Relationship between Immune Cells, Depression, Stress, and Psoriasis: Could the Use of Natural Products Be Helpful?
Abstract
1. Introduction
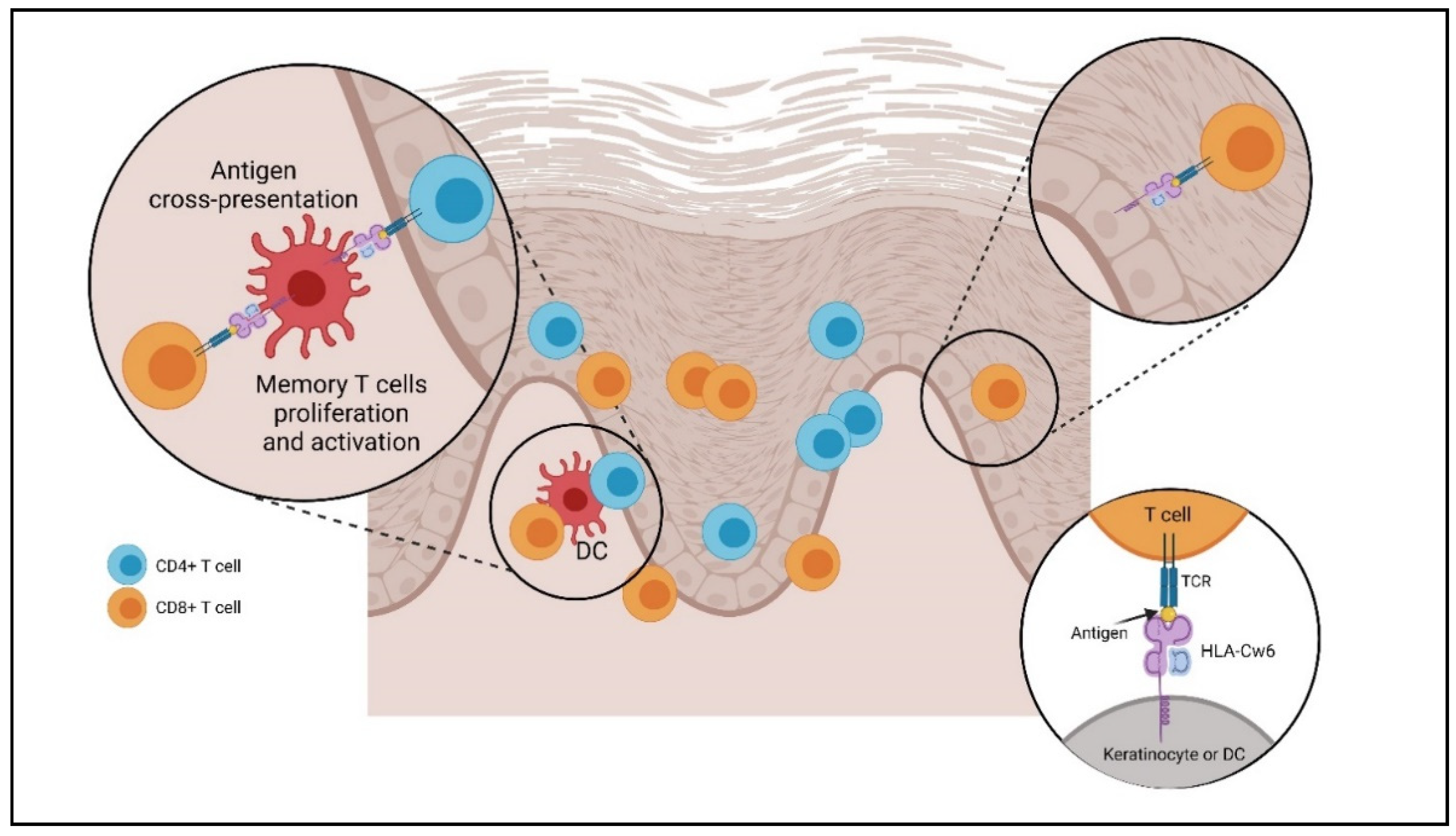
2. Cell Types Involved in the Pathogenesis of Psoriasis
2.1. Keratinocytes
2.2. Dendritic Cells
2.2.1. Plasmacytoid Dendritic Cells (pDCs)
2.2.2. Myeloid Dendritic Cells (mDCs)
2.2.3. Langerhans Cells (LCs)
2.3. Innate Lymphoid Cells (ILCs)
Natural Killer Cells (NKs)
2.4. T Cells (Lymphocyte T Helper, T Cytotoxin γδ T, Natural Killer T Cells)
2.5. Neutrophils
2.6. Mast Cells
3. Psoriasis-Related Psychological Alterations
3.1. Psoriasis and Depression
3.2. Psoriasis and Stress
4. Natural Compounds as an Alternative Treatment for Psoriasis
4.1. Aloe vera L.
4.2. Bergamot Essential Oil
4.3. Quercetin
4.4. Baicalein
4.5. Curcumin
4.6. Resveratrol
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Pergolizzi, S.; Vaccaro, M.; Magaudda, L.; Mondello, M.R.; Arco, A.; Bramanti, P.; Cannavo, S.P.; Guarneri, B. Immunohistochemical study of epidermal nerve fibres in involved and uninvolved psoriatic skin using confocal laser scanning microscopy. Arch. Dermatol. Res. 1998, 290, 483–489. [Google Scholar] [CrossRef]
- Vaccaro, M.; Pergolizzi, S.; Mondello, M.R.; Santoro, G.; Cannavo, S.P.; Guarneri, B.; Magaudda, L. The dermoepidermal junction in psoriatic skin as revealed by scanning electron microscopy. Arch. Dermatol. Res. 1999, 291, 396–399. [Google Scholar] [CrossRef] [PubMed]
- Iskandar, I.Y.K.; Parisi, R.; Griffiths, C.E.M.; Ashcroft, D.M. Systematic review examining changes over time and variation in the incidence and prevalence of psoriasis by age and gender. Br. J. Dermatol. 2020, 184, 243–258. [Google Scholar] [CrossRef]
- Armstrong, A.W. Psoriasis. JAMA Dermatol. 2017, 153, 956. [Google Scholar] [CrossRef] [PubMed]
- Raychaudhuri, S.K.; Maverakis, E.; Raychaudhuri, S.P. Diagnosis and classification of psoriasis. Autoimmun. Rev. 2014, 13, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Dika, E.; Bardazzi, F.; Balestri, R.; Maibach, H.I. Environmental Factors and Psoriasis1. In Environmental Factors in Skin Diseases. Curr. Probl. Dermatol. 2007, 35, 118–135. [Google Scholar] [PubMed]
- Chandra, A.; Ray, A.; Senapati, S.; Chatterjee, R. Genetic and epigenetic basis of psoriasis pathogenesis. Mol. Immunol. 2015, 64, 313–323. [Google Scholar] [CrossRef]
- Fernandes, A.R.; Martins-Gomes, C.; Santini, A.; Silva, A.M.; Souto, E.B. Psoriasis vulgaris—Pathophysiology of the disease and its classical treatment versus new drug delivery systems. In Design of Nanostructures for Versatile Therapeutic Applications; Elsevier: Amsterdam, The Netherlands, 2018; pp. 379–406. [Google Scholar]
- Nestle, F.O.; Kaplan, D.H.; Barker, J. Psoriasis. N. Engl. J. Med. 2009, 361, 496–509. [Google Scholar] [CrossRef] [PubMed]
- Bhosle, M.J.; Kulkarni, A.; Feldman, S.R.; Balkrishnan, R. Quality of life in patients with psoriasis. Health Qual. Life Outcomes 2006, 4, 35. [Google Scholar] [CrossRef] [PubMed]
- Heng, M.C.; Kloss, S.G.; Kuehn, C.S.; Chase, D.G. The sequence of events in psoriatic plaque formation after tape-stripping. Br. J. Dermatol. 1985, 112, 517–532. [Google Scholar] [CrossRef] [PubMed]
- Benhadou, F.; Mintoff, D.; Del Marmol, V. Psoriasis: Keratinocytes or Immune Cells—Which Is the Trigger? Dermatology 2019, 235, 91–100. [Google Scholar] [CrossRef]
- Nestle, F.O.; Turka, L.A.; Nickoloff, B.J. Characterization of dermal dendritic cells in psoriasis. Autostimulation of T lymphocytes and induction of Th1 type cytokines. J. Clin. Investig. 1994, 94, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Iwakura, Y.; Ishigame, H.; Saijo, S.; Nakae, S. Functional specialization of interleukin-17 family members. Immunity 2011, 34, 149–162. [Google Scholar] [CrossRef]
- Di Cesare, A.; Di Meglio, P.; Nestle, F.O. The IL-23/Th17 axis in the immunopathogenesis of psoriasis. J. Investig. Dermatol. 2009, 129, 1339–1350. [Google Scholar] [CrossRef] [PubMed]
- Kastelein, R.A.; Hunter, C.A.; Cua, D.J. Discovery and biology of IL-23 and IL-27: Related but functionally distinct regulators of inflammation. Annu. Rev. Immunol. 2007, 25, 221–242. [Google Scholar] [CrossRef]
- Lowes, M.A.; Kikuchi, T.; Fuentes-Duculan, J.; Cardinale, I.; Zaba, L.C.; Haider, A.S.; Bowman, E.P.; Krueger, J.G. Psoriasis vulgaris lesions contain discrete populations of Th1 and Th17 T cells. J. Investig. Dermatol. 2008, 128, 1207–1211. [Google Scholar] [CrossRef]
- Amin, M.; Darji, K.; No, D.J.; Wu, J.J. Review of phase III trial data on IL-23 inhibitors tildrakizumab and guselkumab for psoriasis. J. Eur. Acad. Dermatol. Venereol. 2017, 31, 1627–1632. [Google Scholar] [CrossRef]
- Sato, Y.; Ogawa, E.; Okuyama, R. Role of Innate Immune Cells in Psoriasis. Int. J. Mol. Sci. 2020, 21, 6604. [Google Scholar] [CrossRef] [PubMed]
- Vanderpuye-Orgle, J.; Zhao, Y.; Lu, J.; Shrestha, A.; Sexton, A.; Seabury, S.; Lebwohl, M. Evaluating the economic burden of psoriasis in the United States. J. Am. Acad. Dermatol. 2015, 72, 961–967.e965. [Google Scholar] [CrossRef]
- Brezinski, E.A.; Dhillon, J.S.; Armstrong, A.W. Economic Burden of Psoriasis in the United States: A Systematic Review. JAMA Dermatol. 2015, 151, 651–658. [Google Scholar] [CrossRef]
- Hawro, T.; Zalewska, A.; Hawro, M.; Kaszuba, A.; Krolikowska, M.; Maurer, M. Impact of psoriasis severity on family income and quality of life. J. Eur. Acad. Dermatol. Venereol. 2015, 29, 438–443. [Google Scholar] [CrossRef]
- Singh, S.; Taylor, C.; Kornmehl, H.; Armstrong, A.W. Psoriasis and suicidality: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2017, 77, 425–440.e422. [Google Scholar] [CrossRef]
- Ferreira, B.I.; Abreu, J.L.; Reis, J.P.; Figueiredo, A.M. Psoriasis and Associated Psychiatric Disorders: A Systematic Review on Etiopathogenesis and Clinical Correlation. J. Clin. Aesthet. Dermatol. 2016, 9, 36–43. [Google Scholar]
- Hall, J.M.; Cruser, D.; Podawiltz, A.; Mummert, D.I.; Jones, H.; Mummert, M.E. Psychological Stress and the Cutaneous Immune Response: Roles of the HPA Axis and the Sympathetic Nervous System in Atopic Dermatitis and Psoriasis. Dermatol. Res. Pract. 2012, 2012, 403908. [Google Scholar] [CrossRef]
- Connor, C.J.; Liu, V.; Fiedorowicz, J.G. Exploring the Physiological Link between Psoriasis and Mood Disorders. Dermatol. Res. Pract. 2015, 2015, 409637. [Google Scholar] [CrossRef]
- Snast, I.; Reiter, O.; Atzmony, L.; Leshem, Y.A.; Hodak, E.; Mimouni, D.; Pavlovsky, L. Psychological stress and psoriasis: A systematic review and meta-analysis. Br. J. Dermatol. 2018, 178, 1044–1055. [Google Scholar] [CrossRef]
- Takeshita, J.; Grewal, S.; Langan, S.M.; Mehta, N.N.; Ogdie, A.; Van Voorhees, A.S.; Gelfand, J.M. Psoriasis and comorbid diseases: Epidemiology. J. Am. Acad. Dermatol. 2017, 76, 377–390. [Google Scholar] [CrossRef]
- Conrad, C.; Gilliet, M. Psoriasis: From Pathogenesis to Targeted Therapies. Clin. Rev. Allergy Immunol. 2018, 54, 102–113. [Google Scholar] [CrossRef]
