Revealing the Combined Effect of Active Sites and Intra-Particle Diffusion on Adsorption Mechanism of Methylene Blue on Activated Red-Pulp Pomelo Peel Biochar
Abstract
1. Introduction
2. Results
2.1. Morphology and Textural Properties of PP and PPCs
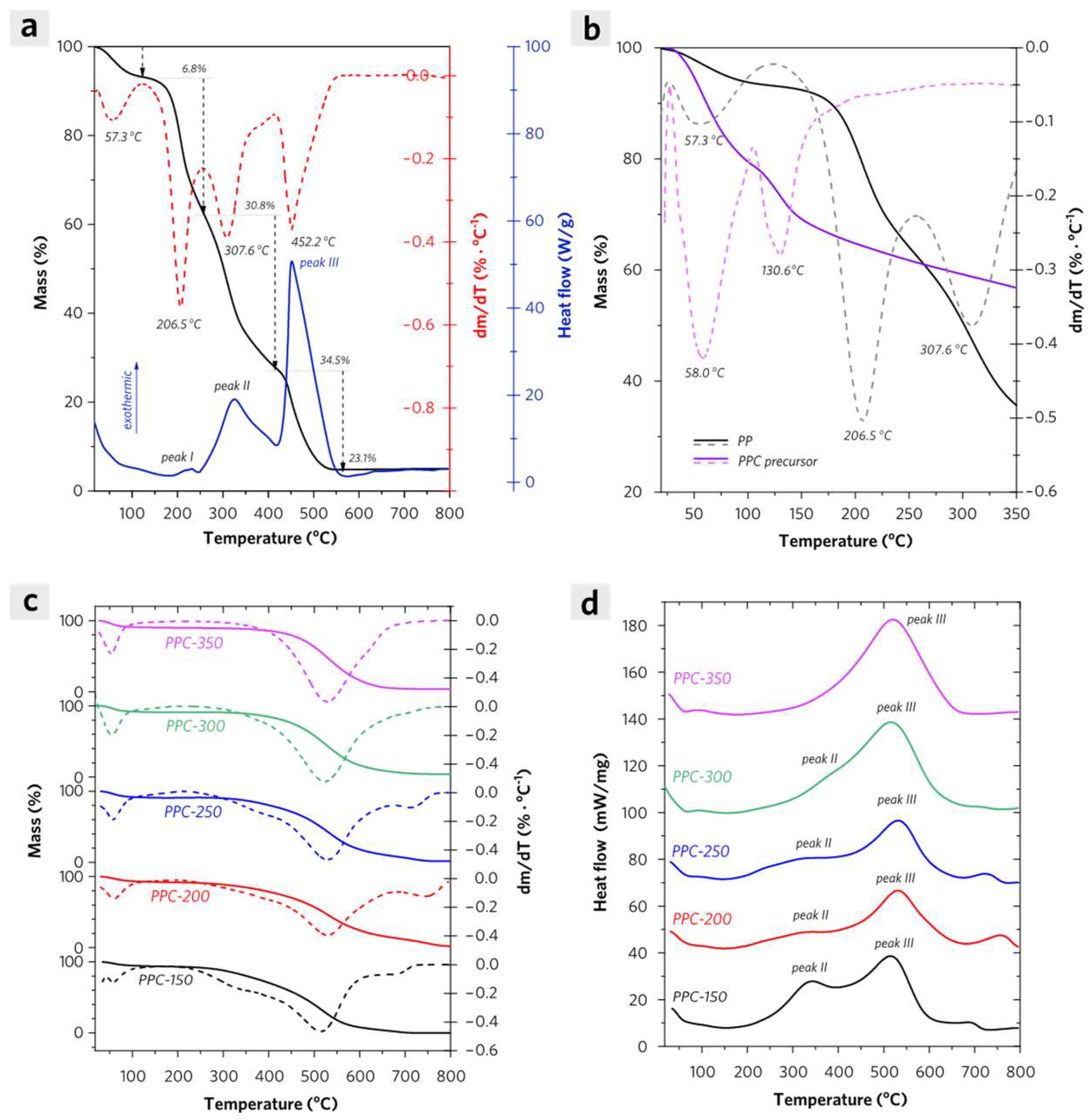
2.2. Thermal Analysis of PP and PPCs
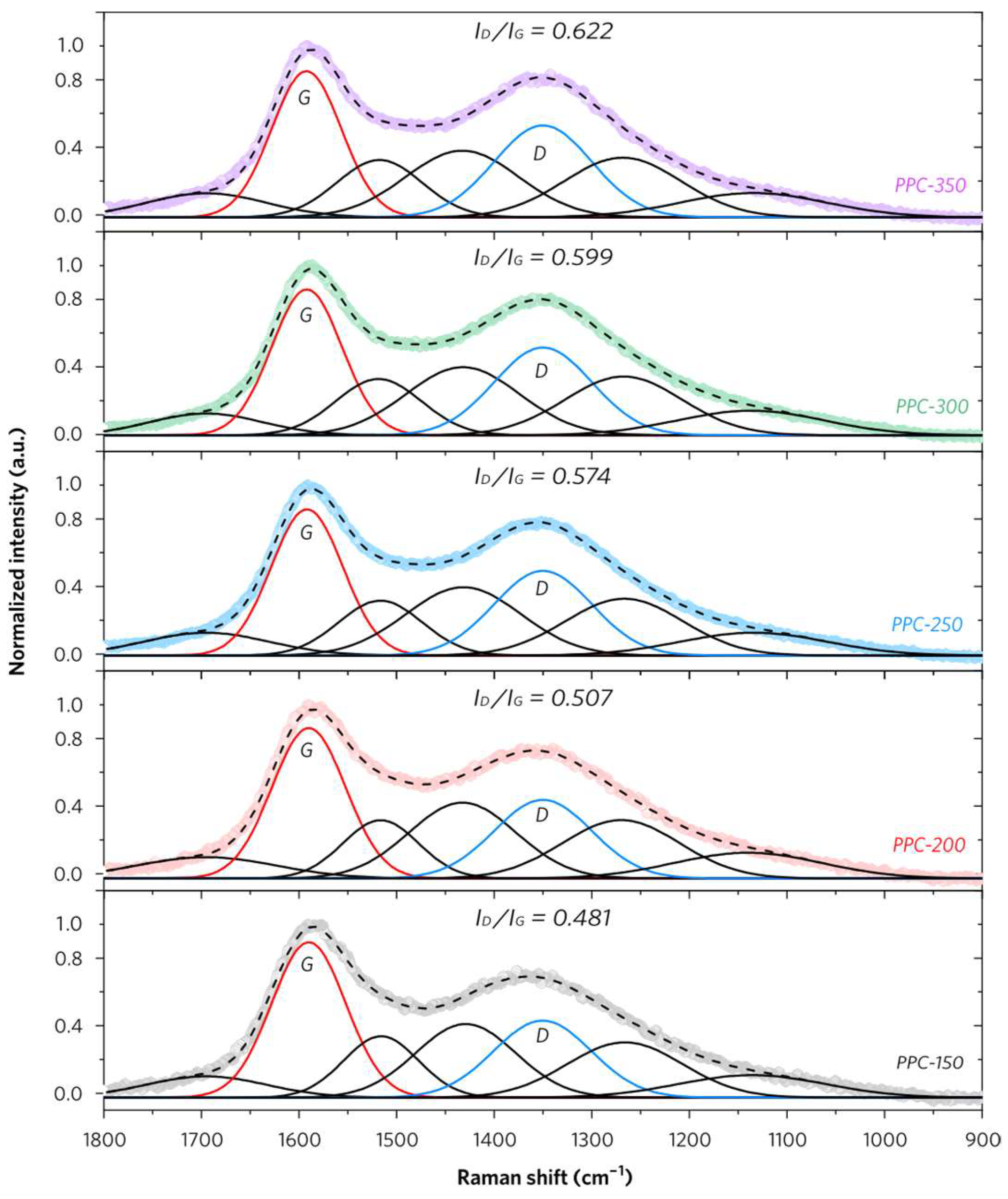
2.3. Surface Chemistry and Characteristics of PPCs
2.4. Adsorption Isotherms and Thermodynamics
2.4.1. Langmuir Isotherm Model
2.4.2. Freundlich Isotherm
2.4.3. Temkin Isotherm
2.5. Adsorption Thermodynamics
2.6. Adsorption Kinetics
2.6.1. Adsorption Empirical and Reaction Models
2.6.2. Adsorption Diffusion Models
3. Materials and Methods
3.1. Materials and Reagents
3.2. Preparation of Pomelo Peel Biochar
3.3. Batch Adsorption Experiments
4. Conclusions
- (1)
- The adsorption of MB on PPC-150 mainly exhibits the character of chemisorption with positive enthalpy energy of adsorption (12.3 kJ/mol), which is likely due to the formation of dipole-dipole H-bonding (N→H–O–adsorbent) and Yoshida H-bonding with electron transfer from aromatic rings (framework structure of MB) to hydroxyl groups (major surface sites of PPC-150);
- (2)
- The adsorption of MB on PPCs except for PPC-150 mainly exhibits characters of physisorption with negative enthalpy energy of adsorption (–25.0 to –32.8 kJ/mol). Desorption becomes significant at high temperatures (above 30 °C), leading to the decrease of equilibrium adsorption quantities of MB on PPCs;
- (3)
- PPC-300 and PPC-350 exhibit much higher apparent adsorption rates than either PPC-250 or PPC-200, leading to a rapid adsorption equilibrium due to synergetic contributions of surface adsorption (active sites) and pore-filling (intra-particle diffusion);
- (4)
- Especially, PPC-300 and PPC-350 exhibit intra-particle-diffusion-controlled adsorption kinetic process with low initial MB concentration (100 ppm) or at the very beginning and final stage of adsorption with high initial MB concentration (300 ppm) at 40 °C.
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Sample Availability
References
