Cell Wall Modifications in Giant Cells Induced by the Plant Parasitic Nematode Meloidogyne incognita in Wild-Type (Col-0) and the fra2 Arabidopsis thaliana Katanin Mutant
Abstract
:1. Introduction
2. Results
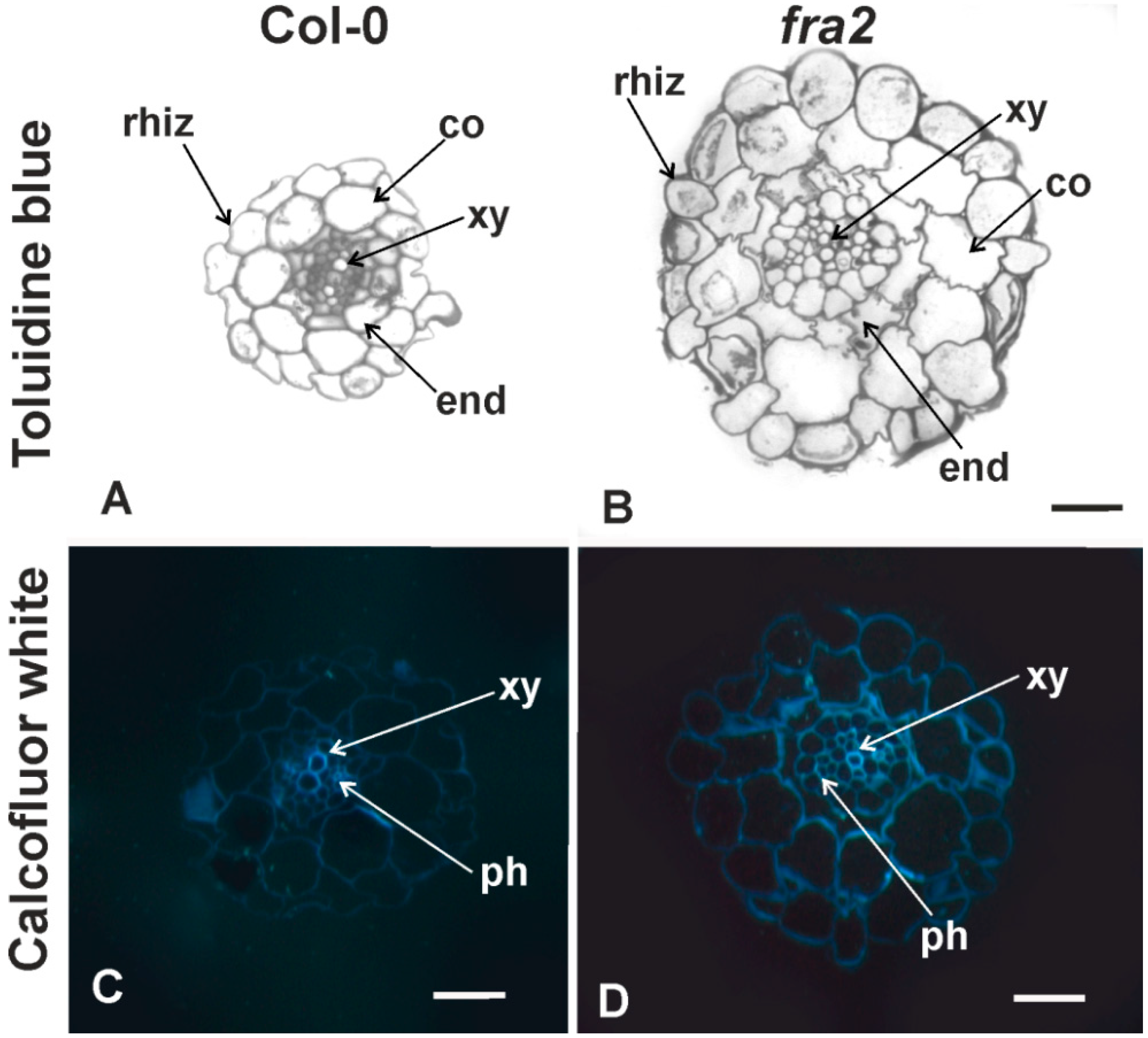
2.1. Cell Wall Hemicellulose and Pectin Epitope Analysis of Uninfected Roots
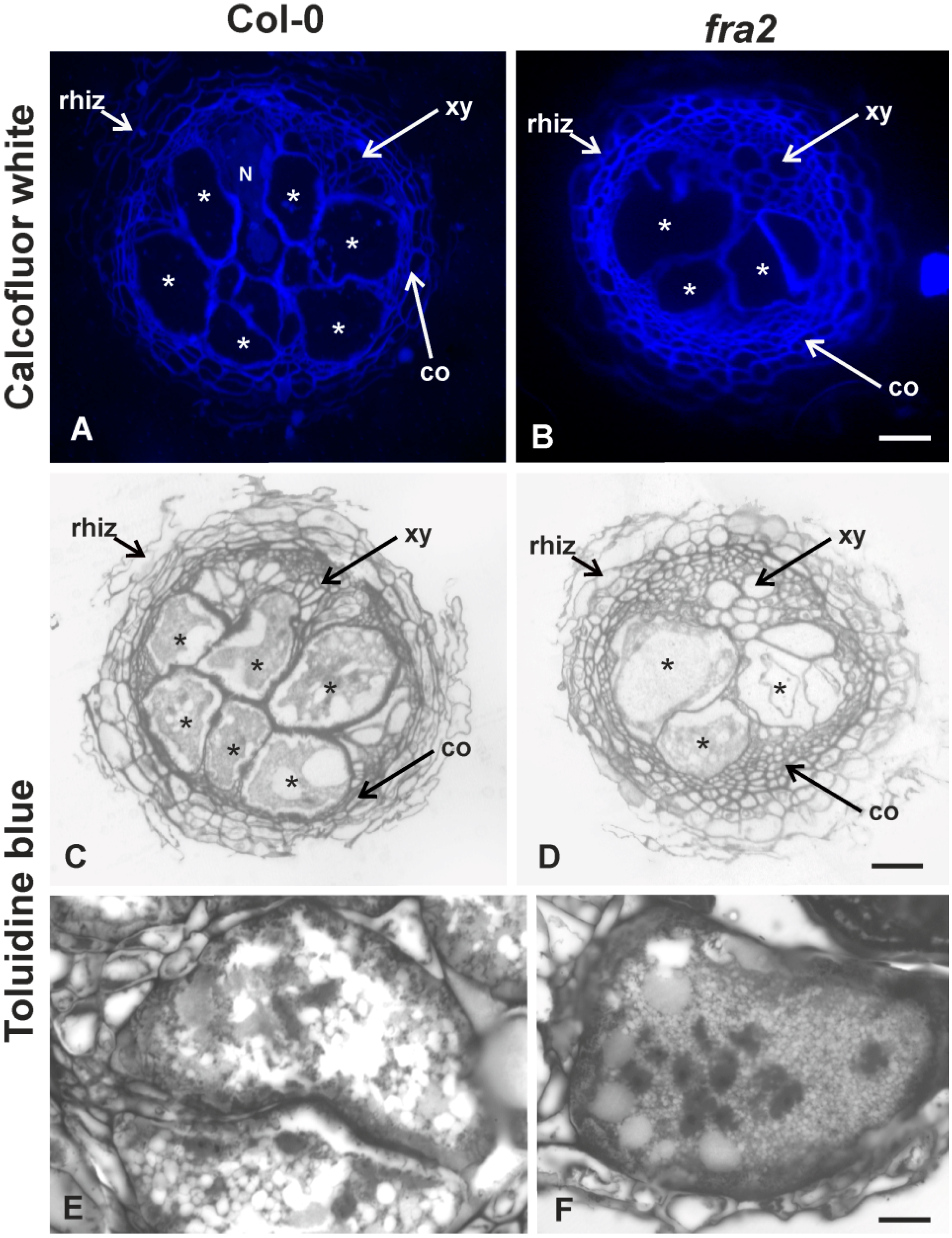
2.2. Hemicellulose and Pectin Distribution Patterns of Giant Cell Walls of Col-0 and fra2 Roots
2.3. The Impact of Katanin Mutation on the Infection of A.thaliana by M. incognita
3. Discussion
4. Materials and Methods
4.1. Plant Growth Conditions, Nematode Rearing and Collection
4.2. Light and Fluorescence Microscopy
4.3. Cell Wall Epitope Immunolabeling
4.4. Arabidopsis Wild-Type and fra2 Mutant Nematode Susceptibility
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| RKN | Root-Knot Nematodes |
| GCs | Giant Cells |
| MT | Microtubules |
| DeSPHG | De-esterified Pectic HomoGalacturonan |
| MPHG | Methylesterified Pectic HomoGalacturonan |
References
- Escobar, C.; Barcala, M.; Cabrera, J.; Fenoll, C. Overview of root-knot nematodes and giant cells. Adv. Bot. Res. 2015, 73, 1–32. [Google Scholar]
- Koenning, S.R.; Wrather, J.A.; Kirkpatrick, T.L.; Walker, N.R.; Starr, J.L.; Mueller, J.D. Plant-parasitic nematodes attacking cotton in the United States: Old and emerging production challenges. Plant Dis. 2004, 88, 100–113. [Google Scholar] [CrossRef] [PubMed]
- Djian-Caporalino, C. Root-knot nematodes (Meloidogyne spp.), a growing problem in French vegetable crops. EPPO Bull. 2012, 42, 127–137. [Google Scholar] [CrossRef]
- Onkendi, E.M.; Kariuki, G.M.; Marais, M.; Moleleki, L.N. The threat of root-knot nematodes (Meloidogyne spp.) in Africa: A review. Plant Pathol. 2014, 63, 727–737. [Google Scholar] [CrossRef]
- Kyndt, T.; Vieira, P.; Gheysen, G.; de Almeida-Engler, J. Nematode feeding sites: Unique organs in plant roots. Planta 2013, 238, 807–818. [Google Scholar] [CrossRef]
- Caillaud, M.C.; Dubreuil, G.; Quentin, M.; Perfus-Barbeoch, L.; Lecomte, P.; de Almeida Engler, J.; Abad, P.; Rosso, M.N.; Favery, B. Root-knot nematodes manipulate plant cell functions during a compatible interaction. J. Plant Physiol. 2008, 165, 104–113. [Google Scholar] [CrossRef]
