Abstract
Studies concerning the role of arachidonic acid (AA) and its metabolites in kidney disease are scarce, and this applies in particular to idiopathic nephrotic syndrome (INS). INS is one of the most frequent glomerular diseases in childhood; it is characterized by T-lymphocyte dysfunction, alterations of pro- and anti-coagulant factor levels, and increased platelet count and aggregation, leading to thrombophilia. AA and its metabolites are involved in several biological processes. Herein, we describe the main fields where they may play a significant role, particularly as it pertains to their effects on the kidney and the mechanisms underlying INS. AA and its metabolites influence cell membrane fluidity and permeability, modulate platelet activity and coagulation, regulate lymphocyte activity and inflammation, preserve the permeability of the glomerular barrier, influence podocyte physiology, and play a role in renal fibrosis. We also provide suggestions regarding dietary measures that are able to prevent an imbalance between arachidonic acid and its parental compound linoleic acid, in order to counteract the inflammatory state which characterizes numerous kidney diseases. On this basis, studies of AA in kidney disease appear as an important field to explore, with possible relevant results at the biological, dietary, and pharmacological level, in the final perspective for AA to modulate INS clinical manifestations.
1. Introduction
Idiopathic nephrotic syndrome (INS) is one of the most frequent glomerular diseases in childhood [1]. It is characterized by proteinuria, caused by podocyte damage, hypoalbuminemia, hyperlipidemia, and edema [1]. While the exact cause of podocyte damage is still not completely understood [1], it is well known that hyperlipidemia is related to urinary loss of transport proteins, which carry free cholesterol, and to the consequent compensatory increase in the synthesis of proteins involved in triglyceride metabolism [1]. Two theories have been proposed to explain the pathogenesis of edema in INS. According to the classical underfill hypothesis, hypoalbuminemia reduces plasma oncotic pressure, which leads to sodium and water retention and water leakage into the interstitium [2]. Meanwhile, the overfill hypothesis postulates proteinuria to be the primary cause of sodium retention, with consequent volume expansion and leakage of excess fluid into the interstitium [3].
Other biochemical alterations were also described in INS, such as changes in pro- and anti-coagulation factors’ levels and increased platelet count and aggregation, leading to a hypercoagulable state [4].
Based on their response to corticosteroid therapy, children with INS are classified as steroid-sensitive patients, which includes those with infrequent relapses, frequently relapsing or steroid-dependent patients who present a favorable prognosis, or steroid-resistant patients, who carry an unfavorable prognosis in the majority of cases. Histopathology usually reveals minimal change of disease, which is characterized by normal glomerular appearance on light microscopy and evidence of podocyte foot processes’ alterations on electron microscopy; focal segmental glomerulosclerosis and interstitial fibrosis may be found in steroid-resistant cases [5,6].
The pathogenesis of INS has not yet been fully clarified. Excluding genetic causes, the main theory for immune-mediated cases involves a dysfunction of T lymphocytes, which would switch to the production of still poorly defined permeability factors that interfere with the expression and/or function of key proteins in the podocyte, thus being the main culprits of proteinuria [7]. Candidates for the circulating factors that affect glomerular permeability include angiopoietin-like 4 (ANGPTL4), cardiotrophin-like cytokine-1 (CLC-1), and soluble urokinase plasminogen activator receptor (suPAR) [1].
Arachidonic acid (AA) is a long-chain polyunsaturated fatty acid of the omega-6 group and represents 7% to 10% of total circulating fatty acids; it is the second most abundant omega-6 fatty acid in the human body [8] (Table 1), with linoleic acid (LA) being the first. AA is synthesized endogenously from LA through three steps mediated by two enzymes, desaturase and elongase, and may also be derived from the diet. In turn, AA is a substrate of elongases for the synthesis of longer fatty acids of the omega-6 series.

Table 1.
Blood omega-6 levels in healthy subjects. Linoleic acid, AA, omega 6, total saturated fatty acid, monounsaturated fatty acids, and total omega-3 levels in human subjects. AA is the second highest fatty acid of the omega-6 series. Data are expressed as percentage of total fatty acids [4].
AA is metabolized by three types of oxygenases: cyclooxygenase (COX), lipoxygenase (LOX), and cytochrome P450, leading to the generation of eicosanoids, namely prostaglandins, thromboxane, leukotrienes, and hydroxyeicosatetraenoic acids.
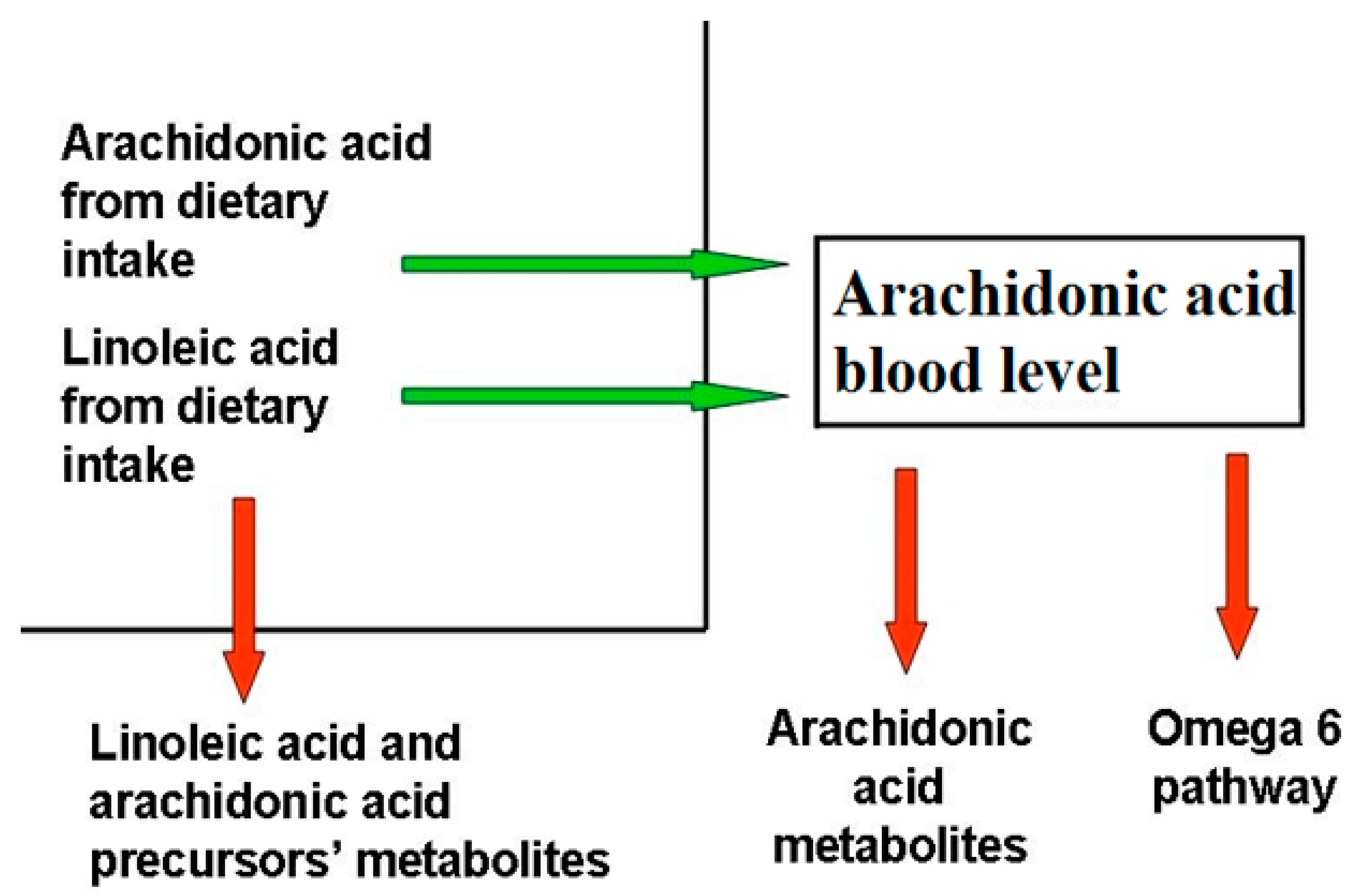
Blood AA levels do not reflect its synthesis and metabolization pathways (Figure 1), as they are maintained as constant, even at the expense of other biological factors, as observed in patients with epidermolysis bullosa [9], where, despite the large amount of active AA metabolites, the AA level is comparable to that of healthy controls. This phenomenon has been observed in several other chronic inflammatory disorders, for instance cystic fibrosis [10], even if the exact mechanism behind it is unclear.
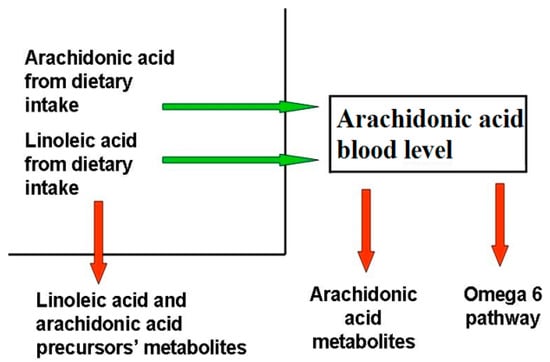
Figure 1.
