Abstract
Most anticancer drugs target mitosis as the most crucial and fragile period of rapidly dividing cancer cells. However the limitations of classical chemotherapeutics drive the search for new more effective and selective compounds. For this purpose structural modifications of the previously characterized pyridine analogue (S1) were incorporated aiming to obtain an antimitotic inhibitor of satisfactory and specific anticancer activity. Structure-activity relationship analysis of the compounds against a panel of cancer cell lines allowed to select a compound with a thiophene ring at C5 of a 3,4-dihydropyridine-2(1H)-thione (S22) with promising antiproliferative activity (IC50 equal 1.71 ± 0.58 µM) and selectivity (SI = 21.09) against melanoma A375 cells. Moreover, all three of the most active compounds from the antiproliferative study, namely S1, S19 and S22 showed better selectivity against A375 cells than reference drug, suggesting their possible lower toxicity and wider therapeutic index. As further study revealed, selected compounds inhibited tubulin polymerization via colchicine binding site in dose dependent manner, leading to aberrant mitotic spindle formation, cell cycle arrest and apoptosis. Summarizing, the current study showed that among obtained mitotic-specific inhibitors analogue with thiophene ring showed the highest antiproliferative activity and selectivity against cancer cells.
1. Introduction
The key function of microtubules and mitotic spindles in cell division make them an attractive target in cancer therapy. Therefore, microtubule-targeting agents (MTAs), also named “spindle poisons” remain the most important of classical chemotherapeutics [1]. These drugs are divided into two main groups: microtubule stabilizing agents (including taxanes, epothilones, laulimalide) and microtubule destabilizing agents (colchicine, vinca alkaloids, combretastatins) with five different tubulin binding sites [2]. Microtubule polymerization and likewise depolymerization are necessary for correct cell division. Therefore, despite different actions, both types of MTAs disrupt microtubule dynamics, leading to aberrant mitotic spindle formation, cell cycle arrest at G2/M phase, activation of the mitotic spindle assembly checkpoint (SAC) and apoptosis [3]. However, classical MTAs are characterized by a narrow therapeutic index and reported resistance of some cancer cells, which encourages a search for new more selective and effective compounds. Most efforts are focused on the new colchicine-binding site inhibitors (CBSIs), because of their ability to overcome P-glycoprotein (P-gp)/class III β-tubulin mediated drug resistance and antiangiogenic/antivascular actions [4]. Colchicine, a prototype drug among CBSIs, that binds to tubulin with high affinity, is not used as an anticancer agent, mainly because of its very narrow therapeutic index [5,6]. Moreover, in spite of the fact hundreds of promising CBSIs have been tested in vitro and in vivo, there are no approved colchicine-binding site drugs for the treatment of cancer. Simultaneously with the search of new potential CBSIs, many efforts are made to obtain small molecules that target kinesins and kinases involved in mitotic spindle organization [7,8].
Our previously published data showed that 4-benzyl-5-phenyl 3,4-dihydropyridine-2(1H)-thione (S1) demonstrated promising antiproliferative activity, with observed cell cycle arrest at G2/M phase, aberrant mitotic spindle formation and inhibition of tubulin polymerization [9]. Pyridine (with piperidine) are two of the most common heterocyclic fragments found in Food and Drug Administration (FDA) approved drugs [10]. Interest in the anticancer activity of dihydropyridines (DHP) and structurally related dihydropyrimidines (DHPM) has been increasing since monastrol was first described [11]. Monastrol is the first known specific inhibitor of kinesin-5 (Eg-5), a motor protein involved in centrosome separation and bipolar spindle formation, required for correct chromosomes segregation [12]. However, monastrol anticancer activity is not satisfactory, encouraging research for more active analogues [13]. Kumar et al., using comparative molecular field analysis (CoMFA), showed that 1,4-dihydropyridine and 1,4-dihydropyrimidine motifs being bioisosteric pharmacophores are common for both compounds and required for their cytotoxicity [14].
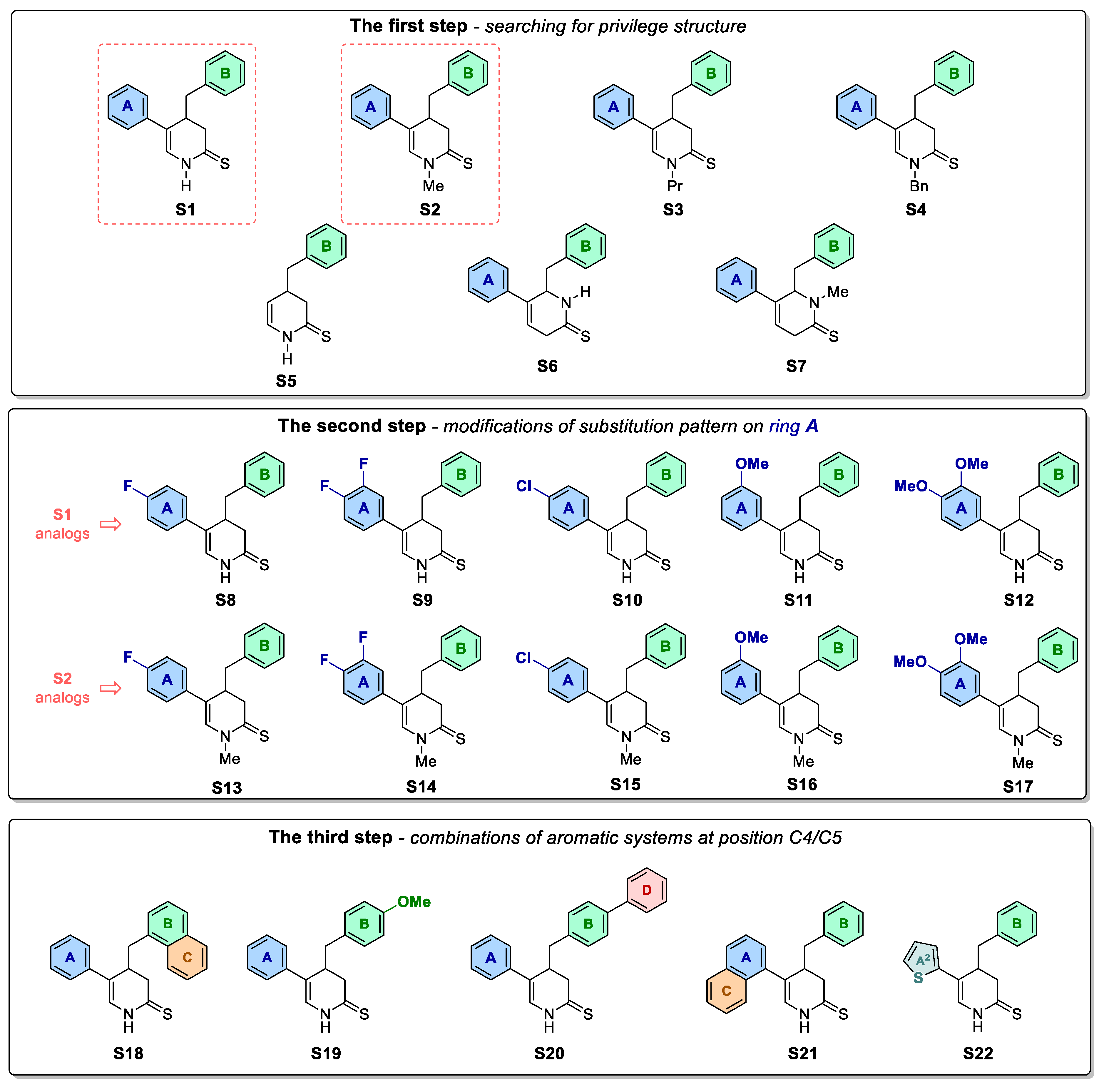
As we found our previously published results quite promising, we conducted further derivatization of the pyridine analogue S1 (Figure 1) aimed to obtain antimitotic inhibitor of satisfactory and specific anticancer activity. For this purpose, series of structural modifications followed by the antiproliferative study were conducted according to three stages: (1) selection of the most active compounds from N-H and N-substituted derivatives of the compound S1; (2) derivatization of the most potent compound from the first stage by adding appropriate substituents to A ring; (3) further modifications of aryl substituents at C4/C5 of 3,4-dihydropyridine-2(1H)-thione. Afterwards, mitotic-specific activity of selected compounds were evaluated against A375 melanoma cell line.
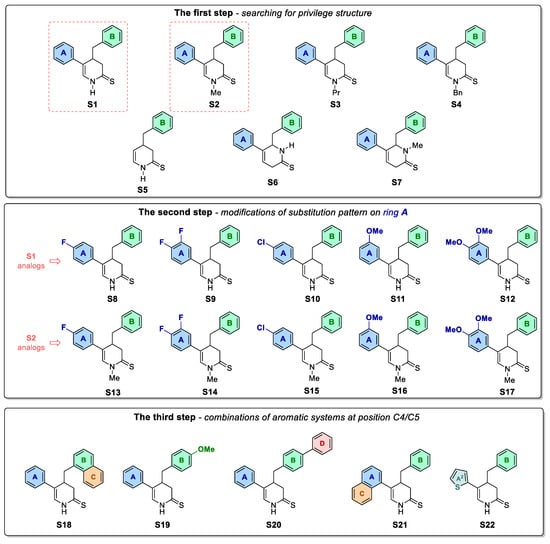
Figure 1.
Compounds S1-S22 obtained and tested within a three-step optimisation process.
2. Results and Discussion
2.1. Synthesis of Dihydropyridine-2-thiones S1-S22
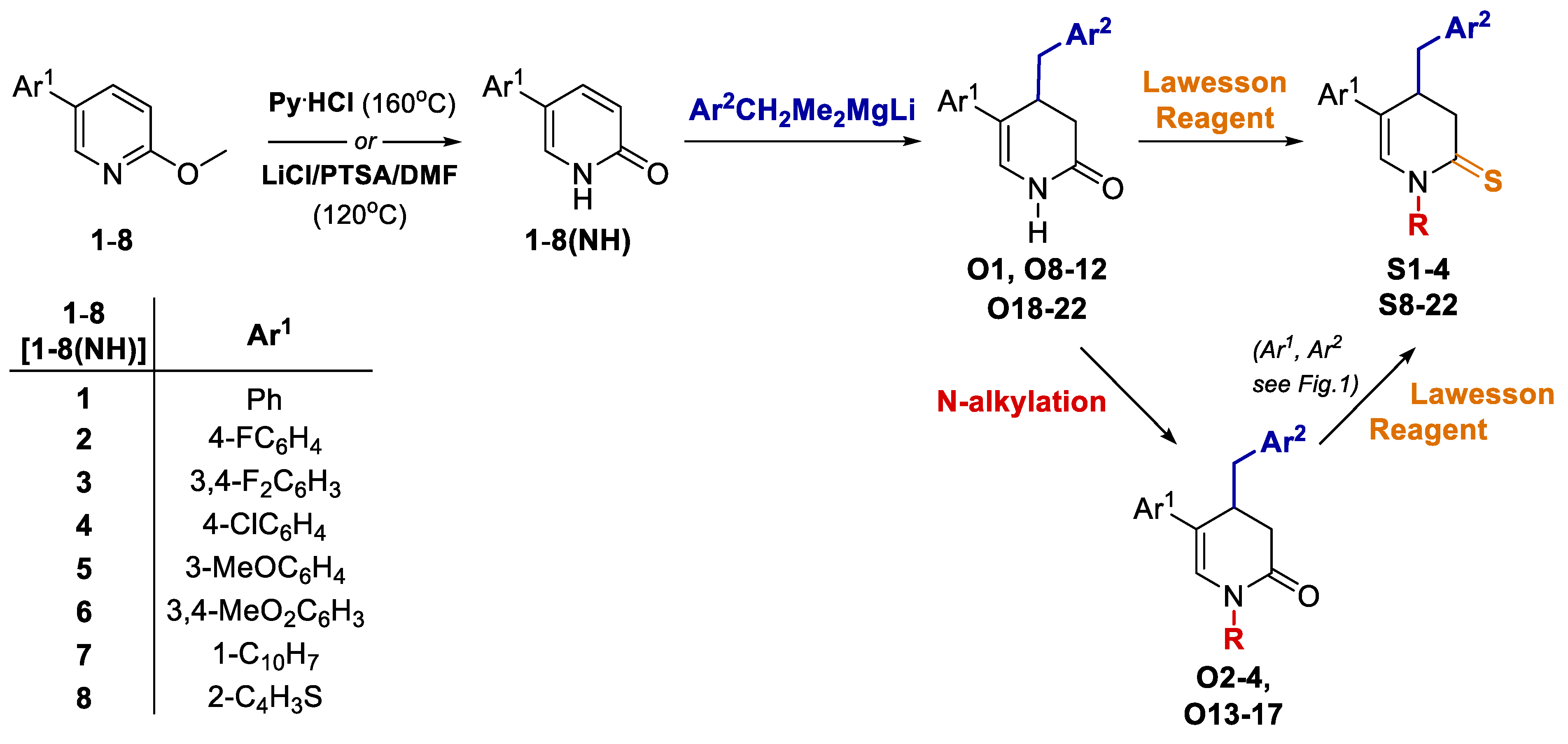
Compounds S1-S4 and S8-S22 (Figure 1) were obtained according to the sequence of reactions depicted in Scheme 1. It comprises demethylation of 5-aryl-2-methoxypyridines 1-8 by heating with the mixture of PTSA and LiCl or with pyridinium hydrochloride leading to corresponding 5-aryl NH 2-pyridones 1(NH)-8(NH). Starting compounds 1-8 were obtained mainly through the cross coupling reactions between 5-bromo-2-methoxypyridine and corresponding arylboronic acids in the presence of PdCl2 as catalyst. The key reaction consisted of the addition of ArCH2Me2MgLi complex to NLi 2-pyridones of 1(NH)-8(NH), yielding O1, O6 (not seen, see Appendix A) O8-O12 and O18-O22. Subsequently, part of obtained 4-benzylated 3,4-dihydropyridin-2-ones were next transformed to N-substituted derivatives O2-O4 and O13-O17. Finally, both NH and N-substituted 3,4-dihydropyridin-2-ones were transformed to their sulphur analogues S1-4 and S8-22 by thionation using Lawesson’s reagent. Compounds S5 was obtained by addition of benzylmagnesiate to 2-thiopyridone as described earlier [15], while compound S7 was achieved by thionation of oxygen analogue O7, obtained by addition of benzylmagnesiate to N-Me 5-phenylpyridin-2-one. Product S6 was gained by thionation of oxygen analogue O6, obtained as by-product during benzylation conducted with bigger amount of MeLi in comparison to a standard procedure. The structures of all compounds were elucidated with the aid of 1D NMR (1H, 13C, 13C-DEPT-135) and basic 2D NMR spectroscopy, low (GC-MS) and high (HRMS) resolution mass spectrometry and elemental analyses. The dihydropyridinones (O-serie) and dihydropyridinethiones (S-serie) were obtained in the form of racemates. Synthesis and spectroscopic data of S1-S4 and S6-S22 is described in the Materials and methods (4.1. Experimental part), while synthesis and spectroscopic data of their precursors is presented in the Appendix A (Synthesis of precursors of compounds S1-S22).
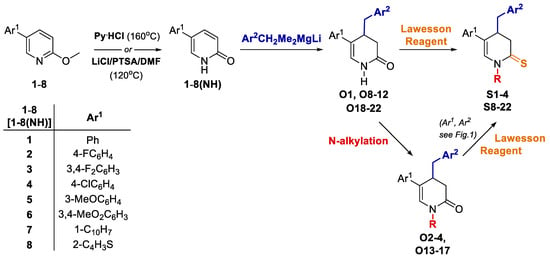
Scheme 1.
Sequence of the synthesis of targeted compounds S1-S4 and S8-S22.
2.2. The Antiproliferative Activity
The compound S1 with previously described promising anticancer activity [9] has been chosen as a starting point for the current study. It should be emphasized that biological activity of S1 was evaluated at single high concentration (100 µM) in the previous study. The Cell Proliferation Reagent (WST-1 assay) was used to evaluate the antiproliferative activity of S1 and its analogues against selected cancer cell lines including breast (MCF7), melanoma (A375), colon (HT-29), ovarian (SK-OV-3) and prostate (PC-3). A combretastatin (CA-4) one of the most extensively researched CBSIs [16], was used as reference control as it was predicted that S1 may bind to colchicine-binding side (which is presented and discussed below). The results are summarized as IC50 values in Table 1. The data revealed that from the following N-H and N-substituted derivatives compound S1 with N-H moiety and S2 with N-Methyl group were the most promising (IC50 values equal 4.33 ± 1.00 µM and 12.55 ± 2.16 µM against A375, respectively). Moreover, structure-activity relationship study of the first step showed that benzene ring is essential for the cytostatic activity of the studied structure since the analogue without 5-phenyl group (S5) lost anticancer activity (IC50 > 100 µM). The importance of that group was previously confirmed in case of dihydropyrimidinethiones derivatives [17]. In spite of increase lipophilic properties, compounds S6 and S7 (isomers of S1 and S2, respectively) were markedly less active, probably as a result of different position of nitrogen atom in relation to phenyl group.

Table 1.
The IC50 values (μM) determined using WST-1 assay after 48 h of treatment. Data are expressed as the mean ± SD from at least three independent experiments.
During the second task of the study compounds S1 and S2 were further derivatized by adding appropriate groups to A-ring. Selected groups were introduced on the basis of their π parameter, determining influence of different substituents on Log p value: two derivatives (S8/S13 and S9/S14) containing respectively one or two fluorine atoms (each of F atoms increases Log P by 0.13), one derivative (S10/S15) containing chlorine, increasing Log p value by 0.51, two derivatives (S11/S16 and S12/S17) containing one or two methoxy groups, respectively (each of the groups decreases Log p value by 0.26 compared to the original compound). In the present study we did not observe direct correlation between Log p values and antiproliferative activity, as it was noted in our previously published data, where the most lipophilic pyrimidine analogues were also the most potent growth inhibitors [17]. The introduction of substituents at the para-position of the phenyl ring (compounds S8-S17) resulted in a reduction of antiproliferative activity against all selected cancer cell lines, as compared with the activity of the unsubstituted A-ring analogue (S1 and S2).
When the effect of electron-releasing (ERG’s) or electron-withdrawing groups (EWG’s) at the phenyl ring was compared, compounds S11 and S16 with electron-donating methoxy groups were generally the most potent among –N-H and –N-Me analogues, respectively. Our data are compliant with Romagnoli et al. study, where the presence of electron-donating group (like methyl, methoxy) rather than electron-withdrawing groups on the phenyl moiety at the position of the 2-alkoxycarbonyl-3-(3′,4′,5′-trimethoxyanilino)thiophene system contributed to increase biological activity [19]. Surprisingly, in the current study two methoxy group (S12 and S17) contributed to decrease anticancer potential. In general, during the second stage of the study, -N-H analogues (S8-S12) were more potent than -N-Me counterparts (S13-S17). Therefore, the antiproliferative activity of -N-H derivatives with aromatic rings at C4/C5 position were evaluated at the third stage. As shown in Table 1, additional modifications of aryl rings at C4 and C5 contributed to increased anticancer activity (IC50 values of compounds S18-S22 ≤ 10 µM), however, an analogue with thiophene ring at C5 pyridine-2(1H)thione (S22) was the most effective one, with IC50 value of 1.71 ± 0.58 µM and 1.67 ± 1.47 µM against A375 and SK-OV-3 cell line, respectively. These findings correspond with several previous studies where thiophene, benzothiophene and fused thienopyridine or thienopyrimidine rings are presented in a variety of anticancer compounds [19,20,21], including CBSIs [22,23,24].
2.3. Selectivity-Index
The starting compound (S1) and its two derivatives defined as the most active compounds in the antiproliferative study (S19 and S22) were selected for further biological study, using A375 cells, that presented the highest sensitivity to the tested compounds. At first, the antiproliferative assay (WST-1) was used to estimate the selectivity-index (SI) defined as ratio of IC50 for normal cells to IC50 for cancer cells. Cancer cell line A375 and a non-tumor human melanocyte cell line (HEM) were used for that purpose. As shown in Table 2, all the newly evaluated compounds showed better selectivity against cancer cells when compared with reference: CA-4. Similar to our study, Amaral et al. presented CA-4 as non-selective cytotoxic agent. Namely, the authors revealed that CA-4 showed higher antiproliferative activity against human lymphocytes than tumor cell lines, except for HL-60 and OVCAR-8 [25]. Whereas, Blay et al. reported that in spite of the combination of ombrabulin (CA-4 analogue) and cisplatin significantly improved progression-free survival in patients with advanced soft-tissue sarcomas, serious adverse effect excluded this strategy from anticancer therapy [26]. According to the “selectivity criteria” (SI > 10 [27]) only the compound S22 could be considered as selective. Similarly, Hura et al. revealed that among twenty-one tested CA-4 derivatives, C-13 analogue with thiophene as ring B was the most potent and selective against cancer cells [24].

Table 2.
Selectivity-index (SI) for selected compounds.
2.4. Apoptosis Detection
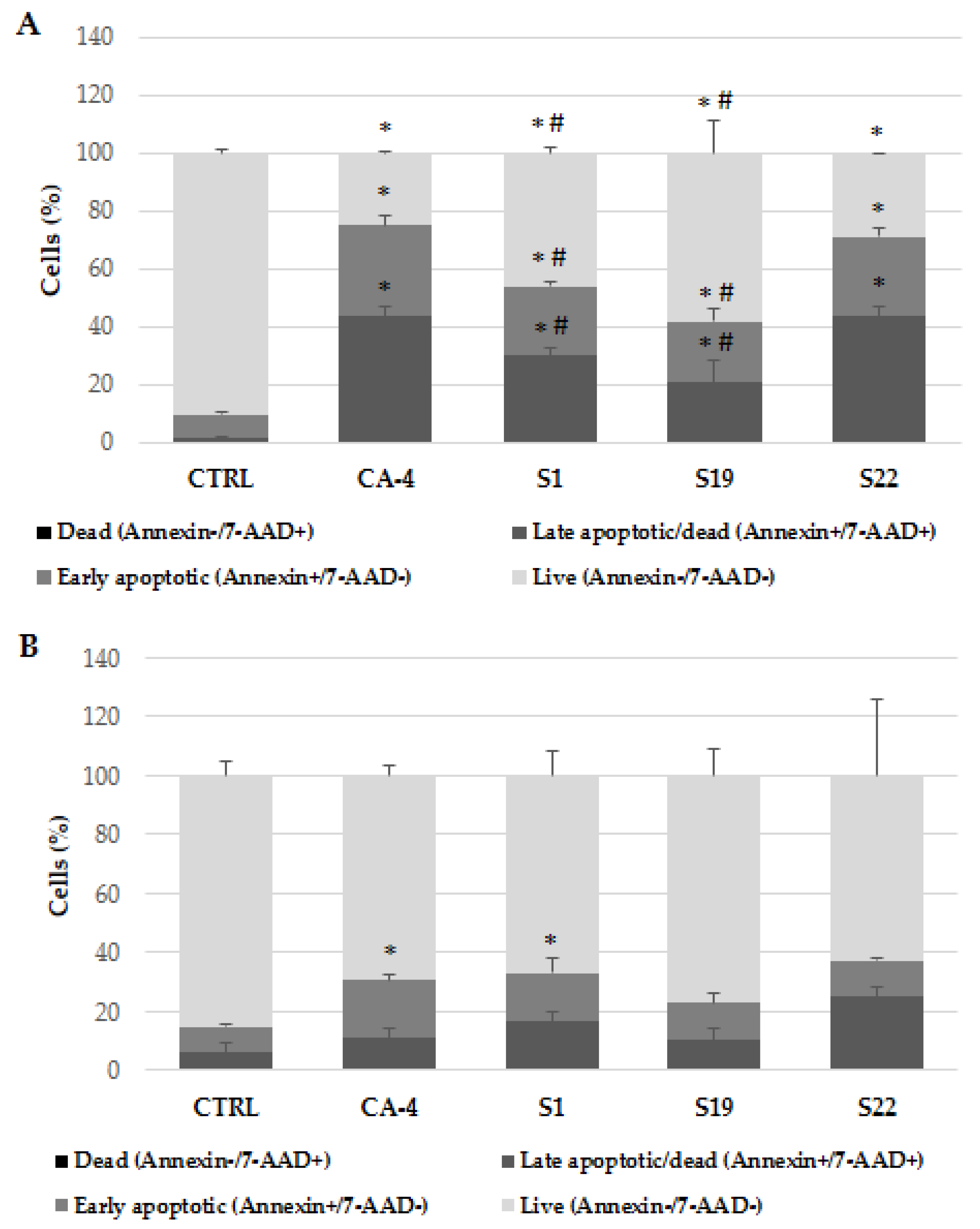
Since apoptosis is identified as one of the most common mechanism of drug induced cell death, an apoptotic marker was evaluated using flow cytometry in the next step of the study. The assay used was based on the detection of phosphatidylserine (PS) on the surface of apoptotic cells. A dead cell marker (7-AAD) is also used as an indicator of the cell membrane structural integrity. Simultaneous staining with two dyes allows to distinguish (after debris excluding) viable (Annexin V-PE− and 7-AAD−), early apoptotic (Annexin V-PE+ and 7-AAD−) and late apoptotic/already dead cells (Annexin V-PE+ and 7-AAD+). The A375 and HEM cells were treated with the compounds: S1, S19 and S22 at 10 µM for 48 h. CA-4 was used as reference. As shown in Figure 2A, the population of living (non-apoptotic) cancer cells significantly decrease after treatment. Moreover, compound S22 showed the most comparable pattern to CA-4 with the percentage of early and late apoptotic cells equal to 27.42 ± 3.20% and 43.58 ± 3.30% for S22 and 31.57 ± 3.16% and 43.42 ± 3.43% for CA-4, in A375 and HEM cells, respectively. As for S1, S19 and S22, more apoptotic cells could have been expected after treatment with 10 µM concentration (based on IC50 values from WST-1 assay), obtained data from apoptosis test suggest that those compounds possess stronger antiproliferative than direct cytotoxic potential. Simultaneously, analysis was performed in non-cancerous cells what allowed to confirm selectivity observed during antiproliferative study (Figure 2B). Namely, only CA-4 and S1 significantly increased the percentage of early apoptotic cells to 19.74 ± 2.19% and 16.43 ± 4.88%, respectively when compared to untreated control (8.8 ± 1.1%). Surprisingly, during apoptosis detection in both cell lines, the differences between reference CA-4 and our compounds seemed less distinct than during IC50 evaluation. Representative individual experiments (scatter diagrams with cell populations divided into quadrant dot plots specific for Annexin V/7-AAD staining) are shown in Figures S1 and S2. Additionally, optical microscopy imaging of A375 and HEM cells was performed just before apoptosis analysis of harvested cells. As shown in Figure S3, A375 treated cells had lower density in comparison with non-treated cells, while increased the number of dead cells could be observed only in CA-4 and S22 exposed cells, which was consistent with flow cytometry results. In the HEM cells, the most striking difference between control and treated cells was in cell morphology, especially in case of CA-4 and S22 exposed cells (Figure S4).