- Shukla, N.; Yadav, R.; Kaur, P.; Rasmussen, S.; Goel, S.; Agarwal, M.; Jagannath, A.; Gupta, R.; Kumar, A. Transcriptome analysis of root-knot nematode (Meloidogyne incognita)-infected tomato (Solanum lycopersicum) roots reveals complex gene expression profiles and metabolic networks of both host and nematode during susceptible and resistance responses. Mol. Plant Pathol. 2018, 19, 615–633. [Google Scholar] [CrossRef]
- Širca, S.; Urek, G.; Karssen, G. Occurrence of the root-knot nematodes Meloidogyne incognita and M. hapla in Slovenia. Plant Dis. 2003, 87, 1150. [Google Scholar] [CrossRef]
- Snyder, D.W.; Opperman, C.H.; Bird, D.M.K. A method for generating Meloidogyne incognita males. J. Nematol. 2006, 38, 192–194. [Google Scholar]
- Holbein, J.; Grundler, F.M.W.; Siddique, S. Plant basal resistance to nematodes: An update. J. Exp. Bot. 2016, 67, 2049–2061. [Google Scholar] [CrossRef]
- Bozbuga, R.; Lilley, C.J.; Knox, J.P.; Urwin, P.E. Host-specific signatures of the cell wall changes induced by the plant parasitic nematode, Meloidogyne incognita. Sci. Rep. 2018, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Veronico, P.; Paciolla, C.; Pomar, F.; De Leonardis, S.; García-Ulloa, A.; Melillo, M.T. Changes in lignin biosynthesis and monomer composition in response to benzothiadiazole and root-knot nematode Meloidogyne incognita infection in tomato. J. Plant Physiol. 2018, 230, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, D.J. Plant cell wall extensibility: Connecting plant cell growth with cell wall structure, mechanics, and the action of wall-modifying enzymes. J. Exp. Bot. 2016, 67, 463–476. [Google Scholar] [CrossRef]
- Zhang, T.; Zheng, Y.; Cosgrove, D.J. Spatial organization of cellulose microfibrils and matrix polysaccharides in primary plant cell walls as imaged by multichannel atomic force microscopy. Plant J. 2016, 85, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Kieliszewski, M.J.; Lamport, D.T.A.; Tan, L.; Cannon, M.C. Hydroxyproline-rich glycoproteins: Form and function. Ann. Plant Rev. 2011, 41, 321–342. [Google Scholar]
- Bohlmann, H.; Sobczak, M. The plant cell wall in the feeding sites of cyst nematodes. Front. Plant Sci. 2014, 5, 1–10. [Google Scholar] [CrossRef]
- Zhang, L.; Lilley, C.J.; Imren, M.; Paul Knox, J.; Urwin, P.E. The complex cell wall composition of syncytia induced by plant parasitic cyst nematodes reflects both function and host plant. Front. Plant Sci. 2017, 8, 1–12. [Google Scholar] [CrossRef]
