Abstract
Green synthesis of silver nanoparticles (AgNPs) has gained greater interest among chemists and researchers in this current scenario. The present research investigates the larvicidal and anti-proliferation activity of AgNPs derived from Knoxia sumatrensis aqueous leaf extract (K. sumatrensis-ALE) as a potential capping and reducing candidate. The synthesized AgNPs were characterized through-UV-spectra absorption peak at 425 nm. The XRD and FT-IR studied displayed the crystalline nature and presence of functional groups in prepared samples. FE-SEM showed the hexagonal shape of NPs with the size of 7.73 to 32.84 nm. The synthesized AgNPs displayed superior antioxidant and anti-proliferative activity (IC50 53.29 µg/mL) of breast cancer cell line (MCF-7). Additionally, larvicidal activity against mosquito vector Culex quinquefasciatus larvae delivered (LC50-0.40, mg/L, and LC90-15.83) significant mortality rate post treatment with synthesized AgNPs. Overall, the present research illustrates that the synthesized AgNPs have high biological potential and present a perfect contender in the pharmacological and mosquitocidal arena.
1. Introduction
Noble metal nanoparticles (MNPs) have received considerable attention in recent years due to their various applications [1]. Various methods can achieve nanoparticle synthesis. Among these techniques, biological synthesis is rapid, simple and low cost and is the best method when compared to other synthesis methods [2]. The green synthesis process has benefits because it is an easy, inexpensive, and safe way to synthesize metal nanoparticles [3]. Additionally, employing plant extracts as a reducing agent makes it suitable for biomedical and pharmaceutical applications because no hazardous chemicals are utilized in the synthesis [4]. Recently, AgNPs have been synthesized using different plant species such as Capsicum chinense and Hypericum perforatum [5,6]. Knoxia sumatrensis belongs to the Rubiaceae. The plant has therapeutic uses in beverage preparation [7] and wound healing [8]. The extract of K. sumatrensis leaves was used to synthesize zinc oxide nanoparticles [9]. However, this is the first report on AgNPs synthesized by K. sumatrensis aqueous leaves extract.
Mosquitoes are considered to be global vectors of crucial diseases across the nation [10]. Dengue is spread by Aedes aegypti [11]. Anopheles stephensi is a carrier of plasmodium, which causes malaria [12]. Culex quinquefasciatus is a vector responsible for lymphatic filariasis [13]. The search for plant-based insecticides is underway, to find alternative mosquito control methods to chemical insecticides. Plant-based insecticides leave the least environmental footprint and can address the problem of insecticide resistance among mosquitoes to chemical insecticides [14]. Cancer is one of the leading diseases in developed countries. Its treatment primarily relies on surgery, chemo- and radio-therapies, although these are high cost, have side effects, and involve the death of normal cells along with cancer cells [15]. Consequently, novel chemotherapy drugs that have specific toxicity towards cancer cells and methods to directly target the tumor tissue are being actively explored. The use of nanoparticles with potential cytotoxic plant-based molecules is considered to be a vital way to treat cancer. Nanoparticles are efficient drug delivery vehicles that can be blended with bio-active plant compounds (secondary metabolites) derived from bio-rational plants for wide cancer therapeutic applications [16]. The present investigation highlights the characterization of green based Ag nanoparticles derived from white horseweed aqueous leaf extracts (K. sumatrensis-ALE) and their biological activity against the MCF-7 cell lines and larvicidal activity against the major mosquito vectors of medical importance.
2. Materials and Methods
2.1. Plant Collection and Extraction
Knoxia sumatrensis (Retz.) DC. was collected from Vytla hills Tamil Nadu, India, and authenticated by BSI. The leaves were washed, dried, and ground. Plant powder (10 g) was mixed with distilled water (100 mL) and boiled for 30 min at 70 °C. The K. sumatrensis-ALE was cooled and filtered with Whatman filter paper no.1 and stored at 4 °C for further use.
2.2. Synthesis
K. sumatrensis-ALE (10 mL) and 90 mL of silver nitrate (1 mM-AgNO3) were mixed and then stirred (3 h at 80 °C). After 3 h the color was changed from yellow into dark brown. The color indicated the formation of the AgNPs. The synthesized NPs were centrifuged at 10,000 for 15 min to obtain pellets, and then stored at 4 °C for further work.
2.3. Characterization
The synthesized AgNPs using K. sumatrensis extract were characterized by UV-Vis spectroscopy (UV-Vis-Shimadzu-1800). X-ray diffraction (XRD-Rigaku Miniflex) and analyzed by its crystalline structure. FT-IR indicated the functional groups, and nanoparticle morphology was analyzed by scanning electron microscopy (SEM- Jeol-6390LA).
2.4. Antioxidant
K. sumatrensis in synthesis of AgNPs was performed in DPPH by Shimada et al. [17], H2O2 Rajeshwar et al. [18], and ABTS assays by Giao et al. [19] with standard protocol and a dosage of 20–100 μg/mL. This assay used the standard of ascorbic acid. The assay inhibition (%) was calculated below.
2.5. Anti-Proliferative Activity
2.5.1. Culture
Breast cancer cell-line (MCF-7) was obtained by NCCS-Pune, India, and it was grown in Dulbecco with DMEM elevated glucose medium.
2.5.2. MTT Assay
Cell viability assays were described by Mosmann [20]. Ninety-six well plates were seeded in MCF-7 cells and maintained at 37 °C for 24 h in the incubator. The AgNPs dosages of 6.5, 12.5, 25, 50 and 100 µg/mL were used. Cell viability was calculated (MTT 10 µL for 4 h at 37 °C) after 24 h treatment. Dimethyl sulfoxide (DMSO) was dissolved in treated cells. The ELISA instrument (2.0-Epoch-USA) measured (OD-540 nm) the formazan in crystals (reference: 630 nm). Cell morphology was captured and calculated below.
Cell viability % = OD of AgNPs/OD of Control (Untreated) × 100%
2.6. Larivicidal Activity
2.6.1. Larval Culture
Ae. aegypti, An. stephensi and Cx. quinquefasciatus were collected from ICMR-VCRC Madurai) and maintained in the laboratory condition.
