1.42 mg/mL against standard Ascorbic acidEC50 value of 0.365 mg/mL | The antioxidant activity was confirmed as 81.63 ± 0.38% by the DPPH assay. | [42] |
| L. laricina | Bark | Phenolic compounds | Ethanol/Water | ORAC | IC50 0.878 μg/mL. | Bark extract of LL shows significant antioxidant activity | [51] |
| Metasequoia glyptostroboides | Cone | Terpenoid | Ethyl acetate | DPPH, NO, superoxide, and H2O2 | 5–250 μg /mL | Sugiol derived from cone extract show good antioxidant activity—78.38, 72.42, 74.45 and 85.04%, respectively. | [26] |
| Picea abies | Bark | Atilbenoids | Ethanol/Water | DPPH | 49.74 μg/mL | UVA-induced modification of the stilbene-rich inner bark extracts increased the antioxidant activity as UVA irradiation decreased the capacity of the extracts to prevent lipid oxidation in the liposome system method | [53] |
| P. smithiana | Leaves | Phenolic compounds | Methanol | DPPH | IC50 (μg/mL)- | Results of the DPPH radical scavenging activity and FRAP study determine that methanol extracts of leaf displayed the highest antiradical efficiency | [150] |
| 228 | |||||||
| FRAP | 494 | ||||||
| Reducing Power assay | 978 | ||||||
| Pinus gerardiana | Bark | Phenolic compounds | Ethanol | DPPH | IC50 value μg/mL | P. gerardiana shows promising H2O2 radical scavenging activity | [104] |
| 102.8 | |||||||
| H2O2 | 81.83 | ||||||
| NO2 | 109.2 | ||||||
| P. halepensis | Bark | Phenolic compounds | Ethanol/Water | IC50 (μg/mL). Ethanol and the water | Ethanol and water extract of bark exhibited significant free radical neutralization capacities, at conc. 0.5–8 μg/mL | [151] | |
| DPPH | 3.28, 3.26 | ||||||
| ABTS | 3.1, 3.59 | ||||||
| P. pinaster | Bark | Phenolic compounds | Ethanol/Water | PB (50%) and (90%) IC50 value μg/mL | PP bark extracts formed from PB 50% (50% ethanol) have maximum (DPPH, ABTS) radical scavenging activity while FRAP shows activity with (PB 90%) | [39] | |
| DPPH | 49.74 | ||||||
| ABTS | 59.41 | ||||||
| FRAP | 101.3 | ||||||
| P. roxburghii | Bark | Phenolic compounds | Ethanol | IC50 value μg/mL | Pine extract shows significant antioxidant activity | [104] | |
| DPPH | 97.54 | ||||||
| H2O2 | 86.90 | ||||||
| NO2 | 111.38 | ||||||
| P. wallichiana | Bark | Phenolic compounds | Ethanol | IC50 (μg/mL) | Pine extract shows significant radical scavenging activity | [104] | |
| DPPH | 111.40 | ||||||
| H2O2 | 84.18 | ||||||
| NO2 | 98.5 | ||||||
| Thuja occidentalis | Leaves | Flavonoids, phenols | Methanol | DPPH, FRAP | 20–100 μg/mL | Crude extract shows significant antioxidant activity | [152] |
| T. occidentalis | Non-woody branches with leaves | Polyphenol, flavonoids | Mother tincture (MT) | DPPH, ORAC, NO | 25 or 50 mg/kg | T. occidentalis mother tincture displayed 88.3% antioxidant activity by DPPH and about 78% by NO assay | [126] |
| Taxus baccata | Leaves and cones | Flavonoids, phenols | Methanol | DPPH | IC50 (μg/mL) 105.41, 518.51 leaves and cones resp. | Acetone and ethyl acetate extract of leaves show good scavenging activity | [153] |
| Water | DPPH | 533.66, >1000 leaves and cones resp. | |||||
| Acetone | DPPH | 25.24, 81.43 leaves and cones resp. | |||||
| Ethyl acetate | DPPH | 29.84, 180.26 leaves and cones resp. | |||||
| Petroleum ether | DPPH | 438.92, > 1000 leaves and cones resp. | |||||
| T. wallichiana | Leaves | Terpenoids, flavonoids | IC50 values (μg/mL) | The maximum DPPH activity was observed in methanol extract (91.25%), followed by water (87.64%), ethanol (85.23%), and ethyl acetate (83.27%) at the highest concentration (700μg/ml) | [154] | ||
| Methanol | Superoxide radical | 170.30 | |||||
| DPPH | 212.00 | ||||||
| LPO | 126.09 | ||||||
| Hydroxyl radical | 82.34 | ||||||
| Ethyl acetate | Superoxide radical | 297.55 | |||||
| DPPH | 301.80 | ||||||
| LPO | 151.96 | ||||||
| Hydroxyl radical | 199.05 | ||||||
| Water | Superoxide radical | 257.00 | |||||
| DPPH | 258.29 | ||||||
| Hydroxyl radical | 175.33 | ||||||
| T. wallichiana | Leaf, stem | Polyphenols, flavanoids, terpenoids | Methanol | DPPH FRAP | IC50 value (μg/mL.) Leaves (23.18) Stem (56.75) | DPPH and FRAP activity of TW leaves and stem extract have high antioxidant activities. | [155] |
| Conifer spp. | Part Used | Nature of Extract | Compounds | Major Method(s) of Testing | Dose. Conc | Main Effect | References |
|---|---|---|---|---|---|---|---|
| Abies chensiensis | Twigs and leaves | Ethanol | Terpenoids | Induce lipopolysaccharide to produce inflammation in RAW 264.7 macrophage cells | 0.2–50.0 μM | 4 compounds—3α-hydroxyl-8,14,22Z,24-tetraenlanosta-26,23-olide; (5R,20R)-8(14→13R)-abeo-17,13-friedo-3-oxolanosta-8,14(30),22Z,24-tetraen-26,23-olide; 8,14,22Z,24-tetraen-3-oxolanosta-26,23-olide; and (23R, 25R)-3,4-seco-9β H-lanosta-4 (28),7-dien-16α-hydroxyl-26,23-olid-3-oate—extracted from extracts showed significant anti-inflammatory activities of inhibition against NO formation with IC50 value of 15.9, 18.7, 20.18, and 10.9 μM | [125] |
| A. georgei | Aerial parts | Chloroform, ethyl acetate, n-butanol | Flavanoids | dimethylbenzene-induced ear oedema in mice | 200 mg/kg | AG ethyl acetate extract shows 18% inhibition against dimethylbenzene-induced ear edema in mice while carrageenin-induced paw edema in rats shows inhibition ratios 28.2% and 35.6%, after 2 and 6h, respectively. | [156] |
| Carrageenin-induced paw oedema rat | 140 mg/kg | ||||||
| A. webbiana | Leaves | Methanol/Petroleum ether extract | Flavanoids | Carrageenan-induced rat hind paw edema model in Albino mice | 400 mg/kg | Plant leaves extract possesses significant anti-inflammatory properties | [157] |
| Agathis robusta | Leaves | Methanol | Flavanoids, tannins and saponins | Heat induced hemolytic method in human red blood cell (HRBC) membrane | 400 μg/kg | Leaves extract shows good antiinflammatory activity | [158] |
| Cedrus deodara | Stem bark | Methanol | Deodarin, quercetin, taxifolin | Carrageenin-induced paw edema in Albino rat | 100 mg/kg | Anti-inflammatory activity with 43.47% inhibition | [159] |
| Cupressus macrocarpa | Leaves | Methanol | Cupressuflavone (CUF) | Carrageenan-induced paw edema model in Mice | 40, 80, and 160 mL/kg | CUF demonstrated antiinflammatory activity by inhibiting paw edema with 55, 60, and 64%, by decreasing the plasma pro-inflammatory mediators PGE2, IL-6, TNF-a and IL-1b | [46] |
| Juniperus communis | Berry | Alcohol/Water | Flavanoids (quercetin rutin, apigenin) chlorogenic acid | Acute-dextran and kaolin subacute inflammation induced in Wistar Rat | 10 mL/kg | The antiinflammatory action of the juniper extract, administered as a microemulsion in acute-dextran model was increased when compared to kaolin subacute inflammation induced model. | [42] |
| J. oxycedrus | Berry | Ethanol, n-butanol | Flavonoids (amentoflavone, cupressuflavone, hinokiflavone, and rutin) | Carrageenan-induced hind paw edema model in mice | 100 mg/kg | Ethanol extract of Joso berries displayed remarkable inflammatory inhibition ranging between 24.5% and 23.7% at 100 mg/kg in carrageenan-induced edema model | [160] |
| J. foetidissima | Berry | Ethanol | Flavonoids (amentoflavone, cupressuflavone, hinokiflavone, and rutin) | carrageenan-induced hind paw edema model in mice | 100 mg/kg | JFB extract at a dose of 100 mg/kg. shows high antiinflammatory effect 26.9% | [160] |
| Pinus gerardiana, P. roxburghii, P. wallichiana | Bark | Ethanol | Flavanoid, tannin | against albumin denaturation, HRBC membrane stabilization assay | 2500 μg/mL | P. roxburghii extract showed highest (%) of inhibition and protection i.e 86.54 and 89.92 against albumin denaturation and HRBC membrane stabilization. However, P. wallichiana have least inhibition and protection percentage, i.e., 76.54 and 81.2% | [104] |
| Taxus baccata | Aerial parts | Methanol | Terpenoids | ear edema induced in mice | 3.2 mg/ear | T. baccata extract displayed best activity | [21] |
| T. baccata | Bark | Ethanol | Alkaloids, terpenoids, flavonoids | carrageenan-induced paw edema in Wistar Albino rat | 200 mg/kg | Percentage of inhibition is 44% at a dose of 200 mg/kg | [161] |
| T. baccata | Heart wood | Ethanol | Taxoids, lignans | carrageenan-induced hind paw edema model inS wiss albino mice | 30–100 mg/kg | TBW shows significant antinociceptive and anti-inflammatory activities | [29] |
| T. wallichiana | Bark | Methanol | Tasumatrol B, 1,13-diacetyl-10-deacetylbaccatin III (10-DAD) and 4-deacetylbaccatin III (4-DAB) | carrageenan-induced paw edema and Cotton-pellet oedema model in Wistar rats and Swiss albino mice | 20 and 40 mg/kg; 40 mg/kg | In a carrageenan-induced inflammation model, tasumatrol B at a dose of 20 mg/kg showed significant activity, while in a cotton-pellet edema model tasumatrol B was found to be highly significant at the dose of 40 mg/kg. | [22] |
| Thuja occidentalis | Non-woody branches with leaves | Mother tincture (MT) | Polyphenols, flavonoids | Administered 2,4,6-trinitrobenzenesulfonic acid to induce intrarectal colitis in mice | 25 or 50 mg/kg | MT manage to relieve intestinal inflammation experimentally induce by TNBS in 7 days. | [126] |
| Conifer spp. | Part Used | Nature of Extract | Compounds | In Vitro and in Vivo Model | Dose. Conc | Main Effects | References |
|---|---|---|---|---|---|---|---|
| Abies georgei | Aerial parts | Chloroform, ethyl acetate, n-butanol | Flavanoids | Human tumor cell lines-A549, QGY-7703, LOVO, 6T-CEM | 77.5, 11.1, 7.8, 32.8 μg/mL | AGC extract has potent tumour and antiproliferative effects in humor tumor cell lines | [156] |
| (Mice) S180 tumours cell lines | 100, 200 and 400 mg/kg | AGC also exhibited activity in tumour growth inhibition in a dose-dependent manner, with ratios of 46.7, 53.1 and 31.0% at doses of 100, 200 and 400 mg/kg, respectively | |||||
| Araucaria angustifolia | Female strobili | Water | Fatty acids and polyphenols | Laryngeal carcinoma HEp-2 cells | 100–500 μg/mL | AAE inhibit the activity of mitochondria complex I and induce redox stress and cytochrome c, which leads cleavage of nuclear proteins of larynx HEp-2 cancer cells | [162] |
| Cedrus deodara | Stem wood | Chloroform | Lignans (Matairesinol, dibenzylbutyrolactol, (−)-Wikstromol) | In vitro human cell lines (cervix, breast, colon, liver, CNS, prostrate) | In vitro cytotoxicity IC 50 value-Wikstromol (71.31–93.63) and Matairesinol (50.84–95.36) μg/mL | CD lignin mixture have potent to show a cytotoxic effect at the maximum in CNS and at the minimum in liver against cancer cell lines in a dose-dependent manner at 100 μg/mL from 49 to 95%. | [34] |
| Human T lymphoblast, acute lymphoblastic leukemia cell line, Molt-4 and human promyelocytic leukemia cell line (HL-60) | IC50 (μg/mL) 15 | AP9-cd-induced endogenous NO production leads to the generation of peroxide and disruption of mitochondrial membrane potential, leading to apoptotic pathway activation Increase in sub-G0 fraction from 35 to 60% in 24 to 48h | [163] | ||||
| In vivo swiss albino mice (K562 cells) | The lignin mixture displays anti-cancer effects by regulating annexin V binding, intracellular caspase activities and DNA fragmentation | ||||||
| C. deodara | Needle | Ethanol | Kaempferol, myricetin, isorhamnetin and quercetin | HepG2 cells | IC50 114.12 μg/mL | TFPNCD shows potent cytotoxicity by inhibiting the growth of HepG2 cells in a dose-dependent manner Regulates cell cycle and apoptosis | [164] |
| Cryptomeria japonica | Leaves | Methanol | Flavonoids | Albino mice of Ehrlich Ascites Carcinoma (EAC). | 100–400 μg/gm | Tumor cell count as well as the amounts of ascetic tumour cells in packed cells were significantly reduced in infected mice treated with MC | [165] |
| Juniper communis | Berry | Methanol and water | Phenolic compounds | CaCo2 and HeLa carcinoma cell lines | IC50 1300–2500 μg/mL | Methanol and water extracts of JCB show potent antiproliferative activity against cancer cell lines | [166] |
| J. taxifolia | Leaves | Chloroform | Polyphenols and lignan | human leukemia (HL-60) cells | 2.5 μg/mL | 7α-hydroxysandaracopimaric acid, a diterpenoid compound obtained from J. taxifolia leaves, shows antitumor effects on HL-60 cells | [24] |
| J. phoenicea | Aerial parts | Chloroform | Polyphenols | IC50 values (μg/mL) | It is found that JPCF disrupts cell cycle progression in the G0/G1phase and shows apoptotic, antiproliferative and necrotic effects on cancer cells lines | [20] | |
| Human lung (A549) | 34.2 | ||||||
| Breast (MCF-7) | 24.5 | ||||||
| Liver (HepG2) cancer cells | 57.6 | ||||||
| J. foetidissima | Needle | Methanol | Quercetin, rutin | Rat brain tumor (C6) cell lines | IC50 values (μg/mL) 10.65 | J. foetidissima needle extract showed significant antiproliferative activity | [50] |
