Abstract
The absolute configuration at two newly formed stereogenic centres (5S, 7S) during the key steps of the total synthesis of naturally occuring goniothalesdiol was established by single-crystal X-ray diffraction analysis relative to stereocentres (1S, 8R) of the title compound (alternatively named 3,6-anhydro-2-deoxy-6-phenyl-l-ido-1,4-hexonolactone, C12H12O4). The conformation of both 5-membered lactone and furanose fused rings is also discussed.
Introduction
As a part of our long-term programme on palladium(II)-catalyzed oxycarbonylation of unsaturated polyols [1], we are interested in the preparation of naturally occuring biologically active lactones [2,3,4,5] structurally related to precursors of goniothalesdiol (1) (Figure 1). Recently, this compound was isolated from the bark of the Malaysian tree Goniothalamus borneensis (Annonaceae) and was shown to have a significant cytotoxicity against P388 mouse leukemia cells and insecticidal activities [6].
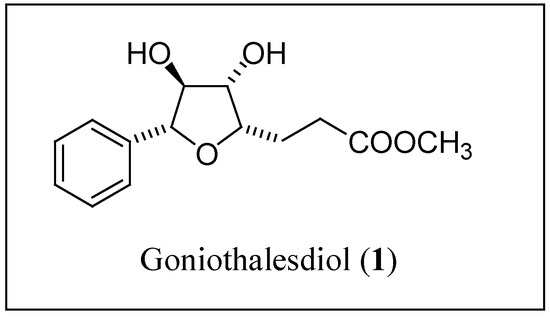
Figure 1.
Figure 1.
Results and Discussion
The title compound 3,6-anhydro-2-deoxy-6-phenyl-l-ido-1,4-hexonolactone (2), representing the key intermediate in the first total synthesis of goniothalesdiol [5], was prepared by palladium(II)-catalyzed oxycarbonylation of diastereomeric mixture of (1S, 2S, 3R) and (1R, 2S, 3R)-1-phenylpent-4-ene-1,2,3-triol (3) (Scheme 1). The products were separated by column chromatography.
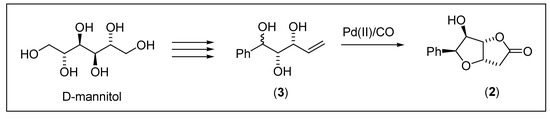
Scheme 1.
Scheme 1.
From the point of view of the synthetic project, it is very important to know its correct configuration at the C-1, C-5, C-7, and C-8 atoms. Because of the obvious difficulties in unambiguously establishing the configuration at two newly formed stereogenic centres C-5 and C-7 (R versus S) by NMR methods, suitable crystals of compound 2 were subjected to X-ray analysis. This confirmed 5-S and 7-S (relatively to the known 1-S and 8-R) configuration of 2 thus indicating a cis-fusion of the five-membered lactone and tetrahydrofuran rings. The relevant 1H-NMR coupling constants J7,8 = 2.6 Hz and J1,5 = 4.3 Hz are also confirmative of the established structural arrangement. A perspective view and the numbering scheme adopted for molecule of 2 is depicted in Figure 2. The relevant crystallographic and structure refinement data for lactone 2 are given in Table 1. The selected bond lengths and bond angles are listed in Table 2. A list of selected torsion angles is given in Table 3. The hydrogen bond geometry is shown in Table 4. Atomic coordinates and equivalent anisotropic displacement parameters have been deposited with CCDC as supplementary information [7].
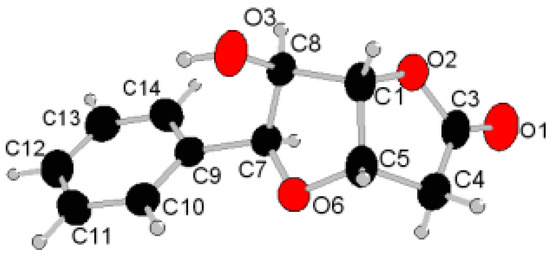
Figure 2.
Thermal ellipsoids plot at 50% probability level and atomic numbering of lactone 2.
Figure 2.
Thermal ellipsoids plot at 50% probability level and atomic numbering of lactone 2.