- Cho, J.S.; Pietras, E.M.; Garcia, N.C.; Ramos, R.I.; Farzam, D.M.; Monroe, H.R.; Magorien, J.E.; Blauvelt, A.; Kolls, J.K.; Cheung, A.L.; et al. IL-17 is essential for host defense against cutaneous Staphylococcus aureus infection in mice. J. Clin. Investig. 2010, 120, 1762–1773. [Google Scholar] [CrossRef]
- Alesci, A.; Cicero, N.; Salvo, A.; Palombieri, D.; Zaccone, D.; Dugo, G.; Bruno, M.; Vadala, R.; Lauriano, E.R.; Pergolizzi, S. Extracts deriving from olive mill waste water and their effects on the liver of the goldfish Carassius auratus fed with hypercholesterolemic diet. Nat. Prod. Res. 2014, 28, 1343–1349. [Google Scholar] [CrossRef]
- Pergolizzi, S.; D’Angelo, V.; Aragona, M.; Dugo, P.; Cacciola, F.; Capillo, G.; Dugo, G.; Lauriano, E.R. Evaluation of antioxidant and anti-inflammatory activity of green coffee beans methanolic extract in rat skin. Nat. Prod. Res. 2018, 34, 1535–1541. [Google Scholar] [CrossRef]
- Alesci, A.; Miller, A.; Tardugno, R.; Pergolizzi, S. Chemical analysis, biological and therapeutic activities of Olea europaea L. Nat. Prod. Res. 2021. [Google Scholar] [CrossRef]
- Gervasi, T.; Pellizzeri, V.; Benameur, Q.; Gervasi, C.; Santini, A.; Cicero, N.; Dugo, G. Valorization of raw materials from agricultural industry for astaxanthin and beta-carotene production by Xanthophyllomyces dendrorhous. Nat. Prod. Res. 2018, 32, 1554–1561. [Google Scholar] [CrossRef]
- Naviglio, D.; Pizzolongo, F.; Ferrara, L.; Aragòn, A.; Santini, A. Extraction of pure lycopene from industrial tomato by-products in water using a new high-pressure process. J. Sci. Food Agric. 2008, 88, 2414–2420. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Camilli, E.; Marconi, S.; Gabrielli, P.; Lisciani, S.; Gambelli, L.; Aguzzi, A.; Novellino, E.; Santini, A.; et al. Dietary Lignans: Definition, Description and Research Trends in Databases Development. Molecules 2018, 23, 3251. [Google Scholar] [CrossRef]
- Li, J.; Li, X.; Hou, R.; Liu, R.; Zhao, X.; Dong, F.; Wang, C.; Yin, G.; Zhang, K. Psoriatic T cells reduce epidermal turnover time and affect cell proliferation contributed from differential gene expression. J. Dermatol. 2015, 42, 874–880. [Google Scholar] [CrossRef]
- Mondello, M.R.; Magaudda, L.; Pergolizzi, S.; Santoro, A.; Vaccaro, M.; Califano, L.; Cannavo, S.P.; Guarneri, B. Behaviour of laminin 1 and type IV collagen in uninvolved psoriatic skin. Immunohistochemical study using confocal laser scanning microscopy. Arch. Dermatol. Res. 1996, 288, 527–531. [Google Scholar] [CrossRef]
- Magaudda, L.; Mondello, M.R.; Vaccaro, M.; Pergolizzi, S.; Cannavo, S.P.; Guarneri, B.; Santoro, A. Changes in the distribution of actin-associated proteins in psoriatic keratinocytes. Immunohistochemical study using confocal laser scanning microscopy. Arch. Dermatol. Res. 1997, 289, 378–383. [Google Scholar] [CrossRef]
- Salmon, J.K.; Armstrong, C.A.; Ansel, J.C. The skin as an immune organ. West. J. Med. 1994, 160, 146–152. [Google Scholar]
- Albanesi, C.; De Pita, O.; Girolomoni, G. Resident skin cells in psoriasis: A special look at the pathogenetic functions of keratinocytes. Clin. Dermatol. 2007, 25, 581–588. [Google Scholar] [CrossRef]
- Lowes, M.A.; Suarez-Farinas, M.; Krueger, J.G. Immunology of psoriasis. Annu. Rev. Immunol. 2014, 32, 227–255. [Google Scholar] [CrossRef]
- Lande, R.; Botti, E.; Jandus, C.; Dojcinovic, D.; Fanelli, G.; Conrad, C.; Chamilos, G.; Feldmeyer, L.; Marinari, B.; Chon, S.; et al. The antimicrobial peptide LL37 is a T-cell autoantigen in psoriasis. Nat. Commun. 2014, 5, 5621. [Google Scholar] [CrossRef]
- Nickoloff, B.J.; Turka, L.A. Immunological functions of non-professional antigen-presenting cells: New insights from studies of T-cell interactions with keratinocytes. Immunol. Today 1994, 15, 464–469. [Google Scholar] [CrossRef]
- Gillitzer, R.; Wolff, K.; Tong, D.; Muller, C.; Yoshimura, T.; Hartmann, A.A.; Stingl, G.; Berger, R. MCP-1 mRNA expression in basal keratinocytes of psoriatic lesions. J. Investig. Dermatol. 1993, 101, 127–131. [Google Scholar] [CrossRef]
- Homey, B.; Wang, W.; Soto, H.; Buchanan, M.E.; Wiesenborn, A.; Catron, D.; Muller, A.; McClanahan, T.K.; Dieu-Nosjean, M.C.; Orozco, R.; et al. Cutting edge: The orphan chemokine receptor G protein-coupled receptor-2 (GPR-2, CCR10) binds the skin-associated chemokine CCL27 (CTACK/ALP/ILC). J. Immunol. 2000, 164, 3465–3470. [Google Scholar] [CrossRef]
- Gangemi, S.; Merendino, R.A.; Guarneri, F.; Minciullo, P.L.; DiLorenzo, G.; Pacor, M.; Cannavo, S.P. Serum levels of interleukin-18 and s-ICAM-1 in patients affected by psoriasis: Preliminary considerations. J. Eur. Acad. Dermatol. Venereol. 2003, 17, 42–46. [Google Scholar] [CrossRef]
- Sugita, K.; Kabashima, K.; Atarashi, K.; Shimauchi, T.; Kobayashi, M.; Tokura, Y. Innate immunity mediated by epidermal keratinocytes promotes acquired immunity involving Langerhans cells and T cells in the skin. Clin. Exp. Immunol. 2007, 147, 176–183. [Google Scholar] [CrossRef]
- Nair, R.P.; Stuart, P.E.; Nistor, I.; Hiremagalore, R.; Chia, N.V.C.; Jenisch, S.; Weichenthal, M.; Abecasis, G.R.; Lim, H.W.; Christophers, E.; et al. Sequence and haplotype analysis supports HLA-C as the psoriasis susceptibility 1 gene. Am. J. Hum. Genet. 2006, 78, 827–851. [Google Scholar] [CrossRef]
- Prinz, J.C. Autoimmune aspects of psoriasis: Heritability and autoantigens. Autoimmun. Rev. 2017, 16, 970–979. [Google Scholar] [CrossRef]
- Lauriano, E.R.; Pergolizzi, S.; Capillo, G.; Kuciel, M.; Alesci, A.; Faggio, C. Immunohistochemical characterization of Toll-like receptor 2 in gut epithelial cells and macrophages of goldfish Carassius auratus fed with a high-cholesterol diet. Fish. Shellfish Immunol. 2016, 59, 250–255. [Google Scholar] [CrossRef]
- Lauriano, E.R.; Silvestri, G.; Kuciel, M.; Zuwala, K.; Zaccone, D.; Palombieri, D.; Alesci, A.; Pergolizzi, S. Immunohistochemical localization of Toll-like receptor 2 in skin Langerhans’ cells of striped dolphin (Stenella coeruleoalba). Tissue Cell 2014, 46, 113–121. [Google Scholar] [CrossRef]
- Lauriano, E.R.; Aragona, M.; Alesci, A.; Lo Cascio, P.; Pergolizzi, S. Toll-like receptor 2 and alpha-Smooth Muscle Actin expressed in the tunica of a urochordate, Styela plicata. Tissue Cell 2021, 71, 101584. [Google Scholar] [CrossRef]
- Alesci, A.; Pergolizzi, S.; Lo Cascio, P.; Fumia, A.; Lauriano, E.R. Neuronal Regeneration: Vertebrates comparative overview and new perspectives for Neurodegenerative diseases. Acta Zool. 2021. [Google Scholar] [CrossRef]
- McInturff, J.E.; Modlin, R.L.; Kim, J. The role of toll-like receptors in the pathogenesis and treatment of dermatological disease. J. Investig. Dermatol. 2005, 125, 1–8. [Google Scholar] [CrossRef]
- Miller, L.S.; Sorensen, O.E.; Liu, P.T.; Jalian, H.R.; Eshtiaghpour, D.; Behmanesh, B.E.; Chung, W.; Starner, T.D.; Kim, J.; Sieling, P.A.; et al. TGF-alpha regulates TLR expression and function on epidermal keratinocytes. J. Immunol. 2005, 174, 6137–6143. [Google Scholar] [CrossRef]
- Curry, J.L.; Qin, J.Z.; Bonish, B.; Carrick, R.; Bacon, P.; Panella, J.; Robinson, J.; Nickoloff, B.J. Innate immune-related receptors in normal and psoriatic skin. Arch. Pathol Lab. Med. 2003, 127, 178–186. [Google Scholar] [CrossRef]
