The Current State of Knowledge about Essential Oil Fumigation for Quality of Crops during Postharvest
Abstract
1. Introduction
2. Effects on Non-Nutrient Compounds and Antioxidant Capacity
2.1. Phenolics, Flavonoids, and Anthocyanins
2.2. Enzymatic Activity
2.3. Antioxidant Activity against Free Radicals
3. Effects on Micronutrient Compounds
4. Sensory Attributes
5. Physicochemical Parameters
5.1. Firmness
5.2. Color Parameters
5.3. Weight Loss
6. Effects on Organic Acids, and Macronutrient Compounds
7. Decay Parameters
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tripathi, P.; Dubey, N.K. Exploitation of Natural Products as an Alternative Strategy to Control Postharvest Fungal Rotting of Fruit and Vegetables. Postharvest Biol. Technol. 2004, 32, 235–245. [Google Scholar] [CrossRef]
- Barkai-Golan, R. Postharvest Diseases of Fruits and Vegetables. In Development and Control, 1st ed.; Elsevier Science: Amsterdam, The Netherlands, 2001. [Google Scholar]
- Abd-Alla, M.A.; Haggag, W.M. Use of Some Plant Essential Oils as Post-Harvest Botanical Fungicides in the Management of Anthracnose Disease of Mango Fruits (Mangi Feraindica L.) Caused by Colletotrichum Gloeosporioides (Penz). Int. J. Agric. For. 2013, 3, 1–6. [Google Scholar] [CrossRef]
- Anaruma, N.D.; Schmidt, F.L.; Duarte, M.C.T.; Figueira, G.M.; Delarmelina, C.; Benato, E.A.; Sartoratto, A. Control of Colletotrichum Gloeosporioides (Penz.) Sacc. In Yellow Passion Fruit Using Cymbopogon Citratus Essential Oil. Braz. J. Microbiol. 2010, 41, 66–73. [Google Scholar] [CrossRef]
- Solgi, M.; Ghorbanpour, M. Application of Essential Oils and Their Biological Effects on Extending the Shelf-Life and Quality of Horticultural Crops. Trakia J. Sci. 2014, 12, 198–210. [Google Scholar]
- Chao, L.K.; Hua, K.F.; Hsu, H.Y.; Cheng, S.S.; Liu, J.Y.; Chang, S.T. Study on the Antiinflammatory Activity of Essential Oil from Leaves of Cinnamomum Osmophloeum. J. Agric. Food Chem. 2005, 53, 7274–7278. [Google Scholar] [CrossRef] [PubMed]
- Ding, C.K.; Wang, C.Y.; Gross, K.C.; Smith, D.L. Reduction of Chilling Injury and Transcript Accumulation of Heat Shock Proteins in Tomato Fruit by Methyl Jasmonate and Methyl Salicylate. Plant Sci. 2001, 161, 1153–1159. [Google Scholar] [CrossRef]
- Harpaz, S.; Glatman, L.; Drabkin, V.; Gelman, A. Effects of Herbal Essential Oils Used to Extend the Shelf Life of Freshwater-Reared Asian Sea Bass Fish (Lates Calcarifer). J. Food Prot. 2003, 66, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Chanjirakul, K.; Wang, S.Y.; Wang, C.Y.; Siriphanich, J. Effect of Natural Volatile Compounds on Antioxidant Capacity and Antioxidant Enzymes in Raspberries. Postharvest Biol. Technol. 2006, 40, 106–115. [Google Scholar] [CrossRef]
- Wang, C.Y. Maintaining Postharvest Quality of Raspberries with Natural Volatile Compounds. Int. J. Food Sci. Technol. 2003, 38, 869–875. [Google Scholar] [CrossRef]
- Wang, C.Y.; Wang, S.Y.; Yin, J.J.; Parry, J.; Yu, L.L. Enhancing Antioxidant, Antiproliferation, and Free Radical Scavenging Activities in Strawberries with Essential Oils. J. Agric. Food Chem. 2007, 55, 6527–6532. [Google Scholar] [CrossRef]
- Chanjirakul, K.; Wang, S.Y.; Wang, C.Y.; Siriphanich, J. Natural Volatile Treatments Increase Free-Radical Scavenging Capacity of Strawberries and Blackberries. J. Sci. Food Agric. 2007, 87, 1463–1472. [Google Scholar] [CrossRef]
- Wang, C.Y.; Wang, S.Y.; Chen, C. Increasing Antioxidant Activity and Reducing Decay of Blueberries by Essential Oils. J. Agric. Food Chem. 2008, 56, 3587–3592. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Fonseca, J.M.; Choi, J.H.; Kubota, C. Effect of Methyl Jasmonate on Phenolic Compounds and Carotenoids of Romaine Lettuce (Lactuca Sativa L.). J. Agric. Food Chem. 2007, 55, 10366–10372. [Google Scholar] [CrossRef] [PubMed]
- Qu, T.; Li, B.; Huang, X.; Li, X.; Ding, Y.; Chen, J.; Tang, X. Effect of Peppermint Oil on the Storage Quality of White Button Mushrooms (Agaricus Bisporus). Food Bioprocess Technol. 2020, 13, 404–418. [Google Scholar] [CrossRef]
