Abstract
Sigma-2 (σ2) is an endoplasmic receptor identified as the Endoplasmic Reticulum (ER) transmembrane protein TMEM97. Despite its controversial identity, which was only recently solved, this protein has gained scientific interest because of its role in the proliferative status of cells; many tumor cells from different organs overexpress the σ2 receptor, and many σ2 ligands display cytotoxic actions in (resistant) cancer cells. These properties have shed light on the σ2 receptor as a potential druggable target to be bound/activated for the diagnosis or therapy of tumors. Additionally, diverse groups have shown how the σ2 receptor can be exploited for the targeted delivery of the anticancer drugs to tumors. As the cancer disease is a multifactorial pathology with multiple cell populations, a polypharmacological approach is very often needed. Instead of the simultaneous administration of different classes of drugs, the use of one molecule that interacts with diverse pharmacological targets, namely MultiTarget Directed Ligand (MTDL), is a promising and currently pursued strategy, that may overcome the pharmacokinetic problems associated with the administration of multiple molecules. This review aims to point out the progress regarding the σ2 ligands in the oncology field, with a focus on MTDLs directed towards σ2 receptors as promising weapons against (resistant) cancer diseases.
1. Introduction
Treatment of cancer, which is a major public health problem worldwide and the second leading cause of death (in the USA) [1], has changed a great deal over the years. The first modern therapeutic approach dates back to the end of 1800s with the discovery of X-rays. From that moment, amazing scientific and medical progresses have furnished a plethora of approaches that have led to increasingly specific and effective treatments. From the birth of chemotherapy, based on cytotoxic antitumor drugs to genetic engineering studies, which provided monoclonal antibodies, immune checkpoint inhibitors, and Chimeric Antigen Receptor T cell therapies (CAR-T), treatment of cancer has drastically changed over the years and life expectancy of people suffering from this pathology has considerably improved [2,3].
Cancer is a complicated pathology because of the many mechanisms responsible for the evasion from the regulatory circuits, which ensure a correct cell growth. Besides the enhanced angiogenesis, the most important mechanisms that sustain the progression of the pathology consist of the production of growth factors and the insensibility to anti-growth factors (which allow a limitless replicative potential); the ability to evade apoptosis and to escape from the primary tumor mass to create metastasis [4].
This plurality of mechanisms justifies the need of a polypharmacological approach to treat the pathology working on two or more targets at the same time, in order to produce synergic effects and increase the efficacy of the treatment. Multifunctional therapies can be based either on the well validated use of combinations of drugs administered together, or on the use of a single multitarget directed ligand (MTDL), whose interaction with different targets exerts more pharmacological effects. Despite the therapeutic success, the main limitation of the former approach lies in the diverse pharmacokinetic and metabolic profiles of the drugs that may lead to multiple toxic metabolites and side effects, compared to a single drug administration. Thus, the MTDL approach is attracting interest as a strategy to be exploited to treat cancer and the other pathologies based on different factors.
Both the Sigma (σ) receptor subtypes, σ1 and σ2, are involved in cancer disease and, have been often exploited as targets for the development of MTDLs to synergize with the antitumor action mediated by other targets. In this review, we only briefly discussed about the σ receptor, while we focused more on the σ subtype and the structural insights of the σ-directed MTLDs in the context of cancer.
2. σ Receptors
σ proteins, which were thought to belong to the opioid receptor family until 1976, were later identified as an independent class of receptors divided into two different subtypes [5]. The σ1 subtype was cloned in the early 1990s, and its crystal structure was recently disclosed [6], while the σ2 receptor was only lately identified as the TMEM97 protein [7] and its crystal structure has been resolved during the preparation of this review [8]. Although the mechanisms of action of the two proteins need to be fully elucidated, they both appear as intriguing targets for the development of therapies useful for a wide range of pathologies [9,10].
2.1. σ1 Receptor
σ1 receptor is a 223-amino-acid protein characterized by a high level of similarity with the ERG2p, a C8-C7 sterol isomerase expressed in yeast, even if no isomerase activity has been attributed to σ1 receptors. The protein structure consists of five α-helices and ten β-strands. The N-terminus crosses the Endoplasmic Reticulum (ER) membrane and protrudes into the lumen forming a transmembrane domain (TMD), while the flat and hydrophobic C-terminus is associated to the cytosolic surface of the ER. The σ1 protein is mainly localized at the interface between mitochondria and ER, a space commonly named mitochondrial associated endoplasmic reticulum membrane (MAM), where it interacts with proteins to modulate Ca2+ fluxes between ER and mitochondria [11,12]. This receptor has been lately defined as a ‘pluripotent chaperone’ that interacts with several client proteins modulating their activity [10]. One of its most studied interactions is with the binding immunoglobulin proteins (BiP), with which it is associated in rest conditions. Upon stress (e.g., Ca2+ depletion from ER), the receptor dissociates from BiP and chaperones the Inositol 1,4,5-triphospate receptor type 3 (IP3R3) at MAM, increasing the Ca2+ flow from ER to mitochondria to guarantee cell energy needs [11].
σ1 receptor’s role in neurodegenerative diseases has been extensively studied and reviewed, and its therapeutic exploitability as druggable target is witnessed by the ligands in clinical trials for treating pathologies such as Alzheimer disease (AD), Huntington disease (HD) etc [13,14]. Less explored, but still intriguing under the therapeutic perspective, is the σ1 receptor role in cancer. The σ1 receptor exploitation as a target for the development of MTDLs addressing the diverse pathologies in which this σ subtype is involved has been recently reviewed [15].
2.1.1. σ1 Receptors Involvement in Cancer
High levels of σ1 receptors have been found in human cancer cell lines of breast, lung, prostate, colon, melanoma, CNS, kidney and pancreas [16]. However, despite controversial results in breast adenocarcinoma [17,18], Xu reported significant correlation between σ1 receptor expression and progression of esophageal squamous cell carcinoma [19] and hilar cholangiocarcinoma [20].
σ1 receptor density was also found to be increased in breast cancer cells with high metastatic potential, supporting a direct correlation between the expression of the receptor and the aggressiveness of the pathology [21].
Moreover, scintigraphy studies performed using (N-[2-(1′-piperidinyl)ethyl]-3-123I-iodo-4-methoxybenzamide), a selective ligand for the σ1 receptor, on patients with primary breast cancer revealed that the ligand was specifically retained in diseased tissues rather than healthy ones [22].
Finally, as reviewed by Kim, both σ1 receptor claimed agonists (i.e., (+)-SKF10047, PRE-084 and (+)-pentazocine) and antagonists (i.e., haloperidol, SR31747A, Rimcazole and BD1047) demonstrated anticancer activities [23]. This data, that may raise doubts about the σ1 receptor mediated cytotoxicity, also emphasizes the likely inconvenience of the word ‘agonist’ and ‘antagonist’ in the σ1 receptor context, as reported by Yano et al. [24].
2.1.2. σ1/σ2 Receptors MTDLs
The still-unclear involvement of the σ1 receptor in cancer justifies the small number of studies performed for the synthesis of σ1 directed MTDLs with anticancer activity. However, ligands with unintentional affinity for both receptors have been found to produce interesting antiproliferative actions.
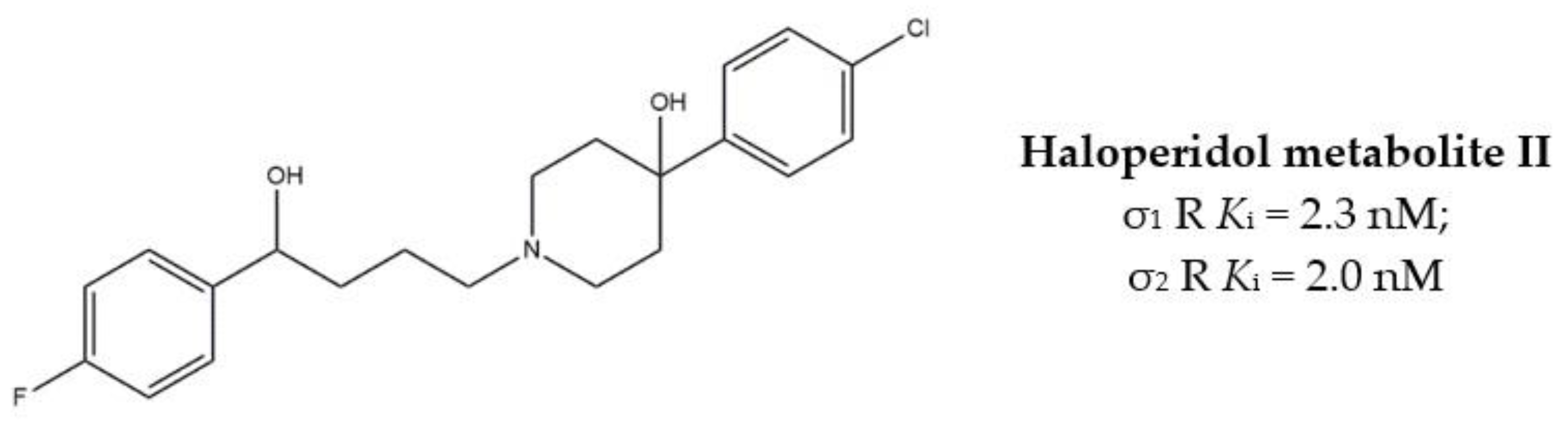
The anticancer effect produced by dual σ receptor ligands was explored by Marrazzo and colleagues, who believed that agonist activity on σ1 receptor and antagonist activity on σ2 receptor may synergize to produce an increased anticancer effect. This pharmacological profile was found in haloperidol metabolite II (HP II) (Figure 1), which was also found to be able to increment intracellular-free calcium [Ca2+]i levels and induce apoptosis [25]. Even if the antiproliferative activity produced by HP II is modest, this compound is the first of a new class of compounds denominated ‘pan-SR ligands class’ [26,27].
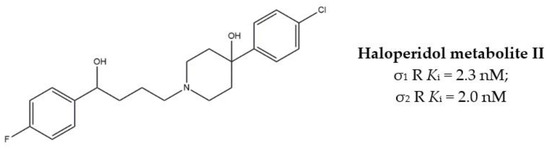
Figure 1.
Haloperidol metabolite II with Ki values for σ receptors.
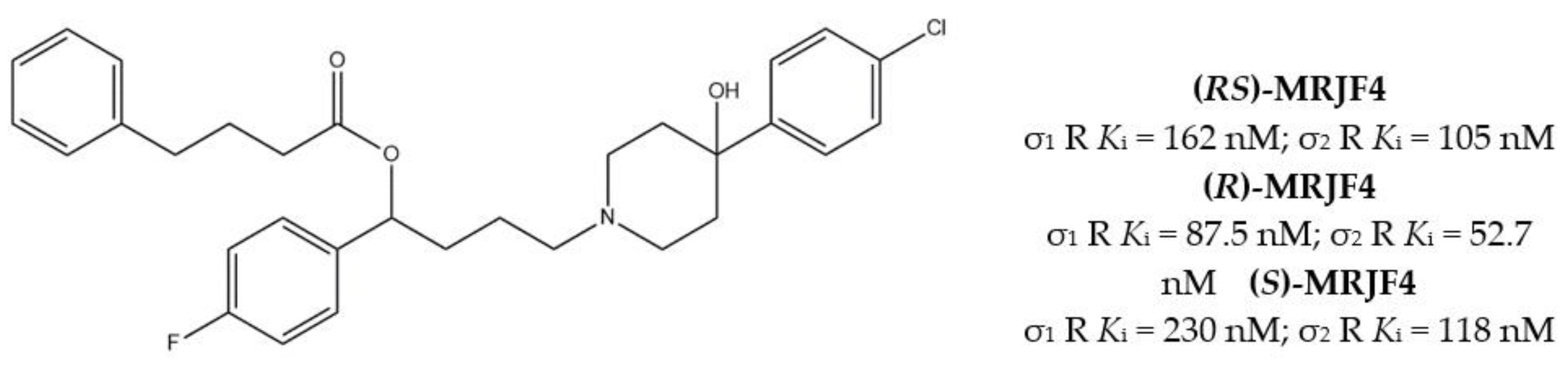
With the aim to extend the pharmacological profile, HP II was conjugated with the Histone Deacetylase (HDAC) inhibitor 4-phenylbutyric acid to produce the ester derivative (R,S)-MRJF4. Despite the higher Ki values for σ receptors (with a slight preference for the σ2 subtype), the MTDL showed a more potent antiproliferative effect in LNCaP and PC3 prostate cancer cells than HP II, administered alone or in co-administration with 4-phenylbutyric acid.
