High Mobility Group Box-1 and Diabetes Mellitus Complications: State of the Art and Future Perspectives
Abstract
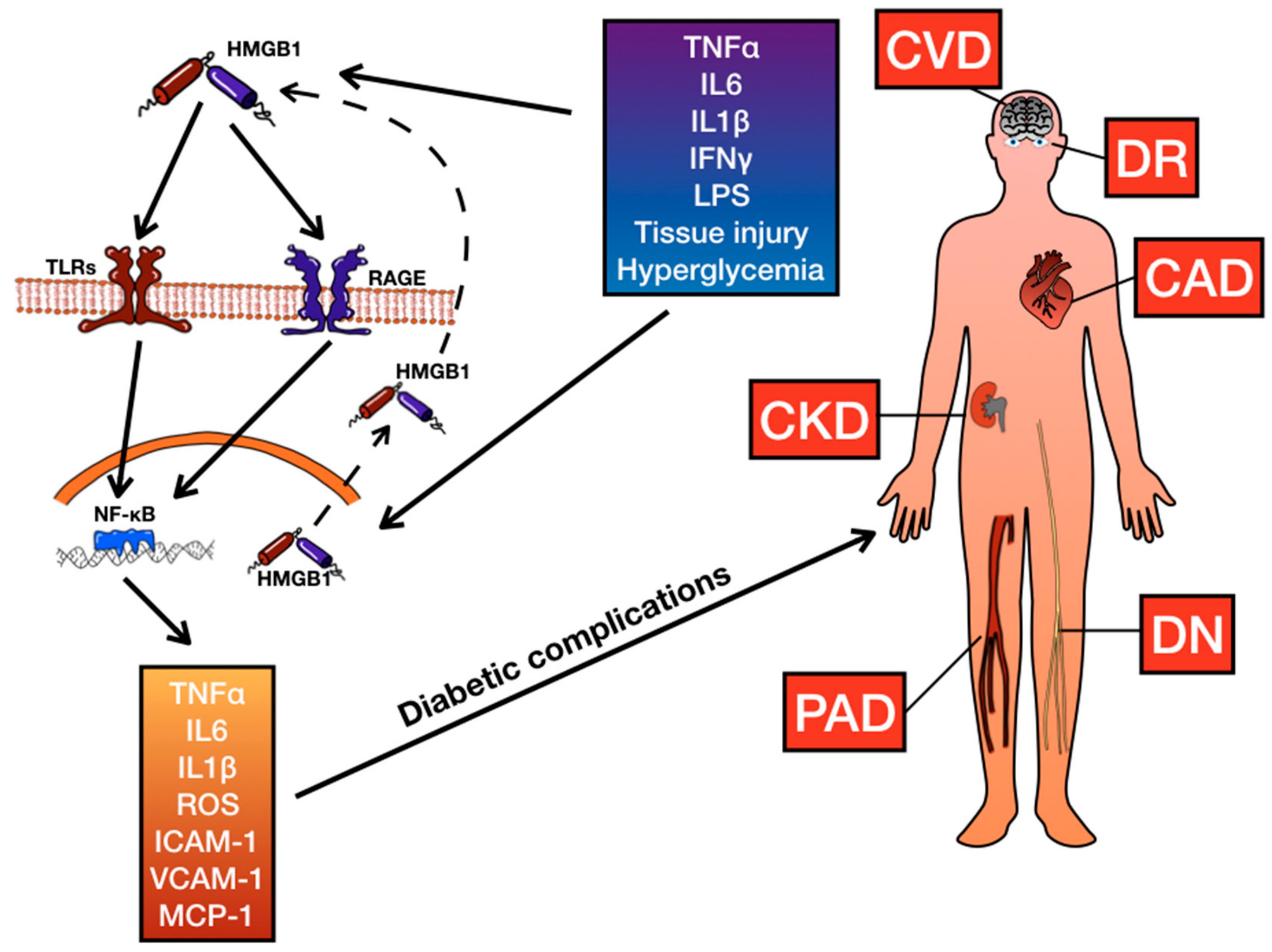
:1. Introduction
2. Diabetes Mellitus
3. HMGB1 and Diabetes
3.1. The Role of RAGE
3.2. The Role of TLRs
4. Diabetic Macroangiopathy
5. HMGB1 and Diabetic Coronary Artery Disease
6. HMGB1, Diabetes Mellitus and Peripheral Arterial Disease
7. HMGB1 and Diabetic Cerebrovascular Disease
8. Diabetic Microangiopathy
9. HMGB1 and Diabetic Nephropathy
10. HMGB1 and Diabetic Neuropathy
11. HMGB1 and Diabetic Retinopathy
12. HMGB1 and Diabetic Cardiomyopathy
13. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2018, 14, 88–98. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. The top 10 Causes of Death (2018). Available online: https://www.who.int/en/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 10 December 2019).
- Wang, Y.; Zhong, J.; Zhang, X.; Liu, Z.; Yang, Y.; Gong, Q.; Ren, B. The Role of HMGB1 in the Pathogenesis of Type 2 Diabetes. J. Diabetes Res. 2016, 2016, 2543268. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pradhan, A.D.; Manson, J.E.; Rifai, N.; Buring, J.E.; Ridker, P.M. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA 2001, 286, 327–334. [Google Scholar] [CrossRef] [PubMed]
- Spranger, J.; Kroke, A.; Möhlig, M.; Hoffmann, K.; Bergmann, M.M.; Ristow, M.; Boeing, H.; Pfeiffer, A.F.H. Inflammatory cytokines and the risk to develop type 2 diabetes: Results of the prospective population-based European Prospective Investigation into Cancer and Nutrition (EPIC)-Potsdam Study. Diabetes 2003, 52, 812–817. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, F.B.; Meigs, J.B.; Li, T.Y.; Rifai, N.; Manson, J.E. Inflammatory markers and risk of developing type 2 diabetes in women. Diabetes 2004, 53, 693–700. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lainampetch, J.; Panprathip, P.; Phosat, C.; Chumpathat, N.; Prangthip, P.; Soonthornworasiri, N.; Puduang, S.; Wechjakwen, N.; Kwanbunjan, K. Association of Tumor Necrosis Factor Alpha, Interleukin 6, and C-Reactive Protein with the Risk of Developing Type 2 Diabetes: A Retrospective Cohort Study of Rural Thais. J. Diabetes Res. 2019, 2019, 9051929. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Feng, X.; Li, Q.; Wang, Y.; Li, Q.; Hua, M. Adiponectin, TNF-α and inflammatory cytokines and risk of type 2 diabetes: A systematic review and meta-analysis. Cytokine 2016, 86, 100–109. [Google Scholar] [CrossRef]
- Wang, X.; Bao, W.; Liu, J.; OuYang, Y.-Y.; Wang, D.; Rong, S.; Xiao, X.; Shan, Z.-L.; Zhang, Y.; Yao, P.; et al. Inflammatory Markers and Risk of Type 2 Diabetes. Diabetes Care 2013, 36, 166–175. [Google Scholar] [CrossRef] [Green Version]
- Hotamisligil, G.S.; Shargill, N.S.; Spiegelman, B.M. Adipose expression of tumor necrosis factor-alpha: Direct role in obesity-linked insulin resistance. Science (New York, N.Y.) 1993, 259, 87–91. [Google Scholar] [CrossRef]
- Hotamisligil, G.S.; Arner, P.; Caro, J.F.; Atkinson, R.L.; Spiegelman, B.M. Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J. Clin. Investig. 1995, 95, 2409–2415. [Google Scholar] [CrossRef]
- Stephens, J.M.; Lee, J.; Pilch, P.F. Tumor necrosis factor-alpha-induced insulin resistance in 3T3-L1 adipocytes is accompanied by a loss of insulin receptor substrate-1 and GLUT4 expression without a loss of insulin receptor-mediated signal transduction. J. Biol. Chem. 1997, 272, 971–976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Massaro, M.; Scoditti, E.; Pellegrino, M.; Carluccio, M.A.; Calabriso, N.; Wabitsch, M.; Storelli, C.; Wright, M.; De Caterina, R. Therapeutic potential of the dual peroxisome proliferator activated receptor (PPAR)α/γ agonist aleglitazar in attenuating TNF-α-mediated inflammation and insulin resistance in human adipocytes. Pharmacol. Res. 2016, 107, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Biscetti, F.; Nardella, E.; Cecchini, A.L.; Landolfi, R.; Flex, A. The Role of the Microbiota in the Diabetic Peripheral Artery Disease. Mediat. Inflamm. 2019, 2019. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nogueira-Machado, J.A.; Volpe, C.M.d.O.; Veloso, C.A.; Chaves, M.M. HMGB1, TLR and RAGE: A functional tripod that leads to diabetic inflammation. Expert Opin. Ther. Targets 2011, 15, 1023–1035. [Google Scholar] [CrossRef]
- Goodwin, G.H.; Sanders, C.; Johns, E.W. A new group of chromatin-associated proteins with a high content of acidic and basic amino acids. Eur. J. Biochem. 1973, 38, 14–19. [Google Scholar] [CrossRef]
- Bustin, M. Regulation of DNA-dependent activities by the functional motifs of the high-mobility-group chromosomal proteins. Mol. Cell. Biol. 1999, 19, 5237–5246. [Google Scholar] [CrossRef] [Green Version]
- Bustin, M. Revised nomenclature for high mobility group (HMG) chromosomal proteins. Trends Biochem. Sci. 2001, 26, 152–153. [Google Scholar] [CrossRef]
- Biscetti, F.; Flex, A.; Alivernini, S.; Tolusso, B.; Gremese, E.; Ferraccioli, G. The Role of High-Mobility Group Box-1 and Its Crosstalk with Microbiome in Rheumatoid Arthritis. Mediat. Inflamm. 2017, 2017, 5230374. [Google Scholar] [CrossRef] [Green Version]
- Biscetti, F.; Ghirlanda, G.; Flex, A. Therapeutic potential of high mobility group box-1 in ischemic injury and tissue regeneration. Curr. Vasc. Pharmacol. 2011, 9, 677–681. [Google Scholar] [CrossRef]
- Tsung, A.; Tohme, S.; Billiar, T.R. High-mobility group box-1 in sterile inflammation. J. Intern. Med. 2014, 276, 425–443. [Google Scholar] [CrossRef]
- Musumeci, D.; Roviello, G.N.; Montesarchio, D. An overview on HMGB1 inhibitors as potential therapeutic agents in HMGB1-related pathologies. Pharmacol. Ther. 2014, 141, 347–357. [Google Scholar] [CrossRef] [PubMed]
- Müller, S.; Ronfani, L.; Bianchi, M.E. Regulated expression and subcellular localization of HMGB1, a chromatin protein with a cytokine function. J. Intern. Med. 2004, 255, 332–343. [Google Scholar] [CrossRef] [Green Version]
- Bianchi, M.E.; Falciola, L.; Ferrari, S.; Lilley, D.M. The DNA binding site of HMG1 protein is composed of two similar segments (HMG boxes), both of which have counterparts in other eukaryotic regulatory proteins. EMBO J. 1992, 11, 1055–1063. [Google Scholar] [CrossRef] [PubMed]