- Giannoutsou, E.; Sotiriou, P.; Nikolakopoulou, T.L.; Galatis, B.; Apostolakos, P. Callose and homogalacturonan epitope distribution in stomatal complexes of Zea mays and Vigna sinensis. Protoplasma 2019. [Google Scholar] [CrossRef]
- Szymanski, D.B.; Cosgrove, D.J. Dynamic coordination of cytoskeletal and cell wall systems during plant cell morphogenesis. Curr. Biol. 2009, 19, R800–R811. [Google Scholar] [CrossRef]
- Luptovčiak, I.; Komis, G.; Takáč, T.; Ovečka, M.; Šamaj, J. Katanin: A sword cutting microtubules for cellular, developmental, and physiological purposes. Front. Plant Sci. 2017, 8, 1982. [Google Scholar] [CrossRef]
- Burk, D.H.; Ye, Z.H. Alteration of oriented deposition of cellulose microfibrils by mutation of a katanin-like microtubule-severing protein. Plant Cell 2002, 14, 2145–2160. [Google Scholar] [CrossRef] [PubMed]
- Burk, D.H.; Liu, B.; Zhong, R.; Morrison, W.H.; Ye, Z.H. A katanin-like protein regulates normal cell wall biosynthesis and cell elongation. Plant Cell 2001, 13, 807–827. [Google Scholar] [CrossRef] [PubMed]
- Burk, D.H.; Zhong, R.; Ye, Z.H. The katanin microtubule severing protein in plants. J. Integr. Plant Biol. 2007, 49, 1174–1182. [Google Scholar] [CrossRef]
- Panteris, E.; Adamakis, I.D.S.; Voulgari, G.; Papadopoulou, G. A role for katanin in plant cell division: Microtubule organization in dividing root cells of fra2 and lue1 Arabidopsis thaliana mutants. Cytoskeleton 2011, 68, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Giannoutsou, E.; Sotiriou, P.; Apostolakos, P.; Galatis, B. Early local differentiation of the cell wall matrix defines the contact sites in lobed mesophyll cells of Zea mays. Ann. Bot. 2013, 112, 1067–1081. [Google Scholar] [CrossRef]
- Sotiriou, P.; Giannoutsou, E.; Panteris, E.; Apostolakos, P.; Galatis, B. Cell wall matrix polysaccharide distribution and cortical microtubule organization: Two factors controlling mesophyll cell morphogenesis in land plants. Ann. Bot. 2016, 117, 401–419. [Google Scholar] [CrossRef]
- Dolan, L.; Davies, J. Cell expansion in roots. Curr. Opin. Plant Biol. 2004, 7, 33–39. [Google Scholar] [CrossRef]
- Arioli, T.; Peng, L.; Betzner, A.S.; Burn, J.; Wittke, W.; Herth, W.; Camilleri, C.; Höfte, H.; Plazinski, J.; Birch, R.; et al. Molecular analysis of cellulose biosynthesis in Arabidopsis. Science 1998, 279, 717–720. [Google Scholar] [CrossRef]