| M. glyptostroboides | Leaf | Water | Polyphenols | PC12 cells | 25 μg/mL | M. glyptostroboides leaf extract shows a cytotoxic effect and prevents oxidative damage of neuronal PC12 cells, protecting them from apoptosis; it was also found to significantly inhibit the release of LDH, which may result from apoptosis or necrosis | [49] |
| Picea wilsonii | Whole plant | DMSO | ND | Human keratinocyte HaCaT cell lines | 1–3 g/mL | PwM extracts inhibit the production of MCP-1 IL-6, IL-13 and but do not inhibit IL-8 production | [115] |
| Pinus kesiya | Woody twig | Ethanol | Phenolic compounds and flavonoids | Human hepatocarcinoma (HepG2) cell lines | IC50 (μg/mL) 52.0 | PK Extract exhibited a potent cytotoxic effect in the HepG2 cell line | [167] |
| P. kesiya | Branch | Ethanol | Phenolic compounds and flavonoids | Human leukemic U937 cancer cells | IC50: 299 μg/mL | PK ethanol extract possesses anticancer activity against U937 human leukemic cells via apoptosis | [168] |
| P. merkusii | Leaves | Methanol | Phenolic compounds | MCF-7, A549, HT 1080 and HepG2 Huh-7 cancer cell lines | IC50 (μg/mL) 4.5, 16, 4.1, 5.6, 9.5 | PM methanol extract possesses anticancer activity against human cancer cell lines | [169] |
| T. baccata | Leaves, cones | Methanol | Phenolic compounds | HCT-116 human colon cancer and MDA-MB-231 human breast cancer cell lines | IC50 μg/mL Leaves: 14.43 and 4.59 cones: 49.69 and 133.53 | Methanol extracts of leaves had better activity on HCT-116 cells than seed cone extract, with IC50 values of 14.3 for 24 h and 4.59 for 72 h. Meanwhile, extracts did not show any significant cytotoxic effects on the cancer cell lines | [153] |
| T. wallichiana | Heartwood | Methanol | Lignans 1 (taxiresinol 1) 2, 3 | colon, ovarian liver, and breast cancer cell lines | IC90 lignan 2 and 3 μg/mL Caco 2:0.08 and 0.056 and 0.251 | Taxiresinol 1 shows anticancer activity against ovary, colon, liver and breast cancer cell lines, while lignans 2 and 3 were found to be most active against Caco-2 cell lines | [170] |
| T. yunnanensis | All parts | ND | α-Conidendrin | MCF-7 andMDA-MB-231 cancer cell lines | 40 μM | α-conidendrin have the potential to inhibit human breast cancer cell lines MDA-MB-231 and MCF-7, showing viability of 73 and 82%, respectively | [31] |
| P. roxburghii | Leaves | Water and ethanol | Phenolic compounds | A549 human lung cancer cell line | 111.2 and 112.7 μg/mL | PRL extract shows potent anticancer activity against cancer cell lines. | [171] |
| Taxus cuspidata | Branches and leaves | Water | Polysaccharides | MCF7 | IC50 μg/mL | Purified polysaccharides (Pe4) on HeLa cells had the highest inhibitory effect, and its IC50 value is 89.9, while (Pe1) shows the best cytotoxic capacity against cancer lines HepG2 and MCF7, with IC50 conc. 132.0 and 169.0 μg/mL, respectively | [172] |
| 169.0 | |||||||
| Hela | 89.9 | ||||||
| HepG2 | 132.0 | ||||||
| Thuja occidentalis | Leaves and non-woody branches | Mother tincture (MT) | Polyphenols including flavonoids | Caco-2 cells | 25 or 50 mg/kg | Caco-2 cells exposed to H2O2 and T. occidentalis MT proves its radical scavenging activity by reducing GSH level by 103% and 98% as compared to TNBS group; MT also managed to reduce the lipid peroxidation | [126] |
| T. occidentalis | Leaves | Ethanol | ND | Human NSCLC (A549) cell lines | IC50 μg/mL | Extract of TO shows both anticancer and antiproliferative activities against NSCLC (A549) cell lines in a dose-dependent manner. | [173] |
| 282 | |||||||
| Human normal embryonic cell lines (L-132) | 376 | ||||||
| T. occidentalis | ND | Mother tincture (MT) Thujone-rich fraction (TRF) | Thujone | A375 human malignant melanoma cell line | 200 μg/mL | TRF as compared with TO MT on exposure to A375 cells exhibited highly cytotoxic, apoptotic and antiproliferative effects, but TRF shows a lower growth inhibitory response towards peripheral blood mononuclear cell (normal cells) | [174] |
4.2.4. Neurodegenerative Diseases
4.2.5. Alzheimer’s Disease (AD)
4.2.6. Parkinson’s Disease
4.2.7. Insomnia
5. Other Activities
5.1. Antidiabetic Activity
5.2. Anticonvulsant Activity
5.3. Analgesic Activity
5.4. Antinociceptive Activity
5.5. Antimicrobial Activity
| Conifers spp. | Compounds with Neuroprotective Potential | Model | Effective Concentration | Relevant Bioactivities | Reference |
|---|---|---|---|---|---|
| Abies holophylla | Holophyllin-D | C6 glioma cells | 20 μM | Diterpenes compound holophyllin D shows neuroprotective potential in C6 glioma cells by inducing nerve growth factor | [25] |
| Araucara angustifolia | Catechin, epicatechin and rutin | Rat | 10 mg/mL | AAE has antioxidant and neuroprotective properties as it decreases the TBARS levels, CAT activity and NO production in the hippocampus region of the brain in rats. | [235] |
| A. angustifolia | Catechin, epicatechin, rutin, quercetin and apigenin | human dopaminergic SH-SY5Y cells | 5 μg/mL | Decrease in the production of neuron (ROS) and lipid peroxidation. | [43] |
| A. angustifolia | Quercetin | cockroach | 200–400 μg/g | Neurotoxicity modulates the behavior of insects by altering the dopaminergic pathways, as quercetin has the ability to induce selective inhibitory actions on NMDA and GABA receptors and inhibit the enzyme acetylcholinesterase (AChE) | [236] |
| Cedrus deodara | Cedrin | PC12 cells | 0.1, 1 and 10 μM | PC12 cells injured by amyloid β1–42 can be improved by cedrin. Cedrin can reduce (ROS) overproduction, enhance the activity of SOD and decrease MDA content and inhibition of oxidative stress, improvement of mitochondrial dysfunction and suppression of apoptosis in PC12 cells | [237] |
| Metasequoia glyptostroboides | Gallic acid, rutin, myricetin, kaempferol, quercitrin, epigallocatechin, epicatechin gallate epigallocatechin gallate and caffeic acid | Neuronal PC12 cells | 2 mg/mL | The extracts effectively reduced the hydrogen peroxide-induced lipid peroxidation in neuronal PC12 cells by decreasing intracellular ROS accumulation | [49] |
| Pinus densiflora | Catechin, quercetin dehydrate, astragalin and kaempferol | Mice | 50–100 mg/kg | Catechin displayed a potential effect protecting mouse brains from oxidative damage via the improvement of the antioxidant capacities of TAC, the GSH-redox system, SOD and CAT in the hippocampus region as well as the inactivation of cytokines such as NF-kB in pyramidal cells of the hippocampal CA1 region, while PNE shows antiamnesic properties and effects in Alzheimer’s, as it attenuated the increase in serum corticosterone level and up-regulation of GR hippocampal gene expression | [238,239] |
| P. eldarica | Needle extract | Mice | 50 mg/kg | Alkanes, sterols, terpenoids, and quercetin, which is found in P. eldarica, help in inducing sleep and alter the sleep–wake cycle partly via activation of GABA receptors | [240] |
| P. massoniana | Polyprenols | Mice | 25 mg/kg | Polyprenols significantly increased T-AOC, GSHPx, damaging peroxide components from cells in order to stop the lipid peroxidation chain reaction and avoid excessive hydrolysis to form NEP, MDA, SOD activity (remove free radicals) and β-site AβPP cleaving enzyme 1 (BACE1) expression, while NOS activity, MDA concentration, NO, concentration of Aβ1-42 and PS1 were reduced | [241] |
| P. pinaster | Pycnogenol (PYC) | Mice | 20 mg/kg | In the MPTP-induced mouse model, PYC could prevent dopaminergic neurons by reducing oxidative loads, suppressing glial cell activation, and inhibiting inflammatory responses | [100,242] |
| P. roxburghii | Quercetin, rutin, gallic acid | Wistar albino | 100–300 mg/kg | Quercetin and gallic acid, both present in stem bark, have been shown to inhibit neuronal toxicity and apoptosis by reversing mitochondrial dysfunction and free radical development | [243] |
| Thuja occidentalis | Water extract | Mice | 100 mg/kg | CNS depressant activity, anticonvulsant and muscle relaxant activity | [244] |
| Torreya nucifera, | Arctigenin | Rat Cortical cells | 0.01 µM to 10.0 µM. | Arctigenin significantly attenuated glutamate-induced neurotoxicity by inhibiting the binding of [3H]-kainate to its receptors | [35] |
| T. semen | Polyphenols, flavonoids | Mice | 0–10 mg/mL | TS increased the level of total glutathiones | [245] |
| Conifer spp. | Part Used | Compounds | Model | Induction of Diabetes | Dose. Conc | Effects | References |
|---|---|---|---|---|---|---|---|
| Araucaria cunninghamii | Seeds | Glucomannan | Albino wistar rats | Streptozotocin | 25 and 50 mg/kg |
| [246] |
| Cedrus deodara | Heart wood | Flavonoids | Wistar albino rat | Alloxan | 500 mg/kg |
| [247] |
| Juniperus communis | Berry | Flavonoids | Wistar rat | Streptozotocin | 250 mg/kg |
| [248] |
| J. communis | NR | NR | Rat | Streptozotocin-nicotinamide | 100–200 mg/kg |
| [249] |
| J. oxycedrus | Leaves | Linolenic acid, oleic acid | Wistar-albino rats | Streptozotocin | 500 and 1000 mg/kg doses |
| [250] |
| Pinus gerardiana | Nut | Flavonoids | Rat | Alloxan | 250, 500, and 750 mg/kg |
| [251] |
| P. halepensis | Bark | Phenolic compounds | Rat | Glucose | 250, 500 mg/kg |
| [151] |
| P. pinaster | Bark | Phenolic compounds | NR | IC50 (µg/mL) at PB 70% and PB 50% |
| [39] | |
| α-amylase | 254.2 | ||||||
| β-Glucosidase | 122.7 | ||||||
| P. pinaster | Bark | Pynogenol | Human | NR | 100 mg |
| [252] |
| P. roxburghii | Bark | Quercetin | NR | Alpha amylase inhibitory activity | 100 μg/mL |
| [171] |
| P. roxburghii | Bark | Quercetin | Rat | Alloxan | 100, 300 and 500 mg/kg |
| [253] |
| Taxus cuspidata | Branches and leaves | Water | Polysaccharides Pe4 (arabinose, galactose, glucose, xylose, mannose) | NR | 10–120 μg/mL |
| [172] |
5.6. Larvicidal Activity
5.7. Cardiovascular Diseases
6. Clinical Trials
| Plant | Part Used | Trade Name | Phytochemicals Composition | Formulation | Dose/Duration | Product |
|---|---|---|---|---|---|---|
| Pinus pinaster | Bark | Pycnogenol | Catechin, taxifolin, procyanidins, caffeic, p-hydroxybenzoic, ferulic, acids | Tablets, liquids, chewing gums, gels, ointments, capsules or lotions | 150 mg/day for 6 months |  |
| P. pinaster | Bark | Oligopin | Caffeic acid, catechin, epicatechin, taxifolin and ferulic acid | Capsules | 150 mg for 10 days |  |
| P. radiata | Bark | Enzogenol | Flavanoids, proanthocyanidins | Tablets | 480–960 mg/day for 5–6 months |  |
| Picea abies | Needles | Ropren | Flavanoids | Tablets, capsules, lotions | 8.6 mg/kg for 28 days |  |
| Taxus brevifolia | Bark | Taxol | Paclitaxel | Injections | 30 mg/m2 every 3 weeks |  |
| P. massiona | Bark | Not found | Polyphenols, flavanoids, proanthocyanidins | Capsules | 1 capsule daily |  |
7. Phytotoxic Effects of Conifer Extract
8. Conclusion and Future Trends
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mustafa, G.; Arif, R.; Atta, A.; Sharif, S.; Jamil, A. Bioactive Compounds from Medicinal Plants and Their Importance in Drug Discovery in Pakistan. Matrix Sci. Pharma 2017, 1, 17–26. [Google Scholar] [CrossRef]
- Abdel-Razek, A.S.; El-Naggar, M.E.; Allam, A.; Morsy, O.M.; Othman, S.I. Microbial natural products in drug discovery. Processes 2020, 8, 470. [Google Scholar] [CrossRef] [Green Version]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The traditional medicine and modern medicine from natural products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nisar, B.; Sultan, A.; Rubab, S.L. Comparison of Medicinally Important Natural Products versus Synthetic Drugs-A Short Commentary. Nat. Prod. Chem. Res. 2018, 06, 308. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M.; Snader, K.M. Natural products as sources of new drugs over the period 1981–2002. J. Nat. Prod. 2003, 66, 1022–1037. [Google Scholar] [CrossRef]
- Galm, U.; Shen, B. Natural Product Drug Discovery: The Times Have Never Been Better. Chem. Biol. 2007, 14, 1098–1104. [Google Scholar] [CrossRef] [Green Version]
- Akaberi, M.; Boghrati, Z.; Amiri, M.S.; Khayyat, M.H.; Emami, S.A. A Review of Conifers in Iran: Chemistry, Biology and their Importance in Traditional and Modern Medicine. Curr. Pharm. Des. 2020, 26, 1584–1613. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, K.; Islam, M.T.; Jayasena, V.; Sharma, B.; Sharma, S.; Sharma, P.; Kuča, K.; Bhardwaj, P. Review on essential oils, chemical composition, extraction, and utilization of some conifers in Northwestern Himalayas. Phyther. Res. 2020, 34, 2889–2910. [Google Scholar] [CrossRef]
- Bhardwaj, K.; Dhanjal, D.S.; Sharma, A.; Nepovimova, E.; Kalia, A.; Thakur, S.; Bhardwaj, S.; Chopra, C.; Singh, R.; Verma, R.; et al. Conifer-derived metallic nanoparticles: Green synthesis and biological applications. Int. J. Mol. Sci. 2020, 21, 9028. [Google Scholar] [CrossRef]
- Conifers. Available online: https://portals.iucn.org/library/sites/library/files/documents/1999-024.pdf (accessed on 15 March 2021).
