Notch Signaling Affects Oral Neoplasm Cell Differentiation and Acquisition of Tumor-Specific Characteristics
Abstract
1. Introduction
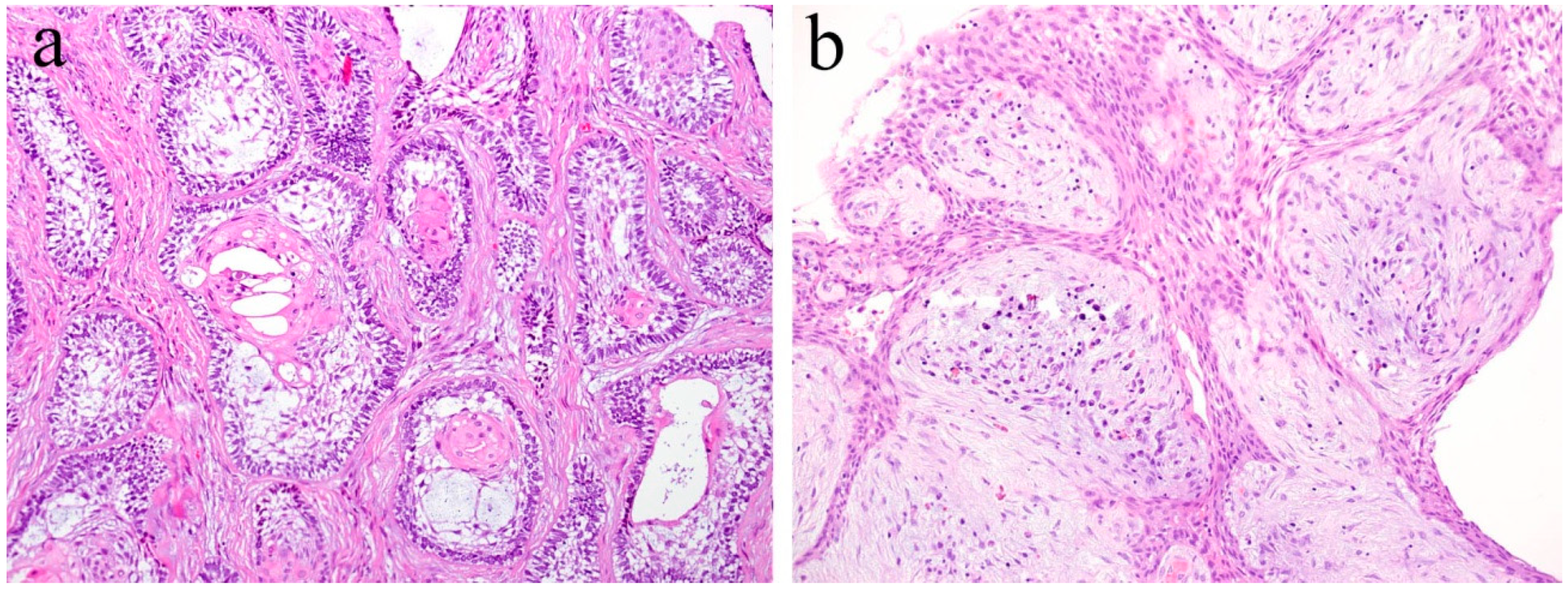
2. Histopathology of Ameloblastoma
3. Notch Signaling in Ameloblastoma
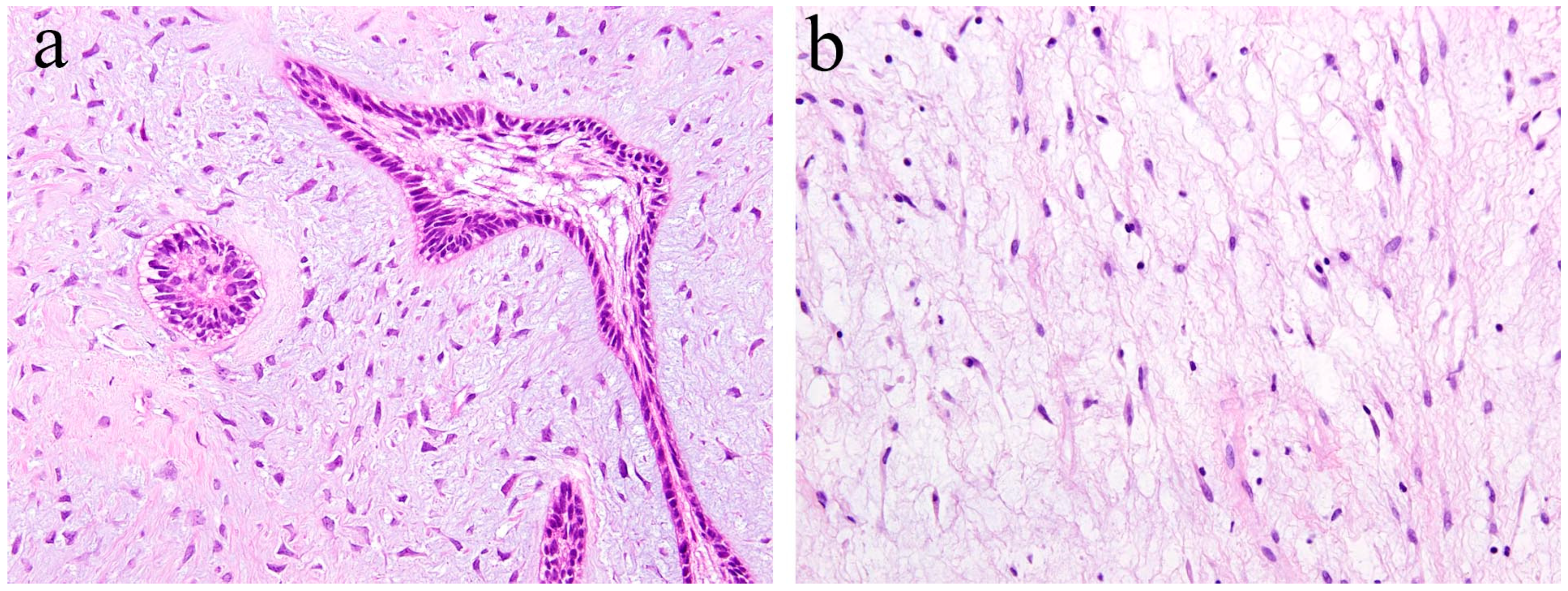
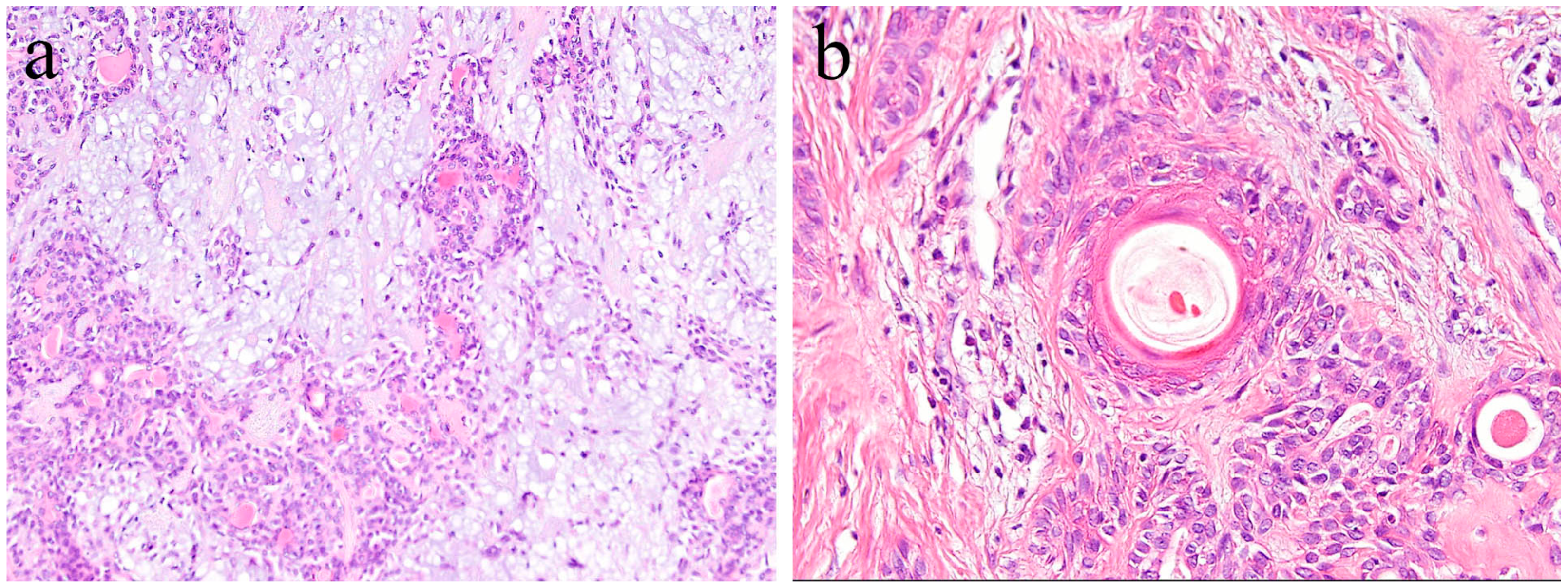
4. Histopathology of Ameloblastic Fibroma and Odontogenic Myxoma
5. Notch Signaling in Ameloblastic Fibroma and Odontogenic Myxoma
6. Histopathology of Calcifying Odontogenic Cyst
7. Notch Signaling in Calcifying Odontogenic Cyst
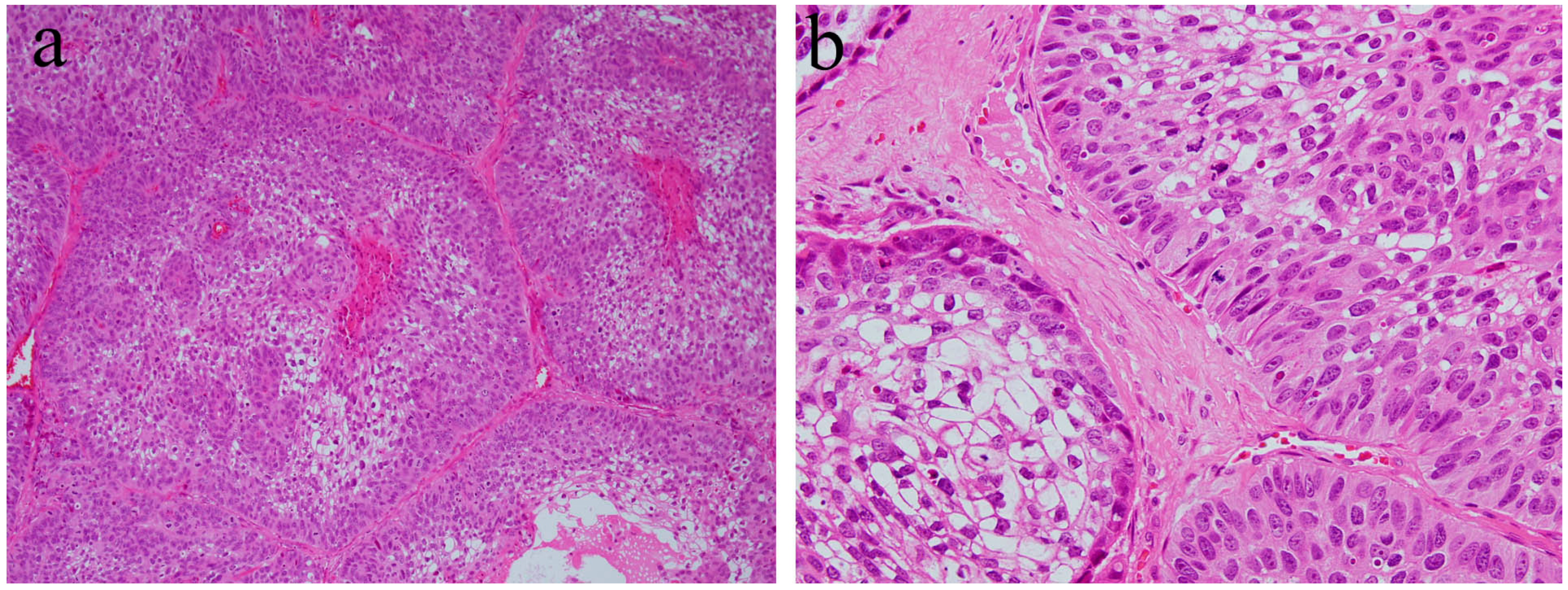
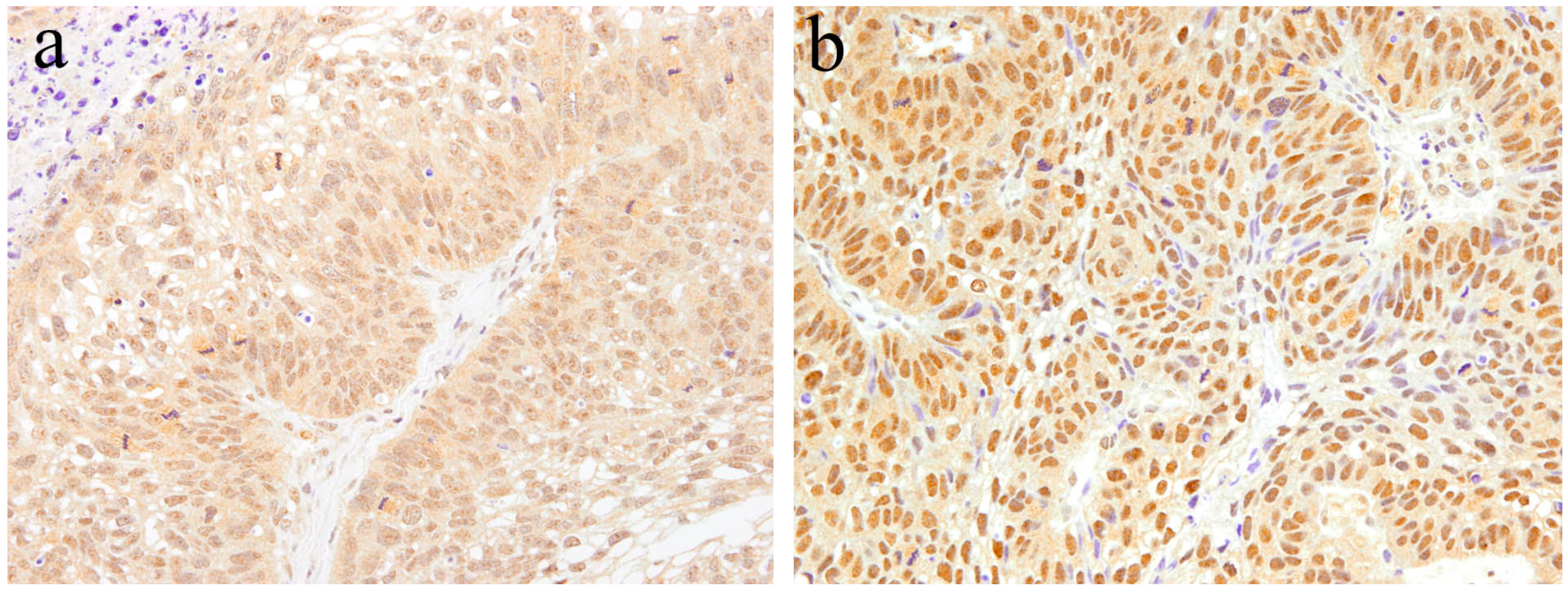
8. Histopathology of Ameloblastic Carcinoma
9. Notch Signaling in Ameloblastic Carcinoma
10. Histopathology of Pleomorphic Adenoma
11. Notch Signaling in Pleomorphic Adenoma
12. Conclusions
Funding
Conflicts of Interest
References
- Vered, M.; Muller, S.; Heikinheimo, K. Ameloblastoma. In WHO Classification of Head and Neck Tumours, 4th ed.; El-Naggar, A.K., Chan, J.K.C., Grandis, J.R., Takata, T., Slootweg, P.G., Eds.; IARC Press: Lyon, France, 2017; pp. 215–218. [Google Scholar]
- Muller, S.; Vered, M. Ameloblastic fibroma. In WHO Classification of Head and Neck Tumours, 4th ed.; El-Naggar, A.K., Chan, J.K.C., Grandis, J.R., Takata, T., Slootweg, P.G., Eds.; IARC Press: Lyon, France, 2017; pp. 222–223. [Google Scholar]
- Speight, P.; Ledesma-Montes, C.; Wright, J.M. Calcifying odontogenic cyst. In WHO Classification of Head and Neck Tumours, 4th ed.; El-Naggar, A.K., Chan, J.K.C., Grandis, J.R., Takata, T., Slootweg, P.G., Eds.; IARC Press: Lyon, France, 2017; pp. 239–241. [Google Scholar]