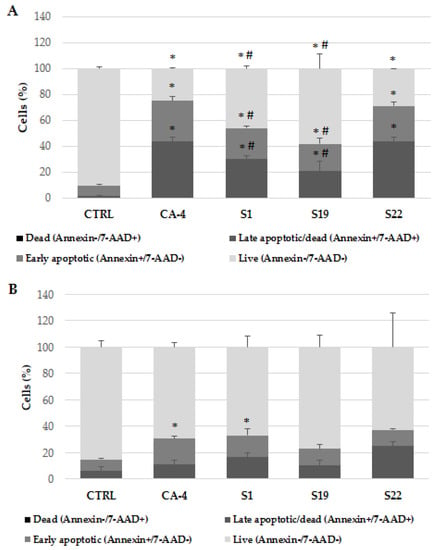
Figure 2.
The effect of indicated compounds (at 10 µM) on the induction of apoptosis in A375 (A) and HEM (B) cells after 48 h treatment, subsequently stained with Annexin V-PE and 7-AAD and analyzed by flow cytometry. Data are expressed as the mean and SD from at least three independent experiments; * p < 0.05 vs. ctrl; # p < 0.05 vs. CA-4.
2.5. Cell Cycle Analysis
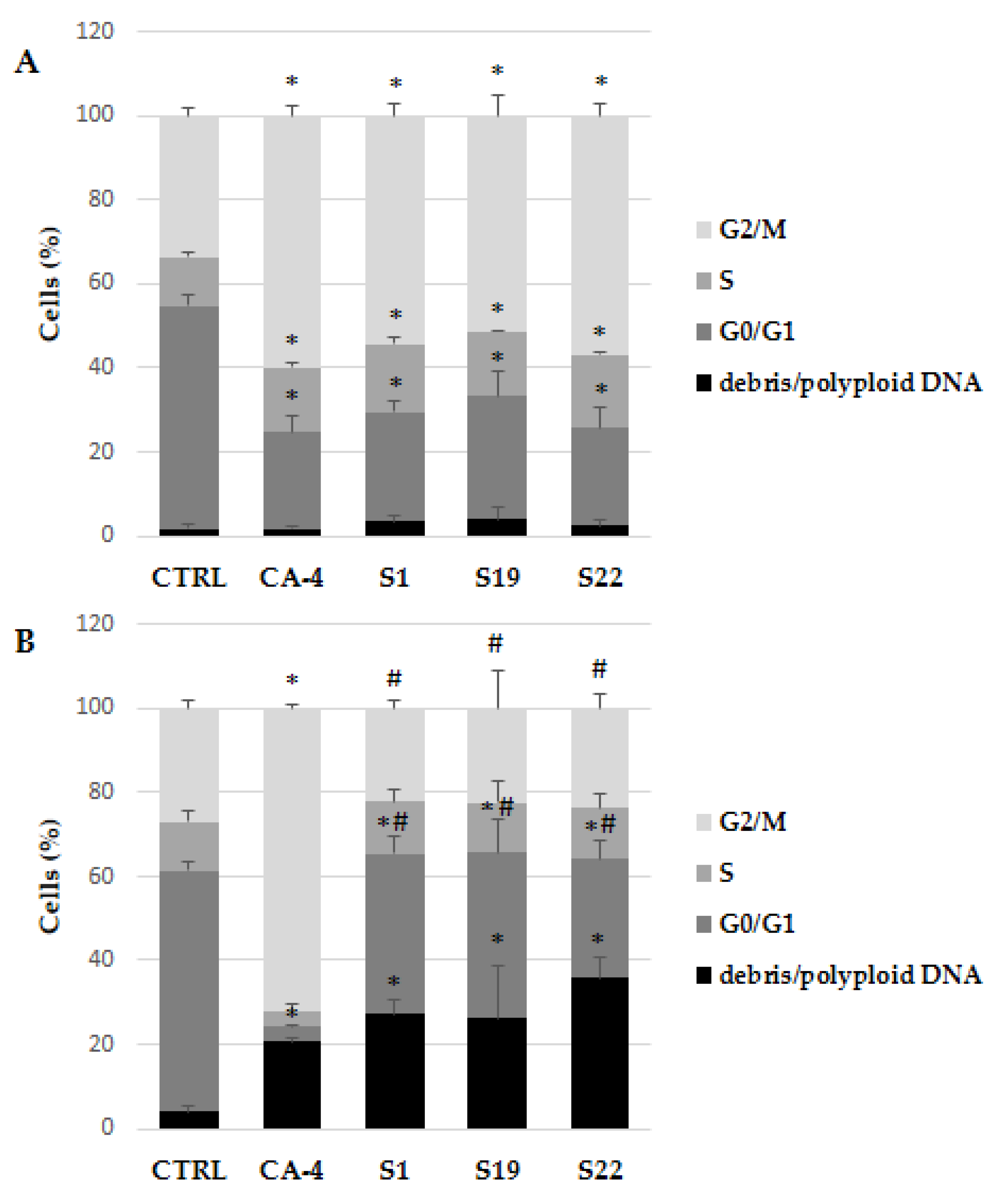
Since growth inhibition may be a result of arrest cell cycle at a particular point, specific antiproliferative activity of the selected compounds was further evaluated by flow cytometry, for the impact on the cell cycle progression in A375 cells. The assay utilizes PI-based staining of DNA content to discriminate and measure the percentage of cells in each cell cycle phase (i.e., G0/G1, S, and G2/M) [28]. In present study, the cells were treated with tested compounds at 10 µM concentration for 6 and 24 h, aimed to detect cell cycle arrest leading to apoptosis at 48 h of treatment. As expected, S1, S19 and S22 compounds caused accumulation of A375 cells in a tetraploid (4N) state, simultaneously decreasing the percentage of cells in G0/G1 phase after 6 h of treatment (Figure 3A) and Figure S5A–E. The percentage of A375 cells in G2/M phase was 54.46 ± 2.94%, 51.43 ± 4.77% and 56.95 ± 3.09% for S1, S19 and S22, respectively, compared to 33.64 ± 1.97% in control. These data were comparable with reference CA-4, where the percentage of arrested cells was 60.00 ± 2.54%. Surprisingly, after 24 h, when CA-4 induced mitotic block increased to 72.36 ± 1.10%, the percentage of G2/M phase cells treated with tested compounds was much lower and not significantly different from control. However, it should be pointed that simultaneously the number of hypodiploid DNA (sub-G0/G1) and/or polyploid DNA (population after G2/M phase) markedly increased (Figure 3B). As shown in Figure S5F–J, the sub-G0/G1 population increased significantly in S1, S19 and S22 treated cells which is considered as characteristic feature of cells undergoing apoptosis [29], whereas in CA-4 treated cells, a DNA index dramatically increased as a result of DNA polyploidization. Polyploidy is also typical for cells blocked in mitosis by colchicine (described as ‘‘c-mitosis’’ or ‘‘colchicine-mitosis’’), as a results of partial or complete absence of spindle apparatus following the breakdown of nuclear envelope, condensed chromosomes, and undivided centromeres [30]. Our data are consistent with Gilson et al. study, where after initial accumulation of H358 and HeLa cells in mitosis as a result of pyrrolopyrimidine (PP-13) treatment, the percentage of G2/M arrested cells was gradually decreasing, concomitantly with the accumulation in the sub-G1 and polyploid fraction [31]. As mentioned above, inhibition of tubulin polymerization and mitotic spindle formation may be one of the reasons for polyploid nuclei formation. Therefore, in the further study, the impact of S1, S19 and S22 compounds on mitotic spindle formation and tubulin polymerization was evaluated.
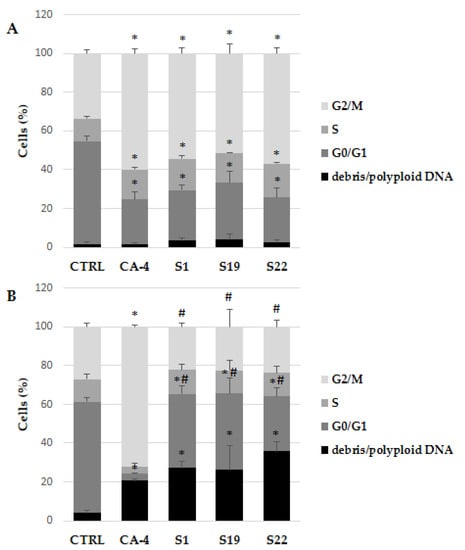
Figure 3.
Flow cytometry analysis of cell cycle in A375 cells using PI staining after exposure to indicated compounds (at 10 µM) for 6 h (A) and 24 h (B). Data are expressed as the mean and SD from three independent experiments; * p < 0.05 vs. ctrl; # p < 0.05 vs. CA-4.
2.6. Impact on Mitotic Spindle Formation
As G2/M cell-cycle arrest is strongly correlated with microtubule disruption [32], in the next step the effect of tested compounds on mitotic spindle formation was evaluated using confocal microscopy imaging. After 6 h-incubation of A375 cells with S1, S19 and S22, the cells were fixed and α-tubulin and chromosomes were stained. At first it should be emphasized that the morphological differences between treated and untreated cells were noticeable only during mitosis, confirming mitotic-specific activity of tested compounds. Similarly, spindle microtubules are more sensitive to colchicine than the interphase microtubules [33], whereas taxanes and vinca-alkaloids also affect interphase cells [34]. Cell-permeable small molecules that perturb mitosis without effects on microtubule/cytoskeleton in interphase may be useful in anticancer therapy and contribute to less pronounced adverse effects. For example, as differentiated neurons are in a post-mitotic state and are not be able to re-enter the cell cycle [35], this phase-specific activity may contribute to reduced neurotoxicity, which is one of the drawbacks of classical MTAs.
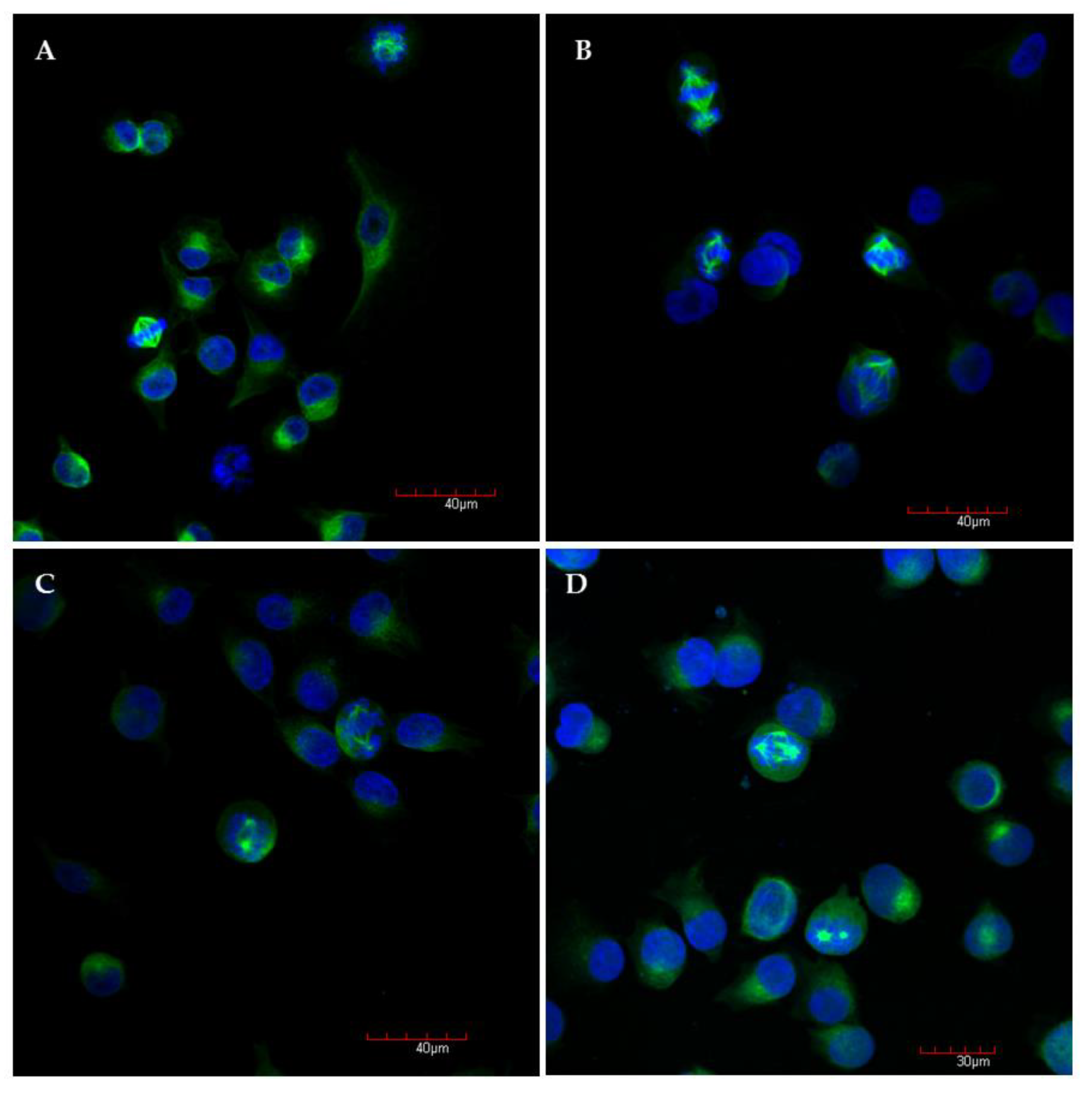
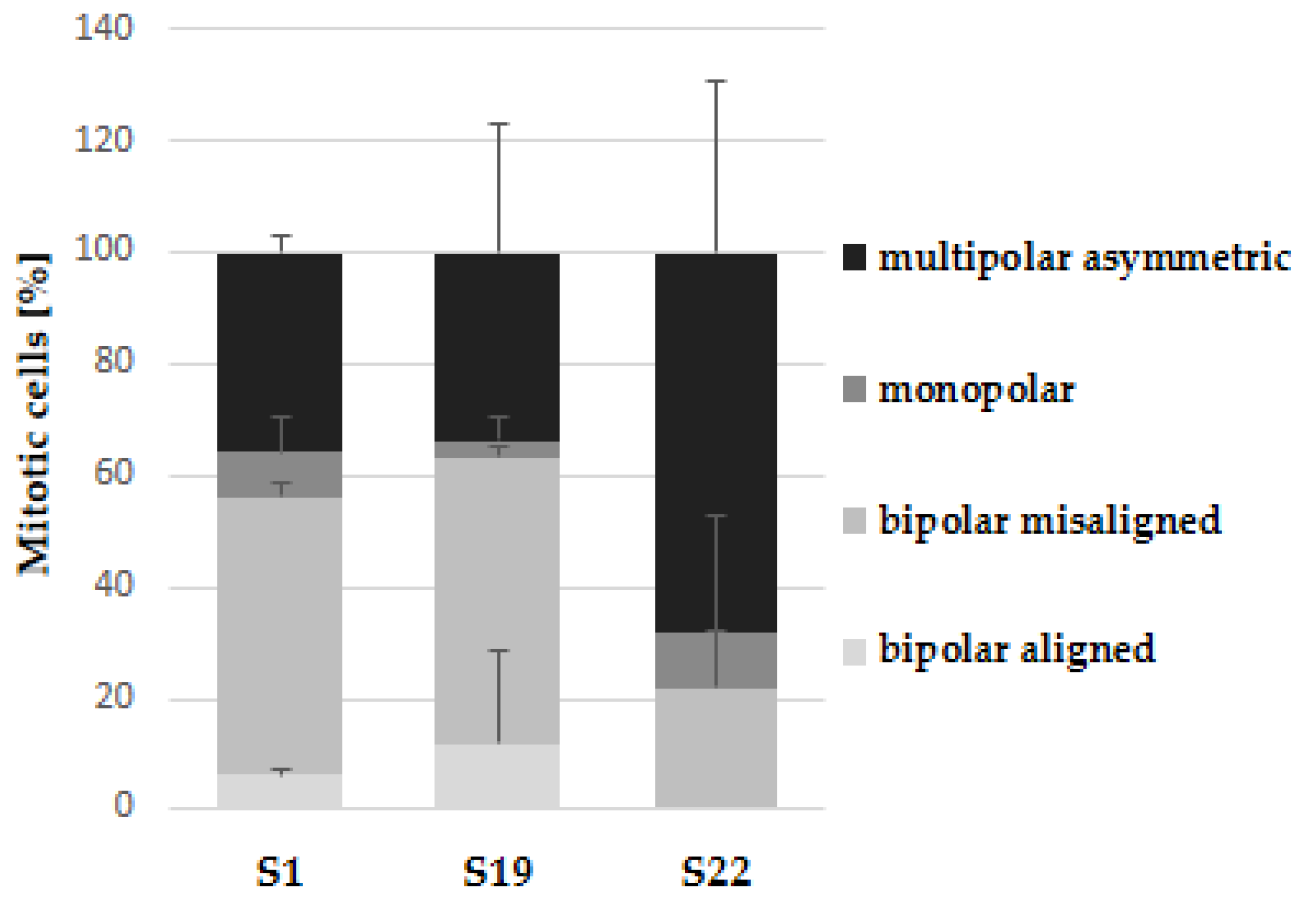
As depicted in Figure 4A, the control cells in mitosis showed bipolar spindle formation with chromosomes arrangement in the central metaphase plate. In contrast, compound S1, S19 and S22 caused improper changes in mitotic spindle organization (Figure 4B–D). We could describe the three most common abnormal phenotypes: (1) bipolar spindles with chromosomes arrangement in the central metaphase and additional “cloud” of chromosomes around of centrioles (misalignment chromosomes), (2) monoastral spindles, (3) multipolar asymmetric asters (Figure 5). Observed aberrations were previously associated with strong defects in chromosome congression and alignment, and may be connected with prometaphase blockade [31]. Multipolar and “bipolar misaligned” spindles were described by Jaunky et al. in cells treated with colchicine and C75 (new CBSIs), depending on concentration and (in the case of C75) cell line used. The authors supposed that the discrepancy in mitotic phenotypes caused by C75 between different cell lines might be connected with genetically-dependent sensitivity to spindle disruption [36]. However, observed aberrations are not only specific for tubulin inhibitors but also other compounds targeting mitotic spindle. Namely, bipolar spindles with chromosomes aligned at metaphase and misaligned chromosomes located close to the spindle poles are also characteristic for inhibitors of kinesin-like protein (KIFC1) and centromere-associated protein E (CENP-E), kinesins essential for bipolar spindle formation and correct chromosome alignment, respectively [37,38]. Therefore, it is not possible to precisely define the molecular target of currently tested compounds on the base of observed phenotypes.
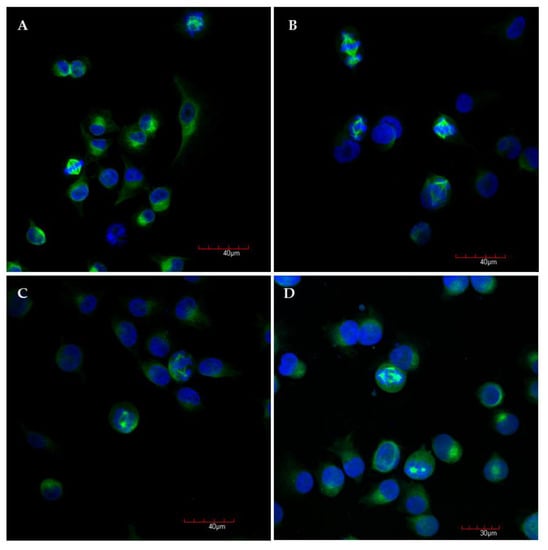
Figure 4.
Representative confocal microscopy images of A375 cells after 6 h incubation with medium and 0.2% DMSO (A), compound S1 (B), S19 (C), S22 (D) at concentration of 10 µM, with stained α-tubulin (green) and chromosomes (blue); magnification ×20 plus zoom ×3 during acquisition.
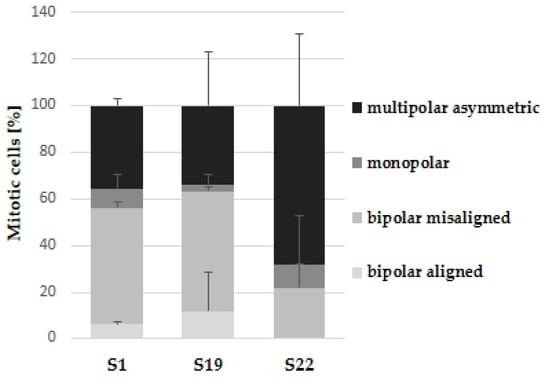
Figure 5.
Quantification of mitotic spindle phenotypes of A375 cells after 6 h incubation with indicated compounds at 10 µM. Data are expressed as the mean and SD from two independent experiments.
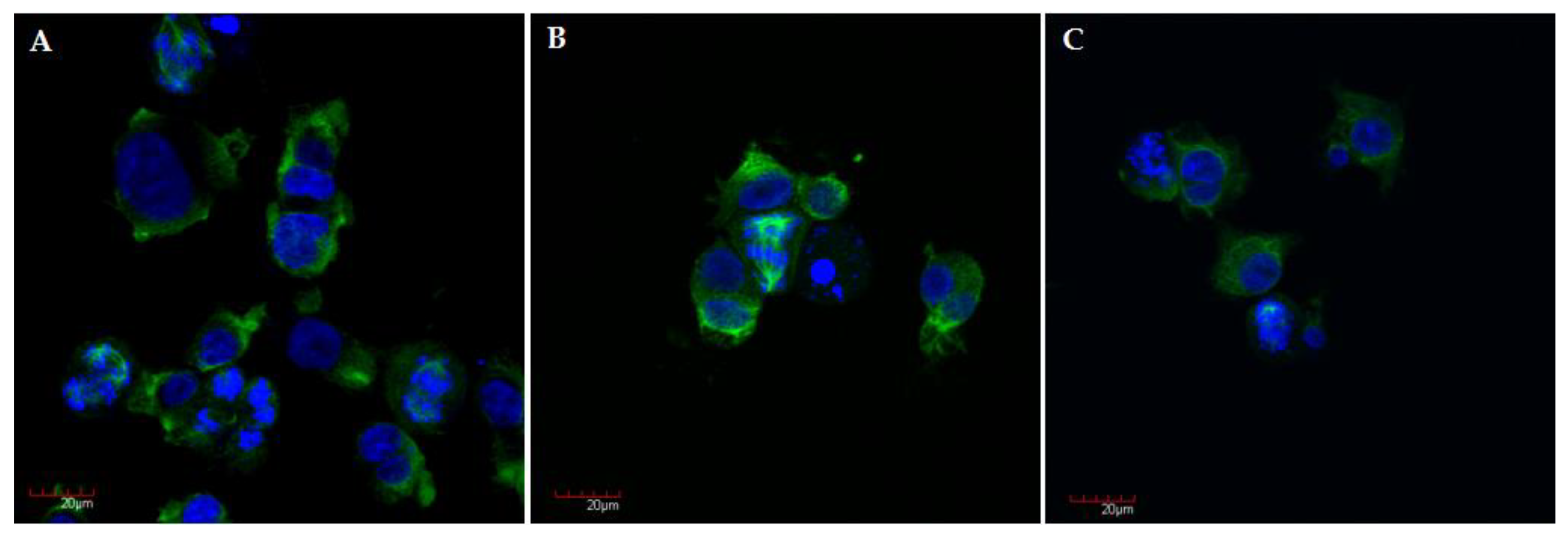
In agreement with the flow cytometry analysis, the number of huge polynucleated cells and apoptotic bodies increased after 24 h of treatment (Figure 6). Observed changes are characteristic for mitotic cell death (MCD) often called a mitotic catastrophe. Typical features of this process include block in mitosis, mitotic spindle disorganization, failed chromosome segregation, and formation of multinucleated cells [39,40]. Mitotic catastrophe has been described as predominant form of cell death mediated by CA-4 and CA-4 analogue [41,42].
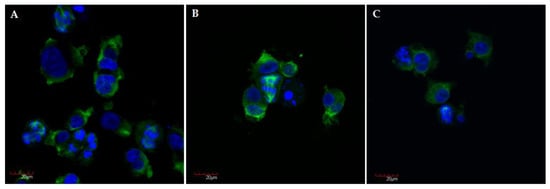
Figure 6.