Factors determining blood arachidonic acid levels. The factors influencing AA levels belong to two categories, those that increase (green arrow) AA blood levels, like diet uptake and the omega-6 pathway from LA, and those that decrease them (red arrow), like the synthesis of omega-6 FA downstream AA, AA metabolization, and the reduction of AA precursors due to their metabolization. Diseases able to modify AA blood levels act through the same mechanisms.
AA is involved in several biological processes, either in health or disease. Herein, we describe its role in nephrotic syndrome from a biological and clinical perspective. AA influences cell membrane fluidity and permeability and modulates platelet function and immune system activation; furthermore, it affects glomerular and tubular function, the physiopathology of podocyte, and the process of renal fibrosis. We also detail the interactions between AA and the common drugs prescribed for INS treatment. Finally, the role of dietary AA balance and its nutritional sources are discussed.
For this review, PubMed (www.pubmed.gov, accessed on 28 February 2021) was the only source of the articles. No limit was given regarding the date of publication of the articles, and the following keywords were used: arachidonic acid, arachidonic acid metabolism, cell membrane, immune system, nephrotic syndrome, membrane receptor, coagulation, platelets, arachidonic acid pathway, TXA2, LTB4, PGE2, CNI pharmacogenomics, cyclosporine A, tacrolimus, kidney disease, arachidonic acid and kidney, podocyte, podocyte and arachidonic acid, 20-HETE, 20-HETE metabolism, renal fibrosis, SNI pharmacogenomics, CYP, and all the key words related to the biological mechanism reported in each chapter.
2. Cell Membrane Fluidity and Permeability
It was recently described that erythrocyte membranes of patients with INS differ from those of normal subjects, particularly due to reduced membrane fluidity [11].
AA is one of the most abundant fatty acids in the cell membrane, to which it endows mobility and flexibility [12,13]. The fatty acid composition determines the viscosity of the cell lipid bilayer and membrane fluidity, thus directly affecting the function of specific membrane proteins, like, for example, those involved in cellular inflammatory signaling, namely lymphocyte function-associated antigen 1 (LFA-1), intercellular adhesion molecule 1 (ICAM-1), and cluster of differentiation 2 (CD2) [12,13].
With regard to membrane permeability, AA acts on Ca2+ cell load [10] with a double effect: at low micromolar concentrations it increases Ca2+-ATPase activity, while at higher concentrations it reduces ATPase activity. This may be due to an unspecific and non-physiological inhibitory effect on the hydrolytic activity of P-type ATPase. ATPases are a superfamily of lipid pumps involved, among other functions, in secretion and absorption at the kidney level; these pumps are blocked by protein kinase C inhibitors [14]. AA increases membrane permeability to calcium, which is a key factor for platelet activation [15].
AA may act on ion channels by either binding to or inserting among the membrane molecules, thus modifying the mechanical properties of the cell membrane and modulating channel function [16].
AA also has a direct effect on several membrane potassium channels, either by accelerating their inactivation (in particular, the A-type channels and delayed rectifier channels), or by inducing the activation of large-conductance voltage-independent channels. The two-pore domain potassium channels are inactivated by AA as well, in contrast to what usually occurs with classical K channel-blocking drugs [16]. Transient receptor potential channels (TPR) are instead activated directly by AA and its lipoxygenase (LOX)-derived metabolites [16] (namely, 12- and 15-(S)-hydroperoxyeicosatetraenoic acids, 5- and 15-(S)-hydroxyeicosatetraenoic acids, and leukotriene B4). LOX metabolites can activate the TPR channel by virtue of their structure that mimics the capsaicin structure [17]. Interestingly, AA and its metabolic byproducts effects on calcium and potassium balance at the membrane level have been hypothesized to underlie the molecular-related derangements in INS [6].
As it concerns membrane fluidity, albumin is the main fatty acid-binding protein in extracellular fluid, having seven fatty acid-binding sites [18]. Albumin increases AA release from cell membranes in a concentration-dependent manner, by interacting with membrane phospholipids on the extracellular surface; in particular, positively charged arginine residues at or near albumin’s binding sites for LCFA interact with AA, determining its release from the phospholipid layer [19]. Thus, albumin decreases cell membrane permeability of endothelial and circulating cells to water and small solutes [19].
In conclusion, the amount of AA in cell membranes regulates several cellular functions and all factors that vary the amount of AA in the membrane may play a significant pathogenetic role in renal disease.
3. Platelet Aggregation and Coagulation
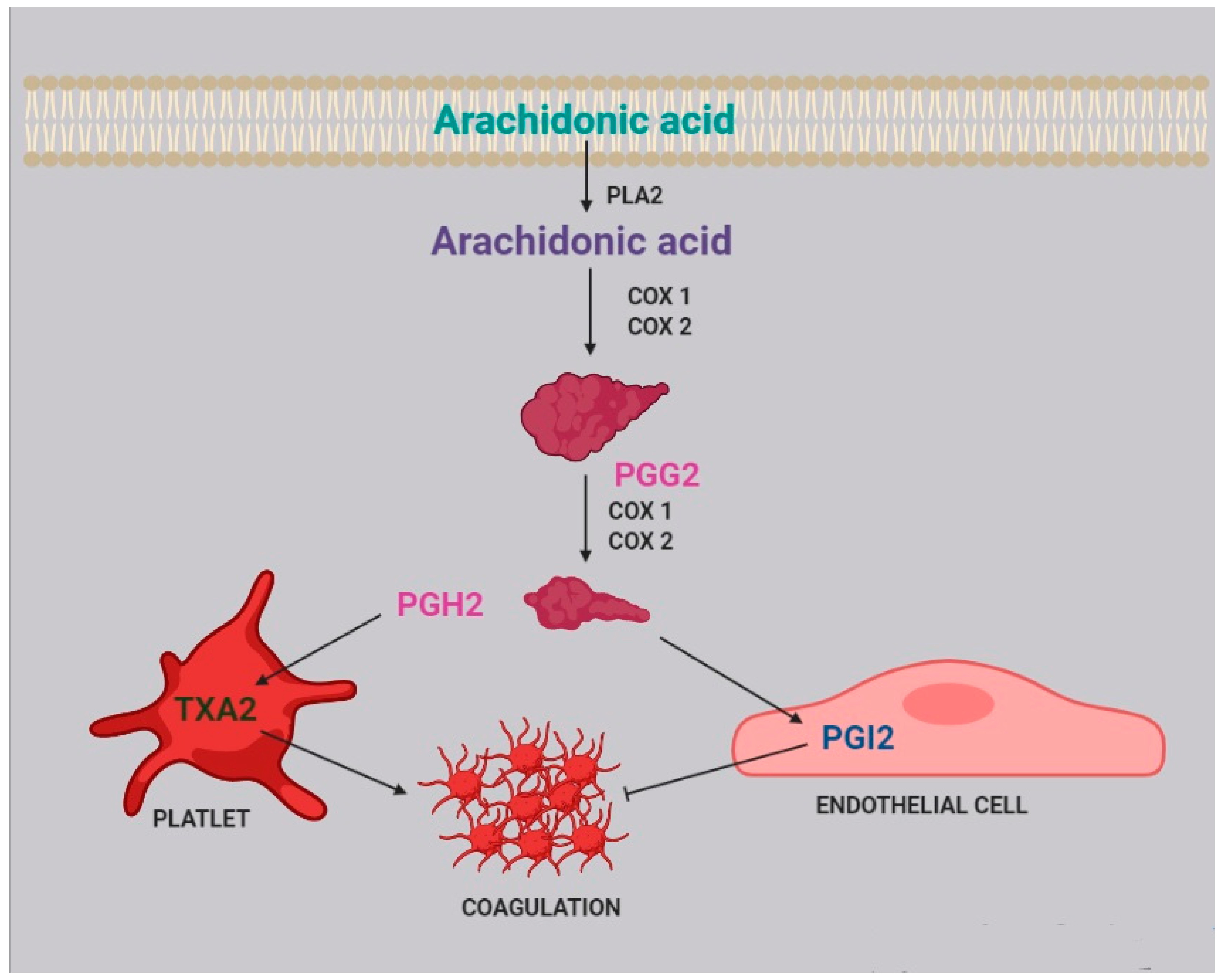
Two of the most active compounds related to platelet function are thromboxane and prostacyclin, both metabolites of AA [20]. AA is released from platelet membrane by phospholipase A2 (PLA2), which hydrolyzes the bond between the second fatty acid of phospholipids and the glycerol molecule. The released AA is then metabolized by cyclooxygenase [21], generating prostaglandin G2, and thereafter prostaglandin H2. Afterwards, two different pathways can take place: the first one, within the platelets, leads to the synthesis of thromboxane A2 (TXA2) and subsequently B2 (TXB2); the second one, within the endothelial cells, leads to the synthesis of prostacyclin (PGI2) (Figure 2).
