Abstract
The most frequent cause of death by cancer worldwide is lung cancer, and the 5-year survival rate is still very poor for patients with advanced stage. Understanding the crosstalk between the signaling pathways that are involved in disease, especially in metastasis, is crucial to developing new targeted therapies. Toll-like receptors (TLRs) are master regulators of the immune responses, and their dysregulation in lung cancer is linked to immune escape and promotes tumor malignancy by facilitating angiogenesis and proliferation. On the other hand, over-activation of the WNT signaling pathway has been reported in lung cancer and is also associated with tumor metastasis via induction of Epithelial-to-mesenchymal-transition (EMT)-like processes. An interaction between both TLRs and the WNT pathway was discovered recently as it was found that the TLR pathway can be activated by WNT ligands in the tumor microenvironment; however, the implications of such interactions in the context of lung cancer have not been discussed yet. Here, we offer an overview of the interaction of TLR-WNT in the lung and its potential implications and role in the oncogenic process.
1. Introduction
Lung cancer is the most frequent cause of death by cancer worldwide [1]. The 5-year survival rate in lung cancer varies from 73% (in stage IA) to 13% (in stage IV). Lung cancer can be classified into two main subtypes according to the histological pattern, namely, small-cell lung carcinomas (SCLC) and non-small cell lung carcinomas (NSCLC) [2]. NSCLC represents 85% of lung cancer cases where adenocarcinoma (ADC), squamous cell carcinoma (SCC), and large-cell carcinoma (LCC) are the main NSCLC subtypes [3]; while SCLC represents 15% of all lung cancers and is classified as small cell carcinoma when it occurs without a combination of large cell carcinoma, that is, with pure histology [4]. Both for the development of mammals, as well as for the regeneration and repair of tissues, the regulation of cell proliferation and survival is very important. What leads to cancer is cell proliferation and survival [5]. In 1863, Rudolf Virchow was the first to identify that leukocytes were present in tumor tissue, and thus he hypothesized that there was a link between inflammation and tumor progression [6]. Host immune responses must induce cell proliferation and survival, which is necessary for host defense after infection, as they stimulate leukocyte proliferation in the bone marrow and induce clonal expansion of lymphocytes. Innate immunity is the first line of defense against microorganisms, it is characterized by not being clonal, and it acts on a very large number of pathogens since it focuses on the recognition of a few conserved structures [7]. Immune cells are capable of mediating pathogen recognition through the expression of the so-called pattern recognition receptors (PRRs), such as the Toll-like receptors (TLRs), which are responsible for the identification of pathogen-associated molecular patterns (PAMPs) and the induction of the inflammatory response [8]. Furthermore, immune-mediated inflammatory responses through TLRs can indirectly initiate tissue repair processes after local infection [5]. The WNT (Wingless/Integrated) pathway is a key developmental pathway involved in tissue repair and regeneration. Upon specific binding to its ligands, the WNT pathway is activated and drives the translocation of transcription factors that regulate gene expression [9]. The role that the TLR family and the WNT signaling pathway play in cancer development and progression has been largely explored in the literature, and alterations of both pathways have been independently reported in lung cancer. However, although an interaction between both pathways exists, there is little reported information about the link between TLR and WNT in lung cancer. In this review, we will summarize the current knowledge of TLR and WNT pathways in lung cancer with a focus on the interaction of both signaling pathways and their involvement in tumor development and progression.
2. Toll-like Receptors (TLRs) in Lung
2.1. TLRs and TLR Signaling
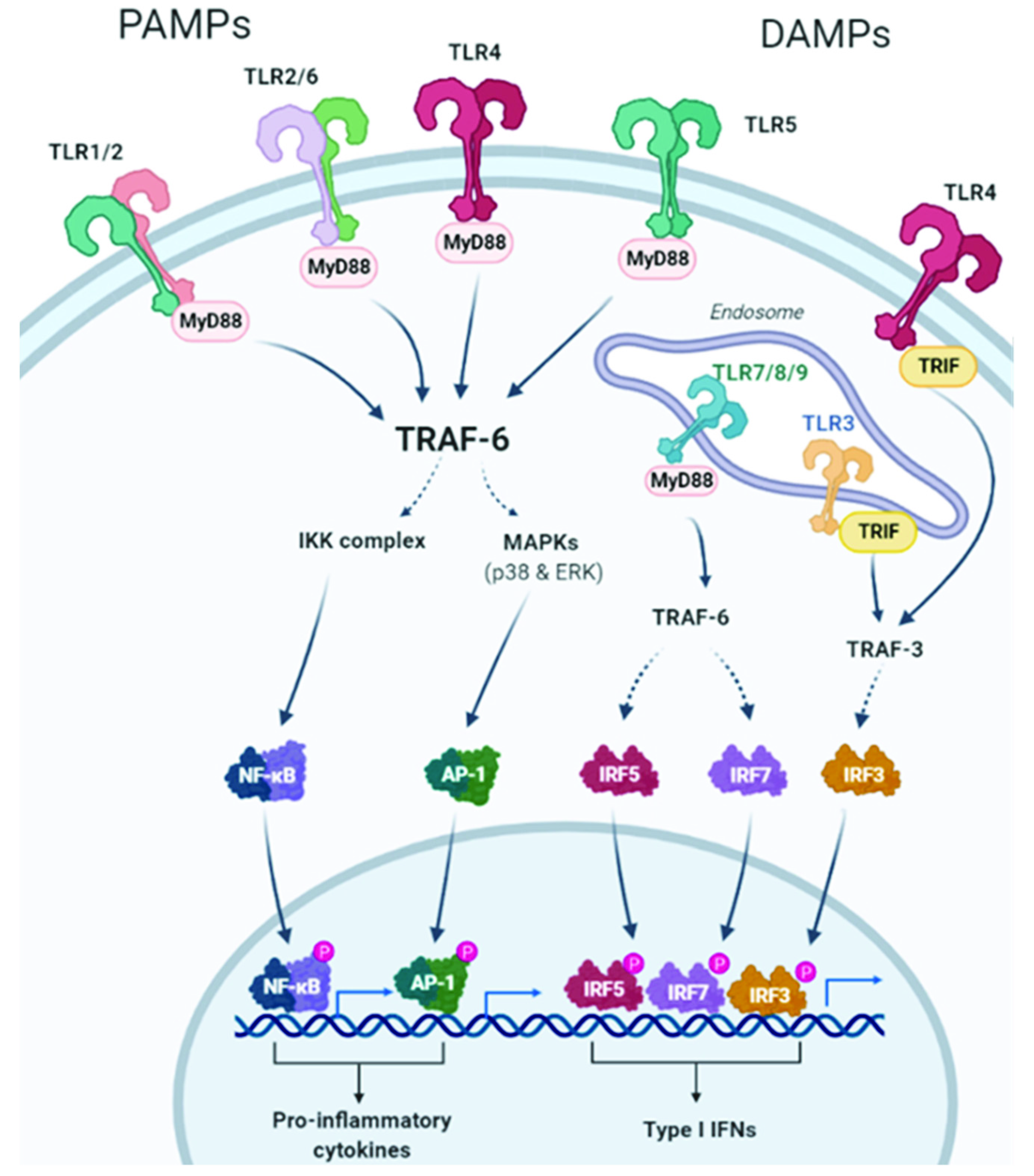
TLRs comprise a family of receptors in the immune system, which are structurally characterized by extracellular leucine-rich repeats (LRRs) and an intracellular Toll/IL-1 receptor (TIR) signaling domain [10]. The TLR family consists of ten members in humans (TLR1-TLR10) [8]. The presence of mRNA or protein of TLR1, TLR2, TLR3, TLR4, TLR7, TLR8, TLR9, and TLR10 has been detected in lung tissue [11,12]. TLRs are activated following the direct recognition of a wide range of pathogen-associated molecular patterns (PAMPs) that include bacterial lipopolysaccharides (LPS), lipoproteins, and flagellin, as well as viral and bacterial nucleic acids [8]. Once PAMPs are recognized by TLRs, a signaling cascade is initiated that triggers the expression of inflammatory mediators, including many cytokines, chemokines, and cell adhesion molecules [13]. In addition to PAMPs, TLRs can also detect the so-called DAMPs (damage-associated molecular patterns), which are a variety of endogenous molecules that are released as a result of cell or tissue damage. Similar to PAMPs, DAMPs can also trigger inflammation even in the absence of infection [14] (Figure 1). In addition to their role in mediating the inflammatory response, TLRs can also regulate both proliferation [15] and cell survival by suppressing apoptosis [5,16,17], thus protecting against injury and initiating tissue repair. However, if this function becomes dysregulated, it can lead to uncontrolled proliferation and tumor development [18]. In fact, different tumor cells seem to express their own set of TLRs, suggesting a role for TLRs in the regulation of tumor growth [19]. Therefore, TLRs act as a double-edged sword where, on the one hand, they identify cancer-specific antigens and activate innate responses, which can serve as a tumor suppressor, and on the other hand, they can promote cell proliferation and survival and latent chronic inflammation by inducing persistent, adaptive responses that facilitate the process of tumorigenesis [20,21,22].
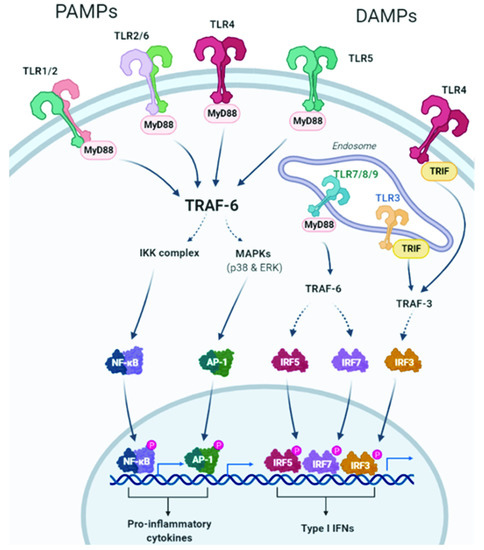
Figure 1.
TLR2, TLR1, TLR4, TLR5, and TLR6 are expressed on the outer cell membrane; they recognize both extracellular pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs). TLR3, TLR7, TLR8, and TLR9 are located in endosomal compartments where they bind to nucleic acids. All TLRs use the myeloid differentiation primary response protein 88 (MyD88) pathway, except TLR3, whose signaling depends on the IFN-α (TRIF) pathway that contains the TIR domain-containing adapter. TLR activates the MyD88-dependent canonical pathway that, through recruitment of TNF receptor-associated factors (TRAF) 6, leads to the activation of the transcription factor NF-KB, mitogen-activated protein kinase (MAPK), and activator protein-1 (AP-1), with the consequent induction of the production of proinflammatory cytokines. Intracellular TLRs are primarily involved in the type I interferon response. TLR7, TLR8, and TLR9 activate interferon regulatory factor 7 (IRF7) through the recruitment of TRAF6. TLR3 and TLR4 use the TRIF-dependent pathway. TRAF3 is activated and consequently activates IRF3, resulting in the induction of type I interferon (IFN) production. Created with BioRender.com (accessed on 24 February 2022).
