Abstract
Floral scents possess high ornamental and economic values to rose production in the floricultural industry. In the past two decades, molecular bases of floral scent production have been studied in the rose as well as their genetic inheritance. Some significant achievements have been acquired, such as the comprehensive rose genome and the finding of a novel geraniol synthase in plants. In this review, we summarize the composition of floral scents in modern roses, focusing on the recent advances in the molecular mechanisms of floral scent production and emission, as well as the latest developments in molecular breeding and metabolic engineering of rose scents. It could provide useful information for both studying and improving the floral scent production in the rose.
1. Introduction
The floral scent is one of the most important traits in plants, which is essential to the fertilization of angiosperm plants by attracting and guiding pollinators [1]. Some volatile compounds in floral scents play important roles in plant defense against detrimental animals [2,3]. To humans, floral scent is an important flower trait; it brings mental pleasure. In addition, floral scent provides essential flavor to the food and perfume industries [4,5].
Most floral scent chemicals are produced by three general metabolic pathways, i.e., terpenoids, phenylpropanoids/benzenoids, and fatty-acid derivatives [1,6]. In plants, terpenes are the largest group of floral scent compounds, which are synthesized by two divergent pathways. One is the 2-C-Methyl-D-Erythritol-4-Phosphate (MEP) pathway, which is mainly located in plastids [7] and is responsible for the production of mono- and diterpenes [8]. The other is the mevalonate (MVA) pathway, which is mainly located in the cytosol, endoplasmic reticulum, and peroxisomes [9,10], and is responsible for the production of volatile sesquiterpenes. Phenylpropanoids/benzenoids represent the second-largest class of floral scent compounds [11], which are exclusively derived from L-phenylalanine (L-Phe). Most phenylpropanoids are not volatile unless they are further acylated or methylated at the C9 position, while benzenoids are volatile, which are synthesized through a branch of the phenylpropanoid pathway, i.e., the cinnamic acid pathway [12]. Fatty acid derivatives constitute the third largest class of flower volatiles, including low-molecular-weight alcohols, aldehydes, and lipids. Biosynthesis of volatile fatty acid derivatives is initiated by stereo-specific oxygenation of unsaturated C18 fatty acids, linolenic, and linoleic, and subsequently catalyzed by the lipoxygenase (LOX) pathway [13,14].
For centuries, roses have been one of the most important crops in the floriculture industry [15], and are highly popular worldwide as garden ornamental plants and cut flowers. Floral scent, as an important characteristic, not only improves the ornamental value of roses, but provides essential fragrances and flavorings for spices, perfumes, and cosmetics in related industries. Additionally, rose essential oil, which is composed of floral scent compounds, can be used as an analgesic or antispasmodic [16,17]. However, the development of fragrances in roses has been at a disadvantage in the breeding program, which has focused on the longevity and visual attributes of cut flowers for centuries [18]. The cause for the loss of fragrance in these flowers remains unknown, but it does not seem to conflict with the increase in the vase life [19]. Unraveling the molecular bases of floral scents is not only a fascinating topic in rose biology, but is also helpful for improving the yield of roses in the floriculture industry.
Here, we will review the current progress of genetic and molecular mechanisms controlling rose scents, including scent composition, production, and emission, and summarize the development of molecular breeding and engineering techniques for the improvement of rose scents.
2. Scent Composition of Modern Roses
The evolution of the floral scent is a complex matter, which is influenced by various factors, including the dynamics between biosynthetic pathways and the balanced selection between pollinators and florivores, which may result in relatively rapid evolution [11].
Roses have been cultivated as early as 3000 BC in China, western Asia, and northern Africa [20]. Since the 14th century, when Chinese roses were first introduced to Europe, the Chinese and European roses began to hybridize extensively, forming the genetic basis of the ‘modern rose cultivars’ Rosa hybrida [21]. Although Rosa comprises approximately 200 species, only 8~20 contribute to the genetic make-up of modern roses, including Chinese rose R. chinensis, R. multiflora, and R. gigantea, and European rose R. moschata, R. gallica, R. canina, and R. Phoenicia [22,23]. Chinese and European roses differ greatly in scent composition [24,25,26,27,28,29,30]. Chinese roses principally produce lipid-derived alcohols and esters (such as hexenol and hexenyl acetate) and aromatic compounds (such as 3,5-dimethoxytoluene (DMT) and 1,3,5-trimethoxybenzene (TMB)), whereas the major scent components of European roses are 2-phenylethanol (2-PE) and a number of monoterpenes (such as rose oxide, geraniol, and nerol) (Table 1).

Table 1.
Major components of rose floral scents.
Many modern rose flowers have earthy and spicy notes due to the presence of DMT [24,36]. DMT is a product of the phenolic methyl ethers (PME) synthesis pathway and is peculiar to Chinese ancient roses. The DMT synthesis pathway in modern roses is speculated to be obtained from Chinese roses, such as R. gigantean [36]. DMT is the basis of the ‘tea scent’ of modern roses. The tea scent is characterized by a combination of phenolic molecules from both Chinese and European lineages, which is reminiscent of ‘black tea’. It is brought to modern roses through an intermediate group of tea and hybrid tea roses, which are derived from the crosses of R. chinensis and R. gigantea with R. moschata. An array of methoxylated phenolics can be produced by these roses, such as DMT, TMB, methyleugenol, and methylisoeugenol, and a variety of alcohols and esters, such as 2-PE, citronellol, geraniol, 2-phenylethylacetate, and geranyl acetate, as well as mono- and sesquiterpenes (predominantly germacrene D), among which, DMT generally represents up to 90% of the total flower volatiles [25,27,28]. cis-3-hexenyl acetate and cis-3-hexenol are also derived from R. chinensis, which give a leafy green note to modern roses [27].
In modern roses, the main components of rose oil, including linalool, citronellol, nerol, and geraniol, are inherited from ancient European roses [27]. The damask rose (R. damascene) is an important intermediate, which is a progeny of ancient European roses by the crosses between R. gallica and R. Phoenicia. Iran is the center of diversity of the damask rose, from where the original oil-bearing cultivars are transferred to Turkey and Bulgaria [37,38,39,40]. The damask rose produces a floral scent characterized by rose oxide, which is rich in alcohols, such as 2-PE, geraniol, and nerol [28,30,41]. Among the components, 2-PE is a dominant aroma compound with a rose-like odor and characterized by the typical floral scent of modern roses [40].
3. Molecular Research Progress on Rose Scent Biosynthesis
Molecular and genetic approaches, including the candidate gene approach, transcriptomic analysis, and genetic mapping, have been used to identify scent-related genes and to unravel the gene expression network and floral scent inheritance in roses [31,33,42,43,44,45,46]. With these tools and approaches, dozens of scent-related genes have been identified and functionally validated, and many pathways of floral scent production have been uncovered in the rose (Figure 1 and Table 2).