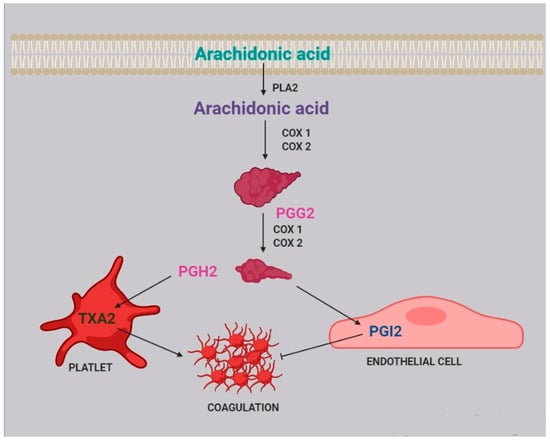
Figure 2.
Role of arachidonic acid in the coagulation process. Arachidonic acid is released from cell membranes by phospholipase A2 (PLA2) and subsequently metabolized by COX1 and COX2 to obtain prostaglandin G2 (PGG2) and H2 (PGH2).
TXA2 stimulates platelet activation and aggregation, via platelet fibrinogen-binding αIIbβ3 receptors [4]. Prostacyclins, in contrast, inhibit platelet activation by activating G protein-coupled receptors on platelets and endothelial cells. Upon binding to the prostacyclin receptor, PGI2 induces adenyl cyclase cAMP production, which in turn inhibits platelet activation [22].
A higher incidence of increased platelet aggregation and thromboembolism has been reported in nephrotic syndrome, in relation to consistently elevated levels of fibrinogen. Moreover, both hyperlipidemia and hypoalbuminemia, which are characteristic findings of nephrotic syndrome, increase thromboxane availability, through the production of TXA2 precursors and the removal of TXA2 inhibitors [23]. The exact mechanism underlying this process is still unknown, but it probably involves an increase in PLA2 activity, related to the abnormally high cholesterol levels [24]. Therefore, arachidonic acid, which is the precursor of thromboxane, may be considered a crucial player in the platelet-related coagulation process.
It is worth noting that in a clinical trial that recruited six healthy male volunteers, fed for 50 days with a diet containing 1.7 gr/day of AA, and six controls, fed with a diet containing 210 mg/day of AA [20], moderate intakes of foods rich in AA, like those of the first group, had only mild effects on blood coagulation, platelet function, and platelet fatty acid composition compared to controls. The authors attributed the poor efficacy of arachidonic acid supplementation to the moderate amount in which it was supplied.
In platelets, PGH2 is metabolized to tromboxane A2, activating coagulation and platelet aggregation, while in endothelial cells, prostaglandin I2 (PGI2), which has an anticoagulant effect, is generated.
4. Immune System
The therapeutic efficacy of Rituximab in modifying the course of steroid-dependent nephrotic syndrome suggested that B cells play a key role in the pathogenesis of INS. This was recently confirmed by evidence of a pathological increase in memory B cells in INS [25]. Moreover, other studies showed a decrease of Treg cells [26], dysregulation of T-cells [27], lower levels of NK and NKT cells, and increased levels of inflammatory markers during proteinuria [28,29]. These studies confirm [1] that the immune system plays a pivotal role in non-genetic INS and specifically in the loss of the glomerular barrier function, by activating the inflammatory process against podocytes.
In immune cells, like lymphocytes, neutrophils, and monocytes, AA constitutes about 20% of total fatty acids, while EPA and DHA constitute 1% and 2.5%, respectively [30]. It was reported that oral administration of omega-3 fatty acids changes the pattern of production of eicosanoids, by increasing resolvins production, thus affecting phagocytosis, T-cell signaling, and antigen presentation capability. These effects seem to be mediated at the membrane level [30].
The distribution of AA within intracellular lipid pools in inflammatory cells has an important role in regulating eicosanoids production. In fact, a pool of AA was identified within the triglycerides of mast cells, eosinophils, monocytes, and platelets [31].
When inflammatory cells are activated, AA is released from membrane phospholipids into the cell and partially incorporated into intracellular triglycerides, ready to supply membrane phospholipids again after cell activation has ended [32].
Thus, AA metabolites can act in several ways on lymphocyte activity, affecting inflammation levels [32,33,34] (Table 2) and possibly the course of INS.

Table 2.
Effects of AA metabolites on immune cells [38]; AA metabolites affect immune cells in various ways, modulating the immune response and inflammation.
Regarding B, NK, and T cells, the main AA metabolites involved are PGE2, LTB4, and TXA2.
PGE2 is produced by nearly all cells within the body [35]. Secreted PGE2 acts in an autocrine or paracrine manner through its four cognate G protein-coupled receptors EP1 to EP4 [36]. It inhibits T-cell and NK-cell proliferation, as well as IFN-γ and IL-12 production [37], binding their cell-surface receptors [38]. PGE2 also inhibits B-cell activation secondary to IL-4 stimulation in a specific manner and enhances IgE and IgG1 production [39].
LTB4 exerts pleiotropic effects on lymphocytes and regulates the immune response in a dynamic, cell type- and context-dependent manner: LTB4 enhances T-cell recruitment, it inhibits de novo iTreg generation and increases interleukin-17 (IL-17) cytokine production during T-cell differentiation. LTB4 also regulates the migration of various lymphoid-derived cell types in different ways that vary depending on disease and tissue. [40].
TXA2, another product of AA metabolism, inhibits naïve T-cell proliferation and exerts several effects on mature T lymphocytes: it inhibits T-cell interaction with dendritic cells, increases T-cell proliferation and activation, and has been shown to topically enhance the cytotoxic activity of immune cells [37].
Moreover, eosinophils, mast cells, macrophages, dendritic cells, and Th2 lymphocytes have surface membrane receptors for arachidonic-derived metabolites, in particular for prostaglandin D2, cysteinyl leukotrienes D4 and E4, and lipoxin A4 [33], but these findings have not been confirmed so far in patients with INS.
A pharmacological modulation of AA metabolites could decrease the inflammatory damage to the podocyte. The pathogenetic role of AA is supported by the fact that medications have been recently administered to target AA metabolism and decrease kidney inflammation [21,34]. They include aspirin, nimesulide, licofelone, baicalein, and others. Some of them are in the early stages of development for kidney diseases like diabetic nephropathy, glomerulonephritis, and idiopathic membranous nephropathy [34].
5. Kidney Glomerular and Tubular Function
Epoxyeicosatrienoic acids (EETs) are produced in several tissues, like the heart, the muscles, the kidneys, the pancreas, the lungs, and the brain [41], but mainly in the vascular endothelium, in response to various PLA2-activating stimuli, EETs activity could be reduced by metabolization made by soluble epoxyde hydrolase (sHE) [42].
EETs modulate kidney function acting directly on tubular ionic transport, vascular tone, and cellular proliferation, and have a nephro-protective role [43] due to their anti-inflammatory properties.
EETs in fact induce vasodilatation in an autocrine manner [44] and have anti-apoptotic activity; it was also reported that their generation is reduced in case of renal disease [42], even if no explanation regarding such a mechanism was given.
Glomerular inflammation is mitigated by EETs, which decrease the influx of neutrophils and macrophages and decrease the production of cytokines, monocyte chemotactic protein-1, TNF-α, macrophage inflammatory protein 2, and ICAM-1 [45]. The protective effect is due to EETs renal vasodilator and antipressor response to salt loading, through the inhibition of renal tubular Na+ reabsorption and the increase of Na+ renal excretion, resulting in an anti-hypertensive effect [46] that is probably mediated by A2A receptors [47].
20-HETE, another eicosanoid derived from AA metabolism, shares the same protective properties as EETs [48]. It plays a predominant role in the regulation of renal tubular and vascular function, and variants in the genes encoding for the enzymes that produce 20-HETE are associated with hypertension [49].
It has been shown that sustained production of 20-HETE in the glomerulus is required to maintain the glomerular permeability barrier to albumin [48]. It is still unclear which cell types in the glomerulus express the CYP enzymes that synthetize 20-HETE, and the exact mechanisms by which this molecule influences the glomerular permeability barrier are yet to be defined as well [48], although it is likely that its effects are mediated by the modulation of Na+-K+-ATPase, Na+-K+- 2Cl− cotransporter, and K+ channel activity in nephrons [49] through the activation of the PKC pathway [50].
20-HETE is also involved in podocyte apoptosis, by regulating the canonical transient receptor potential-6 (TRPC6) channels and increasing the Ca2+ flux [51].
In patients affected by nephrotic syndrome who develop early hypertension [52], a decreased concentration of 20-HETE in the proximal tubule has been observed [53] in association with increased albumin permeability in the glomeruli, which worsens proteinuria and glomerular injury [54]; this finding supports the role of 20-HETE in preserving glomerular permeability barrier to albumin. However, it is unknown if the reduction of 20-HETE causes or is caused by hypertension.
