Abstract
Nitroxyl shows a unique biological profile compared to the gasotransmitters nitric oxide and hydrogen sulfide. Nitroxyl reacts with thiols as an electrophile, and this redox chemistry mediates much of its biological chemistry. This reactivity necessitates the use of donors to study nitroxyl’s chemistry and biology. The preparation and evaluation of a small library of new redox-triggered nitroxyl sources is described. The condensation of sulfonyl chlorides and properly substituted O-benzyl hydroxylamines produced O-benzyl-substituted sulfohydroxamic acid derivatives with a 27–79% yield and with good purity. These compounds were designed to produce nitroxyl through a 1, 6 elimination upon oxidation or reduction via a Piloty’s acid derivative. Gas chromatographic headspace analysis of nitrous oxide, the dimerization and dehydration product of nitroxyl, provides evidence for nitroxyl formation. The reduction of derivatives containing nitro and azide groups generated nitrous oxide with a 25–92% yield, providing evidence of nitroxyl formation. The oxidation of a boronate-containing derivative produced nitrous oxide with a 23% yield. These results support the proposed mechanism of nitroxyl formation upon reduction/oxidation via a 1, 6 elimination and Piloty’s acid. These compounds hold promise as tools for understanding nitroxyl’s role in redox biology.
1. Introduction
The one-electron reduction and protonation of nitric oxide (NO), a well-known biological signaling agent characterized as a gasotransmitter, formally produces nitroxyl (HNO) [1]. These structural/electronic differences give HNO a distinct chemistry from NO, as HNO dimerizes to hyponitrous acid (H2N2O2) that dehydrates to nitrous oxide (N2O) [2]. This reactivity necessitates the use of HNO donors and highlights the extreme electrophilic and oxidizing character of HNO [3]. HNO demonstrates different biological properties from NO [4,5], and at least three drugs that chemically release HNO have been used clinically or evaluated in trials for the treatment of cancer (hydroxyurea), alcoholism (cyanamide) and congestive heart failure (Cimlanod), showing the clinical potential of HNO donors [6,7,8]. Much of our understanding of the pharmacology and therapeutic potential of HNO comes from experiments utilizing HNO donors [3,9]. The most common HNO donors include Angeli’s salt (AS, Na2N2O3) and Piloty’s acid (PA, PhSO2NHOH), which are commercially available solids that rapidly and cleanly release HNO under neutral conditions [3,9].
In addition to dimerization, HNO reacts with thiols, generating a N-hydroxysulfenamide intermediate that can rearrange to a sulfinamide or further react with more thiol to yield a disulfide and hydroxylamine [10]. HNO similarly reacts with hydrogen sulfide (H2S), a second recognized gasotransmitter [1], to give short-chain hydrogen polysulfides (H2Sn) or S8 depending on the relative concentrations of HNO and H2S [11]. Based on this chemistry, HNO can influence thiol-mediated biochemistry and potentially H2S-controlled reactions. For example, HNO inhibits enzymes with active site cysteines, such as aldehyde dehydrogenase (AlDH) and glyceraldehyde 3-phosphate dehydrogenase (GAPDH) [12,13]. HNO modifies cardiac myofilament proteins affecting myocardial contractility by increasing calcium cycling and sensitizing myocardial tissue responsiveness to calcium [14,15]. HNO directly activates the chemosensory TRPA1 channel [16]. In bacteria, the addition of HNO to Staphylococcus aureus increases the formation of the persulfide (RSSH) of the predominant low-molecular-weight thiol, bacillithiol (BSH) [17]. This oxidized thiol derivative may control transcription factors responsible for directing the overall sulfur metabolism in these bacteria [18]. Under oxidative conditions, the addition of HNO to Bacillus subtilis synergistically enhances hydrogen peroxide (H2O2)-mediated cell killing. Angeli’s salt (AS), which decomposes to HNO and nitrite (NO2−) at neutral pH, was used as the HNO source in these redox investigations in B. subtilis [3,19].
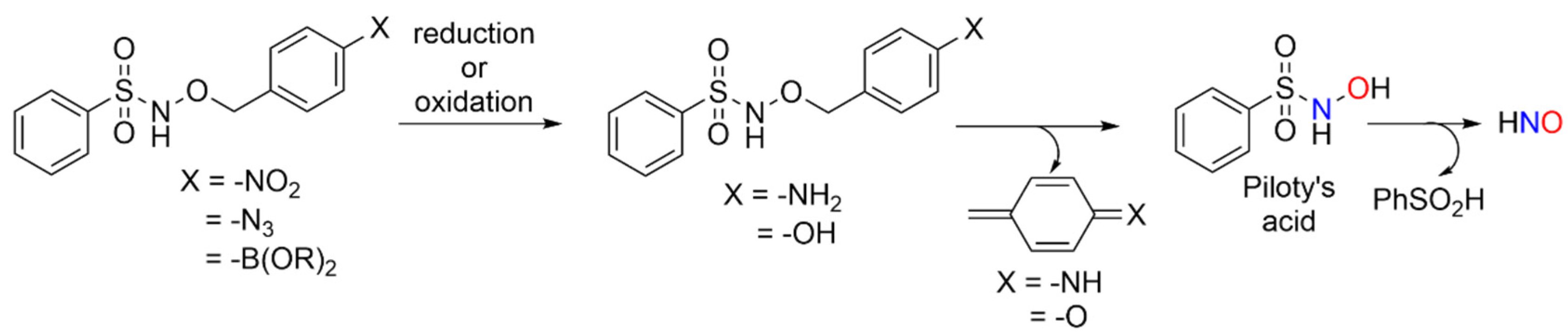
Redox-triggered HNO donors may possess value in probing biological crosstalk between HNO and other small redox active signaling agents, such as H2S or H2O2. The use of AS as a HNO donor is limited by the co-production of NO2−, a relatively fast and pH-insensitive release rate (t1/2~2.8 min, pH 4–8) and limitations in the synthesis of new donors [3]. Piloty’s acid (PA), another common HNO donor, decomposes to HNO and phenyl sulfinic acid (PhSO2H) at a neutral pH [3]. The aryl portion of PA tolerates structural modification while still supporting HNO release, which allows for the installation of designed redox-sensitive elements to initiate a 1, 6-elimination [20]. Scheme 1 shows Piloty’s acid derivatives designed to release HNO via a 1, 6-elimination mechanism upon exposure to either reductants or oxidants. These processes convert the azide, nitro or boronate ester groups into either the aniline or phenol derivative that should decompose to Piloty’s acid with the release of p-quinone methide or its imine (Scheme 1).
