Activation of the Interleukin-33/ST2 Pathway Exerts Deleterious Effects in Myxomatous Mitral Valve Disease
Abstract
:1. Introduction
2. Results
2.1. Clinicopathological Features of the Patients with MVD
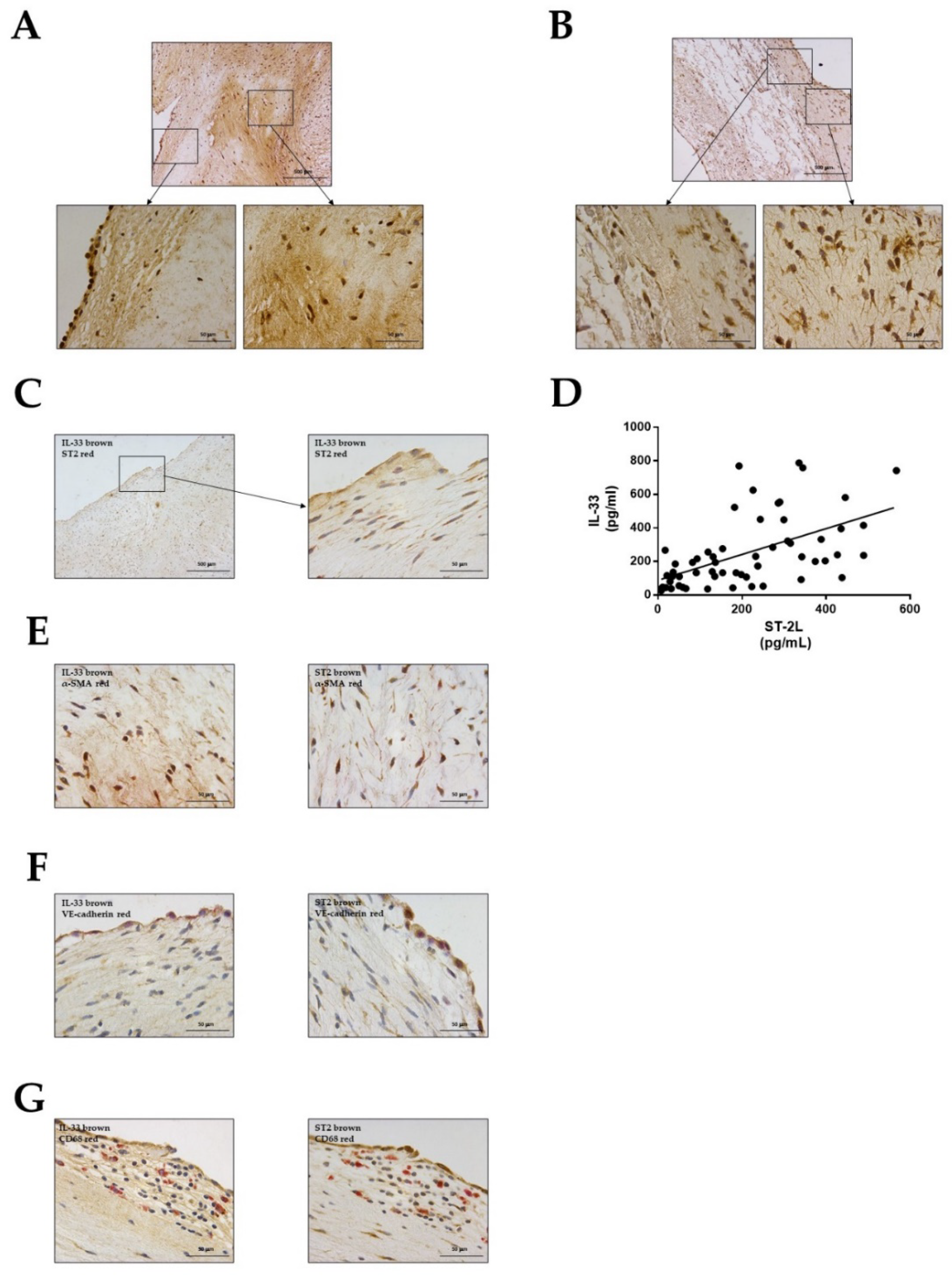
2.2. Distribution of the IL-33 and ST2 within the Mitral Valve
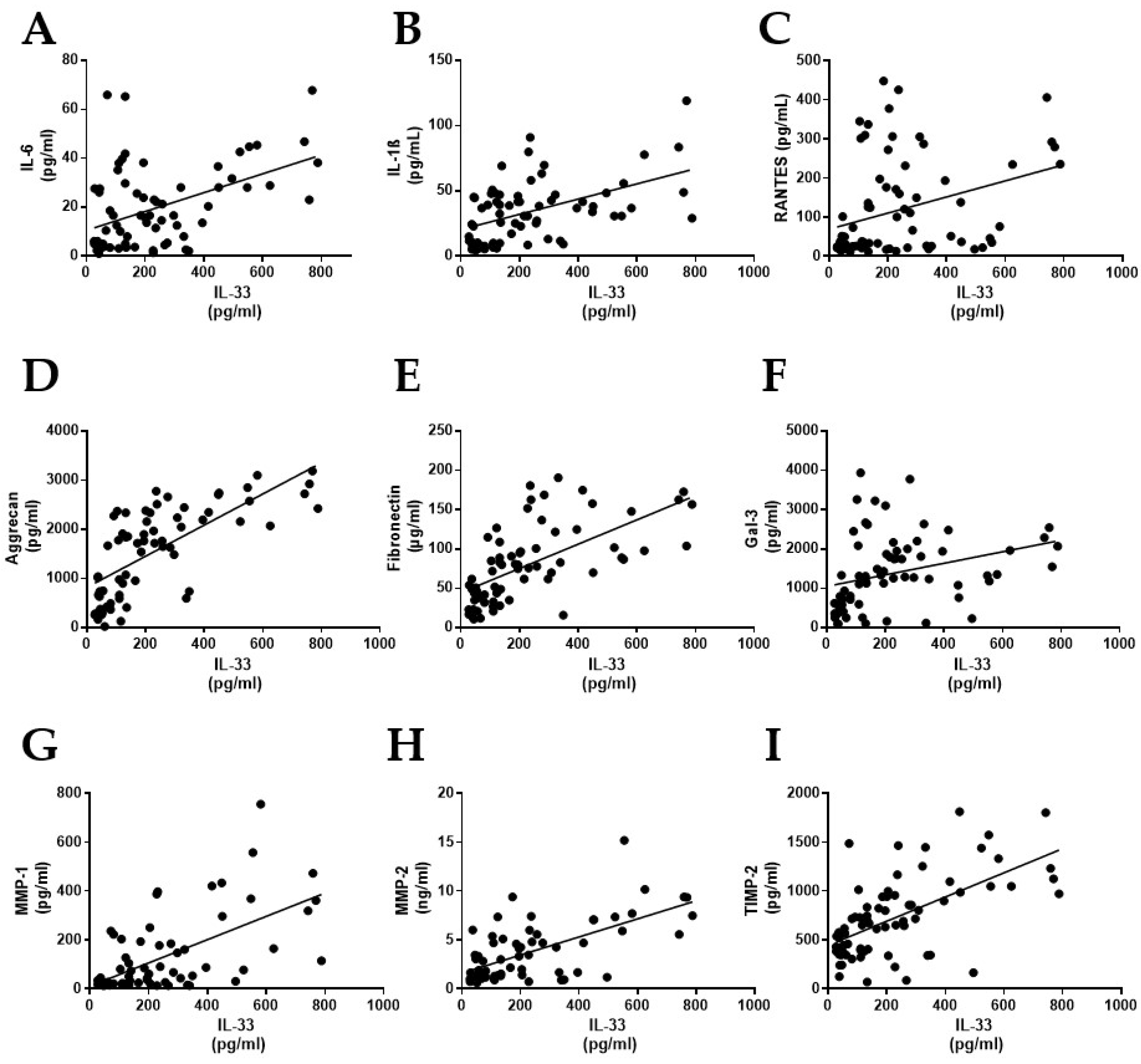
2.3. Correlation of IL-33 in Myxomatous MVs with Inflammatory Mediators, Extracellular Matrix Components, and Matrix Metalloproteinases
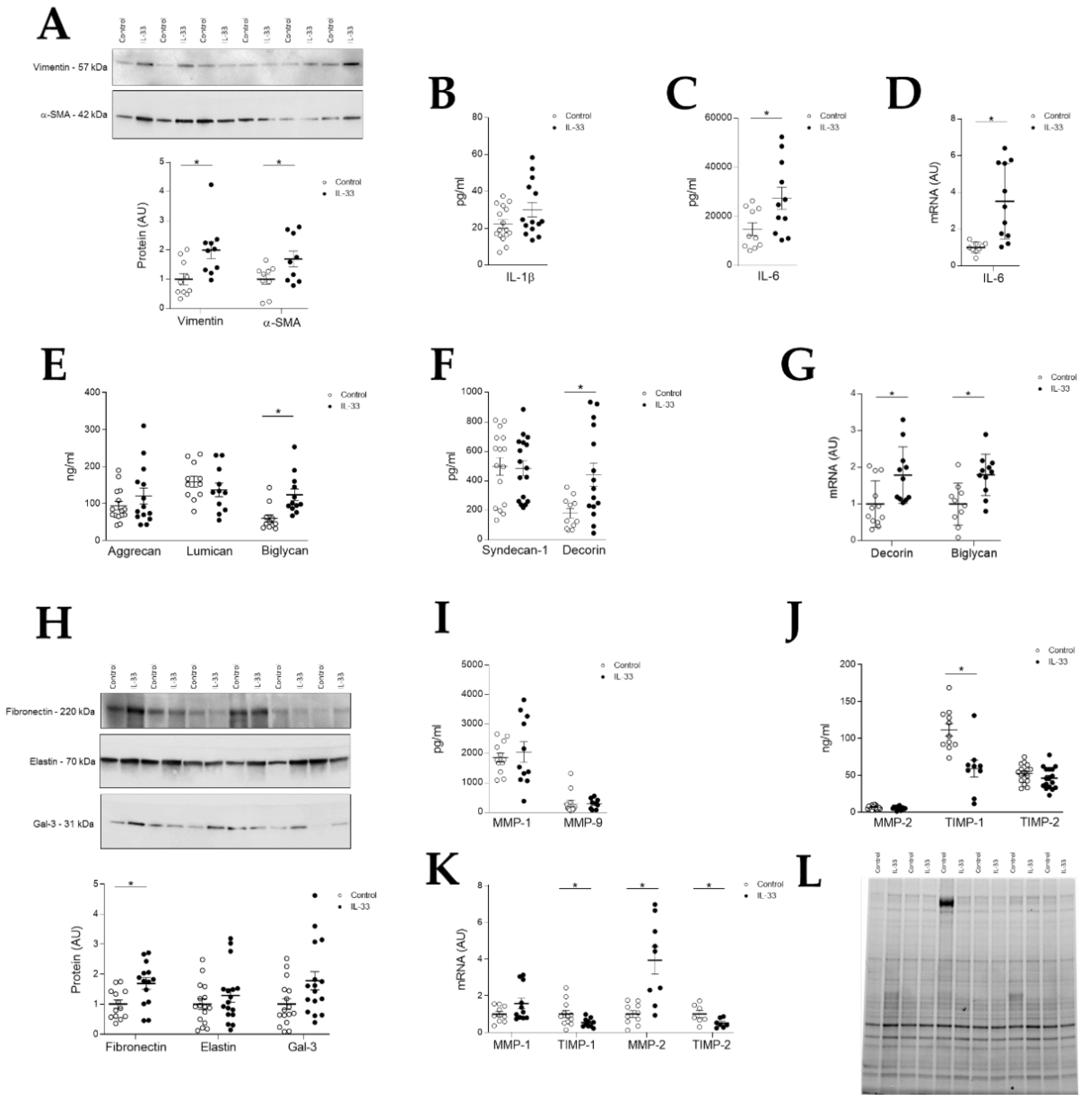
2.4. IL-33 Induces Activation of Human Mitral VICs, Inflammation, Proteoglycans Synthesis and Extracellular Matrix Remodeling
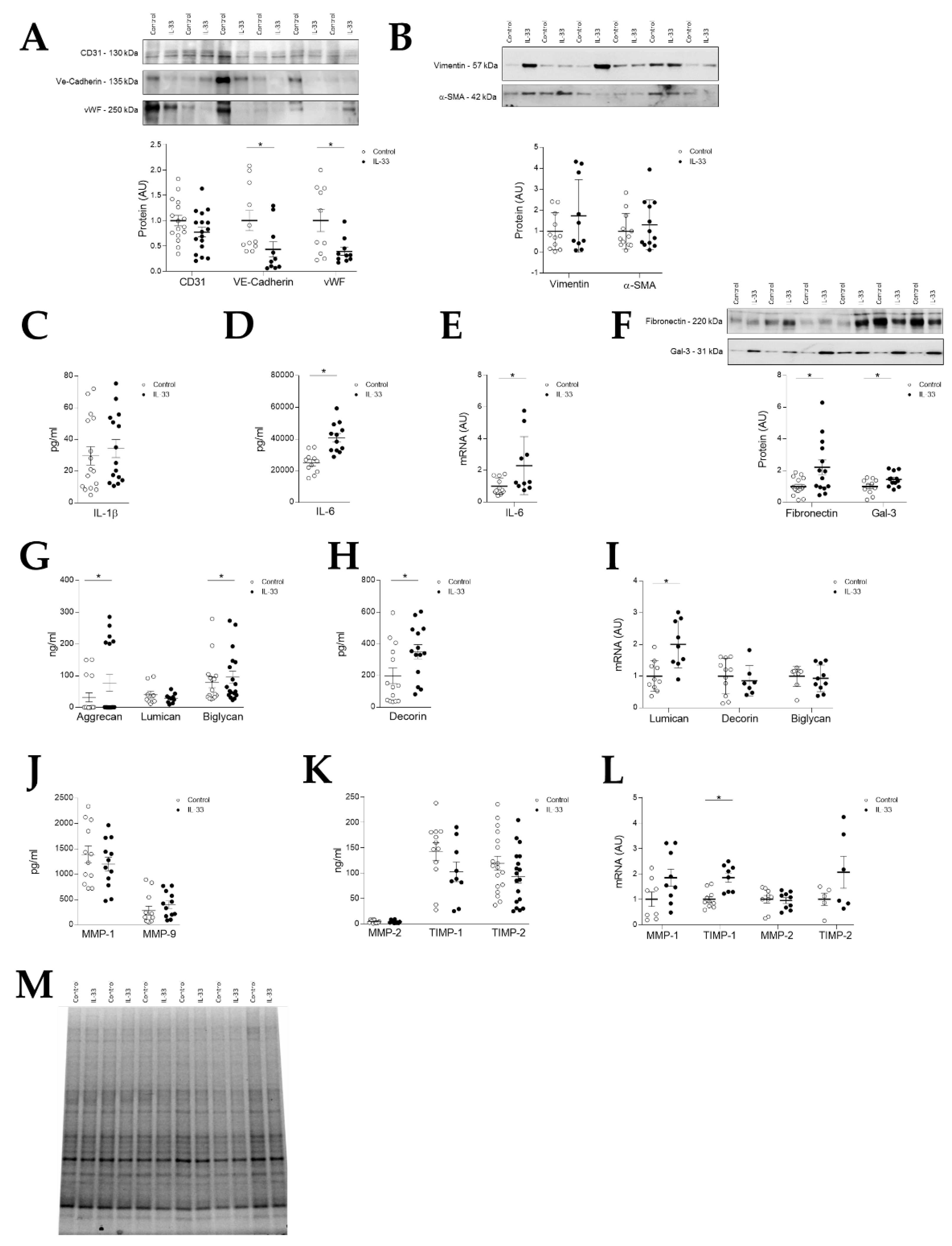
2.5. IL-33 Induces EndMT in Valve Endothelial Cells and Extracellular Matrix Changes
3. Discussion
4. Materials and Methods
4.1. Patient Population
4.2. Immunohistological Evaluation
4.3. ELISA
4.4. Cell Isolation and Culture
4.5. Single Culture Cell Assays
4.6. Real-Time Reverse Transcription PCR
4.7. Western Blot Analysis
4.8. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nkomo, V.T.; Gardin, J.M.; Skelton, T.N.; Gottdiener, J.S.; Scott, C.G.; Enriquez-Sarano, M. Burden of Valvular Heart Diseases: A Population-Based Study. Lancet 2006, 368, 1005–1011. [Google Scholar] [CrossRef]
- Levine, R.A.; Hagége, A.A.; Judge, D.P.; Padala, M.; Dal-Bianco, J.P.; Aikawa, E.; Beaudoin, J.; Bischoff, J.; Bouatia-Naji, N.; Bruneval, P.; et al. Mitral Valve Disease—Morphology and Mechanisms. Nat. Rev. Cardiol. 2015, 12, 689–710. [Google Scholar] [CrossRef] [Green Version]