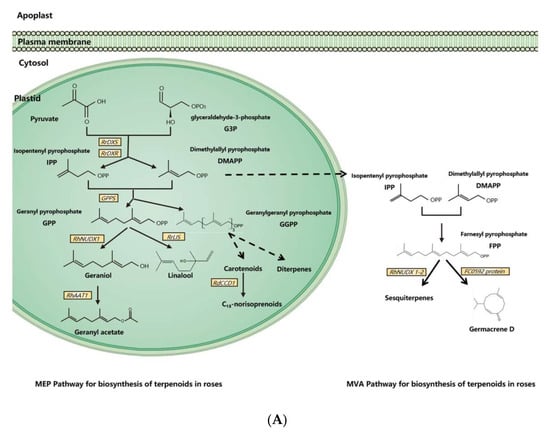
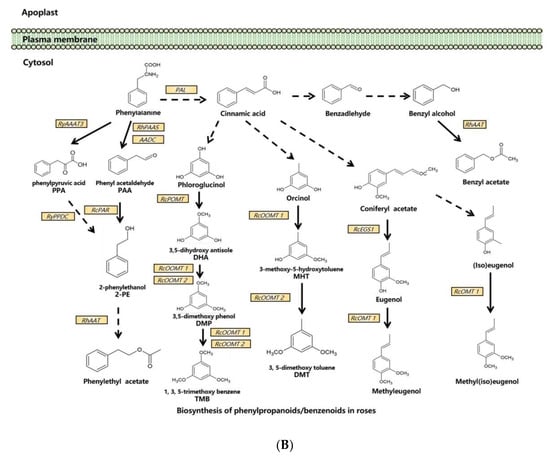
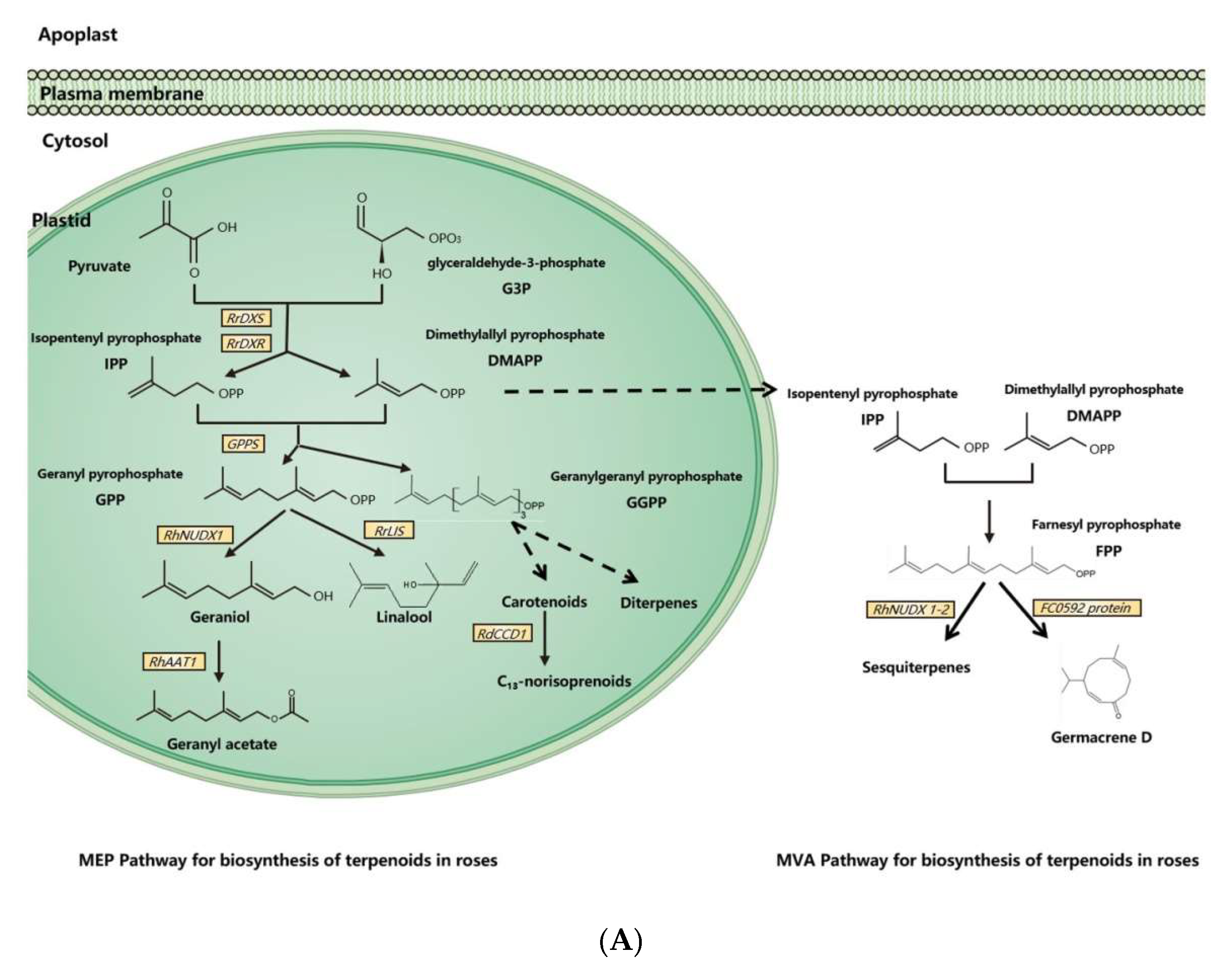
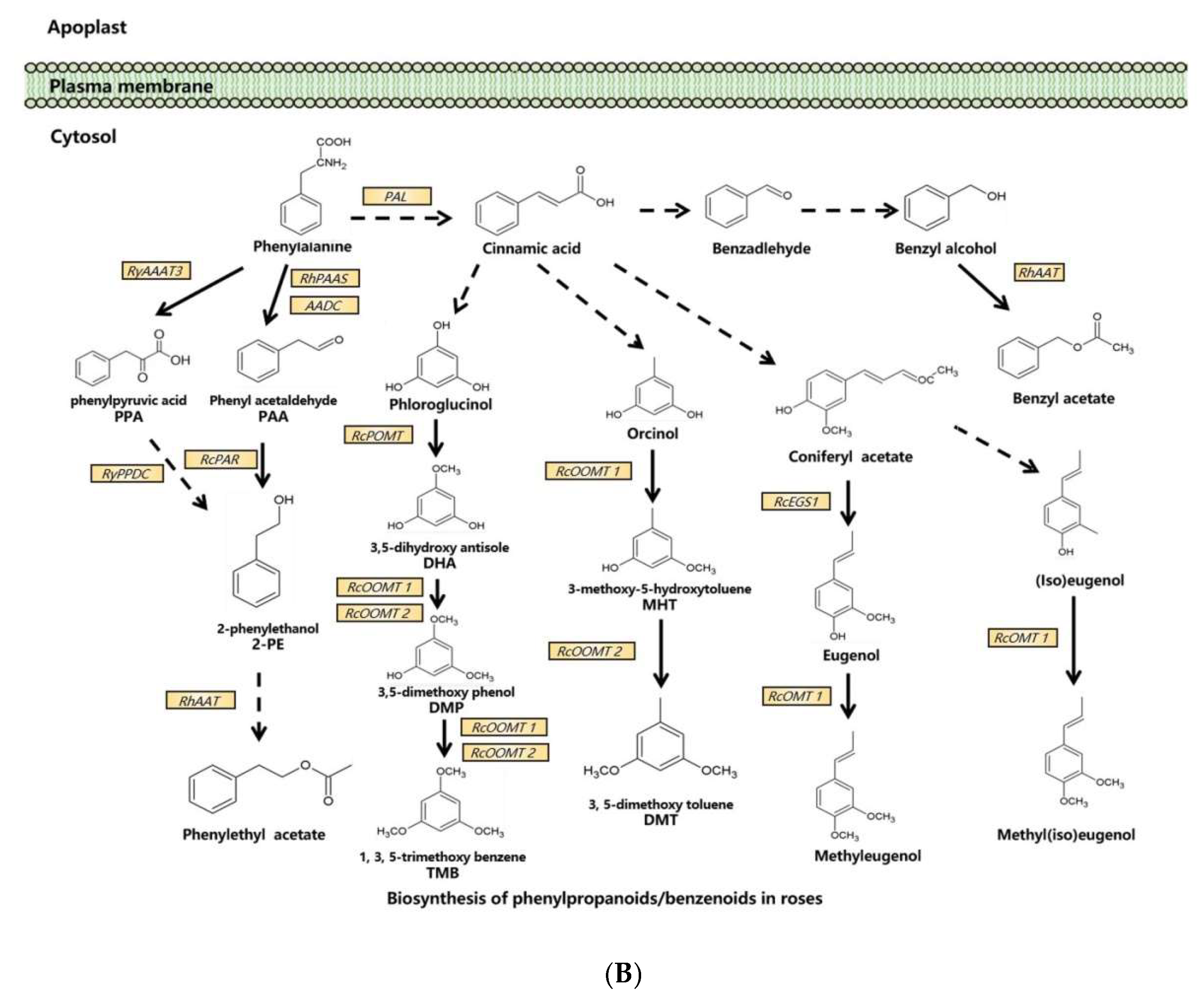
Figure 1.
Synthesis pathways of floral scents in roses. (A) The biosynthesis pathway of terpenoids in roses. (B) Biosynthesis pathway of phenylpropanoids/benzenoids in roses. Solid lines indicate established biochemical reactions, and broken lines indicate possible steps. G3P: glyceraldehyde-3-phosphate; DXS: 1-deoxy-D-xylulose-5-phosphate synthase; DXR: 1-deoxy-D-xylulose-5-phosphate reductoisomerase; IPP: isopentenyl pyrophosphate; DMAPP: dimethylallyl pyrophosphate; GPP: geranyl pyrophosphate; GPPS: GPP synthase; GGPP, geranylgeranyl pyrophosphate; NUDX: nudix hydrolase; LIS: linalool synthase; AAT: alcohol acetyltransferase; CCD: carotenoid cleavage (di-)oxygenase; FPP: farnesyl pyrophosphate; PAL: phenylalanine ammonia lyase; PAAS: phenylacetaldehyde synthase; AADC: aromatic amino acid decarboxylase; AAAT: aromatic amino acid aminotransferase; PPA: phenylpyruvic acid; PAA: phenyl acetaldehyde; PAR: phenyl acetaldehyde reductase; PPDC: phenylpyruvate decarboxylase; 2-PE: 2-phenylethanol; POMT: phloroglucinol O-methyltransferase; OMT: O-methyltransferases; OOMT: orcinol O-methyltransferases; MHT: 3-methoxy-5-hydroxytoluene; DMT: 3,5-dimethoxy toluene; DHA: 3,5-dihydroxy antisole; DMP: 3,5-dimethoxy phenol; TMB: 1,3,5-trimethoxy benzene.

Table 2.
Scent-related genes in roses.
3.1. Biosynthesis of Terpenoids in Rose Floral Scents
Dimethylallyl pyrophosphate (DMAPP) and isopentenyl pyrophosphate (IPP) are synthesized from glyceraldehyde-3-phosphate (G3P) and pyruvate by a series of enzymes in plasmids [11]. As an enzyme for the synthesis of DMAPP and IPP, 1-deoxy-D-xylulose-5-phosphate reductoisomerase (RrDXR) is shown to play a key role in the production of volatile monoterpenes in R. rugosa [50]. Geranyl pyrophosphate (GPP) is synthesized from DMAPP and IPP by the MEP pathway in plastids [60]. GPP is a precursor of monoterpenes in plants, from which various monoterpenes are produced by a series of monoterpene synthases. In roses, a novel monoterpene synthase, designated as RhNUDX1, is responsible for geraniol synthesis. Recombinant RhNUDX1 only shows