Synthesis, Structure, and UV–Vis Characterization of Antimony(III) Phthalocyanine: [(SbPc)2(Sb2I8)(SbBr3)]2
Abstract
:1. Introduction
2. Experimental Section
2.1. General Procedure
2.2. Synthesis [(SbIIIPc)2(Sb2I8)(SbBr3)]2
2.3. X-ray Crystallography
2.4. Hirshfeld Surface Analysis
2.5. Theoretical Calculations
3. Results and Discussion
3.1. Synthesis and Initial Characterization
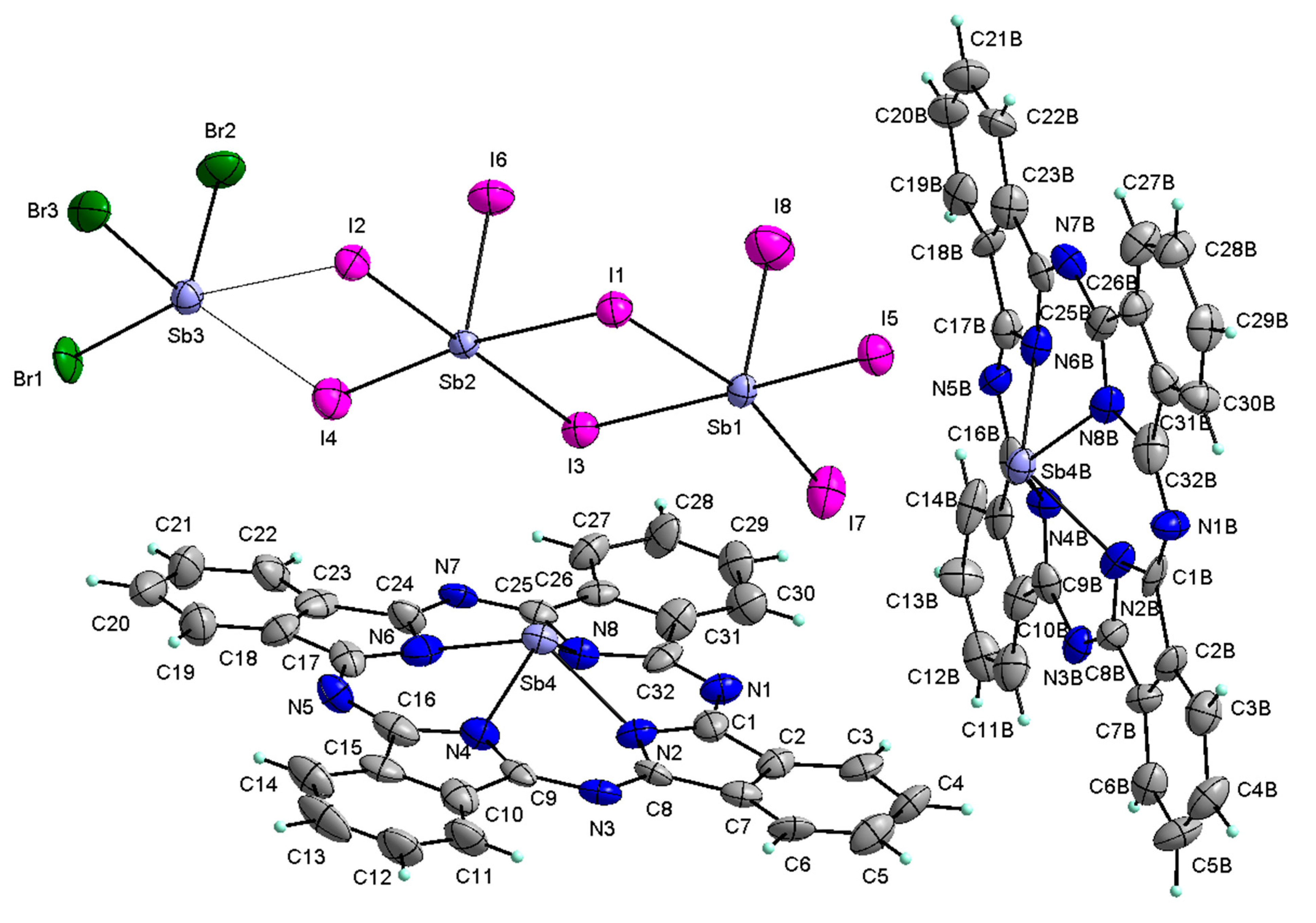
3.2. Structural Characterization
3.3. Hirshfeld Surface Analysis
3.4. UV–Vis Spectrosopy
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Herbst, W.; Hunger, K. Industrial Organic Pigments: Production, Properties, Applications; VCH: New York, NY, USA, 1993. [Google Scholar]
- Gregory, P. Industrial Application of Phthalocyanines. J. Porhyr. Phthalocyanines 2000, 4, 432–437. [Google Scholar] [CrossRef]
- Gregory, P. High-Technology Applications of Organic Colorants; CRC Press: Boca Raton, FL, USA, 1983. [Google Scholar]
- Leznoff, C.C.; Lever, A.B.P. (Eds.) Phthalocyanines: Properties and Applications; VCH Publications: New York, NY, USA, 1989; Volume 1. [Google Scholar]
- Leznoff, C.C.; Lever, A.B.P. (Eds.) Phthalocyanines: Properties and Applications; VCH Publications: New York, NY, USA, 1993; Volume 2. [Google Scholar]
- Leznoff, C.C.; Lever, A.B.P. (Eds.) Phthalocyanines: Properties and Applications; VCH Publications: New York, NY, USA, 1996; Volume 3. [Google Scholar]
- Leznoff, C.C.; Lever, A.B.P. (Eds.) Phthalocyanines: Properties and Applications; VCH Publications: New York, NY, USA, 1996; Volume 4. [Google Scholar]
- Thomas, A. Phthalocyanine Research and Applications; CRS Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Moser, F.H.; Thomas, A.L. The Phthalocyanines; CRC: Boca Raton, FL, USA, 1983. [Google Scholar]
- Gouterman, M. Spectra of Porhyrins. J. Mol. Spectrosc. 1961, 6, 138–163. [Google Scholar] [CrossRef]
- Falk, J.E. Porphyrins and Metalloporphyrins; Elsevier Pub. Co.: Amsterdam, The Netherlands; New York, NY, USA, 1964; p. 232. [Google Scholar]
- Whalley, M. Conjugated macrocycles. Part XXXII. Absorption spectra of tetrazaporphyrins and phthalocyanines. Formation of pyridine salts. J. Chem. Soc. 1961, 866–869. [Google Scholar] [CrossRef]
- Barnett, G.H.; Hudson, M.F.; Smith, K.M. Concerning meso-tetraphenylporphyrin purification. J. Chem. Soc. Perkin Trans. I 1975, 14, 1401–1403. [Google Scholar] [CrossRef]
- Ghani, F.; Kristen, J.; Riegler, H. Solubility Properties of Umsubstituted Metal Phthalocyanines in Different Types of Solvents. J. Chem. Eng. Data 2012, 57, 439–449. [Google Scholar] [CrossRef]
- Knӧr, G. Synthesis and solution spectral properties of antimony(III) phthalocyanine and dihydroxoantimony(V) phthalocyanine complexes. Inorg. Chem. 1996, 35, 7916–7918. [Google Scholar] [CrossRef]
- Modibane, D.K.; Nyokong, T. Synthesis, photophysical and photochemical properties of octa-substituted antimony phthalocyanines. Polyhedron 2009, 28, 479–484. [Google Scholar] [CrossRef]
