Abstract
To investigate the mechanisms by which Moringa oleifera leaves (ML) modulate hepatic lipids, guinea pigs were allocated to either control (0% ML), 10% Low Moringa (LM) or 15% High Moringa (HM) diets with 0.25% dietary cholesterol to induce hepatic steatosis. After 6 weeks, guinea pigs were sacrificed and liver and plasma were collected to determine plasma lipids, hepatic lipids, cytokines and the expression of genes involved in hepatic cholesterol (CH) and triglyceride (TG) metabolism. There were no differences in plasma lipids among groups. A dose-response effect of ML was observed in hepatic lipids (CH and TG) with the lowest concentrations in the HM group (p < 0.001), consistent with histological evaluation of lipid droplets. Hepatic gene expression of diglyceride acyltransferase-2 and peroxisome proliferator activated receptor-γ, as well as protein concentrations interleukin (IL)-1β and interferon-γ, were lowest in the HM group (p < 0.005). Hepatic gene expression of cluster of differentiation-68 and sterol regulatory element binding protein-1c were 60% lower in both the LM and HM groups compared to controls (p < 0.01). This study demonstrates that ML may prevent hepatic steatosis by affecting gene expression related to hepatic lipids synthesis resulting in lower concentrations of cholesterol and triglycerides and reduced inflammation in the liver.
1. Introduction
Non-alcoholic fatty liver disease (NAFLD) is characterized by a variety of liver conditions ranging from hepatic steatosis, to non-alcoholic steatohepatitis (NASH) [1] which, if untreated, can lead to further complications such as hepatic fibrosis, cirrhosis or cancer, and even death [2,3]. There are several dietary interventions that can be used to ameliorate hepatic lipid accumulation, oxidative stress and inflammation associated with NAFLD and NASH [4,5] including the use of foods that are rich in antioxidants, among which polyphenols and carotenoids play a predominate role [6,7].
Moringa oleifera Lam is a softwood tree original from the Himalayas and Northern India, which has been investigated for its uses in human health [8]. Many parts of this plant (leaves, immature pods, flowers and fruits) are edible and are used for their nutritional content in many countries [9]. The leaves of the Moringa, the most utilized part of the plant, are characterized by having a great number of bioactive compounds including vitamins, carotenoids, polyphenols (phenolic acids and flavonoids) [10]. For this reason, Moringa leaves (ML) have been used to treat a number of conditions including insulin resistance, cardiovascular disease, hepatic steatosis, cancer and others [11].
ML have also been shown to have hepatoprotective activity and decrease the plasma lipids of rats fed a high-fat diet [12,13]. The aqueous extract of Moringa leaves has been reported to regulate glucose metabolism in diabetic rats [14,15]. In addition, ML have been found to have a cardioprotective role in isoproterenol-induced myocardial infarction by affecting the activities of several enzymes associated with oxidation [16].
Flavonoids and saponins present in ML are reported to increase HDL (high density lipoprotein) cholesterol (HDL-C) and to lower LDL (low density lipoprotein) cholesterol (LDL-C) and very low-density lipoprotein (VLDL) cholesterol in hypercholesterolemic rats [17]. It appears that flavonoids and saponins lower cholesterol absorption by the inhibition of cholesterol micellar solubility [18]. Thus, both of these bioactive compounds found in the aqueous extract have been reported to have hypolipidemic effects [9,10].
Several bioactive compounds including nitrile, mustard oil glycosides and thiocarbamate present in ML have been shown to stabilize blood pressure [19]. In addition, aqueous extracts of leaves, fruit and seeds of Moringa have antioxidant capacities [20], possibly due to the presence of quercetin and kaempferol in Moringa, as they have been reported to have strong antioxidant activity on hepatocyte growth factor-induced oxidation [21]. The liver protection of Moringa leaves has been found in other reports. Ethanolic extracts of ML have shown a protective effect against antitubercular drug-induced liver damage in rats [22]. Further, histological examinations confirmed a decrease in hepatic damages induced by drugs [23]. ML also showed the ability to reduce carbon tetrachloride-induced liver fibrosis and control the rise of serum aminotransferase activities and globulin level [24] and to protect liver exposed to ionizing radiation by increasing antioxidant enzymes and inhibiting nuclear factor κB (NF-κB) translocation to the nucleus [25].
Phenolic acids, including chlorogenic acid (CGA), from ML have been shown to have antioxidant, anti-inflammatory and anti-hyperglycemic properties [26]. However, the specific mechanisms associated with reduction of hepatic lipids have not been fully elucidated.
The present study was conducted to investigate the effect of ML in a guinea pig model of hepatic steatosis. Our hypothesis was that ML would prevent hepatic steatosis induced by a high cholesterol diet by modulating the gene expression of regulators of hepatic cholesterol and triglyceride homeostasis through the action of bioactive compounds present in ML.
2. Results
2.1. Food Intake and Body and Liver Weight
No differences in food intake, final body weight or liver weight were found among groups during the intervention. Food intake was 24.0 ± 2.2, 20.7 ± 7.8 and 23.6 ± 2.9 g/day for the control, LM and HM groups (p > 0.05). Final body weights for guinea pigs were: 535 ± 44, 543 ± 55 and 507 ± 43 g, respectively (p > 0.05).
2.2. Plasma Lipids, Glucose and Liver Enzymes
There were no differences in plasma total cholesterol, VLDL, LDL, HDL, triglycerides, glucose or insulin among groups. Both Moringa groups had lower activity of ALT than the control group, while no difference in AST between groups was observed (p < 0.05) (Table 1).

Table 1.
Plasma Lipid profile, glucose, insulin and liver enzymes of guinea pigs fed a hypercholesterolemic diet with no Moringa (control), Low (10%), or High Moringa (15%) 1.
2.3. Lipoprotein Size and Subfractions
The NMR method reports the concentration of lipoproteins in plasma and the size [27]. Total VLDL, total LDL and their subfractions (large, medium, and small) did not differ between groups at the end of the intervention. Although total HDL, large and small HDL were not affected, medium HDL was lower in both groups of Moringa compared to the control group (p < 0.05) (Table 2). There were no differences in the VLDL, LDL and HDL size between groups.

Table 2.
Lipoprotein subfractions in guinea pigs fed a hypercholesterolemic diet with no Moringa (control), Low (10%), or High Moringa (15%) 1.
