Abstract
Testicular germ cell tumors (TGCTs) are the leading form of solid cancer and death affecting males between the ages of 20 and 40. Today, their surgical resection and chemotherapy are the treatments of first choice, even if sometimes this is not enough to save the lives of patients with TGCT. As seen for several tumors, the deregulation of microRNAs (miRNAs) is also a key feature in TGCTs. miRNAs are small molecules of RNA with biological activity that are released into biological fluids by testicular cancer cells. Their presence, therefore, can be detected and monitored by considering miRNAs as diagnostic and prognostic markers for TGCTs. The purpose of this review is to collect all the studies executed on miRNAs that have a potential role as biomarkers for testicular tumors.
1. Introduction
One of the most recurrent tumors in adolescent and young adults with the highest incidence between the ages of 20 and 40 years is testicular cancer [1,2]. Different testicular tumors require histological analyses for their therapies and prognosis. Testicular tumors are classified and divided into different subgroups by the International Agency for Research in Cancer of the World Health Organization (WHO). Other than sex cord-stromal tumors (Sertoli cell tumor, Leydig cell tumor), testicular germ cell tumors (TGCTs) occupy the main clinical role considering their frequent occurrence and the possibility of curing them, even when metastases are present, thanks to the introduction of cisplatin-based chemotherapy [3,4,5]. Among all genitourinary carcinomas [6], TGCTs have doubled their incidence because of unidentified environmental factors, undescended testes and positive family history [5]. In spite of the good results obtained with cisplatin treatment, numerous short- and long-term toxicity effects, such as thromboembolism, cardiovascular toxicity and neutropenic complications, are still reported [7,8,9,10]. Once patients tolerate risk-adapted chemotherapy, high cure rates can be achieved in aged patients with metastatic TGCT, as well as in younger patients [10,11]. Even though clinical examination is the diagnostic basis (scrotal ultrasound and high-resolution computed tomography), serum tumor markers maintain their clinical relevance. For instance, when there are suspected cases of testicular cancer, α-fetoprotein (AFP), the β-subunit of chorionic gonadotropin (β-hCG), as well as Lactate dehydrogenase (LDH), are analyzed [12]. In particular, β-hCG and AFP permit us to define TGCT diagnosis, histology, classification and prognosis. These markers are also used to monitor patients during active surveillance and for disease recurrence. Unfortunately, false positive or negative results can always arise [13,14]. Consequently, many studies have been conducted to find and utilize new tumor molecular markers with higher specificity and sensitivity. A new interesting class of tumor molecular markers is blood-circulating miRNAs. miRNAs are 19–22 nucleotides, in a single stranded shape, that thanks to a specific sequence act as post-transcriptional regulators modulating gene expression. miRNA genes are transcribed by RNA polymerase II, which generates a long primary transcript called pri-MiRNA [15]. Subsequently, the miRNA maturation process involves two main steps engaging RNase-III enzymes and double-stranded RNA-binding domain (dsRBD) proteins [15]. The nuclear RNase III-type enzyme Drosha is responsible for the production of a pre-miRNA, a hairpin precursor consisting of around 70 nt, through the maturation of the pri-miRNA. Then, the pre-miRNA is exported to the cytoplasm, where the RNase III protein Dicer processes it into an unstable miRNA duplex structure. Finally, the RNA-induced silencing complex (RISC) loads the less stable strand of the duplex into a nuclease complex that is able to regulate protein expression [15].
Indeed, the miRNA cluster 371–373, but also miRNAs such as miR-223-3, miR-449, miR-383, miR-514a-3p, miR-199a-3p and miR-214, have been proposed as new potential tumor markers [16]. So, the study and the clinical application of these new tumor markers is very interesting for many fields of research worldwide [17]. In this review, the clinical importance of previously offered tumor markers is debated and compared with new serological markers and detection methods, such as the quantification of miRNAs, so as to underline the new diagnostic options.
2. Novel TGCTs Biomarkers
2.1. HMGA Protein Family
The HMGA proteins, meaning proteins with high electrophoretic mobility, are made up of the proteins HMGA1a and HMGA1b, encoded via alternative splicing by the same gene HMGA1, and by the HMGA2 protein, encoded by the HMGA2 gene [18,19]. Their main feature is that of possessing domains, called AT-hooks, that are able to bind DNA at the level of regions rich in adenine and thymine residues [19,20]. Although HMGA proteins participate in the transcriptional regulation of numerous genes, they are not transcriptional factors but chromatin architectural factors, with the ability to form protein complexes at the level of gene promoters and enhancers. The proteins HMGA1 and HMGA2 are strongly expressed during embryogenesis, while their expression becomes almost zero or completely absent in adult tissues. Nevertheless, their expression has been found to be upregulated in several human tumors, and it has also been attributed a causal role in neoplastic transformation [21,22]. Indeed, in vitro and in vivo experiments have clearly demonstrated that HMGA proteins are oncogenes. For example, both hmga1 and hmga2 are capable of transforming rat and murine fibroblasts, and hmga1 or hmga2 transgenic mice develop pituitary adenomas and NK-T lymphomas [19].
In the past, we have shown that HMGA1 is expressed in spermatogonia and primary spermatocytes (mitotic cells), whereas HMGA2 is expressed in secondary spermatocytes and spermatids (meiotic cells) [23]. In support of this, we have concluded that the spermatogenesis differentiation program in hmga2 knock-out mice is strongly compromised. In addition, we have shown that in the event of a controversial TGCT diagnosis, HMGA proteins can be considered an advantageous diagnostic tool [24]. In fact, we have reported that HMGA proteins are expressed depending on the differentiation state of TGCTs: in pluripotential embryonal carcinoma cells, HMGA1 and HMGA2 have been reportedly overexpressed, in mature adult tissue of teratoma areas both proteins have not been detected, in seminomas just HMGA1 have been found to be overexpressed, and in yolk sac tumors just HMGA2 [24].
2.2. PATZ
PATZ1 is a transcriptional repressor that, thanks to its POZ domain, binds to the promoters of numerous genes, regulating their basal activity. In order to study its biological role, a knock-out mouse model for the PATZ1 gene was generated [25]. Interestingly, male patz1 −/− mice were infertile, highlighting a key role for this gene in spermatogenesis. Indeed, adult mice showed just a few apoptotic spermatocytes, other than a lack of spermatids and spermatozoa, leading to an alteration of the normal tubular structure. By northern blot, western blot and immunohistochemistry assays, PATZ was found expressed in Sertoli cells and in the spermatogonia [26]. Considering that PATZ1 is a tumor suppressor gene, its role in TGCTs has been investigated. Indeed, PATZ1 tumor suppressor activity has been found to be impaired in TGCT because of its delocalization into the cytoplasm of cancer cells. This delocalization is likely due to the downregulation of the estrogen receptor β, a PATZ1-interacting molecular partner. In fact, it has been shown that PATZ1 interacts with ERβ in normal germ cells, while the downregulation of ERβ associates with PATZ1 and HMGA1 cytoplasmic delocalization in seminomas [27,28].
