S-Palmitoylation of Synaptic Proteins as a Novel Mechanism Underlying Sex-Dependent Differences in Neuronal Plasticity
Abstract
:1. Introduction
2. Results
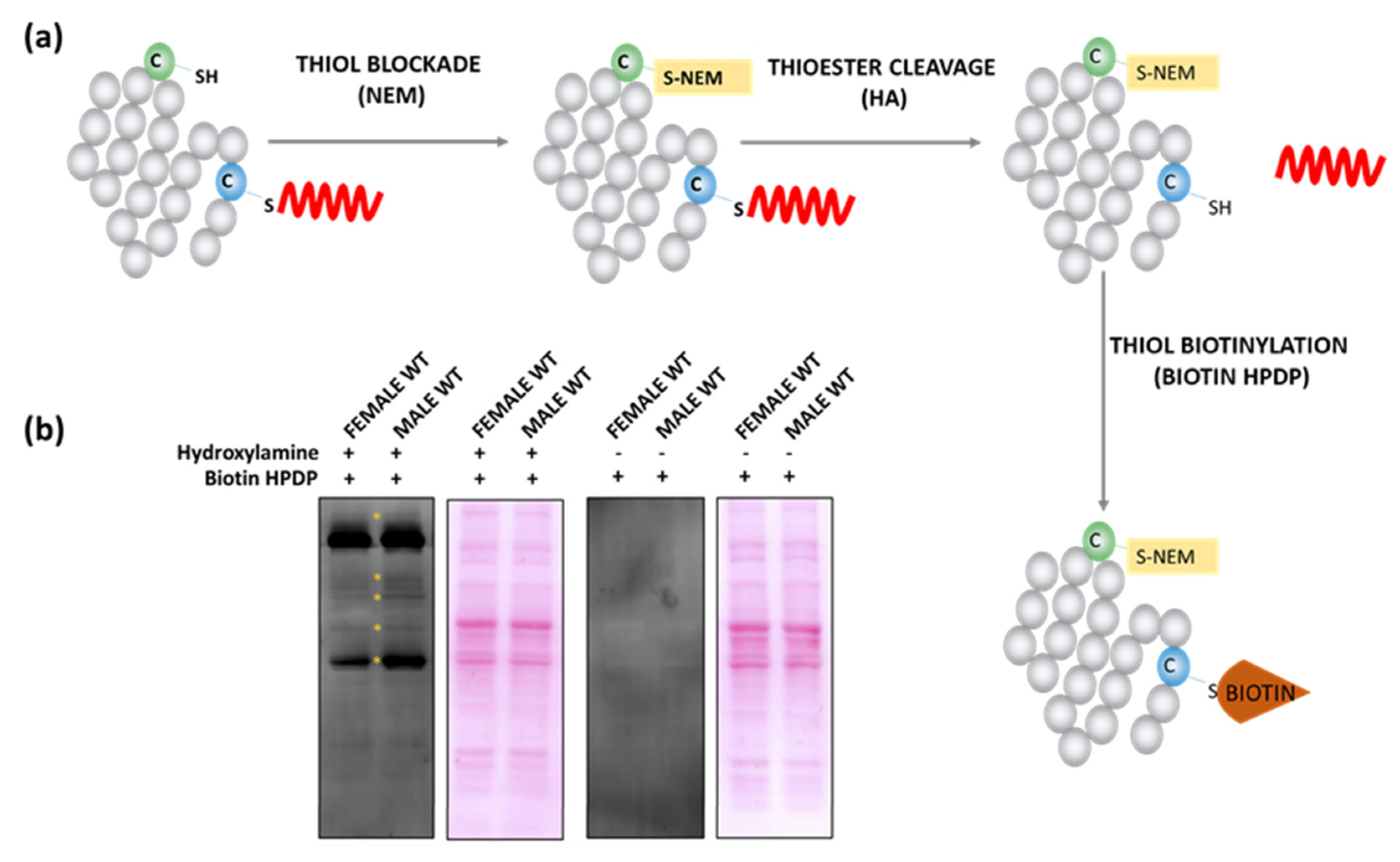
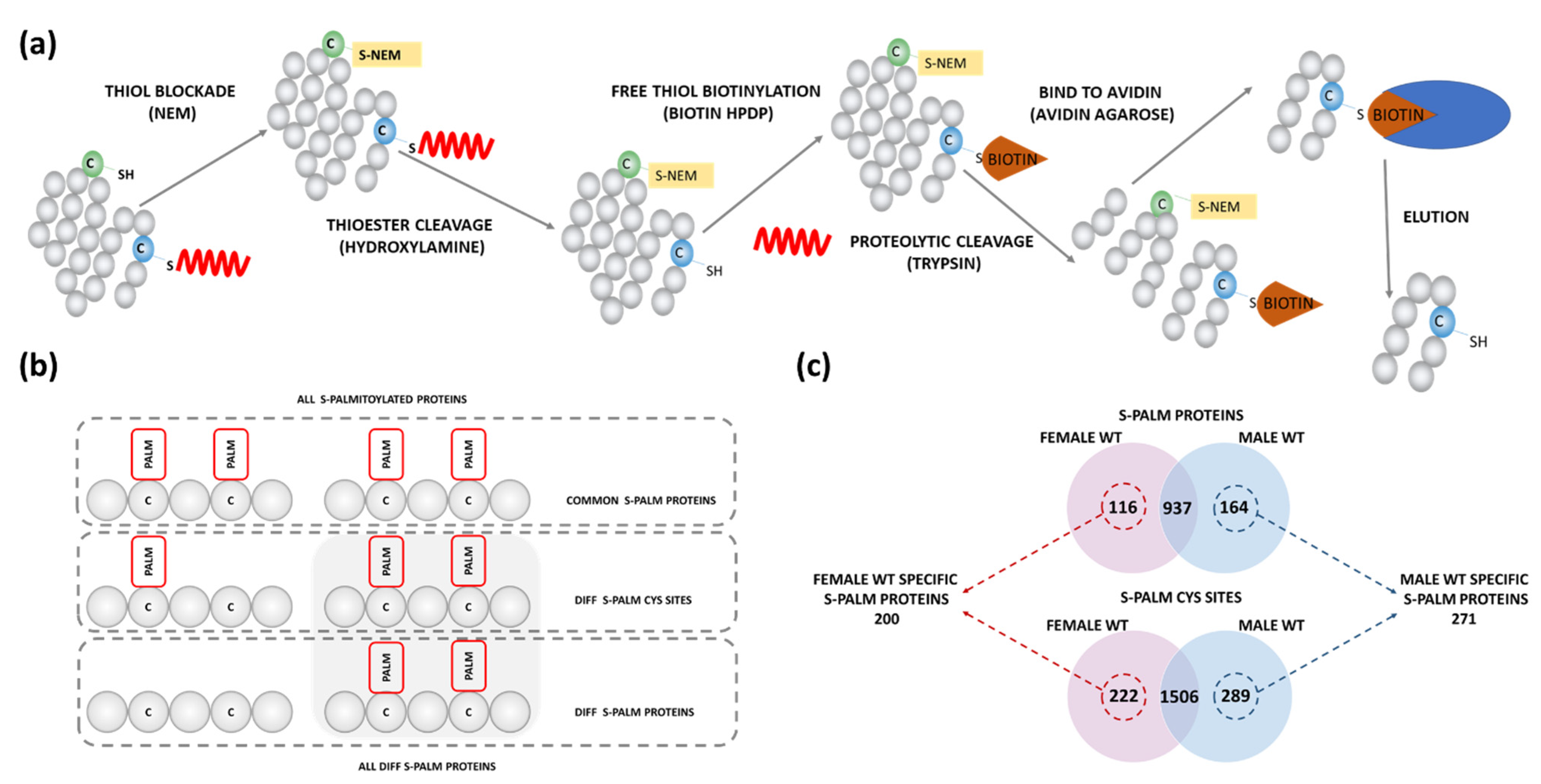
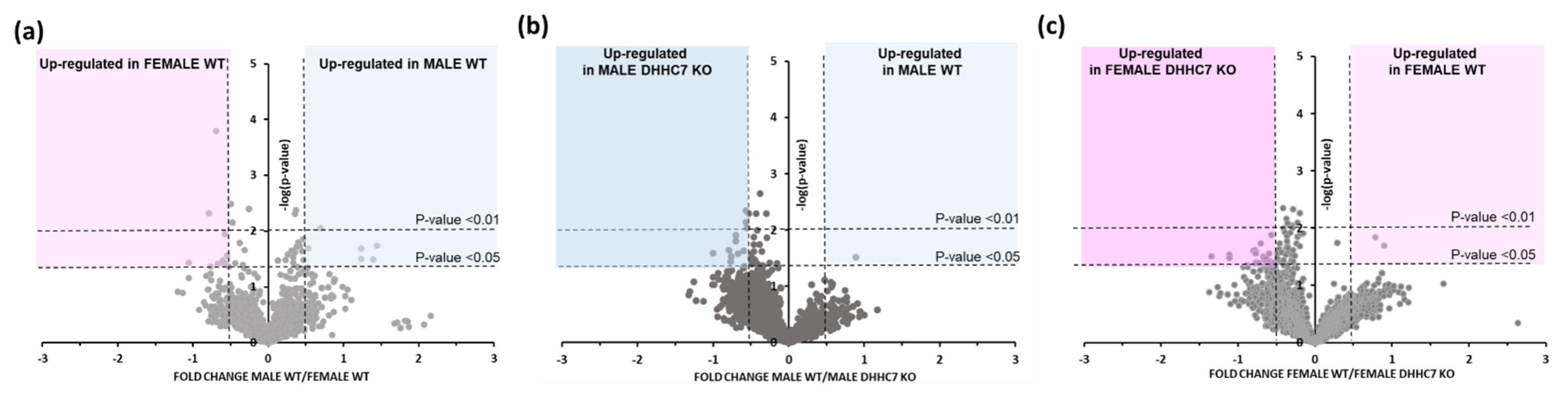
2.1. Sex-Dependent Differences in S-Palmitoylation of Synaptic Proteins
2.2. DHHC7–Dependent Synaptic Proteins S-Palmitoylation in Male and Female Mice
2.3. DHHC7 Operates Differently in Male and Female Mice
3. Discussion
4. Materials and Methods
4.1. Animals and Ethical Statement
4.2. Synaptoneurosomes
4.3. Acyl-Biotin Exchange (ABE)
4.4. PANIMoni
4.5. Mass Spectrometry
4.6. Functional Bioinformatics Analysis
4.7. Semantic Similarity (SS) Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tronson, N.C. Focus on females: A less biased approach for studying strategies and mechanisms of memory. Curr. Opin. Behav. Sci. 2018, 23, 92–97. [Google Scholar] [CrossRef]
- Beery, A.; Zucker, I. Sex bias in neuroscience and biomedical research. Neurosci. Biobehav. Rev. 2011, 35, 565–572. [Google Scholar] [CrossRef] [Green Version]
- Mizuno, K.; Antunes-Martins, A.; Ris, L.; Peters, M.; Godaux, E.; Giese, K.P. Calcium/calmodulin kinase kinase beta has a male-specific role in memory formation. Neuroscience 2007, 145, 393–402. [Google Scholar] [CrossRef]
- Waters, E.M.; Thompson, L.I.; Patel, P.; Gonzales, A.D.; Ye, H.Z.; Filardo, E.J.; Clegg, D.J.; Gorecka, J.; Akama, K.T.; McEwen, B.S.; et al. G-Protein-Coupled Estrogen Receptor 1 Is Anatomically Positioned to Modulate Synaptic Plasticity in the Mouse Hippocampus. J. Neurosci. 2015, 35, 2384–2397. [Google Scholar] [CrossRef] [PubMed]
- Nuñez, J.L.; McCarthy, M.M. Resting intracellular calcium concentration, depolarizing Gamma-Aminobutyric Acid and possible role of local estradiol synthesis in the developing male and female hippocampus. Neuroscience 2009, 158, 623–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monfort, P.; Gómez-Giménez, B.; Llansola, M.; Felipo, V. Gender Differences in Spatial Learning, Synaptic Activity, and Long-Term Potentiation in the Hippocampus in Rats: Molecular Mechanisms. ACS Chem. Neurosci. 2015, 6, 1420–1427. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Le, A.A.; Hou, B.; Lauterborn, J.C.; Cox, C.D.; Levin, E.R.; Lynch, G.; Gall, C.M. Memory-Related Synaptic Plasticity Is Sexually Dimorphic in Rodent Hippocampus. J. Neurosci. 2018, 38, 7935–7951. [Google Scholar] [CrossRef] [Green Version]
- Andreano, J.M.; Cahill, L. Sex influences on the neurobiology of learning and memory. Learn. Mem. 2009, 16, 248–266. [Google Scholar] [CrossRef] [Green Version]
- Hamann, S. Sex Differences in the Responses of the Human Amygdala. Neuroscience 2005, 11, 288–293. [Google Scholar] [CrossRef]
- Wickens, M.M.; Bangasser, D.A.; Briand, L.A. Sex Differences in Psychiatric Disease: A Focus on the Glutamate System. Front. Mol. Neurosci. 2018, 11, 197. [Google Scholar] [CrossRef]
- Altemus, M.; Sarvaiya, N.; Epperson, C.N. Sex differences in anxiety and depression clinical perspectives. Front. Neuroendocr. 2014, 35, 320–330. [Google Scholar] [CrossRef] [Green Version]