In conclusion, 20-HETE may act in different ways (protective or pro-apoptotic) in different cell types and kidney regions.
6. Podocyte Physiopathology and Infections
It is well known that, in the course of nephrotic syndrome, infections lead to an exacerbation of proteinuria [55] and are an important risk factor for relapses [1,56,57].
Infections activate the immune system, triggering the inflammatory cascade. It was recently reported that during inflammation two enzymes are induced: 15-lipoxygenase (15-LO) and secreted phospholipase A2 (sPLA2) [58]. Notably, 15-LO is expressed in human podocytes [59], while sPLA2 is expressed in platelets, neutrophils, eosinophils, and macrophages [60]. sPLA2 releases AA from membrane phospholipids [51,61] acting in a paracrine way.
In glomerular podocytes, intracellular free AA is metabolized to PGE2, which, by interacting with the EP 4 receptor (prostaglandin E2 receptor 4) expressed by podocytes, reduces AA release [52]. This loop regulates podocyte function in both physiological and pathological conditions and is able to change PGE2 synthesis [62].
As described above, during the course of an infection, intracellular AA levels increase due to the action of sPLA2. It was reported that in podocytes, an excess of AA activates protein kinase A, which in turn promotes c-Abl activation and nephrin phosphorylation, thus causing actin cytoskeleton remodeling and podocyte injury [63]. This mechanism could partially explain the frequent recurrence of proteinuria during infectious episodes in children. Moreover, an increase in sPLA2 1B levels and PLA2R expression has been observed to be positively associated to podocyte apoptosis in kidneys of patients with idiopathic membranous nephropathy [64].
Podocyte foot process injury and podocyte apoptosis due to cytoskeleton remodeling were also attributed to a change of Ca2+ efflux [51], driven by 20-HETE, the main AA metabolite. It has also been observed that 20-HETE increases the current Ca2+ flowing through TRPC6 channels in the podocyte [51], which are located at the slit diaphragm, possibly leading to cellular injury.
Studies on the relation between AA and the podocyte are scant, but very promising for increasing our knowledge of the pathogenesis of renal damage in INS.
7. Renal Fibrosis
Renal fibrosis is a process that progresses independently of the primary renal disease [65] and represents a failed wound-healing process of the kidney tissue. Renal biopsies of patients with steroid-resistant nephrotic syndrome often show glomerulosclerosis and interstitial fibrosis, which are associated with progression to end-stage kidney disease in more than 50% of cases [65], a poor prognosis that heightens the necessity of increasing our knowledge of the mechanisms underlying fibrosis.
Renal fibrosis is characterized by connective tissue deposition in the kidney parenchyma, particularly in the interstitial space and within the walls of glomerular capillaries, and by the consequent cellular processes. Fibrosis also interferes with normal tubular function, leading progressively to organ failure [65,66].
The scar tissue contains fibrillar collagen I and III as well as some constituents of the normal capillary basement membrane, like collagen IV and V, fibronectin, laminin, perlecan, and heparin [66].
Fibrosis is associated with leukocyte recruitment, angiogenesis, vascular leak, and the appearance of myofibroblasts. In particular, both the glomerulus and the interstitium attract large numbers of leukocytes, the majority of which are of myeloid lineage, and mostly neutrophiles in acute settings, whereas macrophages and dendritic cells predominate in chronic settings. In the case of chronic immune-mediated diseases, T lymphocytes are predominant [66].
Activated macrophages may either damage the tissue directly or generate profibrotic cytokines, including TGF-β and other growth factors, and are capable of producing some matrix constituents. It is therefore evident that fibrosis and renal inflammation, primarily driven by immune system activation, are closely related.
Beyond its role in immune function regulation, AA is also directly related to fibrosis. In vitro experiments of cell cultures incubated with PUFAs showed that AA is able to induce upregulation of the expression of TGF-β, fibronectin 1 (FN1), connective tissue growth factor (CTGF), and collagen IV, all compounds related to fibrosis [67]. AA also enhances in vitro angiotensin II (AngII)-induced gene expression [67], activating mechanisms that mediate renal damage. Interestingly, omega-3 EPA and DHA, if administered with AA, suppress the effects of both AA and AngII [67]. On the other side, angiotensin II is degraded to form angiotensin-(1-7), which inhibits angiotensin II-stimulated phosphorylation of the mitogen-activated protein kinases (MAPKs) p38, extracellular signal-related kinase (ERK1/ERK2), and C-JUN N-terminal kinase (JNK) in proximal tubular cells, thus exerting a protective role against fibrosis. As a matter of fact, the p38 MAPK phosphorylation leads to the release of AA and the production of TGF-β 1 and extracellular matrix proteins [68].
20-HETE, an AA metabolite, also plays a distinct role in fibrogenesis, by activating the renin-angiotensin-aldosterone system (RAAS), by inducing vascular expression of ACE downstream of NF-κB activation [69,70]. It is well known that the RAAS is involved in renal fibrosis [71], because it increases TGF-β expression, which starts a biomolecular cascade driving to renal fibrosis.
On the contrary, PGE2, another AA metabolite, has been shown to inhibit collagen type 1 production and to induce matrix metalloproteinase 1 (MMP1) expression in dermal fibroblasts [72] by binding to the EP-1 receptor on fibroblasts, starting a pathway-regulated ERK1/2 and IP3 signaling that leads to a reduction in collagen expression and an increase in MMP1 expression [72].
In summary, AA and its metabolites play a significant role in the main mechanisms responsible for irreversible renal damage, within a complex network where the final result may be variable, according to genetic- and environment-associated factors. The metabolic balance between mediators of the intermediate metabolism, more than the individual compounds, may dictate the final effects. Moreover, it is still uncertain if the final, relevant pathologic effect, that is, renal fibrosis, could be regulated by dietary or pharmacological measures addressed to directly modify blood AA levels or indirectly modify the activity of AA metabolites.
8. Drug and Gene Interactions
Idiopathic nephrotic syndrome is usually treated with glucocorticoids or with immunosuppressive drugs, particularly calcineurine inhibitors (CNI), such as cyclosporine A (CsA) and tacrolimus (Fk). CNIs are metabolized mainly by cytochrome P450, encoded by the CYP gene cluster. As seen above, the CYP gene is also involved in AA metabolism, but in the literature, there are no reports of enzymatic competition between these drugs and AA.
With regard to the relationship between CNI and AA blood levels, an in vitro study reported that CsA decreases the activity of Delta 9 desaturase and increases the activity of Delta 6 and Delta 5 desaturases [73] through unknown mechanisms. However, as Delta 5 desaturase is involved in the last step of AA biosynthesis [73], CsA therapy could increase AA blood level of patients with INS.
On the same line, it has been suggested that CsA mostly increased the availability of free AA instead of decreasing AA blood levels through the acceleration of AA conversion by the cyclooxygenase pathway [74], but a further in vitro study concluded that CsA had no effect on AA release and metabolism [75]. This result was confirmed more recently in a study of CsA and glucocorticosteroids in human peripheral blood mononuclear cells [76].
With regard to the role of AA metabolism in determining CNI side effects, it is well known that CsA treatment may cause gingival overgrowth, which depends on PGE2 production in gingival fibroblasts. In fact, CsA potentiates TNF-α to stimulate the release of AA from fibroblasts, with consequent enhanced production of PGE2 and gingival overgrowth [77]. There are no studies reporting the same effect in other tissues.
The nephrotoxicity of CsA is well established, and Fk administration is associated with the same side effect, which has been linked to CYP2C8*3 and CYP2C8*4 polymorphisms and a consequent reduction of EETs: it was observed that a circulating Fk plasma concentration of 10 ng/mL is able to reduce the production of eicosanoids by 35%. It follows that CNIs-induced nephrotoxicity could be due to a reduced activity of CYP2C8*3, which reduces the production of EETs, enhancing drug nephrotoxicity [78].
Pre-treatment with Fk is also known to enhance glucocorticoids to inhibit AA and PGE2 production [79] by inhibiting COX2 expression, but the co-administration of Fk and glucocorticoids does not inhibit COX2 expression, allowing for normal PGE2 production [79].
Therefore, the main drugs that are commonly used in INS may have several effects on AA metabolism, which are possibly related to an increased utilization. While contrasting findings have been reported, some of these may account for some drug side effects, like gingival overgrowth and nephrotoxicity, and even therapeutical actions. Although the incorporation of omega 3 as an oil excipient to CsA, to increase bioavailability and decrease nephrotoxicity, has been investigated with interesting results [80,81], no practical therapeutic strategies have been developed so far.
9. Dietary Balance Between AA and LA and AA Sources
AA, which belongs to the omega-6 series, and docosahexaenoic acid (DHA), which belongs to the omega-3 series, are the most important byproducts of essential fatty acids linoleic and α-linolenic acid, and their imbalance has been associated with inflammatory and chronic disorders [82].