the diphosphohydrolase activity by transforming GPP to geranyl monophosphate (GP) in vitro. However, in a cytosolic context, RhNUDX1 is equivalent to geraniol synthase (GES), which is found in other plants responsible for geraniol production [18]. In rose cultivar Rosa× wichurana, another NUDX1 gene RwNUDX1-2 is involved in the biosynthesis of a group of sesquiterpenoids, especially E,E-farnesol. It is proposed that roses utilize different NUDX1 protein complexes to generate different derivatives [61].
Monoterpene alcohols are normally transformed to acetate esters by alcohol acetyltransferase (AAT), by which the acetyl moiety is transferred from acetyl-CoA to the alcoholic substrate [62,63]. In the rose, an AAT, RhAAT 1, is isolated, which shows limited substrate specificity in vitro. Its preferred substrate is geraniol, while it can also accept other alcohols as substrates, including citronellol, nerol, 1-octanol, 2-PE, and cis-3-hexen-1-ol [32]. Despite the fact that the preferred substrate is geraniol in vitro, the transgenic petunia flower of RhAAT1 mainly produces phenylethyl acetate and phenylmethyl acetate using 2-PE and benzyl alcohol as the substrates. When fed with geraniol or octanol, the transgenic flowers also produce acetates, suggesting its dependence on substrate availability in planta [51].
As a sesquiterpene, germacrene is a common ingredient of rose floral scents and an intermediate in the biosynthesis of other sesquiterpenes [64]. In R. hybrida, a gene (Clone FC0592) for sesquiterpenes has been identified and confirmed to produce germacrene D in an in vitro assay with farnesyl pyrophosphate (FPP) as the substrate [31].
Carotenoids are one class of tetraterpenoids, from which C13-norisoprenoids (such as monoterpenes β-damascenone, α-ionone, and β-ionone) are generated by degradation. A carotenoid cleavage (di-)oxygenase (CCD) gene RdCCD1 was confirmed to be responsible for the accumulation of C13-norisoprenoids in flowers of R. damascene and the cleavage of a variety of carotenoids in in vitro assays [48]. However, as the subclass 4 of CCD genes, RdCCD4 cannot utilize β-carotene as a substrate in in vivo assays, indicating that it is not involved in the production of β-ionone in R. damascene [49].