- Ecker, C.; Andrews, D.S.; Gudbrandsen, C.M.; Marquand, A.F.; Ginestet, C.E.; Daly, E.M.; Murphy, C.M.; Lai, M.-C.; Lombardo, M.V.; Ruigrok, A.N.V.; et al. Association Between the Probability of Autism Spectrum Disorder and Normative Sex-Related Phenotypic Diversity in Brain Structure. JAMA Psychiatry 2017, 74, 329–338. [Google Scholar] [CrossRef]
- Mendrek, A.; Mancini-Marïe, A. Sex/gender differences in the brain and cognition in schizophrenia. Neurosci. Biobehav. Rev. 2016, 67, 57–78. [Google Scholar] [CrossRef]
- Sloan, D.M.; Kornstein, S.G. Gender differences in depression and response to antidepressant treatment. Psychiatr. Clin. N. Am. 2003, 26, 581–594. [Google Scholar] [CrossRef]
- Vierk, R.; Bayer, J.; Freitag, S.; Muhia, M.; Kutsche, K.; Wolbers, T.; Kneussel, M.; Sommer, T.; Rune, G. Structure–function–behavior relationship in estrogen-induced synaptic plasticity. Horm. Behav. 2015, 74, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, K.; Ris, L.; Sánchez-Capelo, A.; Godaux, E.; Giese, K.P. Ca2+/Calmodulin Kinase Kinase α Is Dispensable for Brain Development but Is Required for Distinct Memories in Male, though Not in Female, Mice. Mol. Cell. Biol. 2006, 26, 9094–9104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dachtler, J.; Hardingham, N.R.; Fox, K. The Role of Nitric Oxide Synthase in Cortical Plasticity Is Sex Specific. J. Neurosci. 2012, 32, 14994–14999. [Google Scholar] [CrossRef] [Green Version]
- Zaręba-Kozioł, M.; Figiel, I.; Bartkowiak-Kaczmarek, A.; Włodarczyk, J. Insights Into Protein S-Palmitoylation in Synaptic Plasticity and Neurological Disorders: Potential and Limitations of Methods for Detection and Analysis. Front. Mol. Neurosci. 2018, 11, 175. [Google Scholar] [CrossRef] [PubMed]
- Fukata, Y.; Fukata, M. Protein palmitoylation in neuronal development and synaptic plasticity. Nat. Rev. Neurosci. 2010, 11, 161–175. [Google Scholar] [CrossRef]
- Naumenko, V.S.; Ponimaskin, E. Palmitoylation as a Functional Regulator of Neurotransmitter Receptors. Neural Plast. 2018, 2018, 5701348. [Google Scholar] [CrossRef] [Green Version]
- Kang, R.; Wan, J.; Arstikaitis, P.; Takahashi, H.; Huang, K.; Bailey, A.O.; Thompson, J.X.; Roth, A.F.; Drisdel, R.C.; Mastro, R.; et al. Neural palmitoyl-proteomics reveals dynamic synaptic palmitoylation. Nat. Cell Biol. 2008, 456, 904–909. [Google Scholar] [CrossRef] [Green Version]
- Prescott, G.R.; Gorleku, O.A.; Greaves, J.; Chamberlain, L.H. Palmitoylation of the synaptic vesicle fusion machinery. J. Neurochem. 2009, 110, 1135–1149. [Google Scholar] [CrossRef]
- Pedram, A.; Razandi, M.; Deschenes, R.J.; Levin, E.R. DHHC-7 and -21 are palmitoylacyltransferases for sex steroid receptors. Mol. Biol. Cell 2012, 23, 188–199. [Google Scholar] [CrossRef]
- Balthazart, J.; Ball, G.F. Is brain estradiol a hormone or a neurotransmitter? Trends Neurosci. 2006, 29, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Baudry, M.; Bi, X.; Aguirre, C. Progesterone–estrogen interactions in synaptic plasticity and neuroprotection. Neuroscience 2013, 239, 280–294. [Google Scholar] [CrossRef] [Green Version]