- Wright, Gas adsorption on phthalocyanines and its effects on electrical properties. J. Prog. Surf. Sci. 1989, 31, 1–60. [CrossRef]
- Bassoul, P.; Toupance, T.; Simon, J. Semiconductivity and gas-sensing properties of crown-ether-substituted lutetium bisphthalocyanines. Sens. Actuators B 1995, 26, 150–152. [Google Scholar] [CrossRef]
- Torre, G.; Vázquez, P.; Agulló-López, F.; Torres, T. Phthalocyanines and related compounds: Organic targets for nonlinear optical applications. J. Mater. Chem. 1998, 8, 1671–1683. [Google Scholar] [CrossRef]
- Shirk, J.S.; Pong, R.G.S.; Flom, S.R.; Heckmann, H.; Hanack, M. Effect of Axial Substitution on the Optical Limiting Properties of Indium Phthalocyanines. J. Phys. Chem. A 2000, 104, 1438–1449. [Google Scholar] [CrossRef]
- Torre, G.; Vázquez, P.; Lopez, F.A.; Torres, T. Role of structural factors in the nonlinear optical properties of phthalocyanines and related compounds. Chem. Rev. 2004, 104, 3723–3750. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.; Chen, Q.; Shu, J.; Tang, X.; Fuxi, G.; Sten, S.; Liu, K.; Xu, X. Optical recording performance of thin films of phthalocyanine compounds. Thin Solid Films 1995, 275, 88–93. [Google Scholar] [CrossRef]
- Meng, Z.; Aykanat, A.; Mirica, K.A. Welding Metallophthalocyanines into Bimetallic Molecular Meshes for Ultrasensitive, Low-Power Chemiresistive Detection of Gases. J. Am. Chem. Soc. 2019, 141, 2046–2053. [Google Scholar] [CrossRef] [PubMed]
- Choy, C.H.W.; Liang, Y.J.; Hui, K.N.; Fong, H.H. Improving the efficiency of OLEDs by utilizing metallophthalocyanines. In Proceedings of the Optics and Photonics 2005, San Diego, CA, USA, 31 July–4 August 2005; Volume 5937. [Google Scholar] [CrossRef]
- Pearson, A.J.; Plint, T.; Jones, S.T.E.; Lessard, B.H.; Credgington, D.; Bender, T.P.; Greenham, N.C. Silicon phthalocyanines as dopant red emitters for efficient solution processed OLEDs. J. Mater. Chem. C 2017, 5, 12688–12698. [Google Scholar] [CrossRef]
- Bharati, M.S.S.; Bhattacharya, S.; Krishna, J.V.S.; Giribabu, L.; Rao, S.V. Femtosecond, broadband nonlinear optical studies of a zinc porphyrin and zinc phthalocyanine. Opt. Laser Technol. 2018, 108, 418–425. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Reddy, G.; Paul, S.; Hossain, S.S.; Raavi, S.S.K.; Giribabu, L.; Samanta, A.; Soma, V.R. Comparative photophysical and femtosecond third-order nonlinear optical properties of novel imidazole substituted metal phthalocyanines. Dyes Pigments 2021, 184, 108791. [Google Scholar] [CrossRef]
- Ben-Hur, E.; Chan, W.-S. Phthalocyanines in Photobiology and Their Medical Applications. In The Porphyrins Handbook; Kadish, K.M., Smith, K.M., Guilard, R., Eds.; Academic Press: Boston, FL, USA, 2003; Volume 19, pp. 1–35. [Google Scholar]
- Bonnett, R. Chemical Aspects of Photodynamic Therapy; Gordon and Breach Science Publishers: Amsterdam, The Netherlands, 2000. [Google Scholar]