3.2. Biosynthesis of Phenylpropanoids/Benzenoids in Rose Floral Scents
In one branch pathway of phenylpropanoid synthesis, L-Phe is the direct precursor of 2-PE and its β-D-glucopyranoside (2-PEG) [41]. In roses, L-Phe can be converted into phenyl acetaldehyde (PAA) by both aromatic amino acid decarboxylase (AADC) and phenyl acetaldehyde synthase (PAAS) [52,65], and PAA is subsequently converted to 2-PE by phenyl acetaldehyde reductase (PAR) [53,58]. Another 2-PE biosynthetic pathway via phenylpyruvic acid (PPA) from L-Phe has been reported in roses. The aromatic amino acid aminotransferase (AAAT) catalyzes the production of PPA from L-Phe, and RNAi suppression of the AAAT gene RyAAAT3 decreases 2-PE production in rose protoplasts [54]. In this pathway, phenylpyruvate decarboxylase (RyPPDC) is also shown to be responsible for 2-PE production. However, RyPPDC, as a heat adaptation in the summer, is likely to participate in an alternative principal pathway for rose floral scent production [55].
Multiple branching pathways are involved in the synthesis of rose benzenoids. In the branching pathway for TMB, phloroglucinol O-methyltransferase (POMT) catalyzes the first methylation step of phloroglucinol (PLG) to 3,5-dihdroxyanisole (DHA) in R. chinensis [56]. DHA is subsequently converted to TMB by two orcinol O-methyltransferases, OOMT1 and OOMT2, through two final methylation reactions [28,35]. OOMT1 and OOMT2 are also responsible for DMT synthesis with orcinol as the initial substrate in the first and the second methylation steps, respectively [28,35]. Despite sharing a 96.5% similarity at the amino acid level, OOMT1 and OOMT2 exhibit different substrate specificities in PME biosynthesis. The main sequence difference is the single amino acid polymorphism in the phenolic substrate binding site. OOMT1 is mainly found in Chinese roses instead of European roses. It is speculated that OOMT1 may have evolved from an OOMT2-like gene, and its emergence is a critical step in the evolution of scent production in Chinese roses [36].
OOMTs also catalyze the production of methyleugenol and methyl(iso)eugenol by efficiently methylating eugenol and (iso)eugenol in R. chinensis [34].
In general, the rose floral scent is dominated by terpenoids, phenylpropanoids, and benzenoids. Only a few fatty acid derivatives are involved in rose floral scents. However, no related enzymes or genes have been isolated or characterized from roses to date.
3.3. Transcriptional Regulation of Rose Floral Scent Synthesis
Transcription factors (TFs) have been shown to participate in the coordinated regulation of the scent biosynthetic network [66,67]. Some TFs have been identified for the regulation of floral scents of phenylpropanoids/benzenoids in petunia, including activators of ODO1 [68], EOBI [69,70], EOBII [70,71,72], PH4 [73], and the repressor of PhMYB4 [74]. Recently, only three TFs have been isolated for transcriptional regulation of floral terpenoid production, including GaWRKY1 in Gossypium arboretum [75], MYC2 in Arabidopsis thaliana [76], and PbbHLH4 in Phalaenopsis bellina [77]. Interestingly, some TFs may act upstream of multiple metabolic pathways across terpenoids and phenylpropanoids/benzenoids. When the Arabidopsis transcription factor production of anthocyanin pigment 1 (PAP1) is introduced into the petunia and rose, phenylpropanoid- and terpenoid-derived scent compounds show elevated expressions compared to the control flowers [78,79].
In roses, only one R-type MYB TF, RhMYB1, may play a role in floral scent production, but its function has not been validated [59]. Sun found the expression of NUDX1 is transcriptionally regulated between scented R. chinensis ‘Old blush’ and unscented R.×wichurana, but failed to identify the functionary TFs in the regulation of NUDX1 [80]. In rose petals, the miR156-SPL9 regulatory hub is proposed to orchestrate the production of both colored anthocyanins and certain terpenes, by permitting the complexation of preexisting MYB-bHLH-WD40 proteins [15]. The maximum expression of GDS, which encodes the enzyme catalyzing germacrene D synthesis, is correlated with miR156 activation and with SPL9 downregulation. The miR156-SPL9 regulatory hub might also regulate the expression of terpene synthase genes directly. The absence of the expression of nerolidol synthase (NES) genes is correlated with the downregulation of SPL9 through activation of miR156.
4. Regulation of Floral Scent Production and Emission in Roses
4.1. Floral Scent Production Sites in Roses
The petal represents the major source of scent in R. hybrida. Some scent-related genes are specifically expressed in petals, such as RcEGS 1, RhPAAS, rose-PAR, and RcPOMT [52,53,56,57]. Although the cell morphology between the two epidermal layers of the petal is different, both layers are capable of producing and emitting scent volatiles [81,82], which is different from some other species, such as Antirrhinum majus [83].
However, not all the scent compounds are produced by petals in roses. In R. rugosa, three esters, including citronellyl acetate, geraniol acetate, and nerol acetate, are preferably synthesized in stamens instead of petals [50].
4.2. Flower Developmental Stages for Scent Production in Roses
Petal development can be divided into two phases after its initiation in most plants. The first phase is a slow growth stage mainly resulting from cell division, and the second phase is a rapid growth stage resulting only from cell expansion [84,85]. In roses, floral scent production peaks during the second stage, which usually corresponds to a semi-opening state in the flowering process [31]. Changes in major individual compounds are found to be consistent with the changes in total scents during flower development [86]. Several scent-related genes are expressed during this process, such as RrAAT [50], RhPAAS [52], rose-PAR [53,87], and OOMT2 [88].
4.3. Diurnal Regulation of Scent Production in Rose Flowers
Circadian clock exerts the rhythmic emission of floral scents in numerous species, such as Nicotiana [89], Mirabilis jalapa [90], Petunia axillaris [91], Vaccinium corymbosum [92], etc., presumably synchronized with the activity of pollinators. Most floral volatile emissions are regulated by the circadian clock in roses, including the terpenoids geraniol, E-citral, β-cubebene, geranyl acetate, germacrene D [47,93], phenylpropanoids/benzenoids 2-PE, PAA, DMT [52,93,94], and fatty acid derivative hexylacetate [93]. Glycosylated volatiles stored within petals are believed to be the sources of rhythmically emitted volatiles [94].
However, the regulatory effects on scent compounds vary greatly in roses, showing little to no effect on some compounds. In R. hybrida flowers, the emissions of several scent compounds, such as oxidized monoterpenols, trans-caryophyllene, dihydro-β-ionone, and germacrene D, are completely dependent on light rather than diurnal regulation [47,93], indicating that various independent mechanisms have evolved during diurnal regulation of scent emission in roses.