2.4. LCAT and CETP Activity
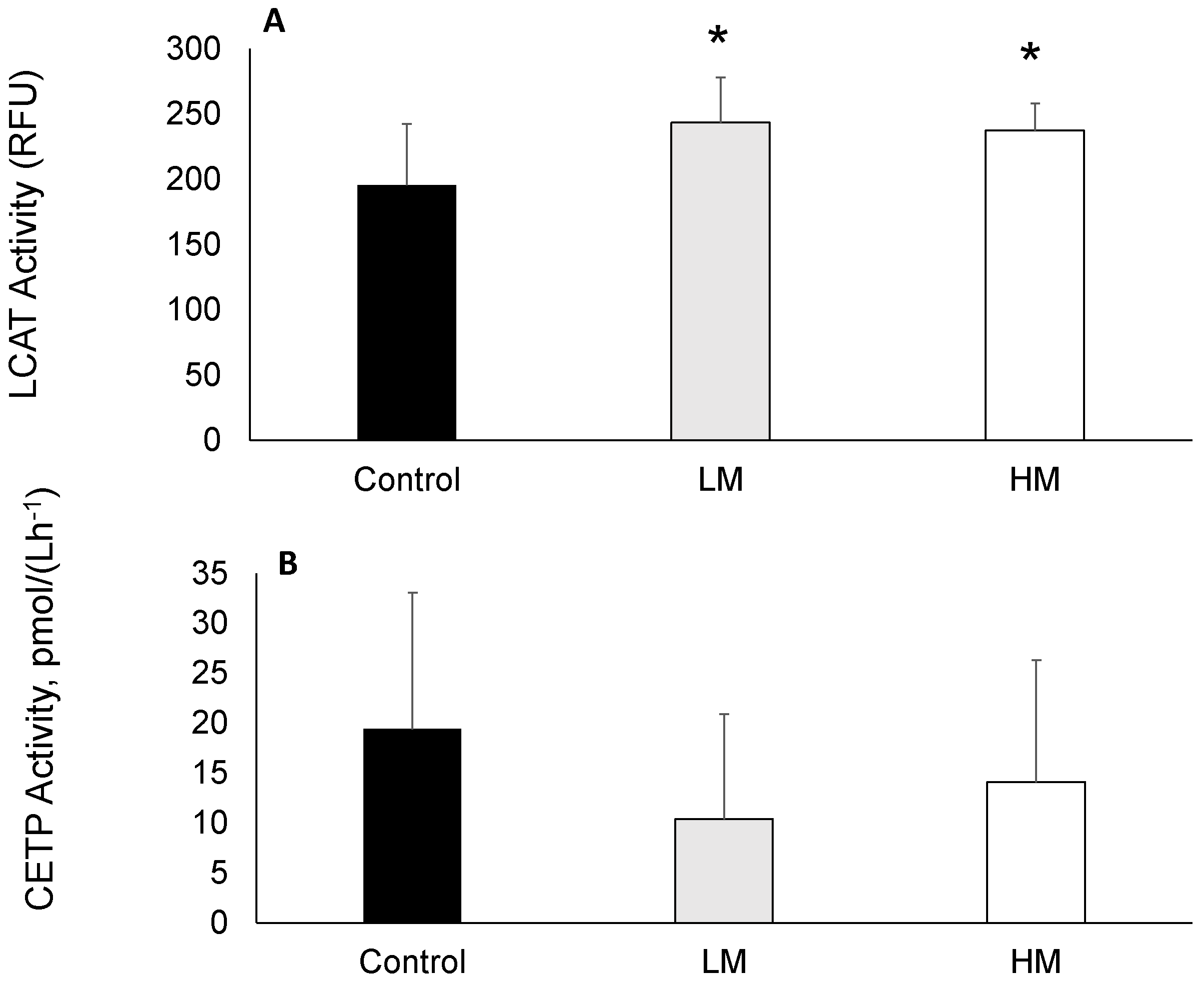
LCAT activity was greater in guinea pigs fed Moringa compared to controls (Figure 1A) while plasma CETP activity was not different among groups (Figure 1B).
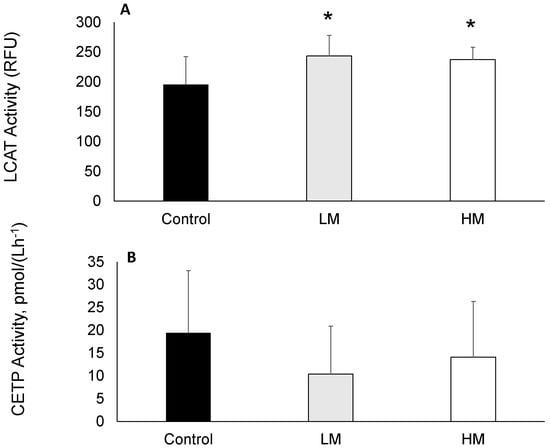
Figure 1.
(A) Lecithin cholesterol acyltransferase (LCAT) and (B) Cholesterol ester transfer protein (CETP) activities of guinea pigs fed control, Low Moringa (LM; 10 g/100 g) and High Moringa (HM; 15 g/100 g) diets. * indicates significantly different from control at p < 0.01. RFU, Reference Units.
2.5. Hepatic Lipids
After the extraction of hepatic lipids [28] and measurement [29,30], TC, CE, and TG were lower in group fed the HM diet (p < 0.05), while the concentrations of hepatic lipids in the LM group were not different compared with HM or control groups (Table 3).

Table 3.
Concentration of hepatic total cholesterol, free cholesterol, cholesteryl ester and inflammatory cytokines of guinea pigs fed a hypercholesterolemic diet with no Moringa (control), Low (10%), or High Moringa (15%) 1.
2.6. Liver Inflammation
Hepatic cytokines were measured simultaneously [31]. The hepatic cytokines IL-1β, 1L-10 and IFNγ were lowest in the HM group, intermediate in the LM group and highest in the control group (p < 0.05). There were no differences in IL-6, MCP-1 and TNFα cytokines between groups (Table 4).

Table 4.
Hepatic cytokines/chemokines of guinea pigs fed a hypercholesterolemic diet with no Moringa (control), Low (10%), or High Moringa (15%) 1.