While LA and AA are mostly known as inflammatory molecules, operating within an interdependent network through their metabolites [82], AA metabolites also have anti-inflammatory and protective roles, while LA metabolites affect immune function by binding cellular receptors and altering signaling molecules [83].
AA and DHA levels depend on both to genetic predisposition and diet intake. Blood AA levels, as shown in Figure 1, can be modulated through dietary habits, taking into account that there is a marked difference between the amount of AA supplied with the diet and the amount synthesized by human metabolic pathways. In the latter case, the main rate-limiting enzymes are the Δ5- and Δ6-desaturases, which are encoded by the genes FADS1 and FADS2, and different polymorphisms in the fatty acid desaturases genes might even increase or decrease the production of these LC-PUFAs [84]. As a matter of fact, looking at the frequencies of the 28 SNPs in the FADS haplotypes, their distribution in the 3 main haplotypes is evident over the world [85].
Unlike other fatty acids, omega-3 and omega-6 precursors (LA and linolenic acid, respectively) cannot be synthesized de novo by mammals (they are essential dietary compounds indeed), so the relative abundance of these PUFAs in the diet has a major influence in humans.
LA is the most represented omega-6 PUFA in most western diets, and is widely distributed in foods: it represents more than 50% of the lipid content in various vegetable oils, including safflower, sunflower, corn, and soybean oils; it is present in high amounts in nuts and seeds, while lower levels are found in whole grains, legumes, some meats, eggs, and dairy products [86]. Notably, it was recently reported that a strong reduction in dietary intake of LA was not associated with a linear decrease in circulating AA levels [87].
The AA state depends on the endogenous synthesis from the essential precursor LA, undergoing desaturation and elongation, and the direct dietary intake [88]. Since LA to AA conversion efficiency is low in humans, AA intake through the diet appears to be significantly more effective in raising its circulating levels.
In contrast to LA, AA is relatively scarce in the diet and is found in meat (both red and white, including fish), organ meats (e.g., liver, kidney, brain), and eggs, with minimal amounts in cow’s milk fat and the products derived from it [89]. The mandatory origin from animal sources is directly connected to the ability of animals to derive it through enzymatic activities, acting on the vegetable essential precursor, LA. In particular, animal sources are the most representative. Diets rich in meat of beef, lamb, pork, and poultry are claimed to contribute to the high tissue AA content [90]. This amount is influenced by the diet composition, the digestive system, and the biosynthetic processes within the animal [91].
Macroalgae, fungi, bacteria, and yeasts can be a source of essential PUFAs, which may provide humans with fatty acids when included in the diet or used as feed for fish and livestock.
Many fungi, yeast, and some bacteria can synthesize great amounts of LC-PUFAs, mostly AA. The most efficient AA-producer fungus is the non-pathogenic Mortierella spp., in which AA production accounts for up to 70% of total lipids [92].
Among algae, AA has been identified in many groups that grow photoautotrophically or heterotrophically. Certain algal species are reported to contain a naturally higher AA content, which may reach 77% of total fatty acids, as in freshwater green microalga Parietochloris incisa, 40% of total fatty acids in the red alga Porphyridium purpureum, and 20–30% in diatoms such as Phaeodactylum tricornutum and Thalassiosira pseudonona. AA has been detected in lesser amounts in some lichen species (symbiosis association between fungi and algae). Lower plants, such as mosses and ferns, have higher amounts of AA than seagrasses and terrestrial higher plants [92].
Over the last century, dietary intake of n-3 LC-PUFA has decreased, while the dietary content of LA has increased, driven by the use of vegetable products that are rich in LA, and industrialized foods produced with LA-rich vegetable sources [83,93].
From a therapeutic point of view, the achievement of a balance between individual polymorphisms in FADS genes modulation and PUFAs dietary intake could improve or prevent inflammatory conditions, especially in those subjects who can take advantage from an exogenous dietary PUFA supply, due to poorer endogenous synthesis rates [93]. The Mediterranean diet, which is balanced in omega-3 fatty acids from vegetal fats and fish, gets few AA through limited amounts of animal products, and is poor in LA, appears today as an optimal reference for dietary pattern as it concerns the prevention of inflammatory states. Indeed, a working hypothesis to be tested indicates that a reduction of LA amounts in diet, together with a balanced AA intake from natural sources, could be a further way to reduce the inflammatory potential of diets, and in parallel to increase of the supply of n-3 fatty acids.
10. Conclusions
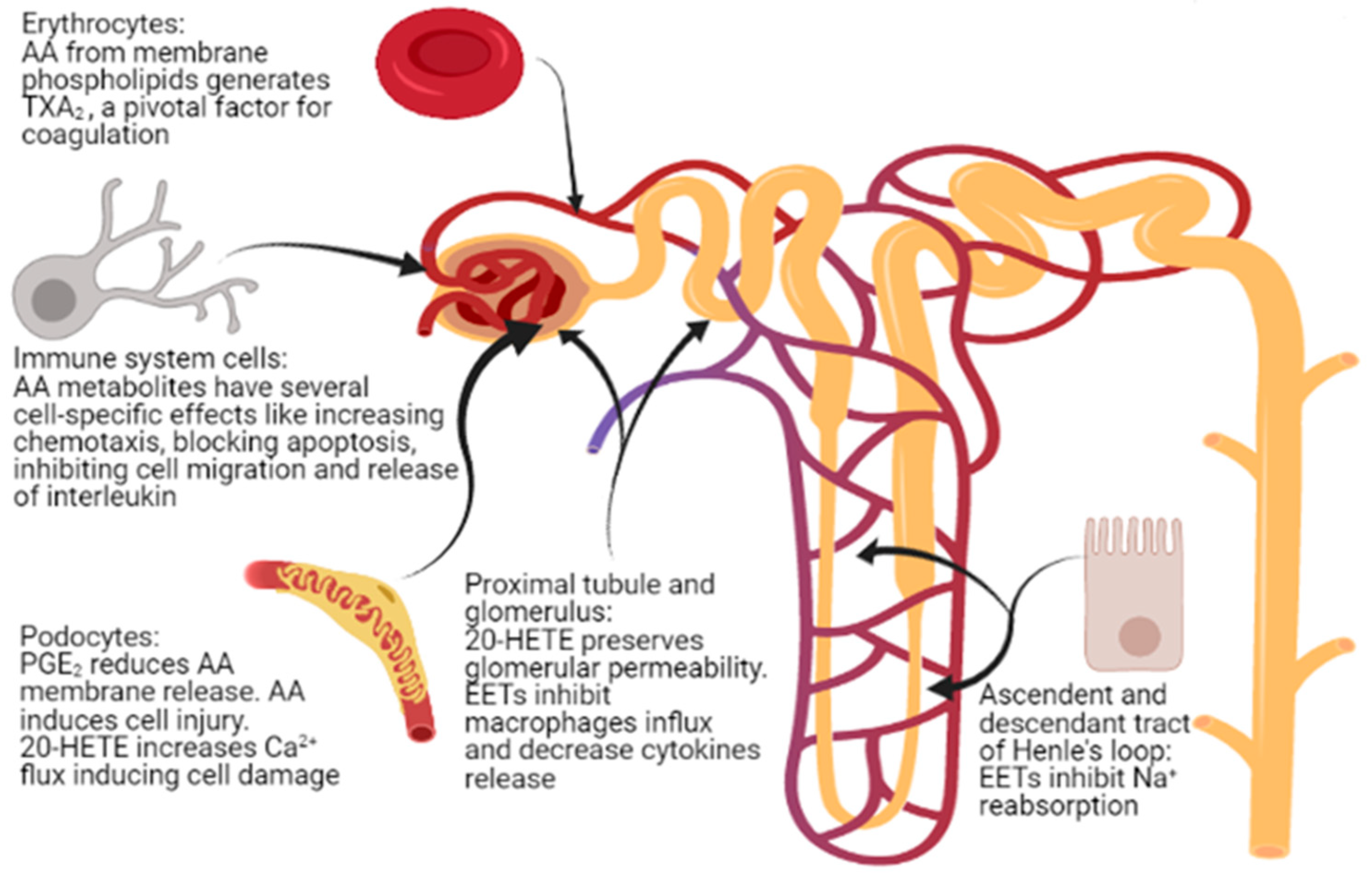
AA and its metabolites play multiple roles, influencing the structure and function of cells such as platelets, lymphocytes, and podocytes (Figure 3), and thus being involved in processes such as coagulation, inflammation, and fibrosis, as well as in maintaining the integrity of the glomerular basement membrane. However, studies concerning the role of AA in kidney disease, including INS, are scarce. Pharmacological and dietary interventions capable of modulating AA and its metabolites are under study, but no clinical trials regarding the role of a diet rich in PUFAs in patients with INS have been published so far. Moreover, a correct dietary balance between AA and LA may represent a further relevant anti-inflammatory measure that should be tested in a controlled context. Accordingly, studies of AA and its metabolites seem an important field to explore, particularly in idiopathic nephrotic syndrome, with possible relevant consequences at the biological, dietary, and pharmacological levels, with the final perspective of obtaining an effective modulation of AA endogenous metabolism, finally counteracting some pathogenetic mechanisms of kidney damage.
