Abstract
A number of pyrrolo[2,3-c]pyridines, pyrrolo[3,2-d]pyrimidines and pyrazolo[4,3-d]pyrimidines were designed and synthesized as antiproliferative agents. The target compounds possessed selected substituents in analogous positions on the central scaffold that allowed the extraction of interesting SARs. The cytotoxic activity of the new derivatives was evaluated against prostatic (PC-3) and colon (HCT116) cell lines, and the most potent analogues showed IC50 values in the nM to low µM range, while they were found to be non-toxic against normal human fibroblasts (WI-38). Flow cytometric analysis of DNA content revealed that the most promising derivative 14b caused a statistically significant accumulation of PC-3 cells at G2/M phase and induced apoptosis in PC-3 cells.
1. Introduction
Azaindole and related nitrogen-containing fused heterocycles, presenting structural analogy to adenine base of DNA, are important building blocks of bioactive compounds, and they have, therefore, been extensively investigated in the field of medicinal chemistry [1,2,3]. Among this structurally diverse class, many pyrazolopyridine and pyrrolopyrimidine derivatives were found to display potent in vitro and in vivo activity, as receptor modifiers or enzyme inhibitors [4,5], possessing interesting antiproliferative [6,7,8], antibacterial [9,10], antiviral [11,12], antimalarial [13], antileishmanial [14], antidiabetic [15], trypanocidal [16] and neuroprotective properties [17]. The exact heterocyclic isomer, depending on the structural arrangement of nitrogen atoms in the central purine-like ring system, along with the introduction of selected substituents, is important for the interaction of each compound with specific cellular substrates. The classical bioisosteric replacement of a CH group with a N atom (vide infra) in heteroaromatic rings can potentially affect a number of the physicochemical parameters of the central scaffold, as well as its intra- and intermolecular orbital, steric, electrostatic and hydrophobic interactions, which can translate into improved pharmacological profiles. This minor structural modification is usually challenging from a synthetic point of view, but it provides an excellent opportunity for lead optimization. [18] Although the antitumor activity of these compounds can be attributed to a broad range of molecular targets and several mechanisms, the more interesting targets and mechanisms are certainly related to the inhibition of receptor or cytoplasmic protein kinases, which are linked to signal transduction networks associated with carcinogenesis, progression and metastasis of a variety of aggressive human cancers [19]. Purine isosteres usually compete successfully with ATP for interaction with the hinge region of the binding pocket of these enzymes and, therefore, are estimated to be non-selective kinase inhibitors. However, a reasonable degree of selectivity can be achieved as a consequence of the introduction of a variety of substituents, mainly on the respective 2-, 6-and 9-positions of the purine nucleus. For example, the pyrrolopyridine derivative vemurafenib is a mutant specific kinase inhibitor currently used for treatment of metastatic BRAFV600E melanoma [20]. Extensive lead optimization studies are essential in each case to enhance interactions with binding pockets, neighboring the hinge region, in order to improve potency and target selectivity. Nevertheless, the great number of protein kinases, the complexity of cellular signal transduction pathways and the possibility of interactions with additional cellular substrates mean that experimental studies aimed at the clarification of the precise pharmacological function of individual derivatives are challenging.
On the other hand, there is an increasing need for the development of novel, safer and tolerable chemotherapeutic agents for more effective cancer treatment. The identification and targeting of the molecular drivers of cancer remain a major scientific subject and contribute towards the discovery of new compounds, able to inhibit uncontrolled cancer cell proliferation, with low toxicity to normal cells and tissues. These targeted therapies are considered a promising approach for the treatment of human malignancies and have already been proven effective in the clinic, providing impressive—though transitory—responses to patients with advanced cancers. They are well-tolerated drugs, characterized by few off-target effects, low toxicity profiles and fairly broad therapeutic windows. However, the development of therapeutic strategies to improve the long-term effectiveness and limit the emerging resistance in targeted therapies remain a vital challenge in this field. [21].
The starting point for the present study was an attractive pyrazolopyridine hit, namely 3-phenyl-7-(3,4,5-trimethoxyphenyl)aminopyrazolo[3,4-c]pyridine, previously identified by our group [22]. This compound was endowed with strong cytotoxic properties against a variety of cancer cell lines, with IC50 values in the medium to high nM range. In the course of our involvement in the discovery of new purine analogues with antiproliferative activity [23,24,25], we considered it interesting to modify the main scaffold of the above-mentioned derivative and insert suitable substituents in order to gain insight into the SARs of this class of compounds. More precisely, we have replaced the pyrazolopyridine central scaffold by three purine-like heterocycles, namely pyrrolo[2,3-c]pyridine, pyrrolo[3,2-d]pyrimidine and pyrazolo[4,3-d]pyrimidine. Besides the 7-(3,4,5-trimethoxyphenyl)- substituent of the lead compound, we have also introduced a phenylamino- and a 4-methylpiperazin-1-yl substituent in the corresponding position of the above-mentioned scaffolds. We preserved the lead compound’s 3-phenyl group, and we also prepared the corresponding 3-(3-fluorophenyl)- analogues. We, thus, present herein the synthesis of these series of derivatives and the evaluation of their biological activity.
2. Results and Discussion
2.1. Chemistry
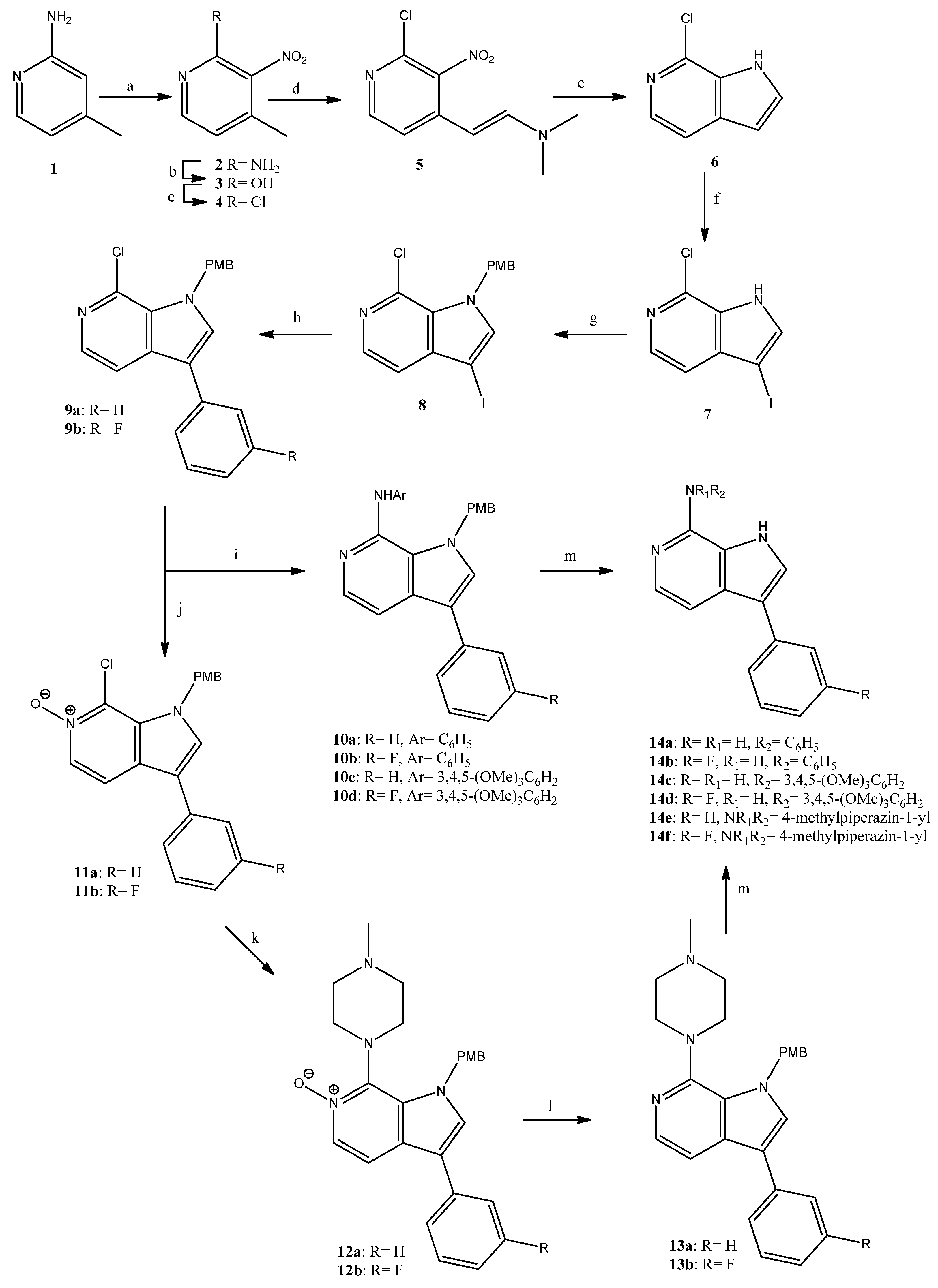
For the preparation of the series of pyrrolopyridines, we used the intermediate chlorides 9a,b, which were synthesized as depicted in Scheme 1. 2-Aminopicoline (1) was successively nitrated, diazotized and chlorinated to provide the nitropyridine 4 [26]. The reaction of 4 with DMF-dimethylacetal provided the enamine 5 that upon reduction was cyclized to result in the 7-chloropyrrolopyridine 6 [27] in 25% overall yield. The latter was iodinated, the pyrrole nitrogen was protected upon treatment with 4-methoxybenzylchloride (PMB-Cl) and the resulting iodide 8 underwent a Suzuki-type coupling with suitable boronic acids to give the chlorides 9a,b. All these reactions proceeded in high yields, and compounds were rationally characterized via 1H-NMR data. It is of interest to notice the characteristic chemical shift of the iodine bearing carbon (C-3) of compounds 7 and 8, which resonated at 56 ppm in the corresponding 13C-NMR spectra. The chlorides 9a,b were then treated with aniline, or 3,4,5-trimethoxyaniline in the presence of potassium tert-butoxide, tris(dibenzylideneacetone)dipalladium(0) [Pd2(dba)3] and 2,2′-bis(diphenylphosphino)-1,1′-dinapthalene (BINAP), to result in the amines 10a–d in good yields (60–91%). In parallel, since the nucleophilic substitution of the 7-chloro group of compounds 9 by alkylamines upon heating in DMF or DMSO was not successful, 9a,b were treated with m-chloroperoxybenzoic acid and were converted in good yield (88%) to the corresponding N-oxides 11a,b. These derivatives underwent nucleophilic substitution by N-methylpiperazine to provide the aminosubstituted compounds 12a,b in moderate yield (50%), which upon reduction gave the intermediates 13a,b. An interesting and indicative feature in the 13C-NMR spectral data of the N-oxides 11a,b and 12a,b was the chemical shift of C-5, which appears upfield, at 133–134 ppm, when compared to the corresponding peak of compounds 9a,b and 13a,b (C-5 resonance in this case is 137–138 ppm); this was obviously due to the presence of the neighboring N-oxide. Finally, the N1-substituted derivatives 10a–d and 13a,b were efficiently deprotected to result in the target pyrrolopyridines 14a–f in good to quantitative yields. In the 1H-NMR spectra of compounds 14a–f, the pyrrole H was detectable as a broad singlet at 10.5–11.8 ppm, whereas the NH of the 7-substituent of 14a–d was detected upfield at 8.6–9.4 ppm.
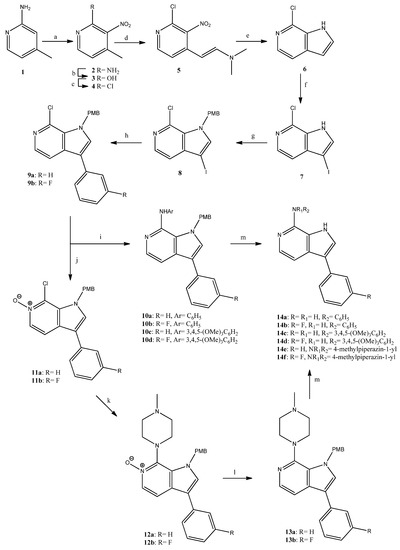
Scheme 1.
Reagents and conditions. a: HNO3, H2SO4, 60 °C, 4 h, 44%; b: NaNO2, H2SO4, rt, 2 h, 90%; c: POCl3, reflux, 4 h, 94%; d: DMF-DMA, DMF, 120 °C, 2 h, 80%; e: Fe, AcOH, AcONa, EtOH/H2O, reflux, 2 h, 82%; f: NIS, MeOH, rt, 1 h, 99%; g: NaH, 4-methoxybenzylchloride (PMB-Cl), DMF, rt, 90 min, 99%; h: phenylboronic acid or 3-fluorophenylboronic acid, K2CO3, tetrakis(triphenylphosphine)palladium, toluene/EtOH, reflux, 3 h, 88–98%; i: aniline or 3,4,5-trimethoxyaniline, Pd2(dba)3, BINAP, t-BuOK, toluene, reflux, 3.5 h, 60–91%; j: m-chloroperoxybenzoic acid, CH2Cl2, rt, 72 h, 88%; k: 1-methylpiperazine, EtOH, autoclave, 120 °C, 24 h, 50%; l: PCl3, CHCl3, rt, 20 h, 72–99%; m: CF3CO2H, 60 °C, 20 h, 91–99%.
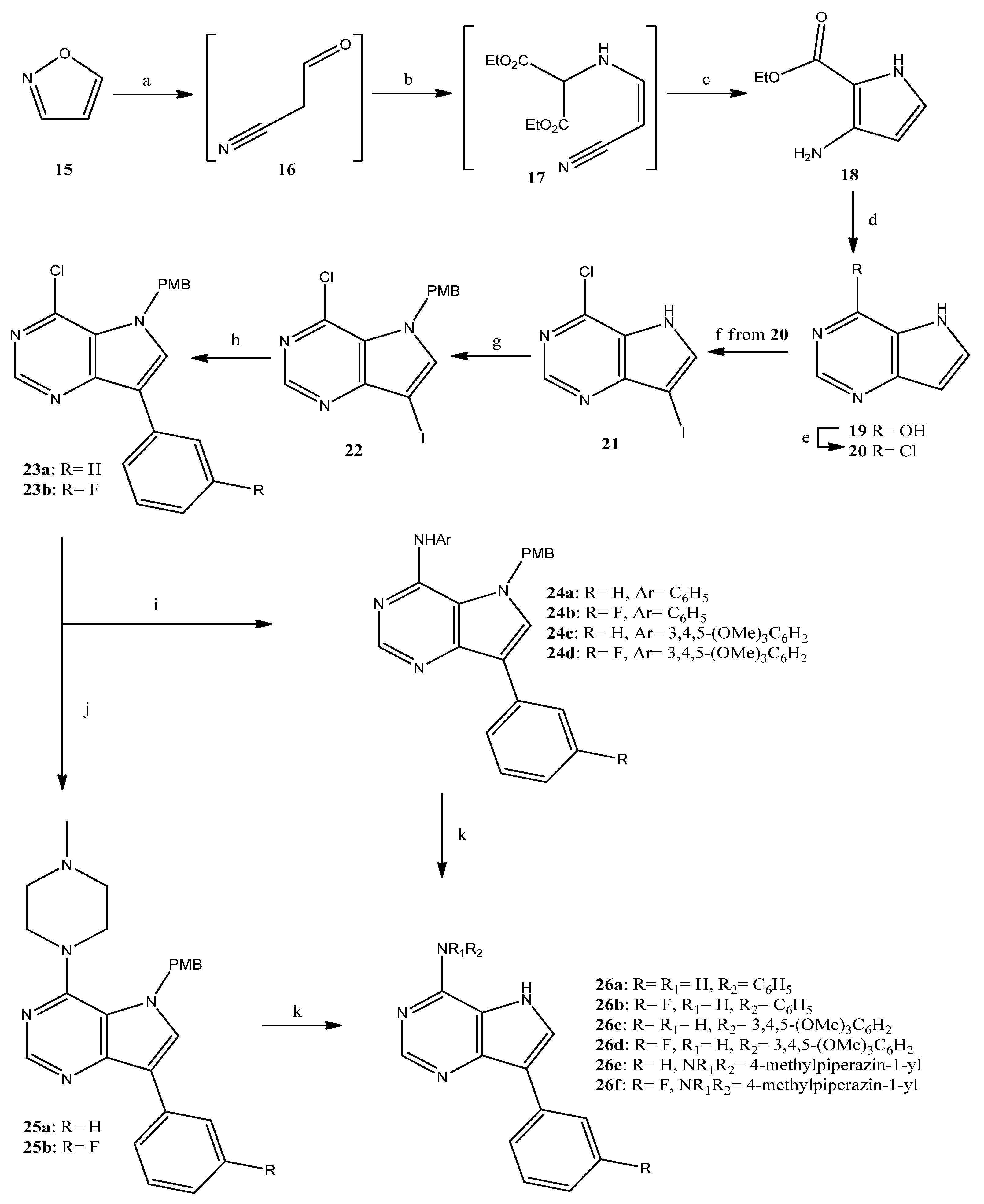
The pyrrolopyrimidine analogues were prepared according to the reaction sequence presented in Scheme 2 from isoxazole (15), which—following a previously reported procedure—was converted to the pyrrolecarboxylate 18 [28]. The latter was, in turn, ring-closed; the resulting pyrimidinone 19 [29] was converted to the chloride 20 [30], which was successively iodinated to give 21 [31]. The iodide 21 was then protected to provide almost quantitatively 22, which was introduced to a Suzuki-type coupling, resulting in high yields of the 3-arylsubstituted chlorides 23a,b. The above-mentioned chlorides were used for the synthesis of the target pyrrolopyrimidines 26a–f, obtained in very good overall yields, following an analogous procedure to that reported for the corresponding pyrrolopyridines. A common pattern was obvious at the 1H-NMR spectra of 26a–f, concerning the pyrrole NH that appeared as a broad singlet at 10.7–11.1 ppm and the NH of the 4-substituent of compounds 26a–d (8.5–8.7 ppm), as well as the characteristic chemical shifts of H-2 (8.4–8.5 ppm) and H-6 (7.9–8.1 ppm).
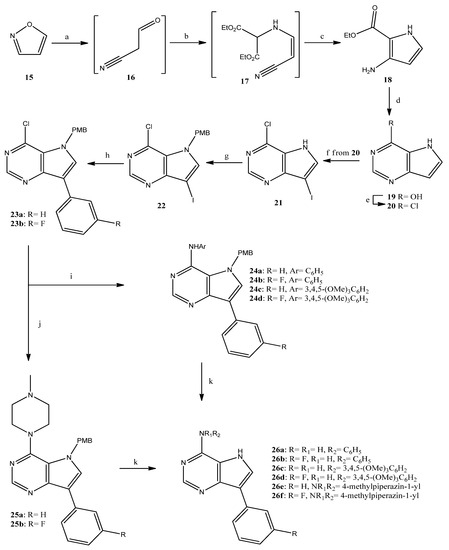
Scheme 2.
Reagents and conditions. a: EtONa, EtOH, rt, 30 min; b: ethyl aminomalonate hydrochloride, AcOH, AcONa, rt, 48 h; c: EtONa, EtOH, rt, 5 days, 37% for 3 steps; d: formamidine acetate, EtOH, reflux, 20 h, 82%; e: POCl3, reflux, 90 min, 88%; f: NIS, MeOH, rt, 1 h, 76%; g: NaH, 4-methoxybenzylchloride (PMB-Cl), DMF, rt, 90 min, 99%; h: phenylboronic acid or 3-fluorophenylboronic acid, K2CO3, tetrakis(triphenylphosphine)palladium, toluene/EtOH, reflux, 3 h, 90–94%; i: aniline or 3,4,5-trimethoxyaniline, Pd2(dba)3, BINAP, t-BuOK, toluene, reflux, 5 h, 60–87%; j: 1-methylpiperazine, DMSO, 120 °C, 20 h, 97–99%; k: CF3CO2H, 60 °C, 20 h, 91–99%.