- Moreira, L.M.; Vieira dos Santos, F.; Pereira Lyon, J.; Maftoum-Costa, M.; Pacheco-Soares, C.; Soares da Silva, N. Photodynamic Therapy: Porphyrins and Pthtalocyanines as Photosensitizers. Aust. J. Chem. 2008, 61, 741–754. [Google Scholar] [CrossRef] [Green Version]
- Lopez, T.; Ortiz, E.; Alvarez, M.; Navarrete, J.; Odriozola, A.; Martinez-Ortega, F.; Páez-Mozo, E.A.; Escobar, P.; Espinoza, K.A.; Rivero, I.A. Study of the stabilization of zinc phthalocyanine in sol-gel TiO2 for photodynamic therapy applications. Nanomed. Nanotechnol. Biol. Med. 2010, 6, 777–785. [Google Scholar] [CrossRef]
- Mfouo-Tynga, I.; Houreld, N.N.; Abrahamse, H. Induced Cell Death Pathway Post Photodynamic Therapy Using a Metallophthalocyanine Photosensitizer in Breast Cancer Cells. Photomed. Laser Surg. 2014, 32, 205–211. [Google Scholar] [CrossRef]
- Neagu, M.; Constantin, C.; Tampa, M.; Matei, C.; Lupu, A.; Manole, E.; Ion, R.M.; Fenga, C.; Tsatsakis, A.M. Toxicological and efficacy assessment of post-transition metal 9indium) phthalocyanine for photodynamic therapy in neuroblastoma. Oncotarget 2016, 7, 69718–69732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oluwole, D.O.; Manoto, S.L.; Malabi, R.; Maphanga, C.; Ombinda-Lemboumba, S.; Mthunzi-Kufa, P.; Nyokong, T. Evaluation of the photophysicochemical properties and photodynamic therapy activity of nanoconjugates of zinc phthalocyanine linked to glutathione capped Au and Au3Ag1 nanoparticles. Dyes Pigments 2018, 150, 139–150. [Google Scholar] [CrossRef]
- Dube, E.; Oluwole, D.O.; Prinsloo, E.; Nyokong, T. A gold-chitosan composite with low symmetry zinc phthalocyanine for enhanced singlet oxygen generation and improved photodynamic therapy activity. New J. Chem. 2018, 42, 10214–10225. [Google Scholar] [CrossRef]
- Dumoulin, F.; Durmus, M.; Ahsen, V.; Nyokong, T. Synthetic pathways to water-soluble phthalocyanines and close analogs. Coord. Chem. Rev. 2010, 254, 2792–2848. [Google Scholar] [CrossRef]
- Terekhov, D.S.; Nolan, K.J.M.; McArthur, C.R.; Leznoff, C.C. Synthesis of 2,3,9,10,16,17,23,24-Octaalkynylphthalocyanines and the Effects of Concentration and Temperature on Their 1H NMR Spectra. J. Org. Chem. 1996, 61, 3034–3040. [Google Scholar] [CrossRef]
- Isago, H.; Leznoff, C.C.; Ryan, M.F.; Metcalfe, R.A.; Davids, R.; Lever, A.B.P. Aggregation Effects on Electrochemical and Spectroelectrochemical Properties O {2,3,9,10,16,17,23,24-Octa(3,3-dimethyl-1-butynyl)phthalocyaninato]cobalt(II) Complex. Bull. Chem. Soc. Jpn. 1998, 71, 1039–1047. [Google Scholar] [CrossRef]
- Isago, H.; Terekhov, D.; Leznoff, C.C. Synthesis and NMR Studies of a Single Isomer of an Alkynyl Substituted Binuclear Phthalocyanine. J. Porphyrins Phthalocyanines 1997, 1, 135–140. [Google Scholar] [CrossRef] [Green Version]
- Würthner, F. (Ed.) Supermolecular Dye Chemistry; Springer: Berlin/Heiderberg, Germany, 2005. [Google Scholar]
- Menzel, E.R.; Rieckhoff, K.E.; Voigt, E.M. Dynamics of the triplet state of phthalocyanine complexes of the platinum metals in zero field. J. Chem. Phys. 1973, 58, 5726–5734. [Google Scholar] [CrossRef]