- Fukata, M.; Fukata, Y.; Adesnik, H.; Nicoll, R.A.; Bredt, D.S. Identification of PSD-95 Palmitoylating Enzymes. Neuron 2004, 44, 987–996. [Google Scholar] [CrossRef] [Green Version]
- Greaves, J.; Gorleku, O.A.; Salaun, C.; Chamberlain, L.H. Palmitoylation of the SNAP25 Protein Family: Specificity and regulation by dhhc palmitoyl transferases. J. Biol. Chem. 2010, 285, 24629–24638. [Google Scholar] [CrossRef] [Green Version]
- Ponimaskin, E.; Dityateva, G.; Ruonala, M.O.; Fukata, M.; Fukata, Y.; Kobe, F.; Wouters, F.S.; Delling, M.; Bredt, D.S.; Schachner, M.; et al. Fibroblast growth factor-regulated palmitoylation of the neural cell adhesion molecule determines neuronal morphogenesis. J. Neurosci. 2008, 28, 8897–8907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dachtler, J.; Fox, K. Do cortical plasticity mechanisms differ between males and females? J. Neurosci. Res. 2016, 95, 518–526. [Google Scholar] [CrossRef] [Green Version]
- Zareba-Koziol, M.; Bartkowiak-Kaczmarek, A.; Figiel, I.; Krzystyniak, A.; Wojtowicz, T.; Bijata, M.; Wlodarczyk, J. Stress-induced Changes in the S-palmitoylation and S-nitrosylation of Synaptic Proteins. Mol. Cell. Proteom. 2019, 18, 1916–1938. [Google Scholar] [CrossRef] [PubMed]
- Bindea, G.; Mlecnik, B.; Hackl, H.; Charoentong, P.; Tosolini, M.; Kirilovsky, A.; Fridman, W.-H.; Pagès, F.; Trajanoski, Z.; Galon, J. ClueGO: A Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 2009, 25, 1091–1093. [Google Scholar] [CrossRef] [Green Version]
- Ashburner, M.; Ball, C.A.; Blake, J.A.; Botstein, D.; Butler, H.; Cherry, J.M.; Davis, A.P.; Dolinski, K.; Dwight, S.S.; Eppig, J.T.; et al. Gene Ontology: Tool for the unification of biology. Nat. Genet. 2000, 25, 25–29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dutta, P.; Basu, S.; Kundu, M. Assessment of Semantic Similarity between Proteins Using Information Content and Topological Properties of the Gene Ontology Graph. IEEE/ACM Trans. Comput. Biol. Bioinform. 2017, 15, 839–849. [Google Scholar] [CrossRef]
- Hohoff, C.; Zhang, M.; Ambrée, O.; Kravchenko, M.; Buschert, J.; Kerkenberg, N.; Gorinski, N.; Galil, D.A.; Schettler, C.; Werth, K.L.V.; et al. Deficiency of the palmitoyl acyltransferase ZDHHC7 impacts brain and behavior of mice in a sex-specific manner. Brain Struct. Funct. 2019, 224, 2213–2230. [Google Scholar] [CrossRef]
- Gorinski, N.; Wojciechowski, D.; Guseva, D.; Galil, D.A.; Mueller, F.E.; Wirth, A.; Thiemann, S.; Zeug, A.; Schmidt, S.; Zareba-Kozioł, M.; et al. DHHC7-mediated palmitoylation of the accessory protein barttin critically regulates the functions of ClC-K chloride channels. J. Biol. Chem. 2020, 295, 5970–5983. [Google Scholar] [CrossRef] [Green Version]
- Greaves, J.; Chamberlain, L.H. DHHC palmitoyl transferases: Substrate interactions and (patho)physiology. Trends Biochem. Sci. 2011, 36, 245–253. [Google Scholar] [CrossRef] [PubMed]