- Farjon, A. The Kew Review Conifers of the World. Kew Bull. 2018, 5974, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Kopaczyk, J.M.; Warguła, J.; Jelonek, T. The variability of terpenes in conifers under developmental and environmental stimuli. Environ. Exp. Bot. 2020, 180, 104197. [Google Scholar] [CrossRef]
- Virjamo, V.; Fyhrquist, P.; Koskinen, A.; Lavola, A.; Nissinen, K.; Julkunen-Tiitto, R. 1,6-Dehydropinidine Is an Abundant Compound in Picea abies (Pinaceae) Sprouts and 1,6-Dehydropinidine Fraction Shows Antibacterial Activity against Streptococcus equi Subsp. equi. Molecules 2020, 25, 4558. [Google Scholar] [CrossRef] [PubMed]
- Mill, R.R.; Chase, M.W. A new classification and linear sequence of extant gymnosperms. Phytotaxa 2011, 19, 55–70. [Google Scholar]
- Sharma, A.; Sharma, L.; Goyal, R. A review on himalayan pine species: Ethnopharmacological, phytochemical and pharmacological aspects. Pharmacogn. J. 2018, 10, 611–619. [Google Scholar] [CrossRef] [Green Version]
- Tiberi, R.; Niccoli, A.; Curini, M.; Epifano, F.; Marcotullio, M.C.; Rosati, O. The role of the monoterpene composition in Pinus spp. needles, in host selection by the pine processionary caterpillar, Thaumetopoea pityocampa. Phytoparasitica 1999, 27, 263–272. [Google Scholar] [CrossRef]
- Naser, B.; Bodinet, C.; Tegtmeier, M.; Lindequist, U. Thuja occidentalis (Arbor vitae): A review of its pharmaceutical, pharmacological and clinical properties. Evid. Based Complement. Altern. Med. 2005, 2, 69–78. [Google Scholar] [CrossRef] [Green Version]
- St-Pierre, A.; Blondeau, D.; Bourdeau, N.; Bley, J.; Desgagné-Penix, I. Chemical Composition of Black Spruce (Picea mariana) Bark Extracts and Their Potential as Natural Disinfectant. Ind. Biotechnol. 2019, 15, 219–231. [Google Scholar] [CrossRef] [Green Version]
- Rafieian-kopaei, M.; Suleimani dehkordi, I.; Ghanadian, M.; Shokrollahi, A.; Aghaei, M.; Ayatollahi, S.A.; Choudhary, M.I. Bioactivity-guided isolation of new antiproliferative compounds from Juniperus foetidissima Willd. Nat. Prod. Res. 2016, 30, 1927–1933. [Google Scholar] [CrossRef]
- Barnawi, I.O.; Nasr, F.A.; Noman, O.M.; Alqahtani, A.S.; Al-Zharani, M.; Alotaibi, A.A.; Daradka, H.M.; Al-Mishari, A.A.; Alobaid, W.A.; Alqahtani, A.; et al. Induction of apoptosis and cell cycle arrest by chloroform fraction of Juniperus phoenicea and chemical constituents analysis. Open Chem. 2021, 19, 119–127. [Google Scholar] [CrossRef]
- Osuna-Torres, L.; García-Martí, X.; Ventura-Zapata, E.; López-Upton, J.; Zamilpa-Alvarez, A.; González-Cortazar, M.; Herrera-Ruiz, M.; Tapia-Barrera, N. Taxus globosa Schltdl. (Mexican yew) and Taxus baccata L. (European yew): Intra and interspecies analysis of taxol content and biological activity according to different sources. For. Syst. 2015, 24, 16. [Google Scholar] [CrossRef] [Green Version]
- Qayum, M.; Nisar, M.; Shah, M.R.; Adhikari, A.; Kaleem, W.A.; Khan, I.; Khan, N.; Gul, F.; Khan, I.A.; Zia-Ul-Haq, M.; et al. Analgesic and antiinflammatory activities of taxoids from Taxus wallichiana Zucc. Phyther. Res. 2012, 26, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Kusumoto, N.; Aburai, N.; Ashitani, T.; Takahashi, K.; Kimura, K. Pharmacological Prospects of Oxygenated Abietane-Type Diterpenoids from Taxodium distichum Cones. Adv. Biol. Chem. 2014, 04, 109–115. [Google Scholar] [CrossRef] [Green Version]
- Muto, N.; Tomokuni, T.; Haramoto, M.; Tatemoto, H.; Nakanishi, T.; Inatomi, Y.; Murata, H.; Inada, A. Isolation of apoptosis- and differentiation-inducing substances toward human promyelocytic leukemia HL-60 cells from leaves of Juniperus taxifolia. Biosci. Biotechnol. Biochem. 2008, 72, 477–484. [Google Scholar] [CrossRef] [Green Version]
- Kim, C.S.; Subedi, L.; Kim, S.Y.; Choi, S.U.; Kim, K.H.; Lee, K.R. Diterpenes from the Trunk of Abies holophylla and Their Potential Neuroprotective and Anti-inflammatory Activities. J. Nat. Prod. 2016, 79, 387–394. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, V.K.; Sharma, A.; Kang, S.C.; Baek, K.H. Antioxidant, lipid peroxidation inhibition and free radical scavenging efficacy of a diterpenoid compound sugiol isolated from Metasequoia glyptostroboides. Asian Pac. J. Trop. Med. 2014, 7, 9–15. [Google Scholar] [CrossRef] [Green Version]
- Tavares, W.; Seca, A. The Current Status of the Pharmaceutical Potential of Juniperus L. Metabolites. Medicines 2018, 5, 81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tawara, J.N.; Blokhin, A.; Foderaro, T.A.; Stermitz, F.R.; Hope, H. Toxic Piperidine Alkaloids from Pine (Pinus) and Spruce (Picea) Trees. New Structures and a Biosynthetic Hypothesis. J. Org. Chem. 1993, 58, 4813–4818. [Google Scholar] [CrossRef]
- Küpeli, E.; Erdemoǧlu, N.; Yeşilada, E.; Şener, B. Anti-inflammatory and antinociceptive activity of taxoids and lignans from the heartwood of Taxus baccata L. J. Ethnopharmacol. 2003, 89, 265–270. [Google Scholar] [CrossRef]
- Juyal, D.; Thawani, V.; Thaledi, S.; Joshi, M. Ethnomedical properties of Taxus wallichiana Zucc. (Himalayan yew). J. Tradit. Complement. Med. 2014, 4, 159–161. [Google Scholar] [CrossRef] [Green Version]
- Hafezi, K.; Hemmati, A.A.; Abbaszadeh, H.; Valizadeh, A.; Makvandi, M. Anticancer activity and molecular mechanisms of α-conidendrin, a polyphenolic compound present in Taxus yunnanensis, on human breast cancer cell lines. Phyther. Res. 2020, 34, 1397–1408. [Google Scholar] [CrossRef]
- Ivanova, D.I.; Tashev, A.N.; Nedialkov, P.T.; Ilieva, Y.E.; Atanassova, T.N.; Olech, M.; Nowak, R.; Angelov, G.; Tsvetanova, F.V.; Iliev, I.A.; et al. Antioxidant and antiproliferative activity of Juniperus L. Species of Bulgarian and foreign origin and their anticancer metabolite identification. Bulg. Chem. Commun. 2018, 50, 144–150. [Google Scholar]
- Kanchan, B.; Prerna, B.; Simran, K. Medicinal value of secondary metabolites of pines grown in Himalayan region of India. Res. J. Biotechnol. 2020, 15, 131–140. [Google Scholar]
- Singh, S.K.; Shanmugavel, M.; Kampasi, H.; Singh, R.; Mondhe, D.M.; Rao, J.M.; Adwankar, M.K.; Saxena, A.K.; Qazi, G.N. Chemically standardized isolates from Cedrus deodara stem wood having anticancer activity. Planta Med. 2007, 73, 519–526. [Google Scholar] [CrossRef]
- Jang, Y.P.; Kim, S.R.; Choi, Y.H.; Kim, J.; Kim, S.G.; Markelonis, G.J.; Oh, T.H.; Kim, Y.C. Arctigenin protects cultured cortical neurons from glutamate-induced neurodegeneration by binding to kainate receptor. J. Neurosci. Res. 2002, 68, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Asmi, K.S.; Lakshmi, T.; Balusamy, S.R.; Parameswari, R. Therapeutic aspects of taxifolin—An update. J. Adv. Pharm. Educ. Res. 2017, 7, 187–189. [Google Scholar]
- Hammerbacher, A.; Kandasamy, D.; Ullah, C.; Schmidt, A.; Wright, L.P.; Gershenzon, J. Flavanone-3-hydroxylase plays an important role in the biosynthesis of spruce phenolic defenses against bark beetles and their fungal associates. Front. Plant Sci. 2019, 10, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Michael, H.N.; Awad, H.M.; El-Sayed, N.H.; Paré, P.W. Chemical and antioxidant investigations: Norfolk pine needles (Araucaria excelsa). Pharm. Biol. 2010, 48, 534–538. [Google Scholar] [CrossRef]
- Ferreira-Santos, P.; Genisheva, Z.; Botelho, C.; Santos, J.; Ramos, C.; Teixeira, J.A.; Rocha, C.M.R. Unravelling the biological potential of Pinus pinaster bark extracts. Antioxidants 2020, 9, 334. [Google Scholar] [CrossRef] [Green Version]
- Gascón, S.; Jiménez-Moreno, N.; Jiménez, S.; Quero, J.; Rodríguez-Yoldi, M.J.; Ancín-Azpilicueta, C. Nutraceutical composition of three pine bark extracts and their antiproliferative effect on Caco-2 cells. J. Funct. Foods 2018, 48, 420–429. [Google Scholar] [CrossRef]
- Dziedzinski, M.; Kobus-Cisowska, J.; Szymanowska, D.; Stuper-Szablewska, K.; Baranowska, M. Identification of polyphenols from coniferous shoots as natural antioxidants and antimicrobial compounds. Molecules 2020, 25, 3527. [Google Scholar] [CrossRef]
- Fierascu, I.; Ungureanu, C.; Avramescu, S.M.; Cimpeanu, C.; Georgescu, M.I.; Fierascu, R.C.; Ortan, A.; Sutan, A.N.; Anuta, V.; Zanfirescu, A.; et al. Genoprotective, antioxidant, antifungal and anti-inflammatory evaluation of hydroalcoholic extract of wild-growing Juniperus communis L. (Cupressaceae) native to Romanian southern sub-Carpathian hills. BMC Complement. Altern. Med. 2018, 18, 1–15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Branco, C.S.; Duong, A.; Machado, A.K.; Wu, A.; Scola, G.; Andreazza, A.C.; Salvador, M. Araucaria angustifolia (Bertol.) Kuntze has neuroprotective action through mitochondrial modulation in dopaminergic SH-SY5Y cells. Mol. Biol. Rep. 2019, 46, 6013–6025. [Google Scholar] [CrossRef]
- Nisar, M.; Khan, I.; Ahmad, B.; Ali, I.; Ahmad, W.; Choudhary, M.I. Antifungal and antibacterial activities of Taxus wallichiana Zucc. J. Enzym. Inhib. Med. Chem. 2008, 23, 256–260. [Google Scholar] [CrossRef] [Green Version]
- Freitas, A.M.; Almeida, M.T.R.; Andrighetti-Fröhner, C.R.; Cardozo, F.T.G.S.; Barardi, C.R.M.; Farias, M.R.; Simões, C.M.O. Antiviral activity-guided fractionation from Araucaria angustifolia leaves extract. J. Ethnopharmacol. 2009, 126, 512–517. [Google Scholar] [CrossRef]
- Al-Sayed, E.; Gad, H.A.; El-Shazly, M.; Abdel-Daim, M.M.; Nasser Singab, A. Anti-inflammatory and analgesic activities of cupressuflavone from Cupressus macrocarpa: Impact on pro-inflammatory mediators. Drug Dev. Res. 2018, 79, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Ferrentino, G.; Haman, N.; Morozova, K.; Tonon, G.; Scampicchio, M. Phenolic compounds extracted from spruce (Picea abies) by supercritical carbon dioxide as antimicrobial agents against gram-positive bacteria assessed by isothermal calorimetry. J. Therm. Anal. Calorim. 2020. [Google Scholar] [CrossRef]
- Hoon, L.Y.; Choo, C.; Watawana, M.I.; Jayawardena, N.; Waisundara, V.Y. Evaluation of the total antioxidant capacity and antioxidant compounds of different solvent extracts of Chilgoza Pine nuts (Pinus gerardiana). J. Funct. Foods 2015, 18, 1014–1021. [Google Scholar] [CrossRef]
- Lee, S.J.; Lee, S.Y.; Hur, S.J.; Bae, Y., II; Jeong, C.H. Neuroprotective and antioxidant effects of Metasequoia glyptostroboides leaf extract. Curr. Top. Nutraceutical Res. 2016, 14, 67–72. [Google Scholar]
- Sahin Yaglioglu, A.; Eser, F. Screening of some Juniperus extracts for the phenolic compounds and their antiproliferative activities. S. Afr. J. Bot. 2017, 113, 29–33. [Google Scholar] [CrossRef]
