Prothoracicostatic Activity of the Ecdysis-Regulating Neuropeptide Crustacean Cardioactive Peptide (CCAP) in the Desert Locust
Abstract
1. Introduction
2. Results
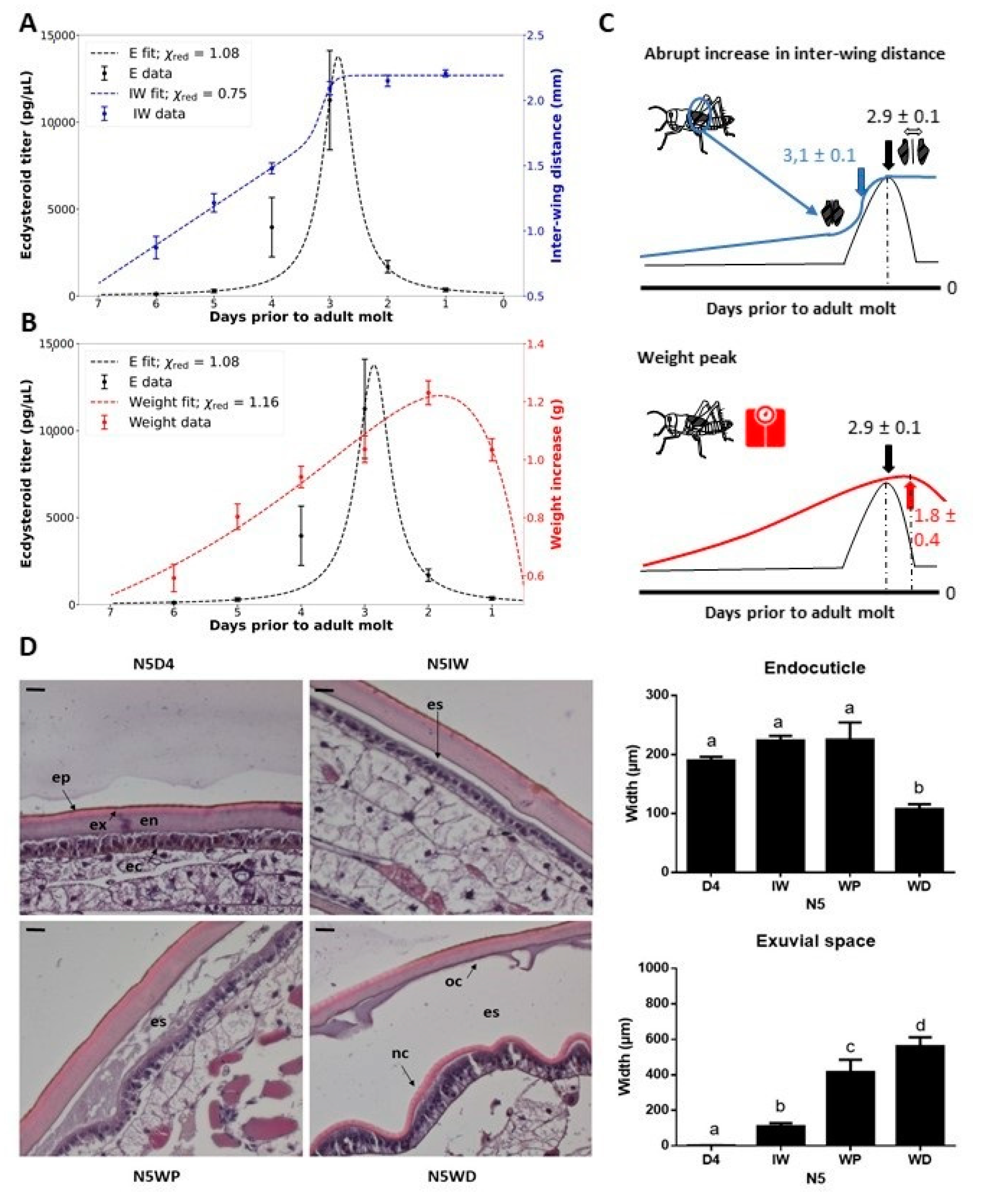
2.1. Fifth Instar Morphology in Correlation to the Ecdysteroid Peak and Structure of the Integument
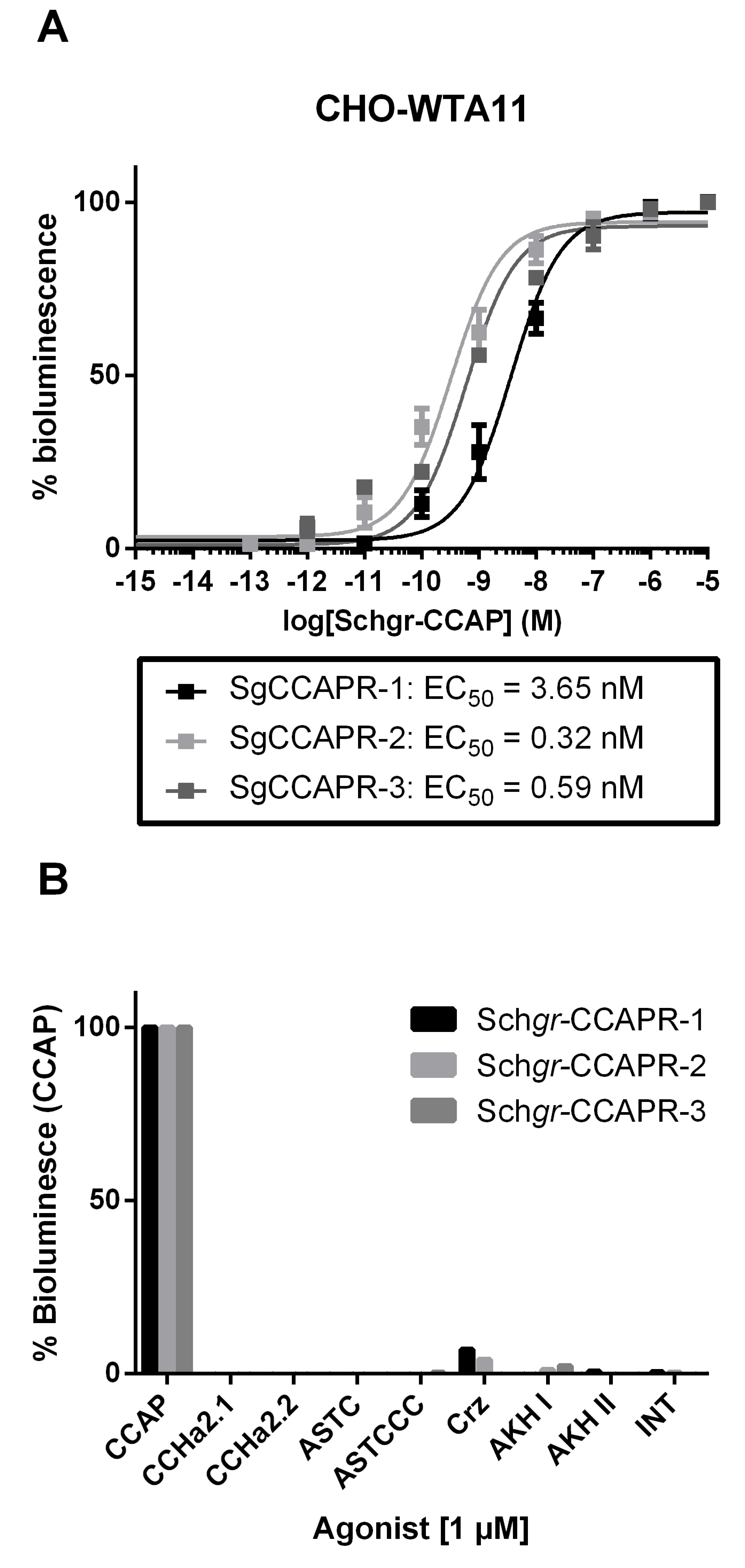
