Abstract
Post-modification of a series of NCN-pincer platinum(II) complexes [PtX(NCN-R-4)] (NCN = [C6H2(CH2NMe2)2-2,6]–, R = C(O)H, C(O)Me and C(O)Et), X = Cl– or Br–) at the para-position using the McMurry reaction was studied. The synthetic route towards two new [PtCl(NCN-R-4)] (R = C(O)Me and C(O)Et) complexes used above is likewise described. The utility and limitations of the McMurry reaction involving these pincer complexes was systematically evaluated. The predicted “homo-coupling” reaction of [PtBr(NCN-C(O)H-4)] led to the unexpected formation of 3,3′,5,5′-tetra[(dimethylamino)methyl]-4,4′-bis(platinum halide)-benzophenone (halide = Br or Cl), referred to hereafter as the bispincer-benzophenone complex 13. This material was further characterized using X-ray crystal structure determination. The applicability of the pincer complexes in the McMurry reaction is shown to open a route towards the synthesis of tamoxifen-type derivatives of which one phenyl ring of Tamoxifen® itself is replaced by an NCN arylplatinum pincer fragment. The newly synthesized derivatives can be used as potential candidates in anti-cancer drug screening protocols. Two NCN-arylpincer platinum tamoxifen type derivatives, 5 and 6, were successfully synthesized and of 5 the separation of the diastereomeric E-/Z-forms was achieved. Compound 6, which is the pivaloyl protected NCN pincer platinum hydroxy-Tamoxifen® derivative, was obtained as a mixture of E-/Z-isomers. The new derivatives were further analyzed and characterized with 1H-, 13C{1H}- and 195Pt{1H}-NMR, IR, exact mass MS and elemental analysis.
1. Introduction
The increasing interest in the application of organometallic complexes in functional materials, for example, in biocatalysis with artificial metalloenzymes [1], in natural enzymes [2], in organic light emitting diodes (OLEDs) [3], in polymers [4], and organometallic pharmaceuticals [5,6,7], has stimulated the development of metal complexes with an increased stability towards air, moisture and high thermal stress. Due to their increased stability, these complexes can be modified or used as a reactant in a chemical reaction without decomposing. The ferrocenes are a good example of organometallic complexes used in compounds with anticancer activity [8]. Another class of stable organometallic compounds that is pertinent to many of the research projects in the career of Dr. Michel Pfeffer, are those that emerged from his pioneering research in the field of cyclometallated organometallic compounds [9]. Starting with the synthesis of the first cyclopalladated [9,10] and ruthenated [11,12] complexes, Pfeffer later extended his interests beyond the synthesis aspects while entering into multidisciplinary collaborations studying the biochemical aspects of his ruthenated compounds [13,14,15,16]. In his cyclometallation research, he focused merely on the use of nitrogen donor atom containing directing groups, e.g., amines, imines, or 2-pyridyl groupings, leading to C,N-cyclometallated aryl, alkenyl and alkyl organometallics. In our longstanding collaborations, we used directed ortho-lithiation [17,18] followed by transmetallation as a useful, alternate approach when direct ortho-metallation through C-H activation using directing nitrogen donor groupings as a synthetic route appeared not to be feasible [19].
The latter approach was extensively explored for the synthesis of bis(ortho-chelated) NCN-aryl-pincer platinum complexes, [PtX(NCN-R-4)] (NCN = [4-RC6H2(CH2NMe2)2-2,6]–; (X = Cl, Br, I) [20], by the Amsterdam and Utrecht research groups. Based on their extraordinary, broad stability towards air, moisture and higher temperatures, these complexes were studied for their properties as, for instance, color biomarkers, a detection label, and as a luminescent material [21]. We also showed the feasibility of post-modification of the NCN backbone in the [MX(NCN-R-4)] pincer system (M = Ni, Pd or Pt; X = Cl, Br or I) with a large library of different para-substituents R (R = –NO2, –C(O)Me, –COOH, –CHO, –SO3H, –PO(OEt)2, –PO(OH)(OEt), –PO(OH)2, –I, –Cl, –H, –CH2OH, –OMe, –SMe or –NH2) [22]. In some cases, these substituents could even be directly used as a reactant in subsequent synthetic reaction protocols [23,24,25].
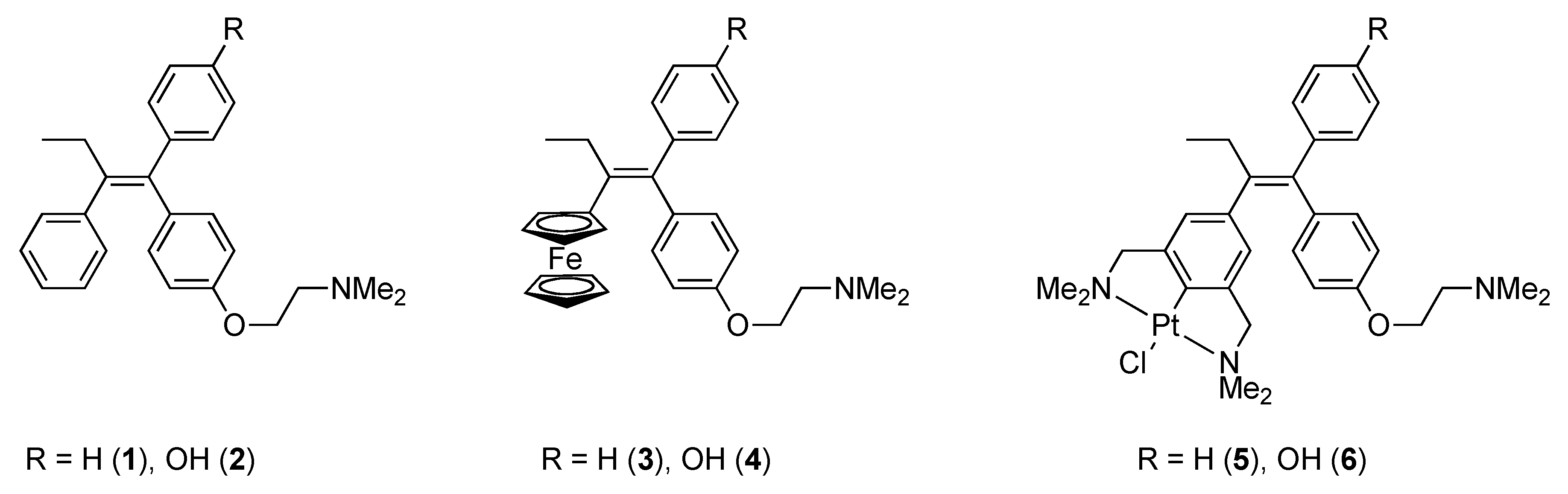
To further explore the scope of the para post-modification of NCN-pincer metal complexes, we were prompted by the extensive work of Jaouen and coworkers on the development of both ferrocifen (3) and hydroxyferrocifen (4) [8,26]. In these organometallic derivatives of the anti-cancer drug Tamoxifen® (1) [26,27], a phenyl ring of Tamoxifen® is replaced by a ferrocene moiety (Chart 1) [26]. Introduction of this metallocene fragment resulted in an enhanced anti-proliferation effect in cancer cells.
Chart 1.
Tamoxifen® (1 and 2), ferrocifens (3 and 4) and its NCN-pincer Pt(II) analogues, named pincercifen (5 and 6).
So, on the one hand prompted by the question to explore the scope of the para post-modification of NCN pincer-metal complexes, and on the other to explore the effect of the replacement of the ferrocenyl group by an NCN pincer-metal halide one, we set out to develop a synthetic methodology in order to synthesize NCN-pincer platinum halide containing tamoxifen-type derivatives. In this manuscript, we report our first results and findings on the synthesis of these molecules.
2. Results
2.1. Synthesis
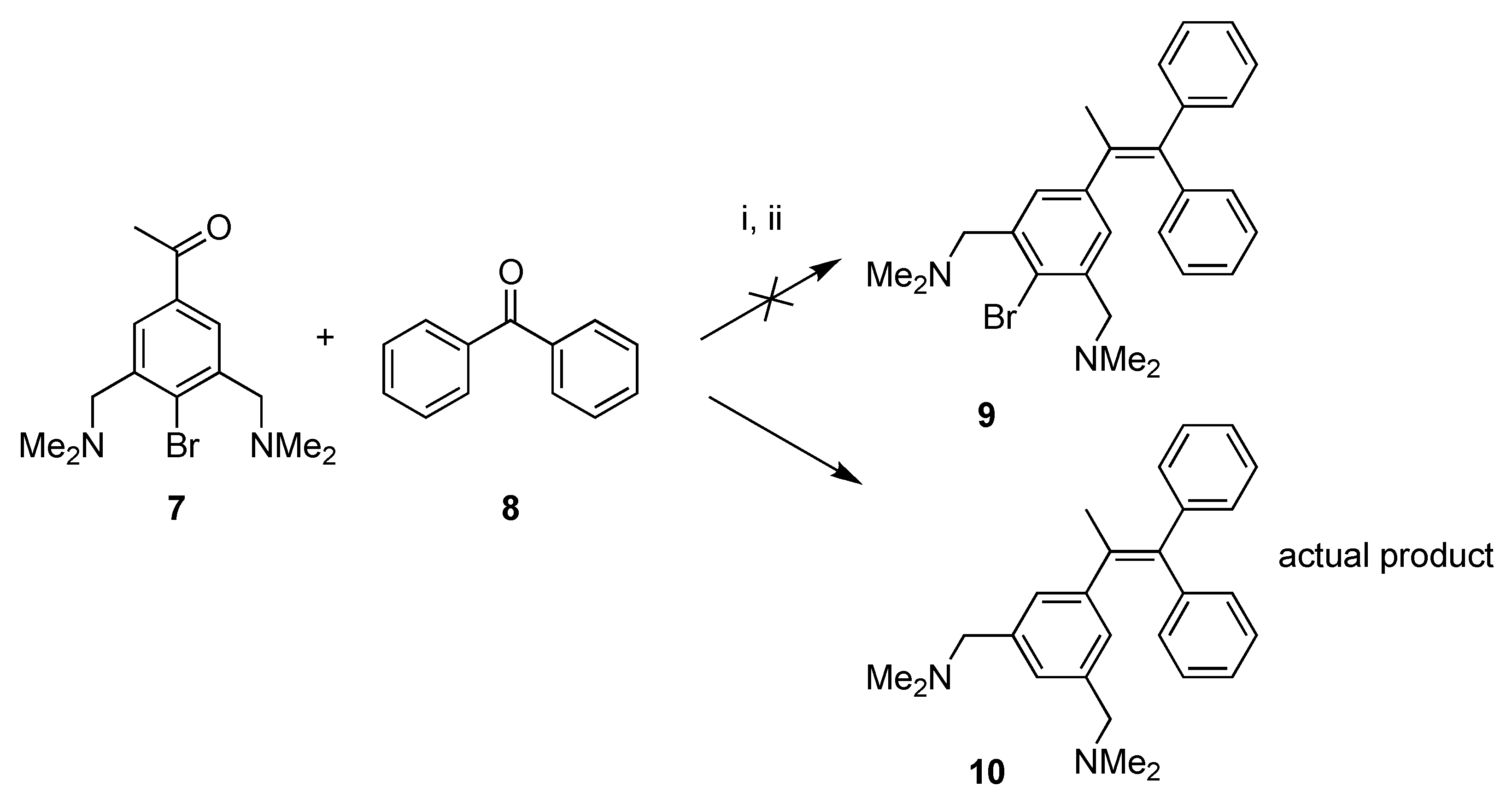
In general, the Tamoxifen® and ferrocifen derivatives are synthesized using the McMurry coupling reaction of ketones [26,28], using low valent titanium as a coupling intermediate. In this study, we will also use this methodology in order to synthesize the NCN-pincer platinum derivatives 5 and 6. To test the applicability of pincer ligands in the McMurry reaction, the acetophenone pincer ligand 7 was synthesized by a known procedure [22]. We thus attempted to couple 7 with benzophenone 8, under typical McMurry conditions. Surprisingly, the product mixture obtained after purification, did not contain desired product 9, but rather consisted mainly of the corresponding alkene derivative 10 (concluded via 1H-NMR spectroscopy). In this dehalogenated product, the bromide had been cleaved from the ligand backbone and was exchanged for a hydrogen atom. Apparently, under the applied McMurry reaction conditions (refluxing in THF for 16 h in the presence of Zn and Ti), the phenyl bromide 7 or 9 is effectively hydrolyzed (Scheme 1). From the literature, it is known that the McMurry reaction should be compatible with organohalides [28]. For example, it has been shown that 4-bromoacetophenone can be used in the McMurry reaction, forming the homocoupled product, without affecting the phenyl bromide bond [29]. A possible explanation for the dehalogenation of 7 or 9 can be the intermediate formation of an NCN-Ti(IV) complex [30]. These complexes are known to be highly sensitive to air and moisture, and readily decompose at higher temperatures thereby forming the resulting protonated NCN ligand. It was therefore decided to use metallated building blocks directly as a reagent in the McMurry reaction in order to avoid this problem.
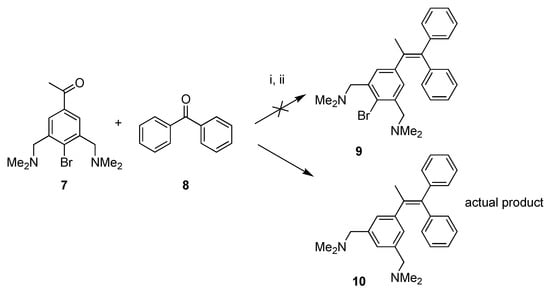
Scheme 1.
McMurry reaction of 7 and 8 yielding de-halogenated product 10. Reagents and conditions: (i) TiCl4(THF)2, Zn(0), THF, reflux, 16 h; (ii) H2O, K2CO3.
