Green Extraction of Polyphenols via Deep Eutectic Solvents and Assisted Technologies from Agri-Food By-Products
Abstract
:1. Introduction
2. Polyphenol-Rich Agri-Food By-Products
2.1. Fruit By-Products
2.2. Vegetable By-Products
2.3. Tree Nut By-Products
2.4. Cereal By-Products
2.5. Oil-Bearing Crop By-Products
2.6. Other Agri-Food By-Products
3. Use of DESs as Green Solvents in the Extraction of Polyphenols from Agri-Food By-Products
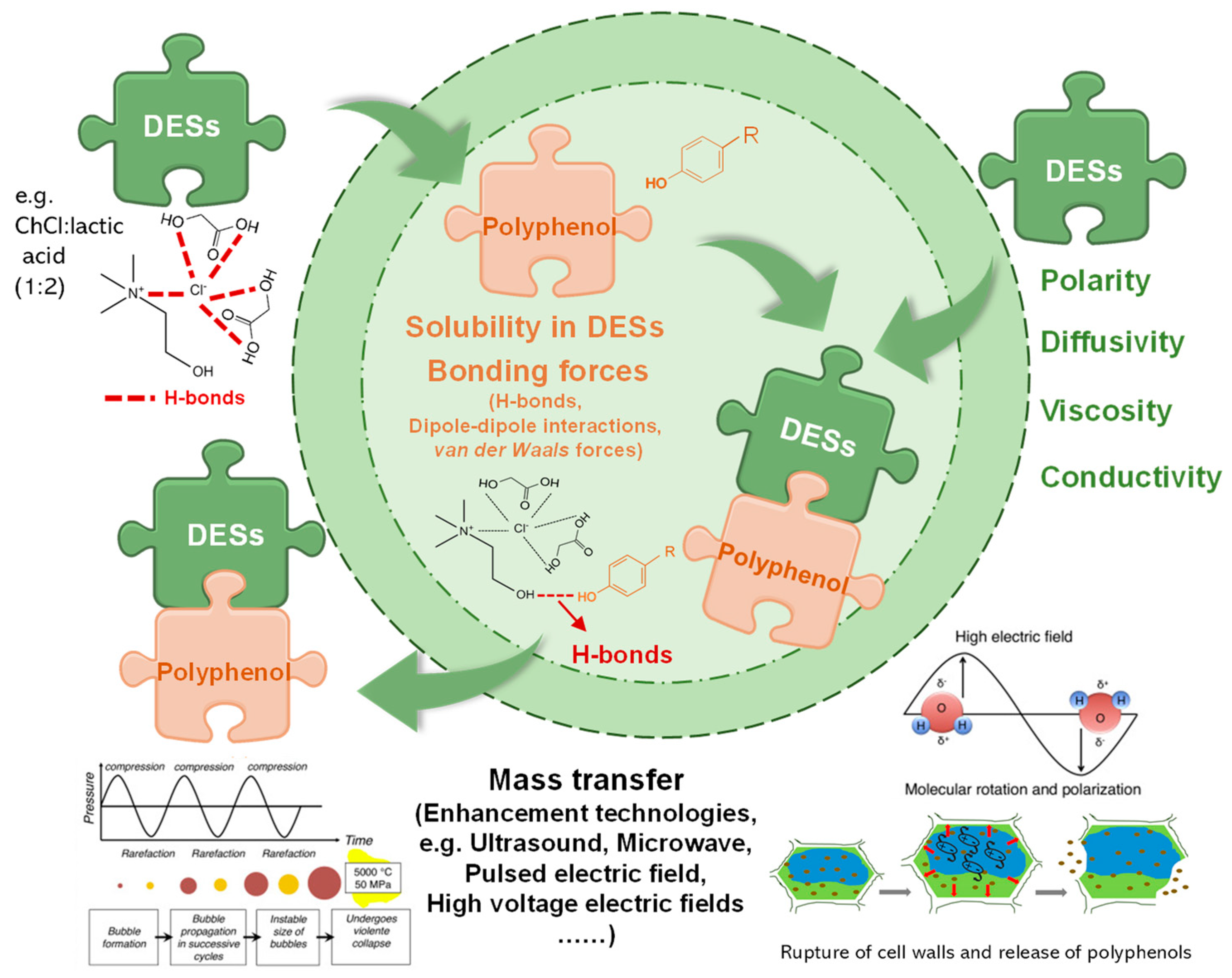
3.1. DESs and Its Mechanism of Polyphenol Extraction
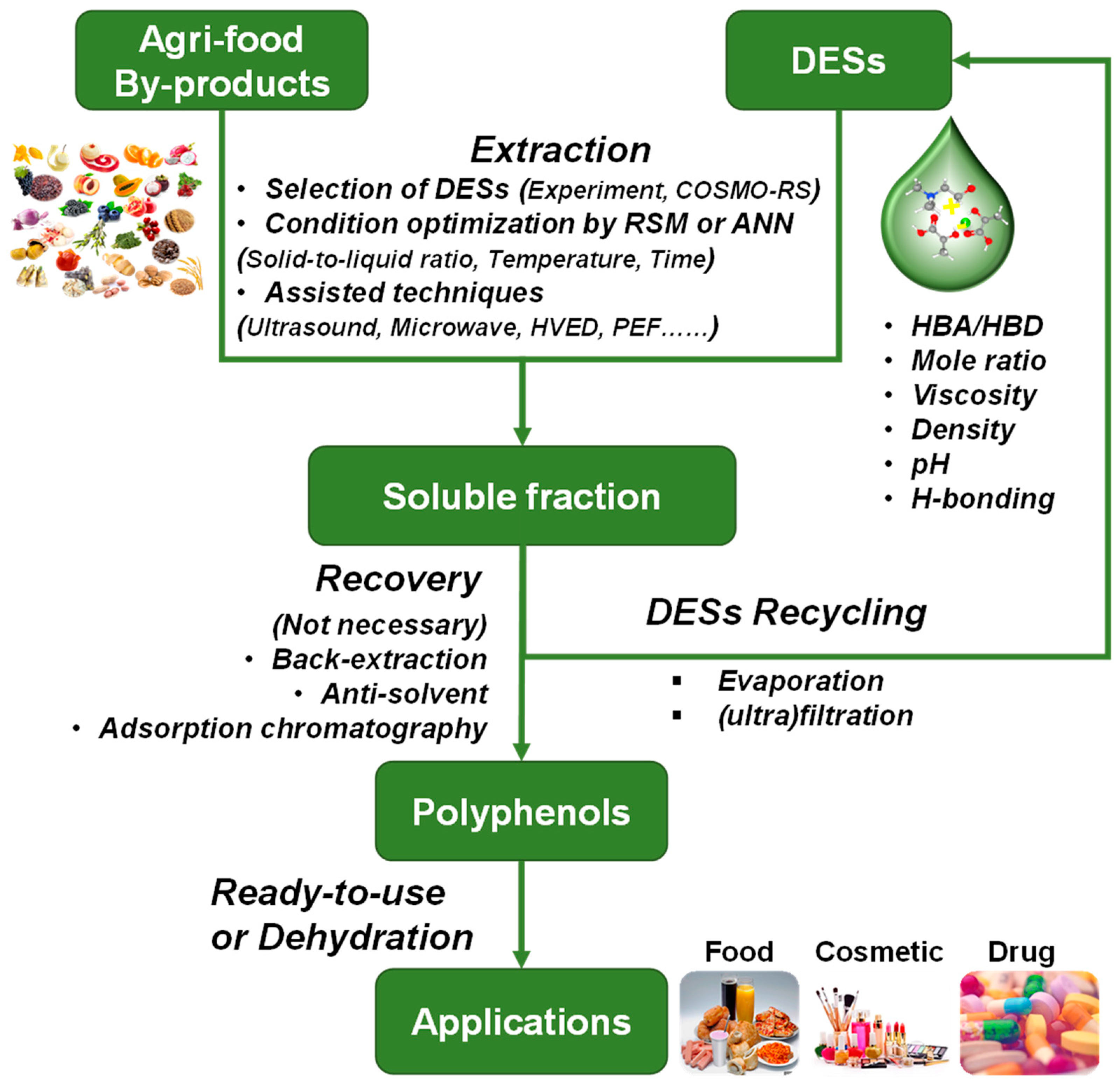
3.2. Process for the Green Extraction of Polyphenols
4. Assisted Technologies of Polyphenols Extraction with DESs
4.1. Ultrasound
4.2. Microwave
4.3. Other or Combined Technologies
4.4. Comparison of These Assisted Technologies
5. Applications of Polyphenol-Rich DESs Extract in the Food, Cosmetic, and Pharmaceutical Industry
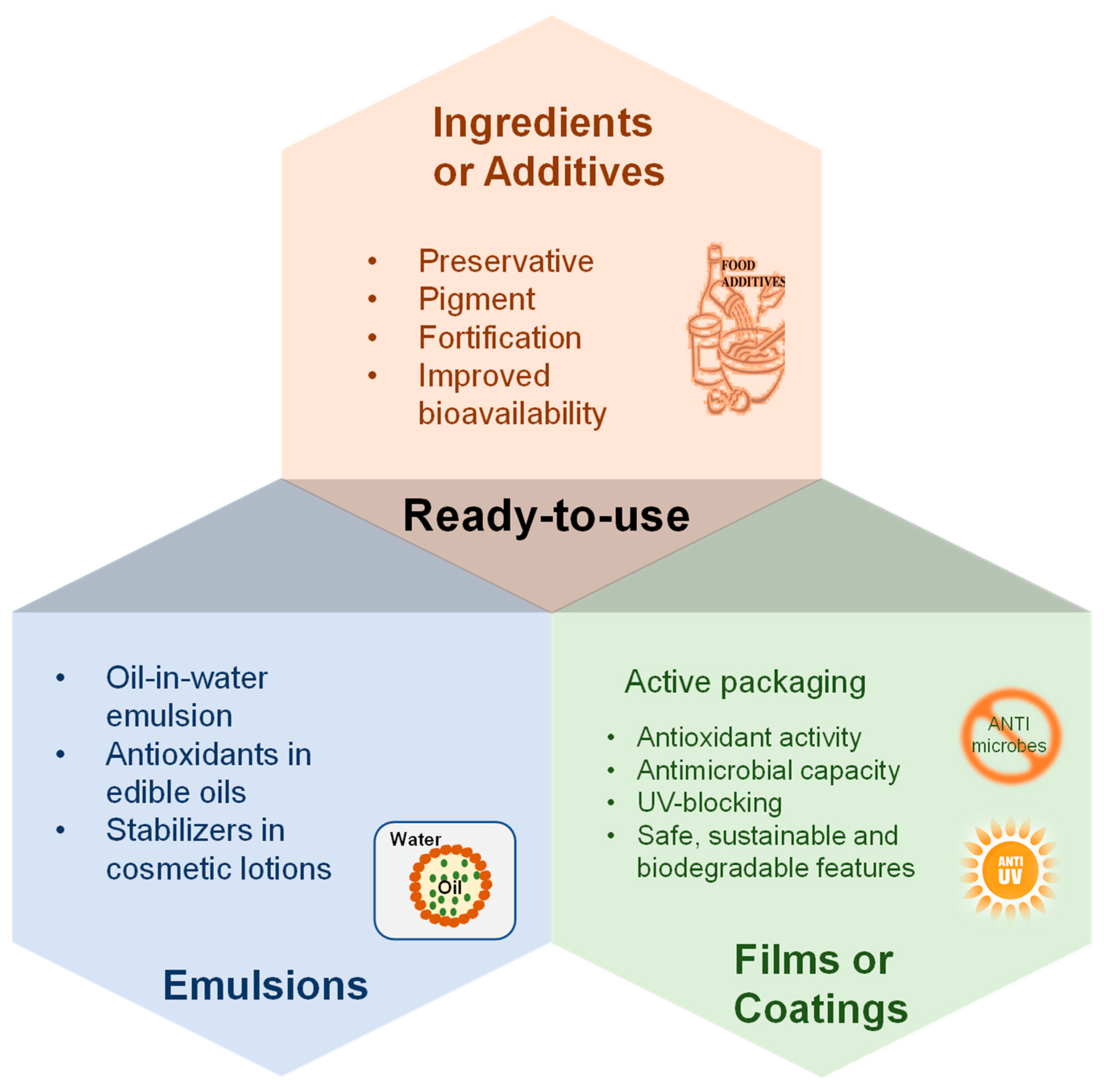
5.1. Ready-to-Use Ingredients or Additives for Products
5.2. Food and Cosmetic Emulsions
5.3. Active Packaging Films or Coatings
6. Concluding Remarks and Future Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- FAO. The State of Food and Agriculture 2019 Moving Forward on Food Loss and Waste Reduction; United Nations: Rome, Italy, 2019; ISBN 9789210046268. [Google Scholar]
- Brglez Mojzer, E.; Knez Hrncic, M.; Skerget, M.; Knez, Z.; Bren, U. Polyphenols: Extraction Methods, Antioxidative Action, Bioavailability and Anticarcinogenic Effects. Molecules 2016, 21, 901. [Google Scholar] [CrossRef]
- Ruesgas-Ramon, M.; Figueroa-Espinoza, M.C.; Durand, E. Application of Deep Eutectic Solvents (DES) for Phenolic Compounds Extraction: Overview, Challenges, and Opportunities. J. Agric. Food Chem. 2017, 65, 3591–3601. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Fakayode, O.A.; Ren, M.; Li, H.; Liang, J.; Zhou, C. Green and sustainable extraction of lignin by deep eutectic solvent, its antioxidant activity, and applications in the food industry. Crit. Rev. Food Sci. Nutr. 2023. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Hu, T.; Gu, J. Edge-cloud cooperation driven self-adaptive exception control method for the smart factory. Adv. Eng. Inform. 2022, 51, 101493. [Google Scholar] [CrossRef]
- Zhou, M.; Fakayode, O.A.; Ahmed Yagoub, A.E.; Ji, Q.; Zhou, C. Lignin fractionation from lignocellulosic biomass using deep eutectic solvents and its valorization. Renew. Sust. Energ. Rev. 2022, 156, 111986. [Google Scholar] [CrossRef]
- Misan, A.; Nadpal, J.; Stupar, A.; Pojic, M.; Mandic, A.; Verpoorte, R.; Choi, Y.H. The perspectives of natural deep eutectic solvents in agri-food sector. Crit. Rev. Food Sci. Nutr. 2020, 60, 2564–2592. [Google Scholar] [CrossRef]
- Benvenutti, L.; Zielinski, A.A.F.; Ferreira, S.R.S. Which is the best food emerging solvent: IL, DES or NADES? Trends Food. Sci. Technol. 2019, 90, 133–146. [Google Scholar] [CrossRef]
- Moni Bottu, H.; Mero, A.; Husanu, E.; Tavernier, S.; Pomelli, C.S.; Dewaele, A.; Bernaert, N.; Guazzelli, L.; Brennan, L. The ability of deep eutectic solvent systems to extract bioactive compounds from apple pomace. Food Chem. 2022, 386, 132717. [Google Scholar] [CrossRef]
- Rashid, R.; Mohd Wani, S.; Manzoor, S.; Masoodi, F.A.; Masarat Dar, M. Green extraction of bioactive compounds from apple pomace by ultrasound assisted natural deep eutectic solvent extraction: Optimisation, comparison and bioactivity. Food Chem. 2023, 398, 133871. [Google Scholar] [CrossRef]
- El Kantar, S.; Rajha, H.N.; Boussetta, N.; Vorobiev, E.; Maroun, R.G.; Louka, N. Green extraction of polyphenols from grapefruit peels using high voltage electrical discharges, deep eutectic solvents and aqueous glycerol. Food Chem. 2019, 295, 165–171. [Google Scholar] [CrossRef]
- Jokić, S.; Šafranko, S.; Jakovljević, M.; Cikoš, A.-M.; Kajić, N.; Kolarević, F.; Babić, J.; Molnar, M. Sustainable Green Procedure for Extraction of Hesperidin from Selected Croatian Mandarin Peels. Processes 2019, 7, 469. [Google Scholar] [CrossRef]
- Xu, M.; Ran, L.; Chen, N.; Fan, X.; Ren, D.; Yi, L. Polarity-dependent extraction of flavonoids from citrus peel waste using a tailor-made deep eutectic solvent. Food Chem. 2019, 297, 124970. [Google Scholar] [CrossRef]
- Kalogiouri, N.P.; Palaiologou, E.; Papadakis, E.N.; Makris, D.P.; Biliaderis, C.G.; Mourtzinos, I. Insights on the impact of deep eutectic solvents on the composition of the extracts from lemon (Citrus limon L.) peels analyzed by a novel RP-LC–QTOF-MS/MS method. Eur. Food Res. Technol. 2022, 248, 2913–2927. [Google Scholar] [CrossRef]
- Gomez-Urios, C.; Vinas-Ospino, A.; Puchades-Colera, P.; Lopez-Malo, D.; Frigola, A.; Esteve, M.J.; Blesa, J. Sustainable Development and Storage Stability of Orange By-Products Extract Using Natural Deep Eutectic Solvents. Foods 2022, 11, 2457. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, B.; Parkinson, C.; Gonzalez-Miquel, M. Extraction of polyphenolic antioxidants from orange peel waste using deep eutectic solvents. Sep. Purif. Technol. 2018, 206, 1–13. [Google Scholar] [CrossRef]
- Jeong, K.M.; Zhao, J.; Jin, Y.; Heo, S.R.; Han, S.Y.; da Yoo, E.; Lee, J. Highly efficient extraction of anthocyanins from grape skin using deep eutectic solvents as green and tunable media. Arch. Pharm. Res. 2015, 38, 2143–2152. [Google Scholar] [CrossRef] [PubMed]
- Cvjetko Bubalo, M.; Curko, N.; Tomasevic, M.; Kovacevic Ganic, K.; Radojcic Redovnikovic, I. Green extraction of grape skin phenolics by using deep eutectic solvents. Food Chem. 2016, 200, 159–166. [Google Scholar] [CrossRef]
