Self-Assembled Monolayers of Push–Pull Chromophores as Active Layers and Their Applications
Abstract
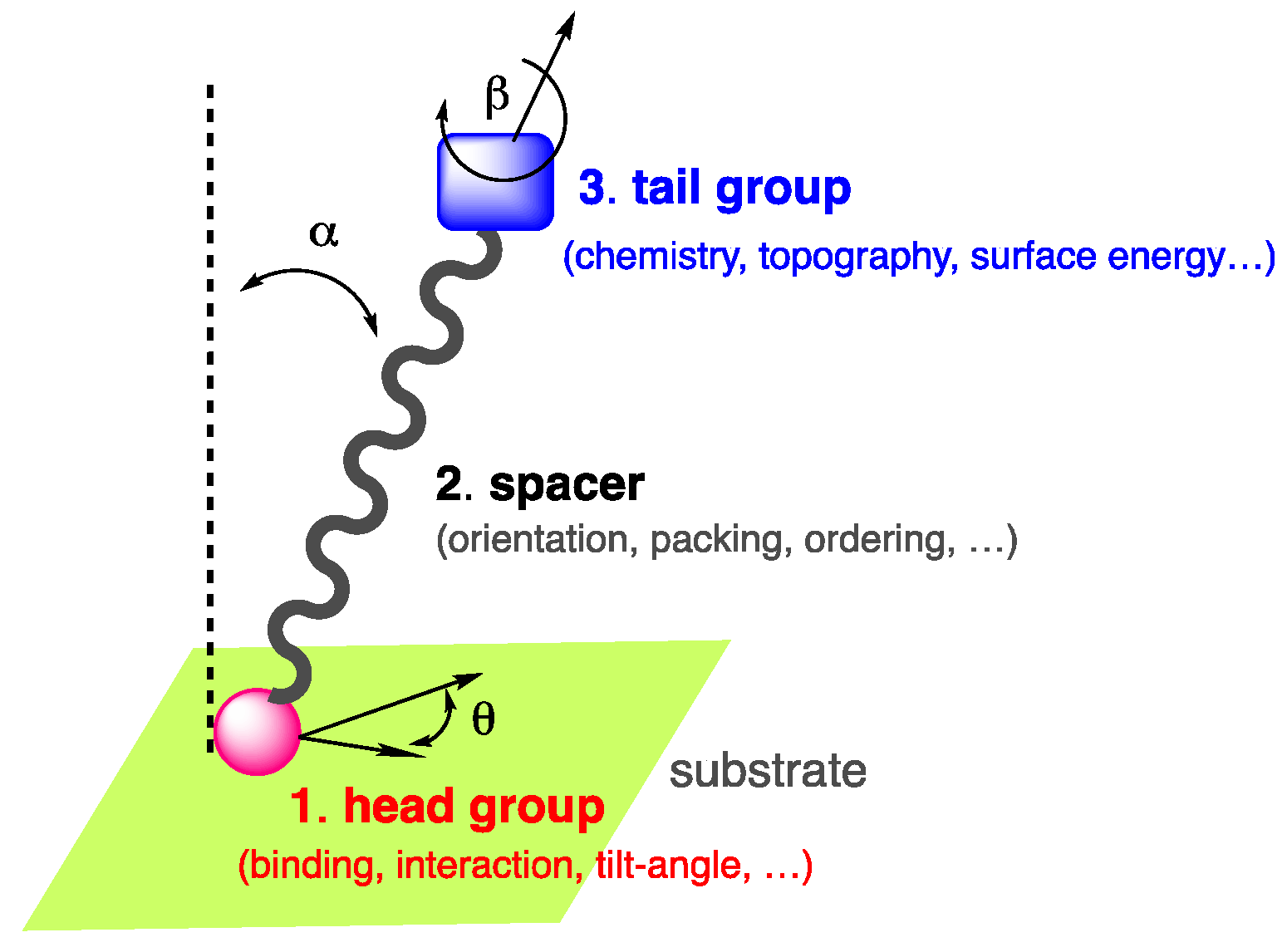
1. Introduction
2. SAMs from Push–Pull Chromophores for Dye-Sensitized Solar Cell Applications
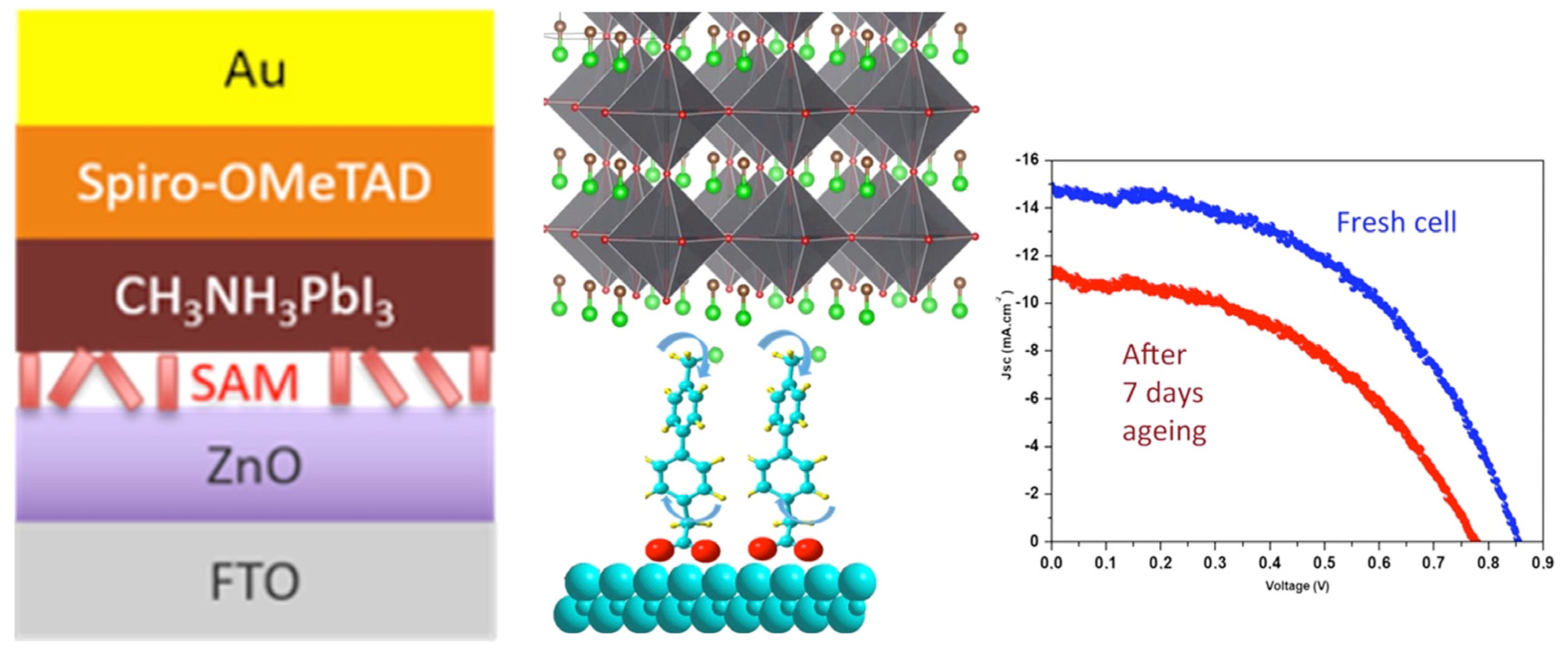
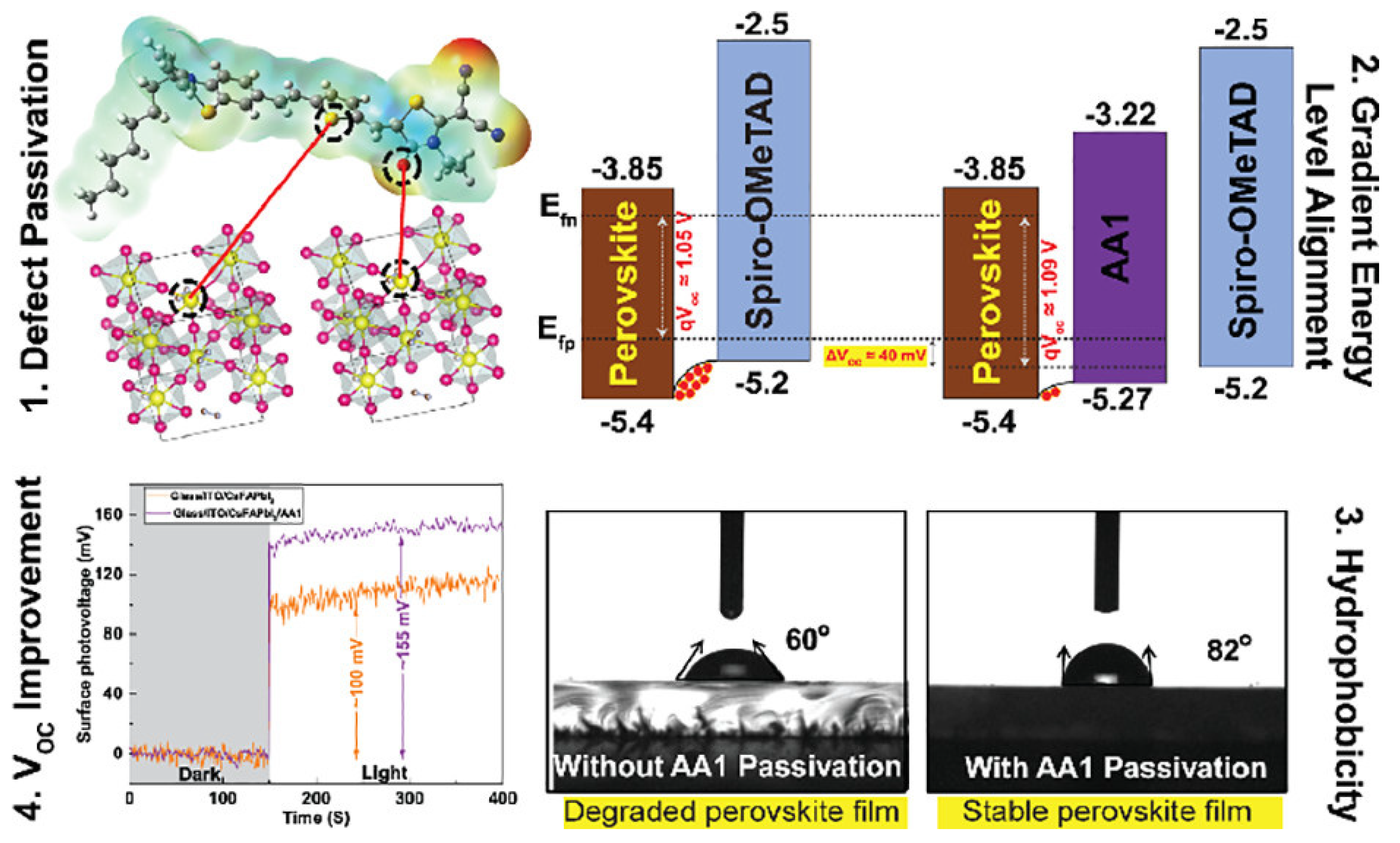
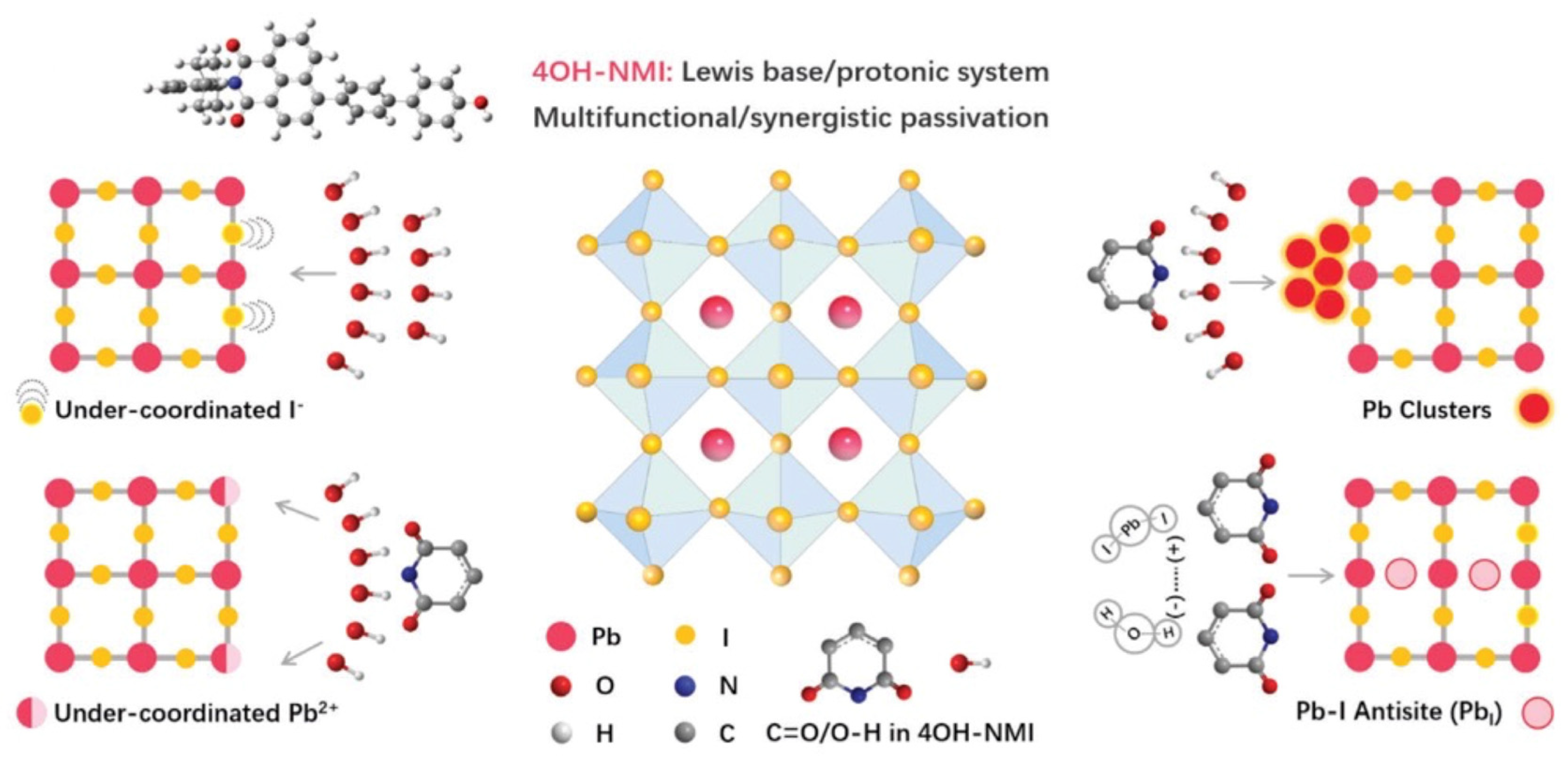
3. SAMs of Push–Pull Chromophores to Improve Perovskite Solar Cell Performances
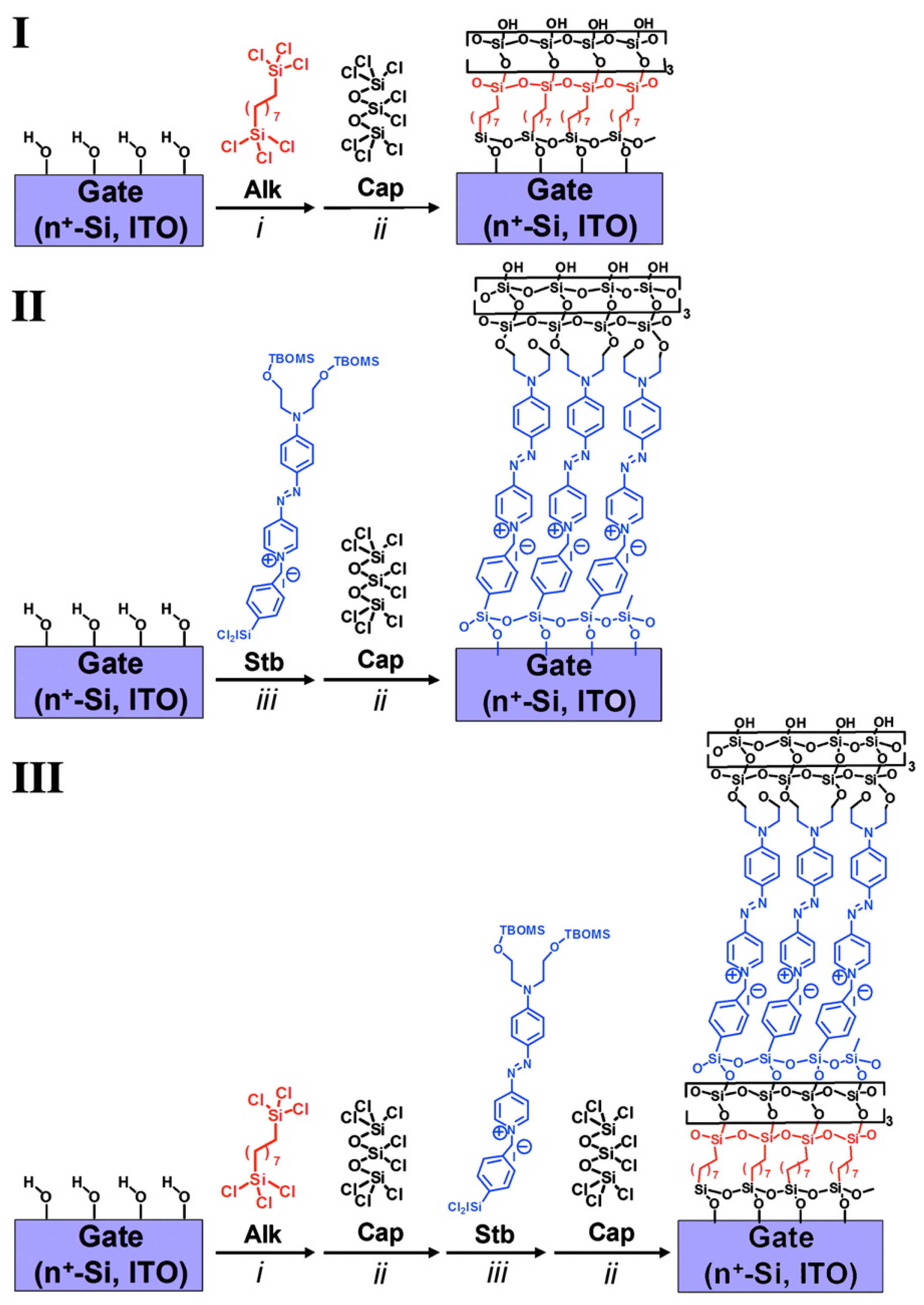
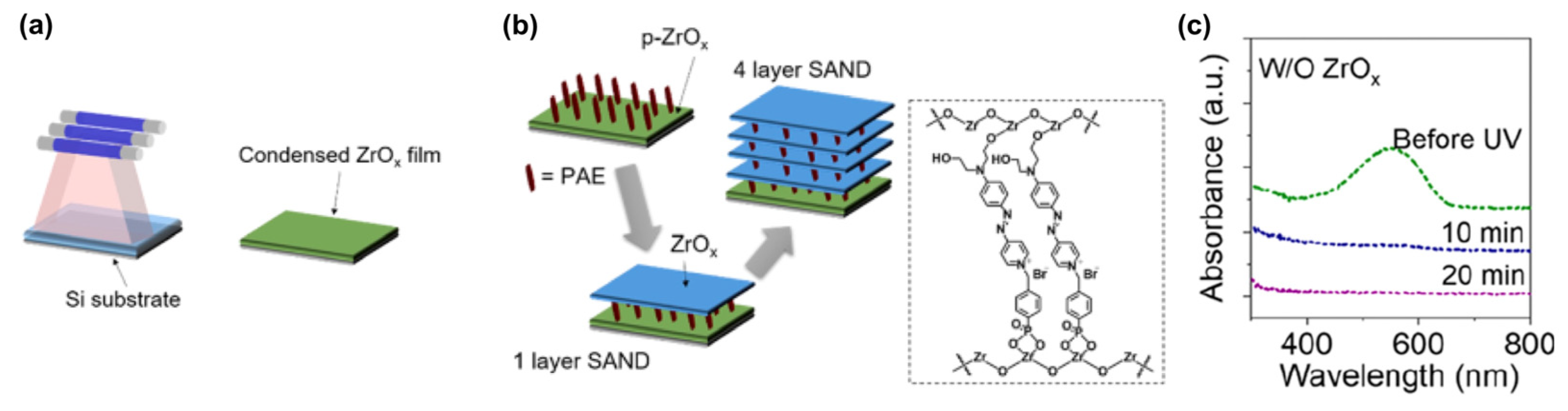
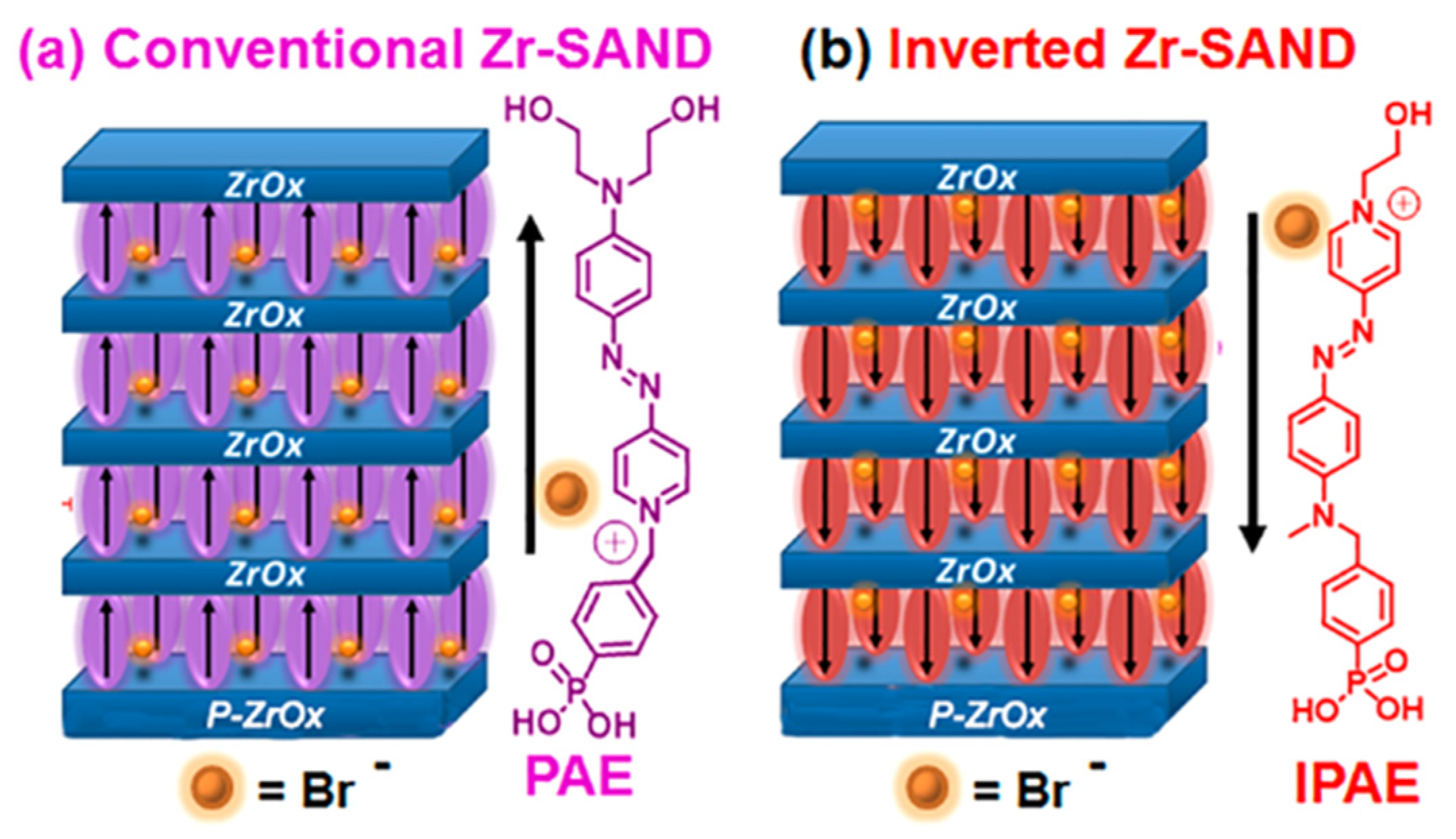
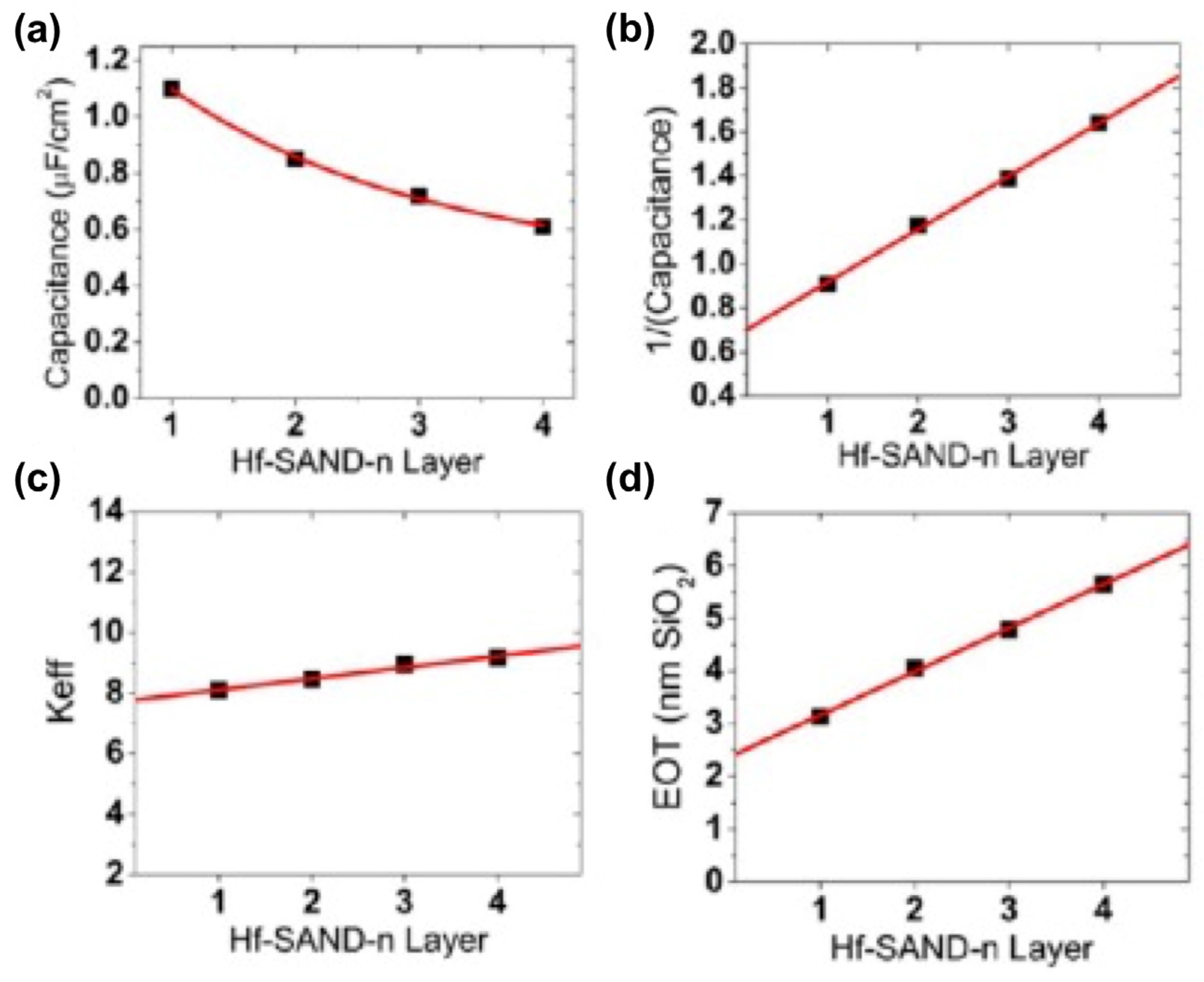
4. SAMs of Push–Pull Chromophores as Dielectric Materials
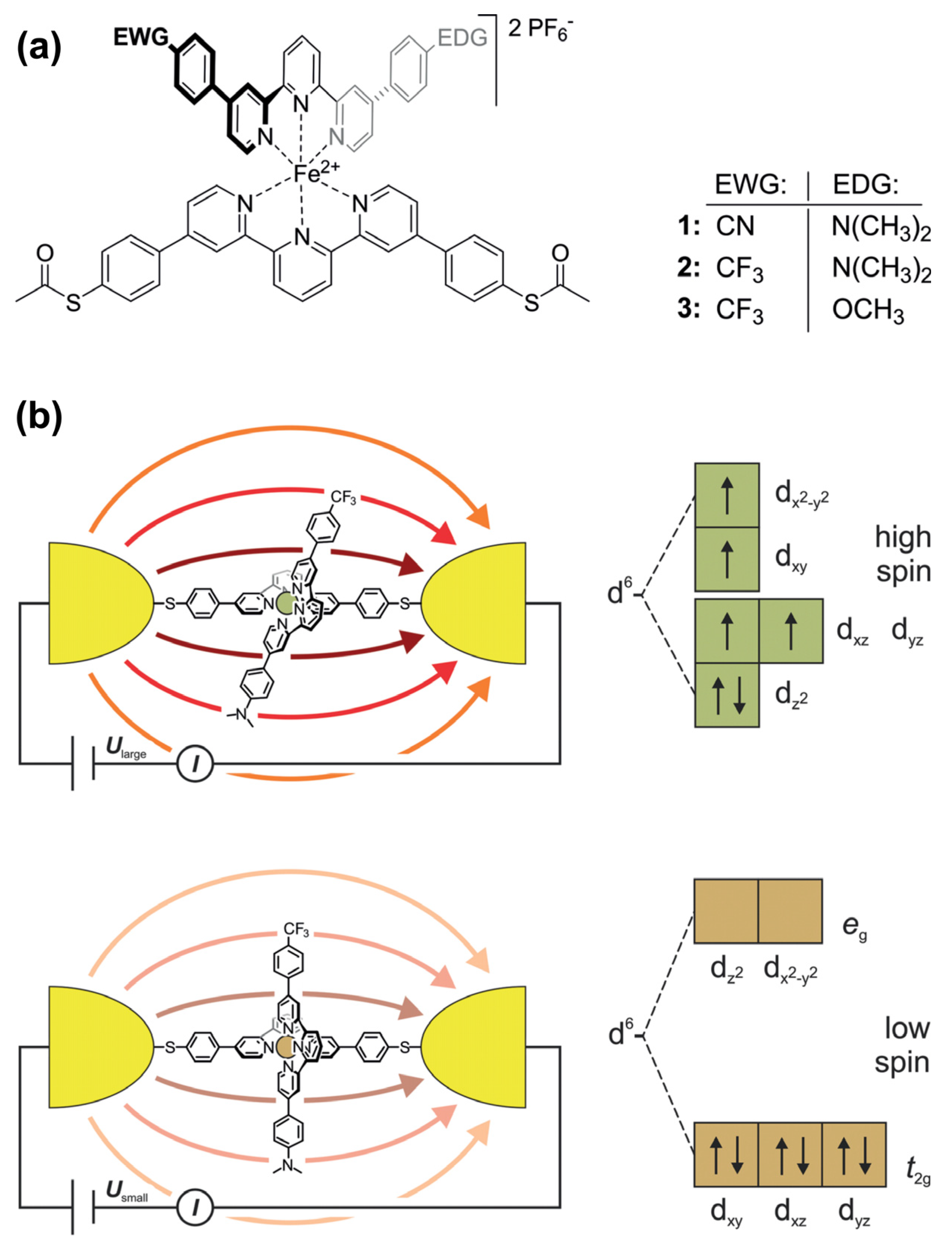
5. Various Electrical and Photonic Properties Generated by Push–Pull Chromophore Assemblies
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kelsall, R.; Hamley, I.W.; Geoghegan, M. Nanoscale Science and Technology; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Whitesides, G.M.; Grzybowski, B. Self-assembly at all scales. Science 2002, 295, 2418–2421. [Google Scholar] [CrossRef]