- Legault, J.; Girard-Lalancette, K.; Dufour, D.; Pichette, A. Antioxidant potential of bark extracts from boreal forest conifers. Antioxidants 2013, 2, 77–89. [Google Scholar] [CrossRef]
- Lantto, T.A.; Colucci, M.; Závadová, V.; Hiltunen, R.; Raasmaja, A. Cytotoxicity of curcumin, resveratrol and plant extracts from basil, juniper, laurel and parsley in SH-SY5Y and CV1-P cells. Food Chem. 2009, 117, 405–411. [Google Scholar] [CrossRef]
- Välimaa, A.L.; Raitanen, J.E.; Tienaho, J.; Sarjala, T.; Nakayama, E.; Korpinen, R.; Mäkinen, S.; Eklund, P.; Willför, S.; Jyske, T. Enhancement of Norway spruce bark side-streams: Modification of bioactive and protective properties of stilbenoid-rich extracts by UVA-irradiation. Ind. Crops Prod. 2020, 145, 112150. [Google Scholar] [CrossRef]
- Raiber, S.; Schröder, G.; Schröder, J. Molecular and enzymatic characterization of two stilbene synthases from Eastern white pine (Pinus strobus) A single Arg/His difference determines the activity and the pH dependence of the enzymes. FEBS Lett. 1995, 361, 299–302. [Google Scholar] [CrossRef] [Green Version]
- Hovelstad, H.; Leirset, I.; Oyaas, K.; Fiksdahl, A. Screening analyses of pinosylvin stilbenes, resin acids and lignans in Norwegian conifers. Molecules 2006, 11, 103–114. [Google Scholar] [CrossRef] [Green Version]
- Francezon, N.; Meda, N.S.B.R.; Stevanovic, T. Optimization of bioactive polyphenols extraction from Picea mariana bark. Molecules 2017, 22, 2118. [Google Scholar] [CrossRef] [Green Version]
- Latva-Mäenpää, H. Bioactive and Protective Polyphenolics From Roots and Stumps of Conifer Trees (Norway Spruce and Scots Pine); Helsingin Yliopisto: Helsinki, Finland, 2017; ISBN 9789515134653. [Google Scholar]
- Brodribb, T.J.; Pittermann, J.; Coomes, D.A. Elegance versus speed: Examining the competition between conifer and angiosperm trees. Int. J. Plant Sci. 2012, 173, 673–694. [Google Scholar] [CrossRef] [Green Version]
- Larter, M. Evolution de la Résistance à la Cavitation Chez les Conifères the Evolution of Cavitation Resistance in Conifers. Ph.D. Thesis, Université de Bordeaux, Bordeaux, France, 2016. [Google Scholar]
- Moriguchi, Y.; Ueno, S.; Hasegawa, Y.; Tadama, T.; Watanabe, M.; Saito, R.; Hirayama, S.; Iwai, J.; Konno, Y. Marker-assisted selection of trees with MALE STERALITY 1 in Cryptomeria japonica D. Don. bioRxiv 2020, 1–10. [Google Scholar] [CrossRef]
- Hussein, R.A.; El-Anssary, A.A. Plants Secondary Metabolites: The Key Drivers of the Pharmacological Actions of Medicinal Plants. Herb. Med. 2019, 1, 13. [Google Scholar] [CrossRef] [Green Version]
- Singh, B.; Sharma, R.A. Plant terpenes: Defense responses, phylogenetic analysis, regulation and clinical applications. 3 Biotech 2015, 5, 129–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Porres-Martínez, M.; González-Burgos, E.; Carretero, M.E.; Pilar Gómez-Serranillos, M. In vitro neuroprotective potential of the monoterpenes α-pinene and 1,8-cineole against H2O2-induced oxidative stress in PC12 cells. Z. fur Naturforsch. Sect. C J. Biosci. 2016, 71, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Da Silveira E Sá, R.D.C.; Andrade, L.N.; De Sousa, D.P. Sesquiterpenes from essential oils and anti-inflammatory activity. Nat. Prod. Commun. 2015, 10, 1767–1774. [Google Scholar] [CrossRef] [Green Version]
- Dey, P.; Kundu, A.; Kumar, A.; Gupta, M.; Lee, B.M.; Bhakta, T.; Dash, S.; Kim, H.S. Analysis of Alkaloids (Indole Alkaloids, Isoquinoline Alkaloids, Tropane Alkaloids); Elsevier Inc.: Amsterdam, The Netherlands, 2020; ISBN 9780128164556. [Google Scholar]
- Thawabteh, A.; Juma, S.; Bader, M.; Karaman, D.; Scrano, L.; Bufo, S.A.; Karaman, R. The biological activity of natural alkaloids against herbivores, cancerous cells and pathogens. Toxins 2019, 11, 656. [Google Scholar] [CrossRef] [Green Version]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef]
- Tanase, C.; Boz, I.; Stingu, A.; Volf, I.; Popa, V.I. Physiological and biochemical responses induced by spruce bark aqueous extract and deuterium depleted water with synergistic action in sunflower (Helianthus annuus L.) plants. Ind. Crops Prod. 2014, 60, 160–167. [Google Scholar] [CrossRef]
- Tanase, C.; Cosarcă, S.; Muntean, D.L. A critical review of phenolic compounds extracted from the bark of woody vascular plants and their potential biological activity. Molecules 2019, 24, 1182. [Google Scholar] [CrossRef] [Green Version]
- El Omari, N.; Ezzahrae Guaouguaou, F.; El Menyiy, N.; Benali, T.; Aanniz, T.; Chamkhi, I.; Balahbib, A.; Taha, D.; Shariati, M.A.; Zengin, G.; et al. Phytochemical and biological activities of Pinus halepensis mill., and their ethnomedicinal use. J. Ethnopharmacol. 2021, 268, 113661. [Google Scholar] [CrossRef]
- Metsämuuronen, S.; Sirén, H. Bioactive phenolic compounds, metabolism and properties: A review on valuable chemical compounds in Scots pine and Norway spruce. Phytochem. Rev. 2019, 18, 623–664, ISBN 0123456789. [Google Scholar]
- Rodríguez-García, C.; Sánchez-Quesada, C.; Gaforio, J.J.; Gaforio, J.J. Dietary flavonoids as cancer chemopreventive agents: An updated review of human studies. Antioxidants 2019, 8, 137. [Google Scholar] [CrossRef] [Green Version]
- Tsao, R. Chemistry and Biochemistry of Dietary Polyphenols. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Nanda, S.; Mohanty, J.N.; Mishra, R.; Joshi, R.K. Metabolic Engineering of Phenylpropanoids in Plants. In Transgenesis and Secondary Metabolism; Springer: Berlin/Heidelberg, Germany, 2017; pp. 485–510. [Google Scholar] [CrossRef]
- Saleem, M.; Kim, J.; Ali, S.; Sup, Y. An update on bioactive plant lignans. Nat. Prod. Rep. 2005, 22, 696–716. [Google Scholar] [CrossRef]
- García-Pérez, M.E.; Royer, M.; Herbette, G.; Desjardins, Y.; Pouliot, R.; Stevanovic, T. Picea mariana bark: A new source of trans-resveratrol and other bioactive polyphenols. Food Chem. 2012, 135, 1173–1182. [Google Scholar] [CrossRef]
- Salminen, J.; Karonen, M. Chemical ecology of tannins and other phenolics: We need a change in approach. Br. Ecol. Soc. 2011, 25, 325–338. [Google Scholar] [CrossRef]
- Raitanen, J.E.; Järvenpää, E.; Korpinen, R.; Mäkinen, S.; Hellström, J.; Kilpeläinen, P.; Liimatainen, J.; Ora, A.; Tupasela, T.; Jyske, T. Tannins of conifer bark as Nordic piquancy—sustainable preservative and aroma? Molecules 2020, 25, 567. [Google Scholar] [CrossRef] [Green Version]
- Koche, D.; Shirsat, R.; Kawale, M. An overview of major classes of phytochemicals: Their type and role in disease prevention. Hislopia J. 2016, 9, 2016. [Google Scholar]
- Prothmann, J.; Sun, M.; Spégel, P.; Sandahl, M.; Turner, C.; Scheuba, J.; Wronski, V.K.; Rollinger, J.M.; Grienke, U.; Santos-Buelga, C.; et al. Relationship between phenolic compounds, anthocyanins content and antioxidant activity in colored barley germplasm. J. Agric. Food Chem. 2017, 53, 1713. [Google Scholar]
- Matthews, S.; Mila, I.; Scalbert, A.; Donnelly, D.M.X. Extractable and non-extractable proanthocyanidins in barks. Phytochemistry 1997, 45, 405–410. [Google Scholar] [CrossRef]
- Koleckar, V.; Kubikova, K.; Rehakova, Z.; Kuca, K.; Jun, D.; Jahodar, L.; Opletal, L. Condensed and Hydrolysable Tannins as Antioxidants Influencing the Health. Mini-Rev. Med. Chem. 2008, 8, 436–447. [Google Scholar] [CrossRef]
- De Bruyne, T.; Pieters, L.; Deelstra, H.; Vlietinck, A. Condensed vegetable tannins: Biodiversity in structure and biological activities. Biochem. Syst. Ecol. 1999, 27, 445–459. [Google Scholar] [CrossRef]
- Scalbert, A. Antimicrobial properties of tannins. Phytochemistry 1991, 30, 3875–3883. [Google Scholar] [CrossRef]
- Bhangale, J.O.; Acharya, S.R. Anti-Parkinson Activity of Petroleum Ether Extract of Ficus religiosa (L.) Leaves. Adv. Pharmacol. Sci. 2016, 2016, 9436106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brijesh, K.; Ruchi, R.; Sanjita, D.; Saumya, D.; June, A. Phytoconstituents and Therapeutic potential of Thuja occidentalis. Res. J. Pharm. Biol. Chem. Sci. 2012, 3, 354–362. [Google Scholar]
- Shuaib, M.; Ali, M.; Ahamad, J.; Naquvi, K.J.; Ahmad, M.I. Pharmacognosy of Pinus roxburghii: A Review. Phytochemistry 2006, 2, 262–268. [Google Scholar]
- Poudel, R.C.; Gao, L.M.; Möller, M.; Baral, S.R.; Uprety, Y.; Liu, J.; Li, D.Z. Yews (Taxus) along the Hindu Kush-Himalayan region: Exploring the ethnopharmacological relevance among communities of Mongol and Caucasian origins. J ethnopharmacol. 2013, 147, 190–203. [Google Scholar] [CrossRef] [PubMed]
- Kunwar, R.M.; Shrestha, K.P.; Bussmann, R.W. Traditional herbal medicine in Far-west Nepal: A pharmacological appraisal. J. Ethnobiol. Ethnomed. 2010, 6, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Sharma, H.; Garg, M. A review of traditional use, phytoconstituents and biological activities of Himalayan yew, Taxus wallichiana. J. Integr. Med. 2015, 13, 80–90. [Google Scholar] [CrossRef]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxid. Med. Cell. Longev. 2016, 2016, 1245049. [Google Scholar] [CrossRef] [PubMed]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free Radicals: Properties, Sources, Targets, and Their Implication in Various Diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, H.; Bhardwaj, K.; Nepovimova, E.; Kuča, K.; Dhanjal, D.S.; Bhardwaj, S.; Bhatia, S.K.; Verma, R.; Kumar, D. Antioxidant functionalized nanoparticles: A combat against oxidative stress. Nanomaterials 2020, 10, 1334. [Google Scholar] [CrossRef]
- Collin, F. Chemical basis of reactive oxygen species reactivity and involvement in neurodegenerative diseases. Int. J. Mol. Sci. 2019, 20, 2407. [Google Scholar] [CrossRef] [Green Version]
- Genestra, M. Oxyl radicals, redox-sensitive signalling cascades and antioxidants. Cell. Signal. 2007, 19, 1807–1819. [Google Scholar] [CrossRef]
- Ricordi, C.; Garcia-Contreras, M.; Farnetti, S. Diet and Inflammation: Possible Effects on Immunity, Chronic Diseases, and Life Span. J. Am. Coll. Nutr. 2015, 34, 10–13. [Google Scholar] [CrossRef]
- Boukhenouna, S.; Wilson, M.A.; Bahmed, K.; Kosmider, B. Reactive oxygen species in chronic obstructive pulmonary disease. Oxid. Med. Cell. Longev. 2018, 2018, 5730395. [Google Scholar] [CrossRef]
- Packer, L.; Rimbach, G.; Virgili, F. Antioxidant activity and biologic properties of a procyanidin-rich extract from pine (Pinus maritima) bark, pycnogenol. Free Radic. Biol. Med. 1999, 27, 704–724. [Google Scholar] [CrossRef]