- Radošević, K.; Ćurko, N.; Gaurina Srček, V.; Cvjetko Bubalo, M.; Tomašević, M.; Kovačević Ganić, K.; Radojčić Redovniković, I. Natural deep eutectic solvents as beneficial extractants for enhancement of plant extracts bioactivity. LWT-Food Sci. Technol. 2016, 73, 45–51. [Google Scholar] [CrossRef]
- Dabetić, N.; Todorović, V.; Panić, M.; Radojčić Redovniković, I.; Šobajić, S. Impact of Deep Eutectic Solvents on Extraction of Polyphenols from Grape Seeds and Skin. Appl. Sci. 2020, 10, 4830. [Google Scholar] [CrossRef]
- Panic, M.; Gunjevic, V.; Radosevic, K.; Cvjetko Bubalo, M.; Ganic, K.K.; Redovnikovic, I.R. COSMOtherm as an Effective Tool for Selection of Deep Eutectic Solvents Based Ready-To-Use Extracts from Grasevina Grape Pomace. Molecules 2021, 26, 4722. [Google Scholar] [CrossRef]
- Punzo, A.; Porru, E.; Silla, A.; Simoni, P.; Galletti, P.; Roda, A.; Tagliavini, E.; Samori, C.; Caliceti, C. Grape Pomace for Topical Application: Green NaDES Sustainable Extraction, Skin Permeation Studies, Antioxidant and Anti-Inflammatory Activities Characterization in 3D Human Keratinocytes. Biomolecules 2021, 11, 1181. [Google Scholar] [CrossRef] [PubMed]
- Panic, M.; Gunjevic, V.; Cravotto, G.; Radojcic Redovnikovic, I. Enabling technologies for the extraction of grape-pomace anthocyanins using natural deep eutectic solvents in up-to-half-litre batches extraction of grape-pomace anthocyanins using NADES. Food Chem. 2019, 300, 125185. [Google Scholar] [CrossRef] [PubMed]
- Panic, M.; Radic Stojkovic, M.; Kraljic, K.; Skevin, D.; Radojcic Redovnikovic, I.; Gaurina Srcek, V.; Radosevic, K. Ready-to-use green polyphenolic extracts from food by-products. Food Chem. 2019, 283, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Bosiljkov, T.; Dujmić, F.; Cvjetko Bubalo, M.; Hribar, J.; Vidrih, R.; Brnčić, M.; Zlatic, E.; Radojčić Redovniković, I.; Jokić, S. Natural deep eutectic solvents and ultrasound-assisted extraction: Green approaches for extraction of wine lees anthocyanins. Food Bioprod. Proces. 2017, 102, 195–203. [Google Scholar] [CrossRef]
- Benvenutti, L.; Zielinski, A.A.F.; Ferreira, S.R.S. Pressurized aqueous solutions of deep eutectic solvent (DES): A green emergent extraction of anthocyanins from a Brazilian berry processing by-product. Food Chem. X 2022, 13, 100236. [Google Scholar] [CrossRef]
- Benvenutti, L.; Sanchez-Camargo, A.D.P.; Zielinski, A.A.F.; Ferreira, S.R.S. NADES as potential solvents for anthocyanin and pectin extraction from Myrciaria cauliflora fruit by-product: In silico and experimental approaches for solvent selection. J. Mol. Liq. 2020, 315, 113761. [Google Scholar] [CrossRef]
- Grillo, G.; Gunjevic, V.; Radosevic, K.; Redovnikovic, I.R.; Cravotto, G. Deep Eutectic Solvents and Nonconventional Technologies for Blueberry-Peel Extraction: Kinetics, Anthocyanin Stability, and Antiproliferative Activity. Antioxidants 2020, 9, 1069. [Google Scholar] [CrossRef]
- Zhang, X.J.; Liu, Z.T.; Chen, X.Q.; Zhang, T.T.; Zhang, Y. Deep eutectic solvent combined with ultrasound technology: A promising integrated extraction strategy for anthocyanins and polyphenols from blueberry pomace. Food Chem. 2023, 422, 136224. [Google Scholar] [CrossRef]
- Fu, X.; Wang, D.; Belwal, T.; Xie, J.; Xu, Y.; Li, L.; Zou, L.; Zhang, L.; Luo, Z. Natural deep eutectic solvent enhanced pulse-ultrasonication assisted extraction as a multi-stability protective and efficient green strategy to extract anthocyanin from blueberry pomace. LWT-Food Sci. Technol. 2021, 144, 111220. [Google Scholar] [CrossRef]
- Xue, H.; Tan, J.; Li, Q.; Tang, J.; Cai, X. Ultrasound-Assisted Deep Eutectic Solvent Extraction of Anthocyanins from Blueberry Wine Residues: Optimization, Identification, and HepG2 Antitumor Activity. Molecules 2020, 25, 5456. [Google Scholar] [CrossRef]
- Alrugaibah, M.; Yagiz, Y.; Gu, L. Use natural deep eutectic solvents as efficient green reagents to extract procyanidins and anthocyanins from cranberry pomace and predictive modeling by RSM and artificial neural networking. Sep. Purif. Technol. 2021, 255, 117720. [Google Scholar] [CrossRef]
- Teslic, N.; Santos, F.; Oliveira, F.; Stupar, A.; Pojic, M.; Mandic, A.; Pavlic, B.; Kljakic, A.C.; Duarte, A.R.C.; Paiva, A.; et al. Simultaneous Hydrolysis of Ellagitannins and Extraction of Ellagic Acid from Defatted Raspberry Seeds Using Natural Deep Eutectic Solvents (NADES). Antioxidants 2022, 11, 254. [Google Scholar] [CrossRef]
- Vázquez-González, M.; Fernández-Prior, Á.; Bermúdez Oria, A.; Rodríguez-Juan, E.M.; Pérez-Rubio, A.G.; Fernández-Bolaños, J.; Rodríguez-Gutiérrez, G. Utilization of strawberry and raspberry waste for the extraction of bioactive compounds by deep eutectic solvents. LWT-Food Sci. Technol. 2020, 130, 109645. [Google Scholar] [CrossRef]
- Zhou, P.; Wang, X.; Liu, P.; Huang, J.; Wang, C.; Pan, M.; Kuang, Z. Enhanced phenolic compounds extraction from Morus alba L. leaves by deep eutectic solvents combined with ultrasonic-assisted extraction. Ind. Crop. Prod. 2018, 120, 147–154. [Google Scholar] [CrossRef]
- Gao, M.-Z.; Cui, Q.; Wang, L.-T.; Meng, Y.; Yu, L.; Li, Y.-Y.; Fu, Y.-J. A green and integrated strategy for enhanced phenolic compounds extraction from mulberry (Morus alba L.) leaves by deep eutectic solvent. Microchem. J. 2020, 154, 104598. [Google Scholar] [CrossRef]
- Hernández-Corroto, E.; Boussetta, N.; Marina, M.L.; García, M.C.; Vorobiev, E. High voltage electrical discharges followed by deep eutectic solvents extraction for the valorization of pomegranate seeds (Punica granatum L.). Innov. Food Sci. Emerg. 2022, 79, 103055. [Google Scholar] [CrossRef]