- Sabzehmeidani, M.M.; Mahnaee, S.; Ghaedi, M.; Heidari, H.; Roy, V.A. Carbon based materials: A review of adsorbents for inorganic and organic compounds. Mater. Adv. 2021, 2, 598–627. [Google Scholar] [CrossRef]
- Reza, M.S.; Yun, C.S.; Afroze, S.; Radenahmad, N.; Bakar, M.S.A.; Saidur, R.; Taweekun, J.; Azad, A.K. Preparation of activated carbon from biomass and its’ applications in water and gas purification, a review. Arab J. Basic Appl. Sci. 2020, 27, 208–238. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Hogland, W.; Marques, M.; Sillanpää, M. An overview of the modification methods of activated carbon for its water treatment applications. Chem. Eng. J. 2013, 219, 499–511. [Google Scholar] [CrossRef]
- Tran, H.N.; Wang, Y.-F.; You, S.-J.; Chao, H.-P. Insights into the mechanism of cationic dye adsorption on activated charcoal: The importance of π–π interactions. Process Saf. Environ. Prot. 2017, 107, 168–180. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Chao, H.-P. Fast and efficient adsorption of methylene green 5 on activated carbon prepared from new chemical activation method. J. Environ. Manag. 2017, 188, 322–336. [Google Scholar] [CrossRef] [PubMed]
- Tran, H.N.; Tomul, F.; Ha, N.T.H.; Nguyen, D.T.; Lima, E.C.; Le, G.T.; Chang, C.-T.; Masindi, V.; Woo, S.H. Innovative spherical biochar for pharmaceutical removal from water: Insight into adsorption mechanism. J. Hazard. Mater. 2020, 394, 122255. [Google Scholar] [CrossRef] [PubMed]
- Tong, Y.; McNamara, P.J.; Mayer, B.K. Adsorption of organic micropollutants onto biochar: A review of relevant kinetics, mechanisms and equilibrium. Environ. Sci. Water Res. Technol. 2019, 5, 821–838. [Google Scholar] [CrossRef]
- Tran, H.N.; You, S.-J.; Hosseini-Bandegharaei, A.; Chao, H.-P. Mistakes and inconsistencies regarding adsorption of contaminants from aqueous solutions: A critical review. Water Res. 2017, 120, 88–116. [Google Scholar] [CrossRef]
- Heidarinejad, Z.; Dehghani, M.H.; Heidari, M.; Javedan, G.; Ali, I.; Sillanpää, M. Methods for preparation and activation of activated carbon: A review. Environ. Chem. Lett. 2020, 18, 393–415. [Google Scholar] [CrossRef]