- Zoghbi, W.A.; Adams, D.; Bonow, R.O.; Enriquez-Sarano, M.; Foster, E.; Grayburn, P.A.; Hahn, R.T.; Han, Y.; Hung, J.; Lang, R.M.; et al. Recommendations for Noninvasive Evaluation of Native Valvular Regurgitation: A Report from the American Society of Echocardiography Developed in Collaboration with the Society for Cardiovascular Magnetic Resonance. J. Am. Soc. Echocardiogr. 2017, 30, 303–371. [Google Scholar] [CrossRef]
- Singh, J.P.; Evans, J.C.; Levy, D.; Larson, M.G.; Freed, L.A.; Fuller, D.L.; Lehman, B.; Benjamin, E.J. Prevalence and Clinical Determinants of Mitral, Tricuspid, and Aortic Regurgitation (the Framingham Heart Study). Am. J. Cardiol. 1999, 83, 897–902. [Google Scholar] [CrossRef]
- Fishbein, G.A.; Fishbein, M.C. Mitral Valve Pathology. Curr. Cardiol. Rep. 2019, 21, 61. [Google Scholar] [CrossRef] [PubMed]
- Gupta, V.; Barzilla, J.E.; Mendez, J.S.; Stephens, E.H.; Lee, E.L.; Collard, C.D.; Laucirica, R.; Weigel, P.H.; Grande-Allen, K.J. Abundance and Location of Proteoglycans and Hyaluronan within Normal and Myxomatous Mitral Valves. Cardiovasc. Pathol. 2009, 18, 191–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bischoff, J.; Aikawa, E. Progenitor Cells Confer Plasticity to Cardiac Valve Endothelium. J. Cardiovasc. Transl. Res. 2011, 4, 710–719. [Google Scholar] [CrossRef]
- Wang, H.; Leinwand, L.A.; Anseth, K.S. Cardiac Valve Cells and Their Microenvironment—Insights from in Vitro Studies. Nat. Rev. Cardiol. 2014, 11, 715–727. [Google Scholar] [CrossRef] [Green Version]
- Rabkin, E.; Aikawa, M.; Stone, J.R.; Fukumoto, Y.; Libby, P.; Schoen, F.J. Activated Interstitial Myofibroblasts Express Catabolic Enzymes and Mediate Matrix Remodeling in Myxomatous Heart Valves. Circulation 2001, 104, 2525–2532. [Google Scholar] [CrossRef] [PubMed]