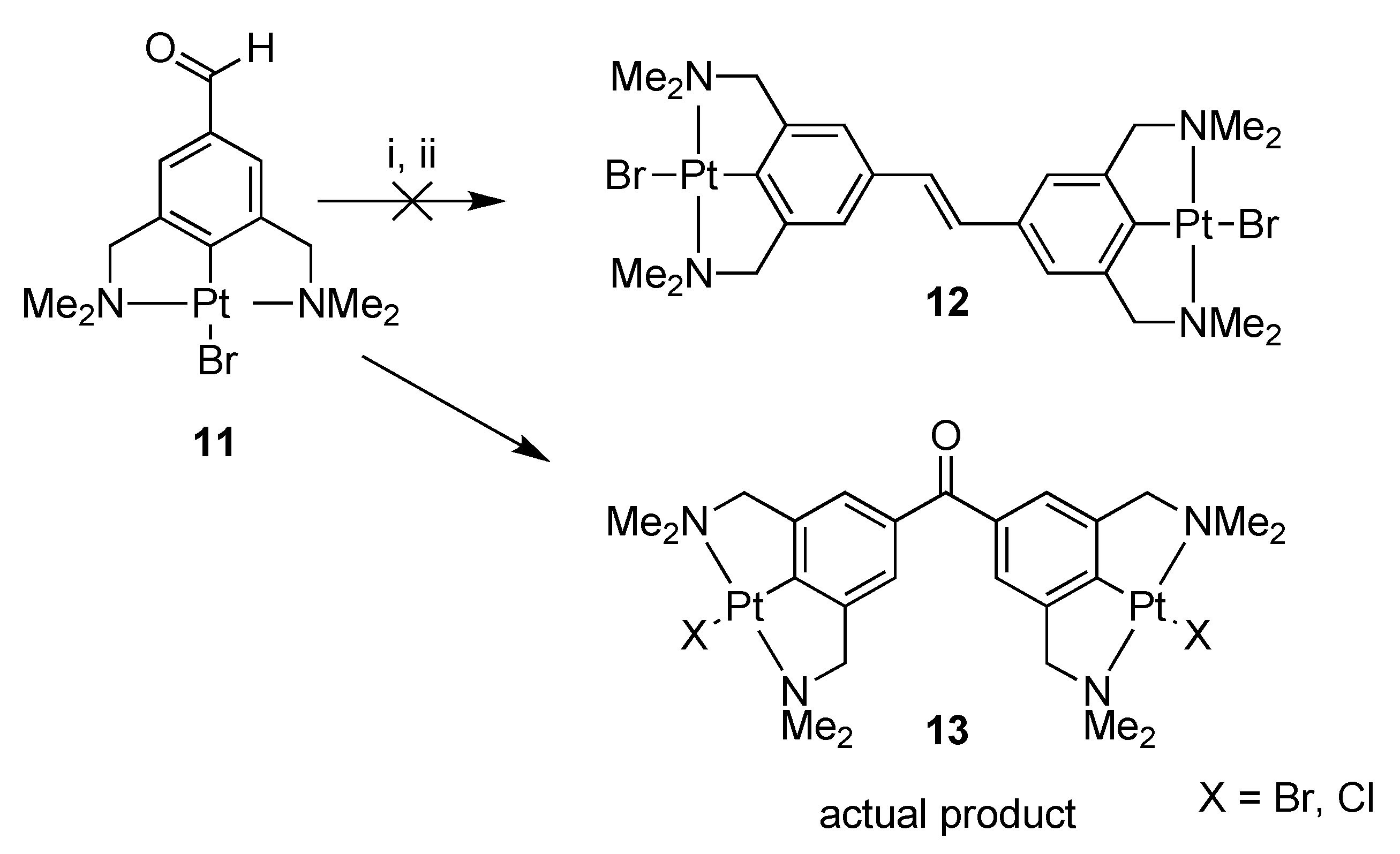
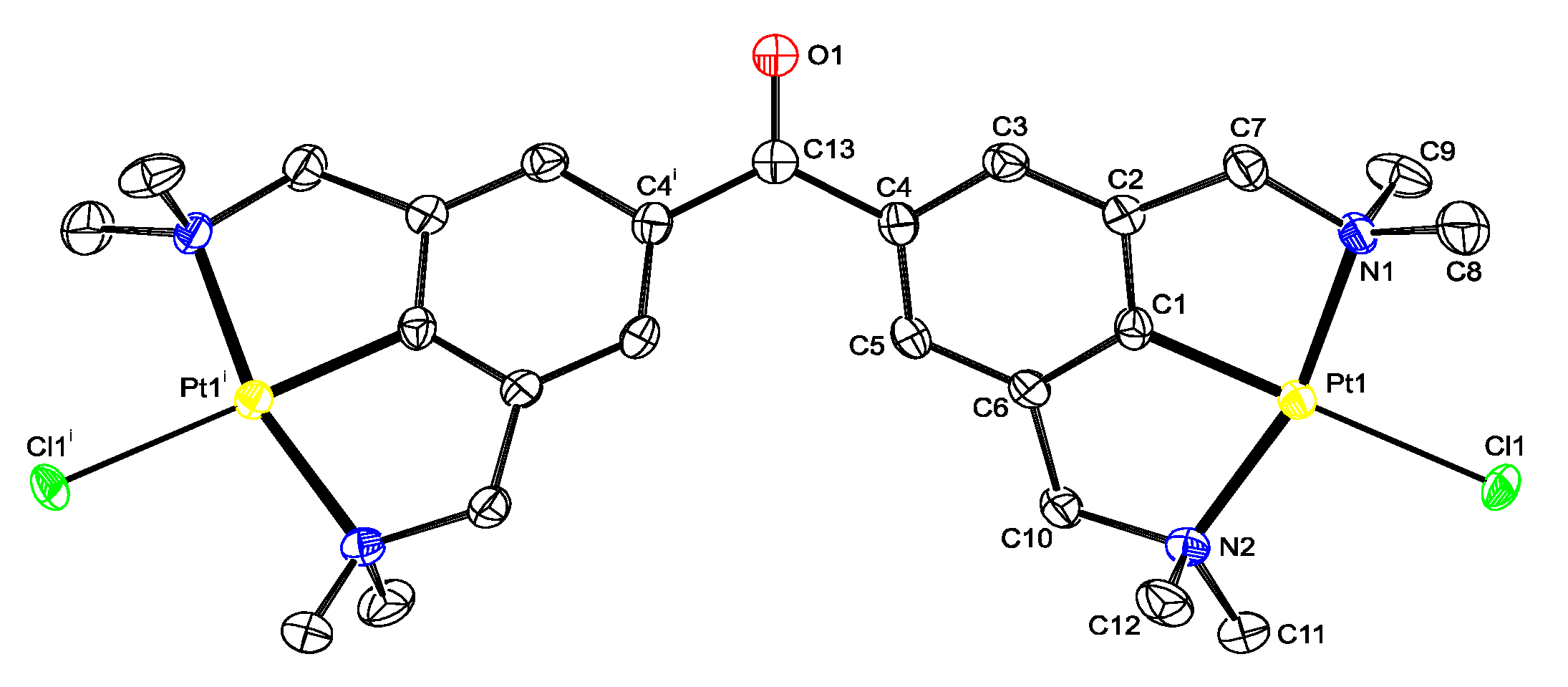
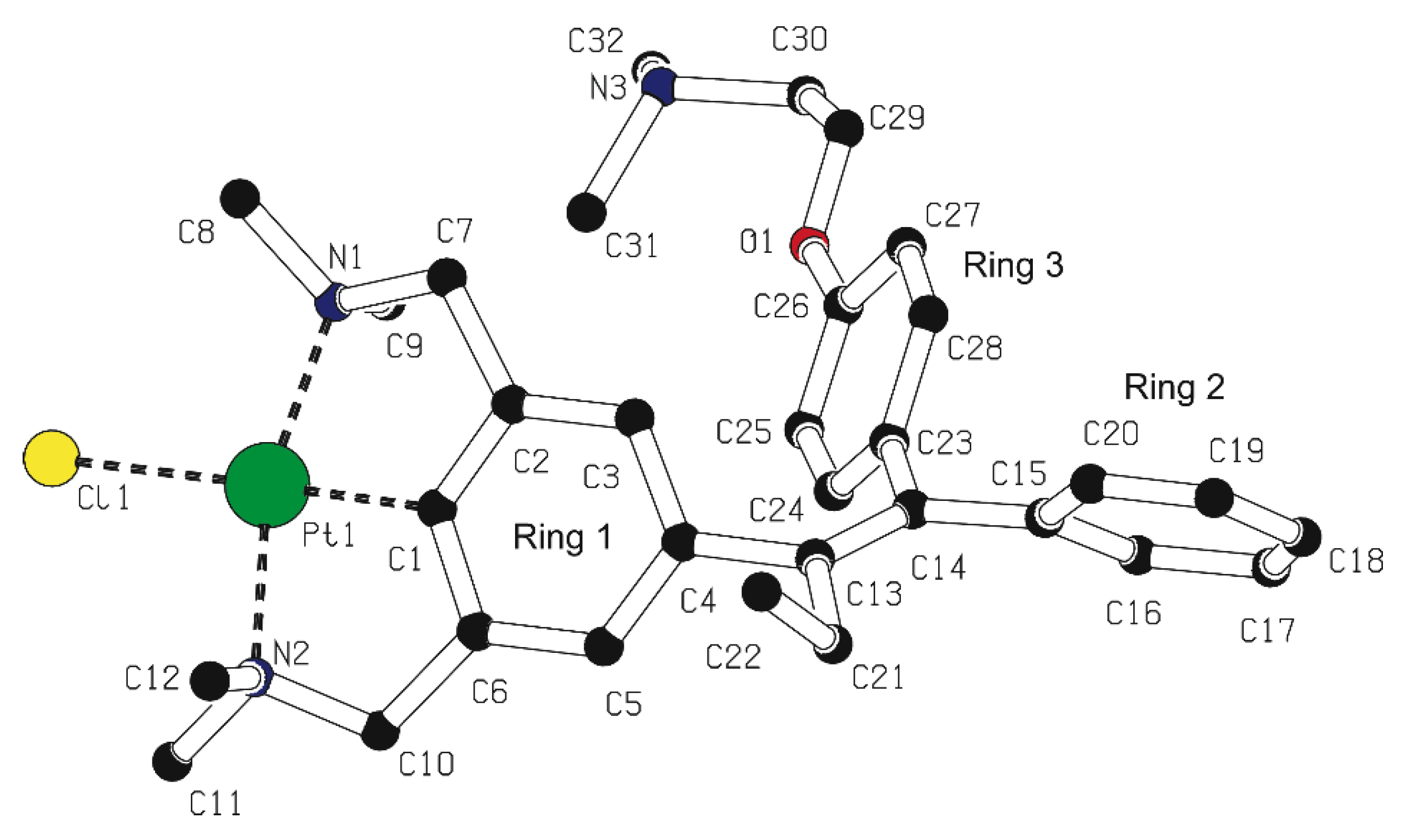
Platinum pincer aldehyde [PtBr(NCN-CHO-4)] 11, a complex developed in a previous study [22,24], was used in a homocoupling reaction under McMurry conditions, in order to synthesize the bisplatinum-stilbene compound 12 (Scheme 2). After workup, the isolated product consisted of a mixture of compounds, also containing the starting aldehyde 11. In attempts to separate the product mixture into separate components, a yellow crystalline solid was obtained after crystallization of the mixture from toluene solution. The structure in the solid state was determined with X-ray crystallography, and, surprisingly, confirmed the crystals to be of the bimetallic benzophenone platinum pincer 13; i.e., rather than 12 the coupling product 13 was obtained in which the two platinum pincer fragments are connected via a carbonyl linker. No signals of the stilbenoid product 12 were found in the residue. During the coupling reaction on the titanium surface, an as yet unclear reaction has occurred. From the crystal structure data (see Figure 1), it was also observed that there is substitutional disorder between bromine and chlorine on the halogen sites, the chlorido ligand likely having been delivered by TiCl4.
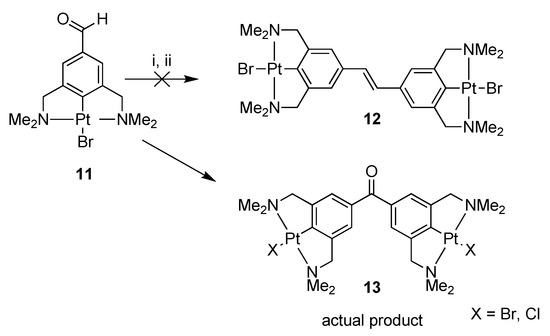
Scheme 2.
Reagents and conditions: (i) TiCl4(THF)2, Zn(0), THF, reflux, 16 h; (ii) H2O, K2CO3.
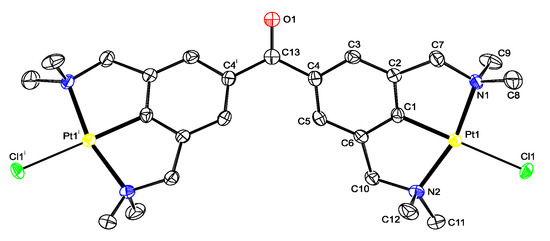
Figure 1.
Displacement ellipsoid plot of 13, drawn at the 50% probability level. Hydrogen atoms and disordered solvent molecules are omitted for clarity. The halogen position is occupied by 60% chlorine and 40% bromine; only chlorine derivative is shown. Symmetry operation i: −x, y, 0.5-z.
Similar halogen scrambling was observed in both the 1H- and 195Pt{1H}-NMR spectra, by the presence of two signals for the dimethylamino protons of the CH2NMe2 groups, and likewise, in the 195Pt-NMR spectrum two signals were found at −3107 and −3130 ppm resulting from the two different platinum halide species. From the mass spectrum, it became clear that for 13 only the X = Br, Cl, and X = Cl species were present, whereas no mass peak for the X = Br only was found (see Figure S1). To avoid halogen scrambling, it was decided to use only PtCl complexes in the rest of our study.