- McCartney, L.; Steele-King, C.G.; Jordan, E.; Knox, J.P. Cell wall pectic (1→4)-β-D-galactan marks the acceleration of cell elongation in the Arabidopsis seedling root meristem. Plant J. 2003, 33, 447–454. [Google Scholar] [CrossRef]
- Dolan, L.; Linstead, P.; Roberts, K. Developmental regulation of pectic polysaccharides in the root meristem of Arabidopsis. J. Exp. Bot. 1997, 48, 713–720. [Google Scholar] [CrossRef]
- Dolan, L.; Janmaat, K.; Willemsen, V.; Linstead, P.; Poethig, S.; Roberts, K.; Scheres, B. Cellular organisation of the Arabidopsis thaliana root. Development 1993, 119, 71–84. [Google Scholar] [PubMed]
- Somssich, M.; Khan, G.A.; Staffan, S.P. Cell wall heterogeneity in root development of arabidopsis. Front. Plant Sci. 2016, 7, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Panteris, E.; Diannelidis, B.-E.; Adamakis, I.-D.S. Cortical microtubule orientation in Arabidopsis thaliana root meristematic zone depends on cell division and requires severing by katanin. J. Biol. Res. 2018, 25, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Saffer, A.M. Expanding roles for pectins in plant development. J. Integr. Plant Biol. 2018, 60, 910–923. [Google Scholar] [CrossRef] [PubMed]
- Cosgrove, D.J. Diffuse growth of plant cell walls. Plant Physiol. 2018, 176, 16–27. [Google Scholar] [CrossRef] [PubMed]
- Uyttewaal, M.; Burian, A.; Alim, K.; Landrein, B.; Borowska-Wykrt, D.; Dedieu, A.; Peaucelle, A.; Ludynia, M.; Traas, J.; Boudaoud, A.; et al. Mechanical stress acts via Katanin to amplify differences in growth rate between adjacent cells in Arabidopsis. Cell 2012, 149, 439–451. [Google Scholar] [CrossRef]
- Zhao, F.; Chen, W.; Sechet, J.; Martin, M.; Bovio, S.; Lionnet, C.; Long, Y.; Battu, V.; Mouille, G.; Monéger, F.; et al. Xyloglucan homeostasis and microtubule dynamics synergistically maintain meristem geometry and robustness of phyllotaxis in Arabidopsis. bioRxiv 2019, 607481. [Google Scholar] [CrossRef]
- Davies, L.J.; Lilley, C.J.; Knox, J.P.; Urwin, P.E. Syncytia formed by adult female Heterodera schachtii in Arabidopsis thaliana roots have a distinct cell wall molecular architecture. New Phytol. 2012, 196, 238–246. [Google Scholar] [CrossRef]