- Dal-Bianco, J.P.; Aikawa, E.; Bischoff, J.; Guerrero, J.L.; Handschumacher, M.D.; Sullivan, S.; Johnson, B.; Titus, J.S.; Iwamoto, Y.; Wylie-Sears, J.; et al. Active Adaptation of the Tethered Mitral Valve: Insights into a Compensatory Mechanism for Functional Mitral Regurgitation. Circulation 2009, 120, 334–342. [Google Scholar] [CrossRef] [Green Version]
- Lu, C.-C.; Liu, M.-M.; Clinton, M.; Culshaw, G.; Argyle, D.J.; Corcoran, B.M. Developmental Pathways and Endothelial to Mesenchymal Transition in Canine Myxomatous Mitral Valve Disease. Vet. J. Lond. Engl. 1997 2015, 206, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, J.; Owyang, A.; Oldham, E.; Song, Y.; Murphy, E.; McClanahan, T.K.; Zurawski, G.; Moshrefi, M.; Qin, J.; Li, X.; et al. IL-33, an Interleukin-1-like Cytokine That Signals via the IL-1 Receptor-Related Protein ST2 and Induces T Helper Type 2-Associated Cytokines. Immunity 2005, 23, 479–490. [Google Scholar] [CrossRef] [Green Version]
- Nishimura, M.; Sharim, J.; Horiuchi, Y.; Barnett, O.; Wettersten, N.; Maisel, A.S. Soluble ST2: A Biomarker to Monitor Heart Failure Progression and Treatment. J. Clin. Prev. Cardiol. 2018, 7, 148–153. [Google Scholar] [CrossRef]
- Seki, K.; Sanada, S.; Kudinova, A.Y.; Steinhauser, M.L.; Handa, V.; Gannon, J.; Lee, R.T. Interleukin-33 Prevents Apoptosis and Improves Survival after Experimental Myocardial Infarction through ST2 Signaling. Circ. Heart Fail. 2009, 2, 684–691. [Google Scholar] [CrossRef] [Green Version]
- Pascual-Figal, D.A.; Januzzi, J.L. The Biology of ST2: The International ST2 Consensus Panel. Am. J. Cardiol. 2015, 115, 3B–7B. [Google Scholar] [CrossRef]