- De, I.; Sadhukhan, S. Emerging Roles of DHHC-mediated Protein S-palmitoylation in Physiological and Pathophysiological Context. Eur. J. Cell Biol. 2018, 97, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Duman, R.S.; Sanacora, G.; Krystal, J.H. Altered Connectivity in Depression: GABA and Glutamate Neurotransmitter Deficits and Reversal by Novel Treatments. Neuron 2019, 102, 75–90. [Google Scholar] [CrossRef]
- Conrad, C.; Jackson, J.; Wise, L. Chronic stress enhances ibotenic acid-induced damage selectively within the hippocampal CA3 region of male, but not female rats. Neuroscience 2004, 125, 759–767. [Google Scholar] [CrossRef] [Green Version]
- Albanesi, J.P.; Barylko, B.; DeMartino, G.N.; Jameson, D.M. Palmitoylated Proteins in Dendritic Spine Remodeling. Front. Synaptic Neurosci. 2020, 12, 22. [Google Scholar] [CrossRef]
- Sohn, H.; Park, M. Palmitoylation-mediated synaptic regulation of AMPA receptor trafficking and function. Arch. Pharm. Res. 2019, 42, 426–435. [Google Scholar] [CrossRef] [Green Version]
- Tu, X.; Yasuda, R.; Colgan, L.A. Rac1 is a downstream effector of PKCα in structural synaptic plasticity. Sci. Rep. 2020, 10, 1777. [Google Scholar] [CrossRef] [Green Version]
- Distler, U.; Schumann, S.; Kesseler, H.-G.; Pielot, R.; Smalla, K.-H.; Sielaff, M.; Schmeisser, M.J.; Tenzer, S. Proteomic Analysis of Brain Region and Sex-Specific Synaptic Protein Expression in the Adult Mouse Brain. Cells 2020, 9, 313. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Hu, L.-L.; Liu, L.-Y.; Zhao, L.-Y.; Hou, N.; Ni, L.; Li, Z.-F.; Wang, A.-Y.; Song, T.-S.; Huang, C. Proteomics Reveals Intersexual Differences in the Rat Brain Hippocampus. Anat. Rec. Adv. Integr. Anat. Evol. Biol. 2013, 296, 462–469. [Google Scholar] [CrossRef]
- Bundy, J.L.; Vied, C.; Nowakowski, R.S. Sex differences in the molecular signature of the developing mouse hippocampus. BMC Genom. 2017, 18, 237. [Google Scholar] [CrossRef] [Green Version]
- Bian, C.; Zhu, K.; Guo, Q.; Xiong, Y.; Cai, W.; Zhang, J. Sex differences and synchronous development of steroid receptor coactivator-1 and synaptic proteins in the hippocampus of postnatal female and male C57BL/6 mice. Steroids 2012, 77, 149–156. [Google Scholar] [CrossRef]
- Pascovici, D.; Wu, J.X.; McKay, M.J.; Joseph, C.; Noor, Z.; Kamath, K.; Wu, Y.; Ranganathan, S.; Gupta, V.; Mirzaie, M. Clinically Relevant Post-Translational Modification Analyses—Maturing Workflows and Bioinformatics Tools. Int. J. Mol. Sci. 2019, 20, 16. [Google Scholar] [CrossRef] [Green Version]
- Karve, T.M.; Cheema, A.K. Small Changes Huge Impact: The Role of Protein Posttranslational Modifications in Cellular Homeostasis and Disease. J. Amino Acids 2011, 2011, 207691. [Google Scholar] [CrossRef] [Green Version]
- Sunyer, B.; Diao, W.; Lubec, G. The role of post-translational modifications for learning and memory formation. Electrophoresis 2008, 29, 2593–2602. [Google Scholar] [CrossRef]
- Khaliulin, I.; Kartawy, M.; Amal, H. Sex Differences in Biological Processes and Nitrergic Signaling in Mouse Brain. Biomedicine 2020, 8, 124. [Google Scholar] [CrossRef]