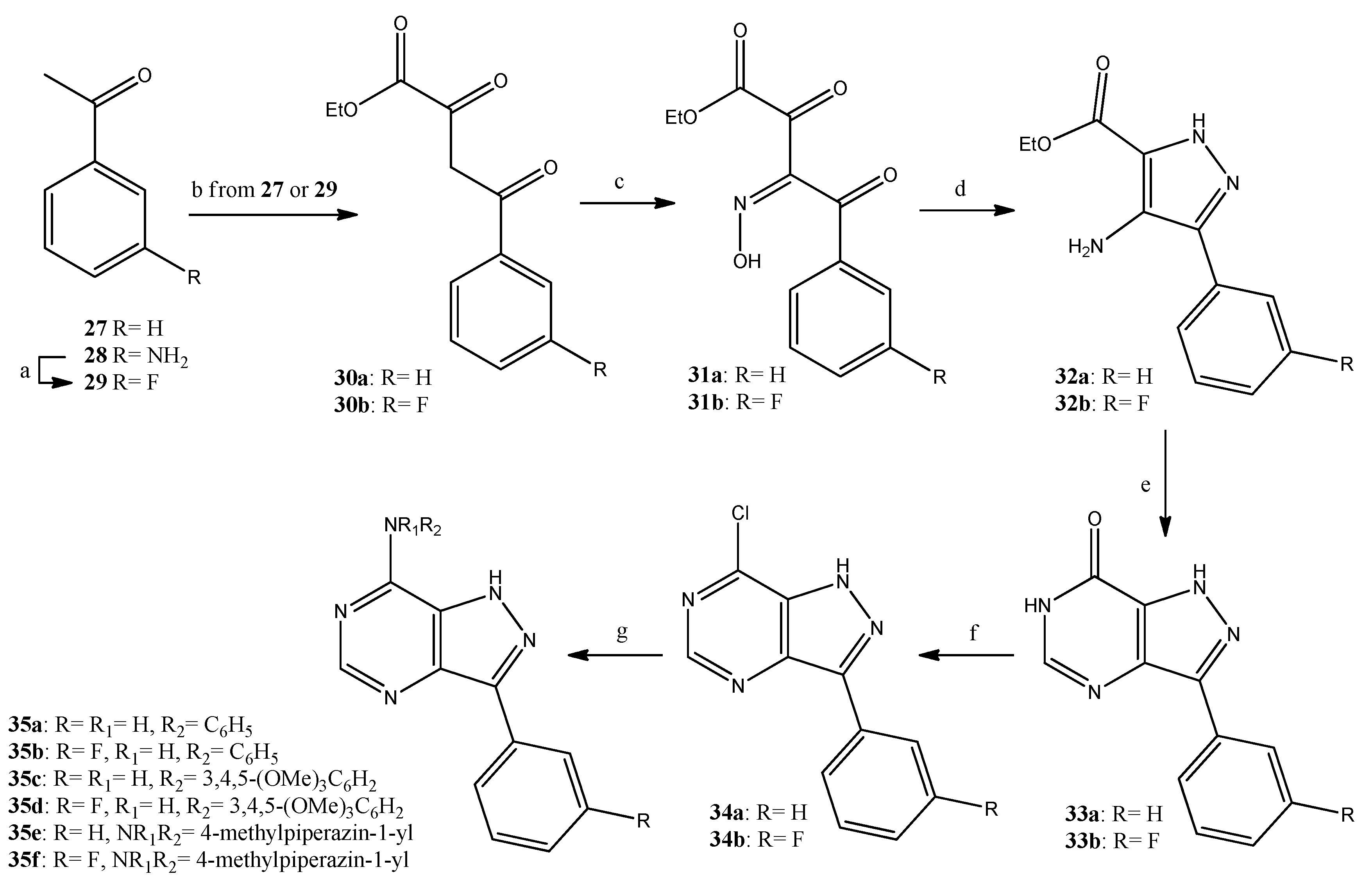
Finally, the pyrazolopyrimidine analogues were synthesized, as shown in Scheme 3, starting from the acetophenones 27 (commercially available) or 29 [32]. The acetophenones were subjected to side chain elongation upon reaction with ethyl oxalate, and the resulting dioxobutyrates 30a [33] and 30b [34] were converted in good yields (85%) to the corresponding α-(hydroxyimino)benzopyruvates 31a [35] and 31b, respectively. Compounds 31a,b were treated with hydrazine hydrate and were ring-closed with the concomitant reduction of the oxime group to result in the aminopyrazole-carboxylates 32a [36] and 32b, in moderate yields (40%). The reaction of the carboxylates 32a,b with formamidine gave the pyrazolopyrimidinones 33a [37] and 33b, which—through the corresponding chlorides 34a,b and the displacement of the chlorine by suitable amines—resulted in the target pyrazolopyrimidines 35a–f in good overall yields (over 40% for three steps). All new compounds were unambiguously identified using NMR and mass spectrometry data.
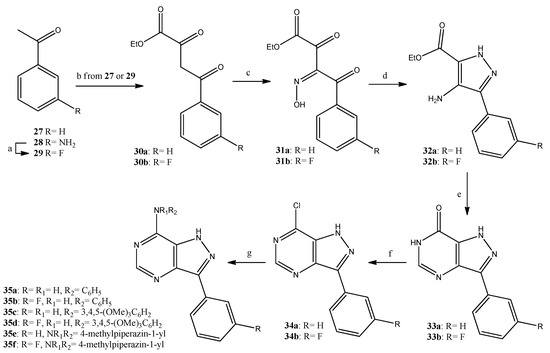
Scheme 3.
Reagents and conditions. a: BF3.(Et2O)2, isoamyl nitrite, 1,2-dichlorobenzene, 90 °C, 45 min, 54%; b: NaH, diethyl oxalate, toluene, 50 °C, 1 h, 99%; c: NaNO2, AcOH, 10 °C, 1 h, 85%; d: hydrazine hydrate, EtOH, 45 °C, 2 h, 40%; e: formamidine acetate, BuOH, reflux, 20 h, 61%; f: POCl3, PCl5, reflux, 3 h, 80%; g: aniline or 3,4,5-trimethoxyaniline or 1-methylpiperazine, EtOH, reflux, 2 h, 57–80%.
2.2. Biological Evaluation
The cytotoxic activity of the new compounds was tested against two cancer cell lines, prostatic (PC-3) and colon (HCT116) cell lines (Table 1), while the most potent derivatives were further tested against normal human fibroblasts (WI-38) (Table 2).

Table 1.
Accumulative results of the antiproliferative activities of all compounds tested.

Table 2.
IC50 values of the most potent compounds in human fibroblasts.
Concerning the N-(p-methoxybenzyl) substituted pyrrolopyridines 10a–d and 13a,b, only a few of them possessed moderate antiproliferative activity, namely the 7-phenylamino derivative 10a, together with both the 7-(N-methylpiperazine) substituted derivatives 13a,b, with IC50 values in the range of 2.4–9.3 µM. On the other hand, the deprotection of the above-mentioned compounds provided derivatives 14a–f, which were endowed with very potent cytotoxic properties, with IC50 values in the nM to low µM range against both cell lines tested. The most potent compound of this group is the 3-(3-fluorophenyl) analogue 14b, possessing IC50 values of 55nM and 90 nM against the prostate and colon cell lines, respectively.
In the pyrrolopyrimidine series, the N-(p-methoxybenzyl) substituted analogues 44a–d and 25a,b are, in analogy to the previous series, devoid of activity, with the sole exception of 24c, which appeared to be cytotoxic against PC-3 cells (IC50: 2.25 µM). Concerning the corresponding deprotected analogues, the 7-(3-fluorophenyl)-4-phenylamino derivative 26b is the most potent compound of this group, followed by the corresponding 7-phenyl-substituted compound 26a. The 4-(3,4,5-trimethoxyphenyl)amino-substituted derivatives 26c,d had moderate activity, while a complete loss of activity was observed in the corresponding 4-(N-methylpiperazine)-substituted analogues.
Finally, in the case of pyrazolopyrimidines, the 3-(3-fluorophenyl)-7-phenylamino derivative 35b possessed very interesting cytotoxicity, whereas among the remaining compounds, only 35a and 35c showed moderate cytotoxicity, and again the N-methylpiperazine substituted analogues were inactive.
Physicochemical characteristics of the thirty compounds tested were calculated using SwissADME platform [38] and are summarized in Table S1. They were further entered as inputs along with the IC50s in the SIMCA software [39]. A multivariate analysis showed that there is a very weak correlation (R2 < 0.25) between activity and molar refractivity, while there was no correlation between activity and logP.
As a general remark, it could be stated that the cytotoxic activity is enhanced in the absence of substituents on the pyrrole or pyrazole nitrogen of the studied compounds and that the pyrrolopyridines possess the most interesting profile, since all six derivatives (14a–f) are potent against both cell lines tested. As already mentioned, 14b is the most active derivative, and at the same time, the analogously substituted pyrrolopyrimidine 26b and pyrazolopyrimidine 35b are also endowed with strong cytotoxic properties. It is of interest to notice that the prostatic cell line appears the most sensitive to the compounds.
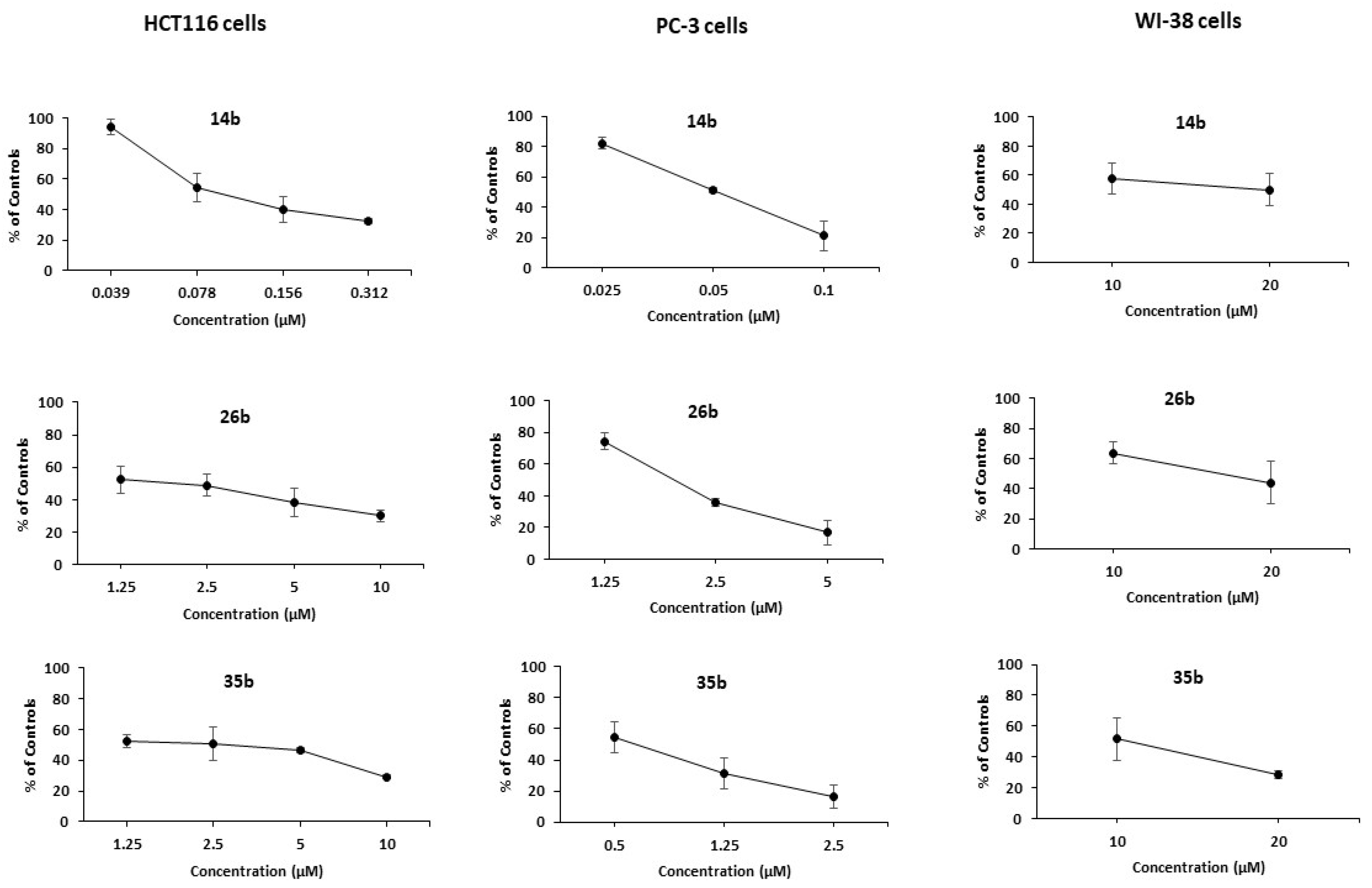
The cytotoxicity of the most potent derivatives, i.e., the majority of the pyrrolopyridines 14, together with 26b and 35b, was then examined towards normal human fibroblasts (WI-38); the results are presented in Figure 1 and Table 2.
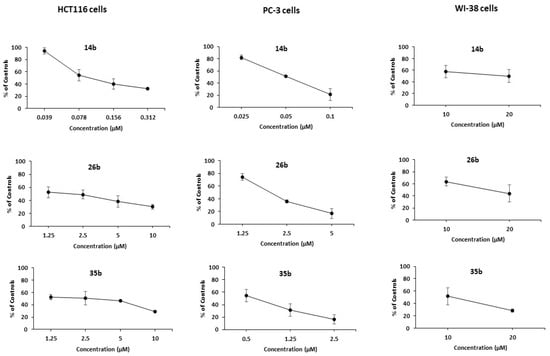
Figure 1.
Dose-response curves for the administration of 14b, 26b, 35b on HCT116, PC-3 and WI-38 cells, as assessed by the MTT assay. Results are expressed as a percentage of controls. Statistical analysis was performed in triplicate determination at p < 0.05.
Among them, 14a and 14c–e appear to be rather cytotoxic to normal cells. By contrast, it is important to note that the 7-phenylamino substituted compound 14b that bears a 3-(3-fluorophenyl)-group, while being highly cytotoxic against both cancer cell lines, appears practically non-toxic against the normal cell line, presenting a selectivity index higher than 200. The derivatives 26b and 35b that possess the same substitution pattern in the central scaffold retain potent activity against the cancer cell lines and appear non-cytotoxic to the normal cell line as well, albeit to a lesser extent than 14b.
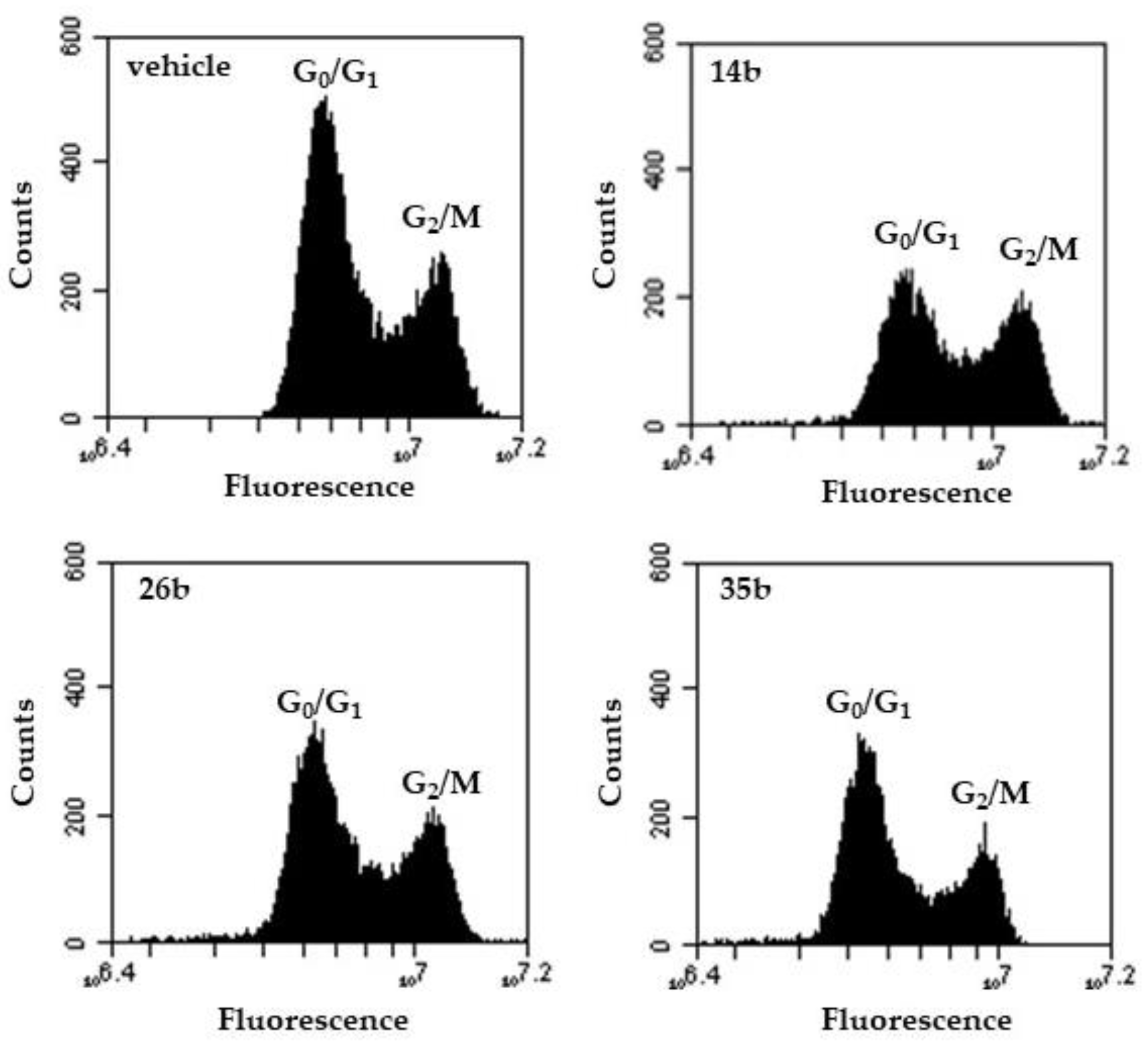
Cell-cycle perturbations induced after the incubation of exponentially growing PC-3 cells with compounds 14b, 26b and 35b for 72 h are given in Figure 2 and Table 3. Compound 14b caused a statistically significant accumulation of PC-3 cells at G2/M phase, significantly reducing, in parallel, the percentage of cells at G0/G1 phase (the accumulation of cells at S phase was marginally non-significant).
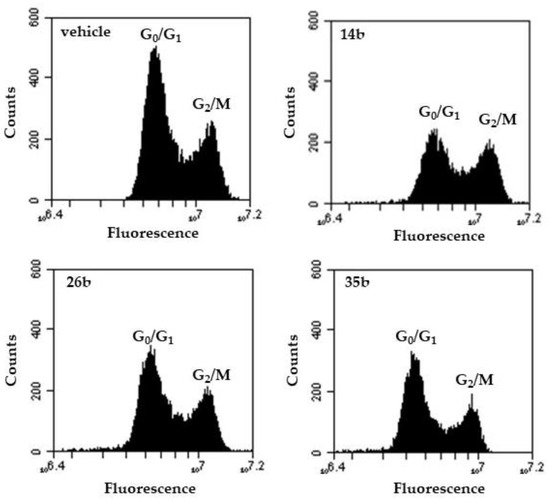
Figure 2.