2.2. TLRs Role in Lung Cancer
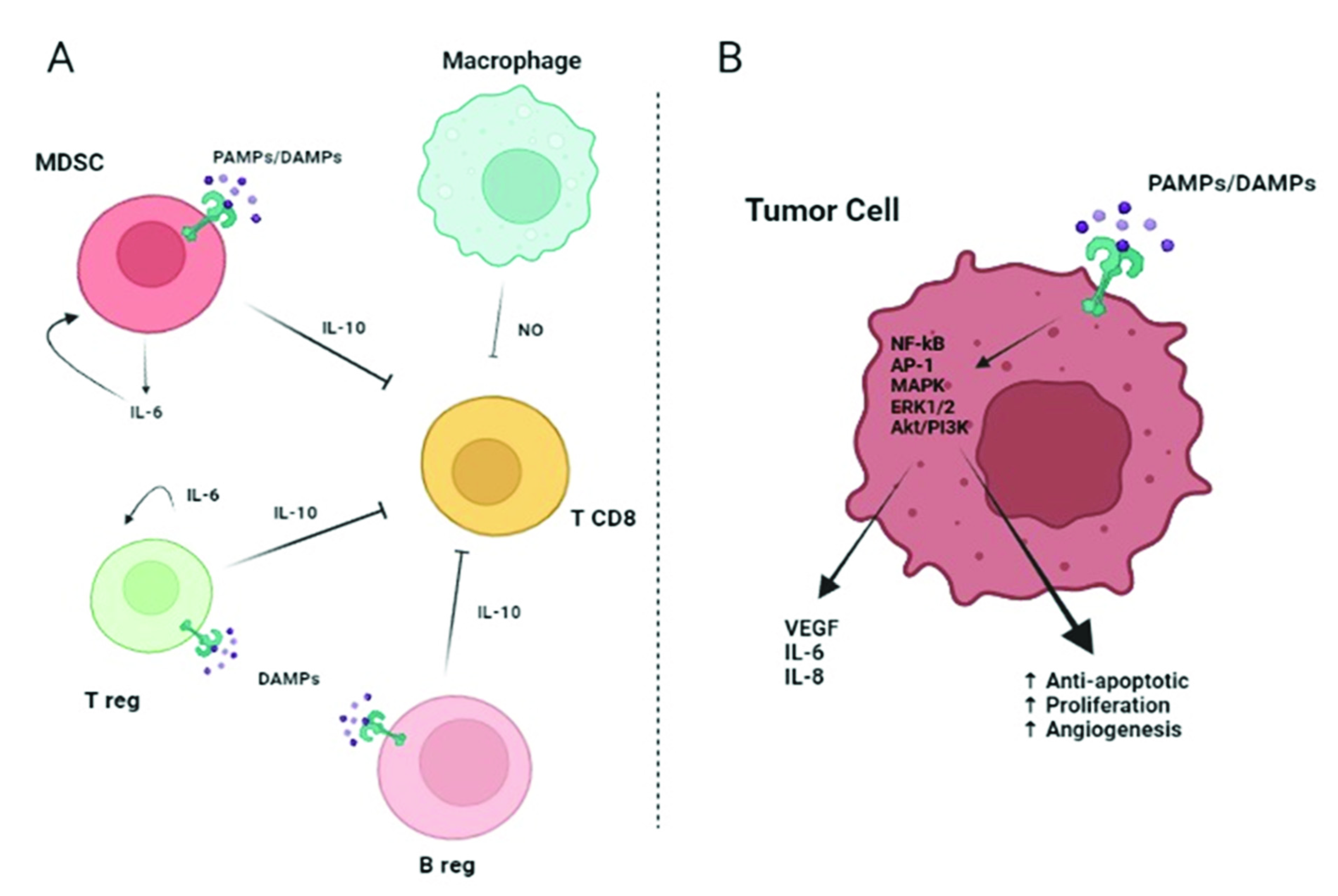
At present, approximately 18% of cancer cases are caused by infection and chronic inflammation, and patients with chronic inflammatory diseases have a higher risk of cancer development [18,23]. In the tumor cells or within the tumor microenvironment, TLRs can promote carcinogenesis through various mechanisms such as proinflammatory, prosurvival, proliferative, and immunosuppression [18]. In the tumor cells, aberrant activation of TLR might occur, thus increasing NF-κB activity, which upregulates pro-apoptotic genes and inhibits JNK-mediated pro-apoptotic signals, therefore favoring tumor survival [24]. The paracrine tumoral effects are basically mediated by the cells of the immune system via TLR-released cytokines that will suppress the T cell response, thus promoting immunoevasion by the tumor (Figure 2, Panel A). TLRs can also activate immune cells, producing a cytokine storm in response to the tumor microenvironment and to the DAMPs derived from cell or tissue damage as a result of harmful processes such as chemotherapy or radiotherapy [25]. In addition, in the context of the tumor, the cytokines released by immune cells can have a paracrine effect on the tumor cells, which causes them to secrete proteins that help keep the cytokine storm ongoing [26]. Moreover, Choi et al. highlighted that tumors associated with overexpression of TLRs result in a poor prognosis for cancer patients [27]. In fact, tumor-derived DAMP proteins may have an effect on the endothelial-to-mesenchymal transition (EMT) mechanism, which is responsible for tumor metastasis [28,29], and can also bind to TLRs on tumor cells activating signaling pathways involved in tumor migration, invasion, and metastasis [30,31]. Activation of TLRs in the tumor cell induces the production of proinflammatory cytokines IL-6 and IL-8, a strong neutrophil chemoattractant, and vascular endothelial-derived growth factor (VEGF), which is a key factor in promoting angiogenesis [32,33] (Figure 2, Panel B).
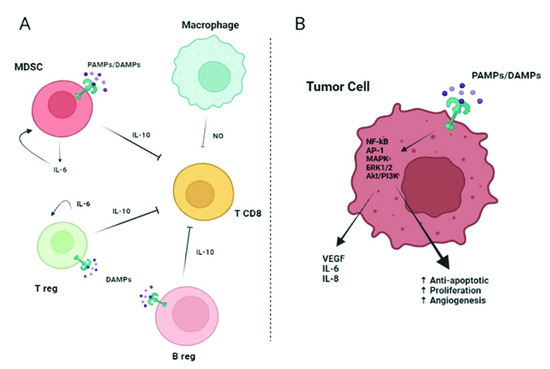
Figure 2.
(A): TLR activation on myeloid-derived suppressor cells (MDSCs), B regulatory cells (Breg), and T regulatory cells (Treg) leads to an immunosuppressive effect. Indeed, MDSCs can induce macrophages to release nitric oxide (NO) that suppresses the activity of T CD8+ lymphocytes. Moreover, activated MDSCs, Bregs, and Tregs release IL-6 and IL-10 with a consequent synergic inhibitory effect on T CD8+ cells. (B): TLR expressed on tumor cells can bind DAMPs/PAMPs and activate intracellular signaling, such as nuclear factor kappa-light-chain-enhancer of activated B cells (NF-kB), AP-1, Akt/PI3K, extracellular signal-regulated kinase (ERK)1/2, and MAPK. The consequence is a pro-tumor effect due to the release of IL-6, IL-8, and VEGF and the promotion of proliferation, angiogenesis, and protection from apoptosis. Continuous arrows indicate activation or secretion. Created with BioRender.com (accessed on 4 May 2022).
The role of IL-6 in lung cancer needs to be taken carefully. Despite the fact that the major role of IL-6 in cancer seems to be protumoral by promoting proliferation, survival, angiogenesis, metastasis, and immunoevasion [34], a new role in inducing anti-tumor immunity has begun to be explored as it appears to help promote the anti-tumoral effects of the T cells although the mechanisms are not yet fully understood [35,36].
However, besides its role in promoting tumor growth, it is known that TLRs are also capable of inducing tumor-suppressive effects, or even a dual role has been proposed for some of the TLRs depending on the context. For instance, TLR5 has been reported to exert an anti-tumoral effect due to its role in the immune response through the regulation of dendritic cells (DC), which can kill tumor cells [37]. On the other hand, some TLRs seem to play a dual role in promoting or suppressing tumor progression depending on the type of cancer; for example, TLR7/8 has been reported to mediate anti-tumor effects due to their role on DC and NK cell activation [38,39]. The role of TLRs in mediating pro- or anti-tumoral effects in the context of each cancer type is summarized in Table 1.

Table 1.
Pro- or anti-tumoral effects mediated by TLRs.
In the lung, the studies so far rather support a role for TLR signaling in promoting the growth and survival of malignant cells. Sureshbabu et al. observed that the release of TGF-β1 by lung epithelial cells facilitates both apoptosis and inflammation [40]. In addition, activation of TLR2 and TLR4 induce extracellular matrix remodeling and Epithelial Growth Factor Receptor (EGFR)-mediated signaling, respectively, stimulating lung carcinoma progression [41]. Furthermore, He et al. showed that stimulation of human lung cancer cells with functional TLR4 resulted in a secretion of immunosuppressive cytokines such as VEGF, TGF-β, and IL-8, which induced resistance to TNFα-induced apoptosis and TRAIL in vitro [42]. TLR7 has been reported to induce cell survival and tumor growth [43], most likely via NF-κB activation and upregulation of Bcl-2 [44]. Droemann et al. observed a high TLR9 expression in both primary lung cancer specimens and tumor cell lines. The activation of the TLR9 pathway resulted in the production of monocyte chemoattractant protein-1 () and the reduction in TNFα-induced apoptosis [45].