- Pelesko, J.A. Self-Assembly: The Science of Things That Put Themselves Together; Chapman & Hall/CRC: London, UK, 2007. [Google Scholar]
- Chai, Z.; Childress, A.; Busnaina, A.A. Directed assembly of nanomaterials for making nanoscale devices and structures: Mechanism and applications. ACS Nano 2022, 16, 17641–17686. [Google Scholar] [CrossRef]
- Ulman, A. Formation and structure of self-assembled monolayers. Chem. Rev. 1996, 96, 1533–1554. [Google Scholar] [CrossRef] [PubMed]
- Lehn, J.-M. Supramolecular chemistry. Science 1993, 260, 1762–1763. [Google Scholar] [CrossRef] [PubMed]
- Blodgett, K.B. Films built by depositing successive monomolecular layers on a solid surface. J. Am. Chem. Soc. 1935, 57, 1007–1022. [Google Scholar] [CrossRef]
- Blodgett, K.B.; Langmuir, I. Built-up films of barium stearate and their optical properties. Phys. Rev. 1937, 51, 0964–0982. [Google Scholar] [CrossRef]
- Bigelow, W.C.; Pickett, D.L.; Zisman, W.A. Oleophobic monolayers: Films adsorbed from solution in non-polar liquids. J. Colloid Sci. 1946, 1, 513–538. [Google Scholar] [CrossRef]
- Bigelow, W.C.; Glass, E.; Zisman, W.A. Oleophobic monolayers: Temperature effects and energy of adsorption. J. Colloid Sci. 1947, 2, 563–591. [Google Scholar] [CrossRef]
- Sagiv, J. Organized monolayers by adsorption. Formation and structure of oleophobic mixed monolayers on solid surfaces. J. Am. Chem. Soc. 1980, 102, 92–98. [Google Scholar] [CrossRef]
- Nuzzo, R.G.; Allara, D.L. Adsorption of bifunctional organic disulfides on gold surfaces. J. Am. Chem. Soc. 1983, 105, 4481–4483. [Google Scholar] [CrossRef]
- Xia, Y.; Whitesides, G.M. Soft litography. Annu. Rev. Mater. Sci. 1998, 28, 153–184. [Google Scholar] [CrossRef]
- Savig, J.; Nuzzo, R.G.; Allara, D.L.; Whitesides, G.M. Self-Assembled Monolayers (SAMs) on Solid Substrates: Molecular Coating to Control Surface Properties, Kavli Prize in Nanoscience. 2022. Available online: https://www.kavliprize.org/prizes/nanoscience/2022 (accessed on 19 January 2024).