- Ho, G.P.H.; Selvakumar, B.; Mukai, J.; Hester, L.D.; Wang, Y.; Gogos, J.A.; Snyder, S.H. S-Nitrosylation and S-Palmitoylation Reciprocally Regulate Synaptic Targeting of PSD-95. Neuron 2011, 71, 131–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stamler, J.S.; Toone, E.J.; Lipton, S.A.; Sucher, N.J. (S) NO Signals: Translocation, Regulation, and a Consensus Motif. Neuron 1997, 18, 691–696. [Google Scholar] [CrossRef] [Green Version]
- Hyer, M.; Phillips, L.L.; Neigh, G.N. Sex Differences in Synaptic Plasticity: Hormones and Beyond. Front. Mol. Neurosci. 2018, 11, 266. [Google Scholar] [CrossRef]
- Garrett, J.; Wellman, C. Chronic stress effects on dendritic morphology in medial prefrontal cortex: Sex differences and estrogen dependence. Neuroscience 2009, 162, 195–207. [Google Scholar] [CrossRef] [Green Version]
- Monteiro-Fernandes, D.; Sousa, N.; Almeida, O.; Sotiropoulos, I. Sex Hormone Depletion Augments Glucocorticoid Induction of Tau Hyperphosphorylation in Male Rat Brain. Neuroscience 2021, 454, 140–150. [Google Scholar] [CrossRef]
- Farrell, M.R.; Gruene, T.M.; Shansky, R.M. The influence of stress and gonadal hormones on neuronal structure and function. Horm. Behav. 2015, 76, 118–124. [Google Scholar] [CrossRef] [Green Version]
- Galea, L.A.M.; Leuner, B.; Slattery, D.A. Hippocampal Plasticity during the Peripartum Period: Influence of Sex Steroids, Stress and Ageing. J. Neuroendocr. 2014, 26, 641–648. [Google Scholar] [CrossRef]
- Kramár, E.A.; Chen, L.Y.; Brandon, N.J.; Rex, C.S.; Liu, F.; Gall, C.M.; Lynch, G.; Christopher, S.R. Cytoskeletal Changes Underlie Estrogen’s Acute Effects on Synaptic Transmission and Plasticity. J. Neurosci. 2009, 29, 12982–12993. [Google Scholar] [CrossRef] [Green Version]
- Rehbein, E.; Hornung, J.; Poromaa, I.S.; Derntl, B. Shaping of the female human brain by sex hormones—A review. Neuroendocrinology 2020, 111, 183–206. [Google Scholar] [CrossRef]
- Lisofsky, N.; Mårtensson, J.; Eckert, A.; Lindenberger, U.; Gallinat, J.; Kühn, S. Hippocampal volume and functional connectivity changes during the female menstrual cycle. Neuroimage 2015, 118, 154–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Protopopescu, X.; Butler, T.; Pan, H.; Root, J.; Altemus, M.; Polanecsky, M.; McEwen, B.; Silbersweig, D.; Stern, E. Hippocampal structural changes across the menstrual cycle. Hippocampus 2008, 18, 985–988. [Google Scholar] [CrossRef]
- Shors, T.J.; Falduto, J.; Leuner, B. The opposite effects of stress on dendritic spines in male vs. female rats are NMDA receptor-dependent. Eur. J. Neurosci. 2004, 19, 145–150. [Google Scholar] [CrossRef] [Green Version]
- Shors, T.J.; Chua, C.; Falduto, J. Sex differences and opposite effects of stress on dendritic spine density in the male ver-sus female hippocampus. J. Neurosci. 2001, 21, 6292–6297. [Google Scholar] [CrossRef] [Green Version]
