Quantitative Analysis and Differential Evaluation of Radix Bupleuri Cultivated in Different Regions Based on HPLC-MS and GC-MS Combined with Multivariate Statistical Analysis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Differential Analysis of Five Saikosaponins in Radix Bupleuri Samples Cultivated in Different Regions
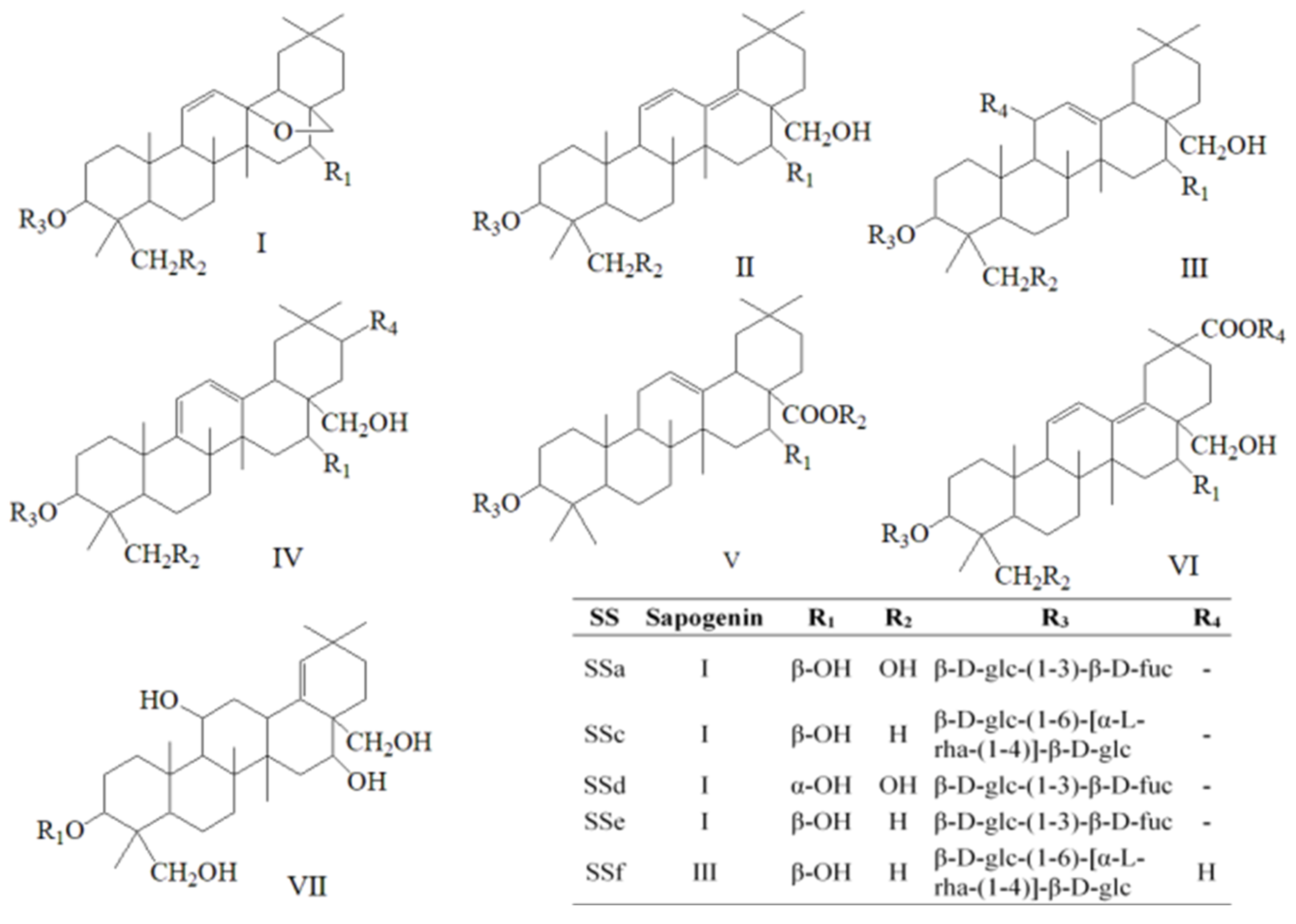
2.1.1. Identification of Five Saikosaponins in Radix Bupleuri
2.1.2. Validation of Quantitative Method
2.1.3. Quantitative Analysis of Five Saikosaponins in Radix Bupleuri Cultivated in Different Regions
2.2. Differential Analysis of Radix Bupleuri from Different Cultivation Regions Based on HPLC-MS and GC-MS Analysis
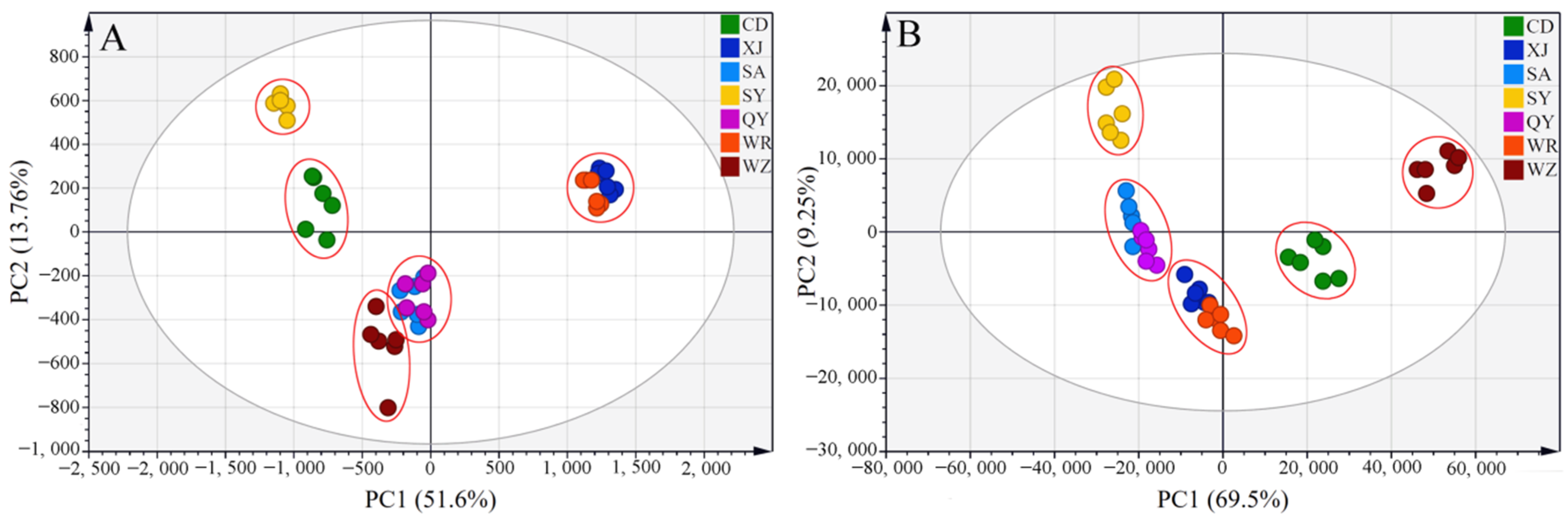
2.2.1. Principal Component Analysis
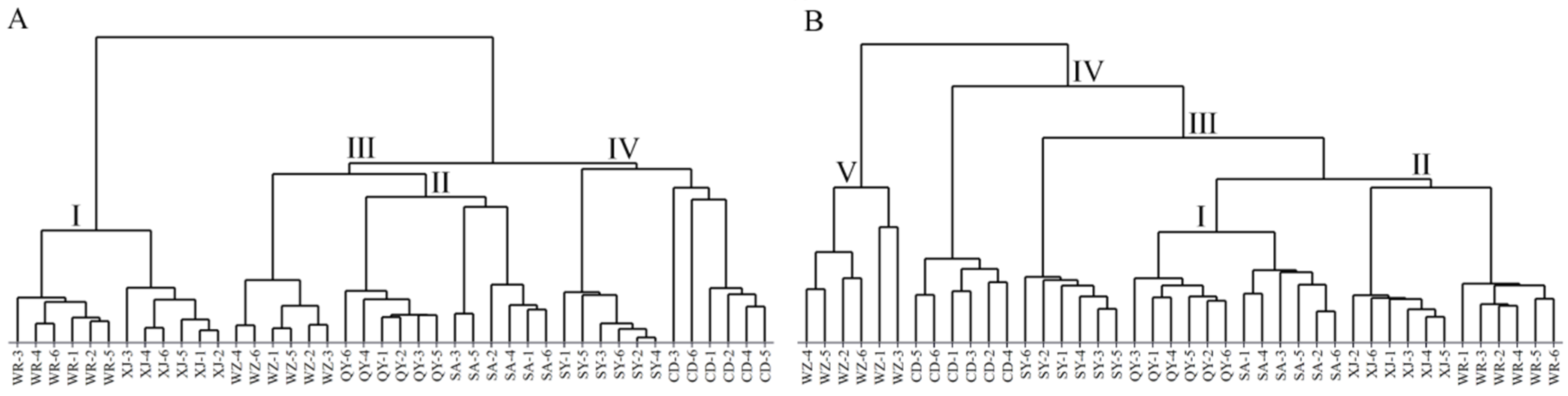
2.2.2. Hierarchical Cluster Analysis
2.2.3. Partial Least Squares-Discriminate Analysis
3. Materials and Methods
3.1. Materials and Chemicals
3.2. Preparation of Sample and Standard Solutions
3.2.1. Extraction of Saikosaponins from Radix Bupleuri
3.2.2. Extraction of Volatile Compounds from Radix Bupleuri
3.2.3. Preparation of Standard Solutions
3.3. Chromatography-Based MS Conditions
3.3.1. HPLC-MS Conditions
3.3.2. GC-MS Conditions
3.4. Validation of Methodology
3.5. Data Processing and Statistical Analysis
3.6. General Process of Compound Identification
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jiang, H.; Yang, L.; Hou, A.; Zhang, J.; Wang, S.; Man, W.; Zheng, S.; Yu, H.; Wang, X.; Yang, B.; et al. Botany, traditional uses, phytochemistry, analytical methods, processing, pharmacology and pharmacokinetics of Bupleuri Radix: A systematic review. Biomed. Pharmacother. 2020, 131, 110679. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, X.; Huang, N.; Liu, R.; Sun, R. A comprehensive review and perspectives on pharmacology and toxicology of saikosaponins. Phytomedicine 2018, 50, 73–87. [Google Scholar] [CrossRef]
- Yang, L.; Zhao, Y.; Zhang, Q.; Cheng, L.; Han, M.; Ren, Y.; Yang, L. Effects of drought-re-watering-drought on the photosynthesis physiology and secondary metabolite production of Bupleurum chinense DC. Plant Cell Rep. 2019, 38, 1181–1197. [Google Scholar] [CrossRef]
- Yang, L.-L.; Yang, L.; Yang, X.; Zhang, T.; Lan, Y.-M.; Zhao, Y.; Han, M.; Yang, L.-M. Drought stress induces biosynthesis of flavonoids in leaves and saikosaponins in roots of Bupleurum chinense DC. Phytochemistry 2020, 177, 112434. [Google Scholar] [CrossRef]