2.2. Identification, Molecular and Functional Characterization of the S. gregaria CCAP Precursor and Receptors
2.3. Localization and Developmental Expression Patterns of the S. gregaria CCAP Precursor and Receptors
2.4. Knockdown of Ecdysteroid Receptor Components Influences Expression of Schgr-CCAPR Transcripts in the Prothoracic Gland
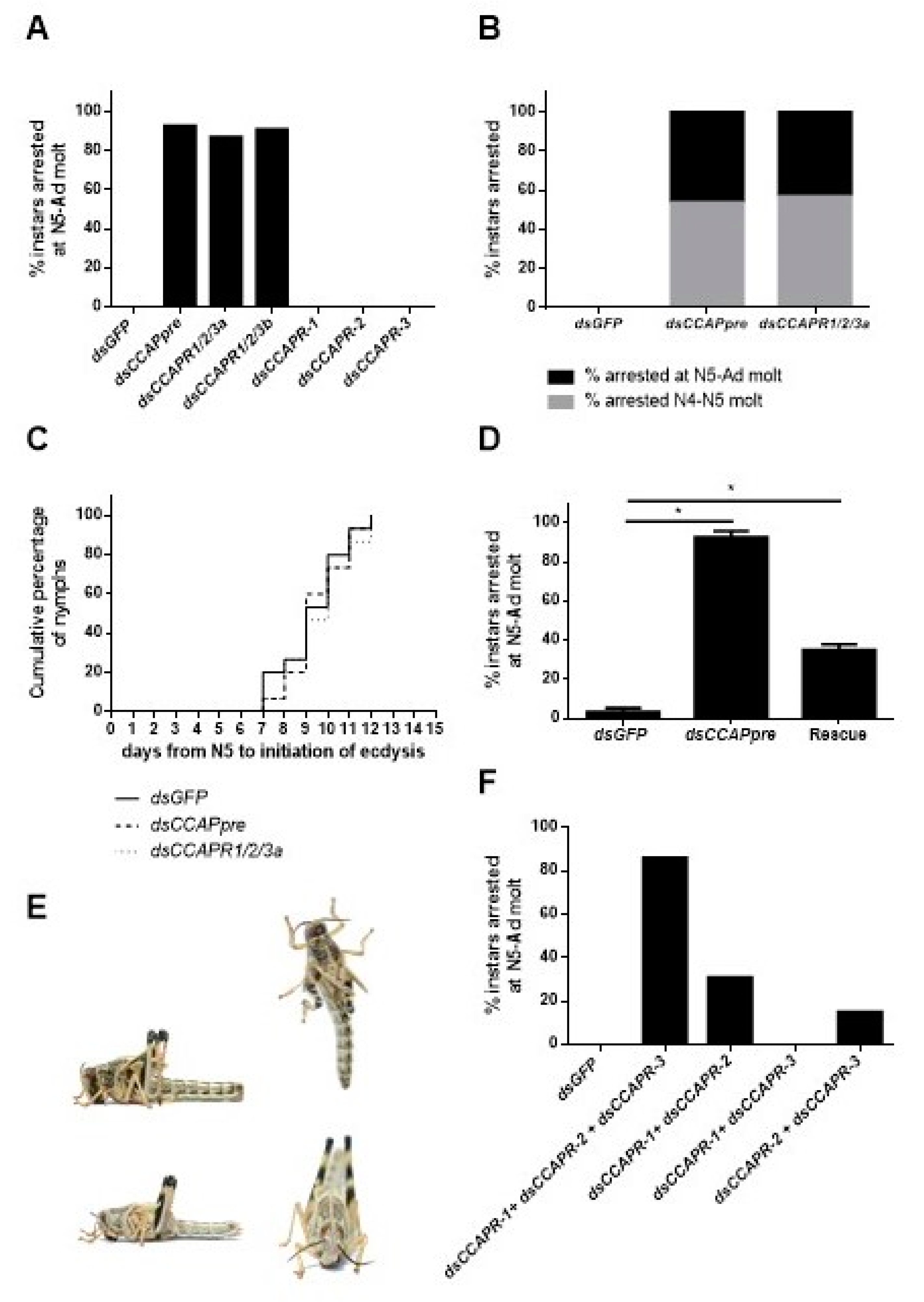