2.7. Histology
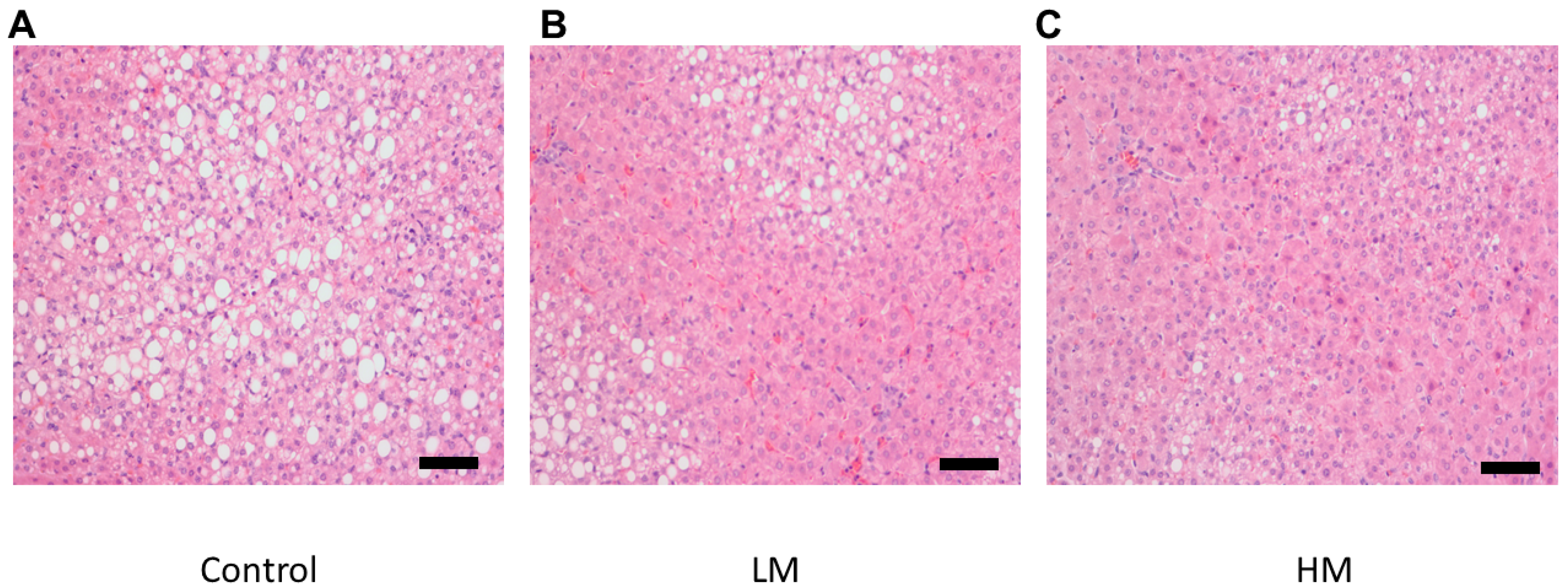
There was a lower degree of steatosis and lipid accumulation in livers from the guinea pigs fed the HM diet compared to the control and the LM group (Figure 2). Liver samples from guinea pigs fed the control diet showed the most lipid accumulation while the LM group exhibited less accumulation than the control in agreement with hepatic cholesterol and TG concentrations.
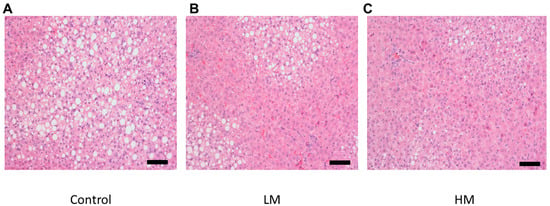
Figure 2.
Histological images of hematoxylin and eosin (H&E) (200×) stained hepatic tissue for (A) Control, (B) Low Moringa (LM; 10%), and (C) High Moringa (HM; 15%). Scale bar = 100 μm.
2.8. Hepatic Gene Expression
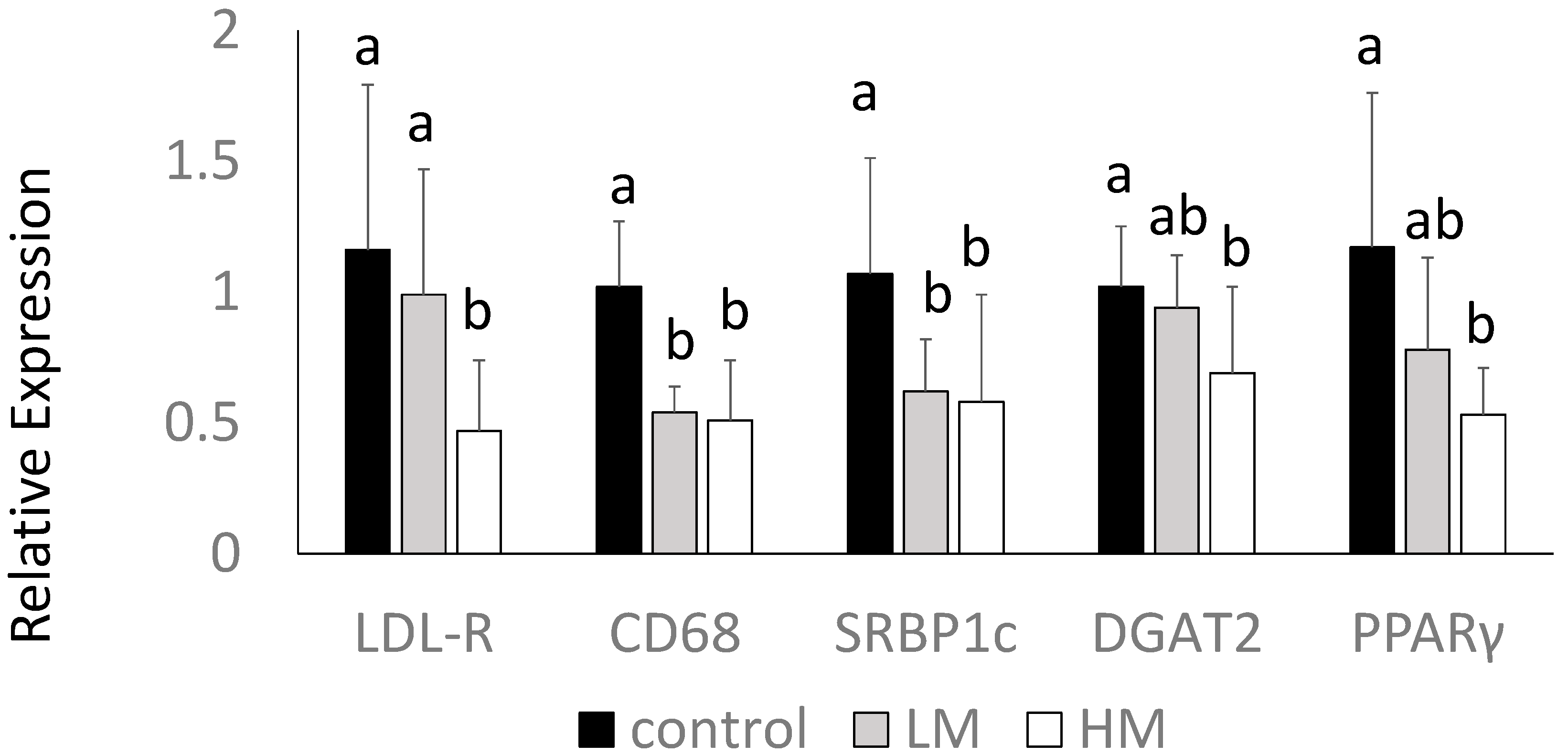
Interestingly, the HM group had lower levels of LDL receptor (LDL-R) gene expression compared to control and LM groups. Guinea pigs fed the ML had lower gene expression of CD68, a macrophage marker, and SREBP1c mRNA compared to control (p < 0.05) (Figure 3). DGAT2 and PPARγ mRNA were lowest in the HM group, intermediate in the LM group, and highest in the control group (p < 0.05) (Figure 3). There were no differences in HMG-CoA reductase, CD36, LXR or IL-1β among groups (data not shown).