2.6.2. Bioassay
This assay was followed by WHO [21] standard procedure with slight change (Thandapani et al. [22]). Larvae (4th instar) in twenty numbers were added from each cup (200 mL). The concentration of K.sumatrensis-ALE and synthesized AgNPs (5–25 mg/L) was added. The dead larva were counted after 12, 24 and 48 h post treatment then mortality (%) was obtained from average of n = 3. Abbott [23] was used in correction of larval mortality.
2.7. Statistical Analysis
The data were analyzed by mean ± SD. Larval mortality was calculated by Probit analysis for finding out LC50, LC90 and chi-square values using SPSS software. All the data were analyzed with analysis of variance (ANOVA), and treatment means were compared by Tukey’s family error test (p < 0.05) for pairwise comparison using Minitab® 16 software package.
3. Results and Discussion
3.1. UV-Vis Spectral Analysis
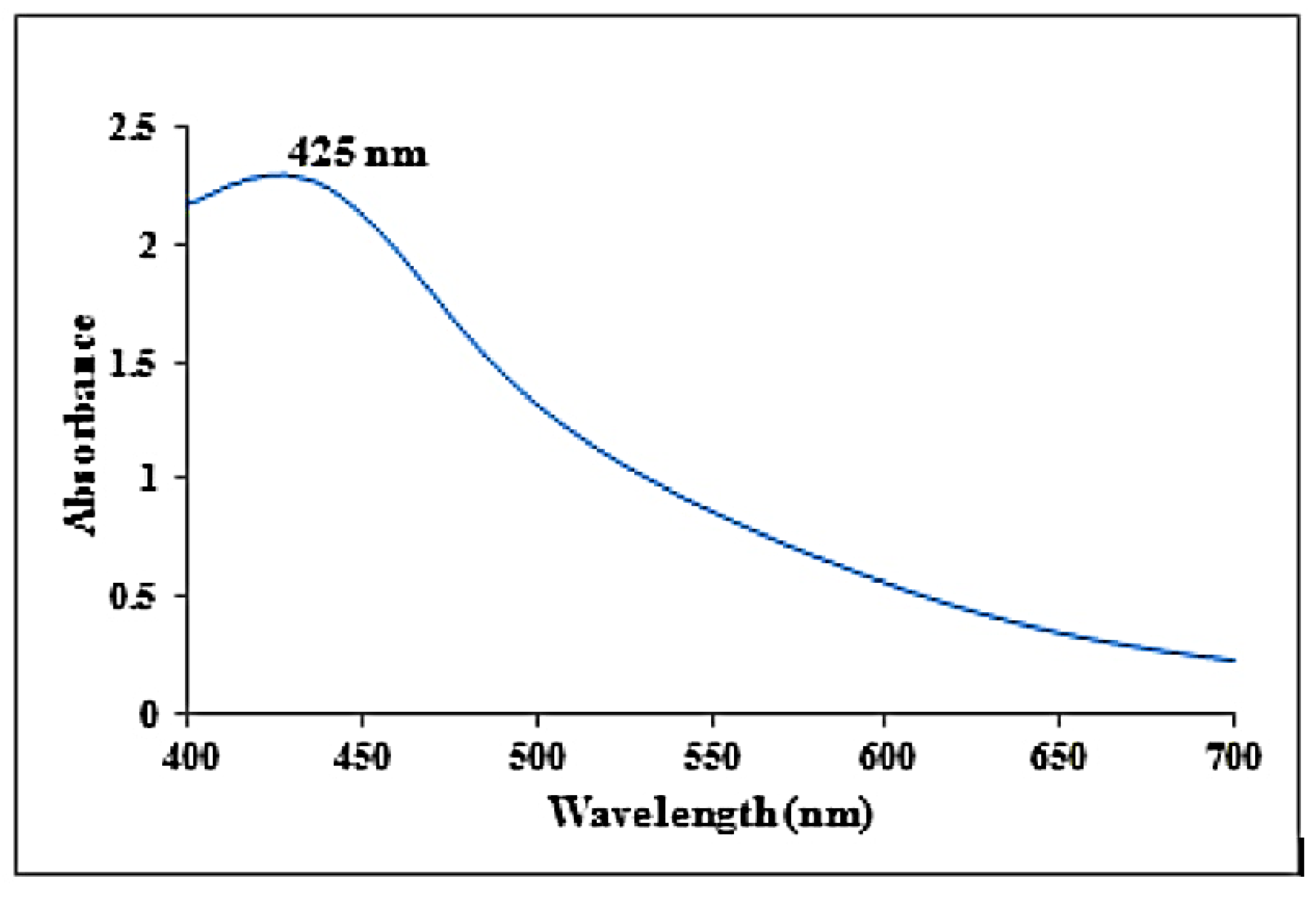
The synthesized AgNPs from K.sumatrensis extract showed the UV absorption peak at 425 nm due to the surface plasmon resonance (SPR), as shown in Figure 1. The mechanism of green synthesis of silver nanoparticles is due the constituent secondary metabolites donating electrons for the reduction of Ag+ ions to Ag° ions [24]. As compared to our results, the recent reports of silver nanoparticles synthesized from Passiflora subpeltata leaf extract demonstrated the UV absorption peak at 456 nm [16]. Additionally, the AgNPs of Acacia concinna leaf extract exhibited the UV absorption peak at 440 nm [25].
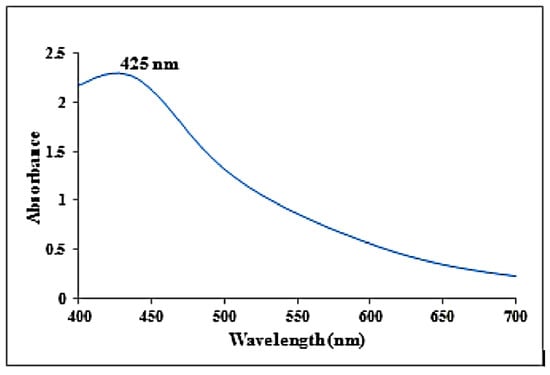
Figure 1.
UV-visible absorbance spectra obtained from synthesized silver nanoparticles using K. sumatrensis aqueous leaf extracts (Ks-ALE).