- Wei, F.; Zhu, Y.; He, T.; Zhu, S.; Wang, T.; Yao, C.; Yu, C.; Huang, P.; Li, Y.; Zhao, Q.; et al. Insights into the pH-Dependent Adsorption Behavior of Ionic Dyes on Phosphoric Acid-Activated Biochar. ACS Omega 2022, 7, 46288–46302. [Google Scholar] [CrossRef] [PubMed]
- Renier, M.L.; Kohn, D.H. Development and characterization of a biodegradable polyphosphate. J. Biomed. Mater. Res. Off. J. Soc. Biomater. Jpn. Soc. Biomater. 1997, 34, 95–104. [Google Scholar] [CrossRef]
- Yang, H.; Yan, R.; Chen, H.; Lee, D.H.; Zheng, C. Characteristics of hemicellulose, cellulose and lignin pyrolysis. Fuel 2007, 86, 1781–1788. [Google Scholar] [CrossRef]
- Kan, T.; Strezov, V.; Evans, T.J. Lignocellulosic biomass pyrolysis: A review of product properties and effects of pyrolysis parameters. Renew. Sustain. Energy Rev. 2016, 57, 1126–1140. [Google Scholar]
- Puziy, A.; Poddubnaya, O.; Martınez-Alonso, A.; Suárez-Garcıa, F.; Tascón, J. Synthetic carbons activated with phosphoric acid: I. Surface chemistry and ion binding properties. Carbon 2002, 40, 1493–1505. [Google Scholar] [CrossRef]
- Guo, Y.; Rockstraw, D.A. Activated carbons prepared from rice hull by one-step phosphoric acid activation. Microporous Mesoporous Mater. 2007, 100, 12–19. [Google Scholar] [CrossRef]
- Bellamy, L. The Infra-Red Spectra of Complex Molecules; Springer Science & Business Media: New York, NY, USA, 2013. [Google Scholar]
- Wang, Y.; Alsmeyer, D.C.; McCreery, R.L. Raman spectroscopy of carbon materials: Structural basis of observed spectra. Chem. Mater. 1990, 2, 557–563. [Google Scholar] [CrossRef]
- Tuinstra, F.; Koenig, J.L. Raman spectrum of graphite. J. Chem. Phys. 1970, 53, 1126–1130. [Google Scholar] [CrossRef]
- Reich, S.; Thomsen, C. Raman spectroscopy of graphite. Philos. Trans. R. Soc. London. Ser. A Math. Phys. Eng. Sci. 2004, 362, 2271–2288. [Google Scholar] [CrossRef]
- Li, X.; Hayashi, J.-i.; Li, C.-Z. FT-Raman spectroscopic study of the evolution of char structure during the pyrolysis of a Victorian brown coal. Fuel 2006, 85, 1700–1707. [Google Scholar] [CrossRef]
- Kim, P.; Johnson, A.; Edmunds, C.W.; Radosevich, M.; Vogt, F.; Rials, T.G.; Labbé, N. Surface functionality and carbon structures in lignocellulosic-derived biochars produced by fast pyrolysis. Energy Fuels 2011, 25, 4693–4703. [Google Scholar] [CrossRef]