The presence of a five-membered lactone ring fused to a furanose ring at the 2,3-position (C-5 and C-1 in Figure 2 or C-3 and C-4 according to carbohydrate nomenclature of 2) imposes some conformational rigidity on this compound. The values of relevant torsion angles O-2–C-1–C-5–C-4 = 15.69(14)°, C-1–C-5–C-4–C-3 = – 12.53(15)°, C-5–C-4–C-3–O-2 = 5.14(16)°, C-4–C-3–O-2–C-1 = 5.32(16)°, C-3–O-2–C-1–C-5= – 13.49(14)° and puckering parameters [8] Q = 0.152(2) Å, ϕ = 55.7(6)° indicate that O-2–C-1–C-5–C-4–C-3 five-membered lactone ring adopts the 2E conformation which is significantly deformed (twisted on C-1–C-5 bond) adopting the shape which is very close to the C-5TC-1 conformation with the C-5 atom oriented endo and C-1 exo to the reference plane defined by the atoms O-2, C-3, and C-4. Analogously, the puckering parameters Q = 0.378(2) Å, ϕ = 124.8(2)° and the relevant dihedral angles O-6–C-5–C-1–C-8 = 12.40(15)°, C-5–C-1–C-8–C-7 = – 30.83(13)°, C-1–C-8–C-7–O-6 = 39.33(13)°, C-8–C-7–O-6–C-5 = – 33.23(15)°, and C-7–O-6–C-5–C-1 = 13.00(15)° are indicative of E3 conformation distorted (twisted on C-7–C-8 bond) almost completely to the C-7TC-8 direction for tetrahydrofuran ring (O-6–C-5–C-1–C-8–C-7) with the C-7 atom lying in the endo and C-8 exo direction with respect to the plane defined by the atoms C-1, C-5, and O-6.

Table 1.
Crystal and experimental data for compound 2a
| Empirical formula | C12H12O4 |
| Formula weight | 220.22 |
| Temperature, T (K) | 183(2) |
| Wavelength, λ (Å) | 0.71073 |
| Crystal system | orthorhombic |
| Space group | P212121 |
| Unit cell dimensions (Å) | a = 5.7069(1) |
| b = 8.4010(1) | |
| c = 21.2427(4) | |
| Unit cell volume, V (Å3) | 1018.45(3) |
| Formula units per unit cell Z | 4 |
| Dcalcd (g/cm3) | 1.436 |
| Absorption coefficient, μ (mm-1) | 0.108 |
| F(000) | 464 |
| Crystal size (mm) | 0.70 × 0.54 × 0.44 |
| Diffractometer | Siemens SMART CCD |
| θ Range (°) | 2.61–28.25 |
| Range of h | –7→7 |
| Range of k | –11→11 |
| Range of l | –27→27 |
| Reflections | 11525 |
| Independent reflections | 2371 (Rint = 0.0328) |
| Completeness to θ = 28.25 (%) | 95.7 |
| Absorption correction | multi-scan |
| Max. and min. transmission | 0.9539 and 0.9281 |
| Refinement method | Full-matrix least-squares on F2 |
| Data / restraints / parameters | 2371 / 0 / 158 |
| Goodness-of-fit on F2 | 1.002 |
| Final R indices [I>2σ(I)] | R1 = 0.0336, wR2 = 0.0798 |
| R indices (all data) | R1 = 0.0400, wR2 = 0.0846 |
| Largest difference peak and hole (e/Å3) | 0.195 and –0.209 |
a Standard deviations in parentheses.

Table 2.