3.2. X-ray Diffraction Studies
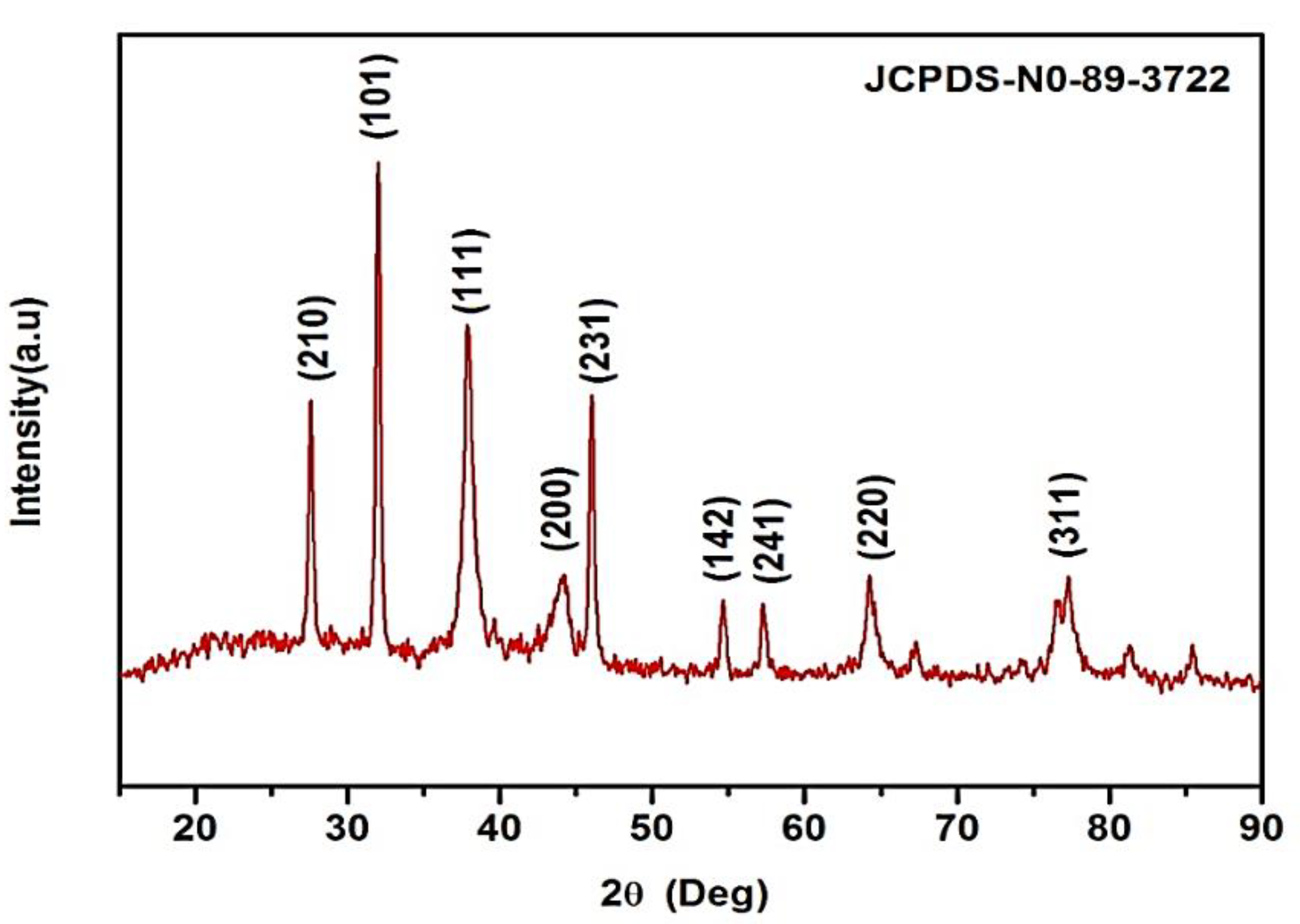
K. sumatrensis of synthesized AgNPs in XRD results showed the nine peaks at 2θ degree ranges of 27.87°, 32.14°, 38.77°, 44.20°, 46.10°, 54.40°, 57.34°, 64.34° and 77.72° corresponding to values 210, 101, 111, 200, 231, 142, 241, 220 and 311 (Figure 2). The lattice planes of pure silver based on the face-center cubic structure (JCPDS No. 89–3722) and the XRD results indicate the crystalline nature of AgNPs. Similar data were found for synthesized AgNPs using Sargassum myriocystum extract [26]. In another report, Drimia polyantha derived AgNPs revealed the presence of four diffraction values at 2θ ranges [27]. The average crystal size of synthesized AgNPs was 15.70 nm using Debye Scherrer’s formula.
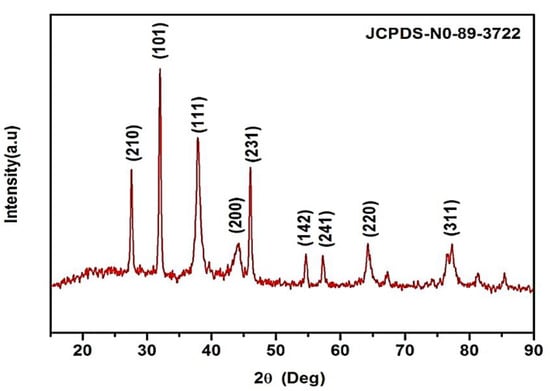
Figure 2.
XRD pattern of synthesized AgNPs using K. sumatrensis aqueous leaf extracts (Ks-ALE) exhibiting the facets of crystalline silver.