- Love, J.C.; Estroff, L.A.; Kriebel, J.K.; Nuzzo, R.G.; Whitesides, G.M. Self-assembled monolayers of thiolates on metals as a form of nanotechnology. Chem. Rev. 2005, 105, 1103–1170. [Google Scholar] [CrossRef]
- Bain, C.D.; Whitesides, G.M. Formation of monolayers by the coadsorption of thiols on gold: Variation in the length of the alkyl chain. J. Am. Chem. Soc. 1989, 111, 7164–7175. [Google Scholar] [CrossRef]
- Claridge, S.A.; Liao, W.-S.; Thomas, J.C.; Zhao, Y.; Cao, H.H.; Cheunkar, S.; Serino, A.C.; Andrews, A.M.; Weiss, P.S. From the bottom up: Dimensional control and characterization in molecular monolayers. Chem. Soc. Rev. 2013, 42, 2725–2745. [Google Scholar] [CrossRef] [PubMed]
- Ariga, K. Organized Organic Ultrathin Films: Fundamentals and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Rogalska, E.; Bilewicz, R.; Brigaud, T.; El Moujahid, C.; Foulard, G.; Portella, C.; Stébé, M.J. Formation and properties of Langmuir and Gibbs monolayers: A comparative study using hydrogenated and partially fluorinated amphiphilic derivatives of mannitol. Chem. Phys. Lipids 2000, 105, 71–91. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Chen, J.; Oyola-Reynoso, S.; Thuo, M. The Porter-Whitesides discrepancy: Revisiting odd-even effects in wetting properties of n-alkanethiolate SAMs. Coatings 2015, 5, 1034–1055. [Google Scholar] [CrossRef]
- Liu, Y.; Katzbach, S.; Asyuda, A.; Das, S.; Terfort, A.; Zharnikov, M. Effect of substitution on the charge transport properties of oligophenylenethiolate self-assembled monolayers. Phys. Chem. Chem. Phys. 2022, 24, 27693–27704. [Google Scholar] [CrossRef]
- Aswal, D.K.; Lenfant, S.; Guerin, D.; Yakhmi, J.V.; Vuillaume, D. Self-assembled monolayers on silicon for molecular electronics. Anal. Chim. Acta 2006, 568, 84–108. [Google Scholar] [CrossRef]
- Cegiełka, D.M.; Kozieł, K.; Zharnikov, M.; Cyganik, P. Molecular engineering of technologically relevant surfaces- carboxylic acids on naturally oxidized Aluminium. Appl. Surf. Sci. 2023, 636, 157798. [Google Scholar] [CrossRef]
- Mamun, A.H.A.; Hahn, J.R. Effect of immersion temperature on self-assembled monolayers of octanethiol on Au(111). Surf. Sci. 2012, 606, 664–669. [Google Scholar] [CrossRef]
- Azzam, W. Effect of immersion time temperature on structure of the self-assembled monolayer of 1,2-diphenyldiselenide on gold surface. Mater. Chem. Phys. 2016, 180, 432–439. [Google Scholar] [CrossRef]
- Lee, N.-S.; Kang, H.; Seong, S.; Noh, J. Effect of immersion time on the structure of octanethiol self-assembled monolayers on Au(111) at elevated solution temperature. Bull. Korean Chem. Soc. 2019, 40, 1152–1153. [Google Scholar] [CrossRef]
- Chen, X.; Luais, E.; Darwish, N.; Ciampi, S.; Thordarson, P.; Gooding, J.J. Studies on the effect of solvents on self-assembled monolayers formed from organophosphonic acids on indium tin oxide. Langmuir 2012, 28, 9487–9495. [Google Scholar] [CrossRef]
- Chen, G.-S.; Chang, W.-H.; Chang, C.-C.; Chien, Y.-H.; Fang, J.-S.; Cheng, Y.-L. Structural models and barrier properties of amine-terminated trialkoxysilane monolayers incubated in nonpolar vs. polar protic solvents. Mater. Chem. Phys. 2021, 274, 125113. [Google Scholar] [CrossRef]
- Bunker, B.C.; Carpick, R.W.; Assink, R.A.; Thomas, M.L.; Hankins, M.G.; Voigt, J.A.; Sipola, D.; de Boer, M.P.; Gulley, G.L. The impact of solution agglomeration on the deposition of self-assembled monolayers. Langmuir 2000, 16, 7742–7751. [Google Scholar] [CrossRef]
- Rozlosnik, N.; Gerstenberg, M.; Larsen, N.B. Effect of solvents and concentration on the formation of a self-assembled monolayer of octodecylsiloxane on silicon(001). Langmuir 2003, 19, 1182–1188. [Google Scholar] [CrossRef]
- Vericat, C.; Vela, M.E.; Corthey, G.; Pensa, E.; Cortés, E.; Fonticelli, M.H.; Ibañez, F.; Benitez, G.E.; Carro, P.; Salvarezza, R.C. Self-assembled monolayers of thiolates on metals: A review article on sulfur-metal chemistry and surface structures. RSC Adv. 2014, 4, 27730–27754. [Google Scholar] [CrossRef]
- Wang, L.; Schubert, U.S.; Hoeppener, S. Surface chemical reactions on self-assembled silane-based monolayers. Chem. Soc. Rev. 2021, 50, 6507–6540. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, S.A. Self-assembled monolayers (SAMs) of carboxylic acids: An overview. Cent. Eur. J. Chem. 2011, 9, 369–378. [Google Scholar] [CrossRef]
- Bauer, T.; Schmaltz, T.; Lenz, T.; Halik, M.; Meyer, B.; Clark, T. Phosphonate- and carboxylate-based self-assembled monolayers for organic devices: A theoretical study of surface binding on aluminium oxide with experimental support. ACS Appl. Mater. Interfaces 2013, 5, 6073–6080. [Google Scholar] [CrossRef]
- Pujari, S.P.; Scheres, L.; Marcelis, A.T.M.; Zuilhof, H. Covalent surface modification of oxide surfaces. Angew. Chem. Int. Ed. 2014, 53, 2–36. [Google Scholar] [CrossRef] [PubMed]
- Engel, S.; Fritz, E.-C.; Ravoo, B.J. New trends in the functionalization of metallic gold: From organosulfur ligands to N-heterocyclic carbenes. Chem. Soc. Rev. 2017, 46, 2057–2075. [Google Scholar] [CrossRef] [PubMed]
- Kaur, G.; Thimes, R.L.; Camden, J.P.; Jenkins, D.M. Fundamentals and applications of N-heterocyclic carbene functionalized gold surfaces and nanoparticles. Chem. Commun. 2022, 58, 13188–13197. [Google Scholar] [CrossRef] [PubMed]
- Chinwangso, P.; Jamison, A.C.; Lee, T.R. Multidente adsorbates for self-assembled monolayer films. Acc. Chem. Res. 2011, 44, 511–519. [Google Scholar] [CrossRef]
- Wilbur, J.L.; Whitesides, G.M. Self-assembly and self-assembled monolayers in micro- and nanofabrication. In Nanotechnology; Timp, G., Ed.; Springer: New York, NY, USA, 1999; pp. 331–369. [Google Scholar]
- Lee, S.H.; Rho, W.-Y.; Park, S.J.; Kim, J.; Kwon, O.S.; Jun, B.-H. Multifunctional self-assembled monolayers via microcontact printing and degas-driven flow guided patterning. Sci. Rep. 2018, 8, 16763. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Bennett, R.K.A.; Xu, Y.; Rather, A.M.; Li, S.; Cheung, T.C.; Bhanji, A.; Kreder, M.J.; Daniel, D.; Adera, S.; et al. Wettability-based ultrasensitive detection of amphiphiles through directed concentration at disordered regions in self-assembled monolayers. Proc. Natl. Acad. Sci. USA 2022, 119, e2211042119. [Google Scholar] [CrossRef] [PubMed]
- Yi, R.; Mao, Y.; Shen, Y.; Chen, L. Self-assembled monolayers for batteries. J. Am. Chem. Soc. 2021, 143, 12897–12912. [Google Scholar] [CrossRef] [PubMed]