- Zhang, W.; Gong, J.; Zhao, J.; Li, Y.; Liu, M.; Yang, L.; Yang, B.; Zhang, Z. What degree of light deficiency is suitable for saikosaponin accumulation by Bupleurum chinense DC. Ind. Crop. Prod. 2018, 122, 392–401. [Google Scholar] [CrossRef]
- Sun, P.; Li, Y.; Wei, S.; Zhao, T.; Wang, Y.; Song, C.; Xue, L.; Wang, F.; Xiao, L.; Wu, J.; et al. Pharmacological Effects and Chemical Constituents of Bupleururn. Mini-Rev. Med. Chem. 2019, 19, 34–55. [Google Scholar] [CrossRef]
- Xia, Z.; Liu, X.; Tong, L.; Wang, H.; Feng, M.; Xi, X.; He, P.; Qin, X. Comparison of chemical constituents of Bupleurum marginatum var. stenophyllum and Bupleurum chinense DC. using UHPLC-Q-TOF-MS based on a metabonomics approach. Biomed. Chromatogr. 2021, 35, e5133. [Google Scholar] [CrossRef]
- Lu, C.N.; Yuan, Z.G.; Zhang, X.L.; Yan, R.; Zhao, Y.Q.; Liao, M.; Chen, J.X. Saikosaponin a and its epimer saikosaponin d exhibit anti-inflammatory activity by suppressing activation of NF-κB signaling pathway. Int. J. Immunopharmacol. 2012, 14, 121–126. [Google Scholar] [CrossRef]
- Wang, A.; Mi, L.; Zhang, Z.; Hu, M.; Zhao, Z.; Liu, B.; Li, Y.; Zheng, S. Saikosaponin A improved depression -like behavior and inhibited hippocampal neuronal apoptosis after cerebral ischemia through p-CREB/BDNF pathway. Behav. Brain Res. 2021, 403, 113138. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.O.; Park, J.Y.; Jeon, S.Y.; Yang, C.H.; Kim, M.R. Saikosaponin a, an active compound of Radix Bupleuri, attenuates inflammation in hypertrophied 3T3-L1 adipocytes via ERK/NF-κB signaling pathways. Int. J. Mol. Cell. Med. 2015, 35, 1126–1132. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.F.; Dai, Z.J.; Wang, X.J.; Bai, M.H.; Lin, S.; Ma, H.B.; Wang, Y.L.; Song, L.Q.; Ma, X.L.; Zan, Y.; et al. Saikosaponin-d increases the radiosensitivity of smmc-7721 hepatocellular carcinoma cells by adjusting the g0/g1 and g2/m checkpoints of the cell cycle. BMC Complem. Altern. Med. 2013, 13, 263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, W.; Cheng, X.; Kang, R.; Wang, Y.; Guo, X.; Jing, W.; Wei, F.; Ma, S. Corrigendum: Systematic Characterization and Identification of Saikosaponins in Extracts from Bupleurum marginatum var. stenophyllum Using UPLC-PDA-Q/TOF-MS. Front. Chem. 2022, 10, 867617. [Google Scholar] [CrossRef] [PubMed]