- Hanack, M.; Schneider, T.; Barthel, M.; Shirk, J.S.; Flom, S.R.; Pong, R.G.S. Indium phthalocyanines and naphthalocyanines for optical limiting. Coord. Chem. Rev. 2001, 219, 235–258. [Google Scholar] [CrossRef]
- Liu, Y.H.; Benassy, M.-F.; Chojnacki, S.; D’Souza, F.; Barbour, T.; Belcher, W.J.; Brothers, P.J.; Kadish, K.M. Electrochemical and Spectroelectrochemical Investigations of [(TpTP)MvL2]+Cl− Where TpTP is the Dianion of Tetra-p-tolyporphrin, M = P or Sb, and L = Cl− or OCH3−. Inorg. Chem. 1994, 33, 4480–4484. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Akiba, K.-y. The chemistry of Group 15 element porphyrins bearing element—Carbon bonds: Synthesis and properties. J. Oraganomet. Chem. 2000, 611, 200–209. [Google Scholar] [CrossRef]
- Kadish, K.M.; Van Caemelbecke, E.; Royal, G. Electrochemistry of Metalloporphyrins in Nonaqueous Media. In The Porphyrin Handbook; Kadish, K.M., Smith, K.M., Guillard, R., Eds.; Academic Press: San Diego, CA, USA, 2000; Volume 8, pp. 83–87. [Google Scholar]
- Satoh, W.; Masumoto, S.; Yamamoto, Y.; Akiba, K. Synthesis and properties of group 15 element porphyrin peroxides. Heteroat. Chem. 2001, 12, 431–443. [Google Scholar] [CrossRef]
- Brothres, P.J. Recent developments in the coordination chemistry of porphyrin complexes containing non-metallic and semi-metallic elements. J. Porphyr. Phthalocyanines 2002, 6, 259–267. [Google Scholar] [CrossRef]
- Isago, H.; Kagaya, Y. Syntheses and Characterization of Bromo- and Chloro(phthalocyaninato)bismuth(III) Complexes. Bull. Chem. Soc. Jpn. 1994, 67, 383–389. [Google Scholar] [CrossRef]
- Isago, H.; Kagaya, Y. Spectrocopic Properties of One-Electron-Reduced Species of Dichloro(phthalaninato)antimony(V) cation. Bull. Chem. Soc. Jpn. 1996, 69, 1281–1288. [Google Scholar] [CrossRef]
- Isago, H.; Kagaya, Y.; Nakajima, S.-I. Spectral Properties of Nonaggregative Antimony(V) Phthalocyanine and Its Film as a Novel Near-Infrred Absorber. Chem. Lett. 2003, 32, 112–113. [Google Scholar] [CrossRef]
- Isago, H.; Miura, K.; Kanesato, M. Unexpected photo-initiated oxidation of antimony in (tetra-tert-butyl)phthalocyaninatoantimony(III) complex in the presence of singlet oxygen acceptors. J. Photochem. Photobiol. A Chem. 2008, 197, 313–320. [Google Scholar] [CrossRef]
- Kubiak, R.; Razik, M. (Phthalocyaninato)antimony Iodide. Acta Cryst. C 1998, 54, 483–485. [Google Scholar] [CrossRef]
- Kubiak, R.; Janczak, J.; Razik, M. Synthesis and X-ray characterization of phthalocyaninato(2-)antimony(III) triiodide. Inorg. Chim. Acta 1999, 293, 155–159. [Google Scholar] [CrossRef]
- Janczak, J.; Kubiak, R.; Jezierski, A. Mixed-Valence, Dosordered Structures and Characterization of Iodine-doped Phthalocyanines: [YbPc2]I2 and [(AsPc)I2. Inorg. Chem. 1999, 38, 2043–2049. [Google Scholar] [CrossRef]
- Janczak, J.; Kubiak, R.; Richter, J.; Fuess, H. Bismuth triple-decer phthalocyanine: Synthesis and structure. Polyhedron 1999, 18, 2775–2780. [Google Scholar] [CrossRef]