- Messmer, D.; Yang, H.; Telusma, G.; Knoll, F.; Li, J.; Messmer, B.; Tracey, K.J.; Chiorazzi, N. High mobility group box protein 1: An endogenous signal for dendritic cell maturation and Th1 polarization. J. Immunol. 2004, 173, 307–313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malarkey, C.S.; Churchill, M.E.A. The high mobility group box: The ultimate utility player of a cell. Trends Biochem. Sci. 2012, 37, 553–562. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, H.; Ochani, M.; Li, J.; Qiang, X.; Tanovic, M.; Harris, H.E.; Susarla, S.M.; Ulloa, L.; Wang, H.; DiRaimo, R.; et al. Reversing established sepsis with antagonists of endogenous high-mobility group box 1. Proc. Natl. Acad. Sci. USA 2004, 101, 296–301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Yang, H.; Tracey, K.J. Extracellular role of HMGB1 in inflammation and sepsis. J. Intern. Med. 2004, 255, 320–331. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Xie, M.; Yang, M.; Yu, Y.; Zhu, S.; Hou, W.; Kang, R.; Lotze, M.T.; Billiar, T.R.; Wang, H.; et al. PKM2 regulates the Warburg effect and promotes HMGB1 release in sepsis. Nat. Commun. 2014, 5, 4436. [Google Scholar] [CrossRef]
- Kang, R.; Zhang, Q.; Zeh, H.J., 3rd; Lotze, M.T.; Tang, D. HMGB1 in cancer: Good, bad, or both? Clin. Cancer Res. 2013, 19, 4046–4057. [Google Scholar] [CrossRef] [Green Version]
- Vijayakumar, E.C.; Bhatt, L.K.; Prabhavalkar, K.S. High Mobility Group Box-1 (HMGB1): A Potential Target in Therapeutics. Curr. Drug Targets 2019, 20, 1474–1485. [Google Scholar] [CrossRef]
- Wu, H.; Chen, Z.; Xie, J.; Kang, L.-N.; Wang, L.; Xu, B. High Mobility Group Box-1: A Missing Link between Diabetes and Its Complications. Mediat. Inflamm. 2016, 2016, 3896147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rovere-Querini, P.; Capobianco, A.; Scaffidi, P.; Valentinis, B.; Catalanotti, F.; Giazzon, M.; Dumitriu, I.E.; Muller, S.; Iannacone, M.; Traversari, C.L.; et al. HMGB1 is an endogenous immune adjuvant released by necrotic cells. EMBO Rep. 2004, 5, 825–830. [Google Scholar] [CrossRef] [PubMed]
- Lotze, M.T.; Tracey, K.J. High-mobility group box 1 protein (HMGB1): Nuclear weapon in the immune arsenal. Nat. Rev. Immunol. 2005, 5, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Lugrin, J.; Rosenblatt-Velin, N.; Parapanov, R.; Liaudet, L. The role of oxidative stress during inflammatory processes. Biol. Chem. 2014, 395, 203–230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, B.; Antoine, D.J.; Kwan, K.; Lundbäck, P.; Wähämaa, H.; Schierbeck, H.; Robinson, M.; Van Zoelen, M.A.D.; Yang, H.; Li, J.; et al. JAK/STAT1 signaling promotes HMGB1 hyperacetylation and nuclear translocation. Proc. Natl. Acad. Sci. USA 2014, 111, 3068–3073. [Google Scholar] [CrossRef] [Green Version]
- Yan, X.X.; Lu, L.; Peng, W.H.; Wang, L.J.; Zhang, Q.; Zhang, R.Y.; Chen, Q.J.; Shen, W.F. Increased serum HMGB1 level is associated with coronary artery disease in nondiabetic and type 2 diabetic patients. Atherosclerosis 2009, 205, 544–548. [Google Scholar] [CrossRef]
- Wang, H.; Qu, H.; Deng, H. Plasma HMGB-1 Levels in Subjects with Obesity and Type 2 Diabetes: A Cross-Sectional Study in China. PLoS ONE 2015, 10, e0136564. [Google Scholar] [CrossRef] [Green Version]
- Dasu, M.R.; Devaraj, S.; Park, S.; Jialal, I. Increased toll-like receptor (TLR) activation and TLR ligands in recently diagnosed type 2 diabetic subjects. Diabetes Care 2010, 33, 861–868. [Google Scholar] [CrossRef] [Green Version]
- Dandona, P.; Ghanim, H.; Green, K.; Sia, C.L.; Abuaysheh, S.; Kuhadiya, N.; Batra, M.; Dhindsa, S.; Chaudhuri, A. Insulin infusion suppresses while glucose infusion induces Toll-like receptors and high-mobility group-B1 protein expression in mononuclear cells of type 1 diabetes patients. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E810–E818. [Google Scholar] [CrossRef] [Green Version]
- Chen, Q.; Wang, Z.Y.; Chen, L.Y.; Hu, H.Y. Roles of High Mobility Group Box 1 in Cardiovascular Calcification. Cell. Physiol. Biochem. 2017, 42, 427–440. [Google Scholar] [CrossRef] [Green Version]
- Yan, S.F.; Ramasamy, R.; Naka, Y.; Schmidt, A.M. Glycation, inflammation, and RAGE: a scaffold for the macrovascular complications of diabetes and beyond. Circ. Res. 2003, 93, 1159–1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gay, N.J.; Symmons, M.F.; Gangloff, M.; Bryant, C.E. Assembly and localization of Toll-like receptor signalling complexes. Nat. Rev. Immunol. 2014, 14, 546. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Zhong, J.; Yang, P.; Gong, F.; Wang, C.Y. HMGB1, an innate alarmin, in the pathogenesis of type 1 diabetes. Int J. Clin. Exp. Pathol. 2010, 3, 24–38. [Google Scholar]
- Biscetti, F.; Straface, G.; De Cristofaro, R.; Lancellotti, S.; Rizzo, P.; Arena, V.; Stigliano, E.; Pecorini, G.; Egashira, K.; De Angelis, G.; et al. High-mobility group box-1 protein promotes angiogenesis after peripheral ischemia in diabetic mice through a VEGF-dependent mechanism. Diabetes 2010, 59, 1496–1505. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, H.; Li, R.; Wei, Z.H.; Zhang, X.L.; Chen, J.Z.; Dai, Q.; Xie, J.; Xu, B. Diabetes-Induced Oxidative Stress in Endothelial Progenitor Cells May Be Sustained by a Positive Feedback Loop Involving High Mobility Group Box-1. Oxid. Med. Cell. Longev. 2016, 2016, 1943918. [Google Scholar] [CrossRef]
- Tang, D.; Billiar, T.R.; Lotze, M.T. A Janus Tale of Two Active High Mobility Group Box 1 (HMGB1) Redox States. Mol. Med. 2012, 18, 1360–1362. [Google Scholar] [CrossRef]
- Venereau, E.; Casalgrandi, M.; Schiraldi, M.; Antoine, D.J.; Cattaneo, A.; De Marchis, F.; Liu, J.; Antonelli, A.; Preti, A.; Raeli, L.; et al. Mutually exclusive redox forms of HMGB1 promote cell recruitment or proinflammatory cytokine release. J. Exp. Med. 2012, 209, 1519–1528. [Google Scholar] [CrossRef] [Green Version]
- Boström, K.I. Where Do We Stand on Vascular Calcification? Vascul. Pharmacol. 2016, 84, 8–14. [Google Scholar] [CrossRef] [Green Version]
- Ruiz-Ortega, M.; Rodriguez-Vita, J.; Sanchez-Lopez, E.; Carvajal, G.; Egido, J. TGF-beta signaling in vascular fibrosis. Cardiovasc. Res. 2007, 74, 196–206. [Google Scholar] [CrossRef] [Green Version]
- Pardali, E.; ten Dijke, P. TGFβ Signaling and Cardiovascular Diseases. Int. J. Biol. Sci. 2012, 8, 195–213. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014; U.S. Department of Health and Human Services: Atlanta, GA, USA, 2014.