Flow cytometric graphs of DNA content. Indicative DNA content histograms of PC-3 cells treated for 72 h with 14b, 26b and 35b, in comparison to control cells (vehicle). Quantification of cell cycle phase distribution was based on staining by the DNA binding dye propidium iodide.

Table 3.
Cell cycle phase distribution at 72 h (%).
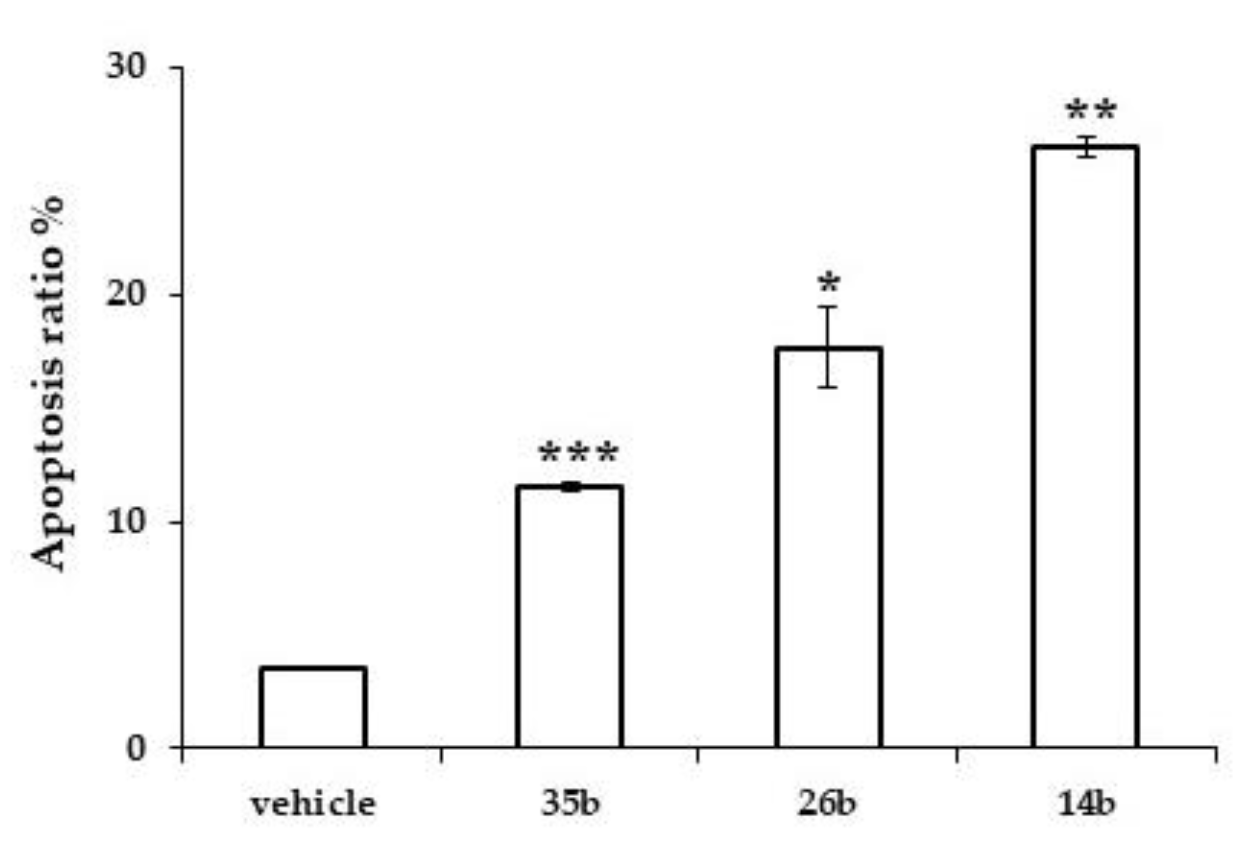
Doxorubicin, which was used as a positive control at a concentration of 25 nM, showed the expected G2/M phase blockade (58%) and the induction of apoptosis (28%), as previously reported [40]. Furthermore, 14b, 26b and 35b induced apoptosis in PC-3 cells after 72 h of treatment, as estimated by AnnexinV-7AAD staining. In particular, compound 14b induced the appearance of 26.5% (±0.47) apoptotic nuclei, 26b induced the appearance of 17.7% (±1.8) apoptotic nuclei and 35b induced the appearance of 11.5% (±0.19) apoptotic nuclei, compared to 3.5% (±0.05) induced by the vehicle (Figure 3). These data give an indication that the mechanism of action of compound 14b may include blocking of the cell cycle at the G2/M phase and inducing apoptosis.
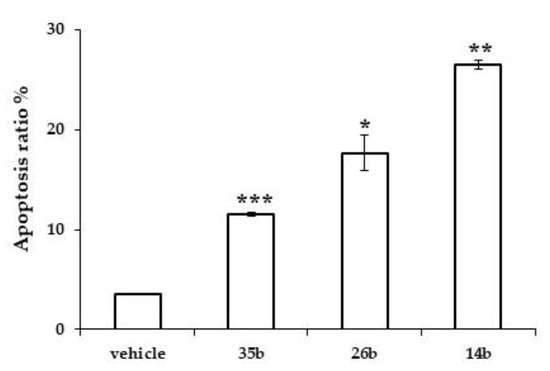
Figure 3.
Induction of apoptosis by the test compounds. Percentage of apoptotic nuclei in PC-3 cells treated for 72 h with the indicated compounds or with vehicle. Bars represent the mean of two independent experiments (error bars: SD); * indicates p < 0.05, ** indicates p < 0.005, *** indicates p < 0.001 (Student’s t-test).
3. Materials and Methods
3.1. General Information
The reagents and solvents were purchased from Sigma-Aldrich Chemical Co. (Darmstadt, Germany) or Fluorochem (Derbyshire’s Peak District, UK). Reagents were used without further purification. Concerning the dry solvents, methanol and dimethylformamide were dried over 3A and 4A molecular sieves, respectively; toluene was pre-dried using CaH2 and then placed over sodium. Hydrazine is considered a suspect carcinogen in humans, may cause serious damage and should be handled carefully. Melting points were determined on a Büchi apparatus (Flawil, Switzerland) and are uncorrected. 1HNMR spectra and 2D spectra were recorded on a Bruker Avance III 600 or a Bruker Avance DRX 400 instrument (Bruker BioSpin, Baden-Württemberg, Germany), whereas 13CNMR spectra were recorded on a Bruker Avance III 600 spectrometer in deuterated solvents and were referenced to TMS (δ scale). The signals of 1H and 13C spectra were unambiguously assigned by using 2D NMR techniques: 1H1H COSY, NOESY, HMQC and HMBC. Mass spectra were recorded with a LTQ Orbitrap Discovery instrument (Thermo Scientific, Dreieich, Germany), possessing an Ionmax ionization source. Flash chromatography was performed on Merck silica gel 60 (0.040–0.063 mm). The purity of all the target compounds that underwent biological evaluation was >95%, as ascertained by elemental analysis. Elemental analyses were undertaken using a PerkinElmer PE 240C elemental analyzer (Norwalk, CT, U.S.) and the measured values for C, H and N were within ± 0.4% of the theoretical values. Analytical thin layer chromatography (TLC) was carried out on precoated (0.25 mm) Merck silica gel F-254 plates (Merck KGa, Darmstadt, Germany). The controlled injection of solutions was performed with the Bioblock Scientific device (Illkirch, France).
3.2. Synthesis
7-Chloro-3-iodo-1H-pyrrolo[2,3-c]pyridine (7): This compound is mentioned in a patent [41]; here, we provide the methodology and identification data. To a solution of 6 (1.40 g, 9.15 mmol) in anhydrous methanol (50 mL), N-iodosuccinimide (2.80 g, 12.4 mmol) was added, and the mixture was stirred at rt for 1 h. Then, the organic solvent was evaporated under vacuo, and the residue was extracted with ethyl acetate. The organic layer was washed with a 10% sodium thiosulfate aqueous solution, dried (Na2SO4) and evaporated to dryness to result in 7 (2.56 g, 99%), m.p. 278–279 °C (CH2Cl2/Et2O). 1H NMR (600 MHz, DMSO-d6) δ 12.56-12.40 (brs, 1H, NH), 8.00 (d, J = 5.4Hz, 1H, H-5), 7.89 (s, 1H, H-2), 7.31 (d, J = 5.4 Hz, 1H, H-4). 13C NMR (151 MHz, DMSO-d6) δ 137.95 (C-5), 136.50 (C-3a), 134.66 (C-2), 133.72 (C-7), 129.68 (C-7a), 114.63 (C-4), 56.54 (C-3). HR-MS (ESI) m/z: Calcd for C7H5ClIN2: [M + H]+ = 278.9181, found 278.9193.
7-Chloro-3-iodo-1-(4-methoxybenzyl)-1H-pyrrolo[2,3-c]pyridine (8): To a solution of 7 (2.40 g, 8.62 mmol) in anhydrous N,N-dimethylformamide (10 mL), sodium hydride (300 mg, 60% in hexanes) was added under cooling in an argon atmosphere, and the resulting solution was stirred at rt for 15 min. Then, 4-methoxybenzyl chloride (1.40 mL, 10.4 mmol) was added, and the solution was stirred at rt for 90 min. The solvent was vacuum-evaporated, ice-water was added to the residue and the precipitate was filtered and air-dried to provide pure 8 (3.40 g, 99%), m.p. 98–99 °C (CH2Cl2/Et2O). 1H NMR (600 MHz, DMSO-d6) δ 8.06 (s, 1H, H-2), 8.02 (d, J = 5.3 Hz, 1H, H-5), 7.34 (d, J = 5.3 Hz, 1H, H-4), 7.04 (d, J = 8.6 Hz, 2H, methoxybenzyl H-2,6), 6.87 (d, J = 8.6 Hz, 2H, methoxybenzyl H-3,5), 5.72 (s, 2H, CH2), 3.69 (s, 3H, CH3O). 13C NMR (151 MHz, DMSO-d6) δ 158.63 (methoxybenzyl C-4), 139.37 (C-2), 138.56 (C-5), 138.22 (C-3a), 132.76 (C-7), 130.14 (methoxybenzyl C-1), 128.39 (C-7a), 127.66 (methoxybenzyl C-2,6), 115.34 (C-4), 114.08 (methoxybenzyl C-3,5), 56.36 (C-3), 55.03 (CH3O), 50.60 (CH2). HR-MS (ESI) m/z: Calcd for C15H13ClIN2O: [M + H]+ = 398.9756, found 398.9766.
7-Chloro-1-(4-methoxybenzyl)-3-phenyl-1H-pyrrolo[2,3-c]pyridine (9a): To a solution of 8 (1.00 g, 2.51 mmol) in toluene (40 mL) and absolute ethanol (5 mL), phenylboronic acid (360 mg, 2.95 mmol), potassium carbonate (700 mg, 5.07 mmol) and tetrakis(triphenylphosphine)palladium (70 mg, 60.6 μmol) were added sequentially, and the solution was heated at reflux for 3 h. The solvents were evaporated under vacuo, water was added to the residue and it was extracted with ethyl acetate. The organic layer was dried (Na2SO4) and concentrated to dryness, and the residue was purified by silica gel column chromatography (cyclohexane/ethyl acetate: 4/1) to give 9a (770 mg, 88%), m.p. 85–86 °C (CHCl3/n-hexane). 1H NMR (600 MHz, CDCl3) δ 8.08 (d, J = 5.5 Hz, 1H, H-5), 7.78 (d, J = 5.5 Hz, 1H, H-4), 7.55 (d, J = 7.4 Hz, 2H, phenyl H-2,6), 7.47-7.44 (m, 3H, H-2, phenyl H-3,5), 7.34 (t, J = 7.4 Hz, 1H, phenyl H-4), 7.11 (d, J = 8.7 Hz, 2H, methoxybenzyl H-2,6), 6.87 (d, J = 8.7 Hz, 2H, methoxybenzyl H-3,5), 5.78 (s, 2H, CH2), 3.78 (s, 3H, CH3O). 13C NMR (151 MHz, CDCl3) δ 159.62 (methoxybenzyl C-4), 137.03 (C-5), 135.24 (C-7), 133.57 (phenyl C-1), 133.25 (C-3a), 132.74 (C-2), 129.42 (methoxybenzyl C-1), 129.21 (phenyl C-3,5), 129.13 (C-7a), 128.45 (methoxybenzyl C-2,6), 127.73 (phenyl C-2,6), 127.23 (phenyl C-4), 118.25 (C-3), 114.57 (methoxybenzyl C-3,5), 114.55 (C-4), 55.46 (CH3O), 51.80 (CH2). HR-MS (ESI) m/z: Calcd for C21H18ClN2O: [M + H]+ = 349.1103, found 349.1115.
7-Chloro-3-(3-fluorophenyl)-1-(4-methoxybenzyl)-1H-pyrrolo[2,3-c]pyridine (9b): This compound was synthesized using a procedure analogous to that of 9a, starting from 8. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 7/3). Oil, yield: 98%. 1H NMR (600 MHz, CDCl3) δ 8.11 (d, J = 5.5 Hz, 1H, H-5), 7.75 (d, J = 5.5 Hz, 1H, H-4), 7.45-7.41 (m, 2H, H-2, fluorophenyl H-5), 7.38-7.34 (m, 1H, fluorophenyl H-6), 7.29-7.26 (m, 1H, fluorophenyl H-2), 7.14 (d, J = 8.9 Hz, 2H, methoxybenzyl H-2,6), 7.06-7.01 (m, 1H, fluorophenyl H-4), 6.90 (d, J= 8.9 Hz, 2H, methoxybenzyl H-3,5), 5.80 (s, 2H, CH2), 3.82 (s, 3H, CH3O). 13C NMR (151 MHz, CDCl3) δ 164.20, 162.57 (fluorophenyl C-3), 159.55 (methoxybenzyl C-4), 138.61 (C-5), 134.48, 134.41 (fluorophenyl C-1), 134.24 (C-7), 131.72 (C-2), 130.64, 130.58 (fluorophenyl C-5), 129.64 (C-7a), 129.37 (methoxybenzyl C-1), 128.55 (C-3a), 128.44 (methoxybenzyl C-2,6), 123.20 (fluorophenyl C-6), 116.70 (C-3), 114.53 (methoxybenzyl C-3,5), 114.43, 114.29 (fluorophenyl C-2), 114.15 (C-4), 113.78, 113.64 (fluorophenyl C-4), 55.44 (CH3O), 51.68 (CH2). HR-MS (ESI) m/z: Calcd for C21H17ClFN2O: [M + H]+ = 367.1008, found 367.0995.
1-(4-Methoxybenzyl)-Ν,3-diphenyl-1H-pyrrolo[2,3-c]pyridin-7-amine (10a): To a solution of 9a (120 mg, 0.34 mmol) in anhydrous toluene (2 mL), tris(dibenzylideneacetone)dipalladium(0) (8.0 mg, 8.7 μmol), 2,2′-bis(diphenylphosphino)-1,1′-dinapthalene (16 mg, 0.26 mmol), potassium tert-butoxide (60 mg, 0.50 mmol) and aniline (46 μL, 0.50 mmol) were added sequentially, and the solution was heated at reflux for 3.5 h. It was then extracted with ethyl acetate,the organic layer was dried (Na2SO4) and concentrated to dryness, and the residue was purified by silica gel column chromatography (cyclohexane/ethyl acetate: 1/1) to give 10a in 87% yield, m.p. 180–181 °C (EtOAc/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 7.87 (d, J = 5.3 Hz, 1H, H-5), 7.81 (s, 1H, H-2), 7.70 (d, J = 7.5 Hz, 2H, 3-phenyl H-2,6), 7.49-7.44 (m, 3H, H-4, 3-phenyl H-3,5), 7.29 (t, J = 7.4 Hz, 1H, 3-phenyl H-4), 7.25-7.18 (m, 6H, N7-phenyl H-2,6, methoxybenzyl H-2,6, N7-phenyl H-3,5), 7.16 (brs, 1H, ΝH), 6.92 (d, J = 8.7 Hz, 2H, methoxybenzyl H-3,5), 6.87 (t, J = 7.0 Hz, 1H, N7-phenyl H-4), 5.69 (s, 2H, CH2), 3.75 (s, 3H, CH3O). 13C NMR (151 MHz, Acetone-d6) δ 160.62 (methoxybenzyl C-4), 144.01 (N7-phenyl C-1), 143.60 (C-7), 138.19 (C-5), 135.83 (3-phenyl C-1), 134.39 (C-3a), 131.95 (C-2), 131.25 (methoxybenzyl C-1), 129.81 (3-phenyl C-3,5), 129.52 (N7-phenyl C-3,5), 128.94 (methoxybenzyl C-2,6), 128.02 (3-phenyl C-2,6), 126.99 (3-phenyl C-4), 124.93 (C-7a), 121.51 (N7-phenyl C-4), 119.09 (N7-phenyl C-2,6), 117.51 (C-3), 115.41 (methoxybenzyl C-3,5), 110.05 (C-4), 55.68 (CH3O), 52.53 (CH2). HR-MS (ESI) m/z: Calcd for C27H24N3O: [M + H]+ = 406.1914, found 406.1900. Anal. Calcd for C27H23N3O: C, 79.97; H, 5.72; N, 10.36. Found: C, 80.06; H, 5.77; N, 10.21.
3-(3-Fluorophenyl)-1-(4-methoxybenzyl)-Ν-phenyl-1H-pyrrolo[2,3-c]pyridin-7-amine (10b): This compound was synthesized using a procedure analogous to that of 10a, starting from 9b. Purification was carried out by silica gel column chromatography (dichloromethane/ethyl acetate: 9/1). Yield: 91%, m.p. 165–166 °C (CH2Cl2/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 7.91 (s, 1H, H-2), 7.89 (d, J = 5.4 Hz, 1H, H-5), 7.58-7.55 (m, 1H, fluorophenyl H-6), 7.51-7.48 (m, 2H, H-4, fluorophenyl H-5), 7.47-7.43 (m, 1H, fluorophenyl H-2), 7.25-7.16 (m, 7H, phenyl H-2,6, methoxybenzyl H-2,6, phenyl H-3,5,NH), 7.07-7.03 (m, 1H, fluorophenyl H-4), 6.92 (d, J = 8.2 Hz, 2H, methoxybenzyl H-3,5), 6.88 (t, J = 7.0 Hz, 1H, phenyl H-4), 5.71 (s, 2H, CH2), 3.76 (s, 3H, CH3O). 13C NMR (151 MHz, Acetone-d6) δ 165.09, 163.48 (fluorophenyl C-3), 160.67 (methoxybenzyl C-4), 143.89 (phenyl C-1), 143.70 (C-7), 138.51 (C-5), 138.32 (fluorophenyl C-1), 134.16 (C-3a), 132.59 (C-2), 131.65, 131.59 (fluorophenyl C-5), 131.07 (methoxybenzyl C-1), 129.52 (phenyl C-3,5), 128.96 (methoxybenzyl C-2,6), 124.90 (C-7a), 123.80 (fluorophenyl C-6), 121.61 (phenyl C-4), 119.18 (phenyl C-2,6), 116.21 (C-3), 115.44 (methoxybenzyl C-3,5), 114.35, 114.21 (fluorophenyl C-2), 113.52, 113.38 (fluorophenyl C-4), 109.81 (C-4), 55.69 (CH3O), 52.65 (CH2). HR-MS (ESI) m/z: Calcd for C27H23FN3O: [M + H]+ = 424.1820, found 424.1804. Anal. Calcd for C27H22FN3O: C, 76.58; H, 5.24; N, 9.92. Found: C, 76.73; H, 5.29; N, 9.66.