4.4. Environmental and Internal Factors Affecting Rose Scent Production
Floral scent production is sensitive to environmental factors [95]. Among the roses, the cultivar ‘Noorjahan’ of R. damascene contains a very different floral aroma complex compared with two cultivars at different altitudes and climatic conditions. A four-fold difference in the main contents of essential oils in cultivars between these two sites has been detected [29].
Light has a great impact on rose floral scent production. In R. hybrida, the production of germacrene D and geranyl acetate is directly regulated by light [47]. Ambient temperature is also found to influence scent production in plants. When the temperature increases from 20 °C to 35 °C, the total endogenous amounts of scent components decrease in P. axillaris [96]. Under high temperatures in the summer, an alternative principal pathway is induced, through which 2-PE is produced via PPA instead of PAA [55].
Hormones are reported to regulate volatile productions in flowers. GA is found to negatively regulate scent production in petunia flowers by transcriptional/post-transcriptional downregulation of regulatory and biosynthetic scent-related genes [97]. Ethylene is also reported to play a negative role in the floral volatile production in Petunia hybrida [98,99] and Lathyrus odoratus [100]. However, in one study using 13 R. hybrida cut cultivars as materials, scent emission does not appear to be regulated by endogenous or exogenous ethylene but occurs independently of petal senescence or abscission [19].
4.5. Internal Mechanisms of Rose Scent Emission after Production
Floral scent composite is controlled by both the endogenous production and emission rate of scent compounds [101]. In roses, not all scent compounds are emitted simultaneously with their production. The emission of germacrene D in R. hybrida oscillates during the daily cycle while its endogenous level is constant throughout the day [47]. A similar pattern has been found for 2-PE in R. damascene [93].
The tissue structures of flowers do not correlate with floral scent emissions in roses, as shown by petal anatomy, which indicates there is no significant difference in optical or ultrathin sections between scented and unscented rose cultivars [82].
The endogenous concentrations of steady-state volatiles are correlated with floral scent emissions in plants [102]. Floral scent compounds, such as 2-PE, geraniol, and benzyl alcohol are present in flower tissues in the forms of monoglycosides and/or diglycosides. After being hydrolyzed by β-glucosidase or endoglycosidase, these glycoconjugates become volatile and emit from petal tissues [103,104,105,106]. Moreover, 2-PEG is a source for the circadian-emitted 2-PE in R. damascene [93]. β-glucosidase is involved in the emission of 2-PE from rose flowers by hydrolyzing 2-PEG [107].
Passive diffusion has been assumed to be the mechanism for floral scent emission. However, studies have shown that exocytosis or specific transporters may be involved in the transport of scent molecules across the plasma membranes [108]. Recently, an adenosine triphosphate-binding cassette (ABC) transporter rather than passive diffusion has been shown to be responsible for floral volatile emission in P. hybrida. Down-regulation of the ABC transporter gene PhABCG results in decreased emission of volatiles and increased toxic accumulation in the plasma membrane [109]. As the petal develops, OOMTs are increasingly associated with membranes, suggesting the involvement of the secretory machinery of cells in scent emission in roses [33].
5. Molecular Breeding and Metabolic Engineering of Rose Floral Scents
Floral volatiles could respond rapidly to artificial selection [110]. However, most modern cultivars used for cut flowers have little fragrance as a result of breeding preference for other traits, such as the flower color and form or longevity in roses [35]. Little is known about the inheritance of rose scents.
Three volatiles, neryl acetate, geranyl acetate, and nerol, show monogenic or oligogenic traits in the process of sexual hybridization of R. hybrida, while three other volatiles, 2-PE, geraniol, and β-citronellol, show quantitative inheritance. All three monogenic volatiles and six QTLs of the three quantitative volatiles have been mapped on the rose chromosome map, together with some scent-related genes for germacrene D, alcohol acetyltransferase 1, and various OMTs [46,111]. To date, dozens of scent-related genes or traits have been mapped to the linkage group (LG) (Table 3) [46,111,112,113,114,115,116].

Table 3.
Phenotypic and genotypic traits, available on the consensus map in roses.
However, it is still far from rationally constructing scent attributes by crossbreeding or molecular approaches. A hybrid tea progeny has been obtained from parents emitting very different floral scents. As a general rule, the quantity and quality of volatile compounds are both low in progeny members [117]. Many uncertainties may be involved in rose scent inheritance, such as gene regulation or mutation, substrate availability, and substrate preference of enzymes [51,78].
It is possible to produce rose scents in model plants, such as petunias. However, in order to regulate the metabolic flow of rose scent compounds, a detailed analysis of available substrates and enzyme activities in petunias is required as a prerequisite [51]. Additionally, yeast has been shown to be another carrier for metabolic engineering of rose scent biosynthesis [52].
6. Obstacles in Further Study for Rose Floral Scents
Floral scents in modern roses are complex combinations of scents from ancient Chinese roses and European roses. Understanding the floral scents can be used to generate the genealogy for Rosa cultivars at the genus level [27,118]. The past decade has witnessed the development of various approaches and tools for molecular studies of rose scents. Metabolic profiling and gene expression profiling are powerful tools for the identification of candidate genes and enzymes in scent synthesis [18,119]. However, some obstacles still hinder the in-depth understanding of the molecular mechanisms of floral scents in roses.
Functional validation of candidate genes is largely limited in roses because no universal transformation system is available. To date, the functions of most rose genes are validated in heterologous systems, such as the petunia, Escherichia coli, and yeast [31,32,33,51,52]. Virus-induced gene silencing (VIGS) is a promising approach for gene knockdown in roses, despite its efficiency remaining low compared to tobacco and tomato [44,120]. Furthermore, an effective method of “graft-accelerated VIGS” was developed especially for research on flower characteristics, such as floral scent, floral color, and so on [121]. Currently, it is still not enough to predict the substrate preference of candidate enzymes in roses based on gene homology with other species, because protein sequence divergence may affect enzyme affinity to the substrates [51,122]. Moreover, the preferred substrates of a candidate enzyme in in vitro assays or heterologous systems may not necessarily be the ones in vivo [32,51,122].
Studies based on the next-generation sequencing (NGS) technology have been reported, including various transcriptome studies, providing a wealth of information about the expression patterns of genes or miRNAs in multiple tissues or biological processes in roses [42,123,124,125,126,127]. A draft genome sequence of a wild diploid rose (R. multiflora Thunb.) has been generated, which provides an important resource for fundamental studies and applications of roses [128]. By generating a homozygous genotype from a heterozygous diploid modern rose progenitor, R. chinensis ‘Old Blush’, the most comprehensive rose genome, is created through single-molecule real-time sequencing and a meta assembly approach [15]. This genome provides a foundation for understanding rose traits. By reconstructing regulatory and secondary metabolism pathways, the regulation of scent is proposed to be interconnected with that of the flower color. However, it still has limitations in studies of cultivated roses because most of the roses are tetraploids.