- The State Pharmacopoeia Commission of P. R. China. Pharmacopoeia of the People’s Repubilic of China; Chemical Industry Press: Beijing, China, 2020; Volume 1, p. 232. [Google Scholar]
- Ashour, M.L.; Wink, M. Genus Bupleurum: A review of its phytochemistry, pharmacology and modes of action. J. Pharm. Pharmacol. 2011, 63, 305–321. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Lu, S.; Su, Y.; Guo, Y. Analysis of Volatile Compounds in Radix Bupleuri Injection by GC-MS-MS. Chromatographia 2011, 74, 497–502. [Google Scholar] [CrossRef]
- Chen, W.; Balan, P.; Popovich, D.G. Ginsenosides analysis of New Zealand-grown forest Panax ginseng by LC-QTOF-MS/MS. J. Ginseng Res. 2020, 44, 552–562. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, Q.; Sun, X.; Li, X.; Miao, R.; Wu, D.; Liu, S.; Xiu, Y. Discrimination of Ginseng Origins and Identification of Ginsenoside Markers Based on HPLC-MS Combined with Multivariate Statistical Analysis. Chem. J. Chin. Univ.-Chin. 2019, 40, 246–253. [Google Scholar] [CrossRef]
- Xiu, Y.; Li, X.; Sun, X.; Xiao, D.; Miao, R.; Zhao, H.; Liu, S. Simultaneous determination and difference evaluation of 14 ginsenosides in Panax ginseng roots cultivated in different areas and ages by high-performance liquid chromatography coupled with triple quadrupole mass spectrometer in the multiple reaction-monitoring mode combined with multivariate statistical analysis. J. Ginseng Res. 2019, 43, 508–516. [Google Scholar] [CrossRef]
- Li, X.Q.; He, Z.G.; Bi, K.S.; Song, Z.H.; Xu, L. Essential oil analyses of the root oils of 10 Bupleurum species from China. J. Essent. Oil Res. 2007, 19, 234–238. [Google Scholar] [CrossRef]
- Tykheev, Z.A.; Taraskin, V.V.; Radnaeva, L.D.; Zhang, F.Q.; Chen, S.L. Composition of Lipids from Roots of Bupleurum scorzonerifolium and B. chinense. Chem. Nat. Compd+ 2017, 53, 937–938. [Google Scholar] [CrossRef]
- Saracoglu, H.T.; Zengin, G.; Akin, M.; Aktumsek, A. Evaluation of oil content and fatty acid composition of five endemic Bupleurum species growing in the Central Anatolia region of Turkey. Nat. Prod. Res. 2012, 26, 1188–1194. [Google Scholar] [CrossRef]
- Tykheev, Z.A.; Zhigzhitzhapova, S.V.; Zhang, F.; Taraskin, V.V.; Anenkhonov, O.A.; Radnaeva, L.D.; Chen, S. Constituents of Essential Oil and Lipid Fraction from the Aerial Part of Bupleurum scorzonerifolium Willd. (Apiaceae) from Different Habitats. Molecules 2018, 23, 1496. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiu, Y.; Zhao, H.; Gao, Y.; Liu, W.; Liu, S. Chemical transformation of ginsenoside Re by a heteropoly acid investigated using HPLC-MSn/HRMS. New J. Chem. 2016, 40, 9073–9080. [Google Scholar] [CrossRef]
