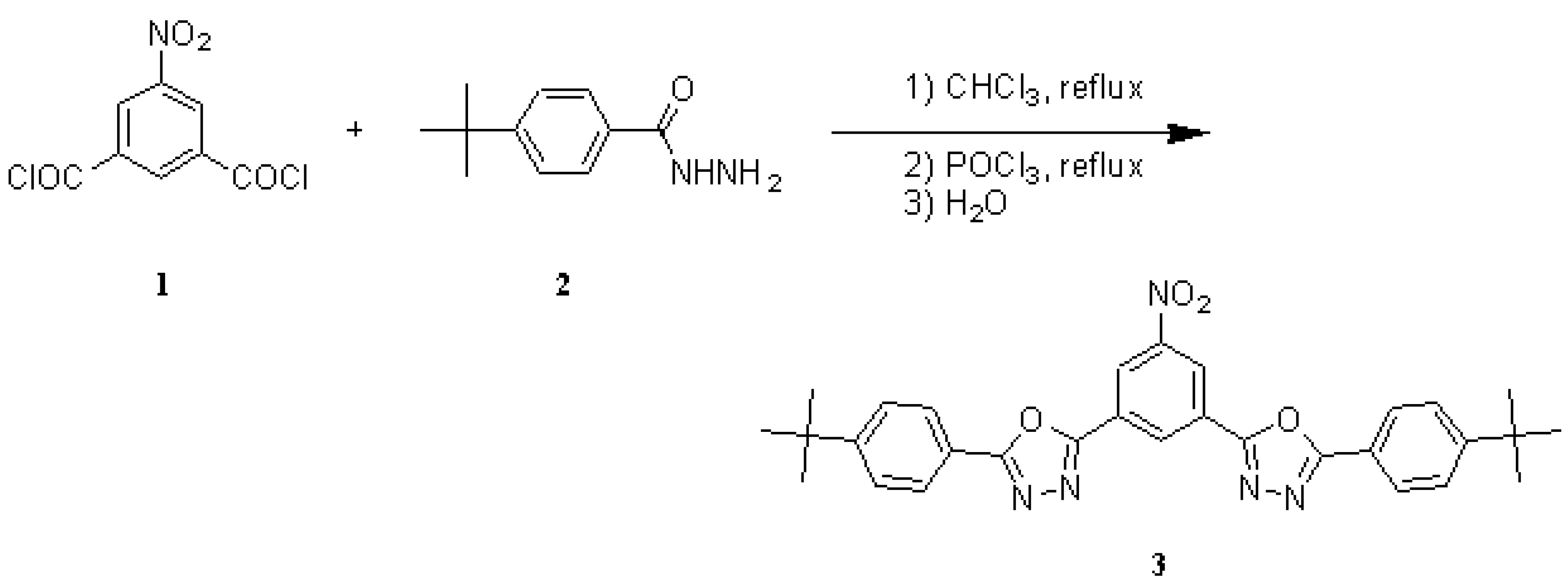
A solution of 5-nitroisophthaloyl dichloride (1) [1] (3.92 g, 15.8 mmol) and 4-tert-butylbenzhydrazide (2) [2] (6.08 g, 31.6 mmol) in CHCl3 (70 mL) was heated to reflux for 12 h. The solvent was then removed in vacuum, the residue was dried and combined with freshly distilled POCl3 (20 mL). The solution was heated to reflux for 3 h until evolution of HCl ceased. The resulting yellow solution was added dropwise to water (300 mL) under vigorous stirring. The precipitate was collected by suction filtration, air-dried, extracted with hot ethanol (200 mL), and dried to afford 3 as a colourless solid (3.11 g, 38%). The yield was not optimised. The extraction with ethanol was responsible for considerable losses in product and should better be replaced by a different method of purification.
Mp. : 288-290°C.
IR (KBr): 2964m; 1615m; 1535s; 1494s; 1349m; 1112m; 884m; 783m; 748m; 717m.
1H NMR (CDCl3, 500 MHz): 9.22 (t, J = 1.5 Hz, 1 H, C6H3); 9.12 (t, J = 1.5 Hz, 2 H, C6H3); 8.14 (AA'XX', 4 H, C6H4); 7.61 (AA'XX', 4 H, C6H4); 1.4 (s, 18 H, CH3).
13C NMR (CDCl3, 125 MHz): 165.9; 161.7; 156.3; 149.2; 129.7; 127.1; 127; 126.3; 123.7; 120.2; 35.2; 31.1.
EI-MS (70 eV): 524 (59, M+); 161 (92); 136 (57); 77 (100).
Anal. calc. for C30H29N5O4 (523.59): C 68.68, H 5.76, N 13.34; found: C 68.59, H 5.65, N 13.26.
Supplementary materials
Supplementary File 1Supplementary File 2References
- Jennings, K. F. J. Chem. Soc. 1957, 1172.
- Yale, H. L.; Losee, K.; Martins, J.; Holsing, M.; Perry, F. M.; Bernstein, J. J. Am. Chem. Soc. 1953, 75, 1933. [CrossRef]
- Sample Availability: available from the authors and from MDPI.
© 1999 MDPI. All rights reserved. Molecules website www.mdpi.org/molecules/
