Agonists and Inhibitors of the cGAS-STING Pathway
Abstract
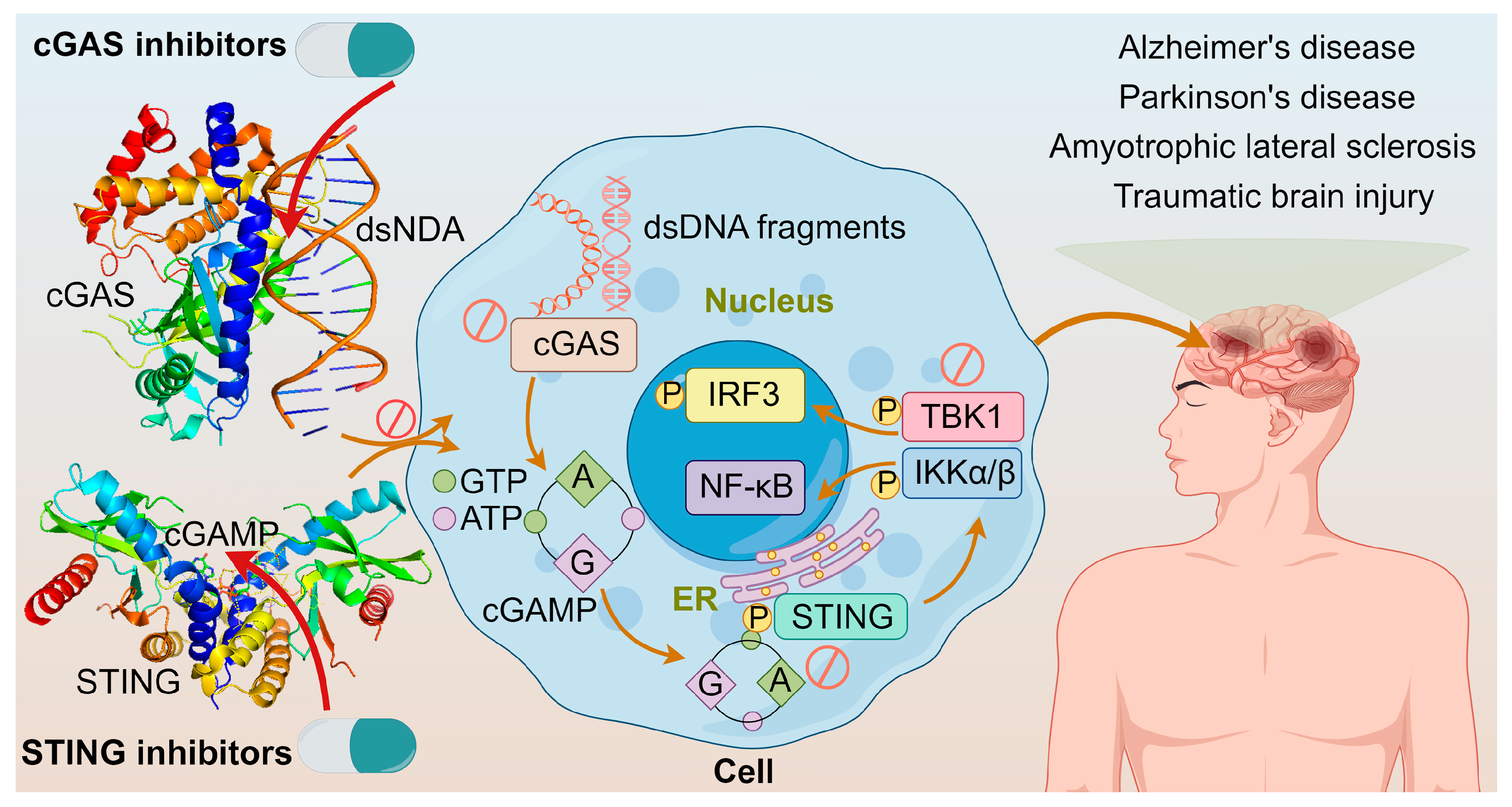
1. Introduction
2. cGAS-STING Agonists
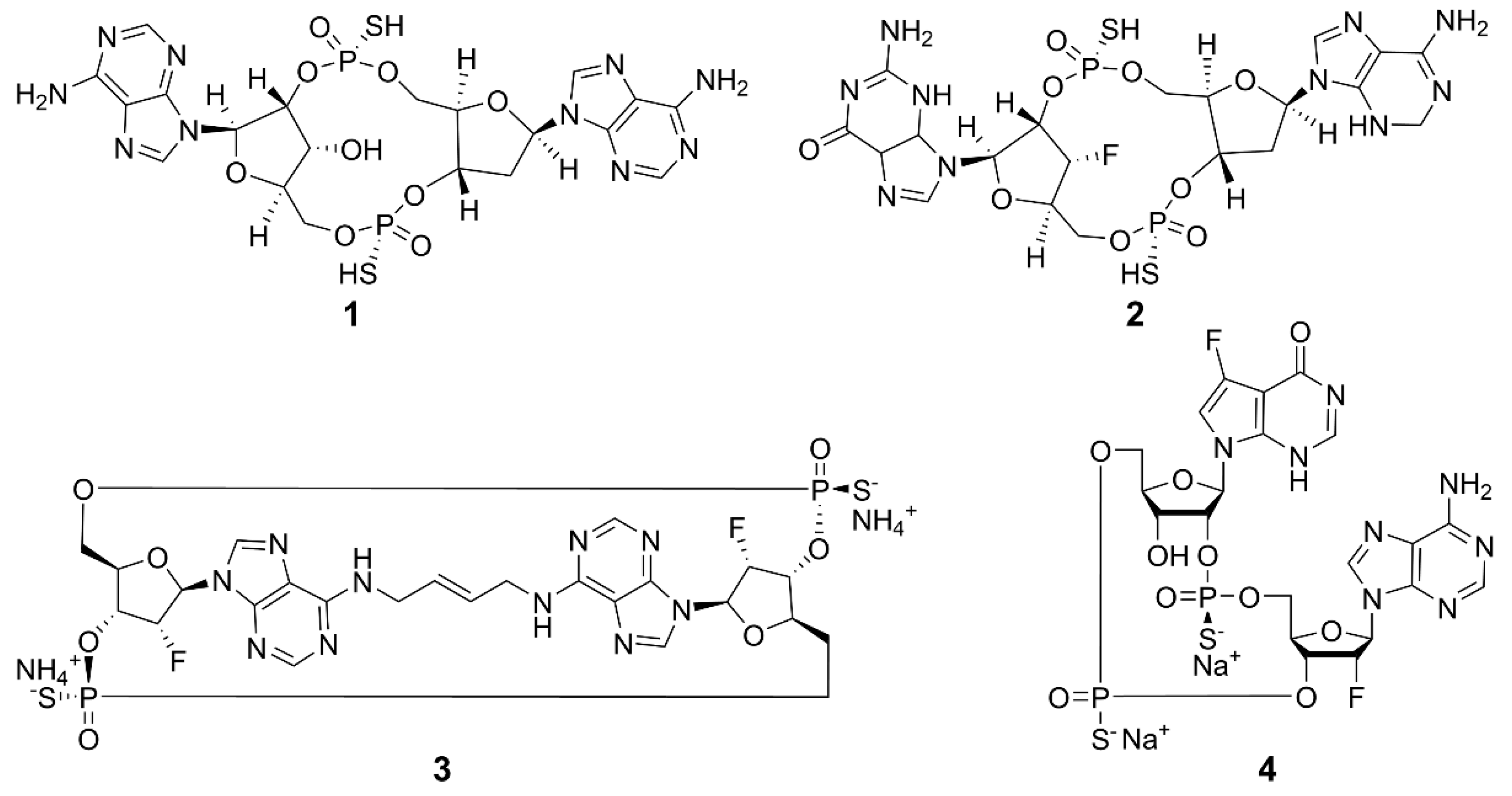
2.1. Cyclic Dinucleotides
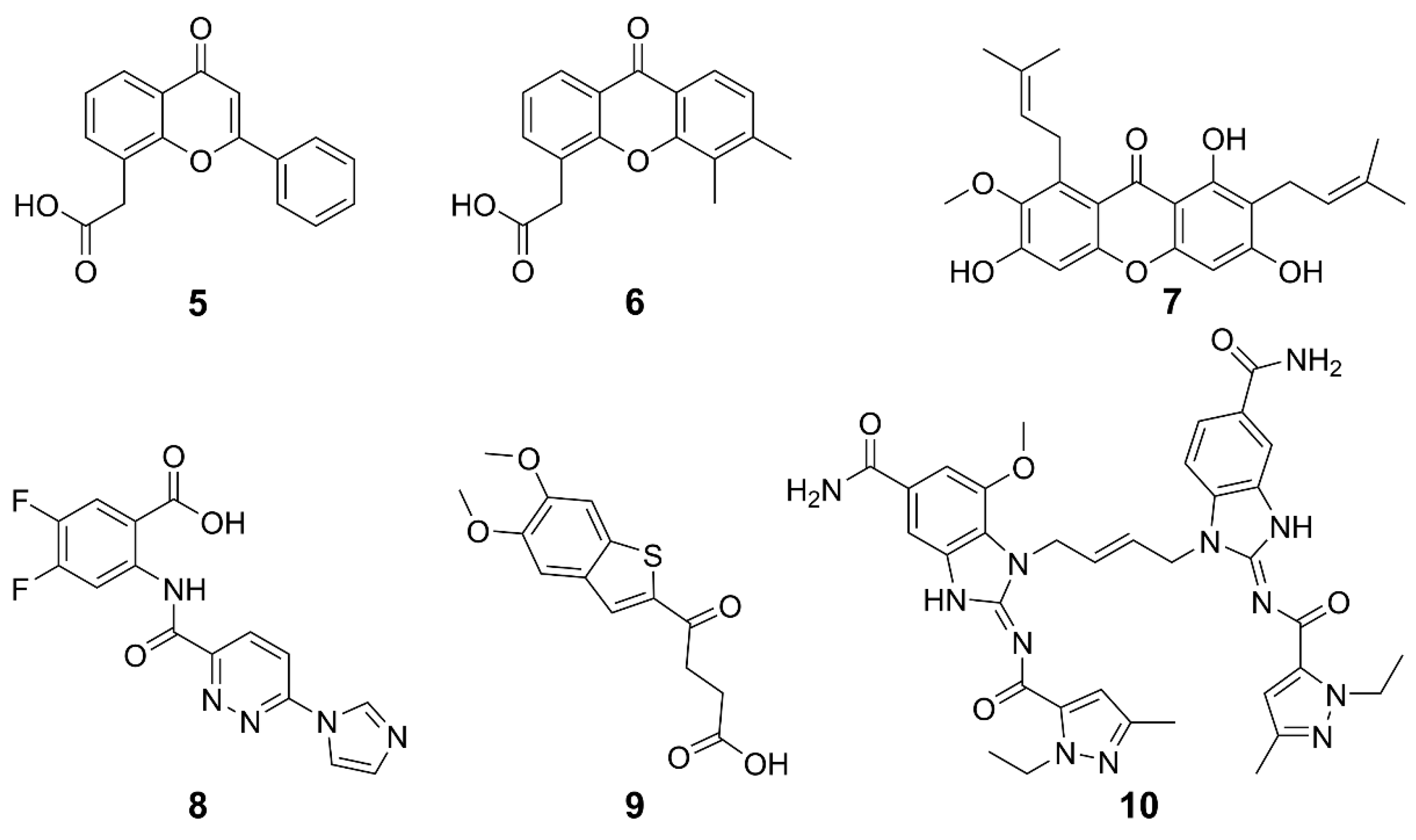
2.2. Non-CDNs
| Compound | Subject Tumors | Adimistration | Combination | Phase | NCT Number | Reference |
|---|---|---|---|---|---|---|
| 1 | Recurrent or metastatic squamous cell carcinoma of the bronchus | i.t.† | Alone | II | NCT03937141 | [58] |
| 2 | Head and neck squamous cell carcinoma | i.t. | Alone and combined with pembrolizumab | II | NCT04220866 | [59] |
| Solid tumors and lymphoma | i.t. | Alone and combined with pembrolizumab | I | NCT03010176 | [58] | |
| 3 | Advanced solid tumor; lymphoma | i.t. | Alone | I | NCT04144140 | [60] |
| Urinary bladder neoplasm | Intravesical injection | Alone | I | NCT04109092 | ||
| 4 | Solid neoplasm | i.v. | Alone and combined with pembrolizumab following radiotherapy | I | NCT04420884 | [61] |
| Carcinoma, non-small-cell lung, triple-negative breast neoplasm, squamous cell carcinoma of head and neck | i.v. | Alone and combined with pembrolizumab following radiotherapy | I | NCT04879849 | ||
| 5 | Various malignant solid tumors | i.v. | Constant infusion with alkalinization | / | / | [78] |
| 6 | Solid tumors | i.v. | Alone | I | NCT00863733 | [79] |
| Non-small cell lung cancer | n.s. | Alone and combined with carboplatin and paclitaxel | I/II | NCT00832494 | ||
| 7 | Cervical cancer stem-like cells | i.g. | Alone and combined with cisplatin | / | / | [80] |
| Colon cancer stem cells | i.p. | Alone and combined with 5-FU | [81] | |||
| 8 | Melanoma | i.p. | / | Preclinical studies | / | [75] |
| 9 | Colorectal carcinoma | i.t., i.h.; p.o. | / | Preclinical studies | / | [76] |
| 10 | Colorectal carcinoma | i.p. | / | Preclinical studies | / | [77] |
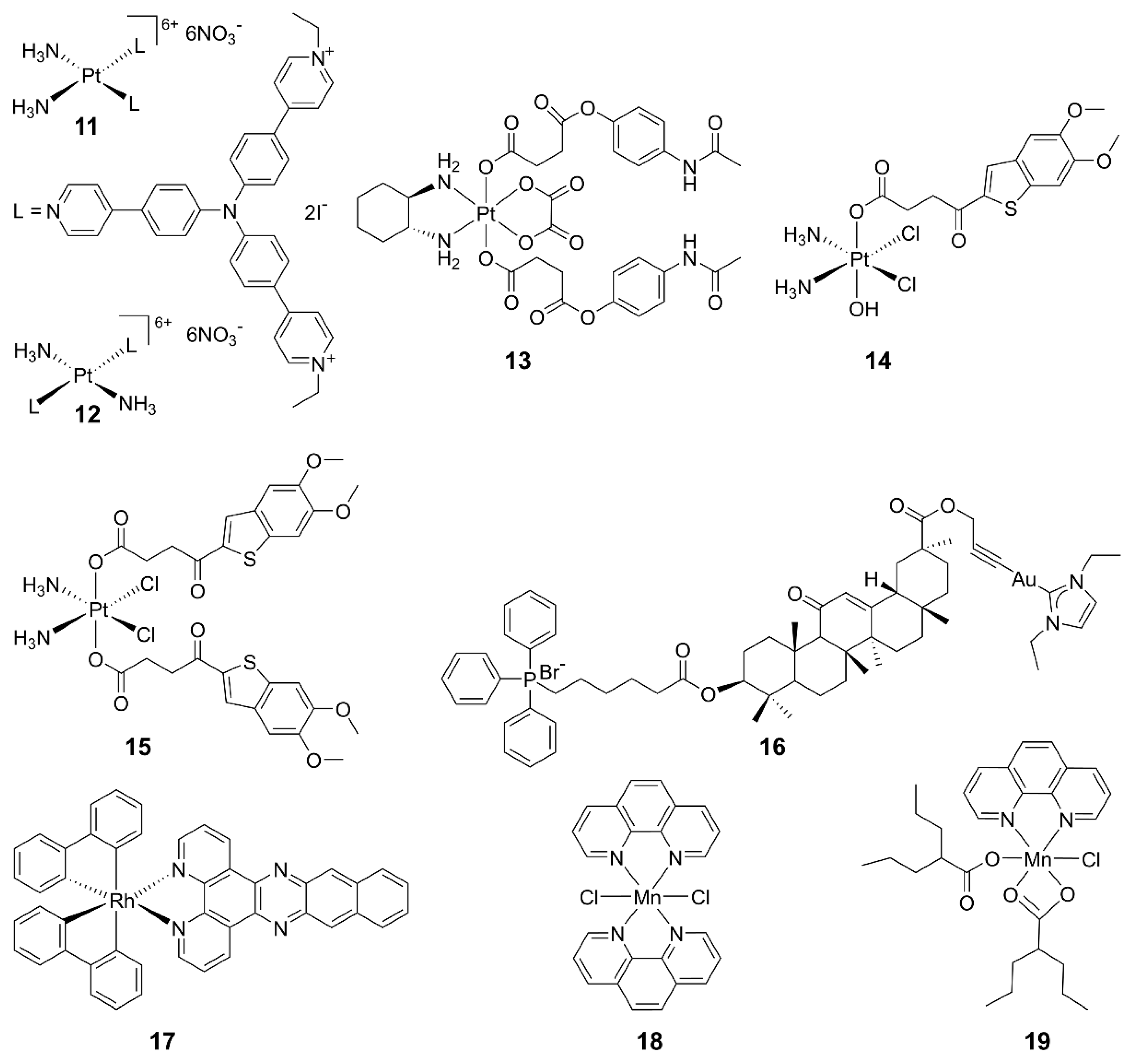
2.3. Metal-Based cGAS-STING Agonists
2.4. Other Agonists
3. cGAS-STING Inhibitors
3.1. STING Inhibitors
| Function | Compound | Subject | Model | Activity | Reference |
|---|---|---|---|---|---|
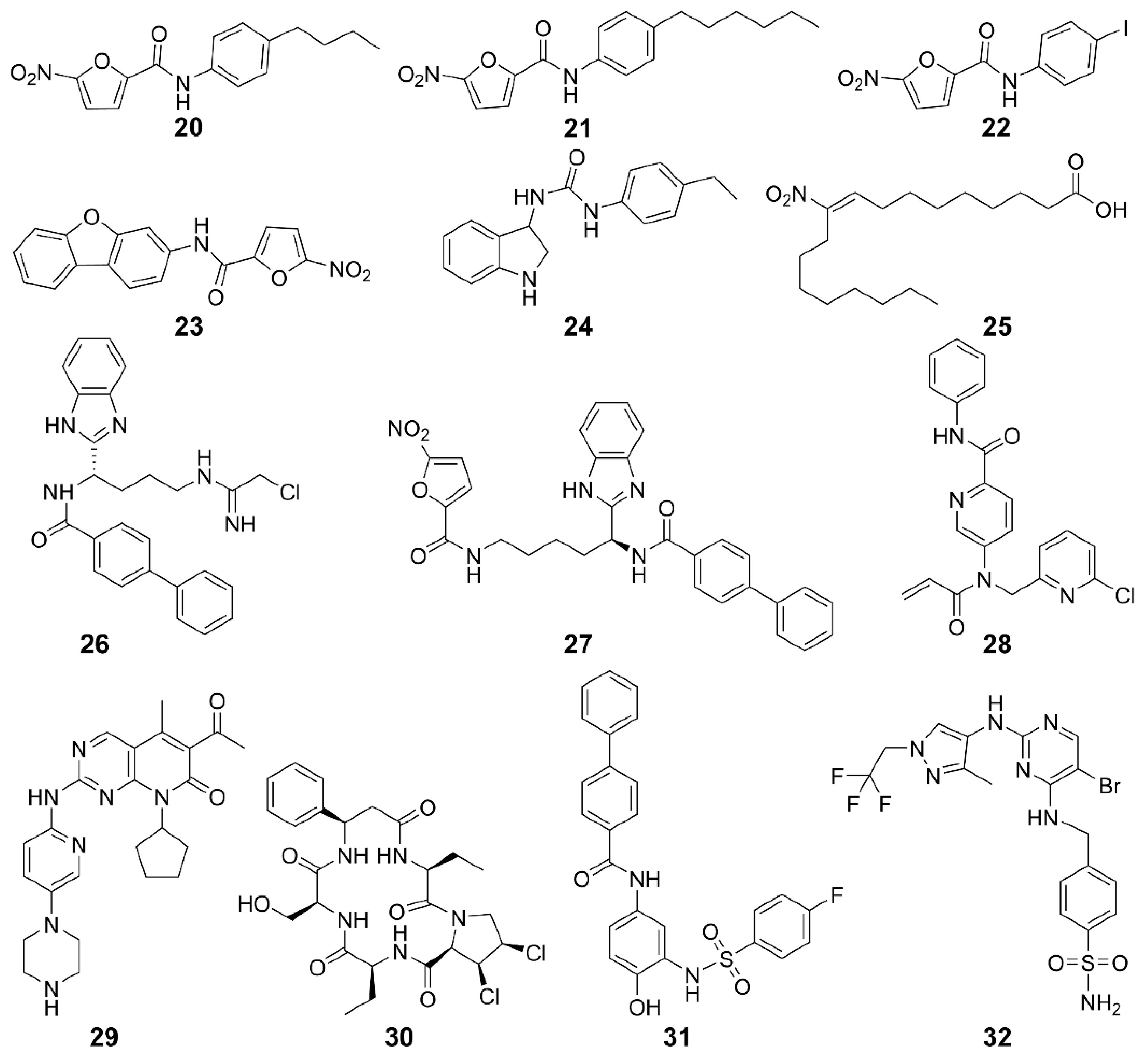
| STING palmitoylation inhibitors | 20, 21 | Human/Mice | Trex1-/- mice | Improves systemic inflammation | [120] |
| 22, 23 | Mice | Trex1-/- mice | Improves systemic inflammation | [120] | |
| 24 | Human/Mice | Trex1-/- mice | Improves systemic inflammation | [120] | |
| 25 | Human/Mice | SAVI patients with fibroblast cells | Inhibits the production of IFN-I | [121] | |
| 26 | Human/Mice | BMDMs cell | Inhibits the activation of STING in mouse and human cells and increases the survival rate of mice | [123] | |
| Peripheral blood monocyte | |||||
| TrexlD18N/D18N mice of AGS | |||||
| 27 | Human/Mice | Monocyte | Inhibits the covalent modification of STING and block the cGAS-STING signaling pathway | [130] | |
| BMDMs cell | |||||
| 28 | Human | Peripheral blood mononuclear cells | Inhibits cGAMP-mediated STING | [124] | |
| 29 | Human/Mice | HEK293T | Lowers the incidence of autoimmune diseases | [125] | |
| Mouse peritoneal macrophages | |||||
| THP1 cells | |||||
| CDN pocket inhibitors | 30 | Human/Mice | Trex1-/- BMDMs cells | Inhibits the expression of IFN-I and pro-inflammatory cytokines and reduces self-inflammatory response | [121] |
| Trex1-/- mice | |||||
| 31 | Human/Mice | Trex1-/- mice | Improves immune system | [131] | |
| TBK1 inhibitors | 32 | Human | Ramos cells | Inhibits Toll-like receptor 3-induced IFN-III phosphorylation | [127] |
| Human primary monocytes | Inhibits IFN-I secretion | ||||
| Monocyte-derived macrophages |
3.2. cGAS inhibitors
| Function | Compound | Subject | Model | Activity | Reference |
|---|---|---|---|---|---|
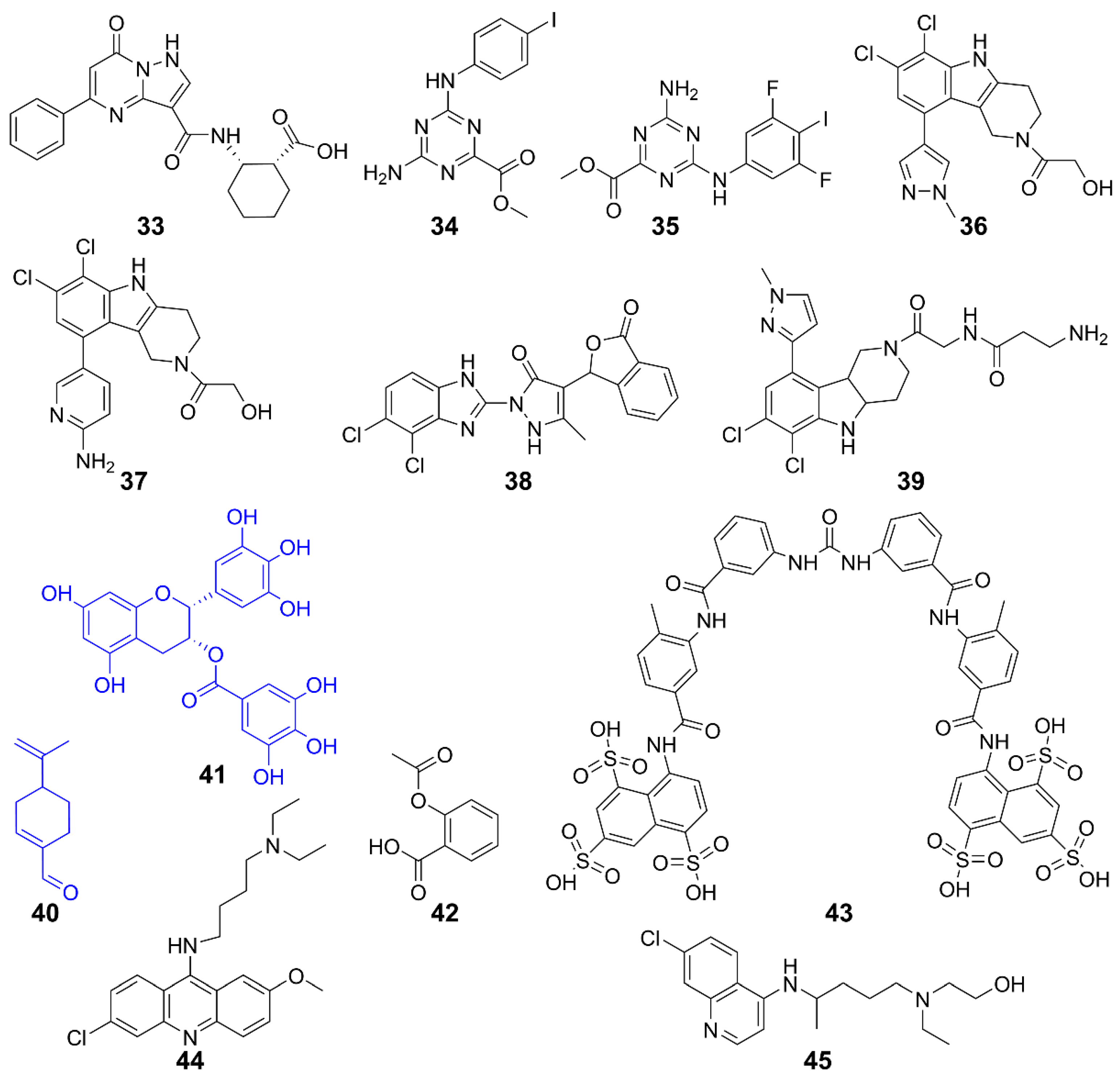
| Inhibits cGAS | 33 | Human cGAS | / | / | [132] |
| 34, 35 | Human cGAS | THP-1 cells | Inhibits DNA pathway | [133] | |
| 36, 37 | Human cGAS | THP-1 cells | Inhibits cGAS activity induced by dsDNA | [135] | |
| RAW 264.7 cells | |||||
| 38 | Mice cGAS | AGS mice | Inhibits IFN-I expression | [134] | |
| 39 | Human/Mice cGAS | THP-1 cells | Inhibits inflammation | [136] | |
| 40 | Human/Mice cGAS | AGS mice | Improves self-induced inflammation response of DNA | [137] | |
| 41 | Mice cGAS | AGS mice | Suppresses DNA-triggered cGAS activation by preventing the formation of G3BP1-cGAS complex, reducing auto-inflammatory response | [139] | |
| 42 | Human/Mice cGAS | AGS patient cells and AGS mice | Inhibits cGAS-mediated immune responses and self-DNA-induced autoimmunity | [140] | |
| Inhibits interaction with dsDNA/cGAS | 43 | Human cGAS | THP-1 cells | Inhibits cGAS-mediated IFN-I response | [141] |
| 44, 45 | Human/Mice cGAS | Trexl-/- mice | Acts on AGS/SLE mice | [142] |
4. Perspectives
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ABZI | Amidobenzimidazole |
| ADCs | Antibody-conjugated drugs |
| AGS | Aicardi–Goutières syndrome |
| APAP | Acetaminophen |
| APCs | Antigen-presenting cells |
| APMP | Amorphous porous manganese phosphate |
| CAFs | Cancer-associated fibroblasts |
| c-di-AMP | Cyclic di-AMP |
| c-di-GMP | Cyclic di-GMP |
| CDK | Cyclin-dependent protein kinase |
| cGAMP | Cyclic GMP-AMP |
| CTLs | Cytotoxic T-lymphocytes |
| Cys88/Cys91 | Cysteine residue 88/91 |
| DCs | Dendritic cells |
| DMBA | 7,12-Dimethyl-benz(a)anthracene |
| DMXAA | 5,6-Dimethylx-antheonone-4-acetic acid |
| DOX | Doxorubicin |
| dsDNA | Double-stranded DNA |
| EGCG | Epigallocatechin gallate |
| ENPP1 | Ectonucleotide pyrophosphatase phosphodiesterase 1 |
| ER | Endoplasmic reticulum |
| FAA | Flavone acetic acid |
| FIH | First-in-human |
| G3BP1 | GTPase-activating protein SH3 domain–binding protein 1 |
| GI | Gastrointestinal |