2.5. Phenotypic and Molecular Effects of CCAP Precursor or Receptor RNAi-Mediated Knockdown
2.5.1. Knockdown Efficiency of the dsCCAP(pre/R) Constructs
2.5.2. RNAi Ecdysis Phenotypes
2.5.3. Effects on Halloween Gene Expression in the Prothoracic Glands
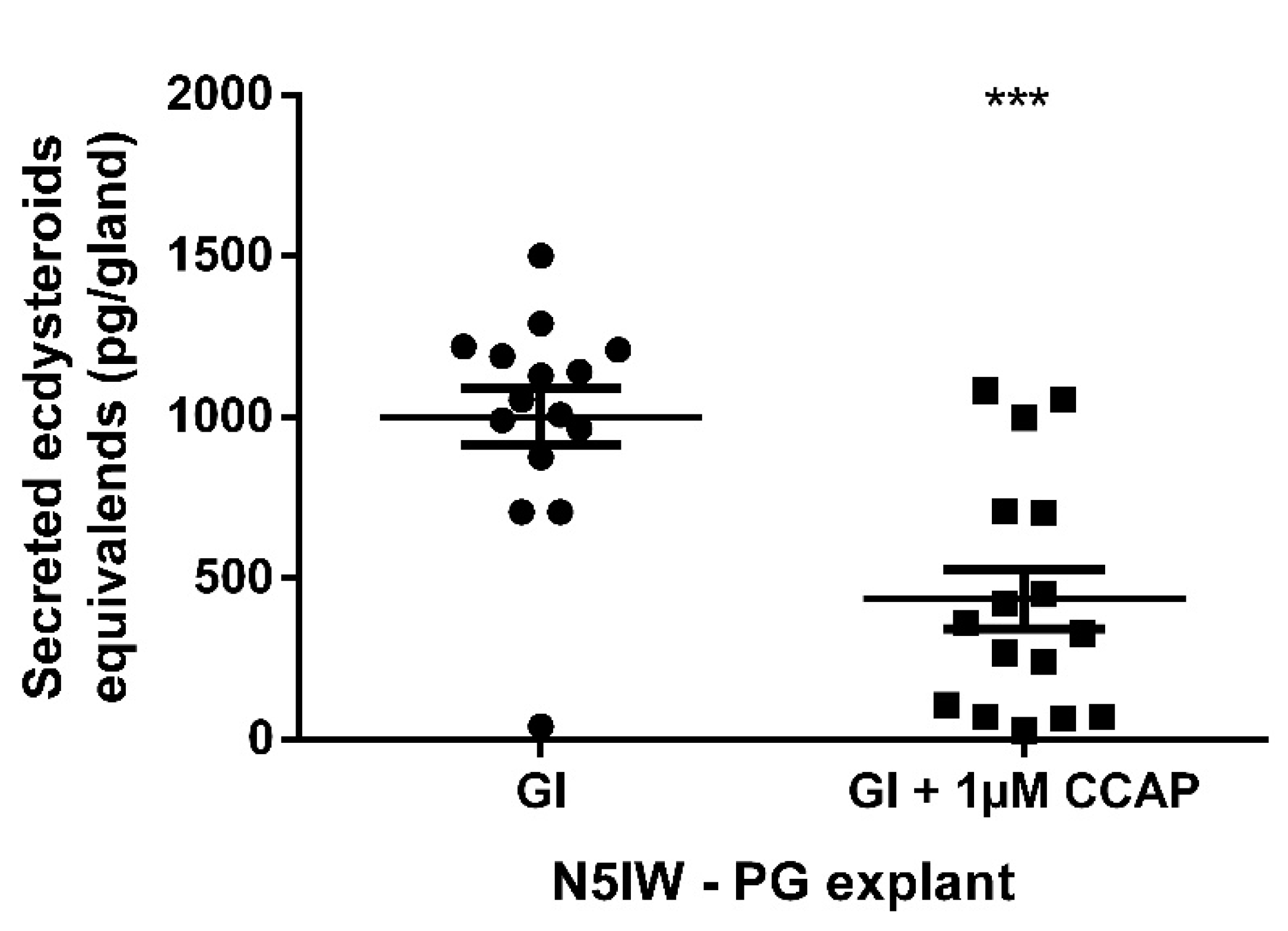
2.6. Effect of CCAP on Ecdysteroidogenesis in the Prothoracic Glands
3. Discussion
3.1. Physical Characteristics Can Be Used to Identify Distinct Points within the Molt Cycle
- The observed rise in interwing distance (IW) correlates with the onset of the molting cycle, approximately marking the peak in circulating ecdysteroids and a significant increase in exuvial space compared to day 4 in the fifth nymphal stage (N5D4).