- Miller, A.M.; Liew, F.Y. The IL-33/ST2 Pathway—A New Therapeutic Target in Cardiovascular Disease. Pharmacol. Ther. 2011, 131, 179–186. [Google Scholar] [CrossRef]
- Aimo, A.; Migliorini, P.; Vergaro, G.; Franzini, M.; Passino, C.; Maisel, A.; Emdin, M. The IL-33/ST2 Pathway, Inflammation and Atherosclerosis: Trigger and Target? Int. J. Cardiol. 2018, 267, 188–192. [Google Scholar] [CrossRef] [PubMed]
- Altara, R.; Ghali, R.; Mallat, Z.; Cataliotti, A.; Booz, G.W.; Zouein, F.A. Conflicting Vascular and Metabolic Impact of the IL-33/SST2 Axis. Cardiovasc. Res. 2018, 114, 1578–1594. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demyanets, S.; Konya, V.; Kastl, S.P.; Kaun, C.; Rauscher, S.; Niessner, A.; Pentz, R.; Pfaffenberger, S.; Rychli, K.; Lemberger, C.E.; et al. Interleukin-33 Induces Expression of Adhesion Molecules and Inflammatory Activation in Human Endothelial Cells and in Human Atherosclerotic Plaques. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 2080–2089. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demyanets, S.; Kaun, C.; Pentz, R.; Krychtiuk, K.A.; Rauscher, S.; Pfaffenberger, S.; Zuckermann, A.; Aliabadi, A.; Gröger, M.; Maurer, G.; et al. Components of the Interleukin-33/ST2 System Are Differentially Expressed and Regulated in Human Cardiac Cells and in Cells of the Cardiac Vasculature. J. Mol. Cell. Cardiol. 2013, 60, 16–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stojkovic, S.; Kaun, C.; Basilio, J.; Rauscher, S.; Hell, L.; Krychtiuk, K.A.; Bonstingl, C.; De Martin, R.; Gröger, M.; Ay, C.; et al. Tissue Factor Is Induced by Interleukin-33 in Human