| GPCR | G protein-coupled receptor |
| GSH | Glutathione |
| hSTING | Human STING |
| HSV-1 | Herpes simplex virus type 1 |
| ICB | Immune checkpoint blockade |
| IDO | Indoleamine 2,3-dioxygenase |
| IFN-I | Type I interferon |
| IKK | IκB kinase |
| IL-6 | Interleukin-6 |
| IRF3 | Interferon regulatory factor 3 |
| ISGs | Interferon-stimulated genes |
| MDS | Myelodysplastic syndromes |
| MDSCs | Myeloid-derived suppressor cells |
| MHC-I | Major histocompatibility complex class I |
| mSTING | Mouse STING |
| NAFLD | Non-alcoholic fatty liver disease |
| NF-κB | Nuclear factor kappa B |
| NK | Natural killer |
| NP | Nanoparticle |
| Oxa | Oxaliplatin |
| PAH | Perillaldehyde |
| PAMPs | Pathogen-associated molecular patterns |
| PCBP1 | Poly-binding protein 1 |
| Pembro | Pembrolizumab |
| Phila | Cancer prev res |
| PL | Phospholipid |
| ROS | Reactive oxygen species |
| SAVI | STING-associated vasculopathy with onset in infancy |
| SLE | systemic lupus erythematosus |
| TBK1 | TANK-binding kinase 1 |
| TCGA | The Cancer Genome Atlas |
| TME | Tumor microenvironment |
| Tregs | Regulatory T cells |
| TRIM21 | Tripartite motif-containing 21 |
| ZCCHC3 | Zinc-finger CCHC-type containing protein 3 |
| ZF | Zinc-finger |
References
- Jenson, J.M.; Chen, Z.J. cGAS goes viral: A conserved immune defense system from bacteria to humans. Mol. Cell 2024, 84, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Cai, X.; Wu, J.; Cong, Q.; Chen, X.; Li, T.; Du, F.; Ren, J.; Wu, Y.T.; Grishin, N.V.; et al. Phosphorylation of innate immune adaptor proteins MAVS, STING, and TRIF induces IRF3 activation. Science 2015, 347, aaa2630. [Google Scholar] [CrossRef] [PubMed]
- Dvorkin, S.; Cambier, S.; Volkman, H.E.; Stetson, D.B. New frontiers in the cGAS-STING intracellular DNA-sensing pathway. Immunity 2024, 57, 718–730. [Google Scholar] [CrossRef]
- Lu, Y.; Zhao, M.; Chen, L.; Wang, Y.; Liu, T.; Liu, H. cGAS: Action in the nucleus. Front. Immunol. 2024, 15, 1380517. [Google Scholar] [CrossRef]
- Hopfner, K.P.; Hornung, V. Molecular mechanisms and cellular functions of cGAS-STING signalling. Nat. Rev. Mol. Cell Biol. 2020, 21, 501–521. [Google Scholar] [CrossRef] [PubMed]
- Decout, A.; Katz, J.D.; Venkatraman, S.; Ablasser, A. The cGAS-STING pathway as a therapeutic target in inflammatory diseases. Nat. Rev. Immunol. 2021, 21, 548–569. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.L.; Kagan, J.C. Targeting innate immune pathways for cancer immunotherapy. Immunity 2023, 56, 2206–2217. [Google Scholar] [CrossRef] [PubMed]
- Fang, R.; Wang, C.; Jiang, Q.; Lv, M.; Gao, P.; Yu, X.; Mu, P.; Zhang, R.; Bi, S.; Feng, J.M.; et al. NEMO-IKKbeta Are essential for IRF3 and NF-kappaB activation in the cGAS-STING pathway. J. Immunol. 2017, 199, 3222–3233. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D. Hallmarks of cancer: New dimensions. Cancer Discov. 2022, 12, 31–46. [Google Scholar] [CrossRef]
- Woo, S.R.; Fuertes, M.B.; Corrales, L.; Spranger, S.; Furdyna, M.J.; Leung, M.Y.; Duggan, R.; Wang, Y.; Barber, G.N.; Fitzgerald, K.A.; et al. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity 2014, 41, 830–842. [Google Scholar] [CrossRef]
- Corrales, L.; Glickman, L.H.; McWhirter, S.M.; Kanne, D.B.; Sivick, K.E.; Katibah, G.E.; Woo, S.R.; Lemmens, E.; Banda, T.; Leong, J.J.; et al. Direct activation of STING in the tumor microenvironment leads to potent and systemic tumor regression and immunity. Cell Rep. 2015, 11, 1018–1030. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Huang, L. Nanodelivery of cGAS-STING activators for tumor immunotherapy. Trends Pharmacol. Sci. 2022, 43, 957–972. [Google Scholar] [CrossRef] [PubMed]
- Lorenzi, S.; Mattei, F.; Sistigu, A.; Bracci, L.; Spadaro, F.; Sanchez, M.; Spada, M.; Belardelli, F.; Gabriele, L.; Schiavoni, G. Type I IFNs control antigen retention and survival of CD8alpha(+) dendritic cells after uptake of tumor apoptotic cells leading to cross-priming. J. Immunol. 2011, 186, 5142–5150. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Khorsandi, S.; Wang, Y.; Santelli, J.; Huntoon, K.; Nguyen, N.; Yang, M.; Lee, D.; Lu, Y.; Gao, R.; et al. Cancer immunotherapy based on image-guided STING activation by nucleotide nanocomplex-decorated ultrasound microbubbles. Nat. Nanotechnol. 2022, 17, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.; Mo, J.; Zhu, T.; Zhuo, W.; Yi, Y.; Hu, S.; Yin, J.; Zhang, W.; Zhou, H.; Liu, Z. Comprehensive elaboration of the cGAS-STING signaling axis in cancer development and immunotherapy. Mol. Cancer 2020, 19, 133. [Google Scholar] [CrossRef] [PubMed]
- Kato, K.; Omura, H.; Ishitani, R.; Nureki, O. Cyclic GMP-AMP as an endogenous second messenger in innate immune signaling by cytosolic DNA. Annu. Rev. Biochem. 2017, 86, 541–566. [Google Scholar] [CrossRef] [PubMed]
- Nicolai, C.J.; Wolf, N.; Chang, I.C. NK cells mediate clearance of CD8+T cell–resistanttumors in response to STING agonists. Sci. Immunol. 2020, 5, 5. [Google Scholar] [CrossRef]
- Marcus, A.; Mao, A.J.; Lensink-Vasan, M.; Wang, L.; Vance, R.E.; Raulet, D.H. Tumor-derived cGAMP triggers a STING-mediated interferon response in non-tumor Cells to activate the NK cell response. Immunity 2018, 49, 754–763.e754. [Google Scholar] [CrossRef]
- Lam, A.R.; Bert, N.L.; Ho, S.S.; Shen, Y.J.; Tang, L.F.; Xiong, G.M.; Croxford, J.L.; Koo, C.X.; Ishii, K.J.; Akira, S.; et al. RAE1 ligands for the NKG2D receptor are regulated by STING-dependent DNA sensor pathways in lymphoma. Cancer Res. 2014, 74, 2193–2203. [Google Scholar] [CrossRef]
- Motwani, M.; Pesiridis, S.; Fitzgerald, K.A. DNA sensing by the cGAS-STING pathway in health and disease. Nat. Rev. Genet. 2019, 20, 657–674. [Google Scholar] [CrossRef]