- Kubiak, R.; Ejsmont, K. Crystal structure of a novel bismuth phthalocyanine-bismuth iodide complex. J. Mol. Struct. 1999, 474, 257–282. [Google Scholar] [CrossRef]
- Hückstüdt, H.; Tutass, A.; Gӧldner, M.; Cornelissen, U.; Homborg, H. Conformational Heterogeneity in Diphthalocyaninato(2-)metalates(III) of Sc, Y, In, Sb, Bi, La, Ce, Pr and Sm. Z. Anorg. Allg. Chem. 2001, 627, 485–497. [Google Scholar] [CrossRef]
- Janczak, J.; Kubiak, R. [Phthalocyaninato(2-)]antimony(III) chloride. Acta Cryst. C 2001, 57, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Janczak, J.; Idemori, Y.M. Tetrakis[phthalocyaninato(2-)antimony(III)] hexadecaiodotetraantimony(III). Acta Cryst. C 2002, 58, m23–m25. [Google Scholar] [CrossRef] [PubMed]
- Janczak, J.; Idemori, Y.M. {Phthalocyaninato(2-)]arsenic(III) triiodide. Acta Cryst. E 2002, 58, m36–m38. [Google Scholar] [CrossRef] [Green Version]
- Janczak, J.; Kubiak, R. Bis{[phthalocyaninato(2-)]arsenic(III)} tetrdecaiodotetraarsenic(III). Acta Cryst. C 2003, 59, m70–m72. [Google Scholar] [CrossRef] [Green Version]
- Perpetuo, G.J.; Janczak, J. Bis{[phthalocyaninato(2-)]antimony(III)} tetradecaiodotetraantimonate(III). Acta Cryst. E 2005, 61, m2003–m2005. [Google Scholar] [CrossRef]
- Janczak, J.; Perpetuo, G.J. Tetrakis[phthalocyaninato(2-)antimony(III)} docosaiodohexaantimony(III). Acta Cryst. C 2006, 62, m323–m326. [Google Scholar] [CrossRef]
- Janczak, J. Synthesis and characterization of bismuth(III) phthalocyaninate complex: [BiPc]4[Bi6I11Cl11]. J. Mol. Struct. 2010, 905, 125–130. [Google Scholar] [CrossRef]
- Kagaya, Y.; Isago, H. Facile Reduction of Dichloro(phthalocyaninato)antimony(V) Cation. Chem. Lett. 1994, 23, 1957–1960. [Google Scholar] [CrossRef]
- Isago, H.; Kagaya, Y. Facile antimony(V/III) interconversion in phthalocyanine complexes. Chem. Lett. 2006, 35, 8–9. [Google Scholar] [CrossRef]
- Kagaya, Y.; Isago, H. Synthesis of Dichloro(phthalocyaninato)antimony(V) Perchlorate, Tetrafkuoroborate, and hexafluorophosphate and Electrochemical Reinvestigation on the New Complex Salts. Bull. Chem. Soc. Jpn. 1997, 70, 2179–2185. [Google Scholar] [CrossRef]
- Janczak, J. The synthesis, structure and spectral properties of antimony(III) phthalocyanine obtained under iodine vapor atmosphere: (SbIIIPc)(I3)∙½(I2). Inorg. Chim. Acta 2022, 532, 120758. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Yan, S.; Jin, L.; Sun, H. 51Sb antimony in medicine. In Metallotherapeutic Drugs and Metal-Based Diagnostic Agents: The Use of Metals in Medicine; Gielen, M., Tiekink, E.R.T., Eds.; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2005. [Google Scholar]
- Carter, K.C.; Hutchinson, S.; Boitelle, A.; Murray, H.W.; Sundar, S.; Mullen, A.B. Sodium stibogluconate resistance in Leishmania donovani correlates with greater tolerance to macrophage antileishmanial responses and trivalent antimony therapy. Parasitology 2005, 131, 747–757. [Google Scholar] [CrossRef] [Green Version]