- Chen, J.F.; Guo, X.Y.; Song, Y.L.; Zhao, M.B.; Tu, P.F.; Jiang, Y. MRM-based strategy for the homolog-focused detection of minor ginsenosides from notoginseng total saponins by ultra-performance liquid chromatography coupled with hybrid triple quadrupole-linear ion trap mass spectrometry. RSC Adv. 2016, 6, 96376–96388. [Google Scholar] [CrossRef]
- Yen, M.H.; Lin, C.C.; Yen, C.M. The immunomodulatory effect of saikosaponin derivatives and the root extract of Bupleurum kaoi in mice. Phytother. Res. 1995, 9, 351–358. [Google Scholar] [CrossRef]
- Liu, C.T.; Wu, C.Y.; Weng, Y.M.; Tseng, C.Y. Ultrasound-assisted extraction methodology as a tool to improve the antioxidant properties of herbal drug Xiao-chia-hu-tang. J. Ethnopharmacol. 2005, 99, 293–300. [Google Scholar] [CrossRef]
- Meng, J.; Chen, X.F.; Yang, W.Y.; Song, J.H.; Zhang, Y.; Li, Z.F.; Yang, X.W.; Yang, Z.G. Gas chromatography-mass spectrometry analysis of essential oils from five parts of Chaihu (Radix Bupleuri Chinensis). J. Tradit. Chin. Med. 2014, 34, 741–748. [Google Scholar] [CrossRef]
- Xing, J.; Sun, H.M.; Li, Z.Y.; Qin, X.M. Comparison of Volatile Components between Raw and Vinegar Baked Radix Bupleuri by GC-MS Based Metabolic Fingerprinting Approach. Evid.-Based Complement. Altern. Med. 2015, 2015, 653791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shan, S.M.; Luo, J.G.; Huang, F.; Kong, L.Y. Chemical characteristics combined with bioactivity for comprehensive evaluation of Panax ginseng C.A. Meyer in different ages and seasons based on HPLC-DAD and chemometric methods. J. Pharmaceut. Biomed. 2014, 89, 76–82. [Google Scholar] [CrossRef]
- Lei, T.; Chen, S.; Wang, K.; Zhang, D.; Dong, L.; Lv, C.; Wang, J.; Lu, J. Characterization and discrimination of raw and vinegar-baked Bupleuri radix based on UHPLC-Q-TOF-MS coupled with multivariate statistical analysis. Biomed. Chromatogr. 2018, 32, e4044. [Google Scholar] [CrossRef]
- Zhu, L.; Liang, Z.T.; Yi, T.; Ma, Y.; Zhao, Z.Z.; Guo, B.L.; Zhang, J.Y.; Chen, H.B. Comparison of chemical profiles between the root and aerial parts from three Bupleurum species based on a UHPLC-QTOF-MS metabolomics approach. BMC Complement. Altern. Med. 2017, 17, 305. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.Q.; Zhang, X.; Lin, M.; Shen, Y.H.; Yan, S.K.; Zhang, W.D. Characterization and identification of saikosaponins in crude extracts from three Bupleurum species using LC-ESI-MS. J. Sep. Sci. 2008, 31, 3190–3201. [Google Scholar] [CrossRef] [PubMed]