- Fu, J.; Kanne, D.B.; Leong, M.; Glickman, L.H.; McWhirter, S.M.; Lemmens, E.; Mechette, K.; Leong, J.J.; Lauer, P.; Liu, W.; et al. STING agonist formulated cancer vaccines can cure established tumors resistant to PD-1 blockade. Sci. Transl. Med. 2015, 7, 283ra252. [Google Scholar] [CrossRef]
- Wang, H.; Hu, S.Q.; Chen, X.; Shi, H.P.; Chen, C.; Sun, L.J.; Chen, Z.J.J. cGAS is essential for the antitumor effect of immune checkpoint blockade. Proc. Natl. Acad. Sci. USA 2017, 114, 1637–1642. [Google Scholar] [CrossRef]
- Vasiyani, H.; Wadhwa, B.; Singh, R. Regulation of cGAS-STING signalling in cancer: Approach for combination therapy. Biochim. Biophys. Acta. Rev. Cancer 2023, 1878, 188896. [Google Scholar] [CrossRef]
- An, X.; Zhu, Y.; Zheng, T.; Wang, G.; Zhang, M.; Li, J.; Ji, H.; Li, S.; Yang, S.; Xu, D.; et al. An analysis of the expression and association with immune cell lnfiltration of the cGAS/STING pathway in pan-cancer. Mol. Ther. Nucleic Acids 2019, 14, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Hubisz, M.J.; Earlie, E.M.; Duran, M.A.; Hong, C.; Varela, A.A.; Lettera, E.; Deyell, M.; Tavora, B.; Havel, J.J.; et al. Non-cell-autonomous cancer progression from chromosomal instability. Nature 2023, 620, 1080–1088. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.; Xia, T.L.; Konno, H.; Konno, K.; Ruiz, P.; Barber, G.N. Inflammation-driven carcinogenesis is mediated through STING. Nat. Commun. 2014, 5, 5166. [Google Scholar] [CrossRef] [PubMed]
- Bakhoum, S.F.; Ngo, B.; Laughney, A.M.; Cavallo, J.A.; Murphy, C.J.; Ly, P.; Shah, P.; Sriram, R.K.; Watkins, T.B.K.; Taunk, N.K.; et al. Chromosomal instability drives metastasis through a cytosolic DNA response. Nature 2018, 553, 467–472. [Google Scholar] [CrossRef]
- Munn, D.H.; Mellor, A.L. IDO in the tumor microenvironment: Inflammation, counter-regulation, and tolerance. Trends Immunol. 2016, 37, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Sun, L.; Chen, Z.J. Regulation and function of the cGAS-STING pathway of cytosolic DNA sensing. Nat. Immunol. 2016, 17, 1142–1149. [Google Scholar] [CrossRef]
- Li, X.D.; Wu, J.; Gao, D.; Wang, H.; Sun, L.; Chen, Z.J. Pivotal roles of cGAS-cGAMP signaling in antiviral defense and immune adjuvant effects. Science 2013, 341, 1390–1394. [Google Scholar] [CrossRef]
- Luo, J.; Solimini, N.L.; Elledge, S.J. Principles of cancer therapy: Oncogene and non-oncogene addiction. Cell 2009, 136, 823–837. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, P.; Yu, Y.; Fu, Y.; Jiang, H.; Lu, M.; Sun, Z.; Jiang, S.; Lu, L.; Wu, M.X. Pulmonary surfactant-biomimetic nanoparticles potentiate heterosubtypic influenza immunity. Science 2020, 367, eaau0810. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Qin, Y.; Yang, B.; Zheng, H.; Qiao, S.; Luo, Z.; Li, R. Pseudorabies virus usurps non-muscle myosin heavy chain IIA to dampen viral DNA recognition by cGAS for antagonism of host antiviral innate immunity. J. Virol. 2024, 98, e0048324. [Google Scholar] [CrossRef]
- Che, X.; Du, X.X.; Cai, X.; Zhang, J.; Xie, W.J.; Long, Z.; Ye, Z.Y.; Zhang, H.; Yang, L.; Su, X.D.; et al. Single mutations reshape the structural correlation network of the DMXAA-human STING complex. J. Phys. Chem. B 2017, 121, 2073–2082. [Google Scholar] [CrossRef] [PubMed]
- Pepin, G.; Nejad, C.; Ferrand, J.; Thomas, B.J.; Stunden, H.J.; Sanij, E.; Foo, C.H.; Stewart, C.R.; Cain, J.E.; Bardin, P.G.; et al. Topoisomerase 1 inhibition promotes cyclic GMP-AMP synthase-dependent antiviral responses. mBio 2017, 8, e01611-17. [Google Scholar] [CrossRef] [PubMed]
- Luthra, P.; Aguirre, S.; Yen, B.C.; Pietzsch, C.A.; Sanchez-Aparicio, M.T.; Tigabu, B.; Morlock, L.K.; Garcia-Sastre, A.; Leung, D.W.; Williams, N.S.; et al. Topoisomerase II inhibitors induce DNA damage-dependent interferon responses circumventing ebola virus immune evasion. mBio 2017, 8, e00368-17. [Google Scholar] [CrossRef] [PubMed]
- Renoult, O.; Laurent-Blond, M.; Awada, H.; Oliver, L.; Joalland, N.; Croyal, M.; Paris, F.; Gratas, C.; Pecqueur, C. Metabolic profiling of glioblastoma stem cells reveals pyruvate carboxylase as a critical survival factor and potential therapeutic target. Neuro Oncol. 2024. [Google Scholar] [CrossRef] [PubMed]
- Maekawa, H.; Inoue, T.; Ouchi, H.; Jao, T.M.; Inoue, R.; Nishi, H.; Fujii, R.; Ishidate, F.; Tanaka, T.; Tanaka, Y.; et al. Mitochondrial damage causes inflammation via cGAS-STING signaling in acute kidney injury. Cell Rep. 2019, 29, 1261–1273. [Google Scholar] [CrossRef] [PubMed]
- Unterholzner, L.; Dunphy, G. cGAS-independent STING activation in response to DNA damage. Mol. Cell. Oncol. 2019, 6, 1558682. [Google Scholar] [CrossRef]
- Chabanon, R.M.; Muirhead, G.; Krastev, D.B.; Adam, J.; Morel, D.; Garrido, M.; Lamb, A.; Henon, C.; Dorvault, N.; Rouanne, M.; et al. PARP inhibition enhances tumor cell-intrinsic immunity in ERCC1-deficient non-small cell lung cancer. J. Clin. Investig. 2019, 129, 1211–1228. [Google Scholar] [CrossRef]
- Pantelidou, C.; Sonzogni, O.; De Oliveria Taveira, M.; Mehta, A.K.; Kothari, A.; Wang, D.; Visal, T.; Li, M.K.; Pinto, J.; Castrillon, J.A.; et al. PARP inhibitor efficacy depends on CD8(+) T-cell recruitment via intratumoral STING pathway activation in BRCA-deficient models of triple-negative breast cancer. Cancer Discov. 2019, 9, 722–737. [Google Scholar] [CrossRef] [PubMed]
- Dillon, M.T.; Bergerhoff, K.F.; Pedersen, M.; Whittock, H.; Crespo-Rodriguez, E.; Patin, E.C.; Pearson, A.; Smith, H.G.; Paget, J.T.E.; Patel, R.R.; et al. ATR inhibition potentiates the radiation-induced inflammatory tumor microenvironment. Clin. Cancer Res. 2019, 25, 3392–3403. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Chen, J.; Hu, J.; Zhang, H.; Xu, F.; He, W.; Wang, X.; Li, M.; Lu, W.; Zeng, G.; et al. cGAS/STING axis mediates a topoisomerase II inhibitor-induced tumor immunogenicity. J. Clin. Investig. 2019, 129, 4850–4862. [Google Scholar] [CrossRef] [PubMed]