- Loomans, C.J.M.; de Koning, E.J.P.; Staal, F.J.T.; Rookmaaker, M.B.; Verseyden, C.; de Boer, H.C.; Verhaar, M.C.; Braam, B.; Rabelink, T.J.; van Zonneveld, A.-J. Endothelial progenitor cell dysfunction: A novel concept in the pathogenesis of vascular complications of type 1 diabetes. Diabetes 2004, 53, 195–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Madonna, R.; Pieragostino, D.; Balistreri, C.R.; Rossi, C.; Geng, Y.-J.; Del Boccio, P.; De Caterina, R. Diabetic macroangiopathy: Pathogenetic insights and novel therapeutic approaches with focus on high glucose-mediated vascular damage. Vasc. Pharmacol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, Z.A.; Armour, C.L.; Phipps, S.; Sukkar, M.B. RAGE and TLRs: Relatives, friends or neighbours? Mol. Immunol. 2013, 56, 739–744. [Google Scholar] [CrossRef] [PubMed]
- Cai, S.; Khoo, J.; Mussa, S.; Alp, N.J.; Channon, K.M. Endothelial nitric oxide synthase dysfunction in diabetic mice: Importance of tetrahydrobiopterin in eNOS dimerisation. Diabetologia 2005, 48, 1933–1940. [Google Scholar] [CrossRef] [Green Version]
- Giovannini, S.; Tinelli, G.; Biscetti, F.; Straface, G.; Angelini, F.; Pitocco, D.; Mucci, L.; Landolfi, R.; Flex, A. Serum high mobility group box-1 and osteoprotegerin levels are associated with peripheral arterial disease and critical limb ischemia in type 2 diabetic subjects. Cardiovasc. Diabetol. 2017, 16, 99. [Google Scholar] [CrossRef] [Green Version]
- Straface, G.; Biscetti, F.; Pitocco, D.; Bertoletti, G.; Misuraca, M.; Vincenzoni, C.; Snider, F.; Arena, V.; Stigliano, E.; Angelini, F.; et al. Assessment of the genetic effects of polymorphisms in the osteoprotegerin gene, TNFRSF11B, on serum osteoprotegerin levels and carotid plaque vulnerability. Stroke 2011, 42, 3022–3028. [Google Scholar] [CrossRef]
- Biscetti, F.; Porreca, C.F.; Bertucci, F.; Straface, G.; Santoliquido, A.; Tondi, P.; Angelini, F.; Pitocco, D.; Santoro, L.; Gasbarrini, A.; et al. TNFRSF11B gene polymorphisms increased risk of peripheral arterial occlusive disease and critical limb ischemia in patients with type 2 diabetes. Acta Diabetol. 2014, 51, 1025–1032. [Google Scholar] [CrossRef]
- Yang, S.-L.; Zhu, L.-Y.; Han, R.; Sun, L.-L.; Li, J.-X.; Dou, J.-T. Pathophysiology of peripheral arterial disease in diabetes mellitus. J. Diabetes 2017, 9, 133–140. [Google Scholar] [CrossRef]
- Biscetti, F.; Straface, G.; Pitocco, D.; Zaccardi, F.; Ghirlanda, G.; Flex, A. Peroxisome proliferator-activated receptors and angiogenesis. Nutr. Metab. Cardiovasc. Dis. NMCD 2009, 19, 751–759. [Google Scholar] [CrossRef]
- Sawada, T.; Uzu, K.; Hashimoto, N.; Onishi, T.; Takaya, T.; Shimane, A.; Taniguchi, Y.; Yasaka, Y.; Ohara, T.; Kawai, H. Empagliflozin’s Ameliorating Effect on Plasma Triglycerides: Association with Endothelial Function Recovery in Diabetic Patients with Coronary Artery Disease. J. Atheroscler. Thromb. 2019. [Google Scholar] [CrossRef] [Green Version]
- James, S.L.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1789–1858. [Google Scholar] [CrossRef] [Green Version]
- Andrassy, M.; Volz, H.C.; Maack, B.; Schuessler, A.; Gitsioudis, G.; Hofmann, N.; Laohachewin, D.; Wienbrandt, A.R.; Kaya, Z.; Bierhaus, A.; et al. HMGB1 is associated with atherosclerotic plaque composition and burden in patients with stable coronary artery disease. PLoS ONE 2012, 7, e52081. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Jiang, H.; Bai, Q.; Zhou, X.; Xu, C.; Lu, Z.; Cui, B.; Wen, H. Increased serum HMGB1 is related to the severity of coronary artery stenosis. Clin. Chim. Acta 2009, 406, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Benlier, N.; Erdoğan, M.B.; Keçioğlu, S.; Orhan, N.; Çiçek, H. Association of high mobility group box 1 protein with coronary artery disease. Asian Cardiovasc. Thorac. Ann. 2019, 27, 251–255. [Google Scholar] [CrossRef]
- Kalinina, N.; Agrotis, A.; Antropova, Y.; DiVitto, G.; Kanellakis, P.; Kostolias, G.; Ilyinskaya, O.; Tararak, E.; Bobik, A. Increased expression of the DNA-binding cytokine HMGB1 in human atherosclerotic lesions: Role of activated macrophages and cytokines. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 2320–2325. [Google Scholar] [CrossRef] [Green Version]
- Dozio, E.; Vianello, E.; Briganti, S.; Lamont, J.; Tacchini, L.; Schmitz, G.; Corsi Romanelli, M.M. Expression of the Receptor for Advanced Glycation End Products in Epicardial Fat: Link with Tissue Thickness and Local Insulin Resistance in Coronary Artery Disease. J. Diabetes Res. 2016, 2016, 2327341. [Google Scholar] [CrossRef]
- Kohno, T.; Anzai, T.; Naito, K.; Miyasho, T.; Okamoto, M.; Yokota, H.; Yamada, S.; Maekawa, Y.; Takahashi, T.; Yoshikawa, T.; et al. Role of high-mobility group box 1 protein in post-infarction healing process and left ventricular remodelling. Cardiovasc. Res. 2009, 81, 565–573. [Google Scholar] [CrossRef] [Green Version]
- Sørensen, M.V.; Pedersen, S.; Møgelvang, R.; Skov-Jensen, J.; Flyvbjerg, A. Plasma high-mobility group box 1 levels predict mortality after ST-segment elevation myocardial infarction. JACC Cardiovasc. Interv. 2011, 4, 281–286. [Google Scholar] [CrossRef] [Green Version]
- Andrassy, M.; Volz, H.C.; Riedle, N.; Gitsioudis, G.; Seidel, C.; Laohachewin, D.; Zankl, A.R.; Kaya, Z.; Bierhaus, A.; Giannitsis, E.; et al. HMGB1 as a predictor of infarct transmurality and functional recovery in patients with myocardial infarction. J. Intern. Med. 2011, 270, 245–253. [Google Scholar] [CrossRef]
- Straino, S.; Di Carlo, A.; Mangoni, A.; De Mori, R.; Guerra, L.; Maurelli, R.; Panacchia, L.; Di Giacomo, F.; Palumbo, R.; Di Campli, C.; et al. High-mobility group box 1 protein in human and murine skin: Involvement in wound healing. J. Invest. Dermatol. 2008, 128, 1545–1553. [Google Scholar] [CrossRef]
- Kitahara, T.; Takeishi, Y.; Harada, M.; Niizeki, T.; Suzuki, S.; Sasaki, T.; Ishino, M.; Bilim, O.; Nakajima, O.; Kubota, I. High-mobility group box 1 restores cardiac function after myocardial infarction in transgenic mice. Cardiovasc. Res. 2008, 80, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Andrassy, M.; Volz, H.C.; Igwe, J.C.; Funke, B.; Eichberger, S.N.; Kaya, Z.; Buss, S.; Autschbach, F.; Pleger, S.T.; Lukic, I.K.; et al. High-mobility group box-1 in ischemia-reperfusion injury of the heart. Circulation 2008, 117, 3216–3226. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santilli, F.; Blardi, P.; Scapellato, C.; Bocchia, M.; Guazzi, G.; Terzuoli, L.; Tabucchi, A.; Silvietti, A.; Lucani, B.; Gioffre, W.R.; et al. Decreased plasma endogenous soluble RAGE, and enhanced adipokine secretion, oxidative stress and platelet/coagulative activation identify non-alcoholic fatty liver disease among patients with familial combined hyperlipidemia and/or metabolic syndrome. Vasc. Pharmacol. 2015, 72, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Jin, D.; Wang, H. Serum glycated albumin is superior to hemoglobin A1c for correlating with HMGB1 in coronary artery disease with type 2 diabetic mellitus patients. Int J. Clin. Exp. Med. 2015, 8, 4821–4825. [Google Scholar]