- The weight peak (WP) coincides with a noticeable and significant increase in the exuvial space compared to N5IW.
- The weight decrease (WD) is associated with an additional noticeable increase in exuvial space compared to N5WP, as well as significant shrinkage of the old endocuticle.
3.2. The Locust’s CCAP Signaling System Is Structurally and Functionally Conserved
3.3. The Imperative Role of the CCAP Signaling System in Ecdysis Is Conserved in S. gregaria
3.4. Identification of a Prothoracicostatic Function of the CCAP Signaling System in S. gregaria
4. Materials and Methods
4.1. Insect Rearing
4.2. Sampling Points Based on Locust Morphology
4.3. 20E Enzyme Immunoassay
4.4. Correlating Locust Morphology to Ecdysteroid Titer
4.5. Histology
4.6. Cell-Based Reporter Assays
4.7. Cloning, Sequence Analysis and Phylogeny of Schgr-CCAPpre and Schgr-CCAPRs
4.8. RNA Interference Experiments
4.9. RNA Extraction, cDNA Synthesis and qRT-PCR
4.10. In Vitro Incubation of Prothoracic Glands
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Marchal, E.; Vandersmissen, H.P.; Badisco, L.; Van de Velde, S.; Verlinden, H.; Iga, M.; Van Wielendaele, P.; Huybrechts, R.; Simonet, G.; Smagghe, G.; et al. Control of ecdysteroidogenesis in prothoracic glands of insects: A review. Peptides 2010, 31, 506–519. [Google Scholar] [CrossRef]
- Pan, X.; Connacher, R.P.; O’Connor, M.B. Control of the insect metamorphic transition by ecdysteroid production and secretion. Curr. Opin. Insect Sci. 2021, 43, 11–20. [Google Scholar] [CrossRef]
- Smith, W.; Rybczynski, R. Prothoracicotropic Hormone; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 9780123847492. [Google Scholar]
- Yamanaka, N.; Hua, Y.J.; Roller, L.; Spalovská-Valachová, I.; Mizoguchi, A.; Kataoka, H.; Tanaka, Y. Bombyx prothoracicostatic peptides activate the sex peptide receptor to regulate ecdysteroid biosynthesis. Proc. Natl. Acad. Sci. USA 2010, 107, 2060–2065. [Google Scholar] [CrossRef]
- Yamanaka, N.; Roller, L.; Žitňan, D.; Satake, H.; Mizoguchi, A.; Kataoka, H.; Tanaka, Y. Bombyx Orcokinins Are Brain-Gut Peptides Involved in the Neuronal Regulation of Ecdysteroidogenesis. J. Comp. Neurol. 2011, 519, 238–246. [Google Scholar] [CrossRef]
- Watanabe, K.; Hull, J.J.; Niimi, T.; Imai, K.; Matsumoto, S.; Yaginuma, T.; Kataoka, H. FXPRL-amide peptides induce ecdysteroidogenesis through a G-protein coupled receptor expressed in the prothoracic gland of Bombyx mori. Mol. Cell. Endocrinol. 2007, 273, 51–58. [Google Scholar] [CrossRef]
- Iga, M.; Nakaoka, T.; Suzuki, Y.; Kataoka, H. Pigment dispersing factor regulates ecdysone biosynthesis via Bombyx neuropeptide G protein coupled receptor-B2 in the prothoracic glands of Bombyx mori. PLoS ONE 2014, 9, e103239. [Google Scholar] [CrossRef]
- Gu, S.H.; Lin, J.L.; Lin, P.L.; Chen, C.H. Insulin stimulates ecdysteroidogenesis by prothoracic glands in the silkworm, Bombyx mori. Insect Biochem. Mol. Biol. 2009, 39, 171–179. [Google Scholar] [CrossRef]