- Curtsinger, J.M.; Valenzuela, J.O.; Agarwal, P.; Lins, D.; Mescher, M.F. Type I IFNs provide a third signal to CD8 T cells to stimulate clonal expansion and differentiation. J. Immunol. 2005, 174, 4465–4469. [Google Scholar] [CrossRef] [PubMed]
- Yi, Y.; Qin, G.; Yang, H.; Jia, H.; Zeng, Q.; Zheng, D.; Ye, S.; Zhang, Z.; Liu, T.M.; Luo, K.Q.; et al. Mesenchymal stromal cells increase the natural killer resistance of circulating tumor cells via intercellular signaling of cGAS-STING-IFNbeta-HLA. Adv. Sci. 2024, 11, e2400888. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.J.; Wu, J.X.; Du, F.H.; Chen, X.; Chen, Z.J.J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 2013, 339, 786–791. [Google Scholar] [CrossRef]
- Ablasser, A.; Goldeck, M.; Cavlar, T.; Deimling, T.; Witte, G.; Rohl, I.; Hopfner, K.P.; Ludwig, J.; Hornung, V. cGAS produces a 2’-5’-linked cyclic dinucleotide second messenger that activates STING. Nature 2013, 498, 380–384. [Google Scholar] [CrossRef]
- Ergun, S.L.; Fernandez, D.; Weiss, T.M.; Li, L. STING polymer structure reveals mechanisms for activation, hyperactivation, and inhibition. Cell 2019, 178, 290–301. [Google Scholar] [CrossRef]
- Zhou, C.; Chen, X.; Planells-Cases, R.; Chu, J.; Wang, L.; Cao, L.; Li, Z.; Lopez-Cayuqueo, K.I.; Xie, Y.; Ye, S.; et al. Transfer of cGAMP into bystander cells via LRRC8 volume-regulated anion channels augments STING-mediated interferon responses and anti-viral immunity. Immunity 2020, 52, 767–781.e766. [Google Scholar] [CrossRef]
- Slavik, K.M.; Morehouse, B.R.; Ragucci, A.E.; Zhou, W.; Ai, X.; Chen, Y.; Li, L.; Wei, Z.; Bahre, H.; Konig, M.; et al. cGAS-like receptors sense RNA and control 3’2’-cGAMP signalling in Drosophila. Nature 2021, 597, 109–113. [Google Scholar] [CrossRef]
- Holleufer, A.; Winther, K.G.; Gad, H.H.; Ai, X.; Chen, Y.; Li, L.; Wei, Z.; Deng, H.; Liu, J.; Frederiksen, N.A.; et al. Two cGAS-like receptors induce antiviral immunity in Drosophila. Nature 2021, 597, 114–118. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Wu, J.; Chen, Z.J.; Chen, C. Molecular basis for the specific recognition of the metazoan cyclic GMP-AMP by the innate immune adaptor protein STING. Proc. Natl. Acad. Sci. USA 2015, 112, 8947–8952. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Shi, H.P.; Wu, J.X.; Zhang, X.W.; Sun, L.J.; Chen, C.; Chen, Z.J.J. Cyclic GMP-AMP containing mixed phosphodiester linkages is an endogenous high-affinity ligand for STING. Mol. Cell 2013, 51, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Kwon, J.; Bakhoum, S.F. The cytosolic DNA-sensing cGAS-STING pathway in cancer. Cancer Discov. 2020, 10, 26–39. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Wang, X.; KuoLee, R.; Chen, W. Synthesis and immunostimulatory properties of the phosphorothioate analogues of cdiGMP. Bioorg. Med. Chem. Lett. 2008, 18, 5631–5634. [Google Scholar] [CrossRef] [PubMed]
- Eckstein, F. Phosphorothioates, essential components of therapeutic oligonucleotides. Nucleic Acid Ther. 2014, 24, 374–387. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Yin, Q.; Kuss, P.; Maliga, Z.; Millan, J.L.; Wu, H.; Mitchison, T.J. Corrigendum: Hydrolysis of 2’3’-cGAMP by ENPP1 and design of nonhydrolyzable analogs. Nat. Chem. Biol. 2015, 11, 235. [Google Scholar] [CrossRef] [PubMed]
- Gogoi, H.; Mansouri, S.; Jin, L. The age of cyclic dinucleotide vaccine adjuvants. Vaccines 2020, 8, 453. [Google Scholar] [CrossRef]
- Chang, W.; Altman, M.D.; Lesburg, C.A.; Perera, S.A.; Piesvaux, J.A.; Schroeder, G.K.; Wyss, D.F.; Cemerski, S.; Chen, Y.; DiNunzio, E.; et al. Discovery of MK-1454: A potent cyclic dinucleotide stimulator of interferon genes agonist for the treatment of cancer. J. Med. Chem. 2022, 65, 5675–5689. [Google Scholar] [CrossRef]
- Kim, D.S.; Endo, A.; Fang, F.G.; Huang, K.C.; Bao, X.; Choi, H.W.; Majumder, U.; Shen, Y.Y.; Mathieu, S.; Zhu, X.; et al. E7766, a macrocycle-bridged stimulator of interferon genes (STING) agonist with potent pan-genotypic activity. ChemMedChem 2021, 16, 1740–1743. [Google Scholar] [CrossRef]
- Carideo Cunniff, E.; Sato, Y.; Mai, D.; Appleman, V.A.; Iwasaki, S.; Kolev, V.; Matsuda, A.; Shi, J.; Mochizuki, M.; Yoshikawa, M.; et al. TAK-676: A novel stimulator of interferon genes (STING) agonist promoting durable IFN-dependent antitumor immunity in preclinical studies. Cancer Res. Commun. 2022, 2, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Fang, R.; Jiang, Q.; Yu, X.; Zhao, Z.; Jiang, Z. Recent advances in the activation and regulation of the cGAS-STING pathway. Adv. Immunol. 2022, 156, 55–102. [Google Scholar] [CrossRef]
- Kerr, D.J.; Kaye, S.B. Flavone acetic acid--preclinical and clinical activity. Eur. J. Cancer Clin. Oncol. 1989, 25, 1271–1272. [Google Scholar] [CrossRef]
- Baguley, B.C.; Ching, L.M. DMXAA: An antivascular agent with multiple host responses. Int. J. Radiat. Oncol. Biol. Phys. 2002, 54, 1503–1511. [Google Scholar] [CrossRef]
- Woon, S.T.; Reddy, C.B.; Drummond, C.J.; Schooltink, M.A.; Baguley, B.C.; Kieda, C.; Ching, L.M. A comparison of the ability of DMXAA and xanthenone analogues to activate NF-kappaB in murine and human cell lines. Oncol. Res. 2005, 15, 351–364. [Google Scholar] [CrossRef] [PubMed]
- Philpott, M.; Baguley, B.C.; Ching, L.M. Induction of tumour necrosis factor-alpha by single and repeated doses of the antitumour agent 5,6-dimethylxanthenone-4-acetic acid. Cancer Chemother. Pharmacol. 1995, 36, 143–148. [Google Scholar] [CrossRef]