1-(4-Methoxybenzyl)-Ν-(3,4,5-trimethoxyphenyl)-3-phenyl-1H-pyrrolo[2,3-c]pyridin-7-amine (10c): This compound was synthesized using a procedure analogous to that of 10a, starting from 9a. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 1/1). Yield: 60%, m.p. 136–137 °C (EtOAc/Et2O). 1H NMR (600 MHz, DMSO-d6) δ 7.99 (s, 1H, H-2), 7.89 (s, 1H, NH), 7.82 (d, J = 5.5 Hz, 1H, H-5), 7.66 (d, J = 7.5 Hz, 2H, phenyl H-2,6), 7.46 (t, J = 7.6 Hz, 2H, phenyl H-3,5), 7.42 (d, J = 5.5 Hz, 1H, H-4), 7.28 (t, J = 7.3 Hz, 1H, phenyl H-4), 7.10 (d, J = 8.5 Hz, 2H, methoxybenzyl H-2,6), 6.83 (d, J = 8.5 Hz, 2H, methoxybenzyl H-3,5), 6.54 (s, 2H, trimethoxyphenyl H-2,6), 5.65 (s, 2H, CH2), 3.69 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.66 (s, 3H, methoxybenzyl CH3O), 3.61 (s, 3H, trimethoxyphenyl CH3O-4). 13C NMR (151 MHz, DMSO-d6) δ 158.66 (methoxybenzyl C-4), 152.75 (trimethoxyphenyl C-3,5), 142.70 (C-7), 138.92 (trimethoxyphenyl C-1), 137.02 (C-5), 134.34 (phenyl C-1), 132.77 (C-3a), 131.59 (trimethoxyphenyl C-4), 131.46 (C-2), 130.23 (methoxybenzyl C-1), 128.92 (phenyl C-3,5), 128.13 (methoxybenzyl C-2,6), 126.59 (phenyl C-2,6), 125.92 (phenyl C-4), 123.40 (C-7a), 115.51 (C-3), 114.07 (methoxybenzyl C-3,5), 108.67 (C-4), 96.24 (trimethoxyphenyl C-2,6), 60.12 (trimethoxyphenyl CH3O-4), 55.64 (trimethoxyphenyl CH3O-3,5), 55.00 (methoxybenzyl CH3O), 51.06 (CH2). HR-MS (ESI) m/z: Calcd for C30H30N3O4: [M + H]+ = 496.2231, found 496.2224. Anal. Calcd for C30H29N3O4: C, 72.71; H, 5.90; N, 8.48. Found: C, 72.80; H, 5.94; N, 8.34.
3-(3-Fluorophenyl)-1-(4-methoxybenzyl)-Ν-(3,4,5-trimethoxyphenyl)-1H-pyrrolo[2,3-c]pyridin-7-amine (10d): This compound was synthesized using a procedure analogous to that of 10a, starting from 9b.The reaction was completed at 20 h. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 1/1). Yield: 60%, m.p. 93–94 °C (CH2Cl2/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 7.91 (s, 1H, H-2), 7.87 (d, J = 5.2 Hz, 1H, H-5), 7.56-7.54 (m, 1H, fluorophenyl H-6), 7.49-7.46 (m, 1H, fluorophenyl H-5), 7.45-7.42 (m, 2H, H-4, fluorophenyl H-2), 7.26 (d, J = 8.8 Hz, 2H, methoxybenzyl H-2,6), 7.07-7.02 (m, 1H, fluorophenyl H-4), 6.98 (d, J = 8.8 Hz, 2H, methoxybenzyl H-3,5), 6.58 (s, 2H, trimethoxyphenyl H-2,6), 5.74 (s, 2H, CH2), 3.77 (s, 3H, methoxybenzyl CH3O), 3.73 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.66 (s, 3H, trimethoxyphenyl CH3O-4). 13C NMR (151 MHz, Acetone-d6) δ 165.08, 163.47 (fluorophenyl C-3), 160.76 (methoxybenzyl C-4), 154.40 (trimethoxyphenyl C-3,5), 143.99 (C-7), 139.50 (trimethoxyphenyl C-1), 138.38, 138.32 (fluorophenyl C-1), 133.95 (trimethoxyphenyl C-4), 133.86 (C-3a), 132.64 (C-2), 131.65, 131.59 (fluorophenyl C-5), 131.01 (methoxybenzyl C-1), 128.92 (methoxybenzyl C-2,6), 124.30 (C-7a), 123.79 (fluorophenyl C-6), 116.18(C-3), 115.56 (methoxybenzyl C-3,5), 114.34, 114.20 (fluorophenyl C-2), 113.51, 113.37 (fluorophenyl C-4), 109.16 (C-4), 97.69 (trimethoxyphenyl C-2,6), 60.71 (trimethoxyphenyl CH3O-4), 56.40 (trimethoxyphenyl CH3O-3,5), 55.72 (methoxybenzyl CH3O), 52.77 (CH2). HR-MS (ESI) m/z: Calcd for C30H29FN3O4: [M + H]+ = 514.2137, found 514.2114. Anal. Calcd for C30H28FN3O4: C, 70.16; H, 5.50; N, 8.18. Found: C, 70.01; H, 5.44; N, 8.37.
7-Chloro-1-(4-methoxybenzyl)-3-phenyl-1H-pyrrolo[2,3-c]pyridin-6-oxide (11a): To a solution of 9a (240 mg, 0.69 mmol) in dichloromethane (5 mL), m-chloroperoxybenzoic acid (180 mg, 1.04 mmol) was added, and the solution was stirred at rt for 72h. The solvent was then vacuum-evaporated, and a saturated sodium bicarbonate solution was added to the residue and extracted with ethyl acetate. The organic layer was dried (Na2SO4) and concentrated to dryness, and the residue was purified by silica gel column chromatography (ethyl acetate/methanol: 95/5) to yield 11a (220 mg, 88%), m.p. 160–161 °C (EtOAc/n-pentane). 1H NMR (600 MHz, CDCl3) δ 8.16 (d, J = 6.8 Hz, 1H, H-5), 7.60 (d, J = 6.8 Hz, 1H, H-4), 7.49 (d, J = 7.6 Hz, 2H, phenyl H-2,6), 7.43 (t, J = 7,5 Hz, 2H, phenyl H-3,5), 7.37 (s, 1H, H-2), 7.33 (t, J = 7.3 Hz, 1H, phenyl H-4), 7.04 (d, J = 7.8 Hz, 2H, methoxybenzyl H-2,6), 6.84 (d, J = 7.8 Hz, 2H, methoxybenzyl H-3,5), 5.66 (s, 2H, CH2), 3.76 (s, 3H, CH3O). 13C NMR (151 MHz, CDCl3) δ 159.68 (methoxybenzyl C-4), 133.45 (C-5), 132.65 (C-2), 132.11 (phenyl C-4), 130.45 (C-7a), 129.28 (phenyl C-1), 128.79 (phenyl C-3,5), 128.23 (methoxybenzyl C-2,6), 127.67 (phenyl C-2,6), 127.50 (C-7), 126.04 (C-3a), 118.83 (C-3), 114.65 (methoxybenzyl C-3,5), 114.47 (methoxybenzyl C-1), 114.03 (C-4), 55.47 (CH3O), 51.83 (CH2). HR-MS (ESI) m/z: Calcd for C21H18ClN2O2: [M + H]+ = 365.1052, found 365.1036.
7-Chloro-3-(3-fluorophenyl)-1-(4-methoxybenzyl)-1H-pyrrolo[2,3-c]pyridin-6-oxide (11b): This compound was synthesized using a procedure analogous to that of 11a, starting from 9b. Purification was carried out by silica gel column chromatography (ethyl acetate/methanol: 9/1). Yield: 88%, m.p. 124–125 °C (EtOAc/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 8.23 (d, J = 6.7 Hz, 1H, H-5), 7.63 (d, J = 6.7 Hz, 1H, H-4), 7.51 (td, J = 7.9, 4.2 Hz, 1H, fluorophenyl H-5), 7.41 (s, 1H, H-2), 7.37 (dt, J = 7.8, 1.2 Hz, 1H, fluorophenyl H-6), 7.28 (ddd, J = 10.0, 2.5, 1.7 Hz, 1H, fluorophenyl H-2), 7.17 (tdd, J = 8.4, 2.7, 0.9 Hz, 1H, fluorophenyl H-4), 7.07 (d, J = 8.1 Hz, 2H, methoxybenzyl H-2,6), 6.87 (d, J = 8.1 Hz, 2H, methoxybenzyl H-3,5), 5.68 (s, 2H, CH2), 3.78 (s, 3H, CH3O). 13C NMR (151 MHz, Acetone-d6) δ 163.73, 162.11 (fluorophenyl C-3), 159.84 (methoxybenzyl C-4), 136.00, 135.94 (fluorophenyl C-1), 134.63 (C-7), 133.21 (C-5), 132.27 (C-2), 130.23 (C-7a), 130.05, 129.99 (fluorophenyl C-5), 128.24 (methoxybenzyl C-2,6), 125.57 (C-3a), 123.33 (fluorophenyl C-6), 119.10 (C-3), 114.77 (methoxybenzyl C-3,5), 114.47 (methoxybenzyl C-1), 114.43, 114.29 (fluorophenyl C-2), 114.25 (C-4), 114.04, 113.90 (fluorophenyl C-4), 55.48 (CH3O), 52.08 (CH2). HR-MS (ESI) m/z: Calcd for C21H17ClFN2O2: [M + H]+ = 383.0958, found 383.0967.
1-(4-Methoxybenzyl)-7-(4-methylpiperazin-1-yl)-3-phenyl-1H-pyrrolo[2,3-c]pyridine-6-oxide (12a): To a solution of 11a (120 mg, 0.33 mmol) in absolute ethanol (5 mL), 1-methylpiperazine (0.20 mL, 2.00 mmol) was added, and the solution was heated in an autoclave at 120 °C for 24 h. The mixture was extracted with ethyl acetate, the organic phase was dried (Na2SO4) and concentrated to dryness and the residue was purified by silica gel column chromatography (dichloromethane/methanol: 8/2) to give 12a (40 mg, 50%), m.p. 127–128 °C (EtOAc/n-pentane). 1H NMR (600 MHz, CD3OD) 8.00 (d, J = 6.9 Hz, 1H, H-5), 7.85 (d, J = 6.9 Hz, 1H, H-4), 7.79 (s, 1H, H-2), 7.62 (d, J = 7.7 Hz, 2H, phenyl H-2,6), 7.46 (t, J = 7.4 Hz, 2H, phenyl H-3,5), 7.32 (t, J = 7.4 Hz, 1H, phenyl H-4), 6.90 (m, 4H, methoxybenzyl H-2,3,5,6), 5.89 (s, 2H, CH2), 4.17-4.14 (m, 2H, methylpiperazine H), 3.75 (s, 3H, CH3O), 2.89-2.85 (m, 2H, methylpiperazine H), 2.80-2.75 (m, 2H, methylpiperazine H), 2.40 (s, 3H, methylpiperazine CH3), 2.24-2.18 (m, 2H, methylpiperazine H). 13C NMR (600 MHz, CD3OD) 160.65 (methoxybenzyl C-4), 142.92 (C-7), 135.49 (C-2), 134.34 (phenyl C-1), 134.17 (C-5), 131.90 (methoxybenzyl C-1), 131.56 (C-3a), 130.43 (C-7a), 130.14 (phenyl C-3,5), 128.41 (phenyl C-2,6), 128.09 (phenyl C-4), 127.62 (methoxybenzyl C-2,6), 119.51 (C-3), 115.69 (C-4), 115.40 (methoxybenzyl C-3,5), 55.78 (CH3O), 55.11 (methylpiperazine C-3,5), 52.30 (CH2), 47.81 (methylpiperazine C-2,6), 45.67 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C26H29N4O2: [M + H]+ = 429.2286, found 429.2273.
3-(3-Fluorophenyl)-1-(4-methoxybenzyl)-7-(4-methylpiperazin-1-yl)-1H-pyrrolo[2,3-c]pyridine-6-oxide (12b): This compound was synthesized using a procedure analogous to that of 12a, starting from 11b. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 85/15). Yield: 50%, m.p. 91–92 °C (EtOAc/n-pentane). 1H NMR (600 MHz, CD3OD) δ 8.01 (d, J = 6.9 Hz, 1H, H-5), 7.85 (d, J = 6.9 Hz, 1H, H-4), 7.84 (s, 1H, H-2), 7.49-7.45 (m, 1H, fluorophenyl H-5), 7.48-7.44 (m, 1H, fluorophenyl H-6), 7.39-7.34 (m, 1H, fluorophenyl H-2), 7.07-7.01 (m, 1H, fluorophenyl H-4), 6.90-6.88 (m, 4H, methoxybenzyl H-2,3,5,6), 5.91 (s, 2H, CH2), 4.14-4.10 (m, 2H, methylpiperazine H), 3.75 (s, 3H, CH3O), 2.77-2.72 (m, 4H, methylpiperazine H), 2.29 (s, 3H, methylpiperazine CH3), 2.10-2.06 (m, 2H, methylpiperazine H). 13C NMR (151 MHz, CD3OD) δ 165.55, 163.93 (fluorophenyl C-3), 160.63 (methoxybenzyl C-4), 143.34 (C-7), 136.81, 136.76 (fluorophenyl C-1), 135.89 (C-2), 134.46 (C-5), 131.95, 131.89 (fluorophenyl C-5), 131.75 (C-7a), 131.69 (C-3a), 129.72 (methoxybenzyl C-1), 127.66 (methoxybenzyl C-2,6), 124.16 (fluorophenyl C-6), 118.20 (C-3), 115.44 (C-4), 115.37 (methoxybenzyl C-3,5), 114.92, 114.77 (fluorophenyl C-2), 114.66, 114.52 (fluorophenyl C-4), 55.78 (CH3O), 55.26 (methylpiperazine C-3,5), 52.31 (CH2), 48.13 (methylpiperazine C-2,6), 46.10 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C26H28FN4O2: [M + H]+ = 447.2191, found 447.2176.
1-(4-Methoxybenzyl)-7-(4-methylpiperazin-1-yl)-3-phenyl-1H-pyrrolo[2,3-c]pyridine (13a): To a solution of 12a (70 mg, 0.16 mmol) in chloroform (3 mL), phosphorus trichloride (0.10 mL, 0.67 mmol) was added, and the solution was stirred at rt for 20 h. The solvent was then vacuum-evaporated and the mixture was neutralized with a sodium bicarbonate solution and extracted with ethyl acetate. The organic phase was dried (Na2SO4) and concentrated to dryness, and the residue was purified by silica gel column chromatography (dichloromethane/methanol: 9/1) to give 13a (50 mg, 72%), m.p. 88–89 °C (Et2O/n-pentane). 1H NMR (600 MHz, CDCl3) 8.04 (d, J = 5.5 Hz, 1H, H-5), 7.59-7.54 (m, 3H, H-4, phenyl H-2,6), 7.42 (t, J = 7.8 Hz, 2H, phenyl H-3,5), 7.30-7.27 (m, 2H, H-2, phenyl H-4), 7.08 (d, J = 8.7 Hz, 2H, methoxybenzyl H-2,6), 6.83 (d, J = 8.7 Hz, 2H, methoxybenzyl H-3,5), 5.70 (s, 2H, CH2), 3.78 (s, 3H, CH3O), 3.32-3.25 (brs, 4H, methylpiperazine H-2,6), 3.02-2.57 (brs, 4H, methylpiperazine H-3,5), 2.38 (s, 3H, methylpiperazine CH3). 13C NMR (600 MHz, CDCl3) 159.25 (methoxybenzyl C-4), 150.88 (C-7), 137.69 (C-5), 134.75 (phenyl C-1), 133.87 (C-3a), 130.62 (methoxybenzyl C-1), 129.77 (C-2), 129.02 (phenyl C-3,5), 128.45 (methoxybenzyl C-2,6), 127.51 (phenyl C-2,6), 126.56 (phenyl C-4), 126.49 (C-7a), 118.30 (C-3), 114.37 (methoxybenzyl C-3,5), 111.55 (C-4), 55.47 (CH3O), 55.26 (methylpiperazine C-3,5), 51.14 (methylpiperazine C-2,6), 50.38 (CH2), 46.38 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C26H29N4O: [M + H]+ = 413.2336, found 413.2319. Anal. Calcd for C26H28N4O: C, 75.70; H, 6.84; N, 13.58. Found: C, 75.52; H, 6.69; N, 13.66.
3-(3-Fluorophenyl)-1-(4-methoxybenzyl)-7-(4-methylpiperazin-1-yl)-1H-pyrrolo[2,3-c]pyridine (13b): This compound was synthesized using a procedure analogous to that of 13a, starting from 12b. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 9/1). Oil, yield: 99%. 1H NMR (600 MHz, Acetone-d6) δ 7.98 (d, J = 5.6 Hz, 1H, H-5), 7.85 (s, 1H, H-2), 7.57 (d, J = 5.5 Hz, 1H, H-4), 7.52-7.50 (m, 1H, fluorophenyl H-6), 7.47-7.45 (m, 1H, fluorophenyl H-5), 7.41-7.37 (m, 1H, fluorophenyl H-2), 7.21 (d, J = 8.5 Hz, 2H, methoxybenzyl H-2,6), 7.06-7.02 (m, 1H, fluorophenyl H-4), 6.83 (d, J = 8.5 Hz, 2H, methoxybenzyl H-3,5), 5.76 (s, 2H, CH2), 3.72 (s, 3H, CH3O), 3.27-3.20 (brs, 4H, methylpiperazine H-2,6), 2.75-2.50 (brs, 4H, methylpiperazine H-3,5), 2.32 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, Acetone-d6) δ 165.04, 163.43 (fluorophenyl C-3), 160.16 (methoxybenzyl C-4), 151.99 (C-7), 138.71 (C-5), 138.32, 138.26 (fluorophenyl C-1), 134.31 (C-3a), 132.02 (C-2), 131.91 (methoxybenzyl C-1), 131.61, 131.55 (fluorophenyl C-5), 129.48 (methoxybenzyl C-2,6), 127.07 (C-7a), 123.70, 123.69 (fluorophenyl C-6), 117.01 (C-3), 114.85 (methoxybenzyl C-3,5), 114.25, 114.10 (fluorophenyl C-2), 113.50, 113.36 (fluorophenyl C-4), 111.79 (C-4), 55.83 (CH3O), 55.55 (methylpiperazine C-3,5), 51.88 (methylpiperazine C-2,6), 51.15 (CH2), 46.44 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C26H28FN4O: [M + H]+ = 431.2242, found 431.2231. Anal. Calcd for C26H27FN4O: C, 72.54; H, 6.32; N, 13.01. Found: C, 72.33; H, 6.19; N, 13.26.