Subsequently, pure enantiomers were also tested and (R)-enantiomer displayed better anticancer activity than racemic mixture, probably because of its lower Ki values for both σ receptors [28] (Figure 2).
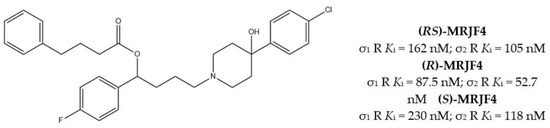
Figure 2.
(RS)-, (R)- and (S)-MRJF4 and Ki values for σ receptors.
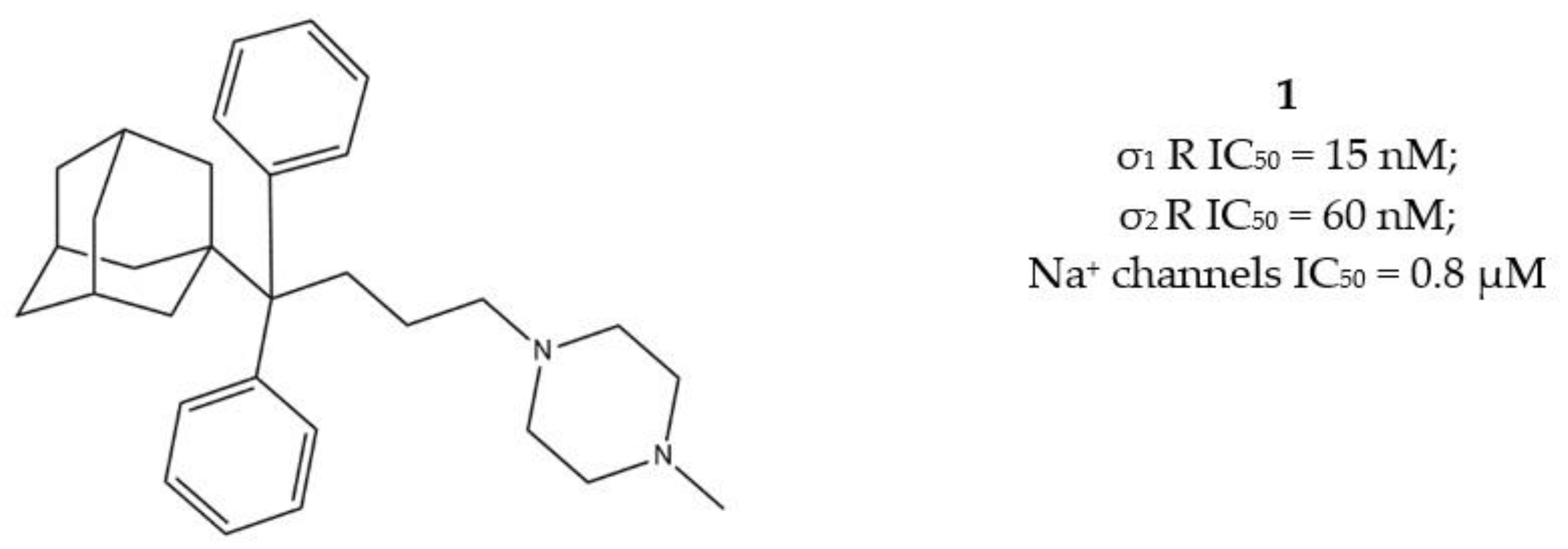
Another step in this direction was taken by Riganas and co-workers, who developed (1-adamantyl)diarylalkylamines in which the adamantyl moiety was introduced with the aim to produce activity at Na+ channels, whose involvement in cancer has been proven [29,30].
Compounds characterized by a butyl chain between the (1-adamantyl)diaryl portion and the amine function, displayed the best profile because of a valuable σ1 receptor antagonist activity, a weak σ2 receptor agonist activity and micromolar affinity for the site 2 of Na+ channels. Overall, piperazine derivative 1 (Figure 3) exerted the best cytotoxic effect on ovarian cancer cells (IGROV-1) and a good in vitro antiangiogenic activity on normal cell lines such as the Human Umbilical Vein Endothelial Cells (HUVEC).
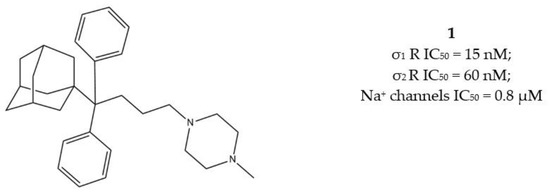
Figure 3.
Compound 1 and its IC50 values for σ receptors and Na+ channels.
Taking into consideration the association of the cytotoxic effect together with the analgesic effect against neuropathic pain obtained by the block of Na+ channels, compound 1 appeared to be endowed with a good therapeutic profile in the treatment of cancer. These few examples show how the σ1 receptor-based development of MTLDs is also a promising approach in the treatment of cancer.
2.2. σ2 Receptor
σ2 receptor was first identified by Hellewell and colleagues who discovered that [3H]-DTG was able to specifically bind two proteins in rat liver: a 25 kDa and a 21.5 kDa protein. Dextrallorphan (DXA) was able to block the binding of the radioligand with the 25 kDa protein (i.e., σ1 receptor), but was unable to block the binding with the 21.5 kD protein, which was named σ2 receptor [5].
In 2006, upon an affinity chromatography purification performed on a lysate from the σ2 overexpressing SK-N-SH neuroblastoma cells, Colabufo et al. advanced the hypothesis of the σ2 receptor identification with histone proteins. Alternatively, the subnanomolar affinity σ2 receptor ligand PB28, whose amino-derivative was used for the functionalization of the chromatographic column, could bind the histones (i.e., H3.3A, H2B, H2A.5 and H2.1) which were identified by MALDI-MS and LC-MS-MS [31].
To support this hypothesis, the interaction between PB28 and the histone proteins was evaluated by computational approaches (homology model and docking) that highlighted two specific high-affinity binding sites for PB28 on the H2A/H2B histone dimer. Experimental supports were provided by: (i) the verified nanomolar affinity binding of [3H]PB28 [32] with the reconstituted human histone dimer H2A/H2B, and (ii) the higher concentration of [3H]PB28 in the nuclei rather than in the cytosol of cancer cells. These data suggested that the effect of PB28 may be exerted through the interaction with nuclear proteins [33]. Nevertheless, studies with σ2 receptor fluorescent ligands did not show nuclear localization [34,35,36], and a new hypothesis about the σ2 receptor identity emerged. Xu and colleagues proposed that the σ2 receptor binding site belonged to the progesterone receptor membrane component 1 (PGRMC1) complex. The study was based on two important pieces of evidence: (i) the fluorescein-azido-derivative WC-21, a σ2 receptor fluorescent ligand, was able to irreversibly bind PGRMC1 in rat liver; (ii) the nitrobenzofurazan-carbamate derivative SW120 colocalized with PGRMC1 and with molecular markers of ER and mitochondria in HeLa cells. The hypothesis was also validated by radioligand binding experiments performed on overexpressing and knockdown PGRMC1 cell lines [37]. On this basis, subsequent studies treated the σ2 receptor and PGRMC1 as the same entity despite some inconsistencies, and pharmacological tools useful to detect PGRMC1 mediated activity were used to define the pharmacological action of σ2 receptor ligands.
The matter about the identity of the σ2 receptor was reopened few years later, when two separate groups conducted independent experiments that led to the same conclusion. Chu and colleagues knocked-out (KO) or overexpressed PGRMC1 in mouse motor neuron cell lines (NSC34). Binding studies with [3H]-DTG and photolabeling studies with [125I]-IAF provided similar results in all cells: wild type, PGRMC1-KO and overexpressing PRGMC1. Moreover, affinities of DTG and haloperidol for PGRMC1, were found to be three orders of magnitude lower than the values reported for the σ2 receptor [38].
At the same time, through a combination of Western blot and radioligand binding assays, Pati et al. demonstrated that the overexpression or knock-down (KD) of PGRMC1 in MCF7 cells (widely used as a model for σ2 receptor activity) did not change the density of σ2 receptors. These results were corroborated by flow cytometry and confocal microscopy experiments in the same cells [39]. The σ2 receptor fluorescent ligands (F412, NO1 and PB385) [34,35,40] used to mark σ2 receptors were displaced by PB28 and DTG, but were not displaced by the PGRMC1 inhibitor AG205 and did not colocalize with the fluorescent anti-PGRMC1 antibody. Importantly, titration calorimetry assays demonstrated that PB28 has no affinity for PGRMC1 dimer or monomer, while DTG showed only modest affinity for the dimer [39].
The last hypothesis about σ2 receptors identity has been proposed by Alon and colleagues in 2017, when the σ2 receptor was identified as the TMEM97 (also known as MAC-30), an ER resident protein involved in cholesterol homeostasis [41] and in the Niemann–Pick type C disease as NPC1-interacting protein [42].
An affinity chromatography purification of calf liver homogenate was performed, and the proteins interacting with the σ2 receptor piperazine ligand JVW-1625 were isolated. Binding studies using [3H]DTG were performed on the detected membrane proteins upon overexpression in HEK293 cells, and TMEM97 emerged as the most likely candidate. Results from several experiments led the authors to conclude that TMEM97 is synonymous of the σ2 receptor. In particular, (i) in PC-12 cells, the KD of TMEM97 produced a decrease in σ2 receptors expression; (ii) in Sf9 insect cells, modified to overexpress the human TMEM97, binding affinity of [3H]DTG was comparable to the values reported in the literature for σ2 receptor; (iii) σ2 ligands belonging to diverse chemical classes, showed binding affinities for TMEM97 consistent with their previously reported σ2 receptor-binding affinities; (iv) the Ki values of two TMEM97 ligands, Elacridar and Ro 48-8071, to Sf9 membranes overexpressing TMEM97, were identical to those measured in MCF7 (that natively overexpress σ2 receptors); (v) site-directed mutagenesis performed on Glutamate (E) and Aspartate (D) residues revealed that D29 and D56 are necessary for ligand binding, similarly to what happens in the σ1 receptor binding site [7].
Moreover, during the drafting of this review, the same group that identified the TMEM97 as the σ2 receptor, published the crystal structure of the protein in complex with roluperidone and PB28, shedding light on the most intriguing mystery of the story of this class of receptors [8].
2.2.1. σ2 Receptor Reference Ligands
Scientific literature reports a plethora of more or less selective ligands for the σ2 receptor. Most of them have been recently reviewed [9] and some interesting classes are briefly described below and reported in Table 1:
- Morphans. They were developed in the 1990s, and emerged as the first class of selective σ2 receptor ligands. These compounds were obtained by insertion of a benzylidene moiety in 8-position of the morphan system that is endowed with mixed affinity for µ and σ2 receptors [43,44].
- Indoles (Siramesine-like compounds). They were developed starting from indole-3-yl-alkyl-arylpiperazines as 5-HT1A agonists [45]. This class of compounds was generated through replacement of the piperazine with constrained arylpiperidines and introduction of a 4-florophenyl group at the indole N-atom leading to siramesine, that despite its subsequently reported lack of selectivity [46], is still widely used as a reference compound. Binding to phosphatidic acid [47], ROS formation [48], lysosomotropic properties [49], release of cytochrome C by mitochondria [50] were reported as mechanisms of action of siramesine, that eventually lead to tumor cells death.