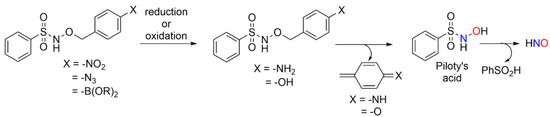
Scheme 1.
Designed redox-triggered HNO donors.
Similar constructs have found extensive use for the detection or generation of other signaling agents, such as H2S, but, to the best of our knowledge, have not been applied to HNO release under redox conditions [21]. A hydrogen-peroxide-based prodrug system that releases a structurally similar hydroxamic acid as a histone deacetylase inhibitor has been described [22]. Such compounds permit HNO formation under specific redox conditions and would form the basis of an improved understanding of the role that HNO plays in redox biochemistry. We describe the preparation and characterization of a small library of redox-triggered HNO donors and an evaluation of their ability to produce HNO under specific redox conditions.
2. Results
2.1. Synthesis
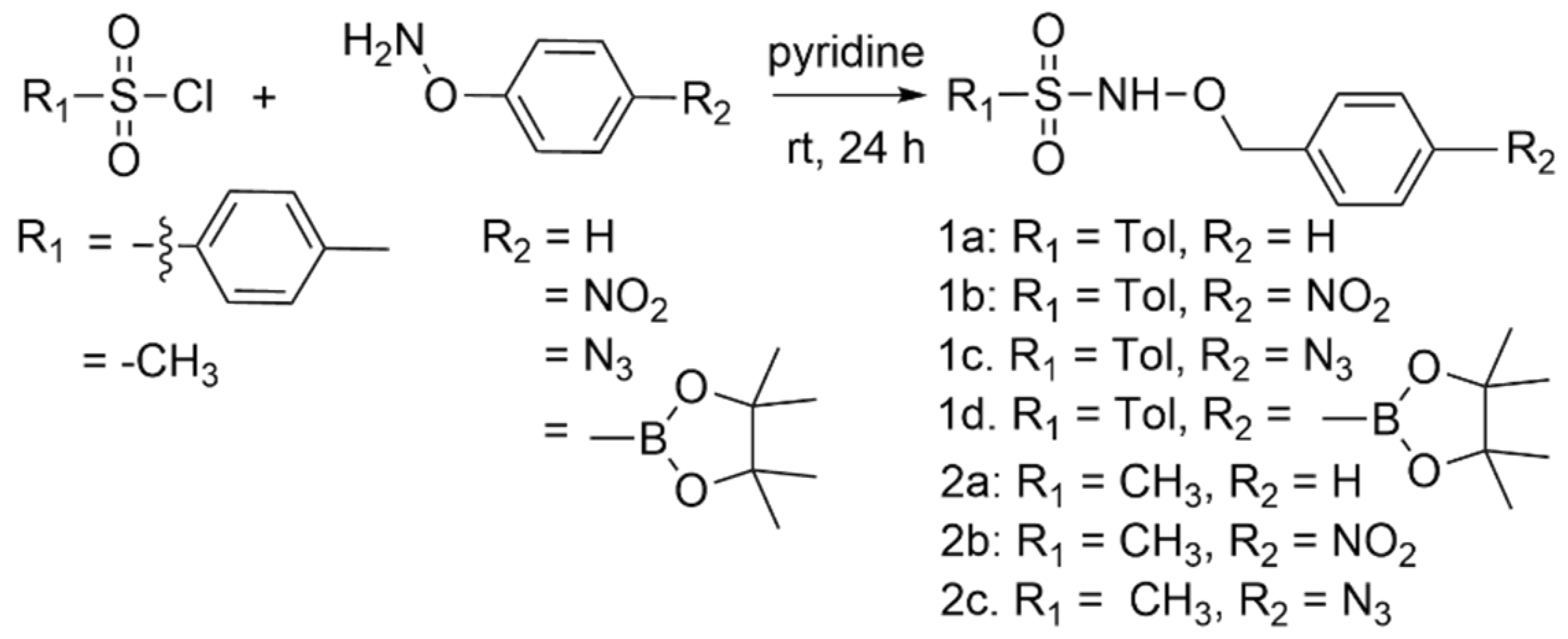
Possible redox-triggered HNO donors (1a–d and 2a–c) were prepared with a 27–79% yield by the condensation of the properly substituted O-benzyl hydroxylamine derivative with either p-toluene or methyl sulfonyl chloride under basic conditions (Scheme 2) [23,24]. The variable yields likely arise from mixtures of O and N-mono alkylated products and N, O-dialkylated products as reported [23,24].
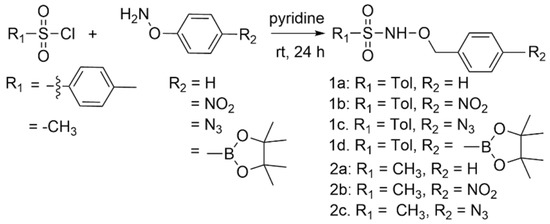
Scheme 2.
Synthesis of potential redox-triggered HNO donors.
This sequence followed by extraction generally gave the desired products with excellent purity, as judged by proton and carbon nuclear magnetic resonance (NMR) spectroscopy and mass spectrometry (MS). Individual compounds could be further purified by recrystallization or silica gel flash chromatography if necessary.