Endothelial Cells: A New Link between Coagulation and Inflammation. Sci. Rep. 2016, 6, 25171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sawada, H.; Naito, Y.; Hirotani, S.; Akahori, H.; Iwasaku, T.; Okuhara, Y.; Miki, K.; Eguchi, A.; Mitsuno, M.; Miyamoto, Y.; et al. Expression of Interleukin-33 and ST2 in Nonrheumatic Aortic Valve Stenosis. Int. J. Cardiol. 2013, 168, 529–531. [Google Scholar] [CrossRef] [PubMed]
- He, Y.B.; Guo, J.H.; Wang, C.; Zhu, D.; Lu, L.M. IL-33 Promotes the Progression of Nonrheumatic Aortic Valve Stenosis via Inducing Differential Phenotypic Transition in Valvular Interstitial Cells. J. Cardiol. 2020, 75, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Tribouilloy, C.M.; Enriquez-Sarano, M.; Schaff, H.V.; Orszulak, T.A.; Bailey, K.R.; Tajik, A.J.; Frye, R.L. Impact of Preoperative Symptoms on Survival after Surgical Correction of Organic Mitral Regurgitation: Rationale for Optimizing Surgical Indications. Circulation 1999, 99, 400–405. [Google Scholar] [CrossRef] [Green Version]
- Yasmin; Al Maskari, R.; McEniery, C.M.; Cleary, S.E.; Li, Y.; Siew, K.; Figg, N.L.; Khir, A.W.; Cockcroft, J.R.; Wilkinson, I.B.; et al. The Matrix Proteins Aggrecan and Fibulin-1 Play a Key Role in Determining Aortic Stiffness. Sci. Rep. 2018, 8, 8550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hulin, A.; Moore, V.; James, J.M.; Yutzey, K.E. Loss of Axin2 Results in Impaired Heart Valve Maturation and Subsequentmyxomatous Valve Disease. Cardiovasc. Res. 2017, 113, 40–51. [Google Scholar] [CrossRef] [PubMed]