In crossbreeding for rose scents, one of the major hindrances to creating novel varieties stems from the lack of allelic variations in modern roses, because their genome bears a massive introgression of R. chinensis alleles [129]. Another hindrance is the polyploidy barrier, which appears when alleles of interest are transferred from wild diploid species to tetraploid modern roses [20]. It has been found that manipulating temperature may be a strategy to overcome the polyploidy barrier [130]. The third hindrance arises from the limitations of genetic mapping. In these studies, due to the inheritance complexity of tetraploid populations, most genetic mappings for locating or tagging scent-related genes are conducted using diploid populations [131,132,133,134], which causes great difficulties when localizing scent characteristics in crossbreeding, because most modern roses are tetraploid.
Our inadequate understanding of odor metabolism pathways greatly limits the genetic engineering of the floral scent-related traits of roses. When a scent enzyme is newly introduced, we need to consider whether appropriate substrates can be found and whether enough products can be produced. Transcriptional factors seem to provide an efficient strategy to manipulate metabolic flux flow for rose scent modification [68,78,79]. Overall, it is clear that genetic manipulation of the rose floral scent is possible but requires a more rational design.
7. Conclusions and Future Perspectives
Over the last two decades, an increasing amount of knowledge on the biosynthesis of rose floral scents has been acquired. Recent advances in the isolation and characterization of genes and enzymes involved in different scent biosynthetic pathways have enhanced our understanding of floral scent synthesis in the rose. Despite the progress, many aspects remain largely unknown. In particular, we still do not know the regulatory networks controlling the synthesis pathways of rose floral scents, including how the majority of floral scents are synthesized and how their orchestrated emissions are regulated, which hinder their molecular breeding and metabolic engineering. Therefore, we anticipate that future research efforts will focus on exploring the regulatory mechanisms of rose floral scents; meanwhile, some more advanced approaches should be developed and applied for elucidating the tetraploid rose genome and function validation of candidate genes.
Author Contributions
Conceptualization, Z.Z.; writing—manuscript preparation, S.S.; writing—review and editing, Z.Z.; funding acquisition, Z.Z. and S.S. All authors have read and agreed to the published version of the manuscript.
Funding
This work was funded by the National Natural Science Foundation of China (grant number 32102430), the Shandong Provincial Natural Science Foundation (grant number ZR2020QC159), and the Innovation Project of the Shandong Academy of Agricultural Sciences (grants numbers CXGC2021B17, CXGC2022A06).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Dudareva, N.; Pichersky, E.; Gershenzon, J. Biochemistry of plant volatiles. Plant Physiol. 2004, 135, 1893–1902. [Google Scholar] [CrossRef]
- Caruso, C.M.; Parachnowitsch, A.L. Do Plsants Eavesdrop on Floral Scent Signals? Trends Plant Sci. 2016, 21, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Picazo-Aragonés, J.; Terrab, A.; Balao, F. Plant Volatile Organic Compounds Evolution: Transcriptional Regulation, Epigenetics and Polyploidy. Int. J. Mol. Sci. 2020, 21, 8956. [Google Scholar] [CrossRef] [PubMed]
- Grammer, K.; Fink, B.; Møller, A.P.; Thornhill, R. Darwinian aesthetics: Sexual selection and the biology of beauty. Biol. Control 2003, 78, 385–407. [Google Scholar] [CrossRef] [PubMed]
- Jianwu, R.; Lina, Y.; Yan, W.; Hongjun, Y. Chemical profile of floral scent at different flower developmental stages of rose de rescht (Rosa damascena Mill.) cultivated in Beijing. J. Essent. Oil Bear. Plants 2016, 19, 433–443. [Google Scholar]
- Croteau, R.; Kutchan, T.; Lewis, N. Natural Products (Secondary Metabolites). In Biochemistry & Molecular Biology of Plants; Buchanan, B.B., Gruissem, W., Jones, R.L., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2000; pp. 1251–1318. [Google Scholar]
- Hsieh, M.-H.; Chang, C.-Y.; Hsu, S.-J.; Chen, J.-J. Chloroplast localization of methylerythritol 4-phosphate pathway enzymes and regulation of mitochondrial genes in ispD and ispE albino mutants in Arabidopsis. Plant Mol. Biol. 2008, 66, 663–673. [Google Scholar] [CrossRef]
- Knudsen, J.T.; Gershenzon, J. The chemical diversity of floral scent. In Biology of PLANT volatiles; Jette, T., Knudsen, J.G., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 57–78. [Google Scholar]
- Pulido, P.; Perello, C.; Rodriguez-Concepcion, M. New insights into plant isoprenoid metabolism. Mol. Plant 2012, 5, 964–967. [Google Scholar] [CrossRef]
- Simkin, A.J.; Guirimand, G.; Papon, N.; Courdavault, V.; Thabet, I.; Ginis, O.; Bouzid, S.; Giglioli-Guivarc’h, N.; Clastre, M. Peroxisomal localisation of the final steps of the mevalonic acid pathway in planta. Planta 2011, 234, 903. [Google Scholar] [CrossRef]
- Knudsen, J.T.; Eriksson, R.; Gershenzon, J.; Ståhl, B. Diversity and distribution of floral scent. Bot. Rev. 2006, 72, 1–120. [Google Scholar] [CrossRef]
- Boatright, J.; Negre, F.; Chen, X.; Kish, C.M.; Wood, B.; Peel, G.; Orlova, I.; Gang, D.; Rhodes, D.; Dudareva, N.; et al. Understanding in vivo benzenoid metabolism in petunia petal tissue. Plant Physiol. 2004, 135, 1993–2011. [Google Scholar] [CrossRef]
- Feussner, I.; Wasternack, C. The lipoxygenase pathway. Annu. Rev. Plant Biol. 2002, 53, 275–297. [Google Scholar] [CrossRef] [PubMed]