- Shen, Y.; Pu, L.J.; Lu, L.; Zhang, Q.; Zhang, R.Y.; Shen, W.F. Glycated albumin is superior to hemoglobin A1c for evaluating the presence and severity of coronary artery disease in type 2 diabetic patients. Cardiology 2012, 123, 84–90. [Google Scholar] [CrossRef]
- Yamashita, A.; Nishihira, K.; Matsuura, Y.; Ito, T.; Kawahara, K.; Hatakeyama, K.; Hashiguchi, T.; Maruyama, I.; Yagi, H.; Matsumoto, M.; et al. Paucity of CD34-positive cells and increased expression of high-mobility group box 1 in coronary thrombus with type 2 diabetes mellitus. Atherosclerosis 2012, 224, 511–514. [Google Scholar] [CrossRef]
- Flex, A.; Gaetani, E.; Angelini, F.; Sabusco, A.; Chillà, C.; Straface, G.; Biscetti, F.; Pola, P.; Castellot, J.J.; Pola, R. Pro-inflammatory genetic profiles in subjects with peripheral arterial occlusive disease and critical limb ischemia. J. Intern. Med. 2007, 262, 124–130. [Google Scholar] [CrossRef]
- Biscetti, F.; Bonadia, N.; Nardella, E.; Cecchini, A.L.; Landolfi, R.; Flex, A. The Role of the Stem Cells Therapy in the Peripheral Artery Disease. Int. J. Mol. Sci. 2019, 20, 2233. [Google Scholar] [CrossRef] [Green Version]
- Fowkes, F.G.; Rudan, D.; Rudan, I.; Aboyans, V.; Denenberg, J.O.; McDermott, M.M.; Norman, P.E.; Sampson, U.K.; Williams, L.J.; Mensah, G.A.; et al. Comparison of global estimates of prevalence and risk factors for peripheral artery disease in 2000 and 2010: A systematic review and analysis. Lancet 2013, 382, 1329–1340. [Google Scholar] [CrossRef]
- Yang, S.L.; Kim, Y.S. Peripheral Arterial Disease in Patients with Type 2 Diabetes Mellitus. Diabetes Metab. J. 2015, 39, 283–290. [Google Scholar] [CrossRef]
- Biscetti, F.; Ferraro, P.M.; Hiatt, W.R.; Angelini, F.; Nardella, E.; Cecchini, A.L.; Santoliquido, A.; Pitocco, D.; Landolfi, R.; Flex, A. Inflammatory Cytokines Associated With Failure of Lower-Extremity Endovascular Revascularization (LER): A Prospective Study of a Population With Diabetes. Diabetes Care 2019, 42, 1939–1945. [Google Scholar] [CrossRef] [PubMed]
- Biscetti, F.; Straface, G.; Pitocco, D.; Angelini, F.; Tinelli, G.; Landolfi, R.; Flex, A. Fibroblast growth factor 23 serum level in type 2 diabetic italian subjects with peripheral arterial disease and critical limb ischemia. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4048–4054. [Google Scholar] [PubMed]
- Xu, J.; Cui, X.; Li, J.; Koutakis, P.; Pipinos, I.; Tzeng, E.; Chen, A.; Sachdev, U. Chloroquine improves the response to ischemic muscle injury and increases HMGB1 after arterial ligation. J. Vasc. Surg. 2018, 67, 910–921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Y.; Zhou, C.; Jiang, J.; Su, Q.; Ding, X. Blockade of HMGB1 preserves vascular homeostasis and improves blood perfusion in rats of acute limb ischemia/reperfusion. Microvasc. Res. 2017, 112, 37–40. [Google Scholar] [CrossRef]
- Tsao, C.F.; Huang, W.T.; Liu, T.T.; Wang, P.W.; Liou, C.W.; Lin, T.K.; Hsieh, C.J.; Weng, S.W. Expression of high-mobility group box protein 1 in diabetic foot atherogenesis. Genet. Mol. Res. GMR 2015, 14, 4521–4531. [Google Scholar] [CrossRef]
- Hafez, Y.M.; El-Deeb, O.S.; Atef, M.M. The emerging role of the epigenetic enzyme Sirtuin-1 and high mobility group Box 1 in patients with diabetic foot ulceration. Diabetes Metab. Syndr. 2018, 12, 1065–1070. [Google Scholar] [CrossRef]
- Biscetti, F.; Gentileschi, S.; Bertucci, F.; Servillo, M.; Arena, V.; Angelini, F.; Stigliano, E.; Bonanno, G.; Scambia, G.; Sacchetti, B.; et al. The angiogenic properties of human adipose-derived stem cells (HASCs) are modulated by the High mobility group box protein 1 (HMGB1). Int. J. Cardiol. 2017, 249, 349–356. [Google Scholar] [CrossRef]
- Chen, R.; Ovbiagele, B.; Feng, W. Diabetes and Stroke: Epidemiology, Pathophysiology, Pharmaceuticals and Outcomes. Am. J. Med. Sci. 2016, 351, 380–386. [Google Scholar] [CrossRef] [Green Version]
- Biscetti, F.; Giovannini, S.; Straface, G.; Bertucci, F.; Angelini, F.; Porreca, C.; Landolfi, R.; Flex, A. RANK/RANKL/OPG pathway: Genetic association with history of ischemic stroke in Italian population. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4574–4580. [Google Scholar]
- Hu, J.; Liu, B.; Zhao, Q.; Jin, P.; Hua, F.; Zhang, Z.; Liu, Y.; Zan, K.; Cui, G.; Ye, X. Bone marrow stromal cells inhibits HMGB1-mediated inflammation after stroke in type 2 diabetic rats. Neuroscience 2016, 324, 11–19. [Google Scholar] [CrossRef]
- Qiu, J.; Nishimura, M.; Wang, Y.; Sims, J.R.; Qiu, S.; Savitz, S.I.; Salomone, S.; Moskowitz, M.A. Early release of HMGB-1 from neurons after the onset of brain ischemia. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2008, 28, 927–938. [Google Scholar] [CrossRef] [PubMed]
- Brea, D.; Blanco, M.; Ramos-Cabrer, P.; Moldes, O.; Arias, S.; Pérez-Mato, M.; Leira, R.; Sobrino, T.; Castillo, J. Toll-like receptors 2 and 4 in ischemic stroke: Outcome and therapeutic values. J. Cereb. Blood Flow Metab. Off. J. Int. Soc. Cereb. Blood Flow Metab. 2011, 31, 1424–1431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, X.; Chopp, M.; Liu, X.; Zacharek, A.; Cui, X.; Yan, T.; Roberts, C.; Chen, J. Niaspan reduces high-mobility group box 1/receptor for advanced glycation endproducts after stroke in type-1 diabetic rats. Neuroscience 2011, 190, 339–345. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yan, T.; Chopp, M.; Ye, X.; Liu, Z.; Zacharek, A.; Cui, Y.; Roberts, C.; Buller, B.; Chen, J. Niaspan increases axonal remodeling after stroke in type 1 diabetes rats. Neurobiol. Dis. 2012, 46, 157–164. [Google Scholar] [CrossRef] [Green Version]
- Chen, J.; Ye, X.; Yan, T.; Zhang, C.; Yang, X.-P.; Cui, X.; Cui, Y.; Zacharek, A.; Roberts, C.; Liu, X.; et al. Adverse effects of bone marrow stromal cell treatment of stroke in diabetic rats. Stroke 2011, 42, 3551–3558. [Google Scholar] [CrossRef] [Green Version]