Ν,3-Diphenyl-1H-pyrrolo[2,3-c]pyridin-7-amine (14a): A solution of 10a (50 mg, 0.12 mmol) in trifluoroacetic acid (2mL) was heated at 60 °C for 20h. The mixture was then vacuum-evaporated, neutralized with a sodium bicarbonate solution, extracted with ethyl acetate, dried (Na2SO4) and concentrated to dryness. The residue was purified by silica gel column chromatography (dichloromethane/methanol: 9/1) to give 14a (yield 99%), m.p. 139-140 °C (EtOAc/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 11.69-11.18 (brs, 1H, pyrrole NH), 9.45-8.50 (brs, 1H, pyridinamine NH), 7.89 (d, J = 8.0 Hz, 2H, N7-phenyl H-2,6), 7.82 (d, J = 5.8 Hz, 1H, H-5), 7.77 (s, 1H, H-2), 7.69 (d, J = 7.8 Hz, 2H, 3-phenyl H-2,6), 7.45 (t, J = 7.5 Hz, 2H, 3-phenyl H-3,5), 7.39 (d, J = 5.8 Hz, 1H, H-4), 7.31 (t, J = 7.5 Hz, 2H, N7-phenyl H-3,5), 7.28 (t, J = 7.4 Hz, 1H, 3-phenyl H-4), 6.99 (t, J = 7.4 Hz, 1H, N7-phenyl H-4). 13C NMR (151 MHz, Acetone-d6) δ 143.78 (C-7), 141.86 (N7-phenyl C-1), 136.08 (C-5), 135.36 (3-phenyl C-1), 131.38 (C-3a), 129.79 (3-phenyl C-3,5), 129.76 (N7-phenyl C-3,5), 127.93 (3-phenyl C-2,6), 126.95 (3-phenyl C-4), 126.12 (N7-phenyl C-4), 122.84 (C-2), 122.52 (C-7a), 120.55 (N7-phenyl C-2,6), 118.92 (C-3), 108.00 (C-4). HR-MS (ESI) m/z: Calcd for C19H16N3: [M + H]+ = 286.1339, found 286.1340. Anal. Calcd for C19H15N3: C, 79.98; H, 5.30; N, 14.72. Found: C, 79.77; H, 5.16; N, 14.89.
3-(3-Fluorophenyl)-Ν-phenyl-1H-pyrrolo[2,3-c]pyridin-7-amine (14b): This compound was synthesized using a procedure analogous to that of 14a, starting from 10b. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 9/1). Yield: 99%, m.p. 168–169 °C (EtOAc/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 11.66-11.23 (brs, 1H, pyrrole NH), 8.94-8.58 (brs, 1H, pyridinamine NH), 7.90-7.85 (m, 4H, H-5, H-2, phenyl H-2,6), 7.57-7.54 (m, 1H, fluorophenyl H-6), 7.51-7.48 (m, 1H, fluorophenyl H-5), 7.46-7.43 (m, 1H, fluorophenyl H-2), 7.40 (d, J = 5.6 Hz, 1H, H-4), 7.31 (t, J = 7.5 Hz, 2H, phenyl H-3,5), 7.06-7.02 (m, 1H, fluorophenyl H-4), 6.98 (t, J = 7.1 Hz, 1H, phenyl H-4). 13C NMR (151 MHz, Acetone-d6) δ 165.10, 163.48 (fluorophenyl C-3), 143.94 (C-5, C-7), 142.19 (phenyl C-1), 138.76, 138.70 (fluorophenyl C-1), 136.40 (C-3a), 131.62, 131.56 (fluorophenyl C-5), 131.14 (C-2), 129.69 (phenyl C-3,5), 126.49 (C-7a), 123.69 (phenyl C-4), 122.72, 122.55 (fluorophenyl C-6), 120.22 (phenyl C-2,6), 117.57 (C-3), 114.24, 114.09 (fluorophenyl C-2), 113.44, 113.30 (fluorophenyl C-4), 107.82 (C-4). HR-MS (ESI) m/z: Calcd for C19H15FN3: [M + H]+ = 304.1245, found 304.1245. Anal. Calcd for C19H14FN3: C, 75.23; H, 4.65; N, 13.85. Found: C, 75.40; H, 4.71; N, 13.69.
N-(3,4,5-trimethoxyphenyl)-3-phenyl-1H-pyrrolo[2,3-c]pyridin-7-amine (14c): This compound was synthesized using a procedure analogous to that of 14a, starting from 10c. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 95/5).Yield: 99%, m.p. 153–154 °C (EtOAc/n-pentane). 1H NMR (600 MHz, DMSO-d6) δ 11.56-11.49 (brs, 1H, pyrrole NH), 8.73-8.65 (brs, 1H, pyridinamine NH), 7.90 (s, 1H, H-2), 7.80 (d, J = 5.7 Hz, 1H, H-5), 7.68 (d, J = 7.6 Hz, 2H, phenyl H-2,6), 7.45 (t, J = 7.6Hz, 2H, phenyl H-3,5), 7.30 (d, J = 5.7Hz, 1H, H-4), 7.28-7.25 (m, 3H, trimethoxyphenyl H-2,6, phenyl H-4), 3.81 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.64 (s, 3H, trimethoxyphenyl CH3O-4).13C NMR (151 MHz, DMSO-d6) δ 152.83 (trimethoxyphenyl C-3,5), 142.93 (C-7), 137.89 (trimethoxyphenyl C-1), 136.24 (C-5), 135.06 (phenyl C-1), 131.83 (trimethoxyphenyl C-4), 129.21 (C-3a), 128.91 (phenyl C-3,5), 126.41 (phenyl C-2,6), 125.70 (phenyl C-4), 124.75 (C-2), 121.31 (C-7a), 116.39 (C-3), 106.62 (C-4), 96.13 (trimethoxyphenyl C-2,6), 60.18 (trimethoxyphenyl CH3O-4), 55.76 (trimethoxyphenyl CH3O-3,5). HR-MS (ESI) m/z: Calcd for C22H20N3O3: [M-H]- = 374.1510, found 374.1508. Anal. Calcd for C22H21N3O3: C, 70.38; H, 5.64; N, 11.19. Found: C, 70.44; H, 5.68; N, 11.11.
3-(3-Fluorophenyl)-N-(3,4,5-trimethoxyphenyl)-1H-pyrrolo[2,3-c]pyridin-7-amine (14d): This compound was synthesized using a procedure analogous to that of 14a, starting from 10d. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 95/5).Yield: 99%, m.p. 149–150 °C (EtOAc/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 11.52-10,85 (brs, 1H, pyrrole NH), 8.90-8.14 (brs, 1H, pyridinamine NH), 7.87 (d, J = 5.7 Hz, 1H, H-5), 7.82 (s, 1H, H-2), 7.56-7.53 (m, 1H, fluorophenyl H-6), 7.50-7.47 (m, 1H, fluorophenyl H-5), 7.46-7.42 (m, 1H, fluorophenyl H-2), 7.37 (d, J = 5.7 Hz, 1H, H-4), 7.27 (s, 2H, trimethoxyphenyl H-2,6), 7.06-7.02 (m, 1H, fluorophenyl H-4), 3.78 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.67 (s, 3H, trimethoxyphenyl CH3O-4). 13C NMR (151 MHz, Acetone-d6) δ 165.08, 163.47 (fluorophenyl C-3), 154.49 (trimethoxyphenyl C-3,5), 144.26 (C-6), 138.81, 138.75 (fluorophenyl C-1), 138.31 (trimethoxyphenyl C-1), 136.86 (C-5), 134.33 (trimethoxyphenyl C-4), 131.59, 131.54 (fluorophenyl C-5), 131.03 (C-3a), 126.22 (C-2), 123.65 (fluorophenyl C-6), 122.64 (C-7a), 117.47 (C-3), 114.18, 114.04 (fluorophenyl C-2), 113.38, 113.24 (fluorophenyl C-4), 107.61 (C-4), 98.40 (trimethoxyphenyl C-2,6), 60.73 (trimethoxyphenyl CH3O-4), 56.40 (trimethoxyphenyl CH3O-3,5). HR-MS (ESI) m/z: Calcd for C22H21FN3O3: [M + H]+ = 394.1562, found 394.1561. Anal. Calcd for C22H20FN3O3: C, 67.17; H, 5.12; N, 10.68. Found: C, 67.33; H, 5.24; N, 10.59.
7-(4-Methylpiperazin-1-yl)-3-phenyl-1H-pyrrolo[2,3-c]pyridine (14e): This compound was synthesized using a procedure analogous to that of 14a, starting from 13a. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 85/15). Yield: 91%, m.p. 220–221 °C (EtOAc/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 10.80-10.70 (brs, 1H, ΝH), 7.88 (d, J = 5.6 Hz, 1H, H-5), 7.70-7.65 (m, 3H, phenyl H-2,6, H-2), 7.47-7.40 (m, 3H, phenyl H-3,5, H-4), 7.26 (t, J = 7.7 Hz, 1H, phenyl H-4), 3.52-3.46 (m, 4H, methylpiperazine H-2,6), 2.64-2.60 (m, 4H, methylpiperazine H-3,5), 2.32 (s, 3H, methylpiperazine CH3). 13C NMR (600 MHz, Acetone-d6) δ 150.27 (C-7), 137.95 (C-5), 136.39 (phenyl C-1), 132.33 (C-3a), 129.72 (phenyl C-3,5), 127.91 (phenyl C-2,6), 126.76 (phenyl C-4), 125.24 (C-7a), 125.08 (C-2), 118.36 (C-3), 109.71 (C-4), 55.98 (methylpiperazine C-3,5), 49.52 (methylpiperazine C-2,6), 46.44 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C18H19N4: [M-H]- =291.1615, found 291.1612. Anal. Calcd for C18H20N4: C, 73.94; H, 6.89; N, 19.17. Found: C, 74.03; H, 6.92; N, 19.02.
3-(3-Fluorophenyl)-7-(4-methylpiperazin-1-yl)-1H-pyrrolo[2,3-c]pyridine (14f): This compound was synthesized using a procedure analogous to that of 14a, starting from 13b. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 85/15). Yield: 93%, m.p. 209–210 °C (EtOAc/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 11.08-10.87 (brs, 1H, NH), 7.90 (d, J = 5.7 Hz, 1H, H-5), 7.77 (s, 1H, H-2), 7.56-7.52 (m, 1H, fluorophenyl H-6), 7.50-7.40 (m, 3H, fluorophenyl H-5, H-4, fluorophenyl H-2), 7.05-6.98 (m, 1H, fluorophenyl H-4), 3.55-3.48 (m, 4H, methylpiperazine H-2,6), 2.72-2.65 (m, 4H, methylpiperazine H-3,5), 2.36 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, Acetone-d6) δ 165.07, 163.46 (fluorophenyl C-3), 150.10 (C-7), 138.92, 138.87 (fluorophenyl C-1), 138.21 (C-5), 132.12 (C-3a), 131.54, 131.48 (fluorophenyl C-5), 125.93 (C-7a), 125.33 (C-2), 123.67 (fluorophenyl C-6), 117.06 (C-3), 114.20, 114.05 (fluorophenyl C-2), 113.28, 113.14 (fluorophenyl C-4), 109.62 (C-4), 55.65 (methylpiperazine C-3,5), 49.14 (methylpiperazine C-2,6), 46.02 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C18H20FN4: [M + H]+ = 311.1667, found 311.1670. Anal. Calcd for C18H19FN4: C, 69.66; H, 6.17; N, 18.05. Found: C, 69.43; H, 6.14; N, 18.21.
4-Chloro-7-iodo-5-(4-methoxybenzyl)-5H-pyrrolo[3,2-d]pyrimidine (22): This compound was synthesized using a procedure analogous to that of 8, starting from 21. Yield: 99%, m.p. 180–181 °C (CH2Cl2/Et2O). 1H NMR (600 MHz, CDCl3) δ 8.78 (s, 1H, H-2), 7.52 (s, 1H, H-6), 7.08 (d, J = 8.6 Hz, 2H, methoxybenzyl H-2,6), 6.88 (d, J = 8.6 Hz, 2H, methoxybenzyl H-3,5), 5.64 (s, 2H, methoxybenzyl CH2), 3.79 (s, 3H, methoxybenzyl CH3O). 13C NMR (151 MHz, CDCl3) δ 159.88 (methoxybenzyl C-4), 152.65 (C-7a), 150.64 (C-2), 142.62 (C-4), 140.09 (C-6), 128.76 (methoxybenzyl C-2,6), 127.81 (methoxybenzyl C-1), 124.45 (C-4a), 114.69 (methoxybenzyl C-3,5), 57.91 (C-7), 55.45 (CH3O), 52.29 (CH2). HR-MS (ESI) m/z: Calcd for C14H12ClIN3O: [M + H]+ = 399.9709, found 399.9718.
4-Chloro-5-(4-methoxybenzyl)-7-phenyl-5H-pyrrolo[3,2-d]pyrimidine (23a): This compound was synthesized using a procedure analogous to that of 9a, starting from 22. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 9/1). Yield: 94%, m.p. 93–94 °C (CHCl3/n-hexane). 1H NMR (600 MHz, CDCl3) δ 8.80 (s, 1H, H-2), 8.00 (d, J = 7.4 Hz, 2H, phenyl H-2,6), 7.71 (s, 1H, H-6), 7.45 (t, J = 7.7 Hz, 2H, phenyl H-3,5), 7.31 (t, J = 7.7 Hz, 1H, phenyl H-4), 7.11 (d, J = 8.7 Hz, 2H, methoxybenzyl H-2,6), 6.88 (d, J = 8.7 Hz, 2H, methoxybenzyl H-3,5), 5.70 (s, 2H, CH2), 3.79 (s, 3H, CH3O). 13C NMR (151 MHz, CDCl3) δ 159.76 (methoxybenzyl C-4), 150.17 (C-2), 149.91 (C-7a), 142.67 (C-4), 133.65 (C-6), 132.13 (phenyl C-1), 128.97 (phenyl C-3,5), 128.53 (methoxybenzyl C-2,6), 127.12 (phenyl C-4), 127.10 (phenyl C-2,6), 124.91 (C-4a), 117.21 (C-7), 114.75 (methoxybenzyl C-1), 114.65 (methoxybenzyl C-3,5), 55.47 (CH3O), 51.89 (CH2). HR-MS (ESI) m/z: Calcd for C20H17ClN3O: [M + H]+ = 350.1055, found 350.1065.
4-Chloro-7-(3-fluorophenyl)-5-(4-methoxybenzyl)-5H-pyrrolo[3,2-d]pyrimidine (23b): This compound was synthesized using a procedure analogous to that of 9a, starting from 22. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 8/2). Yield: 90%, m.p. 88–89 °C (CHCl3/n-hexane). 1H NMR (600 MHz, CDCl3) δ 8.81 (s, 1H, H-2), 7.82-7.79 (m, 1H fluorophenyl H-2), 7.79-7.76 (m, 1H, fluorophenyl H-6), 7.71 (s, 1H, H-6), 7.40-7.37 (m, 1H, fluorophenyl H-5), 7.12 (d, J = 8.5 Hz, 2H, methoxybenzyl H-2,6), 7.00-6.96 (m, 1H, fluorophenyl H-4), 6.89 (d, J = 8.6 Hz, 2H, methoxybenzyl H-3,5), 5.70 (s, 2H, CH2), 3.80 (s, 3H, CH3O). 13C NMR (151 MHz, CDCl3) δ 164.10, 162.48 (fluorophenyl C-3), 159.81 (methoxybenzyl C-4), 150.28 (C-2), 149.73 (C-7a), 142.80 (C-4), 134.28, 134.23 (fluorophenyl C-1) 133.77 (C-6), 130.37, 130.31 (fluorophenyl C-5), 128.59 (methoxybenzyl C-2,6), 124.96 (C-4a), 122.44 (fluorophenyl C-6), 115.92 (C-7), 114.75 (methoxybenzyl C-1), 114.69 (methoxybenzyl C-3,5), 113.94, 113.87 (fluorophenyl C-2), 113.79, 113.73 (fluorophenyl C-4), 55.46 (CH3O), 51.96 (CH2). HR-MS (ESI) m/z: Calcd for C20H16ClFN3O: [M + H]+ = 368.0961, found 368.0970.
5-(4-Methoxybenzyl)-Ν,7-diphenyl-5H-pyrrolo[3,2-d]pyrimidin-4-amine (24a): This compound was synthesized using a procedure analogous to that of 10a, starting from 23a. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 7/3).Yield: 87%, m.p. 57–58 °C (CH2Cl2/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 8.44 (s, 1H, H-2), 8.28 (d, J = 7.6 Hz, 2H, 7-phenyl H-2,6), 8.16 (s, 1H, H-6), 7.49 (d, J = 7.9 Hz, 2H, N4-phenyl H-2,6), 7.41 (t, J = 7.7 Hz, 2H, 7-phenyl H-3,5), 7.33 (brs, 1H, NH), 7.30-7.19 (m, 5H, N4-phenyl H-3,5, methoxybenzyl H-2,6, 7-phenyl H-4), 7.06-6.97 (m, 3H, N4-phenyl H-4, methoxybenzyl H-3,5), 5.81 (s, 2H, CH2), 3.77 (s, 3H, CH3O). 13C NMR (151 MHz, Acetone-d6) δ 160.91 (methoxybenzyl C-4), 151.09 (C-2), 149.36 (C-4), 148.44 (C-7a), 140.77 (N4-phenyl C-1), 134.92 (7-phenyl C-1), 132.90 (C-6), 130.46 (methoxybenzyl C-1), 129.43 (7-phenyl C-3,5), 129.28 (N4-phenyl C-3,5), 128.81 (methoxybenzyl C-2,6), 127.39 (7-phenyl C-2,6), 126.74 (7-phenyl C-4), 123.61 (N4-phenyl C-4), 121.63 (N4-phenyl C-2,6), 117.03 (C-4a), 116.11 (C-7), 115.71 (methoxybenzyl C-3,5), 55.75 (CH3O), 53.24 (CH2). HR-MS (ESI) m/z: Calcd for C26H23N4O: [M + H]+ = 407.1867, found 407.1864. Anal. Calcd for C26H22N4O: C, 76.83; H, 5.46; N, 13.78. Found: C, 76.99; H, 5.45; N, 13.60.
7-(3-Fluorophenyl)-5-(4-methoxybenzyl)-Ν-phenyl-5H-pyrrolo[3,2-d]pyrimidin-4-amine (24b): This compound was synthesized using a procedure analogous to that of 10a, starting from 23b. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 8/2). Yield: 85%, m.p. 131–132 °C (CH2Cl2/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 8.46 (s, 1H, H-2), 8.26-8.22 (m, 2H, fluorophenyl H-2, H-6), 8.06-8.02 (m, 1H, fluorophenyl H-6), 7.48 (d, J = 7.9 Hz, 2H, phenyl H-2,6), 7.46-7.42 (m, 1H, fluorophenyl H-5), 7.33 (brs, 1H, NH), 7.31-7.24 (m, 4H, phenyl H-3,5, methoxybenzyl H-2,6), 7.05-6.96 (m, 4H, phenyl H-4, methoxybenzyl H-3,5, fluorophenyl H-4), 5.80 (s, 2H, CH2), 3.77 (s, 3H, CH3O). 13C NMR (151 MHz, Acetone-d6) δ 164.92, 163.32 (fluorophenyl C-3), 160.95 (methoxybenzyl C-4), 151.30 (C-2), 149.25 (C-4), 148.51 (C-7a), 140.63 (phenyl C-1), 137.44, 137.38 (fluorophenyl C-1), 133.43 (C-6), 131.01, 130.95 (fluorophenyl C-5), 130.26 (methoxybenzyl C-1), 129.44 (phenyl C-3,5), 128.84 (methoxybenzyl C-2,6), 123.74 (phenyl C-4), 122.79 (fluorophenyl C-6), 121.74 (phenyl C-2,6), 117.06 (C-4a), 115.74 (methoxybenzyl C-3,5), 114.69 (C-7), 113.93, 113.78 (fluorophenyl C-2), 113.14, 113.00 (fluorophenyl C-4), 55.75 (CH3O), 53.34 (CH2). HR-MS (ESI) m/z: Calcd for C26H22FN4O: [M + H]+ = 425.1773, found 425.1767. Anal. Calcd for C26H21FN4O: C, 73.57; H, 4.99; N, 13.20. Found: C, 73.73; H, 5.08; N, 13.02.