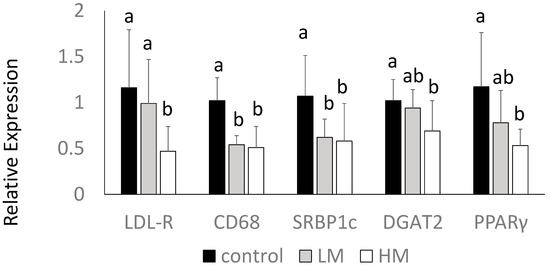
Figure 3.
Hepatic gene expression of LDL receptor (LDL-R), cluster of differentiation 68 (CD68), SREBP1, DGAT2 and PPARγ of guinea pigs fed control (black bars), Low Moringa (LM; 10%, gray bars) or High Moringa (HM; 15%, white bars). Different superscripts, a and b denote significantly different at p < 0.05.
3. Discussion
In this study, we have demonstrated that ML effectively attenuates hepatic steatosis in guinea pigs, leading to the lowering of hepatic lipids and inflammatory markers. We also observed that there was a dose-response with the HM diet having stronger effects on preventing the development of a steatotic phenotype. This could potentially be a consequence of the ingestion of more bioactive compounds present in ML, such as quercetin and CGA, which have been shown to alter gene expression of major regulators of hepatic cholesterol and TG synthesis and uptake [32,33].
3.1. Moringa Leaves and Plasma Lipids and Liver Enzymes
We found no differences in food intake or body weight between controls and guinea pigs fed ML, which suggest that ML did not affect weight gain in guinea pigs. Gebregiorgis et al. [34] used different concentrations of ML in sheep and compared to a control diet of Rhodes grass. Sheep consuming the ML had greater body weight gain and nitrogen retention, also suggesting a non-toxic effect.
ALT and AST are enzymes involved in amino acid metabolism and can be used as indicators of liver damage [35]. In this study, a significant reduction was found in ALT activity in both Moringa groups, but not in AST. Other studies have also shown reductions of serum ALT and AST in animals treated with an extract of ML [36]. Pari et al. [22] reported reductions of these liver enzymes with Moringa intake, and a histological evaluation of the liver further documented a recovery from liver damage with ML. These results suggest that ML exert protective effects in liver without inducing toxicity.
The ML used in this study was composed of 19% fiber; therefore, we were expecting to observe decreases in plasma LDL-C due to fiber intake. However, we observed no changes in plasma total or LDL-C among the three dietary groups, suggesting that the fiber present in Moringa did not disrupt micelle formation and the enterohepatic circulation of bile acids as we have observed in guinea pig fed other sources of dietary fiber [37]. Interestingly, guinea pigs consuming the HM had lower expression of LDL-R compared to the other groups. The hepatic LDL-R is the major mechanism by which the body removes LDL-C from plasma [38], and it has been shown that LDL-R [39] and LDL uptake [40] are up-regulated by fiber. In this study, there were no differences in plasma cholesterol among groups, while there was a very significant lowering of hepatic cholesterol in the HM group. This suggests that the decreased expression of LDL-R might have contributed to the lack of effect in lowering plasma LDL-C and to the very significant lowering of hepatic cholesterol.
Consumption of ML, however, resulted in lower concentrations of the medium HDL particles, which have been shown to be less effective in reverse cholesterol transport (RCT), compared with other HDL particle subclasses [41]. A decrease in medium HDL has also been shown to be accompanied by significant increases in HDL-C in the case of low carbohydrate diets [42]. Further suggesting that RCT may have been altered by ML, we observed a 55% increase in LCAT activity, a key regulator of RCT. The major function of LCAT is to facilitate CE formation through the action of Apolipoprotein I (ApoA-I), and it is believed that larger discoidal HDL particles are the preferred substrate for the enzyme [43]. The lipid transfer protein, CETP, is involved in moving CE, TG and phospholipid molecules between HDL and other lipoprotein particles [44]. Thus, LCAT activity influences the efflux efficiency associated with the removal of cellular cholesterol. Thus, although we did not observe changes in atherogenic lipoproteins, the observed changes in these mediators (LCAT, HDL) of RCT suggest ML may also protect against atherosclerosis. Further studies are necessary to confirm this theory.