These data suggest a crucial role for PATZ1 in normal male gametogenesis, whereas its overexpression and delocalization could be connected to the promotion of TGCTs.
2.3. G Protein-Coupled Oestrogen Receptor (GPR30)
Oestrogens are key hormones playing roles in testis physiology. Indeed, the estrogen receptors α (ERα) and estrogen receptors β (ERβ) are strongly expressed in specific testis cells. Whereas ERβ is almost ubiquitous, ERα is more specific, being expressed in Leydig cells and in the epithelium of efferent ductules [29,30]. Recent studies have underlined the role of other estrogen mediators, such as G protein-coupled estrogen receptor 30 (GPR30). Indeed, GPR30 has been widely accepted as a key regulator of testis pathophysiology [31,32]. Interestingly, its expression has been found in normal human spermatogonia and in spermatocytes, as well as in seminoma samples [32]. Moreover, a strong GPR30 overexpression was found in human carcinoma in situ (CIS) and in seminomas where the ERβ levels were downregulated, thus negatively correlating. Furthermore, GPR30 is able to activate the ERK1/2 signaling pathway by binding to 17β-estradiol in the seminoma-derived cell line (TCam-2) [33]. Thus, ongoing studies are trying to use GPR30 as a biomarker for human seminoma, as well as to design novel therapies to halt TGCT development by targeting this receptor [32,33].
2.4. Aurora B Kinase
Very often, human cancer is induced by the alteration of several cell cycle regulators [34]. In TGCTs, one of the best-studied cell cycle regulators is Aurora B Kinase. Indeed, Aurora B, which is the predominant kinase during G2-M transition, is able to phosphorylate serine 10 of histone H3, a histone modification deeply involved in chromosome condensation, alignment and segregation. Moreover, Aurora B is a key regulator of the spindle check point and cytokinesis [34]. As a molecular marker, this kinase has been used to discriminate the different TGCT histotypes: Aurora B expression has been found in intratubular germ cell neoplasia (IGCNU), seminoma and embryonal carcinoma, whereas it is absent in teratoma and yolk sac tumors (YSTs) [35,36,37]. Furthermore, Aurora B represents a valuable target for therapy. Indeed, its inhibition is able to drastically reduce cell proliferation in two testis cell lines (GC1 and TCam-2) [38]. Several compounds are actually in early clinical evaluation to test their ability to block tumor growth on a wide panel of human cancer types.
2.5. microRNAs in TGCT: From the Pathogenesis to a Novel Tumoral Biomarker Role
In the last few years, more and more research groups have focused their studies on a novel class of short non-coding RNAs, which are transcribed but not translated: miRNAs [39]. This class is constituted of small RNA molecules of about 19–22 nucleotides that are able to bind the 3′unuranslated regions (UTRs) of several transcripts, leading to the degradation of the targeted mRNAs, or to block the protein translation [17,40]. Thus, miRNAs became key molecules involved in both physiological and pathological processes. In particular, cancer research has been revolutionized by miRNAs discovery, since the deregulation of several miRNAs has been involved in the pathogenesis of many human cancer types [41,42,43]. Indeed, this class of short non-coding RNAs is necessary for a variety of cell mechanisms such as cell cycle regulation, migration/invasion, drug resistance and immune response [44,45,46]. Given their importance in cancer phenomena, alterations of the miRNA signature in cancer cells vs. normal cells have been used to better determine the diagnosis, the prognosis and the response to cancer treatment in several human cancers [47]. More recently, miRNAs have been found also in the extracellular milieu with signal transduction functions. Indeed, their structure is highly stable in the blood stream, and they can be used for cell–cell communication in an autocrine, paracrine or endocrine manner [48]. Moreover, they can be released following cell death [49]. Thus, their abundance in serum and the relative easiness of detection makes them suitable also as potential circulating tumor markers.
2.6. miR-302/367 Cluster
The cell cycle of embryonic (ESC) and pluripotent stem cells (PSC) is in part regulated by the miR-302 cluster. In fact, miR-302 induces the fast proliferation of these cells interacting with promoters and inhibitors of genes that regulate the cell cycle [50].
Moreover, the miR-302 cluster influences cell cycle progression by modifying histone methylations and the Akt/PKB pathway [51]. Akt oncogene expression has been found to be suppressed by miR-302 in teratomas. In this context, OCT4, a pluripotent transcription factor, is not still inhibited by Akt. On the contrary, Akt upregulation, as a result of miR-302 downregulation, causes low OCT4 expression. Indeed, OCT4 overexpression is necessary for teratoma formation and promotes PSCs pluripotent skills. It has been demonstrated that miR-302 accelerates the cell cycle switch from the G1 to the S phase, inhibiting cyclin-dependent kinase (CDK) 2 and 4 [52,53]. The blood of TGCT patients contains high levels of miR-302, whereas this is downregulated in liver, stomach and colon cancer [54,55,56,57]. Interestingly, the levels of miR-302a-3p, miR-302b-3p and miR-302c-3p diminished in TGCT cell lines after cisplatin treatment [52].
Sprouty RTK Signaling Antagonist 4 (SPRY4), which alters the PI3K/Akt signaling pathway, has been found overexpressed in TGCT. A possible role for miR-302 in the growth of TGCTs is proven by the fact that its inhibition results in a SPRY4 suppression that in turn decreases TGCT growth and invasion [57]. In conclusion, as demonstrated by Murray et al., miR-302 upregulation has been detected in all blood samples analyzed from TGCT patients [58].
Strictly associated with miR-302, there is miR-367. Whereas their sequences have some differences, they share several mRNAs as targets [51]. Similar to miR-302, miR-367 is able to regulate cancer-related pathways and cell cycles [59]. miR-367-3p serum levels were used to determine TGCT tumoral stage. Interestingly, miR-367-3p was strongly upregulated in TGCT patients compared to healthy ones. Moreover, its serum levels were decreased in Stage I and Stage II TGCTs compared with Stage III, where they strongly diminished after orchiectomy. However, miR-367-3p serum levels failed to detect teratoma and possible TGCT precursors, such as germ cell neoplasia in situ (GCNIS), or benign lesions [60,61]. Although some positive results were achieved by using miR-367-3p as a marker of chemotherapy-resistant germinal tumors [62], several other studies reported that miR-371-3p has a higher sensitivity as a marker of TGCT disease and/or relapse [63,64], thus further studies are needed.