- Anstey, A.; Vivekanandhan, S.; Rodriguez-Uribe, A.; Misra, M.; Mohanty, A.K. Oxidative acid treatment and characterization of new biocarbon from sustainable Miscanthus biomass. Sci. Total Environ. 2016, 550, 241–247. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Wang, J.; Guo, X. Adsorption isotherm models: Classification, physical meaning, application and solving method. Chemosphere 2020, 258, 127279. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Maimaitiniyazi, R.; Wang, Y. Some consideration triggered by misquotation of Temkin model and the derivation of its correct form. Arab. J. Chem. 2022, 15, 104267. [Google Scholar] [CrossRef]
- Azizian, S.; Eris, S.; Wilson, L.D. Re-evaluation of the century-old Langmuir isotherm for modeling adsorption phenomena in solution. Chem. Phys. 2018, 513, 99–104. [Google Scholar] [CrossRef]
- Lima, E.C.; Hosseini-Bandegharaei, A.; Moreno-Piraján, J.C.; Anastopoulos, I. A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t Hoof equation for calculation of thermodynamic parameters of adsorption. J. Mol. Liq. 2019, 273, 425–434. [Google Scholar] [CrossRef]
- Enache, A.-C.; Samoila, P.; Cojocaru, C.; Apolzan, R.; Predeanu, G.; Harabagiu, V. An eco-friendly modification of a walnut shell biosorbent for increased efficiency in wastewater treatment. Sustainability 2023, 15, 2704. [Google Scholar] [CrossRef]
- Sakkayawong, N.; Thiravetyan, P.; Nakbanpote, W. Adsorption mechanism of synthetic reactive dye wastewater by chitosan. J. Colloid Interface Sci. 2005, 286, 36–42. [Google Scholar] [CrossRef]
- Dolphen, R.; Sakkayawong, N.; Thiravetyan, P.; Nakbanpote, W. Adsorption of Reactive Red 141 from wastewater onto modified chitin. J. Hazard. Mater. 2007, 145, 250–255. [Google Scholar] [CrossRef]
- Natarajan, T.S.; Bajaj, H.C.; Tayade, R.J. Preferential adsorption behavior of methylene blue dye onto surface hydroxyl group enriched TiO2 nanotube and its photocatalytic regeneration. J. Hazard. Mater. 2014, 433, 104–114. [Google Scholar]
- Wang, J.; Guo, X. Adsorption kinetic models: Physical meanings, applications, and solving methods. J. Hazard. Mater. 2020, 390, 122156. [Google Scholar] [CrossRef] [PubMed]