- Granatanes. This class of ligands was developed from BIMU-1, a 5-HT3/5-HT4 serotonin receptor ligands with nanomolar affinity for σ2 receptors. The bicyclo-octane core of BIMU-1 was replaced by a 9-azabicyclo[3.3.1]nonane (granatane). In addition, the cyclic urea group was opened and replaced by a phenylcarbamate moiety [51,52]. Moreover, the nitrogen atom was functionalized with a benzyl group (WC26 and WC59 [53]) or ω-aminoalkyl chains (SV119 and SW43 [54]) leading to optimal σ2 ligands.
- Benzamides. Structural modifications on D3 receptor ligands led to σ2 receptor high affinity flexible and selective benzamides. The most successful compounds, i.e., RHM-1 [55] and ISO-1 [56], were also produced as radioligands [57] to perform σ2 receptor binding assays or clinical PET studies for the imaging of tumors [58,59,60,61]. An intramolecular H-bond, which forces the flexible benzamides in a bicyclic conformation, was postulated for the interaction of these compounds with the σ2 binding site. Therefore, compound 2 and analogues, in which such H-bond conformation was mimicked, were produced. The nanomolar affinities shown by the rigid bicyclic benzamides validated the hypothesis [62,63]. Importantly, high selectivity rates characterized flexible and rigid benzamides. Corresponding reverse amides were produced (rigid and flexible anilides) as well as the corresponding flexible and rigid anilines, with agreeing results [64].
- Cyclohexyl piperazines. This class, which is based on its lead compound PB28, was developed starting from serotoninergic arylpiperazines [65]. During the last few decades, several analogues were developed and recently reviewed, mostly with the aim of reducing the lipophilicity, as studies on [11C]-radiolabelled PB28 showed high nonspecific binding in mouse brain in vivo [66,67]. However, within the more polar series no ligand showed affinity values comparable to PB28 [68]. Nonetheless, interesting biological profiles in terms of selectivity (compound 3, Table 1) or cytotoxic activity in cancer cells were obtained. Intriguingly, PB28 showed promising anti-SARS-CoV-2 activity in vitro [69], although the effect was later ascribed to the induction of phospholipidosis as an off-target effect [70].

Table 1.
Affinities for σ1 and σ2 receptors (σ1 R and σ2 R) of reference σ2 R ligands.
Table 1.
Affinities for σ1 and σ2 receptors (σ1 R and σ2 R) of reference σ2 R ligands.
| Name | Structure | σ1 R Ki nM | σ2 R Ki nM | Reference |
|---|---|---|---|---|
| Morphans: | - | |||
| (+)-CB-64D | 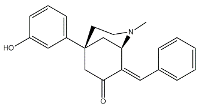 | 3063 | 16.5 | [43] |
| (+)-CB-184 | 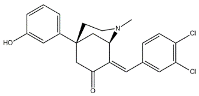 | 7436 | 13.4 | [43] |
| Siramesine | 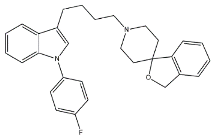 | 10.5 | 12.6 | [46] |
| Granatane: | ||||
| BIMU-1 | 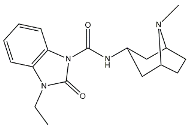 | 6300 | 32 | [51] |
| WC26 | 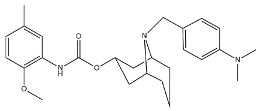 | 1436.5 | 2.58 | [53] |
| WC59 | 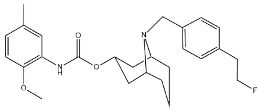 | 1710.5 | 0.82 | [53] |
| SV119 | 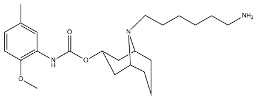 | 1418 | 5.19 | [54] |
| SW43 | 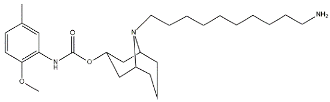 | 134.3 | 7.07 | [54] |
| Benzamides: | ||||
| RHM-1 | 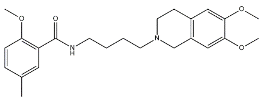 | 3078 | 10.3 | [55] |
| ISO-1 | 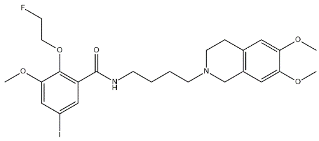 | 2150 | 0.26 | [56] |
| 2 | 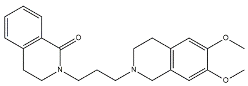 | 709 | 4.74 | [62] |
| Cyclohexyl piperazines: | ||||
| PB28 | 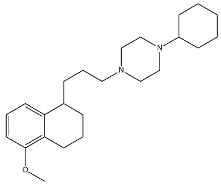 | 0.38 | 0.68 | [68] |
| 3 | 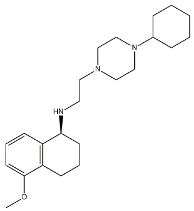 | 23.3 | 8.52 | [68] |
2.2.2. σ2 Receptor Involvement in Cancer
σ2 receptors are associated with a well-established involvement in cell proliferation, although, recently, strong pieces of evidence have also demonstrated the involvement of this receptor in neurodegenerative diseases such as the AD [71]. Indeed, a small σ2 receptor modulator, named CT1812 (ElaytaR), blocks, via the σ2 receptor, the binding of different Aβ oligomers to neuronal receptors and displaces them reducing Aβ-induced synaptic toxicity. Thus, Elayta has undergone clinical trials, and phase II clinical studies are still recruiting [72], highlighting how the σ2 protein is a promising target for cancer and CNS diseases. Many pieces of evidence demonstrate that σ2 receptors are able to induce cell death through different mechanisms, and σ2 ligands inducing cytotoxicity are identified as σ2 agonists. Among the effects induced by σ2 ligands, lysosomal dysfunction and ROS production were proved as cytotoxic mechanisms [49,73]. Additionally, the σ2 receptor modulates ER stress [74], likely through control of Ca2+ release because of direct and indirect interaction with IP3 receptor, ryanodine receptors and Sarco-Endoplasmic Reticulum Calcium ATPase (SERCA) [75]. Additionally, store-operated Ca2+ Entry (SOCE) downregulation [76], activation of caspase and mitochondrial superoxide production [77] and activation of p53- and caspase-independent apoptotic pathways [78] have been associated with the cytotoxicity of σ2 receptor ligands. Nevertheless, Mach et al. showed that the cytotoxicity of σ2 agonists such as PB28 and SW43, is independent from TMEM97 and PGRMC1. Indeed, KO of TMEM97, PGRMC1 or both proteins in HeLa cells, did not alter the cytotoxic effect of these ligands, whose antiproliferative action does not seem to be mediated neither by TMEM97 nor by PGRMC1 [79]. However, the effect of the two proteins on the above described cytotoxic mechanisms, in the presence of the σ2 ligands still needs to be investigated.
2.2.3. MTDLs Based on Granatane SV119 and SW43
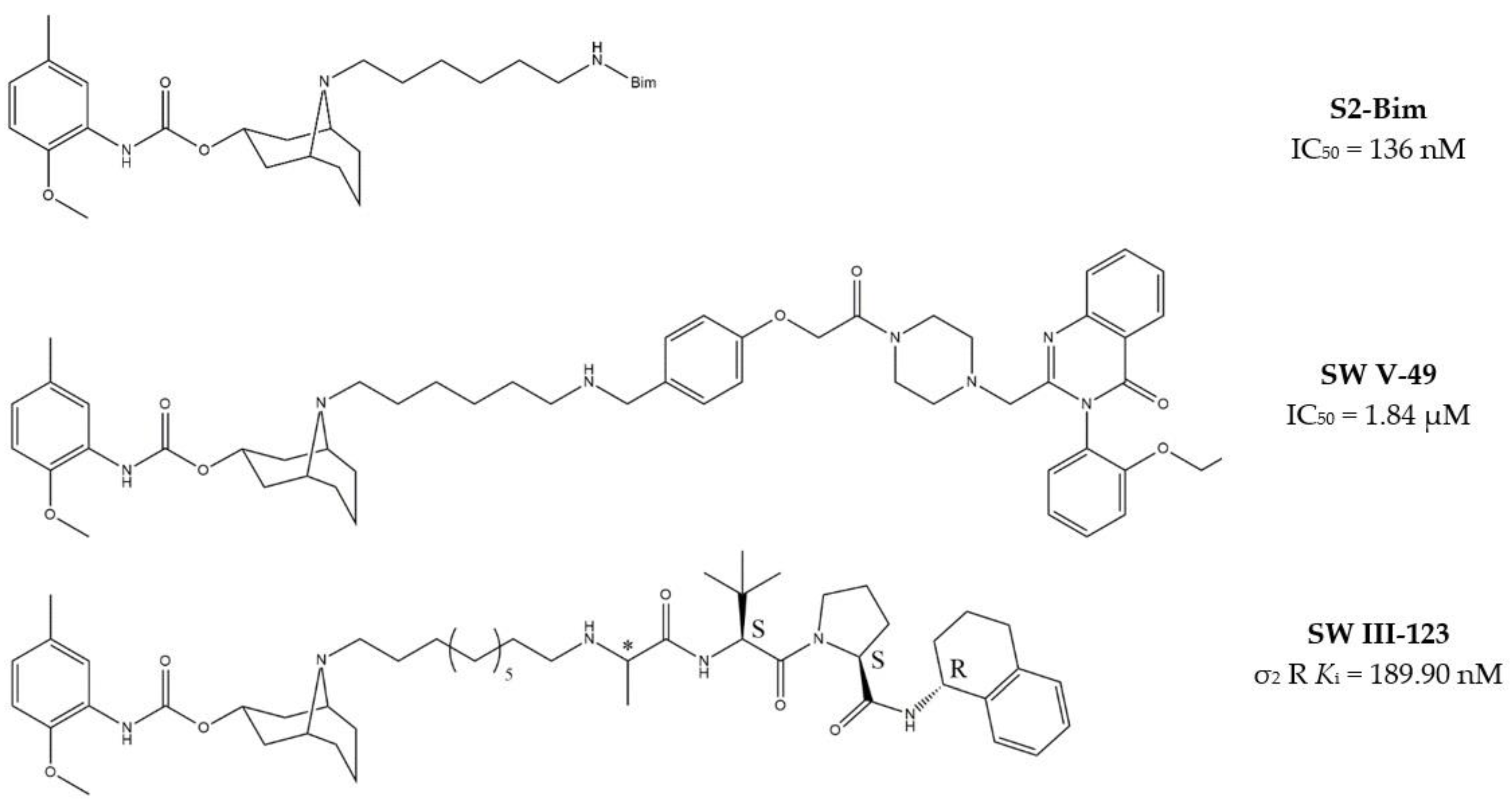
Granatane derivatives are an optimal class of σ2 receptor ligands because of their pharmacological profile in terms of affinity and selectivity. Among all the ligands belonging to this class, analogues bearing an ω-aminoalkyl chain represent ideal tools in the synthesis of MTDLs. The easily conjugatable amino-group can be transformed into the corresponding carbamate and amide derivatives. Among these ligands, Spitzer and colleagues chose granatane derivative SV119 (Table 1), whose intrinsic cytotoxicity, affinity and selectivity for the σ2 receptor prompted its exploitation for the targeted delivery of the MTDL to pancreatic cancer cells. Indeed, SV119 was conjugated with cell-death inducing small molecules such as 1) the pro-apoptotic peptide Bim, (a BH3-only peptide belonging to the Bcl-2 family); 2) the carboxyl-terminal modulator protein (CTMP, a mitochondrial peptide released under apoptotic conditions that inhibits protein kinase B, Akt); 3) rapamycin—a compound with activity on phosphoinositide 3-kinase (PI3K)/Akt system. Among the three MTDLs synthesized, only the peptidomimetic chimera SV119-Bim, named S2-Bim (Figure 4), produced promising effects in in vivo models of pancreatic cancer. Thus, the intrinsic cytotoxic properties of SV119 were reinforced by activation of the Bcl-2 cell death mechanism produced by Bim. However, once the treatment was discontinued, tumor growth restarted and matched the control [80].