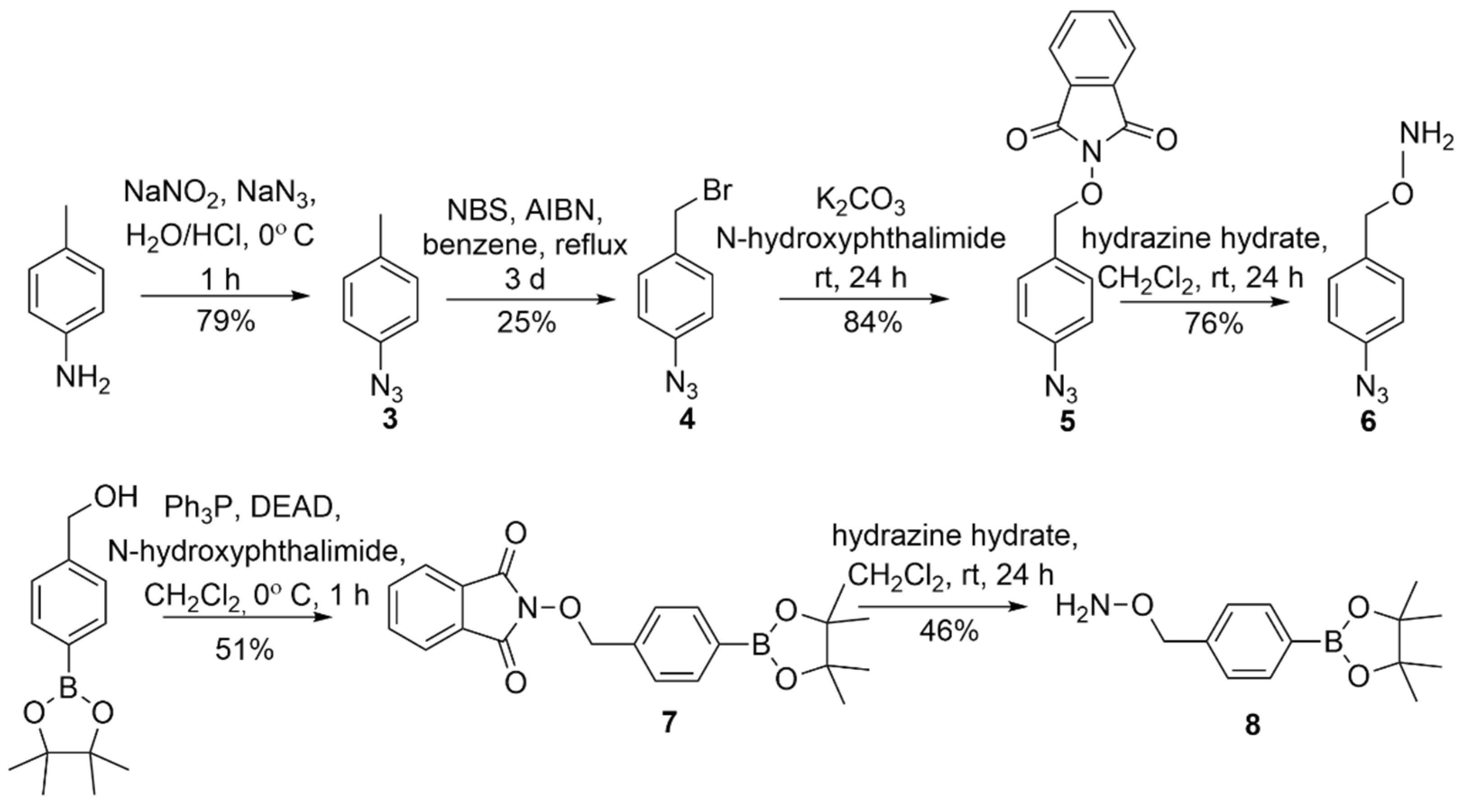
The substituted O-benzyl hydroxylamines required in Scheme 1 were purchased (R = -H or -NO2) or prepared using reported literature procedures for the azide and the pinacol-derived boronate ester [22,25]. Scheme 3 summarizes the preparation of the azide-containing hydroxylamine (6) from p-toluidine through a four-step sequence that features diazotization/azide substitution, bromination, N-hydroxyphthalimde substitution to install the N-O bond and hydrazine deprotection through intermediates 3–5 (Scheme 3) [25].
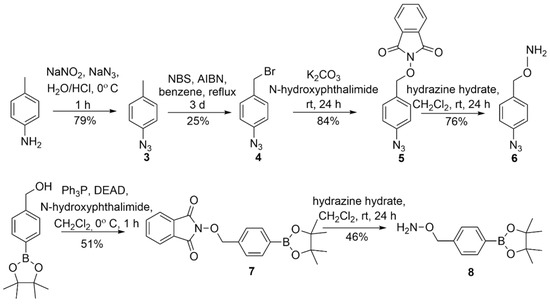
Scheme 3.
Preparation of the azide and boronate ester substituted hydroxylamine (6 and 8) [22,25].
The Mitsonobu coupling of N-hydroxyphthalimide with the commercially available pinacol ester of 4-hydroxymethylphenylboronic acid followed by hydrazine deprotection yields the boronate-ester-substituted hydroxylamine (8) via 7 (Scheme 3) [22]. These intermediates were characterized by NMR spectroscopy and MS and purified by normal phase flash chromatography (see Supplementary Materials).
2.2. HNO Donation Ability
Piloty’s acid (PhSO2NHOH), a common HNO donor, decomposes to HNO and phenyl sulfinic acid (PhSO2H) at a neutral pH. Compounds 1b–d and 2b,c were designed to decompose upon either reduction or oxidation as depicted in Scheme 1 to yield a reactive intermediate that should release a Piloty’s acid derivative (pCH3C4H6SO2NHOH or CH3SO2NHOH) that fragments to HNO and the corresponding sulfinic acid. The gas chromatographic (GC) headspace measurement of nitrous oxide (N2O), the dimerization and dehydration product of HNO, provides a rapid and simple measure of HNO formation from these transformations [26]. The decomposition of the PA derivative pCH3C4H6SO2NHOH, a known HNO donor, in methanol/buffer generates 76% of N2O by this measure after 24 h (Table 1). The sensitivity of N2O formation to the addition of glutathione (GSH), which rapidly reacts with HNO blocking N2O production [10], provides evidence for HNO’s intermediacy (Table 1). Compounds 1a and 2a do not contain reduction or oxidation-sensitive functional groups, preventing the proposed decomposition to a Piloty’s acid derivative, and do not produce N2O under these conditions (Table 1).

Table 1.
Control N2O Production at 24 h from 1a-d and 2a-c (CH3OH/100 mM PBS buffer (pH = 7.4), 1:1, 2 mL).
The incubation of 1b-d and 2b-c in a methanol/100 mM PBS buffer showed ~1% N2O formation, indicating essentially no formation of HNO over 24 h from these compounds, which was as expected, in the absence of any reducing/oxidizing agents. The formation of small amounts of N2O could arise from the slow hydrolysis/methanolysis of the benzyl group producing pCH3C4H6SO2NHOH or CH3SO2NHOH or from the presence of small amounts of these sulfohydroxamic acids in these samples, which could form from the condensation of residual hydroxylamine potentially present in the commercial or synthetic O-benzylhydroxylamine derivatives.