- Mandal, S.; Maharjan, M.; Singh, S.; Chatterjee, M.; Madhubala, R. Assessing aquaglyceroporin gene status and expression profile in antimony-susceptible and resistant clinical isolates of Leishmania donovani from India. J. Antimicrob. Chemother. 2010, 65, 496–507. [Google Scholar] [CrossRef] [Green Version]
- Hadjikakou, S.K.; Ozturk, I.I.; Banti, C.N.; Kourkoumelis, N.; Hadjiliadis, N. Recent advances on antimony(III/V) compounds with potential activity against tumour cells. J. Inorg. Biochem. 2015, 153, 293–305. [Google Scholar] [CrossRef]
- Maes, L.; Beyers, J.; Mondelaers, A.; Van den Kerkhof, M.; Eberhardt, E.; Caljon, G.; Hendrickx, S. In vitro ‘time-to-kill’ assay to assess the cidal activity dynamics of current reference drugs against Leishmania donovani and Leishmania infantum. J. Antimicrob. Chemother. 2017, 72, 428–430. [Google Scholar] [CrossRef] [Green Version]
- Kubiak, R.; Janczak, J. A simple, novel method for the preparation of metallophthalocyanines. J. Alloys Compd. 1993, 200, L7–L8. [Google Scholar] [CrossRef]
- CrysAlis CCD and CrysAlis Red 1.171.38.43; Rigaku Oxford Diffraction: Yaernaton, UK, 2015.
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal structure determination. Acta Cryst. A 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Brandenburg, K.; Putz, H. DIAMOND Version 3.0; Crystal Impact GbR: Bonn, Germany, 2006. [Google Scholar]
- Wolff, S.K.; Grimwood, D.J.; MacKimon, J.J.; Turner, M.J.; Jayatilaka, D. Spackman, Crystal Explorer ver. 3.1; University of Western Australia: Perth, Australia, 2013. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; et al. Gaussian16, Revision B.01; Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Janczak, J.; Kubiak, R. Tris(2-cyanophenyl)-1,3,5-triazine: A By-product of Metallophthalocyanine Synthesis. Acta Chem. Scand. 1999, 53, 602–610. [Google Scholar] [CrossRef] [Green Version]
- Barraclough, C.G.; Bissett, H.; Pitman, P.; Thustlethwaite, P.J. The infrared, Raman and ultraviolet spectra of phthalonitrile, terephthalonitrile and 2,6-dichlorobenzonitrile. Aust. J. Chem. 1977, 30, 753–765. [Google Scholar] [CrossRef]
- Ogunsipe, A.; Opeolu, S. Synthesis, Characterization and Photostability Studies on Aluminium Phthalocyanine Chloride. Chem. Res. J. 2020, 5, 136–143. [Google Scholar]
- Shurvell, H.F.; Pinzuti, L. Sur les spectrers infraeouges des phthalocyanines. Can. J. Chem. 1966, 44, 125–136. [Google Scholar] [CrossRef]
- Janczak, J. Comment on polymorphic forms of metal-free phthalocyanine. The refinement of the crystal structure of α-H2Pc at 160 K. Pol. J. Chem. 2000, 74, 157–162. [Google Scholar]
- Cordero, B.; Gómez, V.; Platero-Prats, A.E.; Revés, M.; Echeverría, J.; Cremades, E.; Barragán, F.; Alvarez, S. Covalent radii revisited. Dalton Trans. 2008, 21, 2832–2838. [Google Scholar] [CrossRef]
- Batsanov, S.S. Van der Waals Radii of Elements. Inorg. Mater. 2001, 37, 871–885, Translated from Neorganicheskie Materialy 2001, 37, 1031–1046. [Google Scholar] [CrossRef]