3.3. FT-IR Study
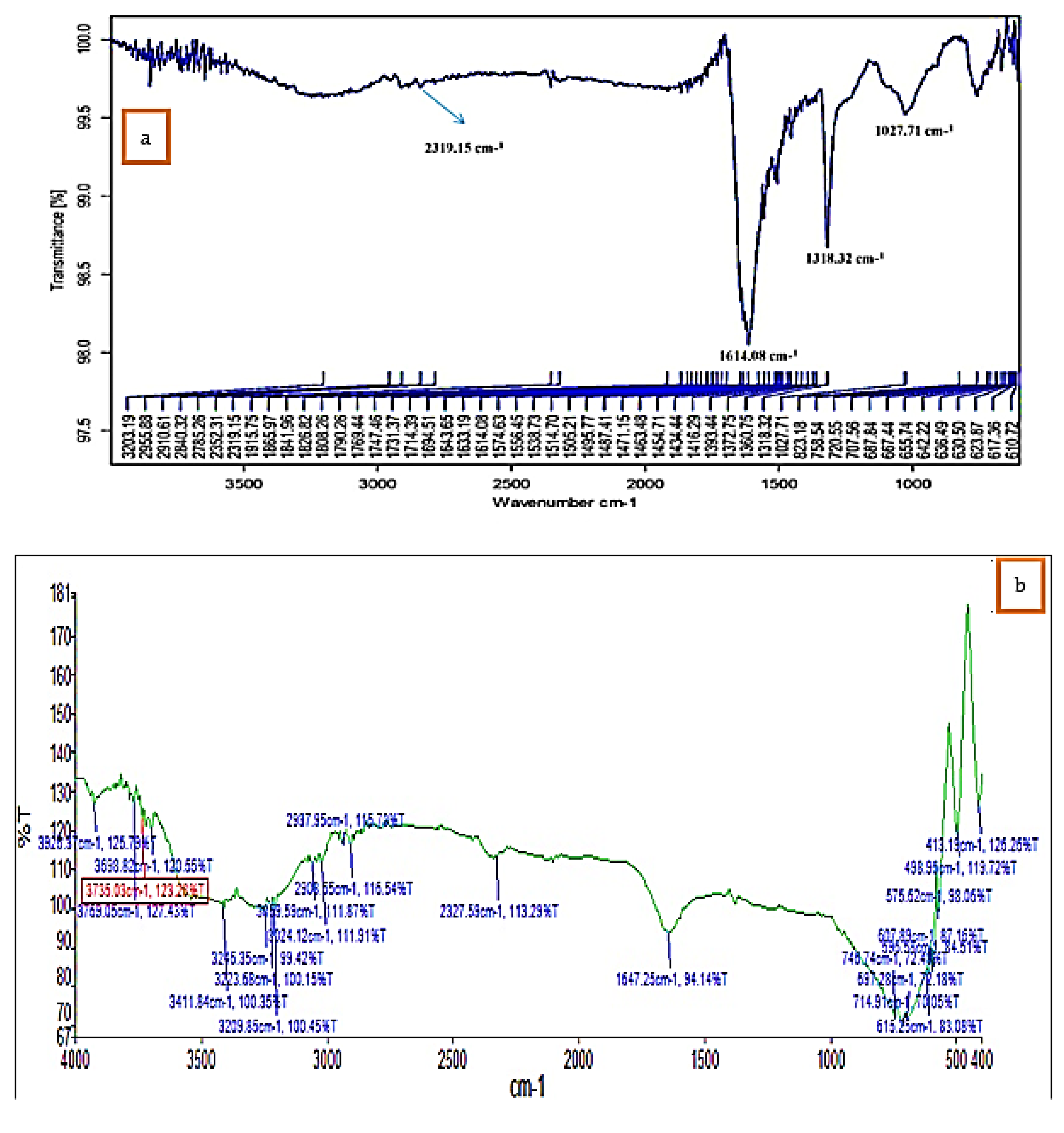
FTIR analysis confirmed that the bioproduction of Ag+ ions to silver nanoparticles was due to the reduction of capping material of green extract. The IR spectra of K. sumatrensis aqueous leaf extract revealed four functional groups: 3411.84 cm−1 for OH (alcohols, phenols), 3209.85 cm−1 for OH (carboxylic acids), 2908.55 cm−1 for C-H (alkanes), 2327.59 cm−1 for P-H (phosphines) and 1647.25 cm−1 for C=C (alkene) (Figure 3; Table 1). The synthesized AgNPs of K.sumatrensis extract in IR spectra showed the four functional groups as: 2319 cm−1 for CH (methylene), 1614.08 cm−1 by C=C stretching (alkenes), 1318.32 cm−1 indicating C-N (amines) and 1027.71 cm−1 C-X stretching (fluoride group) (Figure 3b; Table 2. Kumar et al. [28] reported that the carboxyl (–C=O), hydroxyl (–OH) and amine (–NH) groups of leaf extracts are importantly involved in fabrication of silver nanoparticles. Previous research of Morales-Lozoya et al. [29] illustrates that the synthesized AgNPs from Moringa citrifolia extract showed the presence of four functional groups. Correspondingly, the synthesized AgNPs by Cucumis prophetarum extract indicated the existence of eight functional groups [30]. The essential functional groups, such as alcohol, amides, alkanes, methyl, aliphatic and halides, confirmed the presence of NPs. They were stabilizing, capping and dipping agents of the AgNPs [31]. Thus, in this study, K. Sumatrensis derived AgNPs functional groups may be responsible for the formation of AgNPs.
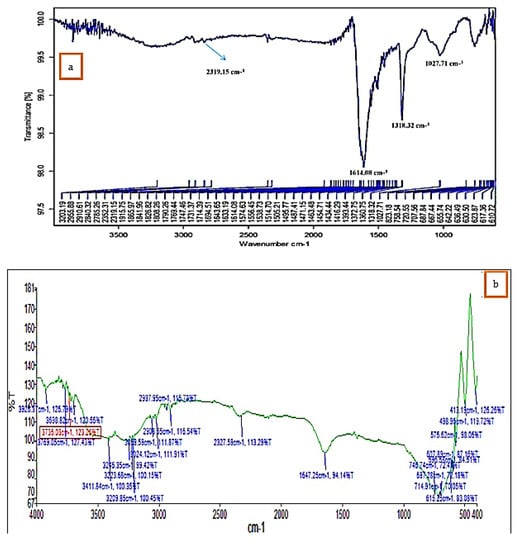
Figure 3.
FT−IR analysis of (a) K. sumatrensis aqueous leaf extracts (Ks-ALE). (b) Synthesized AgNPs.

Table 1.
FT-IR analysis of K. sumatrensis aqueous leaf extract (Ks-ALE).

Table 2.
FT-IR analysis of AgNPs using K. sumatrensis aqueous leaf extract (Ks-ALE).