- Odell, E.W.; Adebiyi, K. Odontogenic myxoma/myxofibroma. In WHO Classification of Head and Neck Tumours, 4th ed.; El-Naggar, A.K., Chan, J.K.C., Grandis, J.R., Takata, T., Slootweg, P.G., Eds.; IARC Press: Lyon, France, 2017; pp. 229–230. [Google Scholar]
- Odell, E.W.; Muller, S.; Richardson, M. Ameloblastic carcinoma. In WHO Classification of Head and Neck Tumours, 4th ed.; El-Naggar, A.K., Chan, J.K.C., Grandis, J.R., Takata, T., Slootweg, P.G., Eds.; IARC Press: Lyon, France, 2017; pp. 206–207. [Google Scholar]
- Bell, D.; Bullerdiek, J.; Gnepp, D.R.; Schwrtz, M.R.; Stenman, G.; Triantafyllou, A. Pleomorphic adenoma. In WHO Classification of Head and Neck Tumours, 4th ed.; El-Naggar, A.K., Chan, J.K.C., Grandis, J.R., Takata, T., Slootweg, P.G., Eds.; IARC Press: Lyon, France, 2017; pp. 185–186. [Google Scholar]
- Nakano, K.; Watanabe, T.; Shimizu, T.; Kawakami, T. Immunohistochemical characteristics of bone forming cell in pleomorphic adenoma. Int. J. Med. Sci. 2007, 4, 264–266. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shayan, O.; Binshtok, U.; Hersch, M.; Rivkin, D.; Weinreb, S.; Amir-Zilberstein, L.; Khamaisi, B.; Oppenheim, O.; Desai, R.A.; Goodyear, R.J.; et al. Cell-Cell Contact Area Affects Notch Signaling and Notch-Dependent Patterning. Dev. Cell 2017, 40, 505–511. [Google Scholar] [CrossRef]