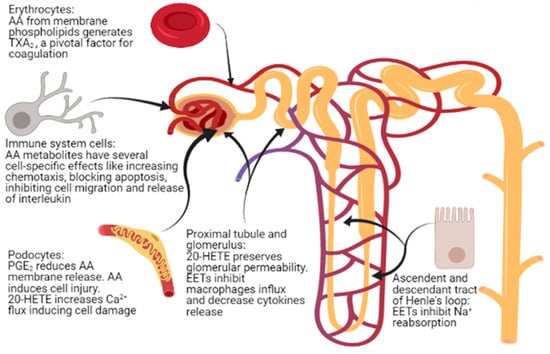
Figure 3.
Sites of action of arachidonic acid (AA) and its metabolites in nephrotic syndrome.
Author Contributions
Writing—original draft preparation, S.T., A.E. and A.M.; writing—review and editing, A.E., M.L.S., W.M., C.A. and G.M.; Supervision, G.M. All authors have read and agreed to the published version of the manuscript.
Funding
The study was supported by a contribution from the Italian Ministry of Health: grant given to a Scientific, Research, and Treatment Institute (IRCCS).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Noone, D.G.; Iijima, K.; Parekh, R. Idiopathic Nephrotic Syndrome in Children. Lancet 2018, 392, 61–74. [Google Scholar] [CrossRef]
- Teoh, C.W.; Robinson, L.A.; Noone, D. Perspectives on edema in childhood nephrotic syndrome. Am. J. Physiol. Ren. Physiol. 2015, 309, F575–F582. [Google Scholar] [CrossRef]
- Dorhout Mees, E.J.; Koomans, H.A. Understanding the nephrotic syndrome: What’s new in a decade? Nephron 1995, 70, 1–10. [Google Scholar] [CrossRef]
- Eneman, B.; Levtchenko, E.; van den Heuvel, B.; Van Geet, C.; Freson, K. Platelet abnormalities in nephrotic syndrome. Pediatr. Nephrol. 2016, 31, 1267–1279. [Google Scholar] [CrossRef] [PubMed]
- Vivarelli, M.; Massella, L.; Ruggiero, B.; Emma, F. Minimal Change Disease. Clin. J. Am. Soc. Nephrol. 2017, 12, 332–345. [Google Scholar] [CrossRef] [PubMed]
- Ranganathan, S. Pathology of Podocytopathies Causing Nephrotic Syndrome in Children. Front. Pediatr. 2016, 4, 32. [Google Scholar] [CrossRef]
- Eddy, A.A.; Symons, J.M. Nephrotic Syndrome in Childhood. Lancet 2003, 362, 629–639. [Google Scholar] [CrossRef]
- Risé, P.; Tragni, E.; Ghezzi, S.; Agostoni, C.; Marangoni, F.; Poli, A.; Catapano, A.L.; Siani, A.; Iacoviello, L.; Galli, C. Different patterns characterize Omega 6 and Omega 3 long chain polyunsaturated fatty acid levels in blood from Italian infants, children, adults and elderly. Prostagland. Leukot. Essent. Fat. Acids 2013, 89, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Cunnnane, S.C.; Kent, E.T.; McAdoo, K.R.; Caldwell, D.; Lin, A.N.; Carter, D.M. Abnormalities of Plasma and Erythrocyte Essential Fatty Acid Composition in Epidermolysis Bullosa: Influence of Treatment with Diphenylhydantoin. J. Investig. Dermatol. 1987, 89, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Strandvik, B. Fatty acid metabolism in cystic fibrosis. Prostagland. Leukot. Essent. Fat. Acids 2010, 83, 121–129. [Google Scholar] [CrossRef]
- Cheng, S.; Chen, Y.L. Study on erythrocyte membrane fluidity by laser Raman spectroscopy. Cell Biol. Int. Rep. 1988, 12, 205–211. [Google Scholar] [PubMed]
- Pompéia, C.; Lopes, L.R.; Miyasaka, C.K.; Procópio, J.; Sannomiya, P.; Curi, R. Effect of fatty acids on leukocyte function. Braz. J. Med. Biol. Res. 2000, 33, 1255–1268. [Google Scholar] [CrossRef]
- Brash, A.R. Arachidonic acid as a bioactive molecule. J. Clin. Investig. 2001, 107, 1339–1345. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, V.H.; Nascimento, K.S.; Freire, M.M.; Moreira, O.C.; Scofano, H.M.; Barrabin, H.; Mignaco, J.A. Mechanism of modulation of the plasma membrane Ca(2+)-ATPase by arachidonic acid. Prostagland. Other Lipid Mediat. 2008, 87, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Varga-Szabo, D.; Braun, A.; Nieswandt, B. Calcium signaling in platelets. J. Thromb. Haemost. 2009, 7, 1057–1066. [Google Scholar] [CrossRef]
- Meves, H. Arachidonic acid and ion channels: An update. Br. J. Pharmacol. 2008, 155, 4–16. [Google Scholar] [CrossRef]
- Hwang, S.W.; Cho, H.; Kwak, J.; Lee, S.Y.; Kang, C.J.; Jung, J.; Cho, S.; Min, K.H.; Suh, Y.G.; Kim, D.; et al. Direct activation of capsaicin receptors by products of lipoxygenases: Endogenous capsaicin-like substances. Proc. Natl. Acad. Sci. USA 2000, 97, 6155–6160. [Google Scholar] [CrossRef]
- Van der Vusse, G.J. Albumin as fatty acid transporter. Drug Metab. Pharmacokinet. 2009, 24, 300–307. [Google Scholar] [CrossRef]
- Beck, R.; Bertolino, S.; Abbot, S.E.; Aaronson, P.I.; Smirnov, S.V. Modulation of arachidonic acid release and membrane fluidity by albumin in vascular smooth muscle and endothelial cells. Circ. Res. 1998, 83, 923–931. [Google Scholar] [CrossRef]
- Nelson, G.J.; Schmidt, P.C.; Bartolini, G.; Kelley, D.S.; Kyle, D. The effect of dietary arachidonic acid on platelet function, platelet fatty acid composition, and blood coagulation in humans. Lipids 1997, 32, 421–425. [Google Scholar] [CrossRef]
- Trostchansky, A.; Moore-Carrasco, R.; Fuentes, E. Oxydative pathways of arachidonic acid as targets for regulation of platelet activation. Prostaglandines Other Lipid Mediat. 2019, 15, 106382. [Google Scholar] [CrossRef]
- Offermanns, S. Activation of platelet function through G protein-coupled receptors. Circ. Res. 2006, 99, 1293–1304. [Google Scholar] [CrossRef]
- Rasedee, A.; Feldman, B.F. Nephrotic syndrome: A platelet hyperaggregability state. Vet. Res. Commun. 1985, 9, 199–211. [Google Scholar] [CrossRef]
- Davì, G.; Averna, M.; Catalano, I.; Barbagallo, C.; Ganci, A.; Notarbartolo, A.; Ciabattoni, G.; Patrono, C. Increased thromboxane biosynthesis in type IIa hypercholesterolemia. Circulation 1992, 85, 1792–1798. [Google Scholar] [CrossRef] [PubMed]
- Colucci, M.; Carsetti, R.; Cascioli, S.; Serafinelli, J.; Emma, F.; Vivarelli, M. B cell phenotype in pediatric idiopathic nephrotic syndrome. Pediatr. Nephrol. 2019, 34, 177–181. [Google Scholar] [CrossRef]
- Chebotareva, N.; Bobkova, I.; Lysenko, L. T regulatory cells in renal tissue of patients with nephrotic syndrome. Pediatr. Int. 2020, 62, 884–885. [Google Scholar] [CrossRef] [PubMed]
- Chambers, E.T.; Gbadegesin, R.A. Aberrant IgM on T cells: Biomarker or pathogenic factor in childhood nephrotic syndrome? Kidney Int. 2019, 96, 818–820. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, F.T.L.; Melo, G.E.B.A.; Cordeiro, T.M.; Feracin, V.; Vieira, E.R.; Pereira, W.F.; Pinheiro, S.V.B.; Miranda, A.S.; Simões-E-Silva, A.C. T-lymphocyte-expressing inflammatory cytokines underlie persistence of proteinuria in children with idiopathic nephrotic syndrome. J. Pediatr. 2018, 94, 546–553. [Google Scholar] [CrossRef]
- Alsharidah, A.S.; Alzogaibi, M.A.; Bayoumy, N.M.; Alghonaim, M. Neutrophil chemokines levels in different stages of nephrotic syndrome. Saudi J. Kidney Dis. Transplant. 2017, 28, 1256–1263. [Google Scholar] [CrossRef]
- Calder, P.C. The relationship between the fatty acid composition of immune cells and their function. Prostagland. Leukot. Essent. Fat. Acids 2008, 79, 101–108. [Google Scholar] [CrossRef]
- Triggiani, M.; Oriente, A.; de Crescenzo, G.; Rossi, G.; Marone, G. Biochemical functions of a pool of arachidonic acid associated with triglycerides in human inflammatory cells. Int. Arch. Allergy Immunol. 1995, 107, 261–263. [Google Scholar] [CrossRef]
- Wei, J.; Gronert, K. Eicosanoid and specialized preresolving mediator regulation of lymphoid cells. Trends Biochem. Sci. 2019, 44, 214–225. [Google Scholar] [CrossRef]