- Hu, Y.H.; Chen, C.M.; Xu, L.; Cui, Y.; Yu, X.Y.; Gao, H.J.; Wang, Q.; Liu, K.; Shi, Y.; Chen, Q.X. Postharvest Application of 4-Methoxy Cinnamic Acid for Extending the Shelf Life of Mushroom (Agaricus Bisporus). Postharvest Biol. Technol. 2015, 104, 33–41. [Google Scholar] [CrossRef]
- Jiang, T.; Luo, Z.; Ying, T. Fumigation with Essential Oils Improves Sensory Quality and Enhanced Antioxidant Ability of Shiitake Mushroom (Lentinus Edodes). Food Chem. 2015, 172, 692–698. [Google Scholar] [CrossRef]
- Gao, M.; Feng, L.; Jiang, T. Browning Inhibition and Quality Preservation of Button Mushroom (Agaricus Bisporus) by Essential Oils Fumigation Treatment. Food Chem. 2014, 149, 107–113. [Google Scholar] [CrossRef]
- Santoro, K.; Maghenzani, M.; Chiabrando, V.; Bosio, P.; Gullino, M.L.; Spadaro, D.; Giacalone, G. Thyme and Savory Essential Oil Vapor Treatments Control Brown Rot and Improve the Storage Quality of Peaches and Nectarines, but Could Favor Gray Mold. Foods 2018, 7, 7. [Google Scholar] [CrossRef]
- Serrano, M.; Martínez-Romero, D.; Guillén, F.; Valverde, J.M.; Zapata, P.J.; Castillo, S.; Valero, D. The Addition of Essential Oils to MAP as a Tool to Maintain the Overall Quality of Fruits. Trends Food Sci. Technol. 2008, 19, 464–471. [Google Scholar] [CrossRef]
- Feldman, E.B. Phenolic: Prooxdants or Antioxidants? Nutr. Rev. 1997, 55, 396. [Google Scholar]
- Cook, N.C.; Samman, S. Flavonoids-Chemistry, Metabolism, Cardioprotective Effects, and Dietary Sources. J. Nutr. Biochem. 1996, 7, 66–76. [Google Scholar] [CrossRef]
- Cheong, J.J.; Choi, Y.D. Methyl Jasmonate as a Vital Substance in Plants. Trends Genet. 2003, 19, 409–413. [Google Scholar] [CrossRef]
- Jin, P.; Zheng, Y.; Tang, S.; Rui, H.; Wang, C.Y. Enhancing Disease Resistance in Peach Fruit with Methyl Jasmonate. J. Sci. Food Agric. 2009, 89, 802–808. [Google Scholar] [CrossRef]
- Wang, K.; Jin, P.; Shang, H.; Zheng, Y. Effect of Methyl Jasmonate in Combination with Ethanol Treatment on Postharvest Decay and Antioxidant Capacity in Chinese Bayberries. J. Agric. Food Chem. 2010, 58, 9597–9604. [Google Scholar] [CrossRef] [PubMed]
- Meng, D.; Song, T.; Shen, L.; Zhang, X.; Sheng, J. Postharvest Application of Methyl Jasmonate for Improving Quality Retention of Agaricus Bisporus Fruit Bodies. J. Agric. Food Chem. 2012, 60, 6056–6062. [Google Scholar] [CrossRef]
- Wang, S.Y.; Chen, C.; Yin, J. Effect of Allyl Isothiocyanate on Antioxidants and Fruit Decay of Blueberries. Food Chem. 2010, 120, 199–204. [Google Scholar] [CrossRef]
- Sellamuthu, P.S.; Sivakumar, D.; Soundy, P.; Korsten, L. Essential Oil Vapours Suppress the Development of Anthracnose and Enhance Defence Related and Antioxidant Enzyme Activities in Avocado Fruit. Postharvest Biol. Technol. 2013, 81, 66–72. [Google Scholar] [CrossRef]
- Valero, D.; Valverde, J.M.; Martínez-Romero, D.; Guillén, F.; Castillo, S.; Serrano, M. The Combination of Modified Atmosphere Packaging with Eugenol or Thymol to Maintain Quality, Safety and Functional Properties of Table Grapes. Postharvest Biol. Technol. 2006, 41, 317–327. [Google Scholar] [CrossRef]
- Geransayeh, M.; Sepahvand, S.; Abdossi, V.; Nezhad, R.A. Effect of Thymol Treatment on Decay, Postharvest Life and Quality of Strawberry (Fragaria Ananassa) Fruit Cv. “Gaviota”. Int. J. Agron. Agric. Res. 2015, 6, 151–162. [Google Scholar]
- Sellamuthu, P.S.; Mafune, M.; Sivakumar, D.; Soundy, P. Thyme Oil Vapour and Modified Atmosphere Packaging Reduce Anthracnose Incidence and Maintain Fruit Quality in Avocado. J. Sci. Food Agric. 2013, 93, 3024–3031. [Google Scholar] [CrossRef]
- Perumal, A.B.; Sellamuthu, P.S.; Nambiar, R.B.; Sadiku, E.R. Effects of Essential Oil Vapour Treatment on the Postharvest Disease Control and Different Defence Responses in Two Mango (Mangifera Indica L.) Cultivars. Food Bioprocess Technol. 2017, 10, 1131–1141. [Google Scholar] [CrossRef]