3.4. SEM and EDAX Analysis
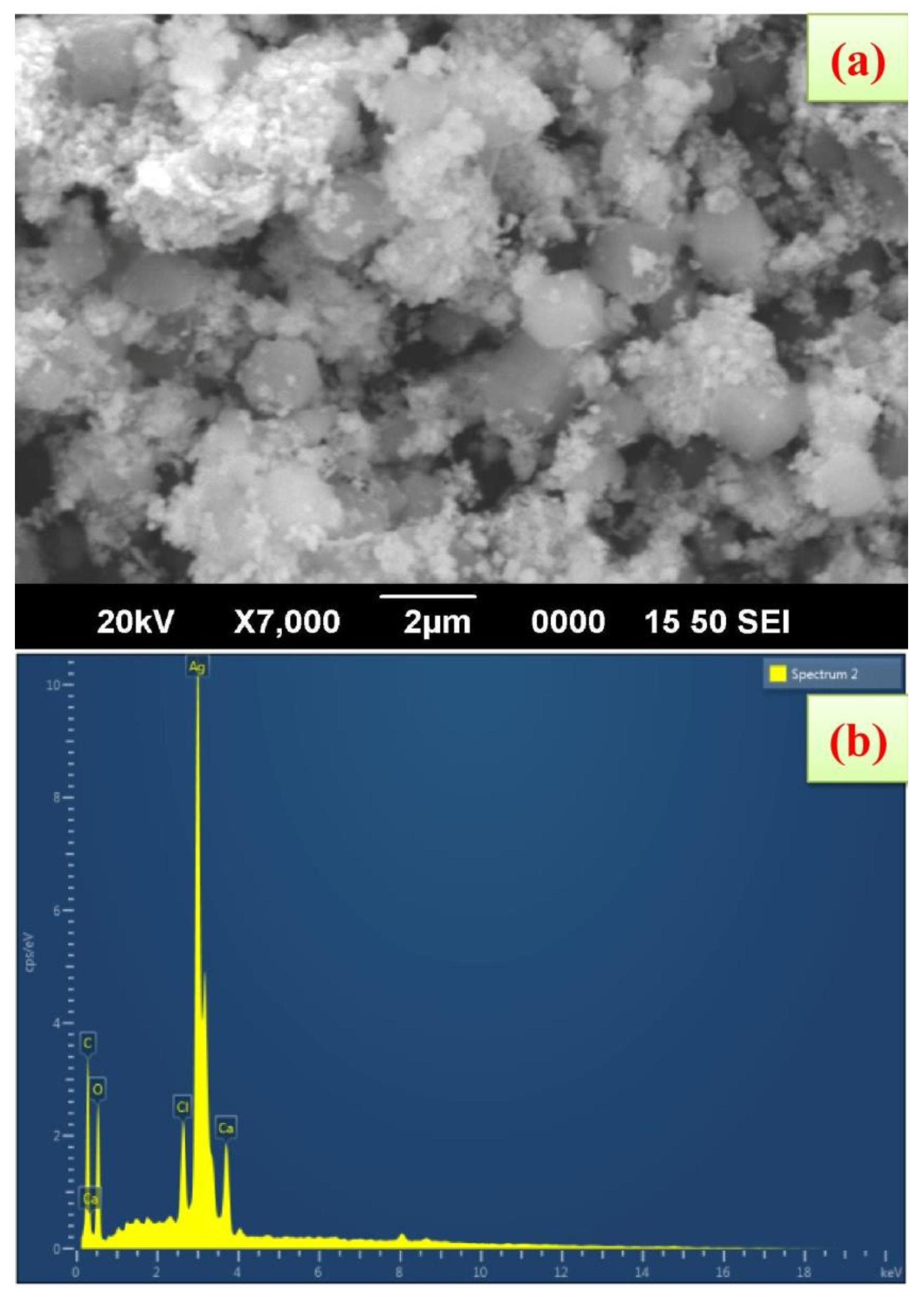
FE-SEM shows that the synthesized AgNPs using K. sumartrensis extract had hexagonal shapes (Figure 4). In parallel, the related shape was obtained by Calotropis gigantean and Sargassum myriocystum in synthesized AgNPs [25,32]. In the present study, the chemical composition was analyzed with EDX, and silver, carbon, oxygen, calcium and chlorine were present (Figure 4). Our research was in agreement with the previous investigation, which found that synthesized AgNPs of Plumeria alba leaf extract in EDX confirmed the presence of silver [33].
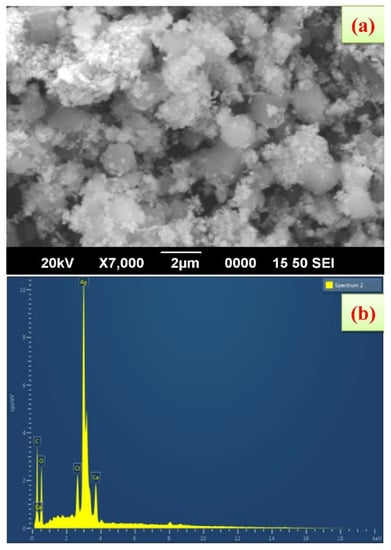
Figure 4.
Scanning electron microscope images of AgNPs synthesized using K. sumatrensis aqueous leaf extracts (Ks-ALE); (a) 2 µm; (b) EDAX spectrum analysis confirmed the presence of silver signals.
3.5. Antioxidant Activity
K. sumatrensis of synthesized AgNPs proved to have superior antioxidant activity against DPPH, ABTS and hydroxyl radical assays. In all three assays, inhibition percentages in synthesized AgNPs (55.20%, 53.15 and 53.70) and ascorbic acid (65.30%, 62.20 and 68.45), respectively, at higher concentrations of 100 µg/mL, were seen (Supplementary Material Figures S1–S3) in dose-dependent manner. Correspondingly, synthesized AgNPs derived from Eucalyptus tereticornis, commonly known as forest red gum, and Embeliaribes (common name: false black pepper), showed potential antioxidant activity [34,35]. The synthesized AgNPs and ascorbic acid (standard) DPPH IC50 values were calculated to be 92.73 and 79.37, respectively. In parallel, the synthesized AgNPs derived from Nepeta leucophylla (common name: white leaved catmint) root extract delivered significant antioxidant activity (IC50 value of 119.28 µg/mL) [36]. Previous research illustrates that the DPPH radical scavenging capacity of the synthesized AgNPs showed the presence of phenolic derivatives that can deliver the H into OH groups [37]. The ABTS inhibitory concentration (IC50) of the synthesized AgNPs displayed 94.68 μg/mL as compared to standard IC50 value of 79.48 µg/mL, and it is in parallel with the previous investigation on Cymbopogon citrates (common name: lemon grass) derived AgNPs, which revealed the IC50 value of 123.89 µg/mL [38]. The H2O2-IC50 value of the synthesized AgNPs showed significant range of 91.46 μg/mL as compared to the standard (IC50:71.47 μg/mL). The present research was well matched with the previous findings that Cassia angustifolia (common name: Indian senn) flowers extract of AgNPs delivered IC50 value of 78.10 µg/mL [39].