Table 2 summarizes N2O formation at 24 h from the treatment of 1a–d and 2a–c with various reducing and oxidizing agents. The addition of sodium borohydride, a reducing agent capable of nitro-to-amine reduction [27], to compounds 1b and 2b generates N2O with a 25 and 51% yield, respectively, providing evidence for initial HNO formation (Table 2). The lower observed amounts of N2O from 1b may reflect the poor solubility of 1b, which did not completely dissolve in 1:1 CH3OH:H2O (2 mL), possibly due to the presence of two para-substituted aromatic rings, including one with the polar nitro group. Compound 1b dissolved with the addition of another 0.5 mL of CH3OH (1.5:1 CH3OH:H2O, 2.5 mL), and the results reported in Table 2 for 1b were obtained under these conditions. Thin-layer chromatography (TLC) and an MS analysis of the reduction of 1b with sodium borohydride provides evidence for the formation of p-amino benzyl alcohol, the product of water addition to the imine of p-quinone methide. The addition of sodium borohydride/copper (II) sulfate, a mixture known to reduce azides to amines [28], to 1c and 2c produces 89 and 92% N2O, respectively (Table 2). These results support Scheme 1 and suggest a reduction of the nitro and azide groups to the amine followed by decomposition to the Piloty’s acid derivative. As expected, the treatment of 1a and 2a with sodium borohydride or sodium borohydride/copper (II) sulfate under these conditions did not generate N2O (Table 2).

Table 2.
N2O Production from 1a-d and 2a-c at 24 h (1 h) upon reduction/oxidation.
The incubation of 1c and 2c with GSH (2.5 or 5 equivalents) does not produce N2O, but the treatment of 1c and 2c with H2S (2.5 equivalents) generated small reproducible amounts (6% and 5%, respectively) of N2O. These results suggest H2S-mediated azide reduction to the amine as described, followed by HNO formation [29]. The lower amounts of N2O observed are likely due to the competition between HNO dimerization and H2S addition. Increasing the amounts of H2S to five equivalents abolished N2O formation, supporting this explanation. Table 2 also shows hydrogen peroxide (H2O2)-mediated N2O release from 1d, a boronate-containing compound designed to release HNO via boronate oxidation to the phenol followed by decomposition. The incubation of 1d with H2O2 resulted in the formation of N2O with a 23% yield, presumably indicating the formation of HNO upon oxidation to the phenol. The treatment of 1a and 2a with H2O2 did not produce N2O (Table 2). Table 2 also shows that the amount of N2O produced increases from the 1 h to 24 h measurements, likely reflecting the kinetics of these model oxidations and reductions.
Bacterial nitroreductases using NADH as a co-substrate act as competent reducing agents of aromatic nitro groups, with the subsequent release of desired compounds via a 1, 6-elimination [30,31]. The treatment of 1b or 2b with Escherichia coli nitroreductase (Sigma) in the presence of NADH failed to generate N2O as initially expected. A previous report indicates that NADH reduces HNO to hydroxylamine, thus blocking N2O formation and suggesting that the lack of observed N2O in these experiments results from HNO reduction by the NADH co-substrate [32]. Further investigation will be necessary to define the practicality of nitroreductase-triggered HNO donors, but these results suggest that HNO may exert a portion of effects through NADH/NADPH depletion.
3. Materials and Methods
All materials and solvents used for extraction and purification were purchased from commercial vendors and used as received. 1H and 13C NMR spectra were recorded using a Bruker Avance 400 MHz NMR spectrometer. Mass spectra were obtained using a Bruker Amazon SL ion trap. An Agilent Technologies 7890A gas chromatograph equipped with a micro-electron capture detector and a 30 m × 0.32 m (25 μm) HP-MOLSIV capillary column was used for the gas chromatographic analysis of N2O. CAUTION: Any experiments preparing alkyl or aryl azides should be performed in a well-ventilated fume hood and behind a blast shield. Sodium azide should not be mixed with strong acids.
3.1. Synthesis of Piloty’s Acid Derivatives
The Piloty’s acid derivatives were prepared by sulfonyl chloride condensation with an O-benzylhydroxylamine derivative using a modified literature procedure [33].
N-(benzyloxy)-4-methylbenzenesulfonamide (1a).O-benzylhydroxylamine hydrochloride (287 mg, 1.80 mmol) was added to a solution of p-toluenesulfonyl chloride (378 mg, 1.98 mmol) in pyridine (5 mL). The reaction mixture reacted overnight at room temperature and was quenched with 2M HCl (25 mL) and extracted with ethyl acetate (25 mL). The organic layer was washed with water (10 mL), brine (10 mL), dried over MgSO4 and concentrated using rotary evaporator to give 396 mg, 79.4% yield of 1a as white solid. 1H NMR (400 MHz, CDCl3): δ 7.82 (d, J = 8.0 Hz, 2H), 7.33–7.35 (m, 6H), 6.88 (s, 1H), 4.97 (s, 2H), 2.44 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 148.02, 145.33, 142.47, 133.33, 129.90, 129.59, 128.56, 123.75, 77.79, 21.72. ESI-MS (positive mode) (m/z) calculated mass for C14H14NO3SNa [M+Na]+ 300.34, found mass 300.13.
4-methyl-N-((4-nitrobenzyl)oxy)benzenesulfonamide (1b). 1-[(Aminooxy)methyl]-4-nitrobenzene hydrochloride (368 mg, 1.80 mmol) was added to a solution of p-toluenesulfonyl chloride (378 mg, 1.98 mmol) in pyridine (5 mL). The reaction mixture reacted overnight at room temperature and was quenched with 2M HCl (25 mL) and extracted with ethyl acetate (25 mL). The organic layer was washed with water (10 mL), brine (10 mL), dried over MgSO4 and concentrated under reduced pressure to give 428 mg, 73.8% yield of 1b as white solid. 1H NMR (400 MHz, CDCl3): δ 8.20 (d, J = 8 Hz, 2H), 7.80 (d, J = 8 Hz, 2H), 7.49 (d, J = 8 Hz, 2H), 7.35 (d, J = 8 Hz, 2H), 6.96 (s, 1H), 5.09 (s, 2H), 2.45 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 147.96, 145.32, 142.56, 133.31, 129.89, 129.54, 128.55, 123.72, 79.74, 21.71. ESI-MS (positive mode) (m/z) calculated mass for C14H14N2O5SNa [M+Na]+ 345.06, found mass 345.20.