- Carrara, S.; Rouvier, F.; Auditto, S.; Brunel, F.; Jeanneau, C.; Camplo, M.; Sergent, M.; About, I.; Bolla, J.-M.; Raimundo, J.-M. Nanoarchitectonics of electrically activable phosphonium self-assembled monolayers to efficiently kill and tackle bacterial infections on demand. Int. J. Mol. Sci. 2022, 23, 2183. [Google Scholar] [CrossRef]
- Ali, F.; Roldán-Carmona, C.; Sohail, M.; Nazeeruddin, M.K. Applications of self-assembled monolayers for perovskite solar cells interface engineering to address efficiency and stability. Adv. Energy Mater. 2020, 10, 2002989. [Google Scholar] [CrossRef]
- He, J.; Tao, L.; Xian, W.; Arbaugh, T.; Li, Y. Molecular self-assembled monolayers anomalously enhance thermal conductance across polymer-semiconductor interfaces. Nanoscale 2022, 14, 17681–17693. [Google Scholar] [CrossRef]
- Casalini, S.; Bortolotti, C.A.; Leonardi, F.; Biscarini, F. Self-assembled monolayers in organic electronics. Chem. Soc. Rev. 2017, 46, 40–71. [Google Scholar] [CrossRef]
- Singh, M.; Kaur, N.; Comini, E. Role of self-assembled monolayers in electronic devices. J. Mater. Chem. C 2020, 8, 3938–3955. [Google Scholar] [CrossRef]
- Hussey, K.A.; Hadyniak, S.E.; Johnston, R.J., Jr. Patterning and development of photoreceptors in the human retina. Front. Cell Dev. Biol. 2022, 10, 878350. [Google Scholar] [CrossRef]
- Borchert, J.W.; Peng, B.Y.; Letzkus, F.; Burghartz, J.N.; Chan, P.K.L.; Zojer, K.; Ludwigs, S.; Klauk, H. Small contact resistance and high-frequency operation of flexible low-voltage inverted coplanar organic transistors. Nat. Commun. 2019, 10, 1119. [Google Scholar] [CrossRef]
- Sauter, E.; Terfort, A.; Zharnikov, M. Pronounced solvent effect on the composition of binary self-assembled monolayers with embedded dipole moments. J. Phys. Chem. C 2020, 124, 28596–28604. [Google Scholar] [CrossRef]
- Abu-Husein, T.; Schuster, S.; Egger, D.A.; Kind, M.; Santowski, T.; Wiesner, A.; Chiechi, R.; Zojer, E.; Terfort, A.; Zharnikov, M. The effect of embedded dipoles in aromatic self-assembled monolayers. Adv. Funct. Mater. 2015, 25, 3943–3957. [Google Scholar] [CrossRef]
- Azzam, W.; Subaihi, A. Influence of an alkyl spacer on the formation and structure of 4-fluorobenthiol and 4-fluorobenzenemethanethiol self-assembled monolayers on Au (111). Surf. Interfaces 2020, 20, 100544. [Google Scholar] [CrossRef]
- Khasbaatar, A.; Xu, Z.; Lee, J.-H.; Campillo-Alvarado, G.; Hwang, C.; Onusaitis, B.N.; Diao, Y. From solution to thin film: Molecular assembly of p-conjugated systems ans impact on (opto) electronic properties. Chem. Rev. 2023, 123, 8395–8487. [Google Scholar] [CrossRef]
- Liu, J.; Ouyang, C.; Huo, F.; He, W.; Cao, A. Progress in the enhancement of electro-optic coefficients and orientation stability for organic second-order nonlinear optical materials. Dyes Pigm. 2020, 181, 108509. [Google Scholar] [CrossRef]
- Gadenne, V.; Grenier, B.; Praveen, C.; Marsal, P.; Valmalette, J.C.; Patrone, L.; Raimundo, J.-M. Combined SERS/DFT studies of push-pull chromophores self-assembled monolayers: Insights of their surface orientation. Phys. Chem. Chem. Phys. 2019, 21, 25865–25871. [Google Scholar] [CrossRef] [PubMed]
- Burěs, F. Fundamental aspects of property tuning in push-pull molecules. RSC Adv. 2014, 4, 58826–58851. [Google Scholar] [CrossRef]
- Klikar, M.; Solanke, P.; Tydlitát, J.; Burěs, F. Alphabet-inspired design of (hetero)aromatic push-pull chromophores. Chem. Rec. 2016, 16, 1886–1905. [Google Scholar] [CrossRef] [PubMed]
- Gámez-Valenzuela, S.; Neusser, D.; Benitez-Martin, C.; Najera, F.; Guadix, J.A.; Moreno-Yruela, C.; Villacampa, B.; Ponce Ortiz, R.; Ludwigs, S.; Andreu, R.; et al. V-shaped pyranylidene/triphenylamine-based chromophores with enhanced photophysical, electrochemical and nonlinear optical properties. Mater. Adv. 2021, 2, 4255–4263. [Google Scholar] [CrossRef]
- Kalinin, A.A.; Islamova, L.N.; Sharipova, S.M.; Fazleeva, G.M.; Gaysin, A.I.; Shmelev, A.G.; Simanchuk, A.E.; Turgeneva, S.A.; Sharipova, A.V.; Mukhtarov, A.S.; et al. Quadratic nonlinear optical response of composite polymer materials based on push-pull quinoxaline chromophores with various groups in the aniline donor moiety. New J. Chem. 2023, 47, 2296–2306. [Google Scholar] [CrossRef]
- Malytskyi, V.; Simon, J.J.; Patrone, L.; Raimundo, J.-M. Thiophene-based push–pull chromophores for small molecule organic solar cells (SMOSCs). RSC Adv. 2015, 5, 354–397. [Google Scholar] [CrossRef]
- Raymakers, J.; Krysova, H.; Artemenko, A.; Cermak, J.; Nicley, S.S.; Verstappen, P.; Gielen, S.; Kromka, A.; Haenen, K.; Kavan, L.; et al. Functionalization of boron-doped diamond with a push–pull chromophore via Sonogashira and CuAAC chemistry. RSC Adv. 2018, 8, 33276–33290. [Google Scholar] [CrossRef]
- Unny, D.; Kandregula, G.R.; Sivanadanam, J.; Ramanujam, K. Molecular engineering of pyrene carbazole dyes with a single bond and double bond as the mode of linkage. New J. Chem. 2020, 44, 16511–16525. [Google Scholar] [CrossRef]
- Lindh, L.; Gordivska, O.; Persson, S.; Michaels, H.; Fan, H.; Chábera, P.; Rosemann, N.W.; Kumar Gupta, A.; Benesperi, I.; Uhlig, J.; et al. Dye-sensitized solar cells based on Fe N-heterocyclic carbene photosensitizers with improved rod-like push-pull functionality. Chem. Sci. 2021, 12, 16035–16053. [Google Scholar] [CrossRef]
- James, R.D.; Algahtani, L.S.; Mallows, J.; Flint, H.V.; Waddell, P.G.; Woodford, O.J.; Gibson, E.A. Pentafluorosulfanyl-functionalised BODIPY push–pull dyes for p-type dye-sensitized solar cells. Sustain. Energy Fuels 2023, 7, 1494–1501. [Google Scholar] [CrossRef]
- Fauvel, S.; Riquelme, A.J.; Castán, J.-M.A.; Mwalukuku, W.M.; Kervella, Y.; Challuri, V.K.; Sauvage, F.; Narbey, S.; Maldivi, P.; Demadrille, R. Push-pull photochromic dyes for semi-transparent solar cells with light-adjustable optical properties and high color-rendering index. Chem. Sci. 2023, 14, 8497–8506. [Google Scholar] [CrossRef]
- D’Annibale, V.; Chen, C.G.; Bonomo, M.; Dini, D.; D’Abramo, M. P1 push-pull dye as a case study in QM/MM theoretical characterization for dye-sensitized solar cell organic chromophores. ChemistrySelect 2023, 8, e20220490. [Google Scholar] [CrossRef]
- Ashwell, G.J.; Tyrell, W.D.; Whittam, A.J. Molecular rectification: Seld-assembled monoloayers in which donor-(p-bridge)-acceptor moities are centrally located and symmetrically coupled to both gold electrodes. J. Am. Chem. Soc. 2004, 126, 7102–7110. [Google Scholar] [CrossRef]
- Malytskyi, V.; Gadenne, V.; Ksari, Y.; Patrone, L.; Raimundo, J.-M. Synthesis and characterization of thiophene-based push-pull chromophores for tuning the electrical and optical properties of surfaces with controlled SAM formation. Tetrahedron 2017, 73, 5738–5744. [Google Scholar] [CrossRef]