- Rajha, H.N.; Mhanna, T.; El Kantar, S.; El Khoury, A.; Louka, N.; Maroun, R.G. Innovative process of polyphenol recovery from pomegranate peels by combining green deep eutectic solvents and a new infrared technology. LWT-Food Sci. Technol. 2019, 111, 138–146. [Google Scholar] [CrossRef]
- Kim, H.J.; Yoon, K.Y. Optimization of ultrasound-assisted deep eutectic solvent extraction of bioactive compounds from pomegranate peel using response surface methodology. Food Sci. Biotechnol. 2023. [Google Scholar] [CrossRef]
- Bertolo, M.R.V.; Martins, V.C.A.; Plepis, A.M.G.; Bogusz, S. Utilization of pomegranate peel waste: Natural deep eutectic solvents as a green strategy to recover valuable phenolic compounds. J. Clean. Prod. 2021, 327, 129471. [Google Scholar] [CrossRef]
- Kyriakidou, A.; Makris, D.P.; Lazaridou, A.; Biliaderis, C.G.; Mourtzinos, I. Physical Properties of Chitosan Films Containing Pomegranate Peel Extracts Obtained by Deep Eutectic Solvents. Foods 2021, 10, 1262. [Google Scholar] [CrossRef]
- Pal, C.B.T.; Jadeja, G.C. Microwave-assisted extraction for recovery of polyphenolic antioxidants from ripe mango (Mangifera indica L.) peel using lactic acid/sodium acetate deep eutectic mixtures. Food Sci. Technol. Int. 2020, 26, 78–92. [Google Scholar] [CrossRef] [PubMed]
- Plaza, M.; Domínguez-Rodríguez, G.; Sahelices, C.; Marina, M.L. A Sustainable Approach for Extracting Non-Extractable Phenolic Compounds from Mangosteen Peel Using Ultrasound-Assisted Extraction and Natural Deep Eutectic Solvents. Appl. Sci. 2021, 11, 5625. [Google Scholar] [CrossRef]
- Fernandez, M.L.A.; Espino, M.; Gomez, F.J.V.; Silva, M.F. Novel approaches mediated by tailor-made green solvents for the extraction of phenolic compounds from agro-food industrial by-products. Food Chem. 2018, 239, 671–678. [Google Scholar] [CrossRef]
- Lee, J.W.; Park, H.Y.; Park, J. Enhanced Extraction Efficiency of Flavonoids from Pyrus ussuriensis Leaves with Deep Eutectic Solvents. Molecules 2022, 27, 2798. [Google Scholar] [CrossRef] [PubMed]
- Şahin, S.; Bilgin, M. Valorization of peach (Prunus persica L.) waste into speciality products via green methods. Biomass Convers. Bior. 2021, 12, 123–132. [Google Scholar] [CrossRef]
- Kurtulbaş, E. Prediction of mass transfer and kinetic behavior during the extraction of high added-value products from sour cherry (Prunus cerasus L.) peels. J. Food Process. Pres. 2022, 46, e16401. [Google Scholar] [CrossRef]
- Popovic, B.M.; Micic, N.; Potkonjak, A.; Blagojevic, B.; Pavlovic, K.; Milanov, D.; Juric, T. Novel extraction of polyphenols from sour cherry pomace using natural deep eutectic solvents—Ultrafast microwave-assisted NADES preparation and extraction. Food Chem. 2022, 366, 130562. [Google Scholar] [CrossRef]
- Cui, Q.; Liu, J.-Z.; Wang, L.-T.; Kang, Y.-F.; Meng, Y.; Jiao, J.; Fu, Y.-J. Sustainable deep eutectic solvents preparation and their efficiency in extraction and enrichment of main bioactive flavonoids from sea buckthorn leaves. J. Clean. Prod. 2018, 184, 826–835. [Google Scholar] [CrossRef]
- Leal, F.C.; Farias, F.O.; do Amaral, W.; Toci, A.T.; Mafra, M.R.; Igarashi-Mafra, L. Green Solvents to Value Annona muricata L. Leaves as Antioxidants Source: Process Optimization and Potential as a Natural Food Additive. Waste Biomass Valori. 2021, 13, 1233–1241. [Google Scholar] [CrossRef]
- Kehili, M.; Isci, A.; Thieme, N.; Kaltschmitt, M.; Zetzl, C.; Smirnova, I. Microwave-assisted deep eutectic solvent extraction of phenolics from defatted date seeds and its effect on solubilization of carbohydrates. Biomass Convers. Bior. 2022. [Google Scholar] [CrossRef]
- Li, J.; Chen, W.; Niu, D.; Wang, R.; Xu, F.-Y.; Chen, B.-R.; Lin, J.-W.; Tang, Z.-S.; Zeng, X.-A. Efficient and green strategy based on pulsed electric field coupled with deep eutectic solvents for recovering flavonoids and preparing flavonoid aglycones from noni-processing wastes. J. Clean. Prod. 2022, 368, 133019. [Google Scholar] [CrossRef]
- Hu, T.; Wang, W.; Gu, J.; Xia, Z.; Zhang, J.; Wang, B. Research on Apple Object Detection and Localization Method Based on Improved YOLOX and RGB-D Images. Agronomy 2023, 13, 1816. [Google Scholar] [CrossRef]
- Panzella, L.; Moccia, F.; Nasti, R.; Marzorati, S.; Verotta, L.; Napolitano, A. Bioactive Phenolic Compounds from Agri-Food Wastes: An Update on Green and Sustainable Extraction Methodologies. Front. Nutr. 2020, 7, 60. [Google Scholar] [CrossRef]
- Pal, C.B.T.; Jadeja, G.C. Microwave-assisted deep eutectic solvent extraction of phenolic antioxidants from onion (Allium cepa L.) peel: A Box-Behnken design approach for optimization. J. Food Sci. Technol. 2019, 56, 4211–4223. [Google Scholar] [CrossRef]
- Shang, X.-c.; Zhang, Y.-q.; Zheng, Y.-f.; Li, Y. Temperature-responsive deep eutectic solvents as eco-friendly and recyclable media for microwave extraction of flavonoid compounds from waste onion (Allium cepa L.) skins. Biomass Convers. Bior. 2022. [Google Scholar] [CrossRef]
- Sukor, N.F.; Selvam, V.P.; Jusoh, R.; Kamarudin, N.S.; Rahim, S.A. Intensified DES mediated ultrasound extraction of tannic acid from onion peel. J. Food Eng. 2021, 296, 110437. [Google Scholar] [CrossRef]
- Stefou, I.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Development of sodium propionate-based deep eutectic solvents for polyphenol extraction from onion solid wastes. Clean Technol. Envir. 2019, 21, 1563–1574. [Google Scholar] [CrossRef]