- Madonna, R.; Balistreri, C.R.; Geng, Y.-J.; De Caterina, R. Diabetic microangiopathy: Pathogenetic insights and novel therapeutic approaches. Vasc. Pharmacol. 2017, 90, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Umanath, K.; Lewis, J.B. Update on Diabetic Nephropathy: Core Curriculum 2018. Am. J. Kidney Dis. Off. J. Natl. Kidney Found. 2018, 71, 884–895. [Google Scholar] [CrossRef]
- Devaraj, S.; Tobias, P.; Kasinath Balakuntalam, S.; Ramsamooj, R.; Afify, A.; Jialal, I. Knockout of Toll-Like Receptor-2 Attenuates Both the Proinflammatory State of Diabetes and Incipient Diabetic Nephropathy. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 1796–1804. [Google Scholar] [CrossRef] [Green Version]
- Hu, C.; Sun, L.; Xiao, L.; Han, Y.; Fu, X.; Xiong, X.; Xu, X.; Liu, Y.; Yang, S.; Liu, F.; et al. Insights into the Mechanisms Involved in the Expression and Regulation of Extracellular Matrix Proteins in Diabetic Nephropathy. Curr. Med. Chem. 2015, 22, 2858–2870. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.; Sohn, E.; Kim, C.-S.; Jo, K.; Kim, J.S. The role of high-mobility group box-1 protein in the development of diabetic nephropathy. Am. J. Nephrol. 2011, 33, 524–529. [Google Scholar] [CrossRef]
- Lin, M.; Yiu, W.H.; Wu, H.J.; Chan, L.Y.Y.; Leung, J.C.K.; Au, W.S.; Chan, K.W.; Lai, K.N.; Tang, S.C.W. Toll-like receptor 4 promotes tubular inflammation in diabetic nephropathy. J. Am. Soc. Nephrol. JASN 2012, 23, 86–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mudaliar, H.; Pollock, C.; Komala, M.G.; Chadban, S.; Wu, H.; Panchapakesan, U. The role of Toll-like receptor proteins (TLR) 2 and 4 in mediating inflammation in proximal tubules. Am. J. Physiol. Renal Physiol. 2013, 305, F143–F154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, J.; Chadban, S.J.; Zhao, C.Y.; Chen, X.; Kwan, T.; Panchapakesan, U.; Pollock, C.A.; Wu, H. TLR4 activation promotes podocyte injury and interstitial fibrosis in diabetic nephropathy. PLoS ONE 2014, 9, e97985. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, J.; Wu, H.; Zhao, C.Y.; Panchapakesan, U.; Pollock, C.; Chadban, S.J. Requirement for TLR2 in the development of albuminuria, inflammation and fibrosis in experimental diabetic nephropathy. Int. J. Clin. Exp. Pathol. 2014, 7, 481–495. [Google Scholar]
- Chen, X.; Ma, J.; Kwan, T.; Stribos, E.G.D.; Messchendorp, A.L.; Loh, Y.W.; Wang, X.; Paul, M.; Cunningham, E.C.; Habib, M.; et al. Blockade of HMGB1 Attenuates Diabetic Nephropathy in Mice. Sci. Rep. 2018, 8, 8319. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, R.; Chen, J.; Shi, M.; Li, W.; Zhang, X. High Mobility Group Box1 Inhibitor Glycyrrhizic Acid Attenuates Kidney Injury in Streptozotocin-Induced Diabetic Rats. Kidney Blood Press. Res. 2017, 42, 894–904. [Google Scholar] [CrossRef] [Green Version]
- Ashrafi Jigheh, Z.; Ghorbani Haghjo, A.; Argani, H.; Roshangar, L.; Rashtchizadeh, N.; Sanajou, D.; Nazari Soltan Ahmad, S.; Rashedi, J.; Dastmalchi, S.; Mesgari Abbasi, M. Empagliflozin alleviates renal inflammation and oxidative stress in streptozotocin-induced diabetic rats partly by repressing HMGB1-TLR4 receptor axis. Iran. J. Basic Med. Sci. 2019, 22, 384–390. [Google Scholar] [CrossRef]
- Dewanjee, S.; Das, S.; Das, A.K.; Bhattacharjee, N.; Dihingia, A.; Dua, T.K.; Kalita, J.; Manna, P. Molecular mechanism of diabetic neuropathy and its pharmacotherapeutic targets. Eur. J. Pharmacol. 2018, 833, 472–523. [Google Scholar] [CrossRef]
- Tracy, J.A.; Dyck, P.J.B. The Spectrum of Diabetic Neuropathies. Phys. Med. Rehabil. Clin. N. Am. 2008, 19, 1–26. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Feng, C.; Qiao, Y.; Zhao, X. Sigma 1 receptor mediated HMGB1 expression in spinal cord is involved in the development of diabetic neuropathic pain. Neurosci. Lett. 2018, 668, 164–168. [Google Scholar] [CrossRef]
- Zhao, H.; Zhang, J.; Yu, J. HMGB-1 as a Potential Target for the Treatment of Diabetic Retinopathy. Med. Sci. Monit. 2015, 21, 3062–3067. [Google Scholar] [CrossRef]
- Zhao, X.; Shen, L.; Xu, L.; Wang, Z.; Ma, C.; Huang, Y. Inhibition of CaMKIV relieves streptozotocin-induced diabetic neuropathic pain through regulation of HMGB1. BMC Anesthesiol. 2016, 16, 27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meng, W.; Deshmukh, H.A.; Donnelly, L.A.; Wellcome Trust Case Control Consortium 2 (WTCCC2); Surrogate markers for Micro- and Macro-vascular hard endpoints for Innovative diabetes Tools (SUMMIT) study group; Torrance, N.; Colhoun, H.M.; Palmer, C.N.A.; Smith, B.H. A Genome-wide Association Study Provides Evidence of Sex-specific Involvement of Chr1p35.1 (ZSCAN20-TLR12P) and Chr8p23.1 (HMGB1P46) With Diabetic Neuropathic Pain. EBioMedicine 2015, 2, 1386–1393. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abu El-Asrar, A.M.; Siddiquei, M.M.; Nawaz, M.I.; Geboes, K.; Mohammad, G. The proinflammatory cytokine high-mobility group box-1 mediates retinal neuropathy induced by diabetes. Mediat. Inflamm. 2014, 2014, 746415. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-B.; Sig Choi, J.; Yu, Y.-M.; Nam, K.; Piao, C.-S.; Kim, S.-W.; Lee, M.-H.; Han, P.-L.; Park, J.-S.; Lee, J.-K. HMGB1, a novel cytokine-like mediator linking acute neuronal death and delayed neuroinflammation in the postischemic brain. J. Neurosci. Off. J. Soc. Neurosci. 2006, 26, 6413–6421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shibasaki, M.; Sasaki, M.; Miura, M.; Mizukoshi, K.; Ueno, H.; Hashimoto, S.; Tanaka, Y.; Amaya, F. Induction of high mobility group box-1 in dorsal root ganglion contributes to pain hypersensitivity after peripheral nerve injury. Pain 2010, 149, 514–521. [Google Scholar] [CrossRef]
- Andersson, A.; Covacu, R.; Sunnemark, D.; Danilov, A.I.; Dal Bianco, A.; Khademi, M.; Wallström, E.; Lobell, A.; Brundin, L.; Lassmann, H.; et al. Pivotal advance: HMGB1 expression in active lesions of human and experimental multiple sclerosis. J. Leukoc. Biol. 2008, 84, 1248–1255. [Google Scholar] [CrossRef] [Green Version]
- Antonetti, D.A.; Klein, R.; Gardner, T.W. Diabetic retinopathy. N. Engl. J. Med. 2012, 366, 1227–1239. [Google Scholar] [CrossRef] [Green Version]
- Heng, L.Z.; Comyn, O.; Peto, T.; Tadros, C.; Ng, E.; Sivaprasad, S.; Hykin, P.G. Diabetic retinopathy: Pathogenesis, clinical grading, management and future developments. Diabet. Med. J. Br. Diabet. Assoc. 2013, 30, 640–650. [Google Scholar] [CrossRef]