- Hanna, V.S.; Hafez, E.A. Synopsis of arachidonic acid metabolism—A review. J. Adv. Res. 2018, 11, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Fu, X.; Chen, Q.; Part, J.K.; Wang, D.; Wang, Z.; Gai, Z. Arachidonic acid metabolism and kidney inflammation. Int. J. Mol. Sci. 2019, 20, 3683. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.L. The eicosanoids and their biochemical mechanisms of action. Biochem. J. 1989, 259, 315–324. [Google Scholar] [CrossRef]
- Legler, D.F.; Bruckner, M.; Uetz-von Allmen, E.; Krause, P. Prostaglandin E2 at new glance: Novel insights in functional diversity offer therapeutic chances. Int. J. Biochem. Cell Biol. 2010, 42, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Harizi, H.; Gualde, N. The impact of eicosanoids on the crosstalk between innate and adaptive immunity: The key roles of dendritic cells. Tissue Antigens 2005, 65, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Nataraj, C.; Thomas, D.W.; Tilley, S.L.; Nguyen, M.T.; Mannon, R.; Koller, B.H.; Coffman, T.M. Receptors for prostaglandin E(2) that regulate cellular immune responses in the mouse. J. Clin. Investig. 2001, 108, 1229–1235. [Google Scholar] [CrossRef]
- Roper, R.L.; Phipps, R.P. Prostaglandin E2 and cAMP inhibit B lymphocyte activation and simultaneously promote IgE and IgG1 synthesis. J. Immunol. 1992, 149, 2984–2991. [Google Scholar]
- Goodarzi, K.; Goodarzi, M.; Tager, A.M.; Luster, A.D.; von Andrian, U.H. Leukotriene B4 and BLT1 control cytotoxic effector T cell recruitment to inflamed tissues. Nat. Immunol. 2003, 4, 965–973. [Google Scholar] [CrossRef]
- Yang, L.; Mäki-Petäjä, K.; Cheriyan, J.; McEniery, C.; Wilkinson, I.B. The role of epoxyeicosatrienoic acids in the cardiovascular system. Br. J. Clin. Pharmacol. 2015, 80, 28–44. [Google Scholar] [CrossRef] [PubMed]
- Fang, Q.; Chen, G.Z.; Wang, Y.; Wang, D.W. Role of cytochrome P450 epoxygenase-dependent arachidonic acid metabolites in kidney physiology and diseases. Sheng Li Xue Bao 2018, 70, 591–599. [Google Scholar] [PubMed]
- Wang, D.; DuBois, R.N. Epoxyeicosatrienoic acids: A double-edged sword in cardiovascular diseases and cancer. J. Clin. Investig. 2012, 122, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Fleming, I. Vascular cytochrome p450 enzymes: Physiology and pathophysiology. Trends Cardiovasc. Med. 2008, 18, 20–25. [Google Scholar] [CrossRef]
- Kim, J.; Yoon, S.P.; Toews, M.L.; Imig, J.D.; Hwang, S.H.; Hammock, B.D.; Padanilam, B.J. Pharmacological inhibition of soluble epoxide hydrolase prevents renal interstitial fibrogenesis in obstructive nephropathy. Am. J. Physiol. Ren. Physiol. 2015, 308, F131–F139. [Google Scholar] [CrossRef] [PubMed]
- Imig, J.D.; Hammock, B.D. Soluble epoxide hydrolase as a therapeutic target for cardiovascular diseases. Nat. Rev. Drug Discov. 2009, 8, 794–805. [Google Scholar] [CrossRef]
- Carroll, M.A.; Cheng, M.K.; Liclican, E.L.; Li, J.; Doumad, A.B.; McGiff, J.C. Purinoceptors in renal microvessels: Adenosine-activated and cytochrome P450 monooxygenase-derived arachidonate metabolites. Pharmacol. Rep. 2005, 57, 191–195. [Google Scholar]
- Williams, J.M.; Sharma, M.; Anjaiahh, S.; Falck, J.R.; Roman, R.J. Role of endogenous CYP450 metabolites of arachidonic acid in maintaining the glomerular protein permeability barrier. Am. J. Physiol. Ren. Physiol. 2007, 293, F501–F505. [Google Scholar] [CrossRef]
- Fan, F.; Roman, R.J. Effect of Cytochrome P450 Metabolites of Arachidonic Acid in Nephrology. J. Am. Soc. Nephrol. 2017, 28, 2845–2855. [Google Scholar] [CrossRef]
- Nowicki, S.; Chen, S.L.; Aizman, O.; Cheng, X.J.; Li, D.; Nowicki, C.; Nairn, A.; Greengard, P.; Aperia, A. 20-Hydroxyeicosa-tetraenoic acid (20 HETE) activates protein kinase C. Role in regulation of rat renal Na+,K+-ATPase. J. Clin. Investig. 1997, 99, 1224–1230. [Google Scholar] [CrossRef]
- Roshanravan, H.; Kim, E.Y.; Dryer, S.E. 20-Hydroxyeicosatetraenoic Acid (20-HETE) Modulates in Podocytes. Front. Physiol. 2016, 7, 351. [Google Scholar] [CrossRef]
- Shatat, I.F.; Becton, L.J.; Woroniecki, R.P. Hypertension in childhood nephrotic syndrome. Front. Pediatr. 2019, 7, 287. [Google Scholar] [CrossRef]
- Quigley, R.; Baum, M.; Reddy, K.M.; Griener, J.C.; Falck, J.R. Effects of 20-HETE and 19(s)-HETE on rabbit proximal straight tubule volume transport. Am. J. Physiol. Ren. Physiol. 2000, 278, 949–953. [Google Scholar] [CrossRef] [PubMed]
- Osama, H.E.; Sheriff, M.S.; Anwar, M.; Ayman, O.S. Clinical implication of 20-hydroeicosatetraenoic acid in the kidney, liver, lung and brain. An emerging therapeutic target. Pharmaceutics 2017, 9, 9. [Google Scholar]
- Han, H.; Wang, S.; Liang, Y.; Lin, J.; Shi, L.; Ye, L.; Song, S.; He, M.; Li, S.; Chen, F.; et al. Respiratory tract infection: A risk factor for the onset and relapse of adult onset minimal changes disease in Southern China. BioMed Res. Int. 2018, 2018, 1657208. [Google Scholar] [CrossRef]
- Mishra, O.P.; Abhinay, A.; Mishra, R.N.; Prasad, R.; Pohl, M. Can we predict relapses in children with idiopathic steroid-sensitive nephrotic syndrome? J. Trop. Pediatr. 2013, 59, 343–349. [Google Scholar] [CrossRef]
- Manta, M.; Singh, S. Infection associated relapses in children with nephrotic syndrome: A short-term outcome study. Saudi J. Kidney Dis. Transplant. 2019, 30, 1245–1253. [Google Scholar] [CrossRef]
- Ha, V.T.; Lainšček, D.; Gesslbauer, B.; Jarc-Jovičić, E.; Hyötyläinen, T.; Ilc, N.; Lakota, K.; Tomšič, M.; van de Loo, F.A.J.; Bochkov, V.; et al. Synergy between 15-lipoxygenase and secreted PLA2 promotes inflammation by formation of TLR4 agonists from extracellular vesicles. Proc. Natl. Acad. Sci. USA 2020, 117, 25679–25689. [Google Scholar] [CrossRef] [PubMed]
- Yiu, S.S.; Zhao, X.; Inscho, E.W.; Imig, J.D. 12-Hydroxyeicosatetraenoic acid participates in angiotensin II afferent arteriolar vasoconstriction by activating L-type calcium channels. J. Lipid Res. 2003, 44, 2391–2399. [Google Scholar] [CrossRef] [PubMed]
- Andreani, M.; Olivier, J.L.; Berenbaum, F.; Raymondjean, M.; Béréziat, G. Transcriptional regulation of inflammatory secreted phospholipases A(2). Biochim. Biophys. Acta 2000, 1488, 149–158. [Google Scholar] [CrossRef]
- Ren, X.; Zhang, M.; Chen, L.; Zhang, W.; Huang, Y.; Luo, H.; Li, L.; He, H. The anti-inflammatory effects of Yunnan Baiyao are involved in regulation of the phospholipase A2/arachidonic acid metabolites pathways in acute inflammation rat model. Mol. Med. Rep. 2017, 16, 4045–4053. [Google Scholar] [CrossRef] [PubMed]