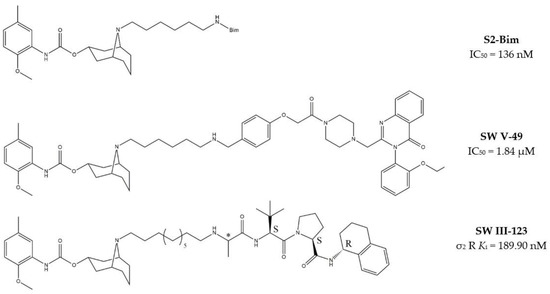
Figure 4.
Granatane-based MTDLs: S2-Bim (peptide sequence: EIWIAQELRRIGDEFNAYYAR-OH), SW V-49 and SW III-123 structures and biological data.
In the following years, granatane SV119 was also linked to Erastin, a 4-quinazolinone able to inhibit cysteine/glutamate antiporter, whose function is to keep the antioxidant glutathione homeostasis. Besides the induction of ROS accumulation, Erastin can influence the activity of mitochondrial voltage-dependent anion channels, although problems of cell uptake limit its efficacy.
With the aim to obtain an Erastin based dual drug, Ohman and colleagues, demethylated the carbon atom between the piperazine ring and the 4-quinazolinone portion in order to get rid of the chirality. The obtained demethylated-Erastin was then connected to the granatane SV119 to obtain the new MTDL named SW V-49 (Figure 4). This dual drug was characterized by a better cell uptake compared to Erastin and encouraging cytotoxicity IC50 values in pancreatic cancer cells. In vivo studies performed on murine models of pancreatic cancer demonstrated that SW V-49 reduces tumor size and improves survival, without side-effects that are typical of gemcitabine, the reference therapeutic for pancreatic tumor treatments [81]. Always with the aim to increase the drug delivery of peptidomimetics to cancer cells, SW43 (Table 1), another granatane derivative with a longer ω-aminoalkychain, was linked to SW IV-52s, that is a mimetic compound (SMC) of the second mitochondria-derived activator of caspase (Smac) (Figure 4). Thus, the peptidomimetic derivative SW III-123 (Figure 4) was obtained and displayed a strong cytotoxicity in all ovarian cancer cell lines studied (SKOV-3, CaOV-3 and BG-1), differently from SW IV-52s, that is active only in SKOV-3. Studies performed in SKOV-3 (EC50 = 4.0 µM (24 h); EC50 = 1.40 µM (48 h)) cells to better understand cell death mechanisms revealed that the peptidomimetic ligand SW III-123 can produce degradation of inhibitor of apoptosis proteins 1 and 2 (cIAP1 and cIAP2), accumulation of NF-κB-inducing kinase (NIK) and phosphorylation of NF-κB p65. All together, these mechanisms suggest the activation of canonical and noncanonical NF-κB pathways of death. In addition, cleavage of caspase-3, -8 and -9 was observed, and the involvement of tumor necrosis factor alpha (TNFα) in the mechanisms of cell death was demonstrated for the SMC-sensitive cells, but not for the SMC-insensitive ones [82]. While in SW III-123 the chiral centers in the proline and tetralin rings and the chirality of the carbon atom bearing the t-butyl group were all defined, the oxopropan-2-yl group was not, so that SW III-123 was a racemic mixture. The promising results obtained with this couple of stereoisomers led to produce the pure stereoisomeric form of SW III-123 in the L-configuration to obtain SW IV-134, which was tested in pancreatic tumor cells and provided better results than the racemate, with a ten-fold higher affinity for σ2 receptors (σ2 Ki = 22.6 nM) [83].
Thus, SW IV-134 was studied in different types of tumors, such as mouse xenograft models of ovarian cancer [84] and triple-negative breast cancer [85] providing promising results.
It is worth noticing that even if these peptidomimetics are characterized by features that are usually detrimental in terms of pharmacokinetic properties, such as high molecular weight and steric hindrance, valuable affinity values and promising preclinical data were obtained. Therefore, the granatane scaffold can be considered as an important tumor-selective delivery system with the potential to reduce the side effects produced by unselective therapies.
2.2.4. Collateral Sensitivity (CS) as Multitarget Strategy to Face Cancer
Collateral sensitivity (CS) was observed for the first time by Szybalaski and Bryson in 1952 during some studies on drug-resistant cultures of Escherichia coli [86]. According to this phenomenon, cancer cells, that show resistance to classical chemotherapy, demonstrate an unusual sensitivity to other drugs. One of the main mechanisms of the resistance to drugs, also called MultiDrug Resistance (MDR), is due the overexpression of efflux pumps that reduce intracellular drug concentrations to ineffective levels [87]. Among all the efflux pumps, P-glycoprotein (P-gp), MultidrugResistance-associated Protein 1 (MRP1) and Breast Cancer Resistance Protein (BCRP), which belong to the ATP-Binding Cassette (ABC) transporters, are the main responsible of this phenomenon.
Although it may sound like a contradiction, the overexpression of the efflux pumps that is responsible of the MDR, may also account for CS. Indeed, the over-working of these transporters can: (i) activate a futile hydrolysis of ATP, increasing ROS levels; (ii) sensitize cells to changes in energy levels; (iii) produce extrusion of essential substrates for cell metabolism; (iv) perturb cell membranes [88]. Drugs able to engage these mechanisms, are able to kill MDR cells more than the non-resistant counterparts, finally leading to select the non-resistant cell population, which can ultimately be treated with the conventional chemotherapeutic.
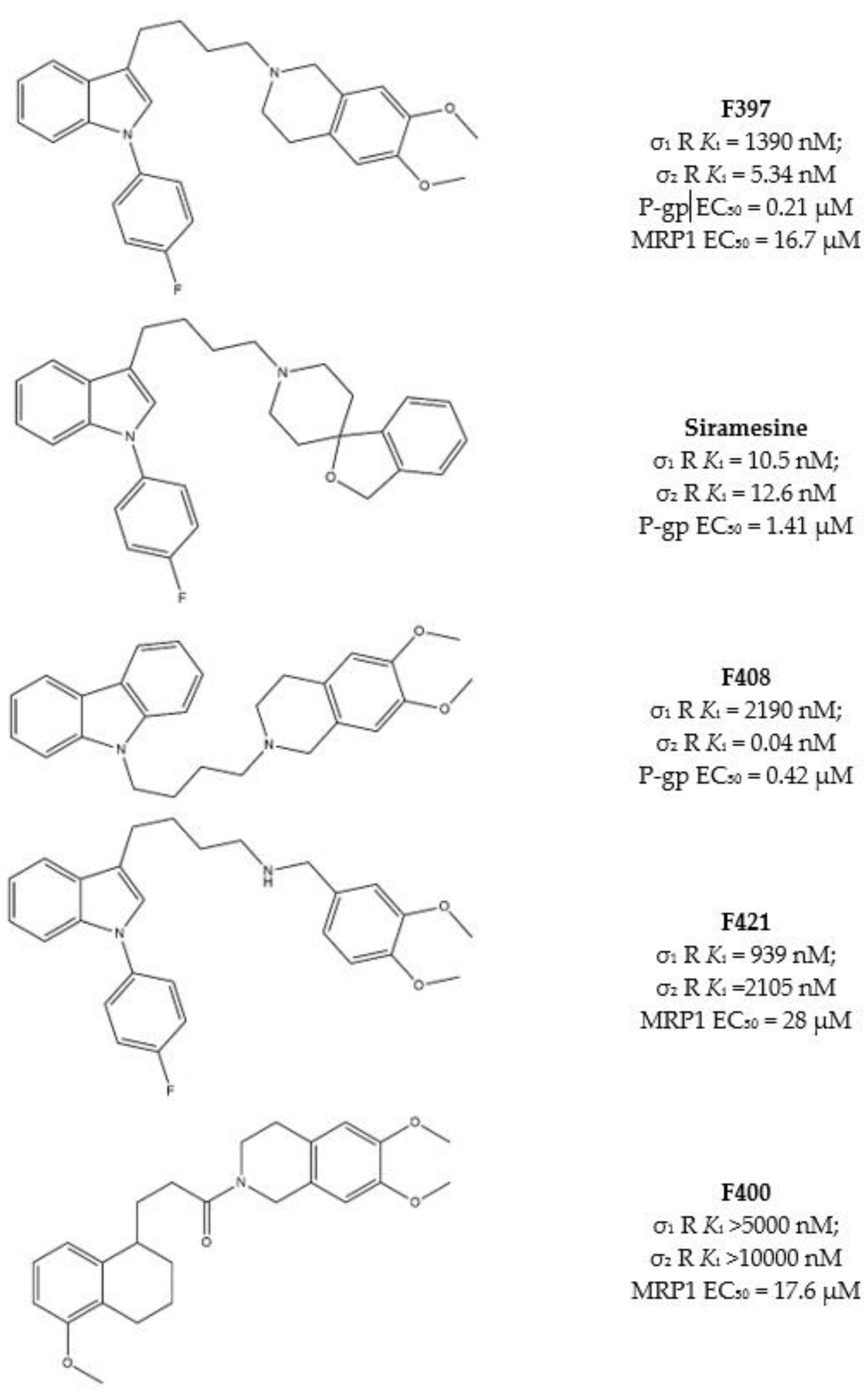
As some σ receptor ligands are P-gp modulators, Niso and colleagues, studied the ability of some σ2 receptor ligands to exert CS, exploiting their interaction with P-gp [46,62,63,89,90]. Ligands were chosen because of the presence of some basic moieties (i.e., 4-(4-fluorophenyl) and 4-cyclohexyl -piperidines or -piperazines and 6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline) and hydrophobic groups (i.e., carbazole, tetraline, indole and N-(4-fluoro-phenyl)indole) with consolidated σ2 receptor affinities together with their likely interaction with P-gp. Combination of these basic and hydrophobic moieties led to several high affinity σ2 receptor ligands, and the indole-based compounds F397, Siramesine and the carbazole derivative F408 (Figure 5) displayed the most promising dual-target profile because of their affinity/activity at σ2 receptor and P-gp. However, CS was only exerted by Siramesine and F408, with a more potent cytotoxicity in MCF7dx (MDR cells) than in the parent MCF7 (non-MDR) cell line. ATP consumption in the same couple of cells (MCF7 and MCF7dx) was evaluated, and while ATP content was lower in MCF7dx than in MCF7 upon F408 and Siramesine administration, the same effect on ATP was not exerted by F397. These data together demonstrated that the CS properties of F408 and Siramesine are due to their activity at P-gp as substrates, with the activation of the futile ATP cycle (to sustain the active efflux of the drug) and increased ROS production. By contrast, F397 was devoid of such an effect because it is a P-gp inhibitor with no ATP consumption and therefore devoid of CS properties mediated by P-gp.
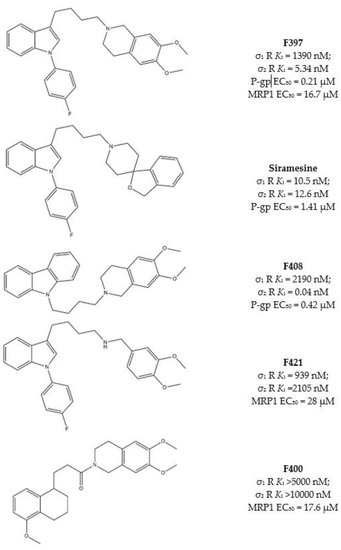
Figure 5.
F397, Siramesine, F408, F421 and F400 structures and Ki values for σ receptors, EC50 for P-gp or MRP1.
These compounds were investigated in other cell lines couples, such as HT29/HT29dx (colorectal cancer) and A549/A549dx (lung cancer) providing results in accordance with the CS exerted in MCF7 cell lines pairs. Again, F408 and Siramesine demonstrated their CS properties, which were exceptionally important in the HT29 cell lines pair for F408. Indeed, in HT29dx the carbazole derivative F408 produced a 60% cell death at 1 µM concentration. Moreover, studies performed on the mitochondrial respiratory chain revealed that treatment of resistant cells with F408 and Siramesine reduced electron flux and ATP supply, so that these ligands are able to activate multiple cytotoxicity mechanisms [91] and appear as MTDLs worthy to be explored to overcome MDR.