3. WNT in Lung Cancer
3.1. WNT Signaling Pathway
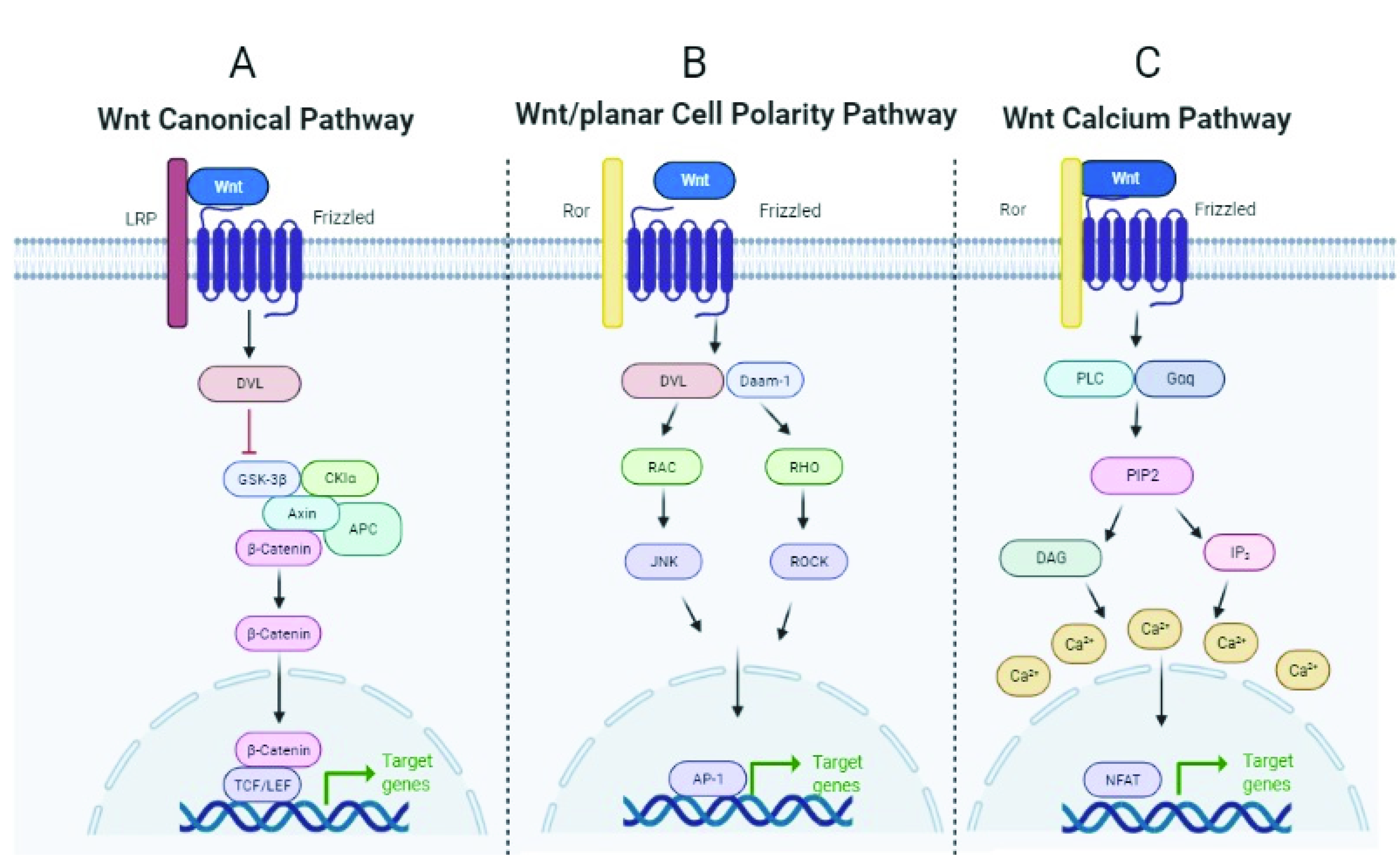
The WNT (Wingless/Integrated) pathway is a developmental pathway involved in repair and regeneration processes, among others, activated by the WNT ligands (19 in humans), which are evolutionarily conserved glycoproteins that are secreted and bind to cell surface receptors termed Frizzled (Fzd). To date, 10 Fzd receptors have been identified in mice and humans [59]. Depending on the context, WNT ligands can initiate at least three different intracellular signaling cascades: the canonical WNT/β-catenin pathway, and the non-canonical WNT/β-catenin pathway (β-catenin-independent pathway), which further divides into the planar cell polarity (PCP), and the WNT/Ca2+ pathway [60,61]. In mammalian cells, the canonical WNT/β-catenin pathway is activated when the WNT ligand binds to the Fzd receptor and the low-density lipoprotein receptor-related protein 5/6 (LRP5/6). Activation of the pathway and recruitment of Axin and associated proteins (GSK3β, CK1α, APC) to Fz-LRP5/6 in the membrane negatively regulates the β-catenin destruction complex. β-catenin can then translocate to the nucleus to act as a transcription factor for genes that encode proteins involved in cell proliferation, differentiation, and stem cell maintenance [62] (Figure 3, panel A). In the non-canonical PCP pathway, WNT ligands bind to Fzd receptors and the RAR-related orphan receptor (ROR) and signals to the cytoplasmic phosphoprotein Dishevelled (DVL), which activates Rho-associated (ROCK) or the JUN-N-terminal kinase (JNK), thus inducing gene expression by the activator protein-1 (AP-1) [63] involved in cytoskeletal rearrangement, cell motility, and coordinated cell polarity (Figure 3, panel B).
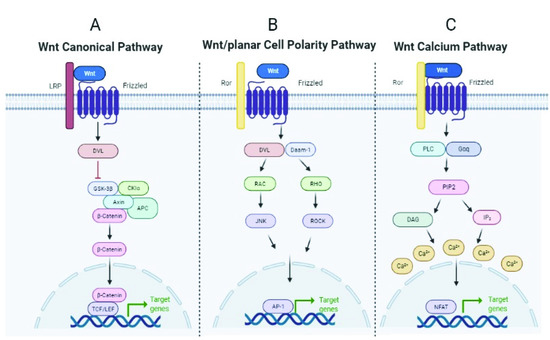
Figure 3.
Scheme of the three WNT pathways. Created by BioRender.com and modified from Koni et al. [66] and Hiremath et al. [67].
In the non-canonical WNT calcium-dependent pathway, WNT ligands bind Fzd and activate phospholipase C (PLC), which hydrolyzes certain lipids, resulting in the release of intracellular calcium and the activation of the nuclear factor of activated T cells (NFAT), which moves to the nucleus and regulates the expression of target genes involved in embryonic patterns and tissue homeostasis [63]. (Figure 3, panel C). The WNT pathway is essential for the proper development of organs and tissues, including the lungs, and also regulates key repair processes in both physiological and pathological situations [64]. Aberrant regulation of the WNT signaling pathway has been a recurrent issue in cancer research [65] due to its contribution to tumorigenesis and metastasis.
3.2. Alterations of the WNT Signaling Pathway in Lung Cancer
The WNT pathway is involved in the maintenance of progenitor stem cells in epithelial tissues, including the lung, playing a major role in mediating processes involved in homeostasis, regeneration, and repair [68,69]. Carcinomas arising from these tissues often exhibit aberrant activation of the WNT pathway by mechanisms such as mutations in APC, β-catenin, or axin [70] and, more recently, through autocrine activation of WNT [71]. At the level of WNT ligands, overexpression of WNT1, WNT3, WNT5A, and WNT11, along with the Fzd8 receptor, has been reported in NSCLC [72,73,74,75]. Moreover, high levels of WNT1, WNT5A [76], and WNT3 [73] in NSCLC patients were correlated with lower survival. Further, WNT3 was observed to promote cell invasion and independent growth of tumor cells, and WNT3A treatment of NSCLC cells facilitated the EMT process, increasing the like-hood of NSCLC metastasis [77]. In fact, overexpression of Glypican-5, which traps WNT3A competing with its Fzd receptor, prevents EMT and metastasis in lung adenocarcinoma [78]. WNT5A ligand expression is highly increased in NSCLC and has been associated with poor prognosis [79,80]. Another study reported a differential overexpression of WNT ligands depending on the NSCLC subtype since overexpression of non-canonical WNT5A was observed in squamous cell carcinomas while canonical WNT7B was found in adenocarcinomas [81]. On the other hand, WNT7A and its Fzd9 receptor were found downregulated in lung tissue from NSCLC patients, most likely preventing the activation of JNK downstream of the non-canonical WNT pathway, which maintains epithelial cell differentiation and suppresses tumor cell proliferation [82]. The O-acyltransferase Porcupine is required in WNT-producing cells for proper ligand maturation and secretion [83], and it was found overexpressed in lung adenocarcinoma tissue [84]. Of notice, WNT ligands were discovered recently to be transported through extracellular vesicles (such as exosomes) in a Porcupine-dependent manner, which are membranous vesicles that mediate intercellular communication by transporting specific cargos (i.e., proteins, lipids, and nucleic acids) between the producer and the receiver cells [85]. It is known that tumor-derived exosomes mediate important roles in tumor progression [86]; in fact, a pro-tumoral role of exosomal WNT5A has been reported in several cancers [87,88].
At the level of the WNT signaling cascade, activation of WNT/β-catenin seems to be a signature in Kras positive lung tumors that predicts the response to pharmacologic treatment and is correlated with faster disease progression [89,90]. The WNT/β-catenin pathway has also been associated with metastasis of NSCLC tumors via induction of key transcription factors (TCF, LEF1, and HOXB9) that will favor EMT-like processes [90,91].
Abnormalities in key pathway modulators were also found to induce aberrant WNT activation. For instance, loss of function mutations in Fzd inhibitors Rnf47 and Znrf3 are present in lung cancer [92], and specific mutation in Lrp6 was found to increase the risk of NSCLC in smokers [93]. Tobacco smoke was also found to induce the secretion of the WNT agonist Dkk1 producing a protumoral effect [94]. Ror1 is upregulated in lung cancer and has been associated with resistance to Tyrosine Kinase Inhibitor (TKIs) therapy in epidermal growth factor receptor (EGFR) positive NSCLC [95,96].
4. TLR/WNT Crosstalk
4.1. TLR/WNT Crosstalk in the Lung
Recent studies suggest that the WNT/β-catenin and NF-κB signaling pathways cross-regulate each of their activities and functions [97], and accumulating evidence has shown that a negative regulation of immune cells such as T regulatory cells (Treg) as well as the reprogramming of dendritic cells (DC) for immunotolerance is due to the fact that inflammatory responses are capable of activating the WNT/β-catenin pathway [98,99].