- Xiao, Z.; Cui, X.; Liu, F.; Wang, Y.; Liu, X.; Zhou, W.; Zhang, Y. Tumor vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid (DMXAA): Suppresses macrophage capping protein beyond STING activation. Biochim. Biophys. Acta Mol. Basis Dis. 2024, 1870, 167149. [Google Scholar] [CrossRef]
- Temizoz, B.; Shibahara, T.; Hioki, K.; Hayashi, T.; Kobiyama, K.; Lee, M.S.J.; Surucu, N.; Sag, E.; Kumanogoh, A.; Yamamoto, M.; et al. 5,6-dimethylxanthenone-4-acetic acid (DMXAA), a partial STING agonist, competes for human STING activation. Front. Immunol. 2024, 15, 1353336. [Google Scholar] [CrossRef]
- Conlon, J.; Burdette, D.L.; Sharma, S.; Bhat, N.; Thompson, M.; Jiang, Z.; Rathinam, V.A.; Monks, B.; Jin, T.; Xiao, T.S.; et al. Mouse, but not human STING, binds and signals in response to the vascular disrupting agent 5,6-dimethylxanthenone-4-acetic acid. J. Immunol. 2013, 190, 5216–5225. [Google Scholar] [CrossRef] [PubMed]
- Graham, P.T.; Nowak, A.K.; Cornwall, S.M.J.; Larma, I.; Nelson, D.J. The STING agonist, DMXAA, reduces tumor vessels and enhances mesothelioma tumor antigen presentation yet blunts cytotoxic T cell function in a murine model. Front. Immunol. 2022, 13, 969678. [Google Scholar] [CrossRef]
- Prantner, D.; Perkins, D.J.; Lai, W.; Williams, M.S.; Sharma, S.; Fitzgerald, K.A.; Vogel, S.N. 5,6-Dimethylxanthenone-4-acetic acid (DMXAA) activates stimulator of interferon gene (STING)-dependent innate immune pathways and is regulated by mitochondrial membrane potential. J. Biol. Chem. 2012, 287, 39776–39788. [Google Scholar] [CrossRef] [PubMed]
- Lara, P.N., Jr.; Douillard, J.Y.; Nakagawa, K.; von Pawel, J.; McKeage, M.J.; Albert, I.; Losonczy, G.; Reck, M.; Heo, D.S.; Fan, X.; et al. Randomized phase III placebo-controlled trial of carboplatin and paclitaxel with or without the vascular disrupting agent vadimezan (ASA404) in advanced non-small-cell lung cancer. J. Clin. Oncol. 2011, 29, 2965–2971. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Zillinger, T.; Wang, W.; Ascano, M.; Dai, P.; Hartmann, G.; Tuschl, T.; Deng, L.; Barchet, W.; Patel, D.J. Binding-pocket and lid-region substitutions render human STING sensitive to the species-specific drug DMXAA. Cell Rep. 2014, 8, 1668–1676. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Sun, Z.; Pei, J.; Luo, Q.; Zeng, X.; Li, Q.; Yang, Z.; Quan, J. Identification of alpha-Mangostin as an Agonist of Human STING. ChemMedChem 2018, 13, 2057–2064. [Google Scholar] [CrossRef]
- Chin, E.N.; Yu, C.; Vartabedian, V.F.; Jia, Y.; Kumar, M.; Gamo, A.M.; Vernier, W.; Ali, S.H.; Kissai, M.; Lazar, D.C.; et al. Antitumor activity of a systemic STING-activating non-nucleotide cGAMP mimetic. Science 2020, 369, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.S.; Perera, S.A.; Piesvaux, J.A.; Presland, J.P.; Schroeder, G.K.; Cumming, J.N.; Trotter, B.W.; Altman, M.D.; Buevich, A.V.; Cash, B.; et al. An orally available non-nucleotide STING agonist with antitumor activity. Science 2020, 369, eaba6098. [Google Scholar] [CrossRef] [PubMed]
- Ramanjulu, J.M.; Pesiridis, G.S.; Yang, J.; Concha, N.; Singhaus, R.; Zhang, S.Y.; Tran, J.L.; Moore, P.; Lehmann, S.; Eberl, H.C.; et al. Design of amidobenzimidazole STING receptor agonists with systemic activity. Nature 2018, 564, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Pratt, C.B.; Relling, M.V.; Meyer, W.H.; Douglass, E.C.; Kellie, S.J.; Avery, L. Phase I study of flavone acetic acid (NSC 347512, LM975) in patients with pediatric malignant solid tumors. Am. J. Clin. Oncol. 1991, 14, 483–486. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhou, H.; Ouyang, X.; Dong, Y.; Sarapultsev, A.; Luo, S.; Hu, D. Multifaceted functions of STING in human health and disease: From molecular mechanism to targeted strategy. Signal Transduct. Target. Ther. 2022, 7, 394. [Google Scholar] [CrossRef]
- Chien, H.J.; Ying, T.H.; Hsieh, S.C.; Lin, C.L.; Yu, Y.L.; Kao, S.H.; Hsieh, Y.H. alpha-Mangostin attenuates stemness and enhances cisplatin-induced cell death in cervical cancer stem-like cells through induction of mitochondrial-mediated apoptosis. J. Cell. Physiol. 2020, 235, 5590–5601. [Google Scholar] [CrossRef]
- Jo, M.K.; Moon, C.M.; Kim, E.J.; Kwon, J.H.; Fei, X.; Kim, S.E.; Jung, S.A.; Kim, M.; Mun, Y.C.; Ahn, Y.H.; et al. Suppressive effect of alpha-mangostin for cancer stem cells in colorectal cancer via the Notch pathway. BMC Cancer 2022, 22, 341. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Guo, Y.; Wang, X. Development of platinum complexes for tumor chemoimmunotherapy. Chemistry 2024, 30, e202302948. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, C.; Ma, Y.; Pan, X.; Chu, R.; Yao, S.; Chen, J.; Liu, C.; Chen, Z.; Sheng, C.; et al. STING inhibitors sensitize platinum chemotherapy in ovarian cancer by inhibiting the CGAS-STING pathway in cancer-associated fibroblasts (CAFs). Cancer Lett. 2024, 588, 216700. [Google Scholar] [CrossRef] [PubMed]
- Ling, Y.Y.; Xia, X.Y.; Hao, L.; Wang, W.J.; Zhang, H.; Liu, L.Y.; Liu, W.; Li, Z.Y.; Tan, C.P.; Mao, Z.W. Simultaneous photoactivation of cGAS-STING pathway and pyroptosis by platinum(II) triphenylamine complexes for cancer immunotherapy. Angew. Chem. Int. Ed. Engl. 2022, 61, e202210988. [Google Scholar] [CrossRef] [PubMed]
- Fan, R.; Lin, R.; Zhang, S.; Deng, A.; Hai, Y.; Zhuang, J.; Liu, Y.; Cheng, M.; Wei, G. Novel Pt(IV) complex OAP2 induces STING activation and pyroptosis via mitochondrial membrane remodeling for synergistic chemo-immunotherapy. Acta Pharm. Sin. B 2024, 14, 1742–1758. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Song, D.; Yu, W.; Li, J.; Wang, X.; Li, Y.; Zhao, Z.; Xue, Q.; Zhao, J.; Li, J.P.; et al. Combining cisplatin and a STING agonist into one molecule for metalloimmunotherapy of cancer. Natl. Sci. Rev. 2024, 11, nwae020. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Wen, Z.; Wu, C.; Yang, Z.; Wang, Z.; Diao, W.; Chen, D.; Xu, Z.; Lu, Y.; Liu, W. Simultaneous activation of immunogenic cell death and cGAS-STING pathway by liver- and mitochondria-targeted gold(I) complexes for chemoimmunotherapy of hepatocellular carcinoma. J. Med. Chem. 2024, 67, 1982–2003. [Google Scholar] [CrossRef]