Subsequently, similar studies were performed on the lesser-studied MRP1, which effluxes glutathione (GSH), the tripeptide essential for the correct redox state of cells. Verapamil, the L-type calcium antagonist, can bind MRP1 and stimulate GSH massive extrusion that leads to the activation of apoptotic mechanisms. These properties render Verapamil a collateral sensitizer upon interaction with MRP1 [88].
Structure similarity between Verapamil and some σ2 receptor ligands prompted Riganti et al. to screen a library of σ2 ligands for their MRP1 activity. Among the most active compounds, the indole-based structures F397 [46] and F421 [90] and the tetralin based amide F400 (Figure 5), [89] were valuable modulators of MRP1 and showed CS in different cells. In particular, F397, F421 and F400 induced CS in MDCK/MDCK-MRP1, A549/A549dx and HT29/HT29dx, and cytotoxicity in MCF7, SKBR3, T74D and MDA-MB-231.
All three compounds were found to deregulate GSH/GSSG ratio and increase ROS production, producing cytotoxicity, especially in MRP1 overexpressing cells.
It is worth noting that F397 did not exert P-gp-mediated CS in MCF7/MCF7dx cells (see above), but emerged as a MRP1-mediated collateral sensitizer in cells where MRP1 is overexpressed. Indeed, MCF7dx cells are devoid of MRP1, so that the data from F397 strongly support the involvement of the P-gp in the CS exerted by F408 and siramesine, while MRP1 is involved in the CS induced by F397, F421 and F400.
These pieces of evidence prompted to further investigate the effect of these σ2 receptor ligands in co-administration with cis-Pt, that is one of the clinical antitumor drugs that suffers from MDR. In vitro and in vivo combinations of F397 or F421 with Cis-Pt re-sensitized A549dx cells to cis-Pt and reduced tumor growth without signs of toxicity [92].
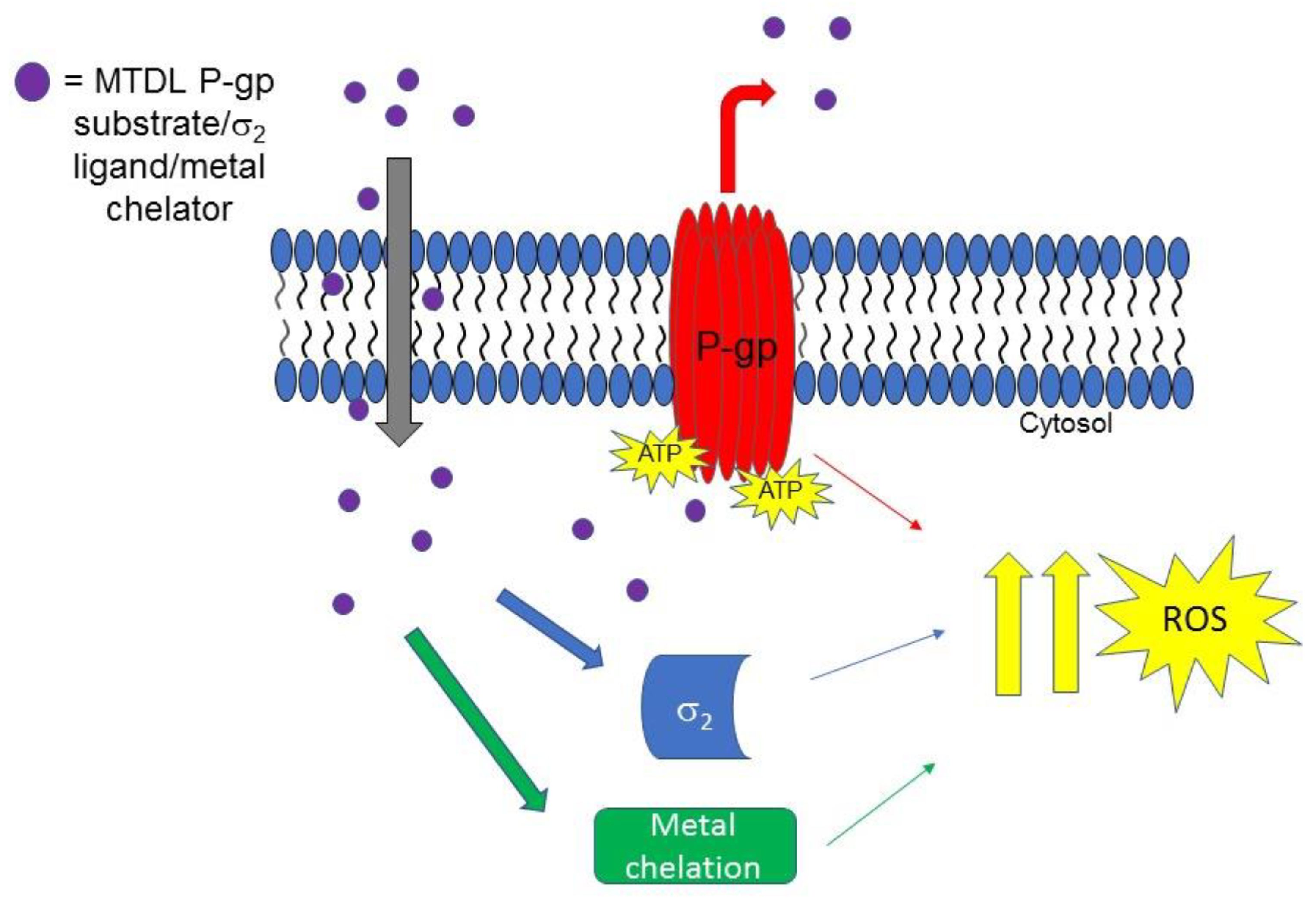
Another step forward on the development of MTDLs based on σ2 receptor for the treatment of tumors was taken when novel isatin-β-thiosemicarbazones (IβTs) were produced. IβTs, upon chelation of metals (such as iron and cupper ions) promote ROS production, and have been previously reported as CS inducer [93]. Therefore, according to an MTLDs approach, IβTs core was functionalized with σ2 targeting basic moieties with the aim to improve the selectivity for cancer cells that overexpress σ2 receptors [94]. In Figure 6, the MTDL strategy targeting σ2 receptors P-gp efflux pump and metal chelation is depicted.
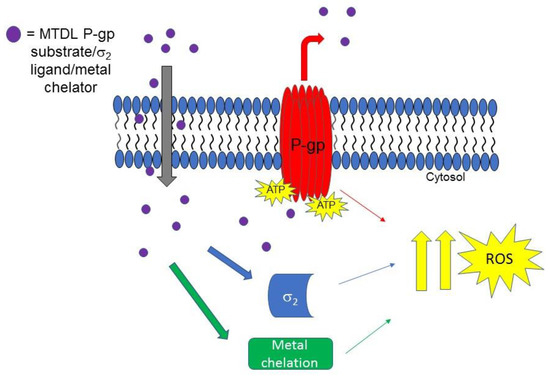
Figure 6.
MTDL acting upon interaction with σ2 and P-gp proteins and metal chelation. The MTDL, that is a P-gp substrate, upon efflux, activates the futile ATP cycle that leads to ROS increase. The effect is synergized by interaction with σ2 receptor and metal ions chelation.
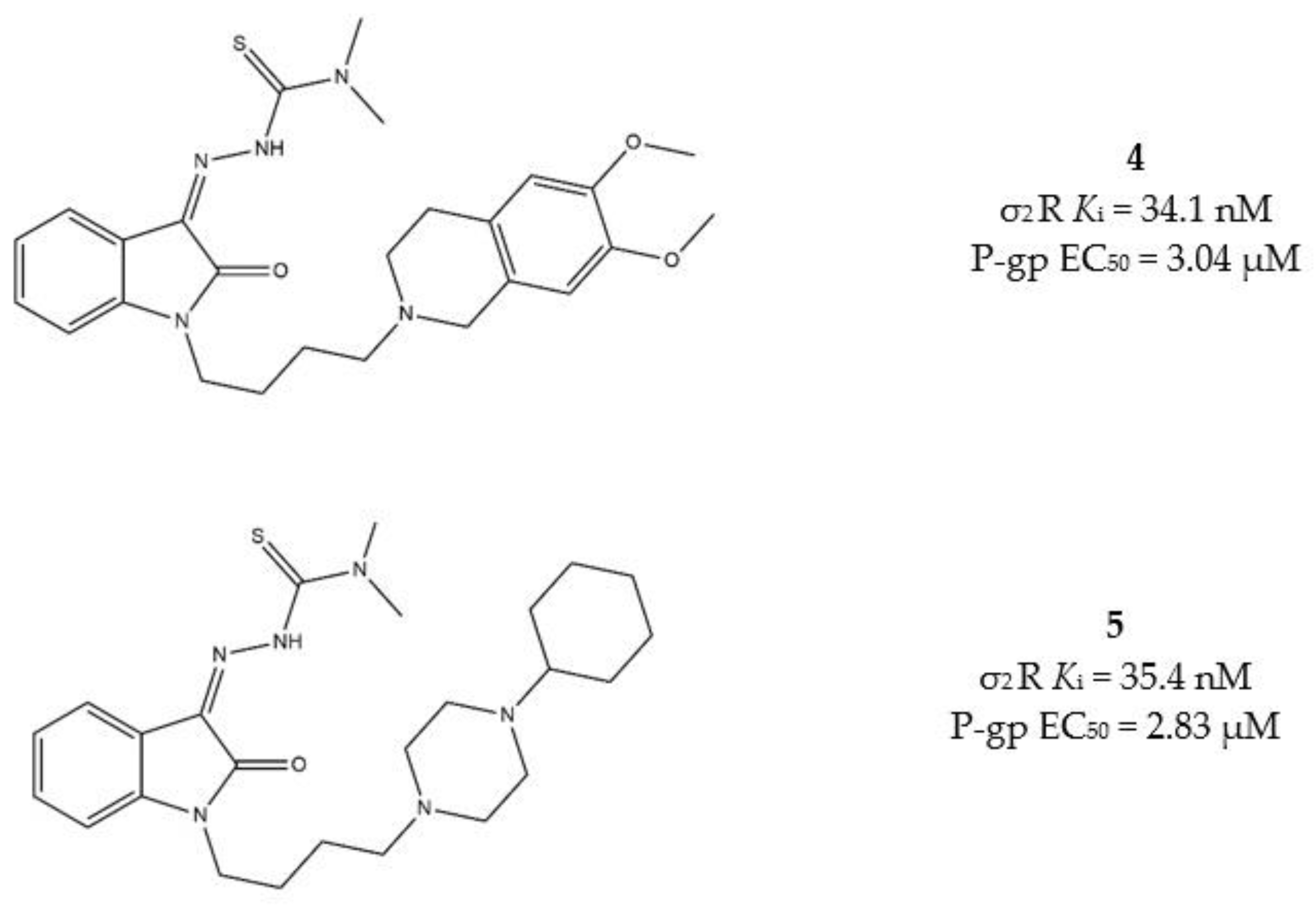
The same basic moiety should also determine the interaction with the P-gp. Among the diverse IβTs, the N,N-dimethylthiosemicarbazones bearing the 1-cyclohexylpiperazine, or the 6,7-dimethoxytetrahydroisoquinoline as the basic moieties (compounds 4 and 5, Figure 7), provided the best results in terms of cytotoxicity in MCF7/MCF7dx and A549/A549dx, showing important activity also against MDR cells. Therefore, these two compounds underwent a rational deconstruction approach in order to elucidate their structure–activity relationships (SAR) and understand the impact of each target in the overall effect [95]. The activity of the novel compounds was evaluated in pancreatic tumors in vitro, in a panel of human (MIAPaCa-2, BxPC3, AsPC1 and Panc-1) and mouse (Panc02, KP02 and KCKO) cancer cell lines, and in vivo in a murine KP02 tumor model (C57BL/6 mice). This study revealed that the sole N,N-dimethylthiosemicarbazone portion is responsible of the cytotoxicity also in the absence of the σ2 receptor targeting moiety. Nevertheless, the IβTs devoid of the σ2 targeting basic moiety resulted in foci of pulmonary metastases in mice, while the σ2 targeting IβTs were equally effective and devoid of side effects. The investigated mechanism of action of σ2 receptors targeting compounds showed ROS increase, caspase-3 activation and mitochondria superoxide production [95].