- Bowler, M.A.; Bersi, M.R.; Ryzhova, L.M.; Jerrell, R.J.; Parekh, A.; Merryman, W.D. Cadherin-11 as a Regulator of Valve Myofibroblast Mechanobiology. Am. J. Physiol. Heart Circ. Physiol. 2018, 315, H1614–H1626. [Google Scholar] [CrossRef] [PubMed]
- Mahler, G.J.; Farrar, E.J.; Butcher, J.T. Inflammatory Cytokines Promote Mesenchymal Transformation in Embryonic and Adult Valve Endothelial Cells. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 121–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhu, J.; Carver, W. Effects of Interleukin-33 on Cardiac Fibroblast Gene Expression and Activity. Cytokine 2012, 58, 368–379. [Google Scholar] [CrossRef] [Green Version]
- Wallby, L.; Lars, W.; Steffensen, T.; Thora, S.; Broqvist, M.; Mats, B. Role of Inflammation in Nonrheumatic, Regurgitant Heart Valve Disease. A Comparative, Descriptive Study Regarding Apolipoproteins and Inflammatory Cells in Nonrheumatic Heart Valve Disease. Cardiovasc. Pathol. Off. J. Soc. Cardiovasc. Pathol. 2007, 16, 171–178. [Google Scholar] [CrossRef]
- Otto, C.M.; Kuusisto, J.; Reichenbach, D.D.; Gown, A.M.; O’Brien, K.D. Characterization of the Early Lesion of “degenerative” Valvular Aortic Stenosis. Histological and Immunohistochemical Studies. Circulation 1994, 90, 844–853. [Google Scholar] [CrossRef] [Green Version]
- Mozaffarian, D.; Benjamin, E.J.; Go, A.S.; Arnett, D.K.; Blaha, M.J.; Cushman, M.; de Ferranti, S.; Després, J.-P.; Fullerton, H.J.; Howard, V.J.; et al. Heart Disease and Stroke Statistics—2015 Update. Circulation 2015, 131, e29–e322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baumgartner, H.; Falk, V.; Bax, J.J.; De Bonis, M.; Hamm, C.; Holm, P.J.; Iung, B.; Lancellotti, P.; Lansac, E.; Rodriguez Muñoz, D.; et al. 2017 ESC/EACTS Guidelines for the Management of Valvular Heart Disease. Eur. Heart J. 2017, 38, 2739–2791. [Google Scholar] [CrossRef] [PubMed]