- Kannangara, J.R.; Mirth, C.K.; Warr, C.G. Regulation of ecdysone production in Drosophila by neuropeptides and peptide hormones. Open Biol. 2021, 11, 200373. [Google Scholar] [CrossRef]
- Hamoudi, Z.; Lange, A.B.; Orchard, I. Identification and Characterization of the Corazonin Receptor and Possible Physiological Roles of the Corazonin-Signaling Pathway in Rhodnius prolixus. Front. Neurosci. 2016, 10, 357. [Google Scholar] [CrossRef] [PubMed]
- Zandawala, M.; Orchard, I. Post-feeding physiology in Rhodnius prolixus: The possible role of FGLamide-related allatostatins. Gen. Comp. Endocrinol. 2013, 194, 311–317. [Google Scholar] [CrossRef]
- Žitňan, D.; Adams, M.E. Neuroendocrine Regulation of Ecdysis; Elsevier: Amsterdam, The Netherlands, 2012; ISBN 9780123847492. [Google Scholar]
- White, B.H.; Ewer, J. Neural and Hormonal Control of Postecdysial Behaviors in Insects. Annu. Rev. Entomol. 2014, 59, 363–381. [Google Scholar] [CrossRef] [PubMed]
- Gammie, S.C.; Truman, J.W. Neuropeptide Hierarchies and the Activation of Sequential Motor Behaviors in the Hawkmoth, Manduca sexta. J. Neurosci. 1997, 17, 4389–4397. [Google Scholar] [CrossRef]
- Arakane, Y.; Li, B.; Muthukrishnan, S.; Beeman, R.W.; Kramer, K.J.; Park, Y. Functional analysis of four neuropeptides, EH, ETH, CCAP and bursicon, and their receptors in adult ecdysis behavior of the red flour beetle, Tribolium castaneum. Mech. Dev. 2008, 125, 984–995. [Google Scholar] [CrossRef]
- Lee, D.; Orchard, I.; Lange, A.B. Evidence for a conserved CCAP-signaling pathway controlling ecdysis in a hemimetabolous insect, Rhodnius prolixus. Front. Neurosci. 2013, 7, 1–9. [Google Scholar] [CrossRef]
- Simpson, S.J.; Sword, G.A. Locusts. Curr. Biol. 2008, 18, 364–366. [Google Scholar] [CrossRef] [PubMed]
- Vanden Broeck, J. Insect G Protein-Coupled Receptors and Signal Transduction. Arch. Insect Biochem. Physiol. 2001, 12, 1–12. [Google Scholar] [CrossRef]
- Fredriksson, R.; Lagerström, M.C.; Lundin, L.G.; Schiöth, H.B. The G-protein-coupled receptors in the human genome form five main families. Phylogenetic analysis, paralogon groups, and fingerprints. Mol. Pharmacol. 2003, 63, 1256–1272. [Google Scholar] [CrossRef] [PubMed]
- Petersen, T.N.; Brunak, S.; von Heijne, G.; Nielsen, H. SignalP 4.0: Discriminating signal peptides from transmembrane regions. Nat. Methods 2011, 8, 785–786. [Google Scholar] [CrossRef]
- Belles, X. Insect Metamorphosis: From Natural History to Regulation of Development and Evolution, 1st ed.; Elsevier (Academic Press): Amsterdam, The Netherlands, 2020; ISBN 978-0-12-813020-9. [Google Scholar]
- Tu, X.; Zhang, Z.; Johnson, D.L.; Cao, G.; Li, Z.; Gao, S.; Nong, X.; Wang, G. Growth, development and daily change in body weight of Locusta migratoria manilensis (Orthoptera: Acrididae) nymphs at different temperatures. J. Orthoptera Res. 2012, 21, 133–140. [Google Scholar] [CrossRef]
- Loi, P.K.; Emmal, S.A.; Park, Y.; Tublitz, N.J. Identification, sequence and expression of a crustacean cardioactive peptide (CCAP) gene in the moth Manduca sexta. J. Exp. Biol. 2001, 204, 2803–2816. [Google Scholar] [CrossRef]