- Zheng, Y.; Chen, X.X.; Zhang, D.Y.; Wang, W.J.; Peng, K.; Li, Z.Y.; Mao, Z.W.; Tan, C.P. Activation of the cGAS-STING pathway by a mitochondrial DNA-targeted emissive rhodium(iii) metallointercalator. Chem. Sci. 2023, 14, 6890–6903. [Google Scholar] [CrossRef]
- Wang, C.G.; Guan, Y.K.; Lv, M.Z.; Zhang, R.; Guo, Z.Y.; Wei, X.M.; Du, X.X.; Yang, J.; Li, T.; Wan, Y.; et al. Manganese increases the sensitivity of the cGAS-STING pathway for double-stranded DNA and is required for the host defense against DNA viruses. Immunity 2018, 48, 675–687.e677. [Google Scholar] [CrossRef]
- Zhao, Z.; Ma, Z.; Wang, B.; Guan, Y.; Su, X.D.; Jiang, Z. Mn2+ directly activates cGAS and structural analysis suggests Mn2+ induces a noncanonical catalytic synthesis of 2’3’-cGAMP. Cell Rep. 2020, 32, 108053. [Google Scholar] [CrossRef]
- Song, Y.; Liu, Y.; Teo, H.Y.; Hanafi, Z.B.; Mei, Y.; Zhu, Y.; Chua, Y.L.; Lv, M.; Jiang, Z.; Liu, H. Manganese enhances the antitumor function of CD8+ T cells by inducing type I interferon production. Cell Mol. Immunol. 2021, 18, 1571–1574. [Google Scholar] [CrossRef] [PubMed]
- Lv, M.Z.; Chen, M.X.; Zhang, R.; Zhang, W.; Wang, C.G.; Zhang, Y.; Wei, X.M.; Guan, Y.K.; Liu, J.J.; Feng, K.C.; et al. Manganese is critical for antitumor immune responses via cGAS-STING and improves the efficacy of clinical immunotherapy. Cell Res. 2020, 30, 966–979. [Google Scholar] [CrossRef]
- Zhang, S.; Kang, L.; Dai, X.; Chen, J.; Chen, Z.; Wang, M.; Jiang, H.; Wang, X.; Bu, S.; Liu, X.; et al. Manganese induces tumor cell ferroptosis through type-I IFN dependent inhibition of mitochondrial dihydroorotate dehydrogenase. Free Radic. Biol. Med. 2022, 193, 202–212. [Google Scholar] [CrossRef]
- Hou, L.; Tian, C.; Yan, Y.; Zhang, L.; Zhang, H.; Zhang, Z. Manganese-based nanoactivator optimizes cancer immunotherapy via enhancing innate immunity. ACS Nano 2020, 14, 3927–3940. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Feng, L.; Wu, Y. Enzymatically synthesised MnO2 nanoparticles for efficient near-infrared photothermal therapy and dual-responsive magnetic resonance imaging. Nanoscale 2021, 13, 11093–11103. [Google Scholar] [CrossRef]
- Li, J.; Li, S.; Li, Y.; Yuan, G.; Shen, Y.; Peng, Y.; Kong, L.; Yang, C.; Zhang, Z.; Li, Z. A magnetic resonance nanoprobe with STING activation character collaborates with platinum-based drug for enhanced tumor immunochemotherapy. J. Nanobiotechnol. 2021, 19, 415. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Wang, Y.; Chen, Y.; Chen, H.; Yang, T.; Zhang, S.; Guo, Z.; Wang, X. Manganese(ii) complexes stimulate antitumor immunity via aggravating DNA damage and activating the cGAS-STING pathway. Chem. Sci. 2023, 14, 4375–4389. [Google Scholar] [CrossRef] [PubMed]
- Englinger, B.; Pirker, C.; Heffeter, P.; Terenzi, A.; Kowol, C.R.; Keppler, B.K.; Berger, W. Metal drugs and the anticancer immune response. Chem. Rev. 2019, 119, 1519–1624. [Google Scholar] [CrossRef]
- Shen, S.; Lim, G.; You, Z.; Ding, W.; Huang, P.; Ran, C.; Doheny, J.; Caravan, P.; Tate, S.; Hu, K.; et al. Gut microbiota is critical for the induction of chemotherapy-induced pain. Nat. Neurosci. 2017, 20, 1213–1216. [Google Scholar] [CrossRef]
- Oun, R.; Moussa, Y.E.; Wheate, N.J. Correction: The side effects of platinum-based chemotherapy drugs: A review for chemists. Dalton Trans. 2018, 47, 7848. [Google Scholar] [CrossRef]
- Lanvers-Kaminsky, C.; Malath, I.; Deuster, D.; Ciarimboli, G.; Boos, J.; Am Zehnhoff-Dinnesen, A.G. Evaluation of pharmacogenetic markers to predict the risk of Cisplatin-induced ototoxicity. Clin. Pharmacol. Ther. 2014, 96, 156–157. [Google Scholar] [CrossRef]
- Hartmann, J.T.; Lipp, H.P. Toxicity of platinum compounds. Expert Opin. Pharmacother. 2003, 4, 889–901. [Google Scholar] [CrossRef]
- Pydi, S.P.; Jain, S.; Tung, W.; Cui, Y.; Zhu, L.; Sakamoto, W.; Jain, S.; Abel, B.S.; Skarulis, M.C.; Liu, J.; et al. Adipocyte beta-arrestin-2 is essential for maintaining whole body glucose and energy homeostasis. Nat. Commun. 2019, 10, 2936. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, M.; Li, L.; Qian, G.; Wang, Y.; Chen, Z.; Liu, J.; Fang, C.; Huang, F.; Guo, D.; et al. beta-arrestin 2 as an activator of cGAS-STING signaling and target of viral immune evasion. Nat. Commun. 2020, 11, 6000. [Google Scholar] [CrossRef]
- Vasiliev, Y.M. Chitosan-based vaccine adjuvants: Incomplete characterization complicates preclinical and clinical evaluation. Expert Rev. Vaccines 2015, 14, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Carroll, E.C.; Jin, L.; Mori, A.; Munoz-Wolf, N.; Oleszycka, E.; Moran, H.B.T.; Mansouri, S.; McEntee, C.P.; Lambe, E.; Agger, E.M.; et al. The vaccine adjuvant chitosan promotes cellular immunity via DNA sensor cGAS-STING-dependent induction of type I interferons. Immunity 2016, 44, 597–608. [Google Scholar] [CrossRef] [PubMed]
- Reineke, L.C.; Lloyd, R.E. The stress granule protein G3BP1 recruits protein kinase R to promote multiple innate immune antiviral responses. J. Virol. 2015, 89, 2575–2589. [Google Scholar] [CrossRef]
- Watkinson, R.E.; McEwan, W.A.; Tam, J.C.; Vaysburd, M.; James, L.C. TRIM21 Promotes cGAS and RIG-I Sensing of Viral Genomes during Infection by Antibody-Opsonized Virus. PLoS Pathog. 2015, 11, e1005253. [Google Scholar] [CrossRef] [PubMed]