- Forlano, P.M.; Woolley, C.S. Quantitative analysis of pre-and postsynaptic sex differences in the nucleus accumbens. J. Comp. Neurol. 2009, 518, 1330–1348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, B.; Dunaevsky, A. Modeling Neurodevelopmental and Neuropsychiatric Diseases with Astrocytes Derived from Human-Induced Pluripotent Stem Cells. Int. J. Mol. Sci. 2021, 22, 1692. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Weiland, H.; Schöfbänker, M.; Zhang, W. Estrogen Receptors Alpha and Beta Mediate Synaptic Transmission in the PFC and Hippocampus of Mice. Int. J. Mol. Sci. 2021, 22, 1485. [Google Scholar] [CrossRef]
- Rabiant, K.; Antol, J.; Naassila, M.; Pierrefiche, O. Sex difference in the vulnerability to hippocampus plasticity impairment after binge-like ethanol exposure in adolescent rat: Is estrogen the key? Addict. Biol. 2021, e13002. [Google Scholar] [CrossRef]
- Skucas, V.A.; Duffy, A.M.; Harte-Hargrove, L.; Magagna-Poveda, A.; Radman, T.; Chakraborty, G.; Schroeder, C.E.; MacLusky, N.J.; Scharfman, H.E. Testosterone Depletion in Adult Male Rats Increases Mossy Fiber Transmission, LTP, and Sprouting in Area CA3 of Hippocampus. J. Neurosci. 2013, 33, 2338–2355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matt, L.; Kim, K.; Chowdhury, D.; Hell, J.W. Role of Palmitoylation of Postsynaptic Proteins in Promoting Synaptic Plasticity. Front. Mol. Neurosci. 2019, 12, 8. [Google Scholar] [CrossRef]
- Han, J.; Wu, P.; Wang, F.; Chen, J. S-palmitoylation regulates ampa receptors trafficking and function: A novel insight into synaptic regulation and therapeutics. Acta Pharm. Sin. B 2015, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Dejanovic, B.; Semtner, M.; Ebert, S.; Lamkemeyer, T.; Neuser, F.; Lüscher, B.; Meier, J.C.; Schwarz, G. Palmitoylation of Gephyrin Controls Receptor Clustering and Plasticity of GABAergic Synapses. PLoS Biol. 2014, 12, e1001908. [Google Scholar] [CrossRef] [Green Version]
- Jeyifous, O.; Lin, E.I.; Chen, X.; Antinone, S.E.; Mastro, R.; Drisdel, R.; Reese, T.S.; Green, W.N. Palmitoylation regulates glutamate receptor distributions in postsynaptic densities through control of PSD95 conformation and orientation. Proc. Natl. Acad. Sci. USA 2016, 113, E8482–E8491. [Google Scholar] [CrossRef] [Green Version]
- Fukata, Y.; Dimitrov, A.; Boncompain, G.; Vielemeyer, O.; Perez, F.; Fukata, M. Local palmitoylation cycles define activity-regulated postsynaptic subdomains. J. Cell Biol. 2013, 202, 145–161. [Google Scholar] [CrossRef] [Green Version]
- Choii, G.; Ko, J. Gephyrin: A central GABAergic synapse organizer. Exp. Mol. Med. 2015, 47, e158. [Google Scholar] [CrossRef] [Green Version]
- Majo, G.; Lorenzo, M.J.; Blasi, J.; Aguado, F. Exocytotic protein components in rat pituitary gland after long-term estrogen administration. J. Endocrinol. 1999, 161, 323–331. [Google Scholar] [CrossRef] [Green Version]
- Verpelli, C.; Schmeisser, M.J.; Sala, C.; Boeckers, T.M. Scaffold Proteins at the Postsynaptic Density. Adv. Exp. Med. Biol. 2012, 970, 29–61. [Google Scholar] [CrossRef]