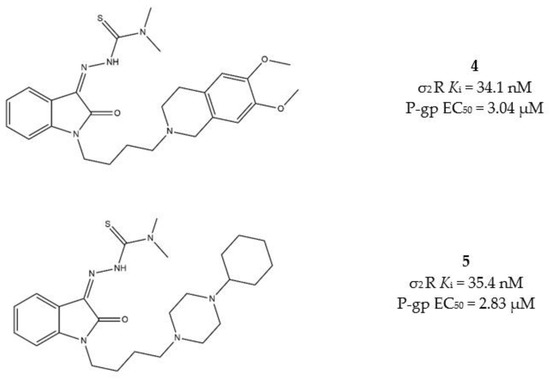
Figure 7.
Structures 4 and 5 and their Ki for σ2 receptors and EC50 values for P-gp.
All these data together support the higher selectivity for cancer cells that the σ2 targeting exerts. Therefore, according to a multitarget perspective, both N,N-dimethyl-thiosemicarbazone and σ2 receptors targeting basic moieties are advisable to produce a strong and selective antitumor activity and poor side effects.
3. Conclusions
The present review summarizes the progress in the knowledge about σ receptors with a focus on the σ2 subtype’s involvement in the oncology field. The role in the proliferative status of cells has directed the σ2-related scientific interest towards cancer research. Overall, we have described herein the most promising σ2 receptor ligands in the perspective of anticancer therapies, with a focus on the development of σ2-based MTDLs ligands. The combination of the σ2 targeting moieties, with molecules that activate different apoptotic pathways results in either a synergistic antitumor action or in a targeted delivery to cancers that overexpress the σ2 protein. The increasing structural knowledge about this receptor, that has culminated with the disclosure of the crystal structure, together with the successful examples reported herein, may spark novel studies to exploit the σ2 subtype as an innovative strategy for the development of MTDLs as anticancer polypharmacological agents, with targeted delivery and improved activity against (resistant) cancers.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer Statistics, 2020. Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Singh, A.; Beechinor, R.J.; Huynh, J.C.; Li, D.; Dayyani, F.; Valerin, J.B.; Hendifar, A.; Gong, J.; Cho, M. Immunotherapy Updates in Advanced Hepatocellular Carcinoma. Cancers 2021, 13, 2164. [Google Scholar] [CrossRef]
- Teoh, P.J. CAR T-Cell Therapy in Multiple Myeloma: More Room for Improvement. Blood Cancer J. 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. The Hallmarks of Cancer Review Douglas. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Hellewell, S.B.; Bowen, W.D. A Sigma-like Binding Site in Rat Pheochromocytoma (PC12) Cells: Decreased Affinity for (+)-benzomorphans and Lower Molecular Weight Suggest a Different Sigma Receptor Form from that of Guinea Pig Brain. Brain Res. 1990, 527, 244–253. [Google Scholar] [CrossRef]
- Schmidt, H.R.; Zheng, S.; Gurpinar, E.; Koehl, A.; Manglik, A.; Kruse, A.C. Crystal Structure of the Human σ1 Receptor. Nature 2016, 532, 527–530. [Google Scholar] [CrossRef]
- Alon, A.; Schmidt, H.R.; Wood, M.D.; Sahn, J.J.; Martin, S.F.; Kruse, A.C. Identification of the Gene That Codes for the σ2 Receptor. Proc. Natl. Acad. Sci. USA 2017, 114, 7160–7165. [Google Scholar] [CrossRef]
- Alon, A.; Lyu, J.; Braz, J.M.; Tummino, T.A.; Craik, V.; Matthew, J.; Webb, C.M.; Radchenko, D.S.; Moroz, Y.S.; Huang, X.; et al. Crystal Structures of the σ2 Receptor Template Large-Library Docking for Selective Chemotypes Active in Vivo. bioRxiv 2021. [Google Scholar] [CrossRef]
- Abate, C.; Niso, M.; Berardi, F. Sigma-2 Receptor: Past, Present and Perspectives on Multiple Therapeutic Exploitations. Future Med. Chem. 2018, 10, 1997–2018. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, H.R.; Kruse, A.C. The Molecular Function of σ Receptors: Past, Present, and Future. Trends Pharmacol. Sci. 2019, 40, 636–654. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, T.; Su, T.P. Sigma-1 Receptor Chaperones at the ER- Mitochondrion Interface Regulate Ca2+ Signaling and Cell Survival. Cell 2007, 131, 596–610. [Google Scholar] [CrossRef]
- Su, T.P.; Hayashi, T.; Maurice, T.; Buch, S.; Ruoho, A.E. The Sigma-1 Receptor Chaperone as an Inter-Organelle Signaling Modulator. Trends Pharmacol. Sci. 2010, 31, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Anavex® Life Sciences Corp. Available online: https://www.anavex.com/pipeline/ (accessed on 28 April 2021).
- PRidopidine’s Outcome on Function in Huntington Disease, PROOF- HD; ClinicalTrials.Gov Identifier: NCT04556656; ClinicalTrials.gov: Bethesda, MA, USA, 2020.
- Abatematteo, F.S.; Niso, M.; Contino, M.; Leopoldo, M.; Abate, C. Multi-Target Directed Ligands (MTDLs) Binding the σ1 Receptor as Promising Therapeutics: State of the Art and Perspectives. Int. J. Mol. Sci. 2021, 22, 6359. [Google Scholar] [CrossRef]
- Sereti, E.; Tsimplouli, C.; Kalaitsidou, E.; Sakellaridis, N.; Dimas, K. Study of the Relationship between Sigma Receptor Expression Levels and Some Common Sigma Ligand Activity in Cancer Using Human Cancer Cell Lines of the Nci-60 Cell Line Panel. Biomedicines 2021, 9, 38. [Google Scholar] [CrossRef]
- Simony-Lafontaine, J.; Esslimani, M.; Bribes, E.; Gourgou, S.; Lequeux, N.; Lavail, R.; Grenier, J.; Kramar, A.; Casellas, P. Immunocytochemical Assessment of Sigma-1 Receptor and Human Sterol Isomerase in Breast Cancer and Their Relationship with a Series of Prognostic Factors. Br. J. Cancer 2000, 82, 1958–1966. [Google Scholar] [CrossRef]
- Wang, B.; Rouzier, R.; Albarracin, C.T.; Sahin, A.; Wagner, P.; Yang, Y.; Smith, T.L.; Bernstam, F.M.; Marcelo, A.C.; Hortobagyi, G.N.; et al. Expression of Sigma 1 Receptor in Human Breast Cancer. Breast Cancer Res. Treat. 2004, 87, 205–214. [Google Scholar] [CrossRef]
- Xu, Q.X.; Li, E.M.; Zhang, Y.F.; Liao, L.D.; Xu, X.E.; Wu, Z.Y.; Shen, J.H.; Xu, L.Y. Overexpression of Sigma1 Receptor and Its Positive Associations With Pathologic TNM Classification in Esophageal Squamous Cell Carcinoma. J. Histochem. Cytochem. 2012, 60, 457–466. [Google Scholar] [CrossRef]
- Xu, D.; Yi, W.; Chen, Y.; Ma, L.; Wang, J.; Yu, G. Overexpression of Sig1R Is Closely Associated with Tumor Progression and Poor Outcome in Patients with Hilar Cholangiocarcinoma. Med. Oncol. 2014, 31, 1–7. [Google Scholar] [CrossRef]
- Aydar, E.; Onganer, P.; Perrett, R.; Djamgoz, M.B.; Palmer, C.P. The Expression and Functional Characterization of Sigma (σ) 1 Receptors in Breast Cancer Cell Lines. Cancer Lett. 2006, 242, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Caveliers, V.; Everaert, H.; Lahoutte, T.; Dierickx, L.O.; John, C.S.; Bossuyt, A. Labelled Sigma Receptor Ligands: Can Their Role in Neurology and Oncology Be Extended? Eur. J. Nucl. Med. 2001, 28, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Kim, F.J.; Maher, C.M. Sigma1 Pharmacology in the Context of Cancer. Handb. Exp. Pharmacol. 2017, 244, 237–308. [Google Scholar] [CrossRef]
- Yano, H.; Bonifazi, A.; Xu, M.; Guthrie, D.A.; Schneck, S.N.; Abramyan, A.M.; Fant, A.D.; Hong, W.C.; Newman, A.H.; Shi, L. Pharmacological Profiling of Sigma 1 Receptor Ligands by Novel Receptor Homomer Assays. Neuropharmacology 2018, 133, 264–275. [Google Scholar] [CrossRef]
- Brent, P.J.; Pang, G.; Little, G.; Dosen, P.J.; Van Helden, D.F. The Sigma Receptor Ligand, Reduced Haloperidol, Induces Apoptosis and Increases Intracellular-Free Calcium Levels [Ca2+]i in Colon and Mammary Adenocarcinoma Cells. Biochem. Biophys. Res. Commun. 1996, 219, 219–226. [Google Scholar] [CrossRef]
- Sozio, P.; Fiorito, J.; Di Giacomo, V.; Di Stefano, A.; Marinelli, L.; Cacciatore, I.; Cataldi, A.; Pacella, S.; Turkez, H.; Parenti, C.; et al. Haloperidol Metabolite II Prodrug: Asymmetric Synthesis and Biological Evaluation on Rat C6 Glioma Cells. Eur. J. Med. Chem. 2015, 90, 1–9. [Google Scholar] [CrossRef]
- Rui, M.; Rossi, D.; Marra, A.; Paolillo, M.; Schinelli, S.; Curti, D.; Tesei, A.; Cortesi, M.; Zamagni, A.; Laurini, E.; et al. Synthesis and Biological Evaluation of New Aryl-alkyl(alkenyl)-4-benzylpiperidines, Novel Sigma Receptor (SR) Modulators, as Potential Anticancer-Agents. Eur. J. Med. Chem. 2016, 124, 649–665. [Google Scholar] [CrossRef] [PubMed]
- Marrazzo, A.; Fiorito, J.; Zappal, L.; Prezzavento, O.; Ronsisvalle, S.; Pasquinucci, L.; Scoto, G.M.; Bernardini, R.; Ronsisvalle, G. Antiproliferative Activity of Phenylbutyrate Ester of Haloperidol Metabolite II [(±)-MRJF4] in Prostate Cancer Cells. Eur. J. Med. Chem. 2011, 46, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Fulgenzi, G.; Graciotti, L.; Faronato, M.; Soldovieri, M.V.; Miceli, F.; Amoroso, S.; Annunziato, L.; Procopio, A.; Taglialatela, M. Human Neoplastic Mesothelial Cells Express Voltage-Gated Sodium Channels Involved in Cell Motility. Int. J. Biochem. Cell Biol. 2006, 38, 1146–1159. [Google Scholar] [CrossRef]
- Brackenbury, W.J.; Djamgoz, M.B.A.; Isom, L.L. An Emerging Role for Voltage-Gated Na+ Channels in Cellular Migration: Regulation of Central Nervous System Development and Potentiation of Invasive Cancers. Neuroscientist 2008, 14, 571–583. [Google Scholar] [CrossRef]