Representative confocal microscopy images of A375 cells after 24 h incubation with compound S1 (A), S19 (B), S22 (C) at concentration of 10 µM, with stained α-tubulin (green) and chromosomes (blue); magnification ×40 plus zoom ×2 during acquisition.
2.7. Tubulin Polymerization in Cells and Cell-Free Conditions
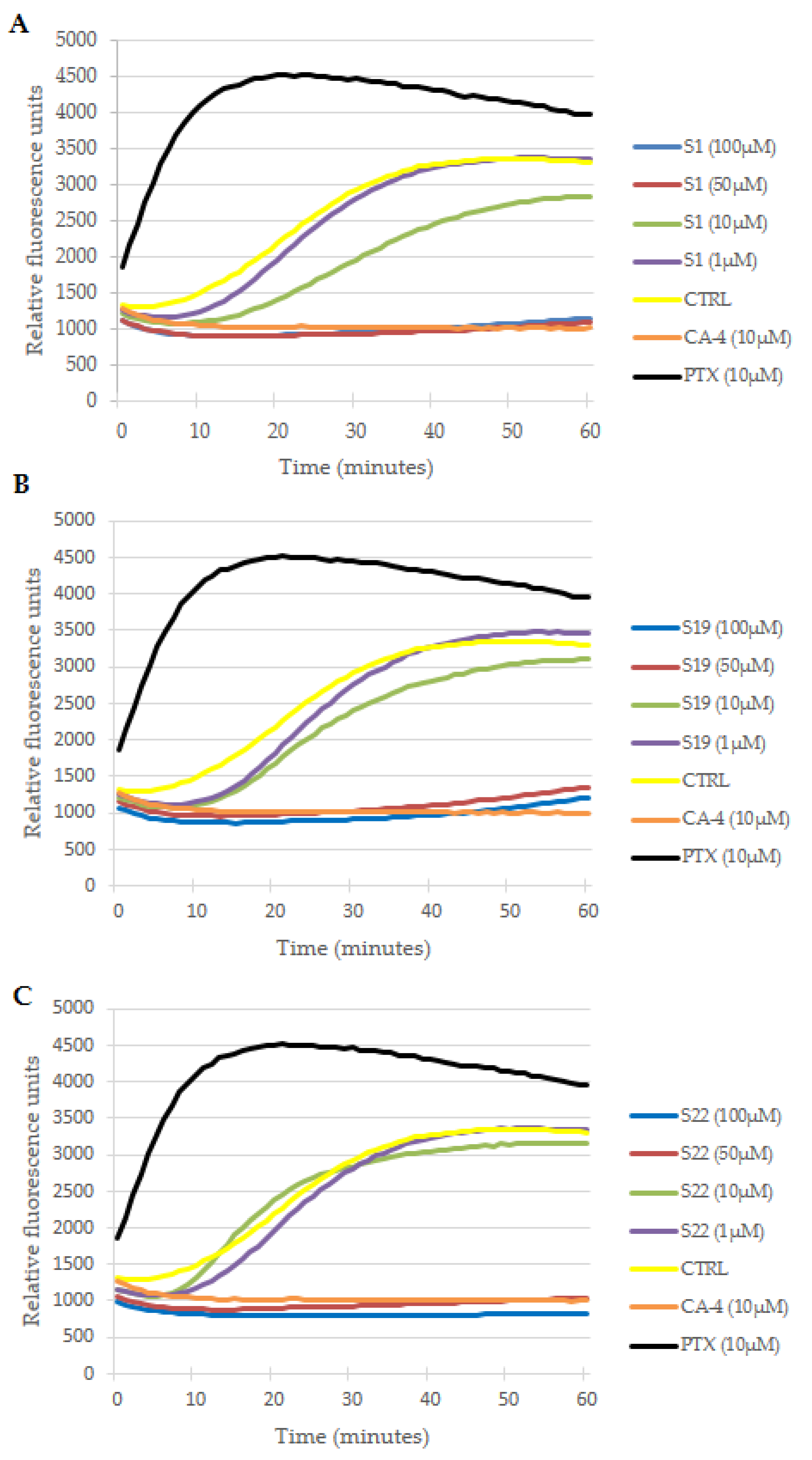
The observed aberrations in mitotic spindle morphology encouraged us to verify whether this effect was a result of interaction with microtubule dynamics, which is essential for a proper mitotic process [43]. A direct effect of S1, S19 and S22 derivatives on tubulin behavior in cell free conditions was evaluated using a fluorescence-based tubulin polymerization assay. The activity of the tested compounds was compared with paclitaxel (PTX), CA-4 and 0.2% DMSO as references and control, respectively. As shown in Figure 7, DMSO in the control sample had no direct effect on spontaneous tubulin self-assemble, where three typical phases of microtubule formation (namely: nucleation, growth and steady state inhibition) could be observed. The reference PTX (10 µM) eliminated the nucleation phase and enhanced the V max (maximal slope values for the growth phase), whereas CA-4 (10 µM) inhibited tubulin polymerization, resulting in a decrease in V max and reduction in final polymer mass protein. The overlapping curves of CA-4 (10 µM) and tested compounds at 50 µM and 100 µM indicated that S1, S19 and S22 act as tubulin polymerization inhibitors. However, inhibition of tubulin assembly by the tested compounds observed only at higher concentrations points that they are less active than reference drug in this regard. At 10 µM (green curves in Figure 7) S1 was the most effective among the tested compounds, as delayed start of tubulin assembly, reduced microtubule polymerization speed and total polymer mass could still be clearly seen at that concentration. At 1 µM none of the investigated derivatives had any significant effect on tubulin polymerization (all purple curves finally overlapped with control in Figure 7). These observations were confirmed by IC50 evaluation presented in Table 3.
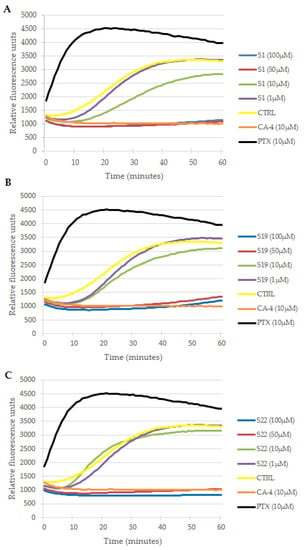
Figure 7.
Effects of S1 (A), S19 (B) and S22 (C) derivatives on tubulin polymerization in cell free conditions. Increasing relative fluorescence units is indicative of tubulin polymerization. Paclitaxel (PTX) and combretastatin (CA-4) were used as references and DMSO (0.2%) as a vehicle control. Curves of tested compounds represent the mean from at least three independent experiments. Error bars are not shown for the sake of clarity.

Table 3.
The IC50 values (μM) of inhibition of tubulin polymerization. Data are expressed as the mean ± SD from at least three independent experiments.
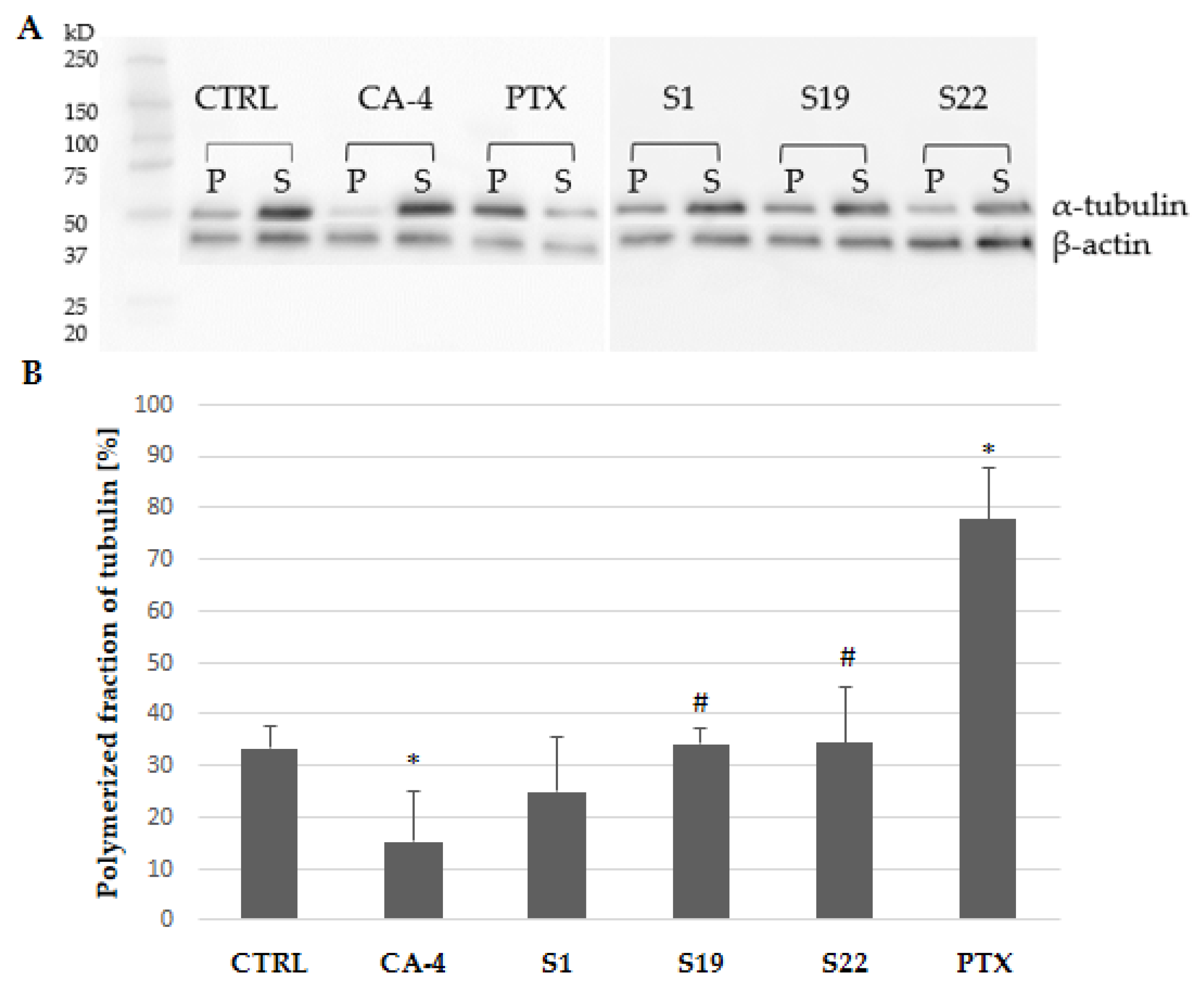
The obtained data are in line with the results from another tubulin polymerization assay used (based on western blot method). Analysis was limited to the single concentration of the tested compounds (10 µM). As expected, CA-4 and PTX (used as references), increased the amount of tubulin in the soluble or polymeric fraction, respectively (Figure 8A). As shown in Figure 8B, α-tubulin polymer fraction was slightly but not significantly reduced in S1-treated A375 cells when compared with the control (24.84% vs. 33.36%). One could expect that the observed effect would be more distinct at higher concentrations, as our previously published data showed a reduction of polymerized tubulin fraction to 0.58% in S1 exposed cells at 100 µM [9]. Compounds S19 and S22 did not influence tubulin polymerization as evaluated by a western blot method.
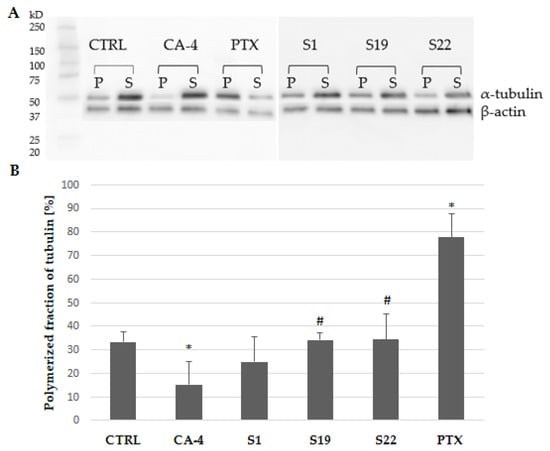
Figure 8.
(A) Western blot analysis of polymerized microtubules (P) and soluble tubulin dimers (S) fraction in the cells after 6 h treatment with indicated compounds. Original images for blots in Supplementary Materials (Figure S6). (B) Percentage of α-tubulin in the pellet fraction after densitometric analysis of western blot gel. Each densitometric value of tubulin was normalized to the densitometric value of β-actin. Data are expressed as the mean and SD from four independent experiments; * p < 0.05 vs. ctrl; # p < 0.05 vs. CA-4).
2.8. Colchicine-Binding Site Study
The aforementioned common behavior of our compounds and colchicine encouraged us to verify the possible binding mode. For this purpose a molecular docking study was performed. Colchicine binds to tubulin at the interface between the α and β subunits. The majority of the compound is buried in the β subunit. As revealed by X-ray analysis of the tubulin-DAMA/colchicine complex structure, the A and C colchicine rings interact with the β-subunit of tubulin, while the B ring of the side chain interacts with the α-subunit. One of its methoxy oxygens of trimethoxyphenyl (TMP) moiety (A ring) is involved in hydrogen bonding with the thiol of Cys241β [2,45]. The Cys241β has been previously indicated as an important target for colchicine site agents [46]. According to Nguyen et al., all of the 15 structurally diverse CBSIs formed hydrogen bond with Cys241β (Cys239β in that study) [47]. Therefore, we focused on this crucial residue during molecular docking study. As shown in Figures S7 and S9 the B-ring of S1 and S22 (first possible mode of action) or A-ring (thiophene) of S22 (Figure S10, second possible docking model) were directed toward Cys239β, but did not form hydrogen bond with sulphur atom of this amino acids. Bhattacharyya et al. indicated that the pi electron clouds of the thiophene and the phenyl rings of nocodazole (well known CBSIs) can act as hydrogen bond acceptors by forming a weak hydrogen bond coupled with hydrogen bond donor such as the thiol group of Cys239β [33]. However, recently data revealed that nocodazole did not interact with the α-tubulin and was situated deeper in β-tubulin binding pocket overlapping very little with colchicine in the regard of binding site [48]. Our findings confirm a crucial role of sulphur atom and -NH group of the pyridine-2(1H)thione (C-ring) moiety in hydrogen bond formation. Namely, the sulphur atom of S1 showed hydrogen bonding interaction with amino acids of tubulin (Asn101α) (Figure S7). The S19 formed two potential hydrogen bonds between sulphur atom and -NH group of compound and Gln247α and Ala354β, respectively (Figure S8). Whereas S22 could form only one hydrogen bond between -S and Gln247β (Figure S9) or -NH and Val238β (Figure S10). Our data are in line with previous studies that pointed to the sulphur atom as an essential factor for anticancer activity of monastrol and other thio-derivaties (3,4-dihydropyrimidin-2(1H)-thiones). The substitution of sulphur by an oxygen led to the loss of the cytotoxic activity, probably due to the “soft nature” of the sulphur atom which makes compounds more nucleophilic [49].
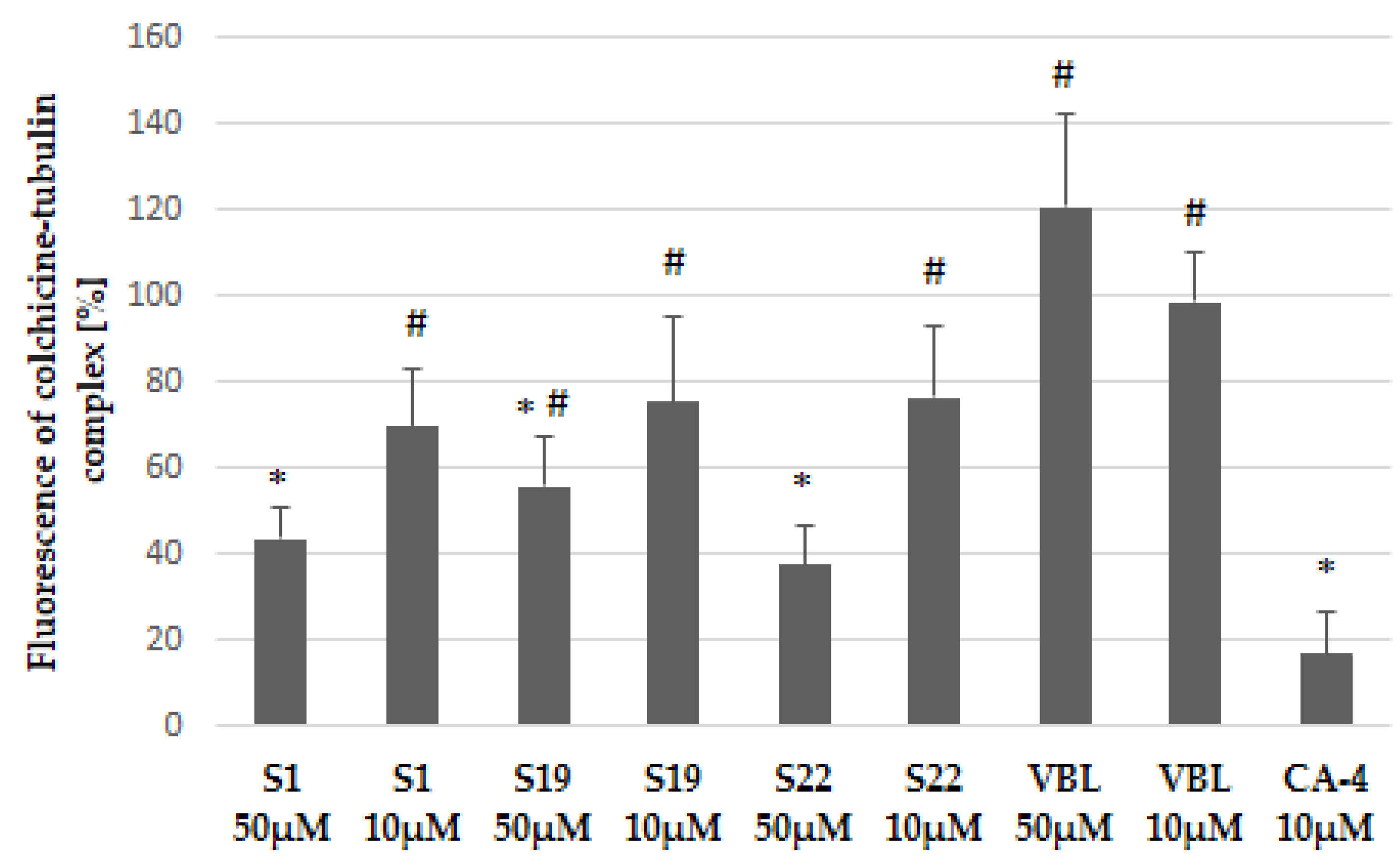
These molecular docking results were further supported by a fluorescence-based competitive binding assay. Namely, we evaluated the ability of compounds S1, S19 and S22 to compete with colchicine which intrinsic fluorescence increases upon binding to tubulin. We chose two the most critical concentration of tested compounds, selected during the cell free tubulin polymerization study (10 µM and 50 µM). As shown in Figure 9 the fluorescence of colchicine-tubulin complex was reduced in the presence of S1, S19 and S22 in dose-dependent manner suggesting that these compounds bind at the colchicine site and may belong to CBSIs. Vinblastine (VBL, used as negative control) did not compete with colchicine because it inhibits tubulin polymerization by binding at different site, located on β-tubulin chain. However, the investigated S1, S19, and S22 compounds had lower affinity to colchicine-binding site than reference CA-4, and those findings were also in line with the results of the tubulin polymerization study described above.
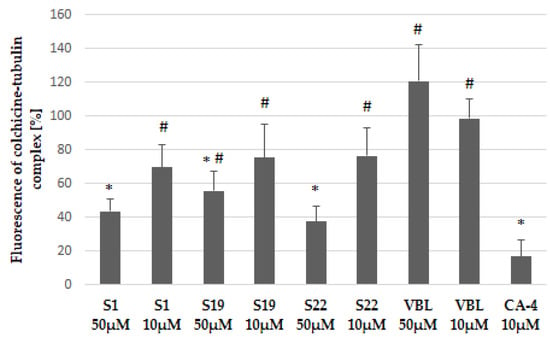
Figure 9.
Colchicine competitive binding assay. Results have been shown as intrinsic fluorescence of colchicine-tubulin complex normalized to control (0.2% DMSO). CA-4 was used as a positive control whereas vinblastine (VBL), which binds at vinblastine site, was used as negative control. Data are expressed as the mean and SD from three independent experiments for S1, S19, S22, CA-4 and two independent experiments for VBL; * p < 0.05 vs. ctrl; # p < 0.05 vs. CA-4.
3. Conclusions
The current study is a part of wider research related to an increasing interest in nitrogen- and sulphur-based heterocycles as promising anticancer agents [10]. A structure-activity relationship study resulted in identification and detailed characterization of 3,4-dihydropyridine-2(1H)-thione derivative with thiophene ring at C5 pyridine-2(1H)-thione (S22) with higher antiproliferative activity (IC50 equal 1.71 ± 0.58 µM) and better selectivity (SI = 21.09) against A375 cells than the starting compound S1 (IC50 = 4.33 ± 1.00 µM, SI = 6.65). Moreover, all three the most active compounds from the antiproliferative study, namely S1, S19 and S22 showed better selectivity against A375 cancer cells than reference CA-4, suggesting their possible lower toxicity and wider therapeutic index. Additionally, mitotic-specific activity of the tested compounds (observed during cell cycle analysis and confocal microscopy imaging), might potentially contribute to decreased side effects compared with classical microtubule targeted agents (MTAs). Further study revealed that all the three tested compounds transiently arrested cells in mitosis, leading to apoptotic cell death. Moreover, we proved that compounds S1, S19 and S22 disrupted mitotic spindle organization and inhibited tubulin polymerization via colchicine binding site in dose-dependent manner. However, direct activity against tubulin was not as high as could be expected from results of cell-based study. Hence, obtained findings may suggest more complexed mechanism of action. The higher anticancer selectivity of S22 compared with the reference microtubule inhibitor (CA-4) may be also connected with multitarget mode of action, including not only microtubules but also kinesins and kinases involved in mitosis and overexpressed in cancer cells [50,51]. The cooperation of kinesins and kinases in microtubule assembly is well documented and pointed as a new opportunity to design potential multitarget antimitotic inhibitors [52]. For example nocodazol, a colchicine binding site agent was found to simultaneously inhibit various cancer-related kinases, including ABL1, c-KIT, BRAF, MEK1 (MAP2K1), MEK2 (MAP2K2), and MET [53]. Frezzato et al., demonstrated that a remarkable ability of nocodazole to discriminate between leukaemic and normal cells was connected with inhibition of Src-kinase Lyn, overexpressed and crucially involved in the neoplastic B-cell survival [54]. We supposed that anticancer selectivity of compound S22 may be also connected with the fact that distribution of β-tubulin isotypes varies among different tumor and normal cells, which modulates their sensitivity to chemotherapeutic drugs [55,56]. However, both hypothesis should be verified in a further study.
4. Materials and Methods
4.1. Experimental Part
Melting points were determined on a Boetius hot stage apparatus. 1H and 13C NMR spectroscopic measurements were performed on a Bruker DPX 400 Avance III HD spectrometer (Billerica, MA, USA, operating at 400.2 and 100.6 MHz, respectively. TMS was used internal standard, δH,C = 0 ppm). For NMR analyses MestReNova (version: 12.0.4 Santiago de Compostela, Spain) program was used. For detailed peak assignments, 2D spectra were acquired using Bruker software (1H,1H DFQCOSY; 13C,1H COSY; 1H,13C HMBC). The 1H,13C HMBC long-range correlations were acquired for JC,H = 10 Hz. The standard abbreviation for multiplicities were used (s = singlet, d =doublet, t = triplet, q =quartet, quint = quintet, m = multiplet, sxt = sextet, spt = septet, etc. Gas chromatography-mass spectrometry (GC-MS) measurements were carried out on a Hewlett-Packard instrument model HP 6890 equipped with a mass detector HP 5973 and on an Agilent 7820A GC system equipped with a mass (Agilent 5977E MSD) detector. HRMS analyses (ESI+) were performed on a Waters LCT premier XE (TOF) using acetonitrile as solvent. Elemental analyses were carried out on Thermo Scientifc™ FLASH 2000 CHNS/O Analyzer (Waltham, MA, USA). n-BuLi (2.5 M in hexane), sec-BuLi (1.4 M in cyclohexane), MeLi (3.1 M in diethoxymethane), BnMgCl (2.0 M in THF) were purchased from Sigma-Aldrich (Merck Group, St. Louis, MO, USA). Anhydrous toluene and THF were purified by distillation over sodium metal under argon prior to use. Products were purified by flash column chromatography on silica gel (63–200 μm, Merck, Darmstadt, Germany) using appropriate solvents.
4.2. Synthesis of Compounds S1-S22
General procedure for the synthesis of 5-aryl-4-benzylpyridine-2(1H)-thiones S1-S22 by the oxygen–sulphur exchange:
To a solution of aryl- and benzyl-substituted pyridin-2(1H)-one (0.308 g, 1.2 mmol) in anhydrous toluene (40 mL) Lawesson reagent (Fluorchem, Derbyshire, United Kingdom; 0.27 g, 0.66 mmol) was added and the mixture was stirred for 4 h at 80 °C (oil bath temperature). After this time the mixture was cooled to room temperature and most of the solvent (2/3 volume) was distilled off in vacuo and the remaining mixture was twice chromatographed on silica gel. The obtained solids were crystallized from appropriate solvents.