2.7. miR-371-3 Cluster
The miR-371-3 locus represents the main miRNA cluster involved in TGCT tumorigenesis. It is settled on chromosome 19, and consists of a polycistronic miRNA transcript (pri-miR-371-3) that is then cleaved in four different molecules: miR-371, miR-372, miR-373 and miR-373* [16,65]. The upregulation of this cluster has been found in a number of human cancers, since it is involved in cell proliferation [66,67,68], drug resistance [69], migration and invasion [70,71]. In TGCTs, this cluster has been found to be mainly overexpressed. Interestingly, it has been reported that miR-372 and miR-373 are able to reduce the expression of the Large Tumor Suppressor Kinase 2 (LATS2), thus inhibiting p53 activity and triggering the accumulation of DNA mutations [72,73]. Moreover, Zhou et al. reported that the miR-371-3 cluster may activate the Wnt/β-catenin pathway and downregulate the Dickkopf-1 (DKK1) protein, thus sustaining cell proliferation and the invasion properties of cancer cells [74]. Although many papers have underlined the molecular mechanisms by which this cluster participates in tumor transformation, only miR-371a-3p has been extensively analyzed as a TGCT biomarker in follow-up, staging and diagnosis, and to estimate TGCT prognosis. Indeed, miR-371a-3p has been reported also as a powerful diagnostic tool able to discriminate various testicular histotypes. Briefly, the miR-371a-3p levels were assessed in the histological sections of different TGCT specimens by qRT-PCR. As expected, miR expression levels were found to be increased in TGCT samples compared to normal testis tissues, and TGCTs derived from GCNIS showed higher miR-371a-3p levels than non-GCNIS tumors. Importantly, seminomas patients displayed the highest miR-371a-3p expression, followed by embryonic carcinomas, teratomas and yolk sac tumors (Table 1) [75]. Furthermore, miR-371a-3p seems to be the first miRNA suitable for assessing the presence of metastatic disease and monitoring treatment success in cisplatin-treated TGCTs patients [16].

Table 1.
miR-371a-3p levels as discriminative diagnostic tool.
2.8. miR-517/519
Near the miR-371-373 cluster, there are three miRNAs, miR-517a-3p, miR-519a-3p, and miR-519c-3p, that belong to the same chromosome miRNA cluster (C19MC) [76]. In several tumors, it has been found that the upregulation of these three miRNAs is responsible for their enhanced migration, invasion, and poor overall survival [77,78]. Flor et al. studied the expressions of miR-517a-3p, miR-519a-3p, and miR-519c-3p in TGCTs. They found the same or a lower expression of these three miRNAs in Stage I seminoma and teratoma mixed tumors in comparison with normal testis. On the contrary, high miRNAs expression was detected in non-seminomatous and Stage III mixed tumors.
These data sustain the role of miR-517a-3p, miR-519a-3p and miR-519c-3p as a novel tumor marker for advanced-stage and non-seminomatous tumors. Interestingly, following tumor resection, their levels in the serum were reduced [79].
2.9. miR-223-3p
The scientific literature reports that miR-223-3p has been found to be deregulated in gastric and esophageal tumors, as well as in acute T-cell lymphoblastic leukemia [80,81,82]. In prostate cancer (PCa) tissue, miR-223-3p expression is recurrently reduced in comparison to the normal tissues, where miR-223-3p works as a tumor suppressor preventing cancer cell migration and invasion [83]. Moreover, miR-223-3p is upregulated in TGCTs with respect to normal testis. In fact, the Cancer Genome Atlas (TCGA) database and published data set reported a higher miR-223-3p expression in tumors than in normal testes. Additionally, a negative correlation was found between miR-223-3p and FBXW7 mRNA expression levels. In TGCT tumors, miR-223-3p upregulation provokes the inhibition of apoptosis mediated by the F-box/WD repeat-containing protein 7 (FBXW7), which belongs to a protein complex acting as a tumor suppressor that sustains the degradation of oncoproteins, including c-Myc, cyclin E, MCL-1, c-JUN, NFκB2 and Notch1 [84]. Additionally, the ectopic expression of the full-length coding sequence of FBXW7 rescued the cell growth and apoptosis mediated by miR-223-3p. As demonstrated in gastric cancer [85], miR-223-3p may influence chemotherapeutic agents’ sensitivity in TGCTs. Finally, more studies are needed to use miR-223-3p as a tumor marker.
2.10. miR-449
A main role in spermatogenesis has been found for miR-449. In fact, even though miR-34b/c and miR-449 are not necessary for male fertility, mutual ablation impedes normal spermatogenesis and provokes male infertility [86].
The transcription factor E2F is able to regulate miR-449a and miR-449b transcription [87]. Cell cycle and apoptosis are strictly modulated by E2F. miR-449a can counteract the progression of the cell cycle by reducing the expression of Cyclin-Dependent Kinase 6 (CDK6) [88]. miR-449a/b structurally calls to mind the miRNA 34 family, which is induced by p53. Consistently with a supposed tumor-suppressive role, miR-449a, along with miR-34a, inhibited proliferation and promoted apoptosis by at least partially p53-independent mechanisms. In testicular cancer, miR-449a expression was low or absent, whereas it is expressed in healthy testicular tissue. This is likely due to retinoblastoma mutations (pRB), which are not still able to bind to E2F. Therefore, a low amount of E2F brings about a downregulation of miR-449a that promotes cell cycle progression in tumor cells [89].
2.11. miR-383
miR-383 regulates the different biological activities of TGCTs, such as apoptosis, proliferation and cell cycle regulations [90,91]. It has been found to be overexpressed in embryonic carcinomas, while it is downregulated in infertile men. miR-383 binds to interferon regulatory factor-1 (IRF1), leading to its downregulation. Decreased IRF1 expression provokes the downregulation of its molecular partners involved in cell cycle regulation, such as cyclin D1, CDK2 and p21 [92]. As described for miR-449, miR-383 regulates the pRB protein by binding to IRF1 mRNA in embryonic carcinomas [90]. In embryonic carcinomas, the inhibition of H2AX, the DNA damage marker, is likely due to miR-383 overexpression. This phenomenon increases the tumor’s sensitivity to cisplatin and suggests for miR-383 the role of potential target in embryonic carcinomas [93].
2.12. Let-7a and miR-26a
Let-7a and miR-26a act as tumor suppressor miRNAs in different human cancer types [94,95,96]. Consistently, De Martino et al. showed that Let-7a and miR-26a were downregulated also in human seminoma. Moreover, they demonstrated that Let-7a and miR-26a were able to inhibit seminoma cell growth and mobility by inhibiting HMGA1 levels [97].
Intriguingly, Let-7a and miR-26a’s inhibitory effects on seminoma cell growth could produce new understandings from therapeutic perspectives. In reality, new therapy approaches may restore the normal levels of Let-7a and miR-26a in seminomas by administering synthetic miRNAs [97].