- Jin, P.; Wu, X.; Xu, F.; Wang, X.; Wang, J.; Zheng, Y. Enhancing Antioxidant Capacity and Reducing Decay of Chinese Bayberries by Essential Oils. J. Agric. Food Chem. 2012, 60, 3769–3775. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.; Amini, J.; Saba, M.K.; Karimi, K.; Pertot, I. Preharvest and Postharvest Application of Garlic and Rosemary Essential Oils for Controlling Anthracnose and Quality Assessment of Strawberry Fruit During Cold Storage. Front. Microbiol. 2020, 11, 1–15. [Google Scholar] [CrossRef]
- Mrázová, A.; Belay, S.A.; Eliášová, A.; Perez-Delgado, C.; Kaducová, M.; Betti, M.; Vega, J.M.; Palove-Balang, P. Expression, Activity of Phenylalanine-Ammonia-Lyase and Accumulation of Phenolic Compounds in Lotus Japonicus under Salt Stress. Biology 2017, 72, 36–42. [Google Scholar] [CrossRef]
- Medda, S.; Dessena, L.; Mulas, M. Monitoring of the PAL Enzymatic Activity and Polyphenolic Compounds in Leaves and Fruits of Two Myrtle Cultivars during Maturation. Agriculture 2020, 10, 389. [Google Scholar] [CrossRef]
- Sikora, N.; Znajewska, Z.; Dąbrowska, G. Wybrane Mechanizmy Obronne Organizmów Przed Stresem Oksydacyjnym Ze Szczególnym Uwzględnieniem Dysmutaz Ponadtlenkowych. Eduk. Biol. i Środowiskowa 2017, 4, 19–31. [Google Scholar] [CrossRef]
- Song, X.; Verma, K.K.; Tian, D.; Zhang, X.; Liang, Y.; Huang, X.; Li, C.; Li, Y. Exploration of Silicon Functions to Integrate with Biotic Stress Tolerance and Crop Improvement. Biol. Res. 2021, 54. [Google Scholar] [CrossRef]
- Malinowska-Pańczyk, E.; Kołodziejska, I. Możliwości Zastosowania Wysokiego Ciśnienia W Przemyśle Owocowo-Warzywnym. ŻYWNOŚĆ. Nauk. Technol. Jakość 2010, 2, 5–15. [Google Scholar]
- Nandi, A.; Yan, L.; Jana, C.K.; Das, N. Review Article Role of Catalase in Oxidative Stress- and Age-Associated Degenerative Diseases. Oxid. Med. Cell. Longev. 2019, 2019, 9613090. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Shao, X.; Wei, Y.; Li, Y.; Xu, F.; Wang, H. Solidago Canadensis L. Essential Oil Vapor Effectively Inhibits Botrytis Cinerea Growth and Preserves Postharvest Quality of Strawberry as a Food Model System. Front. Microbiol. 2016, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Karimirad, R.; Behnamian, M.; Dezhsetan, S.; Sonnenberg, A. Chitosan Nanoparticles-Loaded Citrus Aurantium Essential Oil: A Novel Delivery System for Preserving the Postharvest Quality of Agaricus Bisporus. J. Sci. Food Agric. 2018, 98, 5112–5119. [Google Scholar] [CrossRef]
- Chen, H.; Gao, H.; Fang, X.; Ye, L.; Zhou, Y.; Yang, H. Effects of Allyl Isothiocyanate Treatment on Postharvest Quality and the Activities of Antioxidant Enzymes of Mulberry Fruit. Postharvest Biol. Technol. 2015, 108, 61–67. [Google Scholar] [CrossRef]
- Forkmann, G.; Heller, W.; Forschungszentrum, G.S.F. Biosynthesis of Flavonoids. In Comprehensive Natural Products Chemistry; Springer: Berlin/Heidelberg, Germany, 1999; p. 719. [Google Scholar] [CrossRef]
- Wang, K.; Jin, P.; Cao, S.; Shang, H.; Yang, Z.; Zheng, Y. Methyl Jasmonate Reduces Decay and Enhances Antioxidant Capacity in Chinese Bayberries. J. Agric. Food Chem. 2009, 57, 5809–5815. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.H.; Xia, L.X.; Zhou, H.B.; Qiu, G.Z. Chemical Composition and Antioxidant Activities of Russula Griseocarnosa Sp. Nov. J. Agric. Food Chem. 2010, 58, 6966–6971. [Google Scholar] [CrossRef]
- Wang, S.Y.; Chen, C.T. Effect of Allyl Isothiocyanate on Antioxidant Enzyme Activities, Flavonoids and Post-Harvest Fruit Quality of Blueberries (Vaccinium Corymbosum L., Cv. Duke). Food Chem. 2010, 122, 1153–1158. [Google Scholar] [CrossRef]
- Melgarejo-Flores, B.G.; Ortega-Ramírez, L.A.; Silva-Espinoza, B.A.; González-Aguilar, G.A.; Miranda, M.R.A.; Ayala-Zavala, J.F. Antifungal Protection and Antioxidant Enhancement of Table Grapes Treated with Emulsions, Vapors, and Coatings of Cinnamon Leaf Oil. Postharvest Biol. Technol. 2013, 86, 321–328. [Google Scholar] [CrossRef]