- Colabufo, N.A.; Berardi, F.; Abate, C.; Contino, M.; Niso, M.; Perrone, R. Is the σ2 Receptor a Histone Binding Protein? J. Med. Chem. 2006, 49, 4153–4158. [Google Scholar] [CrossRef] [PubMed]
- Colabufo, N.A.; Abate, C.; Contino, M.; Inglese, C.; Ferorelli, S.; Berardi, F.; Perrone, R. Tritium Radiolabelling of PB28, a Potent Sigma-2 Receptor Ligand: Pharmacokinetic and Pharmacodynamic Characterization. Bioorganic Med. Chem. Lett. 2008, 18, 2183–2187. [Google Scholar] [CrossRef] [PubMed]
- Abate, C.; Elenewski, J.; Niso, M.; Berardi, F.; Colabufo, N.A.; Azzariti, A.; Perrone, R.; Glennon, R.A. Interaction of the σ2 Receptor Ligand PB28 with the Human Nucleosome: Computational and Experimental Probes of Interaction with the H2A/H2B Dimer. ChemMedChem 2010, 5, 268–273. [Google Scholar] [CrossRef]
- Abate, C.; Hornick, J.R.; Spitzer, D.; Hawkins, W.G.; Niso, M.; Perrone, R.; Berardi, F. Fluorescent Derivatives of σ Receptor Ligand 1-Cyclohexyl-4-[3-(5-methoxy-1,2,3,4-tetrahydronaphthalen-1-yl)propyl]piperazine (PB28) as a Tool for Uptake and Cellular Localization Studies in Pancreatic Tumor Cells. J. Med. Chem. 2011, 54, 5858–5867. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Abate, C.; Niso, M.; Marottoli, R.; Riganti, C.; Ghigo, D.; Ferorelli, S.; Ossato, G.; Perrone, R.; Lacivita, E.; Lamb, D.C.; et al. Novel Derivatives of 1-Cyclohexyl-4-[3-(5-methoxy-1,2,3,4-tetrahydronaphthalen-1-yl)propyl]piperazine (PB28) with Improved Fluorescent and σ Receptors Binding Properties. J. Med Chem 2014, 57, 3314–3323. [Google Scholar] [CrossRef]
- Zeng, C.; Vangveravong, S.; Jones, L.A.; Hyrc, K.; Chang, K.C.; Xu, J.; Rothfuss, J.M.; Goldberg, M.P.; Hotchkiss, R.S.; Mach, R.H. Characterization and Evaluation of Two Novel Fluorescent Sigma-2 Receptor Ligands as Proliferation Probes. Mol. Imaging 2011, 10, 420–433. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Zeng, C.; Chu, W.; Pan, F.; Rothfuss, J.M.; Zhang, F.; Tu, Z.; Zhou, D.; Zeng, D.; Vangveravong, S.; et al. Identification of the PGRMC1 Protein Complex as the Putative Sigma-2 Receptor Binding Site. Nat. Commun. 2011, 2, 380–387. [Google Scholar] [CrossRef]
- Chu, U.B.; Mavlyutov, T.A.; Chu, M.L.; Yang, H.; Schulman, A.; Mesangeau, C.; McCurdy, C.R.; Guo, L.W.; Ruoho, A.E. The Sigma-2 Receptor and Progesterone Receptor Membrane Component 1 Are Different Binding Sites Derived From Independent Genes. EBioMedicine 2015, 2, 1806–1813. [Google Scholar] [CrossRef]
- Pati, M.L.; Groza, D.; Riganti, C.; Kopecka, J.; Niso, M.; Berardi, F.; Hager, S.; Heffeter, P.; Hirai, M.; Tsugawa, H.; et al. Sigma-2 receptor and Progesterone Receptor Membrane Component 1 (PGRMC1) are two Different proteins: Proofs by Fluorescent Labeling and Binding of Sigma-2 Receptor Ligands to PGRMC1. Pharmacol Res. 2017, 117, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Niso, M.; Riganti, C.; Pati, M.L.; Ghigo, D.; Berardi, F.; Abate, C. Novel and Selective Fluorescent σ2-Receptor Ligand with a 3,4-Dihydroisoquinolin-1-one Scaffold: A Tool to Study σ2 Receptors in Living Cells. ChemBioChem 2015, 16, 1078–1083. [Google Scholar] [CrossRef]
- Bartz, F.; Kern, L.; Erz, D.; Zhu, M.; Gilbert, D.; Meinhof, T.; Wirkner, U.; Erfle, H.; Muckenthaler, M.; Pepperkok, R.; et al. Identification of Cholesterol-Regulating Genes by Targeted RNAi Screening. Cell Metab. 2009, 10, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi-Fakhari, D.; Wahlster, L.; Bartz, F.; Werenbeck-Ueding, J.; Praggastis, M.; Zhang, J.; Joggerst-Thomalla, B.; Theiss, S.; Grimm, D.; Ory, D.S.; et al. Reduction of TMEM97 Increases NPC1 Protein Levels and Restores Cholesterol Trafficking in Niemann-Pick Type C1 Disease Cells. Hum. Mol. Genet. 2016, 25, 3588–3599. [Google Scholar] [CrossRef]
- Bowen, W.D.; Bertha, C.M.; Vilner, B.J.; Rice, K.C. CB-64D and CB-184: Ligands with High σ2 Receptor Affinity and Subtype Selectivity. Eur. J. Pharmacol. 1995, 278, 257–260. [Google Scholar] [CrossRef]
- Bertha, C.M.; Mattson, M.V.; Flippen-anderson, J.L.; Rothman, R.B.; Xu, H.; Cha, X.; Becketts, K.; Rice, K.C. A Marked Change of Receptor Affinity of the 2-Methyl-5-(3-hydroxyphenyl)morphans upon Attachment of an (E)-8-benzylidene Moiety: Synthesis and Evaluation of a New Class of Receptor Ligands. J. Med. Chem. 1994, 37, 3163–3170. [Google Scholar] [CrossRef]
- Perregaard, J.; Moltzen, E.K.; Meier, E.; Sánchez, C. Sigma Ligands with Subnanomolar Affinity and Preference for the Sigma 2 Binding Site. 1. 3-(Omega-aminoalkyl)-1H-indoles. J. Med. Chem. 1995, 38, 1998–2008. [Google Scholar] [CrossRef]
- Niso, M.; Abate, C.; Contino, M.; Ferorelli, S.; Azzariti, A.; Perrone, R.; Colabufo, N.A.; Berardi, F. Sigma-2 Receptor Agonists as Possible Antitumor Agents in Resistant Tumors: Hints for Collateral Sensitivity. ChemMedChem 2013, 8, 2026–2035. [Google Scholar] [CrossRef]
- Parry, M.J.; Alakoskela, J.M.I.; Khandelia, H.; Kumar, S.A.; Jäättelä, M.; Mahalka, A.K.; Kinnunen, P.K.J. High-Affinity Small Molecule-Phospholipid Complex Formation: Binding of Siramesine to Phosphatidic Acid. J. Am. Chem. Soc. 2008, 130, 12953–12960. [Google Scholar] [CrossRef]
- Ostenfeld, M.S.; Fehrenbacher, N.; Hoyer-Hansen, M.; Thomsen, C.; Farkas, T.; Jäättelä, M. Effective Tumor Cell Death by σ-2 Receptor Ligand Siramesine Involves Lysosomal Leakage and Oxidative Stress. Cancer Res. 2005, 65, 8975–8983. [Google Scholar] [CrossRef] [PubMed]
- Ostenfeld, M.S.; Høyer-Hansen, M.; Bastholm, L.; Fehrenbacher, N.; Olsen, O.D.; Groth-Pedersen, L.; Puustinen, P.; Kirkegaard-Sørensen, T.; Nylandsted, J.; Farkas, T.; et al. Anti-Cancer Agent Siramesine Is a Lysosomotropic Detergent That Induces Cytoprotective Autophagosome Accumulation. Autophagy 2008, 4, 487–499. [Google Scholar] [CrossRef] [PubMed]
- Hafner, Ĉ.M.; Repnik, U.; Turk, V.; Turk, B. Siramesine Triggers Cell Death through Destabilization of Mitochondria, but Not Lysosomes. Cell Death Dis. 2013, 4, 1–13. [Google Scholar] [CrossRef]
- Mach, R.H.; Yang, B.; Wu, L.; Kuhner, R.J.; West, T. Synthesis and Sigma Receptor Binding Affinities of 8-azabicyclo[3.2.1]octan-3α-yl and 9-azabicyclo[3.2.1]nonan-3α-yl phenylcarbamates. Med. Chem. Res. 2001, 10, 339–355. [Google Scholar]
- Mach, R.H.; Vangvervong, S.; Huang, Y.; Yang, B.; Blair, J.B.; Wu, L. Synthesis of N-Substituted 9-azabicyclo[3.2.1]nonan-3α-yl phenylcarbamates Analogs as Sigma-2 Receptor Ligands. Med. Chem. Res. 2003, 11, 380–398. [Google Scholar]
- Chu, W.; Xu, J.; Zhou, D.; Zhang, F.; Jones, L.A.; Wheeler, K.T.; Mach, R.H. New N-Substituted 9-azabicyclo[3.3.1]nonan-3α-yl phenylcarbamate Analogs as σ2 Receptor Ligands: Synthesis, in Vitro Characterization, and Evaluation as PET Imaging and Chemosensitization Agents. Bioorganic Med. Chem. 2009, 17, 1222–1231. [Google Scholar] [CrossRef] [PubMed]
- Vangveravong, S.; Xu, J.; Zeng, C.; Mach, R.H. Synthesis of N-Substituted 9-azabicyclo[3.3.1]nonan-3α-yl carbamate Analogs as σ2 Receptor Ligands. Bioorganic Med. Chem. 2006, 14, 6988–6997. [Google Scholar] [CrossRef]
- Mach, R.H.; Huang, Y.; Freeman, R.A.; Wu, L.; Vangveravong, S.; Luedtke, R.R. Conformationally-Flexible Benzamide Analogues as Dopamine D3 and σ2 Receptor Ligands. Bioorganic Med. Chem. Lett. 2004, 14, 195–202. [Google Scholar] [CrossRef]
- Tu, Z.; Xu, J.; Jones, L.A.; Li, S.; Dumstorff, C.; Vangveravong, S.; Chen, D.L.; Wheeler, K.T.; Welch, M.J.; Mach, R.H. Fluorine-18-Labeled Benzamide Analogues for Imaging the σ2 Receptor Status of Solid Tumors with Positron Emission Tomography. J. Med. Chem. 2007, 50, 3194–3204. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tu, Z.; Jones, L.A.; Vangveravong, S.; Wheeler, K.T.; Mach, R.H. [3H]N-[4-(3,4-dihydro-6,7-dimethoxyisoquinolin-2(1H)-yl)butyl]-2-methoxy-5-methylbenzamide: A Novel Sigma-2 Receptor Probe. Eur. J. Pharmacol. 2005, 525, 8–17. [Google Scholar] [CrossRef]
- [18F]ISO-1 Positron Emission Tomography (PET/CT) in Primary Breast Cancer; ClinicalTrials.Gov Identifier: NCT02284919; ClinicalTrials.gov: Bethesda, MA, USA, 2014.
- Assessment of Cellular Proliferation in Tumors by Positron Emission Tomography (PET) Using [18F]ISO-1; ClinicalTrials.Gov Identifier: NCT00968656; ClinicalTrials.gov: Bethesda, MA, USA, 2009.
- Imaging of in Vivo Sigma-2 Receptor Expression With 18F-ISO-1 Positron Emission Tomography in Metastatic Breast Cancer; ClinicalTrials.Gov Identifier: NCT03057743; ClinicalTrials.gov: Bethesda, MA, USA, 2021.
- PET Assessment of Acute Lung Transplant Rejection; ClinicalTrials.Gov Identifier: NCT02204202; ClinicalTrials.gov: Bethesda, MA, USA, 2014.