- Shi, Y.; Liu, T.Y.; Pei, Y.X.; Jiang, H.B.; Dou, W.; Smagghe, G.; Wang, J.J. Crustacean cardioactive peptide (CCAP) of the oriental fruit fly, Bactrocera dorsalis (Diptera: Tephritidae): Molecular characterization, distribution and its potential roles in larva-pupa ecdysis. Peptides 2018, 122, 7. [Google Scholar] [CrossRef]
- Lee, D.H.; Paluzzi, J.P.; Orchard, I.; Lange, A.B. Isolation, cloning and expression of the crustacean cardioactive peptide gene in the Chagas’ disease vector, Rhodnius prolixus. Peptides 2011, 32, 475–482. [Google Scholar] [CrossRef]
- Li, B.; Beeman, R.; Park, Y. Functions of duplicated genes encoding CCAP receptors in the red flour beetle, Tribolium castaneum. J. Insect Physiol. 2011, 57, 1190–1197. [Google Scholar] [CrossRef]
- Cazzamali, G.; Hauser, F.; Kobberup, S.; Williamson, M.; Grimmelikhuijzen, C.J.P. Molecular identification of a Drosophila G protein-coupled receptor specific for crustacean cardioactive peptide. Biochem. Biophys. Res. Commun. 2003, 303, 146–152. [Google Scholar] [CrossRef]
- Lee, D.; Vanden Broeck, J.; Lange, A.B. Identification and Expression of the CCAP Receptor in the Chagas’ Disease Vector, Rhodnius prolixus, and Its Involvement in Cardiac Control. PLoS ONE 2013, 8, e68897. [Google Scholar] [CrossRef]
- Estévez-lao, T.Y.; Boyce, D.S.; Honegger, H.; Hillyer, J.F. Cardioacceleratory function of the neurohormone CCAP in the mosquito Anopheles gambiae. J. Exp. Biol. 2013, 216, 601–613. [Google Scholar] [CrossRef][Green Version]
- Belmont, M.; Cazzamali, G.; Williamson, M.; Hauser, F.; Grimmelikhuijzen, C.J.P. Identification of four evolutionarily related G protein-coupled receptors from the malaria mosquito Anopheles gambiae. Biochem. Biophys. Res. Commun. 2006, 344, 160–165. [Google Scholar] [CrossRef]
- Mirabeau, O.; Joly, J. Molecular evolution of peptidergic signaling systems in bilaterians. Proc. Natl. Acad. Sci. USA 2013, 110, 2028–2037. [Google Scholar] [CrossRef]
- Donini, A.; Lange, A.B. Crustacean cardioactive peptide is a modulator of oviduct contractions in Locusta migratoria. J. Insect Physiol. 2001, 47, 277–285. [Google Scholar] [CrossRef]
- Kim, Y.J.; Žitňan, D.; Cho, K.H.; Schooley, D.A.; Misoguchi, A.; Adams, M.E. Central peptidergic ensembles associated with organization of an innate behavior. Proc. Natl. Acad. Sci. USA 2006, 103, 14211–14216. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Schroeder, A.J.; Helfrich-förster, C.; Jackson, F.R.; Ewer, J. Targeted ablation of CCAP neuropeptide-containing neurons of Drosophila causes specific defects in execution and circadian timing of behavior. Development 2003, 130, 2645–2656. [Google Scholar] [CrossRef]
- Kamruzzaman, A.S.M.; Mikani, A.; Mohamed, A.A.; Elgendy, A.M.; Takeda, M. Crosstalk among Indoleamines, Neuropeptides and JH/20E in Regulation of Reproduction in the American Cockroach, Periplaneta americana. Insects 2020, 11, 155. [Google Scholar] [CrossRef] [PubMed]