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. [Google Scholar] [CrossRef] [Green Version]
- Iravani, S.; Zolfaghari, B. Pharmaceutical and nutraceutical effects of Pinus pinaster bark extract. Res. Pharm. Sci. 2011, 6, 1–11. [Google Scholar]
- Senthilmohan, S.T.; Zhang, J.; Stanley, R.A. Effects of flavonoid extract Enzogenol with vitamin C on protein oxidation and DNA damage in older human subjects. Nutr. Res. 2003, 23, 1199–1210. [Google Scholar] [CrossRef]
- Azqueta, A.; Collins, A. Polyphenols and DNA damage: A mixed blessing. Nutrients 2016, 8, 785. [Google Scholar] [CrossRef]
- Kukreja, A.; Wadhwa, N. Therapeutic Role of Resveratrol and Piceatannol in Disease Prevention. J. Blood Disord. Transfus. 2014, 5, 9. [Google Scholar] [CrossRef] [Green Version]
- Sharma, A.; Goyal, R.; Sharma, L. Potential biological efficacy of Pinus plant species against oxidative, inflammatory and microbial disorders. BMC Complement. Altern. Med. 2016, 16, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Azab, A.; Nassar, A.; Azab, A.N. Anti-inflammatory activity of natural products. Molecules 2016, 21, 1321. [Google Scholar] [CrossRef]
- Artis, D.; Spits, H. The biology of innate lymphoid cells. Nature 2015, 517, 293–301. [Google Scholar] [CrossRef]
- Fernandes, J.V.; Cobucci, R.N.O.; Jatobá, C.A.N.; de Medeiros Fernandes, T.A.A.; de Azevedo, J.W.V.; de Araújo, J.M.G. The Role of the Mediators of Inflammation in Cancer Development. Pathol. Oncol. Res. 2015, 21, 527–534. [Google Scholar] [CrossRef]
- Heppner, F.L.; Ransohoff, R.M.; Becher, B. Immune attack: The role of inflammation in Alzheimer disease. Nat. Rev. Neurosci. 2015, 16, 358–372. [Google Scholar] [CrossRef]
- Rock, K.L.; Rock, K.L. Innate and adaptive immune responses to cell death. Immunol. Rev. 2011, 243, 191–205. [Google Scholar] [CrossRef] [Green Version]
- Waisman, A.; Liblau, R.S.; Becher, B. Innate and adaptive immune responses in the CNS. Lancet Neurol. 2015, 14, 945–955. [Google Scholar] [CrossRef]
- Vignali, D.A.A.; Kuchroo, V.K. Review IL-12 family cytokines: Immunological playmakers. Nat. Immunol. 2012, 13, 722–728. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montgomery, S.L.; Bowers, W.J. Tumor Necrosis Factor-alpha and the Roles it Plays in Homeostatic and Degenerative Processes Within the Central Nervous System. J. Neuroimmune Pharmacol. 2012, 7, 42–59. [Google Scholar] [CrossRef] [PubMed]
- Fenton, M.J. Review: Transcriptional and post-transcriptional regulation of interleukin 1 gene expression. Int. J. Immunopharm. 1992, 14, 401–411. [Google Scholar] [CrossRef]
- Rider, P.; Carmi, Y.; Voronov, E.; Apte, R.N. Interleukin-1α. Semin. Immunol. 2013, 25, 430–438. [Google Scholar] [CrossRef]
- Cha, K.-J. The Anti-Inflammatory Effects of Picea wilsonii Mast on HaCaT Cells. Korean J. Clin. Lab. Sci. 2016, 48, 365–370. [Google Scholar] [CrossRef] [Green Version]
- Langrish, C.L.; Mckenzie, B.S.; Wilson, N.J.; Kastelein, R.A.; Cua, D.J. IL-12 and IL-23: Master regulators of innate and adaptive immunity. Immunol. Rev. 2004, 202, 96–105. [Google Scholar] [CrossRef]
- Duvallet, E.; Semerano, L.; Assier, E.; Falgarone, G.; Duvallet, E.; Semerano, L.; Assier, E.; Falgarone, G.; Duvallet, E.; Semerano, L.; et al. Interleukin-23: A key cytokine in inflammatory diseases. Ann. Med. 2011, 3890, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Sabat, R. Cytokine & Growth Factor Reviews IL-10 family of cytokines. Cytokine Growth Factor Rev. 2010, 21, 315–324. [Google Scholar] [CrossRef]
- Ng, T.H.S.; Britton, G.J.; Hill, E.V.; Verhagen, J.; Burton, B.R.; Wraith, D.C. Regulation of adaptive immunity; the role of interleukin-10. Front. Immunol. 2013, 4, 1–14. [Google Scholar] [CrossRef] [Green Version]
- Kwilasz, A.J.; Grace, P.M.; Serbedzija, P.; Maier, S.F.; Watkins, L.R. Neuropharmacology The therapeutic potential of interleukin-10 in neuroimmune diseases. Neuropharmacology 2014, 2, 55–69. [Google Scholar] [CrossRef] [Green Version]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.W.; Lin, L.G.; Ye, W.C. Techniques for extraction and isolation of natural products: A comprehensive review. Chin. Med. 2018, 13, 1–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bucar, F.; Wube, A.; Schmid, M. Natural product isolation-how to get from biological material to pure compounds. Nat. Prod. Rep. 2013, 30, 525–545. [Google Scholar] [CrossRef] [Green Version]
- Gopalasatheeskumar, K. Significant Role of Soxhlet Extraction Process in Phytochemical. Mintage J. Pharm. Med. Sci. 2018, 7, 43–47. [Google Scholar]
- Zhao, Q.Q.; Wang, S.F.; Li, Y.; Song, Q.Y.; Gao, K. Terpenoids with anti-inflammatory activity from Abies chensiensis. Fitoterapia 2016, 111, 87–94. [Google Scholar] [CrossRef]
- Stan, M.S.; Voicu, S.N.; Caruntu, S.; Nica, I.C.; Olah, N.K.; Burtescu, R.; Balta, C.; Rosu, M.; Herman, H.; Hermenean, A.; et al. Antioxidant and anti-inflammatory properties of a Thuja occidentalis mother tincture for the treatment of ulcerative colitis. Antioxidants 2019, 8, 416. [Google Scholar] [CrossRef] [Green Version]
- Kim, D.S.; Kim, M.S.; Kang, S.W.; Sung, H.Y.; Kang, Y.H. Pine bark extract enzogenol attenuated tumor necrosis factor-α- induced endothelial cell adhesion and monocyte transmigration. J. Agric. Food Chem. 2010, 58, 7088–7095. [Google Scholar] [CrossRef]
- Schäfer, A.; Chovanová, Z.; Muchová, J.; Sumegová, K.; Liptáková, A.; Högger, P. Inhibition of COX-1 and COX-2 activity by plasma of human volunteers after ingestion of French maritime pine bark extract (Pycnogenol). Biomed. Pharmacother. 2005, 60, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Latest Global Cancer Data_ Cancer Burden Rises to 18 2018. WHO. Available online: https://www.iarc.who.int/featured-news/latest-global-cancer-data-cancer-burden-rises-to-18-1-million-new-cases-and-9-6-million-cancer-deaths-in-2018/PDF (accessed on 15 February 2021).
- Yan, S.H. An early history of human breast cancer: West meets East. Chin. J. Cancer 2013, 32, 475–477. [Google Scholar] [CrossRef] [PubMed]
- Sudhakar, A. History of Cancer, Ancient and Modern Treatment Methods. J. Cancer Sci. Ther. 2009, 01, i–iv. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, S.K. Giovanni Battista Morgagni (1682–1771): Father of pathologic anatomy and pioneer of modern medicine. Anat. Sci. Int. 2017, 92, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Cancer. Available online: https://www.niehs.nih.gov/health/materials/cancer_and_the_environment_508.pdf (accessed on 20 February 2021).
- Ma, X.; Wang, Z. Anticancer drug discovery in the future: An evolutionary perspective. Drug Discov. Today 2009, 14, 1136–1142. [Google Scholar] [CrossRef]
- Widmer, N.; Bardin, C.; Chatelut, E.; Paci, A.; Beijnen, J.; Levêque, D.; Veal, G.; Astier, A. Review of therapeutic drug monitoring of anticancer drugs part two—Targeted therapies. Eur. J. Cancer 2014, 50, 2020–2036. [Google Scholar] [CrossRef]
- Kuczynski, E.A.; Sargent, D.J.; Grothey, A.; Kerbel, R.S. Drug rechallenge and treatment beyond progression-implications for drug resistance. Nat. Rev. Clin. Oncol. 2013, 10, 571–587. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lichota, A.; Gwozdzinski, K. Anticancer activity of natural compounds from plant and marine environment. Int. J. Mol. Sci. 2018, 19, 3533. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.A.H.; Kim, D.H.; Hossain, M.A.; Kim, M.Y.; Sung, B.; Yoon, J.H.; Suh, H.; Jeong, T.C.; Chung, H.Y.; Kim, N.D. HS-1793, a resveratrol analogue, induces cell cycle arrest and apoptotic cell death in human breast cancer cells. Int. J. Oncol. 2014, 44, 473–480. [Google Scholar] [CrossRef] [Green Version]
- Sharifi-Rad, J.; Ozleyen, A.; Tumer, T.B.; Adetunji, C.O.; El Omari, N.; Balahbib, A.; Taheri, Y.; Bouyahya, A.; Martorell, M.; Martins, N.; et al. Natural products and synthetic analogs as a source of antitumor drugs. Biomolecules 2019, 9, 679. [Google Scholar] [CrossRef] [PubMed]
- Tafrihi, M.; Imran, M.; Tufail, T.; Gondal, T.A.; Caruso, G.; Sharma, S.; Sharma, R.; Atanassova, M.; Atanassov, L.; Valere, P.; et al. The Wonderful Activities of the Genus Mentha: Not Only Antioxidant Properties. Molecules 2021, 26, 1118. [Google Scholar] [CrossRef] [PubMed]
- Birinci, H.; Şen, B.; Sayğılı, S.; Ölmez, E.; Uluer, E.T.; Özbilgin, K. The Effect of Pycnogenol and Paclitaxel on DNA Damage in Human Breast Cancer Cell Line. Proceedings 2017, 1, 1023. [Google Scholar] [CrossRef] [Green Version]
- Dinić, J.; Ríos-Luci, C.; Karpaviciene, I.; Cikotiene, I.; Fernandes, M.X.; Pešić, M.; Padrón, J.M. CKT0353, a novel microtubule targeting agent, overcomes paclitaxel induced resistance in cancer cells. Investig. New Drugs 2020, 38, 584–598. [Google Scholar] [CrossRef] [PubMed]
- Binarová, P.; Tuszynski, J. Tubulin: Structure, Functions and Roles in Disease. Cells 2019, 8, 1294. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Kanakkanthara, A. Beyond the paclitaxel and vinca alkaloids: Next generation of plant-derived microtubule-targeting agents with potential anticancer activity. Cancers 2020, 12, 1721. [Google Scholar] [CrossRef]
- Harshita; Barkat, M.A.; Beg, S.; Pottoo, F.H.; Ahmad, F.J. Nanopaclitaxel therapy: An evidence based review on the battle for next-generation formulation challenges. Nanomedicine 2019, 14, 1323–1341. [Google Scholar] [CrossRef]
- Ganguly, A.; Yang, H.; Cabral, F. Paclitaxel-dependent cell lines reveal a novel drug activity. Mol. Cancer Ther. 2010, 9, 2914–2923. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, S.; Das, A.; Chakraborty, P.; Suthindhiran, K.; Jayasri, M.A. Antioxidant and antimicrobial activity of Araucaria cookii and Brassaia actinophyla. Pak. J. Biol. Sci. 2014, 17, 715–719. [Google Scholar] [CrossRef] [Green Version]
- Jain, S.; Kumar, D.; Malviya, N.; Jain, A.; Jain, S.; Jain, V. Estimation of total phenolic, tannins, and flavonoid contents and antioxidant activity of Cedrus deodara heart wood extracts. Egypt. Pharm. J. 2015, 14, 10. [Google Scholar] [CrossRef]
- Horiba, H.; Nakagawa, T.; Zhu, Q.; Ashour, A.; Watanabe, A.; Shimizu, K. Biological activities of extracts from different parts of cryptomeria japonica. Nat. Prod. Commun. 2016, 11, 1337–1342. [Google Scholar] [CrossRef] [Green Version]