- Nasirifar, S.Z.; Maghsoudlou, Y.; Oliyaei, N. Effect of Active Lipid-Based Coating Incorporated with Nanoclay and Orange Peel Essential Oil on Physicochemical Properties of Citrus Sinensis. Food Sci. Nutr. 2018, 6, 1508–1518. [Google Scholar] [CrossRef] [PubMed]
- Farag, R.S.; Shalaby, A.S.; El-Baroty, G.A.; Ibrahim, N.A.; Ali, M.A.; Hassan, E.M. Chemical and Biological Evaluation of the Essential Oils of Different Melaleuca Species. Phyther. Res. 2004, 18, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Ismahan, G.; Serpil, E.; Huseyin, A.; Nazli, E.; Erol, D. Malondialdehyde and Some Biochemical Parameters of Rats *. Braz. J. Poult. Sci. 2019, 21, 001–006. [Google Scholar]
- Alikhani-Koupaei, M.; Mazlumzadeh, M.; Sharifani, M.; Adibian, M. Enhancing Stability of Essential Oils by Microencapsulation for Preservation of Button Mushroom during Postharvest. Food Sci. Nutr. 2014, 2, 526–533. [Google Scholar] [CrossRef] [PubMed]
- Tzortzakis, N.G.; Tzanakaki, K.; Economakis, C.D. Effect of Origanum Oil and Vinegar on the Maintenance of Postharvest Quality of Tomato. Food Nutr. Sci. 2011, 02, 974–982. [Google Scholar] [CrossRef][Green Version]
- Maghenzani, M.; Chiabrando, V.; Santoro, K.; Spadaro, D.; Giacalone, G. Effects of Treatment by Vapour of Essential Oil from Thymus Vulgaris and Satureja Montana on Postharvest Quality of Sweet Cherry (Cv. Ferrovia). J. Food Nutr. Res. 2018, 57, 161–169. [Google Scholar]
- Raafat, S.M.; Abou-Zaid, M.I.; Aly, A.Z.; Tohamy, M.R.A. Safety Control of Strawberry Fruits Gray Mold Fungus by Plant Volatile Oils. Zagazig J. Agric. Res. 2012, 39, 181–187. [Google Scholar]
- Leong, S.Y.; Oey, I. Effects of Processing on Anthocyanins, Carotenoids and Vitamin C in Summer Fruits and Vegetables. Food Chem. 2012, 133, 1577–1587. [Google Scholar] [CrossRef]
- Mariod, A.A. Effect of Essential Oils on Organoleptic (Smell, Taste, and Texture) Properties of Food; Elsevier Inc.: Amsterdam, The Netherlands, 2016. [Google Scholar] [CrossRef]
- Božik, M.; Císarová, M.; Tančinová, D.; Kouřimská, L.; Hleba, L.; Klouček, P. Selected Essential Oil Vapours Inhibit Growth of Aspergillus Spp. in Oats with Improved Consumer Acceptability. Ind. Crops Prod. 2017, 98, 146–152. [Google Scholar] [CrossRef]
- Bill, M.; Sivakumar, D.; Beukes, M.; Korsten, L. Expression of Pathogenesis-Related (PR) Genes in Avocados Fumigated with Thyme Oil Vapours and Control of Anthracnose. Food Chem. 2016, 194, 938–943. [Google Scholar] [CrossRef] [PubMed]
- Sonker, N.; Pandey, A.K.; Singh, P. Efficiency of Artemisia Nilagirica (Clarke) Pamp. Essential Oil as a Mycotoxicant against Postharvest Mycobiota of Table Grapes. J. Sci. Food Agric. 2015, 95, 1932–1939. [Google Scholar] [CrossRef]
- González-Aguilar, G.A.; Buta, J.G.; Wang, C.Y. Methyl Jasmonate and Modified Atmosphere Packaging (MAP) Reduce Decay and Maintain Postharvest Quality of Papaya “Sunrise”. Postharvest Biol. Technol. 2003, 28, 361–370. [Google Scholar] [CrossRef]
- Valverde, J.M.; Guillén, F.; Martínez-Romero, D.; Castillo, S.; Serrano, M.; Valero, D. Improvement of Table Grapes Quality and Safety by the Combination of Modified Atmosphere Packaging (MAP) and Eugenol, Menthol, or Thymol. J. Agric. Food Chem. 2005, 53, 7458–7464. [Google Scholar] [CrossRef] [PubMed]
- Perdones, A.; Sánchez-González, L.; Chiralt, A.; Vargas, M. Effect of Chitosan-Lemon Essential Oil Coatings on Storage-Keeping Quality of Strawberry. Postharvest Biol. Technol. 2012, 70, 32–41. [Google Scholar] [CrossRef]
- Conway, W.S.; Gross, K.C.; Sams, C.E. Relationship of Bound Calcium and Inoculum Concentration to the Effect of Postharvest Calcium Treatment on Decay of Apples by Penicillium Expansum. Plant Dis. 1987, 71, 78–80. [Google Scholar] [CrossRef]
- Wang, Y.; Shan, T.; Yuan, Y.; Yue, T. Overall Quality Properties of Kiwifruit Treated by Cinnamaldehyde and Citral: Microbial, Antioxidant Capacity during Cold Storage. J. Food Sci. 2016, 81, H3043–H3051. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.T.; Chu, C.L.; Zhou, T. Thymol and Acetic Acid Vapors Reduce Postharvest Brown Rot of Apricots and Plums. HortScience 2002, 37, 151–156. [Google Scholar] [CrossRef]