3.2. Moringa Leaves and Hepatic Lipids, Inflammation, and Gene Expression
Previous studies in our laboratory have shown that a cholesterol challenge of 0.25% (w/w) dietary cholesterol leads to hepatic steatosis in guinea pigs [45]. This accumulation of lipids was observed in the control animals, while the intake of ML resulted in lower concentrations of cholesterol and TG in a dose-dependent manner. CGA is one of the bioactive components present in ML in sufficient amounts to exert hypolipidemic effects [46]. Other investigators have reported that CGA markedly decreases the concentration of total cholesterol and TG in the liver [47], similar to our findings. In addition, Cho et al. [48] reported that CGA significantly inhibited the activities of fatty acid synthase, 3-hydroxy-3-methylglutaryl CoA reductase, and acyl-CoA cholesterol acyltransferase, while it increased fatty acid β-oxidation and the expression of PPARα in mouse livers compared to a control group fed a high-fat diet. We observed decreases in SREBP-1c expression in guinea pigs fed ML. Lipid biosynthesis is regulated at the transcriptional level by SREBP-1 and -2. SREBP-1c is associated with the synthesis of fatty acids and TG, while SREBP-2 with that of cholesterol [49]. CGA, as has been shown in other studies [32], could have also had an effect in decreasing TG via reductions in SREBP1c.
Excess free cholesterol is toxic to cells and induces pro-inflammatory responses which increase the production of cytokines and chemokines [50]. In our study, the control group had 41% higher concentrations of free cholesterol than the Moringa groups, which was accompanied by higher concentrations of the inflammatory cytokines IL-1β and IFNγ and higher expression of the macrophage marker CD68, suggesting that increases in free cholesterol led to hepatic inflammation [50]. Recently, Waterman et al. [51] evaluated the effect of supplementation of a ML concentrate in high fat diet-induced obese mice for 12 weeks. They also found a reduction of gene expression of pro-inflammatory markers, IL-6 and IL-1β in the liver and ileum tissues in mice treated with ML compared to control. CGA may contribute to such effects, as it has shown to suppress the transcription of inflammatory cytokines and inhibit activation of the NF-κB signaling pathway [52]. Histological evaluation of livers also demonstrated less lipid droplet accumulation in the ML groups, in total agreement with hepatic lipids and inflammatory makers.
The decreases in hepatic TG can also be associated with the lower expression of DGAT2. DGAT2, is a key microsomal enzyme involved in TG biosynthesis, and its function is to acylates diacylglycerol at the sn-3 position, using fatty acyl CoAs [53]. Quercetin, a flavonoid, has been shown to reduce TG synthesis in Caco-2 cells, in part via the reduction of DGAT2 activity [54]. Since ML have significant concentrations of quercetin [46], we can speculate that this flavonoid might have been involved in the decreased expression of DGAT2, further contributing to the observed lower concentrations of TG in the guinea pigs fed the ML.
We conclude that ML regulate key genes involved in lipid synthesis, resulting in lower concentrations of lipids and inflammatory cytokines in the liver, demonstrating the effectiveness of ML in reducing fatty liver in cholesterol-fed guinea pigs. Further studies are needed to examine the effects of ML on liver health in humans.
4. Materials and Methods
4.1. Experimental Design
Male Hartley guinea pigs (n = 8 per group) were allocated into three groups: Control, Low Moringa (LM) (10 g/100 g), and High Moringa (HM) (15 g/100 g) for 6 weeks. This amount of Moringa was chosen based on 20 g of food consumed by guinea pigs per day to have 2 or 3.5 g or Moringa per day similar to human studies (Vergara-Jimenez, personal communication). At the end of the 6 weeks, guinea pigs were killed by exsanguination following heart puncture and blood was centrifuged immediately (2000× g) to separate plasma. Liver tissue was also collected, snap-frozen and stored at −80 °C until further analysis. All animal protocols were approved by the Institutional Animal Care and Use Committee (Assurance A3124-01) at the University of Connecticut (Protocol #A16-003) on 1 March 2016. Moringa leaves were a gift from Scientech from Health International (Mexico City, Mexico). Macronutrient concentration of the diet was adjusted to account for the inclusion of Moringa in the diet. Moringa composition is presented in (Table 5). A detailed description of the diets is presented in (Table 6).

Table 5.
Composition of Moringa oleifera leaves.

Table 6.
Composition of experimental diets.
4.2. Plasma Lipids, Glucose and Liver Enzymes
The concentrations of glucose, alanine aminotransferase (ALT), aspartate aminotransferase (AST) and plasma lipids, including total cholesterol (TC), LDL-C, HDL-C and triglycerides (TG) were measured by using Cobas C 111 Analyzer (Roche Diagnostics, Indianapolis, IN, USA).
4.3. Lipoprotein Subfraction and Size
The concentrations of large, medium and small VLDL, LDL and HDL particles were measured by nuclear magnetic resonance (NMR). NMR spectroscopy was performed on a 400-MHz NMR analyzer (Bruker Biospin, LipoScience Inc., Raleigh, NC, USA), as described previously [27].
4.4. Lecithin Cholesterol Acyl Transferase (LCAT) and Choleterol Ester Protein (CETP) Acitivities
The activity of LCAT was evaluated by use of a commercially available fluorometric assay kit (Cell Biolabs Inc., San Diego, CA, USA). The relative LCAT activity was assessed by the strength of the fluorescence signal following an incubation period. CETP activity was measured by use of a commercially available assay kit (BioVision Inc., Milpitas, CA, USA). The decrease in fluorescence intensity over time was used to calculate CETP activity after incubation of plasma with a self-quenching donor molecule and an acceptor molecule.
4.5. Hepatic Lipids
Hepatic tissue (100 mg) from each sample was homogenized with 200 μL of chloroform/isopropanol/MP40 (7/11/1). The mixtures were centrifuged and the supernatant dried at 50 °C overnight. The quantification for TC and free cholesterol (FC) was done with enzymatic assays kits (Wako Diagnostics, Mountain View, CA, USA) [28]. Esterified cholesterol (CE) was calculated manually by subtracting FC from TC. Liver TG were extracted (100 mg of tissue) with chloroform:methanol (2:1), dried under nitrogen at 60 °C and solubilized in 1% Triton X-100. The solubilized lipid isolates were analyzed by enzymatic methods (Wako Diagnostics, Mountain View, CA, USA), as previously described [29].
4.6. Histologic Evaluation
Small pieces taken from the same liver section were immersed in 10% buffered formalin. Formalin-fixed livers were paraffin embedded and sections measuring 3–5 μm were stained with hematoxylin. The stained tissue sections were viewed under bright field microscopy at 200× magnification. An AxioCam ICc3 camera (Zeiss, Thornwood, NY, USA) was used to take the pictures.