3. Conclusions
This paper aimed to review the canonical and emerging tumoral markers for TGCT disease. Indeed, until now, several markers have been used in diagnosis and prognosis; however, novel molecules, such as miRNAs, may be of great potential as tumor markers or therapeutics, even though larger studies are needed (Figure 1). Indeed, the advantages of using miRNAs as tumoral markers are numerous. First of all, they are easy to obtain without severe damage for the patients. They can be simply collected from blood, urine and semen. Moreover, the current miRNAs detection methods, such as polymerase chain reaction (PCR), make circulating miRNAs more sensitive biomarkers in comparison with traditional biopsies and/or image examinations. Finally, the expression pattern of miRNAs gives the opportunity to assess the stage and progression of tumors in real time and in a dynamic manner, from tumorigenesis throughout the following progression. Therefore, miRNAs show a prospective capacity to serve as non-invasive cancer biomarkers for clinical application. They may be applied in many aspects, such as cancer screening, subtype classification and drug sensitivity prediction for treatment strategy selection.
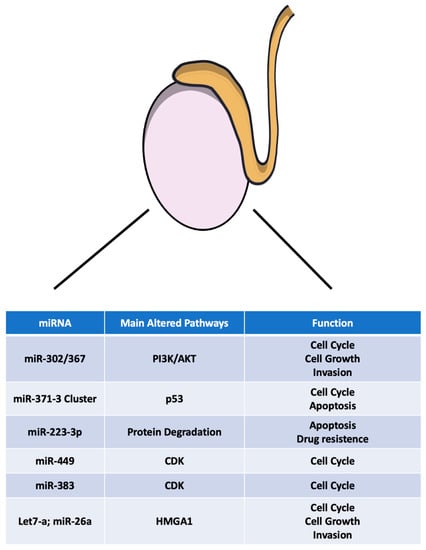
Figure 1.
Main microRNAs deregulated in TGCTs. PI3K: Phosphoinositide 3-kinase; AKT: CDK: cyclin-dependent kinase.
However, numerous independent validation studies are still required to improve the use of miRNAs in clinical applications. A main contribution could come from the combination of miRNAs with other canonical biomarkers, which could also result in a comprehensive view of miRNAs, known and unknown, long non-coding RNAs (lncRNAs), circular-RNAs, and other ncRNAs.
Funding
This paper has not received external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Chieffi, P. Molecular targets for the treatment of testicular germ cell tumors. Mini Rev. Med. Chem. 2007, 7, 755–759. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.; De Martino, M.; Esposito, F. New Anti-Cancer Strategies in Testicular Germ Cell Tumors. Recent Pat. Anticancer Drug Discov. 2019, 14, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Moch, H.; Cubilla, A.L.; Humphrey, P.A.; Reuter, V.E.; Ulbright, T.M. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs-Part A: Renal, Penile, and Testicular Tumours. Eur. Urol. 2016, 70, 93–105. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.; Franco, R.; Portella, G. Molecular and cell biology of testicular germ cell tumors. Int. Rev. Cell Mol. Biol. 2009, 278, 277–308. [Google Scholar] [CrossRef]
- Chieffi, P.; Chieffi, S.; Franco, R.; Sinisi, A.A. Recent advances in the biology of germ cell tumors: Implications for the diagnosis and treatment. J. Endocrinol. Investig. 2012, 35, 1015–1020. [Google Scholar] [CrossRef]
- Marra, L.; Cantile, M.; Scognamiglio, G.; Perdona, S.; La Mantia, E.; Cerrone, M.; Gigantino, V.; Cillo, C.; Caraglia, M.; Pignata, S.; et al. Deregulation of HOX B13 expression in urinary bladder cancer progression. Curr. Med. Chem. 2013, 20, 833–839. [Google Scholar]
- Bezan, A.; Posch, F.; Ploner, F.; Bauernhofer, T.; Pichler, M.; Szkandera, J.; Hutterer, G.C.; Pummer, K.; Gary, T.; Samonigg, H.; et al. Risk stratification for venous thromboembolism in patients with testicular germ cell tumors. PLoS ONE 2017, 12, e0176283. [Google Scholar] [CrossRef]
- Terbuch, A.; Posch, F.; Annerer, L.M.; Bauernhofer, T.; Pichler, M.; Szkandera, J.; Hutterer, G.C.; Pummer, K.; Partl, R.; Kapp, K.S.; et al. Long-term cardiovascular complications in stage I seminoma patients. Clin. Transl. Oncol. 2017, 19, 1400–1408. [Google Scholar] [CrossRef]
- Terbuch, A.; Posch, F.; Partl, R.; Zurl, B.; Bauernhofer, T.; Pichler, M.; Szkandera, J.; Hutterer, G.C.; Pummer, K.; Kapp, K.S.; et al. Risk stratification for febrile neutropenia in patients with testicular germ cell tumors. Cancer Med. 2018, 7, 508–514. [Google Scholar] [CrossRef]
- Terbuch, A.; Posch, F.; Bauernhofer, T.; Pichler, M.; Peinsith, H.; Szkandera, J.; Riedl, J.; Hutterer, G.C.; Pummer, K.; Partl, R.; et al. Age as a Predictor of Treatment Outcome in Metastatic Testicular Germ Cell Tumors. Anticancer Res. 2019, 39, 5589–5596. [Google Scholar] [CrossRef]
- Chieffi, P. Recent advances in molecular and cell biology of testicular germ-cell tumors. Int. Rev. Cell Mol. Biol. 2014, 312, 79–100. [Google Scholar] [CrossRef] [PubMed]
- Khan, O.; Protheroe, A. Testis cancer. Postgrad. Med. J. 2007, 83, 624–632. [Google Scholar] [CrossRef] [PubMed]
- Batool, A.; Karimi, N.; Wu, X.N.; Chen, S.R.; Liu, Y.X. Testicular germ cell tumor: A comprehensive review. Cell Mol. Life Sci. 2019, 76, 1713–1727. [Google Scholar] [CrossRef] [PubMed]
- Dieckmann, K.P.; Simonsen-Richter, H.; Kulejewski, M.; Anheuser, P.; Zecha, H.; Isbarn, H.; Pichlmeier, U. Serum Tumour Markers in Testicular Germ Cell Tumours: Frequencies of Elevated Levels and Extents of Marker Elevation Are Significantly Associated with Clinical Parameters and with Response to Treatment. Biomed. Res. Int. 2019, 2019, 5030349. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell 2004, 116, 281–297. [Google Scholar] [CrossRef]