4.3. Purification Conditions and Spectroscopic Data
(4RS)-4-Benzyl-5-phenyl-3,4-dihydropyridine-2(1H)-thione [9] (S1): The crude product purified by column chromatography on silica gel using a mixture of ethyl acetate and n-hexane yielded pale yellow solid in 68%. Mp. 143–144 °C (n-hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3) δ 2.58 (dd, J = 13.6, 10.9 Hz, 1 H, 4-CHH), 2.79 (dd, J = 17.3, 7.1 Hz, 1 H, CHH-3), 2.83 (dd, J = 13.6, 3.7 Hz, 1 H, 4-CHH), 2.98–3.05 (m, 1 H, CH-4), 3.21 (d, J = 17.3 Hz, 1 H, CHH-3), 6.56 (d, J = 4.6 Hz, 1 H, =CH-6), 7.21–7.48 (m, 10 H, 2× C6H5), 9.66 (br. s., 1 H, NH). 13C NMR (100.6 MHz CDCl3): δ 35.73 (CH-4), 37.11 (4-CH2), 41.68 (CH2-3), 120.13 (=CH-6), 124.95, 126.53 (C6H5), 126.99 (C-5), 127.71, 128.50, 128.98, 129.55, 136.12, 138.48 (C6H5), 197.96 (C=S). GC-MS (EI, 70 eV): m/z = 279 (36), [M+], 188 (100), 154 (19), 128 (30), 91 (15). HRMS (ESI-TOF): calcd. for C18H18NS 280.1160 [M + H]+; found 280.1166.
(4RS)-4-Benzyl-1-methyl-5-phenyl-3,4-dihydropyridine-2(1H)-thione (S2): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 9: 1, the second column: SiO2, chloroform), gave yellow solid in 96% yield. Mp. 127–129 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3) δ 2.53 (dd, J = 13.5, 10.3 Hz, 1H, 4-CHH), 2.79 (dd, J = 13.5, 3.6 Hz, 1H, 4-CHH), 2.88 (ddq, J = 16.3, 6.5 Hz, 0.9 1H, CHH-3), 2.95 (dddd, 10.3, 6.5, 3.6, 1.5, 1H, CH-4), 3.34 (dd, J = 16.3, 1.5 Hz, 1H, CHH-3), 3.59 (d, J = 0.9 Hz, 3H, NCH3), 6.64 (s, 1H, =CH-6), 7.16–7.26 (m, 3H, C6H5), 7.26–7.34 (m, 3H, C6H5), 7.35–7.43 (m, 2H, C6H5), 7.43–7.48 (m, 2H, C6H5). 13C NMR (101 MHz CDCl3) δ 35.86 (CH-4), 37.29 (4-CH2), 42.27 (N-CH3), 44.19 (CH2-3), 125.07 (C6H5), 126.40 (=CH-6), 126.49, 127.75 (C6H5), 128.18 (=C-5), 128.39 128.99, 129.61, 136.18, 138.59 (C6H5), 195.72 (C=S). GC-MS (EI, 70 eV): m/z = 293 (19), [M+], 204 (6), 203 (15), 202 (100), 201 (7), 186 (4), 170 (4), 168 (5), 128 (14), 127 (5), 115 (7), 91(9). HRMS (ESI-TOF): m/z [M++H] calcd. for C19H20NS 294.1316; found: 294.1323.
(4RS)-4-Benzyl-5-phenyl-1-propyl-3,4-dihydropyridine-2(1H)-thione (S3): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 10: 1, the second column: SiO2, chloroform), gave yellow solid in 89% yield. Mp. 105–107 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3) δ 1.02 (t, J = 7.4 Hz, 3H, N-CH3), 1.72–1.89 (m, 2H, CH2) 2.53 (dd, J = 13.5, 10.3 Hz, 1H, 4-CHH), 2.78 (dd, J = 13.5, 3.6 Hz, 1H, 4-CHH), 2.86 (dd, J = 16.2, 6.6 Hz, 1H, CHH-3), 2.89–3.00 (m, 1H, CH-4), 3.34 (dd, J = 16.2, 1.5, 1H, CHH-3), 3.83 (ddd, J = 13.1, 8.7, 6.5 Hz, 1H, NCHH), 4.38 (ddd, J = 13.0, 8.8, 6.4 Hz, 1H, NCHH), 6.63 (s, 1H, =CH-6), 7.14–7.34 (m, 6H, C6H5), 7.37–7.48 (m, 4H, C6H5). 13C NMR (101 MHz CDCl3): δ 11.36 (CH3), 20.57 (CH2), 35.76 (CH-4), 37.03 (4-CH2), 44.55 (CH2-3), 55.24 (NCH2), 125.06 (C6H5), 125.47(=CH-6), 126.46, 127.69 (C6H5), 128.36 (=C-5), 128.41, 128.95, 129.60, 136.31, 138.64 (C6H5), 195.29 (C=S). GC-MS (EI, 70 eV): m/z = 321 (27), [M+], 230 (100), 188 (44), 154 (15), 128 (19), 91 (16), 65 (5). HRMS (ESI-TOF): m/z calcd for C21H24NS [M+H]+, 322.1629; found: 322.1635.
(4RS)-1,4-Dibenzyl-5-phenyl-3,4-dihydropyridine-2(1H)-thione (S4): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 9: 1, the second column: SiO2, chloroform), gave yellow solid in 85% yield. Mp. 73–76 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.54 (dd, J = 13.6, 9.8 Hz, 1H, 4-CHH), 2.77 (dd, J = 13.6, 3.5 Hz, 1H, 4-CHH), 2.92–2.99 (m, 2H, CHH-3, CH-4), 3.41–3.48 (m, 1H, CHH-3), 5.39 (d, J = 14.8 Hz, 1H, NCHH), 5.53 (d, J = 14.8 Hz, 1H, NCHH), 6.64 (s, 1H, =CH-6), 7.20–7.45 (m, 15H, C6H5). 13C NMR (101 MHz CDCl3): δ 35.91 (CH-4), 37.02 (4-CH2), 44.56 (CH2-3), 55.88 (NCH2), 124.77 (=CH-6), 125.10, 126.49, 127.72, 127.78, 127.96, 128.43 (C6H5), 128.71 (=C-5), 128.85, 128.88, 129.58, 135.70, 136.16, 138.61 (C6H5), 196.49 (C=S). GC-MS (EI, 70 eV): m/z = 369 (11), [M+], 279 (8), 278 (40), 123 (5), 92 (8), 91 (100), 65 (7). HRMS (ESI-TOF): m/z calcd for C25H24NS [M+H]+, 370.1629; found: 370.1633.
(6RS)-6-Benzyl-5-phenyl-3,6-dihydropyridine-2(1H)-thione (S6): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 6: 1, the second column: SiO2, chloroform), gave pale yellow solid in 90% yield. Mp. 137–138 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.72 (dd, J = 13.8, 6.4 Hz 1H, 6-CHH), 2.79 (dt, J = 23.2, 3.1 Hz, 1H, CHH-3), 3.07 (dd, J = 13.8, 3.8 Hz, 1H, 6-CHH), 3.48 (ddd, J = 23.2, 5.1, 3.1 Hz, 1H, CHH-3), 4.86 (ddt, J = 6.4, 3.1, 3.8 Hz, 1H, CH-6), 5.90 (dd, J = 5.1, 3.1 Hz, 1H, =CH-4), 7.00–7.14 (m, 2H, C6H5), 7.24–7.34 (m, 3H, C6H5), 7.34–7.53 (m, 5H, C6H5), 9.26 (br. s, 1H, NH). 13C NMR (101 MHz CDCl3): δ 40.00 (CH2-3), 40.83 (6-CH2), 58.3 (CH-6), 120.60 (=CH-4), 126.00, 127.23, 128.55, 129.03, 130.05 (C6H5), 134.27 (=C-5), 135.22, 137.27 (C6H5), 199.5 (C=S). GC-MS (EI, 70 eV): m/z = 279 (27), [M+], 188 (100), 154 (19), 128 (29), 91 (16). HRMS (ESI-TOF): m/z calcd for C18H18NS [M+H]+, 280.1160; found: 280.1161.
(6RS)-6-Benzyl-1-methyl-5-phenyl-3,6-dihydropyridine-2(1H)-thione (S7): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 6: 1, the second column: SiO2, chloroform), gave dark red oil in 78% yield. 1H NMR (400 MHz, CDCl3): δ 2.22 (ddd, J = 22.6, 3.5, 2.2 Hz, 1H, CHH-3), 2.78 (dd, J = 13.9, 4.1 Hz, 1H, 6-CHH), 3.18 (dd, J = 13.9, 4.5 Hz, 1H, 6-CHH), 3.51 (ddd, J = 22.4, 5.7, 1.9 Hz, 1H, CHH-3), 3.62 (d, J = 0.8 Hz, 3H, N-CH3), 4.92 (dddd, J = 4.5, 4.1, 3.4, 1.9 Hz, 1H, CH-6), 5.9 (dd, J = 5.7, 2.2 Hz, 1H, =CH-4), 6.78–7.06 (m, 2H, C6H5), 7.12–7.31 (m, 3H, C6H5), 7.32–7.48 (m, 5H, C6H5). 13C NMR (101 MHz CDCl3): δ 37.64 (6-CH2), 42.46 (CH2-3), 42.73 (NCH3), 66.24 (CH-6), 121.50 (=CH-4), 125.85, 127.39, 128.22, 128.29, 129.04, 130.13 (C6H5), 134.66 (=C-5), 134.73, 137.06 (C6H5), 197.40 (C=S). GC-MS (EI, 70 eV): m/z = 293 (22), [M+], 202 (100), 170 (6), 128 (11), 91 (8), 65 (63), 42 (8). HRMS (ESI-TOF): m/z calcd for C19H20NS [M+H]+, 294,1316; found: 294,1314.
(4RS)-4-Benzyl-5-(4-fluorophenyl)-3,4-dihydropyridine-2(1H)-thione (S8): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 10: 1, the second column: SiO2, chloroform), gave yellow solid in 90% yield. Mp. 144–147 °C (hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.57 (dd, J = 13.7, 10.7 Hz, 1H, 4-CHH), 2.73–2.84 (m, 2H, 4-CHH, CHH-3), 2.96 (dddd, J = 10.7, 6.2, 4.0, 1.8 Hz, 1H, CH-4), 3.20 (dd, J = 17.2, 1.5 Hz, 1H, CHH-3), 6.50 (d, J = 4.6 Hz, 1H, =CH-6), 7.02–7.12 (m, 2H, C6H5), 7.15–7.46 (m, 7H, C6H5), 9.95 (br. s, 1H, NH). 13C NMR (101 MHz CDCl3): δ 35.99 (CH-4), 37.16 (4-CH2), 41.85 (CH2-3), 115.89 (d,2JC-F = 21.6 Hz, CH-3′, CH-5′), 120.05 (=CH-6), 126.22 (=C-5), 126.59 (C6H5), 126.73 (d,3 JC-F = 8.0 Hz, CH-2′, CH-6′), 128.50 (C6H5), 129.50 (C6H5), 132.40 (d, 4JC-F = 3.4 Hz, C-1′), 138.32 (C6H5), 162.24 (d, 1JC-F = 247.9 Hz, C-4′), 197.71 (C=S). GC-MS (EI, 70 eV): m/z = 279 (37) [M+], 207(14), 206 (100), 173 (9), 172 (21), 146 (30), 91 (17). HRMS (ESI-TOF): m/z calcd for C18H17FNS [M+H]+, 298.1066; found: 298.1064.
(4RS)-4-Benzyl-5-(3,4-difluorophenyl)-3,4-dihydropyridine-2(1H)-thione (S9): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 8: 1, the second column: SiO2, chloroform), gave yellow solid in 95% yield. Mp. 142–149 °C (hexan: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ = 2.58 (dd, J = 13.6, 10.5 Hz, 1H, 4-CHH), 2.73–2.84 (m, 2H, CHH-3, 4-CHH), 2.92 (dddd, J = 10.5, 6.4, 4.2, 1.5, 1H, CH-4), 3.22 (dd, J = 16.8, 1.5 Hz, 1H, CHH-3), 6.51 (d, J = 4.6 Hz, 1H, =CH-6), 7.00–7.46 (m, 8H, ArH), 9.76 (s, 1H, NH). 13C NMR (101 MHz CDCl3): δ 35.93 (CH-4), 37.29 (4-CH2), 41.99 (CH2-3), 114.02 (d, 2J = 18.2 Hz, CH-2′), 117.75 (d, 2J = 17.4 Hz, CH-5′), 120.85 (d, 5J = 1.5 Hz, =CH-6), 121.06 (dd, J = 6.1, 3.4 Hz, CH-6′), 125.11 (=C-5), 126.73, 128.58, 129.51 (C6H5), 133.62 (dd, J = 5.9, 4.3 Hz, C-1′), 138.11 (C6H5), 148.41 (dd, J = 250.7, 12.6 Hz, C-3′), 150.68 (dd, J = 248.9, 12.5 Hz, C-4′), 198.1 (C=S). GC/MS (EI, 70 eV): m/z = 316 (6) [M+], 315 (27), [M+], 225 (14), 224 (100), 191 (8), 190 (20), 164 (25), 163 (6), 91 (21), 65 (7). HRMS (ESI-TOF): m/z calcd for C18H16F2NS [M+H]+, 316.0972; found: 316.0981.
(4RS)-4-Benzyl-5-(4-chlorophenyl)-3,4-dihydropyridine-2(1H)-thione (S10): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 3: 1, the second column: SiO2, chloroform), gave yellow solid in 78% yield. Mp. 161–163 °C (hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.57 (dd, J = 13.6, 10.7 Hz, 1H, 4-CHH), 2.73–2.84 (m, 2H, 4-CHH, CHH-3), 2.97 (dddd, J = 10.7, 6.1, 4.0, 1.8 Hz, 1H, CH-4), 3.21 (ddd, J = 17.1, 1.8, 0.9 Hz, 1H, CHH-3), 6.55 (d, J = 4.6 Hz, 1H, =CH-6), 7.13–7.44 (m, 9H, C6H5, C6H4), 9.53 (br. s, 1H, NH). 13C NMR (101 MHz CDCl3): δ 35.78 (CH-4), 37.21 (4-CH2), 41.82 (CH2-3), 120.45 (=CH-6), 125.83 (=C-5), 126.22, 126.64, 128.54, 129.11, 129.51, 133.38, 134.73, 138.25 (C6H5, C6H4), 198.12 (C=S). GC-MS (EI, 70 eV): m/z = 313 (31) [M+], 225 (5), 224 (37), 222 (100), 221 (12), 188 (16), 162 (17), 128 (9), 115 (10), 91 (23), 65 (8). HRMS (ESI-TOF): m/z calcd for C18H17ClNS [M+H]+, 314.0770; found: 314.0782.
(4RS)-4-Benzyl-5-(3-methoxyphenyl)-3,4-dihydropyridine-2(1H)-thione (S11): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 6: 1, the second column: SiO2, chloroform), gave yellow solid in 96% yield. Mp. 137–139 °C (n-hexane: ethyl acetate). 1H NMR NMR (400 MHz, CDCl3): δ 2.57 (dd, J = 13.6, 11.0 Hz, 1H, 4-CHH), 2.78 (dd, J = 17.0, 6.7 Hz, 1H, CHH-3), 2.83 (dd, J = 13.6, 3.7 Hz, 1H, 4-CHH), 2.99 (dddd J = 11.0, 6.7, 3.7, 1.8 Hz 1H, CH-4), 3.20 (ddd, J = 17.0, 1.8, 1.0 Hz, 1H, CHH-3), 3.85 (s, 3H, OCH3), 6.56 (d, J = 4.6 Hz, 1H, =CH-6), 6.85 (ddd, J = 8.2, 2.5, 0.8 Hz, 1H, C6H4), 6.97 (t, J = 2.1 Hz, 1H, C6H4), 7.05 (ddd, J = 7.8, 1.8, 0.9 Hz, 1H, C6H4), 7.16–7.39 (m, 6H, C6H5, C6H4), 9.69 (br. d, J = 4.6 Hz, 1H); 13C NMR (100 MHz, CDCl3): δ 35.83 (CH-4), 37.17 (4-CH2), 41.70 (CH2-3), 55.33 (OCH3), 111.10, 112.73, 117.49 (C6H4), 120.39 (=CH-6),126.56 (C6H5), 126.84 (=C-5), 128.52, 129.57, 129.98, 137.67, 138.50, 160.06 (C6H5, C6H4), 198.07 (C=S). GC-MS (EI, 70 eV): m/z = 309 (30) [M+], 219 (15), 218 (100), 203 (6), 185 (7), 158 (14), 115 (11), 91 (17). HRMS (ESI-TOF): m/z calcd for C19H20NOS [M+H]+, 310.1266; found: 310.1252.
(4RS)-4-Benzyl-5-(3,4-dimethoxyphenyl)-3,4-dihydropyridine-2(1H)-thione (S12): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 3: 1, the second column: SiO2, chloroform), gave yellow solid in 88% yield. Mp. 135–138 °C. 1H NMR (400 MHz, CDCl3): δ 2.58 (dd, J = 13.6, 10.8 Hz, 1H, 4-CHH), 2.73–2.85 (m, 2H, 4-CHH, CHH-3), 2.97 (dddd, J = 10.8, 6.4, 3.8, 1.8 Hz, 1H, CH-4), 3.21 (ddd, J = 17.1, 1.8, 1.0 Hz, 1H, CHH-3), 3.92 (s, 3H, OCH3), 3.92 (s, 3H, OCH3), 6.50 (d, J = 4.5 Hz, 1H, CH-6), 6.87–6.93 (m, 2H, C6H3), 7.03 (dd, J = 8.3, 2.2 Hz, 1H, C6H3), 7.19–7.34 (m, 5H, C6H5), 9.76 (br. d, J = 4.5 Hz, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 36.08 (CH-4), 37.24 (4-CH2), 41.78 (CH2-3), 55.99 (two OCH3), 108.28, 111.44, 117.79 (C6H3), 119.11 (=CH-6), 126.57 (C6H5), 127.18 (=C-5), 128.54, 129.01, 129.53, 138.65, 148.95, 149.26 (C6H3, C6H5), 197.42 (C=S). GC-MS (EI, 70 eV): m/z = 339 (64) [M+], 249 (16), 248 (100), 233 (7), 232 (18), 215 (77), 200 (7), 188 (13), 172 (6), 115 (8), 91 (31), 77 (4), 65 (8). HRMS (ESI-TOF): m/z calcd for C20H21NNaO2S [M+Na]+ 362.1191; found: 362.1197.
(4RS)-4-Benzyl-5-(4-fluorophenyl)-1-methyl-3,4-dihydropyridine-2(1H)-thione (S13): The crude product purified twice by column chromatography the first column (SiO2, n-hexane: ethyl acetate, 8: 1, then chloroform), gave yellow solid in 89% yield. Mp. 143–147 °C. 1H NMR (400 MHz, CDCl3): δ 2.53(dd, J = 13.7, 9.5 Hz, 1H, 4-CHH), 2.74 (dd, J = 13.7, 3.6 Hz, 1H, 4-CHH), 2.84–2.93 (m, 2H, CH-4, CHH-3), 3.29–3.39 (m, 1H, CHH-3), 3.59 (s, 3H, N-CH3), 6.57 (s, 1H, CH-6), 7.04–7.12 (m, 2H, C6H5), 7.16–7.24 (m, 3H, C6H5), 7.25– 7.30 (m, 2H), 7.35–7.44 (m, 2H, C6H5). 13C NMR (100 MHz, CDCl3): δ 36.13 (CH-4), 37.31 (4-CH2), 42.25 (N-CH3), 44.28 (CH2-3), 115.9 (d, 2JC-F = 21,8 Hz, CH-3′, CH-5′), 126.28 (d, 6J = 1.9 Hz, =CH-6), 126.54 (C6H5), 126.83 (d, 3JC-F = 8.0 Hz, CH-2′, CH-6′), 127.36 (=C-5), 128.39 (2C), 129.55 (2C, C6H5), 132.40 (d, 4JC-F = 3.3 Hz, C-1′), 138.42 (C6H5), 162.28 (d, 1JC-F = 248.0 Hz, C-4′), 195.56 (C=S). GC/MS GC-MS (EI, 70 eV): m/z = 311 (20) [M+], 220 (100), 146 (15), 91 (44). HRMS (ESI-TOF): m/z calcd for C19H19FNS [M+H]+, 312.1222; found: 312.1216.
(4RS)-4-Benzyl-5-(3,4-difluorophenyl)-1-methyl-3,4-dihydropyridine-2(1H)-thione (S14): The crude product purified twice by column chromatography the first (SiO2, n-hexane: ethyl acetate, 10: 1, then chloroform), gave yellow solid in 96% yield. Mp. 128–132 °C. 1H NMR (400 MHz, CDCl3): δ 2.55 (dd, J = 13.4, 9.7 Hz, 1H, 4-CHH), 2.73 (dd, J = 13.4, 3.9 Hz, 1H, 4-CHH), 2.81–2.87 (m, 1H, CH-4), 2.90 (dd, J = 15.1, 6.6 Hz, 1H, CHH-3), 3.36 (d, J = 15.1 Hz, 1H, CHH-3), 3.59 (s, 3H, NCH3), 6.59 (s, 1H, =CH-6), 7.07–7.33 (m, 8H, C6H5). 13C NMR (100 MHz, CDCl3): δ 36.03 (CH-4), 37.43 (4-CH2), 42.29 (N-CH3), 44.40 (CH2-3), 114.09 (d, 2J = 18.2 Hz, CH-2′), 117.74 (d, 2J = 17.5 Hz, CH-5′), 121.13 (dd, J = 6.2, 3.3 Hz, CH-6′), 126.20 (=C-5), 126.66 (C6H5), 127.02 (=CH-6), 128.45, 129.53 (C6H5), 133.62 (dd, J = 6.2, 3.8 Hz, C-1′), 138.21 (C6H5), 149.66 (dd, J = 249.9, 13.8 Hz, C-3′), 150.53 (dd, J = 248.6, 13.2 Hz, C-2′), 195.97 (C=S). GC-MS (EI, 70 eV): m/z = 329 (23) [M+], 238 (100), 164 (10). HRMS (ESI-TOF): m/z calcd. for C19H17F2NNaS [M+Na]+ 352.0947; found: 352.0946.
(4RS)-4-Benzyl-5-(4-chlorophenyl)-1-methyl-3,4-dihydropyridine-2(1H)-thione (S15): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 10: 1, the second column SiO2, chloroform), gave yellow solid in 84% yield. Mp. 154–156 °C. 1H NMR (400 MHz, CDCl3): δ 2.53 (dd, J = 13.5, 9.8 Hz, 1H, 4-CHH), 2.67–2.79 (m, 1H, CHH-3), 2.84–3.00 (m, 2H, CH-4, 4-CHH), 3.22–3.43 (m, 1H, CHH-3), 3.58 (s, 3H, NCH3), 6.62 (s, 1H, =CH-6), 7.17–7.23 (m, 3H, C6H5), 7.25–7.31 (m, 2H, C6H5), 7.35 (s, 4H, C6H4). 13C NMR (100 MHz, CDCl3): δ 35.84 (CH-4), 37.35 (4-CH2), 42.27 (NCH3), 44.27 (CH2-3), 126.31, 126.57 (ArH), 126.66 (=CH-6), 126.95 (=C-5), 128.40, 129.10 (ArH), 129.54 (ArH), 133.39, 134.75, 138.34 (Ar), 195.82 (C=S). GC-MS (EI, 70 eV): m/z = 327 (23) [M+], 239 (5), 238 (38), 236 (100), 235 (7), 201 (6), 128 (6), 115 (6), 91 (10), 65 (5). HRMS (ESI-TOF): m/z calcd. for C19H18ClNNaS [M+Na]+ 350.0746; found: 350.0755.
(4-RS)-4-Benzyl-5-(3-methoxyphenyl)-1-methyl-3,4-dihydropyridine-2(1H)-thione (S16): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 10: 1, the second column: SiO2, chloroform), gave yellow solid in 98% yield. Mp. 106–109 °C (hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.53 (dd, J = 13.5, 10.2 Hz, 1H, 4-CHH), 2.79–2.94 (m, 3H, CH-4, 4-CHH, CHH-3), 3.24–3.40 (m, 1H, CHH-3), 3.58 (s, 3H, NCH3), 3.85 (s, 3H, OCH3), 6.64 (s, 1H, =CH-6), 6.86 (ddd, J = 8.2, 2.5, 0.8 Hz, 1H, ArH), 6,97 (t, J = 2.1 Hz, 1H, ArH), 7.05 (ddd, J = 7.8, 1.8, 0.9 Hz, 1H, ArH), 7.16–7.37 (m, 6H, ArH). 13C NMR (100 MHz, CDCl3): δ = 35.94 (CH-4), 37.33 (4-CH2), 42.27 (NCH3), 44.18 (CH2-3), 55.33 (O-CH3), 111.25, 112.68, 117.57 (ArH), 126.48 (=C-6), 126.61 (ArH), 127.98 (=C-5), 128.37, 129.61, 129.96 (ArH), 137.68, 138.57, 160.06 (Ar), 195.81 (C=S). GC-MS (EI, 70 eV): m/z = 323 (21) [M+], 233 (15), 232 (100), 217 (5), 189 (7), 158 (8), 115 (7), 91 (9). HRMS (ESI-TOF): m/z calcd. for C20H21NnaOS [M+Na]+ 346.1242; found: 346.1236.