- Önal, Y.; Akmil-Başar, C.; Sarıcı-Özdemir, Ç. Investigation kinetics mechanisms of adsorption malachite green onto activated carbon. J. Hazard. Mater. 2007, 146, 194–203. [Google Scholar] [CrossRef] [PubMed]









| Sample Name | SBET (m2/g) | Smeso (m2/g) | Vtotal (cm3/g) | Vmicro (cm3/g) | Vmeso (cm3/g) | Mid-Value Dmicro (nm) | Average Dmeso (nm) |
|---|---|---|---|---|---|---|---|
| PP | 0.5503 | – | 0.00143 | – | – | – | – |
| PPC-150 | 3.065 | – | 0.015 | – | – | – | – |
| PPC-200 | 29.463 | 25.854 | 0.039 | – | 0.038 | – | – |
| PPC-250 | 511.380 | 101.325 | 0.267 | 0.2017 | 0.101 | 0.6005 | 3.994 |
| PPC-300 | 1274.577 | 646.040 | 0.927 | 0.5105 | 0.661 | 0.6444 | 4.095 |
| PPC-350 | 1094.909 | 824.592 | 1.298 | 0.4559 | 1.181 | 0.6726 | 5.728 |
| Isotherm Model | Temperature | Parameters | Samples | ||||
|---|---|---|---|---|---|---|---|
| PPC-150 | PPC-200 | PPC-250 | PPC-300 | PPC-350 | |||
| Langmuir | 293 K | Qmax | 262.08 | 318.05 | 359.72 | 385.23 | 351.40 |
| KL | 0.271 | 1.360 | 1.783 | 2.750 | 1.797 | ||
| Adj. R2 | 0.974 | 0.968 | 0.955 | 0.946 | 0.936 | ||
| 313 K | Qmax | 286.57 | 304.30 | 345.06 | 356.90 | 322.68 | |
| KL | 0.374 | 0.545 | 0.829 | 1.162 | 0.932 | ||
| Adj. R2 | 0.967 | 0.969 | 0.946 | 0.961 | 0.958 | ||
| Isotherm Model | Temperature | Parameters | Samples | ||||
|---|---|---|---|---|---|---|---|
| PPC-150 | PPC-200 | PPC-250 | PPC-300 | PPC-350 | |||
| Freundlich | 293 K | KL | 104.39 | 153.46 | 171.12 | 187.50 | 163.17 |
| n | 5.79 | 6.90 | 6.45 | 6.60 | 6.37 | ||
| Adj. R2 | 0.951 | 0.919 | 0.947 | 0.966 | 0.935 | ||
| 313 K | KL | 113.15 | 131.80 | 142.33 | 155.62 | 149.56 | |
| n | 5.62 | 6.19 | 5.60 | 6.04 | 6.47 | ||
| Adj. R2 | 0.944 | 0.926 | 0.971 | 0.932 | 0.922 | ||
| Isotherm Model | Temperature | Parameters | Samples | ||||
|---|---|---|---|---|---|---|---|
| PPC-150 | PPC-200 | PPC-250 | PPC-300 | PPC-350 | |||
| Temkin | 293 K | Q | 33.29 | 32.90 | 38.68 | 38.72 | 37.10 |
| Kt | 13.00 | 110.51 | 101.26 | 201.67 | 104.66 | ||
| bt (kJ/mol) | 20.212 | 25.130 | 25.613 | 26.631 | 25.657 | ||
| Adj. R2 | 0.992 | 0.974 | 0.986 | 0.992 | 0.968 | ||
| 313 K | Q | 36.11 | 35.53 | 40.94 | 39.80 | 35.84 | |