- Regouc, M.; Belge, G.; Lorch, A.; Dieckmann, K.P.; Pichler, M. Non-Coding microRNAs as Novel Potential Tumor Markers in Testicular Cancer. Cancers 2020, 12, 749. [Google Scholar] [CrossRef]
- Pichler, M.; Calin, G.A. MicroRNAs in cancer: From developmental genes in worms to their clinical application in patients. Br. J. Cancer 2015, 113, 569–573. [Google Scholar] [CrossRef]
- D’Angelo, D.; Borbone, E.; Palmieri, D.; Uboldi, S.; Esposito, F.; Frapolli, R.; Pacelli, R.; D’Incalci, M.; Fusco, A. The impairment of the High Mobility Group A (HMGA) protein function contributes to the anticancer activity of trabectedin. Eur. J. Cancer 2013, 49, 1142–1151. [Google Scholar] [CrossRef]
- Fusco, A.; Fedele, M. Roles of HMGA proteins in cancer. Nat. Rev. Cancer 2007, 7, 899–910. [Google Scholar] [CrossRef]
- De Martino, M.; Fusco, A.; Esposito, F. HMGA and Cancer: A Review on Patent Literatures. Recent Pat. Anticancer Drug Discov. 2019, 14, 258–267. [Google Scholar] [CrossRef]
- Palumbo, A., Jr.; De Martino, M.; Esposito, F.; Fraggetta, F.; Neto, P.N.; Valverde Fernandes, P.; Santos, I.C.; Dias, F.L.; Nasciutti, L.E.; Meireles Da Costa, N.; et al. HMGA2, but not HMGA1, is overexpressed in human larynx carcinomas. Histopathology 2018, 72, 1102–1114. [Google Scholar] [CrossRef] [PubMed]
- Puca, F.; Tosti, N.; Federico, A.; Kuzay, Y.; Pepe, A.; Morlando, S.; Savarese, T.; D’Alessio, F.; Colamaio, M.; Sarnataro, D.; et al. HMGA1 negatively regulates NUMB expression at transcriptional and post transcriptional level in glioblastoma stem cells. Cell Cycle 2019, 18, 1446–1457. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.; Battista, S.; Barchi, M.; Di Agostino, S.; Pierantoni, G.M.; Fedele, M.; Chiariotti, L.; Tramontano, D.; Fusco, A. HMGA1 and HMGA2 protein expression in mouse spermatogenesis. Oncogene 2002, 21, 3644–3650. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Franco, R.; Esposito, F.; Fedele, M.; Liguori, G.; Pierantoni, G.M.; Botti, G.; Tramontano, D.; Fusco, A.; Chieffi, P. Detection of high-mobility group proteins A1 and A2 represents a valid diagnostic marker in post-pubertal testicular germ cell tumours. J. Pathol. 2008, 214, 58–64. [Google Scholar] [CrossRef]
- Pero, R.; Lembo, F.; Chieffi, P.; Del Pozzo, G.; Fedele, M.; Fusco, A.; Bruni, C.B.; Chiariotti, L. Translational regulation of a novel testis-specific RNF4 transcript. Mol. Reprod. Dev. 2003, 66, 1–7. [Google Scholar] [CrossRef]
- Fedele, M.; Franco, R.; Salvatore, G.; Paronetto, M.P.; Barbagallo, F.; Pero, R.; Chiariotti, L.; Sette, C.; Tramontano, D.; Chieffi, G.; et al. PATZ1 gene has a critical role in the spermatogenesis and testicular tumours. J. Pathol. 2008, 215, 39–47. [Google Scholar] [CrossRef]
- Esposito, F.; Boscia, F.; Franco, R.; Tornincasa, M.; Fusco, A.; Kitazawa, S.; Looijenga, L.H.; Chieffi, P. Down-regulation of oestrogen receptor-beta associates with transcriptional co-regulator PATZ1 delocalization in human testicular seminomas. J. Pathol. 2011, 224, 110–120. [Google Scholar] [CrossRef]
- Esposito, F.; Boscia, F.; Gigantino, V.; Tornincasa, M.; Fusco, A.; Franco, R.; Chieffi, P. The high-mobility group A1-estrogen receptor beta nuclear interaction is impaired in human testicular seminomas. J. Cell. Physiol. 2012, 227, 3749–3755. [Google Scholar] [CrossRef]
- Vicini, E.; Loiarro, M.; Di Agostino, S.; Corallini, S.; Capolunghi, F.; Carsetti, R.; Chieffi, P.; Geremia, R.; Stefanini, M.; Sette, C. 17-beta-estradiol elicits genomic and non-genomic responses in mouse male germ cells. J. Cell. Physiol. 2006, 206, 238–245. [Google Scholar] [CrossRef]
- Staibano, S.; Franco, R.; Mezza, E.; Chieffi, P.; Sinisi, A.; Pasquali, D.; Errico, M.E.; Nappi, C.; Tremolaterra, F.; Somma, P.; et al. Loss of oestrogen receptor beta, high PCNA and p53 expression and aneuploidy as markers of worse prognosis in ovarian granulosa cell tumours. Histopathology 2003, 43, 254–262. [Google Scholar] [CrossRef]
- Chieffi, P.; Colucci-D’Amato, G.L.; Staibano, S.; Franco, R.; Tramontano, D. Estradiol-induced mitogen-activated protein kinase (extracellular signal-regulated kinase 1 and 2) activity in the frog (Rana esculenta) testis. J. Endocrinol. 2000, 167, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Franco, R.; Boscia, F.; Gigantino, V.; Marra, L.; Esposito, F.; Ferrara, D.; Pariante, P.; Botti, G.; Caraglia, M.; Minucci, S.; et al. GPR30 is overexpressed in post-puberal testicular germ cell tumors. Cancer Biol. Ther. 2011, 11, 609–613. [Google Scholar] [CrossRef]
- Boscia, F.; Passaro, C.; Gigantino, V.; Perdona, S.; Franco, R.; Portella, G.; Chieffi, S.; Chieffi, P. High levels of GPR30 protein in human testicular carcinoma in situ and seminomas correlate with low levels of estrogen receptor-beta and indicate a switch in estrogen responsiveness. J. Cell. Physiol. 2015, 230, 1290–1297. [Google Scholar] [CrossRef] [PubMed]
- Mayer, F.; Stoop, H.; Sen, S.; Bokemeyer, C.; Oosterhuis, J.W.; Looijenga, L.H. Aneuploidy of human testicular germ cell tumors is associated with amplification of centrosomes. Oncogene 2003, 22, 3859–3866. [Google Scholar] [CrossRef] [PubMed]