- Sumalan, R.M.; Kuganov, R.; Obistioiu, D.; Popescu, I.; Radulov, I.; Alexa, E.; Negrea, M.; Salimzoda, A.F.; Sumalan, R.L.; Cocan, I. Assessment of Mint, Basil, and Lavender Essential Oil Vapor-Phase in Antifungal Protection and Lemon Fruit Quality. Molecules 2020, 25, 1831. [Google Scholar] [CrossRef]
- Tombesi, A.; Antognozzi, E.; Palliotti, A. Influence of Light Exposure on Characteristics and Storage Life of Kiwifruit. New Zeal. J. Crop Hortic. Sci. 1993, 21, 87–92. [Google Scholar] [CrossRef]
- Serrano, M.; Martínez-Romero, D.; Castillo, S.; Guillén, F.; Valero, D. The Use of Natural Antifungal Compounds Improves the Beneficial Effect of MAP in Sweet Cherry Storage. Innov. Food Sci. Emerg. Technol. 2005, 6, 115–123. [Google Scholar] [CrossRef]
- Guillén, F.; Zapata, P.J.; Martínez-Romero, D.; Castillo, S.; Serrano, M.; Valero, D. Improvement of the Overall Quality of Table Grapes Stored under Modified Atmosphere Packaging in Combination with Natural Antimicrobial Compounds. J. Food Sci. 2007, 72, S185–S190. [Google Scholar] [CrossRef] [PubMed]
- Itai, A.; Tanahashi, T. Inhibition of Sucrose Loss during Cold Storage in Japanese Pear (Pyrus Pyrifolia Nakai) by 1-MCP. Postharvest Biol. Technol. 2008, 48, 355–363. [Google Scholar] [CrossRef]
- Jiang, D.Q.; Guo, Y.; Xu, D.H.; Huang, Y.S.; Yuan, K.; Lv, Z.Q. Antioxidant and Anti-Fatigue Effects of Anthocyanins of Mulberry Juice Purification (MJP) and Mulberry Marc Purification (MMP) from Different Varieties Mulberry Fruit in China. Food Chem. Toxicol. 2013, 59, 1–7. [Google Scholar] [CrossRef]
- Lawless, H.T.; Heymann, H. Sensory Evaluation of Food: Principles and Practices, 2nd ed.; Springer Science+ Business Media: New York, NY, USA, 1999. [Google Scholar] [CrossRef]
- Weatherall, I.L.; Coombs, B.D. Skin Color Measurements in Terms of CIELAB Color Space Values. J. Invest. Dermatol. 1992, 99, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Gormley, R. Chill Storage of Mushrooms. J. Sci. Food Agric. 1975, 26, 401–411. [Google Scholar] [CrossRef]
- Cantos, E.; Espín, J.C.; Tomás-Barberán, F.A. Varietal Differences among the Polyphenol Profiles of Seven Table Grape Cultivars Studied by LC-DAD-MS-MS. J. Agric. Food Chem. 2002, 50, 5691–5696. [Google Scholar] [CrossRef]
- González-Barrio, R.; Salmenkallio-Marttila, M.; Tomás-Barberán, F.A.; Cantos, E.; Espín, J.C. Etiology of UV-C-Induced Browning in Var. Superior White Table Grapes. J. Agric. Food Chem. 2005, 53, 5990–5996. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Qian, X.; Jiang, T.; Zheng, X. Effect of Eugenol Fumigation Treatment on Chilling Injury and CBF Gene Expression in Eggplant Fruit during Cold Storage. Food Chem. 2019, 292, 143–150. [Google Scholar] [CrossRef] [PubMed]
- De la Peña Moreno, F.; Monagas, M.; Blanch, G.P.; Bartolomé, B.; Ruiz del Castillo, M.L. Enhancement of Anthocyanins and Selected Aroma Compounds in Strawberry Fruits through Methyl Jasmonate Vapor Treatment. Eur. Food Res. Technol. 2010, 230, 989–999. [Google Scholar] [CrossRef]
- Fan, X.; Mattheis, J.P. Responses of Apples to Postharvest Jasmonate Treatments. J. Am. Soc. Hortic. Sci. 1998, 123, 421–425. [Google Scholar] [CrossRef]
- Matsuoka, K. Anthocyanins in Apple Fruit and Their Regulation for Health Benefits. In Flavonoids: A Coloring Model for Cheering Up Life; IntechOpen: London, UK, 2020; pp. 1–15. [Google Scholar] [CrossRef]
- Wills, R.B.H.; Golding, J.B. Postharvest: An Introduction to the Physiology and Handling of Fruit and Vegetables, 6th ed.; Cabi: Wallingford, UK, 2016. [Google Scholar]