A special relationship between TLRs and WNT signaling happens through the non-canonical WNT5A ligand. WNT5A transcription is regulated by many proteins, including nuclear factor NF-κB. Katoh et al. found that within the B region of the WNT5A promoter, there was a conserved NF-κB binding site. This explains the mechanisms by which WNT5A are upregulated through TNFα and TLR signals [100]. In the last decade, Trinath et al. identified a novel role for the WNT signaling pathway by observing cross-regulation with TLRs in macrophages where the TLR proinflammatory signal is downregulated by the WNT5A ligand. In more detail, they found that upon microbial challenge, activation of dectin-1 (a receptor present in macrophages) stabilizes β-catenin and upregulates non-canonical WNT5A secretion, which mediates downregulation of MyD88 (a major effector of the TLR pathway), thus resulting in decreased expression of IL-12, IL1-β and TNF-α [101]. In fact, Mehmeti et al. discovered a novel role of WNT5A in inflammatory cells by direct binding to TLR2/4, inducing L-10 expression and immune tolerance [102]. Upon infection by mycobacteria, WNT5A was induced in antigen-presenting T cells in a TLR-NF-κB-dependent manner, where it mediates the secretion of IL-12 and IFN [103], and both WNT5A and WNT3A were found to inhibit TLR-induced secretion of proinflammatory cytokines in DC [98,104]. Another study by Neumann et al. also suggested a negative regulation of TLR by WNT signaling as they observed that proinflammatory mediators induced by TLR were increased while WNT/β-catenin signaling was decreased in macrophages following bacterial infection [98,104]. The negative regulation between both signals is also observed in lung disease. For instance, downregulation of the WNT/β-catenin pathway is found in the lung epithelium o COPD smokers, which are known to be susceptible to bacterial infection [80]. Moreover, in the context of infection by pathogens, it has been reported that the WNT/β-catenin pathway is constitutively activated in lung epithelial cells, thus mediating the recruitment of macrophages, specifically at the sites of bacterial infection [104,105]. In fact, TLR-4-MyD88 signaling is also associated with WNT5A to induce IL-12p40 and IL-6 expression in macrophages [106]. Similarly, LPS/IFN gamma induces WNT5A expression in macrophages, which involves the activation of TLR signaling and NF-κB by mediating the release of IL-1b, IL-6, IL-8, and MIP1b [107]. In contrast, in the context of viral infection, Oderup et al. found that WNT5A rather inhibits the production of IL-6 [98,104]. In addition, it was found that infected macrophages seem to undergo differentiation and proliferation in response to WNT6 [104,105]. Altogether, these studies support the role of WNT signaling in regulating the TLR inflammatory response in immune cells and in mediating immunotolerance and macrophages homing upon lung infection.
4.2. TLR/WNT in Lung Cancer
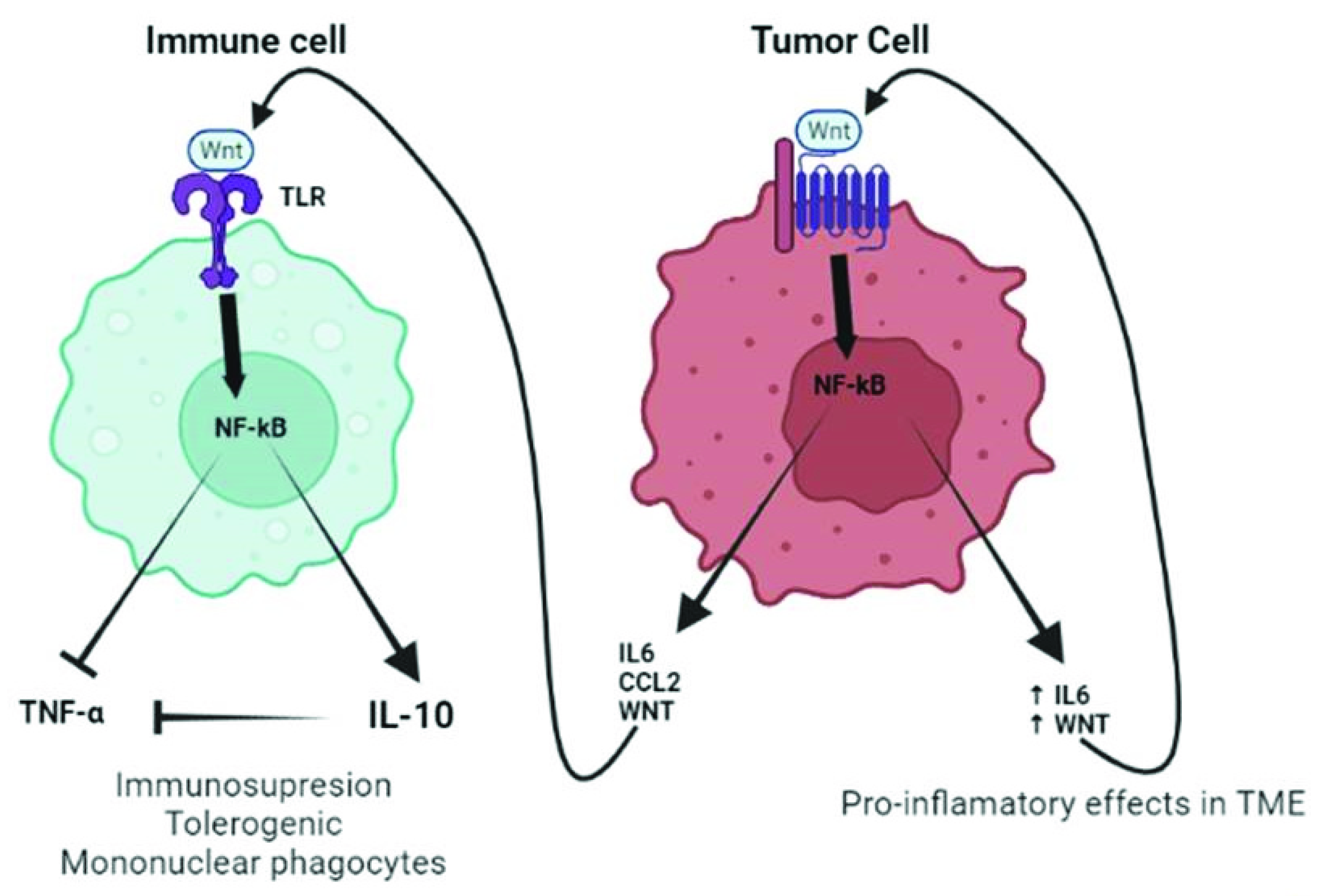
As previously mentioned, TLRs and the WNT signaling have been observed to play a role in promoting carcinogenesis and tumor cell proliferation. Increased/aberrant WNT signaling in the tumor cell usually leads to a great release of WNT ligands and proinflammatory mediators (i.e., IL-6 and MCP-1), which is sustained over time due to a self-feeding loop. These mediators will then attract the immune cells to the tumor, which are susceptible to TLR activation through direct binding with the tumor-derived WNT ligands. TLR active monocytes would then secrete IL-10, thus inducing immunosuppression, which helps tumor cells evade the immune system (Figure 4).
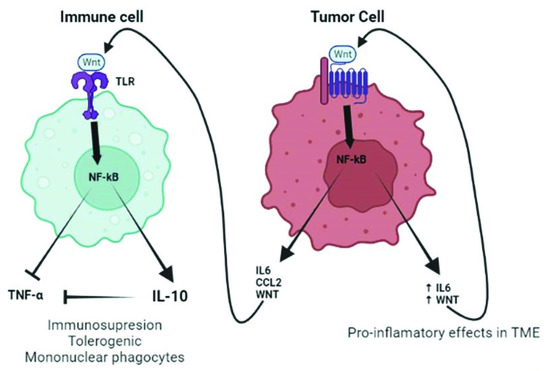
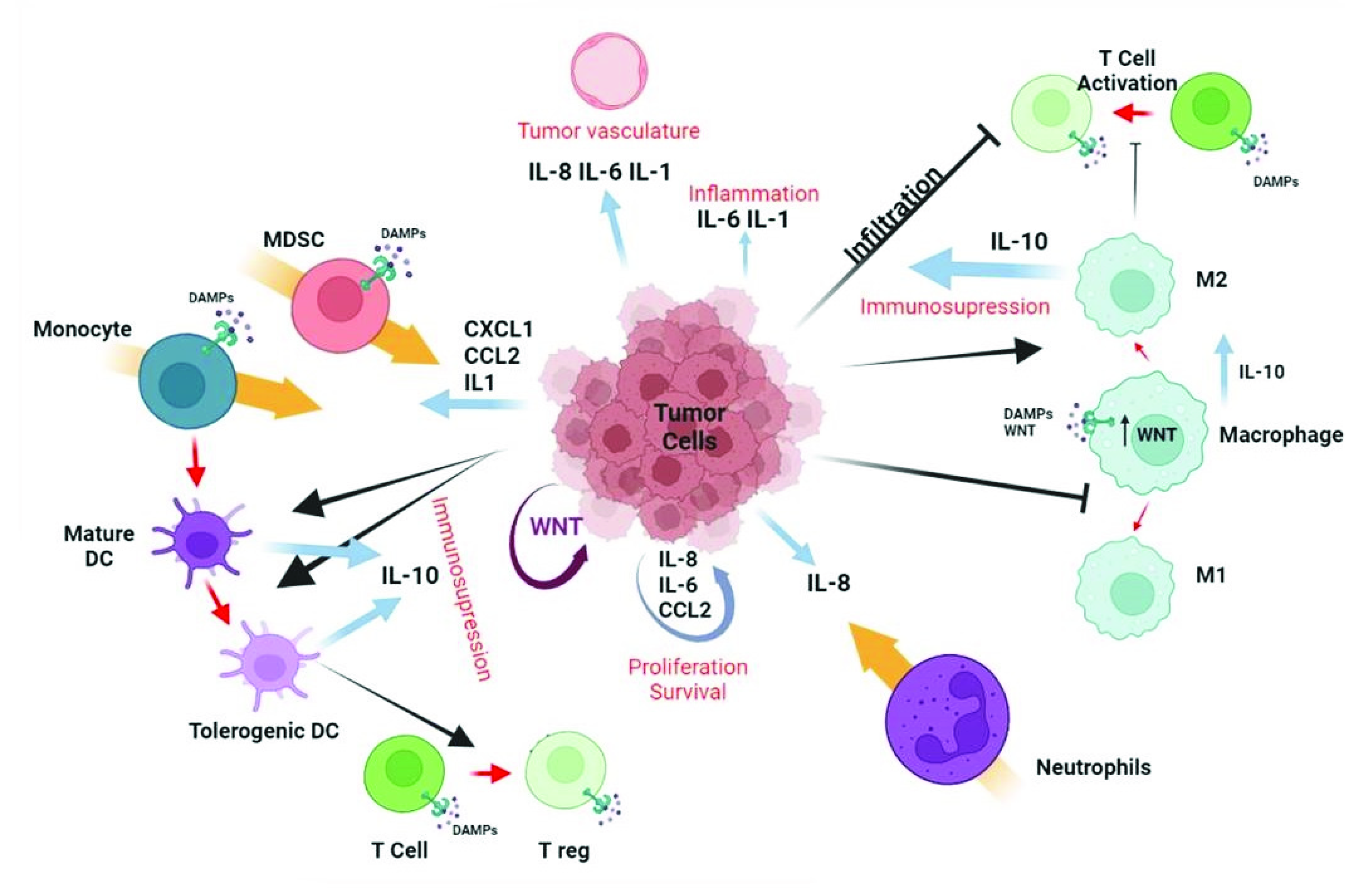
Figure 4.