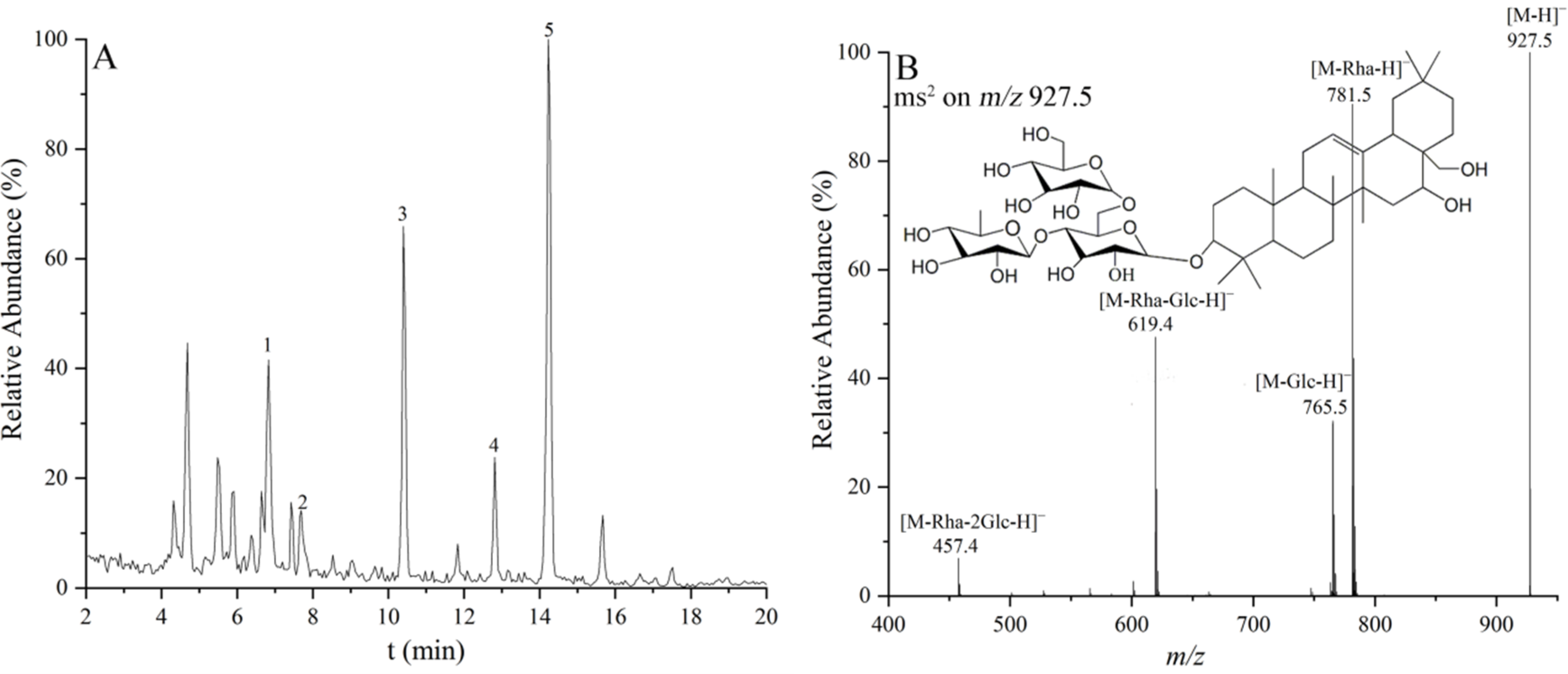




| Peak | tR (min) | Identification | Measured Mass | Molecular Formula | Product Ions |
|---|---|---|---|---|---|
| 1 | 6.83 | SSc | 926.5 | C48H78O17 | 925.5 [M-H]−, 779.5 [M-Rha-H]−, 763.5 [M-Glc-H]−, 617.4 [M-Rha-Glc-H]−, 455.6 [M-Rha-2Glc-H]− |
| 2 | 7.69 | SSf | 928.5 | C48H80O17 | 927.5 [M-H]−, 781.5 [M-Fuc-H]−, 619.4 [M-Fuc-Glc-H]−, 457.4 [M-Fuc-2Glc-H]− |
| 3 | 10.39 | SSa | 780.5 | C42H68O13 | 779.5 [M-H]−, 617.4 [M-Glc-H]−, 471.4 [M-Glc-Fuc-H]− |
| 4 | 12.81 | SSe | 764.5 | C42H68O12 | 763.5 [M-H]−, 601.4 [M-Glc-H]−, 455.4 [M-Rha-2Glc-H]− |
| 5 | 14.22 | SSd | 780.5 | C42H68O13 | 779.5 [M-H]−, 617.4 [M-Glc-H]−, 471.6 [M-Glc-Fuc-H]− |
| Saikosaponin | Calibration Curve | R2 | Linear Range (μg/mL) | LOD (μg/mL) | LOQ (μg/mL) |
|---|---|---|---|---|---|
| SSa | y = 13,888.3 + 92,655.3x | 0.9986 | 0.1–100.0 | 0.009 | 0.030 |
| SSc | y = 64,383 + 755,798x | 0.9974 | 0.05–10.0 | 0.006 | 0.020 |
| SSd | y = 36,478.4 + 89,231.8x | 0.9953 | 0.1–100.0 | 0.015 | 0.050 |
| SSe | y = 133,789 + 313,062x | 0.9949 | 0.03–3.0 | 0.009 | 0.030 |
| SSf | y = 46,408.3 + 909,911x | 0.9966 | 0.05–10.0 | 0.005 | 0.017 |
| Saikosaponin | Intraday Precision RSD (n = 9, %) | Interday Precision RSD (n = 6, %) | Repeatability RSD (n = 6, %) | Stability RSD (n = 7, %) | Recovery (%)/RSD (n = 3, %) | ||
|---|---|---|---|---|---|---|---|
| 80% | 100% | 120% | |||||
| SSa | 2.93 | 1.78 | 2.04 | 1.98 | 101.32/1.57 | 101.96/2.03 | 99.63/2.19 |
| SSc | 2.06 | 1.90 | 1.68 | 1.53 | 100.91/1.95 | 102.46/1.47 | 95.55/2.09 |
| SSd | 1.53 | 2.23 | 1.46 | 1.46 | 101.13/1.32 | 102.45/1.22 | 98.10/1.82 |
| SSe | 3.14 | 2.77 | 3.23 | 3.28 | 97.47/1.65 | 103.20/2.59 | 98.46/1.48 |
| SSf | 3.45 | 3.23 | 3.18 | 2.89 | 98.83/2.61 | 102.05/1.78 | 96.12/2.57 |
| No. | tR (min) | VIP | Compound Name | [M-H]− Ion (m/z) | Fragment Ions (m/z) | Molecular Formula | References |
|---|---|---|---|---|---|---|---|
| 1 | 4.01 | 2.0 | 3′,4′-dimethoxy quercetin | 329 | 314, 299 | C18H34O5 | [30] |
| 2 | 4.17 | 1.3 | hydroxy-SSc or its isomer | 943 | 811, 649, 503, 471, 453 | C48H80O18 | [12] |
| 3 | 4.49 | 1.6 | rotundioside w | 941 | 795, 777 | C48H78O18 | [31] |
| 4 | 4.86 | 1.6 | hydroxy-SSa | 797 | 635, 559, 489 | C42H70O14 | [12] |