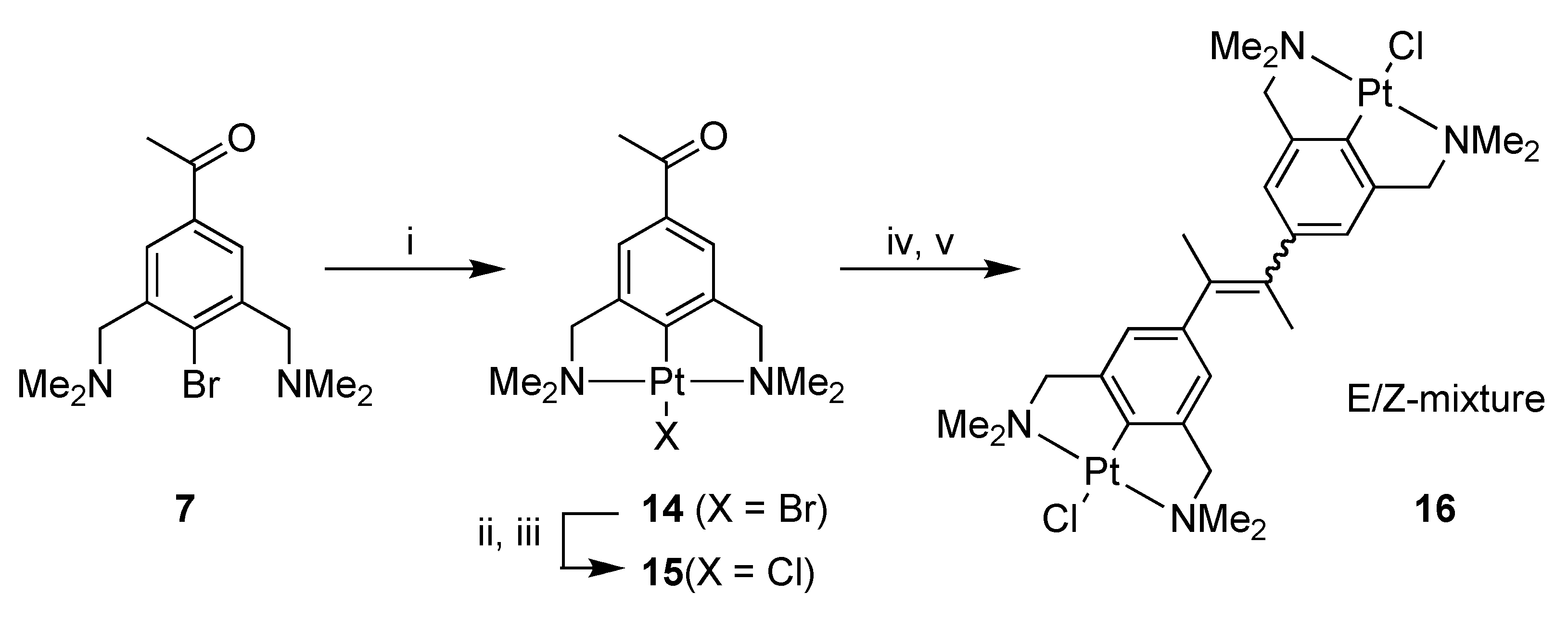
Platinum pincer acetophenone 14 was prepared by platination of 4-bromo-3,5-bis[(dimethylamino)methyl]acetophenone (7) [22,24] with Canty’s reagent, [Pt(4-Tol)2(SEt2)]2 [31]. The PtBr complex 14 was subsequently converted into the PtCl complex 15 by the use of silver salts (AgOTf), and subsequent addition of NaCl (Scheme 3).
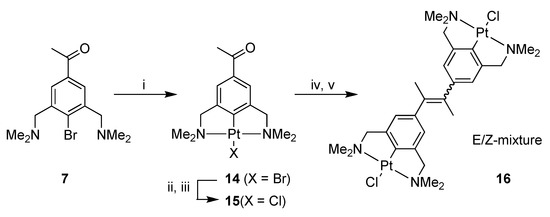
Scheme 3.
Synthesis of 14, 15 and 16. Reagents and Conditions: (i) [Pt(4-Tol)2(SEt2)]2, benzene, reflux, 3 h, 92%; (ii) AgOTf, acetone, rt, 30 min; (iii) NaCl, rt, 1 h, 82%; (iv) TiCl4(THF)2, Zn(0), THF, reflux, 16 h; (v) H2O, K2CO3.
Platinum pincer acetophenone 15 was used in the McMurry reaction, in order to achieve the homocoupled platinum alkene 16 (Scheme 3). The reaction resulted in the isolation of a product mixture consisting of an equimolar amount of the corresponding E- and Z-isomers of 16 combined with unreacted 15. The formation of 16 showed that it was possible to use the platinum complex as a reactant in a McMurry reaction. Attempts to remove 15 from the product mixture by crystallization were, unfortunately, not successful.
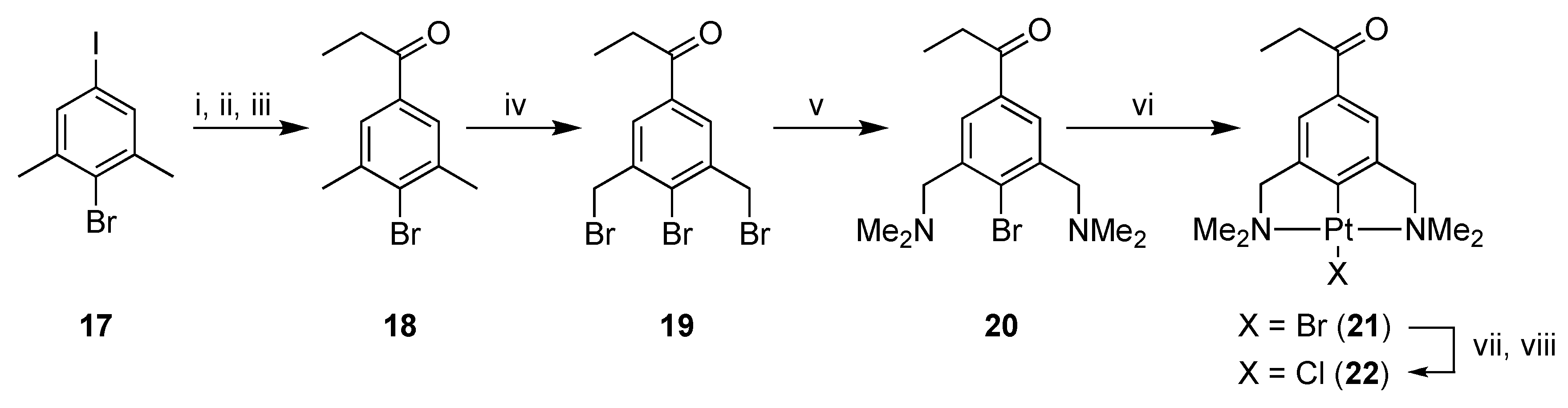
Proceeding on our quest to synthesize 5 and 6, platinum-containing ketone 22 was synthesized starting from 4-iodo-1-bromo-2,6-dimethylbenzene (17) [22,24] via a multistep reaction procedure (Scheme 4). In the first step, the iodo group of 17 was selectively substituted with lithium by a metallation reaction with n-BuLi at low temperature. The lithiated species was reacted in situ with N,N-dimethylpropionamide and after hydrolysis 4-bromo-3,5-dimethylphenyl ethyl ketone 18 was obtained, which was converted to bis-benzyl bromide 19 via a radical bromination reaction [32].
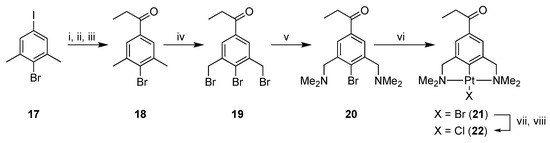
Scheme 4.
Reagents and Conditions: (i) n-BuLi, THF, −78 °C; (ii) N,N-dimethylpropionamide, −78 °C → rt; (iii) H2O, 43%; (iv) NBS, AIBN, MeOAc, 12 h, hν, 49%; (v) HNMe2, Et2O, 12 h, rt, 94%; (vi) [Pt(4-Tol)2(SEt2)]2, toluene, 70 °C, 3 h, 96%; (vii) AgBF4, acetone, rt; (viii) NaCl, 96%.
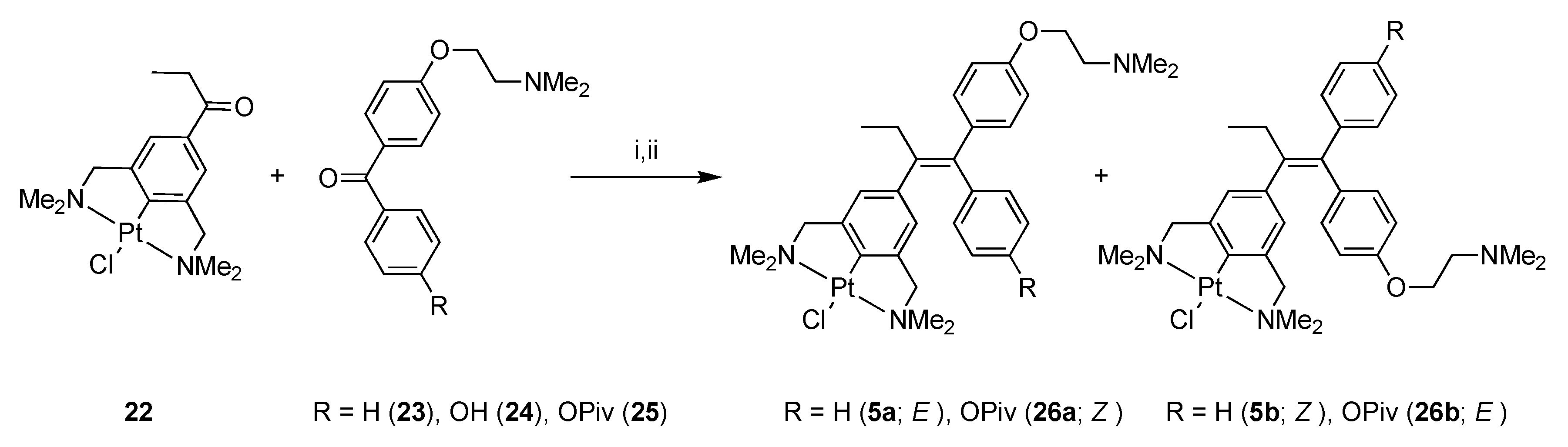
Compound 19 was then converted into 2,6-bis(dimethylamino)benzyl bromide 20 by an amination reaction using excess dimethylamine. Propiophenone pincer ligand 20 was selectively platinated with Canty’s reagent, [Pt(4-Tol)2(SEt2)]2, affording platinum pincer complex 21 in a good overall yield (19% starting from 17). Complex 21 was then converted to PtCl complex 22 via a halogen exchange reaction using AgBF4 and NaCl, subsequently, as described earlier. The organic benzophenone fragment 23 was synthesized according to a literature procedure [33] (Scheme 5).
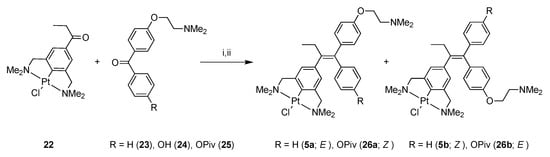
Scheme 5.
Synthesis of E/Z complexes 5 and 26 via McMurry coupling. Reagents and Conditions: (i) TiCl4(THF)2, Zn(0), THF, reflux, 16h; (ii) H2O, K2CO3 (for 5 36%).
Finally, the ketones 22 and 23 were coupled, using the McMurry protocol, giving a mixture of products containing the E/Z mixture 5a and 5b, respectively, and the homo-coupled products derived from 23. The inorganic compounds were partly separated from the organics by precipitation from CH2Cl2, with pentane, and the inorganics were isolated after centrifugation; this resulted in the isolation of an E/Z mixture 5a and 5b (3:7). However, this material still contained some impurities. Fortunately, the isomers could be separated by silica gel column chromatography, although mainly yielding fractions containing different ratios of both isomers but also some fractions containing the separated isomers in pure form. The isomers 5a and 5b were characterized with 1H-, 13C{1H}-, and 195Pt{1H}-NMR, IR, exact mass MS and elemental analysis. For the Z-isomer 5b, an X-ray crystal structure [34] was obtained (see Figure 2 below).
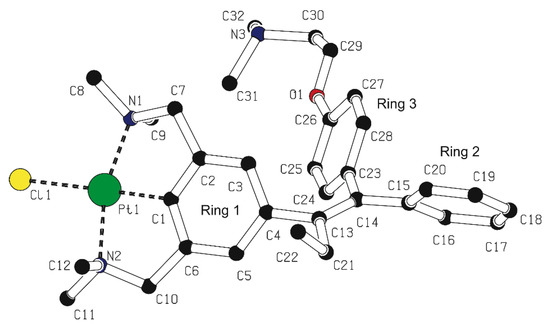
Figure 2.
PLUTON visualization of 5b in the crystal [34]. Hydrogen atoms are omitted for clarity.
With the successful synthesis of 5 in mind, we tried to synthesize 6, which is an analogue of both 2 and 4, i.e., the hydroxy analogues of Tamoxifen® and ferrocifen. These compounds are also active metabolites when tested for anti-cancer activity.
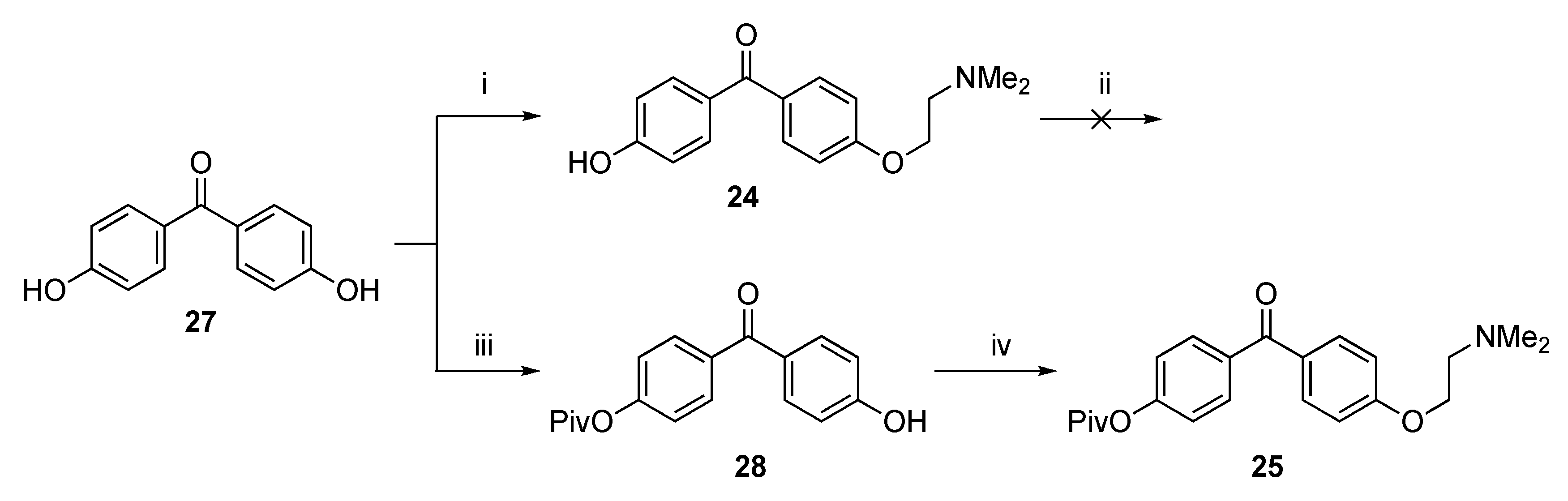
In order to be able to prepare 6, we first functionalized 4,4′-dihydroxybenzophenone 27 with a single 2-(dimethylamino)ethoxy group, obtaining 24 according to a modified literature procedure [35]. Repeated attempts to couple 22 and 24 in order to obtain 6′ i.e., the precursor of 6 (as E-/Z-mixture) failed, and only the starting metal complex 22 was recovered from the reaction mixtures. Presumably, the free phenol group in 24 hampers the coupling reaction. Therefore, it was decided to protect the phenol group of 24 with a tert-butyldimethylsilyl (TBDMS) group (Scheme 6). When 24 was reacted with TBDMS-Cl using imidazole as a base, no reaction took place to form the desired product. The use of NaH was likewise not successful. It was decided to prepare the single protected pivaloyl (OPiv) hydroxybenzophenone 28 instead using a literature procedure [35]. Next, 28 was functionalized with a dimethylaminoethoxy group offering 25.