Interaction between tumor-derived WNT and immune cells. Increased production of WNT by tumor cells (right part of the figure) stimulates an autocrine pathway that promotes the secretion of proinflammatory cytokines (IL-6) and chemokines (MCP-1). Both cytokines and WNT are maintained at a high level of expression by a self-feeding loop. Chemokines are critical for populating the tumor microenvironment (TME) with immune cells. The different cytokines induced by WNT, together with the WNT ligand itself, would stimulate other cells in the TME, such as tumor-associated fibroblasts or endothelial cells that further amplify the effects of WNT. At a later stage, once immune cells are recruited to the TME, WNT begins to play a different role (left part of the figure). In this new scenario, WNT activates a TLR/MyD88/p50 pathway, promoting the synthesis of the anti-inflammatory cytokine IL-10. As a result, WNT induces immunosuppression and the formation of tolerogenic mononuclear phagocytes. Created with BioRender.com (accessed on 21 March 2022).
The WNT/β-catenin pathway components modulate inflammatory and immune responses via the interaction with NF-κB and, thus, significantly influence the progression of inflammation and cancer. In this line, Li et al. found that NF-κB-dependent and smoke-induced inflammation activates numerous growth pathways in lung cancer cells, including the WNT/β-catenin signaling pathway [108]. Moreover, in a recent analysis of the correlation between the expression of WNT ligands and 23 immunosuppressive genes across all cancer types in the TCGA dataset, high levels of WNT1 were found to significantly negatively correlate with CD8 + T cells, suggesting that it induces immune resistance in lung adenocarcinoma cells [109].
The WNT non-canonical WNT5A ligand has shown important roles in both innate and adaptive immunity to infections, being associated with several inflammatory processes such as induction of proinflammatory cytokines, regulation of migration, and recruitment of various immune effector cells, and inducement of T cell differentiation [110]. In cancer, WNT5A has mainly been implicated as an oncogenic protein that is involved in the invasion and metastasis of many cancers [103,111]. Tobacco smoke is a very potent inducer of lung cancer [112], and exposure to cigarette smoke-extract induces WNT5A expression in human bronchial epithelial cells [113] and increases the expression of TLR in macrophages [114]. In the lung, WNT5A is overexpressed, correlating with tumor aggressiveness, lower survival, and the development of resistance to therapy [80,115].
It is known that patients suffering from other chronic lung diseases where WNT and/or TLR play an important role are often at higher risk of developing lung cancer. For instance, an overactivation of the non-canonical WNT signal, mostly represented by a WNT5A overexpression, has been reported in alveolar epithelial cells from COPD patients, and inhibition of WNT5A results in attenuation and amelioration of elastase-induced emphysema in mice [116]. Upregulation of TLR signaling also takes place in COPD, where it seems to contribute to prolonging the oxidant/antioxidant imbalance [117], and overactivation of TLRs often happens in COPD due to common exacerbations caused by bacterial infections [118]. Idiopathic Pulmonary Fibrosis (IPF) has been largely related to the development of lung cancer [119]. Aberrant WNT activation and overexpression of WNT ligands (WNT3A in epithelium and WNT5A in fibroblasts) is a hallmark in IFP [120,121], and TLR also plays a role in disease since there is an ongoing inflammation due to an upregulation of proinflammatory cytokines IL-1 and IL-6 in part in response to an increased WNT signaling [122]. The fact that both WNT and TLR signals coexist not only in lung cancer but also in other lung diseases related to cancer highlights the prominent role of the interaction of these two signals in tumor development.
5. Conclusions
Lung cancer is a frequent tumor worldwide and is associated with poor survival. TLRs and WNT signaling play important roles in the immune response to various inflammatory and tumorigenic processes (Figure 5). Various subtypes of TLRs and WNT signaling members have been involved in tumor cell proliferation, tumor invasion, metastasis, and poor survival, especially in NSCLC. In the context of lung cancer, autocrine WNT/TLR promotes tumor growth and survival and further recruits monocytes and mediates immunosuppressive effects that support tumor progression. More research is still required to assess the role of TLRs and WNT pathways in lung cancer and explore the possibility of dual therapeutic options.
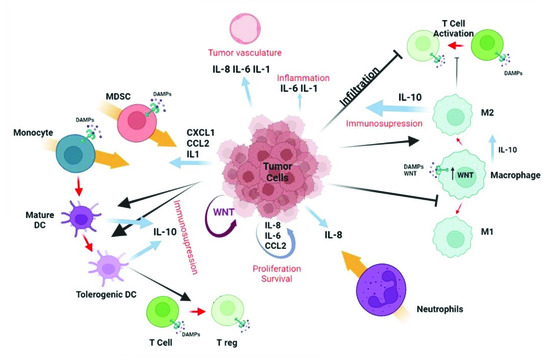
Figure 5.
Immunomodulatory effects of WNT on the tumor microenvironment. The autocrine effect of WNT (garnet arrow) drives the secretion of various cytokines and chemokines by tumor cells (blue arrows), which in turn promote different processes (in red) and chemotaxis of various cell types (orange arrow). WNT also promotes IL-10 secretion by dendritic cells (DC) and macrophages. WNT released by tumor cells promotes (black lines with arrowheads) or inhibits (black lines) maturation and/or differentiation (red arrows) of immune cells. The effects associated with WNT are angiogenesis, inflammation, and immunosuppression, as well as proliferation and survival of tumor cells. Created with BioRender.com (accessed on 21 March 2022).
Author Contributions
Conceptualization, A.M.-M. and A.I.; funding acquisition, A.I.; investigation, A.I., A.M.-M., H.S., E.M.-F. and N.C.-P.; methodology, A.I., A.M.-M., H.S., E.M.-F. and N.C.-P.; project administration, A.I.; resources, A.I., A.M.-M., H.S., E.M.-F. and N.C.-P.; supervision, A.I.; writing—original draft preparation, A.I., A.M.-M., H.S., E.M.-F. and N.C.-P.; writing—review and editing, A.I., A.M.-M., H.S., E.M.-F., A.O.-H., N.C.-P. and J.S. All authors have read and agreed to the published version of the manuscript.
Funding
The APC was funded by 1046-2020 SEPAR (Sociedad Española de Neumología y Cirugía Torácica).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Lemjabbar-Alaoui, H.; Hassan, O.U.; Yang, Y.-W.; Buchanan, P. Lung cancer: Biology and treatment options. Biochim. Biophys. Acta 2015, 1856, 189–210. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Morgensztern, D.; Boshoff, C. The biology and management of non-small cell lung cancer. Nature 2018, 553, 446–454. [Google Scholar] [CrossRef]
- Travis, W.D. Update on small cell carcinoma and its differentiation from squamous cell carcinoma and other non-small cell carcinomas. Mod. Pathol. 2012, 25, S18–S30. [Google Scholar] [CrossRef]
- Li, X.; Jiang, S.; Tapping, R.I. Toll-like receptor signaling in cell proliferation and survival. Cytokine 2010, 49, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Balkwill, F.; Mantovani, A. Inflammation and cancer: Back to Virchow? Lancet 2001, 357, 539–545. [Google Scholar] [CrossRef]
- Martin, T.R.; Frevert, C.W. Innate Immunity in the Lungs. Proc. Am. Thorac. Soc. 2005, 2, 403–411. [Google Scholar] [CrossRef]
- Takeda, K.; Akira, S. Toll-Like Receptors. Curr. Protoc. Immunol. 2015, 109, 14.12.1–14.12.10. [Google Scholar] [CrossRef]
- Menck, K.; Heinrichs, S.; Baden, C.; Bleckmann, A. The WNT/ROR Pathway in Cancer: From Signaling to Therapeutic Intervention. Cells 2021, 10, 142. [Google Scholar] [CrossRef]
- Roach, J.C.; Glusman, G.; Rowen, L.; Kaur, A.; Purcell, M.K.; Smith, K.D.; Hood, L.E.; Aderem, A. The evolution of vertebrate Toll-like receptors. Proc. Natl. Acad. Sci. USA 2005, 102, 9577–9582. [Google Scholar] [CrossRef]
- Droemann, D.; Goldmann, T.; Tiedje, T.; Zabel, P.; Dalhoff, K.; Schaaf, B. Toll-like receptor 2 expression is decreased on alveolar macrophages in cigarette smokers and COPD patients. Respir. Res. 2005, 6, 68. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, M.; Naito, S. Tissue-Specific mRNA Expression Profiles of Human Toll-Like Receptors and Related Genes. Biol. Pharm. Bull. 2005, 28, 886–892. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.; Gerondakis, S. Coordinating TLR-activated signaling pathways in cells of the immune system. Immunol. Cell Biol. 2007, 85, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H. Endogenous TLR Ligands and Autoimmunity. Adv. Immunol. 2006, 91, 159–173. [Google Scholar] [CrossRef]
- Rakoff-Nahoum, S.; Paglino, J.; Eslami-Varzaneh, F.; Edberg, S.; Medzhitov, R. Recognition of Commensal Microflora by Toll-Like Receptors Is Required for Intestinal Homeostasis. Cell 2004, 118, 229–241. [Google Scholar] [CrossRef]