- Spackman, M.A.; Jayatilaka, D. Hirshfeld surface analysis. CrystEngComm 2009, 11, 19–32. [Google Scholar] [CrossRef]
- McKinnon, J.J.; Spackman, M.A.; Mitchell, A.S. Novel tools for visualizing and exploring intermolecular interactions in molecular crystals. Acta Cryst. B 2004, 60, 627–668. [Google Scholar] [CrossRef] [PubMed]
- McKinnon, J.J.; Mitchell, A.S.; Spackman, M.A. Hirshfeld surfaces: A new tool for visualising and exploring molecular crystals. Chem. Eur. J. 1998, 4, 2136–2141. [Google Scholar] [CrossRef]
- McKinnon, J.J.; Jayatilaka, D.; Spackman, M.A. Towards quantitative analysis of intermolecular interactions with Hirshfeld surfaces. Chem. Commun. 2007, 37, 3814–3816. [Google Scholar] [CrossRef] [PubMed]
- Janczak, J. Coordination properties of diethylenetriamine in relation to zinc phthalocyanine. Polyhedron 2020, 178, 114313. [Google Scholar] [CrossRef]
- Isago, H. Spectral properties of a novel antimony(III)-phthalocyanine complex that behaves like J-aggregates in non-aqueous media. Chem. Commun. 2003, 15, 1864–1865. [Google Scholar] [CrossRef]
- Zahir, M.H.; Kagaya, Y.; Isago, H.; Furubayashi, T. Preparation of phthalocyanine(3-) species as bulk material by reduction of the dichloro(phthalocyaninato)antimony(V) complex cation. Inorg. Chim. Acta 2004, 357, 2755–2758. [Google Scholar] [CrossRef]
- Bayo, K.; Mossoyan, J.C.; Ouedrago, G.V. Preparation and analysis by UV-Vis of zinc phthalocyanine complexes. Spectrochim. Acta A 2004, 60, 653–657. [Google Scholar] [CrossRef]
- Staicu, A.; Pascu, A.; Nuta, A.; Sorescu, A.; Raditoiu, V.; Pascu, M.L. Studies about phthalocyanine photosensitizeres to be used in photodynamic therapy. Rom. Rep. Phys. 2013, 65, 1032–1051. [Google Scholar]
- Zhang, X.-F.; Guo, W. Imidazole functionalized magnesium phthalocyanine photosensitizer: Modified photophysics, singlet oxygen generation and photooxidation mechanism. J. Phys. Chem. A 2012, 116, 7651–7657. [Google Scholar] [CrossRef]
- Tackley, D.R.; Dent, G.; Smith, W.E. Phthalocyanines: Structure and vibrations. Phys. Chem. Chem. Phys. 2001, 3, 1419–1426. [Google Scholar] [CrossRef]
- Stillman, M.J.; Nyokong, T. Nonlinear Optical Properties of Phthalocyanines. In Phthalocyanines, Properties and Applications; Lever, A.B.P., Leznoff, C.C., Eds.; VCH: New York, NY, USA, 1989; Volume 1, Chapter 3. [Google Scholar]
- Li, C.-P.; Du, M. Role of solvents in coordination supramolecular systems. Chem. Commun. 2011, 47, 5958–5972. [Google Scholar] [CrossRef] [PubMed]
- Shelnutt, J.A. Correlation between metal stability, charge transfer, and Raman frequencies in metalloporphyrins and their π–π complexes. J. Am. Chem. Soc. 1983, 105, 774–778. [Google Scholar] [CrossRef]










| [(SbIIIPc)2(Sb2I8)(SbBr3)]—(1) | |
|---|---|
| Formula | C64H32Br3I8N16Sb5 |
| Molecular weight | 2888.73 |
| Temperature (K) | 295(2) |
| Crystal system | Triclinic |
| Space group | |
| a, b, c (Å) | 14.399(2), 15.344(3), 18.992(3) |
| α, β, γ (°) | 95.64(2), 96.89(2), 113.93(2) |