- Lian, H.; Wei, J.; Zang, R.; Ye, W.; Yang, Q.; Zhang, X.N.; Chen, Y.D.; Fu, Y.Z.; Hu, M.M.; Lei, C.Q.; et al. Author Correction: ZCCHC3 is a co-sensor of cGAS for dsDNA recognition in innate immune response. Nat. Commun. 2021, 12, 5526. [Google Scholar] [CrossRef]
- Liao, C.Y.; Lei, C.Q.; Shu, H.B. PCBP1 modulates the innate immune response by facilitating the binding of cGAS to DNA. Cell Mol. Immunol. 2021, 18, 2334–2343. [Google Scholar] [CrossRef]
- Ding, C.; Song, Z.; Shen, A.; Chen, T.; Zhang, A. Small molecules targeting the innate immune cGAS-STING-TBK1 signaling pathway. Acta Pharm. Sin. B 2020, 10, 2272–2298. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Ma, G.; Li, X.; Zhao, J.; Zhao, Z.; Zeng, J. Activation of innate immune cGAS-STING pathway contributes to Alzheimer’s pathogenesis in 5xFAD mice. Nat. Aging 2023, 3, 202–212. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.M.; Zhao, Y.; Liu, J.; Fan, R.R.; Tang, Y.Q.; Guo, Z.Y.; Li, T. The role of the cGAS-STING signaling pathway in viral infections, inflammatory and autoimmune diseases. Acta Pharmacol. Sin. 2024. [CrossRef] [PubMed]
- Huang, Y.; Liu, B.; Sinha, S.C.; Amin, S.; Gan, L. Mechanism and therapeutic potential of targeting cGAS-STING signaling in neurological disorders. Mol. Neurodegener. 2023, 18, 79. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Li, T.; Li, X.D.; Chen, X.; Li, Q.Z.; Wight-Carter, M.; Chen, Z.J. Activation of cyclic GMP-AMP synthase by self-DNA causes autoimmune diseases. Proc. Natl. Acad. Sci. USA 2015, 112, E5699–E5705. [Google Scholar] [CrossRef] [PubMed]
- Chu, T.T.; Tu, X.; Yang, K.; Wu, J.; Repa, J.J.; Yan, N. Tonic prime-boost of STING signalling mediates Niemann-Pick disease type C. Nature 2021, 596, 570–575. [Google Scholar] [CrossRef] [PubMed]
- Mukai, K.; Konno, H.; Akiba, T.; Uemura, T.; Waguri, S.; Kobayashi, T.; Barber, G.N.; Arai, H.; Taguchi, T. Activation of STING requires palmitoylation at the Golgi. Nat. Commun. 2016, 7, 11932. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wu, P.; Du, Q.; Hanif, U.; Hu, H.; Li, K. cGAS-STING, an important signaling pathway in diseases and their therapy. MedComm 2024, 5, e511. [Google Scholar] [CrossRef]
- Kang, J.; Wu, J.; Liu, Q.; Wu, X.; Zhao, Y.; Ren, J. Post-translational modifications of STING: A potential therapeutic target. Front. Immunol. 2022, 13, 888147. [Google Scholar] [CrossRef]
- Haag, S.M.; Gulen, M.F.; Reymond, L.; Gibelin, A.; Abrami, L.; Decout, A.; Heymann, M.; van der Goot, F.G.; Turcatti, G.; Behrendt, R.; et al. Targeting STING with covalent small-molecule inhibitors. Nature 2018, 559, 269–273. [Google Scholar] [CrossRef]
- Hansen, A.L.; Buchan, G.J.; Ruhl, M.; Mukai, K.; Salvatore, S.R.; Ogawa, E.; Andersen, S.D.; Iversen, M.B.; Thielke, A.L.; Gunderstofte, C.; et al. Nitro-fatty acids are formed in response to virus infection and are potent inhibitors of STING palmitoylation and signaling. Proc. Natl. Acad. Sci. USA 2018, 115, E7768–E7775. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Cheng, T.; Huang, J.; Zhang, T.; Yin, H. 4-Octyl itaconate restricts STING activation by blocking its palmitoylation. Cell Rep. 2023, 42, 113040. [Google Scholar] [CrossRef]
- Humphries, F.; Shmuel-Galia, L.; Jiang, Z.; Zhou, J.Y.; Barasa, L.; Mondal, S.; Wilson, R.; Sultana, N.; Shaffer, S.A.; Ng, S.L.; et al. Targeting STING oligomerization with small-molecule inhibitors. Proc. Natl. Acad. Sci. USA 2023, 120, e2305420120. [Google Scholar] [CrossRef]
- Vinogradova, E.V.; Zhang, X.; Remillard, D.; Lazar, D.C.; Suciu, R.M.; Wang, Y.; Bianco, G.; Yamashita, Y.; Crowley, V.M.; Schafroth, M.A.; et al. An Activity-Guided Map of Electrophile-Cysteine Interactions in Primary Human T Cells. Cell 2020, 182, 1009–1026.e1029. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Zheng, M.; Wu, X.; Zhang, H.; Su, H.; Dang, Y.; Ma, M.; Wang, F.; Xu, J.; Chen, L.; et al. CDK inhibitor Palbociclib targets STING to alleviate autoinflammation. EMBO Rep. 2022, 23, e53932. [Google Scholar] [CrossRef] [PubMed]
- Abdolmaleky, H.M.; Zhou, J.R. Underlying mechanisms of brain aging and neurodegenerative diseases as potential targets for preventive or therapeutic strategies using phytochemicals. Nutrients 2023, 15, 3456. [Google Scholar] [CrossRef]
- Thomson, D.W.; Poeckel, D.; Zinn, N.; Rau, C.; Strohmer, K.; Wagner, A.J.; Graves, A.P.; Perrin, J.; Bantscheff, M.; Duempelfeld, B.; et al. Discovery of GSK8612, a highly selective and potent TBK1 inhibitor. ACS Med. Chem. Lett. 2019, 10, 780–785. [Google Scholar] [CrossRef]
- Gao, C.Q.; Chu, Z.Z.; Zhang, D.; Xiao, Y.; Zhou, X.Y.; Wu, J.R.; Yuan, H.; Jiang, Y.C.; Chen, D.; Zhang, J.C.; et al. Serine/threonine kinase TBK1 promotes cholangiocarcinoma progression via direct regulation of beta-catenin. Oncogene 2023, 42, 1492–1507. [Google Scholar] [CrossRef]
- Zeng, H.; Gao, Y.; Yu, W.; Liu, J.; Zhong, C.; Su, X.; Wen, S.; Liang, H. Pharmacological inhibition of STING/TBK1 signaling attenuates myeloid fibroblast activation and macrophage to myofibroblast transition in renal fibrosis. Front. Pharmacol. 2022, 13, 940716. [Google Scholar] [CrossRef]
- Barasa, L.; Chaudhuri, S.; Zhou, J.Y.; Jiang, Z.; Choudhary, S.; Green, R.M.; Wiggin, E.; Cameron, M.; Humphries, F.; Fitzgerald, K.A.; et al. Development of LB244, an irreversible STING antagonist. J. Am. Chem. Soc. 2023, 145, 20273–20288. [Google Scholar] [CrossRef]
- Hong, Z.; Mei, J.; Li, C.; Bai, G.; Maimaiti, M.; Hu, H.; Yu, W.; Sun, L.; Zhang, L.; Cheng, D.; et al. STING inhibitors target the cyclic dinucleotide binding pocket. Proc. Natl. Acad. Sci. USA 2021, 118, e2105465118. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.; Brault, A.; Vincent, F.; Weng, S.; Wang, H.; Dumlao, D.; Aulabaugh, A.; Aivazian, D.; Castro, D.; Chen, M.; et al. Discovery of PF-06928215 as a high affinity inhibitor of cGAS enabled by a novel fluorescence polarization assay. PLoS ONE 2017, 12, e0184843. [Google Scholar] [CrossRef] [PubMed]