N-((4-azidobenzyl)oxy)-4-methylbenzenesulfonamide (1c). p-Toluenesulfonyl chloride (2.24 g, 11.8 mmol) was added to a solution of O-(4-azidobenzyl) hydroxylamine (1.78 g, 10.8 mmol) in pyridine (30 mL). The reaction mixture was allowed to stir overnight at room temperature and quenched with 2M HCl (150 mL) and extracted using ethyl acetate (150 mL). The organic layer was washed with water (100 mL), brine (100 mL), dried over MgSO4 and concentrated under reduced pressure to give 1.32 g, 38.4% yield of 1c as a light yellow solid. 1H NMR (400 MHz, CDCl3): δ 7.73 (d, J = 8 Hz, 2H), 7.26 (d, J = 8 Hz, 4H), 6.93 (d, J = 8 Hz, 2H), 6.85 (s, 1H), 4.87 (s, 2H), 2.37 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 145.00, 140.53, 133.60, 131.96, 131.04, 129.79, 128.55, 119.09, 78.66, 21.69. ESI-MS (positive mode) (m/z) calculated mass for C14H14N4O3SNa [M+Na]+ 341.34, found mass 341.28.
4-methyl-N-((4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzyl)oxy)benzenesulfonamide (1d). p-Toluenesulfonyl chloride (340 mg, 1.79 mmol) was added to a solution of O-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzyl)hydroxylamine (410 g, 1.64 mmol) in pyridine (5 mL). The reaction mixture reacted overnight and concentrated under reduced pressure with the addition of excess toluene. The resulting product was acidified using 2M HCl and extracted with chloroform (25 mL). The organic layer was washed with water (30 mL), brine (30 mL), dried over MgSO4 and concentrated under pressure to give 420 mg, 63.5% of 1d as a white solid. 1H NMR (400 MHz, CDCl3): δ 7.70–7.75 (m, 4H), 7.24–7.27 (m, 4H), 6.83 (s, 1H), 4.91 (s, 2H), 2.36 (s, 3H), 1.27 (s, 12H). 13C NMR (101 MHz, CDCl3): δ144.91, 138.23, 134.94, 133.62, 129.76, 128.59, 128.41, 83.92, 79.23, 24.88, 21.68. ESI-MS (positive mode) (m/z) calculated mass for C20H26BNO5SNa [M+Na]+ 426.15, found mass 426.29.
N-(benzyloxy)methanesulfonamide (2a). O-benzylhydroxylamine hydrochloride (510 mg, 2.50 mmol) was added to a solution of methanesulfonyl chloride (210 µL, 2.71 mmol) in pyridine (12 mL). The reaction mixture reacted overnight at room temperature and was quenched with 2M HCl (60 mL) and extracted with ethyl acetate (60 mL). The organic layer was washed with water (40 mL), brine (40 mL), dried over MgSO4 and concentrated using rotary evaporator to give 404 mg, 80.4% yield of 2a as a white solid. 1H NMR (400 MHz, CDCl3): δ 7.37–7.41 (m, 5H), 6.86 (s, 1H), 5.00 (s, 2H), 3.04 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 135.02, 129.52, 128.96, 128.88, 79.66, 37.03. ESI-MS (positive mode) (m/z) calculated mass for C8H11NO3SNa [M+Na]+ 224.06, found mass 224.01.
N-((4-nitrobenzyl)oxy)methanesulfonamide (2b). 1-[(Aminooxy)methyl]-4-nitrobenzene hydrochloride (613 mg, 3.0 mmol) was added to a solution of methanesulfonyl chloride (210 µL, 2.71 mmol) in pyridine (12 mL). The reaction mixture reacted overnight under room temperature and was quenched with 2M HCl (50 mL) and extracted with ethyl acetate (50 mL). The organic layer was washed with water (30 mL), brine (30 mL), dried over MgSO4 and concentrated using reduced pressure to give 516 mg, 77.5% yield of 2b as white solid. mp 111–113 ⁰C; IR (film) 3211, 1608, 1517, 1320, 1151 cm−1; 1H NMR (400 MHz, CDCl3): δ 8.17 (d, J = 8 Hz, 2H), 7.49 (d, J = 8 Hz, 2H), 7.10 (s, 1H), 5.03 (s, 2H), 3.03 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 148.13, 142.19, 129.74, 123.84, 78.08, 37.09. ESI-MS (positive mode) (m/z) calculated mass for C18H10N2O5SNa [M+Na]+ 269.23, found mass 269.06.
N-((4-azidobenzyl)oxy)methanesulfonamide (2c). O-(4-azidobenzyl) hydroxylamine (1.3g, 7.93 mmol) was added to a solution of methanesulfonyl chloride (675 µL, 8.72 mmol) in pyridine (30 mL). The reaction mixture reacted overnight at room temperature and was quenched with 2M HCl (150 mL) and extracted with ethyl acetate (150 mL). The organic layer was washed with water (100 mL), brine (100 mL), dried over MgSO4 and concentrated using rotary evaporator to give a crude product that was purified using column chromatography (Pet Ether/Ethyl Acetate, 3:1) to give 530 mg, 27.6% yield of 2c as a white solid. mp 108–110 ⁰C; IR (film) 3207, 3024, 2943, 2111, 1505, 1304, 1157 cm−1; 1H NMR (400 MHz, CDCl3): δ 7.38 (d, J = 10 Hz, 2H), 7.04 (d, J = 10 Hz, 2H), 6.98 (s, 1H), 4.96 (s, 2H), 3.05 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 140.78, 131.71, 131.17, 119.19, 78.91, 37.03. ESI-MS (positive mode) (m/z) calculated mass for C8H11NO3SNa [M+Na]+ 265.04, found mass 265.03.
1-azido-4-methylbenzene (3). Was prepared following a published procedure [23]. A solution of sodium nitrite (6.3g, 92.8 mmol) in 26 mL of cold water and a solution of sodium azide [25] (6.1g, 92.8 mmol) in 58mL of cold water were added slowly to a solution of p-toluidine (10g, 92.8 mmol) in 50mL HCl: H2O (1:1) at 0 °C. After 1 h of reaction, the mixture was extracted with CHCl3 (100 mL), the remaining aqueous layer was washed with CHCl3 (2 × 50 mL) and the combined organic layers were washed with water (100 mL), brine (100 mL), dried over MgSO4 and concentrated under reduced pressure to give 9.5 g, 79.3% yield of 3 as a brown oil. 1H NMR (400 MHz, CDCl3): δ 7.16 (d, J = 8 Hz, 2H), 6.93 (d, J = 8 Hz, 2H), 2.33 (s, 3H). 13C NMR (101 MHz, CDCl3): δ 137.15, 134.63, 130.35, 118.85, 20.85.