- Pal, C.B.T.; Jadeja, G.C. Deep eutectic solvent-based extraction of polyphenolic antioxidants from onion (Allium cepa L.) peel. J. Sci. Food Agric. 2019, 99, 1969–1979. [Google Scholar] [CrossRef]
- Ciardi, M.; Ianni, F.; Sardella, R.; Di Bona, S.; Cossignani, L.; Germani, R.; Tiecco, M.; Clementi, C. Effective and Selective Extraction of Quercetin from Onion (Allium cepa L.) Skin Waste Using Water Dilutions of Acid-Based Deep Eutectic Solvents. Materials 2021, 14, 6465. [Google Scholar] [CrossRef]
- Vasyliev, G.; Lyudmyla, K.; Hladun, K.; Skiba, M.; Vorobyova, V. Valorization of tomato pomace: Extraction of value-added components by deep eutectic solvents and their application in the formulation of cosmetic emulsions. Biomass Convers. Bior. 2022, 12 (Suppl. S1), 95–111. [Google Scholar] [CrossRef]
- Grillo, G.; Tabasso, S.; Capaldi, G.; Radosevic, K.; Radojcic-Redovnikovic, I.; Gunjevic, V.; Calcio Gaudino, E.; Cravotto, G. Food-Waste Valorisation: Synergistic Effects of Enabling Technologies and Eutectic Solvents on the Recovery of Bioactives from Violet Potato Peels. Foods 2023, 12, 2214. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Liang, Y.N.; Stuckey, D.C.; Hu, X. Single-step extraction of bioactive compounds from cruciferous vegetable (kale) waste using natural deep eutectic solvents. Sep. Purif. Technol. 2023, 317, 123677. [Google Scholar] [CrossRef]
- Zannou, O.; Pashazadeh, H.; Ghellam, M.; Ali Redha, A.; Koca, I. Enhanced ultrasonically assisted extraction of bitter melon (Momordica charantia) leaf phenolic compounds using choline chloride-acetic acid–based natural deep eutectic solvent: An optimization approach and in vitro digestion. Biomass Convers. Bior. 2022. [Google Scholar] [CrossRef]
- Aviles-Betanzos, K.A.; Oney-Montalvo, J.E.; Cauich-Rodriguez, J.V.; Gonzalez-Avila, M.; Scampicchio, M.; Morozova, K.; Ramirez-Sucre, M.O.; Rodriguez-Buenfil, I.M. Antioxidant Capacity, Vitamin C and Polyphenol Profile Evaluation of a Capsicum chinense By-Product Extract Obtained by Ultrasound Using Eutectic Solvent. Plants 2022, 11, 2060. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Yan, Z.; Li, L.; Zhang, L.; Zhao, M.; Yi, H.; Wang, Z.; Li, G.; Wang, Z.; Li, M.; et al. Green Extraction of Phenolic Compounds from Lotus (Nelumbo nucifera Gaertn) Leaf Using Deep Eutectic Solvents: Process Optimization and Antioxidant Activity. Separations 2023, 10, 272. [Google Scholar] [CrossRef]
- González-Rivera, J.; Mero, A.; Husanu, E.; Mezzetta, A.; Ferrari, C.; D’Andrea, F.; Bramanti, E.; Pomelli, C.S.; Guazzelli, L. Combining acid-based deep eutectic solvents and microwave irradiation for improved chestnut shell waste valorization. Green Chem. 2021, 23, 10101–10115. [Google Scholar] [CrossRef]
- An, J.-Y.; Wang, L.-T.; Lv, M.-J.; Wang, J.-D.; Cai, Z.-H.; Wang, Y.-Q.; Zhang, S.; Yang, Q.; Fu, Y.-J. An efficiency strategy for extraction and recovery of ellagic acid from waste chestnut shell and its biological activity evaluation. Microchem. J. 2021, 160, 105616. [Google Scholar] [CrossRef]
- Fu, X.; Wang, D.; Belwal, T.; Xu, Y.; Li, L.; Luo, Z. Sonication-synergistic natural deep eutectic solvent as a green and efficient approach for extraction of phenolic compounds from peels of Carya cathayensis Sarg. Food Chem. 2021, 355, 129577. [Google Scholar] [CrossRef]
- Fu, X.; Belwal, T.; He, Y.; Xu, Y.; Li, L.; Luo, Z. UPLC-Triple-TOF/MS characterization of phenolic constituents and the influence of natural deep eutectic solvents on extraction of Carya cathayensis Sarg. peels: Composition, extraction mechanism and in vitro biological activities. Food Chem. 2022, 370, 131042. [Google Scholar] [CrossRef]
- Bener, M.; Sen, F.B.; Onem, A.N.; Bekdeser, B.; Celik, S.E.; Lalikoglu, M.; Asci, Y.S.; Capanoglu, E.; Apak, R. Microwave-assisted extraction of antioxidant compounds from by-products of Turkish hazelnut (Corylus avellana L.) using natural deep eutectic solvents: Modeling, optimization and phenolic characterization. Food Chem. 2022, 385, 132633. [Google Scholar] [CrossRef]
- Lopez-Linares, J.C.; Campillo, V.; Coca, M.; Lucas, S.; Garcia-Cubero, M.T. Microwave-assisted deep eutectic solvent extraction of phenolic compounds from brewer’s spent grain. J. Chem. Technol. Biot. 2021, 96, 481–490. [Google Scholar] [CrossRef]
- Mouratoglou, E.; Malliou, V.; Makris, D.P. Novel Glycerol-Based Natural Eutectic Mixtures and Their Efficiency in the Ultrasound-Assisted Extraction of Antioxidant Polyphenols from Agri-Food Waste Biomass. Waste Biomass Valori. 2016, 7, 1377–1387. [Google Scholar] [CrossRef]
- Thakur, R.; Gupta, V.; Dhar, P.; Deka, S.C.; Das, A.B. Ultrasound-assisted extraction of anthocyanin from black rice bran using natural deep eutectic solvents: Optimization, diffusivity, and stability. J. Food Process. Pres. 2022, 46, e16309. [Google Scholar] [CrossRef]
- Huang, Y.; Feng, F.; Jiang, J.; Qiao, Y.; Wu, T.; Voglmeir, J.; Chen, Z.G. Green and efficient extraction of rutin from tartary buckwheat hull by using natural deep eutectic solvents. Food Chem. 2017, 221, 1400–1405. [Google Scholar] [CrossRef]
- Zheng, B.; Yuan, Y.; Xiang, J.; Jin, W.; Johnson, J.B.; Li, Z.; Wang, C.; Luo, D. Green extraction of phenolic compounds from foxtail millet bran by ultrasonic-assisted deep eutectic solvent extraction: Optimization, comparison and bioactivities. LWT-Food Sci. Technol. 2022, 154, 112740. [Google Scholar] [CrossRef]
- Santos, M.C.B.; Barouh, N.; Baréa, B.; Villeneuve, P.; Bourlieu-Lacanal, C.; Ferreira, M.S.L.; Durand, E. Sequential one-pot NaDES assisted extraction and biotransformation of rice bran: A new strategy to boost antioxidant activity of natural extracts. Process Biochem. 2022, 117, 110–116. [Google Scholar] [CrossRef]