- Padilla-Salinas, R.; Sun, L.; Anderson, R.; Yang, X.; Zhang, S.; Chen, Z.J.; Yin, H. Discovery of small-molecule cyclic GMP-AMP synthase inhibitors. J. Org. Chem. 2020, 85, 1579–1600. [Google Scholar] [CrossRef] [PubMed]
- Vincent, J.; Adura, C.; Gao, P.; Luz, A.; Lama, L.; Asano, Y.; Okamoto, R.; Imaeda, T.; Aida, J.; Rothamel, K.; et al. Small molecule inhibition of cGAS reduces interferon expression in primary macrophages from autoimmune mice. Nat. Commun. 2017, 8, 750. [Google Scholar] [CrossRef] [PubMed]
- Lama, L.; Adura, C.; Xie, W.; Tomita, D.; Kamei, T.; Kuryavyi, V.; Gogakos, T.; Steinberg, J.I.; Miller, M.; Ramos-Espiritu, L.; et al. Development of human cGAS-specific small-molecule inhibitors for repression of dsDNA-triggered interferon expression. Nat. Commun. 2019, 10, 2261. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Wu, B.; Chen, T.; Fan, C.; Zhao, J.; Xiong, C.; Feng, C.; Xiao, R.; Ding, C.; Tang, W.; et al. Synthesis and pharmacological evaluation of tetrahydro-gamma-carboline derivatives as potent anti-inflammatory agents targeting cyclic GMP-AMP synthase. J. Med. Chem. 2021, 64, 7667–7690. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.; Li, C.; Li, Y.; Yu, Q.; Yu, H.; Li, C.; Meng, W.; Zhu, J.; Wang, Q.; Wang, C.; et al. Perillaldehyde inhibition of cGAS reduces dsDNA-induced interferon response. Front. Immunol. 2021, 12, 655637. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.H.; Su, Z.Y.; Chae, J.I.; Kim, D.J.; Zhu, F.; Ma, W.Y.; Bode, A.M.; Yang, C.S.; Dong, Z. Epigallocatechin gallate suppresses lung cancer cell growth through Ras-GTPase-activating protein SH3 domain-binding protein 1. Cancer Prev. Res. 2010, 3, 670–679. [Google Scholar] [CrossRef]
- Liu, Z.S.; Cai, H.; Xue, W.; Wang, M.; Xia, T.; Li, W.J.; Xing, J.Q.; Zhao, M.; Huang, Y.J.; Chen, S.; et al. G3BP1 promotes DNA binding and activation of cGAS. Nat. Immunol. 2019, 20, 18–28. [Google Scholar] [CrossRef]
- Dai, J.; Huang, Y.J.; He, X.; Zhao, M.; Wang, X.; Liu, Z.S.; Xue, W.; Cai, H.; Zhan, X.Y.; Huang, S.Y.; et al. Acetylation blocks cGAS activity and inhibits self-DNA-induced autoimmunity. Cell 2019, 176, 1447–1460. [Google Scholar] [CrossRef]
- Wang, M.; Sooreshjani, M.A.; Mikek, C.; Opoku-Temeng, C.; Sintim, H.O. Suramin potently inhibits cGAMP synthase, cGAS, in THP1 cells to modulate IFN-beta levels. Future Med. Chem. 2018, 10, 1301–1317. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Woodward, J.J.; Sasaki, T.; Minie, M.; Elkon, K.B. Cutting edge: Antimalarial drugs inhibit IFN-beta production through blockade of cyclic GMP-AMP synthase-DNA interaction. J. Immunol. 2015, 194, 4089–4093. [Google Scholar] [CrossRef] [PubMed]
- Meric-Bernstam, F.; Sandhu, S.K.; Hamid, O.; Spreafico, A.; Kasper, S.; Dummer, R.; Shimizu, T.; Steeghs, N.; Lewis, N.; Talluto, C.C.; et al. Phase Ib study of MIW815 (ADU-S100) in combination with spartalizumab (PDR001) in patients (pts) with advanced/metastatic solid tumors or lymphomas. J. Clin. Oncol. 2019, 37, 2507. [Google Scholar] [CrossRef]
- Harrington, K.J.; Brody, J.; Ingham, M.; Strauss, J.; Cemerski, S.; Wang, M.; Tse, A.; Khilnani, A.; Marabelle, A.; Golan, T. Preliminary results of the first-in-human (FIH) study of MK-1454, an agonist of stimulator of interferon genes (STING), as monotherapy or in combination with pembrolizumab (pembro) in patients with advanced solid tumors or lymphomas. Ann. Oncol. 2018, 29, viii712. [Google Scholar] [CrossRef]
- Irvine, D.J.; Aung, A.; Silva, M. Controlling timing and location in vaccines. Adv. Drug. Deliv. Rev. 2020, 158, 91–115. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.R.; Corrales, L.; Gajewski, T.F. The STING pathway and the T cell-inflamed tumor microenvironment. Trends Immunol. 2015, 36, 250–256. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Dobbs, N.; Yang, K.; Yan, N. Interferon-ondependent activities of mammalian STING mediate antiviral response and tumor immune evasion. Immunity 2020, 53, 115–126.e115. [Google Scholar] [CrossRef] [PubMed]
- Le, R.Q.; Li, L.; Yuan, W.; Shord, S.S.; Nie, L.; Habtemariam, B.A.; Przepiorka, D.; Farrell, A.T.; Pazdur, R. FDA approval summary: Tocilizumab for treatment of chimeric antigen receptor T cell-Induced severe or life-threatening cytokine release syndrome. Oncologist 2018, 23, 943–947. [Google Scholar] [CrossRef]
- Xue, Y.; Bai, H.; Peng, B.; Fang, B.; Baell, J.; Li, L.; Huang, W.; Voelcker, N.H. Stimulus-cleavable chemistry in the field of controlled drug delivery. Chem. Soc. Rev. 2021, 50, 4872–4931. [Google Scholar] [CrossRef]
- Garland, K.M.; Sheehy, T.L.; Wilson, J.T. Chemical and biomolecular strategies for STING pathway activation in cancer immunotherapy. Chem. Rev. 2022, 122, 5977–6039. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, X.; Cai, L.; Yao, J.; Li, C.; Wang, X. Agonists and Inhibitors of the cGAS-STING Pathway. Molecules 2024, 29, 3121. https://doi.org/10.3390/molecules29133121
Yu X, Cai L, Yao J, Li C, Wang X. Agonists and Inhibitors of the cGAS-STING Pathway. Molecules. 2024; 29(13):3121. https://doi.org/10.3390/molecules29133121
Chicago/Turabian StyleYu, Xiaoxuan, Linxiang Cai, Jingyue Yao, Cenming Li, and Xiaoyong Wang. 2024. "Agonists and Inhibitors of the cGAS-STING Pathway" Molecules 29, no. 13: 3121. https://doi.org/10.3390/molecules29133121
APA StyleYu, X., Cai, L., Yao, J., Li, C., & Wang, X. (2024). Agonists and Inhibitors of the cGAS-STING Pathway. Molecules, 29(13), 3121. https://doi.org/10.3390/molecules29133121