- Xiao, C.Y.; Zhu, Z.L.; Zhang, C.; Fu, M.; Qiao, H.J.; Wang, G.; Dang, E.L. Small interfering RNA targeting of keratin 17 reduces inflammation in imiquimod-induced psoriasis-like dermatitis. Chin. Med. J. 2020, 133, 2910–2918. [Google Scholar] [CrossRef]
- Zhang, X.; Yin, M.; Zhang, L.J. Keratin 6, 16 and 17-Critical Barrier Alarmin Molecules in Skin Wounds and Psoriasis. Cells 2019, 8, 807. [Google Scholar] [CrossRef]
- Zaba, L.C.; Krueger, J.G.; Lowes, M.A. Resident and “inflammatory” dendritic cells in human skin. J. Investig. Dermatol. 2009, 129, 302–308. [Google Scholar] [CrossRef]
- Pergolizzi, S.; Rizzo, G.; Favaloro, A.; Alesci, A.; Pallio, S.; Melita, G.; Cutroneo, G.; Lauriano, E.R. Expression of VAChT and 5-HT in Ulcerative Colitis dendritic cells. Acta Histochem. 2021, 123, 151715. [Google Scholar] [CrossRef]
- Chiricozzi, A.; Romanelli, P.; Volpe, E.; Borsellino, G.; Romanelli, M. Scanning the Immunopathogenesis of Psoriasis. Int. J. Mol. Sci. 2018, 19, 179. [Google Scholar] [CrossRef]
- Liu, Y.J. IPC: Professional type 1 interferon-producing cells and plasmacytoid dendritic cell precursors. Annu. Rev. Immunol. 2005, 23, 275–306. [Google Scholar] [CrossRef]
- Cella, M.; Jarrossay, D.; Facchetti, F.; Alebardi, O.; Nakajima, H.; Lanzavecchia, A.; Colonna, M. Plasmacytoid monocytes migrate to inflamed lymph nodes and produce large amounts of type I interferon. Nat. Med. 1999, 5, 919–923. [Google Scholar] [CrossRef]
- Fonteneau, J.F.; Gilliet, M.; Larsson, M.; Dasilva, I.; Munz, C.; Liu, Y.J.; Bhardwaj, N. Activation of influenza virus-specific CD4+ and CD8+ T cells: A new role for plasmacytoid dendritic cells in adaptive immunity. Blood 2003, 101, 3520–3526. [Google Scholar] [CrossRef] [PubMed]
- Blanco, P.; Palucka, A.K.; Gill, M.; Pascual, V.; Banchereau, J. Induction of dendritic cell differentiation by IFN-alpha in systemic lupus erythematosus. Science 2001, 294, 1540–1543. [Google Scholar] [CrossRef]
- Gilliet, M.; Conrad, C.; Geiges, M.; Cozzio, A.; Thurlimann, W.; Burg, G.; Nestle, F.O.; Dummer, R. Psoriasis triggered by toll-like receptor 7 agonist imiquimod in the presence of dermal plasmacytoid dendritic cell precursors. Arch. Dermatol. 2004, 140, 1490–1495. [Google Scholar] [CrossRef]
- Farkas, L.; Beiske, K.; Lund-Johansen, F.; Brandtzaeg, P.; Jahnsen, F.L. Plasmacytoid dendritic cells (natural interferon- alpha/beta-producing cells) accumulate in cutaneous lupus erythematosus lesions. Am. J. Pathol. 2001, 159, 237–243. [Google Scholar] [CrossRef]
- Van Krinks, C.H.; Matyszak, M.K.; Gaston, J.S. Characterization of plasmacytoid dendritic cells in inflammatory arthritis synovial fluid. Rheumatology 2004, 43, 453–460. [Google Scholar] [CrossRef][Green Version]
- Kadowaki, N.; Ho, S.; Antonenko, S.; Malefyt, R.W.; Kastelein, R.A.; Bazan, F.; Liu, Y.J. Subsets of human dendritic cell precursors express different toll-like receptors and respond to different microbial antigens. J. Exp. Med. 2001, 194, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Amakawa, R.; Inaba, M.; Hori, T.; Ota, M.; Nakamura, K.; Takebayashi, M.; Miyaji, M.; Yoshimura, T.; Inaba, K.; et al. Plasmacytoid dendritic cells regulate Th cell responses through OX40 ligand and type I IFNs. J. Immunol. 2004, 172, 4253–4259. [Google Scholar] [CrossRef] [PubMed]
- Nestle, F.O.; Conrad, C.; Tun-Kyi, A.; Homey, B.; Gombert, M.; Boyman, O.; Burg, G.; Liu, Y.J.; Gilliet, M. Plasmacytoid predendritic cells initiate psoriasis through interferon-alpha production. J. Exp. Med. 2005, 202, 135–143. [Google Scholar] [CrossRef]
- Randolph, G.J.; Ochando, J.; Partida-Sanchez, S. Migration of dendritic cell subsets and their precursors. Annu. Rev. Immunol 2008, 26, 293–316. [Google Scholar] [CrossRef] [PubMed]
- Sozzani, S. Dendritic cell trafficking: More than just chemokines. Cytokine Growth Factor Rev. 2005, 16, 581–592. [Google Scholar] [CrossRef] [PubMed]
- Vinter, H.; Iversen, L.; Steiniche, T.; Kragballe, K.; Johansen, C. Aldara(R)-induced skin inflammation: Studies of patients with psoriasis. Br. J. Dermatol. 2015, 172, 345–353. [Google Scholar] [CrossRef]
- Takeda, K.; Kaisho, T.; Akira, S. Toll-like receptors. Annu. Rev. Immunol. 2003, 21, 335–376. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Ohba, Y.; Yanai, H.; Negishi, H.; Mizutani, T.; Takaoka, A.; Taya, C.; Taniguchi, T. Spatiotemporal regulation of MyD88-IRF-7 signalling for robust type-I interferon induction. Nature 2005, 434, 1035–1040. [Google Scholar] [CrossRef] [PubMed]
- Lowes, M.A.; Turton, J.A.; Krueger, J.G.; Barnetson, R.S. Psoriasis vulgaris flare during efalizumab therapy does not preclude future use: A case series. BMC Dermatol. 2005, 5, 9. [Google Scholar] [CrossRef] [PubMed]
- Zaba, L.C.; Fuentes-Duculan, J.; Eungdamrong, N.J.; Abello, M.V.; Novitskaya, I.; Pierson, K.C.; Gonzalez, J.; Krueger, J.G.; Lowes, M.A. Psoriasis is characterized by accumulation of immunostimulatory and Th1/Th17 cell-polarizing myeloid dendritic cells. J. Investig. Dermatol. 2009, 129, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Johnson-Huang, L.M.; McNutt, N.S.; Krueger, J.G.; Lowes, M.A. Cytokine-producing dendritic cells in the pathogenesis of inflammatory skin diseases. J. Clin. Immunol. 2009, 29, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.C.; Di Meglio, P.; Nestle, F.O. Harnessing dendritic cells in inflammatory skin diseases. Semin. Immunol. 2011, 23, 28–41. [Google Scholar] [CrossRef]
- Lowes, M.A.; Chamian, F.; Abello, M.V.; Fuentes-Duculan, J.; Lin, S.L.; Nussbaum, R.; Novitskaya, I.; Carbonaro, H.; Cardinale, I.; Kikuchi, T.; et al. Increase in TNF-alpha and inducible nitric oxide synthase-expressing dendritic cells in psoriasis and reduction with efalizumab (anti-CD11a). Proc. Natl. Acad. Sci. USA 2005, 102, 19057–19062. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.G.; Jee, H.; Fuentes-Duculan, J.; Wu, W.H.; Byamba, D.; Kim, D.S.; Kim, D.Y.; Lew, D.H.; Yang, W.I.; Krueger, J.G.; et al. Dermal clusters of mature dendritic cells and T cells are associated with the CCL20/CCR6 chemokine system in chronic psoriasis. J. Investig. Dermatol. 2014, 134, 1462–1465. [Google Scholar] [CrossRef] [PubMed]
- Alesci, A.; Lauriano, E.R.; Aragona, M.; Capillo, G.; Pergolizzi, S. Marking vertebrates langerhans cells, from fish to mammals. Acta Histochem. 2020, 122, 151622. [Google Scholar] [CrossRef]
- Sundberg, J.P.; Boggess, D.; Sundberg, B.A.; Beamer, W.G.; Shultz, L.D. Epidermal dendritic cell populations in the flaky skin mutant mouse. Immunol. Investig. 1993, 22, 389–401. [Google Scholar] [CrossRef]
- Schon, M.; Behmenburg, C.; Denzer, D.; Schon, M.P. Pathogenic function of IL-1 beta in psoriasiform skin lesions of flaky skin (fsn/fsn) mice. Clin. Exp. Immunol. 2001, 123, 505–510. [Google Scholar] [CrossRef]
- Cumberbatch, M.; Singh, M.; Dearman, R.J.; Young, H.S.; Kimber, I.; Griffiths, C.E. Impaired Langerhans cell migration in psoriasis. J. Exp. Med. 2006, 203, 953–960. [Google Scholar] [CrossRef]