- Kim, J.; Kim, C.-S.; Sohn, E.; Kim, J.S. Cytoplasmic translocation of high-mobility group box-1 protein is induced by diabetes and high glucose in retinal pericytes. Mol. Med. Rep. 2016, 14, 3655–3661. [Google Scholar] [CrossRef] [Green Version]
- Mohammad, G.; Siddiquei, M.M.; Othman, A.; Al-Shabrawey, M.; Abu El-Asrar, A.M. High-mobility group box-1 protein activates inflammatory signaling pathway components and disrupts retinal vascular-barrier in the diabetic retina. Exp. Eye Res. 2013, 107, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Chen, X. Expression of High-Mobility Group Box 1 Protein (HMGB1) and Toll-Like Receptor 9 (TLR9) in Retinas of Diabetic Rats. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2017, 23, 3115–3122. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abu El-Asrar, A.M.; Nawaz, M.I.; Kangave, D.; Abouammoh, M.; Mohammad, G. High-mobility group box-1 and endothelial cell angiogenic markers in the vitreous from patients with proliferative diabetic retinopathy. Mediat. Inflamm. 2012, 2012, 697489. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Asrar, A.M.A.; Nawaz, M.I.; Kangave, D.; Geboes, K.; Ola, M.S.; Ahmad, S.; Al-Shabrawey, M. High-mobility group box-1 and biomarkers of inflammation in the vitreous from patients with proliferative diabetic retinopathy. Mol. Vis. 2011, 17, 1829–1838. [Google Scholar] [PubMed]
- Gong, Y.; Jin, X.; Wang, Q.-S.; Wei, S.-H.; Hou, B.-K.; Li, H.-Y.; Zhang, M.-N.; Li, Z.-H. The involvement of high mobility group 1 cytokine and phospholipases A2 in diabetic retinopathy. Lipids Health Dis. 2014, 13, 156. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, A.R.C.; Dvoriantchikova, G.; Li, Y.; Mohammad, G.; Abu El-Asrar, A.M.; Wen, R.; Ivanov, D. Cellular mechanisms of high mobility group 1 (HMGB-1) protein action in the diabetic retinopathy. PLoS ONE 2014, 9, e87574. [Google Scholar] [CrossRef] [PubMed]
- Fu, D.; Tian, X. Effect of high mobility group box 1 on the human retinal pigment epithelial cell in high-glucose condition. Int. J. Clin. Exp. Med. 2015, 8, 17796–17803. [Google Scholar]
- Chang, Y.-C.; Lin, C.-W.; Hsieh, M.-C.; Wu, H.-J.; Wu, W.-S.; Wu, W.-C.; Kao, Y.-H. High mobility group B1 up-regulates angiogenic and fibrogenic factors in human retinal pigment epithelial ARPE-19 cells. Cell. Signal. 2017, 40, 248–257. [Google Scholar] [CrossRef]
- Jiang, Y.; Steinle, J.J. HMGB1 inhibits insulin signalling through TLR4 and RAGE in human retinal endothelial cells. Growth Factors (Chur, Switz.) 2018, 36, 164–171. [Google Scholar] [CrossRef]
- Yu, Y.; Yang, L.; Lv, J.; Huang, X.; Yi, J.; Pei, C.; Shao, Y. The role of high mobility group box 1 (HMGB-1) in the diabetic retinopathy inflammation and apoptosis. Int. J. Clin. Exp. Pathol. 2015, 8, 6807–6813. [Google Scholar]
- Sohn, E.; Kim, J.; Kim, C.-S.; Lee, Y.M.; Kim, J.S. Extract of Polygonum cuspidatum Attenuates Diabetic Retinopathy by Inhibiting the High-Mobility Group Box-1 (HMGB1) Signaling Pathway in Streptozotocin-Induced Diabetic Rats. Nutrients 2016, 8, 140. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, G.; Alam, K.; Nawaz, M.I.; Siddiquei, M.M.; Mousa, A.; Abu El-Asrar, A.M. Mutual enhancement between high-mobility group box-1 and NADPH oxidase-derived reactive oxygen species mediates diabetes-induced upregulation of retinal apoptotic markers. J. Physiol. Biochem. 2015, 71, 359–372. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Chen, X. HMGB1 siRNA can reduce damage to retinal cells induced by high glucose in vitro and in vivo. Drug Des. Dev. Ther. 2017, 11, 783–795. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, W.; Wang, Y.; Kong, Y. Exosomes Derived From Mesenchymal Stem Cells Modulate miR-126 to Ameliorate Hyperglycemia-Induced Retinal Inflammation Via Targeting HMGB1. Investig. Ophthalmol. Vis. Sci. 2019, 60, 294–303. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Patel, P.; Steinle, J.J. PKA regulates HMGB1 through activation of IGFBP-3 and SIRT1 in human retinal endothelial cells cultured in high glucose. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. 2018, 67, 1013–1019. [Google Scholar] [CrossRef]
- Raucci, A.; Di Maggio, S.; Scavello, F.; D’Ambrosio, A.; Bianchi, M.E.; Capogrossi, M.C. The Janus face of HMGB1 in heart disease: A necessary update. Cell. Mol. Life Sci. 2019, 76, 211–229. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.-K.; Wang, B.; Lu, Q.-H.; Zhang, W.; Qin, W.-D.; Liu, X.-J.; Liu, X.-Q.; An, F.-S.; Zhang, Y.; Zhang, M.-X. Inhibition of high-mobility group box 1 improves myocardial fibrosis and dysfunction in diabetic cardiomyopathy. Int. J. Cardiol. 2014, 172, 202–212. [Google Scholar] [CrossRef]
- Volz, H.C.; Seidel, C.; Laohachewin, D.; Kaya, Z.; Muller, O.J.; Pleger, S.T.; Lasitschka, F.; Bianchi, M.E.; Remppis, A.; Bierhaus, A.; et al. HMGB1: The missing link between diabetes mellitus and heart failure. Basic Res. Cardiol. 2010, 105, 805–820. [Google Scholar] [CrossRef]
- Wang, W.K.; Lu, Q.H.; Zhang, J.N.; Wang, B.; Liu, X.J.; An, F.S.; Qin, W.D.; Chen, X.Y.; Dong, W.Q.; Zhang, C.; et al. HMGB1 mediates hyperglycaemia-induced cardiomyocyte apoptosis via ERK/Ets-1 signalling pathway. J. Cell. Mol. Med. 2014, 18, 2311–2320. [Google Scholar] [CrossRef]
- Tao, A.; Song, J.; Lan, T.; Xu, X.; Kvietys, P.; Kao, R.; Martin, C.; Rui, T. Cardiomyocyte-fibroblast interaction contributes to diabetic cardiomyopathy in mice: Role of HMGB1/TLR4/IL-33 axis. Biochim. Biophys. Acta 2015, 1852, 2075–2085. [Google Scholar] [CrossRef] [Green Version]
- Song, J.; Liu, Q.; Tang, H.; Tao, A.; Wang, H.; Kao, R.; Rui, T. Activation of PI3Kgamma/Akt pathway increases cardiomyocyte HMGB1 expression in diabetic environment. Oncotarget 2016, 7, 80803–80810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, H.; Morgan, B.; Potter, B.J.; Ma, L.; Dellsperger, K.C.; Ungvari, Z.; Zhang, C. Resveratrol improves left ventricular diastolic relaxation in type 2 diabetes by inhibiting oxidative/nitrative stress: In vivo demonstration with magnetic resonance imaging. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H985–H994. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Ref. | Year | Aim | HMGB1 |
|---|---|---|---|
| Yan et al. | 2009 | Role of HMGB1 in T2DM patients with CAD e in non-T2DM patients with CAD. | Increased serum levels of HMGB1 and hsCRP in non-T2DM patients with CAD and in T2DM patients with CAD. HMGB1 levels correlate with hsCRP, TNF-α, IL-6 levels. |