- Hasan, U.A.; Trinchieri, G.; Vlach, J. Toll-like Receptor Signaling Stimulates Cell Cycle Entry and Progression in Fibroblasts. J. Biol. Chem. 2005, 280, 20620–20627. [Google Scholar] [CrossRef]
- Jego, G.; Bataille, R.; Geffroyluseau, A.; Descamps, G.; Deceunynck, C. Pathogen-associated molecular patterns are growth and survival factors for human myeloma cells through Toll-like receptors. Leukemia 2006, 20, 1130–1137. [Google Scholar] [CrossRef]
- Pradere, J.-P.; Dapito, D.H.; Schwabe, R.F. The Yin and Yang of Toll-like receptors in cancer. Oncogene 2013, 33, 3485–3495. [Google Scholar] [CrossRef]
- Huang, B.; Zhao, J.; Unkeless, J.C.; Feng, Z.H.; Xiong, H. TLR signaling by tumor and immune cells: A double-edged sword. Oncogene 2008, 27, 218–224. [Google Scholar] [CrossRef]
- Igney, F.H.; Krammer, P.H. Immune escape of tumors: Apoptosis resistance and tumor counterattack. J. Leukoc. Biol. 2002, 71, 907–920. [Google Scholar]
- Colotta, F.; Allavena, P.; Sica, A.; Garlanda, C.; Mantovani, A. Cancer-related inflammation, the seventh hallmark of cancer: Links to genetic instability. Carcinogenesis 2009, 30, 1073–1081. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Mohan, C. Toll-Like Receptor Signaling Pathways—Therapeutic Opportunities. Mediat. Inflamm. 2010, 2010, 781235. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, H.; Nair, J. Chronic inflammation and oxidative stress in the genesis and perpetuation of cancer: Role of lipid peroxidation, DNA damage, and repair. Langenbeck’s Arch. Surg. 2006, 391, 499–510. [Google Scholar] [CrossRef]
- Dutta, J.; Fan, Y.; Gupta, N.; Fan, G.; Gélinas, C. Current insights into the regulation of programmed cell death by NF-κB. Oncogene 2006, 25, 6800–6816. [Google Scholar] [CrossRef]
- Patidar, A.; Selvaraj, S.; Sarode, A.; Chauhan, P.; Chattopadhyay, D.; Saha, B. DAMP-TLR-cytokine axis dictates the fate of tumor. Cytokine 2018, 104, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Jang, G.-Y.; Lee, J.W.; Kim, Y.S.; Lee, S.E.; Han, H.D.; Hong, K.-J.; Kang, T.H.; Park, Y.-M. Interactions between tumor-derived proteins and Toll-like receptors. Exp. Mol. Med. 2020, 52, 1926–1935. [Google Scholar] [CrossRef]
- Choi, C.H.; Kang, T.H.; Song, J.S.; Kim, Y.S.; Chung, E.J.; Ylaya, K.; Kim, S.; Koh, S.S.; Chung, J.-Y.; Kim, J.-H.; et al. Elevated expression of pancreatic adenocarcinoma upregulated factor (PAUF) is associated with poor prognosis and chemoresistance in epithelial ovarian cancer. Sci. Rep. 2018, 8, 12161. [Google Scholar] [CrossRef]
- Srikrishna, G.; Freeze, H.H. Endogenous Damage-Associated Molecular Pattern Molecules at the Crossroads of Inflammation and Cancer. Neoplasia 2009, 11, 615–628. [Google Scholar] [CrossRef]
- Zhang, J.; Shao, S.; Han, D.; Xu, Y.; Jiao, D.; Wu, J.; Yang, F.; Ge, Y.; Shi, S.; Li, Y.; et al. High mobility group box 1 promotes the epithelial-to-mesenchymal transition in prostate cancer PC3 cells via the RAGE/NF-κB signaling pathway. Int. J. Oncol. 2018, 53, 659–671. [Google Scholar] [CrossRef]
- Kelly, M.G.; Alvero, A.B.; Chen, R.; Silasi, D.-A.; Abrahams, V.M.; Chan, S.; Visintin, I.; Rutherford, T.; Mor, G. TLR-4 Signaling Promotes Tumor Growth and Paclitaxel Chemoresistance in Ovarian Cancer. Cancer Res. 2006, 66, 3859–3868. [Google Scholar] [CrossRef]
- Park, H.D.; Lee, Y.; Oh, Y.K.; Jung, J.G.; Park, Y.W.; Myung, K.; Kim, K.-H.; Koh, S.S.; Lim, D.-S. Pancreatic adenocarcinoma upregulated factor promotes metastasis by regulating TLR/CXCR4 activation. Oncogene 2011, 30, 201–211. [Google Scholar] [CrossRef] [PubMed]
- Kluwe, J.; Mencin, A.; Schwabe, R.F. Toll-like receptors, wound healing, and carcinogenesis. J. Mol. Med. 2009, 87, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, R.; De Souza, P.M.; Sriskandan, S.; Duffin, C.; Paul-Clark, M.J.; Mitchell, J.A. Pattern recognition receptors and interleukin-8 mediate effects of Gram-positive and Gram-negative bacteria on lung epithelial cell function. Br. J. Pharmacol. 2008, 154, 864–871. [Google Scholar] [CrossRef] [PubMed]
- Fisher, D.T.; Appenheimer, M.M.; Evans, S.S. The two faces of IL-6 in the tumor microenvironment. Semin. Immunol. 2014, 26, 38–47. [Google Scholar] [CrossRef]
- Angell, H.; Galon, J. From the immune contexture to the Immunoscore: The role of prognostic and predictive immune markers in cancer. Curr. Opin. Immunol. 2013, 25, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Fridman, W.H.; Pagès, F.; Sautes-Fridman, C.; Galon, J. The immune contexture in human tumours: Impact on clinical outcome. Nat. Rev. Cancer 2012, 12, 298–306. [Google Scholar] [CrossRef]
- Garaude, J.; Kent, A.; van Rooijen, N.; Blander, J.M. Simultaneous Targeting of Toll- and Nod-Like Receptors Induces Effective Tumor-Specific Immune Responses. Sci. Transl. Med. 2012, 4, 120ra16. [Google Scholar] [CrossRef]
- Drobits, B.; Holcmann, M.; Amberg, N.; Swiecki, M.; Grundtner, R.; Hammer, M.; Colonna, M.; Sibilia, M. Imiquimod clears tumors in mice independent of adaptive immunity by converting pDCs into tumor-killing effector cells. J. Clin. Investig. 2012, 122, 575–585. [Google Scholar] [CrossRef]
- Peng, G.; Guo, Z.; Kiniwa, Y.; Voo, K.S.; Peng, W.; Fu, T.; Wang, D.Y.; Li, Y.; Wang, H.Y.; Wang, R.-F. Toll-Like Receptor 8-Mediated Reversal of CD4+ Regulatory T Cell Function. Science 2005, 309, 1380–1384. [Google Scholar] [CrossRef]
- Sureshbabu, A.; Syed, M.A.; Boddupalli, C.S.; Dhodapkar, M.V.; Homer, R.J.; Minoo, P.; Bhandari, V. Conditional overexpression of TGFβ1 promotes pulmonary inflammation, apoptosis and mortality via TGFβR2 in the developing mouse lung. Respir. Res. 2015, 16, 4. [Google Scholar] [CrossRef]
- Arora, S.; Ahmad, S.; Irshad, R.; Goyal, Y.; Rafat, S.; Siddiqui, N.; Dev, K.; Husain, M.; Ali, S.; Mohan, A.; et al. TLRs in pulmonary diseases. Life Sci. 2019, 233, 116671. [Google Scholar] [CrossRef] [PubMed]
- He, W.; Liu, Q.; Wang, L.; Chen, W.; Li, N.; Cao, X. TLR4 signaling promotes immune escape of human lung cancer cells by inducing immunosuppressive cytokines and apoptosis resistance. Mol. Immunol. 2007, 44, 2850–2859. [Google Scholar] [CrossRef] [PubMed]
- Fabbri, M.; Paone, A.; Calore, F.; Galli, R.; Gaudio, E.; Santhanam, R.; Lovat, F.; Fadda, P.; Mao, C.; Nuovo, G.J.; et al. MicroRNAs bind to Toll-like receptors to induce prometastatic inflammatory response. Proc. Natl. Acad. Sci. USA 2012, 109, E2110–E2116. [Google Scholar] [CrossRef] [PubMed]
- Cherfils-Vicini, J.; Platonova, S.; Gillard, M.; Laurans, L.; Validire, P.; Caliandro, R.; Magdeleinat, P.; Mami-Chouaib, F.; Dieu-Nosjean, M.-C.; Fridman, W.H.; et al. Triggering of TLR7 and TLR8 expressed by human lung cancer cells induces cell survival and chemoresistance. J. Clin. Investig. 2010, 120, 1285–1297. [Google Scholar] [CrossRef] [PubMed]
- Droemann, D.; Albrecht, D.; Gerdes, J.; Ulmer, A.J.; Branscheid, D.; Vollmer, E.; Dalhoff, K.; Zabel, P.; Goldmann, T. Human lung cancer cells express functionally active Toll-like receptor 9. Respir. Res. 2005, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Grivennikov, S.I.; Karin, M. Inflammatory cytokines in cancer: Tumour necrosis factor and interleukin 6 take the stage. Ann. Rheum. Dis. 2011, 70, i104–i108. [Google Scholar] [CrossRef]
- Tye, H.; Kennedy, C.L.; Najdovska, M.; McLeod, L.; McCormack, W.; Hughes, N.; Dev, A.; Sievert, W.; Ooi, C.-H.; Ishikawa, T.-O.; et al. STAT3-Driven Upregulation of TLR2 Promotes Gastric Tumorigenesis Independent of Tumor Inflammation. Cancer Cell 2012, 22, 466–478. [Google Scholar] [CrossRef]
- Kim, S.; Takahashi, H.; Lin, W.W.; Descargues, P.; Grivennikov, S.; Kim, Y.; Luo, J.-L.; Karin, M. Carcinoma-produced factors activate myeloid cells through TLR2 to stimulate metastasis. Nature 2009, 457, 102–106. [Google Scholar] [CrossRef]
- Jia, D.; Yang, W.; Li, L.; Liu, H.; Tan, Y.; Ooi, S.; Chi, L.; Filion, L.G.; Figeys, D.; Wang, L. β-Catenin and NF-κB co-activation triggered by TLR3 stimulation facilitates stem cell-like phenotypes in breast cancer. Cell Death Differ. 2015, 22, 298–310. [Google Scholar] [CrossRef]
- Dapito, D.H.; Mencin, A.; Gwak, G.-Y.; Pradère, J.-P.; Jang, M.-K.; Mederacke, I.; Caviglia, J.M.; Khiabanian, H.; Adeyemi, A.; Bataller, R.; et al. Promotion of Hepatocellular Carcinoma by the Intestinal Microbiota and TLR4. Cancer Cell 2012, 21, 504–516. [Google Scholar] [CrossRef]
- Fukata, M.; Hernandez, Y.; Conduah, D.; Cohen, J.; Chen, A.; Breglio, K.; Goo, T.; Hsu, D.; Xu, R.; Abreu, M.T. Innate immune signaling by Toll-like receptor-4 (TLR4) shapes the inflammatory microenvironment in colitis-associated tumors. Inflamm. Bowel Dis. 2009, 15, 997–1006. [Google Scholar] [CrossRef] [PubMed]