- Fujita, H.; Shemer, A.; Suarez-Farinas, M.; Johnson-Huang, L.M.; Tintle, S.; Cardinale, I.; Fuentes-Duculan, J.; Novitskaya, I.; Carucci, J.A.; Krueger, J.G.; et al. Lesional dendritic cells in patients with chronic atopic dermatitis and psoriasis exhibit parallel ability to activate T-cell subsets. J. Allergy Clin. Immunol. 2011, 128, 574–582.e12. [Google Scholar] [CrossRef]
- Martini, E.; Wiken, M.; Cheuk, S.; Gallais Serezal, I.; Baharom, F.; Stahle, M.; Smed-Sorensen, A.; Eidsmo, L. Dynamic Changes in Resident and Infiltrating Epidermal Dendritic Cells in Active and Resolved Psoriasis. J. Investig. Dermatol. 2017, 137, 865–873. [Google Scholar] [CrossRef] [PubMed]
- Eidsmo, L.; Martini, E. Human Langerhans Cells with Pro-inflammatory Features Relocate within Psoriasis Lesions. Front. Immunol. 2018, 9, 300. [Google Scholar] [CrossRef] [PubMed]
- Bos, J.D.; Hulsebosch, H.J.; Krieg, S.R.; Bakker, P.M.; Cormane, R.H. Immunocompetent cells in psoriasis. In situ immunophenotyping by monoclonal antibodies. Arch. Dermatol. Res. 1983, 275, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Terhorst, D.; Chelbi, R.; Wohn, C.; Malosse, C.; Tamoutounour, S.; Jorquera, A.; Bajenoff, M.; Dalod, M.; Malissen, B.; Henri, S. Dynamics and Transcriptomics of Skin Dendritic Cells and Macrophages in an Imiquimod-Induced, Biphasic Mouse Model of Psoriasis. J. Immunol. 2015, 195, 4953–4961. [Google Scholar] [CrossRef] [PubMed]
- Cupedo, T.; Crellin, N.K.; Papazian, N.; Rombouts, E.J.; Weijer, K.; Grogan, J.L.; Fibbe, W.E.; Cornelissen, J.J.; Spits, H. Human fetal lymphoid tissue-inducer cells are interleukin 17-producing precursors to RORC+ CD127+ natural killer-like cells. Nat. Immunol. 2009, 10, 66–74. [Google Scholar] [CrossRef]
- Lo Presti, E.L.; De Gaetano, A.D.; Pioggia, G.; Gangemi, S. Comprehensive Analysis of the ILCs and Unconventional T Cells in Virus Infection: Profiling and Dynamics Associated with COVID-19 Disease for a Future Monitoring System and Therapeutic Opportunities. Cells 2022, 11, 542. [Google Scholar] [CrossRef]
- Mashiko, S.; Mehta, H.; Bissonnette, R.; Sarfati, M. Increased frequencies of basophils, type 2 innate lymphoid cells and Th2 cells in skin of patients with atopic dermatitis but not psoriasis. J. Dermatol. Sci. 2017, 88, 167–174. [Google Scholar] [CrossRef]
- Mjosberg, J.; Eidsmo, L. Update on innate lymphoid cells in atopic and non-atopic inflammation in the airways and skin. Clin. Exp. Allergy 2014, 44, 1033–1043. [Google Scholar] [CrossRef]
- Parodi, M.; Favoreel, H.; Candiano, G.; Gaggero, S.; Sivori, S.; Mingari, M.C.; Moretta, L.; Vitale, M.; Cantoni, C. NKp44-NKp44 Ligand Interactions in the Regulation of Natural Killer Cells and Other Innate Lymphoid Cells in Humans. Front. Immunol. 2019, 10, 719. [Google Scholar] [CrossRef]
- Teunissen, M.B.M.; Munneke, J.M.; Bernink, J.H.; Spuls, P.I.; Res, P.C.M.; Te Velde, A.; Cheuk, S.; Brouwer, M.W.D.; Menting, S.P.; Eidsmo, L.; et al. Composition of innate lymphoid cell subsets in the human skin: Enrichment of NCR(+) ILC3 in lesional skin and blood of psoriasis patients. J. Investig. Dermatol. 2014, 134, 2351–2360. [Google Scholar] [CrossRef]
- Villanova, F.; Flutter, B.; Tosi, I.; Grys, K.; Sreeneebus, H.; Perera, G.K.; Chapman, A.; Smith, C.H.; Di Meglio, P.; Nestle, F.O. Characterization of innate lymphoid cells in human skin and blood demonstrates increase of NKp44+ ILC3 in psoriasis. J. Investig. Dermatol. 2014, 134, 984–991. [Google Scholar] [CrossRef]
- Moretta, L.; Bottino, C.; Pende, D.; Mingari, M.C.; Biassoni, R.; Moretta, A. Human natural killer cells: Their origin, receptors and function. Eur J. Immunol. 2002, 32, 1205–1211. [Google Scholar] [CrossRef]
- Belizario, J.E.; Neyra, J.M.; Setubal Destro Rodrigues, M.F. When and how NK cell-induced programmed cell death benefits immunological protection against intracellular pathogen infection. Innate Immun. 2018, 24, 452–465. [Google Scholar] [CrossRef]
- Vissers, W.H.; Arndtz, C.H.; Muys, L.; Van Erp, P.E.; de Jong, E.M.; van de Kerkhof, P.C. Memory effector (CD45RO+) and cytotoxic (CD8+) T cells appear early in the margin zone of spreading psoriatic lesions in contrast to cells expressing natural killer receptors, which appear late. Br. J. Dermatol. 2004, 150, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, C.; Nasorri, F.; Bedini, C.; de Pita, O.; Girolomoni, G.; Cavani, A. CD56brightCD16(-) NK cells accumulate in psoriatic skin in response to CXCL10 and CCL5 and exacerbate skin inflammation. Eur. J. Immunol. 2006, 36, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, A.C.; Chaurasia, S.; Mishra, S.K.; Aggarwal, A.; Misra, R. IL-17 and IFN-gamma producing NK and gammadelta-T cells are preferentially expanded in synovial fluid of patients with reactive arthritis and undifferentiated spondyloarthritis. Clin. Immunol. 2017, 183, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Tian, Z. Diversity of tissue-resident NK cells. Semin Immunol. 2017, 31, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Barten, R.; Torkar, M.; Haude, A.; Trowsdale, J.; Wilson, M.J. Divergent and convergent evolution of NK-cell receptors. Trends Immunol. 2001, 22, 52–57. [Google Scholar] [CrossRef]
- Rajagopalan, S.; Long, E.O. KIR2DL4 (CD158d): An activation receptor for HLA-G. Front. Immunol. 2012, 3, 258. [Google Scholar] [CrossRef] [PubMed]
- Girardi, M. Immunosurveillance and immunoregulation by gammadelta T cells. J. Investig. Dermatol. 2006, 126, 25–31. [Google Scholar] [CrossRef]
- Laggner, U.; Di Meglio, P.; Perera, G.K.; Hundhausen, C.; Lacy, K.E.; Ali, N.; Smith, C.H.; Hayday, A.C.; Nickoloff, B.J.; Nestle, F.O. Identification of a novel proinflammatory human skin-homing Vgamma9Vdelta2 T cell subset with a potential role in psoriasis. J. Immunol. 2011, 187, 2783–2793. [Google Scholar] [CrossRef]
- Cai, Y.; Xue, F.; Quan, C.; Qu, M.; Liu, N.; Zhang, Y.; Fleming, C.; Hu, X.; Zhang, H.G.; Weichselbaum, R.; et al. A Critical Role of the IL-1beta-IL-1R Signaling Pathway in Skin Inflammation and Psoriasis Pathogenesis. J. Investig. Dermatol. 2019, 139, 146–156. [Google Scholar] [CrossRef]
- Hijnen, D.; Knol, E.F.; Gent, Y.Y.; Giovannone, B.; Beijn, S.J.; Kupper, T.S.; Bruijnzeel-Koomen, C.A.; Clark, R.A. CD8(+) T cells in the lesional skin of atopic dermatitis and psoriasis patients are an important source of IFN-gamma, IL-13, IL-17, and IL-22. J. Investig. Dermatol. 2013, 133, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Nickoloff, B.J.; Wrone-Smith, T. Injection of pre-psoriatic skin with CD4+ T cells induces psoriasis. Am. J. Pathol. 1999, 155, 145–158. [Google Scholar] [CrossRef]
- Gunderson, A.J.; Mohammed, J.; Horvath, F.J.; Podolsky, M.A.; Anderson, C.R.; Glick, A.B. CD8(+) T cells mediate RAS-induced psoriasis-like skin inflammation through IFN-gamma. J. Investig. Dermatol. 2013, 133, 955–963. [Google Scholar] [CrossRef] [PubMed]