- Bhagat, M.; Gupta, S.; Sudan, R. In vitro Evaluation of Antioxidant Activity of Picea smithiana Growing in Bhaderwah Region of Jammu and Kashmir. Cell. Life Sci. J. 2017, 2. [Google Scholar] [CrossRef]
- Salhi, N.; Bouyahya, A.; El Guourrami, O.; El Jemli, M.; Bourais, I.; Zellou, A.; Cherrah, Y.; El Abbes Faouzi, M. Investigation of in vitro and in vivo antioxidant and antidiabetic activities of Pinus halepensis extracts. J. Herbmed Pharmacol. 2021, 10, 123–131. [Google Scholar] [CrossRef]
- Tekaday, D.; Antony, B.; Jain, S. Antimicrobial, antioxidant and phytochemical investigation of Thuja occidentalis (Arbor vitae) leave extract. GSC Biol. Pharm. Sci. 2020, 12, 108–116. [Google Scholar] [CrossRef]
- Milutinović, M.G.; Stanković, M.S.; Cvetković, D.M.; Topuzović, M.D.; Mihailović, V.B.; Marković, S.D. Antioxidant and anticancer properties of leaves and seed cones from European yew (Taxus baccata L.). Arch. Biol. Sci. 2015, 67, 525–534. [Google Scholar] [CrossRef]
- Bhat, M.A.; Ganie, S.A.; Dar, K.B.; Ali, R.; Hamid, R. In Vitro antioxidant potential and hepatoprotective activity of Taxus Wallichiana. Asian J. Pharm. Clin. Res. 2018, 11, 237–243. [Google Scholar] [CrossRef]
- Subba, B. Analysis of Phytochemical Constituents and Biological Activity of Taxus Wallichiana Zucc. Dolakha District of Nepal. Int. J. Appl. Sci. Biotechnol. 2018, 6, 110–114. [Google Scholar] [CrossRef]
- Yang, X.-W.; Zeng, H.-W.; Liu, X.-H.; Li, S.-M.; Xu, W.; Shen, Y.-H.; Zhang, C.; Zhang, W.-D. Anti-inflammatory and anti-tumour effects of Abies georgei extracts. J. Pharm. Pharmacol. 2008, 60, 937–941. [Google Scholar] [CrossRef]
- Nayak, S.S.; Ghosh, A.K.; Debnath, B.; Vishnoi, S.P.; Jha, T. Synergistic effect of methanol extract of Abies webbiana leaves on sleeping time induced by standard sedatives in mice and anti-inflammatory activity of extracts in rats. J. Ethnopharmacol. 2004, 93, 397–402. [Google Scholar] [CrossRef]
- Bisht, B.; Nainwal, P.; Saini, P. Evaluation of in vitro anti-inflammatory activity of Agathis robusta. J. Pharma. Res. 2012, 2, 1304–1306. [Google Scholar]
- Journal, A.I. An Indian Journal Note. Anal. Chem. 2007, 6, 4–8. [Google Scholar]
- Orhan, N.; Akkol, E.; Ergun, F. Evaluation of antiinflammatory and antinociceptive effects of some juniperus species growing in Turkey. Turk. J. Biol. 2012, 36, 719–726. [Google Scholar] [CrossRef]
- Science, A. Assessment of Anti-Inflammatory Activity of Taxus baccata Linn. Bark Extract Satyajit Dutta * G. Mariappan ** Dipankar Sarkar ** Piyali Sarkar ** Table 1: Effect of Taxus baccata (L) bark extracts on Carrageenan-induced paw edema method in rats. Anc. Sci. Life 2010, 29, 19–21. [Google Scholar]
- Branco, C.D.S.; De Lima, É.D.; Rodrigues, T.S.; Scheffel, T.B.; Scola, G.; Laurino, C.C.F.C.; Moura, S.; Salvador, M. Mitochondria and redox homoeostasis as chemotherapeutic targets of Araucaria angustifolia (Bert.) O. Kuntze in human larynx HEp-2 cancer cells. Chem. Biol. Interact. 2015, 231, 108–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shashi, B.; Jaswant, S.; Madhusudana, R.J.; Kumar, S.A.; Nabi, Q.G. A novel lignan composition from Cedrus deodara induces apoptosis and early nitric oxide generation in human leukemia Molt-4 and HL-60 cells. Nitric Oxide Biol. Chem. 2006, 14, 72–88. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Liu, D.; Zhang, J.; Hu, P.; Shen, W.; Fan, B.; Ma, Q.; Wang, X. Extraction and purification of total flavonoids from pine needles of Cedrus deodara contribute to anti-tumor in vitro. BMC Complement. Altern. Med. 2016, 16, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basu, L.R.; De, A.; Sarkar, P.; Karak, P.; Dastidar, S.G. Possibilities of developing novel potent antitumor agents from the leaves of Cryptomaria japonica. Int. J. Phytomed. 2016, 8, 404–410. [Google Scholar] [CrossRef] [Green Version]
- Fernandez, A.; Cock, I.E. The therapeutic properties of juniperus communis L.: Antioxidant capacity, bacterial growth inhibition, anticancer activity and toxicity. Pharmacogn. J. 2016, 8, 273–280. [Google Scholar] [CrossRef] [Green Version]
- Machana, S.; Weerapreeyakul, N.; Barusrux, S.; Nonpunya, A.; Sripanidkulchai, B.; Thitimetharoch, T. Cytotoxic and apoptotic effects of six herbal plants against the human hepatocarcinoma (HepG2) cell line. Chin. Med. 2011, 6, 2–9. [Google Scholar] [CrossRef] [Green Version]
- MacHana, S.; Weerapreeyakul, N.; Barusrux, S.; Thumanu, K.; Tanthanuch, W. FTIR microspectroscopy discriminates anticancer action on human leukemic cells by extracts of Pinus kesiya; Cratoxylum formosum ssp. pruniflorum and melphalan. Talanta 2012, 93, 371–382. [Google Scholar] [CrossRef] [PubMed]
- Thu, N.B.; Trung, T.N.; Ha, D.T.; Khoi, N.M.; Hung, T.V.; Hien, T.T.; Namhui, Y.; Bae, K. Screening of Vietnamese medicinal plants for cytotoxic activity. Nat. Prod. Sci. 2010, 16, 43–49. [Google Scholar]
- Chattopadhyay, S.K.; Kumar, T.R.S.; Maulik, P.R.; Srivastava, S.; Garg, A.; Sharon, A.; Negi, A.S.; Khanuja, S.P.S. Absolute configuration and anticancer activity of taxiresinol and related lignans of Taxus wallichiana. Bioorg. Med. Chem. 2003, 11, 4945–4948. [Google Scholar] [CrossRef] [PubMed]
- Kaushik, P.; Lal Khokra, S.; Rana, A.C.; Kaushik, D. Evaluation of anticancer activity of Pinus roxburghii sarg. Against IMR-32 human neuroblastoma cancer cell line. Int. J. Pharm. Clin. Res. 2015, 7, 105–108. [Google Scholar]
- Jiang, P.; Zhang, Q.; Zhao, Y.; Xiong, J.; Wang, F.; Zhang, T.; Zhang, C. Extraction, Purification, and Biological Activities of Polysaccharides from Branches and Leaves of Taxus cuspidata S. Et Z. Molecules 2019, 24, 2926. [Google Scholar] [CrossRef] [Green Version]
- Mukherjee, A.; Sikdar, S.; Bishayee, K.; Paul, A.; Ghosh, S.; Boujedaini, N.; Khuda-Bukhsh, A.R. Ethanolic extract of Thuja occidentalis blocks proliferation of A549 cells and induces apoptosis in vitro. J. Chin. Integr. Med. 2012, 10, 1451–1459. [Google Scholar] [CrossRef]
- Khuda-Bukhsh, A.R.; Biswas, R.; Mandal, S.K.; Dutta, S.; Bhattacharyya, S.S.; Boujedaini, N. Thujone-rich fraction of Thuja occidentalis demonstrates major anti-cancer potentials: Evidences from in vitro studies on A375 cells. Evid. Based Complement. Altern. Med. 2011, 2011, 568148. [Google Scholar] [CrossRef] [Green Version]
- Velmurugan, B.K.; Rathinasamy, B.; Lohanathan, B.P.; Thiyagarajan, V.; Weng, C.F. Neuroprotective role of phytochemicals. Molecules 2018, 23, 2485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gitler, A.D.; Dhillon, P.; Shorter, J. Neurodegenerative disease: Models, mechanisms, and a new hope. DMM Dis. Model. Mech. 2017, 10, 499–502. [Google Scholar] [CrossRef] [Green Version]
- Venkatesan, R.; Ji, E.; Kim, S.Y. Phytochemicals that regulate neurodegenerative disease by targeting neurotrophins: A comprehensive review. Biomed Res. Int. 2015, 2015, 814068. [Google Scholar] [CrossRef]
- Johri, A.; Beal, M.F. Mitochondrial dysfunction in neurodegenerative diseases. J. Pharmacol. Exp. Ther. 2012, 342, 619–630. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.L.; Lin, Y.T.; Chuang, P.C.; Bohr, V.A.; Mattson, M.P. BDNF and exercise enhance neuronal DNA repair by stimulating CREB-mediated production of apurinic/apyrimidinic endonuclease 1. NeuroMol. Med. 2014, 16, 161–174. [Google Scholar] [CrossRef]
- Huang, E.J.; Reichardt, L.F. Neurotrophins: Roles in neuronal development and function. Annu. Rev. Neurosci. 2001, 24, 677–736. [Google Scholar] [CrossRef] [Green Version]
- Agrawal, M.; Biswas, A.; Levy, C.E. Molecular diagnostics of neurodegenerative disorders. Front. Mol. Biosci. 2015, 2, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Olivares, D.; Deshpande, V.K.; Shi, Y.; Lahiri, D.K.; Greig, N.H.; Rogers, J.T.; Huang, X. N-Methyl D-Aspartate (NMDA) Receptor Antagonists and Memantine Treatment for Alzheimer’s Disease, Vascular Dementia and Parkinson’s Disease. Curr. Alzheimer Res. 2012, 9, 746–758. [Google Scholar] [CrossRef] [PubMed]
- Briffa, M.; Ghio, S.; Neuner, J.; Gauci, A.J.; Cacciottolo, R.; Marchal, C.; Caruana, M.; Cullin, C.; Vassallo, N.; Cauchi, R.J. Extracts from two ubiquitous Mediterranean plants ameliorate cellular and animal models of neurodegenerative proteinopathies. Neurosci. Lett. 2017, 638, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Physiology, G.; Waczulikova, I.; Kilanczyk, E.; Bryszewska, M. The effect of Pycnogenol on the erythrocyte membrane fluidity. Gen. Physiol. Biophys. 2004, 23, 39–51. [Google Scholar]
- Voss, P.; Horakova, L.; Jakstadt, M.; Kiekebusch, D.; Grune, T. Ferritin oxidation and proteasomal degradation: Protection by antioxidants. Free Radic. Res. 2006, 40, 673–683. [Google Scholar] [CrossRef]
- Deture, M.A.; Dickson, D.W. The neuropathological diagnosis of Alzheimer’s disease. Mol. Neurodegener. 2019, 14, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Jahn, H. Memory loss in Alzheimer’s disease. Dialogues Clin. Neurosci. 2013, 15, 445–454. [Google Scholar] [PubMed]
- Tanvir Kabir, M.; Sahab Uddin, M.; Al Mamun, A.; Jeandet, P.; Aleya, L.; Mansouri, R.A.; Md Ashraf, G.; Mathew, B.; Bin-Jumah, M.N.; Abdel-Daim, M.M. Combination drug therapy for the management of alzheimer’s disease. Int. J. Mol. Sci. 2020, 21, 3272. [Google Scholar] [CrossRef]
- Durães, F.; Pinto, M.; Sousa, E. Old drugs as new treatments for neurodegenerative diseases. Pharmaceuticals 2018, 11, 44. [Google Scholar] [CrossRef] [Green Version]
- Barbier, P.; Zejneli, O.; Martinho, M.; Lasorsa, A.; Belle, V.; Smet-Nocca, C.; Tsvetkov, P.O.; Devred, F.; Landrieu, I. Role of tau as a microtubule-associated protein: Structural and functional aspects. Front. Aging Neurosci. 2019, 10, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cory, H.; Passarelli, S.; Szeto, J.; Tamez, M.; Mattei, J. The Role of Polyphenols in Human Health and Food Systems: A Mini-Review. Front. Nutr. 2018, 5, 1–9. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rensink, A.A.M.; De Waal, R.M.W.; Kremer, B.; Verbeek, M.M. Pathogenesis of cerebral amyloid angiopathy. Brain Res. Rev. 2003, 43, 207–223. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Saito, S.; Inoue, T.; Satoh-Asahara, N.; Ihara, M. Novel therapeutic potentials of taxifolin for amyloid-β-associated neurodegenerative diseases and other diseases: Recent advances and future perspectives. Int. J. Mol. Sci. 2019, 20, 2139. [Google Scholar] [CrossRef] [Green Version]