- Ali, A.; Wee Pheng, T.; Mustafa, M.A. Application of Lemongrass Oil in Vapour Phase for the Effective Control of Anthracnose of “Sekaki” Papaya. J. Appl. Microbiol. 2015, 118, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Jia, B.; Xu, L.; Guan, W.; Lin, Q.; Brennan, C.; Yan, R.; Zhao, H. Effect of Citronella Essential Oil Fumigation on Sprout Suppression and Quality of Potato Tubers during Storage. Food Chem. 2019, 284, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Bosquez-Molina, E.; de Jesús, E.R.; Bautista-Baños, S.; Verde-Calvo, J.R.; Morales-López, J. Inhibitory Effect of Essential Oils against Colletotrichum Gloeosporioides and Rhizopus Stolonifer in Stored Papaya Fruit and Their Possible Application in Coatings. Postharvest Biol. Technol. 2010, 57, 132–137. [Google Scholar] [CrossRef]
- Sangsuwan, J.; Pongsapakworawat, T.; Bangmo, P.; Sutthasupa, S. Effect of Chitosan Beads Incorporated with Lavender or Red Thyme Essential Oils in Inhibiting Botrytis Cinerea and Their Application in Strawberry Packaging System. LWT-Food Sci. Technol. 2016, 74, 14–20. [Google Scholar] [CrossRef]
- Borsani, J.; Budde, C.O.; Porrini, L.; Lauxmann, M.A.; Lombardo, V.A.; Murray, R.; Andreo, C.S.; Drincovich, M.F.; Lara, M.V. Carbon Metabolism of Peach Fruit after Harvest: Changes in Enzymes Involved in Organic Acid and Sugar Level Modifications. J. Exp. Bot. 2009, 60, 1823–1837. [Google Scholar] [CrossRef] [PubMed]
- Ziedan, E.H.E.; Farrag, E.S.H. Fumigation of Peach Fruits with Essential Oils to Control Postharvest Decay. Res. J. Agric. Biol. Sci. 2008, 4, 512–519. [Google Scholar]
- Naidu, A.S. Natural Food Antimicrobial Systems, 1st ed.; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar] [CrossRef]
- Juglal, S.; Govinden, R.; Odhav, B. Spice Oils for the Control of Co-Occurring Mycotoxin-Producing Fungi. J. Food Prot. 2002, 65, 683–687. [Google Scholar] [CrossRef] [PubMed]
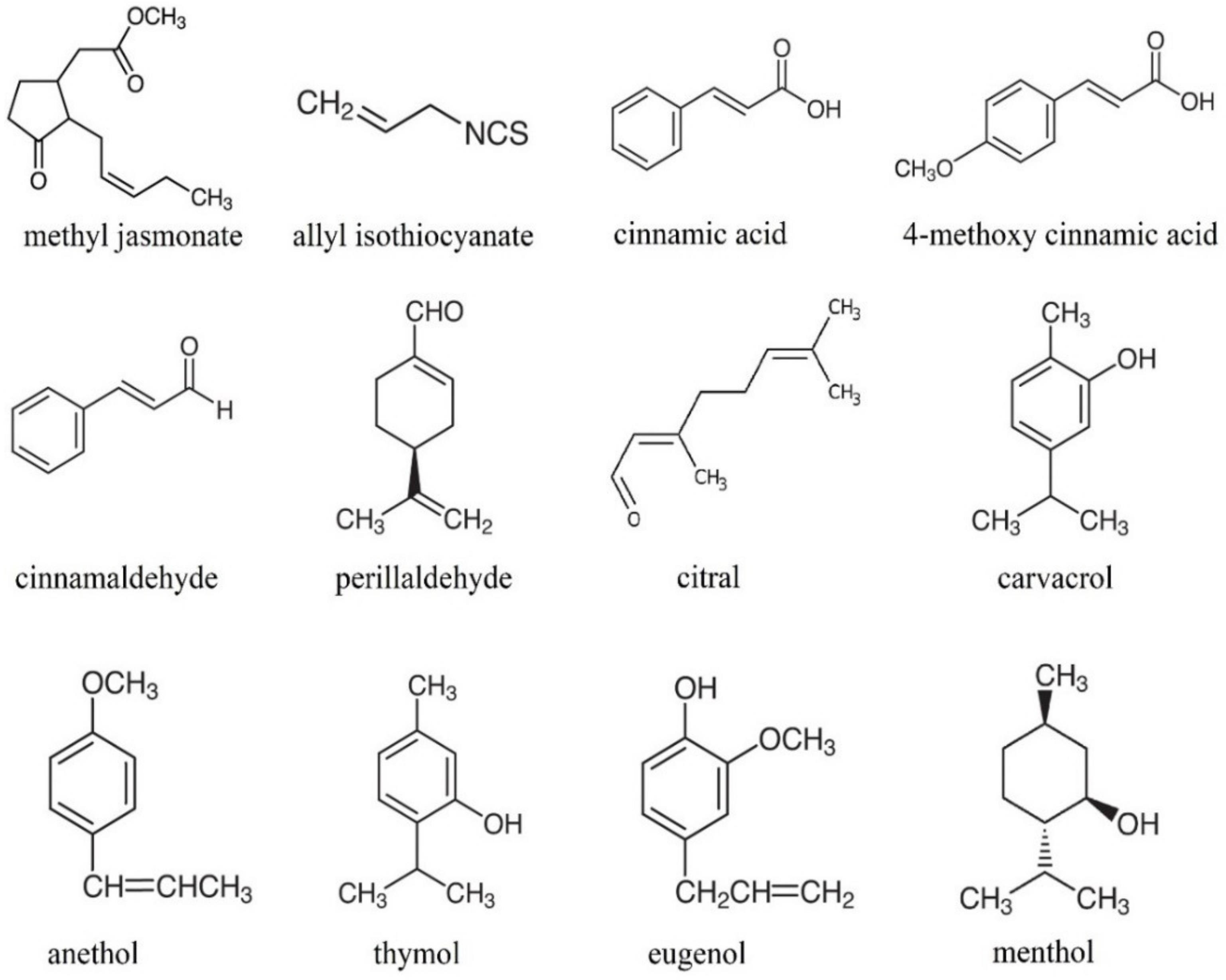
| Volatile | Parameter | Fruit | Reference |
|---|---|---|---|
| Methyl jasmonate | Total phenolics content ↑ | Prunus persica L. Myrica rubra Seib and Zucc. Agaricus bisporus L. Rubus idaeus L. | [24] [25] [26] [9] |
| PPO, POD activity ↑ | Prunus persica L. Myrica rubra Seib and Zucc. | [24] [25] | |
| SOD activity ↑ | Rubus idaeus L. Prunus persica L. Agaricus bisporus | [9] [24] [26] | |
| Ascorbic acid content ↑ | Rubus idaeus L. | [9] | |
| Decay incidence * ↓ | Myrica rubra Seib and Zucc. Solanum lycopersicum L. Fragaria x ananassas Duch. Rubus L. Prunus persica L. | [25,45] [7] [12] [12] [24] | |
| Sensory attributes ↑ | Myrica rubra Seib and Zucc. Carica papaya L. | [25] [61] | |
| Decay severity ** ↓ | Rubus idaeus L. Carica papaya L. Prunus persica L. | [10] [61] [24] | |
| Fructose, glucose, malic, and citric acids content ↑ Total sugars ↑ | Rubus idaeus L. Agaricus bisporus | [10] [26] | |
| Total carotenoids, lutein, β-carotene content ↑ | Lactuca sativa L. var. longifolia | [14] | |
| Allyl isothiocyanate | Firmness ↑ | Morus alba L. | [43] |
| Malic acid content ↑ Decay severity ** ↓ | Rubus idaeus L. | [10] | |
| Decay incidence * ↓ | Morus alba L. Fragaria x ananassas Duch. Rubus L. | [43] [12] [12] | |
| Glucose and fructose content ↑ | Vaccinium corymbosum L. | [27] | |
| 4-methoxy cinnamic acid | Ascorbic acid content ↑ Weight loss, percentage open caps ↓ | Agaricus bisporus L. | [16] |
| Carvacrol | Total phenolics content ↑ | Myrica rubra Seib and Zucc. Vaccinium corymbosum L. | [33] [13] |
| Decay incidence * ↓ | Myrica rubra Seib and Zucc. Vitis vinifera L. | [33] [69] | |
| Citric acid content ↑ Fructose and glucose content ↑ | Vaccinium corymbosum L. | [13] | |
| POD, CAT, APX, SOD activity ↑ | Myrica rubra Seib and Zucc. | [33] | |
| Anethole | Total phenolics content ↑ Citric acid content ↑ Fructose and glucose content ↑ | Vaccinium corymbosum L. | [13] |
| Menthol | Total phenolics content ↑ Fructose, glucose, and sucrose content ↑ Malic and citric acids content ↑ | Fragaria x ananassas Duch. | [11] |
| Decay incidence * ↓ | Vitis vinifera L. Fragaria x ananassas Duch. | [29] [11,62] | |
| Firmness ↑ | Prunus avium L. cv Ferrovia | [20] | |
| Thymol | Total phenolics content ↑ | Fragaria x ananassas Duch. Vitis vinifera L. | [11] [29] |