Selected bond lengths [Å] and bond angles [º] for compound 2a
| O(1)-C(3) | 1.2065(17) | C(5)-C(1)-C(8) | 104.37(11) | |
| O(2)-C(3) | 1.3533(18) | O(1)-C(3)-O(2) | 120.39(15) | |
| O(2)-C(1) | 1.4474(16) | O(1)-C(3)-C(4) | 128.97(14) | |
| O(3)-C(8) | 1.4211(15) | O(2)-C(3)-C(4) | 110.63(12) | |
| O(6)-C(7) | 1.4379(17) | C(3)-C(4)-C(5) | 105.48(12) | |
| O(6)-C(5) | 1.4382(17) | O(6)-C(5)-C(1) | 106.39(11) | |
| C(1)-C(5) | 1.540(2) | O(6)-C(5)-C(4) | 112.97(13) | |
| C(1)-C(8) | 1.5206(18) | C(1)-C(5)-C(4) | 103.71(12) | |
| C(3)-C(4) | 1.499(2) | O(6)-C(7)-C(9) | 112.15(12) | |
| C(4)-C(5) | 1.524(2) | O(6)-C(7)-C(8) | 104.29(11) | |
| C(7)-C(9) | 1.5076(19) | C(9)-C(7)-C(8) | 114.36(11) | |
| C(7)-C(8) | 1.5315(19) | O(3)-C(8)-C(1) | 105.62(11) | |
| C(3)-O(2)-C(1) | 111.22(11) | O(3)-C(8)-C(7) | 112.58(12) | |
| C(7)-O(6)-C(5) | 108.55(11) | C(1)-C(8)-C(7) | 101.00(11) | |
| O(2)-C(1)-C(5) | 106.51(11) | C(10)-C(9)-C(7) | 123.04(13) | |
| O(2)-C(1)-C(8) | 109.74(11) | C(14)-C(9)-C(7) | 117.78(13) |
a Standard deviations in parentheses.

Table 3.
Selected torsion angles [º] for compound 2a
| O(2)-C(1)-C(5)-C(4) | 15.69(14) | O(2)-C(1)-C(5)-O(6) | –103.68(12) | |
| C(1)-C(5)-C(4)-C(3) | –12.53(15) | C(8)-C(1)-C(5)-C(4) | 131.77(11) | |
| C(5)-C(4)-C(3)-O(2) | 5.14(16) | C(3)-C(4)-C(5)-O(6) | 102.24(14) | |
| C(4)-C(3)-O(2)-C(1) | 5.32(16) | C(5)-O(6)-C(7)-C(9) | –157.52(11) | |
| C(3)-O(2)-C(1)-C(5) | –13.49(14) | O(2)-C(1)-C(8)-O(3) | –159.61(11) | |
| O(6)-C(5)-C(1)-C(8) | 12.40(15) | C(5)-C(1)-C(8)-O(3) | 86.59(13) | |
| C(5)-C(1)-C(8)-C(7) | –30.83(13) | O(2)-C(1)-C(8)-C(7) | 82.97(13) | |
| C(1)-C(8)-C(7)-O(6) | 39.33(13) | O(6)-C(7)-C(8)-O(3) | –72.88(14) | |
| C(8)-C(7)-O(6)-C(5) | –33.23(15) | C(9)-C(7)-C(8)-O(3) | 49.98(16) | |
| C(7)-O(6)-C(5)-C(1) | 13.00(15) | C(9)-C(7)-C(8)-C(1) | 162.18(12) | |
| C(3)-O(2)-C(1)-C(8) | –125.91(12) | C(8)-C(7)-C(9)-C(10) | –106.15(16) | |
| C(7)-O(6)-C(5)-C(4) | –100.14(14) | O(6)-C(7)-C(9)-C(14) | –168.01(12) |
a Standard deviations in parentheses.
Analysis of the molecular packing in the unit cell revealed two hydrogen bonds (Table 4). Atom O(1) acts as a bifurcated acceptor of both hydrogen bonds. The first-level descriptors based on the graph-set theory [9] give chain C1,1(7) for O...O hydrogen bond while C...O hydrogen bond forms chain C1,1(5). On the second-level, C2,2(12) and C1,2(6) chains are formed by both types of hydrogen bonds. For convenience, the notation Xa,d(n) has also been adopted in this paper, in which (X) is the pattern descriptor, (a) is number of acceptors, (d) is number of donors and (n) is the number of atoms comprising the pattern.
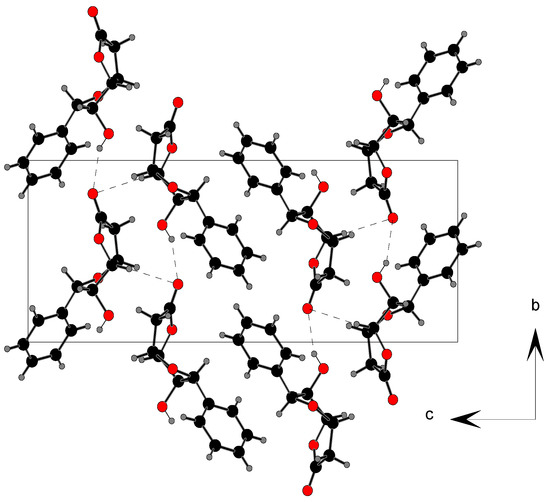
Figure 3.