- Chieffi, P.; Troncone, G.; Caleo, A.; Libertini, S.; Linardopoulos, S.; Tramontano, D.; Portella, G. Aurora B expression in normal testis and seminomas. J. Endocrinol. 2004, 181, 263–270. [Google Scholar] [CrossRef]
- Esposito, F.; Libertini, S.; Franco, R.; Abagnale, A.; Marra, L.; Portella, G.; Chieffi, P. Aurora B expression in post-puberal testicular germ cell tumours. J. Cell. Physiol. 2009, 221, 435–439. [Google Scholar] [CrossRef]
- Portella, G.; Passaro, C.; Chieffi, P. Aurora B: A new prognostic marker and therapeutic target in cancer. Curr. Med. Chem. 2011, 18, 482–496. [Google Scholar] [CrossRef]
- Keen, N.; Taylor, S. Aurora-kinase inhibitors as anticancer agents. Nat. Rev. Cancer 2004, 4, 927–936. [Google Scholar] [CrossRef]
- Jansson, M.D.; Lund, A.H. MicroRNA and cancer. Mol. Oncol. 2012, 6, 590–610. [Google Scholar] [CrossRef]
- Gutschner, T.; Richtig, G.; Haemmerle, M.; Pichler, M. From biomarkers to therapeutic targets-the promises and perils of long non-coding RNAs in cancer. Cancer Metastasis Rev. 2018, 37, 83–105. [Google Scholar] [CrossRef]
- Barth, D.A.; Slaby, O.; Klec, C.; Juracek, J.; Drula, R.; Calin, G.A.; Pichler, M. Current Concepts of Non-Coding RNAs in the Pathogenesis of Non-Clear Cell Renal Cell Carcinoma. Cancers 2019, 11, 1580. [Google Scholar] [CrossRef] [PubMed]
- Klec, C.; Prinz, F.; Pichler, M. Involvement of the long noncoding RNA NEAT1 in carcinogenesis. Mol. Oncol. 2019, 13, 46–60. [Google Scholar] [CrossRef] [PubMed]
- Forzati, F.; De Martino, M.; Esposito, F.; Sepe, R.; Pellecchia, S.; Malapelle, U.; Pellino, G.; Arra, C.; Fusco, A. miR-155 is positively regulated by CBX7 in mouse embryonic fibroblasts and colon carcinomas, and targets the KRAS oncogene. BMC Cancer 2017, 17, 170. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.; Mayr, C.; Bach, D.; Illig, R.; Plaetzer, K.; Berr, F.; Pichler, M.; Neureiter, D.; Kiesslich, T. MicroRNAs associated with the efficacy of photodynamic therapy in biliary tract cancer cell lines. Int. J. Mol. Sci. 2014, 15, 20134–20157. [Google Scholar] [CrossRef] [PubMed]
- Pehserl, A.M.; Ress, A.L.; Stanzer, S.; Resel, M.; Karbiener, M.; Stadelmeyer, E.; Stiegelbauer, V.; Gerger, A.; Mayr, C.; Scheideler, M.; et al. Comprehensive Analysis of miRNome Alterations in Response to Sorafenib Treatment in Colorectal Cancer Cells. Int. J. Mol. Sci. 2016, 17, 2011. [Google Scholar] [CrossRef]
- Smolle, M.A.; Prinz, F.; Calin, G.A.; Pichler, M. Current concepts of non-coding RNA regulation of immune checkpoints in cancer. Mol. Asp. Med. 2019, 70, 117–126. [Google Scholar] [CrossRef]
- Paranjape, T.; Slack, F.J.; Weidhaas, J.B. MicroRNAs: Tools for cancer diagnostics. Gut 2009, 58, 1546–1554. [Google Scholar] [CrossRef]
- Turchinovich, A.; Samatov, T.R.; Tonevitsky, A.G.; Burwinkel, B. Circulating miRNAs: Cell-cell communication function? Front. Genet. 2013, 4, 119. [Google Scholar] [CrossRef]
- Xu, L.; Yang, B.F.; Ai, J. MicroRNA transport: A new way in cell communication. J. Cell. Physiol. 2013, 228, 1713–1719. [Google Scholar] [CrossRef]
- Li, H.L.; Wei, J.F.; Fan, L.Y.; Wang, S.H.; Zhu, L.; Li, T.P.; Lin, G.; Sun, Y.; Sun, Z.J.; Ding, J.; et al. miR-302 regulates pluripotency, teratoma formation and differentiation in stem cells via an AKT1/OCT4-dependent manner. Cell Death Dis. 2016, 7, e2078. [Google Scholar] [CrossRef]
- Gao, Z.; Zhu, X.; Dou, Y. The miR-302/367 cluster: A comprehensive update on its evolution and functions. Open Biol. 2015, 5, 150138. [Google Scholar] [CrossRef] [PubMed]
- Das, M.K.; Evensen, H.S.F.; Furu, K.; Haugen, T.B. miRNA-302s may act as oncogenes in human testicular germ cell tumours. Sci. Rep. 2019, 9, 9189. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Go, Y.; Kang, I.; Han, Y.M.; Kim, J. Oct-4 controls cell-cycle progression of embryonic stem cells. Biochem. J. 2010, 426, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Ma, G.; Li, Q.; Dai, W.; Yang, X.; Sang, A. Prognostic Implications of miR-302a/b/c/d in Human Gastric Cancer. Pathol. Oncol. Res. 2017, 23, 899–905. [Google Scholar] [CrossRef]
- Wang, L.; Yao, J.; Shi, X.; Hu, L.; Li, Z.; Song, T.; Huang, C. MicroRNA-302b suppresses cell proliferation by targeting EGFR in human hepatocellular carcinoma SMMC-7721 cells. BMC Cancer 2013, 13, 448. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, H.; Wu, X.; Kawamoto, K.; Nishida, N.; Konno, M.; Koseki, J.; Matsui, H.; Noguchi, K.; Gotoh, N.; Yamamoto, T.; et al. MicroRNAs Induce Epigenetic Reprogramming and Suppress Malignant Phenotypes of Human Colon Cancer Cells. PLoS ONE 2015, 10, e0127119. [Google Scholar] [CrossRef]
- Das, M.K.; Furu, K.; Evensen, H.F.; Haugen, O.P.; Haugen, T.B. Knockdown of SPRY4 and SPRY4-IT1 inhibits cell growth and phosphorylation of Akt in human testicular germ cell tumours. Sci. Rep. 2018, 8, 2462. [Google Scholar] [CrossRef]
- Murray, M.J.; Halsall, D.J.; Hook, C.E.; Williams, D.M.; Nicholson, J.C.; Coleman, N. Identification of microRNAs From the miR-371~373 and miR-302 clusters as potential serum biomarkers of malignant germ cell tumors. Am. J. Clin. Pathol. 2011, 135, 119–125. [Google Scholar] [CrossRef]