- De Storme, N.; Geelen, D. The impact of environmental stress on male reproductive development in plants: Biological processes and molecular mechanisms. Plant Cell Environ. 2014, 37, 1–18. [Google Scholar] [CrossRef]
- Piršelová, B.; Matušíková, I. Callose: The plant cell wall polysaccharide with multiple biological functions. Acta Physiol. Plant. 2013, 35, 635–644. [Google Scholar] [CrossRef]
- Eleftheriou, E.P.; Adamakis, I.D.S.; Melissa, P. Effects of hexavalent chromium on microtubule organization, ER distribution and callose deposition in root tip cells of Allium cepa L. Protoplasma 2012, 249, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Voigt, C.A. Callose-mediated resistance to pathogenic intruders in plant defense-related papillae. Front. Plant Sci. 2014, 5, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Fangel, J.U.; Ulvskov, P.; Knox, J.P.; Mikkelsen, M.D.; Harholt, J.; Popper, Z.A.; Willats, W.G.T. Cell wall evolution and diversity. Front. Plant Sci. 2012, 3, 152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Caillaud, M.C.; Lecomte, P.; Jammes, F.; Quentin, M.; Pagnotta, S.; Andrio, E.; Engler, J.D.A.; Marfaing, N.; Gounon, P.; Abad, P.; et al. MAP65-3 microtubule-associated protein is essential for nematode-induced giant cell ontogenesis in Arabidopsis. Plant Cell 2008, 20, 423–437. [Google Scholar] [CrossRef]
- De Almeida Engler, J.; Van Poucke, K.; Karimi, M.; De Groodt, R.; Gheysen, G.; Engler, G.; Gheysen, G. Dynamic cytoskeleton rearrangements in giant cells and syncytia of nematode-infected roots. Plant J. 2004, 38, 12–26. [Google Scholar] [CrossRef]
- Parre, E.; Geitmann, A. More than a leak sealant. The mechanical properties of callose in pollen tubes. Plant Physiol. 2005, 137, 274–286. [Google Scholar] [CrossRef]
- Adamakis, I.D.S.; Panteris, E.; Eleftheriou, E.P. Tungsten disrupts root growth in Arabidopsis thaliana by PIN targeting. J. Plant Physiol. 2014, 171, 1174–1187. [Google Scholar] [CrossRef]
- Ntalli, N.; Monokrousos, N.; Rumbos, C.; Kontea, D.; Zioga, D.; Argyropoulou, M.D.; Menkissoglu-Spiroudi, U.; Tsiropoulos, N.G. Greenhouse biofumigation with Melia azedarach controls Meloidogyne spp. and enhances soil biological activity. J. Pest Sci. 2018, 91, 29–40. [Google Scholar] [CrossRef]
- Kimenju, J.; Sibanda, Z.; Talwana, H.; Wanjohi, W. Nematology Training Manual; NIESA: Harare, Zimbabwe, 2004; pp. 1–128. [Google Scholar]