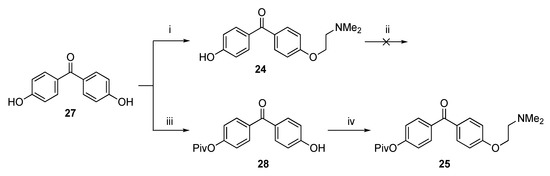
Scheme 6.
Functionalization of 4,4′-hydroxybenzophenone 27. Reagents and Conditions: (i) ClCH2CH2NMe2·HCl, K2CO3, acetone, reflux, 20 h, 21%.; (ii) TBDMS-Cl, imidazole or NaH, THF; (iii) NaH, PivOCl, THF; (iv) ClCH2CH2NMe2·HCl, K2CO3, acetone, reflux, 20 h, 20%.
The doubly functionalized benzophenone 25 was then successfully coupled with 22 via the McMurry reaction, yielding a product mixture after workup. Purification by silica gel chromatography gave 26 as a mixture of E- and Z-isomers (62% yield). In a second run, attempts were made to separate the E/Z isomers using silica gel chromatography, yielding a fraction in which one of the isomers of 26 was present in a 49:1 ratio, see Experimental section. Regrettably, synthetic attempts to de-protect 26 in order to obtain 6, were unsuccessful up to this time. Synthetic methodologies are currently under development to address this issue.
2.2. Structural Features of 13, and 5b in the Solid State
Suitable crystals for single crystal X-ray structure determination for 13 were obtained by slow evaporation of a toluene solution containing the product mixture from the reaction depicted in Scheme 2 (Figure 1). In the crystals the molecule is positioned on an exact, crystallographic two-fold axis. For 13, the overall geometric conformation is similar to that found for other benzophenone solid state structures [28]. An interplanar angle of 23.47(13)° is observed between the plane of the phenyl ring and the plane containing the C=O fragment and the C4 and C4i atoms. The non-planarity of the molecule results from the minimization of the intramolecular interactions between the two rings. The length of the C=O bond of 1.237(5) Å is in the expected bond length order of 1.22 Å.
Crystals of 5b were obtained by slow evaporation of a toluene/ethanol/dichloromethane mixture containing the compound. For 5b, the overall conformation of the structure is similar to that of other tamoxifen-based structures in the solid state (Figure 2) [26,36]. The triphenylethylene system containing the platinum pincer fragment (ring 1), the unsubstituted phenyl (ring 2) and the 4-[2-(dimethylamino)ethoxy]phenyl (ring 3), adopts a propeller conformation.
In 5b, angles of 74.9(7), 48.6(7), and 88.4(6)° are found between the planes of ring 1 and ring 2, ring 1 and ring 3, ring 2 and ring 3, respectively. A dihedral angle of 52.5(7)° is found between ring 1 and the plane formed by C4, C13, C14, C21. Angles of 48.8(9) and 56.9(9)° are found for ring 2 and ring 3, respectively, with the plane formed by C13, C14, C15, and C23. The phenyl rings are not coplanar with the ethylene bond C13-C14, and an angle of 6.4(9)° is found between the planes formed by C4, C13, C14, C21, and C13, C14, C15, C23, which both contain the ethylene fragment.
Angles for C4-C13-C21 and C15-C14-C23 of 113.7(12) and 117.0(11)°, respectively, are found, which are shorter than the angle of 120° expected to be found for formal sp2 hybridization, but which is commonly found in tamoxifen analogs [20,37].
Comparison of the molecular structures of 5b and 13 with earlier reported [PtX(NCN)] (X = Br or Cl) pincer complexes (see the various mer-NCN pincer-metal complexes reported in refs 20 and 37) show a similar distorted square planar geometry around the Pt nucleus, in which the carbon atoms of the benzylic arms (C7 and C10) are positioned above and below the plane defined by C1-N1-Pt1-N2-Cl1. For the two five-membered metallacycles, which are slightly puckered, torsion angles for the Pt1-N1-C7-C2 and Pt1-N2-C10-C6 bonds amounting to −29.0(3)° to −29.2(2)° were found in 13. The Pt-C distances of 5b, and 13 are as expected [20,37].
3. Discussion
The success of combining an organometallic complex with a selective estrogen receptor modulator (SERM) [38] is clearly demonstrated by the creative and extensive work of Jaouen and coworkers on ferrocifen (3) and hydroxyferrocifen (4) (Figure 3) [8,25,39,40,41,42]. In these organometallic derivatives of the anticancer drug Tamoxifen® (1) [43], a phenyl ring of the original Tamoxifen® is replaced by a ferrocenyl moiety. The fixation of a ferrocenyl group onto an anti-estrogen vector hydroxytamoxifen (2), provided hydroxyferrocifen (4) which showed an anti-estrogenic effect and also cytotoxic activity [44]. The cytotoxic activity of the ferrocifen derivatives is ascribed to the redox properties of the ferrocene moiety [45]. The anti-estrogenic effect of this compound was ascribed to a conformational change of the α-receptor site of the estrogen receptor containing protein, present in the tumor cell, upon binding of hydroxy-ferrocifen [44,46]. Unfortunately, the ferrocifens show a reduced relative binding affinity for the estrogen receptor, compared to that of hydroxy-Tamoxifen®, due to the steric size of the ferrocenyl group [39].
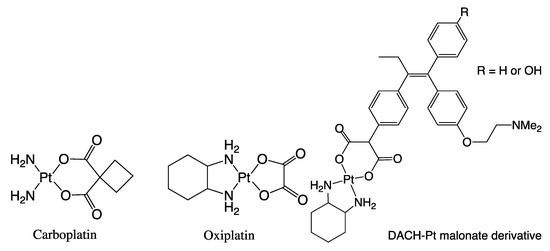
Figure 3.
Example of a coordination compound containing Pt(II) used in the screening for anti-cancer activity.
The present result shows that the use of the McMurry reaction for post-modification of the para aryl-ring position of parent NCN-pincer platinum is possible [22,37]. This reaction protocol provides a direct, short synthetic route to the platinum(II) containing tamoxifen derivatives 5 and 6 (abbreviated as pincercifens, cf. Figure 3). To accomplish this, new NCN platinum pincer acetophenone and propiophenone building blocks were developed. Both the applications and limitations of the McMurry coupling protocol were established using the free NCN ligands and their platinum complexes as starting compounds.
Similar to ferrocifen, the pincercifens 5 and 6 contain a redox active metal center, but like the ferrocenyl fragment in ferrocifen, the NCN-pincer platinum fragment is geometrically different from that of the Z-phenyl group, which is present in Tamoxifen®, cf. the structures of these compounds in the solid state, see Supporting Materials, 5 [42,47]. Both properties make the pincercifens potential interesting candidates for screening on anticancer activity.
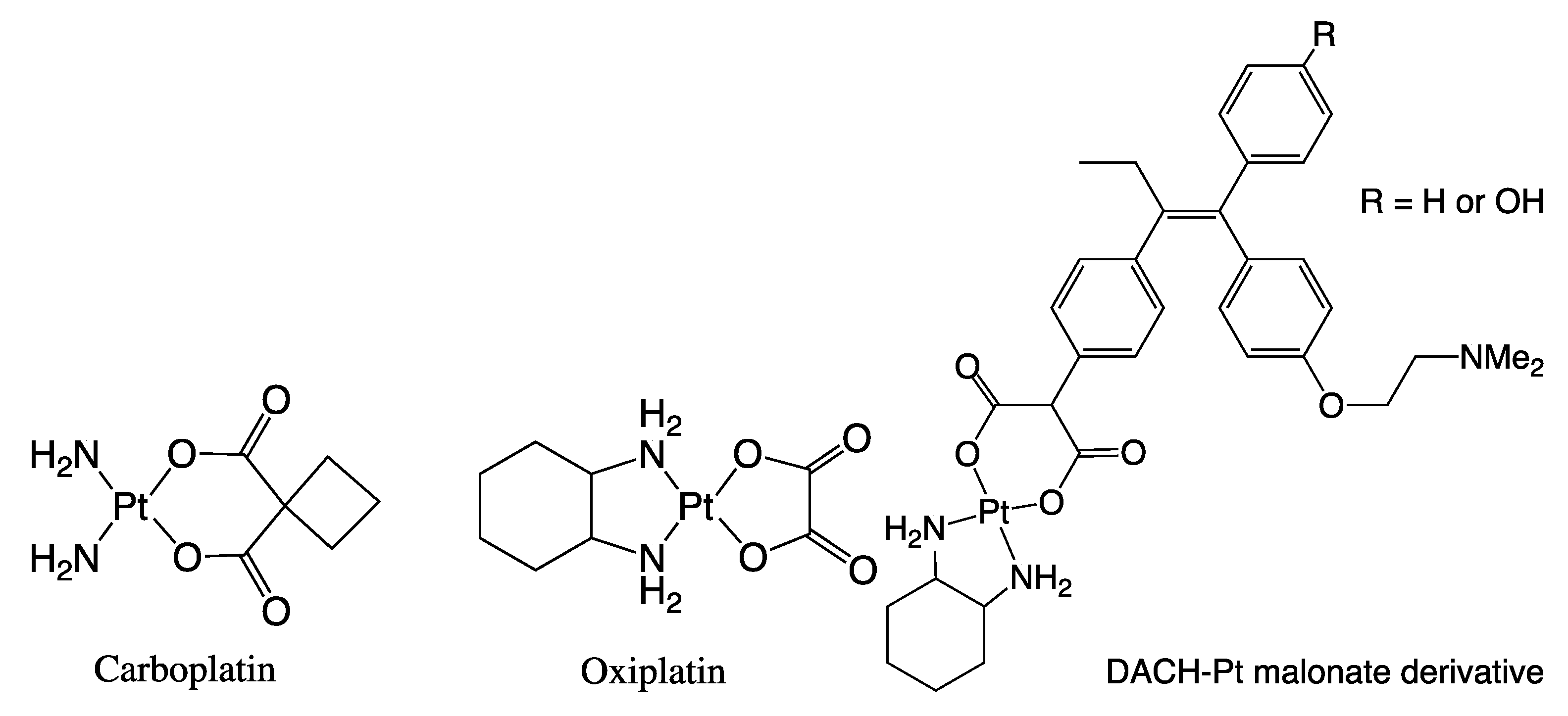
Based on the bonding motifs present in the well-known and clinically used Pt-based antitumor drugs [48,49] such as cisplatin (i.e., cis-[PtCl2(NH3)2]) [50,51] and other platinum containing complexes (Carboplatin® and Oxiplatin®: Figure 3), the Jaouen group also synthesized and screened diaminocyclohexane-malonate-platinum(II) derivatives of Tamoxifen®: DACH-Pt (Figure 3) [27]. In contrast to the stability of the organometallic fragments in ferrocifen and pincercifen, the lability of this type of platinum complexes shown in Figure 3 under physiological conditions is sometimes used to release and deliver an active metal center close to the target site [27]. However, this lability can also be problematic, especially when the metal center is released before reaching the receptor or target site [5]. In the case of ferrocifen and pincercifen, it is the stability of the respective organometallic groupings that can be an advantage, because it is shown for both complexes that the covalently bound metal center is compatible under a diversity of reaction conditions without the cleavage of the metal-C sigma bond [24].
In the mode of action of some Pt-based antitumor drugs, a cationic [PtCl(H2O)(L)2]+ species (L = coordinating ligand) that is formed in the cell by aquation of the [PtCl2(L)2] complex, binds to DNA [51,52]. Here, the pincercifens offer potentially interesting features. For an NCN-PtCl complex, it was found that the Pt-Cl grouping is able to form intermolecular non-covalent hydrogen bonds, for example, with a phenol group, i.e., forming O–H•••Cl–Pt bonds [53]. Furthermore, it was shown that [Pt(NCN)(H2O)]+ cations, either as such or as irreversibly embedded by covalent bonding in lipases, under chloride poor reaction conditions, strongly bind to, for example, pyridyl functionalized substrates [54,55]. Therefore, it is not unlikely that, under physiological conditions, an NCN-PtCl or an aquated NCN-Pt complex [55] will bind to DNA via coordination and/or hydrogen bonding, inducing cytotoxic activity.
4. Materials and Methods
4.1. Synthesis
4.1.1. General Information
All reactions involving air- or moisture-sensitive reagents were performed by standard Schlenk techniques unless stated otherwise. Pentane, THF, benzene, toluene and Et2O were distilled from Na/benzophenone, CH2Cl2 was distilled from CaH2 prior to use. The platinum precursor [Pt(p-Tol)2(SEt2)]2 [36], [PtBr(NCN-CHO-4)] (11) [20], 4-bromo-3,5-bis(dimethylamino)methyl]acetophenone (7) [22], 3,5-dimethyl-4-bromo-iodobenze (17) [20], 4-[2-(dimethylamino)ethoxy]benzophenone (23) [25], 4-[2-(dimethylamino)ethoxy]-4′-hydroxybenzophenone (24) [54], and 4-trimethylacetoxy-4′-hydroxybenzophenone (28) [54] were synthesized according to literature procedures. All other reagents were commercially available and used without further purification. 1H- and 13C{1H}-NMR spectra were recorded at 25 °C on AC 300 NMR (Bruker) or Inova 300 or 400 spectrometers (Varian) (operating frequencies: for 1H spectra at 300 and 400 MHz; for 13C spectra at 75 and 101 MHz), chemical shifts are depicted in ppm and referenced to residual solvents resonances. 195Pt{1H}-NMR spectra were recorded on a Varian Inova 300 MHz NMR spectrometer (operating at 64.4 MHz), referenced to external Na2PtCl6 (1 M in D2O, δ = 0 ppm). Elemental analyses were performed by Kolbe, Mikroanalytisches Laboratorium (Mülheim a.d. Ruhr, Germany). ESI-MS spectra were obtained from the Biomolecular Mass Spectrometry Group at Utrecht University. Infrared spectra were recorded with a Spectrum 1 FT-IR spectrometer (Perkin Elmer).