Packing scheme along a-axis for lactone 2. Hydrogen bonds are shown as broken lines.
Figure 3.
Packing scheme along a-axis for lactone 2. Hydrogen bonds are shown as broken lines.

Table 4.
Hydrogen bond geometry in compound 2a
| X–H…Y | Symmetry code | X–H(Å) | H…Y(Å) | X…Y(Å) | X–H…Y(°) |
| O(3)–H(3)…O(1) | x, y+1, z | 0.84 | 2.12 | 2.8885(14) | 151.2 |
| C(1)–H(1)…O(1) | –x, y+0.5, –z+0.5 | 1.00 | 2.55 | 3.4976(19) | 157.3 |
a Standard deviations in parentheses.
Experimental
General
1H- and 13C-NMR spectra (in CDCl3 with Me4Si as an internal standard) were recorded on a Bruker Avance DPX 300 instrument operating at working frequencies of 300.13 and 75.46 MHz, respectively. For the assignments of signals, 1D NOESY and C–H heterocorrelated experiments were used. The EI mass spectrum (70 eV) was obtained on a Finnigan MAT SSQ 710 instrument. Specific rotation was determined on a Perkin–Elmer 241 polarimeter (10 cm cell). Microanalyses were performed on a Fisons EA 1108 analyser. Melting point was determined with a Boetius PHMK 05 microscope. Column chromatography was performed as flash chromatography on Silica Gel 60 (E. Merck, 0.063–0.200 mm).
X-ray techniques
Crystal and experimental data for lactone 2 are summarized in Table 1. Preliminary orientation matrix was obtained from the first frames using Siemens SMART software [10]. Final cell parameters were obtained by refinement of 7739 reflections using Siemens SAINT software [10]. The data were empirically corrected for absorption and other effects using SADABS program [11] based on the method of Blessing [12]. The structure was solved by direct methods and refined by full-matrix least-squares on all F2 data using Bruker SHELXTL [13]. The non-H atoms were refined anisotropically. Hydrogen atoms were constrained to the ideal geometry using an appropriate riding model. Molecular graphics were obtained using the program DIAMOND [14].
(1S, 5S, 7S, 8R)-8-Hydroxy-7-phenyl-2,6-dioxabicyclo[3.3.0]octan-3-one (2).
A 25-mL flask, purged with CO and connected to a balloon with CO gas, was charged with PdCl2 (10 mg, 0.05 mol), anhydrous CuCl2 (200 mg, 1.56 mmol), NaOAc (130 mg, 1.56 mmol), a mixture of (1S, 2S, 3R) and (1R, 2S, 3R)-1-phenylpent-4-en-1,2,3-triol (3) (100 mg, 0.52 mmol), and AcOH (10 mL). The mixture was stirred for 20 h at room temperature, then filtered through a short tube filled with cellulose (2 g). The solvent was evaporated under diminished pressure and the residue was purified by chromatography on a column of silica gel using hexane–ethyl acetate (5 : 2, v/v) as an eluent. The fractions with Rf = 0.4 (1:1 hexane–ethyl acetate) were collected, evaporated and the product recrystallized from 1:1 EtOAc–hexane affording the title compound 2 (37 mg, 32 %, colourless crystals). M.p. 177–180 °C; [α]D + 38° (c 0.31, MeOH); 1H-NMR (300 MHz, CDCl3): δ 7.46–7.35 (m, 5H, Ph), 5.23 (d, 1 H, J7,8 = 2.6 Hz, H-7), 5.20 (ddd, 1 H, J1,5 = 4.3 Hz, J4a,5 = 5.6 Hz, J4b,5 = 1.4 Hz, H-5), 5.07 (d, 1 H, J1,5 = 4.3 Hz, H-1), 4.47 (d, 1 H, J7,8 = 2.6 Hz, H-8), 2.88 (dd, 1 H, J4a,4b = 18.8 Hz, J4a,5 = 5.6 Hz, H-4a), 2.79 (dd, 1 H, J4a,4b = 18.8 Hz, J4b,5 = 1.4 Hz, H-4b); 13C-NMR (75 MHz, CDCl3): δ 175.4 (C-3), 134.2 (C-1 in Ph), 128.9 (C-3 and C-5 in Ph), 128.6 (C-4 in Ph), 126.6 (C-2 and C-6 in Ph), 87.2 (C-1), 82.9 (C-7), 77.1 (C-5), 75.8 (C-8), 36.0 (C-4); EIMS (70 eV): m/z 220 [M]+, 202, 192, 176, 143, 114, 106, 85, 84. Anal. Calcd for C12H12O4 (220.22): C, 65.40; H, 5.49. Found: C, 65.29; H, 5.52.