(4RS)-4-Benzyl-5-(3,4-dimethoxyphenyl)-1-methyl-3,4-dihydropyridine-2(1H)-thione (S17): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 3: 1, the second column SiO2, chloroform), gave yellow solid in 98% yield. Mp. 115–119 °C (n-hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.54 (dd, J = 13.6, 9.9 Hz, 1H, 4-CHH), 2.77 (dd, J = 13.6, 3.4 Hz, 1H, 4-CHH), 2.82–3.03 (m, 2H, CHH-3, CH-4), 3.25–3.46 (m, 1H, CHH-3), 3.61 (s, 3H, NCH3), 3.92 (d, J = 4.6 Hz, 6H, two OCH3), 6.56 (s, 1H, =CH-6), 6.85–6.95 (m, 2H, C6H3), 7.03 (dd, J = 8.3, 2.2 Hz, 1H, C6H3), 7.12–7.48 (m, 5H, C6H5). 13C NMR (100 MHz, CDCl3): δ 36.22 (CH-4), 37.38 (4-CH2), 42.29 (N-CH3), 44.23 (CH2-3), 56.00 (two OCH3), 108.32, 108.36, 111.42, 117.91 (C6H3), 125.33 (=CH-6), 126.50 (C6H5), 128.38 (C6H3), 128.43 (C6H5), 128.98 (=C-5), 129.57 (C6H5), 138.74 (C6H5), 148.98, 149.24 (C6H3), 195.17 (C=S). GC-MS (EI, 70 eV): m/z = 353 (38) [M+], 264 (6), 263 (17), 262 (100), 247 (5), 246 (18), 231 (6), 206 (4), 188 (9), 115 (4), 91 (10). HRMS (ESI-TOF): m/z calcd. for C21H23NNaO2S [M+Na]+ 376.1347; found: 376.1340.
(4RS)-4-(Naphthalen-1-ylmethyl)-5-phenyl-3,4-dihydropyridine-2(1H)-thione (S18): The crude product purified twice by column chromatography (the first column SiO2, n-hexane: ethyl acetate, 6: 1, the second column SiO2, chloroform), gave yellow solid in 68% yield. Mp. 160–162 °C (n-hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.78 (dd, J = 16.9, 7.1 Hz, 1H, CHH-3), 2.89 (dd, J = 12.1, 3.0 Hz, 1H, 4-CHH), 3.22 (d, J = 16.9 Hz, 1H, CHH-3), 3.26–3.32 (m, 2H, 4-CHH, CH-4), 6.55 (d, J = 4.5, 1H, =CH-6), 7.29–7.55 (m, 9H, ArH), 6.67–7.77 (m, 1H, ArH), 7.80–7.98 (m, 2H, ArH), 9.54 (s, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 34.18 (CH2–4), 34.48 (4-CH), 42.21 (CH2-3), 120.26 (=CH-6), 123.61, 125.51, 125.79, 125.92, 127.41, 127.91 (C10H7, C6H5), 128.02 (=C-5), 128.74, 128.95, 128.97, 131.69, 134.01, 134.19, 136.41 (C10H7, C6H5, two signal overlapped), 198.14 (C=S). GC-MS (EI, 70 eV); m/z = 330 (5), 329 (20), [M+], 189 (15), 188 (100), 187 (34), 154 (15), 142 (7), 141 (28), 128 (25), 127 (10), 115 (18). HRMS (ESI-TOF): m/z calcd. for C22H20NS [M+H]+ 330.1316; found: 330.1318.
(4RS)-4-(4-Methoxybenzyl)-5-phenyl-3,4-dihydropyridine-2(1H)-thione (S19): The crude product purified twice by column chromatography (the first column SiO2, n-hexane: ethyl acetate, 10: 1, the second column SiO2 chloroform), gave yellow solid in 86% yield. Mp. 149–151 °C (n-hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.52 (dd, J = 13.9, 11.0 Hz, 1H, 4-CHH), 2.70–2.89 (m, 2H, 4-CHH, CHH-3), 2.96 (dddd, J = 11.0, 6.9, 3.7, 1.9 Hz, 1H, 4-CH), 3.21 (ddd, J = 17.0,1.9, 1.0 Hz, 1H, CHH-3), 3.79 (s, 3H, OCH3), 6.55 (d, J = 4.6 Hz, 1H, =CH-6), 6.73–6.97 (m, 2H, C6H4), 7.04–7.21 (m, 2H, C6H4), 7.27–7.35 (m, 1H, C6H5), 7.36–7.50 (m, 4H, C6H5), 9.73 (s, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 35.90 (CH-4), 36.26 (4-CH2), 41.69 (CH2-3), 55.28 (OCH3), 113.91 (ArH), 120.10 (=CH-6), 124.97 (ArH), 127.09 (=C-5), 127.69, 128.98, 130.54, 130.55, 136.23, 158.32 (ArH, Ar), 198.01 (C=S). GC-MS (EI, 70 eV); m/z = 309 (13), [M+], 189 (7), 188 (43), 187 (23), 154 (12), 128 (17), 121 (100), 115 (5), 91 (5), 77 (8). HRMS (ESI-TOF): m/z calcd. for C19H20NOS [M+H]+ 310.1266; found: 310.1267.
4-([1,1′-Biphenyl]-4-ylmethyl)-5-phenyl-3,4-dihydropyridine-2(1H)-thione (S20): Yield 98%. The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 6: 1, the second column: SiO2, chloroform), gave yellow solid in 98% yield. Mp. 173–174 °C. 1H NMR (400 MHz, DMSO-d6): δ 2.43 (dd, J = 13.6, 11.2 Hz, 1H, 4-CHH), 2.67–2.82 (m, 2H, 4-CHH, 3-CHH), 2.91 (ddd, J = 17.0, 1.9, 1.0 Hz, 1H, CHH-3), 3.04–3.12 (m, 1H, CH-4), 6.75 (d, J = 4.6 Hz, 1H, =CH-6), 7.21–7.55 (m, 8H, ArH), 7.54–7.79 (m, 6H, ArH), 11.67 (d, J = 4.6 Hz, 1H, NH). 13C NMR (100 MHz, DMSO-d6): δ 34.06 (CH-4), 36.09 (4-CH2) 41.90 (CH2-3), 120.97 (=CH-6), 124.51 (Ar), 124.83 (=C-5), 126.42, 126.43, 126.95, 127.16, 128.75, 128.80, 129.89, 135.91, 137.77, 138.10, 139.84 (Ar), 196.37 (C=S). GC-MS (EI, 70 eV); m/z = 355 (15), [M+], 281 (8), 208, 207 (24), 189 (15), 188 (100), 187 (25), 168 (7), 154 (17), 128 (25), 127 (11). Anal. calcd for C24H21NS: C 81.09, H 5.95, N 3.94, S 9.02. Found: C 80.98, H 5.90, N 4.04.
(4RS)-4-Benzyl-5-(naphthalen-1-yl)-3,4-dihydropyridine-2(1H)-thione (S21): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate, 6: 1, the second column SiO2, chloroform), gave yellow solid in 95% yield. Mp. 173–175 °C (n-hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.55 (dd, J = 13.5, 11.4 Hz, 1H, 4-CHH), 2.76 (dd, J = 13.5, 3.8 Hz 1H, 4-CHH), 2.95 (dddd, J = 11.4, 6.6, 3.8, 3.4 Hz, 1H, CH-4), 3.08 (dd, J = 17.0, 6.6 Hz, 1H, CHH-3) 3.20 (dd, J = 17.0, 3.4 Hz, 1H, CHH-3), 6.27 (d, J = 4.4 Hz, 1H, =CH-6), 7.02–7.07 (m, 2H, C6H5), 7.11–7.23 (m, 3H, C6H5), 7.34 (dd, J = 7.0, 1.3 Hz, 1H, C10H7), 7.48 (dd, J = 8.3, 7.0 Hz, 1H, C10H7), 7.51–7.56 (m, 2H, C10H7), 7.86 (dt, J = 8.4, 1.0 Hz, 1H, C10H7), 7.89–7.95 (m, 2H, C10H7), 9.43 (s, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 36.94 (4-CH2), 38.89 (CH-4), 41.91 (CH2-3), 123.00 (=CH-6), 125.08, 125.35, 126.10, 126,40, 126.48, 126.75 (C10H7), 127.56 (=C-5), 128.39, 128.49 (C6H5), 128.69 (C10H7), 129.43 (C6H5), 131.80, 133.90, 135.73 (C10H7), 138.27(C6H5), 198.39 (C=S). GC-MS (EI, 70 eV): m/z = 330 (7), 329 (28), [M+], 240 (6), 239 (19), 238 (100), 221 (9), 205 (22), 204 (35), 179 (10), 178 (21), 152 (10), 91 (18). HRMS (ESI-TOF): m/z calcd. for C22H19NNaS [M+Na]+ 352.1136; found: 352.1132.
(4RS)-4-Benzyl-5-(thiophen-2-yl)-3,4-dihydropyridine-2(1H)-thione (S22): The crude product purified twice by column chromatography (the first column: SiO2, n-hexane: ethyl acetate), 10:1, the second column SiO2, chloroform), gave yellow solid in 95% yield. Mp. 130–132 °C (n-hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3) δ 2.60 (dd, J = 14.9, 12.0 Hz, 1H, 4-CHH), 2.78 (dd, J = 17.1, 6.9 Hz, 1H, CHH-3), 2.85–2.97 (m, 2H, 4-CHH, CH-4), 3.19 (d, J = 17.1 Hz, 1H, CHH-3), 6.58 (d, J = 4.5 Hz, 1H, =CH-6), 7.03 (dd, J = 5.1, 3.6 Hz, 1H, C4H3S), 7.08 (dd, J = 3.7, 1.2 Hz, 1H, C4H3S), 7.20–7.28 (m, 4H, C4H3S, C6H5), 7.29–7.35 (m, 2H, C6H5), 9.78 (s, 1H, NH). 13C NMR (101 MHz, CDCl3) δ 37.28 (CH-4), 37.32 (4-CH2), 41.53 (CH2-3), 118.82 (=CH-6), 122.05 (=C-5), 122.96, 124.20 (C4H3S), 126.62 (C6H5), 127.91 (C4H3S), 128.55 (2C, C6H5), 129.58 (2C, (C6H5), 138.43 ((C6H5), 140.61 (C4H3S), 197.42 (C=S). GC-MS (EI, 70 eV): m/z = 285 (48), [M+], 195 (13), 194 (100), 193 (14), 161 (23), 134 (30), 91 (21). HRMS (ESI-TOF): m/z calcd. for C16H16NS2 [M+H]+ 286.0724; found: 286.0724.
4.4. Cell Culture
Human breast adenocarcinoma cells (MCF7 ECACC 86012803), malignant melanoma cells (A375 ECACC 88113005), colon adenocarcinoma (HT-29 ECACC 91072201), ovary adenocarcinoma (SK-OV-3 ECACC 91091004) and prostate adenocarcinoma (PC-3 ECACC 90112714) were purchased from European Collection of Authenticated Cell Cultures (ECACC). Human Epidermal Melanocytes HEM (neonatal, 104K-05n) were purchased from Cells Applications (San Diego, CA, USA). All cells were cultured in a humidified incubator (5% CO2, 37 °C) in recommended culture medium supplemented with heat-inactivated fetal bovine serum (FBS, EURx, Gdansk, Poland), L-glutamine (2 mM, Sigma-Aldrich Merck Group, St. Louis, MO, USA) and penicillin-streptomycin (Sigma-Aldrich Merck Group, St. Louis, MO, USA). Cell lines were routinely tested for the presence of mycoplasma.
4.5. Antiproliferative Activity
The antiproliferative activity of S1 and S1 analogues (S2-S22), with combretastatin (CA-4, Sigma-Aldrich Merck Group, St. Louis, MO, USA) as reference, were evaluated using the Cell Proliferation Reagent WST-1 assay (Sigma-Aldrich Merck Group, St. Louis, MO, USA). It should be noted that this kit is commonly used for proliferation measurement, but it does not directly measure DNA replication, and may be influenced by cell metabolic activity. WST-1 assay is based on the reduction of the tetrazolium salt to formazan which amount is directly correlated to the number of metabolically active cells. In the present study, MCF7, A375, HT-29, SK-OV-3 and PC-3 cells were seeded in 96-well plate (at plating densities depending on the doubling time of individual cell lines) and then cultured in appropriate medium in standard conditions. After 24 h the culture medium was removed and replaced with fresh medium containing compounds at final concentrations equal: 1, 10, 50, 100 µM for 48 h. All the tested compounds were dissolved in DMSO purchased from Sigma-Aldrich Merck Group, St. Louis, MO, USA (final concentrations of DMSO did not exceed 0.2%; initially impact of 0.2% DMSO on cell viability was negatively verified). The cells without tested compounds (but in medium with DMSO) were used as control and tested compounds in medium without cells as blank. After 48 h, WST-1 reagent was added, incubated with the cells for 30 min and absorbance was measured at 450 nm (with 620 nm background correction), using a spectrophotometric microplate reader (Infinite 200 Pro, Tecan, Männedorf, Switzerland). The cell viability was calculated using the following formula: [(Atest − Ablank)/(Acontrol − Ablank)] × 100%. The readings were acquired from at least three independent experiments (each conducted in triplicate). The IC50 values (the inhibitory concentration causing 50% growth inhibition) were evaluated using an online calculator (AAT Bioquest, Inc., Quest Graph™ IC50 Calculator (v.1). Retrieved from: https://www.aatbio.com/tools/ic50-calculator-v1 (accessed on: 10 September 2020).
4.6. Selectivity-Index
The malignant melanoma (A375) and non-tumor melanocytes (HEM) cells were used to estimate the anticancer selectivity of the three the most active compounds from the earlier study (S1, S19 and S22; CA-4 was used as reference). The WST-assay and IC50 calculation were performed, as described above. Then the selectivity index (SI) was calculated according to the following formula: SI = IC50 normal cells/IC50 cancer cells.
4.7. Apoptosis Detection
A375 and HEM cells were seeded in 24-well plates (1 × 104 and 2 × 104 cells/well, respectively). After 24 h, the culture medium was removed and the cells were treated with S1, S19, S22 and CA-4 (as reference) at final concentrations of 10 µM and incubated for another 48 h. Untreated cells growing in medium with DMSO (0.2%) was used as control. After treatment the cells were washed with PBS (both the cell medium and PBS were collected) and harvested by trypsinization. After centrifugation, the supernatant was discarded and cells pellet was resuspended in appropriate medium. Next, equal volume of the Muse Annexin V & Dead Cell Reagent (Luminex Corporation, Austin, TX, USA) was added, mixed thoroughly by pipetting up and down and stained for 20 min (at RT in the dark). Samples were analyzed by Muse Cell Analyzer (Luminex Corporation, Austin, TX, USA). The assay is based on the detection of phosphatidylserine (PS) on the surface of apoptotic cells by binding with Annexin V-PE. A dead cell marker (7-AAD) is also used as an indicator of cell membrane structural integrity. Simultaneous staining with these two dyes allow to distinguish four populations of cells: viable (Annexin V-PE− and 7-AAD−), early apoptotic (Annexin V-PE+ and 7-AAD−), late apoptotic/already dead cells (Annexin V-PE+ and 7-AAD+), mostly nuclear debris (Annexin V-PE− and 7-AAD+). According to the manufacturer’s protocol (on the base of cell size index) debris were excluded from further analysis. The readings were acquired from at least three independent experiments. Additionally, optical microscopy imaging of A375 and HEM cells was performed just before apoptosis analysis of harvested cells using Smart Fluorescent Cell Analyzer Microscope JuLi (Seoul, Korea).
4.8. Cell Cycle Analysis
For cell cycle analysis, A375 cells were seeded in 6-well plates (1 × 105 cells/well) and cultured for 24 h. Then the medium was removed and replaced with fresh medium containing S1, S19, S22 and CA-4 at final concentrations of 10 µM and incubated for additional 6 and 24 h. Afterwards, cells were washed with PBS and harvested by trypsinization. Then, cell pellet was washed with PBS and fixed with 70% cold ethanol at −20 °C for at least 3 h. Before the analysis ethanol-fixed cells were centrifuged, washed with PBS and stained with Muse Cell Cycle Reagent (Luminex Corporation, Austin, TX, USA), containing propidium ioide (PI) and RNAse, for 30 min at RT in the dark. The assay utilizes PI-based staining of DNA content to discriminate and measure the percentage of cells in each cell cycle phase (G0/G1, S, and G2/M) using a Muse Cell Analyzer (Luminex Corporation, Austin, TX, USA). The readings were acquired from three independent experiments.
4.9. Confocal Microscopy Imaging
To visualize the microtubule and DNA, A375 cells were seeded on cover slips at density of 1 × 105 cells/well and cultured as above for 24 h. Afterwards, 10 µM of compounds: S1, S2 and S22 were added, and incubated for additional 6 h. Next, the cells were fixed in 4% buffered formalin for 5 min at 37 °C, washed twice with PBS and permeabilized with 0.01% Triton X-100 (Sigma-Aldrich Merck Group, St. Louis, MO, USA) in PBS for 10 min at RT. Nonspecific antibody binding was blocked (0.01% Triton X-100, 2% BSA, 1.5% FBS in PBS) for 10 min (RT) and subsequently incubated with mouse anti-α-tubulin monoclonal antibody (Sigma-Aldrich Merck Group, St. Louis, MO, USA), diluted 1:2000 for overnight (4 °C). The cover slips were then washed tree times with 0.05% Triton X-100 in PBS and the cells were incubated with secondary antibodies anti-mouse IgG-FITC (Sigma-Aldrich Merck Group, St. Louis, MO, USA) diluted 1:64 for 1 h at RT, washed and counterstained with DAPI (Sigma-Aldrich Merck Group, St. Louis, MO, USA). Finally, cells were washed with PBS and mounted with mounting medium (Dako-Agilent, CA, USA) on glass slides. The slides were examined with a FV1000 confocal microscope (Olympus, Hamburg, Germany) in two separated channels: for DAPI (405 nm laser), FITC (488 nm laser). To calculate the proportion of different mitotic spindle phenotypes cells in mitosis from ten random fields (×20 objective magnification) of the slide were counted (from two independent experiments).
4.10. Cell Free Tubulin Polymerization Assay
Tubulin polymerization was monitored using a >99% pure tubulin and fluorescence-based tubulin polymerization kit (Cytoskeleton, Denver, CO, USA), where tubulin polymerization is followed by fluorescence enhancement due to the incorporation of a fluorescence reporter into microtubules as polymerization proceeds. Compounds S1, S19 and S22 (at final concentrations: 100, 50, 10 and 1 µM) were evaluated for their effect on tubulin polymerization (in cell free conditions). Paclitaxel (Cytoskeleton, Denver, CO, USA) and CA-4 (10 µM) were used as references and DMSO (0.2%) as vehicle control. According to the manufacturer’s protocol after incubation of tested compounds at 37 °C for 1 min, the icy tubulin reaction mixture (2 mg/mL tubulin, 1.0 mM GTP, 15% glycerol in buffer containing 80 mM PIPES, 2.0 mM MgCl2, 0.5 mM EGTA, pH 6.9 and 10 µM fluorescent reporter) was added. The samples were mixed and tubulin assembly was monitored (excitation: 360 nm, emission: 450 nm) at 1 min intervals for 1 h at 37 °C using a spectrophotometric microplate reader (Infinite 200 Pro, Tecan, Männedorf, Switzerland). The inhibition of tubulin polymerization was expressed as IC50 values and quantified according to Cytoskeleton, Inc. instruction. Firstly, maximal slope (V max) of the growth phase expressed as RFU/min was calculated using a software installed on the aforementioned plate reader. Next, V max of each polymerization curve was normalized to V max of appropriate control’s curve and IC50 values were evaluated using an online calculator (AAT Bioquest, Inc. Quest Graph™ IC50 Calculator. Retrieved from https://www.aatbio.com/tools/ic50-calculator (accessed on: 16 February 2021).
4.11. Cell-Based Tubulin Polymerization Assay
Western blot based tubulin polymerization assay was modified by previously described method [57,58]. A375 cells were seeded in 6-well plates (5 × 104 cells/well), grown to subconfluency and next additional 6 h with 10 µM of compounds S1, S19, S22, CA-4 and PTX (as references), and DMSO (0.2%) as control. After treatment, cells were washed twice with warm PBS, lysed shaking for 5 min with hypotonic buffer (20 mM Tris–HCl pH 6.8, 1 mM MgCl2, 2 mM, EGTA, 0.5% NP-40; Sigma-Aldrich Merck Group, St. Louis, MO, USA) containing 1X protease inhibitor cocktail (Thermo Fisher Scientific, Waltham, MA, USA), then scraped and transferred to tubes. The samples were centrifuged at 13,200 rpm for 10 min at RT, and supernatants containing soluble (cytosolic) tubulin were separated from the pellets containing polymerized (cytoskeletal) tubulin. The pellets were resuspended in equal volumes of hypotonic buffer. Total protein concentration was then determined in each fraction by BCA method (Thermo Fisher Scientific, Waltham, MA, USA). The equal amount of protein (6 µg per well) from supernatant and pellet fractions were resuspended in 2× Laemmli buffer (Bio-Rad, Hercules, CA, USA), heated for 5 min at 95 °C and analyzed by SDS-PAGE using 4–20% mini-PROTEAN electrophoresis system (Bio-Rad, Hercules, CA, USA), then transferred to PVD membrane (Bio-Rad, Hercules, CA, USA). Immunoblotting was performed using a primary mouse anti-α-tubulin monoclonal antibody (Sigma-Aldrich Merck Group, USA) diluted 1:1000 for overnight (4 °C) and secondary mouse IgGκ BP conjugated to horseradish peroxidase (HRP) (Santa Cruz Biotechnology, Dallas, TX, USA) diluted 1:2000. The obtained products were detected using the Western Bright Sirius Chemiluminescent Detection Kit (Advansta, San Jose, CA, USA) and bands were subsequently visualized in UVP camera (UVP BioImaging system EpiChemi 3, Houston TX, USA). Equal loading in the lanes was evaluated by stripping the blots according to manufacturer’s protocol (Restore Western Blot Stripping Buffer, Thermo Fisher Scientific, Waltham, MA, USA) and reprobing them with mouse β-actin monoclonal antibody 1:1000 (Santa Cruz Biotechnology, Dallas, TX, USA) and then secondary HRP-conjugated antibody (as above). Percentage of α-tubulin in the pellet fractions was calculated as a densitometric value of the pellet band divided by the total densitometric value of the pellet and supernatant bands using ImageJ Software 1.53c (Maryland, VA, USA). Each densitometric value of tubulin was before normalized to the densitometric value of β-actin The readings were acquired from four independent experiments.
4.12. Colchicine Binding-Site Assay
Colchicine binding-site assay was performed as previously described [44] by using >99% pure tubulin protein (Cytoskeleton, Denver, CO, USA) reconstituted in buffer containing 80 mM PIPES pH 6.9, 2 mM MgCl2, 0.5 mM EGTA supplemented with 1 mM GTP. The compounds S1, S19 and S22 at 50 and 10 µM were incubated with 3 µM tubulin in the presence and absence of 3 µM colchicine (Sigma-Aldrich Merck Group, St. Louis, MO, USA) in 30 mM Tris buffer for 60 min at 37 °C. CA-4 (10 µM) was used as a positive control whereas vinblastine (VBL, Sigma-Aldrich Merck Group, St. Louis, MO, USA) binding at distinct site on tubulin (10 µM) was used as negative control. This method is based on the fact that the intrinsic fluorescence of colchicine increases upon binding to tubulin. Hence, tubulin binding site inhibitors that compete with colchicine may decrease fluorescence of colchicine-tubulin complex. After incubation fluorescence was determined (excitation 350 nm, emission 435 nm) by using a spectrophotometric microplate reader (Infinite 200 Pro, Tecan, Männedorf, Switzerland). The raw fluorescence values were normalized first by subtracting the fluorescence of the blank (tubulin with tested compounds without colchicine) and setting the fluorescence of 3 μM tubulin with 3 μM colchicine to 100% (control with 0.2% DMSO).
4.13. Molecular Docking
Computational docking was carried out using the genetic algorithm-based ligand docking program Chimera and AutoDock Vina version 1.1.2 (San Diego, CA, USA). The X-ray crystal structure of αβ-tubulin complexed colchicine (PDB code: 4o2b) was used in this study. The stathmin-like domain and the C and D subunits were removed from the model. Binding modes for each colchicine analogue were investigated to understand the steric, electrostatic, and hydropathic features of the colchicine binding site. The top 10 conformations with different scores (between −5 to −9) were acquired, and the best ranking pose was visualized.
4.14. Statistical Analysis
Results are presented as mean ± standard deviation (SD). Statistical analysis was carried out using Statistica 13.3 (StatSoft Inc., Tulsa, OK, USA) and ANOVA test followed by a post hoc Tukey’s multiple comparison. A p-value level of < 0.05 was considered statistically significant.
Supplementary Materials
The following are available online at https://www.mdpi.com/1422-0067/22/5/2462/s1, Figure S1. Representative scatter diagrams of A375 cells exposed to medium with 0.2% DMSO (A), CA-4 (B), S1 (C), S19 (D), S22 (E) at 10 µM for 48 h. Figure S2. Representative scatter diagrams of HEM cells exposed to medium with 0.2% DMSO (A), CA-4 (B), S1 (C), S19 (D), S22 (E) at 10 µM for 48 h; Figure S3: Optical microscopy images of A375 cells, controls (A) and after 48 h incubation with 10 µM of compounds: CA-4 (B), S1(C), S19 (D) and S22 (E); Figure S4: Optical microscopy images of HEM cells, controls (A) and after 48 h incubation with 10 µM of compounds: CA-4 (B), S1(C), S19 (D) and S22 (E); Figure S5. Representative individual experiment of cell cycle phase distribution of A375 cells exposed to medium with 0.2% DMSO (A), CA-4 (B), S1 (C), S19 (D), S22 (E) for 6 h and DMSO (F), CA-4 (G), S1 (H), S19 (I), S22 (J) for 24 h. Figure S6: Original western blot images for Figure 8 (wells shown as white). A, C-α-tubulin, B,D- β-actin used as a loading control in stripped and reprobed membrane; Figure S7: Binding model of compound S1 at the colchicine binding-site of tubulin. The hydrogen bond is shown as blue line; Figure S8: Binding model of compound S19 at the colchicine binding-site of tubulin. The hydrogen bonds are shown as blue line; Figure S9: First possible binding model of compound S22 at the colchicine binding-site of tubulin. The hydrogen bond is shown as blue line; Figure S10: Second possible binding model of compound S22 at the colchicine binding-site of tubulin. The hydrogen bond is shown as blue line.