- Lemieux, L.I.; Rahal, S.S.; Kennedy, C.R. PGE2 reduces arachidonic acid release in murine podocytes: Evidence for an autocrine feedback loop. Am. J. Physiol. Cell Physiol. 2003, 284, C302–C309. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Pan, Y.; Wu, Y.; Lin, S.; Dai, B.; Chen, H.; Wan, J. Excessive arachidonic acid induced actin bunching remodeling and podocyte injury via a PKA-c-Abl dependent pathway. Exp. Cell Res. 2020, 388, 111808. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Wan, J.; Liu, Y.; Yang, Q.; Liang, W.; Singhal, P.C.; Saleem, M.A.; Ding, G. sPLA2 IB induces human podocyte apoptosis via the M-type phospholipase A2 receptor. Sci. Rep. 2014, 4, 6660. [Google Scholar] [CrossRef]
- Djudjaj, S.; Boor, P. Cellular and molecular mechanisms of kidney fibrosis. Mol. Asp. Med. 2019, 65, 16–36. [Google Scholar] [CrossRef]
- Duffield, J.S. Cellular and molecular mechanisms in kidney fibrosis. J. Clin. Investig. 2014, 124, 2299–2306. [Google Scholar] [CrossRef]
- Priante, G.; Musacchio, E.; Valvason, C.; Baggio, B. EPA and DHA suppress AngII- and arachidonic acid-induced expression of profibrotic genes in human mesangial cells. J. Nephrol. 2009, 22, 137–143. [Google Scholar]
- Zimpelmann, J.; Burns, K.D. Angiotensin-(1-7) activates growth-stimulatory pathways in human mesangial cells. Am. J. Physiol. Ren. Physiol. 2009, 296, F337–F346. [Google Scholar] [CrossRef]
- Zhang, C.; Booz, G.W.; Yu, Q.; He, X.; Wang, S.; Fan, F. Conflicting roles of 20-HETE in hypertension and renal end organ damage. Eur. J. Pharmacol. 2018, 833, 190–200. [Google Scholar] [CrossRef]
- Cheng, J.; Garcia, V.; Ding, Y.; Wu, C.C.; Thakar, K.; Falck, J.R.; Ramu, E.; Schwartzman, M.L. Induction of angiotensin-converting enzyme and activation of the renin-angiotensin system contribute to 20-hydroxyeicosatetraenoic acid-mediated endothelial dysfunction. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1917–1924. [Google Scholar] [CrossRef]
- Balakumar, P.; Sambathkumar, R.; Mahadevan, N.; Muhsinah, A.B.; Alsayari, A.; Venkateswaramurthy, N.; Jagadeesh, G. A potential role of the renin-angiotensin-aldosterone system in epithelial-to-mesenchymal transition-induced renal abnormalities: Mechanisms and therapeutic implications. Pharmacol. Res. 2019, 146, 104314. [Google Scholar] [CrossRef] [PubMed]
- Shim, J.H. Prostaglandin E2 Induces Skin Aging via E-Prostanoid 1 in Normal Human Dermal Fibroblasts. Int. J. Mol. Sci. 2019, 20, 5555. [Google Scholar] [CrossRef] [PubMed]
- Lausada, N.; de Gómez Dumm, I.N.; Raimondi, J.C.; de Alaniz, M.J. Effect of cyclosporine and sirolimus on fatty acid desaturase activities in cultured HEPG2 cells. Transplant. Proc. 2009, 41, 1865–1870. [Google Scholar] [CrossRef] [PubMed]
- Whisler, R.L.; Lindsey, J.A.; Proctor, K.V.; Morisaki, N.; Cornwell, D.G. Characteristics of cyclosporine induction of increased prostaglandin levels from human peripheral blood monocytes. Transplantation 1984, 38, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, O.H.; Bukhave, K.; Ahnfelt-Rønne, I.; Elmgreen, J. Arachidonic acid metabolism in human neutrophils: Lack of effect of cyclosporine A. Int. J. Immunopharmacol. 1986, 8, 419–426. [Google Scholar] [CrossRef]
- Sipka, S.; Szücs, K.; Szántó, S.; Kovács, I.; Lakos, G.; Antal-Szalmás, P.; Szegedi, G.; Gergely, P. Inhibition of calcineurin activity and protection against cyclosporine A induced cytotoxicity by prednisolone sodium succinate in human peripheral mononuclear cells. Immunopharmacology 2000, 48, 87–92. [Google Scholar] [CrossRef]
- Wondimu, B.; Modéer, T. Cyclosporin A upregulates prostaglandin E2 production in human gingival fibroblasts challenged with tumor necrosis factor alpha in vitro. J. Oral Pathol. Med. 1997, 26, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Smith, H.E.; Jones, J.P., 3rd; Kalhorn, T.F.; Farin, F.M.; Stapleton, P.L.; Davis, C.L.; Perkins, J.D.; Blough, D.K.; Hebert, M.F.; Thummel, K.E.; et al. Role of cytochrome P450 2C8 and 2J2 genotypes in calcineurin inhibitor-induced chronic kidney disease. Pharmacogenet. Genom. 2008, 18, 943–953. [Google Scholar] [CrossRef]
- Croxtall, J.D.; Paul-Clark, M.; Van Hal, P.T. Differential modulation of glucocorticoid action by FK506 in A549 cells. Biochem. J. 2003, 376 Pt 1, 285–290. [Google Scholar] [CrossRef]
- Mariee, A.D.; Abd-Ellah, M.F. Protective effect of docosahexaenoic acid against cyclosporine A-induced nephrotoxicity in rats: A possible mechanism of action. Ren. Fail. 2011, 33, 66–71. [Google Scholar] [CrossRef]
- Hirunpanich, V.; Sato, H. Improvement of cyclosporine A bioavailability by incorporating ethyl docosahexaenoate in the microemulsion as an oil excipient. Eur. J. Pharm. Biopharm. 2009, 73, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Mazzocchi, A.; Agostoni, C. Long-Chain ω-3 Polyunsaturated Fatty Acids: Do Genetic Steps Match Metabolic Needs? J. Nutr. 2019, 149, 1690–1691. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. Evolutionary aspects of diet: The omega-6/omega-3 ratio and the brain. Mol. Neurobiol. 2011, 44, 203–215. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.T.; Nara, T.Y. Structure, function, and dietary regulation of delta6, delta5, and delta9 desaturases. Annu. Rev. Nutr. 2004, 24, 345–376. [Google Scholar] [CrossRef]
- Ameur, S.; Enroth, S.; Johansson, A.; Zaboli, G.; Igl, W.; Johansson, A.C.V.; Rivas, M.A.; Daly, M.J.; Schmitz, G.; Hicks, A.A.; et al. Genetic adaptation of fatty acid metabolism: A human specific haplotype increasing the biosyntesis of long-chain omega-3 and omega -6 fatty acid. Am. J. Hum. Genet. 2012, 90, 809–820. [Google Scholar] [CrossRef]
- Marangoni, F.; Agostoni, C.; Borghi, C.; Catapano, A.L.; Cena, H.; Ghiselli, A.; La Vecchia, C.; Lercker, G.; Manzato, E.; Pirillo, A.; et al. Dietary linoleic acid and human health: Focus on cardiovascular and cardiometabolic effects. Atherosclerosis 2020, 292, 90–98. [Google Scholar] [CrossRef]
- Ramsden, C.E.; Zamora, D.; Makriyannis, A.; Wood, J.T.; Mann, J.D.; Faurot, K.R.; MacIntosh, B.A.; Majchrzak-Hong, S.F.; Gross, J.R.; Courville, A.B.; et al. Diet-induced changes in n-3- and n-6-derived endocannabinoids and reductions in headache pain and psychological distress. J. Pain 2015, 16, 707–716. [Google Scholar] [CrossRef]
- Forsyth, S.; Gautier, S.; Salem, N., Jr. Global Estimates of Dietary Intake of Docosahexaenoic Acid and Arachidonic Acid in Developing and Developed Countries. Ann. Nutr. Metab. 2016, 68, 258–267. [Google Scholar] [CrossRef]
- US National Institutes of Health. Food Sources of Arachidonic Acid; US National Institutes of Health: Bethesda, MD, USA, 2005.
- Li, D.; Ng, A.; Mann, N.J.; Sinclair, A.J. Contribution of meat fat to dietary arachidonic acid. Lipids 1998, 33, 437–440. [Google Scholar] [CrossRef]
- Abedi, E.; Sahari, M.A. Long-chain polyunsaturated fatty acid sources and evaluation of their nutritional and functional properties. Food Sci. Nutr. 2014, 2, 443–463. [Google Scholar] [CrossRef]
- Shanab, S.M.M.; Hafez, R.M.; Fouad, A.S. A review on algae and plants as potential source of arachidonic acid. J. Adv. Res. 2018, 11, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp. Biol. Med. 2008, 233, 674–688. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).