4-Bromo-3,5-bis[(dimethylamino)methyl]acetophenone (7)
IR (ATR): ῦ = 3074, 2976, 2948, 2858, 2822, 2772, 1683 (C=O), 1577, 1455, 1428, 1401, 1361, 1348, 1296, 1277, 1265, 1218, 1189, 1153, 1097, 1057, 1041, 1012, 983, 939, 912, 897, 856, 710, 685 cm−1 [15].
[PtBr(NCN-C(O)Me-4)] (14)
A mixture of 4-bromo-3,5-bis[(dimethylamino)methyl]acetophenone (7, 0.85 g, 2.71 mmol) and [Pt(Tol-4)2(SEt2)]2 (1.26 g, 1.35 mmol) in dry benzene (50 mL) was refluxed for 3 h. The slightly yellow solution was allowed to cool to RT, and all volatiles were removed in vacuo. The residue was dissolved in a minimal amount of CH2Cl2 and the product precipitated from the solution upon addition of pentane. Product 14 was isolated as a yellow powder (1.27 g, 2.50 mmol, 92%). 1H-NMR (400 MHz, CD2Cl2): δ = 7.45 (s, 2H; ArH), 4.07 (s, 3J(H,Pt) = 46.0 Hz, 4H; CH2N), 3.09 (s, 3J(H,Pt) = 38.4 Hz, 12H; CH3N), 2.48 (s, 3H; CH3C(O)); 13C{1H}-NMR (101 MHz, CD2Cl2): δ = 197.7 (C=O), 155.3 (C ipso to Pt), 143.9 (ArCC(O)), 133.5 (ArCCH2), 119.9 (3J(C,Pt) = 34.9 Hz; ArCH), 77.2 (2J(C,Pt) = 64.1 Hz; (CH2N), 55.0 (CH3N), 26.6 (CH3C(O)); IR (ATR): ῦ = 3008, 2982, 2920, 2832, 1663 (C=O), 1581, 1570, 1454, 1432, 1401, 1352, 1301, 1270, 1199, 1140, 1113, 1049, 1030, 1015, 972, 961, 943, 896, 887, 870, 836, 711, 672 cm−1; MS (ES+; CH2Cl2) m/z: 509.0188 [M + H]+; elemental analysis calcd (%) for C14H21BrN2OPt (508.31): C 33.08, H 4.16, N 5.51; found: C 33.23, H 4.20, N 5.42.
[PtCl(NCN-C(O)Me-4)] (15)
A mixture of 14 (0.29 g, 0.57 mmol) and silver triflate (AgOTf, 0.16 g, 0.62 mmol) in wet acetone (20 mL) was stirred for 30 min. The formed suspension was filtered over Celite and the filtrate was evaporated to dryness in vacuo. The residue was dissolved in wet acetone (20 mL) and an excess of NaCl was added. The resulting suspension was stirred for 1h after which the solvent was removed in vacuo. The residue was dissolved in CH2Cl2 and subsequently washed with water and brine. The organic layer was separated, dried over MgSO4, and filtered. The filtrate was evaporated to dryness and dissolved in a minimal amount of CH2Cl2. The product precipitated from the solution upon addition of pentane and afforded 15 as a pale white powder (0.22 g, 0.47 mmol, 82%). 1H-NMR (300 MHz, CD2Cl2): δ = 7.45 (s, 2H; ArH), 4.07 (s, 3J(H,Pt) = 46.2 Hz, 4H; CH2N), 3.05 (s, 3J(H,Pt) = 38.1 Hz, 12H; CH3N), 2.48 (s, 3H; CH3C(O)); 13C{1H}-NMR (75 MHz, CDCl3): δ = 197.7 (C=O), 154.2 (C ipso to Pt), 143.5 (ArCC(O)), 133.2 (ArCCH2), 119.7 (ArCH), 77.2 (NCH2), 54.3 (NCH3), 26.4 (CH3C(O)); 195Pt{1H}-NMR (64 MHz, CD2Cl2): δ = –3101; IR (ATR): ῦ = 3046, 3009, 2981, 2921, 1663 (C=O), 1580, 1568, 1456, 1433, 1397, 1355, 1300, 1274, 1233, 1197, 1141, 1115, 1049, 1017, 973, 961, 944, 896, 882, 873, 836, 712, 670 cm−1; MS (ES+; CH2Cl2) m/z: 464.0717 [M + H]+; elemental analysis calcd (%) for C14H21ClN2OPt (463.86): C 36.25, H 4.56, N 6.04; found: C 36.39, H 4.51, N 6.11.
3,5-Dimethyl-4-bromopropiophenone (18)
To a solution of 17 (10.2 g, 32.8 mmol) in dry Et2O (250 mL) at −78 °C a solution of n-BuLi (1.6M in hexane, 22 mL, 35.2 mmol) was added dropwise, and the mixture was stirred at this temperature for 30 min. Next, N,N-dimethylpropionamide (9 mL, 82.8 mmol) was added dropwise and the reaction mixture was stirred at the same temperature for 1 h. The mixture was allowed to warm to rt, and the reaction was quenched by adding an aqeous HCl solution (1 M, 40 mL). The layers were separated and the water layer was extracted with Et2O (2 × 40 mL). The combined organic fractions were subsequently washed with an aqueous HCl solution (1 M, 2 × 40 mL), a saturated sodium bicarbonate solution (2 × 40 mL), and brine (40 mL). The organic layer was dried using MgSO4, and evaporated to dryness. The residual oil was crystallized from hexane to afford 18 as colorless crystals (3.39 g, 14.1 mmol, 43%). 1H-NMR (200 MHz, CDCl3): δ = 7.64 (s, 2H; ArH), 2.95 (q, 3J(H,H) = 7.3 Hz, 2H; CH2), 2.47 (s, 6H; ArCH3), 1.21 (t, 3J(H,H) = 7.3 Hz, 3H; CH3); 13C{1H}-NMR (75 MHz, CDCl3): δ = 200.5 (C=O), 138.9 (ArCCH2), 135.4 (ArCC(O)), 133.1 (ArCBr), 127.3 (ArCH), 32.0 (CH2C(O)), 24.2 (ArCH3), 8.4 (CH3CH2); IR (ATR): ῦ = 2988, 2941, 2907, 1674 (C=O), 1586, 1464, 1436, 1410, 1377, 1349, 1291, 1177, 1093, 1044, 1028, 1007, 978, 946, 891, 855, 801, 703 cm−1; MS (ES+; CH2Cl2) m/z: 481.0164 [2M + H]+, 240.9976 [M + H]+; elemental analysis calcd (%) for C11H13BrO (241.12): C 54.79, H 5.43; found: C 54.67, H 5.38.
3,5-Bis(bromomethyl)-4-bromopropiophenone (19)
In a 100 mL one necked round-bottom flask, 18 (1.81 g, 7.5 mmol) was dissolved in MeOAc (40 mL). To the solution N-bromosuccinimide (3.18 g, 17.9 mmol) and 2,2’-azobisisobutyronitrile (0.08 g, 0.5 mmol) were added. After the connection of a reflux condenser, the stirred mixture was irradiated for 12 h with a lamp (80 W). The solvent was evaporated and the residue was dissolved in a mixture of CHCl3/hexane (1:9). The formed precipitate was filtered off and the filtrate was evaporated to dryness, and subsequently the residue was crystallized from a mixture of CHCl3/hexane (1:2) to obtain 19 as a white solid (1.48 g, 3.7 mmol, 49%). 1H-NMR (400 MHz, CDCl3): δ = 7.96 (s, 2H, ArH), 4.66 (s, 4H, CH2Br), 2.99 (q, 3J(H,H) = 7.2 Hz, 2H; CH2C(O)), 1.22 (t, 3J(H,H) = 7.2 Hz, 3H; CH3CH2); 13C{1H}-NMR (101 MHz, CDCl3): δ = 199.0 (C=O), 139.4 (ArCCH2), 136.6 (ArCC(O)), 131.7 (ArCBr), 130.5 (ArCH), 33.5 (CH2Br), 32.2 (CH2C(O)), 8.2 (CH3CH2); IR (ATR): ῦ = 3058, 2978, 2935, 2902, 2874, 1689 (C=O), 1586, 1461, 1438, 1409, 1375, 1350, 1291, 1259, 1213, 1185, 1120, 1091, 1040, 1022, 981, 936, 900, 890, 878, 853, 804, 756, 737, 726, 695, 661 cm−1; MS (ES+; CH2Cl2) m/z: 398.8243 [M + H]+; elemental analysis calcd (%) for C11H11Br3O (398.92): C 33.12, H 2.78; found: C 33.22, H 2.71.
3,5-Bis[(dimethylamino)methyl]-4-bromopropiophenone (20)
To a cold solution (0 °C) of 19 (1.25 gr, 3.1 mmol) in dry diethyl ether (50 mL), an excess of anhydrous dimethylamine (10 mL) was added and the reaction mixture was allowed to warm to rt and stirred for 3 h. The resulting white suspension was washed with an aqueous NaOH solution (2M, 2 × 50 mL) and with brine (50 mL). The organic layer was dried using MgSO4 and evaporated to dryness to yield 20 as a yellow oil (0.96 g, 2.9 mmol, 94%). 1H-NMR (400 MHz, CDCl3): δ = 7.91 (s, 2H; ArH), 3.58 (s, 4H; NCH2), 3.01 (q, 3J (H,H) = 7.2 Hz, 2H; CH2CH3), 2.31 (s, 12H; N(CH3)2), 1.20 (t, 3J (H,H) = 7.2 Hz, 3H; CH3CH2); 13C{1H}-NMR (101 MHz, CDCl3): δ = 200.7 (C=O), 139.4 (ArCCH2), 135.6 (ArCC(O)), 132.4 (ArCBr), 129.0 (ArCH), 64.0 (CH2N), 45.8 (N(CH3)2), 32.2 (CH2C(O)), 8.4 (CH3CH2); IR (ATR): ῦ = 2974, 2941, 2904, 2857, 2818, 2770, 1686 (C=O), 1585, 14,456, 1406, 1377, 1358, 1343, 1265, 1174, 1093, 1040, 1019, 986, 949, 916, 900, 853, 840, 806, 730, 700 cm−1; MS (ES+; CH2Cl2) m/z: 327.0954 [M + H]+; elemental analysis calcd (%) for C15H23BrN2O (327.26): C 55.05, H 7.08, N 8.56; found: C 54.92, H 6.93, N 8.44.
[PtBr(NCN-C(O)Et-4)] (21)
A mixture of 20 (0.96 g, 2.9 mmol) and [Pt(tol-4)2(SEt2)]2 (1.31 g, 1.4 mmol) in toluene (30 mL) was heated at 75 °C for 3 h, after which the solvent was evaporated on the rotary evaporator. The residue was dissolved in a minimal amount of CH2Cl2 and upon the addition of pentane the product precipitated. After centrifugation, 21 was obtained as a light-yellow powder (1.40 g, 2.7 mmol, 96%). 1H-NMR (300 MHz, CDCl3): δ = 7.47 (s, 2H; ArH). 4.06 (s, 3J(H,Pt) = 45.9 Hz, 4H; NCH2), 3.13 (s, 3J(H,Pt) = 37.9 Hz, 12H; N(CH3)2), 2.89 (q, 3J(H,H) = 7.4 Hz, 2H; CH2CH3), 1.20 (t, 3J(H,H) = 7.4 Hz, 3H; CH2CH3); 13C{1H}-NMR (75 MHz, CDCl3): δ = 200.9 (C=O), 154.9 (C ipso Pt), 143.8, 133.4, 119.9, 77.4 (NCH2), 55.3 (N(CH3)2), 31.8 (CH2CH3), 8.9 (CH2CH3); IR (ATR): ῦ = 3057, 3013, 2969, 2916, 2889, 2830, 2783, 1655 (C=O), 1582, 1573, 1452, 1430, 1418, 1398, 1353, 1287, 1258, 1230, 1169, 1104, 1068, 1015, 990, 971, 943, 900, 863, 838, 801, 733, 705 cm−1; MS (ES+; CH2Cl2) m/z: 523.0468 [M + H]+; elemental analysis calcd (%) for C15H23BrN2OPt (522.34): C 34.49, H 4.44, N 5.36; found: C 34.61, H 4.43, N 5.27.
[PtCl(NCN-C(O)Et-4)] (22)
To a mixture of 21 (1.40 g, 2.7 mmol) in acetone (30 mL) AgBF4 (0.56 g, 2.9 mmol) was added, causing a white suspension. The mixture was covered with aluminum foil and stirred for 30 min, after which the aluminum foil was removed and the grey suspension was stirred for another 30 min. The suspension was filtered over Celite and the residue was subsequently washed with CH2Cl2 (20 mL), acetone (10 mL) and demineralized water (10 mL). A solution of NaCl (0.47 g, 8.0 mmol) in demineralized water (10 mL) was added, and the formed layers were separated. The aqueous layer was washed with CH2Cl2 (2 × 15 mL), and the combined organic fractions were dried using MgSO4 and evaporated to dryness. The residue was dissolved in a minimal amount of CH2Cl2, from which the product precipitated upon addition of pentane. After centrifugation 22 was isolated as a light-yellow powder (1.25 g, 2.6 mmol, 96%). 1H-NMR (300 MHz, CDCl3): δ = 7.46 (s, 2H; ArH). 4.06 (s, 3J(H,Pt) = 46.4 Hz, 4H; NCH2), 3.09 (s, 3J(H,Pt) = 37.9 Hz, 12H; N(CH3)2), 2.89 (q, 3J(H,H) = 7.4 Hz, 2H; CH2CH3), 1.21 (t, 3J(H,H) = 7.4 Hz, 3H; CH2CH3); 13C{1H}-NMR (75 MHz, CDCl3): δ = 200.6 (C=O), 153.9 (C ipso Pt), 143.5, 133.1, 119.5, 77.8 (NCH2), 54.4 (N(CH3)2), 31.5 (CH2CH3), 8.6 (CH2CH3); 195Pt{1H}-NMR (64 MHz, CD2Cl2): δ = –3116; IR (ATR): ῦ = 2976, 2920, 1661 (C=O), 1582, 1570, 1454, 1432, 1398, 1350, 1317, 1287, 1168, 1105, 1068, 1014, 978, 964, 944, 899, 880, 863, 836, 805, 735, 710 cm−1; MS (ES+; CH2Cl2) m/z: 479.1310 [M + H]+, 442.1285 [M–Cl]+; elemental analysis calcd (%) for C15H23N2OPtCl (477.89): C 37.70, H 4.85, N 5.86, found: C 37.86, H 4.95, N 5.76.