- Schaller, F. Enzymes of the biosynthesis of octadecanoid-derived signalling molecules. J. Exp. Bot. 2001, 52, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Raymond, O.; Gouzy, J.; Just, J.; Badouin, H.; Verdenaud, M.; Lemainque, A.; Vergne, P.; Moja, S.; Choisne, N.; Pont, C. The Rosa genome provides new insights into the domestication of modern roses. Nat. Genet. 2018, 50, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Basim, E.; Basim, H. Antibacterial activity of Rosa damascena essential oil. Fitoterapia 2003, 74, 394–396. [Google Scholar] [CrossRef]
- Elliott, J. Delmar’s Integrated Herb Guide for Nurses. Northeast. Nat. 2002, 9, 360. [Google Scholar]
- Magnard, J.-L.; Roccia, A.; Caissard, J.-C.; Vergne, P.; Sun, P.; Hecquet, R.; Dubois, A.; Hibrand-Saint Oyant, L.; Jullien, F.; Nicolè, F. Biosynthesis of monoterpene scent compounds in roses. Science 2015, 349, 81–83. [Google Scholar] [CrossRef]
- Borda, A.M.; Clark, D.G.; Huber, D.J.; Welt, B.A.; Nell, T.A. Effects of ethylene on volatile emission and fragrance in cut roses: The relationship between fragrance and vase life. Postharvest. Biol. Technol. 2011, 59, 245–252. [Google Scholar] [CrossRef]
- Bendahmane, M.; Dubois, A.; Raymond, O.; Bris, M.L. Genetics and genomics of flower initiation and development in roses. J. Exp. Bot. 2013, 64, 847–857. [Google Scholar] [CrossRef]
- Raymond, O. Domestication et Sélection Dirigée Chez le Rosier: Analyse Historique via les Phénotypes Morphologique, Chimique et Biochimique. Ph.D. Thesis, Université Claude Bernard Lyon 1, Lyon, France, 1999. [Google Scholar]
- De Vries, D.; Dubois, L.A. Rose breeding: Past, present, prospects. In Proceedings of the II International Rose Symposium 424, Antibes, France, 20–24 February 1995. [Google Scholar]
- Reynders-Aloisi, S.; Bollereau, P. Characterisation of genetic diversity in genus Rosa by randomly amplified polymorphic DNA. In Proceedings of the II International Rose Symposium 424, Antibes, France, 20–24 February 1995; ISHS Acta: Antibes, France, 1995. [Google Scholar]
- Flament, I.; Debonneville, C.; Furrer, A. Volatile compounds of roses: Characterization of cultivars based on the headspace analysis of living flower emissions. In ACS Symposium Series (USA); AGRIS: San Francisco, CA, USA, 1993. [Google Scholar]
- Gang, D.R. Evolution of flavors and scents. Annu. Rev. Plant Biol. 2005, 56, 301–325. [Google Scholar] [CrossRef]
- Jirovetz, L.; Buchbauer, G.; Stoyanova, A.; Balinova, A.; Guangjiun, Z.; Xihan, M. Solid phase microextraction/gas chromatographic and olfactory analysis of the scent and fixative properties of the essential oil of Rosa damascena L. from China. Flavour Fragr. J. 2005, 20, 7–12. [Google Scholar] [CrossRef]
- Joichi, A.; Yomogida, K.; Awano, K.i.; Ueda, Y. Volatile components of tea-scented modern roses and ancient Chinese roses. Flavour Fragr. J. 2005, 20, 152–157. [Google Scholar] [CrossRef]
- Scalliet, G.; Journot, N.; Jullien, F.; Baudino, S.; Magnard, J.-L.; Channelière, S.; Vergne, P.; Dumas, C.; Bendahmane, M.; Cock, J.M. Biosynthesis of the major scent components 3,5-dimethoxytoluene and 1,3,5-trimethoxybenzene by novel rose O-methyltransferases. FEBS Lett. 2002, 523, 113–118. [Google Scholar] [CrossRef]
- Verma, R.S.; Padalia, R.C.; Chauhan, A.; Singh, A.; Yadav, A.K. Volatile constituents of essential oil and rose water of damask rose (Rosa damascena Mill.) cultivars from North Indian hills. Nat. Prod. Res. 2011, 25, 1577–1584. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Washio, H.; Straubinger, M.; Knapp, H.; Winterhalter, P. Occurrence of a glucosidic progenitor of rose oxide in rose flowers, Rosa damascena Mill. Nat. Prod. Lett. 1998, 12, 5–10. [Google Scholar] [CrossRef]
- Guterman, I. Rose Scent: Genomics Approach to Discovering Novel Floral Fragrance-Related Genes. Plant Cell 2002, 14, 2325–2338. [Google Scholar] [CrossRef]
- Shalit, M.; Guterman, I.; Volpin, H.; Bar, E.; Tamari, T.; Menda, N.; Adam, Z.; Zamir, D.; Vainstein, A.; Weiss, D. Volatile ester formation in roses. Identification of an acetyl-coenzyme A. Geraniol/Citronellol acetyltransferase in developing rose petals. Plant Physiol. 2003, 131, 1868–1876. [Google Scholar] [CrossRef] [PubMed]
- Scalliet, G.; Lionnet, C.; Le Bechec, M.; Dutron, L.; Magnard, J.-L.; Baudino, S.; Bergougnoux, V.; Jullien, F.; Chambrier, P.; Vergne, P. Role of petal-specific orcinol O-methyltransferases in the evolution of rose scent. Plant Physiol. 2006, 140, 18–29. [Google Scholar] [CrossRef]
- Wu, S.; Watanabe, N.; Mita, S.; Ueda, Y.; Shibuya, M.; Ebizuka, Y. Two O-Methyltransferases isolated from flower petals of Rosa chinensis var. spontanea involved in scent biosynthesis. J. Biosci. Bioeng. 2003, 96, 119–128. [Google Scholar] [CrossRef]
- Lavid, N.; Wang, J.; Shalit, M.; Guterman, I.; Bar, E.; Beuerle, T.; Menda, N.; Shafir, S.; Zamir, D.; Adam, Z. O-methyltransferases involved in the biosynthesis of volatile phenolic derivatives in rose petals. Plant Physiol. 2002, 129, 1899–1907. [Google Scholar] [CrossRef]
- Scalliet, G.; Piola, F.; Douady, C.J.; Réty, S.; Raymond, O.; Baudino, S.; Bordji, K.; Bendahmane, M.; Dumas, C.; Cock, J.M. Scent evolution in Chinese roses. Proc. Natl. Acad. Sci. USA 2008, 105, 5927–5932. [Google Scholar]
- Babaei, A.; Tabaei-Aghdaei, S.R.; Khosh-Khui, M.; Omidbaigi, R.; Naghavi, M.R.; Esselink, G.D.; Smulders, M.J. Microsatellite analysis of Damask rose (Rosa damascena Mill.) accessions from various regions in Iran reveals multiple genotypes. BMC Plant Biol. 2007, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Baydar, N.G.; Baydar, H.; Debener, T. Analysis of genetic relationships among Rosa damascena plants grown in Turkey by using AFLP and microsatellite markers. J. Biotechnol. 2004, 111, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Kiani, M.; Zamani, Z.; Khalighi, A.; Fatahi, R.; Byrne, D.H. Wide genetic diversity of Rosa damascena Mill. germplasm in Iran as revealed by RAPD analysis. Sci. Hortic. 2008, 115, 386–392. [Google Scholar] [CrossRef]