| V (Å3) | 3757.4(12) |
| Z | 2 |
| Dcal (gcm−3) | 2.553 |
| Crystal size (mm3) | 0.250 × 0.125 × 0.080 |
| μ (mm−1) | 6.707 |
| Absorption correction | multi-scan |
| Tmax./Tmin. | 0.4754/1.000 |
| Total/Unique/Obs refls | 23,647/14,738/7721 |
| Rint | 0.0271 |
| R, [F2 > 2σ(F2)] 1 | 0.0476 |
| wR [F2 all refls] 2 | 0.0957 |
| S | 1.001 |
| Δρmax, Δρmin (e·Å−3) | 2.072, −1.286 |
| (SbPc)+ Cations | |||||
|---|---|---|---|---|---|
| Sb4–N2 | 2.191(8) | Sb4B–N2B | 2.202(7) | ||
| Sb4–N4 | 2.178(8) | Sb4B–N4B | 2.181(7) | ||
| Sb4–N6 | 2.190(9) | Sb4B–N6B | 2.206(8) | ||
| Sb4–N8 | 2.206(8) | Sb4B–N8B | 2.146(8) | ||
| N2–Sb4–N4 | 78.0(3) | N2B–Sb4B–N4B | 77.6(3) | ||
| N4–Sb4–N6 | 79.4(3) | N4B–Sb4B–N6B | 77.7(3) | ||
| N6–Sb4–N8 | 77.5(3) | N6B–Sb4B–N8B | 78.5(3) | ||
| N2–Sb4–N8 | 77.7(3) | N2B–Sb4B–N8B | 78.4(3) | ||
| Deviation of Sb from N4-plane | 0.992(1) | Deviation of Sb from N4-plane | 0.994(1) | ||
| (Sb6I16Br6)4− Anion | |||||
| Terminal Sb–I | |||||
| Sb1–I7 | 2.7668(13) | Sb1–I8 | 2.6965(12) | Sb2–I6 | 2.7374(11) |
| Bridged μ2 | |||||
| Sb1–I3 | 3.2329(11) | Sb2–I3 | 2.9833(13) | Sb1–I5 | 2.8744(11) |
| Sb3–I5 i | 3.4481(10) | Sb2–I4 | 2.8302(10) | Sb3–I4 | 3.31901(10) |
| Bridged μ3 | |||||
| Sb1–I1 | 3.1765(14) | Sb2–I1 | 3.1856(11) | Sb2i–I1 | 3.3610(10) |
| Sb2–I2 | 3.0061(13) | Sb1i–I2 | 3.4281(12) | Sb3–I2 | 3.3531(11) |
| Terminal Sb–Br | |||||
| Sb3–Br1 | 2.6888(12) | Sb3–Br2 | 2.5659(14) | Sb3–Br3 | 2.6321(15) |
| I8–Sb1–I7 | 96.78(4) | I8–Sb1–I5 | 90.48(4) | I7–Sb1–I5 | 98.42(4) |
| I8–Sb1–I1 | 93.88(4) | I7–Sb1–I1 | 166.21(3) | I5–Sb1–I1 | 90.20(3) |
| I8–Sb1–I3 | 91.83(3) | I7–Sb1–I3 | 85.88(4) | I5–Sb1–I3 | 174.85(3) |
| I1–Sb1–I3 | 85.05(3) | I6–Sb2–I4 | 97.06(3) | I6–Sb2–I3 | 90.66(4) |
| I4–Sb2–I3 | 88.68(3) | I6–Sb2–I2 | 91.49(3) | I4–Sb2–I2 | 93.83(4) |
| I3–Sb2–I2 | 176.48(3) | I6–Sb2–I1 | 90.88(3) | I4–Sb2–I1 | 171.80(3) |
| I3–Sb2–I1 | 89.16(3) | I2–Sb2–I1 | 88.02(3) | Br2–Sb3–Br3 | 92.11(5) |
| Br2–Sb3–Br1 | 97.60(5) | Br3–Sb3–Br1 | 96.8 (5) | Sb1–I1–Sb2 | 91.46(3) |
| Sb2–I3–Sb1 | 94.17(3) | ||||
| Short Contacts between (SbPc)+ Units and (Sb6I16Br6)4− Anion | |||||
| Sb4–I1 i | 3.5083(13) | Sb4–I2i | 3.6471(13) | Sb4–I3 | 3.6115(14) |
| Sb4B–I5 | 3.6283(14) | Sb4B–I7 | 3.4969(11) | Sb4B–Br3i | 3.5941(18) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubiak, R.; Janczak, J. Synthesis, Structure, and UV–Vis Characterization of Antimony(III) Phthalocyanine: [(SbPc)2(Sb2I8)(SbBr3)]2. Molecules 2022, 27, 1839. https://doi.org/10.3390/molecules27061839
Kubiak R, Janczak J. Synthesis, Structure, and UV–Vis Characterization of Antimony(III) Phthalocyanine: [(SbPc)2(Sb2I8)(SbBr3)]2. Molecules. 2022; 27(6):1839. https://doi.org/10.3390/molecules27061839
Chicago/Turabian StyleKubiak, Ryszard, and Jan Janczak. 2022. "Synthesis, Structure, and UV–Vis Characterization of Antimony(III) Phthalocyanine: [(SbPc)2(Sb2I8)(SbBr3)]2" Molecules 27, no. 6: 1839. https://doi.org/10.3390/molecules27061839