- Conrad, C.; Boyman, O.; Tonel, G.; Tun-Kyi, A.; Laggner, U.; de Fougerolles, A.; Kotelianski, V.; Gardner, H.; Nestle, F.O. Alpha1beta1 integrin is crucial for accumulation of epidermal T cells and the development of psoriasis. Nat. Med. 2007, 13, 836–842. [Google Scholar] [CrossRef]
- Di Meglio, P.; Villanova, F.; Navarini, A.A.; Mylonas, A.; Tosi, I.; Nestle, F.O.; Conrad, C. Targeting CD8(+) T cells prevents psoriasis development. J. Allergy Clin. Immunol. 2016, 138, 274–276.e276. [Google Scholar] [CrossRef]
- Rebora, A. Cyclosporine A in psoriasis. Clin. Dermatol. 1991, 9, 515–522. [Google Scholar] [CrossRef]
- Mondello, M.R.; Califano, L.; Cannavo, S.P.; Di Mauro, D.; Guarneri, B.; Magaudda, L.; Pergolizzi, S.; Santoro, G.; Vaccaro, M. Psoriasis and cyclosporin: Immunohistochemical aspects of the basement membrane. Acta Derm. Venereol. Suppl. 1994, 186, 96–98. [Google Scholar]
- Volpe, E.; Servant, N.; Zollinger, R.; Bogiatzi, S.I.; Hupe, P.; Barillot, E.; Soumelis, V. A critical function for transforming growth factor-beta, interleukin 23 and proinflammatory cytokines in driving and modulating human T(H)-17 responses. Nat. Immunol. 2008, 9, 650–657. [Google Scholar] [CrossRef]
- Duhen, T.; Geiger, R.; Jarrossay, D.; Lanzavecchia, A.; Sallusto, F. Production of interleukin 22 but not interleukin 17 by a subset of human skin-homing memory T cells. Nat. Immunol. 2009, 10, 857–863. [Google Scholar] [CrossRef]
- Sumaria, N.; Roediger, B.; Ng, L.G.; Qin, J.; Pinto, R.; Cavanagh, L.L.; Shklovskaya, E.; Fazekas de St Groth, B.; Triccas, J.A.; Weninger, W. Cutaneous immunosurveillance by self-renewing dermal gammadelta T cells. J. Exp. Med. 2011, 208, 505–518. [Google Scholar] [CrossRef]
- Cosmi, L.; De Palma, R.; Santarlasci, V.; Maggi, L.; Capone, M.; Frosali, F.; Rodolico, G.; Querci, V.; Abbate, G.; Angeli, R.; et al. Human interleukin 17-producing cells originate from a CD161+CD4+ T cell precursor. J. Exp. Med. 2008, 205, 1903–1916. [Google Scholar] [CrossRef]
- Nickoloff, B.J.; Bonish, B.; Huang, B.B.; Porcelli, S.A. Characterization of a T cell line bearing natural killer receptors and capable of creating psoriasis in a SCID mouse model system. J. Dermatol. Sci. 2000, 24, 212–225. [Google Scholar] [CrossRef]
- Nickoloff, B.J.; Wrone-Smith, T.; Bonish, B.; Porcelli, S.A. Response of murine and normal human skin to injection of allogeneic blood-derived psoriatic immunocytes: Detection of T cells expressing receptors typically present on natural killer cells, including CD94, CD158, and CD161. Arch. Dermatol. 1999, 135, 546–552. [Google Scholar] [CrossRef]
- Kriegsmann, K.; Kriegsmann, M.; von Bergwelt-Baildon, M.; Cremer, M.; Witzens-Harig, M. NKT cells—New players in CAR cell immunotherapy? Eur. J. Haematol. 2018, 101, 750–757. [Google Scholar] [CrossRef]
- Tecchio, C.; Cassatella, M.A. Neutrophil-derived chemokines on the road to immunity. Semin. Immunol. 2016, 28, 119–128. [Google Scholar] [CrossRef]
- Leliefeld, P.H.; Koenderman, L.; Pillay, J. How Neutrophils Shape Adaptive Immune Responses. Front. Immunol. 2015, 6, 471. [Google Scholar] [CrossRef]
- Rosales, C.; Lowell, C.A.; Schnoor, M.; Uribe-Querol, E. Neutrophils: Their Role in Innate and Adaptive Immunity 2017. J. Immunol. Res. 2017, 2017, 9748345. [Google Scholar] [CrossRef]
- Chiang, C.C.; Cheng, W.J.; Korinek, M.; Lin, C.Y.; Hwang, T.L. Neutrophils in Psoriasis. Front. Immunol. 2019, 10, 2376. [Google Scholar] [CrossRef]
- Cannavo, S.P.; Riso, G.; Casciaro, M.; Di Salvo, E.; Gangemi, S. Oxidative stress involvement in psoriasis: A systematic review. Free Radic. Res. 2019, 53, 829–840. [Google Scholar] [CrossRef]
- Polat, M.; Bugdayci, G.; Kaya, H.; Oguzman, H. Evaluation of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio in Turkish patients with chronic plaque psoriasis. Acta. Dermatovenerol. Alp. Pannonica Adriat. 2017, 26, 97–100. [Google Scholar] [CrossRef]
- Mrowietz, U. Neutrophils’ sexiness is independent of trendy fashion. Exp. Dermatol. 2017, 26, 312–313. [Google Scholar] [CrossRef]
- Alesci, A.; Pergolizzi, S.; Fumia, A.; Calabrò, C.; Lo Cascio, P.; Lauriano, E.R. Mast cells in goldfish (Carassius auratus) gut: Immunohistochemical characterization. Acta Zool. 2022, 114, 166–171. [Google Scholar] [CrossRef]
- Lauriano, E.R.; Calo, M.; Silvestri, G.; Zaccone, D.; Pergolizzi, S.; Lo Cascio, P. Mast cells in the intestine and gills of the sea bream, Sparus aurata, exposed to a polychlorinated biphenyl, PCB 126. Acta Histochem. 2012, 114, 166–171. [Google Scholar] [CrossRef]
- Mashiko, S.; Bouguermouh, S.; Rubio, M.; Baba, N.; Bissonnette, R.; Sarfati, M. Human mast cells are major IL-22 producers in patients with psoriasis and atopic dermatitis. J. Allergy Clin. Immunol. 2015, 136, 351–359.e351. [Google Scholar] [CrossRef] [PubMed]
- Lim, D.S.; Bewley, A.; Oon, H.H. Psychological Profile of Patients with Psoriasis. Ann. Acad. Med. Singap. 2018, 47, 516–522. [Google Scholar] [PubMed]
- Emons, W.H.; Meijer, R.R.; Denollet, J. Negative affectivity and social inhibition in cardiovascular disease: Evaluating type-D personality and its assessment using item response theory. J. Psychosom. Res. 2007, 63, 27–39. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mizara, A.; Papadopoulos, L.; McBride, S.R. Core beliefs and psychological distress in patients with psoriasis and atopic eczema attending secondary care: The role of schemas in chronic skin disease. Br. J. Dermatol. 2012, 166, 986–993. [Google Scholar] [CrossRef]
- Dehghani, F.; Dehghani, F.; Kafaie, P.; Taghizadeh, M.R. Alexithymia in different dermatologic patients. Asian J. Psychiatr. 2017, 25, 42–45. [Google Scholar] [CrossRef]
- DiBonaventura, M.; Carvalho, A.V.E.d.; Souza, C.d.S.; Squiassi, H.B.; Ferreira, C.N. The association between psoriasis and health-related quality of life, work productivity, and healthcare resource use in Brazil. An. Bras. Dermatol. 2018, 93, 197–204. [Google Scholar] [CrossRef]
- Singer, M.; Bulled, N.; Ostrach, B.; Mendenhall, E. Syndemics and the biosocial conception of health. Lancet 2017, 389, 941–950. [Google Scholar] [CrossRef]
- Larsen, M.H.; Krogstad, A.L.; Aas, E.; Moum, T.; Wahl, A.K. A telephone-based motivational interviewing intervention has positive effects on psoriasis severity and self-management: A randomized controlled trial. Br. J. Dermatol. 2014, 171, 1458–1469. [Google Scholar] [CrossRef]
- Xiao, Y.; Zhang, X.; Luo, D.; Kuang, Y.; Zhu, W.; Chen, X.; Shen, M. The efficacy of psychological interventions on psoriasis treatment: A systematic review and meta-analysis of randomized controlled trials. Psychol. Res. Behav. Manag. 2019, 12, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Saraceno, R.; Giunta, A.; Maccarone, M.; Chimenti, S. An Italian study on psoriasis and depression. Dermatology 2006, 212, 123–127. [Google Scholar] [CrossRef] [PubMed]
- Kurd, S.K.; Troxel, A.B.; Crits-Christoph, P.; Gelfand, J.M. The risk of depression, anxiety, and suicidality in patients with psoriasis: A population-based cohort study. Arch. Dermatol. 2010, 146, 891–895. [Google Scholar] [CrossRef] [PubMed]