- Mitsiadis, T.A.; Lardelli, M.; Lendahl, U.; Thesleff, I. Expression of Notch 1, 2 and 3 is regulated by epithelial-mesenchymal interactions and retinoic acid in the developing mouse tooth and associated with determination of ameloblast cell fate. J. Cell Biol. 1995, 130, 407–418. [Google Scholar] [CrossRef]
- Abbaszadegan, M.R.; Riahi, A.; Forghanifard, M.M.; Moghbeli, M. WNT and NOTCH signaling pathways as activators for epidermal growth factor receptor in esophageal squamous cell carcinoma. Cell. Mol. Biol. Lett. 2018, 23, 42. [Google Scholar] [CrossRef]
- Frank, S.B.; Berger, P.L.; Ljungman, M.; Miranti, C.K. Human prostate luminal cell differentiation requires NOTCH3 induction by p38-MAPK and MYC. J. Cell Sci. 2017, 130, 1952–1964. [Google Scholar] [CrossRef] [PubMed]
- Muraki, E.; Nakano, K.; Maeda, H.; Takayama, M.; Jinno, M.; Kubo, K.; Yoshida, W.; Hasegawa, H.; Kawakami, T. Immunohistochemical localization of Notch signaling molecules in ameloblastomas. Eur. J. Med. Res. 2011, 16, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Takamine, K.; Ueda, Y.; Nakano, K.; Ochiai, T.; Sugita, Y.; Kubo, K.; Maeda, H.; Hasegawa, H.; Kawakami, T. Notch as a Possible Cell Differentiation Factor in Pleomorphic Adenomas. Int. J. Med. Sci. 2015, 12, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, T.; Nakano, K.; Tsujigiwa, H.; Nagatsuka, H.; Watanabe, T.; Okafuji, N.; Kurihara, S.; Hasegawa, H.; Nagai, N.; Kawakami, T. Notch signaling in mandibular condylar cartilage development. Eur. J. Med. Res. 2007, 12, 515–519. [Google Scholar]
- Nakano, K.; Siar, C.H.; Tsujigiwa, H.; Nagatsuka, H.; Nagai, N.; Kawakami, T. Notch signaling in benign and malignant ameloblastic neoplasms. Eur. J. Med. Res. 2008, 13, 476–480. [Google Scholar]