Author Contributions
Conceptualization, J.G.S. and M.P.; methodology, M.P. and J.G.S.; software, P.Ł.; validation, J.G.S., M.P. and M.K.; formal analysis, M.P. J.G.S. and G.M.; investigation, A.B.-L., Ł.S. (Łukasz Struk), T.J.I., M.P., Ł.S. (Łukasz Skalski), and K.P.; resources, A.B.-L. and M.P.; data curation, A.B.-L. and M.P.; writing—original draft preparation, M.P. and J.G.S.; writing—review and editing, J.G.S., M.K. and M.D.; visualization, M.P.; supervision, J.G.S., M.D. and M.K.; project administration, M.P. and J.G.S.; funding acquisition, M.P. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by National Science Center, Poland (Grant No. 2016/23/N/NZ7/02599).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Appendix A
Appendix A.1. Synthesis of Precursors of Compounds S1-S22
Synthesis and Spectroscopic Data of Compounds 1-8
Synthesis of 5-Ar-2-methoxypyridines (1-7) were performed from 5-bromo-2-methoxypyridine according to procedure described earlier, [59] with some modifications:
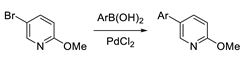 |
To the mixture of DMF (60 mL) and water (60 mL) in a 250 mL flask 5-bromo-2-methoxypyridine (3 g, 16.0 mmol), arylboronic acid (18.4 mmol), (K3PO4.5H2O (3.19 g; 12 mmol) and PdCl2 (0.057 g; 0.32 mmol). The resulting solution was stirred for 24 h at room temperature in open flask. After this time brine (100 mL) was added and the aqueous layer was extracted with diethyl ether (4 × 100 mL) and the organic layer were washed with brine and dried with MgSO4. The mixture was filtered, and the solvents were evaporated under reduced pressure. The crude product was purified by column chromatography on SiO2.
5-(4-Fluorophenyl)-2-methoxypyridine (2): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 11:1), gave white solid in 86% yield. Mp. 76–77 °C (petroleum ether: ethyl acetate). 1H and 13C NMR spectra of 2 matched those previously reported [60].
5-(3,4-Difluorophenyl)-2-methoxypyridine (3): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 10:1), gave white solid in 96% yield. Mp. 86–88 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3) δ 3.98 (s, 3H, OCH3), 6.82 (dd, J = 8.6 Hz, 1H, CH-3), 7.15–7.37 (m, 3H, C6H3F2), 7.71 (dd, J = 8.6, 2.6 Hz, 1H, CH-4), 8.32 (d, J = 2.6 Hz, 1H, CH-6). 13C NMR (101 MHz, CDCl3) δ 53.65 (O-CH3), 111.05 (CH-3), 115.60 (d, J = 17.8 Hz, CH-2′), 117.83 (d, J = 17.6 Hz, CH-5′), 122.62 (dd, J = 6.2, 3.5 Hz, CH-6′), 128.19 (C-5), 135.08 (dd, J = 6.2, 3.9 Hz, C-1′), 137.24 (CH-4), 144.91 (CH-6), 149.94 (dd, J = 248.6, 12.3 Hz, C-3′), 150.65 (dd, J = 248.2, 13.3 Hz, C-4′), 163.92 (C-2). GC-MS (EI, 70 eV): m/z = 221 (100), [M+], 220 (97), 192 (45), 190 (46), 164 (15), 151 (34), 138 (9). HRMS (ESI-TOF): m/z calcd. for C12H10F2NO [M+H]+ 222.0730; found: 222.0728.
5-(4-Chlorophenyl)-2-methoxypyridine [61] (4): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 11:1), gave white semisolid in 90% yield. 1H NMR (400.1 MHz, CDCl3): δ 3.98 (3 H, s, OCH3), 6.81 (dd, J = 8.6, 0.7 Hz,1H CH-3), 7.3 -7.49 (m, 4H, C6H5), 7.74 (dd, J = 8.6, 2.5 Hz, 1H, CH-4), 8.35 (dd, J = 2.5, 0.8 Hz, 1H, CH-6). 13C NMR (100.6 MHz, CDCl3): δ 53.59 (OCH3), 110.95 (CH-3), 127.89 (C6H5), 128.91 (C-5), 129.13 (2× C6H5), 133.44, 136.38 (C6H5), 137.23 (CH-4), 144.89 (CH-6), 163.78 (C-2). GC-MS (EI, 70 eV): m/z = 221 (33), 220 (44), 218 (100), 191 (18), 190 (55), 189 (29), 188 (18), 154 (30), 140 (10), 127 (16), 114 (16), 75 (10), 63 (14).
2-Methoxy-5-(3-methoxyphenyl)pyridine [62] (5): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 15:1), gave colorless oil in 91% yield. 1H NMR (400.1 MHz, CDCl3): δ 3.85 (s, 3H, OCH3), 3.98 (s, 3H, OMe), 6.80 (dd, J = 8.6, 0.8 Hz,1H, CH-3), 6.89 (ddd, J = 8.2, 2.6, 1.0 Hz, 1H, C6H4), 7.05 (dd, J = 2.6, 1.7 Hz, 1H, C6H4), 7.10 (dt, J = 7.7, 1.3 Hz, 1H, C6H4), 7.35 (dd, J = 8.2, 7.7 Hz, 1H, C6H4), 7.77 (dd, J = 8.6, 2.6 Hz, 1H, CH-4), 8.39 (dd, J = 2.6, 0.8 1H, CH-6). 13C NMR (100.6 MHz, CDCl3): δ = 55.54 (OCH3), 55.28 (OCH3), 110.78 (CH-3), 112.53, 112.56, 119.15 (C6H4), 129.94 (=C-5), 130.00 (C6H4), 137.47 (CH-4), 139.38 C6H4), 145.02 (CH-6), 160.07 (C6H4), 163.69 (C-2). GC-MS (EI, 70 eV): m/z = 216 (17), 215(100) [M+], 186 (42), 185 (27), 184 (12), 171 (12), 154 (11), 142 (11), 115 (17), 102 (15), 93 (9).
5-(3,4-Dimethoxyphenyl)-2-methoxypyridine [63] (6): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 3:1), gave colorless solid in 93% yield. Mp. 85–86 °C (petroleum ether:ethyl acetate). 1H NMR (400.1 MHz, CDCl3): δ 3.92 (s, 3H, OCH3), 3.94 (s, 3H OCH3), 3.98 (s, 3 H, OCH3), 6.80 (dd, J = 8.5, 0.7 Hz,1H, CH-3), 6.95 (d, J = 8.2, 1H, C6H3), 7.02 (d, J = 2.1 Hz, 1H, C6H3), 7.07 (dd, J = 8.2, 2.1 Hz, 1H, C6H3), 7.75 (dd, J = 8.6, 2.6 Hz, 1H, CH-4), 8.35 (dd, J = 2.5, 0.6 Hz, 1H, CH-6). 13C NMR (100.6 MHz, CDCl3): δ 55.52, 55.96, 56.01 (three OCH3), 110.03 (C6H3), 110.72 (CH-3), 111.68, 119.01 (C6H3), 130.03 (C-5), 130.89 (C6H3), 137.28 (CH-4), 144.64 (CH-6), 148.67, 149.35 (C6H3), 163.78 (C-2). GC-MS (EI, 70 eV); m/z = 246 (16), 245 (100), [M+], 230 (26), 202 (19), 177 (17), 170 (8), 159 (8).
2-Methoxy-5-(naphthalen-1-yl)pyridine [64] (7): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 10:1), gave white semisolid in 89% yield. 1H NMR (400.1 MHz, CDCl3): δ 4.02 (s, 3 H, OCH3), 6.87 (dd, J = 8.4, 0.8 Hz, 1 H, CH-3), 7.34–7.57 (m. 4 H, C10H7), 7.70 (dd, J = 8.4, 2.4 Hz, 1H, CH-4), 7.79–7.99 (m, 3H, C10H7), 8.29 (dd, J = 2.4, 0.8 Hz, 1H, CH-6). 13C NMR (100.6 MHz, CDCl3): δ 53.55 (OCH3), 110.35 (CH-3), 125.49 (=C-5), 125.49, 125.95, 126.31, 127.22, 128.05, 128.42, 129.45, 131.81, 133.84, 136.40 (C10H7), 140.33 (CH-4), 147.25 (CH-6), 163.52 (C-2). GC-MS (EI, 70 eV); m/z = 235 (100) [M+], 234 (74), 206 (33), 204 (35), 191 (9), 165 (24), 102 (13), 88 (8).
2-Methoxy-5-phenylpyridine [59] (1) was prepared on multigram scale as follows:
To the mixture of DMF (300 mL) and water (300 mL) in a 1 l flask 5-bromo-2-methoxypyridine (20 g, 0.106 mol), arylboronic acid (14.3 g, 0.117 mol), (K3PO4.5H2O (22 g; 0.0827 mol) and PdCl2 (0.374 g, 2.1 mmol). The resulting solution was stirred for 22 h at room temperature in open flask. After this time brine (300 mL) was added and the aqueous layer was extracted with diethyl ether (4 × 300 mL) and the organic layer were washed with brine (100 mL) and dried with MgSO4. The mixture was filtered, and the solvents were evaporated under reduced pressure. The crude product was purified by vacuum distillation (bp 68–70 °C, 0.01 mbar) afforded 1 in 95% yield (18.57 g) as colorless oil. 1H and 13C NMR spectra matched those previously reported [65].
2-Methoxy-5-(thiophen-2-yl)pyridine [66] (8) was obtained as follows:
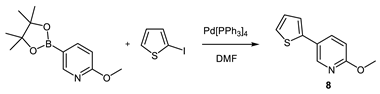 |
To a 100-mL Schlenk flask, equipped with a magnetic stir bar and a condenser crowned with argon balloon, charged with 40 mL of degassed DMF, 2-iodothiophene (Apollo Scientific, 1 g, 4.76 mmol), 2-methoxy-5-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)pyridin [67] (1.679 g, 7.14 mmol, 1.5 eqiv.) and 0.275 g tetrakis(triphenylphosphino)palladium(0) (Sigma-Aldrich Merck Group, St. Louis, MO, USA) was added, and the mixture was stirred at rt for 60 min. Next 21.5 mL of degassed 1.0 M aqueous Na2CO3 solution was added, and the reaction mixture was heated under argon at 80 °C for 3 h. After this time the reaction mixture was cooled to rt. and solvent was removed under reduced pressure. The residue was extracted by ethyl acetate, and the organic layer was washed with brine, and dried over MgSO4. The crude product was purified by column chromatography on silica gel using a mixture n-hexane and ethyl acetate (9:1 v/v) as the eluent, yielding 8 as yellow oil 0.827g (90%).
1H NMR (400 MHz, CDCl3) δ 3.96 (s, 3H, OCH3), 6.76 (dd, J = 8.6, 0.8 Hz, 1H, CH-3), 7.07 (dd, J = 5.1, 3.5 Hz, 1H, C4H3S), 7.21 (dd, J = 3.5, 1.2 Hz, 1H, C4H3S), 7.27 (dd, J = 5.1, 1.2 Hz, 1H, C4H3S), 7.76 (dd, J = 8.6, 2.6 Hz, 1H, CH-4), 8.42 (d, J = 2.6 Hz, 1H, CH-6). 13C NMR (101 MHz, CDCl3) δ 53.59 (t), 110.94 (d), 122.93 (d), 124.10 (s), 124.66 (d), 128.06 (d), 136.54 (d), 140.77 (s), 143.96 (d), 163.56 (s). GC-MS (EI, 70 eV); m/z = 191 (100) [M+], 190 (64), 162 (57), 161 (25), 160 (25), 148 (12), 121 (45), 77 (12).
Synthesis of compounds 1-8(NH):
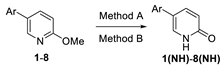 |
Method A: A mixture of 2-methoxy(5-aryl)pyridine (0.016 mol) and pyridine hydrochloride (18.4 g, 0.16 mol) was heated in a 100-mL flask at 160 °C (oil bath) on continuous stirring for 20 min. After cooling to rt brine (40 mL) was added followed with of water (20 mL). White solid was filtered off, dissolved in ethyl acetate and passed through a pad of SiO2 and washed with ethyl acetate till complete product was obtained (TLC-control) [1-2(NH)] or chromatographed on SiO2 (ethyl acetate) [3-4(NH)], [7-8(NH)].
5-Phenylpyridin-2(1H)-one [60] [1(NH)]: Yield 84%. Mp.177–178 °C. 1H and 13C NMR spectra matched those previously reported [60].
5-(4-Fluorophenyl)pyridin-2(1H)-one [60] [2(NH)]: Yield 94%. Mp. 178–180 °C (petroleum ether:ethyl acetate). 1H and 13C NMR spectra matched those previously reported [60].
5-(3,4-Difluorophenyl)pyridin-2(1H)-one [3(NH)]: Yield 90%. Mp. 210–213 °C (petroleum ether:ethyl acetate). 1H NMR (400 MHz, DMSO-d6) δ 6.57 (d, J = 9.8 Hz, 1H, CH-3), 6.97–7.42 (m, 3H, C6H3F2), 7.53 (d, J = 2.9 Hz, 1H, CH-6), 7.66 (dd, J = 9.8, 2.9 Hz, CH-4), 12.05 (br s, 1H, NH). 13C NMR (101 MHz, DMSO-d6) δ 114.49 (d, J = 17.9 Hz, CH-2′), 117.49 (C-5), 117.73 (d, J = 17.4 Hz, CH-5′), 120.75 (CH-3), 121.66 (dd, J = 6.2, 3.3 Hz, CH-6′), 132.43 (CH-6), 133.85 (dd, J = 6.2, 3.7, Hz, C-1′), 139.98 (CH-4), 149.20 (dd, J = 248.9, 12.2 Hz, C-3′), 150.12 (dd, J = 248.9, 12.2 Hz, C-4′), 162.92 (C=O). GC-MS (EI, 70 eV); m/z = 207 (100), [M+], 179 (65), 151 (72), 125 (8). HRMS (ESI-TOF): m/z calcd. for C11H8F2NO [M+H]+ 208.0574; found: 208.0565.
5-(4-Chlorophenyl)pyridin-2(1H)-one [68] [4(NH)]: Yield 96%. Mp. 185–186 °C (ethyl acetate: petroleum ether). 1H NMR (400.1 MHz, CDCl3): δ 6.70 (1 H, d, J = 9.4 Hz, CH-3), 7.32–7.43 (m, 4 H, C6H4), 7.62 (d, J = 2.7 Hz, 1H, CH-6), 7.75 (dd, J = 9.4, 2.6 Hz, 1 H, CH-4), 13.37 (s, 1 H, br, NH). 13C NMR (100.6 MHz, CDCl3): δ 120.18 (C=5), 120.45 (CH-3), 127.04, 129.31 (C6H4), 132.05 (CH-6), 133.50, 134.79 (C6H4), 141.15 (CH-4), 164.80 (C=O). GC/MS decomposition.
5-(Naphthalen-1-yl)pyridin-2(1H)-one [7(NH)]: Yield 77%. Mp. 185–187 °C (ethyl acetate). 1H NMR (400.1 MHz, CDCl3): δ 6.76 (d, J = 9.3 Hz, 1 H, CH-3), 7.38 (dd. J = 7.0, 1.3 Hz, 1 H, C10H7), 7.46–7.57 (m, 3H, C10H7), 7.60 (d, J = 2.5, 1H, CH-6), 7.70 (dd, J = 9.3, 2.5 Hz, 1H, CH-4), 7.83–7.97 (m, 3H, C10H7), 13.52 (br s, 1H, NH). 13C NMR (100.6 MHz, CDCl3): δ 119.58 (CH-3), 120.61 (=C-5), 125.06, 125.49, 126.16, 126.58, 127.00, 128.45, 128.60, 131.54, 133.91 (C10H7), 134.42 (CH-4), 134.70 (C10H7), 144.49 (CH-6), 164.86 (C-2). GC-MS (EI, 70 eV); m/z = 221 (100) [M+], 220 (25), 192 (37), 191 (16), 165 (44), 164 (14), 95 (13). HRMS (ESI-TOF): m/z calcd. for C15H12NO [M+H]+ 222.0919; found: 222.0917.
5-(Thiophen-2-yl)pyridin-2(1H)-one [8(NH)]: Yield 65%. Mp. 179–183 °C. 1H NMR (400 MHz, CDCl3) δ 6.67 (dd, J = 9.4, 0.7 Hz, 1H, CH-3), 7.05 (dd, J = 5.1, 3.6 Hz, 1H, CH-4′), 7.10 (dd, J = 3.6, 1.2 Hz, 1H, CH-3′), 7.24 (dd, J = 5.1, 1.2 Hz, 1H, CH-5′), 7.69 (dd, J = 2.7, 0.7 Hz, 1H, CH-6), 7.76 (dd, J = 9.4, 2.7 Hz, 1H, CH-4). 13C NMR (101 MHz, CDCl3) δ 115.78, (=C-5) 120.35 (CH-3), 122.70 (CH-3′), 124.36 (CH-5′), 128.11 (CH-4′), 131.07 (=CH-6), 139.27 (C-2′), 140.83 (CH-4), 164.65 (C=O). GC-MS (EI, 70 eV); m/z = 178 (11), [M+], 177 (100), 149 (39), 148 (12), 121 (23). HRMS (ESI-TOF): m/z calcd. for C9H8NOS [M+H]+ 178.0327; found: 178.0325.
Method B: A solution of 2-methoxy-5-arylpyridine (0.0106 mol), LiCl (2.24 g, 0.053 mol) and PTSA (9.1 g, 0.053 mol) in DMF (35 mL) was stirred at 120° C for 3 h. After this time, next portion of LiCl (2.97 g, 32.5 mmol) and PTSA (9.1 g, 0.053 mol) was added and the mixture was heated next 3 h at 120 °C. After cooling the reaction mixture was quenched by the addition of water (15 mL) and extracted with EtOAc (100 mL × 3) and washed with brine (15 mL × 3). The combined organic phase was dried over Na2SO4, filtered and concentrated. The residue was purified by column chromatography (SiO2, ethyl acetate).
5-(3-Methoxyphenyl)pyridin-2(1H)-one [69] [5(NH)]. Yield 80%. 1H and 13C NMR spectra matched those previously reported [69].
5-(3,4-Dimethoxyphenyl)pyridin-2(1H)-one [6(NH)]: Yield 71%. 1H NMR (400 MHz, CDCl3) δ 3.91 (s, 3H, OCH3), 3.93 (s, 3H, OCH3), 6.69 (d, J = 9.4 Hz, 1H, CH-3), 6.88–6.94 [m, 2H, C6H3(OCH3)2], 6.98 (dd, J = 8.3, 2.1 Hz, 1H, C6H3(OCH3)2), 7.62 (d, J = 2.6 Hz, 1H, CH-6), 7.78 (dd, J = 9.4, 2.6 Hz, 1H, CH-4), 13.86 (br. s, 1H, NH). 13C NMR (101 MHz, CDCl3) 55.97, 56.01 (two OCH3), 109.11, 111.74, 118.17 [C6H3(OCH3)2], 120.09 (CH-3), 121.30 (C-5), 129.27 [C6H3(OCH3)2], 131.52 (CH-6), 141.49 (CH-4), 148.69, 149.46 [C6H3(OCH3)2], 164.83 (C=O). GC-MS (EI, 70 eV); decomposition. HRMS (ESI-TOF): m/z calcd. for C13H14NO3 [M+H]+ 232.0974; found: 232.0978.
Appendix A.2. Synthesis and Spectroscopic Data of Compounds O1, O8-12, O18-22
General procedure for the benzylation of 5-arylpyridones:
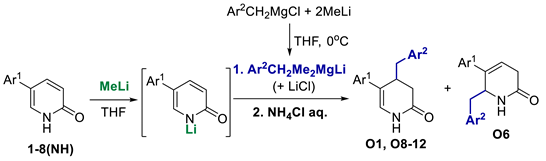 |
To 100 mL Schlenk flask the solution of BnMgCl (2.0 M in THF; 9.7 mmol, 4.85 mL) and dry THF (15 mL) was added under argon and cooled to 0 °C, followed by addition of MeLi (3.1 M in diethoxymethane; 19.4 mmol, 6.3 mL) with a syringe over 5 min. The resulting solution was stirred for 5 min at 0 °C. In a second Schlenk flask (250 mL), N-lithium 2-pyridone solution was prepared by adding with a syringe a solution of MeLi (3.1 M in diethoxymethane; 7.08 mmol, 2.29 mL) to 5-aryl-2-pyridone [1-8(NH)] solution (6.44 mmol) in THF (100 mL) at 0 °C. The solution prepared in the first Schlenk flask was then transferred to a cooled (0 °C) solution prepared in the second Schlenk flask. The resulting solution was stirred for 15 min at 0 °C and 120–180 min at rt (TLC or GC-MS control). After this time, the mixture was carefully quenched with saturated aqueous NH4Cl (30 mL). The aqueous layer was extracted with ethyl acetate (3 × 90 mL) and the combined organic layer were dried with MgSO4. The mixture was filtered, and the solvents were evaporated under reduced pressure. The crude product was purified by column chromatography on silica gel using appropriate solvents.
NOTE: BnMgCl (2.0 M in THF) was commercially available, while α-NaphCH2MgCl (0.05 M in THF) was prepared and titrated according to the procedure described earlier. [70]
p-MeOC6H4CH2MgCl in THF (A) and p-PhC6H4CH2MgCl in THF (B) were prepared as follows:
A Schlenk flask (100 mL) equipped with a 3 cm long stir bar was heated twice with a heat gun under argon. Then magnesium turnings (Sigma-Aldrich Merck Group, St. Louis, MO, USA) (A: 1.19 g, 0.0489 mol, B: 1.52 g, 0.0624 mol), porcelain finely broken (about half the volume of magnesium used) and iodine (catalytic amount) was added and heated again three short times, next vigorous stirring was started to grind the magnesium turnings (1 h at the speed of 600 rpm). Next dry THF (A: 42 mL B: 52 mL) was added (rt). In a second Schlenk flask the solution of [A: p-MeOC6H4CH2Cl (1.31 g, 8.87 mmol) in THF (5 mL), B: p-PhC6H4CH2Cl (2.1 g, 0.01 mol) in THF (7 mL)] was prepared, and the content of the second flask was added dropwise with a syringe to a suspension of Mg in THF over 120 min stirred at the speed of 500 rpm. The content of the flask was then allowed to stand for 30 min, and after this time a clear Grignard solution was transferred into separate Schlenk with a syringe from above the residue of Mg. The clear solution was titrated using a method described in the literature [71]. Grignard reagent A was obtained in 64% yield (0.13 M), B 29 % yield (0,06 M).
(4RS)-4-Benzyl-5-phenyl-3,4-dihydropyridin-2(1H)-one (O1) [15] Yield 62%. Spectra of compound O1 are in agreement with literature data [15]. Note: In the step of lithation of 2-pyridone 1.7-fold excess was used and in this conditions apart from O1 also by-product O6 was obtained.
(6RS)-6-Benzyl-5-phenyl-3,6-dihydropyridin-2(1H)-one (O6): The crude product purified by column chromatography (SiO2, ethyl acetate), gave colorless solid in 14% yield. Mp. 187–189 °C. 1H NMR (400 MHz, CDCl3): δ 2.50 (ddd, J = 22.2, 3.8, 2.7 Hz, 1 H, CHH-3), 2.63 (dd, J = 13.6, 7.2 Hz, 1 H, 6-CHH),), 2.90 (ddd, J = 22.2, 4.9, 3.0 Hz, 1H, CHH-3), 2.96 (dd, J = 13.6, 3.5 Hz, 1H, 6-CHH), 4.81 (dp, J = 7.2, 3.5 Hz, 1H, CH-6), 5.91 (dd, J = 4.9, 2.7 Hz, 1 H, =CH-4), 6.55–6.64 (br. s, 1 H, NH), 7.05–7.16 (m, 2 H, C6H5), 7.19–7.48 (m, 8H, C6H5). 13C NMR (100 MHz, CDCl3): δ 31.79 (CH2-3), 41.88 (6-CH2), 56.59 (CH-6), 120.99 (=CH-4), 126.29, 126.96, 128.03, 128.49, 128.92, 129.90 (two C6H5), 135.86 (=C-5), 136.10, 138.04 (two C6H5), 170.21 (C=O). GC-MS (EI, 70 eV): m/z = 263(3), [M+], 173 (12), 172 (100), 145 (10), 127 (11), 115 (16), 91 (16). HRMS (ESI-TOF): m/z calcd. for C18H17NNaO [M+Na]+ 286.1208; found: 286.1212.