4-[2-(Dimethylamino)ethoxy]-4′-hydroxybenzophenone (24)
The compound was synthesized according to a modified reaction procedure of Gauthier and coworkers [35], i.e., K2CO3 was used as the base and acetone was used as the solvent (instead of Cs2CO3 and DMF, respectively). 4,4′-Dihydroxybenzophenone (5.0 g, 23.3 mmol) was dissolved in a mixture of acetone (240 mL) and water (10 mL). To this mixture K2CO3 (10.0 g, 72.4 mmol), and 2-(dimethylamino)ethyl chloride hydrochloride (3.8 gr, 26.4 mmol) were added and the mixture was heated at reflux for 20 h. After cooling, the mixture was evaporated to dryness and taken up in a mixture of EtOAc:MeOH (1:1, 100 mL) and filtered. The filtrate was evaporated to dryness and the residue was triturated from CH2Cl2 with pentane. The residue was purified by silicagel column chromatography (EtOAc:MeOH, 1:1). The second fraction contained the desired product 24, which was triturated from CH2Cl2 with pentane to yield the 24 as a light-yellow powder (1.4 g, 4.9 mmol; 21%). The analytical data obtained for 24 is in agreement with the published data. 1H-NMR (300 MHz, CD3OD): δ = 7.78 (d, 3J = 8.5 Hz, 2H; ArH), 7.71 (d, 3J = 8.5 Hz, 2H; ArH), 7.12 (d, 3J = 8.5 Hz; ArH), 6.92 (d, 3J = 8.5 Hz, 2H; ArH), 4.31 (t, 3J = 5.2 Hz, 2H; CH2O), 3.05 (t, 3J = 5.2 Hz, 2H; CH2N), 2.55 (s, 6H; CH3); 13C{1H}-NMR (300 MHz, CD3OD): δ = 196.8 (C=O), 163.4, 163.4, 133.7, 133.3, 132.3, 130.3, 116.2, 115.2, 66.3, 58.7, 45.5.
Attempted Protection of 24 with TBDMS-Cl
To a solution of 24 (0.26 g, 0.93 mmol) in dry THF (20 mL), t-butyldimethylsilyl chloride (TBDMS-Cl, 0.14 g, 0.94 mmol) and imidazole (64 mg, 0.94 mmol) were added. The mixture was stirred for 3 h and the suspension was filtered over Celite. The filtrate was extracted with ethyl acetate (3 × 20 mL) and the combined organic layers were washed with brine (2 × 100 mL), dried using MgSO4, filtered and evaporated to dryness. Only starting compound 24 was isolated. The same reaction was performed using NaH (60% dispersion in mineral oil) as base, again only starting compound 24 was isolated.
4-Trimethylacetoxy-4′-[2-(dimethylamino)ethoxy]benzophenone (25)
A mixture of 28 (7.0 gr, 23.5 mmol), 2-(dimethylamino)ethyl chloride hydrochloride (8.5 g, 59.0 mmol), and K2CO3 (15.0 g, 0.11 mol) in acetone (250 mL) was heated at reflux for 20 h. After cooling to rt the mixture was evaporated to dryness, and subsequently EtOAc (150 mL) and water (100 mL) were added. The layers were separated and the aqueous layer was extracted with EtOAc (2 × 100 mL). The combined organic fractions were washed with brine (2 × 100 mL), dried using MgSO4, and evaporated to dryness. The residue was purified using column chromatography (Al2O3, CH2Cl2). The second fraction that came off the column contained the product 25 as a white solid (1.74 g, 4.7 mmol, 20%). 1H-NMR (400 MHz, CDCl3): δ = 7.81 (d, 3J = 8.8 Hz, 2H; ArH), 7.80 (d, 3J = 8.8 Hz, 2H; ArH), 7.17 (d, 3J = 8.8 Hz, 2H; ArH), 6.98 (d, 3J = 8.8 Hz, 2H; ArH), 4.24 (t, 3J = 5.4 Hz, 2H; CH2), 2.91 (t, 3J = 5.4 Hz, 2H; CH2), 2.47 (s, 6H; N(CH3)2), 1.38 (s, 9H; C(CH3)3); 13C{1H}-NMR (101 MHz, CDCl3): δ = 194.4 (Ar2C=O), 176.7, 162.1, 154.1, 135.5, 132.5, 131.3, 130.4, 121.4, 114.2, 65.7 (OCH2), 57.8 (NCH2CH2), 45.5 ((CH3)2NCH2CH2), 39.2 (C(CH3)3, 27.1 (C(CH3)3; IR (ATR): ῦ = 2956, 2818, 2769, 1750 (C=O pivaloyl), 1639 (C=O benzophenone), 1600 and 1575 (C–O), 1461, 1252, 1108, 1030, 928, 899, 845, 759, 682 cm−1; MS (ES+; CH2Cl2) m/z: 370.13 [M + H]+; elemental analysis calcd (%) for C22H27NO4 (369.45): C 71.51, H 7.37, N 3.79, found: C 71.39, H 7.32, N 3.71.
4.1.2. McMurry Reaction
In a flame dried Schlenk tube an amount of TiCl4(THF)2 (6 equiv. in respect to the platinum aldehyde or ketone) was transferred via a glass bend from the TiCl4(THF)2 storage Schlenk. The complex was dissolved in dry THF (20 mL), and the yellow solution was added via a cannula to a stirred suspension of Zn (10 equiv. with respect to the platinum aldehyde or ketone) in dry THF (10 mL) at −20 °C. The mixture was heated at reflux for 2 h, and then allowed to cool to rt. To this solution a mixture of the platinum complex (1 equiv.) and the ketone (6 equiv. with respect to the platinum aldehyde or ketone) in dry THF (20 mL) was added dropwise, and the resulting mixture was heated at reflux for 16 h. After cooling to rt the mixture was hydrolyzed with a 10% K2CO3 solution (50 mL). Then EtOAc (100 mL) was added and the mixture was stirred for 15 min. The formed slurry was filtered over Celite, and from the filtrate the organic layer was separated. The organic layer was washed with brine (20 mL) and dried using MgSO4 and evaporated to dryness to afford the product with the reported yields. When necessary the compounds were further purified as indicated below.
Attempted Synthesis of 1-(1,1-Diphenyl-1-propenyl)-4-bromo-3,5-bis[(dimethylamino)methyl]Benzene (9); Formation of 1-(1,1-Diphenyl-1-propenyl)-3,5-bis[(dimethylamino)methyl]Benzene (10)
The reaction was performed according to the general McMurry procedure, using 4-bromo-3,5-bis[(dimethylamino)methyl]acetophenone 7 (0.28 g, 0.89 mmol) (instead of the platinum ketone) and benzophenone (as the ketone). After hydrolysis, the reaction mixture was extracted using diethyl ether, and the organic layer was dried over MgSO4. After filtration the volatiles were removed on the rotary evaporator leaving a product mixture. The crude product (0.9 g) was further purified by silica gel column chromatography, first eluting with CH2Cl2 to remove the excess of benzophenone, secondly a gradient elution starting with a CH2Cl2:MeOH (49:1) mixture changing to CH2Cl2:MeOH (9:1) ending with a solvent mixture of CH2Cl2:MeOH:pyridine (44:5:1). From the 1H-NMR spectrum it became clear that the isolated fraction did not contain the desired compound 9, but mainly consisted of the dehalogenated compound 10, together with a small amount of other impurities. For 10: 1H-NMR (300 MHz, CDCl3): δ = 7.39-7.22 (m, ArH), 7.14 (s, 2H; ArH), 7.06–6.92 (m, ArH), 6.81 (s, 1H; ArH), 3.41 (s, 4H; CH2), 2.11 (s, 12H; (CH3)2N), 2.01 (s, 3H; CH3).
Attempted Synthesis of 3,3′,5,5′-Tetra(dimethylamino)methyl-4,4′-bisplatinumbromide-stilbene (12); Formation of 3,3′,5,5′-Tetra(dimethylaminomethyl)-4,4′-bisplatinum Halide-Benzophenone (13), (Halide = Br and Cl)
The reaction was performed according to the general McMurry procedure, using [PtBr(NCN-CHO-4)] 11 (0.60 g, 1.2 mmol), yielding a product mixture (340 mg) containing the starting aldehyde 11 and other compounds. The product mixture was precipitated from CH2Cl2 with pentane (2 ×), yielding a yellow powder (220 mg), consisting of multiple compounds. The mixture was crystallized from toluene giving yellow crystals (26 mg), which were determined with X-ray crystallography to be the bis(platinum halide)benzophenone 13 (halide = Br and Cl). Analytical data obtained for 13: 1H-NMR (400 MHz, CD2Cl2): δ = 7.26 (s, 4H; ArH), 4.08 (s, 3J(H,Pt) = 42.0 Hz, 8H; CH2N), 3.12 and 3.07 (s, 24H; CH3N, X = Br or Cl); 13C{1H}-NMR (101 MHz, CD2Cl2): δ = 196.8 (C=O), 153.8 and 153.3 (C ipso to Pt, X = Br or Cl), 143.6 (ArCCH2), 134.2 (ArCC(O)), 121.5 (ArCH), 77.5 and 77.3 (CH2N, X = Br or Cl), 55.1 and 54.4 (CH3N); 195Pt{1H}-NMR (64 MHz, CD2Cl2): δ = −3107 and −3130; IR (ATR): ῦ = 3047, 3016, 2976, 2921, 2889, 2820, 2790, 1614 (C=O), 1579, 1557, 1467, 1456, 1425, 1402, 1390, 1346, 1317, 1265, 1177, 1144, 1098, 1068, 1017, 984, 960, 949, 885, 838, 777, 761, 718, 703 cm−1; MS (ES+; CH2Cl2) m/z: 949.0305 [M + H + Cl]+ (X = Cl, Br), 914.1157 [M + H]+ (X = Cl, Br), 905.1085 [M+H+Cl]+ (X = Cl), 869.1773 [M + H]+ (X = Cl), 833.1945 [M + H–Cl]+ (X = Cl).
1-(2-(1-[(4-Dimethylaminoethoxy)phenyl]-1-phenyl-1-butenyl))-4-PtCl-3,5-bis[(dimethylamino)methyl]) Benzene (5)
The reaction was performed according to the general McMurry procedure, using 22 (0.58 g, 1.2 mmol) and 23 (1.96 g, 7.3 mmol), yielding a crude reaction product (2.29 g). The crude product was dissolved in a minimal amount of CH2Cl2, and upon addition of pentane a solid precipitated, which was isolated after centrifugation. This procedure was repeated and a solid was isolated (510 mg) containing 5 as the E/Z mixture (3:7 according to NMR), which was further purified with silica gel chromatography. A side product was eluted from the column with CH2Cl2, followed by a gradient elution, changing the eluent from pure CH2Cl2 to CH2Cl2:MeOH (9:1). One of the first fractions and one of the final fractions contained the separate E- (5a, 20 mg) and Z-isomer (5b, 15 mg), respectively, in pure form. The rest of the product fractions contained the product in a changing E:Z ratio. In total 310 mg (0.43 mmol, 36%) of product 5 was isolated, indicating the sum of the separately isolated product fractions. For 5a: 1H-NMR (300 MHz, CDCl3): δ = 7.12 (d, 3J(H,H) = 8.5 Hz, 2H, ArH), 6.97 (m, 3H; ArH), 6.89–6.82 (m, 4H, ArH), 6.51 (s, 2H; ArH), 4.13 (t, 3J(H,H) = 5.8 Hz, 2H; OCH2), 3.86 (s, 3J(H,Pt) = 46.4 Hz, 4H; NCH2), 3.02 (s, 3J(H,Pt) = 35.7 Hz, 12H; N(CH3)2), 2.82 (t, 3J(H,H) = 5.8 Hz, 2H; NCH2CH2), 2.45 (q, 3J(H,H) = 7.4 Hz, 2H; CH2CH3), 2.41 (s, 6H; CH2CH2N(CH3)2), 0.97 (t, 3J(H,H) = 7.4 Hz, 3H; CH3CH2); 13C{1H}- NMR (75 MHz, CDCl3): δ = 157.3 (ArCOCH2), 143.8, 143.0 (C ipso to Pt), 142.8, 142.5, 137.4, 137.3, 136.5, 130.8, 130.5, 127.1, 125.3, 120.7, 114.1, 77.5 (ArCH2N), 65.6 (OCH2), 58.2 (NCH2CH2), 54.2 (ArCH2N(CH3)2), 45.6 ((CH3)2NCH2CH2), 29.7 (C=CCH2CH3), 13.9 (CH3CH2C=C); 195Pt{1H}-NMR (64 MHz, CDCl3): δ = −3208; IR (ATR): ῦ = 3007, 2966, 2923, 2869, 2853, 2779, 1683, 1604, 1508, 1463, 1450, 1397, 1370, 1337, 1295, 1284, 1270, 1251, 1234, 1190, 1174, 1137, 1124, 1104, 1074, 1024, 982, 965, 941, 863, 835, 811, 763, 698, 662 cm−1; MS (ES+; CH2Cl2) m/z: 716.21 [M+H]+, 679.31 [M–Cl]+; elemental analysis calcd (%) for C32H42ClN3OPt (715.23): C 53.74 0, H 5.92, N 5.88, found: C 53.66, H 6.05, N 5.79.