- Wu, J.; Su, M.; Hu, A.; Wang, H. Extraction and recovery of chlorogenic acid from sunflower disks using a high-efficiency system composed of deep eutectic solvents and macroporous resins. J. Food Process. Pres. 2022, 46, e16856. [Google Scholar] [CrossRef]
- Balaraman, H.B.; Sivasubramaniyam, A.; Rathnasamy, S.K. High selective purification of Quercetin from Peanut hull using protic deep eutectic mixture based liquid–liquid microextraction. Microchem. J. 2020, 152, 104444. [Google Scholar] [CrossRef]
- Chanioti, S.; Tzia, C. Extraction of phenolic compounds from olive pomace by using natural deep eutectic solvents and innovative extraction techniques. Innov. Food Sci. Emerg. 2018, 48, 228–239. [Google Scholar] [CrossRef]
- Pontes, P.V.d.A.; Czaikoski, A.; Almeida, N.A.; Fraga, S.; Rocha, L.d.O.; Cunha, R.L.; Maximo, G.J.; Batista, E.A.C. Extraction optimization, biological activities, and application in O/W emulsion of deep eutectic solvents-based phenolic extracts from olive pomace. Food Res. Int. 2022, 161, 111753. [Google Scholar] [CrossRef]
- Duru, K.C.; Slesarev, G.P.; Aboushanab, S.A.; Kovalev, I.S.; Zeidler, D.M.; Kovaleva, E.G.; Bhat, R. An eco-friendly approach to enhance the extraction and recovery efficiency of isoflavones from kudzu roots and soy molasses wastes using ultrasound-assisted extraction with natural deep eutectic solvents (NADES). Ind. Crop. Prod. 2022, 182, 114886. [Google Scholar] [CrossRef]
- Wang, W.; Zhang, Y.; Gu, J.; Wang, J. A Proactive Manufacturing Resources Assignment Method Based on Production Performance Prediction for the Smart Factory. IEEE T. Ind. Inform. 2022, 18, 46–55. [Google Scholar] [CrossRef]
- Athanasiadis, V.; Grigorakis, S.; Lalas, S.; Makris, D.P. Highly Efficient Extraction of Antioxidant Polyphenols from Olea europaea Leaves Using an Eco-friendly Glycerol/Glycine Deep Eutectic Solvent. Waste Biomass Valori. 2018, 9, 1985–1992. [Google Scholar] [CrossRef]
- Unlu, A.E. Green and Non-conventional Extraction of Bioactive Compounds from Olive Leaves: Screening of Novel Natural Deep Eutectic Solvents and Investigation of Process Parameters. Waste Biomass Valori. 2021, 12, 5329–5346. [Google Scholar] [CrossRef] [PubMed]
- de Almeida Pontes, P.V.; Ayumi Shiwaku, I.; Maximo, G.J.; Caldas Batista, E.A. Choline chloride-based deep eutectic solvents as potential solvent for extraction of phenolic compounds from olive leaves: Extraction optimization and solvent characterization. Food Chem. 2021, 352, 129346. [Google Scholar] [CrossRef]
- Zurob, E.; Cabezas, R.; Villarroel, E.; Rosas, N.; Merlet, G.; Quijada-Maldonado, E.; Romero, J.; Plaza, A. Design of natural deep eutectic solvents for the ultrasound-assisted extraction of hydroxytyrosol from olive leaves supported by COSMO-RS. Sep. Purif. Technol. 2020, 248, 117054. [Google Scholar] [CrossRef]
- Mir-Cerda, A.; Granados, M.; Saurina, J.; Sentellas, S. Green Extraction of Antioxidant Compounds from Olive Tree Leaves Based on Natural Deep Eutectic Solvents. Antioxidants 2023, 12, 995. [Google Scholar] [CrossRef]
- Plaza, A.; Tapia, X.; Yañez, C.; Vilches, F.; Candia, O.; Cabezas, R.; Romero, J. Obtaining Hydroxytyrosol from Olive Mill Waste Using Deep Eutectic Solvents and Then Supercritical CO2. Waste Biomass Valori. 2020, 11, 6273–6284. [Google Scholar] [CrossRef]
- da Silva, M.R.; Jelley, R.E.; Carneiro, R.L.; Fedrizzi, B.; Weber, C.C.; Funari, C.S. Green solvents for the selective extraction of bioactive compounds from by-products of the coffee production chain. Innov. Food Sci. Emerg. 2023, 86, 103365. [Google Scholar] [CrossRef]
- Manuela, P.; Drakula, S.; Cravotto, G.; Verpoorte, R.; Hruškar, M.; Radojčić Redovniković, I.; Radošević, K. Biological activity and sensory evaluation of cocoa by-products NADES extracts used in food fortification. Innov. Food Sci. Emerg. 2020, 66, 102514. [Google Scholar] [CrossRef]
- Ruesgas-Ramon, M.; Suarez-Quiroz, M.L.; Gonzalez-Rios, O.; Barea, B.; Cazals, G.; Figueroa-Espinoza, M.C.; Durand, E. Biomolecules extraction from coffee and cocoa by- and co-products using deep eutectic solvents. J. Sci. Food Agric. 2020, 100, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Fanali, C.; Della Posta, S.; Dugo, L.; Gentili, A.; Mondello, L.; De Gara, L. Choline-chloride and betaine-based deep eutectic solvents for green extraction of nutraceutical compounds from spent coffee ground. J. Pharm. Biomed. Anal. 2020, 189, 113421. [Google Scholar] [CrossRef] [PubMed]
- García-Roldán, A.; Piriou, L.; Jauregi, P. Natural deep eutectic solvents as a green extraction of polyphenols from spent coffee ground with enhanced bioactivities. Front. Plant Sci. 2023, 13, 1072592. [Google Scholar] [CrossRef] [PubMed]
- Grudniewska, A.; Popłoński, J. Simple and green method for the extraction of xanthohumol from spent hops using deep eutectic solvents. Sep. Purif. Technol. 2020, 250, 117196. [Google Scholar] [CrossRef]
- Lu, W.; Liu, S. Choline chloride–based deep eutectic solvents (Ch-DESs) as promising green solvents for phenolic compounds extraction from bioresources: State-of-the-art, prospects, and challenges. Biomass Convers. Bior. 2020, 12, 2949–2962. [Google Scholar] [CrossRef]
- da Silva, D.T.; Smaniotto, F.A.; Costa, I.F.; Baranzelli, J.; Muller, A.; Somacal, S.; Monteiro, C.S.; Vizzotto, M.; Rodrigues, E.; Barcia, M.T.; et al. Natural deep eutectic solvent (NADES): A strategy to improve the bioavailability of blueberry phenolic compounds in a ready-to-use extract. Food Chem. 2021, 364, 130370. [Google Scholar] [CrossRef]