- Sharma, L.; Sharma, A.; Goyal, R.; Alam, J. Pinus roxburghii Sarg. Ameliorates alzheimer’s disease-type neurodegeneration and cognitive deficits caused by intracerebroventricular-streptozotocin in rats: An in vitro and in vivo study. Indian J. Pharm. Sci. 2020, 82, 861–870. [Google Scholar] [CrossRef]
- Hassaan, Y.; Handoussa, H.; El-Khatib, A.H.; Linscheid, M.W.; El Sayed, N.; Ayoub, N. Evaluation of plant phenolic metabolites as a source of Alzheimer’s drug leads. Biomed Res. Int. 2014, 2014, 843263. [Google Scholar] [CrossRef] [PubMed]
- Arbo, B.D.; André-Miral, C.; Nasre-Nasser, R.G.; Schimith, L.E.; Santos, M.G.; Costa-Silva, D.; Muccillo-Baisch, A.L.; Hort, M.A. Resveratrol Derivatives as Potential Treatments for Alzheimer’s and Parkinson’s Disease. Front. Aging Neurosci. 2020, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.; Javed, S.; Javed, S.; Tariq, A.; Šamec, D.; Tejada, S.; Nabavi, S.F.; Braidy, N.; Nabavi, S.M. Resveratrol and Alzheimer’s Disease: Mechanistic Insights. Mol. Neurobiol. 2017, 54, 2622–2635. [Google Scholar] [CrossRef]
- Maimoona, A.; Naeem, I.; Saddiqe, Z.; Jameel, K. A review on biological, nutraceutical and clinical aspects of French maritime pine bark extract. J. Ethnopharmacol. 2011, 133, 261–277. [Google Scholar] [CrossRef]
- Peng, Q.L.; Zard, A.R.B.; Lau, B.H.S. Pycnogenol protects neurons from amyloid- b peptide-induced apoptosis. Mol. Brain Res. 2002, 104, 55–65. [Google Scholar] [CrossRef]
- Paarmann, K.; Prakash, S.R.; Krohn, M.; Möhle, L.; Brackhan, M.; Brüning, T.; Eiriz, I.; Pahnke, J. French maritime pine bark treatment decelerates plaque development and improves spatial memory in Alzheimer’s disease mice. Phytomedicine 2019, 57, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Fang, C.; Lv, L.; Mao, S.; Dong, H.; Liu, B. Cognition Deficits in Parkinson’s Disease: Mechanisms and Treatment. Parkinsons Dis. 2020, 2020, 2076942. [Google Scholar] [CrossRef]
- Dias, V.; Junn, E.; Mouradian, M.M. The role of oxidative stress in parkinson’s disease. J. Parkinsons Dis. 2013, 3, 461–491. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Ding, Y.; Cagniard, B.; Van Laar, A.D.; Mortimer, A.; Chi, W.; Hastings, T.G.; Un, J.K.; Zhuang, X. Unregulated cytosolic dopamine causes neurodegeneration associated with oxidative stress in mice. J. Neurosci. 2008, 28, 425–433. [Google Scholar] [CrossRef] [Green Version]
- Zoccarato, F.; Toscano, P.; Alexandre, A. Dopamine-derived dopaminochrome promotes H2O2 release at mitochondrial Complex I: Stimulation by rotenone, control by Ca2+, and relevance to Parkinson disease. J. Biol. Chem. 2005, 280, 15587–15594. [Google Scholar] [CrossRef] [Green Version]
- Ebrahimi-Fakhari, D.; Wahlster, L.; McLean, P.J. Protein degradation pathways in Parkinson’s disease: Curse or blessing. Acta Neuropathol. 2012, 124, 153–172. [Google Scholar] [CrossRef] [Green Version]
- Javed, H.; Nagoor Meeran, M.F.; Azimullah, S.; Adem, A.; Sadek, B.; Ojha, S.K. Plant Extracts and Phytochemicals Targeting α-Synuclein Aggregation in Parkinson’s Disease Models. Front. Pharmacol. 2019, 9, 1555. [Google Scholar] [CrossRef] [Green Version]
- Corona, J.C. Natural Compounds for the Management of Parkinson’s Disease and Attention-Deficit/Hyperactivity Disorder. Biomed Res. Int. 2018, 2018, 4067597. [Google Scholar] [CrossRef] [Green Version]
- Ríos, J.L.; Onteniente, M.; Picazo, D.; Montesinos, M.C. Medicinal Plants and Natural Products as Potential Sources for Antiparkinson Drugs. Planta Med. 2016, 82, 942–951. [Google Scholar] [CrossRef] [Green Version]
- Bais, S.; Gill, N.S.; Kumar, N. Neuroprotective Effect of Juniperus communis on Chlorpromazine Induced Parkinson Disease in Animal Model. Chin. J. Biol. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Zhang, F.; Shi, J.S.; Zhou, H.; Wilson, B.; Hong, J.S.; Gao, H.M. Resveratrol protects dopamine neurons against lipopolysaccharide-induced neurotoxicity through its anti-inflammatory actions. Mol. Pharmacol. 2010, 78, 466–477, reprinted in Mol. Pharmacol. 2010, 78, 981. [Google Scholar] [CrossRef] [Green Version]
- Fang, X.S.; Hao, J.F.; Zhou, H.Y.; Zhu, L.X.; Wang, J.H.; Song, F.Q. Pharmacological studies on the sedative-hypnotic effect of Semen Ziziphi spinosae (Suanzaoren) and Radix et Rhizoma Salviae miltiorrhizae (Danshen) extracts and the synergistic effect of their combinations. Phytomedicine 2010, 17, 75–80. [Google Scholar] [CrossRef]
- Akram, M.; Daniyal, M.; Munir, N.; Mohiuddin, E.; Sultana, S. Medicinal Plants Combating Against Insomnia: A Green Anti-Insomnia Approach. J. Nerv. Ment. Dis. 2019, 207, 927–935. [Google Scholar] [CrossRef]
- Gooneratne, N.S.; Vitiello, M.V. Sleep in Older Adults. Normative Changes, Sleep Disorders, and Treatment Options. Clin. Geriatr. Med. 2014, 30, 591–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Atkin, T.; Comai, S.; Gobbi, G. Drugs for insomnia beyond benzodiazepines: Pharmacology, clinical applications, and discovery. Pharmacol. Rev. 2018, 70, 197–245. [Google Scholar] [CrossRef]
- Sateia, M.J.; Buysse, D.J.; Krystal, A.D.; Neubauer, D.N. Adverse effects of hypnotic medications. J. Clin. Sleep Med. 2017, 13, 839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woo, J.; Yang, H.; Yoon, M.; Gadhe, C.G.; Pae, A.N.; Cho, S.; Justin Lee, C. 3-Carene, a phytoncide from pine tree has a sleep-enhancing effect by targeting the GABAA-benzodiazepine receptors. Exp. Neurobiol. 2019, 28, 593–601. [Google Scholar] [CrossRef] [Green Version]
- Arumugam, G.; Manjula, P.; Paari, N. A review: Anti diabetic medicinal plants used for diabetes mellitus. J. Acute Dis. 2013, 2, 196–200. [Google Scholar] [CrossRef] [Green Version]
- Salehi, B.; Ata, A.; Kumar, N.V.A.; Sharopov, F.; Ramírez-Alarcón, K.; Ruiz-Ortega, A.; Ayatollahi, S.A.; Fokou, P.V.T.; Kobarfard, F.; Zakaria, Z.A.; et al. Antidiabetic potential of medicinal plants and their active components. Biomolecules 2019, 9, 551, ISBN 5641266167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Upendra Rao, M.; Sreenivasulu, M.; Chengaiah, B.; Jaganmohan Reddy, K.; Madhusudhana Chetty, C. Herbal medicines for diabetes mellitus: A review. Int. J. PharmTech Res. 2010, 2, 1883–1892. [Google Scholar]
- Warren, R.E. The stepwise approach to the management of type 2 diabetes. Diabetes Res. Clin. Pract. 2004, 65, S3. [Google Scholar] [CrossRef] [PubMed]
- Wadkar, K.A.; Magdum, C.S.; Patil, S.S.; Naikwade, N.S. Anti-diabetic potential and Indian medicinal plants. J. Herbmed Toxicol. 2014, 2, 45–50. [Google Scholar]
- Afrisham, R.; Aberomand, M.; Ghaffari, M.A.; Siahpoosh, A.; Jamalan, M. Inhibitory Effect of Heracleum persicum and Ziziphus jujuba on Activity of Alpha-Amylase. J. Bot. 2015, 2015, 824683. [Google Scholar] [CrossRef] [Green Version]
- Jeong, S.O.; Son, Y.; Lee, J.H.; Cheong, Y.K.; Park, S.H.; Chung, H.T.; Pae, H.O. Resveratrol analog piceatannol restores the palmitic acid-induced impairment of insulin signaling and production of endothelial nitric oxide via activation of anti-inflammatory and antioxidative heme oxygenase-1 in human endothelial cells. Mol. Med. Rep. 2015, 12, 937–944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vallianou, N.G.; Evangelopoulos, A.; Kazazis, C. Resveratrol and diabetes. Rev. Diabet. Stud. 2013, 10, 236–242. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szkudelski, T.; Szkudelska, K. Resveratrol and diabetes: From animal to human studies. Biochim. Biophys. Acta Mol. Basis Dis. 2015, 1852, 1145–1154. [Google Scholar] [CrossRef] [Green Version]
- Bagul, P.K.; Banerjee, S.K. Application of Resveratrol in Diabetes: Rationale, Strategies and Challenges. Curr. Mol. Med. 2015, 15, 312–330. [Google Scholar] [CrossRef]
- Sciences, D. The New Antiepileptic Drugs: Their Neuropharmacology and Clinical Indications. Neurol. Med. Chir. 2016, 56, 205–220. [Google Scholar] [CrossRef] [Green Version]
- Kaushik, D.; Kumar, A.; Kaushik, P.; Rana, A.C. Anticonvulsant activity of alcoholic extract of bark of Pinus roxburghii Sarg. J. Chin. Integr. Med. 2012, 10, 1056–1060. [Google Scholar] [CrossRef]
- Dhayabaran, D.; Florance, E.J.; Nandakumar, K.; Shanmugarathinam, A.; Puratchikody, A. Anticonvulsant activity of fraction isolated from ethanolic extract of heartwood of Cedrus deodara. J. Nat. Med. 2014, 68, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.W.; Yang, C.T.; Gong, C.L.; Chen, Y.H.; Chen, Y.W.; Wu, K.C.; Cheng, T.H.; Kuo, Y.H.; Chen, Y.F.; Leung, Y.M. Inhibition of voltage-gated Na+ channels by hinokiol in neuronal cells. Pharmacol. Rep. 2015, 67, 1049–1054. [Google Scholar] [CrossRef] [PubMed]
- Vasconcelos, S.M.M.; Lima, S.R.; Soares, P.M.; Assreuy, A.M.S.; de Sousa, F.C.F.; Lobato, R.d.F.G.; Vasconcelos, G.S.; Santi-Gadelha, T.; Bezerra, E.H.S.; Cavada, B.S.; et al. Central action of Araucaria angustifolia seed lectin in mice. Epilepsy Behav. 2009, 15, 291–293. [Google Scholar] [CrossRef] [PubMed]
- Hijazi, M.A.; El-Mallah, A.; Aboul-Ela, M.; Ellakany, A. Evaluation of Analgesic Activity of Papaver libanoticum Extract in Mice: Involvement of Opioids Receptors. Evid. Based Complement. Altern. Med. 2017, 2017, 8935085. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.S.; Chi, C.S.; Huang, S.S.; Shie, P.H.; Lin, T.H.; Huang, G.J. Antioxidant, analgesic, and anti-inflammatory activities of the ethanolic extracts of Taxillus liquidambaricola. J. Ethnopharmacol. 2011, 137, 1161–1171. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, A.; Morankar, P.G.; Kumbhare, M.R. Review on Analgesic Activity and Determination Methods. Pharmtechmedica 2014, 3, 425–428. [Google Scholar]
- Machado, F.D.; Kuo, J.; Ongaratti, B.R.; Medeiros, N.D.; Salvador, M.; Dani, C.; Funchal, C. Antioxidant and neuroprotective potential of extract of Brazilian pine Araucaria angustifolia bracts against oxidative stress induced by sodium azide in hippocampus. Integr. Pharmacol. Toxicol. Genotoxicol. 2015, 1, 16–20. [Google Scholar]
- Thiago, C.; Patrícia de Brum, V.; Patrícia Gomes da, S.; Marines de Avila, H.; Graziele Daiane, S.; Michele Stach, C.; Antônio Batista, P.; Sidnei, M.; Andreas Sebastian, M.; Chariston André Dal, B. Mechanism of the Entomotoxic Activity Induced by Araucaria Angustifolia Methanolic Extract in Nauphoeta Cinerea Lobster Cockroaches. J. Bot. Res. 2017, 1, 38–49. [Google Scholar] [CrossRef]