- Abdellaha, I.M.; El-Shafei, A. Synthesis and characterization of novel tetra anchoring A2-D-D-D-A2 architecture sensitizers for efficient dye-sensitized solar cells. Sol. Energy 2020, 198, 25–35. [Google Scholar] [CrossRef]
- Gholamrezaie, F.; Kirkus, M.; Mathijissen, S.G.J.; de Leeuw, D.M.; Meskers, S.C.J. Photophysics of self-assembled monolayers of a π-conjugated quinquethiophene derivative. J. Phys. Chem. A 2012, 116, 7645–7650. [Google Scholar] [CrossRef][Green Version]
- Vijayaraghavan, R.K.; Gholamrezaie, F.; Meskers, S.C.J. Photovoltaic effect in self-assembled molecular monolayers on gold: Influence of orbital energy level alignment on short-circuits current generation. J. Phys. Chem. C 2013, 117, 16820–16829. [Google Scholar] [CrossRef]
- Keremane, K.S.; Planchat, A.; Pellegrin, Y.; Jacquemin, D.; Odobel, F.; Adhikari, A.V. Push-pull phenoxazine-based sensitizers for p-type DSSCs: Effect of acceptor units on photovoltaic performance. ChemSusChem 2022, 15, e202200520. [Google Scholar] [CrossRef]
- Paul, D.; Sarkar, U. Designing of PC31BM-based acceptors for dye-sensitized solar cell. J. Phys. Org. Chem. 2022, 36, e4419. [Google Scholar] [CrossRef]
- Mustafa, F.M.; Latif, M.K.A.; Khalek, A.A.A. Theoretical investigation on the influence of electron-accepting substitutions in modulating the optoelectronic properties of dye-sensitized solar cells. Mol. Phys. 2023, 121, e2245060/1–e2245060/12. [Google Scholar] [CrossRef]
- Avhad, K.; Jadhav, M.; Patil, D.; Chowdhury, T.H.; Bedja, I.; Sekar, N. Rhodanine-3-acetic acid containing D–π–A push-pull chromophores: Effect of methoxy group on the performance of dye-sensitized solar cells. Org. Electron. 2019, 65, 386–393. [Google Scholar] [CrossRef]
- Belen Marco, A.; Martinez de Baroja, N.; Andres-Castan, J.M.; Franco, S.; Andreu, R.; Villacampa, B.; Orduna, J.; Garin, J. Pyranylidene/thienothiophene-based organic sensitizers for dye-sensitized solar cells. Dyes Pigm. 2019, 161, 205–213. [Google Scholar] [CrossRef]
- Gümüşgöz Çelik, G.; Tunç, G.; Lafzi, F.; Saracoglu, N.; Seçkin Arslan, B.; Nebioğlu, M.; Şişman, I.; Gül Gürek, A. Influence of spacer and donor groups as tetraphenylethylene or triphenylamine in asymmetric zinc phthalocyanine dyes for dye-sensitized solar cells. J. Photochem. Photobiol. A 2023, 444, 114962. [Google Scholar] [CrossRef]
- Trabelsi, S.; Kouki, N.; Seydou, M.; Maurel, F.; Tangour, B. Intramolecular path determination of active electrons on push-pull oligocarbazole dyes-sensitized solar cells. ChemistryOpen 2019, 8, 580–588. [Google Scholar] [CrossRef]
- Dovaa, D.; Cauteruccioa, S.; Manfredib, N.; Pragerc, S.; Dreuwc, A.; Arnaboldia, S.; Mussinia, P.R.; Licandroa, E.; Abbotto, A. An unconventional helical push-pull system for solar cells. Dyes Pigm. 2019, 161, 382–388. [Google Scholar] [CrossRef]
- Lee, M.W.; Kim, J.Y.; Lee, H.G.; Gil Cha, H.; Lee, D.H.; Jae Ko, M. Effects of structure and electronic properties of D–π–A organic dyes on photovoltaic performance of dye-sensitized solar cells. J. Energy Chem. 2021, 54, 208–216. [Google Scholar] [CrossRef]
- An, J.; Tian, Z.; Zhang, L.; Yang, X.; Cai, B.; Yu, Z.; Zhang, L.; Hagfeldt, A.; Sun, L. Supramolecular Co-adsorption on TiO2 to enhance the efficiency of dye-sensitized solar cells. J. Mater. Chem. A 2021, 9, 13697–13703. [Google Scholar] [CrossRef]
- Maffeis, V.; Dogan, H.; Cassette, E.; Jousselme, B.; Gustavsson, T. Role of electronic relaxation in the injection process of organic push–pull dyes in complete dye-sensitized solar cells. J. Phys. Chem. Lett. 2019, 10, 5076–5081. [Google Scholar] [CrossRef]
- Kim, C.; Kim, T.W.; Kim, S.; Oh, I.; Wonneberger, H.; Yoon, K.; Kwak, M.; Kim, J.; Kim, J.; Li, C.; et al. Molecular-level understanding of excited states of N-annulated rylene dye for dye-sensitized solar sells. J. Phys. Chem. C 2020, 124, 22993–23003. [Google Scholar] [CrossRef]
- Prakash, K.; Alsaleh, A.Z.; Neeraj; Rathi, P.; Sharma, A.; Sankar, M.; D’Souza, F. Synthesis, spectral, electrochemical and photovoltaic studies of A3B porphyrinic dyes having peripheral donors. Chem. Phys. Chem. 2019, 20, 2627–2634. [Google Scholar] [CrossRef]
- Demirak, K.; Can, M.; Özsoy, C.; Yiğit, M.Z.; Gültekin, B. Synthesis and photovoltaic characterization of triarylamine-substituted quinoxaline push-pull push-pull dyes to improve the performance of dye-sensitized solar cells. Turk. J. Chem. 2017, 41, 309–322. [Google Scholar] [CrossRef]
- Fernandez-Ariza, J.; Urbani, M.; Graetzel, M.; Rodriguez-Morgade, M.S.; Nazeeruddin, M.K.; Torres, T. An unsymmetrical, push–pull porphyrazine for dye-sensitized solar cells. ChemPhotoChem 2017, 1, 164–166. [Google Scholar] [CrossRef]
- Medina, D.-P.; Fernández-Ariza, J.; Urbani, M.; Sauvage, F.; Torres, T.; Rodríguez-Morgade, M.S. Tuning the acceptor unit of push–pull porphyrazines for dye-sensitized solar cells. Molecules 2021, 26, 2129. [Google Scholar] [CrossRef]
- Shi, W.J.; Kinoshita, T.; Ng, D.K.P. Push–pull distyryl boron dipyrromethenes as near-infrared sensitizers for dye-sensitized solar cells. Asian J. Org. Chem. 2017, 6, 1476–1485. [Google Scholar] [CrossRef]
- Urbani, M.; Ragoussi, M.E.; Nazeeruddin, M.K.; Torres, T. Phthalocyanines for dye-sensitized solar cells. Coord. Chem. Rev. 2019, 381, 1–64. [Google Scholar] [CrossRef]
- Ghazal, B.; Azizi, K.; Ewies, E.F.; Youssef, A.S.A.; Mwalukuku, V.M.; Demadrille, R.; Torres, T.; Makhseed, S. Push–pull zinc phthalocyanine bearing hexa-tertiary substituted carbazolyl donor groups for dye-sensitized solar cells. Molecules 2020, 25, 1692. [Google Scholar] [CrossRef]
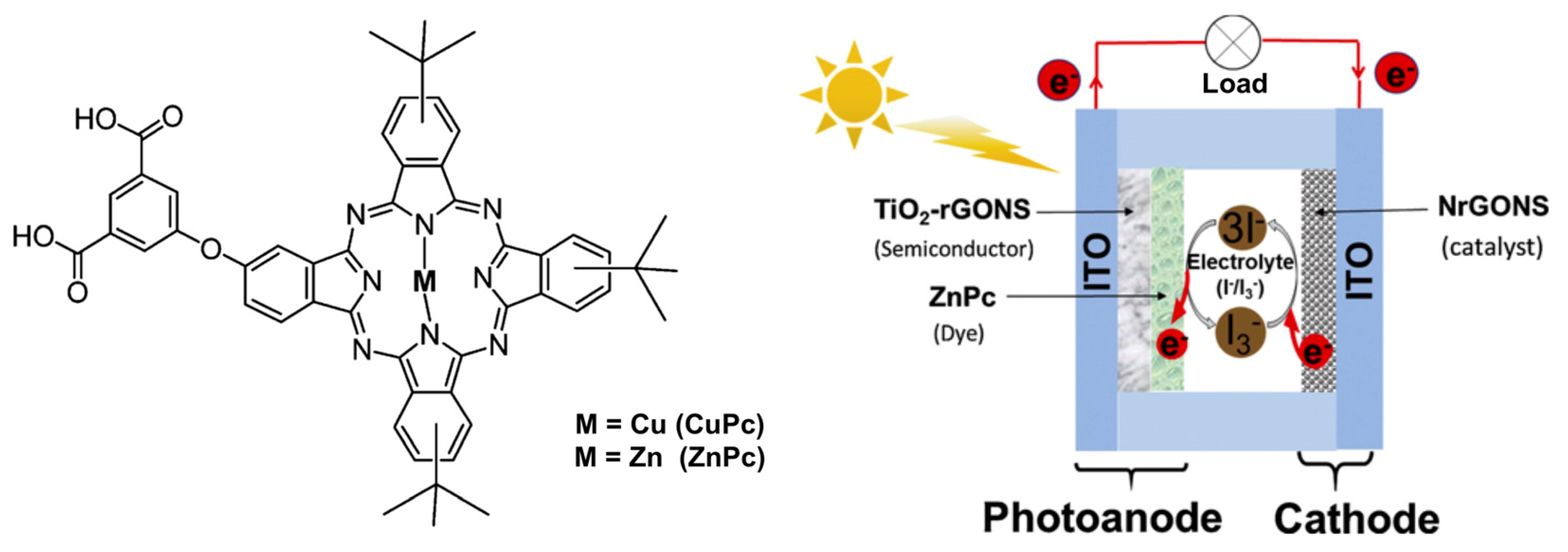
- Chindeka, F.; Mashazi, P.; Britton, J.; Oluwole, D.O.; Mapukata, S.; Nyokong, T. Fabrication of dye-sensitized solar cells based on push-pull asymmetrical substituted zinc and copper phthalocyanines and reduced graphene oxide nanosheets. J. Photochem. Photobiol. A 2020, 399, 112612. [Google Scholar] [CrossRef]
- Urbani, M.; Gratzel, M.; Nazeeruddin, M.K.; Torres, T. Meso-substituted porphyrins for dye-sensitized solar cells. Chem. Rev. 2014, 114, 12330–12396. [Google Scholar] [CrossRef]
- Birel, O.; Nadeem, S.; Duman, H. Porphyrin-based dye-sensitized solar cells (DSSCs): A review. J. Fluoresc. 2017, 27, 1075–1085. [Google Scholar] [CrossRef]
- Lu, J.; Liu, S.; Wang, M. Push-pull zinc porphyrins as light-harvesters for efficient dye-sensitized solar cells. Front. Chem. 2018, 6, 541. [Google Scholar] [CrossRef]