- Siar, C.H.; Nagatsuka, H.; Chuah, K.S.; Rivera, R.S.; Nakano, K.; Ng, K.H.; Kawakami, T. Notch4 overexpression in ameloblatoma correlates with the solid/multicystic phenotype. Oral Surg Oral Med Oral Pathol. Oral. Radiol. Endod. 2010, 110, 224–233. [Google Scholar] [CrossRef]
- Nakano, K.; Chelvanayagam, P.; Born, K.; Siar, C.H.; Ng, K.H.; Nagatsuka, H.; Kawakami, T. A study of recurrent giant cell odontogenic myxoma of the mandible with immunohistochemical examination of Notch. Oral. Med. Pathol. 2008, 12, 53–56. [Google Scholar] [CrossRef]
- Nakano, K.; Siar, C.H.; Tomida, M.; Matsuura, S.; Tsujigiwa, H.; Nagastuka, H.; Kawakami, T. Immunohistochemical observation of Notch signaling in a case of calcifying cystic odontogenic tumor. J. Hard. Tissue. Biol. 2010, 19, 147–152. [Google Scholar] [CrossRef]
- Cai, X.; Gong, P.; Huang, Y.; Lin, Y. Notch signalling pathway in tooth development and adult dental cells. Cell. Prolif. 2011, 44, 495–507. [Google Scholar] [CrossRef] [PubMed]
- Siar, C.H.; Kawakami, T.; Buery, R.R.; Nakano, K.; Tomida, M.; Tsujigiwa, H.; Han, P.P.; Nagatsuka, H.; Ng, K.H. Notch signaling and ghost cell fate in the calcifying cystic odontogenic tumor. Eur. J. Med. Res. 2011, 16, 501–506. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, T.; Shimizu, T.; Kimura, A.; Hasegawa, H.; Siar, C.H.; Ng, K.H.; Nagatsuka, H.; Nagai, N.; Kanda, H. Immunohistochemical examination of cytological differentiation in osteosarcomas. Eur. J. Med. Res. 2005, 10, 475–479. [Google Scholar] [PubMed]