- Pancar Yuksel, E.; Durmus, D.; Sarisoy, G. Perceived stress, life events, fatigue and temperament in patients with psoriasis. J. Int. Med. Res. 2019, 47, 4284–4291. [Google Scholar] [CrossRef] [PubMed]
- Dowlati, Y.; Herrmann, N.; Swardfager, W.; Liu, H.; Sham, L.; Reim, E.K.; Lanctot, K.L. A meta-analysis of cytokines in major depression. Biol. Psychiatry 2010, 67, 446–457. [Google Scholar] [CrossRef]
- Jokela, M.; Virtanen, M.; Batty, G.D.; Kivimaki, M. Inflammation and Specific Symptoms of Depression. JAMA Psychiatry 2016, 73, 87–88. [Google Scholar] [CrossRef]
- Catena-Dell’Osso, M.; Bellantuono, C.; Consoli, G.; Baroni, S.; Rotella, F.; Marazziti, D. Inflammatory and neurodegenerative pathways in depression: A new avenue for antidepressant development? Curr. Med. Chem. 2011, 18, 245–255. [Google Scholar] [CrossRef]
- Capuron, L.; Miller, A.H. Immune system to brain signaling: Neuropsychopharmacological implications. Pharmacol. Ther. 2011, 130, 226–238. [Google Scholar] [CrossRef]
- Bortolato, B.; Carvalho, A.F.; Soczynska, J.K.; Perini, G.I.; McIntyre, R.S. The Involvement of TNF-alpha in Cognitive Dysfunction Associated with Major Depressive Disorder: An Opportunity for Domain Specific Treatments. Curr. Neuropharmacol. 2015, 13, 558–576. [Google Scholar] [CrossRef]
- Gonzalez-Parra, S.; Dauden, E. Psoriasis and Depression: The Role of Inflammation. Actas Dermosifiliogr. 2019, 110, 12–19. [Google Scholar] [CrossRef]
- Arborelius, L.; Owens, M.J.; Plotsky, P.M.; Nemeroff, C.B. The role of corticotropin-releasing factor in depression and anxiety disorders. J. Endocrinol. 1999, 160, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Quevedo, M.E.; Slominski, A.; Pinto, W.; Wei, E.; Wortsman, J. Pleiotropic effects of corticotropin releasing hormone on normal human skin keratinocytes. In Vitro Cell Dev. Biol. Anim. 2001, 37, 50–54. [Google Scholar] [CrossRef]
- Jacobson, L.; Sapolsky, R. The role of the hippocampus in feedback regulation of the hypothalamic-pituitary-adrenocortical axis. Endocr. Rev. 1991, 12, 118–134. [Google Scholar] [CrossRef]
- Beurel, E.; Lowell, J.A. Th17 cells in depression. Brain. Behav. Immun. 2018, 69, 28–34. [Google Scholar] [CrossRef]
- Davami, M.H.; Baharlou, R.; Ahmadi Vasmehjani, A.; Ghanizadeh, A.; Keshtkar, M.; Dezhkam, I.; Atashzar, M.R. Elevated IL-17 and TGF-beta Serum Levels: A Positive Correlation between T-helper 17 Cell-Related Pro-Inflammatory Responses with Major Depressive Disorder. Basic Clin. Neurosci. 2016, 7, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Sahi, F.M.; Masood, A.; Danawar, N.A.; Mekaiel, A.; Malik, B.H. Association Between Psoriasis and Depression: A Traditional Review. Cureus 2020, 12, e9708. [Google Scholar] [CrossRef] [PubMed]
- Moynihan, J.; Rieder, E.; Tausk, F. Psychoneuroimmunology: The example of psoriasis. G Ital. Dermatol. Venereol. 2010, 145, 221–228. [Google Scholar] [PubMed]
- Zaccone, G.; Lauriano, E.R.; Silvestri, G.; Kenaley, C.; Icardo, J.M.; Pergolizzi, S.; Alesci, A.; Sengar, M.; Kuciel, M.; Gopesh, A. Comparative neurochemical features of the innervation patterns of the gut of the basal actinopterygian, Lepisosteus oculatus, and the euteleost, Clarias batrachus. Acta Zool. 2015, 96, 127–139. [Google Scholar] [CrossRef]
- Lauriano, E.R.; Żuwała, K.; Kuciel, M.; Budzik, K.A.; Capillo, G.; Alesci, A.; Pergolizzi, S.; Dugo, G.; Zaccone, G. Confocal immunohistochemistry of the dermal glands and evolutionary considerations in the caecilian, Typhlonectes natans(Amphibia: Gymnophiona). Acta Zool. 2016, 97, 154–164. [Google Scholar] [CrossRef]
- Lauriano, E.R.; Icardo, J.M.; Zaccone, D.; Kuciel, M.; Satora, L.; Alesci, A.; Alfa, M.; Zaccone, G. Expression patterns and quantitative assessment of neurochemical markers in the lung of the gray bichir, Polypterus senegalus (Cuvier, 1829). Acta Histochem. 2015, 117, 738–746. [Google Scholar] [CrossRef]
- Zaccone, D.; Icardo, J.M.; Kuciel, M.; Alesci, A.; Pergolizzi, S.; Satora, L.; Lauriano, E.R.; Zaccone, G. Polymorphous granular cells in the lung of the primitive fish, the bichir Polypterus senegalus. Acta Zool. 2017, 98, 13–19. [Google Scholar] [CrossRef]
- Zaccone, G.; Lauriano, E.R.; Kuciel, M.; Capillo, G.; Pergolizzi, S.; Alesci, A.; Ishimatsu, A.; Ip, Y.K.; Icardo, J.M. Identification and distribution of neuronal nitric oxide synthase and neurochemical markers in the neuroepithelial cells of the gill and the skin in the giant mudskipper, Periophthalmodon schlosseri. Zoology 2017, 125, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Maina, J.N.; Icardo, J.M.; Zaccone, G.; Aragona, M.; Lauriano, E.R.; Alesci, A.; Albano, M.; Guerrera, M.C.; Germana, A.; Fernandes, J.M.O.; et al. Immunohistochemical and ultrastructural study of the immune cell system and epithelial surfaces of the respiratory organs in the bimodally-breathing African sharptooth catfish (Clarias gariepinus Burchell, 1822). Anat. Rec. 2022. [Google Scholar] [CrossRef]
- Chalinee, R.; Tewin, T. Psoriasis: A review of the role of serotonergic system. Afr. J. Biotechnol. 2010, 9, 1528–1534. [Google Scholar] [CrossRef]
- Shajib, M.S.; Khan, W.I. The role of serotonin and its receptors in activation of immune responses and inflammation. Acta Physiol. 2015, 213, 561–574. [Google Scholar] [CrossRef]
- Capuron, L.; Neurauter, G.; Musselman, D.L.; Lawson, D.H.; Nemeroff, C.B.; Fuchs, D.; Miller, A.H. Interferon-alpha-induced changes in tryptophan metabolism. relationship to depression and paroxetine treatment. Biol. Psychiatry 2003, 54, 906–914. [Google Scholar] [CrossRef]
- Muller, N.; Schwarz, M.J. The immune-mediated alteration of serotonin and glutamate: Towards an integrated view of depression. Mol. Psychiatry 2007, 12, 988–1000. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Dunn, A.J. Mouse interleukin-6 stimulates the HPA axis and increases brain tryptophan and serotonin metabolism. Neurochem. Int. 1998, 33, 143–154. [Google Scholar] [CrossRef]
- Mori, T.; Kabashima, K.; Fukamachi, S.; Kuroda, E.; Sakabe, J.; Kobayashi, M.; Nakajima, S.; Nakano, K.; Tanaka, Y.; Matsushita, S.; et al. D1-like dopamine receptors antagonist inhibits cutaneous immune reactions mediated by Th2 and mast cells. J. Dermatol. Sci. 2013, 71, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Wardhana, M.; Windari, M.; Puspasari, N.; Suryawati, N. Role of Serotonin and Dopamine in Psoriasis: A Case-Control Study. Open Access Maced. J. Med. Sci. 2019, 7, 1138–1142. [Google Scholar] [CrossRef] [PubMed]
- Portou, M.J.; Baker, D.; Abraham, D.; Tsui, J. The innate immune system, toll-like receptors and dermal wound healing: A review. Vascul. Pharmacol. 2015, 71, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Rosenblat, J.D.; Cha, D.S.; Mansur, R.B.; McIntyre, R.S. Inflamed moods: A review of the interactions between inflammation and mood disorders. Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 53, 23–34. [Google Scholar] [CrossRef] [PubMed]
- Ginhoux, F.; Jung, S. Monocytes and macrophages: Developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 2014, 14, 392–404. [Google Scholar] [CrossRef]