- Abate, C.; Ferorelli, S.; Contino, M.; Marottoli, R.; Colabufo, N.A.; Perrone, R.; Berardi, F. Arylamides Hybrids of Two High-Affinity σ 2 Receptor Ligands as Tools for the Development of PET Radiotracers. Eur. J. Med. Chem. 2011, 46, 4733–4741. [Google Scholar] [CrossRef] [PubMed]
- Abate, C.; Selivanova, S.V.; Müller, A.; Krämer, S.D.; Schibli, R.; Marottoli, R.; Perrone, R.; Berardi, F.; Niso, M.; Ametamey, S.M. Development of 3,4-dihydroisoquinolin-1(2H)-one Derivatives for the Positron Emission Tomography (PET) Imaging of σ2 Receptors. Eur. J. Med. Chem. 2013, 69, 920–930. [Google Scholar] [CrossRef] [PubMed]
- Niso, M.; Pati, M.L.; Berardi, F.; Abate, C. Rigid: Versus Flexible Anilines or Anilides Confirm the Bicyclic Ring as the Hydrophobic Portion for Optimal σ2 Receptor Binding and Provide Novel Tools for the Development of Future σ2 Receptor PET Radiotracers. RSC Adv. 2016, 6, 88508–88518. [Google Scholar] [CrossRef]
- Perrone, R.; Berardi, F.; Colabufo, N.A.; Leopoldo, M.; Abate, C.; Tortorella, V. N-aryl or N-alkylpiperazine Derivatives: The Role of N-Substituent on σ1, σ2, 5-HT1A and D2 Receptor Affinity. Med. Chem. Res. 2000, 10, 201–207. [Google Scholar]
- Abate, C.; Niso, M.; Abatematteo, F.S.; Contino, M.; Colabufo, N.A.; Berardi, F. PB28, the Sigma-1 and Sigma-2 Receptors Modulator With Potent Anti–SARS-CoV-2 Activity: A Review About Its Pharmacological Properties and Structure Affinity Relationships. Front. Pharmacol. 2020, 11, 1–11. [Google Scholar] [CrossRef]
- Kassiou, M.; Dannals, R.F.; Liu, X.; Wong, D.F.; Ravert, H.T.; Scheffel, U.A. Synthesis and in Vivo Evaluation of a New PET Radioligand for Studying Sigma-2 Receptors. Bioorganic Med. Chem. 2005, 13, 3623–3626. [Google Scholar] [CrossRef]
- Abate, C.; Niso, M.; Lacivita, E.; Mosier, P.D.; Toscano, A.; Perrone, R. Analogues of σ Receptor Ligand 1-cyclohexyl-4-[3-(5-methoxy-1,2,3,4-tetrahydronaphthalen-1-yl)propyl]piperazine (PB28) with Added Polar Functionality and Reduced Lipophilicity for Potential Use as Positron Emission Tomography Radiotracers. J. Med. Chem. 2011, 54, 1022–1032. [Google Scholar] [CrossRef] [PubMed]
- Gordon, D.E.; Jang, G.M.; Bouhaddou, M.; Xu, J.; Obernier, K.; White, K.M.; O’Meara, M.J.; Rezelj, V.V.; Guo, J.Z.; Swaney, D.L.; et al. A SARS-CoV-2 Protein Interaction Map Reveals Targets for Drug Repurposing. Nature 2020, 583, 459–468. [Google Scholar] [CrossRef]
- Tummino, T.A.; Rezelj, V.V.; Fischer, B.; Fischer, A.; Meara, M.J.; Monel, B.; Vallet, T.; Zhang, Z.; Alon, A.; Donnell, H.R.; et al. Phospholipidosis Is a Shared Mechanism Underlying the in Vitro Antiviral Activity of Many Repurposed Drugs against SARS-CoV-2. bioRxiv 2021. [Google Scholar] [CrossRef]
- Izzo, N.J.; Colom-Cadena, M.; Riad, A.A.; Xu, J.; Singh, M.; Abate, C.; Cahill, M.A.; Spires-Jones, T.L.; Bowen, W.D.; Mach, R.H.; et al. Proceedings from the Fourth International Symposium on σ-2 Receptors: Role in Health and Disease. eNeuro 2020, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.Gov. Available online: https://clinicaltrials.gov/ct2/results?cond=&term=CT1812&cntry=&state=&city=&dist= (accessed on 28 April 2021).
- Hornick, J.R.; Vangveravong, S.; Spitzer, D.; Abate, C.; Berardi, F.; Goedegebuure, P.; MacH, R.H.; Hawkins, W.G. Lysosomal Membrane Permeabilization Is an Early Event in Sigma-2 Receptor Ligand Mediated Cell Death in Pancreatic Cancer. J. Exp. Clin. Cancer Res. 2012, 31, 1–11. [Google Scholar] [CrossRef]
- Colgan, S.M.; Al-Hashimi, A.A.; Austin, R.C. Endoplasmic Reticulum Stress and Lipid Dysregulation. Expert Rev. Mol. Med. 2011, 13, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Mach, R.H.; Zeng, C.; Hawkins, W.G. The σ2 Receptor: A Novel Protein for the Imaging and Treatment of Cancer. J. Med. Chem. 2013, 56, 7137–7160. [Google Scholar] [CrossRef]
- Cantonero, C.; Camello, P.J.; Abate, C.; Berardi, F.; Salido, G.M.; Rosado, J.A.; Redondo, P.C. NO1, a New Sigma 2 Receptor/TMEM97 Fluorescent Ligand, Downregulates SOCE and Promotes Apoptosis in the Triple Negative Breast Cancer Cell Lines. Cancers 2020, 12, 257. [Google Scholar] [CrossRef]
- Pati, M.L.; Hornick, J.R.; Niso, M.; Berardi, F.; Spitzer, D.; Abate, C.; Hawkins, W. Sigma-2 Receptor Agonist Derivatives of 1-cyclohexyl-4-[3-(5-methoxy-1,2,3,4-tetrahydronaphthalen-1-yl)propyl]piperazine (PB28) Induce Cell Death via Mitochondrial Superoxide Production and Caspase Activation in Pancreatic Cancer. BMC Cancer 2017, 17, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Crawford, K.W.; Bowen, W.D. Sigma-2 Receptor Agonists Activate a Novel Apoptotic Pathway and Potentiate Antineoplastic Drugs in Breast Tumor Cell Lines. Cancer Res. 2002, 62, 313–322. [Google Scholar] [PubMed]
- Zeng, C.; Weng, C.C.; Schneider, M.E.; Puentes, L.; Riad, A.; Xu, K.; Makvandi, M.; Jin, L.; Hawkins, W.G.; Mach, R.H. TMEM97 and PGRMC1 Do Not Mediate Sigma-2 Ligand-Induced Cell Death. Cell Death Discov. 2019, 5. [Google Scholar] [CrossRef]
- Spitzer, D.; Simon, P.O.; Kashiwagi, H.; Xu, J.; Zeng, C.; Vangveravong, S.; Zhou, D.; Chang, K.; McDunn, J.E.; Hornick, J.R.; et al. Use of Multifunctional Sigma-2 Receptor Ligand Conjugates to Trigger Cancer-Selective Cell Death Signaling. Cancer Res. 2012, 72, 201–209. [Google Scholar] [CrossRef]
- Ohman, K.A.; Hashim, Y.M.; Vangveravong, S.; Nywening, T.M.; Cullinan, D.R.; Goedegebuure, S.P.; Liu, J.; Van Tine, B.A.; Tiriac, H.; Tuveson, D.A.; et al. Conjugation to the Sigma-2 Ligand SV119 Overcomes Uptake Blockade and Converts Dm-Erastin into a Potent Pancreatic Cancer Therapeutic. Oncotarget 2016, 7, 33529–33541. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Vangveravong, S.; Mcdunn, J.E.; Hawkins, W.G.; Mach, R.H. Sigma-2 Receptor Ligand as a Novel Method for Delivering a SMAC Mimetic Drug for Treating Ovarian Cancer. Br. J. Cancer 2013, 109, 2368–2377. [Google Scholar] [CrossRef]
- Hashim, Y.M.; Spitzer, D.; Vangveravong, S.; Hornick, M.C.; Garg, G.; Hornick, J.R.; Goedegebuure, P.; Mach, R.H.; Hawkins, W.G. Targeted Pancreatic Cancer Therapy with the Small Molecule Drug Conjugate SW IV-134. Mol. Oncol. 2014, 8, 956–967. [Google Scholar] [CrossRef]
- Garg, G.; Vangveravong, S.; Zeng, C.; Collins, L.; Hornick, M.; Hashim, Y.; Piwnica-Worms, D.; Powell, M.A.; Mutch, D.G.; Mach, R.H.; et al. Conjugation to a SMAC Mimetic Potentiates Sigma-2 Ligand Induced Tumor Cell Death in Ovarian Cancer. Mol. Cancer 2014, 13, 1–13. [Google Scholar] [CrossRef]
- Makvandi, M.; Tilahun, E.D.; Lieberman, B.P.; Anderson, R.C.; Zeng, C.; Xu, K.; Hou, C.; McDonald, E.S.; Pryma, D.A.; Mach, R.H. The Sigma-2 Receptor as a Therapeutic Target for Drug Delivery in Triple Negative Breast Cancer. Biochem. Biophys. Res. Commun. 2015, 467, 1070–1075. [Google Scholar] [CrossRef] [PubMed]
- Szybalski, W.; Bryson, V. Genetic Studies on Microbial Cross Resistance To Toxic Agents, I: Cross resistance of Escherichia coli to fifteen antibiotics1, 2. J. Bacteriol. 1952, 64, 489–499. [Google Scholar] [CrossRef]
- Gottesman, M.M.; Fojo, T.; Bates, S.E. Multidrug Resistance in Cancer: Role of ATP-Dependent Transporters. Nat. Rev. Cancer 2002, 2, 48–58. [Google Scholar] [CrossRef]
- Pluchino, K.M.; Hall, M.D.; Goldsborough, A.S.; Callaghan, R.; Gottesman, M.M. Collateral Sensitivity as a Strategy against Cancer Multidrug Resistance. Drug Resist. Updat. 2012, 15, 98–105. [Google Scholar] [CrossRef]
- Abate, C.; Pati, M.L.; Contino, M.; Colabufo, N.A.; Perrone, R.; Niso, M.; Berardi, F. From Mixed Sigma-2 Receptor/P-Glycoprotein Targeting Agents to Selective P-Glycoprotein Modulators: Small Structural Changes Address the Mechanism of Interaction at the Efflux Pump. Eur. J. Med. Chem. 2015, 89, 606–615. [Google Scholar] [CrossRef] [PubMed]
- Pati, M.L.; Abate, C.; Contino, M.; Ferorelli, S.; Luisi, R.; Carroccia, L.; Niso, M.; Berardi, F. Deconstruction of 6,7-Dimethoxy-1,2,3,4-tetrahydroisoquinoline Moiety to Separate P-Glycoprotein (P-gp) Activity from Sigma-2 (σ2) Receptor Affinity in Mixed P-gp/σ2 Receptor Agents. Eur. J. Med. Chem. 2015, 89, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Niso, M.; Riganti, C.; Abate, C. Collateral Sensitivity of Sigma-2 Receptor Ligands: Potentials in the Treatment of Multidrug Resistant Tumors. Recept. Clin. Investig. 2014, 1–8. [Google Scholar] [CrossRef]
- Riganti, C.; Giampietro, R.; Kopecka, J.; Costamagna, C.; Abatematteo, F.S.; Contino, M.; Abate, C. MRP1-Collateral Sensitizers as a Novel Therapeutic Approach in Resistant Cancer Therapy: An in Vitro and in Vivo Study in Lung Resistant Tumor. Int. J. Mol. Sci. 2020, 21, 3333. [Google Scholar] [CrossRef] [PubMed]
- Hall, M.D.; Salam, N.K.; Hellawell, J.L.; Fales, H.M.; Kensler, C.B.; Ludwig, J.A.; Szakács, G.; Hibbs, D.E.; Gottesman, M.M. Synthesis, Activity, and Pharmacophore Development for Isatin-β- Thiosemicarbazones with Selective Activity toward Multidrug-Resistant Cells. J. Med. Chem. 2009, 52, 3191–3204. [Google Scholar] [CrossRef]
- Pati, M.L.; Niso, M.; Ferorelli, S.; Abate, C.; Berardi, F. Novel Metal Chelators Thiosemicarbazones with Activity at the σ2 Receptors and P-Glycoprotein: An Innovative Strategy for Resistant Tumor Treatment. RSC Adv. 2015, 5, 103131–103146. [Google Scholar] [CrossRef]
- Pati, M.L.; Niso, M.; Spitzer, D.; Berardi, F.; Contino, M.; Riganti, C.; Hawkins, W.G.; Abate, C. Multifunctional Thiosemicarbazones and Deconstructed Analogues as a Strategy to Study the Involvement of Metal Chelation, Sigma-2 (σ2) Receptor and P-gp Protein in the Cytotoxic Action: In Vitro and in Vivo Activity in Pancreatic Tumors. Eur. J. Med. Chem. 2018, 144, 359–371. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).