1-azido-4-(bromomethyl)benzene (4). Was prepared following a published procedure [25] N-Bromosuccinimide (10.51 g, 59 mmol) and 2,2′-Azobis(2-methylpropionitrile) (1 g, 6.1 mmol) were added under N2 atmosphere to a solution of 1-azido-4-methylbenzene (8 g, 60.1 mmol) in benzene (200 mL). The reaction mixture was heated under reflux for 3 days and concentrated under reduced pressure. The resulting product was partitioned between CH2Cl2 (100 mL) and washed with water (100 mL), brine (2 × 50 mL), dried over MgSO4 and concentrated to give a crude product that was purified by column chromatography (petroleum ether) to give 3.1g, 24.6% yield of 4 as a yellow-orange oil. 1H NMR (400 MHz, CDCl3): δ 7.40 (d, J = 12 Hz, 2H), 7.02 (d, J = 12 Hz, 2H), 4.50 (s, 2H).
2-((4-azidobenzyl)oxy)-isoindoline-1,3-dione (5). Was prepared following a published procedure [25] K2CO3 (2.34 g, 16.9 mmol) was added slowly to a solution of 1-azido-4-(bromomethyl)benzene (3.1 g, 14.6 mmol) and N-Hydroxyphthalimide (2.76 g, 16.9 mmol) in 45 mL DMF. The reaction mixture reacted overnight at room temperature and partitioned between CH2Cl2 (150 mL) and H2O (100 mL). The organic layer was washed with brine (100 mL), dried over MgSO4 and concentrated under reduced pressure to give 4.2g, 84.5% yield of 5 as a white solid. 1H NMR (400 MHz, CDCl3): δ 7.75–7.76 (m, 4H), 7.45 (d, J = 8 Hz, 2H), 6.96 (d, J = 8 Hz, 2H), 5.11 (s, 2H). 13C NMR (101 MHz, CDCl3): δ 163.51, 141.17, 134.51, 131.57, 130.41, 128.83, 123.57, 119.14 79.11.
O-(4-azidobenzyl) hydroxylamine (6). Was prepared following a modified procedure [25] Hydrazine monohydrate (1.04 mL, 32.4 mmol) was added slowly to a solution of 2-((4-azidobenzyl)oxy)-isoindoline-1,3-dione (4.2 g, 14.3 mmol) in CH2Cl2 (45 mL) and the reaction mixture reacted overnight at room temperature. The resulting mixture was quenched with 1M NaOH (50 mL) and the separated organic layer was washed with water (30 mL), brine (30 mL), dried over MgSO4 and concentrated under reduced pressure to give 1.78g, 76.4% yield of 6 as a yellow oil. 1H NMR (400 MHz, CDCl3): δ 7.27 (d, J = 8 Hz, 2H), 6.94 (d, J = 8 Hz, 2H), 5.35 (broad s, 2H), 4.56 (s, 2H). 13C NMR (101 MHz, CDCl3): δ 139.75, 134.29, 130.01, 119.06, 77.24.
2-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy)isoindoline-1,3-dione (7). Was prepared following a published procedure [22]. Diethyl azodicarboxylate (1 mL, 5.2 mmol) was added slowly at 0 °C to a solution of triphenylphosphine (1.23 g, 4.7 mmol), N-hydroxyphthalimide (0. 77g, 4.7mmol), and 4-hydroxymethylphenylboronic acid pinacol ester (1 g, 4.3 mmol) in CH2Cl2 (10 mL). After 1 h of reaction, the reaction mixture was concentrated under reduced pressure and purified by column chromatography (Pet Ether/Ethyl Acetate, 10:1) to give 900 mg, 50.5% yield of 7 as a white solid. 1H NMR (400 MHz, CDCl3): δ 7.75–7.71 (m, 4H), 7.66–7.62 (m, 2H), 7.45 (d, J = 8 Hz, 2H), 5.15 (s, 2H), 1.26 (s, 12H).
O-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)benzyl)hydroxylamine (8). Was prepared following a modified procedure [22]. Hydrazine hydrate (120 µL, 3.75 mmol) was added slowly to a solution of 2-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)phenoxy)isoindoline-1,3-dione (900 mg, 3.61 mmol) in ethanol (20 mL). The reaction mixture reacted overnight and the solid was filtered and rinsed with cold ethanol. The filtrate was concentrated under reduced pressure to give 410 mg, 45.6% yield of 8 as a yellow oil. 1H NMR (400 MHz, CDCl3): δ 7.64 (d, J = 8 Hz, 2H), 7.45 (d, J = 8 Hz, 2H), 5.15 (s, 2H), 1.26 (s, 12H).
3.2. Gas Chromatographic N2O Analysis
For headspace analysis, substrate (1a–d, 2a–c, 0.04 mmol) was placed in a 10 mL round-bottom flask with a stir bar and sealed with a rubber septum. Solvent (methanol:water or methanol:PBS (100 mM), pH = 7.4; 1:1, 2 mL) was added using a syringe, and headspace aliquots (0.1 mL) from each experiment were injected at 1 and 24 h onto a 7890A Agilent Technologies gas chromatograph equipped with a micro-electron capture detector and a 30 m × 0.32 m (25 μm) HP-MOLSIV capillary column. The oven was operated at 200 °C for the duration of the run (4.5 min). The inlet was held at 250 °C and run in split mode (split ratio 1:1) with a total flow (N2 as carrier gas) of 4 mL/min and a pressure of 37.9 psi. The μECD was held at 325 °C with a makeup flow (N2) of 5 mL/min. The retention time of nitrous oxide was 3.4 min, and yields were calculated based on a standard curve for nitrous oxide gas (Gasco Precision Calibration Mixtures). To some samples, sodium borohydride (1.1 equivalents), sodium borohydride (1.1 equivalents) and copper (II) sulfate (0.1 equivalents), GSH (5 equivalents) or sodium sulfide (2–5 equivalents) were added. For oxidation of 1d, hydrogen peroxide (5 equivalents) was added to a solution (2 mL) of the substrate in a mixture of 3:2:0.5 acetonitrile, water, PBS (100 mM, pH = 7.4).