- Lenaerts, C.; Cools, D.; Verdonck, R.; Verbakel, L.; Vanden Broeck, J.; Marchal, E. The ecdysis triggering hormone system is essential for successful moulting of a major hemimetabolous pest insect, Schistocerca gregaria. Sci. Rep. 2017, 7, 46502. [Google Scholar] [CrossRef]
- Marchal, E.; Badisco, L.; Verlinden, H.; Vandersmissen, T.; Van Soest, S.; Van Wielendaele, P.; Vanden Broeck, J. Role of the Halloween genes, Spook and Phantom in ecdysteroidogenesis in the desert locust, Schistocerca gregaria. J. Insect Physiol. 2011, 57, 1240–1248. [Google Scholar] [CrossRef]
- Verlinden, H.; Sterck, L.; Li, J.; Li, Z.; Yssel, A.; Gansemans, Y.; Verdonck, R.; Holtof, M.; Song, H.; Behmer, S.T.; et al. First draft genome assembly of the desert locust, Schistocerca gregaria. F1000Research 2020, 9, 775. [Google Scholar] [CrossRef]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Lenaerts, C.; Van Wielendaele, P.; Peeters, P.; Vanden Broeck, J.; Marchal, E. Ecdysteroid signalling components in metamorphosis and development of the desert locust, Schistocerca gregaria. Insect Biochem. Mol. Biol. 2016, 75, 10–23. [Google Scholar] [CrossRef]
- Mikani, A.; Watari, Y.; Takeda, M. Brain-midgut cross-talk and autocrine metabolastat via the sNPF/CCAP negative feed-back loop in the American cockroach, Periplaneta americana. Cell Tissue Res. 2015, 362, 481–496. [Google Scholar] [CrossRef]
- Williams, M.J.; Akram, M.; Barkauskaite, D.; Patil, S.; Kotsidou, E.; Kheder, S.; Vitale, G.; Filaferro, M.; Blemings, S.W.; Maestri, G.; et al. CCAP regulates feeding behavior via the NPF pathway in Drosophila adults. Proc. Natl. Acad. Sci. USA 2020, 117, 7401–7408. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verbakel, L.; Lenaerts, C.; Abou El Asrar, R.; Zandecki, C.; Bruyninckx, E.; Monjon, E.; Marchal, E.; Vanden Broeck, J. Prothoracicostatic Activity of the Ecdysis-Regulating Neuropeptide Crustacean Cardioactive Peptide (CCAP) in the Desert Locust. Int. J. Mol. Sci. 2021, 22, 13465. https://doi.org/10.3390/ijms222413465
Verbakel L, Lenaerts C, Abou El Asrar R, Zandecki C, Bruyninckx E, Monjon E, Marchal E, Vanden Broeck J. Prothoracicostatic Activity of the Ecdysis-Regulating Neuropeptide Crustacean Cardioactive Peptide (CCAP) in the Desert Locust. International Journal of Molecular Sciences. 2021; 22(24):13465. https://doi.org/10.3390/ijms222413465
Chicago/Turabian StyleVerbakel, Lina, Cynthia Lenaerts, Rania Abou El Asrar, Caroline Zandecki, Evert Bruyninckx, Emilie Monjon, Elisabeth Marchal, and Jozef Vanden Broeck. 2021. "Prothoracicostatic Activity of the Ecdysis-Regulating Neuropeptide Crustacean Cardioactive Peptide (CCAP) in the Desert Locust" International Journal of Molecular Sciences 22, no. 24: 13465. https://doi.org/10.3390/ijms222413465
APA StyleVerbakel, L., Lenaerts, C., Abou El Asrar, R., Zandecki, C., Bruyninckx, E., Monjon, E., Marchal, E., & Vanden Broeck, J. (2021). Prothoracicostatic Activity of the Ecdysis-Regulating Neuropeptide Crustacean Cardioactive Peptide (CCAP) in the Desert Locust. International Journal of Molecular Sciences, 22(24), 13465. https://doi.org/10.3390/ijms222413465