| Yamashita et al. | 2012 | Presence of CD34-positive cells and HMGB1 in acute coronary thrombi in T2DM patients vs non-T2DM patients. | Extracellular HMGB1 area in the thrombi larger in T2DM patients. HMGB1 facilitates thrombus formation. |
| Yin et al. | 2015 | Relationship between blood glucose levels and HMGB1 levels T2DM patients with CAD. | Increased serum levels of HMGB1 in T2DM patients with CAD. Levels of HMGB1 correlate with glucose levels and HbA1c levels. |
| Ref. | Year | Aim | HMGB1 |
|---|---|---|---|
| Biscetti et al. | 2010 | Role of HMGB1 in diabetic angiogenesis. | Lower HMGB1 protein expression in the ischemic tissue of STZ-induced mice. HMGB1 administration ameliorates the blood flow recovery and capillary density in the ischemic muscle of STZ-induced mice. Reduced HMGB1-induced angiogenesis by inhibiting VEGF activity. |
| Tsao et al. | 2015 | Expression of HMGB1 in diabetic foot atherogenesis. | Increased HMGB1 expression in vessels of T2DM patients and T2DM patients with PAD compared to non-T2DM patients. |
| Giovannini et al. | 2017 | Role of HMGB1, OPG, TNF-α, IL-6, CRP in diabetic patients with PAD. | Increased serum levels of HMGB1 in T2DM patients with PAD. Levels of HMGB1 positively correlates with severity of PAD in T2DM patients. HMGB1 independent risk factor for PAD in T2DM patients. |
| Biscetti et al. | 2017 | Role of HMGB1 in cell therapy with HASCs in PAD. | Improved blood flow recovery in mice co-treated with HASCs and HMGB1 protein, compared to HASCs-treated mice. Reduced post-ischemic angiogenesis with HMGB1 inhibition in mice co-treated with HASCs and HMGB1. |
| Hafez et al. | 2018 | Role of Sirtuin-1 and HMGB1 in DFU. | Increased serum levels HMGB1 and AGEs in T2DM patients compared to non-T2DM patients with the highest levels in T2DM patients with DFU. |
| Ref. | Year | Aim | HMGB1 |
|---|---|---|---|
| Ye et al. | 2011 | Role of HMGB1 and RAGE in stroke in T1DM rats and role of niaspan on pro-inflammatory proteins expression. | Increased HMGB1 expression after stroke in brains of STZ-induced MCAo rats. Niaspan treatment in STZ-induced MCAo rats decreased HMGB1 expression. |
| Hu et al. | 2016 | Role of HMGB1 in stroke in T2DM rats and role of BMSCs in HMGB1 inflammation. | MCAo STZ/N-induced rats: increased HMGB1 and RAGE expression, increased BBB leakage, decreased functional outcome after stroke. Injection of BMSCs in STZ/N-induced rats: decreased HMGB1 and RAGE expression, attenuated BBB leakage and improved functional outcome after stroke, decreased inflammation after stroke. |
| Ref. | Year | Aim | HMGB1 |
|---|---|---|---|
| Kim et al. | 2011 | Role of HMGB1, RAGE and NF-kB in DN. | Increased HMGB1 levels in cytoplasmic and nuclear patterns of glomerular cells of STZ-induced rats vs non STZ-induced rats (nuclear HMGB1 only). |
| Lin et al. | 2012 | Role of TLR-4 in DN. | Increased HMGB1 expression in renal biopsies of T2DM patients with DN. |
| Mudaliar et al. | 2013 | Role of TLR-2 and -4 in DN. | Upregulation of HMGB1 and TLR-2 levels in tubules cells of STZ-induced mice compared to non STZ induced mice. Increased HMGB1 secretion and NF-kB activation in response to high level of glucose. Reduced HMGB1 secretion and NF-kB activation with TLR-2 siRNA and TLR-4 siRNA. |
| Ma et al. | 2014 | Role of TLR-4 in DN. | Upregulation of HMGB1 in STZ-induced WT mice and STZ-induced TLR-4 deficient mice compared to non STZ-induced mice. |
| Ma et al. | 2014 | Role of TLR-2 in DN. | Upregulation of HMGB1 in STZ-induced WT mice compared to non STZ-induced WT mice. |
| Chen et al. | 2015 | Role of high glucose on HMGB1 expression in T2DM patients and in mesangial cells. | Increased serum HMGB1 levels in T2DM patients and in mesangial cells stimulated with high glucose. Knockdown of HMGB1 in mesangial cells reduces HMGB1 mRNA levels. |
| Zhang et al. | 2017 | Role of HMGB1 inhibitor GA in DN. | Increased HMGB1 expression in kidney tissue of STZ-induced rats and lower HMGB1 expression in kidney tissue of STZ-induced rats treated with GA. |
| Jigheh et al. | 2018 | Empaglifozin role in reduction of HMGB1 and TLR-4 levels in DN. | Empaglifozin reduces renal levels of HMGB1 in STZ-induced rats. |
| Ref. | Year | Aim | HMGB1 |
|---|---|---|---|
| El Asrar et al. | 2014 | Role of HMGB1 in retinal neuropathy. | Increased HMGB1 retinal levels in STZ-induced rats. Vitreal injection of HMGB1in non STZ-induced rats increased HMGB1 mRNA levels. GA intake in STZ-induced rats decreased HMGB1 mRNA levels. |
| Meng et al. | 2015 | GWAS to clarify the role of sex specific involvement of Chr1p35.1 (ZSCAN-TLR12P) and Chr8p23.1 (HMGB1P46) in diabetic NP. | Involvement of Chr8p23.1 (HMGB1P46) in NP, with high heritability of Chr8p23.1 (HMGB1P46) in males. |
| Zhao et al. | 2015 | Role of HMGB1 in RGC in high glucose environment. | Increased levels of HMGB1 mRNA and protein in RGC and decreased cells survival in high glucose environment. Decreased levels of HMGB1 mRNA and protein in RGC and increased cells survival after siRNA HMGB1 injection. |
| Zhao et al. | 2016 | Role of CaMKIV in diabetic NP and its relationship with HMGB1 expression in DRG. | Increased pCAMKIV and HMGB1 levels in DRG of STZ-induced rats. Inhibition of CAMKIV reduced CAMKIV and HMGB1 expression in DRG of STZ-induced rats. |
| Wang et al. | 2018 | Role of sigma 1 receptor and its relationship with HMGB1 expression in DRG in NP. | In STZ-induced rats TA and TH correlate with increased HMGB1 expression in DRG. Stimulation of Sigma-1R induces TA and TH and increases HMGB1 levels. Blockade of Sigma-1R and HMGB1 reduce TA and TH. STZ-induced Sigma-1R knockdown rats have modest NP and no variation in HMGB1 levels. |
| Ref. | Year | Aim | HMGB1 |
|---|---|---|---|
| El-Asrar et al. | 2011 | Levels of HMGB1, RAGE in patients with PDR and correlation with MCP-1, sICAM-1, IL-1 beta, GM-CSF. | Increased HGMB1 levels in vitreous samples of patients with PDR. Increased HMGB1 levels in patients with PDR and hemorrhages. |
| El-Asrar et al. | 2012 | Levels of HMGB1 in patients with PDR and correlation with VEGF, G-CSF, sVE-cadherin, sEng. | Increased HMGB1 levels in vitreous samples of patients with PDR. |
| Mohammad et al. | 2012 | Role of HMGB1 in DR. | Increased HMGB1 levels in retinas of STZ-induced rats. Intravitreal administration of HMGB1 in non STZ-induced rats increases levels of ICAM-1, sICAM-1, HMGB1, RAGE, ERK1/2, NFkB and retinal permeability. Administration of GA reduced upregulation of HMGB1 in STZ-induced rats. |