- Maes, M.; Van der Planken, M.; Stevens, W.J.; Peeters, D.; DeClerck, L.S.; Bridts, C.H.; Schotte, C.; Cosyns, P. Leukocytosis, monocytosis and neutrophilia: Hallmarks of severe depression. J. Psychiatr. Res. 1992, 26, 125–134. [Google Scholar] [CrossRef]
- Mueller, S.N.; Mackay, L.K. Tissue-resident memory T cells: Local specialists in immune defence. Nat. Rev. Immunol. 2016, 16, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Talamonti, M.; Galluzzo, M.; Chiricozzi, A.; Quaglino, P.; Fabbrocini, G.; Gisondi, P.; Marzano, A.V.; Potenza, C.; Conti, A.; Parodi, A.; et al. Management of biological therapies for chronic plaque psoriasis during COVID-19 emergency in Italy. J. Eur. Acad. Dermatol. Venereol. 2020, 34, e770–e772. [Google Scholar] [CrossRef]
- Geng, W.; Zhao, J.; Fu, J.; Zhang, H.; Qiao, S. Efficacy of several biological therapies for treating moderate to severe psoriasis: A network meta-analysis. Exp. Ther. Med. 2018, 16, 5085–5095. [Google Scholar] [CrossRef]
- Ronholt, K.; Iversen, L. Old and New Biological Therapies for Psoriasis. Int. J. Mol. Sci. 2017, 18, 2297. [Google Scholar] [CrossRef]
- Sbidian, E.; Chaimani, A.; Afach, S.; Doney, L.; Dressler, C.; Hua, C.; Mazaud, C.; Phan, C.; Hughes, C.; Riddle, D.; et al. Systemic pharmacological treatments for chronic plaque psoriasis: A network meta-analysis. Cochrane Database Syst. Rev. 2020, 1, CD011535. [Google Scholar] [CrossRef]
- Huang, T.H.; Lin, C.F.; Alalaiwe, A.; Yang, S.C.; Fang, J.Y. Apoptotic or Antiproliferative Activity of Natural Products against Keratinocytes for the Treatment of Psoriasis. Int. J. Mol. Sci. 2019, 20, 2558. [Google Scholar] [CrossRef]
- Fumia, A.; Cicero, N.; Gitto, M.; Nicosia, N.; Alesci, A. Role of nutraceuticals on neurodegenerative diseases: Neuroprotective and immunomodulant activity. Nat. Prod. Res. 2021, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Alesci, A.; Fumia, A.; Lo Cascio, P.; Miller, A.; Cicero, N. Immunostimulant and Antidepressant Effect of Natural Compounds in the Management of COVID-19 Symptoms. J. Am. Coll. Nutr. 2021, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Bonesi, M.; Loizzo, M.R.; Provenzano, E.; Menichini, F.; Tundis, R. Anti-Psoriasis Agents from Natural Plant Sources. Curr Med. Chem. 2016, 23, 1250–1267. [Google Scholar] [CrossRef]
- Alessio, A.; Pergolizzi, S.; Gervasi, T.; Aragona, M.; Lo Cascio, P.; Cicero, N.; Lauriano, E.R. Biological effect of astaxanthin on alcohol-induced gut damage in Carassius auratus used as experimental model. Nat. Prod. Res. 2020, 35, 5737–5743. [Google Scholar] [CrossRef] [PubMed]
- Alesci, A.; Salvo, A.; Lauriano, E.R.; Gervasi, T.; Palombieri, D.; Bruno, M.; Pergolizzi, S.; Cicero, N. Production and extraction of astaxanthin from Phaffia rhodozyma and its biological effect on alcohol-induced renal hypoxia in Carassius auratus. Nat. Prod. Res. 2015, 29, 1122–1126. [Google Scholar] [CrossRef] [PubMed]
- Miroddi, M.; Navarra, M.; Calapai, F.; Mancari, F.; Giofre, S.V.; Gangemi, S.; Calapai, G. Review of Clinical Pharmacology of Aloe vera L. in the Treatment of Psoriasis. Phytother Res. 2015, 29, 648–655. [Google Scholar] [CrossRef]
- Leng, H.; Pu, L.; Xu, L.; Shi, X.; Ji, J.; Chen, K. Effects of aloe polysaccharide, a polysaccharide extracted from Aloe vera, on TNFalphainduced HaCaT cell proliferation and the underlying mechanism in psoriasis. Mol. Med. Rep. 2018, 18, 3537–3543. [Google Scholar] [CrossRef] [PubMed]
- Niculet, E.; Radaschin, D.S.; Nastase, F.; Draganescu, M.; Baroiu, L.; Miulescu, M.; Arbune, M.; Tatu, A.L. Influence of phytochemicals in induced psoriasis (Review). Exp. Ther. Med. 2020, 20, 3421–3424. [Google Scholar] [CrossRef]
- Foadoddini, M.; Alinejad Mofrad, S. Effect of Aloe vera Extract on Depression in People with Prediabetes. Modern Care J. 2020, 17, e100927. [Google Scholar] [CrossRef]
- Maurya, A.K.; Amrita, B.Y.; Pandey, D.K. To examine antidepressant impact of Aloe Vera and Fluoxetine in Mice. Eur. J. Mol. Clin. Med. 2021, 8, 3612–3619. [Google Scholar]
- Chen, J.; Wu, G.; Zhu, C.; Wu, Y.; Wei, R.; Ma, Q. Structurally Diverse Flavonoids from Citrus medica L. Var. sarcodactylis Swingle and Their Multiple Bioactivities: A Review. J. Food Nutr. Res. 2022, 10, 81–87. [Google Scholar] [CrossRef]
- Cirmi, S.; Bisignano, C.; Mandalari, G.; Navarra, M. Anti-infective potential of Citrus bergamia Risso et Poiteau (bergamot) derivatives: A systematic review. Phytother. Res. 2016, 30, 1404–1411. [Google Scholar] [CrossRef]
- Mannucci, C.; Navarra, M.; Calapai, F.; Squeri, R.; Gangemi, S.; Calapai, G. Clinical Pharmacology of Citrus bergamia: A Systematic Review. Phytother. Res. 2017, 31, 27–39. [Google Scholar] [CrossRef] [PubMed]
- Valkova, S. UVB phototherapeutic modalities. Comparison of two treatments for chronic plaque psoriasis. Acta Dermatovenerol. Alp. Pannonica Adriat. 2007, 16, 26–30. [Google Scholar]
- Forlot, P.; Pevet, P. Bergamot (Citrus bergamia Risso et Poiteau) essential oil: Biological properties, cosmetic and medical use. A review. J. Essent. Oil Res. 2012, 24, 195–201. [Google Scholar] [CrossRef]
- Valussi, M.; Donelli, D.; Firenzuoli, F.; Antonelli, M. Bergamot Oil: Botany, Production, Pharmacology. Encyclopedia 2021, 1, 152–176. [Google Scholar] [CrossRef]
- Alesci, A.; Aragona, M.; Cicero, N.; Lauriano, E.R. Can neutraceuticals assist treatment and improve COVID-19 symptoms? Nat. Prod. Res. 2021. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.K.; Hwang, J.T.; Kwon, D.Y.; Surh, Y.J.; Park, O.J. Induction of apoptosis by quercetin is mediated through AMPKalpha1/ASK1/p38 pathway. Cancer Lett. 2010, 292, 228–236. [Google Scholar] [CrossRef]
- Shen, S.C.; Lee, W.R.; Yang, L.Y.; Tsai, H.H.; Yang, L.L.; Chen, Y.C. Quercetin enhancement of arsenic-induced apoptosis via stimulating ROS-dependent p53 protein ubiquitination in human HaCaT keratinocytes. Exp. Dermatol. 2012, 21, 370–375. [Google Scholar] [CrossRef]
- Mestry, M.; Rane, M.; Bajaj, A. Commiphora mukul and Quercetin Loaded Liposphere Gel: Potential Treatment for Psoriasis. Indian J. Pharm. Educ. Res. 2020, 54, 654–667. [Google Scholar] [CrossRef]
- Chen, H.; Lu, C.; Liu, H.; Wang, M.; Zhao, H.; Yan, Y.; Han, L. Quercetin ameliorates imiquimod-induced psoriasis-like skin inflammation in mice via the NF-kappaB pathway. Int. Immunopharmacol. 2017, 48, 110–117. [Google Scholar] [CrossRef]
- Samad, N.; Saleem, A.; Yasmin, F.; Shehzad, M.A. Quercetin protects against stress-induced anxiety- and depression-like behavior and improves memory in male mice. Physiol. Res. 2018, 67, 795–808. [Google Scholar] [CrossRef]
- Kosari-Nasab, M.; Shokouhi, G.; Ghorbanihaghjo, A.; Mesgari-Abbasi, M.; Salari, A.A. Quercetin mitigates anxiety-like behavior and normalizes hypothalamus-pituitary-adrenal axis function in a mouse model of mild traumatic brain injury. Behav. Pharmacol. 2019, 30, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Chen, X.Y.; Martin, C. Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Sci. Bull. 2016, 61, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.F.; Ma, K.H.; Liu, P.S.; Chen, B.W.; Chueh, S.H. Baicalein increases keratin 1 and 10 expression in HaCaT keratinocytes via TRPV4 receptor activation. Exp. Dermatol 2016, 25, 623–629. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.J.; Ma, X.C.; Gao, X.Y.; Xue, X.H.; Zhang, S.Q. Latest research progress in the correlation between baicalein and breast cancer invasion and metastasis. Mol. Clin. Oncol. 2016, 4, 472–476. [Google Scholar] [CrossRef]