3.6. Anti-Proliferative Activity
Figures S4 and S5 show the cell viability (%) and treated images of MCF-7 cell line by the synthesized AgNPs from K. sumatrensis extract in a dose dependent manner. The percentage of viability is directly proportional to the synthesized AgNPs dosages (6.5 to 100 µg/mL) treatments. The inhibitory dosage IC50 of synthesized K. sumatrensis was 5.3 at 29 µg/mL at the maximum dosage of 100 µg/mL. Correspondingly, the synthesized AgNPs from Syzgium aromaticum (common name: clove) delivered profound MCF-7 activity (IC50-60 µg/mL) [40]. Similar results were obtained in the treatment of Camellia sinensis (common name: tea shrub) (IC50-59.2 µg/mL) [41] and also in Cassia angustifolia (Indian senna) (IC50-73.82 µg/mL) [39]. All the above findings were in line with our present investigation, which reveals that the K. sumatrensis of AgNPs delivered significant MCF-7 activity.
3.7. Larvicidal Activity
K. sumatrensis-ALE and the synthesized AgNPs displayed significant larvicidal activity against all three crucial mosquito vectors and the mortality rate was significant in the filarial vector Cx. quinquefasciatus as compared to dengue (Ae. aegypti) and malarial vector (A. stephensi) in the preliminary screening. The lethal concentrations (LC50 and LC90) were displayed at 0.40 mg/L and 15.83 mg/L, respectively, post 48 h treatment with K. sumatrensis-ALE (Tables S1–S3). In parallel, the synthesized AgNPs derived from Ixorabrachiata (Gorbale) and Carmona retusa (Fukien tea tree) extract delivered significant larvicidal activity against the filarial vector Cx. quinquefasciatus [42,43].
4. Conclusions
As an endnote, the present biological screening of green synthesized K. sumatrensis-ALE displayed multipotent biological activity as an anti-cancer and larvicidal agent and the present baseline toxicological screening suggests further interest in this eco-friendly green extract derived from white horseweed, as a potential eco-friendly drug and mosquitocide. Future prospective research is highly required to determine the molecular mechanisms and pharmacokinetic activity of bioactive molecules derived from K. sumatrensis using an in vivo rat model and field trial to determine the mosquitocidal actions and their non-target toxicity against beneficial insects which share the same ecological niche of mosquito vectors.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/molecules27227854/s1. Figure S1: Antioxidant activity of AgNPs synthesized using K. sumatrensis aqueous leaf extracts (Ks-ALE). DPPH radical scavenging activity, (b) ABTS radical scavenging activity and (c) Hydroxyl scavenging activity. Figure S4: MTT assay confirming the Anti-proliferative effects of AgNPs using aqueous leaf extracts of K. sumatrensis (Ks-ALE) against MCF-7 cell line. Figure S5: Anti-proliferative observed from confocal microscope (340 pixel); Control and various concentrations (6.5, 12.5, 25, 50 and 100 µg/mL) of K. sumatrensis aqueous leaf extract (Ks-ALE) of AgNPstreated on breast cancer cells (MCF-7). Table S1: Larvicidal activity of K. sumatrensis aqueous leaf extract (Ks-ALE) and Synthesized AgNPs against Aedes aegypti. TableS2: Larvicidal activity of K. sumatensis aqueous leaf extract (Ks-ALE) and synthesized AgNPs against Anopheles stephensi. Table S3: Larvicidal activity of K. sumatrensis aqueous leaf extract (Ks-ALE) and synthesized AgNPs against Culex quinquefasciatus.
Author Contributions
S.L. and K.S. designed and drafted the manuscript and also performed the experimental works; M.S.S. and S.S.-N. coordinated the work and discussed the results; P.V.-S., S.K., S.M., F.A.-M., A.A.-M., A.G. and D.G.P. performed data compilation and revised the manuscript; P.K. revised the manuscript and organized funding for the research. All authors have read and agreed to the published version of the manuscript.
Funding
This research work was partially supported by Chiang Mai University, Thailand. The authors express their sincere appreciation to the Researchers Supporting Project No. (RSP-2021-24) the King Saud University, Riyadh, Saudi Arabia. This research was also supported by the department of science and technology (DST-FIST), India under FIST program (SR/FIST/LS-1/2019/522) to S.S.-N.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All relevant data are within the paper and its Supporting Information files.
Acknowledgments
The authors acknowledge Periyar University, Salem, for providing University Research Fellowship (Ref. No. PU/AD-3/URF/2016). The authors also thank the Department of Botany, School of Life Sciences, Periyar University, Salem, Tamil Nadu-636 011, India, for providing the infrastructural facility. The authors express their sincere appreciation to the Researchers Supporting Project No. (RSP-2021-24) the King Saud University, Riyadh, Saudi Arabia.
Conflicts of Interest
The authors declare no conflict to interest.
Sample Availability
Samples of the compounds Silver Nanoparticle (AgNPs) of Knoxia Sumatrensis are currently not available from the authors.