- Higashino, T.; Imahori, H. Porphyrins as excellent dyes for dye-sensitized solar cells: Recent developments and insights. Dalton Trans. 2015, 44, 448–463. [Google Scholar] [CrossRef]
- Song, H.; Li, X.; Ågren, H.; Xie, Y. Branched and linear alkoxy chains-wrapped push-pull porphyrins for developing efficient dye-sensitized solar cells. Dyes Pigm. 2017, 137, 421–429. [Google Scholar] [CrossRef]
- Higashino, T.; Kawamoto, K.; Sugiura, K.; Fujimori, Y.; Tsuji, Y.; Kurotobi, K.; Ito, S.; Imahori, H. Effects of bulky substituents of push–pull porphyrins on photovoltaic properties of dye-sensitized solar cells. ACS Appl. Mater. Interfaces 2016, 8, 15379–15390. [Google Scholar] [CrossRef] [PubMed]
- Yella, A.; Lee, H.-W.; Tsao, H.N.; Yi, C.; Chandiran, A.K.; Nazeeruddin, M.K.; Diau, E.W.-G.; Yeh, C.-Y.; Zakeeruddin, S.M.; Gratzel, M. Porphyrin-sensitized solar cells with cobalt (II/III)–based redox electrolyte exceed 12 percent efficiency. Science 2011, 334, 629–634. [Google Scholar] [CrossRef] [PubMed]
- Higashino, T.; Iiyama, H.; Nishimura, I.; Imahori, H. Exploration on the combination of push-pull porphyrin dyes and copper(I/II) redox shuttles toward high-performance dye-sensitized solar cells. Chem. Lett. 2020, 49, 936–939. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, J.C.; Li, R.; Ci, C.G. Different metal upper porphyrin based self-assembly sensitizers for application in efficient dye-sensitized solar cells. Polyhedron 2022, 211, 115573. [Google Scholar] [CrossRef]
- Higashino, T.; Sugiura, K.; Namikawa, K.; Imahori, H. Energy level tuning of push-pull porphyrin sensitizer by trifluoromethyl group for dye-sensitized solar cells. J. Porphyr. Phthalocyanines 2023, 27, 145–156. [Google Scholar] [CrossRef]
- Hu, Y.; Webre, W.A.; Thomas, M.B.; Moss, A.; Hancock, S.N.; Schaffner, J.; D’Souza, F.; Wang, H. β-Functionalized push–pull opp-dibenzoporphyrins as sensitizers for dye-sensitized solar cells: The role of the phenylethynyl bridge. J. Mater. Chem. A 2019, 7, 10712–10722. [Google Scholar] [CrossRef]
- Mattew, S.; Yella, A.; Gao, P.; Humphry-Baker, R.; Curchod, B.F.E.; Ashari-Astani, N.; Tavernelli, I.; Rothlisberger, U.; Nazeeruddin, M.K.; Gratzel, M. Dye-sensitized solar cells with 13% efficiency achieved through the molecular engineering of porphyrin sensitizers. Nat. Chem. 2014, 6, 242–247. [Google Scholar] [CrossRef]
- Yella, A.; Mai, C.-L.; Zakeeruddin, S.M.; Chang, S.-N.; Hsieh, C.-H.; Yeh, C.-Y.; Gratzel, M. Molecular engineering of push–pull porphyrin dyes for highly efficient dye-sensitized solar cells: The role of benzene spacers. Angew. Chem. Int. Ed. 2014, 53, 2973–2977. [Google Scholar] [CrossRef]
- Panagiotakis, S.; Giannoudis, E.; Charisiadis, A.; Paravatou, R.; Lazaridi, M.-E.; Kandyli, M.; Ladomenou, K.; Angaridis, P.A.; Bertrand, H.; Sharma, G.D.; et al. Increased efficiency of dye-sensitized solar cells by incorporation of a π-spacer in donor–acceptor zinc porphyrins bearing cyanoacrylic acid as an anchoring group. Eur. J. Inorg. Chem. 2018, 20–21, 2369–2379. [Google Scholar] [CrossRef]
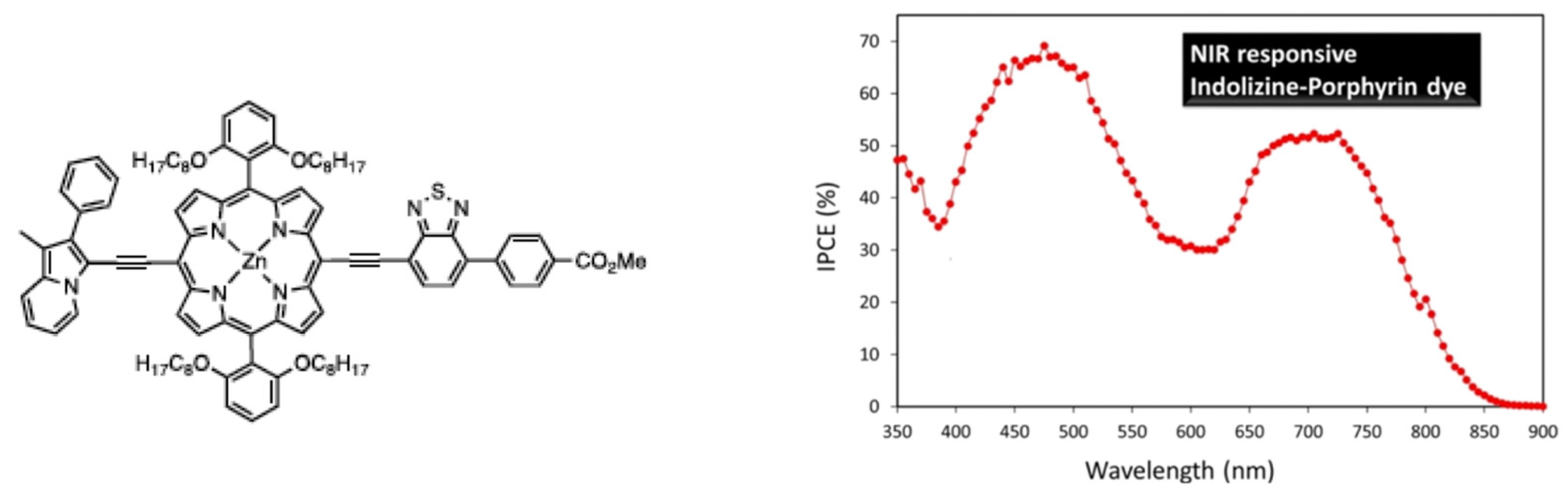
- Cheema, H.; Baumann, A.; Loya, E.K.; Brogdon, P.; McNamara, L.E.; Carpenter, C.A.; Hammer, N.I.; Mathew, S.; Risko, C.; Delcamp, J.H. Near-infrared-absorbing indolizine-porphyrin push–pull dye for dye-sensitized solar cells. ACS Appl. Mater. Interfaces 2019, 11, 16474–16489. [Google Scholar] [CrossRef] [PubMed]
- Milan, R.; Selopal, G.S.; Cavazzini, M.; Orlandi, S.; Boaretto, R.; Caramori, S.; Concina, I.; Pozzi, G. Zinc phthalocyanines as light harvesters for SnO2-based solar cells: A case study. Sci. Rep. 2020, 10, 1176. [Google Scholar] [CrossRef] [PubMed]
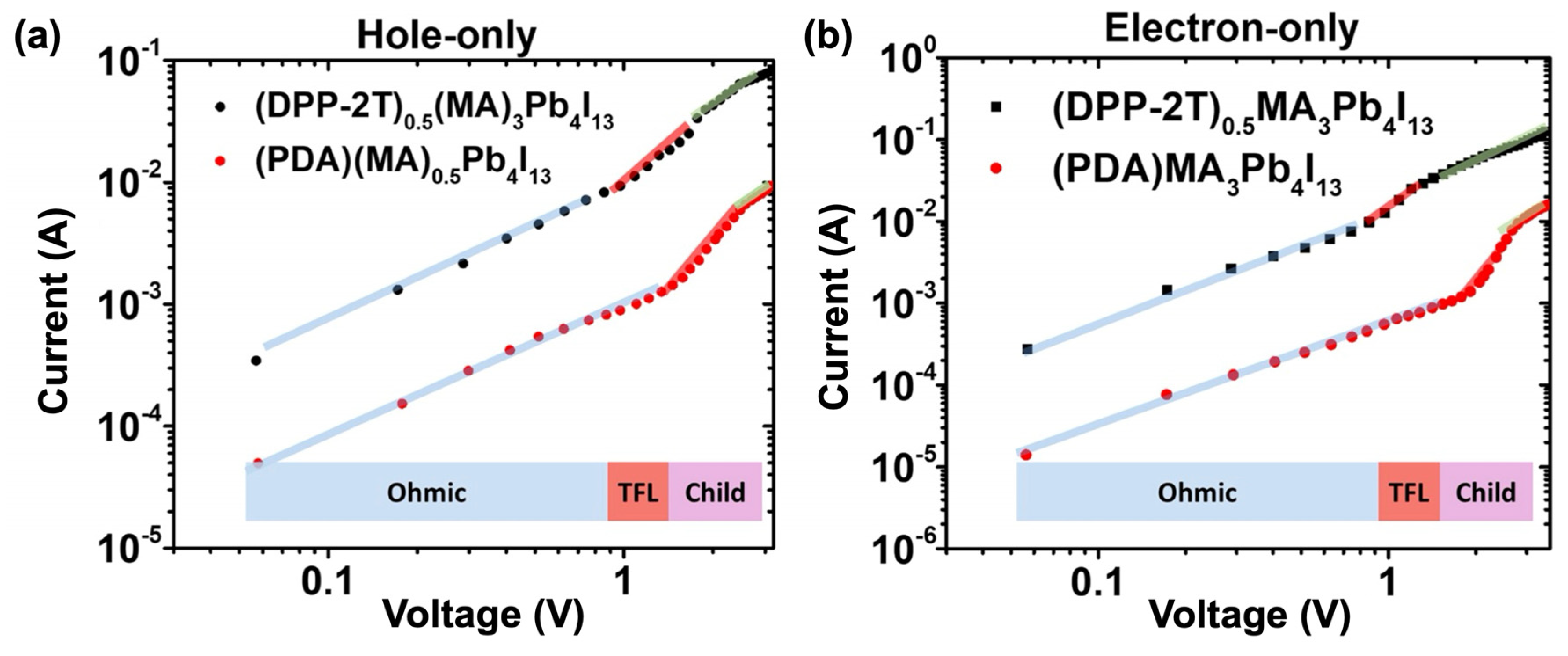
- Liu, C.; Fang, Z.; Sun, J.; Shang, M.; Zheng, K.; Yang, W.; Ge, Z. Donor-acceptor-donor type organic spacer for regulating the quantum wells of Dion-Jacobson 2D perovskites. Nano Energy 2022, 93, 106800. [Google Scholar] [CrossRef]
- Gkini, K.; Verykios, A.; Balis, N.; Kaltzoglou, A.; Papadakis, M.; Adamis, K.S.; Armadorou, K.-K.; Soultati, A.; Drivas, C.; Gardelis, S.; et al. Enhanced Organic and Perovskite Solar Cell Performance through Modification of the Electron-Selective Contact with a Bodipy−Porphyrin Dyad. ACS Appl. Mater. Interfaces 2020, 12, 1120–1131. [Google Scholar] [CrossRef] [PubMed]
- Kouki, H.; Pitié, S.; Torkhani, A.; Mamèche, F.; Decorse, P.; Seydou, M.; Kouki, F.; Lang, P. Tailor-Made Amino-Based Self-Assembled Monolayers Grafted on Electron Transport ZnO Layers: Perovskite Solar Cell Performance and Modified Interface Relationship. ACS Appl. Energy Mater. 2022, 5, 1635–1645. [Google Scholar] [CrossRef]
- Alagumalai, A.; Rajendran, M.V.; Ganesan, S.; Menon, V.S.; Raman, R.K.; Chelli, S.M.; Vijayasayee, S.M.; Gurusamy Thangavelu, S.A.; Krishnamoorthy, A. Interface Modification with Holistically Designed Push−Pull D−π−A Organic Small Molecule Facilitates Band Alignment Engineering, Efficient Defect Passivation, and Enhanced Hydrophobicity in Mixed Cation Planar Perovskite Solar Cells. ACS Appl. Energy Mater. 2022, 5, 6783–6796. [Google Scholar] [CrossRef]
- Liu, C.; Su, H.; Pu, Y.; Guo, M.; Zhai, P.; Liu, L.; Fu, H. 3D Polydentate Complexing Agents for Passivating Defects and Modulating Crystallinity for High-Performance Perovskite Solar Cells. Adv. Funct. Mater. 2023, 33, 2212577. [Google Scholar] [CrossRef]
- Zhang, Z.; Gao, Y.; Li, Z.; Qiao, L.; Xiong, Q.; Deng, L.; Zhang, Z.; Long, R.; Zhou, Q.; Du, Y.; et al. Marked Passivation Effect of Naphthalene-1,8-Dicarboximides in High-Performance Perovskite Solar Cells. Adv. Mater. 2021, 33, 2008405. [Google Scholar] [CrossRef]
- Yoon, M.-H.; Facchetti, A.; Marks, T.J. σ-π Molecular Dielectric Multilayers for Low-Voltage Organic Thin-Film Transistors. Proc. Natl. Acad. Sci. USA 2005, 102, 4678–4682. [Google Scholar] [CrossRef]
- DiBenedetto, S.A.; Frattarelli, D.; Ratner, M.A.; Facchetti, A.; Marks, T.J. Vapor Phase Self-Assembly of Molecular Gate Dielectrics for Thin Film Transistors. J. Am. Chem. Soc. 2008, 130, 7528–7529. [Google Scholar] [CrossRef]
- Li, L.; Lu, N.; Liu, M. Effect of dipole layer on the density-of-states and charge transport in organic thin film transistors. Appl. Phys. Lett. 2013, 103, 253303. [Google Scholar] [CrossRef]
- Ashkenasy, G.; Cahen, D.; Cohen, R.; Shanzer, A.; Vilan, A. Molecular engineering of semiconductor surfaces and devices. Acc. Chem. Res. 2002, 35, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Cahen, D.; Naaman, R.; Vager, Z. The Cooperative Molecular Field Effect. Adv. Funct. Mater. 2005, 15, 1571–1578. [Google Scholar] [CrossRef]