- Locatelli, M.; Curigliano, G. Notch inhibitors and their role in the treatment of triple negative breast cancer: Promises and failures. Curr. Opin. Oncol. 2017, 29, 411–427. [Google Scholar] [CrossRef]
- Lubin, D.J.; Mick, R.; Shroff, S.G.; Stashek, K.; Furth, E.E. The notch pathway is activated in neoplastic progression in esophageal squamous cell carcinoma. Hum. Pathol. 2018, 72, 66–70. [Google Scholar] [CrossRef]
- Doi, K.; Imai, T.; Kressler, C.; Yagita, H.; Agata, Y.; Vooils, M.; Hamazaki, Y.; Inoue, J.; Minato, N. Crucial role of the Rap G protein signal in Notch activation and leukemogenicity of T-cell acute lymphoblasticleukemia. Sci. Rep. 2015, 5, 7978. [Google Scholar] [CrossRef]
- Ding, L.C.; She, L.; Zheng, D.L.; Huang, Q.L.; Wang, J.F.; Zheng, F.F.; Lu, Y.G. Notch-4 contributes to the metastasis of salivary adenoid cystic carcinoma. Oncol. Rep. 2010, 24, 363–368. [Google Scholar]
- Luo, K. Signaling Cross Talk between TGF-β/Smad and Other Signaling Pathways. Cold Spring Harb Perspect. Biol. 2017, 9, a022137. [Google Scholar] [CrossRef] [PubMed]
- Fujii, S.; Nagata, K.; Matsumoto, S.; Kohashi, K.I.; Kikuchi, A.; Oda, Y.; Kiyoshima, T.; Wada, N. Wnt/β-catenin signaling, which is activated in odontomas, reduces Sema3A expression to regulate odontogenic epithelial cell proliferation and tooth germ development. Sci. Rep. 2019, 9, 4257. [Google Scholar] [CrossRef] [PubMed]
- Estrach, S.; Ambler, C.A.; Lo Celso, C.; Hozumi, K.; Watt, F.M. Jagged 1 is a beta-catenin target gene required for ectopic hair follicle formation in adult epidermis. Development 2006, 133, 4427–4438. [Google Scholar] [CrossRef] [PubMed]