For 5b: 1H-NMR (300 MHz, CDCl3): δ = 7.35–7.19 (m, 5H; C6H5), 6.75 (d, 3J = 8.8 Hz, 2H; ArH), 6.56 (d, 3J = 8.8 Hz, 2H; ArH), 6.55 (s, 2H; ArH), 3.99 (t, 3J(H,H) = 5.6 Hz, 2H; OCH2), 3.89 (s, 3J(H,Pt) = broad, 4H; NCH2), 3.04 (s, 3J(H,Pt) = 34.6 Hz, 12H; N(CH3)2), 2.74 (t, 3J(H,H) = 5.6 Hz, 2H; NCH2CH2), 2.39 (q, 3J(H,H) = 7.4 Hz, 2H; CH2CH3), 2.36 (s, 6H; CH2CH2N(CH3)2), 0.94 (t, 3J(H,H) = 7.4 Hz, 3H; CH3CH2); 13C{1H}-NMR (75 MHz, CDCl3): δ = 156.4 (ArCOCH2), 144.1, 143.0 (Cipso to Pt), 142.6, 142.2, 137.5, 137.2, 136.1, 131.9, 129.4, 128.1, 126.4, 120.7, 113.3, 77.6 (ArCH2N), 65.5 (OCH2), 58.1 (NCH2CH2), 54.3 (ArCH2N(CH3)2), 45.6 ((CH3)2NCH2CH2), 29.7 (C=CCH2CH3), 13.9 (CH3CH2C=C); 195Pt{1H}- NMR (64 MHz, CDCl3): δ = −3207; IR (ATR): ῦ = 3032, 2955, 2922, 2868, 2855, 2767, 1667, 1603, 1571, 1508, 1492, 1449, 1402, 1372, 1335, 1286, 1244, 1173, 1138, 1124, 1024, 960, 945, 913, 865, 829, 804, 774, 727, 703, 667 cm−1; MS (ES+; CH2Cl2) m/z: 716.25 [M + H]+, 679.26 [M–Cl]+; elemental analysis calcd (%) for C32H42ClN3OPt (715.23): C 53.74 0, H 5.92, N 5.88, found: C 53.82, H 6.08, N 5.81.
1-(4-[2-(Dimethylamino)ethoxy]phenyl)-1-(4-trimethylacetoxyphenyl)-2-(NCN-PtCl)but-1-ene (26)
The reaction was performed according to the general McMurry procedure, using 22 (378 mg, 0.79 mmol) and 25 (1.7 g, 4.7 mmol). After extraction of the reaction mixture with CH2Cl2, the crude product was isolated after evaporation and was purified with silica gel chromatography with a gradient elution. The eluent was changed from CH2Cl2:MeOH (9:1) to CH2Cl2:MeOH (5:1) to CH2Cl2:MeOH:Et3N (80:19:1). The second fraction collected contained the desired product 26 (E-:Z-mixture, 7:9, based on NMR) as a white solid (0.40 g, 0.49 mmol, 62%). A subsequent separation of the isomers was performed by silica gel flash chromatography (SiO2, CH2Cl2:MeOH, 9:1) None of the fractions contained the pure (either E- or Z-) isomer, best ratio was 1:49 (based on NMR). Major isomer of 26. 1H-NMR (400 MHz, C6D6): δ = 7.27 (d, 3J(H,H) = 8.8 Hz, 2H; ArH), 7.07 (d, 3J(H,H) = 8.4 Hz, 2H; ArH), 6.86 (d, 3J(H,H) = 8.8 Hz, 2H; ArH), 6.75 (d, 3J(H,H) = 8.4 Hz, 2H; ArH), 6.54 (s, 2H; ArH), 3.83 (t, 3J(H,H) = 6.0 Hz, 2H; OCH2CH2), 3.27 (s, 3J(H,Pt) = broad, 4H; ArCH2N(CH3)2), 2.69 (q, 3J(H,H) = 7.6 Hz, 2H; CH2CH3), 2.67 (s, 3J(H,Pt) = broad, 12H; ArCH2N(CH3)2), 2.50 (t, 3J(H,H) = 5.6 Hz, 2H; OCH2CH2), 2.08 (s, 6H; CH2CH2N(CH3)2), 1.18 (t, 3J(H,H) = 7.6 Hz, 3H; CH3CH2), 1.07 (s, 9H; C(O)(CH3)3); 13C{1H}-NMR (101 MHz, C6D6): δ = 177.2 (C=O), 157.6, 149.1, 143.7 (Cipso to Pt), 143.1, 137.3, 136.4, 131.8, 130.7, 130.5, 121.3, 120.9, 120.4, 114.3, 113.4, 77.7 (ArCH2N), 65.8 (OCH2), 58.2 (NCH2CH2), 54.3 (ArCH2N(CH3)2), 45.5 ((CH3)2NCH2CH2), 39.0 (C(CH3)3), 29.9 (C=CCH2CH3), 27.0 (C(CH3)3), 13.7 (CH3CH2C=C); δ = 195Pt{1H}-NMR (64 MHz, CD2Cl2): δ = −3192; IR (ATR): ῦ = 2971, 2928, 1748 (C=O stretch of pivaloyl group), 1606 (C–O stretch), 1505, 1462, 1241, 1198, 1164, 1116, 1029, 1016, 833 cm−1. Minor isomer of 26. 1H-NMR (400 MHz, C6D6): δ = 7.33 (d, 3J(H,H) = 8.8 Hz, 2H; ArH), 7.08 (d, 3J(H,H) = 8.4 Hz) 7.00 (d, 3J = 8.8 Hz, 2H; ArH), 6.60 (d, 3J = 8.4 Hz, 2H; ArH), 6.59 (s, 2H; ArH), 3.65 (t, 3J = 5.8 Hz, OCH2CH2), 3.20 (s, 3J(H,Pt) = broad, 4H; ArCH2N(CH3)2), 2.63 (s, 3J(H,Pt) = broad, 12H; ArCH2N(CH3)2), 2.58 (q, 3J(H,H) = 7.6 Hz, 2H; CH2CH3), 2.37 (t, 3J(H,H) = 6.0 Hz, 2H; OCH2CH2), 1.99 (s, 6H; CH2CH2N(CH3)2), 1.21 (s, 9H; C(O)(CH3)3), 0.83 (t, 3J(H,H) = 7.0 Hz, 3H; CH3CH2).
4.2. X-ray Crystal Structure Determination of Compound 13
C25H36Br0.82Cl1.18N4OPt2 + disordered solvent, Fw = 906.11[*], yellow needle, 0.39 × 0.12 × 0.09 mm3, monoclinic, C2/c (no. 15), a = 21.3275(11), b = 13.8471(9), c = 11.9515(8) Å, β = 93.981(5)°, V = 3521.1(4) Å3, Z = 4, Dx = 1.709 g/cm3[*], μ = 8.98 mm−1[*]. The diffraction experiment was performed on a Nonius KappaCCD diffractometer with rotating anode and graphite monochromator (λ = 0.71073 Å) at a temperature of 150(2) K up to a resolution of (sin θ/λ)max = 0.65 Å−1. The Eval-14 software [56] was used for the intensity integration. An analytical absorption correction was performed with PLATON [57] (correction range 0.06–0.58). The SortAV software [58] was used for scaling and merging. A total of 34116 reflections was measured, 4050 reflections were unique (Rint = 0.042), 3435 reflections were observed [I > 2σ(I)]. The structure was solved with Patterson methods using DIRDIF99 [59]. Structure refinement was performed with SHELXL-2018 [60] on F2 of all reflections. The crystal structure contains large voids (1068 Å3/unit cell) filled with severely disordered solvent molecules. Their contribution to the structure factors was secured by the SQUEEZE algorithm [61] resulting in 344 electrons/unit cell. Non-hydrogen atoms were refined freely with anisotropic displacement parameters. Hydrogen atoms were introduced in calculated positions and refined with a riding model. On the halogen site there is substitutional disorder between bromine and chlorine. 163 Parameters were refined with two restraints (Pt-Br and Pt-Cl distances). R1/wR2 [I > 2σ(I)]: 0.0169/0.0357. R1/wR2 [all refl.]: 0.0242/0.0375. S = 1.038. Residual electron density between −0.68 and 0.73 e/Å3. Geometry calculations and checking for higher symmetry was performed with the PLATON program [57]. Derived values do not contain the contribution of the disordered solvent. For detailed information on 5b, see [34].
Supplementary Materials
The following are available online, Figure S1: ESI mass spectrum of bis(NCN-pincer platinum halide)benzophenone 13, showing the halogen scrambling (Br− vs. Cl−) on the platinum centers, Table S1: Relevant 13C{1H}, 195Pt{1H} NMR and IR data including those of the NCN arylpincer platinum halide substituted compoundsa (for all data see Experimental Section). Table S2: Selected bond lengths [Å] and angles [°] of 13. CCDC 2062971 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/structures. Comparison of the structural features of 5b, 1 and 3.
Author Contributions
Conceptualization, G.D.B., G.P.M.v.K. and G.v.K.; methodology, R.A.G., G.G. and G.R.; realization, G.D.B. and T.J.K.; X-ray analysis, M.L.; writing/original draft preparation, G.D.B. and G.v.K.; supervision, G.P.M.v.K. and G.v.K.; funding acquisition, G.v.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research was partly funded by the Council for Chemical Sciences of the Netherlands Organization for Scientific Research (NWO/CW) G.D.B., M.L.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data are contained within the article and the Supplementary Materials.
Acknowledgments
R.A.G., is indebted to Ryerson University and the Dean’s Research Fund for support of this work.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds are not available from the authors.
References
- Collot, J.; Gradinaru, J.; Humbert, N.; Skander, M.; Zocchi, A.; Ward, T.R. Artificial Metalloenzymes for Enantioselective Catalysis Based on Biotin-Avidin. J. Am. Chem. Soc. 2003, 125, 9030–9031. [Google Scholar] [CrossRef]
- Desguin, B.; Zhang, T.; Soumillion, P.; Hols, P.; Hu, J.; Hausinger, R.P. A tethered niacin-derived pincer complex with a nickel-carbon bond in lactate racemase. Science 2015, 349, 66–69. [Google Scholar] [CrossRef]
- Ho, C.-L.; Wong, W.-Y. Organometallic Versus Organic Molecules for Energy Conversion in Organic Light-Emitting Diodes and Solar Cells. In Organometallics and Related Molecules for Energy Conversion; Wong, W.-Y., Ed.; Springer: Berlin/Heidelberg, Germany, 2015; pp. 1–29. [Google Scholar] [CrossRef]
- Hussein, M.A.; Asiri, A.M. Organometallic Ferrocene- and Phosphorus-Containing Polymers: Synthesis and Characterization. Des. Monomers Polym. 2012, 15, 207–251. [Google Scholar] [CrossRef]
- Allardyce, C.S.; Dorcier, A.; Scolaro, C.; Dyson, P.J. Development of Organometallic (Organo-Transition Metal) Pharmaceuticals. Appl. Organometal. Chem. 2005, 19, 1–10. [Google Scholar] [CrossRef]
- Hirao, T.; Moriuchi, T. Advances in Bioorganometallic Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; pp. 1–472. [Google Scholar]
- Bruijnincx, P.C.A.; Sadler, P.J. New trends for metal complexes with anticancer activity. Curr. Opin. Chem. Biol. 2008, 12, 197–206. [Google Scholar] [CrossRef]
- Jaouen, G.; Vessières, A.; Top, S. Ferrocifen type anticancer drugs. Chem. Soc. Rev. 2015, 44, 8802–8817. [Google Scholar] [CrossRef]
- Dehand, J.; Pfeffer, M. Cyclometallated Compounds. Coord. Chem. Rev. 1976, 18, 327–352. [Google Scholar] [CrossRef]
- Dehand, J.; Jordanov, J.; Pfeffer, M. Reactivity of primary amines co-ordinated to palladium and platinum. Part II. cyclometallation and rearrangement of the co-ordinated ligands. J. Chem. Soc. Dalton Trans. 1976, 1553–1556. [Google Scholar] [CrossRef]
- Abbenhuis, H.C.L.; Pfeffer, M.; Sutter, J.P.; De Cian, A.; Fischer, J.; Li, J.H.; Nelson, J.H. Carbon-Carbon and Carbon-Nitrogen Bond Formation Mediated by Ruthenium(II) Complexes: Synthesis of (1H)-Isoquinolinium Derivatives. Organometallics 1993, 12, 4464–4472. [Google Scholar] [CrossRef]
- Pfeffer, M.; Sutter, J.P.; Urriolabeitia, E.P. Ruthenium Mediated Carbon-Carbon and Carbon-Nitrogen bond formation: Parameters governing the reactivity of the metal centre. Bull. Soc. Chim. Fr. 1997, 134, 947–954. [Google Scholar]
- Ryabov, A.D.; Sukharev, V.S.; Alexandrova, L.; Le Lagadec, R.; Pfeffer, M. New Synthesis and New Bio-Application of Cylometalated Ruthenium(II) Complexes for Fast Mediated Electron transfer with Peroxidase and Glucose Oxidase. Inorg. Chem. 2001, 40, 6529–6532. [Google Scholar] [CrossRef] [PubMed]
- Gaiddon, C.; Jeannequin, P.; Bishoff, P.; Pfeffer, M.; Sirlin, C.; Loeffler, J.P. Ruthenium(II)-derived organometallic compounds induce cytostatic and cytotoxic effects on mammalian cancer cell lines through p53-dependent and p53-independent mechanisms. J. Pharma. Exper. Ther. 2005, 315, 1403–1411. [Google Scholar] [CrossRef]
- Meng, X.; Leyva, L.M.; Jenny, M.; Gross, I.; Benosman, S.; Fricker, B.; Harlepp, S.; Hebraud, P.; Boos, A.; Wlosik, P.; et al. A ruthenium containing organometallic compound reduces tumor growth through induction of the endoplasmic reticulum stress gene CHOP. Cancer Res. 2009, 69, 5458–5466. [Google Scholar] [CrossRef] [PubMed]
- Licona, C.; Delhorme, J.-B.; Riegel, G.; Vidimar, V.; Cerón-Camacho, R.; Boff, B.; Venkatasamy, A.; Tomasetto, C.; da Silva Figueirdo Celestino Gomes, P.; Rognan, D.; et al. Anticancer activity of ruthenium and osmium cyclometalated compounds: Identification of ABCB1 and EGFR as resistance mechanisms. Inorg. Chem. Front. 2020, 7, 678–688. [Google Scholar] [CrossRef]
- Van Koten, G.; Leusink, A.J.; Noltes, J.G. Stable arylcopper compounds containing 2-(dimethylamino)methyl or 2-methoxymethyl groups at the aryl nucleus. J. Chem. Soc. Chem. Commun. 1970, 1107–1108. [Google Scholar] [CrossRef]
- Jastrzebski, J.T.B.H.; van Koten, G.; Goubitz, K.; Arlen, C.; Pfeffer, M. Synthesis of 8-(dimethylamino)-1-naphthyllithium Etherate: Its Structure in the Solid (X-ray) and in Solution (7Li and 1H NMR). J. Organomet. Chem. 1983, 246, C75–C79. [Google Scholar] [CrossRef]