| 5 | 6.57 | 3.3 | SSi | 925 | 779, 763, 617 | C48H78O17 | [12] |
| 6 | 6.69 | 3.3 | SSc | 971 | 925, 779, 763, 617, 455 | C48H78O17 | [12] |
| 7 | 6.77 | 1.2 | rotundioside n | 941 | 779, 763, 618 | C48H78O18 | [31] |
| 8 | 7.14 | 1.7 | hydroxy-SSd | 797 | 635, 559, 489 | C42H70O14 | [12] |
| 9 | 7.56 | 5.2 | SSf | 973 | 927, 781, 765, 619, 457 | C48H80O17 | [12] |
| 10 | 8.72 | 1.3 | rotundioside p | 943 | 811, 649, 503, 471 | C48H80O18 | [12] |
| 11 | 9.36 | 4.2 | 2″-O-acetyl SSb3 | 853 | 811, 793, 649 | C45H74O15 | [32] |
| 12 | 10.33 | 2.5 | SSa | 825 | 779, 617, 541, 471 | C42H68O13 | [12] |
| 13 | 10.5 | 13.4 | SSb2 | 825 | 779, 617, 541, 471 | C42H68O13 | [12] |
| 14 | 11.11 | 5.2 | isomer of SSf | 927 | 781, 765, 619 | C48H80O17 | [12] |
| 15 | 11.28 | 1.6 | chinoposaponin XVIII | 941 | 779, 617 | C48H78O18 | [7] |
| 16 | 11.43 | 1.2 | 2″-O-acetyl SSa | 821 | 779, 761, 617 | C44H70O14 | [12] |
| 17 | 11.49 | 1.0 | diacetyl SSa | 863 | 821, 761 | C46H72O15 | [30] |
| 18 | 11.54 | 1.5 | 3″-O-acetyl SSa | 821 | 779, 761, 617, 541, 471 | C44H70O14 | [30] |
| 19 | 11.66 | 1.5 | malonyl-SSa | 865 | 821, 779, 761, 617 | C45H70O16 | [7] |
| 20 | 11.85 | 12.0 | SSb1 | 825 | 779, 617, 471 | C42H68O13 | [12] |
| 21 | 11.89 | 2.3 | 3β, 23, 28-trihydroxyolean-11, 13(18)-diene-16-one 3-O-β-D-glucopyranosyl-(1-3)-β-D-fucopyranoside | 777 | 615, 539, 469, 437 | C42H66O13 | [12] |
| 22 | 11.92 | 3.6 | acetyl SSa | 821 | 779, 617 | C44H70O14 | [31] |
| 23 | 12.75 | 3.9 | SSe | 809 | 763, 601, 455 | C42H68O12 | [12] |
| 24 | 13.84 | 1.2 | 3″-O-acetyl SSb2 | 821 | 779, 761, 617 | C44H70O14 | [12] |
| 25 | 14.14 | 2.4 | SSd | 825 | 779, 617 | C42H68O13 | [12] |
| 26 | 15.51 | 1.5 | malonyl-SSd | 865 | 821, 779, 761, 617 | C45H70O16 | [32] |
| 27 | 16.07 | 1.0 | diacetyl SSd | 863 | 821, 779, 76 | C46H72O15 | [30] |
| 28 | 16.13 | 1.5 | 6″-O-acetyl SSd | 821 | 779, 761, 617 | C44H70O14 | [12] |
| No. | tR (min) | VIP | Compound Name | M+• Ion (m/z) | Molecular Formula |
|---|---|---|---|---|---|
| 1 | 2.02 | 1.4 | L-isoleucine | 130.1 | C6H13NO2 |
| 2 | 2.02 | 1.4 | 3, 4-dimethyl-1-hexene | 111.2 | C8H16 |
| 3 | 2.55 | 1.2 | cyclobutene, 2-propenylidene | 91.1 | C7H8 |
| 4 | 2.56 | 1.0 | n-hexane | 85.2 | C6H14C6H14 |
| 5 | 2.85 | 1.6 | 2, 2-dimethyl heptane | 127.3 | C9H20 |
| 6 | 2.88 | 1.6 | 2-(1-methylbutyl)-oxirane | 113.2 | C7H14O |
| 7 | 4.03 | 1.3 | chloromethyl 2-chloroundecanoate | 268.2 | C12H22Cl2O2 |