| Kt | 15.50 | 30.74 | 33.76 | 55.06 | 59.52 | ||
| bt (kJ/mol) | 22.704 | 24.259 | 24.821 | 25.519 | 25.318 | ||
| Adj. R2 | 0.985 | 0.977 | 0.987 | 0.974 | 0.973 | ||
| Samples | Parameters | ∆Ho (kJ/mol) | ∆So (J/mol K) | ||
|---|---|---|---|---|---|
| T (K) | Kc | ∆Go (kJ/mol) | |||
| PPC-150 | 293 | 13,811 | −23.223 | 12.281 | 121.2 |
| 313 | 19,060 | −25.646 | |||
| PPC-200 | 293 | 69,309 | −27.152 | −34.862 | −26.3 |
| 313 | 27,774 | −26.626 | |||
| PPC-250 | 293 | 90,866 | −27.812 | −29.196 | −4.7 |
| 313 | 42,248 | −27.718 | |||
| PPC-300 | 293 | 140,147 | −28.868 | −32.841 | −13.6 |
| 313 | 59,218 | −28.596 | |||
| PPC-350 | 293 | 91,580 | −27.831 | −25.030 | 9.6 |
| 313 | 47,497 | −28.022 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, F.; Jin, S.; Yao, C.; Wang, T.; Zhu, S.; Ma, Y.; Qiao, H.; Shan, L.; Wang, R.; Lian, X.; et al. Revealing the Combined Effect of Active Sites and Intra-Particle Diffusion on Adsorption Mechanism of Methylene Blue on Activated Red-Pulp Pomelo Peel Biochar. Molecules 2023, 28, 4426. https://doi.org/10.3390/molecules28114426
Wei F, Jin S, Yao C, Wang T, Zhu S, Ma Y, Qiao H, Shan L, Wang R, Lian X, et al. Revealing the Combined Effect of Active Sites and Intra-Particle Diffusion on Adsorption Mechanism of Methylene Blue on Activated Red-Pulp Pomelo Peel Biochar. Molecules. 2023; 28(11):4426. https://doi.org/10.3390/molecules28114426
Chicago/Turabian StyleWei, Fang, Shenglong Jin, Chunyi Yao, Tianhao Wang, Shengpu Zhu, Yabiao Ma, Heng Qiao, Linxi Shan, Rencong Wang, Xiaoxue Lian, and et al. 2023. "Revealing the Combined Effect of Active Sites and Intra-Particle Diffusion on Adsorption Mechanism of Methylene Blue on Activated Red-Pulp Pomelo Peel Biochar" Molecules 28, no. 11: 4426. https://doi.org/10.3390/molecules28114426
APA StyleWei, F., Jin, S., Yao, C., Wang, T., Zhu, S., Ma, Y., Qiao, H., Shan, L., Wang, R., Lian, X., Tong, X., Li, Y., Zhao, Q., & Song, W. (2023). Revealing the Combined Effect of Active Sites and Intra-Particle Diffusion on Adsorption Mechanism of Methylene Blue on Activated Red-Pulp Pomelo Peel Biochar. Molecules, 28(11), 4426. https://doi.org/10.3390/molecules28114426