- Faggian, M.; Sut, S.; Perissutti, B.; Baldan, V.; Grabnar, I.; Dall’Acqua, S. Natural Deep Eutectic Solvents (NADES) as a Tool for Bioavailability Improvement: Pharmacokinetics of Rutin Dissolved in Proline/Glycine after Oral Administration in Rats: Possible Application in Nutraceuticals. Molecules 2016, 21, 1531. [Google Scholar] [CrossRef]
- Thakur, R.; Gupta, V.; Ghosh, T.; Das, A.B. Effect of anthocyanin-natural deep eutectic solvent (lactic acid/fructose) on mechanical, thermal, barrier, and pH-sensitive properties of polyvinyl alcohol based edible films. Food Packag. Shelf 2022, 33, 100914. [Google Scholar] [CrossRef]
- Boiteux, J.; Espino, M.; Azcarate, S.; Silva, M.F.; Gomez, F.J.V.; Pizzuolo, P.; Fernandez, M.L.A. NADES blend for bioactive coating design as a sustainable strategy for postharvest control. Food Chem. 2023, 406, 135054. [Google Scholar] [CrossRef]
- Braham, F.; Amaral, L.; Biernacki, K.; Carvalho, D.O.; Guido, L.F.; Magalhaes, J.; Zaidi, F.; Souza, H.K.S.; Goncalves, M.P. Phenolic Extraction of Moringa oleifera Leaves in DES: Characterization of the Extracts and Their Application in Methylcellulose Films for Food Packaging. Foods 2022, 11, 2641. [Google Scholar] [CrossRef]
- Zhao, P.; Wang, J.; Yan, X.; Cai, Z.; Fu, L.; Gu, Q.; Liu, L.; Jin, H.; Fu, Y. Functional chitosan/zein films with Rosa roxburghii Tratt leaves extracts prepared by natural deep eutectic solvents. Food Packag. Shelf 2022, 34, 101001. [Google Scholar] [CrossRef]
- Mostafa, H.; Airouyuwaa, J.O.; Hamed, F.; Wang, Y.; Maqsood, S. Structural, mechanical, antioxidant and antibacterial properties of soy protein isolate (SPI)-based edible food packaging films as influenced by nanocellulose (NC) and green extracted phenolic compounds from date palm leaves. Food Packag. Shelf 2023, 38, 101124. [Google Scholar] [CrossRef]
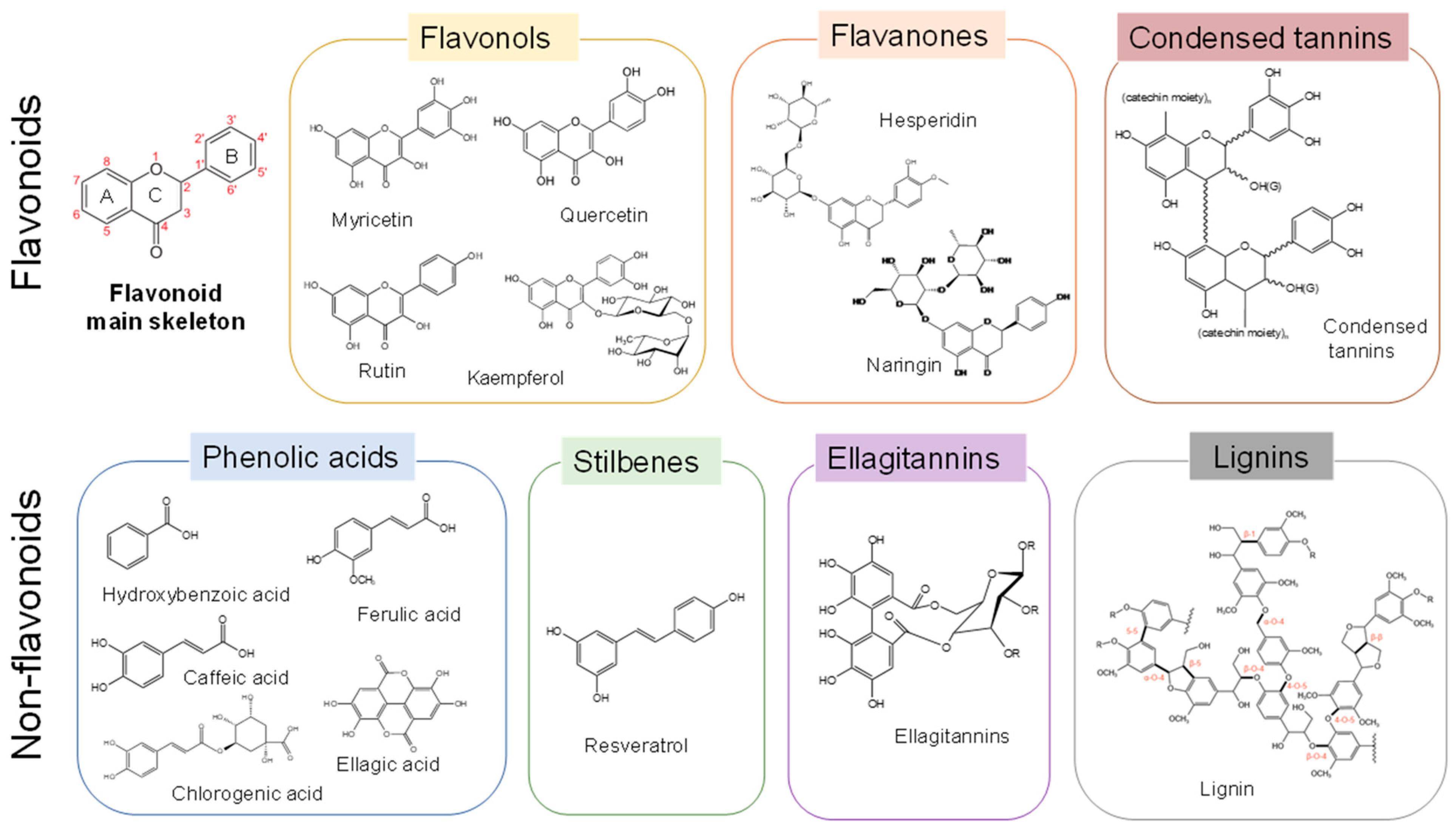


| Fruit By-Products | Optimal DESs/NADESs | Value-Added Compounds | Assisted Techniques and Optimal Conditions | Yields (mg/g) | Reference |
|---|---|---|---|---|---|
| Apple pomace | ChCl:ethylene glycol (1:4) | Polyphenols (Procyanidin B2, chlorogenic acid, epicatechin hydrate, vanillin, and phloridzin) | ~ | ~ | [9] |
| ChCl:glycerol (1:2) | Polyphenols (Quercetin, chlorogenic acid, gallic acid, phloretin, phloridizin, and rutin) | Ultrasound (acoustic intensity: 83.2 W/cm2, duty cycle: 75%) | TPC (GAE): 5.8 | [10] | |
| Citrus fruits industry by-products (orange, grapefruit, mandarin, lemon, pomelo peels) | Lactic acid:glucose (5:1) | Polyphenols (naringin) | HVED (Energy: 7.27~218 kJ/kg, 40 kV, 10 kA, 0.5 Hz, 160 J/pulse) | 1.864 | [11] |
| ChCl:citric acid (1:1) | Hesperidin | ~ | 112 | [12] | |
| ChCl:levulinic acid:N-methyl urea (1:1.2:0.8) | Polymethoxylated flavonoids and glycosides of flavonoids | Ultrasound (200 W, 35 kHz) | 18.8, 47.1 | [13] | |
| ChCl:glycerol (1:3) | Polyphenols (quercetin, p-coumaric acid) | ~ | TPC (GAE): 24.2 | [14] | |
| Lactic acid:glucose (5:1), L-proline:malic acid (1:1) | Polyphenols and flavonoids | ~ | TPC (GAE): 21.6, TFC (ECE): 0.97 | [15] | |
| ChCl:ethylene glycol (1:4) | Polyphenols (p-coumaric acid and gallic acid) | ~ | TPC (GAE): 5.84 | [16] | |
| Grape solid wastes (stem, skin, seed, pomace, and lee) | Citric acid:maltose (4:1) | Anthocyanins | Ultrasound | TAC: 63.36 | [17] |
| ChCl:oxalic acid (1:2) | Polyphenols (catechin and quercetin-3-Oglucoside) | Microwave and ultrasound | ~ | [18] | |
| ChCl:malic acid (1:1) | Polyphenols and anthocyanins (malvidin-3-O-monoglucoside) | Ultrasound | TPC (GAE): 91, TAC (C3GE): 24 | [19] | |
| ChCl:citric acid (1:2) | Flavan-3-ols, catechin, epicatechin, protocatechuic acid | Ultrasound (59 kHz) | TPC (GAE): 146.69 | [20] | |
| Betaine:Glucose (1:1) | Polyphenols (flavan-3-ols) | Ultrasound (100 W) | ~ | [21] | |
| Betaine:citric acid (1:1) | Malvidin | ~ | 56.66 µg/mL | [22] | |
| ChCl:citric acid (1:2) | Anthocyanins (malvidin-3-O-monoglucoside) | SUMAE (microwave 300 W + ultrasound 50 W) | TAC (M3GE): 1.77 | [23] | |
| Polyphenols (anthocyanins, gallic acid, catechin and quercetin-3-O-glucoside) | TPC (GAE): 2.89 | [24] | |||
| ChCl:malic acid (1:1) | Anthocyanins (anthocyanin-3-O-monoglucosides and anthocyanin-3-(6-O-p-coumaroyl)monoglucosides) | Ultrasound (341.5 W, 37 kHz) | TAC (M3GE): 6.55 | [25] | |
| Blueberry processing wastes | ChCl:malic acid (1:1) | Anthocyanin | ~ | TAC (C3GE): 173 | [26] |
| ChCl:propylene glycol (1:2) | Anthocyanin | ~ | ~ | [27] | |
| ChCl:lactic acid (1:1) | Anthocyanins | Ultrasound and microwave (500 W, 15 min; 500 W, 20 kHz, 40 °C, 30 min) | TAC: 25.83 and 21.18 | [28] | |
| ChCl:1,4-butanediol (1:3) | Polyphenols and Anthocyanins | Ultrasound (200 W, 63 °C, 20 min) | TPC (GAE): 41.6, TAC (C3GE): 11.40 | [29] | |
| ChCl:oxlaic acid (1:1) | Anthocyanins | Ultrasound (325 W, 76 °C, 20 kHz, 3.2 min) | TAC (C3GE): 24.3 | [30] | |
| ChCl:1,4-butanediol (1:3) | Anthocyanins (cyanidin-3-rutinoside) | Ultrasound (380 W, 37 kHz, 55 °C, 40 min) | TAC (C3GE): 9.32 | [31] | |
| Cranberry, strawberry, and raspberry extrudate waste | ChCl:betaine hydrochloride: levulinic acid (1:1:2) | Procyanidins and anthocyanins | Ultrasound | TAC: 1.30 | [32] |
| Citric acid:betaine (1:1) | Ellagic Acid | Ultrasound | TPC (GAE): 44.8 | [33] | |
| ChCl:glycolic acid:oxalic acid (1:1.7:0.3) | Anthocyanins | ~ | ~ | [34] | |
| Mulberry (Morus alba L.) leaves | ChCl:citric acid (2:1) | Chlorogenic acid | Ultrasound (600 W, 25 kHz) | 22.7 | [35] |