- Zhao, Z.; Dong, Z.; Ming, J.; Liu, Y. Cedrin identified from Cedrus deodara (Roxb.) G. Don protects PC12 cells against neurotoxicity. Nat. Prod. Res. 2018, 6419, 1455–1458. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, H.G.; Lee, H.W.; Han, J.M.; Lee, S.K.; Kim, D.W.; Saravanakumar, A.; Son, C.G. Hippocampal memory enhancing activity of pine needle extract against scopolamine-induced amnesia in a mouse model. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.S.; Kim, H.G.; Lee, H.W.; Kim, W.Y.; Ahn, Y.C.; Son, C.G. Pine needle extract prevents hippocampal memory impairment in acute restraint stress mouse model. J. Ethnopharmacol. 2017, 207, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Forouzanfar, F.; Ghorbani, A.; Hosseini, M. Hydroalcoholic extract of needles of Pinus eldarica enhances pentobarbital-induced sleep: Possible involvement of GABAergic system. Avicenna J. Phytomed. 2016, 6, 449. [Google Scholar] [PubMed]
- Wang, C.; He, L.; Yan, M. Effects of polyprenols from pine needles of Pinus massoniana on ameliorating cognitive impairment in a D -galactose-induced mouse model. Age 2014, 36, 9676. [Google Scholar] [CrossRef] [Green Version]
- Khan, M.M.; Kempuraj, D.; Thangavel, R.; Zaheer, A. Protection of MPTP-induced neuroinflammation and neurodegeneration by Pycnogenol. Neurochem. Int. 2013, 62, 379–388. [Google Scholar] [CrossRef] [Green Version]
- Kabra, A.; Baghel, U.S.; Hano, C.; Martins, N.; Khalid, M.; Sharma, R. Neuroprotective potential of Myrica esulenta in Haloperidol induced Parkinson’s disease. J. Ayurveda Integr. Med. 2020, 11, 448–454. [Google Scholar] [CrossRef]
- Lokesh, D.; Amitabha, D.; Sachin, A.; Avijeet, J. Neuropharmacological Exploration of Thuja Occidentalis Linn. Int. Res. J. Pharm. 2011, 2, 143–148. [Google Scholar]
- Lee, S.; Choi, C.; Kim, J.; Lim, S.; Jung, H. The Antioxidant Activities and Neuroprotective Effects of Hot Water Extracts from Torreyae Semen. Korea J. Herbol. 2017, 32, 41–48. [Google Scholar]
- Pradeep Kumar, C.; Lokesh, T.; Gobinath, M.; Kumar, B.; Saravanan, D. Anti-diabetic and anti-hyperlipidemic activities of glucomannan isolated from Araucaria cunninghamii seeds. J. Chem. Pharm. Sci. 2013, 6, 204–209. [Google Scholar]
- Jain, S.; Jain, A.; Malviya, N.; Kumar, D.; Jain, V.; Jain, S. Antidiabetic Activity of Cedrus deodara Aqueous Extract and Its Relationship with Its Antioxidant Properties. J. Pharm. Sci. Pharmacol. 2015, 1, 187–194. [Google Scholar] [CrossRef]
- De Medina, F.S.; Gamez, M.J.; Jimenez, I.; Jimenez, J.; Osuna, J.I.; Zarzuelo, A. Hypoglycemic activity of juniper “berries”. Planta Med. 1994, 60, 197–200. [Google Scholar] [CrossRef]
- Esmail Al-Snafi, A.; Majid, W.J.; Ali Talab, T.; Author, C. Medicinal Plants with Antidiabetic Effects-An Overview (Part 1) Clinically tested medicinal plants View project Medicinal plants with antimicrobial effects View project Medicinal Plants with Antidiabetic Effects-An Overview (Part 1). IOSR J. Pharm. 2019, 9, 9–46. [Google Scholar]
- Orhan, N.; Aslan, M.; Demirci, B.; Ergun, F. A bioactivity guided study on the antidiabetic activity of Juniperus oxycedrus subsp. oxycedrus L. leaves. J. Ethnopharmacol. 2012, 140, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Zulfqar, F.; Akhtar, M.F.; Saleem, A.; Akhtar, B.; Sharif, A.; Saleem, U. Chemical characterization, antioxidant evaluation, and antidiabetic potential of Pinus gerardiana (Pine nuts) extracts. J. Food Biochem. 2020, 44, 1–12. [Google Scholar] [CrossRef]
- Liu, X.; Wei, J.; Tan, F.; Zhou, S.; Würthwein, G.; Rohdewald, P. Antidiabetic effect of Pycnogenol® French maritime pine bark extract in patients with diabetes type II. Life Sci. 2004, 75, 2505–2513. [Google Scholar] [CrossRef]
- Kaushik, P.; Khokra, D. Evaluation of Antidiabetic Potential of Pinus roxburghii Bark Extract in Alloxan Induced Diabetic Rats. J. Pharmacogn. Nat. Prod. 2016, 1, 1–5. [Google Scholar] [CrossRef] [Green Version]
- Haman, N.; Morozova, K.; Tonon, G.; Scampicchio, M.; Ferrentino, G. Antimicrobial effect of Picea abies extracts on E. coli growth. Molecules 2019, 24, 4053. [Google Scholar] [CrossRef] [Green Version]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial agents from plants: Antibacterial activity of plant volatile oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef]
- Céspedes, C.L.; Avila, J.G.; García, A.M.; Becerra, J.; Flores, C.; Aqueveque, P.; Bittner, M.; Hoeneisen, M.; Martinez, M.; Silva, M. Antifungal and antibacterial activities of Araucaria araucana (Mol.) K. Koch heartwood lignans. Z. fur Naturforsch. Sect. C J. Biosci. 2006, 61, 35–43. [Google Scholar] [CrossRef]
- Hedenström, E.; Fagerlund Edfeldt, A.; Edman, M.; Jonsson, B.G. Resveratrol, piceatannol, and isorhapontigenin from Norway spruce (Picea abies) debarking wastewater as inhibitors on the growth of nine species of wood-decaying fungi. Wood Sci. Technol. 2016, 50, 617–629. [Google Scholar] [CrossRef]
- De Souza Wuillda, A.C.J.; Martins, R.C.C.; Costa, F.D.N. Larvicidal activity of secondary plant metabolites in aedes aegypti control: An overview of the previous 6 years. Nat. Prod. Commun. 2019, 14, 1934578X19862893. [Google Scholar] [CrossRef] [Green Version]
- Hales, S.; De Wet, N.; Maindonald, J.; Woodward, A. Potential effect of population and climate changes on global distribution of dengue fever: An empirical model. Lancet 2002, 360, 830–834. [Google Scholar] [CrossRef]
- Wondimu, E.; Hirpasa, T.; Tadele, A. Larvicidal activity of Juniperus procera extract against anopheles mosquito in in vitro, North Western Ethiopia. J. Med. Plants Res. 2020, 14, 445–450. [Google Scholar] [CrossRef]
- Nayak, J.B.; Mohan, B. Larvicidal activity of Rauvolfia serpentina L. fruits against Aedes aegypti Mosquito larvae. Int. Res. J. Biol. Sci. 2015, 4, 54–56. [Google Scholar]
- Phambala, K.; Tembo, Y.; Kasambala, T.; Kabambe, V.H.; Stevenson, P.C.; Belmain, S.R. Bioactivity of common pesticidal plants on fall Armyworm Larvae (Spodoptera frugiperda). Plants 2020, 9, 112. [Google Scholar] [CrossRef] [Green Version]
- Sharma, P.; Mohan, L.; Srivastava, C.N. Larvicidal potential of Nerium indicum and Thuja oriertelis extracts against malaria and Japanese encephalitis vector. J. Environ. Biol. 2005, 26, 657–660. [Google Scholar]
- Hasaballah, A.; Shehata, A.; Fouda, M.; Hassan, M.; Gad, M. The Biological Activity of Cupressus sempervirens Extracts against Musca domestica. Asian J. Biol. 2018, 5, 1–12. [Google Scholar] [CrossRef]
- Setiawan, S.; Koerniasari, K.; Ngadino, N.; Sudjarwo, S.A. Bioinsecticide effect of Pinus merkusii tree bark extract on aedes aegypti larvae. J. Young Pharm. 2017, 9, 127–130. [Google Scholar] [CrossRef] [Green Version]
- Tang, Y.L.; Chan, S.W. A review of the pharmacological effects of piceatannol on cardiovascular diseases. Phyther. Res. 2014, 28, 1581–1588. [Google Scholar] [CrossRef]
- Rohdewald, P. Pleiotropic Effects of French Maritime Pine Bark Extract to Promote Healthy Aging. Rejuvenation Res. 2019, 22, 210–217. [Google Scholar] [CrossRef]
- Markus, M.A.; Morris, B.J. Resveratrol in prevention and treatment of common clinical conditions of aging. Clin. Interv. Aging 2008, 3, 331–339. [Google Scholar] [CrossRef] [PubMed]
- Shaito, A.; Posadino, A.M.; Younes, N.; Hasan, H.; Halabi, S.; Alhababi, D.; Al-Mohannadi, A.; Abdel-Rahman, W.M.; Eid, A.H.; Nasrallah, G.K.; et al. Potential adverse effects of resveratrol: A literature review. Int. J. Mol. Sci. 2020, 21, 2084. [Google Scholar] [CrossRef] [Green Version]
- Kee, H.J.; Park, S.; Kang, W.; Lim, K.S.; Kim, J.H.; Ahn, Y.; Jeong, M.H. Piceatannol attenuates cardiac hypertrophy in an animal model through regulation of the expression and binding of the transcription factor GATA binding factor 6. FEBS Lett. 2014, 588, 1529–1536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malekahmadi, M.; Moradi Moghaddam, O.; Islam, S.M.S.; Tanha, K.; Nematy, M.; Pahlavani, N.; Firouzi, S.; Zali, M.R.; Norouzy, A. Evaluation of the effects of pycnogenol (French maritime pine bark extract) supplementation on inflammatory biomarkers and nutritional and clinical status in traumatic brain injury patients in an intensive care unit: A randomized clinical trial protocol. Trials 2020, 21, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Shand, B.; Strey, C.; Scott, R.; Morrison, Z.; Gieseg, S. Pilot study on the clinical effects of dietary supplementation with Enzogenol, a flavonoid extract of pine bark and vitamin C. Phyther. Res. 2003, 17, 490–494. [Google Scholar] [CrossRef]
- Divvela, H.N.D.; Duppala, L.; Kolapalli, V.R.M. Isolation and acute oral toxicity studies of Araucaria heterophylla novel natural polysaccharide gum in albino mice. World J. Pharm. Pharm. Sci. 2016, 5, 702–711. [Google Scholar] [CrossRef]
- Grobosch, T.; Schwarze, B.; Stoecklein, D.; Binscheck, T. Fatal poisoning with Taxus baccata. quantification of paclitaxel (taxol A), 10-deacetyltaxol, baccatin III, 10-deacetylbaccatin III, cephalomannine (taxol B), and 3,5-dimethoxyphenol in body fluids by liquid chromatography-tandem mass spectrometry. J. Anal. Toxicol. 2012, 36, 36–43. [Google Scholar] [CrossRef]

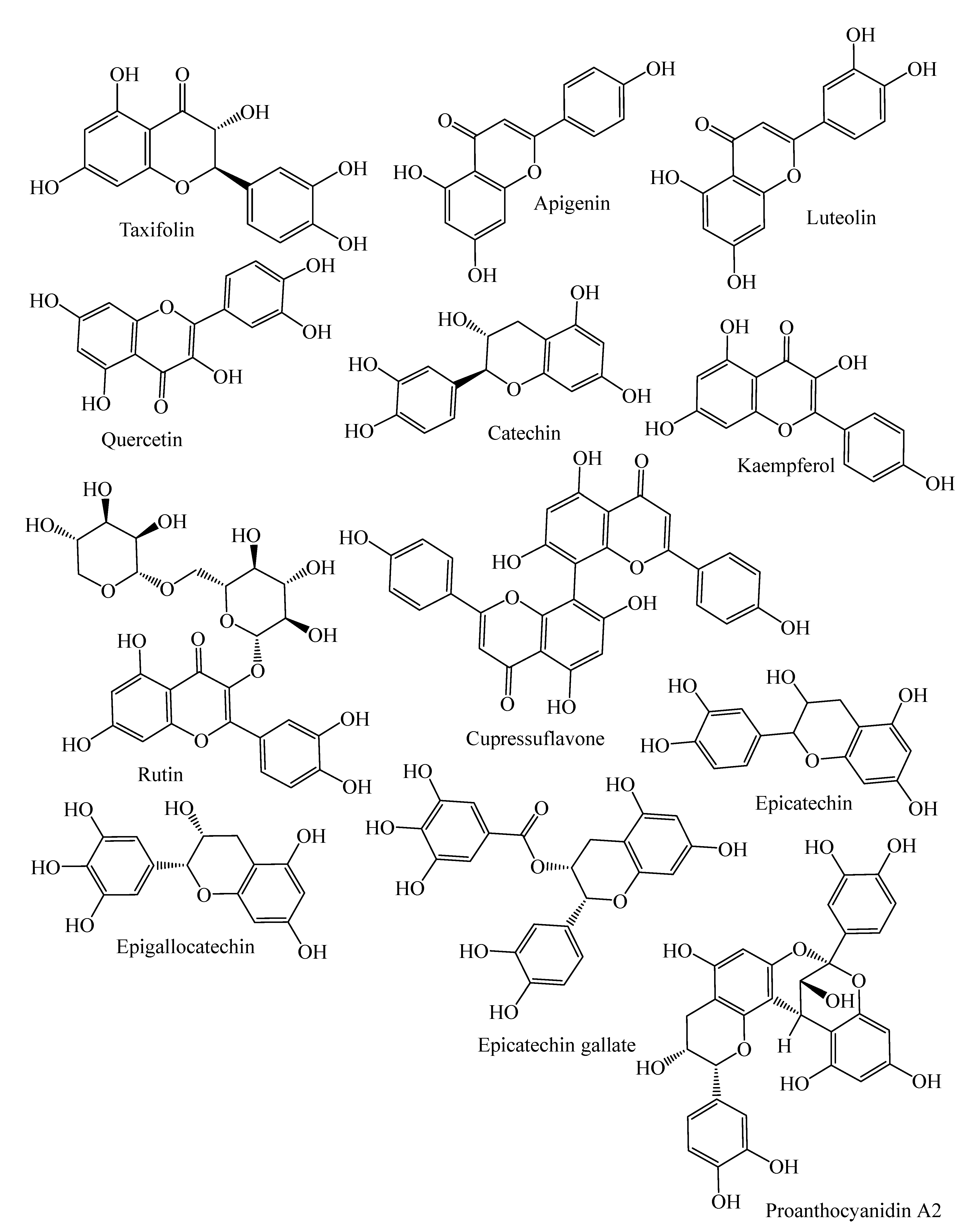


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bhardwaj, K.; Silva, A.S.; Atanassova, M.; Sharma, R.; Nepovimova, E.; Musilek, K.; Sharma, R.; Alghuthaymi, M.A.; Dhanjal, D.S.; Nicoletti, M.; et al. Conifers Phytochemicals: A Valuable Forest with Therapeutic Potential. Molecules 2021, 26, 3005. https://doi.org/10.3390/molecules26103005
Bhardwaj K, Silva AS, Atanassova M, Sharma R, Nepovimova E, Musilek K, Sharma R, Alghuthaymi MA, Dhanjal DS, Nicoletti M, et al. Conifers Phytochemicals: A Valuable Forest with Therapeutic Potential. Molecules. 2021; 26(10):3005. https://doi.org/10.3390/molecules26103005
Chicago/Turabian StyleBhardwaj, Kanchan, Ana Sanches Silva, Maria Atanassova, Rohit Sharma, Eugenie Nepovimova, Kamil Musilek, Ruchi Sharma, Mousa A. Alghuthaymi, Daljeet Singh Dhanjal, Marcello Nicoletti, and et al. 2021. "Conifers Phytochemicals: A Valuable Forest with Therapeutic Potential" Molecules 26, no. 10: 3005. https://doi.org/10.3390/molecules26103005
APA StyleBhardwaj, K., Silva, A. S., Atanassova, M., Sharma, R., Nepovimova, E., Musilek, K., Sharma, R., Alghuthaymi, M. A., Dhanjal, D. S., Nicoletti, M., Sharma, B., Upadhyay, N. K., Cruz-Martins, N., Bhardwaj, P., & Kuča, K. (2021). Conifers Phytochemicals: A Valuable Forest with Therapeutic Potential. Molecules, 26(10), 3005. https://doi.org/10.3390/molecules26103005
