- Kuo, C.H.; Deng, J.H.; Deng, Q.; Ying, S.Y. A novel role of miR-302/367 in reprogramming. Biochem. Biophys. Res. Commun. 2012, 417, 11–16. [Google Scholar] [CrossRef]
- Syring, I.; Bartels, J.; Holdenrieder, S.; Kristiansen, G.; Muller, S.C.; Ellinger, J. Circulating serum miRNA (miR-367-3p, miR-371a-3p, miR-372-3p and miR-373-3p) as biomarkers in patients with testicular germ cell cancer. J. Urol. 2015, 193, 331–337. [Google Scholar] [CrossRef]
- van Agthoven, T.; Looijenga, L.H.J. Accurate primary germ cell cancer diagnosis using serum based microRNA detection (ampTSmiR test). Oncotarget 2017, 8, 58037–58049. [Google Scholar] [CrossRef] [PubMed]
- Rosas Plaza, X.; van Agthoven, T.; Meijer, C.; van Vugt, M.; de Jong, S.; Gietema, J.A.; Looijenga, L.H.J. miR-371a-3p, miR-373-3p and miR-367-3p as Serum Biomarkers in Metastatic Testicular Germ Cell Cancers Before, During and After Chemotherapy. Cells 2019, 8, 1221. [Google Scholar] [CrossRef] [PubMed]
- Terbuch, A.; Adiprasito, J.B.; Stiegelbauer, V.; Seles, M.; Klec, C.; Pichler, G.P.; Resel, M.; Posch, F.; Lembeck, A.L.; Stoger, H.; et al. MiR-371a-3p Serum Levels Are Increased in Recurrence of Testicular Germ Cell Tumor Patients. Int. J. Mol. Sci. 2018, 19, 3130. [Google Scholar] [CrossRef] [PubMed]
- Leao, R.; van Agthoven, T.; Figueiredo, A.; Jewett, M.A.S.; Fadaak, K.; Sweet, J.; Ahmad, A.E.; Anson-Cartwright, L.; Chung, P.; Hansen, A.; et al. Serum miRNA Predicts Viable Disease after Chemotherapy in Patients with Testicular Nonseminoma Germ Cell Tumor. J. Urol. 2018, 200, 126–135. [Google Scholar] [CrossRef]
- Murray, M.J.; Huddart, R.A.; Coleman, N. The present and future of serum diagnostic tests for testicular germ cell tumours. Nat. Rev. Urol. 2016, 13, 715–725. [Google Scholar] [CrossRef]
- Lee, K.H.; Goan, Y.G.; Hsiao, M.; Lee, C.H.; Jian, S.H.; Lin, J.T.; Chen, Y.L.; Lu, P.J. MicroRNA-373 (miR-373) post-transcriptionally regulates large tumor suppressor, homolog 2 (LATS2) and stimulates proliferation in human esophageal cancer. Exp. Cell Res. 2009, 315, 2529–2538. [Google Scholar] [CrossRef]
- Tanaka, T.; Arai, M.; Wu, S.; Kanda, T.; Miyauchi, H.; Imazeki, F.; Matsubara, H.; Yokosuka, O. Epigenetic silencing of microRNA-373 plays an important role in regulating cell proliferation in colon cancer. Oncol. Rep. 2011, 26, 1329–1335. [Google Scholar] [CrossRef]
- Cairo, S.; Wang, Y.; de Reynies, A.; Duroure, K.; Dahan, J.; Redon, M.J.; Fabre, M.; McClelland, M.; Wang, X.W.; Croce, C.M.; et al. Stem cell-like micro-RNA signature driven by Myc in aggressive liver cancer. Proc. Natl. Acad. Sci. USA 2010, 107, 20471–20476. [Google Scholar] [CrossRef]
- Adi Harel, S.; Bossel Ben-Moshe, N.; Aylon, Y.; Bublik, D.R.; Moskovits, N.; Toperoff, G.; Azaiza, D.; Biagoni, F.; Fuchs, G.; Wilder, S.; et al. Reactivation of epigenetically silenced miR-512 and miR-373 sensitizes lung cancer cells to cisplatin and restricts tumor growth. Cell Death Differ. 2015, 22, 1328–1340. [Google Scholar] [CrossRef]
- Huang, Q.; Gumireddy, K.; Schrier, M.; le Sage, C.; Nagel, R.; Nair, S.; Egan, D.A.; Li, A.; Huang, G.; Klein-Szanto, A.J.; et al. The microRNAs miR-373 and miR-520c promote tumour invasion and metastasis. Nat. Cell Biol. 2008, 10, 202–210. [Google Scholar] [CrossRef]
- Lu, S.; Zhu, Q.; Zhang, Y.; Song, W.; Wilson, M.J.; Liu, P. Dual-Functions of miR-373 and miR-520c by Differently Regulating the Activities of MMP2 and MMP9. J. Cell. Physiol. 2015, 230, 1862–1870. [Google Scholar] [CrossRef] [PubMed]
- Wei, F.; Cao, C.; Xu, X.; Wang, J. Diverse functions of miR-373 in cancer. J. Transl. Med. 2015, 13, 162. [Google Scholar] [CrossRef] [PubMed]
- Voorhoeve, P.M.; le Sage, C.; Schrier, M.; Gillis, A.J.; Stoop, H.; Nagel, R.; Liu, Y.P.; van Duijse, J.; Drost, J.; Griekspoor, A.; et al. A genetic screen implicates miRNA-372 and miRNA-373 as oncogenes in testicular germ cell tumors. Cell 2006, 124, 1169–1181. [Google Scholar] [CrossRef] [PubMed]
- Zhou, A.D.; Diao, L.T.; Xu, H.; Xiao, Z.D.; Li, J.H.; Zhou, H.; Qu, L.H. beta-Catenin/LEF1 transactivates the microRNA-371-373 cluster that modulates the Wnt/beta-catenin-signaling pathway. Oncogene 2012, 31, 2968–2978. [Google Scholar] [CrossRef]
- Vilela-Salgueiro, B.; Barros-Silva, D.; Lobo, J.; Costa, A.L.; Guimaraes, R.; Cantante, M.; Lopes, P.; Braga, I.; Oliveira, J.; Henrique, R.; et al. Germ cell tumour subtypes display differential expression of microRNA371a-3p. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2018, 373, 20140338. [Google Scholar] [CrossRef]
- Bentwich, I.; Avniel, A.; Karov, Y.; Aharonov, R.; Gilad, S.; Barad, O.; Barzilai, A.; Einat, P.; Einav, U.; Meiri, E.; et al. Identification of hundreds of conserved and nonconserved human microRNAs. Nat. Genet. 2005, 37, 766–770. [Google Scholar] [CrossRef]
- Toffanin, S.; Hoshida, Y.; Lachenmayer, A.; Villanueva, A.; Cabellos, L.; Minguez, B.; Savic, R.; Ward, S.C.; Thung, S.; Chiang, D.Y.; et al. MicroRNA-based classification of hepatocellular carcinoma and oncogenic role of miR-517a. Gastroenterology 2011, 140, 1618–1628. [Google Scholar] [CrossRef]