- Wehman, E.; van Koten, G.; Knaap, C.T.; Ossor, H.; Pfeffer, M.; Spek, A.L. Synthesis of Cyclometalated (8-methoxynaphthyl)- and {8-(dimethylamino)naphthyl} Platinum Compounds and Their Reactivity Toward Electrophiles. X-ray Molecular Structures of Square- planar (8-methoxynaphthyl)platinum(II) Chloride bis (diethyl sulfide) and bis (8-methoxy naphthyl)platinum(II) Diethyl Sulfide and Octahedral fac-bis(8-methoxynaphthyl)Methyl platinum(IV) Iodide. Inorg. Chem. 1988, 27, 4409–4417. [Google Scholar] [CrossRef]
- Albrecht, M.; van Koten, G. Platinum Group Organometallics Based on ‘Pincer’ Ligands: Sensors, Switchses and Catalysis. Angew. Chem. Int. Ed. Engl. 2001, 40, 3750–3781. [Google Scholar] [CrossRef]
- Wieczorek, B.; Dijkstra, H.P.; Egmond, M.R.; Klein Gebbink, R.J.M.; van Koten, G. Incorporating ECE-Pincer Metal Complexes as Functional Building Blocks in Semisynthetic Metalloenzymes, Supramolecular Polypeptide Hybrids, Tamoxifen Derivatives, Biomarkers and Sensors. J. Organomet. Chem. 2009, 694, 812–822. [Google Scholar] [CrossRef]
- van de Kuil, L.A.; Luitjes, J.; Grove, D.M.; Zwikker, J.W.; van der Linden, J.G.M.; Roelofsen, A.M.; Jenneskens, L.W.; Drenth, W.; van Koten, G. Electronic Tuning of Arylnickel(II) Complexes by Para Substitution of the Terdentate Monoanionic 2,6-Bis[(dimethylamino)methyl]phenyl Ligand. Organometallics 1994, 13, 468–477. [Google Scholar] [CrossRef]
- Valdés, H.; González-Sebastián, L.; Morales-Morales, D. Aromatic para-functionalized NCN pincer compounds. J. Organometal. Chem. 2017, 845, 229–257. [Google Scholar] [CrossRef]
- Rodríguez, G.; Albrecht, M.; Schoenmaker, J.; Ford, A.; Lutz, M.; Spek, A.L.; van Koten, G. Bifunctional Pincer-type Organometallics as Substrates for Organic Transformations and as Novel Building Blocks for Polymetallic Materials. J. Am. Chem. Soc. 2002, 124, 5127–5138. [Google Scholar] [CrossRef]
- Canty, A.J.; Ariafard, A.; van Koten, G. Computational Analysis of Mesomerism in para-Substituted mer- NCN Pincer Platinum(II) Complexes, Including its Relationships with Hammett σp Substituent Parameters. Chem. Eur. J. 2020, 67, 15629–15635. [Google Scholar] [CrossRef] [PubMed]
- Top, S.; Dauer, B.; Vaissermann, J.; Jaouen, G. Facile route to ferrocifen, 1-[4-(2-dimethylaminoethoxy)]-1-(phenyl-2-ferrocenyl-but- 1-ene), first organometallic analogue of tamoxifen, by the McMurry reaction. J. Organometal. Chem. 1997, 541, 355–361. [Google Scholar] [CrossRef]
- Top, S.; Kaloun, E.B.; Vessieres, A.; Leclercq, G.; Laios, I.; Ourevitch, I.; Deuschel, C.; McGlinchey, M.J.; Jaouen, G. Tamoxifen Derivatives for Delivery of the Antitumoral (DACH)Pt Group: Selective Synthesis by McMurry Coupling, and Biochemical Behaviour. ChemBioChem 2003, 4, 754–761. [Google Scholar] [CrossRef]
- McMurry, J.E. Carbonyl-coupling reactions using low-valent titanium. Chem. Rev. 1989, 89, 1513–1524. [Google Scholar] [CrossRef]
- Richardson, W.H. Haloaromatic Substituted Olefins by the MC Murry Olefin Synthesis. Synth. Commun. 1981, 11, 895–899. [Google Scholar] [CrossRef]
- Donkervoort, J.G.; Jastrzebski, J.T.B.H.; Deelman, B.J.; Kooijman, H.; Veldman, N.; Spek, A.L.; van Koten, G. Novel Aryltitanium(IV) Complexes Containing η3-NCN-Pseudofacial- and η3-NCN-Meridional-Bonded [C6H3(CH2NMe2)2-2,6]- (Abbreviated as NCN) Ligands. The Crystal Structures of [TiCl2(η3-fac-NCN)(O-i-Pr)], [TiCl(η3-mer-NCN)(O-i-Pr)(OTf)] Containing an η1-O-Bonded Triflate Anion, and the Seven-Coordinated [Ti(η3-mer-NCN)(O-i-Pr)(OTf)2] Containing η1-O- and η2-O,O‘-Bonded Triflate Anions. Organometallics 1997, 16, 4174–4184. [Google Scholar] [CrossRef]
- Casado Lacabra, M.A.; Canty, A.M.; Lutz, M.; Patel, J.; Spek, A.L.; Sun, H.; van Koten, G. Mono(p-tolyl)platinum(II) and bis(p-tolyl)platinum(II) complexes of diethylsulfide as reagents for organoplatinum synthesis. Structures of [Pt(p-Tol)2(μ-SEt2)]2 and PtCl(p-Tol)(bpy) (bpy = 2,2′-bipyridine). Inorg. Chim. Acta 2016, 442, 167–171. [Google Scholar]
- Amijs, C.H.M.; van Klink, G.P.M.; van Koten, G. Carbon tetrachloride free benzylic brominations of methyl aryl halides. Green Chem. 2003, 5, 470–474. [Google Scholar] [CrossRef]
- Meegan, M.J.; Hughes, R.B.; Lloyd, D.G.; Williams, D.C.; Zisterer, D.M. Flexible Estrogen Receptor Modulators: Design, Synthesis, and Antagonistic Effects in Human MCF-7 Breast Cancer Cells. J. Med. Chem. 2001, 44, 1072–1084. [Google Scholar] [CrossRef]
- Kooijman, H.; Spek, A.L.; Batema, G.D.; van Koten, G. CSD Communications Is a Collection of Small Molecule Crystallographic Data Which Has Been Shared by Depositors. 2020. Available online: https://www.ccdc.cam.ac.uk/Community/csd-communications/CSDCommunicationsInformation/ (accessed on 24 March 2021).
- Gauthier, S.; Mailhot, J.; Labrie, J. New Highly Stereoselective Synthesis of (Z)-4-Hydroxytamoxifen and (Z)-4-Hydroxytoremifen via McMurry Reaction. J. Org. Chem. 1996, 61, 3890–3893. [Google Scholar] [CrossRef]
- Nikitin, K.; Ortin, Y.; Müller-Bunz, H.; Plamont, M.-A.; Jaouen, G.; Vessières, A.; McGlinchey, M.J. Organometallic SERMs (selective estrogen receptor modulators): Cobaltifens, the (cyclobutadiene)cobalt analogues of hydroxytamoxifen. J. Organometal. Chem. 2010, 695, 595–608. [Google Scholar] [CrossRef]
- Slagt, M.Q.; Rodríguez, G.; Grutters, M.M.P.; Klein Gebbink, R.J.M.; Klopper, W.; Jenneskens, L.W.; Lutz, M.; Spek, A.L.; van Koten, G. Synthesis and Properties of para-Substituted NCN-Pincer Palladium and Platinum Complexes. Chem. Eur. J. 2004, 10, 1331–1344. [Google Scholar] [CrossRef]
- Top, S.; Tang, J.; Vessieres, A.; Carrez, D.; Provot, C.; Jaouen, G. Ferrocenyl hydroxytamoxifen: A prototype for a new range of oestradiol receptor site-directed cytotoxics. Chem. Commun. 1996, 955–956. [Google Scholar] [CrossRef]
- Vessières, A.; Top, S.; Beck, W.; Hillard, E.; Jaouen, G. Metal complex SERMs (selective oestrogen receptor modulators). The influence of different metal units on breast cancer cell antiproliferative effects. J. Chem. Soc. Dalton Trans. 2006, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Top, S.; Vessières, A.; Leclercq, G.; Quivy, J.; Tang, J.; Vaissermann, J.; Huche, M.; Jaouen, C. Synthesis, Biochemical Properties and Molecular Modelling Studies of Organometallic Specific Estrogen Receptor Modulators (SERMs), the Ferrocifens and Hydroxyferrocifens: Evidence for an Antiproliferative Effect of Hydroxyferrocifens on both Hormone-Dependent and Hormone-Independent Breast Cancer Cell Lines. Chem. Eur. J. 2003, 9, 5223–5236. [Google Scholar] [CrossRef]
- Jaouen, G.; Top, S.; Vessières, A.; Alberto, R. New paradigms for synthetic pathways inspired by bioorganometallic chemistry. J. Organomet. Chem. 2000, 600, 23–36. [Google Scholar] [CrossRef]
- Fus, F.; Yang, Y.; Lee, H.Z.; Top, S.; Carriere, M.; Bouron, A.; Pacureanu, A.; da Silva, J.C.; Salmain, M.; Vessières, A.; et al. Intracellular Localization of an Osmocenyl-Tamoxifen Derivative in Breast Cancer Cells Revealed by Synchrotron Radiation X-ray Fluorescence Nanoimaging. Angew. Chem. Int. Ed. Engl. 2019, 58, 3461–3465. [Google Scholar] [CrossRef]
- Dhingra, K. Antiestrogens–Tamoxifen, SERMs and Beyond. Investig. New Drugs 1999, 17, 285–311. [Google Scholar] [CrossRef]
- Top, S.; Vessières, A.; Cabestaing, C.; Laios, I.; Leclercq, G.; Provot, C.; Jaouen, G. Studies on organometallic selective estrogen receptor modulators. (SERMs) Dual activity in the hydroxy-ferrocifen series. J. Organomet. Chem. 2001, 637, 500–506. [Google Scholar] [CrossRef]
- Hillard, E.; Vessières, A.; Thouin, G.; Jaouen, G.; Amotore, C. Ferrocene-Mediated Proton-Coupled Electron Transfer in a Series of Ferrocifen-Type Breast-Cancer Drug Candidates. Angew. Chem. Int. Ed. Eng. 2006, 45, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Fish, R.H.; Jaouen, G. Bioorganometallic Chemistry: Structural Diversity of Organometallic Complexes with Bioligands and Molecular Recognition Studies of Several Supramolecular Hosts with Biomolecules, Alkali-Metal Ions, and Organometallic Pharmaceuticals. Organometallics 2003, 22, 2166–2177. [Google Scholar] [CrossRef]
- Precigoux, G.; Courseille, C.; Geoffre, S. Hospital, [p-(Diméthylamino-2 éthoxy)phényl]-1 trans-Diphenyl-1,2 Butène-1 (Tamoxifène) (ICI-46474). Acta Crystallogr. Sect. B Struct. Sci. 1979, 35, 3070–3072. [Google Scholar] [CrossRef]
- Dyson, P.J.; Sava, G. Metal-based antitumour drugs in the post genomic era. J. Chem. Soc. Dalton Trans. 2006, 1929–1933. [Google Scholar] [CrossRef]
- Wong, E.; Giandomenico, C.M. Current Status of Platinum-Based Antitumor Drugs. Chem. Rev. 1999, 99, 2451–2466. [Google Scholar] [CrossRef]
- Rosenberg, B.; van Camp, L.; Krigas, T. Inhibition of Cell Division in Escherichia coli by Electrolysis Products from a Platinum Electrode. Nature 1965, 205, 698–699. [Google Scholar] [CrossRef] [PubMed]
- Alderden, R.A.; Hall, M.D.; Hambley, T.W. The Discovery and Development of Cisplatin. J. Chem. Educ. 2006, 83, 728–734. [Google Scholar] [CrossRef]
- Van Zutphen, S.; Reedijk, J. Targeting platinum anti-tumour drugs: Overview of strategies employed to reduce systemic toxicity. Coord. Chem. Rev. 2005, 249, 2845–2853. [Google Scholar] [CrossRef]
- Davies, P.J.; Veldman, N.; Grove, D.M.; Spek, A.L.; Lutz, B.T.G.; van Koten, G. Organoplatinum Building Blocks for one-Dimensional Hydrogen-Bonded Polymeric structures. Angew. Chem. Int. Ed. 1996, 35, 1959–1961. [Google Scholar] [CrossRef]
- Chuchuryukin, A.V.; Chase, P.A.; Mills, A.M.; Lutz, M.; Spek, A.L.; van Klink, G.P.M.; van Koten, G. Hydroxy- and Mercaptopyridine Pincer Platinum and Palladium Complexes Generated by Silver-Free Halide Abstraction. Inorg. Chem. 2006, 45, 2045–2054. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, B.; Snelders, D.J.M.; Dijkstra, H.P.; Versluis, K.; Lutz, M.; Spek, A.L.; Egmond, M.R.; Klein Gebbink, R.J.M.; van Koten, G. Coordination Chemistry in Water of a Free and a Lipase-Embedded Cationic NCN-Pincer Platinum Center with Neutral and Ionic Triarylphosphines. Organometallics 2012, 31, 2810–2820. [Google Scholar] [CrossRef]
- Duisenberg, A.J.M.; Kroon-Batenburg, L.M.J.; Schreurs, A.M.M. An intensity evaluation method: EVAL-14. J. Appl. Cryst. 2003, 36, 220–229. [Google Scholar] [CrossRef]
- Spek, A.L. Structure validation in chemical crystallography. Acta Cryst. 2009, D65, 148–155. [Google Scholar] [CrossRef]
- Blessing, R.H. Data Reduction and Error Analysis for Accurate Single Crystal Diffraction Intensities. Crystallogr. Rev. 1987, 1, 3–58. [Google Scholar] [CrossRef]
- Beurskens, P.T.; Admiraal, G.; Beurskens, G.; Bosman, W.P.; Garcia-Granda, S.; Gould, R.O.; Smits, J.M.M.; Smykalla, C. The DIRDIF99 Program System; Technical Report of the Crystallography; Laboratory at University of Nijmegen, University of Nijmegen: Nijmegen, The Netherlands, 1999. [Google Scholar]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. 2015, C71, 3–8. [Google Scholar]
- Spek, A.L. PLATON SQUEEZE: A tool for the calculation of the disordered solvent contribution to the calculated structure factors. Acta Cryst. 2015, C71, 9–18. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).