(4RS)-4-Benzyl-5-(4-fluorophenyl)-3,4-dihydropyridin-2(1H)-one (O8): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1), gave white solid in 72% yield. Mp. 124–125 °C (petroleum ether:ethyl acetate). 1H NMR (400.1 MHz, CDCl3): δ 2.51 (ddd, J = 16.5, 2.1, 1.0 Hz, 1H, CHH-3), 2.58 (dd, J = 10.7, 6.3 Hz, 1H, 4-CHH), 2.63 (ddd, J = 16.2, 6.5, 0.6 Hz, 1H, CHH-3), 2.86 (dd, J = 13.7, 4.2 Hz, 1H, 4-CHH), 3.08 (dddd, J = 10.7, 6.5, 4.2, 2.2 Hz, 1H, CH-4), 6.47 (d, J = 4.8 Hz, 1H, =CH-6), 7.00–7.12 (m, 2 H, C6H5), 7.14–7.24 (m, 3 H, C6H5), 7.24–7.41 (m, 4 H, C6H5), 8.13 (br s, 1 H, NH). 13C NMR (100.6 MHz, CDCl3): δ 34.19 (CH2-3), 36.71 (CH-4), 37.86 (4-CH2), 115.69 (d, 2JC-F = 21.6 Hz, CH-3′, CH-5′), 120.74 (=C-5), 120.91 (=CH-6), 126.53 (C6H5), 126.55 (d, 3JC-F = 7.5 Hz, CH-2′, CH-6′) 126.50, 129.38, (C6H5), 133.33 (d, 4JC-F = 3.3 Hz, C-1′), 138.47 (C6H5), 161.82 (d, 1JC-F = 246.6 Hz, C-4′), 170.52 (C=O). GC-MS (EI, 70 eV); m/z = 281 (11), [M+], 191 (13), 190 (100), 163 (18), 145 (9), 133 (20), 115 (15), 91 (28), 65 (12). Anal. calcd for C18H16FNO: C 76.85, H 5.73, N 4.98. Found: C 76.86, H 5.75, N 5.06.
(4RS)-4-Benzyl-5-(3,4-difluorophenyl)-3,4-dihydropyridin-2(1H)-one (O9): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1), gave colorless semisolid in 41% yield. 1H NMR (400 MHz, CDCl3): δ 2.46–2.71 (m, 3H, CH2-3, 4-CHH), 2.85 (dd, J = 13.7, 4.6 Hz, 1H, 4-CHH), 2.98–3.13 (m, 1H, CH-4), 6.50 (d, J = 4.8 Hz, 1H, CH-6), 7.03–7.35 (m, 8H, C6H5, C6H3F2), 8.27 (br s, 1H, NH). 13C NMR (101 MHz, CDCl3): δ 34.25(CH2-3), 36.54 (CH-4), 37.94 (4-CH2), 113.84 (d, J = 18.0 Hz, CH-2′), 117.54 (d, J = 17.4 Hz, CH-5′), 119.76 (C-5), 120.82 (dd, J = 6.1, 3.5 Hz, CH-6′), 121.86 (CH-6), 126.65, 128.55, 129.38 (C6H5), 134.52 (dd, J = 6.4, 3.9 Hz, C-1′), 138.23 (C6H5), 149.19 (dd, J = 248.8, 12.8 Hz, C-3′), 150.50 (dd, J = 247.3, 12.8 Hz, C-4′), 170.54 (C=O). GC-MS (EI, 70 eV); m/z = 299 (8), [M+], 208 (100), 207 (42), 179 (18), 151 (21), 133 (23), 91 (43), 65 (18). HRMS (ESI-TOF): m/z calcd. for C18H15NNaO [M+Na]+ 322.1019; found: 322.1027.
(4RS)-4-Benzyl-5-(4-chlorophenyl)-3,4-dihydropyridin-2(1H)-one (O10): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 3:1), gave white solid in 65% yield. Mp. 139–142 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.51 (dt, J = 16.4, 1.3 Hz, 1H, CHH-3), 2.57–2.64 (m, 2H, 4-CHH, CHH-3), 2.86 (dd, J = 13.7, 4.1 Hz, 1H, 4-CHH), 3.05–3.09 (m, 1H, CH-4), 6.55 (d, J = 4,8 Hz, 1H, CH-6), 7.16–7.31 (m, 9H, C6H5, C6H4), 8.61 (d, J = 4.8, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 34.13 (CH2-3), 36.32 (CH-4), 37.86 (4-CH2), 120.33 (=C-5), 121.53 (=CH-6), 126.08, 126.56, 128.52, 128.9, 129.38, 132.46, 135.66, 138.39 (C6H5, C6H4), 198.1 (C=O). GC-MS (EI, 70 eV): m/z = 297 (9), [M+], 208 (33), 206 (100), 205 (9), 144 (14), 143 (27), 115 (21), 91 (17). Anal. calcd for C18H16ClNO: C 72.60, H 5.42, N 4.70. Found: C 72.73, H 5.35, N 4.81.
(4RS)-4-Benzyl-5-(3-methoxyphenyl)-3,4-dihydropyridin-2(1H)-one (O11): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1), gave colorless solid in 65% yield. Mp. 133–135 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): = δ 2.51 (ddd, J = 16.5, 2.1, 1.0 Hz, 1H, CHH-3), 2.56–2.67 (m, 2H, CHH-3, 4-CHH), 2.91 (dd, J = 13.7, 3.9 Hz, 1H, 4-CHH), 3.11 (dddd, J = 10.6, 6.5, 3.9, 2.1 Hz, 1H, CH-4), 3.84 (s, 3H, OCH3), 6.56 (d, J = 4.8 Hz, 1H, =CH-6), 6.81 (ddd, J = 8.2, 2.6, 0.9 Hz, 1H, C6H4), 6.95 (dd, J = 2.5, 1.7 Hz, 1H, C6H4), 7.02 (ddd, J = 7.7, 1.8, 0.9 Hz, 1H, C6H4), 7.16–7.35 (m, 6H, C6H4, C6H5, 8.06 (br s, 1H, NH). 13C NMR (100 MHz, CDCl3) δ 34.06 (CH2-3), 36.45 (CH-4), 37.88 (4-CH2), 55.27 (OCH3), 110.95, 111.93, 117.38 (C6H4), 121.26 (=CH-6), 121.30 (=C-5), 126.48, 128.49, 129.43, 129.81, 138.62 (overlapped signals), 159.99 (C6H4, C6H5), 170.59 (C=O); GC-MS (EI, 70 eV): m/z = 293(13), [M+], 203 (15), 202 (100), 159 (8), 130 (8), 91 (12). HRMS (ESI-TOF): m/z calcd. for C19H19NNaO2 [M+Na]+ 316.1313; found: 316.1319.
(4RS)-4-Benzyl-5-(3,4-dimethoxyphenyl)-3,4-dihydropyridin-2(1H)-one (O12). The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1), gave white solid in 61% yield. Mp. 164–166 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.50 (ddd, J = 16.5, 2.0, 1.0 Hz, 1H, CHH-3), 2.56–2.68 (m, 2H, CHH-3, 4-CHH), 2.89 (dd, J = 13.7, 3.9 Hz, 1H, 4-CHH), 3.08 (qd, J = 6.6, 5.2, 2.0 Hz, 1H, CH-4), 3.90 and 3.91 (two s, 6H, two OCH3), 6.50 (d, J = 4,7 Hz, 1H, =CH-6), 6.85–6.92 (m, 2H, C6H3), 6.98 (dd, J = 8.3, 2.2 Hz, 1H, C6H3), 7.14–7.36 (m, 5H, C6H5), 8.64 (d, J = 4.7 Hz, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 34.15 (CH2-3), 36.69 (CH-4), 37.90 (4-CH2), 55.91 and 55.96 (two OCH3), 108.31, 111.40, 117.40 (C6H3), 119.96 (=CH-6), 121.49 (=C-5), 126.46, 128.49, 129.39, 130.07, 138.79, 148.26, 149.14 (C6H3, C6H5), 170.95 (C=O). GC-MS (EI, 70 eV); m/z = 323 (23), [M+], 233 (15), 232 (100), 207 (11), 91 (17). Anal. calcd for C20H21NO3: C 74.28, H 6.55, N 4.33. Found: C 74.34, H 6.73, N 4.50.
4-(Naphthalen-2-ylmethyl)-5-phenyl-3,4-dihydropyridin-2(1H)-one (O18) was obtained with 5% of binaphtyl, which could not be removed and contaminated O18 was submitted for thionation.
(4RS)-4-(4-Methoxybenzyl)-5-phenyl-3,4-dihydropyridin-2(1H)-one (O19): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1) gave colorless solid in 88% yield. Mp. 149–152 °C (petroleum ether: ethyl acetate). 1H NMR NMR (400 MHz, CDCl3): δ 2.44–2.67 (m, 3H, CH2-3, 4-CHH), 2.84 (dd, J = 13.9, 3.8 Hz, 1H, 4-CHH), 3.02–3.14 (m, 1H, CH-4), 3.77 (s, 3H, OCH3), 6.55 (d, J = 4.4 Hz, 1H, =CH-6), 6.78 (m, 2H, C6H4), 7.05–7.17 (m, 2H, C6H4), 7.26 (tt, J = 7.0, 1.5 Hz, 1H, C6H5), 7.33–7.47 (m, 4H, C6H5), 8.37 (br s, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 34.01 (CH2-3), 36.48 (CH-4), 36.95 (4-CH2), 55.25 (O-CH3), 113.87 (2C, C6H4), 120.96 (=CH-6), 121.53 (=C-5), 124.82, 126.83, 128.83 (C6H5), 130.36 (2C), 130.65 (C6H4), 137.14, (C6H5), 158.26 (C6H4), 170.91 (C=O). GC-MS (EI, 70 eV); m/z = 293 (11) [M+], 172 (55), 171 (21), 121 (100), 115 (11). Anal. calcd for C19H19NO2: C 77.79, H 6.53, N 4.77. Found: C 77.64, H 6.73, N 4.95.
(4RS)-4-([1,1′-biphenyl]-4-ylmethyl)-5-phenyl-3,4-dihydropyridin-2(1H)-one (O20): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1) gave colorless solid in 48% yield. Mp. 207–209 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.52–2.70 (m, 3H, CH2-3, 4-CHH), 2.94 (dd, J = 13.8, 3.9, 1H, 4-CHH), 3.11–3.21 (m, 1H, CH-4) 6.56 (d, J = 4.8 Hz,1H, =CH-6), 7.23–7.47 m, 10H, ArH), 7.49–7.61 (m, 4H, ArH), 7.98 (br. s, 1H, NH). 13C NMR (100 MHz, CDCl3): δ 34.15 (CH2-3), 36.41 (CH-4), 37.50 (4-CH2), 121.01 (=CH-6), 121.48 (=C-5), 124.86 (2C), 126.88, 127.00 (2C), 127.14, 127.18 (2C), 128.74 (2C), 128.85 (2C), 129.84 (2C), 137.09, 137.71, 139.40, 140.89 (ArH), 170.52 (C=O). GC-MS (EI, 70 eV): m/z = 339 (5) [M+], 172 (100), 167 (22), 165 (11), 115 (10). Anal. calcd for C24H21NO: C 84.92, H 6.24, N 4.13. Found: C 84.62, H 6.16, N 4.21.
(4RS)-4-Benzyl-5-(naphthalen-1-yl)-3,4-dihydropyridin-2(1H)-one (O21): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 3:1) gave colorless solid in 41% yield. Mp. 174–76 °C (ethyl acetate). 1H NMR NMR (400 MHz, CDCl3): δ 2.49–2.64 (m, 2H, CHH-3, 4-CHH), 2.76–2.90 (m, 2H, CHH-3, 4-CHH), 3.04 (ddt, J = 10.8, 7.1, 3.6 Hz, 1H, CH-4), 6.25 (d, J = 4.6 Hz, 1H, =CH-6), 6.87–7.06 (m, 2H, ArH), 7.07–7.22 (m, 3H, ArH), 7.35 (dd, J = 7.0, 1.3 Hz, 1H, ArH), 7.40–7.56 (m, 3H, ArH), 7.83 (dt, J = 8.2, 1.0 Hz, 1H, ArH), 7.87–7.93 (m, 1H, ArH), 7.98 (dd, J = 6.2, 3.5 Hz, 1H, ArH), 8.38 (d, J = 4.6 Hz, 1H, NH).13C NMR (100 MHz, CDCl3): δ 34.29 (CH2-3), 37.59 (4-CH2), 39.51 (CH-4), 121.95 (=C-5), 123.61 (=CH-6), 125.32, 125.42, 125.94, 126.24, 126.30, 127.00, 128.01, 128.38 (2C), 128.62, 129.27 (2C), 132.23, 133.90, 136.51, 138.58, (C10H7, C6H5) 170.73 (C=O). GC-MS (EI, 70 eV); m/z = 313 (15) [M.+], 222 (100), 205 (14), 193 (10), 167 (12), 165 (25), 152 (11), 91 (13). HRMS (ESI-TOF): m/z calcd. for C22H20NO [M+H]+ 314.1545; found: 314.1546.
(4RS)-4-benzyl-5-(thiophen-2-yl)-3,4-dihydropyridin-2(1H)-one (O22): The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 2:1) gave white solid in 59% yield. Mp. 137–139 °C (hexane: ethyl acetate). 1H NMR (400 MHz, CDCl3) δ 2.50 (dt, J = 16.5, 1.5 Hz, 1H, CHH-3), 2.58–2.68 (m, 2H, CHH-3, 4-CHH), 2.97 (dd, J = 13.5, 3.9 Hz, 1H, 4-CHH), 3.04 (dddd, J = 10.5, 6.5, 3.9, 1.5 Hz, 1H, CH-4), 6.58 (d, J = 4.8 Hz, 1H, =CH-6), 6.98 (dd, J = 3.6, 1.2 Hz, 1H, C4H3S), 7.01 (dd, J = 5.1, 3.6 Hz, 1H, C4H3S), 7.16 (dd, J = 5.0, 1.2 Hz, 1H, C4H3S), 7.19–7.25 (m, 3H, C6H5), 7.31 (ddt, J = 7.7, 6.3, 1.2 Hz, 2H, C6H5), 7.90 (d, J = 4.8 Hz, 1H, NH). 13C NMR (101 MHz, CDCl3) δ 33.92 (CH2-3), 37.89 (CH-4), 37.99 (4-CH2), 116.75 (=C-5), 119.98 (=CH-6), 121.89 (C4H3S), 123.07 (C4H3S), 126.57 (C6H5), 127.71 (C4H3S), 128.53 (2C, C6H5), 129.48 (2C, C6H5), 138.48 (C6H5), 141.56 (C4H3S), 170.18 (C=O). GC-MS (EI, 70 eV); m/z = 269 (54), [M+], 179 (19), 178 (100), 177 (14), 151 (22), 149 (11), 117 (17), 91 (16). HRMS (ESI-TOF): m/z calcd. for C16H15NNaOS [M+Na]+ 292.0772; found: 292.0772.
Appendix A.3. Synthesis and Spectroscopic Data of N-alkylated O2, O3, O7, O13-17
General procedure for the N-alkylation of NH, 4-benzyl-3,4-aryl-3,4-dihydropyridin-2(1H)-ones O2, O3, O13-17
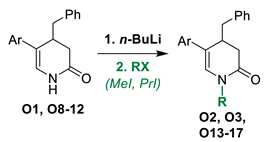 |
The solution of 5-aryl-4-benzyl-3,4-dihydropyridin-2(1H)-one (O1, O8-12) (1.82 mmol) in dry THF (30 mL) was placed in a 50 mL Schlenk flask under argon. To the solution a portion of n-BuLi (2.5 M in hexane, 2.0 mmol, 0.8 mL) was slowly added under argon with a syringe at 0 °C under stirring. The mixture was stirred 5 min at 0 °C, then MeI (4.56 mmol, 0.647 g) or PrI (2.55 mmol, 0.433 g) was added with a syringe. The resulting solution was warmed up to rt and stirred for 2 h (MeI) or 48h (PrI). After this time saturated aqueous NH4Cl (10 mL) was added and the aqueous layer was extracted with ethyl acetate (3 × 60 mL), and the combined organic layers were dried with MgSO4. The mixture was filtered, and the solvents were evaporated under reduced pressure. The crude product was purified by column chromatography on SiO2 using appropriate solvent as eluent.
4-Benzyl-1-methyl-5-phenyl-3,4-dihydropyridin-2(1H)-one (O2): (From O1.) The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate, 3: 1), gave white solid in 79% yield. Mp. 86–88 °C. 1H NMR (400 MHz, CDCl3): δ 2.51–2.65 (m, 3H, CH2–3, 4-CHH), 2.87 (dd, J = 13.7, 3.9 Hz, 1H, 4-CHH), 3.07 (s, 3H, NCH3), 3.08 (m, CH-4), 6.44 (d, J = 0.6 Hz, 1H, =CH-6), 7.15–7.22 (m, 3H, C6H5), 7.23–7.32 (m, 3H, C6H5), 7.34–7.44 (m, 4H, C6H5). 13C NMR (101 MHz, CDCl3): δ 33.90 (CH-4), 34.97 (CH2–3), 36.51 (NCH3), 38.22 (4-CH2), 121.82 (=C-5), 124.80, 126.44 126.82, (C6H5), 127.12 (=CH-6), 128.34, 128.80, 129.46, 137.11, 138.53 (C6H5), 168.41 (C=O). GC-MS (EI, 70 eV): m/z = 277 (7), [M+], 187 (13), 186 (100), 158 (5), 143 (11), 115 (11), 91 (10). HRMS (ESI-TOF): m/z calcd. for C19H19NNaO [M+Na]+ 300.1364; found: 300.1371.
4-Benzyl-5-phenyl-1-propyl-3,4-dihydropyridin-2(1H)-one (O3): (From O1.) The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate, 3: 1), gave pale yellow oil in 37% yield. 1H NMR (400 MHz, CDCl3): δ 1.01 (t, J = 7.4 Hz, 3H, CH3), 1.61 (h, J = 7.4 Hz, 2H, CH2), 2.48–2.67 (m, 3H, 4-CHH, CH2-3) 2.88 (dd, J = 13.6, 4.0 Hz, 1H, 4-CHH), 3.00–3.13 (m, 1H, CH-4) 3.28 (dt, J = 13.5, 7.3 Hz, 1H, NCHH), 3.69 (dt, J = 13.4, 7.4 Hz, 1H, NCHH), 6.47 (d, J = 0.6 Hz, 1H, =CH-6), 7.11–7.31 (m, 6H, C6H5), 7.33–7.45 (m, 4H, C6H5). 13C NMR (101 MHz, Chloroform) δ (ppm) 11.35 (CH3), 22.08 (CH2), 35.10 (CH2-3), 36.47 (CH-4), 38.05 (4-CH2), 48.06 (NCH2), 121.88 (=C-5), 124.77 (C6H5), 126.21 (=CH-6), 126.42, 126.75, 128.42, 128.81, 129.44, 137.29, 138.68 (two C6H5), 168.03 (C=O). GC-MS (EI, 70 eV): m/z = 305 (8), [M+], 215 (17), 214 (100), 172 (36), 115 (10), 91 (12). HRMS (ESI-TOF): m/z calcd. for C21H23NNaO [M+Na]+ 328.1677; found: 328.1673.
(4RS)-4-Benzyl-5-(4-fluorophenyl)-1-methyl-3,4-dihydropyridin-2(1H)-one (O13). (From O8.) The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1), gave white solid in 90% yield. Mp. 110–112 °C (petroleum ether: ethyl acetate). 1H NMR (400.1 MHz, CDCl3): δ 2.52–2.62 (m, 3H, CH2-3, 4-CHH), 2.82 (dd, J = 13.6, 4.2, 1H, 4-CHH), 3.01–3.07 (m, 1H, CH-4), 3.07 (s, 3H, NCH3), 6.36 (s, 1H, =CH-6), 7.00–7.09 (m, 2 H, C6H5), 7.12–7.17 (m, 2 H, C6H5), 7.17–7.24 (m, 2 H, C6H5), 7.25–7.31 (m, 1H, C6H5), 7.33–7.39 (m, 2H, C6H5). 13C NMR (100.6 MHz, CDCl3): δ 33.88 (CH-4), 35.06 (CH2-3), 36.83 (NCH3), 38.22 (4-CH2), 115.68 (d, 2JC-F = 21.5 Hz, CH-3′, CH-5′), 121.09 (=C-5), 126.52 (d, 3JC-F = 7.8 Hz, CH-2′, CH-6′), 126.55 (C6H5), 127 (=CH-6), 128.35, 129.41 (C6H5), 133.34 (d, 4JC-F = 3.4 Hz, C-1′), 138.37 (C6H5), 161.81 (d, 1JC-F = 246.5 Hz, C-4′), 168.25 (C=O). GC-MS (EI, 70 eV); m/z = 295 (7), [M+], 205 (13), 204 (100), 176 (9), 161 (13). HRMS (ESI-TOF): m/z calcd. for C19H18NNaO [M+Na]+ 318.127; found: 318.1262.
(4RS)-4-Benzyl-5-(3,4-difluorophenyl)-1-methyl-3,4-dihydropyridin-2(1H)-one (O14): (From O9.) The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate) gave white solid in 96% yield. Mp 89–91 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.52–2.66 (m, 3H, CH2-3, 4-CHH), 2.81 (dd, J = 13.6, 4.4 Hz, 1H, 4-CHH), 2.96–3.05 (m, 1H, CH-4), 3.07 (s, 3H, N-CH3), 6.39 (s, 1H, CH-6), 6.99–7.43 (m, 8H, C6H5, C6H3). 13C NMR (101 MHz, CDCl3): δ 33.97 (NCH3), 35.10 (CH2-3), 36.65 (CH-4), 38.29 (4-CH2), 113.75 (d, J = 18.0 Hz, CH-2′), 117.51 (d, J = 17.4 Hz, CH-5′), 120.01 (=C-5), 120.76 (dd, J = 5.8, 3.6 Hz, CH-6′), 126.62 (=CH-6), 127.89, 128.39, 129.39 (C6H5), 134.53 (dd, J = 6.6, 3.8 Hz, C-1′), 138.13 (C6H5), 149.16 (dd, J = 248.8, 12.6 Hz, C-3′), 151.65 (dd, J = 247.5, 13.2 Hz, C-4′), 168.22 (C=O). GC-MS (EI, 70 eV): m/z = 313 (6) [M+], 222 (100), 194 (11) 179 (16), 151 (8), 91 (7). Anal. calcd for C19H17F2NO: C 72.83, H 5.47, N 4.47. Found: C 73.09, H 5.75, N 4.52.
(4RS)-4-Benzyl-5-(4-chlorophenyl)-1-methyl-3,4-dihydropyridin-2(1H)-one (O15): (From O10.) The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1) gave white solid in 71% yield. Mp. 100–101 °C (petroleum ether: ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.53–2.60 (m, 3H, 4-CHH, CH2-3), 2.82 (dd, J = 13.6, 4.1 Hz, 1H, 4-CHH), 3.01–3.06 (m, 1H, CH-4), 3.06 (s, 3H, NCH3), 6.43 (s, 1H, CH-6), 7.13–7.32 (m, 9H, C6H5, C6H4). 13C NMR (100 MHz, CDCl3): δ 33.94 (CH-4), 35.01 (CH2-3), 36.48 (NCH3), 38.24 (4-CH2), 120.67 (=C-5), 126.03, 126.53 (C6H4, C6H5), 127.51 (=CH-6), 128.36, 128.91, 129.40, 132.40, 132.42, 135.66, 138.27 (C6H4, C6H5), 168.28 (C=O). GC-MS (EI, 70 eV); m/z = 311 (8), [M+], 222 (33), 220 (100), 185 (6), 157 (17), 156 (6), 115 (11), 91 (8). HRMS (ESI-TOF): m/z calcd. for C19H18ClNNaO [M+Na]+ 334.0975; found: 334.0972.
(4RS)-4-Benzyl-5-(3-methoxyphenyl)-1-methyl-3,4-dihydropyridin-2(1H)-one (O16): (From O11.) The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 1:1, gave pale yellow oil in 96% yield. 1H NMR (400 MHz, CDCl3) δ 2.49–2.67 (m, 3H, CH2-3, 4-CHH), 2.87 (dd, J = 13.6, 3.8 Hz, 1H, 4-CHH), 3.04–3.11 (m, 1H, CH-4), 3.06 (s, 3H, NCH3), 3.85 (s, 3H, OCH3), 6.45 (s, 1H, =CH-6), 6.81 (ddd, J = 8.2, 2.5, 0.8 Hz, 1H, C6H4), 6.95 (t, J = 2.1 Hz, 1H, C6H4), 7.03 (dt, J = 7.7, 1.8, 0.9 Hz, 1H, C6H4), 7.14–7.24 (m, 3H, C6H4, C6H5), 7.24–7.33 (m, 3H, C6H5). 13C NMR (101 MHz, CDCl3) δ 33.91 (CH-4), 34.95(CH2-3), 36.57 (NCH3), 38.26 (4-CH2), 55.29 (OMe), 111.03, 111.74, 117.35 (C6H4), 121.62 (=C-5), 126.45 (C6H5), 127.38 (=CH-6), 128.33, 129.46, 129.79, 138.50, 138.64, 159.99 (C6H4, C6H5), 168.46 (C=O). GC-MS (EI, 70 eV): m/z = 307 (11), [M+], 217 (16), 216 (100), 173 (6), 144 (6), 115 (6) 91 (8). HRMS (ESI-TOF): m/z calcd. for C20H21NNaO2 [M+Na]+ 330.1470; found: 330.1465.