4.7. Inflammatory Cytokine Concentration in Liver
Tissue total protein was extracted and protein concentration of the lysates was evaluated by use of the bicinchoninicc acid assay rotein Assay Kit (Cell Signaling Technologies, Inc., Beverly, MA, USA). Using the same concentration of protein for all samples, the following cytokines were measured using Luminex technology (Luminex 200 System, Austin, TX, USA) with the MILLIPLEX MAP Rat Cytokine Immunoassay kit (Millipore corporation, Charles, MO, USA): interleukin (IL)-1β, IL-6, IL-10, interferon (IFN)-γ, monocyte chemotactic protein 1 (MCP-1), and tumor necrosis factor (TNF)-α, as previously described [30].
4.8. Gene Expression
Primer design: The National Center for Biotechnology Information website was used to design the specific primers of Cavia porcellus. The following genes involved in lipid metabolism and inflammation were analyzed: LDL receptor (LDL-R), cluster of differentiation-68 (CD68); HMG-CoA reductase (HMG-CoA-R); peroxisome proliferator-activated receptor (PPARγ), cluster of differentiation-36 (CD36); diacyl glycerol acyltransferase-2 (DGAT-2), Primer sequences are available upon request.
4.9. RNA Isolation, cDNA Synthesis and Real-Time qRT-PCR
RNA was extracted with TRIzol (Thermo Fisher Scientific, Pittsburgh, PA, USA) after liver tissues were homogenized. RNA was reverse transcribed to cDNA and then amplified and quantified by real-time quantitative polymerase chain reaction using a Bio-Rad CFX96 Real-Time system (BioRad, Hercules, CA, USA), as described [31]. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) expression was used in normalization using the 2−ΔΔCt method.
4.10. Statistical Analysis
Differences among groups were analyzed by one-way ANOVA and Tukey post hoc analysis. p < 0.05 was considered to be significant. All analyses were conducted on SPSS for Windows, Version 20 (IBM Corp., Armonk, NY, USA). All data are presented as mean ± Standard Deviation (SD).
Acknowledgments
Supported by funds provided Taif University, Taif, Saudi Arabia.
Author Contributions
Manal Mused Almatrafi fed and took care of the guinea pigs, contributed to all lab experiment, participated in the writing of the manuscript and analyzed and interpreted the data; Marcela Vergara-Jimenez participated in experimental design, did all the analysis related to gene expression and contributed to data analysis and interpretation and revised the written document; Ana Gabriela Murillo helped with taking care of guinea pigs and some of the analysis and provided input in data analysis and interpretation; Gregory H. Norris helped with laboratory assays and data interpretation; provided input in data analysis and the writing of the manuscript; Christopher N. Blesso participated in experimental design, data analysis and in providing input for the final version of the manuscript. Maria Luz Fernandez was responsible for the design of the experiment, for supervising all assays, data analysis, interpretation and the production of the final manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Angulo, P. Nonalcoholic fatty liver disease. N. Engl. J. Med. 2014, 346, 1221–1231. [Google Scholar] [CrossRef] [PubMed]
- Tolman, K.G.; Dalpiaz, A.S. Treatment of non-alcoholic fatty liver disease. Ther. Clin. Risk Manag. 2007, 3, 1153–1163. [Google Scholar] [PubMed]
- Krawczyk, M.; Bonfrate, L.; Portincasa, P. Nonalcoholic fatty liver disease. Best Pract. Res. Clin. Gastroenterol. 2010, 24, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Abenavoli, L.; Milic, N.; Peta, V.; Alfieri, F.; de Lorenzo, A.; Bellentani, S. Alimentary regimen in non-alcoholic fatty liver disease: Mediterranean diet. World J. Gastroenterol. 2014, 20, 16831–16840. [Google Scholar] [CrossRef] [PubMed]
- Masarone, M.; Federico, A.; Abenavoli, L.; Loguercio, C.; Persico, M. Non alcoholic fatty liver: Epidemiology and natural history. Rev. Recent Clin. Trials 2014, 9, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Murillo, A.; DiMarco, D.; Fernandez, M. The potential of non-provitamin A carotenoids for the prevention and treatment of non-alcoholic fatty liver disease. Biology 2016, 5, 42. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tan, H.-Y.; Wang, N.; Zhang, Z.-J.; Lao, L.; Wong, C.-W.; Feng, Y. The role of oxidative stress and antioxidants in liver diseases. Int. J. Mol. Sci. 2015, 16, 26087–26124. [Google Scholar] [CrossRef] [PubMed]
- Leone, A.; Spada, A.; Battezzati, A.; Schiraldi, A.; Aristil, J.; Bertoli, S. Moringa oleifera seeds and oil: Characteristics and uses for human health. Int. J. Mol. Sci. 2016, 17, 2141. [Google Scholar] [CrossRef] [PubMed]
- Leone, A.; Spada, A.; Battezzati, A.; Schiraldi, A.; Aristil, J.; Bertoli, S. Cultivation, genetic, ethnopharmacology, phytochemistry and pharmacology of Moringa oleifera leaves: An overview. Int. J. Mol. Sci. 2015, 16, 12791–12835. [Google Scholar] [CrossRef] [PubMed]
- Adedapo, A.A.; Falayi, O.O.; Oyagbemi, A.A. Evaluation of the analgesic, anti-inflammatory, anti-oxidant, phytochemical and toxicological properties of the methanolic leaf extract of commercially processed Moringa oleifera in some laboratory animals. J. Basic Clin. Physiol. Pharmacol. 2015, 26, 491–499. [Google Scholar] [CrossRef] [PubMed]
- Al-Asmari, A.K.; Al-Elaiwi, A.M.; Athar, M.T.; Tariq, M.; Al Eid, A.; Al-Asmary, S.M. A review of hepatoprotective plants used in Saudi traditional medicine. Evid. Based Complement. Altern. Med. 2014, 2014, 890842. [Google Scholar] [CrossRef] [PubMed]