Acknowledgements
Financial support of this work by the Scientific Grant Agency (VEGA, Slovak Academy of Sciences and Ministry of Education, Bratislava, projects Nos. 2/7204/20, 1/9251/02 and 1/7314/20) is gratefully appreciated.
References and Notes
- Gracza, T.; Hasenöhrl, T.; Stahl, U.; Jäger, V. Synthesis of 3,5-Anhydro-2-deoxy-1,4-glyconolactones by Palladium(II)-Catalyzed, Regioselective Oxycarbonylation of C5- and C6-Enitols. ω-Homologation of Aldoses to Produce Intermediates for C-Glycoside/C-Nucleoside Synthesis. Synthesis 1991, 1108–1118. [Google Scholar]
- Gracza, T.; Jäger, V. Palladium(II)-Catalyzed Oxycarbonylation of Unsaturated Polyols: Synthesis of (-)-Goniofufurone and Assignment of Absolute Configuration to the Natural (+)-Enantiomer, a Cytotoxic Styryllactone. Synlett 1992, 191–193. [Google Scholar]
- Gracza, T.; Jäger, V. Synthesis of Natural and Unnatural Enantiomers of Goniofufurone and Its 7-Epimers from D-Glucose. Application of Palladium(II)-Catalyzed Oxycarbonylation of Unsaturated Polyols. Synthesis 1994, 1359–1368. [Google Scholar]
- Dixon, D. J.; Ley, S.V.; Gracza, T.; Szolcsanyi, P. Total synthesis of the polyenoyltetramic acid mycotoxin erythroskyrine. J. Chem. Soc., Perkin Trans. 1 1999, 839–841. [Google Scholar]
- Babjak, M.; Kapitán, P.; Gracza, T. The First Total Synthesis of Goniothalesdiol. Tetrahedron Lett. 2002, 43, 6983–6985. [Google Scholar] [CrossRef]
- Cao, Sh.-G.; Wu, X.-H.; Sim, K.-Y.; Tan, B. K. H.; Pereira, J. T.; Goh, S.-H. Styryl-Lactone Derivatives and Alkaloids from Goniothalamus borneensis (Annonaceae). Tetrahedron 1998, 54, 2143–2148. [Google Scholar] [CrossRef]
- CCDC 194248 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from the Director, CCDC, 12 Union Road, Cambridge CB2 1EZ, UK (Fax: +44-1223-336030; e-mail: deposit@ccdc.cam.ac.uk or www: http://www.ccdc.cam.ac.uk).
- Cremer, D.; Pople, J. A. A General Definition of Ring Puckering Coordinates. J. Am. Chem. Soc. 1975, 97, 1354–1573. [Google Scholar] [CrossRef]
- Bernstein, J.; Davis, R. E.; Shimoni, L.; Chang, L.-N. Patterns in Hydrogen Bonding: Functionality and Graph Set Analysis in Crystals. Angew. Chem., Int. Ed. Engl. 1995, 34, 1555–1573. [Google Scholar]
- Siemens AXS. SMART & SAINT: Madison, WI, USA, 1995.
- Sheldrick, G. M. Program SADABS; University of Göttingen: Germany, 2001. [Google Scholar]
- Blessing, R. H. An Empirical Correction for Absorption Anisotropy. Acta Crystallogr., Sect. A 1995, 51, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Bruker AXS Inc. SHELXTL Version 6.10; Madison, WI, USA, 2001. [Google Scholar]
- Brandenburg, K. DIAMOND: Visual Crystal Structure Information System, Version 2.1d; Crystal Impact GbR: Bonn, Germany, 2000. [Google Scholar]
- Samples Availability: Compound 2 reported in this paper is available from MDPI.
© 2003 by MDPI ( http://www.mdpi.org). Reproduction is permitted for non commercial purposes.