References
- Csakvari, A.C.; Moisa, C.; Radu, D.G.; Olariu, L.M.; Lupitu, A.I.; Panda, A.O.; Copolovici, D.M. Green synthesis, characterization, and antibacterial properties of silver nanoparticles obtained by using diverse varieties of Cannabis sativa leaf extracts. Molecules 2021, 26, 4041. [Google Scholar] [CrossRef] [PubMed]
- Nasrollahzadeh, M.; Sajjadi, M.; Dadashi, J.; Ghafuri, H. Pd-based nanoparticles: Plant-assisted biosynthesis, characterization, mechanism, stability, catalytic and antimicrobial activities. Adv. Colloid. Interface Sci. 2020, 276, 102103. [Google Scholar] [CrossRef]
- Taha, A.; Ben Aissa, M.; Da’na, E. Green synthesis of an activated carbon-supported Ag and ZnO nanocomposite for photocatalytic degradation and its antibacterial activities. Molecules 2020, 25, 1586. [Google Scholar] [CrossRef] [PubMed]
- Da’na, E.; Taha, A.; Afkar, E. Green synthesis of iron nanoparticles by Acacia nilotica pods extract and its catalytic, adsorption, and antibacterial activities. Appl. Sci. 2018, 8, 1922. [Google Scholar] [CrossRef]
- Lomelí-Rosales, D.A.; Zamudio-Ojeda, A.; Reyes-Maldonado, O.K.; López-Reyes, M.E.; Basulto-Padilla, G.C.; Lopez-Naranjo, E.J.; Velázquez-Juárez, G. Green synthesis of gold and silver nanoparticles using leaf extract of Capsicum chinense plant. Molecules 2022, 27, 1692. [Google Scholar] [CrossRef] [PubMed]
- Alahmad, A.; Feldhoff, A.; Bigall, N.C.; Rusch, P.; Scheper, T.; Walter, J.G. Hypericum perforatum L. -mediated green synthesis of silver nanoparticles exhibiting antioxidant and anticancer activities. Nanomaterials 2021, 11, 487. [Google Scholar] [CrossRef] [PubMed]
- Vivek, K.; Rao, R. Some interesting indigenous beverages among the tribals of Central India. Ind. J. Trad. Knowl. 2007, 6, 141–143. [Google Scholar]
- Ayyanar, M.; Ignacimuthu, S. Herbal medicines for wound healing among tribal people in Southern India: Ethnobotanical and Scientific evidences. Int. J. Appl. Res. Nat. Prod. 2009, 2, 29–42. [Google Scholar]
- Loganathan, S.; Shivakumar, M.S.; Karthi, S.; Senthil-Nathan, S.; Selvam, K. Metal oxide nanoparticle synthesis (ZnO-NPs) of Knoxia sumatrensis (Retz.) DC. Aqueous leaf extract and It’s evaluation of their antioxidant, anti-proliferative and larvicidal activities. Toxicol. Repor. 2021, 8, 64–72. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Dengue and Severe Dengue. 2009. Available online: https://apps.who.int/mediacentre/factsheets/fs117/en/index.html (accessed on 14 April 2021).
- World Health Organization (WHO). Promoting Dengue Vector Surveillance and Control. 2018. Available online: https://www.who.int/denguecontrol/disease/en/ (accessed on 23 April 2021).
- World Health Organization (WHO). World Malaria Report 2018. Available online: https://www.who.int/publications/i/item/9789241565653 (accessed on 23 April 2021).
- World Health Organization (WHO). Lymphatic filariasis: Managing morbidity and preventing disability: An aide-mémoire for national programme managers, 2nd ed. 2018. Available online: https://www.who.int/publications/i/item/lymphatic-filariasis-managing-morbidity-and-preventing-disability-an-aide-m%C3%A9moire-for-national-programme-managers-2nd-ed (accessed on 23 April 2021).
- Senthil-Nathan, S. A review of resistance mechanisms of synthetic insecticides and botanicals, phytochemicals, and essential oils as alternative larvicidal agents against mosquitoes. Front. Physiol. 2019, 10, 1591. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Vaseeharan, B.; Sudhakaran, R.; Jeyakandan, J.; Ramasamy, P.; Sonawane, A.; Faggio, C. Bioinspired zinc oxide nanoparticles using Lycopersicon esculentum for antimicrobial and anticancer applications. J. Clust. Sci. 2019, 30, 1465–1479. [Google Scholar] [CrossRef]
- Loganathan, S.; Selvam, K.; Padmavathi, G.; Shivakumar, M.S.; Senthil-Nathan, S.; Gounder, S.A.; Ali, M.A.; Almutairi, S.M. Biological synthesis and characterization of Passiflora subpeltata ortega aqueous leaf extract in silver nanoparticles and their evaluation of antibacterial, antioxidant, anti-cancer and larvicidal activities. J. King Saud Univ. Sci. 2022, 22, 101846. [Google Scholar] [CrossRef]
- Shimada, K.; Fujikawa, K.; Yahara, K.; Nakamura, T. Antioxidative properties of xanthan on the autoxidation of soybean oil in cyclodextrin emulsion. J. Agric. Food. Chem. 1992, 40, 945–948. [Google Scholar] [CrossRef]
- Rajeshwar, Y.; Senthilkumar, G.P.; Malay, A.G.; Mazumder, U.K. Studies on in vitro antioxidant activities of methanol extract of Mucuma pruriens (Fabaceae) seeds. Europ. Bull. Drug. Res. 2005, 13, 131–138. [Google Scholar]
- Senthil-Nathan, S. Physiological and biochemical effect of neem and other Meliaceae plants secondary metabolites against Lepidopteran insects. Front. Physiol. 2013, 4, 359. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods. 1983, 65, 55–63. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Communicable Disease Tool Kit, Sudan; WHO/CDS/2005; World Health Organization: Geneva, Switzerland, 2005; p. 26. [Google Scholar]
- Senthil-Nathan, S. A review of biopesticides and their mode of action against insect pest’s. In Environmental Sustainability—Role of Green Technologies; Springer: Berlin/Heidelberg, Germany, 2015; pp. 49–63. [Google Scholar]
- Abbott, W.S. A method of computing the effectiveness of an insecticide. J. Econ. Entomol. 1925, 18, 265–267. [Google Scholar] [CrossRef]
- Ssekatawa, K.; Byarugaba, D.K.; Kato, C.D.; Wampande, E.M.; Ejobi, F.; Nakavuma, J.L.; Kirabira, J.B. Green strategy–based synthesis of silver nanoparticles for antibacterial applications. Front. Nanotechnol. 2021, 3, 697303. [Google Scholar] [CrossRef]