(4RS)-4-Benzyl-5-(3,4-dimethoxyphenyl)-1-methyl-3,4-dihydropyridin-2(1H)-one (O17): (From O12.) The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 3:1) gave white solid in 91% yield. Mp. 109–110 °C (petroleum ether:ethyl acetate). 1H NMR (400 MHz, CDCl3): δ 2.52–2.66 (m, 3H, 4-CHH, CH2-3), 2.85 (dd, J = 13.6, 4.0, 1H, 4-CHH), 3.01–3.07 (m, 1H, CH-4), 3.09 (s, 3H, N-CH3), 3.91 (s, 3H, O-CH3), 3.92 (s, 3H, OCH3), 6.33–6.37 (m, 1H, =CH-6), 6.85–6.91 (m, 2H, C6H5), 6.98 (dd, J = 8.4, 2.2 Hz, 1H, C6H5), 7.14–7.24 (m, 3H, C6H5), 7.25–7.33 (m, 2H C6H5). 13C NMR (100 MHz, CDCl3): δ 33.88 (CH-4), 35.05 (CH2-3), 36.95 (N-CH3), 38.31 (4-CH2), 55.98 (2 × O-CH3), 108.40, 111.43, 117.51 (C6H5), 122.04 (=C-5), 125.97 (=CH-6), 126.46, 128.38, 129.44, 130.12, 138.68, 148.31, 149.16 (C6H5), 168.35 (C=O). GC-MS (EI, 70 eV); m/z = 337 (18) [M+], 247 (16), 246 (100), 205 (9), 91 (10). Anal. calcd for C21H23NO3: C 74.75, H 6.87, N 4.15. Found: C 74.67, H 7.06, N 4.35.
1,4-Dibenzyl-5-phenyl-3,4-dihydropyridin-2(1H)-one [15](O4): Prepared according to the procedure described earlier [15].
6-Benzyl-1-methyl-5-phenyl-3,6-dihydropyridin-2(1H)-one (O7): Prepared according to the procedure described earlier [15]. The crude product purified by column chromatography (SiO2, n-hexane: ethyl acetate 3:1), gave light cream semisolid in 82 % yield. 1H NMR (400 MHz, CDCl3): δ 1.69 (dt, J = 21.5,2.9 Hz, 1 H, CHH-3), 2.66–2.72 (m, 2 H, CHH-3, 6-CHH), 3.09 (dd, J = 13.8, 4.6 Hz, 1H, 6-CHH), 3.19 (s, 3H, NCH3), 4.61 (dtd, J = 4.9, 3.4, 1.6 Hz 1 H, CH-6), 5.86 (dd, J = 5.5, 2.4 Hz, 1H, =CH-4), 6.87–7.02 (m, 2 H, C6H5), 7.09–7.60 (m, 8H, C6H5). 13C NMR (100 MHz, CDCl3) δ = 33.25 (CH2-3), 33.56 (NCH3), 36.76 (6-CH2), 62.95 (CH-6), 122.12 (=CH-4), 126.18, 127.00, 128.00, 128.06, 128.93, 130.21 (C6H5), 135.21 (=C-5), 135.51, 137.71 (C6H5), 169.01 (C=O). GC-MS (EI, 70 eV): m/z = 277 (2), [M+], 187 (13), 186 (100), 158, 143 (11), 115 (13), 91 (13). HRMS (ESI-TOF): m/z calcd. for C19H19NNaO [M+Na]+ 300.1364; found: 300.1368.
References
- Wood, K.W.; Cornwell, W.D.; Jackson, J.R. Past and future of the mitotic spindle as an oncology target. Curr. Opin. Pharmacol. 2001, 1, 370–377. [Google Scholar] [CrossRef]
- Liu, Y.M.; Chen, H.L.; Lee, H.Y.; Liou, J.P. Tubulin inhibitors: A patent review. Expert Opin. Ther. Pat. 2014, 24, 69–88. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.S.; Koh, C.G.; Li, H.Y. Mitosis-targeted anti-cancer therapies: Where they stand. Cell Death Dis. 2012, 3, e411. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.T.; Liu, Y.N.; Liu, Z.P. Tubulin colchicine binding site inhibitors as vascular disrupting agents in clinical developments. Curr. Med. Chem. 2015, 22, 1348–1360. [Google Scholar] [CrossRef]
- Finkelstein, Y.; Aks, S.E.; Hutson, J.R.; Juurlink, D.N.; Nguyen, P.; Dubnov-Raz, G.; Pollak, U.; Koren, G.; Bentur, Y. Colchicine poisoning: The dark side of an ancient drug. Clin. Toxicol. (Phila) 2010, 48, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Chen, J.; Xiao, M.; Li, W.; Miller, D.D. An overview of tubulin inhibitors that interact with the colchicine binding site. Pharm. Res. 2012, 29, 2943–2971. [Google Scholar] [CrossRef] [PubMed]
- Henriques, A.C.; Ribeiro, D.; Pedrosa, J.; Sarmento, B.; Silva, P.M.A.; Bousbaa, H. Mitosis inhibitors in anticancer therapy: When blocking the exit becomes a solution. Cancer Lett. 2019, 440–441, 64–81. [Google Scholar] [CrossRef] [PubMed]
- Penna, L.S.; Henriques, J.A.P.; Bonatto, D. Anti-mitotic agents: Are they emerging molecules for cancer treatment? Pharmacol. Ther. 2017, 173, 67–82. [Google Scholar] [CrossRef]
- Perużyńska, M.; Piotrowska, K.; Tkacz, M.; Kurzawski, M.; Struk, Ł.; Borzyszkowska, A.; Idzik, T.J.; Sośnicki, J.G.; Droździk, M. Comparative evaluation of new dihydropyrimidine and dihydropyridine derivatives perturbing mitotic spindle formation. Future Med. Chem. 2018, 10, 2395–2410. [Google Scholar] [CrossRef]
- Martins, P.; Jesus, J.; Santos, S.; Raposo, L.R.; Roma-Rodrigues, C.; Baptista, P.V.; Fernandes, A.R. Heterocyclic Anticancer Compounds: Recent Advances and the Paradigm Shift towards the Use of Nanomedicine’s Tool Box. Molecules 2015, 20, 16852–16891. [Google Scholar] [CrossRef] [PubMed]
- Mayer, T.U.; Kapoor, T.M.; Haggarty, S.J.; King, R.W.; Schreiber, S.L.; Mitchison, T.J. Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen. Science 1999, 286, 971–974. [Google Scholar] [CrossRef]
- DeBonis, S.; Simorre, J.P.; Crevel, I.; Lebeau, L.; Skoufias, D.A.; Blangy, A.; Ebel, C.; Gans, P.; Cross, R.; Hackney, D.D.; et al. Interaction of the mitotic inhibitor monastrol with human kinesin Eg5. Biochemistry 2003, 42, 338–349. [Google Scholar] [CrossRef]
- El-Nassan, H.B. Advances in the discovery of kinesin spindle protein (Eg5) inhibitors as antitumor agents. Eur. J. Med. Chem. 2013, 62, 614–631. [Google Scholar] [CrossRef]
- Prashantha Kumar, B.R.; Masih, P.; Karthikeyan, E.; Bansal, A.; Suja Vijayan, P. Synthesis of novel Hantzsch dihydropyridines and Biginelli dihydropyrimidynes of biological interest: A 3D-QSAR study on their cytotoxicity. Med. Chem. Res. 2010, 19, 344–363. [Google Scholar] [CrossRef]
- Sośnicki, G.J.; Idzik, T.; Borzyszkowska, A.; Wróblewski, E.; Maciejewska, G.; Struk, Ł. Addition of novel benzylmagnesium “ate” complexes of BnR2MgLi type to 2-(thio)pyridones and related compounds. Tetrahedron 2017, 73, 481–493. [Google Scholar]
- McLoughlin, E.C.; O’Boyle, N.M. Colchicine-Binding Site Inhibitors from Chemistry to Clinic: A Review. Pharmaceuticals (Basel) 2020, 13, 8. [Google Scholar] [CrossRef]
- Sośnicki, J.G.; Struk, Ł.; Kurzawski, M.; Perużyńska, M.; Maciejewska, G.; Droździk, M. Regioselective synthesis of novel 4,5-diaryl functionalized 3,4-dihydropyrimidine-2(1H)-thiones via a non-Biginelli-type approach and evaluation of their in vitro anticancer activity. Org. Biomol. Chem. 2014, 12, 3427–3440. [Google Scholar] [CrossRef]
- Ghose, A.K.; Pritchett, A.; Crippen, G.M. Atomic physicochemical parameters for three dimensional structure directed quantitative structure-activity relationships III: Modeling hydrophobic interactions. J. Comput. Chem. 1988, 9, 80. [Google Scholar] [CrossRef]
- Hafez, H.N.; Alsalamah, S.A.; El-Gazzar, A.B.A. Synthesis of thiophene and N-substituted thieno[3,2-d] pyrimidine derivatives as potent antitumor and antibacterial agents. Acta Pharm. 2017, 67, 275–292. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, F.A.; Pereira, M.C.; de Oliveira, T.B.; Mendonça Junior, F.J.B.; de Lima, M.D.C.A.; Pitta, M.G.D.R.; Pitta, I.D.R.; de Melo Rêgo, M.J.B.; da Rocha Pitta, M.G. Anticancer properties of thiophene derivatives in breast cancer MCF-7 cells. Anticancer Drugs. 2018, 29, 157–166. [Google Scholar] [CrossRef]
- Gulipalli, K.C.; Bodige, S.; Ravula, P.; Endoori, S.; Vanaja, G.R.; Suresh Babu, G.; Narendra Sharath Chandra, J.N.; Seelam, N. Design, synthesis, in silico and in vitro evaluation of thiophene derivatives: A potent tyrosine phosphatase 1B inhibitor and anticancer activity. Bioorg. Med. Chem. Lett. 2017, 27, 3558–3564. [Google Scholar] [CrossRef]
- Romagnoli, R.; Kimatrai Salvador, M.; Schiaffino Ortega, S.; Baraldi, P.G.; Oliva, P.; Baraldi, S.; Lopez-Cara, L.C.; Brancale, A.; Ferla, S.; Hamel, E.; et al. 2-Alkoxycarbonyl-3-arylamino-5-substituted thiophenes as a novel class of antimicrotubule agents: Design, synthesis, cell growth and tubulin polymerization inhibition. Eur. J. Med. Chem. 2018, 143, 683–698. [Google Scholar] [CrossRef]
- Romagnoli, R.; Baraldi, P.G.; Kimatrai Salvador, M.; Preti, D.; Aghazadeh Tabrizi, M.; Bassetto, M.; Brancale, A.; Hamel, E.; Castagliuolo, I.; Bortolozzi, R.; et al. Synthesis and biological evaluation of 2-(alkoxycarbonyl)-3-anilinobenzo[b]thiophenes and thieno[2,3-b]pyridines as new potent anticancer agents. J. Med. Chem. 2013, 56, 2606–2618. [Google Scholar] [CrossRef] [PubMed]
- Hura, N.; Sawant, A.V.; Kumari, A.; Guchhait, S.K.; Panda, D. Combretastatin-Inspired Heterocycles as Antitubulin Anticancer Agents. ACS Omega. 2018, 3, 9754–9769. [Google Scholar] [CrossRef] [PubMed]
- Do Amaral, D.N.; Cavalcanti, B.C.; Bezerra, D.P.; Ferreira, P.M.; Castro Rde, P.; Sabino, J.R.; Machado, C.M.; Chammas, R.; Pessoa, C.; Sant’Anna, C.M.; et al. Docking, synthesis and antiproliferative activity of N-acylhydrazone derivatives designed as combretastatin A4 analogues. PLoS ONE 2014, 9, e85380. [Google Scholar] [CrossRef] [PubMed]
- Blay, J.Y.; Pápai, Z.; Tolcher, A.W.; Italiano, A.; Cupissol, D.; López-Pousa, A.; Chawla, S.P.; Bompas, E.; Babovic, N.; Penel, N.; et al. Ombrabulin plus cisplatin versus placebo plus cisplatin in patients with advanced soft-tissue sarcomas after failure of anthracycline and ifosfamide chemotherapy: A randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. 2015, 16, 531–540. [Google Scholar] [CrossRef]
- Peña-Morán, O.A.; Villarreal, M.L.; Álvarez-Berber, L.; Meneses-Acosta, A.; Rodríguez-López, V. Cytotoxicity, Post-Treatment Recovery, and Selectivity Analysis of Naturally Occurring Podophyllotoxins from Bursera fagaroides var. fagaroides on Breast Cancer Cell Lines. Molecules 2016, 21, 1013. [Google Scholar] [CrossRef]
- Darzynkiewicz, Z.; Gloria Juan, G.; Bedner, E. Determining Cell Cycle Stages by Flow Cytometry. Curr. Protoc. Cell Biol. 2001, 1, 8.4.1–8.4.18. [Google Scholar] [CrossRef]
- Kajstura, M.; Halicka, H.D.; Pryjma, J.; Darzynkiewicz, Z. Discontinuous fragmentation of nuclear DNA during apoptosis revealed by discrete “sub-G1” peaks on DNA content histograms. Cytom. A 2007, 71, 125–131. [Google Scholar] [CrossRef]
- Fankhauser, G.; Humphrey, R.R. The Rare Occurrence of Mitosis Without Spindle Apparatus (“Colchicine Mitosis”) Producing Endopolyploidy in Embryos of the Axolotl. Proc. Natl. Acad. Sci. USA 1952, 38, 1073–1082. [Google Scholar] [CrossRef]
- Gilson, P.; Josa-Prado, F.; Beauvineau, C.; Naud-Martin, D.; Vanwonterghem, L.; Mahuteau-Betzer, F.; Moreno, A.; Falson, P.; Lafanechère, L.; Frachet, V.; et al. Identification of pyrrolopyrimidine derivative PP-13 as a novel microtubule-destabilizing agent with promising anticancer properties. Sci. Rep. 2017, 7, 10209. [Google Scholar] [CrossRef]
- Romagnoli, R.; Baraldi, P.G.; Cara, C.L.; Hamel, E.; Basso, G.; Bortolozzi, R.; Viola, G. Synthesis and biological evaluation of 2-(3′,4′,5′-trimethoxybenzoyl)-3-aryl/arylaminobenzo[b]thiophene derivatives as a novel class of antiproliferative agents. Eur. J. Med. Chem. 2010, 45, 5781–5791. [Google Scholar] [CrossRef]
- Bhattacharyya, B.; Panda, D.; Gupta, S.; Banerjee, M. Anti-mitotic activity of colchicine and the structural basis for its interaction with tubulin. Med. Res. Rev. 2008, 28, 155–183. [Google Scholar] [CrossRef] [PubMed]
- Ogden, A.; Rida, P.C.; Reid, M.D.; Aneja, R. Interphase microtubules: Chief casualties in the war on cancer? Drug Discov. Today. 2014, 19, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Iyama, T.; Wilson, D.M., 3rd. DNA repair mechanisms in dividing and non-dividing cells. DNA Repair (Amst.) 2013, 12, 620–636. [Google Scholar] [CrossRef] [PubMed]
- Jaunky, D.B.; Husser, M.; Larocque, K.; Liu, P.; Thampipillai, S.; Forgione, P.; Piekny, A.J. A novel compound that disrupts mitotic spindle poles in human cells. bioRxiv 2020. [Google Scholar] [CrossRef]
- Zhu, C.; Zhao, J.; Bibikova, M.; Leverson, J.D.; Bossy-Wetzel, E.; Fan, J.B.; Abraham, R.T.; Jiang, W. Functional analysis of human microtubule-based motor proteins, the kinesins and dyneins, in mitosis/cytokinesis using RNA interference. Mol. Biol. Cell. 2005, 16, 3187–3199. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.; Bechi, B.; Tighe, A.; Thompson, S.; Procter, D.J.; Taylor, S.S. Cenp-E inhibitor GSK923295: Novel synthetic route and use as a tool to generate aneuploidy. Oncotarget 2015, 6, 20921–20932. [Google Scholar] [CrossRef]
- Blank, M.; Shiloh, Y. Programs for cell death: Apoptosis is only one way to go. Cell Cycle 2007, 6, 686–695. [Google Scholar] [CrossRef]
- Castedo, M.; Perfettini, J.L.; Roumier, T.; Andreau, K.; Medema, R.; Kroemer, G. Cell death by mitotic catastrophe: A molecular definition. Oncogene 2004, 23, 2825–2837. [Google Scholar] [CrossRef]
- Kanthou, C.; Greco, O.; Stratford, A.; Cook, I.; Knight, R.; Benzakour, O.; Tozer, G. The tubulin-binding agent combretastatin A-4-phosphate arrests endothelial cells in mitosis and induces mitotic cell death. Am. J. Pathol. 2004, 165, 1401–1411. [Google Scholar] [CrossRef]
- Vitale, I.; Antoccia, A.; Cenciarelli, C.; Crateri, P.; Meschini, S.; Arancia, G.; Pisano, C.; Tanzarella, C. Combretastatin CA-4 and combretastatin derivative induce mitotic catastrophe dependent on spindle checkpoint and caspase-3 activation in non-small cell lung cancer cells. Apoptosis 2007, 12, 155–166. [Google Scholar] [CrossRef]
- Risinger, A.L.; Giles, F.J.; Mooberry, S.L. Microtubule dynamics as a target in oncology. Cancer Treat. Rev. 2009, 35, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Kamal, A.; Shaik, B.; Nayak, V.L.; Nagaraju, B.; Kapure, J.S.; Shaheer Malik, M.; Shaik, T.B.; Prasad, B. Synthesis and biological evaluation of 1,2,3-triazole linked aminocombretastatin conjugates as mitochondrial mediated apoptosis inducers. Bioorg. Med. Chem. 2014, 22, 5155–5167. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, A.; Fornabaio, M.; Kellogg, G.E.; Gupton, J.T.; Gewirtz, D.A.; Yeudall, W.A.; Vega, N.E.; Mooberry, S.L. Docking and hydropathic scoring of polysubstituted pyrrole compounds with antitubulin activity. Bioorg. Med. Chem. 2008, 16, 2235–2242. [Google Scholar] [CrossRef]
- Da, C.; Telang, N.; Hall, K.; Kluball, E.; Barelli, P.; Finzel, K.; Jia, X.; Gupton, J.T.; Mooberry, S.L.; Kellogg, G.E. Developing novel C-4 analogues of pyrrole-based antitubulin agents: Weak but critical hydrogen bonding in the colchicine site. Medchemcomm 2013, 4, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.L.; McGrath, C.; Hermone, A.R.; Burnett, J.C.; Zaharevitz, D.W.; Day, B.W.; Wipf, P.; Hamel, E.; Gussio, R. A common pharmacophore for a diverse set of colchicine site inhibitors using a structure-based approach. J. Med. Chem. 2005, 48, 6107–6116. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, H.; Gigant, B.; Yu, Y.; Wu, Y.; Chen, X.; Lai, Q.; Yang, Z.; Chen, Q.; Yang, J. Structures of a diverse set of colchicine binding site inhibitors in complex with tubulin provide a rationale for drug discovery. FEBS J. 2016, 283, 102–111. [Google Scholar] [CrossRef]
- Russowsky, D.; Canto, R.F.; Sanches, S.A.; D’Oca, M.G.; de Fátima, A.; Pilli, R.A.; Kohn, L.K.; Antônio, M.A.; de Carvalho, J.E. Synthesis and differential antiproliferative activity of Biginelli compounds against cancer cell lines: Monastrol, oxo-monastrol and oxygenated analogues. Bioorg. Chem. 2006, 34, 173–182. [Google Scholar] [CrossRef]
- Ardito, F.; Giuliani, M.; Perrone, D.; Troiano, G.; Lo Muzio, L. The crucial role of protein phosphorylation in cell signaling and its use as targeted therapy (Review). Int. J. Mol. Med. 2017, 40, 271–280. [Google Scholar] [CrossRef]
- Rath, O.; Kozielski, F. Kinesins and cancer. Nat. Rev. Cancer. 2012, 12, 527–539. [Google Scholar] [CrossRef] [PubMed]
- Daire, V.; Poüs, C. Kinesins and protein kinases: Key players in the regulation of microtubule dynamics and organization. Arch. Biochem. Biophys. 2011, 510, 83–92. [Google Scholar] [CrossRef]
- Park, H.; Hong, S.; Hong, S. Nocodazole is a high-affinity ligand for the cancer-related kinases ABL, c-KIT, BRAF, and MEK. ChemMedChem 2012, 7, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Frezzato, F.; Trimarco, V.; Martini, V.; Gattazzo, C.; Ave, E.; Visentin, A.; Cabrelle, A.; Olivieri, V.; Zambello, R.; Facco, M.; et al. Leukaemic cells from chronic lymphocytic leukaemia patients undergo apoptosis following microtubule depolymerization and Lyn inhibition by nocodazole. Br. J. Haematol. 2014, 165, 659–672. [Google Scholar] [CrossRef] [PubMed]
- Ravanbakhsh, S.; Gajewski, M.; Greiner, R.; Tuszynski, J.A. Determination of the optimal tubulin isotype target as a method for the development of individualized cancer chemotherapy. Theor. Biol. Med. Model. 2013, 10, 29. [Google Scholar] [CrossRef] [PubMed]
- Scott, C.A.; Walker, C.C.; Neal, D.A.; Harper, C.E.; Bloodgood, R.A.; Somers, K.D.; Mills, S.E.; Rebhun, L.I.; Levine, P.A. Beta-tubulin epitope expression in normal and malignant epithelial cells. Arch. Otolaryngol. Head Neck Surg. 1990, 116, 583–589. [Google Scholar] [CrossRef]
- Blagosklonny, M.V.; Giannakakou, P.; el-Deiry, W.S.; Kingston, D.G.; Higgs, P.I.; Neckers, L.; Fojo, T. Raf-1/bcl-2 phosphorylation: A step from microtubule damage to cell death. Cancer Res. 1997, 57, 130–135. [Google Scholar]
- Sung, M.; Giannakakou, P. BRCA1 regulates microtubule dynamics and taxane-induced apoptotic cell signaling. Oncogene 2014, 33, 1418–1428. [Google Scholar] [CrossRef]
- Liu, C.; Ni, Q.; Bao, F.; Qiu, J. A simple and efficient protocol for a palladium-catalyzed ligand-free Suzuki reaction at room temperature in aqueous DMF. Green Chem. 2011, 13, 1260–1266. [Google Scholar] [CrossRef]
- Sośnicki, J.G.; Struk, Ł.; Idzik, T.; Maciejewska, G. Scope and limitations of the synthesis of functionalized quinolizidi-nones and related compounds by a simple precursor approach via addition of lithium allylmagnesates to 2-pyridones and RCM as key steps. Tetrahedron 2014, 70, 8624–8635. [Google Scholar] [CrossRef]
- Tan, X.; Zhou, Z.J.; Zhang, J.X.; Duan, X.H. Efficient One-Pot Cross-Coupling of Two Aryl Halides by Stannylation/Stille Reaction in Water under Microwave Irradiation. Eur. J. Org. Chem. 2014, 24, 5153–5157. [Google Scholar] [CrossRef]
- Markovic, T.; Rocke, B.N.; Blakemore, D.C.; Mascitti, V.; Willis, M.C. Pyridine sulfinates as general nucleophilic cou-pling partners in palladium-catalyzed cross-coupling reactions with aryl halides. Chem. Sci. 2017, 8, 4437–4442. [Google Scholar] [CrossRef] [PubMed]
- Spada, A.P.; Myers, M.R.; Maguire, M.P.; Persons, P.E. Bis Mono- and Bicyclic Aryl and Heteroaryl Compounds Which Inhibit EGF and/or PDGF Receptor Tyrosine Kinase. U.S. Patent Grant US-5480883-A, 2 January 1996. [Google Scholar]
- Bolliger, J.L.; Frech, C.M. [Pd(Cl)2{P(NC5H10)(C6H11)2}2]—A Highly Effective and Extremely Versatile Palladi-um-Based Negishi Catalyst that Efficiently and Reliably Operates at Low Catalyst Loadings. Chem.—Eur. J. 2010, 16, 11072–11081. [Google Scholar] [CrossRef] [PubMed]
- Duong, A.H.; Wu, W.; Teo, Y.-Y. Cobalt-Catalyzed Cross-Coupling Reactions of Arylboronic Esters and Aryl Halides. Organometallics 2017, 36, 4363–4366. [Google Scholar] [CrossRef]
- Parry, P.R.; Wang, C.; Batsanov, A.S.; Bryce, M.R.; Tarbit, B. Functionalized Pyridylboronic Acids and Their Su-zuki Cross-Coupling Reactions To Yield Novel Heteroarylpyridines. J. Org. Chem. 2002, 67, 7541–7543. [Google Scholar] [CrossRef]
- Struk, Ł.; Sośnicki, J.G. Noncryogenic Synthesis of Functionalized 2-Methoxypyridines by Halogen-Magnesium Exchange Using Lithium Dibutyl (isopropyl) magnesate (1-) and Lithium Chloride. Synthesis 2012, 44, 735–746. [Google Scholar]
- Church, R.; Trust, R.; Albright, J.D.; Powell, D. New Synthetic Routes to 3-, 5-, and 6-Aryl-2-chloropyridines. J. Org. Chem. 1995, 60, 3750–3758. [Google Scholar] [CrossRef]
- Idzik, T.J.; Borzyszkowska-Ledwig, A.; Struk, Ł.; Sośnicki, J.G. Magnesiate-Utilized/Benzyne-Mediated Approach to Indenopyridones from 2-Pyridones: An Attempt To Synthesize the Indenopyridine Core of Haouamine. Org. Lett. 2019, 21, 9667–9671. [Google Scholar] [CrossRef]
- Idzik, T.J.; Myk, Z.M.; Sośnicki, J.G. Diversity-Oriented Synthesis toward Fused and Bridged Benzobicyclic Piperidin(on)es. J. Org. Chem. 2019, 84, 8046–8066. [Google Scholar] [CrossRef]
- Krasovskiy, A.; Knochel, P. Convenient Titration Method for Organometallic Zinc, Magnesium, and Lanthanide-Reagents. Synthesis 2006, 5, 890–891. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).