- Mastro, T.L.; Preza, A.; Basu, S.; Chattarji, S.; Till, S.M.; Kind, P.C.; Kennedy, M.B. A sex difference in the response of the rodent postsynaptic density to synGAP haploinsufficiency. Elife 2020, 9, e52656. [Google Scholar] [CrossRef]
- Ventura-Clapier, R.; Moulin, M.; Piquereau, J.; Lemaire, C.; Mericskay, M.; Veksler, V.; Garnier, A. Mitochondria: A central target for sex differences in pathologies. Clin. Sci. 2017, 131, 803–822. [Google Scholar] [CrossRef]
- Velarde, M.C. Mitochondrial and sex steroid hormone crosstalk during aging. Longev. Healthspan 2014, 3, 2. [Google Scholar] [CrossRef] [Green Version]
- Arias-Reyes, C.; Losantos-Ramos, K.M.; Gonzales, M.; Furrer, D.; Soliz, J.; Christian, A.-R.; Losantos, R.K.; Marcelino, G.; Daniela, F.; Jorge, S. NADH-linked mitochondrial respiration in the developing mouse brain is sex-, age- and tissue-dependent. Respir. Physiol. Neurobiol. 2019, 266, 156–162. [Google Scholar] [CrossRef]
- Irwin, R.W.; Yao, J.; Hamilton, R.T.; Cadenas, E.; Brinton, R.D.; Nilsen, J. Progesterone and Estrogen Regulate Oxidative Metabolism in Brain Mitochondria. Endocrinology 2008, 149, 3167–3175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harish, G.; Venkateshappa, C.; Mahadevan, A.; Pruthi, N.; Bharath, M.M.S.; Shankar, S.K. Mitochondrial function in human brains is affected bypre-andpost mortemfactors. Neuropathol. Appl. Neurobiol. 2013, 39, 298–315. [Google Scholar] [CrossRef]
- Gaignard, P.; Savouroux, S.; Liere, P.; Pianos, A.; Thérond, P.; Schumacher, M.; Slama, A.; Guennoun, R. Effect of Sex Differences on Brain Mitochondrial Function and Its Suppression by Ovariectomy and in Aged Mice. Endocrinology 2015, 156, 2893–2904. [Google Scholar] [CrossRef] [PubMed]
- Bosch, M.; Castro, J.; Saneyoshi, T.; Matsuno, H.; Sur, M.; Hayashi, Y. Structural and Molecular Remodeling of Dendritic Spine Substructures during Long-Term Potentiation. Neuron 2014, 82, 444–459. [Google Scholar] [CrossRef] [Green Version]
- Ehansberg-Pastor, V.; Egonzález-Arenas, A.; Piña-Medina, A.G.; Ecamacho-Arroyo, I. Sex Hormones Regulate Cytoskeletal Proteins Involved in Brain Plasticity. Front. Psychiatry 2015, 6, 165. [Google Scholar] [CrossRef] [Green Version]
- Kalpachidou, T.; Spiecker, L.; Kress, M.; Quarta, S. Rho GTPases in the Physiology and Pathophysiology of Peripheral Sensory Neurons. Cells 2019, 8, 591. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spillane, M.; Gallo, G. Involvement of Rho-family GTPases in axon branching. Small GTPases 2014, 5, e27974. [Google Scholar] [CrossRef] [Green Version]
- Moutin, E.; Nikonenko, I.; Stefanelli, T.; Wirth, A.; Ponimaskin, E.; De Roo, M.; Muller, D. Palmitoylation of cdc42 Promotes Spine Stabilization and Rescues Spine Density Deficit in a Mouse Model of 22q11.2 Deletion Syndrome. Cereb. Cortex 2016, 27, 3618–3629. [Google Scholar] [CrossRef]
- Wirth, A.; Chen-Wacker, C.; Wu, Y.-W.; Gorinski, N.; Filippov, M.A.; Pandey, G.; Ponimaskin, E. Dual lipidation of the brain-specific Cdc42 isoform regulates its functional properties. Biochem. J. 2013, 456, 311–322. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, D.P.; Woolfrey, K.M.; Liu, F.; Brandon, N.; Penzes, P. Estrogen Receptor Activity Modulates Synaptic Signaling and Structure. J. Neurosci. 2010, 30, 13454–13460. [Google Scholar] [CrossRef] [Green Version]