- Mu, Y.P.; Zhang, X.; Xu, Y.; Fan, W.W.; Li, X.W.; Chen, J.M.; Chen, G.F.; Liu, P. Notch signaling pathway participates in the differentiation of hepatic progenitor cells into bile duct epithelial cells and progression of hepatic fibrosis in cholestatic liver fibrosis rat. Zhonghua Bing Li Xue Za Zhi 2017, 46, 400–405. [Google Scholar]
- Thélu, J.; Rossio, P.; Favier, B. Notch signalling is linked to epidermal cell differentiation level in basal cell carcinoma, psoriasis and wound healing. BMC Derm. 2002, 2, 7. [Google Scholar] [CrossRef]
- Kasagi, Y.; Chandramouleeswaran, P.M.; Whelan, K.A.; Tanaka, K.; Giroux, V.; Sharma, M.; Wang, J.; Benitez, A.J.; DeMarshall, M.; Tobias, J.W.; et al. The Esophageal Organoid System Reveals Functional Interplay Between Notch and Cytokines in Reactive Epithelial Changes. Cell Mol. Gastroenterol. Hepatol. 2018, 5, 333–352. [Google Scholar] [CrossRef] [PubMed]
- Okuda, Y.; Nakano, K.; Suzuki, K.; Sugita, Y.; Kubo, K.; Maeda, H.; Okafuji, N.; Hasegawa, H.; Kawakami, T. Wnt signaling as a possible promoting factor of cell differentiation in pleomorphic adenomas. Int. J. Med. Sci. 2014, 11, 971–978. [Google Scholar] [CrossRef]
- Ueda, Y.; Nakano, K.; Ochiai, T.; Yoshida, W.; Sugita, Y.; Kubo, K.; Maeda, H.; Hasegawa, H.; Kawakami, T. A possible functional role of HSP27 as a molecular chaperone of Wnt in cell differentiation of pleomorphic adenomas. Int. J. Dent. Oral Sci. 2016, 3, 340–343. [Google Scholar]