- Dinda, B.; Dinda, S.; DasSharma, S.; Banik, R.; Chakraborty, A.; Dinda, M. Therapeutic potentials of baicalin and its aglycone, baicalein against inflammatory disorders. Eur. J. Med. Chem. 2017, 131, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.; Sur, B.; Park, J.; Kim, S.H.; Kwon, S.; Yeom, M.; Shim, I.; Lee, H.; Hahm, D.H. Chronic administration of baicalein decreases depression-like behavior induced by repeated restraint stress in rats. Korean J. Physiol. Pharmacol. 2013, 17, 393–403. [Google Scholar] [CrossRef]
- Zhao, X.; Kong, D.; Zhou, Q.; Wei, G.; Song, J.; Liang, Y.; Du, G. Baicalein alleviates depression-like behavior in rotenone- induced Parkinson’s disease model in mice through activating the BDNF/TrkB/CREB pathway. Biomed. Pharmacother. 2021, 140, 111556. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, A.; Alves, H.; Marques, V.; Durazzo, A.; Lucarini, M.; Alves, T.F.; Morsink, M.; Willemen, N.; Eder, P.; Chaud, M.V.; et al. Properties, Extraction Methods, and Delivery Systems for Curcumin as a Natural Source of Beneficial Health Effects. Medicina 2020, 56, 336. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Zhao, Y.; Hu, J. Curcumin inhibits imiquimod-induced psoriasis-like inflammation by inhibiting IL-1beta and IL-6 production in mice. PLoS ONE 2013, 8, e67078. [Google Scholar] [CrossRef] [PubMed]
- Nardo, V.D.; Gianfaldoni, S.; Tchernev, G.; Wollina, U.; Barygina, V.; Lotti, J.; Daaboul, F.; Lotti, T. Use of Curcumin in Psoriasis. Open Access Maced. J. Med. Sci. 2018, 6, 218–220. [Google Scholar] [CrossRef]
- Tizabi, Y.; Hurley, L.L.; Qualls, Z.; Akinfiresoye, L. Relevance of the anti-inflammatory properties of curcumin in neurodegenerative diseases and depression. Molecules 2014, 19, 20864–20879. [Google Scholar] [CrossRef]
- Polese, B.; Zhang, H.; Thurairajah, B.; King, I.L. Innate Lymphocytes in Psoriasis. Front. Immunol. 2020, 11, 242. [Google Scholar] [CrossRef] [PubMed]
- Alesci, A.; Nicosia, N.; Fumia, A.; Giorgianni, F.; Santini, A.; Cicero, N. Resveratrol and Immune Cells: A Link to Improve Human Health. Molecules 2022, 27, 424. [Google Scholar] [CrossRef] [PubMed]
- Di Stefano, V.; Avellone, G.; Pitonzo, R.; Capocchiano, V.G.; Mazza, A.; Cicero, N.; Dugo, G. Natural co-occurrence of ochratoxin A, ochratoxin B and aflatoxins in Sicilian red wines. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2015, 32, 1343–1351. [Google Scholar] [CrossRef]
- Rigon, R.B.; Fachinetti, N.; Severino, P.; Durazzo, A.; Lucarini, M.; Atanasov, A.G.; El Mamouni, S.; Chorilli, M.; Santini, A.; Souto, E.B. Quantification of Trans-Resveratrol-Loaded Solid Lipid Nanoparticles by a Validated Reverse-Phase HPLC Photodiode Array. Appl. Sci. 2019, 9, 4961. [Google Scholar] [CrossRef]
- Yeung, A.W.K.; Aggarwal, B.B.; Orhan, I.E.; Barreca, D.; Battino, M.; Belwal, T.; Bishayee, A.; Daglia, M.; Devkota, H.P.; Echeverria, J.; et al. Resveratrol, a popular dietary supplement for human and animal health: Quantitative research literature analysis-a review. Anim. Sci. Pap. Rep. 2019, 37, 103–118. [Google Scholar]
- Oliveira, A.L.B.; Monteiro, V.V.S.; Navegantes-Lima, K.C.; Reis, J.F.; Gomes, R.S.; Rodrigues, D.V.S.; Gaspar, S.L.F.; Monteiro, M.C. Resveratrol Role in Autoimmune Disease-A Mini-Review. Nutrients 2017, 9, 1306. [Google Scholar] [CrossRef] [PubMed]
- Khurana, B.; Arora, D.; Narang, R.K. QbD based exploration of resveratrol loaded polymeric micelles based carbomer gel for topical treatment of plaque psoriasis: In vitro, ex vivo and in vivo studies. J. Drug Deliv. Sci. Technol. 2020, 59, 101901. [Google Scholar] [CrossRef]
- Zhu, X.; Li, W.; Li, Y.; Xu, W.; Yuan, Y.; Zheng, V.; Zhang, H.; O’Donnell, J.M.; Xu, Y.; Yin, X. The antidepressant- and anxiolytic-like effects of resveratrol: Involvement of phosphodiesterase-4D inhibition. Neuropharmacology 2019, 153, 20–31. [Google Scholar] [CrossRef] [PubMed]




| Natural Compounds | Biological Effects | References |
|---|---|---|
| Aloe vera L. | Immunomodulant and stimulant effect on macrophages, lymphocytes, and dendritic cells. | [187] |
| Antidepressant effect. | [190,191] | |
| Bergamot essential oil | Antibacterial and anti-psoriatic properties. | [193,195] |
| Moderation of mood disorders and stress-induced anxiety. Neuroprotective and sleep-inducing effect. | [194] | |
| Quercetin | Decrease in TNF-α, IL-6, and IL-17 levels. | [202] |
| Regulation of cholinergic and sero-tonergic functions, anxiolytic and anti-depressant effects, enhances memory performance. | [203,204] | |
| Baicalein | Antioxidant effect, inhibits the release of IL-17 and the expression of IL-6 and TNF-α. | [207,208] |
| Antidepressive and neuroprotective effect. | [183,209] | |
| Curcumin | Decrease in IL-1β, IL-6, IL-22, and TNF-α levels. | [212] |
| Modulates serotonin, monoamine, noradrenaline, and dopamine. Regu-lates the function of the HPA axis. | [214,215] | |
| Resveratrol | Reduces the secretion of inflammatory cytokines and induces apoptosis in keratinocytes. Inhibits the release of IL-17. | [220] |
| Anxiolytic and antidepressant effect by inhibiting the expression of PDE4D, which regulates cAMP expression at the intracellular level. | [222] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alesci, A.; Lauriano, E.R.; Fumia, A.; Irrera, N.; Mastrantonio, E.; Vaccaro, M.; Gangemi, S.; Santini, A.; Cicero, N.; Pergolizzi, S. Relationship between Immune Cells, Depression, Stress, and Psoriasis: Could the Use of Natural Products Be Helpful? Molecules 2022, 27, 1953. https://doi.org/10.3390/molecules27061953
Alesci A, Lauriano ER, Fumia A, Irrera N, Mastrantonio E, Vaccaro M, Gangemi S, Santini A, Cicero N, Pergolizzi S. Relationship between Immune Cells, Depression, Stress, and Psoriasis: Could the Use of Natural Products Be Helpful? Molecules. 2022; 27(6):1953. https://doi.org/10.3390/molecules27061953
Chicago/Turabian StyleAlesci, Alessio, Eugenia Rita Lauriano, Angelo Fumia, Natasha Irrera, Enza Mastrantonio, Mario Vaccaro, Sebastiano Gangemi, Antonello Santini, Nicola Cicero, and Simona Pergolizzi. 2022. "Relationship between Immune Cells, Depression, Stress, and Psoriasis: Could the Use of Natural Products Be Helpful?" Molecules 27, no. 6: 1953. https://doi.org/10.3390/molecules27061953
APA StyleAlesci, A., Lauriano, E. R., Fumia, A., Irrera, N., Mastrantonio, E., Vaccaro, M., Gangemi, S., Santini, A., Cicero, N., & Pergolizzi, S. (2022). Relationship between Immune Cells, Depression, Stress, and Psoriasis: Could the Use of Natural Products Be Helpful? Molecules, 27(6), 1953. https://doi.org/10.3390/molecules27061953