5-(4-Methoxybenzyl)-N-(3,4,5-trimethoxyphenyl)-7-phenyl-5H-pyrrolo[3,2-d]pyrimidin-4-amine (24c): This compound was synthesized using a procedure analogous to that of 10a, starting from 23a. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 7/3). Yield: 60%, m.p. 187–188 °C (CH2Cl2/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 8.45 (s, 1H, H-2), 8.28 (d, J = 8.1 Hz, 2H, phenyl H-2,6), 8.15 (s, 1H, H-6), 7.41 (t, J = 7.8 Hz, 2H, phenyl H-3,5), 7.31 (d, J = 8.7 Hz, 2H, methoxybenzyl H-2,6), 7.23 (t, J = 7.4 Hz, 1H, phenyl H-4), 7.12 (brs, 1H, NH), 7.05 (d, J = 8.7 Hz, 2H, methoxybenzyl H-3,5), 6.78 (s, 2H, trimethoxyphenyl H-2,6), 5.80 (s, 2H, CH2), 3.79 (s, 3H, methoxybenzyl CH3O), 3.78 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.68 (s, 3H, trimethoxyphenyl CH3O-4). 13C NMR (151 MHz, Acetone-d6) δ 161.00 (methoxybenzyl C-4), 154.22 (trimethoxyphenyl C-3,5), 151.16 (C-2), 149.17 (C-4), 148.48 (C-7a), 136.56 (trimethoxyphenyl C-1), 135.08 (trimethoxyphenyl C-4), 134.93 (phenyl C-1), 132.94 (C-6), 130.45 (methoxybenzyl C-1), 129.28 (phenyl C-3,5), 128.84 (methoxybenzyl C-2,6), 127.39 (phenyl C-2,6), 126.74 (phenyl C-4), 116.91 (C-4a), 116.07 (C-7), 115.83 (methoxybenzyl C-3,5), 99.69 (trimethoxyphenyl C-2,6), 60.71 (trimethoxyphenyl CH3O-4), 56.51 (trimethoxyphenyl CH3O-3,5), 55.77 (methoxybenzyl CH3O), 53.29 (CH2). HR-MS (ESI) m/z: Calcd for C29H29N4O4: [M + H]+ = 497.2184, found 497.2165. Anal. Calcd for C29H28N4O4: C, 70.15; H, 5.68; N, 11.28. Found: C, 69.99; H, 5.51; N, 11.44.
7-(3-Fluorophenyl)-5-(4-methoxybenzyl)-N-(3,4,5-trimethoxyphenyl)-5H-pyrrolo[3,2-d]pyrimidin-4-amine (24d): This compound was synthesized using a procedure analogous to that of 10a, starting from 23b. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 6/4). Yield: 63%, m.p. 96–97 °C (CH2Cl2/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 8.47 (s, 1H, H-2), 8.26-8.20 (m, 2H, H-6, fluorophenyl H-2), 8.07-8.03 (m, 1H, fluorophenyl H-6), 7.47-7.40 (m, 1H, fluorophenyl H-5), 7.30 (d, J = 8.7 Hz, 2H, methoxybenzyl H-2,6), 7.15 (brs, 1H, NH), 7.05 (d, J = 8.7 Hz, 2H, methoxybenzyl H-3,5), 7.02-6.97 (m, 1H, fluorophenyl H-4), 6.77 (s, 2H, trimethoxyphenyl H-2,6), 5.81 (s, 2H, CH2), 3.79 (s, 3H, methoxybenzyl CH3O), 3.78 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.67 (s, 3H, trimethoxyphenyl CH3O-4). 13C NMR (151 MHz, Acetone-d6) δ 164.92, 163.32 (fluorophenyl C-3), 161.05 (methoxybenzyl C-4), 154.23 (trimethoxyphenyl C-3,5), 151.39 (C-2), 149.07 (C-4), 148.53 (C-7a), 137.46, 137.40 (fluorophenyl C-1), 136.41 (trimethoxyphenyl C-1), 135.17 (trimethoxyphenyl C-4), 133.47 (C-6), 131.02, 130.96 (fluorophenyl C-5), 130.24 (methoxybenzyl C-1), 128.86 (methoxybenzyl C-2,6), 122.78 (fluorophenyl C-6), 116.94 (C-4a), 115.87 (methoxybenzyl C-3,5), 114.65 (C-7), 11393, 113.78 (fluorophenyl C-2), 113.15, 113.01 (fluorophenyl C-4), 99.78 (trimethoxyphenyl C-2,6), 60.71 (trimethoxyphenyl CH3O-4), 56.51 (trimethoxyphenyl CH3O-3,5), 55.78 (methoxybenzyl CH3O), 53.39 (CH2). HR-MS (ESI) m/z: Calcd for C29H28FN4O4: [M + H]+ = 515.2090, found 515.2075. Anal. Calcd for C29H27FN4O4: C, 67.69; H, 5.29; N, 10.89. Found: C, 67.48; H, 5.16; N, 11.03.
5-(4-Methoxybenzyl)-4-(4-methylpiperazin-1-yl)-7-phenyl-5H-pyrrolo[3,2-d]pyrimidine (25a): To a solution of 23a (30 mg, 0.09 mmol) in dimethylsulfoxide (1 mL), 1-methylpiperazine (0.1 mL, 0.90 mmol) was added, and the solution was heated at 120 °C for 20 h. Then, the mixture was diluted with water and extracted with ethyl acetate. The organic phase was dried (Na2SO4) and concentrated to dryness, and the residue was purified by silica gel column chromatography (ethyl acetate/methanol: 8/2) to give 25a in almost quantitative yield. M.p. 150–151 °C (CH2Cl2/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 8.57 (s, 1H, H-2), 8.22 (d, J = 7.6 Hz, 2H, phenyl H-2,6), 8.11 (s, 1H, H-6), 7.37 (t, J = 7.7 Hz, 2H, phenyl H-3,5), 7.23-7.19 (m, 3H, phenyl H-4, methoxybenzyl H-2,6), 6.82 (d, J = 8.6 Hz, 2H, methoxybenzyl H-3,5), 5.52 (s, 2H, CH2), 3.71 (s, 3H, CH3O), 3.46-3.37 (brs, 4H, methylpiperazine H-2,6), 2.68-2.60 (brs, 4H, methylpiperazine H-3,5), 2.32 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, Acetone-d6) δ 160.31 (methoxybenzyl C-4), 156.02 (C-4), 151.09 (C-2), 150.79 (C-7a), 134.67 (phenyl C-1), 132.92 (C-6), 131.00 (methoxybenzyl C-1), 129.67 (phenyl C-3,5), 129.25 (methoxybenzyl C-2,6), 127.29 (phenyl C-2,6), 126.86 (phenyl C-4), 120.77 (C-4a), 117.66 (C-7), 114.90 (methoxybenzyl C-3,5), 55.53 (CH3O), 55.46 (methylpiperazine C-3,5), 51.63 (methylpiperazine C-2,6), 50.90 (CH2), 46.42 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C25H28N5O: [M + H]+ = 414.2289, found 414.2286. Anal. Calcd for C25H27N5O: C, 72.61; H, 6.58; N, 16.94. Found: C, 72.90; H, 6.69; N, 16.63.
7-(3-Fluorophenyl)-5-(4-methoxybenzyl)-4-(4-methylpiperazin-1-yl)-5H-pyrrolo[3,2-d]pyrimidine (25b): This compound was synthesized using a procedure analogous to that of 25a, starting from 23b. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 9/1). Yield: 97%, m.p. 151–152 °C (CH2Cl2/n-pentane). 1H NMR (600 MHz, Acetone-d6) δ 8.58 (s, 1H, H-2), 8.20 (s, 1H, H-6), 8.18-8.14 (m, 1H, fluorophenyl H-2), 8.02-7.97 (m, 1H, fluorophenyl H-6), 7.42-7.35 (m, 1H, fluorophenyl H-5), 7.20 (d, J = 8.6 Hz, 2H, methoxybenzyl H-2,6), 6.98-6.93 (m, 1H, fluorophenyl H-4), 6.82 (d, J = 8.6 Hz, 2H, methoxybenzyl H-3,5), 5.53 (s, 2H, CH2), 3.71 (s, 3H,CH3O), 3.50-3.38 (brs, 4H, methylpiperazine H-2,6), 2.67-2.58 (brs, 4H, methylpiperazine, H-3,5), 2.30 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, Acetone-d6) δ 164.86, 163.26 (fluorophenyl C-3), 160.38 (methoxybenzyl C-4), 156.14 (C-4), 151.28 (C-2), 150.65 (C-7a), 137.18, 137.12 (fluorophenyl C-1), 133.49 (C-6), 131.01, 130.95 (fluorophenyl C-5), 130.85 (methoxybenzyl C-1), 129.69 (methoxybenzyl C-2,6), 122.77 (fluorophenyl C-6), 121.89 (C-7), 116.27 (C-4a), 114.96 (methoxybenzyl C-3,5), 113.84, 113.69 (fluorophenyl C-2), 113.29, 113.15 (fluorophenyl C-4), 55.55 (CH3O, methylpiperazine C-3,5), 51.79 (CH2), 50.96 (methylpiperazine C-2,6), 46.46 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C25H27FN5O: [M + H]+ = 432.2195, found 432.2181. Anal. Calcd for C25H26FN5O: C, 69.59; H, 6.07; N, 16.23. Found: C, 69.80; H, 6.11; N, 16.00.
Ν,7-Diphenyl-5H-pyrrolo[3,2-d]pyrimidin-4-amine (26a): This compound was synthesized using a procedure analogous to that of 14a, starting from 24a. Purification was carried out by silica gel column chromatography (dichloromethane/ethyl acetate: 7/3). Yield: 99%, m.p. > 250 °C (EtOAc). 1H NMR (600 MHz, Acetone-d6) δ 10.76-10.67 (brs, 1H, pyrrole NH), 8.64-8.58 (brs, 1H, pyrimidinamine NH), 8.54 (s, 1H, H-2), 8.26 (dd, J = 8.2, 1.1 Hz, 2H, 7-phenyl H-2,6), 8.02 (s, 1H, H-6), 7.93 (dd, J = 7.7, 1.0 Hz, 2H, N-phenyl H-2,6), 7.39 (t, J = 7.8 Hz, 2H, 7-phenyl H-3,5), 7.35 (t, J = 7.5 Hz, 2H, N-phenyl H-3,5), 7.21 (t, J = 7.4 Hz, 1H, 7-phenyl H-4), 7.05 (t, J = 7.3 Hz, 1H, N-phenyl H-4). 13C NMR (151 MHz, Acetone-d6) δ 151.09 (C-2), 148.19 (C-4), 147.05 (C-7a), 141.33 (N-phenyl C-1), 135.34 (7-phenyl C-1), 129.64 (7-phenyl C-3,5), 129.24 (N-phenyl C-3,5), 127.27 (7-phenyl C-2,6), 126.63 (7-phenyl C-4), 126.42 (C-6), 123.32 (N-phenyl C-4), 120.81 (N-phenyl C-2,6), 117.10 (C-4a), 116.53 (C-7). HR-MS (ESI) m/z: Calcd for C18H15N4: [M + H]+ =287.1292, found 287.1296. Anal. Calcd for C18H14N4: C, 75.50; H, 4.93; N, 19.57. Found: C, 75.36; H, 4.90; N, 19.69.
7-(3-Fluorophenyl)-Ν-phenyl-5H-pyrrolo[3,2-d]pyrimidin-4-amine (26b): This compound was synthesized using a procedure analogous to that of 14a, starting from 24b. Purification was carried out by silica gel column chromatography (dichloromethane/ethyl acetate: 75/25). Yield: 96%, m.p. > 250 °C (EtOAc). 1H NMR (600 MHz, Acetone-d6) δ 10.87-10.71 (brs, 1H, pyrrole NH), 8.63-8.58 (brs, 1H, pyrimidinamine NH), 8.55 (s, 1H, H-2), 8.24-8.20 (m, 1H, fluorophenyl H-2), 8.12 (s, 1H, H-6), 8.06-8.02 (m, 1H, fluorophenyl H-6), 7.93 (d, J = 7.7 Hz, 2H, phenyl H-2,6), 7.39-7.36 (m, 1H, fluorophenyl H-5), 7.34 (t, J = 7.5 Hz, 2H, phenyl H-3,5), 7.06 (t, J = 7.3 Hz, 1H, phenyl H-4), 6.99-6.93 (m 1H, fluorophenyl H-4). 13C NMR (151 MHz, Acetone-d6) δ 164.93, 163.33 (fluorophenyl C-3), 151.35 (C-2), 148.25 (C-4), 147.07 (C-7a), 141.23 (phenyl C-2,6), 137.90, 137.84 (fluorophenyl C-1), 130.97, 130.91 (fluorophenyl C-5), 129.67 (phenyl C-3,5), 127.06 (C-6), 123.44 (phenyl C-4), 122.69 (fluorophenyl C-6), 120.88 (phenyl C-2,6), 116.58 (C-4a), 115.74 (C-7), 113.79, 113.64 (fluorophenyl C-2), 113.03, 112.89 (fluorophenyl C-4). HR-MS (ESI) m/z: Calcd for C18H14FN4: [M + H]+ = 305.1197, found 305.1192. Anal. Calcd for C18H13FN4: C, 71.04; H, 4.31; N, 18.41. Found: C, 70.79; H, 4.22; N, 18.60.
N-(3,4,5-Trimethoxyphenyl)-7-phenyl-5H-pyrrolo[3,2-d]pyrimidin-4-amine (26c): This compound was synthesized using a procedure analogous to that of 14a, starting from 24c. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 3/7). Yield: 97%, m.p. > 250 °C (EtOAc). 1H NMR (600 MHz, Acetone-d6) δ 10.85-10.79 (brs, 1H, pyrrole NH), 8.70-8.63 (brs, 1H, pyrimidinamine NH), 8.53 (s, 1H, H-2), 8.26 (d, J = 7.9 Hz, 2H, phenyl H-2,6), 8.01 (s, 1H, H-6), 7.39 (t, J = 7.5 Hz, 2H, phenyl H-3,5), 7.31 (s, 2H, trimethoxyphenyl H-2,6), 7.21 (t, J = 7.2 Hz, 1H, phenyl H-4), 3.84 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.71 (s, 3H, CH3O-4). 13C NMR (151 MHz, Acetone-d6) δ 154.46 (trimethoxyphenyl C-3,5), 151.17 (C-2), 148.32 (C-4), 147.07 (C-7a), 137.37 (trimethoxyphenyl C-1), 135.43 (phenyl C-1), 134.98 (trimethoxyphenyl C-4), 129.24 (phenyl C-3,5), 127.27 (phenyl C-2,6), 126.59 (phenyl C-4), 126.29 (C-6), 117.05 (C-4a), 116.48 (C-7), 99.14 (trimethoxyphenyl C-2,6), 60.75 (trimethoxyphenyl CH3O-4), 56.52 (trimethoxyphenyl CH3O-3,5). HR-MS (ESI) m/z: Calcd for C21H19N4O3: [M-H]- =375.1462, found 375.1456. Anal. Calcd for C21H20N4O3: C, 67.01; H, 5.36; N, 14.88. Found: C, 66.84; H, 5.29; N, 15.09.
7-(3-Fluorophenyl)-N-(3,4,5-trimethoxyphenyl)-5H-pyrrolo[3,2-d]pyrimidin-4-amine (26d): This compound was synthesized using a procedure analogous to that of 14a, starting from 24d. Purification was carried out by silica gel column chromatography (chloroform/ethyl acetate: 4/6). Yield: 93%, m.p. 139–140 °C (EtOAc/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 10.82-10.66 (brs, 1H, pyrrole NH), 8.58-8.54 (m, 2H, pyrimidinamine NH, H-2), 8.25-8.19 (m, 1H, fluorophenyl H-2), 8.11 (s, 1H, H-6), 8.06-8.02 (m, 1H, fluorophenyl H-6), 7.44-7.38 (m, 1H, fluorophenyl H-5), 7.28 (s, 2H, trimethoxyphenyl H-2,6), 7.00-6.93 (m, 1H, fluorophenyl H-4), 3.84 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.71 (s, 3H, CH3O-4). 13C NMR (151 MHz, Acetone-d6) δ 164.92, 163.32 (fluorophenyl C-3), 154.46 (trimethoxyphenyl C-3,5), 151.38 (C-2), 148.34 (C-4), 146.94 (C-7a), 137.91, 137.85 (fluorophenyl C-1), 137.16 (trimethoxyphenyl C-1), 135.07 (trimethoxyphenyl C-4), 130.97, 130.91 (fluorophenyl C-5), 126.97 (C-6), 122.68 (fluorophenyl C-6), 116.48 (C-4a), 115.69 (C-7), 113.77, 113.62 (fluorophenyl C-2), 113.02, 112.87 (fluorophenyl C-4), 99.19 (trimethoxyphenyl C-2,6), 60.75 (trimethoxyphenyl CH3O-4), 56.52 (trimethoxyphenyl CH3O-3,5). HR-MS (ESI) m/z: Calcd for C21H18FN4O3: [M-H]- = 393.1368, found 393.1370. Anal. Calcd for C21H19FN4O3: C, 63.95; H, 4.86; N, 14.21. Found: C, 64.12; H, 4.89; N, 14.05.
4-(4-Methylpiperazin-1-yl)-7-phenyl-5H-pyrrolo[3,2-d]pyrimidine (26e): This compound was synthesized using a procedure analogous to that of 14a, starting from 25a. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 85/15). Yield: 92%, m.p. > 250 °C (EtOAc/Et2O). 1H NMR (600 MHz, Acetone-d6) δ 11.18-10.91 (brs, 1H, NH), 8.44 (s, 1H, H-2), 8.25 (d, J = 7.5 Hz, 2H, phenyl H-2,6), 7.96 (s, 1H, H-6), 7.37 (t, J = 7.7 Hz, 2H, phenyl H-3,5), 7.19 (t, J = 7.4 Hz, 1H, phenyl H-4), 3.96-3.90 (m, 4H, methylpiperazine H-2,6), 2.82-2.76 (m, 4H, methylpiperazine H-3,5), 2.47 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, Acetone-d6) δ 151.98 (C-2), 150.94 (C-4), 148.69 (C-7a), 135.32 (phenyl C-1), 129.17 (phenyl C-3,5), 127.38 (phenyl C-2,6), 126.79 (C-6), 126.54 (phenyl C-4), 116.95 (C-4a), 116.67 (C-7), 55.05 (methylpiperazine C-3,5), 46.24 (methylpiperazine C-2,6), 45.45 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C17H20N5: [M + H]+ = 294.1714, found 294.1715. Anal. Calcd for C17H19N5: C, 69.60; H, 6.53; N, 23.87. Found: C, 69.82; H, 6.58; N, 23.57.