| ChCl:glycerol (1:2) | Neochlorogenic acid, chlorogenic acid, cryptochlorogenic acid, caffeic acid, rutin, isoquercetin, astragalin | Microwave (600 W) | 0.240, 4.51, 0.342, 0.286, 1.47, 0.971 and 0.538 | [36] | |
| Pomegranate waste (peel, mesocarp, and seeds) | ChCl:acetic acid (1:1) | Polyphenols (mainly ellagitannins) | HVED (Energy: 7.27~218 kJ/kg, 40 kV, 10 kA, 0.5 Hz, 160 J/pulse, electrical pulses: 10~800) | TPC (GAE): 9.5 | [37] |
| Eight different DESs | Polyphenols (tannin) | Infrared (70~170 W) | TPC (GAE): 152 | [38] | |
| ChCl:urea (1:2) | Punicalagin and ellagic acid | Ultrasound (120 W and 300 W, 20 min), | 130.65 and 2.04 | [39] | |
| ChCl:lactic acid (1:1) | Polyphenols | Ultrasound | TPC (GAE): 6.4 mg/mL | [40] | |
| ChCl:glycerol (1:11) | Polyphenols and flavonoids | ~ | TPC (GAE): 273, TFC (QE): 20.1 | [41] | |
| Mango peel | Sodium acetate:lactic acid (1:3) | Polyphenols (mangiferin) | Microwave (436.45 W) | TPC (GAE): 56.17 | [42] |
| Mangosteen peel | ChCl:lactic acid (1:2) | Anthocyanins | Ultrasound (amplitude: 60%) | TAC (ECE): 2.38 | [43] |
| Pear industrial by-products | Lactic acid:glucose (5:1) | Gallic acid and rutin | Ultrasound (20 kHz, 200 W) | ~ | [44] |
| ChCl:glutaric acid (1:1) | Rutin, hyperoside, and isoquercitrin | ~ | ~ | [45] | |
| Peach peels | Lactic acid:glycerol (1:1) | Polyphenols and chlorogenic acid) | ~ | TPC (GAE): 10.5 and 2.70 | [46] |
| Sour cherry pomace | Citric acid:ethylene glycol (1:4) | Polyphenols | Microwave (500 W) | TPC (GAE): 33.61 | [47] |
| ChCl:malic acid (1:1) | Neochlorogenic acid | Ultrasound and microwave (37 kHz, 30 W, 50 °C; 180 W within 30 s) | 0.145 and 0.178 | [48] | |
| Sea buckthorn leaves | ChCl:1,4-butanediol (1:3) | Rutin, quercetin-3-O-glucoside, quercetin, kaempferol, isorhamnetin | Microwave (600 W) | 8.99, 1.81, 9.11, 0.45, 0.49 | [49] |
| Annona muricata L. Leaves | Linium chloride:glycerol (1:2), Linium chloride:xylitol (1:2) | Rutin and catechin | Ultrasound | ~ | [50] |
| Date palm seeds | ChCl:formic acid (1:2) | Polyphenols | Microwave (400 W) | TPC (GAE): 128 | [51] |
| Noni-processing waste | ChCl:oxalic acid (1:2) | Rutin and quercetin | PEF (field strength: 0.64–1.84 kV/cm), number of pulses: 25–125) | 16.21 and 19.85 | [52] |
| Vegetable By-Products | Optimal DESs/NADESs | Value-Added Compounds | Assisted Techniques and Optimal Conditions | Yields (mg/g) | Reference |
|---|---|---|---|---|---|
| Onion solid wastes | ChCl:urea: H2O (1:2:4) | Polyphenols (Quercetin, kaempferol, and myricetin) | Microwave (100 W) | TPC (GAE): 80.5 | [55] |
| ChCl:urea (1:2) | Microwave (300 W) | TPC (GAE): 223 | [59] | ||
| Temperature-responsive DESs | Flavonoids (Quercetin, kaempferol, luteolin, and quercetin-3-O-β-D-glucoside) | Microwave (554 W) | 47.83 | [56] | |
| ChCl:urea (1:1) | Tannic acid | Ultrasound (Duty cycle: 10%, amplitude: 13%, 26 W, intensity 20.47 w/cm2, 20 kHz) | 1.71 | [57] | |
| Sodium propionate-based DESs | Polyphenol (quercetin 4′-O-glucoside and quercetin) and flavonoids | ~ | TPC (CAE): 137.5, TPC (RtE): 136.5 | [58] | |
| Betaine:glycolic acid (1:2) | Quercetin | Ultrasound | 14.79 µg/mL | [60] | |
| Tomato industry by-products | Lactic acid:glucose (5:1) | Polyphenols (caffeic acid, naringenin, catechin, quercetin, rutin) | Ultrasound (20 kHz, 200 W) | ~ | [44] |
| ChCl:lactic acid (1:2) | Phenolic acids (chlorogenic acid) | ~ | 52.3 | [61] | |
| Violet potato peels | ChCl:lactic acid | Polyphenols | Microwave and ultrasound | ~ | [62] |
| Kale waste | Betaine:glycerol (1:3) | Polyphenols | ~ | TPC (GAE): 16.8 | [63] |
| Bitter melon leaves | ChCl:acetic acid (1:2) | Polyphenols (gallic acid, chlorogenic acid, vanillic acid, quercetin-3-glucoside) and flavonoid (epicatechin) | Ultrasound | TPC (GAE): 82.1, TFC (ECE): 1.26 | [64] |
| Pepper leaves | ChCl:glucose (1:1) | Polyphenols (gallic, protocatechuic, chlorogenic, cinnamic, and coumaric acids) | Ultrasound (42 kHz) | TPC (GAE): 22.4 | [65] |
| Lotus leaf | Lactic acid:glycerol (1:2) | Flavonoids (quercetin 3-O-glucoside) | ~ | TPC (GAE): 113, TFC (RtE): 126 | [66] |
| Tree Nut By-Products | Optimal DESs/NADESs | Value-Added Compounds | Assisted Techniques and Optimal Conditions | Yields (mg/g) | Reference |
|---|---|---|---|---|---|
| Chestnut shell | ChCl:oxalic acid (1:1) | Polyphenols (gallic acid, ellagic acid, catechin hydrate, and procyanidin B2), total condensed tannin | Microwave | TPC (GAE): 295, total condensed tannins: 229.6 | [67] |
| ChCl:n-propanol (1:1) | Ellagic acid | Ultrasound | 4.64 | [68] | |
| Carya cathayensis Sarg peel | ChCl:malic acid (1:1) | Polyphenols (catechin, procyanidin B1, 2,3-dihydroxybenzoic acid, pinocembrin, procyanidin B3, and myricetrin) and flavonoids | Pulse ultrasound (20 kHz, 400 W) | TPC (GAE): 60.8 | [69] |
| TPC (GAE): 76.3, TFC (QE): 793 | [70] | ||||
| Hazelnut pomace | ChCl:1,2-propylene glycol (1:4) | Polyphenols (quinic acid, gallic acid, catechin, protocatechuic acid, and quercetin-3-O-rhamnoside) | Microwave | ~ | [71] |
| Cereal By-products | Optimal DESs/NADESs | Value-Added Compounds | Assisted Techniques and Optimal Conditions | Yields (mg/g) | Reference |
|---|---|---|---|---|---|
| Wheat bran | ChCl:glycerol (1:3) | Polyphenols | Ultrasound (37 KHz; 140 W; Power density: 35 W/L) | TPC (FAE): 17.78, TFC (RtE): 7.27 | [73] |
| Brewer’s spent grain | ChCl:glycerol (1:2) | Polyphenols (ferulic and coumaric acids) | Microwave | TPC (GAE): 2.30 | [72] |
| Black rice bran | Lactic acid:fructose (5:1) | Anthocyanin | Ultrasound (amplitude: 21.31%) | TAC (C3GE): 109 mg/L | [74] |