- Ward, A.; Shukla, K.; Balwierz, A.; Soons, Z.; Konig, R.; Sahin, O.; Wiemann, S. MicroRNA-519a is a novel oncomir conferring tamoxifen resistance by targeting a network of tumour-suppressor genes in ER+ breast cancer. J. Pathol. 2014, 233, 368–379. [Google Scholar] [CrossRef]
- Flor, I.; Spiekermann, M.; Loning, T.; Dieckmann, K.P.; Belge, G.; Bullerdiek, J. Expression of microRNAs of C19MC in Different Histological Types of Testicular Germ Cell Tumour. Cancer Genom. Proteom. 2016, 13, 281–289. [Google Scholar]
- Li, J.; Guo, Y.; Liang, X.; Sun, M.; Wang, G.; De, W.; Wu, W. MicroRNA-223 functions as an oncogene in human gastric cancer by targeting FBXW7/hCdc4. J. Cancer Res. Clin. Oncol. 2012, 138, 763–774. [Google Scholar] [CrossRef]
- Kurashige, J.; Watanabe, M.; Iwatsuki, M.; Kinoshita, K.; Saito, S.; Hiyoshi, Y.; Kamohara, H.; Baba, Y.; Mimori, K.; Baba, H. Overexpression of microRNA-223 regulates the ubiquitin ligase FBXW7 in oesophageal squamous cell carcinoma. Br. J. Cancer 2012, 106, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Mavrakis, K.J.; Van Der Meulen, J.; Wolfe, A.L.; Liu, X.; Mets, E.; Taghon, T.; Khan, A.A.; Setty, M.; Rondou, P.; Vandenberghe, P.; et al. A cooperative microRNA-tumor suppressor gene network in acute T-cell lymphoblastic leukemia (T-ALL). Nat. Genet. 2011, 43, 673–678. [Google Scholar] [CrossRef] [PubMed]
- Kurozumi, A.; Goto, Y.; Matsushita, R.; Fukumoto, I.; Kato, M.; Nishikawa, R.; Sakamoto, S.; Enokida, H.; Nakagawa, M.; Ichikawa, T.; et al. Tumor-suppressive microRNA-223 inhibits cancer cell migration and invasion by targeting ITGA3/ITGB1 signaling in prostate cancer. Cancer Sci. 2016, 107, 84–94. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shi, H.; Li, X.; Chen, G.; Larsson, C.; Lui, W.O. miR2233p regulates cell growth and apoptosis via FBXW7 suggesting an oncogenic role in human testicular germ cell tumors. Int. J. Oncol. 2017, 50, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Jin, W.; Jia, H.; Yan, J.; Zhang, G. MiR-223 promotes the cisplatin resistance of human gastric cancer cells via regulating cell cycle by targeting FBXW7. J. Exp. Clin. Cancer Res. 2015, 34, 28. [Google Scholar] [CrossRef]
- Yuan, S.; Tang, C.; Zhang, Y.; Wu, J.; Bao, J.; Zheng, H.; Xu, C.; Yan, W. mir-34b/c and mir-449a/b/c are required for spermatogenesis, but not for the first cleavage division in mice. Biol. Open 2015, 4, 212–223. [Google Scholar] [CrossRef]
- Lize, M.; Pilarski, S.; Dobbelstein, M. E2F1-inducible microRNA 449a/b suppresses cell proliferation and promotes apoptosis. Cell Death Differ. 2010, 17, 452–458. [Google Scholar] [CrossRef]
- Yang, X.; Feng, M.; Jiang, X.; Wu, Z.; Li, Z.; Aau, M.; Yu, Q. miR-449a and miR-449b are direct transcriptional targets of E2F1 and negatively regulate pRb-E2F1 activity through a feedback loop by targeting CDK6 and CDC25A. Genes Dev. 2009, 23, 2388–2393. [Google Scholar] [CrossRef]
- Yong-Ming, H.; Ai-Jun, J.; Xiao-Yue, X.; Jian-Wei, L.; Chen, Y.; Ye, C. miR-449a: A potential therapeutic agent for cancer. Anticancer Drugs 2017, 28, 1067–1078. [Google Scholar] [CrossRef]
- Lian, J.; Tian, H.; Liu, L.; Zhang, X.S.; Li, W.Q.; Deng, Y.M.; Yao, G.D.; Yin, M.M.; Sun, F. Downregulation of microRNA-383 is associated with male infertility and promotes testicular embryonal carcinoma cell proliferation by targeting IRF1. Cell Death Dis. 2010, 1, e94. [Google Scholar] [CrossRef]
- Tian, H.; Cao, Y.X.; Zhang, X.S.; Liao, W.P.; Yi, Y.H.; Lian, J.; Liu, L.; Huang, H.L.; Liu, W.J.; Yin, M.M.; et al. The targeting and functions of miRNA-383 are mediated by FMRP during spermatogenesis. Cell Death Dis. 2013, 4, e617. [Google Scholar] [CrossRef] [PubMed]
- Alsamman, K.; El-Masry, O.S. Interferon regulatory factor 1 inactivation in human cancer. Biosci. Rep. 2018, 38, BSR20171672. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Tian, H.; Duan, Z.; Cao, Y.; Zhang, X.S.; Sun, F. microRNA-383 impairs phosphorylation of H2AX by targeting PNUTS and inducing cell cycle arrest in testicular embryonal carcinoma cells. Cell. Signal. 2014, 26, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhang, X.F.; Lu, X.; Jia, H.L.; Liang, L.; Dong, Q.Z.; Ye, Q.H.; Qin, L.X. MicroRNA-26a suppresses angiogenesis in human hepatocellular carcinoma by targeting hepatocyte growth factor-cMet pathway. Hepatology 2014, 59, 1874–1885. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.; Liu, F.; Liang, X.; Liu, S.; Huang, X.; Sang, M.; Geng, C. Tumor suppressor let-7a inhibits breast cancer cell proliferation, migration and invasion by targeting MAGE-A1. Neoplasma 2019, 66, 54–62. [Google Scholar] [CrossRef] [PubMed]
- De Martino, M.; Esposito, F.; Chieffi, P. An update on microRNAs as potential novel therapeutic targets in testicular germ cell tumors. Intractable Rare Dis. Res. 2020, 9, 184–186. [Google Scholar] [CrossRef] [PubMed]
- De Martino, M.; Esposito, F.; Pellecchia, S.; Cortez Cardoso Penha, R.; Botti, G.; Fusco, A.; Chieffi, P. HMGA1-Regulating microRNAs Let-7a and miR-26a are Downregulated in Human Seminomas. Int. J. Mol. Sci. 2020, 21, 3014. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).