7-(3-Fluorophenyl)-4-(4-methylpiperazin-1-yl)-5H-pyrrolo[3,2-d]pyrimidine (26f): This compound was synthesized using a procedure analogous to that of 14a, starting from 25b. Oil purification was carried out by silica gel column chromatography (dichloromethane/methanol: 7/3). Yield: 91%. 1H NMR (600 MHz, Acetone-d6) δ 11.78-11.05 (brs, 1H, NH), 8.44 (s, 1H, H-2), 8.22-8.16 (m, 1H, fluorophenyl H-2), 8.02 (s, 1H, H-6), 8.00-7.96 (m, 1H, fluorophenyl H-6), 7.42-7.34 (m, 1H, fluorophenyl H-5), 6.97-6.89 (m, 1H, fluorophenyl H-4), 3.98-3.87 (m, 4H, methylpiperazine H-2,6), 2.75-2.67 (m, 4H, methylpiperazine H-3,5), 2.39 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, Acetone-d6) δ 164.86, 163.26 (fluorophenyl C-3), 152.03 (C-4), 151.14 (C-2), 148.55 (C-7a), 137.89, 137.83 (fluorophenyl C-1), 130.84, 130.78 (fluorophenyl C-5), 127.27 (C-6), 122.76 (fluorophenyl C-6), 116.85 (C-4a), 115.21 (C-7), 113.87, 113.72 (fluorophenyl C-2), 112.89, 112.75 (fluorophenyl C-4), 55.21 (methylpiperazine C-3,5), 46.38 (methylpiperazine C-2,6), 45.68 (methylpiperazine CH3). HR-MS (ESI) m/z: Calcd for C17H19FN5: [M + H]+ = 312.1619, found 312.1610. Anal. Calcd for C17H18FN5: C, 65.58; H, 5.83; N, 22.49. Found: C, 65.66; H, 5.87; N, 22.34.
Ethyl 4-(3-fluorophenyl)-3-(hydroxyimino)-2,4-dioxobutanoate (31b): To a solution of 30b (1.28 g, 5.36 mmol) in glacial acetic acid (10 mL), a sodium nitrite solution (3.5 mL, 3.80 M) was added at 10 °C, under the surface of the solution with a flow rate of 0.15 mL/min via a controlled infusion device. The solution was stirred at rt for 1 h and then was neutralized with 25% ammonia solution and extracted with ethyl acetate; the solvent was dried (Na2SO4) and concentrated to dryness to give pure 31b that corresponded practically to a sole isomer. Yield: 85%. Amorphous solid. 1H NMR (600 MHz, DMSO-d6) δ 12.59 (s, 1H, OH), 7.81-7.78 (m, 1H, fluorophenyl H-6), 7.67-7.63 (m, 1H, fluorophenyl H-2), 7.58-7.55 (m, 1H, fluorophenyl H-5), 7.50-7.45 (m, 1H, fluorophenyl H-4), 4.38 (q, J = 7.2 Hz, 2H, ethylester H-1), 1.26 (t, J = 7.2 Hz, 3H, ethylester H-2). 13C NMR (151 MHz, DMSO-d6) δ 192.03 (C-4), 166.20 (C-2), 164.47 (C-1), 162.79, 161.17 (fluorophenyl C-3), 152.09 (C-3), 133.28, 133.23 (fluorophenyl C-1), 130.84, 130.78 (fluorophenyl C-5), 125.44, 125.42 (fluorophenyl C-6), 119.94, 119.80 (fluorophenyl C-4), 115.81, 115.66 (fluorophenyl C-2), 62.76 (ethylester C-1), 13.72 (ethylester C-2). HR-MS (ESI) m/z: Calcd for C12H9FNO5: [M-H]- =266.0470, found 266.0469.
Ethyl 4-amino-3-(3-fluorophenyl)-1H-pyrazole-5-carboxylate (32b): To a solution of 31b (1.22 g, 4.57 mmol) in absolute ethanol (8 mL), hydrazine hydrate (0.5 mL, 9.14 mmol) was added dropwise, under cooling, and the resulting solution was heated at 45 °C for 2 h. The solvent was then vacuum evaporated ethyl acetate was added to the residue and it was washed with a 3N hydrochloric acid solution. The aqueous phase was neutralized with a 2N sodium hydroxide solution and extracted with ethyl acetate; the organic phase was dried (Na2SO4) and concentrated to dryness to give pure 32b as an amorphous solid. Yield: 40%. 1H NMR (600 MHz, DMSO-d6) δ 13.50–13.15 (brs, 1H, NH), 7.62-7.58 (m, 1H, fluorophenyl H-6), 7.55-7.51 (m, 1H, fluorophenyl H-2), 7.49-7.45 (m, 1H, fluorophenyl H-5), 7.14-7.09 (m, 1H, fluorophenyl H-4), 5.15-4.90 (brs, 2H, NH2), 4.32 (q, J = 7.2 Hz, 2H, ethylester H-1), 1.33 (t, J = 7.2 Hz, 3H, ethylester H-2). 13C NMR (151 MHz, DMSO-d6) δ 163.23, 161.62 (fluorophenyl C-3), 160.32 (C=O), 136.90 (C-5), 135.21 (C-3), 133.44 (fluorophenyl C-1), 130.58, 130.53 (fluorophenyl C-5), 121.49 (fluorophenyl C-6), 118.59 (C-4), 113.46, 113.32 (fluorophenyl C-4), 112.08, 111.93 (fluorophenyl C-2), 59.88 (ethylester C-1), 14.28 (ethylester C-2). HR-MS (ESI) m/z: Calcd for C12H11FN3O2: [M-H]- = 248.0840, found 248.0836.
3-(3-Fluorophenyl)-1H-pyrazolo[4,3-d]pyrimidin-7(6H)-one (33b): To a solution of 32b (540 mg, 2.16 mmol) in butanol (8 mL), formamidine acetate (500 mg, 4.80 mmol) was added, and the solution was refluxed for 20 h. Upon completion of the reaction, chloroform (10 mL) was added into the flask, and the precipitate was filtered and dried to give pure 33b. Yield: 61%, m.p. > 250 °C (MeOH). 1H NMR (600 MHz, DMSO-d6) δ 14.70–14.24 (brs, 1H, pyrazole NH), 12.67–12.30 (brs, 1H, pyrimidinone NH), 8.15-8.13 (m, 1H, fluorophenyl H-6), 8.11-8.07 (m, 1H, fluorophenyl H-2), 8.00 (s, 1H, H-5), 7.56-7.51 (m, 1H, fluorophenyl H-5), 7.23-7.18 (m, 1H, fluorophenyl H-4). 13C NMR (151 MHz, DMSO-d6) δ 163.15, 161.54 (fluorophenyl C-3), 153.49 (C-7), 143.65 (C-5), 140.13 (C-3), 136.50 (C-3a), 133.91 (fluorophenyl C-1), 130.75, 130.69 (fluorophenyl C-5), 129.55 (C-7a), 121.86 (fluorophenyl C-6), 114.73, 114.59 (fluorophenyl C-4), 112.43, 112.27 (fluorophenyl C-2). HR-MS (ESI) m/z: Calcd for C11H6FN4O: [M-H]- = 229.0531, found 229.0540.
7-Chloro-3-phenyl-1H-pyrazolo[4,3-d]pyrimidine (34a): Compound 33a (100 mg, 0.47 mmol) was added under cooling to phosphorous oxychloride (1.5 mL). Then, N,N-diisopropylethylamine (122 µL, 0.71 mmol) was added, and the mixture was refluxed for 3 h under argon. The bulk of the volatile material was then vacuum-evaporated, water was added into the flask and the pH was adjusted to 5 upon addition of a saturated sodium bicarbonate solution. The precipitate was filtered, washed with water and air-dried. The residue was purified by silica gel column chromatography (cyclohexane/ethyl acetate: 7/3) to result in 34a (85 mg, 80%), m.p. 220–221 °C (acetone). 1H NMR (600 MHz, DMSO-d6) δ 14.80–14.55 (brs, 1H, NH), 8.94 (s, 1H, H-5), 8.43 (d, J = 7.7 Hz, 2H, phenyl H-2,6), 7.56 (t, J = 7.2 Hz, 2H, phenyl H-3,5), 7.46 (t, J = 7.5 Hz, 1H, phenyl H-4). 13C NMR (151 MHz, DMSO-d6) δ 150.43 (C-5), 143.94 (C-7), 143.02 (C-3), 142.28 (C-3a), 131.03 (C-7a), 130.65 (phenyl C-1), 128.79 (phenyl C-3,5), 128.76 (phenyl C-4), 126.28 (phenyl C-2,6). HR-MS (ESI) m/z: Calcd for C11H6ClN4: [M-H]- = 229.0286, found 229.0301.
7-Chloro-3-(3-fluorophenyl)-1H-pyrazolo[4,3-d]pyrimidine (34b): This compound was synthesized using a procedure analogous to that of 34a, starting from 33b. Purification was carried out by silica gel column chromatography (cyclohexane/ethyl acetate: 7/3). Yield: 80%, m.p. > 250 °C (EtOAc). 1H NMR (600 MHz, DMSO-d6) δ 13.80–13.54 (brs, 1H, NH), 8.96 (s, 1H, H-5), 8.30-8.27 (m, 1H, fluorophenyl H-6), 8.22-8.18 (m, 1H, fluorophenyl H-2), 7.65-7.59 (m, 1H, fluorophenyl H-5), 7.33-7.27 (m, 1H, fluorophenyl H-4). 13C NMR (151 MHz, DMSO-d6) δ 163.19, 161.58 (fluorophenyl C-3), 150.76 (C-5), 144.41 (C-7), 142.10 (C-3a), 141.52 (C-3), 133.18 (fluorophenyl C-1), 131.29 (C-7a), 131.08, 131.02 (fluorophenyl C-5), 122.27 (fluorophenyl C-6), 115.62, 115.48 (fluorophenyl C-4), 112.74, 112.58 (fluorophenyl C-2). HR-MS (ESI) m/z: Calcd for C11H5ClFN4: [M-H]- = 247.0192, found 247.1002.
N,3-Diphenyl-1H-pyrazolo[4,3-d]pyrimidin-7-amine (35a): Aniline (0.1 mL, 0.85 mmol) was added into a solution of the chloride 34a (80 mg, 0.34 mmol) in absolute ethanol (5 mL), and the mixture was refluxed under argon for 2 hrs. Upon completion of the reaction, water (20 mL) was added, and the precipitate was filtered under vacuum, washed with water and air-dried. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 9/1). Yield: 61%, m.p. 247–248 °C (EtOAc). 1H NMR (600 MHz, DMSO-d6) δ 13.20-12.90 (brs, 1H, pyrazole NH), 9.80-9.55 (brs, 1H, pyrimidinamine NH), 8.53 (brs, 1H, H-5), 8.44 (brs, 2H, 3-phenyl H-2,6), 7.93 (brs, 2H, N-phenyl H-2,6), 7.53 (t, J = 7.5 Hz, 2H, 3-phenyl H-3,5), 7.45-7.38 (m, 3H, N-phenyl H-3,5, 3-phenyl H-4), 7.13 (t, J = 7.4 Hz, 1H, N-phenyl H-4). 13C NMR (151 MHz, DMSO-d6) δ 151.42 (C-5), 146.78 (C-7), 142.13 (C-3), 139.74 (C-3a), 138.91 (N-phenyl C-1), 132.16 (3-phenyl C-1), 128.89 (N-phenyl C-3,5), 128.62 (3-phenyl C-3,5), 128.08 (3-phenyl C-4), 126.15 (3-phenyl C-2,6), 123.23 (N-phenyl C-4), 122.78 (C-7a), 120.10 (N-phenyl C-2,6). HR-MS (ESI) m/z: Calcd for C17H12N5: [M-H]- = 286.1098, found 286.1095. Anal. Calcd for C17H13N5: C, 71.06; H, 4.56; N, 24.38. Found: C, 70.88; H, 4.65; N, 24.44.
3-(3-Fluorophenyl)-Ν-phenyl-1H-pyrazolo[4,3-d]pyrimidin-7-amine (35b): This compound was synthesized using a procedure analogous to that of 35a starting from 34b. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 98/2). Yield: 65%, m.p. 237–238 °C (EtOAc). 1H NMR (600 MHz, DMSO-d6) δ 13.35-12.95 (brs, 1H, pyrazole NH), 9.85-9.60 (brs, 1H, pyrimidinamine NH), 8.55 (s, 1H, H-5), 8.30-8.26 (m, 1H, fluorophenyl H-6), 8.25-8.21 (m, 1H, fluorophenyl H-2), 7.93 (brs, 2H, phenyl H-2,6), 7.61-7.55 (m, 1H, fluorophenyl H-5), 7.43 (t, J = 7.4 Hz, 2H, phenyl H-3,5), 7.26-7.21 (m, 1H, fluorophenyl H-4), 7.13 (t, J = 7.4 Hz, 1H, phenyl H-4). 13C NMR (151 MHz, DMSO-d6) δ 163.18, 161.57 (fluorophenyl C-3), 151.65 (C-5), 146.79 (C-7), 140.81 (C-3), 139.73 (C-3a), 138.83 (phenyl C-1), 134.43 (fluorophenyl C-1), 130.65 (fluorophenyl C-5), 128.86 (phenyl C-3,5), 123.27 (phenyl C-4), 122.87 (C-7a), 121.99 (fluorophenyl C-6), 120.13 (phenyl C-2,6), 114.75, 114.61 (fluorophenyl C-4), 112.57, 112.42 (fluorophenyl C-2). HR-MS (ESI) m/z: Calcd for C17H11FN5: [M-H]- = 304.1003, found 304.0998. Anal. Calcd for C17H12FN5: C, 66.88; H, 3.96; N, 22.94. Found: C, 66.61; H, 4.02; N, 23.09.
3-Phenyl-N-(3,4,5-trimethoxyphenyl)-1H-pyrazolo[4,3-d]pyrimidin-7-amine (35c): This compound was synthesized using a procedure analogous to that of 35a starting from 34a. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 95/5). Yield: 68%, m.p. > 250 °C (EtOAc). 1H NMR (600 MHz, DMSO-d6) δ 13.10-12.80 (brs, 1H, pyrazole NH), 9.70-9.50 (brs, 1H, pyrimidinamine NH), 8.53 (s, 1H, H-5), 8.43 (brs, 2H, phenyl H-2,6), 7.53 (t, J = 7.1 Hz, 2H, phenyl H-3,5), 7.40 (t, J = 7.4 Hz, 1H, phenyl H-4), 7.35-7.20 (brs, 2H, trimethoxyphenyl H-2,6), 3.83 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.68 (s, 3H, trimethoxyphenyl CH3O-4). 13C NMR (151 MHz, DMSO-d6) δ 152.88 (trimethoxyphenyl C-3,5), 151.44 (C-5), 146.79 (C-7), 142.13 (C-3), 139.67 (C-3a), 134.96 (C-7a), 133.70 (trimethoxyphenyl C-4), 132.18 (phenyl C-1), 128.62 (phenyl C-3,5), 128.08 (phenyl C-4), 126.18 (phenyl C-2,6), 122.75 (trimethoxyphenyl C-1), 98.25 (trimethoxyphenyl C-2,6), 60.13 (trimethoxyphenyl CH3O-4), 55.88 (trimethoxyphenyl CH3O-3,5). HR-MS (ESI) m/z: Calcd for C20H18N5O3: [M-H]- =376.1415, found 376.1408. Anal. Calcd for C20H19N5O3: C, 63.65; H, 5.07; N, 18.56. Found: C, 63.90; H, 5.01; N, 18.69.
3-(3-Fluorophenyl)-N-(3,4,5-trimethoxyphenyl)-1H-pyrazolo[4,3-d]pyrimidin-7-amine (35d): This compound was synthesized using a procedure analogous to that of 35a starting from 34b. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 98/2). Yield: 57%, m.p. > 250 °C (EtOAc). 1H NMR (600 MHz, DMSO-d6) δ 13.12-12.96 (brs, 1H, pyrazole NH), 9.66-9.54 (brs, 1H, pyrimidinamine NH), 8.54 (s, 1H, H-5), 8.30-8.19 (m, 2H, fluorophenyl H-6, fluorophenyl H-2), 7.60-7.53 (m, 1H, fluorophenyl H-5), 7.30-7.19 (m, 3H, trimethoxyphenyl H-2,6, fluorophenyl H-4), 3.83 (s, 6H, trimethoxyphenyl CH3O-3,5), 3.68 (s, 3H, trimethoxyphenyl CH3O-4). 13C NMR (151 MHz, DMSO-d6) δ 163.19, 161.58 (fluorophenyl C-3), 152.85 (C-5), 151.73 (trimethoxyphenyl C-3,5), 146.79 (C-7), 140.82 (C-3), 139.65 (C-3a), 134.85 (C-7a), 133.78 (trimethoxyphenyl C-4), 130.76 (trimethoxyphenyl C-1, fluorophenyl C-5), 122.80 (fluorophenyl C-1), 121.97 (fluorophenyl C-6), 114.81, 114.67 (fluorophenyl C-4), 112.52, 112.36 (fluorophenyl C-2), 98.36 (trimethoxyphenyl C-2,6), 60.12 (trimethoxyphenyl CH3O-4), 55.88 (trimethoxyphenyl CH3O-3,5). HR-MS (ESI) m/z: Calcd for C20H19FN5O3: [M + H]+ = 396.1467, found 396.1455. Anal. Calcd for C20H18FN5O3: C, 60.75; H, 4.59; N, 17.71. Found: C, 60.94; H, 4.46; N, 17.59.
7-(4-Methylpiperazin-1-yl)-3-phenyl-1H-pyrazolo[4,3-d]pyrimidine (35e): This compound was synthesized using a procedure analogous to that of 35a starting from 34a. Upon completion of the reaction, the solvent was evaporated and water (20 mL) was added into the flask, followed by extraction with ethyl acetate (3 × 20 mL). The combined organic layers were dried (Na2SO4) and evaporated to dryness. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 9/1). Yield: 80%, m.p. 184–185 °C (EtOAc). 1H NMR (600 MHz, DMSO-d6) δ 8.35 (d, J = 7.9 Hz, 2H, phenyl H-2,6), 8.30 (s, 1H, H-5), 7.52 (t, J = 7.2 Hz, 2H, phenyl H-3,5), 7.38 (t, J = 7.5Hz, 1H, phenyl H-4), 4.25-4.10 (brs, 4H, methylpiperazine H-2,6), 2.49-2.46 (m, 4H, methylpiperazine H-3,5), 2.24 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, DMSO-d6) δ 152.80 (C-7), 151.80 (C-5), 138.33 (C-3a), 134.43 (C-3), 129.56 (C-7a, phenyl C-1), 128.76 (phenyl C-3,5), 128.01 (phenyl C-4), 125.73 (phenyl C-2,6), 54.53 (methylpiperazine C-3,5), 45.59 (methylpiperazine CH3), 44.89 (methylpiperazine C-2,6). HR-MS (ESI) m/z: Calcd for C16H17N6: [M-H]- =293.1520, found 293.1499. Anal. Calcd for C16H18N6: C, 65.29; H, 6.16; N, 28.55. Found: C, 65.44; H, 6.08; N, 28.31.