- Mittal, D.; Saccheri, F.; Venereau, E.; Pusterla, T.; Bianchi, M.E.; Rescigno, M. TLR4-mediated skin carcinogenesis is dependent on immune and radioresistant cells. EMBO J. 2010, 29, 2242–2252. [Google Scholar] [CrossRef] [PubMed]
- Ochi, A.; Graffeo, C.S.; Zambirinis, C.P.; Rehman, A.; Hackman, M.; Fallon, N.; Barilla, R.; Henning, J.R.; Jamal, M.; Rao, R.; et al. Toll-like receptor 7 regulates pancreatic carcinogenesis in mice and humans. J. Clin. Investig. 2012, 122, 4118–4129. [Google Scholar] [CrossRef] [PubMed]
- Di Lorenzo, A.; Bolli, E.; Tarone, L.; Cavallo, F.; Conti, L. Toll-Like Receptor 2 at the Crossroad between Cancer Cells, the Immune System, and the Microbiota. Int. J. Mol. Sci. 2020, 21, E9418. [Google Scholar] [CrossRef]
- Chin, A.I.; Miyahira, A.K.; Covarrubias, A.; Teague, J.; Guo, B.; Dempsey, P.W.; Cheng, G. Toll-like Receptor 3–Mediated Suppression of TRAMP Prostate Cancer Shows the Critical Role of Type I Interferons in Tumor Immune Surveillance. Cancer Res. 2010, 70, 2595–2603. [Google Scholar] [CrossRef]
- Chew, V.; Tow, C.; Huang, C.; Bard-Chapeau, E.; Copeland, N.G.; Jenkins, N.A.; Weber, A.; Lim, K.H.; Toh, H.C.; Heikenwalder, M.; et al. Toll-Like Receptor 3 Expressing Tumor Parenchyma and Infiltrating Natural Killer Cells in Hepatocellular Carcinoma Patients. JNCI J. Natl. Cancer Inst. 2012, 104, 1796–1807. [Google Scholar] [CrossRef] [PubMed]
- Cai, Z.; Sanchez, A.; Shi, Z.; Zhang, T.; Liu, M.; Zhang, D. Activation of Toll-like Receptor 5 on Breast Cancer Cells by Flagellin Suppresses Cell Proliferation and Tumor Growth. Cancer Res. 2011, 71, 2466–2475. [Google Scholar] [CrossRef]
- Rhee, S.H.; Im, E.; Pothoulakis, C. Toll-Like Receptor 5 Engagement Modulates Tumor Development and Growth in a Mouse Xenograft Model of Human Colon Cancer. Gastroenterology 2008, 135, 518–528. [Google Scholar] [CrossRef]
- He, X.; Semenov, M.; Tamai, K.; Zeng, X. LDL receptor-related proteins 5 and 6 in Wnt/beta-catenin signaling: Arrows point the way. Development 2004, 131, 1663–1677. [Google Scholar] [CrossRef]
- Macdonald, B.T.; Tamai, K.; He, X. Wnt/β-Catenin Signaling: Components, Mechanisms, and Diseases. Dev. Cell 2009, 17, 9–26. [Google Scholar] [CrossRef]
- Van Amerongen, R.; Nusse, R. Towards an integrated view of Wnt signaling in development. Development 2009, 136, 3205–3214. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xiao, Q.; Xiao, J.; Niu, C.; Li, Y.; Zhang, X.; Zhou, Z.; Shu, G.; Yin, G. Wnt/β-catenin signalling: Function, biological mechanisms, and therapeutic opportunities. Signal Transduct. Target. Ther. 2022, 7, 3. [Google Scholar] [CrossRef] [PubMed]
- Gajos-Michniewicz, A.; Czyz, M. WNT Signaling in Melanoma. Int. J. Mol. Sci. 2020, 21, 4852. [Google Scholar] [CrossRef]
- Kotton, D.N.; Morrisey, E.E. Lung regeneration: Mechanisms, applications and emerging stem cell populations. Nat. Med. 2014, 20, 822–832. [Google Scholar] [CrossRef]
- Anastas, J.N.; Moon, R.T. WNT signalling pathways as therapeutic targets in cancer. Nat. Rev. Cancer 2013, 13, 11–26. [Google Scholar] [CrossRef]
- Koni, M.; Pinnarò, V.; Brizzi, M.F. The Wnt Signalling Pathway: A Tailored Target in Cancer. Int. J. Mol. Sci. 2020, 21, 7697. [Google Scholar] [CrossRef] [PubMed]
- Hiremath, I.S.; Goel, A.; Warrier, S.; Kumar, A.P.; Sethi, G.; Garg, M. The multidimensional role of the Wnt/β-catenin signaling pathway in human malignancies. J. Cell. Physiol. 2022, 237, 199–238. [Google Scholar] [CrossRef]
- Radtke, F.; Clevers, H. Self-Renewal and Cancer of the Gut: Two Sides of a Coin. Science 2005, 307, 1904–1909. [Google Scholar] [CrossRef]
- Aros, C.J.; Pantoja, C.J.; Gomperts, B.N. Wnt signaling in lung development, regeneration, and disease progression. Commun. Biol. 2021, 4, 601. [Google Scholar] [CrossRef]
- Giles, R.H.; van Es, J.H.; Clevers, H. Caught up in a Wnt storm: Wnt signaling in cancer. Biochim. Biophys. Acta 2003, 1653, 1–24. [Google Scholar] [CrossRef]
- Akiri, G.; Cherian, M.M.; Vijayakumar, S.; Liu, G.; Bafico, A.; Aaronson, S.A. Wnt pathway aberrations including autocrine Wnt activation occur at high frequency in human non-small-cell lung carcinoma. Oncogene 2009, 28, 2163–2172. [Google Scholar] [CrossRef] [PubMed]
- Rapp, J.; Kiss, E.; Meggyes, M.; Szabo-Meleg, E.; Feller, D.; Smuk, G.; Laszlo, T.; Sarosi, V.; Molnar, T.F.; Kvell, K.; et al. Increased Wnt5a in squamous cell lung carcinoma inhibits endothelial cell motility. BMC Cancer 2016, 16, 915. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, N.; Liu, D.; Huang, C.-L.; Ueno, M.; Zhang, X.; Yokomise, H. Wnt3 gene expression promotes tumor progression in non-small cell lung cancer. Lung Cancer 2012, 76, 228–234. [Google Scholar] [CrossRef]
- Stewart, D.J. Wnt Signaling Pathway in Non-Small Cell Lung Cancer. JNCI J. Natl. Cancer Inst. 2014, 106, djt356. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.; Ma, R.; Xu, Y.; Li, N.; Li, Z.; Yue, J.; Li, H.; Guo, Y.; Qi, D. Wnt2 promotes non-small cell lung cancer progression by activating WNT/β-catenin pathway. Am. J. Cancer Res. 2015, 5, 1032–1046. [Google Scholar]
- Jin, J.; Zhan, P.; Qian, H.; Wang, X.; Katoh, M.; Phan, K.; Chung, J.-H.; Lv, T.; Song, Y.; Written on behalf of the AME Lung Cancer Collaborative Group. Prognostic value of wingless-type proteins in non-small cell lung cancer patients: A meta-analysis. Transl. Lung Cancer Res. 2016, 5, 436–442. [Google Scholar] [CrossRef]
- Li, C.; Song, G.; Zhang, S.; Wang, E.; Cui, Z. Wnt3a Increases the Metastatic Potential of Non-Small Cell Lung Cancer Cells in Vitro in Part via Its Upregulation of Notch3. Oncol. Rep. 2015, 33, 1207–1214. [Google Scholar] [CrossRef]
- Wang, S.; Qiu, M.; Xia, W.; Xu, Y.; Mao, Q.; Wang, J.; Dong, G.; Xu, L.; Yang, X.; Yin, R. Glypican-5 suppresses Epithelial-Mesenchymal Transition of the lung adenocarcinoma by competitively binding to Wnt3a. Oncotarget 2016, 7, 79736–79746. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, G.; Zhang, B.; Xu, G.; Xiong, W.; Yang, H. Wnt-5a regulates proliferation in lung cancer cells. Oncol. Rep. 2010, 23, 177–181. [Google Scholar] [CrossRef]
- Huang, C.-L.; Liu, D.; Nakano, J.; Ishikawa, S.; Kontani, K.; Yokomise, H.; Ueno, M. Wnt5a Expression Is Associated With the Tumor Proliferation and the Stromal Vascular Endothelial Growth Factor—An Expression in Non–Small-Cell Lung Cancer. J. Clin. Oncol. 2005, 23, 8765–8773. [Google Scholar] [CrossRef]
- Vesel, M.; Rapp, J.; Feller, D.; Kiss, E.; Jaromi, L.; Meggyes, M.; Miskei, G.; Duga, B.; Smuk, G.; Laszlo, T.; et al. ABCB1 and ABCG2 drug transporters are differentially expressed in non-small cell lung cancers (NSCLC) and expression is modified by cisplatin treatment via altered Wnt signaling. Respir. Res. 2017, 18, 52. [Google Scholar] [CrossRef]
- Winn, R.A.; Marek, L.; Han, S.-Y.; Rodriguez, K.; Rodriguez, N.; Hammond, M.; Van Scoyk, M.; Acosta, H.; Mirus, J.; Barry, N.; et al. Restoration of Wnt-7a Expression Reverses Non-small Cell Lung Cancer Cellular Transformation through Frizzled-9-mediated Growth Inhibition and Promotion of Cell Differentiation. J. Biol. Chem. 2005, 280, 19625–19634. [Google Scholar] [CrossRef] [PubMed]
- Clevers, H.; Loh, K.M.; Nusse, R. Stem Cell Signaling. An integral program for tissue renewal and regeneration: Wnt signaling and stem cell control. Science 2014, 346, 1248012. [Google Scholar] [CrossRef] [PubMed]
- Tammela, T.; Sánchez-Rivera, F.J.; Cetinbas, N.M.; Wu, K.; Joshi, N.; Helenius, K.; Park, Y.; Azimi, R.; Kerper, N.R.; Wesselhoeft, R.A.; et al. A Wnt-producing niche drives proliferative potential and progression in lung adenocarcinoma. Nature 2017, 545, 355–359. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367, eaau6977. [Google Scholar] [CrossRef] [PubMed]
- Fujita, Y.; Kosaka, N.; Araya, J.; Kuwano, K.; Ochiya, T. Extracellular vesicles in lung microenvironment and pathogenesis. Trends Mol. Med. 2015, 21, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Shamskhou, E.A.; Orcholski, M.E.; Nathan, A.; Reddy, S.; Honda, H.; Mani, V.; Zeng, Y.; Ozen, M.O.; Wang, L.; et al. Loss of Endothelium-Derived Wnt5a Is Associated With Reduced Pericyte Recruitment and Small Vessel Loss in Pulmonary Arterial Hypertension. Circulation 2019, 139, 1710–1724. [Google Scholar] [CrossRef] [PubMed]
- Harada, T.; Yamamoto, H.; Kishida, S.; Kishida, M.; Awada, C.; Takao, T.; Kikuchi, A. Wnt5b-associated exosomes promote cancer cell migration and proliferation. Cancer Sci. 2017, 108, 42–52. [Google Scholar] [CrossRef] [PubMed]