- Eisenback, J.D.; Triantaphyllou, H.H. Root-knot Nematodes: Meloidogyne species and races. Man. Agric. Nematol. 1991, 191–274. [Google Scholar]
- Panteris, E.; Komis, G.; Adamakis, I.D.S.; Šamaj, J.; Bosabalidis, A.M. MAP65 in tubulin/colchicine paracrystals of Vigna sinensis root cells: Possible role in the assembly and stabilization of atypical tubulin polymers. Cytoskeleton 2010, 67, 152–160. [Google Scholar]
- Verbelen, J.-P.; Cnodder, T.D.; Le, J.; Vissenberg, K.; Baluška, F. The root apex of Arabidopsis thaliana consists of four distinct zones of growth activities. Plant Signal. Behav. 2006, 1, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Palomares-Rius, J.E.; Escobar, C.; Cabrera, J.; Vovlas, A.; Castillo, P. Anatomical alterations in plant tissues induced by plant-parasitic nematodes. Front. Plant Sci. 2017, 8, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Schenk, S.T.; Schikora, A. Staining of callose depositions in root and leaf tissues. Bio Protoc. 2015, 5, e1429. [Google Scholar] [CrossRef]
- Eleftheriou, E.P.; Adamakis, I.D.S.; Panteris, E.; Fatsiou, M. Chromium-induced ultrastructural changes and oxidative stress in roots of Arabidopsis thaliana. Int. J. Mol. Sci. 2015, 16, 15852–15871. [Google Scholar] [CrossRef] [PubMed]
- Kondori, N.; Edebo, L.; Mattsby-Baltzer, I. A novel monoclonal antibody recognizing β(1-3) glucans in intact cells of Candida and Cryptococcus. APMIS 2008, 116, 867–876. [Google Scholar] [CrossRef]
- Falconer, M.M.; Seagull, R.W. Immunofluorescent and calcofluor white staining of developing tracheary elements in Zinnia elegans L. suspension cultures. Protoplasma 1985, 125, 190–198. [Google Scholar] [CrossRef]
- Adamakis, I.D.S.; Panteris, E.; Eleftheriou, E.P. Tubulin acetylation mediates bisphenol a effects on the microtubule arrays of Allium cepa and Triticum turgidum. Biomolecules 2019, 9, 185. [Google Scholar] [CrossRef]
- Verhertbruggen, Y.; Marcus, S.E.; Haeger, A.; Ordaz-Ortiz, J.J.; Knox, J.P. An extended set of monoclonal antibodies to pectic homogalacturonan. Carbohydr. Res. 2009, 344, 1858–1862. [Google Scholar] [CrossRef]
- Knox, J.P.; Linstead, P.J.; King, J.; Cooper, C.; Roberts, K. Pectin esterification is spatially regulated both within cell walls and between developing tissues of root apices. Planta 1990, 181, 512–521. [Google Scholar] [CrossRef]