- Suri, R.M.; Vanoverschelde, J.-L.; Grigioni, F.; Schaff, H.V.; Tribouilloy, C.; Avierinos, J.-F.; Barbieri, A.; Pasquet, A.; Huebner, M.; Rusinaru, D.; et al. Association Between Early Surgical Intervention vs Watchful Waiting and Outcomes for Mitral Regurgitation Due to Flail Mitral Valve Leaflets. JAMA 2013, 310, 609. [Google Scholar] [CrossRef] [PubMed]
| Clinicopathological Features | n |
|---|---|
| Total number of patients | 103 |
| Age (years ± SD) | 68 ± 10 |
| Male (N, % of total) | 68 (65) |
| Hypertension (N, % of total) | 59 (56) |
| Hypercholesterolemia (N, % of total) | 48 (46) |
| Diabetes mellitus (N, % of total) | 9 (9) |
| Coronary artery disease (N, % of total) | 22 (21) |
| Atrial fibrillation (N, % of total) | 52 (50) |
| Heart failure | |
| NYHA functional class at the time of surgery (N, % of total) | |
| I | 9 (9) |
| II | 51 (49) |
| III | 42 (40) |
| IV | 1 (1) |
| Previous admission for heart failure (N, % of total) | 20 (19) |
| BNP (pg/mL ± SD) | 266 ± 342 |
| CRP (µg/mL ± SD) | 2.84 ± 1.67 |
| Medications | |
| ACE inhibitors (N, % of total) | 38 (37) |
| ARB (N, % of total) | 21 (20) |
| Spironolactone (N, % of total) | 6 (6) |
| Eplerenone (N, % of total) | 3 (3) |
| Diuretics (N, % of total) | 80 (78) |
| Betablockers (N, % of total) | 52 (51) |
| Digoxin (N, % of total) | 18 (17) |
| Statins (N, % of total) | 44 (43) |
| Echocardiographic parameters | |
| LVEF (mean ± SD) | 60.6 ± 10.4 |
| Systolic dysfunction (LVEF <60 %) (N, % of total) | 36 (35) |