| Tartary buckwheat hull | ChCl:glycerol (1:1) | Rutin | Ultrasound (200 W, 20 kHz) | 9.5 | [75] |
| Red rice bran | ChCl:xylitol (1:1) | Polyphenols | ~ | ~ | [77] |
| Foxtail millet bran | Betaine:glycerol (1:2) | Polyphenols and flavonoids (coumaric acid, apigenin-C-dihexoside, and coumaroylspermidine) | Ultrasound (247 W) | TPC (FAE): 7.80 | [76] |
| Oil-Bearing Crop By-products | Optimal DESs/NADESs | Value-Added Compounds | Assisted Techniques and Optimal Conditions | Yields (mg/g) | Reference |
|---|---|---|---|---|---|
| Olive oil industry by-products | Glycerol:glycine: water (7:1:3) | Oleuropein, flavone glycosides | ~ | TPC (GAE): 106 | [84] |
| ChCl:citric acid (1:2) | Polyphenols (Oleuropein, hydroxytyrsol) | Microwave (200 W) and ultrasound (60 kHz, 280 W) | TPC (GAE): 34.1 (12.9, 3.37) | [80] | |
| ChCl:citric acid (1:1), ChCl:ethylene glycol (1:2) | Hydroxytyrosol, tyrosol, and oleuropein | Ultrasound (40 KHz) | ~ | [89] | |
| Citric acid:glycine:water (2:1:1) | Hydroxytyrosol | ~ | ~ | [87] | |
| ChCl:acetic acid (1:2), ChCl:malic acid (1:1), ChCl:malonic acid (1:1), and ChCl:citric acid (2:1) | Ferulic acid, kaempferol, luteolin, oleuropein, tyrosol | ~ | TPC (GAE): 34.6 | [86] | |
| Polyphenols | ~ | TPC (GAE): 19.8 | [81] | ||
| ChCl:fructose:water (5:2:5) | Oleuropein, caffeic acid | Ultrasound (37 KHz; 140 W; Power density: 35 W/L) | 1.63 and 0.113 | [85] | |
| ChCl:glycerol (1:5) | Luteolin-7-oglucoside, lleuropein, 3-hydroxytyrosol, rutin, and luteolin | ~ | ~ | [88] | |
| Soy molasses | ChCl:citric acid (1:2) | Isoflavone (daidzein, genistein and puerarin) | Ultrasound (37 KHz; 100 W) | ~, 2.49, 7.23 | [82] |
| Kudzu roots | 12.13, 2.51, 0.37 | ||||
| Peanut hull | Four Imidazole-based DESs (1:1) | Quercetin | Microwave (50 kHz) | 0.43 | [79] |
| Sunflower disks | ChCl:1,4-butanediol (1:2) | Chlorogenic acids | Ultrasound (40 KHz; 300 W; Power density: 0.5 W/cm2) | 6.16 | [78] |
| Other Agri-Food By-Products | Optimal DESs/NADESs | Value-Added Compounds | Assisted Techniques and Optimal Conditions | Yields (mg/g) | Reference |
|---|---|---|---|---|---|
| Coffee and cocoa industry residues | Fourteen hydrophilic and hydrophobic DESs | Chlorogenic acid | ~ | ~ | [90] |
| ChCl:citric acid (2:1), ChCl:glycerol (1:2), ChCl:glucose (1:1), betaine:citric acid (1:1), betaine:glycerol (1:2) and betaine:glucose (1:1) | Polyphenols | Ultrasound (150 W) | TPC (GAE): 20.3, 19.2, 21.6, 18.2, 15.3, 22.8 | [91] | |
| ChCl:lactic acid (1:2) | Polyphenols and chlorogenic acid | ~ | TPC (GAE): 52.9 | [92] | |
| Betaine:triethylene glycol (1:2) | Chlorogenic acid | Ultrasound (200 W, 37 kHz) | 4.64 | [93] | |
| ChCl:1,2-propanediol (1:2) | Gallic acid | ~ | 138 | [94] | |
| Spent hops | ChCl:ethylene glycol (1:2), ChCl:propylene glycol (1:2), ChCl:glycerol (1:2), ChCl:lactic acid (1:2) | Xanthohumol | ~ | 0.42–0.75 | [95] |
| Polyphenol-Rich NADESs Extracts | Film Matrix | Effects on Film Properties | Purpose | Reference | ||
|---|---|---|---|---|---|---|
| Main Compounds | By-Product | NADESs | ||||
| Polyphenols and flavonoids | Pomegranate peel | ChCl:glycerol | Chitosan | Reduced water sorption and shade color of films | Edible packaging film | [41] |
| Anthocyanin | Black rice bran | Lactic acid:fructose | Polyvinyl alcohol | Reduced the glass transition temperature, transparency, and young modulus, whereas increased water vapor permeability, elasticity, and water solubility. | Edible pH indicators for evaluating food quality during storage | [99] |
| Cinnamic acid, nordihidroguiaretic acid, and quercetin | Larrea divaricata | Lactic acid:glucose:water | Pectin | Achieved an in vitro antimicrobial activity of 72% against Monilinia fructicola. | Bioactive coating | [100] |
| Phenolic compounds | Moringa oleifera leaves | ChCl:glycerol, ChCl:lactic acid | Methylcellulose | All films are uniform, clear, and transparent with smooth, homogeneous surfaces. | Packaging films for sliced wheat bread | [101] |
| Ellagic acid, quercitrin, isoquercitrin | Rosa roxburghii Tratt leaves | ChCl:lactic acid | Chitosan/zein | Improved the mechanical properties, light barrier properties, water vapor permeability, antioxidant, and antibacterial activities of the chitosan/zein films. | Active packaging for fruit preservation | [102] |
| Catechin, protocatechuic acid ethyl ester, benzoic acid, syringic acid, and vanillin | Date palm leaves | ChCl:glycerol | Cellulose nanoparticles and soy protein isolate | Significantly increased antioxidant and antibacterial properties of films. | Active packaging films | [103] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, M.; Fakayode, O.A.; Li, H. Green Extraction of Polyphenols via Deep Eutectic Solvents and Assisted Technologies from Agri-Food By-Products. Molecules 2023, 28, 6852. https://doi.org/10.3390/molecules28196852
Zhou M, Fakayode OA, Li H. Green Extraction of Polyphenols via Deep Eutectic Solvents and Assisted Technologies from Agri-Food By-Products. Molecules. 2023; 28(19):6852. https://doi.org/10.3390/molecules28196852
Chicago/Turabian StyleZhou, Man, Olugbenga Abiola Fakayode, and Haoxin Li. 2023. "Green Extraction of Polyphenols via Deep Eutectic Solvents and Assisted Technologies from Agri-Food By-Products" Molecules 28, no. 19: 6852. https://doi.org/10.3390/molecules28196852
APA StyleZhou, M., Fakayode, O. A., & Li, H. (2023). Green Extraction of Polyphenols via Deep Eutectic Solvents and Assisted Technologies from Agri-Food By-Products. Molecules, 28(19), 6852. https://doi.org/10.3390/molecules28196852