| Ascorbic acid content ↑ | Fragaria x ananassas Duch. Vitis vinifera L. | [30] [29] | |
| Malic and citric acids content ↑ Fructose, glucose, and sucrose content ↑ | Fragaria x ananassas Duch. | [11] | |
| Decay incidence * ↓ | Fragaria x ananassas Duch. Vitis vinifera L. | [11,30] [29,69] | |
| Tartaric acid content ↑ | Vitis vinifera L. | [29] | |
| Sensory attributes ↑ | Fragaria x ananassas Duch. Vitis vinifera L | [30] [62] | |
| Firmness ↑ | Prunus armeniaca L. Prunus avium L. cv Ferrovia Vitis vinifera L. Persea americana Mill. | [66] [20] [29] [31] | |
| Weight loss ↓ | Fragaria x ananassas Duch. Vitis vinifera L. Prunus avium L. cv Ferrovia | [30] [29] [20] | |
| Citral | Firmness ↑ | Fragaria x ananassas Duch. | [65] |
| Cinnamic acid | Glucose content ↑ | Vaccinium corymbosum L. | [13] |
| Eugenol | Total phenolics content ↑ | Fragaria x ananassas Duch. Vitis vinifera L. | [11] [29] |
| Firmness ↑ | Prunus avium L. cv Ferrovia Vitis vinifera L. | [20] [29] | |
| Sensory attributes ↑ | Vitis vinifera L | [62] | |
| Decay incidence * ↓ | Fragaria x ananassas Duch. Vitis vinifera L. | [11] [29,69] | |
| Ascorbic acid content ↑ Tartaric acid content ↑ | Vitis vinifera L. | [29] | |
| Malic and citric acid content ↑ Fructose, glucose, and sucrose content ↑ | Fragaria x ananassas Duch. | [11] | |
| Weight loss ↓ | Solanum melongena L. Vitis vinifera L. Prunus avium L. cv Ferrovia | [77] [29] [20] | |
| Fructose and glucose content ↑ | Vaccinium corymbosum L. | [13] | |
| Decay incidence ↓ POD, CAT, APX activity ↑ Total phenolics content ↑ | Myrica rubra Seib and Zucc. | [33] | |
| Perillaldehyde | Total phenolics content ↑ | Myrica rubra Seib and Zucc. Vaccinium corymbosum L. | [33] [13] |
| POD, CAT, APX activity ↑ | Myrica rubra Seib and Zucc. | [33] | |
| Citric acid content ↑ Fructose and glucose content ↑ | Vaccinium corymbosum L. | [13] | |
| Tea tree EO Melaleuca alternifolia L. | Total phenolics content ↑ Ascorbic acid content ↑ | Rubus idaeus L. | [9] |
| Malic and citric acid content ↑ Glucose, fructose, and sucrose content ↑ | Rubus idaeus L. | [10] | |
| Decay incidence * ↓ | Rubus idaeus L., Rubus L. | [12] | |
| Thyme EO Thymus vulgaris L. | Total phenolics content ↑ | Persea americana Mill. Agaricus bisporus L. Lentinus edodes L Mangifera indica L. | [28] [18] [17] [32] |
| Decay incidence * ↓ | Prunus persica L. var. nucipersica Prunus persica L. | [19] | |
| Sensory attributes ↓ | Prunus avium L. (cv. “Ferrovia”) Avena sativa L. | [54] [58] | |
| Sensory attributes ↑ | Lentinus edodes L. Persea americana Mill. | [17] [59] | |
| Anthracnose severity and incidence ↓ | Mangifera indica L. Persea americana Mill. | [32] [31,59] | |
| Weight loss ↓ | Prunus persica L. var. nucipersica Prunus persica L. Prunus avium L. (cv. “Ferrovia”) | [19] [19] [54] | |
| POD, SOD activity ↑ | Persea americana Mill. Mangifera indica L. | [28] [32] | |
| PPO activity ↑ | Agaricus bisporus L. | [18] | |
| Persea americana Mill. | [31] [28] | ||
| Ascorbic acid content ↑ | Prunus persica L. var. nucipersica Agaricus bisporus L. | [19] [15] | |
| Total carotenoids content ↑ | Prunus persica L. var. nucipersica | [19] | |
| Firmness ↑ | Agaricus bisporus L. Lentinus edodes L. | [18] [17] | |
| Cinnamaldehyde | Total phenolics content ↑ | Myrica rubra Seib and Zucc. Vaccinium corymbosum L. Agaricus bisporus L. Lentinus edodes L. | [33] [13] [18] [17] |
| POD, CAT, APX activity ↑ | Myrica rubra Seib and Zucc. Lentinus edodes L. | [33] [17] | |
| PPO activity ↑ Weight loss, percentage open caps ↓ | Agaricus bisporus L. | [18] | |
| Ascorbic acid content ↑ | Agaricus bisporus L. | [16] | |
| Sensory attributes ↑ | Lentinus edodes L. | [17] | |
| Firmness ↑ | Fragaria x ananassas Duch. Agaricus bisporus L. Lentinus edodes L. | [65] [18] [17] | |
| Citric acid content ↑ Fructose and glucose content ↑ | Vaccinium corymbosum L. | [13] | |