- Sun, W.; Gaykalova, D.A.; Ochs, M.F.; Mambo, E.; Arnaoutakis, D.; Liu, Y.; Loyo, M.; Agrawal, N.; Howard, J.; Li, R.; et al. Activation of the Notch pathway in head and neck cancer. Cancer Res. 2014, 15, 1091–1104. [Google Scholar] [CrossRef]
- Yap, L.; Lee, D.; Khairuddin, A.; Pairan, M.; Puspita, B.; Siar, C.; Paterson, I. The opposing roles of Notch signaling in head 8and neck cancer: A mini review. Oral Dis. 2015. [Google Scholar] [CrossRef]





| Clinical Features | Histological Features | |
|---|---|---|
| Odontogenic tumor/cyst | ||
| Ameloblastoma | Locally invasive tumor. Clinically benign but has a high risk of recurrence. | Epithelial tumor nests are similar to the enamel organ. Tumor cells arranged at periphery of tumor nests are columnar or cuboidal. |
| Ameloblastic fibroma | Usually grows slowly and painlessly, causing swelling of the jawbone. | Tumor composed of ameloblastoma like epithelial tissue and dental papilla like mesenchyme. |
| Odonotogenic myxoma | Usually grows slowly and painlessly, causing swelling of the jawbone. Clinically benign but has a high risk of recurrence. | Spindle or stellate tumor cells proliferating in an abundant myxoid extracellular matrix. |
| Calcifying odontogenic cyst | Usually grows slowly and painlessly, causing swelling of the jawbone. | Ameloblastoma-like lining epithelium including ghost cells and calcified foci. Dentin-like hard tissue and dysplastic dentin may be observed. |
| Ameloblastic carcinoa | Rare odontogenic carcinoma. Most cases arise in posterior segments of the jaws. | Histological similarity of ameloblastoma with strong cytological atypism. |