- Ghasi, S.; Nwobodo, E.; Ofili, J.O. Hypocholesterolemic effects of crude extract of leaf of Moringa oleifera Lam in high-fat diet fed wistar rats. J. Ethnopharmacol. 2000, 69, 21–25. [Google Scholar] [CrossRef]
- Fakurazi, S.; Hairuszah, I.; Nanthini, U. Moringa oleifera Lam prevents acetaminophen induced liver injury through restoration of glutathione level. Food Chem. Toxicol. 2008, 46, 2611–2615. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, D.; Rai, P.K.; Kumar, A.; Mehta, S.; Watal, G. Effect of Moringa oleifera Lam leaves aqueous extract therapy on hyperglycemic rats. J. Ethnopharmacol. 2009, 123, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, D.; Rai, P.K.; Mehta, S.; Chatterji, S.; Shukla, S.; Rai, D.K.; Sharma, G.; Sharma, B.; Khair, S.; Watal, G. Role of Moringa oleifera in regulation of diabetes-induced oxidative stress. Asian Pac. J. Trop. Med. 2013, 6, 426–432. [Google Scholar] [CrossRef]
- Nandave, M.; Ojha, S.K.; Joshi, S.; Kumari, S.; Arya, D.S. Moringa oleifera leaf extract prevents isoproterenol-induced myocardial damage in rats: Evidence for an antioxidant, antiperoxidative, and cardioprotective intervention. J. Med. Food 2009, 12, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Mehta, L.K.; Balaraman, R.; Amin, A.H.; Bafna, P.A.; Gulati, O.D. Effect of fruits of Moringa oleifera on the lipid profile of normal and hypercholesterolaemic rabbits. J. Ethnopharmacol. 2003, 86, 191–195. [Google Scholar] [CrossRef]
- Chávez-Santoscoy, R.A.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Effect of flavonoids and saponins extracted from black bean (Phaseolus vulgaris L.) seed coats as cholesterol micelle disruptors. Plant Foods Hum. Nutr. 2013, 68, 416–423. [Google Scholar] [CrossRef] [PubMed]
- Minaiyan, M.; Asghari, G.; Taheri, D.; Saeidi, M.; Nasr-Esfahani, S. Anti-inflammatory effect of Moringa oleifera Lam seeds on acetic acid-induced acute colitiis in rats. Avicenna J. Phytomed. 2014, 4, 127–136. [Google Scholar] [PubMed]
- Singh, B.N.; Singh, B.R.; Singh, R.L.; Prakash, D.; Dhakarey, R.; Upadhyay, G.; Singh, H.B. Oxidative DNA damage protective activity, antioxidant and anti-quorum sensing potentials of Moringa oleifera. Food Chem. Toxicol. 2009, 47, 1109–1116. [Google Scholar] [CrossRef] [PubMed]
- Labbe, D.; Provencal, M.; Lamy, S.; Boivin, D.; Gingras, D.; Beliveau, R.; Be, R.; Provenc, M. The flavonols quercetin, kaempferol, and myricetin inhibit hepatocyte growth factor-induced medulloblastoma cell migration. J. Nutr. 2009, 139, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Pari, L.; Kumar, N.A. Hepatoprotective activity of Moringa oleifera on antitubercular drug-induced liver damage in rats. J. Med. Food 2002, 5, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Sharifudin, S.A.; Fakurazi, S.; Hidayat, M.T.; Hairuszah, I.; Aris Mohd Moklas, M.; Arulselvan, P. Therapeutic potential of Moringa oleifera extracts against acetaminophen-induced hepatotoxicity in rats. Pharm. Biol. 2013, 51, 279–288. [Google Scholar] [CrossRef] [PubMed]
- Hamza, A.A. Ameliorative effects of Moringa oleifera Lam seed extract on liver fibrosis in rats. Food Chem. Toxicol. 2010, 48, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Sinha, M.; Das, D.K.; Bhattacharjee, S.; Majumdar, S.; Dey, S. Leaf extract of Moringa oleifera prevents ionizing radiation-induced oxidative stress in mice. J. Med. Food 2011, 14, 1167–1172. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Hartman, M.J. Review of the safety and efficacy of Moringa oleifera. Phytother. Res. 2015, 29, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Al-Sarraj, T.; Saadi, H.; Volek, J.S.; Fernandez, M.L. Carbohydrate restriction favorably alters lipoprotein metabolism in Emirati subjects classified with the metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2010, 20, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Carr, T.P.; Andresen, C.J.; Rudel, L.L. Enzymatic determination of triglyceride, free cholesterol, and total cholesterol in tissue lipid extracts. Clin. Biochem. 1993, 26, 39–42. [Google Scholar] [CrossRef]
- Kim, J.E.; Leite, J.O.; Smyth, J.A.; Clark, R.M.; Fernandez, M.L. A lutein-enriched diet prevents cholesterol accumulation and decreases oxidized LDL and inflammatory cytokines in the aorta of guinea pigs. J. Nutr. 2011, 141, 1458–1463. [Google Scholar] [CrossRef] [PubMed]
- Murillo, A.G.; Aguilar, D.; Norris, G.H.; Dimarco, D.M.; Missimer, A.; Hu, S.; Smyth, J.A.; Gannon, S.; Blesso, C.N.; Luo, Y.; et al. Compared with powdered lutein, a lutein nanoemulsion increases plasma and liver lutein, protects against hepatic steatosis, and affects lipoprotein metabolism in guinea pigs. J. Nutr. 2016, 146, 1961–1969. [Google Scholar] [CrossRef] [PubMed]
- Farrell, N.; Norris, G.; Lee, S.G.; Chun, O.K.; Blesso, C.N. Anthocyanin-rich black elderberry extract improves markers of HDL function and reduces aortic cholesterol in hyperlipidemic mice. Food Funct. 2015, 6, 1278–1287. [Google Scholar] [CrossRef] [PubMed]
- Li, X.J.; Mu, Y.M.; Li, T.T.; Yang, Y.L.; Zhang, M.T.; Li, Y.S.; Zhang, W.K.; Tang, H.B.; Shang, H.C. Gynura procumbens reverses acute and chronic ethanol-induced liver steatosis through MAPK/SREBP-1c-dependent and -independent pathways. J. Agric. Food Chem. 2015, 63, 8460–8471. [Google Scholar] [CrossRef] [PubMed]