| Cinnamon EO Cinnamomum zeylanicum L. | Total phenolics content ↑ POD activity ↑ Anthracnose severity and incidence ↓ | Mangifera indica L. | [32] |
| Decay severity ** ↓ | Vitis vinifera L. | [48] | |
| Sensory attributes ↓ | Avena sativa L. | [58] | |
| Lavender EO Lavandula angustifolia L. | Firmness ↑ Decay severity ** ↓ Weight loss ↓ | Citrus limon L. | [67] |
| Clove EO Syzygium aromatic L. | Total phenolics content ↑ | Lentinus edodes L. Mangifera indica L. | [17] [32] |
| Weight loss, percentage open caps ↓ | Agaricus bisporus L. | [18] | |
| Anthracnose severity and incidence ↓ POD activity ↑ | Mangifera indica L. | [32] | |
| APX, GR activity ↑ | Lentinus edodes L. | [17] | |
| PPO activity ↑ | Agaricus bisporus Mangifera indica L. | [18] [32] | |
| Sensory attributes ↓ Sensory attributes ↑ | Avena sativa L. Lentinus edodes L. | [58] [17] | |
| Ascorbic acid content ↑ | Agaricus bisporus L. | [15] | |
| Firmness ↑ | Agaricus bisporus L. Lentinus edodes L. | [18] [17] | |
| Oregano EO Origanum vulgare L. | Ascorbic acid content ↑ Lycopene ↑ Firmness ↑ Total sugars, glucose, and fructose content ↑ Decay severity ** ↓ | Solanum lycopersicum L. | [53] |
| Decay incidence * ↓ | Avena sativa L. | [58] | |
| Peppermint EO Mentha piperita L. | Total phenolics content ↑ SOD activity ↑ PPO activity ↑ | Agaricus bisporus Persea americana Mill. | [15] [28] |
| Decay incidence * ↓ | Prunus persica L. | [87] | |
| Anthracnose severity and incidence ↓ | Persea americana Mill. | [28] | |
| Firmness ↑ | Agaricus bisporus L. | [15] | |
| Weight loss ↓ Decay severity ** | Citrus limon L. | [67] | |
| Basil EO Ocimum basilicum L. | Weight loss ↓ Decay severity ** | Citrus limon L. | [67] |
| Decay incidence * | Prunus persica L. | [87] | |
| Anthracnose incidence ↓ | Mangifera indica L. | [3] | |
| Rosemary EO Rosmarinus officinalis L. | Firmness ↑ Anthracnose severity and incidence ↓ Sensory attributes ↑ | Fragaria x ananassas Duch. | [34] |
| Garlic EO Allium sativum L. | Firmness ↑ Anthracnose severity and incidence ↓ Sensory attributes (taste) ↑ Sensory attributes (scent) ↓ | Fragaria x ananassas Duch. | [34] |
| Savory EO Satureja montana L. | Ascorbic acid content ↑ Total carotenoids content ↑ | Prunus persica L. var. nucipersica | [19] |
| Weight loss ↓ | Prunus avium L. (cv. “Ferrovia”) Prunus persica L. var. nucipersica Prunus persica L. | [54] [19] [19] | |
| Sensory attributes ↓ | Prunus avium L. (cv. “Ferrovia”) | [54] | |
| Decay incidence * ↓ | Prunus persica L. var. nucipersica Prunus persica L. | [19] | |
| Lemongrass EO Cymbopogon citratus L. | Decay incidence * ↓ Sensory attributes ↓ | Avena sativa L. | [58] |
| Citronella EO Cymbopogen nardus L. | Total phenolics content ↑ POD activity ↑ Anthracnose severity and incidence ↓ | Persea americana Mill. | [28] |
| Weight loss ↓ | Solanum tuberosum L. | [83] | |
| Goldenrod EO Solidago canadensis L. | Decay severity ** ↓ Sensory attributes ↑ | Fragaria x ananassas Duch. | [41] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Namiota, M.; Bonikowski, R. The Current State of Knowledge about Essential Oil Fumigation for Quality of Crops during Postharvest. Int. J. Mol. Sci. 2021, 22, 13351. https://doi.org/10.3390/ijms222413351
Namiota M, Bonikowski R. The Current State of Knowledge about Essential Oil Fumigation for Quality of Crops during Postharvest. International Journal of Molecular Sciences. 2021; 22(24):13351. https://doi.org/10.3390/ijms222413351
Chicago/Turabian StyleNamiota, Małgorzata, and Radosław Bonikowski. 2021. "The Current State of Knowledge about Essential Oil Fumigation for Quality of Crops during Postharvest" International Journal of Molecular Sciences 22, no. 24: 13351. https://doi.org/10.3390/ijms222413351
APA StyleNamiota, M., & Bonikowski, R. (2021). The Current State of Knowledge about Essential Oil Fumigation for Quality of Crops during Postharvest. International Journal of Molecular Sciences, 22(24), 13351. https://doi.org/10.3390/ijms222413351