4. Conclusions
Nitroxyl (HNO) demonstrates a unique biological profile compared to NO that deserves more detailed studies that have been afforded to other nitrogen oxides and gasotransmitters. The high chemical reactivity of HNO requires the use of donors and relatively complex detection methods, which complicate such studies and the confirmation of endogenous HNO production. HNO exhibits rich redox reactivity with thiols, hydrogen sulfide and heme proteins, suggesting a potential role in various redox-mediated processes. We report a small library of derivatives (1a–d and 2a–c) of the HNO donor, Piloty’s acid, that liberate HNO upon oxidation or reduction through a 1, 6 elimination mechanism. These compounds were quickly constructed by the condensation of a sulfonyl chloride and an appropriately substituted hydroxylamine derivative. The nitro and azide-containing molecules (1b, c and 2b, c) demonstrated HNO release, as measured by headspace GC for N2O, upon chemical treatment with reducing agents. Similarly, a boronate ester derivative (1d) generates HNO upon hydrogen peroxide oxidation. Control experiments with 1a and 2a show that only compounds with redox active groups generate HNO under these conditions, supporting the mechanism. Overall, these results show the ability of these redox-sensitive HNO donors to release HNO upon oxidation/reduction, and could find use in further defining the role of HNO in redox-based biological processes.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules27165305/s1, Materials; Figure S1. 1H NMR and 13C NMR of 1a; Figure S2. 1H NMR and 13C NMR of 1b; Figure S3. 1H NMR and 13C NMR of 2b; Figure S4. IR of 2b; Figure S5. 1H NMR and 13C NMR of 3; Figure S6. 1H NMR of 4; Figure S7. 1H NMR of 5; Figure S8. 1H NMR and 13C NMR of 6; Figure S9. 1H NMR and 13C NMR of 1c; Figure S10. 1H NMR of 7; Figure S11. 1H NMR of 8; Figure S12. 1H NMR and 13C NMR of 1d; Figure S13. 1H NMR and 13C NMR of 2a; Figure S14. 1H NMR and 13C NMR of 2c; Figure S15. IR of 2c; Figure S16. MS of 1a; Figure S17. MS of 1b; Figure S18. MS of 2b; Figure S19. MS of 1c; Figure S20. MS of 1d; Figure S21. MS of 2a; Figure S22. MS of 2c.
Author Contributions
Compound synthesis, purification, characterization and evaluation of nitrous oxide release, Y.L. and Z.X.; evaluation of nitrous oxide release, student mentorship and manuscript preparation, A.M.R.; project conception and direction and manuscript preparation, S.B.K. All authors have read and agreed to the published version of the manuscript.
Funding
This research received internal research support from Wake Forest University.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data available in Supplementary Materials.
Acknowledgments
The authors wish to acknowledge the Wake Forest University Center for Molecular Signaling for helpful discussion organizing this manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Yang, G.; Sener, A.; Ji, Y.; Pei, Y.; Pluth, M.D. Gasotransmitters in Biology and Medicine: Molecular Mechanisms and Drug Targets. Oxid. Med. Cell. Longev. 2016, 2016, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Bonner, F.T.; Hughes, M.N. The Aqueous Solution Chemistry of Nitrogen in Low Positive Oxidation States. Comments Inorg. Chem. 1988, 7, 215–234. [Google Scholar] [CrossRef]
- Dumond, J.F.; King, S.B. The chemistry of nitroxyl-releasing compounds. Antioxid. Redox Signal. 2011, 14, 1637–1648. [Google Scholar] [CrossRef] [Green Version]
- Miranda, K.M.; Paolocci, N.; Katori, T.; Thomas, D.D.; Ford, E.; Bartberger, M.D.; Espey, M.G.; Kass, D.A.; Feelisch, M.; Fukuto, J.M.; et al. A biochemical rationale for the discrete behavior of nitroxyl and nitric oxide in the cardiovascular system. Proc. Natl. Acad. Sci. USA 2003, 100, 9196–9201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fukuto, J.M.; Chiang, K.; Hszieh, R.; Wong, P.; Chaudhuri, G. The pharmacological activity of nitroxyl: A potent vasodilator with activity similar to nitric oxide and/or endothelium-derived relaxing factor. J. Pharmacol. Exp. Ther. 1992, 263, 546–551, PMID: 1331403. [Google Scholar] [PubMed]
- King, S.B. The Nitric Oxide Producing Reactions of Hydroxyurea. Curr. Med. Chem. 2003, 10, 437–452. [Google Scholar] [CrossRef]
- Nagasawa, H.T.; DeMaster, E.G.; Redfern, B.; Shirota, F.N.; Goon, D.J.W. Evidence for Nitroxyl in the Catalase-Mediated Bioactivation of the Alcohol Deterrent Agent Cyanamide. J. Med. Chem. 1990, 33, 3122–3124. [Google Scholar] [CrossRef]
- Lang, N.N.; Ahmad, F.A.; Cleland, J.G.; O’Connor, C.M.; Teerlink, J.R.; Voors, A.A.; Taubel, J.; Hodes, A.R.; Anwar, M.; Karra, R.; et al. Haemodynamic effects of the nitroxyl donor cimlanod (BMS-986231) in chronic heart failure: A randomized trial. Eur. J. Heart Fail. 2021, 23, 1147–1155. [Google Scholar] [CrossRef]
- Switzer, C.H.; Flores-Santana, W.; Mancardi, D.; Donzelli, S.; Basudhar, D.; Ridnour, L.A.; Miranda, K.M.; Fukuto, J.M.; Paolocci, N.; Wink, D.A. The emergence of nitroxyl (HNO) as a pharmacological agent. Biochim. Biophys. Acta Bioenerg. 2009, 1787, 835–840. [Google Scholar] [CrossRef] [Green Version]
- Miranda, K.M. The chemistry of nitroxyl (HNO) and implications in biology. Coord. Chem. Rev. 2005, 249, 433–455. [Google Scholar] [CrossRef]
- Zarenkiewicz, J.; Khodade, V.S.; Toscano, J.P. Reaction of Nitroxyl (HNO) with Hydrogen Sulfide and Hydropersulfides. J. Org. Chem. 2021, 86, 868–877. [Google Scholar] [CrossRef] [PubMed]