- Papadopoulou, N.; Charalampopoulos, I.; Alevizopoulos, K.; Gravanis, A.; Stournaras, C. Rho/ROCK/actin signaling regulates membrane androgen receptor induced apoptosis in prostate cancer cells. Exp. Cell Res. 2008, 314, 3162–3174. [Google Scholar] [CrossRef]
- De Pins, B.; Montalban, E.; Vanhoutte, P.; Giralt, A.; Girault, J.-A. The non-receptor tyrosine kinase Pyk2 modulates acute locomotor effects of cocaine in D1 receptor-expressing neurons of the nucleus accumbens. Sci. Rep. 2020, 10, 6619. [Google Scholar] [CrossRef] [Green Version]
- Giralt, A.; Brito, V.; Chevy, Q.; Simonnet, C.; Otsu, Y.; Diaz, C.C.; De Pins, B.; Coura, R.; Alberch, J.; Ginés, S.; et al. Pyk2 modulates hippocampal excitatory synapses and contributes to cognitive deficits in a Huntington’s disease model. Nat. Commun. 2017, 8, 15592. [Google Scholar] [CrossRef] [PubMed]
- Fourie, C.; Li, D.; Montgomery, J.M. The anchoring protein SAP97 influences the trafficking and localisation of multiple membrane channels. Biochim. Biophys. Acta Biomembr. 2014, 1838, 589–594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Waites, C.L.; Specht, C.G.; Härtel, K.; Leal-Ortiz, S.; Genoux, D.; Li, D.; Drisdel, R.C.; Jeyifous, O.; Cheyne, J.; Green, W.N.; et al. Synaptic SAP97 Isoforms Regulate AMPA Receptor Dynamics and Access to Presynaptic Glutamate. J. Neurosci. 2009, 29, 4332–4345. [Google Scholar] [CrossRef] [PubMed]
- Zaręba-Kozioł, M.; Szwajda, A.; Dadlez, M.; Wysłouch-Cieszyńska, A.; Lalowski, M. Global Analysis of S-nitrosylation Sites in the Wild Type (APP) Transgenic Mouse Brain-Clues for Synaptic Pathology. Mol. Cell. Proteom. 2014, 13, 2288–2305. [Google Scholar] [CrossRef] [Green Version]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaręba-Kozioł, M.; Bartkowiak-Kaczmarek, A.; Roszkowska, M.; Bijata, K.; Figiel, I.; Halder, A.K.; Kamińska, P.; Müller, F.E.; Basu, S.; Zhang, W.; et al. S-Palmitoylation of Synaptic Proteins as a Novel Mechanism Underlying Sex-Dependent Differences in Neuronal Plasticity. Int. J. Mol. Sci. 2021, 22, 6253. https://doi.org/10.3390/ijms22126253
Zaręba-Kozioł M, Bartkowiak-Kaczmarek A, Roszkowska M, Bijata K, Figiel I, Halder AK, Kamińska P, Müller FE, Basu S, Zhang W, et al. S-Palmitoylation of Synaptic Proteins as a Novel Mechanism Underlying Sex-Dependent Differences in Neuronal Plasticity. International Journal of Molecular Sciences. 2021; 22(12):6253. https://doi.org/10.3390/ijms22126253
Chicago/Turabian StyleZaręba-Kozioł, Monika, Anna Bartkowiak-Kaczmarek, Matylda Roszkowska, Krystian Bijata, Izabela Figiel, Anup Kumar Halder, Paulina Kamińska, Franziska E. Müller, Subhadip Basu, Weiqi Zhang, and et al. 2021. "S-Palmitoylation of Synaptic Proteins as a Novel Mechanism Underlying Sex-Dependent Differences in Neuronal Plasticity" International Journal of Molecular Sciences 22, no. 12: 6253. https://doi.org/10.3390/ijms22126253