- Shaikh, I.A.; Muddapur, U.M.; Bagewadi, Z.K.; Chiniwal, S.; Ghoneim, M.M.; Mahnashi, M.H.; Iqubal, S.M. Characterization of bioactive compounds from Acacia concinna and Citrus limon, silver nanoparticles’ production by A. concinna extract, and their biological properties. Molecules 2022, 27, 2715. [Google Scholar] [CrossRef]
- Balaraman, P.; Balasubramanian, D.; Kaliannan, D.; Durai, M.; Kamyab, H.; Park, S.; Maruthupandian, A. Phyco-synthesis of silver nanoparticles mediated from marine algae Sargassum myriocystum and its potential biological and environmental applications. Waste. Biomass. Valori. 2020, 11, 5255–5271. [Google Scholar] [CrossRef]
- Daphedar, A.; Taranath, T.C. Characterization and cytotoxic effect of biogenic silver nanoparticles on mitotic chromosomes of Drimia polyantha (Blatt. & McCann) Stearn. Toxicol. Repor. 2018, 5, 910–918. [Google Scholar]
- Kumar, S.P.; Darshit, P.; Ankita, P.; Palak, D.; Ram, P.; Pradip, P.; Kaliaperumal, S. Biogenic synthesis of silver nanoparticles using Nicotiana tobaccum leaf extract and study of their antibacterial effect. Afr. J. Biotechnol. 2011, 10, 8122–8130. [Google Scholar] [CrossRef]
- Morales-Lozoya, V.; Espinoza-Gómez, H.; Flores-López, L.Z.; Sotelo-Barrera, E.L.; Núñez-Rivera, A.; Cadena-Nava, R.D.; Rivero, I.A. Study of the effect of the different parts of Morinda citrifolia L. (noni) on the green synthesis of silver nanoparticles and their antibacterial activity. Appl. Surf. 2021, 537, 147855. [Google Scholar] [CrossRef]
- Hemlata, P.; Meena, R.; Singh, A.P.; Tejavath, K.K. Biosynthesis of silver nanoparticles using Cucumis prophetarum aqueous leaf extract and their antibacterial and antiproliferative activity against cancer cell lines. ACS Omega 2020, 5, 5520–5528. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Nalini, S.K.; Prakash, N.U.; Madhankumar, D. Biomimetic synthesis of silver nanoparticles by aqueous extract of Syzygium aromaticum. Mater. Lett. 2012, 75, 33–35. [Google Scholar] [CrossRef]
- Kumar, C.R.; Betageri, V.S.; Nagaraju, G.; Pujar, G.H.; Onkarappa, O.H.; Latha, M.S. Synthesis of core/shell (zno/ag) nanoparticles using Calotropis gigantea and their applications in photocatalytic and antibacterial studies. J. Inorg. Organomet. Polym. 2020, 30, 3410–3417. [Google Scholar] [CrossRef]
- Rudrappa, M.; Rudayni, H.A.; Assiri, R.A.; Bepari, A.; Basavarajappa, D.S.; Nagaraja, S.K.; Nayaka, S. Plumeria alba-mediated green synthesis of silver nanoparticles exhibits antimicrobial effect and anti-oncogenic activity against glioblastoma u118 mg cancer cell line. Nanomaterials 2022, 12, 493. [Google Scholar] [CrossRef]
- Kiran, M.S.; Betageri, V.S.; Kumar, C.R.; Vinay, S.P.; Latha, M.S. In-vitro antibacterial, antioxidant and cytotoxic potential of silver nanoparticles synthesized using novel Eucalyptus tereticornis leaves extract. J. Inorg. Organomet. Polym. 2020, 30, 2916–2925. [Google Scholar] [CrossRef]
- Dhayalan, M.; Denison, M.I.J.; Krishnan, K. In vitro antioxidant, antimicrobial, cytotoxic potential of gold and silver nanoparticles prepared using Embelia ribes. Nat. Prod. Res. 2017, 31, 465–468. [Google Scholar] [CrossRef]
- Singh, J.; Dhaliwal, A.S. Novel green synthesis and characterization of the antioxidant activity of silver nanoparticles prepared from Nepeta leucophylla root extract. Anal. Biochem. 2019, 52, 213–230. [Google Scholar] [CrossRef]
- Edwin, E.; Vasantha-Srinivasan, P.; Senthil-Nathan, S.; Thanigaivel, A.; Ponsankar, A.; Pradeepa, V.; Selin-Rani, S.; Kalaivani, K.; Hunter, W.B.; Abel-Megeed, A.; et al. Anti-dengue efficacy of bioactive andrographolide from Andrographis paniculata (Lamiales: Acanthaceae) against the primary dengue vector Aedes aegypti (diptera: Culicidae). Acta Trop. 2016, 163, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Ajayi, E.; Afolayan, A. Green synthesis, characterization and biological activities of silver nanoparticles from alkalinized Cymbopogon citratus Stapf. Adv. Nat. Sci. Nanosci. Nanotechnol. 2017, 8, 015017. [Google Scholar] [CrossRef]
- Bharathi, D.; Bhuvaneshwari, V. Evaluation of the cytotoxic and antioxidant activity of phyto-synthesized silver nanoparticles using Cassia angustifolia flowers. Bionanoscience 2019, 9, 155–163. [Google Scholar] [CrossRef]
- Venugopal, K.; Rather, H.A.; Rajagopal, K.; Shanthi, M.P.; Sheriff, K.; Illiyas, M.; Maaza, M. Synthesis of silver nanoparticles (Ag NPs) for anticancer activities (MCF 7 breast and A549 lung cell lines) of the crude extract of Syzygium aromaticum. J. Photochem. Photobiol. B 2017, 167, 282–289. [Google Scholar] [CrossRef]
- Yadav, A.; Mendhulkar, V.D. Antiproliferative activity of Camellia sinensis mediated silver nanoparticles on three different human cancer cell lines. J. Cancer Res. Ther. 2018, 14, 1316–1324. [Google Scholar] [PubMed]
- Sivasakthi, V.; Selvam, K.; Prakash, P.; Shivakumar, M.S.; Senthil-Nathan, S. Characterization of silver nanoparticles using Ixora brachiate Roxb. and its biological application. Curr. Opin. Green. Sustain. Chem. 2022, 28, 100257. [Google Scholar] [CrossRef]
- Rajkumar, R.; Shivakumar, M.S.; Senthil-Nathan, S.; Selvam, K. Preparation and characterization of chitosan nanocomposites material using silver nanoparticle synthesized Carmona retusa (Vahl) Masam leaf extract for antioxidant, anti-cancerous and insecticidal application. J. Clust. Sci. 2019, 30, 1145–1155. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).