- DeMaster, E.G.; Redfern, B.; Nagasawa, H.T. Mechanisms of inhibition of aldehyde dehydrogenase by nitroxyl, the active metabolite of the alcohol deterrent agent cyanamide. Biochem. Pharmacol. 1998, 55, 2007–2015. [Google Scholar] [CrossRef]
- Lopez, B.E.; Wink, D.A.; Fukuto, J.M. The inhibition of glyceraldehyde-3-phosphate dehydrogenase by nitroxyl (HNO). Arch. Biochem. Biophys. 2007, 465, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Dai, T.; Tian, Y.; Tocchetti, C.G.; Katori, T.; Murphy, A.M.; Kass, D.A.; Paolocci, N.; Gao, W.D. Nitroxyl increases force development in rat cardiac muscle. J. Physiol. 2007, 580, 951–960. [Google Scholar] [CrossRef] [PubMed]
- Ge, Y.; Moss, R.L. HNO, Redox Switches, Cardiac Myofilaments, and Heart Failure: A Prequel to Novel Therapeutics. Circ. Res. 2012, 23, 954–956. [Google Scholar] [CrossRef] [Green Version]
- Eberhardt, M.; Dux, M.; Namer, B.; Miljkovic, J.; Cordasic, N.; Will, C.; Kichko, T.I.; De La Roche, J.; Fischer, M.; Suárez, S.A.; et al. H2S and NO cooperatively regulate vascular tone by activating a neuroendocrine HNO-TRPA1-CGRP signalling pathway. Nat. Commun. 2014, 5, 4381. [Google Scholar] [CrossRef]
- Peng, H.; Shen, J.; Edmonds, K.A.; Luebke, J.L.; Hickey, A.K.; Palmer, L.D.; Chang, F.-M.J.; Bruce, K.A.; Kehl-Fie, T.E.; Skaar, E.P.; et al. Sulfide Homeostasis and Nitroxyl Intersect via Formation of Reactive Sulfur Species in Staphylococcus aureus. mSphere 2017, 2, e00082-17. [Google Scholar] [CrossRef] [Green Version]
- Shen, J.; Walsh, B.J.C.; Flores-Mireles, A.L.; Peng, H.; Zhang, Y.; Zhang, Y.; Trinidad, J.C.; Hultgren, S.J.; Giedroc, D.P. Hydrogen Sulfide Sensing through Reactive Sulfur Species (RSS) and Nitroxyl (HNO) in Enterococcus faecalis. ACS Chem. Biol. 2018, 13, 1610–1620. [Google Scholar] [CrossRef]
- Yadav, R.; Goldstein, S.; Nasef, M.O.; Lee, W.; Samuni, U. Synergistic activity of acetohydroxamic acid on prokaryotes under oxidative stress: The role of reactive nitrogen species. Free Radic. Biol. Med. 2014, 77, 291–297. [Google Scholar] [CrossRef]
- Sirsalmath, K.; Suárez, S.A.; Bikiel, D.E.; Doctorovich, F. The pH of HNO donation is modulated by ring substituents in Piloty’s acid derivatives: Azanone donors at biological pH. J. Inorg. Biochem. 2013, 118, 134–139. [Google Scholar] [CrossRef]
- Chauhan, P.; Bora, P.; Ravikumar, G.; Jos, S.; Chakrapani, H. Esterase activated carbonyl sulfide/hydrogen sulfide (H2S) donors. Org. Lett. 2017, 19, 62–65. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Xu, L.; Ou, S.; Edwards, H.; Luedtke, D.; Ge, Y.; Qin, Z. H2O2/Peroxynitrite-Activated Hydroxamic Acid HDAC Inhibitor Prodrugs Show Antileukemic Activities against AML Cells. ACS Med. Chem. Lett. 2018, 9, 635–640. [Google Scholar] [CrossRef] [PubMed]
- Kazuyuki, A.; Hidehiko, N.; Kazuya, M.; Kodai, K.; Naoya, I.; Takayoshi, S.; Naoki, M. Piloty’s acid derivative with improved nitroxyl-releasing characteristics. Bioorg. Med. Chem. Lett. 2013, 23, 2340–2343. [Google Scholar] [CrossRef]
- Porcheddu, A.; De Luca, L.; Giacomelli, G. A Straightforward Route to Piloty’s Acid Derivatives: A Class of Potential Nitroxyl-Generating Prodrugs. Synlett 2009, 13, 2149–2153. [Google Scholar] [CrossRef]
- Gjonaj, L.; Roelfes, G. Selective chemical modification of DNA with alkoxy- and benzyloxyamines. Org. Biomol. Chem. 2015, 13, 6059–6065. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Sommers, E.M.; Kim-Shapiro, D.B.; King, S.B. Horseradish peroxidase catalyzed nitric oxide formation from hydroxyurea. J. Am. Chem. Soc. 2002, 124, 3473–3480. [Google Scholar] [CrossRef] [PubMed]
- Rice, A.M.; Faig, A.; Wolff, D.E.; King, S.B. Sodium borohydride and thiol mediated nitrite release from nitroaromatic antibiotics. Bioorganic Med. Chem. Lett. 2021, 48, 128245. [Google Scholar] [CrossRef]
- Tomaszewska, J.; Koroniak-Szejn, K.; Koroniak, H. Fluorinated organic azides—their preparation and synthetic applications. Arkivoc 2016, 2017, 421–432. [Google Scholar] [CrossRef] [Green Version]
- Montoya, L.A.; Pluth, M.D. Selective turn-on fluorescent probes for imaging hydrogen sulfide in living cells. Chem. Commun. 2012, 48, 4767–4769. [Google Scholar] [CrossRef]
- Ji, Y.; Wang, Y.; Zhang, N.; Xu, S.; Zhang, L.; Wang, Q.; Zhang, Q.; Hu, H.Y. Cell-Permeable Fluorogenic Probes for Identification and Imaging Nitroreductases in Live Bacterial Cells. J. Org. Chem. 2019, 84, 1299–1309. [Google Scholar] [CrossRef]
- Dillon, K.M.; Morrison, H.A.; Powell, C.R.; Carrazzone, R.J.; Ringel-Scaia, V.M.; Winckler, E.W.; Council-Troche, R.M.; Allen, I.C.; Matson, J.B. Targeted Delivery of Persulfides to the Gut: Effects on the Microbiome. Angew. Chemie 2021, 133, 6126–6132. [Google Scholar] [CrossRef]
- Jackson, M.I.; Han, T.H.; Serbulea, L.; Dutton, A.; Ford, E.; Miranda, K.M.; Houk, K.N.; Wink, D.A.; Fukuto, J.M. Kinetic feasibility of nitroxyl reduction by physiological reductants and biological implications. Free Radic. Biol. Med. 2009, 47, 1130–1139. [Google Scholar] [CrossRef] [PubMed]
- Toscano, J.P.; Brookfield, F.A.; Cohen, A.D.; Courtney, S.M.; Frost, L.M.; Kalish, V.J. N-Hydroxylsulfonamide Derivatives as New Physiologically Useful Nitroxyl Donors. World Intellect. Prop. Organ. Int. Bur. 2007, 1–75. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).