| Salivary gland tumour | ||
| Pleomorphic adenoma | Tumor shows slowly and painless growing. Most cases arise in the parotid. | Admixture of polygonal epithelial and neoplastic myoepithelial cells in a variable background stroma. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nakano, K.; Takabatake, K.; Kawai, H.; Yoshida, S.; Maeda, H.; Kawakami, T.; Nagatsuka, H. Notch Signaling Affects Oral Neoplasm Cell Differentiation and Acquisition of Tumor-Specific Characteristics. Int. J. Mol. Sci. 2019, 20, 1973. https://doi.org/10.3390/ijms20081973
Nakano K, Takabatake K, Kawai H, Yoshida S, Maeda H, Kawakami T, Nagatsuka H. Notch Signaling Affects Oral Neoplasm Cell Differentiation and Acquisition of Tumor-Specific Characteristics. International Journal of Molecular Sciences. 2019; 20(8):1973. https://doi.org/10.3390/ijms20081973
Chicago/Turabian StyleNakano, Keisuke, Kiyofumi Takabatake, Hotaka Kawai, Saori Yoshida, Hatsuhiko Maeda, Toshiyuki Kawakami, and Hitoshi Nagatsuka. 2019. "Notch Signaling Affects Oral Neoplasm Cell Differentiation and Acquisition of Tumor-Specific Characteristics" International Journal of Molecular Sciences 20, no. 8: 1973. https://doi.org/10.3390/ijms20081973
APA StyleNakano, K., Takabatake, K., Kawai, H., Yoshida, S., Maeda, H., Kawakami, T., & Nagatsuka, H. (2019). Notch Signaling Affects Oral Neoplasm Cell Differentiation and Acquisition of Tumor-Specific Characteristics. International Journal of Molecular Sciences, 20(8), 1973. https://doi.org/10.3390/ijms20081973