| 8 | 4.03 | 1.3 | 2, 4, 6-trimethyl-heptane | 141.3 | C10H22 |
| 9 | 4.07 | 1.1 | 2, 5, 6-trimethyl-decane | 183.4 | C13H28 |
| 10 | 4.07 | 1.1 | trimethylene oxide | 57.1 | C3H6O |
| 11 | 4.40 | 1.1 | hexyl-oxirane | 71.1 | C4H8O |
| 12 | 4.40 | 1.1 | D-sphingosine | 298.5 | C18H37NO2 |
| 13 | 4.40 | 1.1 | 1-methyl-4-(1-methylethenyl)- 2-cyclohexene | 135.2 | C10H16 |
| 14 | 4.8 | 1.2 | phenacyl thiocyanate | 176.2 | C9H7NOS |
| 15 | 4.95 | 1.6 | 2-butenoic acid, 3-methylbutyl ester | 155.2 | C9H16O2 |
| 16 | 4.95 | 1.5 | 5-methyl-2-hexanamine | 114.2 | C7H17N |
| 17 | 5.02 | 2.0 | α-pinene | 135.2 | C10H16 |
| 18 | 5.05 | 1.7 | 4-octyl acetate | 171.3 | C10H20O2 |
| 19 | 5.05 | 1.7 | 4, 4-dimethyl-1-hexene | 111.2 | C8H16 |
| 20 | 5.05 | 1.7 | ethyl-cyclohexane | 111.2 | C8H16 |
| 21 | 5.09 | 1.4 | di-t-butylacetylene | 137.3 | C10H18 |
| 22 | 5.24 | 1.4 | 4-methyl-cyclopentadecanone | 237.4 | C16H30O |
| 23 | 5.36 | 1.3 | trans-4, 5-epoxydecane | 155.3 | C10H20O |
| 24 | 5.81 | 1.7 | 7-methylene-tridecane | 195.4 | C14H28 |
| 25 | 5.81 | 1.7 | benzyl 2-chloroethyl sulfone | 217.7 | C9H11ClO2S |
| 26 | 5.87 | 1.7 | n-nonane | 267.5 | C19H40 |
| 27 | 6.10 | 1.0 | acetophenone | 119.2 | C8H8O |
| 28 | 6.16 | 2.0 | 1-nitro-2-octanone | 172.2 | C8H15NO3 |
| 29 | 6.17 | 2.1 | 1-heptadecyne | 235.4 | C17H32 |
| 30 | 6.19 | 1.1 | cis-linaloloxide | 169.3 | C10H18O2 |
| 31 | 6.31 | 1.6 | heptanoic acid | 128.2 | C7H13O2 |
| 32 | 6.35 | 1.6 | cyclopropylacetic acid | 99.1 | C5H8O2 |
| 33 | 6.86 | 1.2 | 1-(3, 7-dimethyl-1-octenyl)-cyclopropanol | 195.3 | C13H24O |
| 34 | 6.90 | 1.4 | benzyl nitrile | 116.2 | C8H7N |
| 35 | 7.31 | 1.1 | 1-(ethenylthio)-octane | 171.3 | C10H20S |
| 36 | 7.32 | 1.0 | 1, 2, 3, 4, 5-cyclopentanepentol | 149.1 | C5H10O5 |
| 37 | 7.61 | 1.1 | 1, 4-dimethyl-adamantane | 163.3 | C12H20 |
| 38 | 7.90 | 1.4 | 1-tridecanol | 199.4 | C13H28O |
| 39 | 8.03 | 1.1 | 1-pentanol | 87.2 | C5H12O |
| 40 | 8.11 | 1.3 | methyl-cycloheptane | 111.2 | C8H16 |
| 41 | 8.11 | 1.3 | 1, 2-dimethyl-cyclohexane | 111.2 | C8H16 |
| 42 | 8.36 | 1.1 | 2, 4-bis(diazo)adamantane | 187.2 | C10H12N4 |
| 43 | 8.36 | 1.1 | isopulegol | 153.3 | C10H18O |
| 44 | 8.6 | 1.9 | cyclopentadecanol | 225.4 | C15H30O |
| 45 | 8.68 | 1.3 | 2, 5-diethylphenol | 149.2 | C10H14O |
| 46 | 8.69 | 1.1 | thymol | 149.2 | C10H14O |
| 47 | 8.70 | 1.0 | 3-methyl-5-(1-methylethyl)-phenol methylcarbamate | 206.3 | C12H17NO2 |