- Yin, N.; Liu, Y.; Khoor, A.; Wang, X.; Thompson, E.A.; Leitges, M.; Justilien, V.; Weems, C.; Murray, N.R.; Fields, A.P. Protein Kinase Cι and Wnt/β-Catenin Signaling: Alternative Pathways to Kras/Trp53-Driven Lung Adenocarcinoma. Cancer Cell 2019, 36, 156–167. [Google Scholar] [CrossRef]
- Pacheco-Pinedo, E.C.; Durham, A.C.; Stewart, K.M.; Goss, A.M.; Lu, M.M.; DeMayo, F.J.; Morrisey, E.E. Wnt/β-catenin signaling accelerates mouse lung tumorigenesis by imposing an embryonic distal progenitor phenotype on lung epithelium. J. Clin. Investig. 2011, 121, 1935–1945. [Google Scholar] [CrossRef]
- Nguyen, D.X.; Chiang, A.C.; Zhang, X.H.-F.; Kim, J.Y.; Kris, M.G.; Ladanyi, M.; Gerald, W.L.; Massagué, J. WNT/TCF Signaling through LEF1 and HOXB9 Mediates Lung Adenocarcinoma Metastasis. Cell 2009, 138, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Tu, K.; Xia, L.; Luo, K.; Luo, W.; Tang, J.; Lu, K.; Hu, X.; He, Y.; Qiao, W.; et al. The Open Chromatin Landscape of Non–Small Cell Lung Carcinoma. Cancer Res. 2019, 79, 4840–4854. [Google Scholar] [CrossRef] [PubMed]
- Deng, D.; Zhang, Y.; Bao, W.; Kong, X. Low-Density Lipoprotein Receptor-Related Protein 6 (LRP6) rs10845498 Polymorphism Is Associated with a Decreased Risk of Non-Small Cell Lung Cancer. Int. J. Med. Sci. 2014, 11, 685–690. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Rao, M.; Humphries, A.E.; Hong, J.A.; Liu, F.; Yang, M.; Caragacianu, D.; Schrump, D.S. Tobacco Smoke Induces Polycomb-Mediated Repression of Dickkopf-1 in Lung Cancer Cells. Cancer Res. 2009, 69, 3570–3578. [Google Scholar] [CrossRef] [PubMed]
- Hojjat-Farsangi, M.; Moshfegh, A.; Daneshmanesh, A.H.; Khan, A.S.; Mikaelsson, E.; Österborg, A.; Mellstedt, H. The receptor tyrosine kinase ROR1—An oncofetal antigen for targeted cancer therapy. Semin. Cancer Biol. 2014, 29, 21–31. [Google Scholar] [CrossRef]
- Khaledian, B.; Taguchi, A.; Shin-Ya, K.; Kondo-Ida, L.; Kagaya, N.; Suzuki, M.; Kajino, T.; Yamaguchi, T.; Shimada, Y.; Takahashi, T. Inhibition of heat shock protein 90 destabilizes receptor tyrosine kinase ROR1 in lung adenocarcinoma. Cancer Sci. 2021, 112, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Hottiger, M.O. Crosstalk between Wnt/β-Catenin and NF-κB Signaling Pathway during Inflammation. Front. Immunol. 2016, 7, 378. [Google Scholar] [CrossRef]
- Oderup, C.; LaJevic, M.; Butcher, E.C. Canonical and Noncanonical Wnt Proteins Program Dendritic Cell Responses for Tolerance. J. Immunol. 2013, 190, 6126–6134. [Google Scholar] [CrossRef]
- van Loosdregt, J.; Fleskens, V.; Tiemessen, M.M.; Mokry, M.; van Boxtel, R.; Meerding, J.; Pals, C.E.G.M.; Kurek, D.; Baert, M.R.M.; Delemarre, E.M.; et al. Canonical Wnt Signaling Negatively Modulates Regulatory T Cell Function. Immunity 2013, 39, 298–310. [Google Scholar] [CrossRef]
- Katoh, M. Transcriptional mechanisms of WNT5A based on NF-κB, Hedgehog, TGFβ, and Notch signaling cascades. Int. J. Mol. Med. 2009, 23, 763–769. [Google Scholar] [CrossRef]
- Trinath, J.; Holla, S.; Mahadik, K.; Prakhar, P.; Singh, V.; Balaji, K.N. The WNT Signaling Pathway Contributes to Dectin-1-Dependent Inhibition of Toll-Like Receptor-Induced Inflammatory Signature. Mol. Cell. Biol. 2014, 34, 4301–4314. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mehmeti, M.; Bergenfelz, C.; Källberg, E.; Millrud, C.R.; Björk, P.; Ivars, F.; Johansson-Lindbom, B.; Kjellström, S.; André, I.; Leandersson, K. Wnt5a is a TLR2/4-ligand that induces tolerance in human myeloid cells. Commun. Biol. 2019, 2, 176. [Google Scholar] [CrossRef]
- Blumenthal, A.; Ehlers, S.; Lauber, J.; Buer, J.; Lange, C.; Goldmann, T.; Heine, H.; Brandt, E.; Reiling, N. The Wingless homolog WNT5A and its receptor Frizzled-5 regulate inflammatory responses of human mononuclear cells induced by microbial stimulation. Blood 2006, 108, 965–973. [Google Scholar] [CrossRef] [PubMed]
- Neumann, J.; Schaale, K.; Farhat, K.; Endermann, T.; Ulmer, A.J.; Ehlers, S.; Reiling, N. Frizzled1 is a marker of inflammatory macrophages, and its ligand Wnt3a is involved in reprogramming Mycobacterium tuberculosis-infected macrophages. FASEB J. 2010, 24, 4599–4612. [Google Scholar] [CrossRef] [PubMed]
- Schaale, K.; Brandenburg, J.; Kispert, A.; Leitges, M.; Ehlers, S.; Reiling, N. Wnt6 Is Expressed in Granulomatous Lesions of Mycobacterium tuberculosis—Infected Mice and Is Involved in Macrophage Differentiation and Proliferation. J. Immunol. 2013, 191, 5182–5195. [Google Scholar] [CrossRef]
- Zhao, C.; Bu, X.; Wang, W.; Ma, T.; Ma, H. GEC-derived SFRP5 Inhibits Wnt5a-Induced Macrophage Chemotaxis and Activation. PLoS ONE 2014, 9, e85058. [Google Scholar] [CrossRef]
- Pereira, C.; Schaer, D.J.; Bachli, E.B.; Kurrer, M.O.; Schoedon, G. Wnt5A/CaMKII Signaling Contributes to the Inflammatory Response of Macrophages and Is a Target for the Antiinflammatory Action of Activated Protein C and Interleukin-10. Arter. Thromb. Vasc. Biol. 2008, 28, 504–510. [Google Scholar] [CrossRef]
- Li, D.; Beisswenger, C.; Herr, C.; Hellberg, J.; Han, G.; Zakharkina, T.; Voss, M.; Wiewrodt, R.; Bohle, R.M.; Menger, M.D.; et al. Myeloid cell RelA/p65 promotes lung cancer proliferation through Wnt/β-catenin signaling in murine and human tumor cells. Oncogene 2014, 33, 1239–1248. [Google Scholar] [CrossRef][Green Version]
- Kerdidani, D.; Chouvardas, P.; Arjo, A.R.; Giopanou, I.; Ntaliarda, G.; Guo, Y.A.; Tsikitis, M.; Kazamias, G.; Potaris, K.; Stathopoulos, G.T.; et al. Wnt1 silences chemokine genes in dendritic cells and induces adaptive immune resistance in lung adenocarcinoma. Nat. Commun. 2019, 10, 1405. [Google Scholar] [CrossRef]
- Kumawat, K.; Gosens, R. WNT-5A: Signaling and functions in health and disease. Cell. Mol. Life Sci. 2016, 73, 567–587. [Google Scholar] [CrossRef]
- Du, Q.; Geller, D.A. Cross-Regulation Between Wnt and NF-κB Signaling Pathways. Forum Immunopathol. Dis. Ther. 2010, 1, 155–181. [Google Scholar] [CrossRef]
- Hecht, S.S. Tobacco Smoke Carcinogens and Lung Cancer. JNCI J. Natl. Cancer Inst. 1999, 91, 1194–1210. [Google Scholar] [CrossRef] [PubMed]
- Whang, Y.M.; Jo, U.; Sung, J.S.; Ju, H.J.; Kim, H.K.; Park, K.H.; Lee, J.W.; Koh, I.S.; Kim, Y.H. Wnt5a Is Associated with Cigarette Smoke-Related Lung Carcinogenesis via Protein Kinase C. PLoS ONE 2013, 8, e53012. [Google Scholar] [CrossRef]
- Haw, T.J.; Starkey, M.R.; Pavlidis, S.; Fricker, M.; Arthurs, A.L.; Nair, P.M.; Liu, G.; Hanish, I.; Kim, R.Y.; Foster, P.S.; et al. Toll-like receptor 2 and 4 have Opposing Roles in the Pathogenesis of Cigarette Smoke-induced Chronic Obstructive Pulmonary Disease. Am. J. Physiol. bLung Cell. Mol. Physiol. 2018, 314, L298–L317. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.; Sun, B.; Zhao, X.; Zhao, X.; Gu, Q.; Dong, X.; Zheng, Y.; Sun, J.; Cheng, R.; Qi, H.; et al. Overexpression of Wnt5a Promotes Angiogenesis in NSCLC. BioMed Res. Int. 2014, 2014, 832562. [Google Scholar] [CrossRef]
- Baarsma, H.; Skronska-Wasek, W.; Mutze, K.; Ciolek, F.; Wagner, D.E.; John-Schuster, G.; Heinzelmann, K.; Günther, A.; Bracke, K.R.; Dagouassat, M.; et al. Noncanonical WNT-5A signaling impairs endogenous lung repair in COPD. J. Exp. Med. 2017, 214, 143–163. [Google Scholar] [CrossRef]
- Zhang, X.; Shan, P.; Jiang, G.; Cohn, L.; Lee, P.J. Toll-like receptor 4 deficiency causes pulmonary emphysema. J. Clin. Investig. 2006, 116, 3050–3059. [Google Scholar] [CrossRef]
- Teng, F.; Slavik, V.; Duffy, K.E.; Mateo, L.S.; Goldschmidt, R. Toll-like receptor 3 is involved in airway epithelial cell response to nontypeable Haemophilus influenzae. Cell. Immunol. 2010, 260, 98–104. [Google Scholar] [CrossRef]
- Carobene, L.; Spina, D.; Disanto, M.G.; Micheletto, C.; Mazzei, M.A.; Paladini, P.; Ghiribelli, C.; Bargagli, E.; Rottoli, P. Lung cancer and interstitial lung diseases: The lack of prognostic impact of lung cancer in IPF. Intern. Emerg. Med. 2022, 17, 457–464. [Google Scholar] [CrossRef]
- Königshoff, M.; Balsara, N.; Pfaff, E.-M.; Kramer, M.; Chrobak, I.; Seeger, W.; Eickelberg, O. Functional Wnt Signaling Is Increased in Idiopathic Pulmonary Fibrosis. PLoS ONE 2008, 3, e2142. [Google Scholar] [CrossRef]
- Martin-Medina, A.; Lehmann, M.; Burgy, O.; Hermann, S.; Baarsma, H.; Wagner, D.E.; De Santis, M.M.; Ciolek, F.; Hofer, T.P.; Frankenberger, M.; et al. Increased Extracellular Vesicles Mediate WNT5A Signaling in Idiopathic Pulmonary Fibrosis. Am. J. Respir. Crit. Care Med. 2018, 198, 1527–1538. [Google Scholar] [CrossRef] [PubMed]
- Aumiller, V.; Balsara, N.; Wilhelm, J.; Günther, A.; Königshoff, M. WNT/β-Catenin Signaling Induces IL-1β Expression by Alveolar Epithelial Cells in Pulmonary Fibrosis. Am. J. Respir. Cell Mol. Biol. 2013, 49, 96–104. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).