| Gong et al. | 2014 | Role of HMGB1 and PLA2 in DR. | Increased HMGB1 levels in retinal tissue of STZ-induced rats; HMGB1 induces endothelial cells death directly and pericytes death through cytotoxic activity of glial cells. |
| Santos et al. | 2014 | Role of HMGB1 in vulnerability of endothelial cells and pericytes. | HMGB1 induces endothelial cells death directly and pericytes death through glial cells. No differences between neovascularization and levels of VEGF after HMGB1 subretinal administration. No involvement of HMGB1 in rats with oxygen induced retinopathy. |
| Fu et al. | 2015 | Serum HMGB1 and VEGF levels in DR patients. | Increased serum HMGB1 levels in DR patients. In vitro HMGB1 inhibits human retina pigment epithelium cells growth and it induces apoptosis. |
| Mohammad et al. | 2015 | Relationship between HMGB1 and NADPH oxidase-derived ROS in DR. | Increased HMGB1 levels and oxidative stress in vitreous fluid of PDR patients. HMGB1 enhances IL-1β, ROS, Nox2, PARP-1, and cleaved caspase-3 production by HRMEC. Diabetes and intra-vitreal injection of HMGB1 in normal rats induce significant upregulation of ROS, Nox2, PARP-1, and cleaved caspase-3 in the retina. |
| Yu et al. | 2015 | Role of HMGB1 in DR inflammation and cellular apoptosis. | Increased HMGB1 expression in retinas of STZ-induced rats. HMGB1 accelerates apoptosis of diabetic retinal cells. |
| Kim et al. | 2016 | Role of HMGB1, RAGE, NFkB in DR. | Increased HMGB1 cytoplasmic translocation in high glucose environment. |
| Jiang et al. | 2016 | Role of HMGB1 and TLR-9 in DR. | Increased HMGB1 and TLR-9 expression in retinas tissues and in RGC of STZ-induced rats. |
| Sohn et al. | 2016 | Protective effect of PCE in DR by inhibition of HMGB1 pathway. | Increased levels of HMGB1 in retinas of STZ-induced rats. Treatment with PCE reduces HMGB1 and RAGE expression in retinas of STZ-rats. |
| Chang et al. | 2017 | Role of hypoxia in HMGB1 release in DR. | Hypoxia induces HMGB1 cytoplasmic release. HMGB1 upregulates expression of VEGF, bFGF, TGF-β2, CTCF and phosphorylation of Akt, p38MAPK and NFkB but not ERK, JNK, Smad2. HMGB1 causes growth suppression and G1 cell cycle arrest in ARPE-19 cells. Neutralization of TLR4 and RAGE reduces HMGB1-driven cytokines production. |
| Jiang et al. | 2017 | Role of HMGB1siRNA in DR. | HMGB1siRNA reduces apoptosis and oxidative damage of retinal cells in STZ-induced rats (intravitreal injection) and in HRECs (HMGBsiRNA pre-treatment) of treated with high. Decreased IKKβ and NFκB protein expression after HMGB1 silencing. |
| Jiang et al. | 2018 | Role of HMGB1on the proteins involved in insulin signaling. | Recombinant HMGB1 blocks insulin receptor and Akt phosphorylation through RAGE and TLR-4. |
| Liu et al. | 2018 | Role of PKA in HMGB1 inhibition. | PKA inhibits cytoplasmic HMGB1, activating IGFBP-3 and SIRT1. |
| Zhang et al. | 2018 | Role of exosome derived from mesenchymal stem cells in retinal inflammation reduction. | MSC-exos overexpressing miR-126 suppresses HMGB1 expression and NLRP3 inflammasome activity in human retinal endothelial cells. |
| Ref. | Year | Aim | HMGB1 |
|---|---|---|---|
| Volz et al. | 2010 | Role of HMGB1 in diabetic heart disease. | High glucose treatment of cardiac fibroblasts, macrophages and cardiomyocytes increased HMGB1 mRNA expression and protein levels. Increased cardiac HMGB1 mRNA expression and protein levels in STZ-induced mice with post-myocardial infarction remodeling and HMGB1 blockage reduces post-myocardial infarction remodeling and markers of tissue damage. |
| Wang et al. | 2014 | Role of HMGB1 in high glucose-induced apoptosis of cardiomyocytes. | HMGB1siRNA reduces cell apoptosis in high glucose milieu through Ets-1/ERK1/2 signaling. |
| Wang et al. | 2014 | Role of HMGB1 in fibrosis and myocardial dysfunction. | HMGB1 silencing ameliorated LV dysfunction and remodeling in STZ-induced mice. High glucose milieu induced HMGB1 translocation and secretion in isolated cardiac fibroblasts. Administration of HMGB1 increased expression of collagens I and III and TGF-β1 in cardiac fibroblasts. HMGB1 inhibition reduced high glucose-induced collagen production, MMP activity, proliferation, MAPK signaling. |
| Tao et al. | 2015 | Extracellular communication pathways between cardiomyocytes and fibroblasts in DC. | Increased myocardial expression of HMGB1, collagen deposition and myocardial dysfunction and reduced IL-33 in STZ-induced mice. Inhibition of HMGB1 prevents myocardial collagen deposition and dysfunction. Increased HMGB1 secretion and collagen I production in high glucose induced cardiomyocytes/fibroblasts. HMGB1 inhibition reduces collagen I expression in the fibroblasts. |
| Song et al. | 2016 | Intracellular signaling pathway leading to cardiomyocyte HMGB1 expression in hyperglycemia. | Increased HMGB1 expression in high glucose-conditioned cardiomyocytes by PI3Kγ and Akt pathway. Treatment of cardiomyocytes with an antioxidant abolished high glucose-induced PI3Kγ and Akt activation and HMGB1 production. |
| Wu et al. | 2016 | Expression of HMGB1 pathway and oxidative stress in resveratrol-treated diabetic mice. | Lower serum and bone marrow-derived monocytes HMGB1 levels in STZ-induced rats treated with resveratrol. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Biscetti, F.; Rando, M.M.; Nardella, E.; Cecchini, A.L.; Pecorini, G.; Landolfi, R.; Flex, A. High Mobility Group Box-1 and Diabetes Mellitus Complications: State of the Art and Future Perspectives. Int. J. Mol. Sci. 2019, 20, 6258. https://doi.org/10.3390/ijms20246258
Biscetti F, Rando MM, Nardella E, Cecchini AL, Pecorini G, Landolfi R, Flex A. High Mobility Group Box-1 and Diabetes Mellitus Complications: State of the Art and Future Perspectives. International Journal of Molecular Sciences. 2019; 20(24):6258. https://doi.org/10.3390/ijms20246258
Chicago/Turabian StyleBiscetti, Federico, Maria Margherita Rando, Elisabetta Nardella, Andrea Leonardo Cecchini, Giovanni Pecorini, Raffaele Landolfi, and Andrea Flex. 2019. "High Mobility Group Box-1 and Diabetes Mellitus Complications: State of the Art and Future Perspectives" International Journal of Molecular Sciences 20, no. 24: 6258. https://doi.org/10.3390/ijms20246258