| 48 | 9.11 | 1.1 | 2-carbonitrile-cyclopentanone | 108.1 | C6H7NO |
| 49 | 9.18 | 1.5 | 2-undecenal | 167.3 | C11H20O |
| 50 | 9.68 | 1.3 | n-decanoic acid | 171.3 | C10H20O2 |
| 51 | 9.68 | 1.3 | cubenol | 221.4 | C15H26O |
| 52 | 9.70 | 1.1 | 3-methyl-heptanedioic acid dimethyl ester | 201.2 | C10H18O4 |
| 53 | 9.73 | 1.3 | chamigrene | 203.4 | C15H24 |
| 54 | 9.80 | 1.0 | cyclodecane | 139.3 | C10H20 |
| 55 | 9.94 | 1.4 | 15(S)-hydroxy-(5Z, 8Z, 11Z, 13E)- eicosatetraenoic acid | 319.5 | C20H32O3 |
| 56 | 9.95 | 1.0 | 1, 8-cyclopentadecadiyne | 201.3 | C15H22 |
| 57 | 23.2 | 2.9 | 12, 15-octadecadienoic acid, methyl ester | 293.5 | C19H34O2 |
| 58 | 24.06 | 3.1 | linoleic acid ethyl ester | 307.5 | C20H36O2 |
| Saikosaponin | Precursor Ion (m/z) | Product Ion I (m/z)/Collision Energy (eV) | Product Ion II (m/z)/Collision Energy (eV) |
|---|---|---|---|
| SSa | 779.5 | 439.6/52.0 | 617.3/32.2 |
| SSc | 971.5 | 779.3/38.9 | 925.4/23.7 |
| SSd | 779.5 | 439.2/55.0 | 617.3/33.1 |
| SSe | 809.5 | 601.3/33.0 | 763.3/21.3 |
| SSf | 973.5 | 781.3/39.9 | 927.4/23.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Z.; Zhao, H.; Tian, L.; Zhao, M.; Xiao, Y.; Liu, S.; Xiu, Y. Quantitative Analysis and Differential Evaluation of Radix Bupleuri Cultivated in Different Regions Based on HPLC-MS and GC-MS Combined with Multivariate Statistical Analysis. Molecules 2022, 27, 4830. https://doi.org/10.3390/molecules27154830
Wang Z, Zhao H, Tian L, Zhao M, Xiao Y, Liu S, Xiu Y. Quantitative Analysis and Differential Evaluation of Radix Bupleuri Cultivated in Different Regions Based on HPLC-MS and GC-MS Combined with Multivariate Statistical Analysis. Molecules. 2022; 27(15):4830. https://doi.org/10.3390/molecules27154830
Chicago/Turabian StyleWang, Zhenhuan, Huanxi Zhao, Lu Tian, Mengya Zhao, Yusheng Xiao, Shuying Liu, and Yang Xiu. 2022. "Quantitative Analysis and Differential Evaluation of Radix Bupleuri Cultivated in Different Regions Based on HPLC-MS and GC-MS Combined with Multivariate Statistical Analysis" Molecules 27, no. 15: 4830. https://doi.org/10.3390/molecules27154830
APA StyleWang, Z., Zhao, H., Tian, L., Zhao, M., Xiao, Y., Liu, S., & Xiu, Y. (2022). Quantitative Analysis and Differential Evaluation of Radix Bupleuri Cultivated in Different Regions Based on HPLC-MS and GC-MS Combined with Multivariate Statistical Analysis. Molecules, 27(15), 4830. https://doi.org/10.3390/molecules27154830