| Severe systolic dysfunction (LVEF <35 %) (N, % of total) | 2 (2) |
| End diastolic diameter (mm ± SD) | 59 ± 7 |
| Indexed end diastolic diameter (mm/m2 ± SD) | 33 ± 4 |
| End systolic diameter (mm ± SD) | 40 ± 8 |
| Indexed end systolic diameter (mm/m2 ± SD) | 23 ± 7 |
| Systolic pulmonary artery pressure (mmHg ± SD) | 47 ± 12 |
| MVP etiologic subgroups | |
| Barlow disease (N, % of total) | 70 (68) |
| Fibroelastic deficiency (N, % of total) | 30 (29) |
| Syndromic MVP | 1 (1) |
| Associated to hypertrophic cardiomyopathy | 2 (2) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Pena, A.; Ibarrola, J.; Navarro, A.; Sadaba, A.; Tiraplegui, C.; Garaikoetxea, M.; Arrieta, V.; Matilla, L.; Fernández-Celis, A.; Sadaba, R.; et al. Activation of the Interleukin-33/ST2 Pathway Exerts Deleterious Effects in Myxomatous Mitral Valve Disease. Int. J. Mol. Sci. 2021, 22, 2310. https://doi.org/10.3390/ijms22052310
Garcia-Pena A, Ibarrola J, Navarro A, Sadaba A, Tiraplegui C, Garaikoetxea M, Arrieta V, Matilla L, Fernández-Celis A, Sadaba R, et al. Activation of the Interleukin-33/ST2 Pathway Exerts Deleterious Effects in Myxomatous Mitral Valve Disease. International Journal of Molecular Sciences. 2021; 22(5):2310. https://doi.org/10.3390/ijms22052310
Chicago/Turabian StyleGarcia-Pena, Amaia, Jaime Ibarrola, Adela Navarro, Alba Sadaba, Carolina Tiraplegui, Mattie Garaikoetxea, Vanessa Arrieta, Lara Matilla, Amaya Fernández-Celis, Rafael Sadaba, and et al. 2021. "Activation of the Interleukin-33/ST2 Pathway Exerts Deleterious Effects in Myxomatous Mitral Valve Disease" International Journal of Molecular Sciences 22, no. 5: 2310. https://doi.org/10.3390/ijms22052310