3-(3-Fluorophenyl)-7-(4-methylpiperazin-1-yl)-1H-pyrazolo[4,3-d]pyrimidine (35f): This compound was synthesized using a procedure analogous to that of 35a starting from 34b. The work-up procedure was similar to that of the derivative 35e. Purification was carried out by silica gel column chromatography (dichloromethane/methanol: 93/7). Yield: 80%, m.p. 212–213 °C (EtOAc). 1H NMR (600 MHz, DMSO-d6) δ 8.32 (s, 1H, H-5), 8.22-8.19 (m, 2H, fluorophenyl H-2, fluorophenyl H-6), 7.58-7.53 (m, 1H, fluorophenyl H-5), 7.22-7.18 (m, 1H, fluorophenyl H-4), 4.18-4.12 (brs, 4H, methylpiperazine H-2,6), 2.48-2.45 (m, 4H, methylpiperazine H-3,5), 2.23 (s, 3H, methylpiperazine CH3). 13C NMR (151 MHz, DMSO-d6) δ 163.17, 161.56 (fluorophenyl C-3), 152.52 (C-7), 152.03 (C-5), 138.82 (C-3a), 133.93 (C-3), 132.01, 131.96 (fluorophenyl C-5), 130.84, 130.78 (fluorophenyl C-1), 129.23 (C-7a), 121.58 (fluorophenyl C-6), 114.64, 114.50 (fluorophenyl C-4), 112.21, 112.06 (fluorophenyl C-2), 54.48 (methylpiperazine C-3,5), 45.55 (methylpiperazine CH3), 44.92 (methylpiperazine C-2,6). HR-MS (ESI) m/z: Calcd for C16H16FN6: [M-H]- =311.1425, found 311.1455. Anal. Calcd for C16H17FN6: C, 61.53; H, 5.49; N, 26.91. Found: C, 61.61; H, 5.36; N, 27.00.
3.3. Cell Viability Assays
The human HCT116 colon cancer cell line and PC-3 prostate cancer cell line were obtained from the American Type Cell Culture (ATCC, Bethesda, Md). Both cell lines were grown in 75 cm2 culture flasks at 37 °C in 5% CO2 using Roswell Park Memorial Institute 1640 medium (RPMI 1640, Gibco) and Dulbecco’s modified Eagle’s medium F/12 (DMEM/F-12, Gibco), respectively, containing 10% fetal bovine serum (FBS, Gibco). To test the inhibitory activities of compounds using a cell-based assay, MTT assay was performed for cell viability, as described previously [42]. Briefly, HCT116 cells were plated at a density of 1500 per well, while PC-3 cells were plated at a density of 750 per well in a 96-well plate. After 24 h, cells were treated with the indicated compounds in a dose-dependent manner for 72 h and 96 h (all tested compounds provided clear solutions). MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide] (Sigma M-5655) was added at a final concentration of 0.5 mg/mL directly to each well for 4 h at 37 °C. The medium was aspirated, and the blue MTT formazan precipitate was dissolved in dimethyl sulfoxide (DMSO). Absorbance was determined in a Powerwave microplate spectrophotometer (Biotek Instruments, Inc.) at 540 nm. Viable cell numbers were determined by tetrazolium conversion to its formazan dye. The IC50 was calculated by Microsoft Excel equation and confirmed by GraphPad Prism (7.0). Each experiment was performed in triplicate and mean values ± SD are reported.
3.4. DNA Staining and Flow Cytometric Analysis
Exponentially growing PC-3 cells were treated with the IC50 values of the compounds 14b, 26b and 35b or the corresponding DMSO concentration (vehicle) for 72 h. For cell cycle analysis, cell culture supernatants and attached cells were collected, centrifuged, washed in PBS, fixed in 50% ethanol and stained with an RNase-containing propidium iodide solution (50 μg/mL) (all reagents from Sigma). For cell apoptosis assay, cells were harvested and stained with Annexin V binding buffer, Annexin V–FITC and 7-AAD (Annexin V-FITC Apoptosis Detection Kit, BD Systems). DNA content was analyzed on a BD Accuri C6 Flow Cytometer using the BD CSampler software (BD Biosciences, USA). Non-apoptotic events were used to calculate the percentage of cells distributed in each phase. A p value < 0.05 was considered to be statistically significant (Student’s t-test).
4. Conclusions
In conclusion, we have designed and synthesized a number of new substituted pyrrolo[2,3-c]pyridines, pyrrolo[3,2-d]pyrimidines and pyrazolo[4,3-d]pyrimidines and have evaluated their antiproliferative activity against two cancer cell lines. We preserved an analogous substitution pattern around the fused ring-system of all three scaffolds in order to assist the extraction of SARs. We have identified a number of derivatives with potent cytotoxic properties—more precisely, the pyrrolopyridines 14a–f, the pyrrolopyrimidines 26a,b and the pyrazolopyrimidines 35a,b, all of which possess IC50 values in the nM to low µM level against both cell lines tested. We assume that in all cases the simultaneous presence of a 3-fluorophenyl moiety and a phenylamino substituent at analogous positions resulted in highly active compounds which induce apoptosis in PC-3 cells and, importantly, are devoid of toxicity against normal cells. These interesting observations have triggered the continuation of research efforts in our laboratory for the investigation of the exact mechanism of action of this promising class of compounds.
Supplementary Materials
The following supporting information can be downloaded online: Figures S1–S55: 1H-NMR and 13C NMR spectra of new compounds, HRMS spectra of active compounds; Table S1: Calculated physicochemical characteristics of the tested compounds using SwissADME.
Author Contributions
Conceptualization, P.M. and N.P.; methodology, S.D., E.-S.G., A.P., N.L., K.G. and R.T.; validation, N.L., R.T., P.M. and N.P.; formal analysis, S.D., E.-S.G., A.P. and K.G.; data curation, N.L., P.M., N.P. and R.T.; writing—original draft preparation, N.L., P.M., N.P. and R.T.; writing—review and editing, N.L., P.M., N.P. and R.T.; supervision, P.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Acknowledgments
The authors are grateful to Emmanuel Mikros who performed the physicochemical calculations and participated in useful discussion concerning the evaluation of the results of this work.
Conflicts of Interest
The authors declare no conflict of interest.
Sample Availability
Samples of the compounds 14a-f, 26a-f, 35a-f are available from the authors.
References
- Motati, D.R.; Amaradhi, R.; Ganesh, T. Azaindole therapeutic agents. Bioorg. Med. Chem. 2020, 28, 115830. [Google Scholar] [CrossRef] [PubMed]
- Irie, T.; Sawa, M. 7-Azaindole: A Versatile Scaffold for Developing Kinase Inhibitors. Chem. Pharm. Bull. 2018, 66, 29–36. [Google Scholar] [CrossRef] [Green Version]
- Han, Y.; Dong, W.; Guo, Q.; Li, X.; Huang, L. The importance of indole and azaindole scaffold in the development of antitumor agents. Eur. J. Med. Chem. 2020, 203, 112506. [Google Scholar] [CrossRef] [PubMed]
- Maemoto, T.; Finlayson, K.; Olverman, H.J.; Akahane, A.; Horton, R.W.; Butcher, S.P. Species differences in brain adenosine A1 receptor pharmacology revealed by use of xanthine and pyrazolopyridine based antagonists. Br. J. Pharmacol. 1997, 122, 1202–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamajima, T.; Takahasi, F.; Kato, K.; Sugano, Y.; Yamaki, S.; Moritomo, A.; Kubo, S.; Nakamura, K.; Yamagami, K.; Hamakawa, N.; et al. Optimization and in vivo evaluation of pyrazolopyridines as a potent and selective PI3Kδ inhibitor. Bioorg. Med. Chem. 2018, 26, 3917–3924. [Google Scholar] [CrossRef]
- Eissa, I.H.; El-Naggar, A.M.; El-Hashash, M.A. Design, synthesis, molecular modeling and biological evaluation of novel 1H-pyrazolo[3,4-b]pyridine derivatives as potential anticancer agents. Bioorg. Chem. 2016, 67, 43–56. [Google Scholar] [CrossRef]
- Michailidou, M.; Giannouli, V.; Kotsikoris, V.; Papadodima, O.; Kontogianni, G.; Kostakis, I.K.; Lougiakis, N.; Chatziioannou, A.; Kolisis, F.N.; Marakos, P.; et al. Novel pyrazolopyridine derivatives as potential angiogenesis inhibitors: Synthesis, biological evaluation and transcriptome-based mechanistic analysis. Eur. J. Med. Chem. 2016, 121, 143–157. [Google Scholar] [CrossRef]
- Pathania, S.; Rawal, R.K. Pyrrolopyrimidines: An update on recent advancements in their medicinal attributes. Eur. J. Med. Chem. 2018, 157, 503–526. [Google Scholar] [CrossRef]
- El-Gohary, N.S.; Shaaban, M.I. New pyrazolopyridine analogs: Synthesis, antimicrobial, antiquorum-sensing and antitumor screening. Eur. J. Med. Chem. 2018, 152, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Salem, M.S.; Ali, M.A.M. Novel Pyrazolo[3,4-b]pyridine Derivatives: Synthesis, Characterization, Antimicrobial and Antiproliferative Profile. Biol. Pharm. Bull. 2016, 39, 473–483. [Google Scholar] [CrossRef] [Green Version]
- McGowan, D.C.; Balemans, W.; Embrechts, W.; Motte, M.; Keown, J.R.; Buyck, C.; Corbera, J.; Funes, M.; Moreno, L.; Cooymans, L.; et al. Design, Synthesis, and Biological Evaluation of Novel Indoles Targeting the Influenza PB2 Cap Binding Region. J. Med. Chem. 2019, 62, 9680–9690. [Google Scholar] [CrossRef] [PubMed]
- Patnaik, S.; Basu, D.; Southall, N.; Dehdashti, S.; Wan, K.K.; Zheng, W.; Ferrer, M.; Taylor, M.; Engel, D.A.; Marugan, J.J. Identification, design and synthesis of novel pyrazolopyridine influenza virus nonstructural protein 1 antagonists. Bioorg. Med. Chem. Lett. 2019, 29, 1113–1119. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, L.C.S.; Feitosa, L.M.; Gandi, M.O.; Silveira, F.F.; Boechat, N. The Development of Novel Compounds Against Malaria: Quinolines, Triazolpyridines, Pyrazolopyridines and Pyrazolopyrimidines. Molecules 2019, 24, 4095. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anand, D.; Yadav, P.K.; Patel, O.P.S.; Parmar, N.; Maurya, R.K.; Vishwakarma, P.; Raju, K.S.R.; Taneja, I.; Wahajuddin, M.; Kar, S.; et al. Antileishmanial Activity of Pyrazolopyridine Derivatives and Their Potential as an Adjunct Therapy with Miltefosine. J. Med. Chem. 2017, 60, 1041–1059. [Google Scholar] [CrossRef]
- Pfefferkorn, J.A.; Tu, M.; Filipski, K.J.; Guzman-Perez, A.; Bian, J.; Aspnes, G.E.; Sammons, M.F.; Song, W.; Li, J.C.; Jones, C.S.; et al. The design and synthesis of indazole and pyrazolopyridine based glucokinase activators for the treatment of type 2 diabetes mellitus. Bioorg. Med. Chem. Lett. 2012, 22, 7100–7105. [Google Scholar] [CrossRef] [PubMed]
- da Silva Lima, C.H.; de Araujo Vanelis Soares, J.C.; de Sousa Ribeiro, J.L.; Muri, E.M.F.; de Albuquerque, S.; Dias, L.R.S. Anti-Trypanosoma cruzi Activity and Molecular Docking Studies of 1H-pyrazolo[ 3, 4-b]pyridine Derivatives. Lett. Drug Des. Discov. 2020, 17, 184–191. [Google Scholar] [CrossRef]
- Jouha, J.; Loubidi, M.; Bouali, J.; Hamri, S.; Hafid, A.; Suzenet, F.; Guillaumet, G.; Dagcı, T.; Khouili, M.; Aydın, F.; et al. Synthesis of new heterocyclic compounds based on pyrazolopyridine scaffold and evaluation of their neuroprotective potential in MPP+-induced neurodegeneration. Eur. J. Med. Chem. 2017, 129, 41–52. [Google Scholar] [CrossRef]
- Pennington, L.D.; Moustakas, D.T. The necessary nitrogen atom: A versatile high-impact design element for multiparameter optimization. J. Med. Chem. 2017, 60, 3552–3579. [Google Scholar] [CrossRef]
- Bedard, P.L.; Hyman, D.M.; Davids, M.S.; Siu, L.L. Small molecules, big impact: 20 years of targeted therapy in oncology. Lancet 2020, 395, 1078–1088. [Google Scholar] [CrossRef]
- Agianian, B.; Gavathiotis, E. Current Insights of BRAF Inhibitors in Cancer. J. Med. Chem. 2018, 61, 5775–5793. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Lagarón, N.O.; McGowan, E.M.; Parmar, I.; Jha, A.; Hubbard, B.P.; Vasantha Rupasinghe, H.P. Kinase-targeted cancer therapies: Progress, challenges and future directions. Mol. Cancer 2018, 17, 48. [Google Scholar] [CrossRef] [PubMed]
- Giannouli, V.; Lougiakis, N.; Kostakis, I.K.; Pouli, N.; Marakos, P.; Skaltsounis, A.-L.; Nam, S.; Jove, R.; Horne, D.; Tenta, R.; et al. The discovery of new cytotoxic pyrazolopyridine derivatives. Bioorg. Med. Chem. Lett. 2016, 26, 5229–5233. [Google Scholar] [CrossRef]
- Gavriil, E.-S.; Lougiakis, N.; Pouli, N.; Marakos, P.; Skaltsounis, A.-L.; Nam, S.; Jove, R.; Horne, D.; Gioti, K.; Pratsinis, H.; et al. Synthesis and antiproliferative activity of new pyrazolo[3,4-c]pyridines. Med. Chem. 2017, 13, 365–374. [Google Scholar] [CrossRef]
- Gavriil, E.-S.; Doukatas, A.; Karampelas, T.; Myrianthopoulos, V.; Dimitrakis, S.; Mikros, E.; Marakos, P.; Tamvakopoulos, C.; Pouli, N. Design, Synthesis and Biological Evaluation of Novel Substituted Purine Isosters as EGFR kinase inhibitors, with promising pharmacokinetic profile and in vivo efficacy. Eur. J. Med. Chem. 2019, 176, 393–409. [Google Scholar] [CrossRef]
- Papastathopoulos, A.; Lougiakis, N.; Kostakis, I.K.; Marakos, P.; Pouli, N.; Pratsinis, H.; Kletsas, D. New bioactive 5-arylcarboximidamidopyrazolo[3,4-c]pyridines: Synthesis, cytotoxic activity, mechanistic investigation and structure-activity relationships. Eur. J. Med. Chem. 2021, 218, 113387. [Google Scholar] [CrossRef]
- Brown, E.V. Syntheses and Decarboxylation of the Isomeric Nitropyridinecarboxylic Acids. J. Am. Chem. Soc. 1954, 76, 3167–3168. [Google Scholar] [CrossRef]
- Linz, S.; Müller, J.; Hübner, H.; Gmeiner, P.; Troschütz, R. Design, synthesis and dopamine D4 receptor binding activities of new N-heteroaromatic 5/6-ring Mannich bases. Bioorg. Med. Chem. 2009, 17, 4448–4458. [Google Scholar] [CrossRef]
- Theoclitou, M.-E.; Aquila, B.; Block, M.H.; Brassil, P.J.; Castriotta, L.; Code, E.; Collins, M.P.; Davies, A.M.; Deegan, T.; Ezhuthachan, J.; et al. Discovery of (+)-N-(3-Aminopropyl)-N-[1-(5-benzyl-3-methyl-4-oxo-[1,2]thiazolo[5,4-d]pyrimidin-6-yl)-2-methylpropyl]-4-methylbenzamide (AZD4877), a Kinesin Spindle Protein Inhibitor and Potential Anticancer Agent. J. Med. Chem. 2011, 54, 6734–6750. [Google Scholar] [CrossRef] [PubMed]
- Furneaux, R.H.; Tyler, P.C. Improved Syntheses of 3H,5H-Pyrrolo[3,2-d]pyrimidines. J. Org. Chem. 1999, 64, 8411–8412. [Google Scholar] [CrossRef]
- Haraguchi, K.; Horii, C.; Yoshimura, Y.; Ariga, F.; Tadokoro, A.; Tanaka, H. An Access to the β-Anomer of 4′-Thio-C-ribonucleosides: Hydroboration of 1-C-Aryl- or 1-C-Heteroaryl-4-thiofuranoid Glycals and Its Regiochemical Outcome. J. Org. Chem. 2011, 76, 8658–8669. [Google Scholar] [CrossRef]
- Kaiser, M.M.; Baszczyňski, O.; Hocková, D.; Poštová-Slavětínská, L.; Dračínský, M.; Keough, D.T.; Guddat, L.W.; Janeba, Z. Acyclic Nucleoside Phosphonates Containing 9-Deazahypoxanthine and a Five-Membered Heterocycle as Selective Inhibitors of Plasmodial 6-Oxopurine Phosphoribosyltransferases. Chem. Med. Chem. 2017, 12, 1133–1141. [Google Scholar] [CrossRef]
- Garel, L.; Saint-Jalmes, L. One-pot fluoro-de-diazoniation of anilines in organic medium. Tet. Lett. 2006, 47, 5705–5708. [Google Scholar] [CrossRef]
- Tabrizi, M.A.; Baraldi, P.G.; Baraldi, S.; Ruggiero, E.; De Stefano, L.; Rizzolio, F.; Di Cesare Mannelli, L.; Ghelardini, C.; Chicca, A.; Lapillo, M.; et al. Discovery of 1,5-Diphenylpyrazole-3-Carboxamide Derivatives as Potent, Reversible, and Selective Monoacylglycerol Lipase (MAGL) Inhibitors. J. Med. Chem. 2018, 61, 1340–1354. [Google Scholar] [CrossRef] [Green Version]
- Hoveyda, H.R.; Roy, M.-O.; Blanc, S.; Noël, S.; Salvino, J.M.; Ator, M.A.; Fraser, G. Discovery of 3-aryl-5-acylpiperazinyl-pyrazoles as antagonists to the NK3 receptor. Bioorg. Med. Chem. Lett. 2011, 21, 1991–1996. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Gulianello, M.; De Lombaert, S.; Brodbeck, R.; Kieltyka, A.; Hodgetts, K.J. 3-Aryl pyrazolo[4,3-d]pyrimidine derivatives: Nonpeptide CRF-1 antagonists. Bioorg. Med. Chem. Lett. 2002, 12, 2133–2136. [Google Scholar] [CrossRef]
- Majid, T.; Hopkins, C.R.; Pedgrift, B.; Collar, N. Convenient synthesis of 4-amino-3,5-disubstituted pyrazoles in one step from the corresponding diketo oximes. Tet. Lett. 2004, 45, 2137–2139. [Google Scholar] [CrossRef]
- Geffken, D.; Soliman, R.; Soliman, F.S.G.; Abdel-Khalek, M.M.; Issa, D.A.E. Synthesis of new series of pyrazolo[4,3-d]pyrimidin-7-ones and pyrido[2,3-d]pyrimidin-4-ones for their bacterial and cyclin-dependent kinases (CDKs) inhibitory activities. Med. Chem. Res. 2011, 20, 408–420. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef] [Green Version]
- SIMCA, version 14.1; Umetrics: Malmö, Sweden, 2015.
- Gioti, K.; Papachristodoulou, A.; Benaki, D.; Beloukas, A.; Vontzalidou, A.; Aligiannis, N.; Skaltsounis, A.L.; Mikros, E.; Tenta, R. Glycyrrhiza glabra-Enhanced Extract and Adriamycin Antiproliferative Effect on PC-3 Prostate Cancer Cells. Nutr. Cancer 2020, 72, 320–332. [Google Scholar] [CrossRef]
- Hanney, B.; Manley, P.; Rudd, M.T.; Sanders, J.M.; Stachel, S.J.; Henze, D. Merck Sharp & Dohme Corp. WO2013/9582 (A1), 17 January 2013.
- Vistica, D.T.; Skehan, P.; Scudiero, D.; Monks, A.; Pittman, A.; Boyd, M.R. Tetrazolium-based assays for cellular viability: A critical examination of selected parameters affecting formazan production. Cancer Res. 1991, 51, 2515–2520. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).