- Salinas, M.; Jäger, C.M.; Amin, A.Y.; Dral, P.O.; Meyer-Friedrichsen, T.; Hirsch, A.; Clark, T.; Halik, M. The Relationship between Threshold Voltage and Dipolar Character of Self-Assembled Monolayers in Organic Thin-Film Transistors. J. Am. Chem. Soc. 2012, 134, 12648–12652. [Google Scholar] [CrossRef]
- Huang, W.; Yu, X.; Zeng, L.; Wang, B.; Takai, A.; Di Carlo, G.; Bedzyk, M.J.; Marks, T.J.; Facchetti, A. Ultraviolet Light-Densified Oxide-Organic Self-Assembled Dielectrics: Processing Thin-Film Transistors at Room Temperature. ACS Appl. Mater. Interfaces 2021, 13, 3445–3453. [Google Scholar] [CrossRef]
- Kim, Y.-H.; Heo, J.-S.; Kim, T.-H.; Park, S.; Yoon, M.-H.; Kim, J.; Oh, M.S.; Yi, G.-R.; Noh, Y.-Y.; Park, S.K. Flexible Metal-Oxide Devices Made by Room-Temperature Photochemical Activation of Sol–Gel Films. Nature 2012, 489, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Di Carlo, G.; Turrisi, R.; Zeng, L.; Stallings, K.; Huang, W.; Bedzyk, M.J.; Beverina, L.; Marks, T.J.; Facchetti, A. The Dipole Moment Inversion Effects in Self-Assembled Nanodielectrics for Organic Transistors. Chem. Mater. 2017, 29, 9974–9980. [Google Scholar] [CrossRef]
- Lu, B.; Wang, B.; Chen, Y.; Facchetti, A.; Marks, T.J.; Balogun, O. Cross-Plane Thermal Conductance of Phosphonate-Based Self-Assembled Monolayers and Self-Assembled Nanodielectrics. ACS Appl. Mater. Interfaces 2020, 12, 34901–34909. [Google Scholar] [CrossRef]
- Everaerts, K.; Emery, J.D.; Jariwala, D.; Karmel, H.J.; Sangwan, V.K.; Prabhumirashi, P.L.; Geier, M.L.; McMorrow, J.J.; Bedzyk, M.J.; Facchetti, A.; et al. Ambient-Processable High Capacitance Hafnia-Organic Self-Assembled Nanodielectrics. J. Am. Chem. Soc. 2013, 135, 8926–8939. [Google Scholar] [CrossRef]
- Veres, J.; Ogier, S.; Lloyd, G.; de Leeuw, D. Gate Insulators in Organic Field-Effect Transistors. Chem. Mater. 2004, 16, 4543–4555. [Google Scholar] [CrossRef]
- Ting, G.G.; Acton, O.; Ma, H.; Ka, J.W.; Jen, A.K.-Y. Study on the Formation of Self-Assembled Monolayers on Sol−Gel Processed Hafnium Oxide as Dielectric Layers. Langmuir 2009, 25, 2140–2147. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.; Kline, R.J.; Fischer, D.A.; Lin, E.K.; Heeney, M.; McCulloch, I.; DeLongchamp, D.M. The Effect of Interfacial Roughness on the Thin Film Morphology and Charge Transport of High-Performance Polythiophenes. Adv. Funct. Mater. 2008, 18, 742–750. [Google Scholar] [CrossRef]
- Kim, M.-G.; Kim, H.S.; Ha, Y.-G.; He, J.; Kanatzidis, M.G.; Facchetti, A.; Marks, T.J. High-Performance Solution-Processed Amorphous Zinc−Indium−Tin Oxide Thin-Film Transistors. J. Am. Chem. Soc. 2010, 132, 10352–10364. [Google Scholar] [CrossRef] [PubMed]
- Alaboson, J.M.P.; Wang, Q.H.; Emery, J.D.; Lipson, A.L.; Bedzyk, M.J.; Elam, J.W.; Pellin, M.J.; Hersam, M.C. Seeding Atomic Layer Deposition of High- k Dielectrics on Epitaxial Graphene with Organic Self-Assembled Monolayers. ACS Nano 2011, 5, 5223–5232. [Google Scholar] [CrossRef] [PubMed]
- Stallings, K.; Smith, J.; Chen, Y.; Zeng, L.; Wang, B.; Di Carlo, G.; Bedzyk, M.J.; Facchetti, A.; Marks, T.J. Self-Assembled Nanodielectrics for Solution-Processed Top-Gate Amorphous IGZO Thin-Film Transistors. ACS App. Mater. Interfaces 2021, 13, 15399–15408. [Google Scholar] [CrossRef]
- Hupfer, M.L.; Kaufmann, M.; Roussille, L.; Preiß, J.; Weiß, D.; Hinrichs, K.; Deckert, V.; Dietzek, B.; Beckert, R.; Presselt, M. Arylic ve rsus Alkylic-Hydrophobic Linkers Determine the Supramolecular Structure and Optoelectronic Properties of Tripodal Amphiphilic Push−Pull Thiazoles. Langmuir 2019, 35, 2561–2570. [Google Scholar] [CrossRef] [PubMed]
- Malytskyi, V.; Simon, J.-J.; Patrone, L.; Raimundo, J.-M. Synthesis, self-assembly and characterization of a novel push–pull thiophene-based chromophore on a gold surface. RSC Adv. 2015, 5, 26308. [Google Scholar] [CrossRef]
- Lissau, H.; Frisenda, R.; Olsen, S.T.; Jevric, M.; Parker, C.R.; Kadziola, A.; Hansen, T.; van der Zant, H.S.J.; Nielsen, M.B.; Mikkelsen, K.V. Tracking molecular resonance forms of donor–acceptor push–pull molecules by single-molecule conductance experiments. Nat. Commun. 2015, 6, 10233. [Google Scholar] [CrossRef]
- Brandl, T.; El Abbassi, M.; Stefani, D.; Frisenda, R.; Harzmann, G.D.; van der Zant, H.S.J.; Mayor, M. Enhanced Separation Concept (ESC): Removing the Functional Subunit from the Electrode by Molecular Design. Eur. J. Org. Chem. 2019, 2019, 5334–5343. [Google Scholar] [CrossRef]
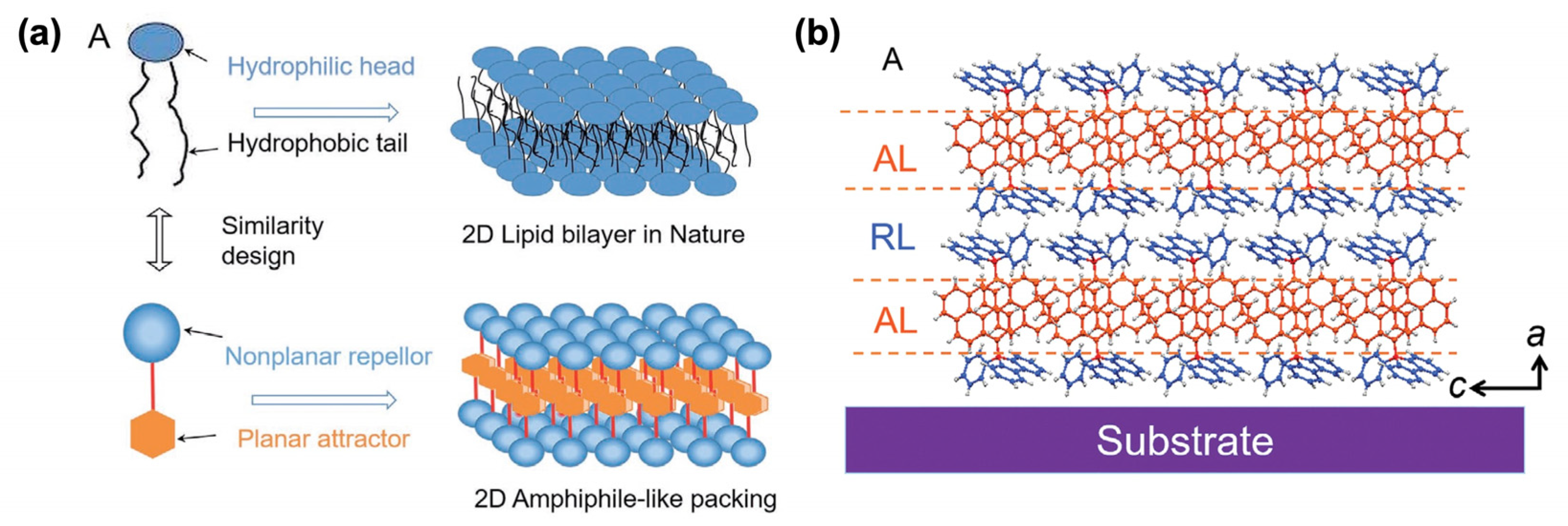
- Li, Y.-X.; Dong, X.-M.; Yu, M.-N.; Liu, W.; Nie, Y.-J.; Zhang, J.; Xie, L.-H.; Xu, C.-X.; Liu, J.-Q.; Huang, W. A Bio-Inspired Molecular Design Strategy toward 2D Organic Semiconductor Crystals with Superior Integrated Optoelectronic Properties. Small 2021, 17, 2102060. [Google Scholar] [CrossRef]
- Xu, J.; Wen, L.; Zhou, W.; Lv, J.; Guo, Y.; Zhu, M.; Liu, H.; Li, Y.; Jiang, L. Asymmetric and Symmetric Dipole-Dipole Interactions Drive Distinct Aggregation and Emission Behavior of Intramolecular Charge-Transfer Molecules. J. Phys. Chem. C 2009, 113, 5924–5932. [Google Scholar] [CrossRef]
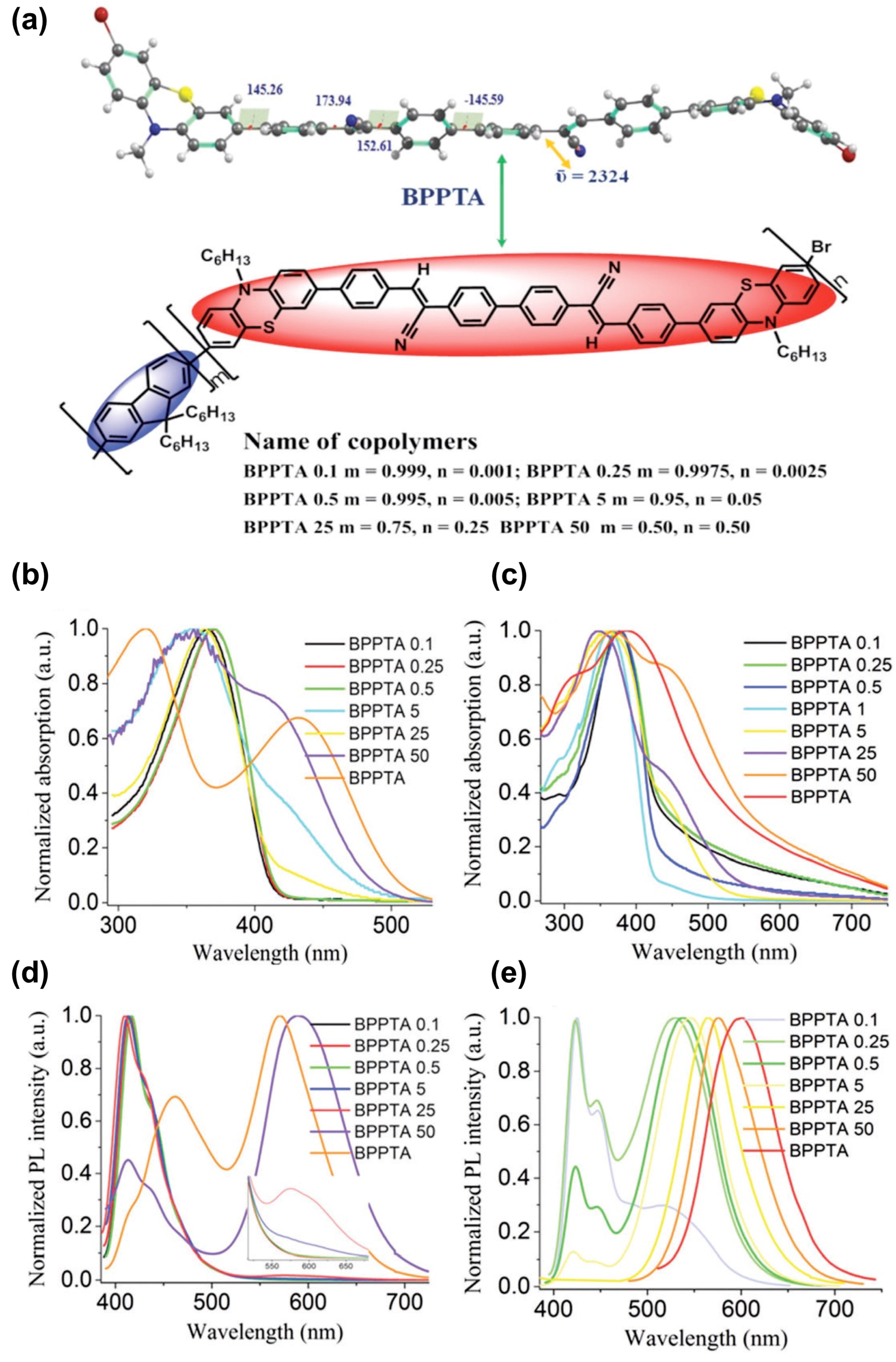
- Ravindran, E.; Somanathan, N. Efficient white-light emission from a single polymer system with ‘‘spring-like’’ self-assemblies induced emission enhancement and intramolecular charge transfer characteristics. J. Mater. Chem. C 2017, 5, 4763. [Google Scholar] [CrossRef]
- Kim, J.H.; Oh, S.; Park, S.K.; Park, S.Y. Green-Sensitive Phototransistor Based on Solution-Processed 2D n-Type Organic Single Crystal. Adv. Electron. Mater. 2019, 5, 1900478. [Google Scholar] [CrossRef]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, J.; Gadenne, V.; Patrone, L.; Raimundo, J.-M. Self-Assembled Monolayers of Push–Pull Chromophores as Active Layers and Their Applications. Molecules 2024, 29, 559. https://doi.org/10.3390/molecules29030559
Wang J, Gadenne V, Patrone L, Raimundo J-M. Self-Assembled Monolayers of Push–Pull Chromophores as Active Layers and Their Applications. Molecules. 2024; 29(3):559. https://doi.org/10.3390/molecules29030559
Chicago/Turabian StyleWang, Junlong, Virginie Gadenne, Lionel Patrone, and Jean-Manuel Raimundo. 2024. "Self-Assembled Monolayers of Push–Pull Chromophores as Active Layers and Their Applications" Molecules 29, no. 3: 559. https://doi.org/10.3390/molecules29030559
APA StyleWang, J., Gadenne, V., Patrone, L., & Raimundo, J.-M. (2024). Self-Assembled Monolayers of Push–Pull Chromophores as Active Layers and Their Applications. Molecules, 29(3), 559. https://doi.org/10.3390/molecules29030559