- Meng, S.; Cao, J.; Feng, Q.; Peng, J.; Hu, Y. Roles of chlorogenic acid on regulating glucose and lipids metabolism: A review. Evid. Based Complement. Altern. Med. 2013, 2013, 801457. [Google Scholar] [CrossRef] [PubMed]
- Gebregiorgis, F.; Negesse, T.; Nurfeta, A. Feed intake and utilization in sheep fed graded levels of dried Moringa (Moringa stenopetala) leaf as a supplement to Rhodes grass hay. Trop. Anim. Health Prod. 2012, 44, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Giannini, E.G.; Testa, R.; Savarino, V. Liver enzyme alteration: A guide for clinicians. Can. Med. Assoc. J. 2005, 172, 367–379. [Google Scholar] [CrossRef] [PubMed]
- Ouédraogo, M.; Lamien-Sanou, A.; Ramdé, N.; Ouédraogo, A.S.; Ouédraogo, M.; Zongo, S.P.; Goumbri, O.; Duez, P.; Guissou, P.I. Protective effect of Moringa oleifera leaves against gentamicin-induced nephrotoxicity in rabbits. Exp. Toxicol. Pathol. 2013, 65, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.L.; Vergara-Jimenez, M.; Conde, K.; Behr, T.; Abdel-Fattah, G. Regulation of apolipoprotein B-containing lipoproteins by dietary soluble fiber in guinea pigs. Am. J. Clin. Nutr. 1997, 65, 814–822. [Google Scholar] [PubMed]
- Osono, Y.; Woollett, L.A.; Herz, J.; Dietschy, J.M. Role of the low density lipoprotein receptor in the flux of cholesterol through the plasma and across the tissues of the mouse. J. Clin. Invest. 1995, 95, 1124–1132. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.L.; Ruiz, L.R.; Conde, A.K.; Sun, D.M.; Erickson, S.K.; McNamara, D.J. Psyllium reduces plasma LDL in guinea pigs by altering hepatic cholesterol homeostasis. J. Lipid Res. 1995, 36, 1128–1138. [Google Scholar] [PubMed]
- Fernandez, M.L.; Vergara-Jimenez, M.; Conde, K.; Abdel-Fattah, G. Dietary carbohydrate type and fat amount alter VLDL and LDL metabolism in guinea pigs. J. Nutr. 1996, 126, 2494–2504. [Google Scholar] [PubMed]
- Duprez, D.A.; Otvos, J.; Tracy, R.P.; Feingold, K.R.; Gross, M.D.; Lima, J.A.C.; Mackey, R.H.; Neaton, J.D.; Sanchez, O.A.; Jacobs, D.R. High-density lipoprotein subclasses and noncancer chronic inflammatory-related events versus cardiovascular events: The multi-ethnic study of atherosclerosis. J. Am. Heart Assoc. 2015, 4, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Mutungi, G.; Waters, D.; Ratliff, J.; Puglisi, M.; Clark, R.M.; Volek, J.S.; Luz, M. Eggs distinctly modulate plasma carotenoid and lipoprotein subclasses in adult men following a carbohydrate-restricted diet. J. Nutr. Biochem. 2009, 21, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Ossoli, A.; Pavanello, C.; Calabresi, L. High-density lipoprotein, lecithin: Cholesterol acyltransferase, and atherosclerosis. Endocrinol. Metab. 2016, 31, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Barter, P.J.; Rye, K.-A. Cholesteryl ester transfer protein inhibition as a strategy to reduce cardiovascular risk. J. Lipid Res. 2012, 53, 1755–1766. [Google Scholar] [CrossRef] [PubMed]
- De Ogburn, R.; Leite, J.O.; Ratliff, J.; Volek, J.S.; McGrane, M.M.; Fernandez, M.L. Effects of increased dietary cholesterol with carbohydrate restriction on hepatic lipid metabolism in guinea pigs. Comp. Med. 2012, 62, 109–115. [Google Scholar]
- Atawodi, S.E.; Atawodi, J.C.; Idakwo, G.A.; Pfundstein, B.; Haubner, R.; Wuntele, G.; Bartsch, H.; Owen, R.W. Evaluation of the polypehnol content and antioxidant properties of methanol extracts of the leaves, stem and root barks of Moringa oleifera Lam. J. Med. Food 2010, 13, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.; Dong, L.; Dang, X.; Liu, Y.; Jiang, J.; Wang, Y.; Lu, X.; Guo, X. Effect of chlorogenic acid on LPS-induced proinflammatory signaling in hepatic stellate cells. Inflamm. Res. 2013, 62, 581–587. [Google Scholar] [CrossRef] [PubMed]
- Cho, A.S.; Jeon, S.M.; Kim, M.J.; Yeo, J.; Seo, K.I.; Choi, M.S.; Lee, M.K. Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem. Toxicol. 2010, 48, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Horton, J.D. Sterol regulatory element-binding proteins: Transcriptional activators of lipid synthesis. Biochem. Soc. Trans. 2002, 30, 1091–1095. [Google Scholar] [CrossRef] [PubMed]
- Ioannou, G.N. The role of cholesterol in the pathogenesis of NASH. Trends Endocrinol. Metab. 2016, 27, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Waterman, C.; Cheng, D.M.; Rojas-Silva, P.; Poulev, A.; Dreifus, J.; Lila, M.A.; Raskin, I. Stable, water extractable isothiocyanates from Moringa oleifera leaves attenuate inflammation in vitro. Phytochemistry 2014, 103, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.; Shi, L.; Sheng, Y.; Zhang, J.; Lu, B.; Ji, L. Chlorogenic acid suppresses monocrotaline-induced sinusoidal obstruction syndrome: The potential contribution of NFκB, Egr1, Nrf2, MAPKs and PI3K signals. Environ. Toxicol. Pharmacol. 2016, 46, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, R.; Tobe, K.; Aoyama, M.; Sakamoto, K.; Ohsugi, M.; Kamei, N.; Nemoto, S.; Inoue, A.; Ito, Y.; Uchida, S.; et al. Expression of DGAT2 in white adipose tissue is regulated by central leptin action. J. Biol. Chem. 2005, 280, 3331–3337. [Google Scholar] [CrossRef] [PubMed]
- Casaschi, A.; Wang, Q.; Dang, K.O.; Richards, A.; Theriault, A. Intestinal Apolipoprotein B secretion is inhibited by the flavonoid quercitin: Potential role of microsomal triglyceride transfer protein and dicaylglycerol acyltranferase. Lipids 2002, 37, 547–652. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).