- Pedersen, H.L.; Fangel, J.U.; McCleary, B.; Ruzanski, C.; Rydahl, M.G.; Ralet, M.C.; Farkas, V.; Von Schantz, L.; Marcus, S.E.; Andersen, M.C.F.; et al. Versatile high resolution oligosaccharide microarrays for plant glycobiology and cell wall research. J. Biol. Chem. 2012, 287, 39429–39438. [Google Scholar] [CrossRef]
- Bybd, D.W.; Kirkpatrick, T.; Barker, K.R. An improved technique for clearing and staining plant tissues for detection of nematodes. J. Nematol. 1983, 15, 142–143. [Google Scholar] [PubMed]





| Cell Wall Components | A. thaliana | ||
|---|---|---|---|
| Col-0 GCs | fra2 GCs | ||
| Hemicelluloses | Xyloglucans | +/+ | +/+ |
| Callose | +/+ | − | |
| Pectins | MPHG | +/− | +/+ |
| DeSPHG | +/+ | +/+ | |
| Arabinan | +/− | +/+ | |
| Antibody | Epitope | References |
|---|---|---|
| Hemicelluloses | ||
| LM25 | Recognizes XLLG, XXLG and XXXG motifs of xyloglucans | [61] |
| Anti-β-1,3-glucan | Recognizes 1,3 β-glucan | [57] |
| Pectins | ||
| LM20 | HG domain in pectic polysaccharides, requires methyl esters for recognition of HG and does not bind to unesterified HG | [59] |
| JIM5 | HG domain of pectic polysaccharides, recognizes partially methyl-esterified epitopes of HG, can also bind to unesterified HG | [60] |
| LM6 | Recognizes a linear pentasaccharide in (1-5)-α-L-arabinans | [59] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meidani, C.; Ntalli, N.G.; Giannoutsou, E.; Adamakis, I.-D.S. Cell Wall Modifications in Giant Cells Induced by the Plant Parasitic Nematode Meloidogyne incognita in Wild-Type (Col-0) and the fra2 Arabidopsis thaliana Katanin Mutant. Int. J. Mol. Sci. 2019, 20, 5465. https://doi.org/10.3390/ijms20215465
Meidani C, Ntalli NG, Giannoutsou E, Adamakis I-DS. Cell Wall Modifications in Giant Cells Induced by the Plant Parasitic Nematode Meloidogyne incognita in Wild-Type (Col-0) and the fra2 Arabidopsis thaliana Katanin Mutant. International Journal of Molecular Sciences. 2019; 20(21):5465. https://doi.org/10.3390/ijms20215465
Chicago/Turabian StyleMeidani, Christianna, Nikoletta G. Ntalli, Eleni Giannoutsou, and Ioannis-Dimosthenis S. Adamakis. 2019. "Cell Wall Modifications in Giant Cells Induced by the Plant Parasitic Nematode Meloidogyne incognita in Wild-Type (Col-0) and the fra2 Arabidopsis thaliana Katanin Mutant" International Journal of Molecular Sciences 20, no. 21: 5465. https://doi.org/10.3390/ijms20215465