- Rusanov, K.; Kovacheva, N.; Vosman, B.; Zhang, L.; Rajapakse, S.; Atanassov, A.; Atanassov, I. Microsatellite analysis of Rosa damascena Mill. accessions reveals genetic similarity between genotypes used for rose oil production and old Damask rose varieties. Theor. Appl. Genet. 2005, 111, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Hayashi, K.; Yagi, K.; Asai, T.; MacTavish, H.; Picone, J.; Turnbull, C.; Watanabe, N. Biogenesis of 2-phenylethanol in rose flowers: Incorporation of [2H8]L-phenylalanine into 2-phenylethanol and its beta-D-glucopyranoside during the flower opening of Rosa ’Hoh-Jun’ and Rosa damascena Mill. Biosci. Biotechnol. Biochem. 2002, 66, 943–947. [Google Scholar] [CrossRef]
- Dubois, A.; Carrere, S.; Raymond, O.; Pouvreau, B.; Cottret, L.; Roccia, A.; Onesto, J.-P.; Sakr, S.; Atanassova, R.; Baudino, S. Transcriptome database resource and gene expression atlas for the rose. BMC Genom. 2012, 13, 638. [Google Scholar] [CrossRef]
- Dubois, A.; Raymond, O.; Maene, M.; Baudino, S.; Langlade, N.B.; Boltz, V.; Vergne, P.; Bendahmane, M. Tinkering with the C-function: A molecular frame for the selection of double flowers in cultivated roses. PLoS ONE 2010, 5, e9288. [Google Scholar] [CrossRef]
- Ma, N.; Xue, J.; Li, Y.; Liu, X.; Dai, F.; Jia, W.; Luo, Y.; Gao, J. Rh-PIP2; 1, a rose aquaporin gene, is involved in ethylene-regulated petal expansion. Plant Physiol. 2008, 148, 894–907. [Google Scholar] [CrossRef]
- Nakamura, N.; Fukuchi-Mizutani, M.; Katsumoto, Y.; Togami, J.; Senior, M.; Matsuda, Y.; Furuichi, K.; Yoshimoto, M.; Matsunaga, A.; Ishiguro, K.; et al. Environmental risk assessment and field performance of rose (Rosa × hybrida) genetically modified for delphinidin production. Plant Biotechnol. 2011, 28, 251–261. [Google Scholar] [CrossRef]
- Spiller, M.; Berger, R.G.; Debener, T. Genetic dissection of scent metabolic profiles in diploid rose populations. Theor. Appl. Genet. 2010, 120, 1461–1471. [Google Scholar] [CrossRef]
- Hendel-Rahmanim, K.; Masci, T.; Vainstein, A.; Weiss, D. Diurnal regulation of scent emission in rose flowers. Planta 2007, 226, 1491–1499. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.-C.; Horváth, G.; Molnár, P.; Turcsi, E.; Deli, J.; Schrader, J.; Sandmann, G.; Schmidt, H.; Schwab, W. Substrate promiscuity of RdCCD1, a carotenoid cleavage oxygenase from Rosa damascena. Phytochemistry 2009, 70, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.-C.; Molnár, P.; Schwab, W. Cloning and functional characterization of carotenoid cleavage dioxygenase 4 genes. J. Exp. Bot. 2009, 60, 3011–3022. [Google Scholar] [CrossRef]
- Feng, L.; Chen, C.; Li, T.; Wang, M.; Tao, J.; Zhao, D.; Sheng, L. Flowery odor formation revealed by differential expression of monoterpene biosynthetic genes and monoterpene accumulation in rose (Rosa rugosa Thunb.). Plant Physiol. Biochem. 2014, 75, 80–88. [Google Scholar] [CrossRef]
- Guterman, I.; Masci, T.; Chen, X.; Negre, F.; Pichersky, E.; Dudareva, N.; Weiss, D.; Vainstein, A. Generation of phenylpropanoid pathway-derived volatiles in transgenic plants: Rose alcohol acetyltransferase produces phenylethyl acetate and benzyl acetate in petunia flowers. Plant Mol. Biol. 2006, 60, 555–563. [Google Scholar] [CrossRef]
- Farhi, M.; Lavie, O.; Masci, T.; Hendel-Rahmanim, K.; Weiss, D.; Abeliovich, H.; Vainstein, A. Identification of rose phenylacetaldehyde synthase by functional complementation in yeast. Plant Mol. Biol. 2010, 72, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.M.; Kobayashi, H.; Sakai, M.; Hirata, H.; Asai, T.; Ohnishi, T.; Baldermann, S.; Watanabe, N. Functional characterization of rose phenylacetaldehyde reductase (PAR), an enzyme involved in the biosynthesis of the scent compound 2-phenylethanol. J. Plant Physiol. 2011, 168, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Hirata, H.; Ohnishi, T.; Ishida, H.; Tomida, K.; Sakai, M.; Hara, M.; Watanabe, N. Functional characterization of aromatic amino acid aminotransferase involved in 2-phenylethanol biosynthesis in isolated rose petal protoplasts. J. Plant Physiol. 2012, 169, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Hirata, H.; Ohnishi, T.; Tomida, K.; Ishida, H.; Kanda, M.; Sakai, M.; Yoshimura, J.; Suzuki, H.; Ishikawa, T.; Dohra, H. Seasonal induction of alternative principal pathway for rose flower scent. Sci. Rep. 2016, 6, 20234. [Google Scholar] [CrossRef]
- Wu, S.; Watanabe, N.; Mita, S.; Dohra, H.; Ueda, Y.; Shibuya, M.; Ebizuka, Y. The key role of phloroglucinol O-methyltransferase in the biosynthesis of Rosa chinensis volatile 1, 3, 5-trimethoxybenzene. Plant Physiol. 2004, 135, 95–102. [Google Scholar] [CrossRef]
- Wang, H.; Yan, H.; Zhang, H. Cloning and expression analysis of eugenol synthase gene RcEGS1 in Rosa chinensis Pallida. Acta Hortic. Sin. 2012, 39, 1387–1394. [Google Scholar]
- Sakai, M.; Hirata, H.; Sayama, H.; Sekiguchi, K.; Itano, H.; Asai, T.; Dohra, H.; Hara, M.; Watanabe, N. Production of 2-phenylethanol in roses as the dominant floral scent compound from L-phenylalanine by two key enzymes, a PLP-dependent decarboxylase and a phenylacetaldehyde reductase. Biosci. Biotechnol. Biochem. 2007, 71, 2408–2419. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhang, H.; Wang, Q.; Jian, H.; Qiu, X.; Wang, J.; Tang, K. Isolation and identification of a putative scent-related gene RhMYB1 from rose. Mol. Biol. Rep. 2011, 38, 4475–4482. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Concepción, M.; Boronat, A. Elucidation of the methylerythritol phosphate pathway for isoprenoid biosynthesis in bacteria and plastids. A metabolic milestone achieved through genomics. Plant Physiol. 2002, 130, 1079–1089. [Google Scholar] [CrossRef]
- Sun, P.; Dégut, C.; Réty, S.; Caissard, J.C.; Hibrand-Saint Oyant, L.; Bony, A.; Paramita, S.N.; Conart, C.; Magnard, J.L.; Jeauffre, J. Functional diversification in the Nudix hydrolase gene family drives sesquiterpene biosynthesis in Rosa× wichurana. Plant J. 2020, 104, 185–199. [Google Scholar] [CrossRef]
- Aharoni, A.; Keizer, L.C.; Bouwmeester, H.J.; Sun, Z.; Alvarez-Huerta, M.; Verhoeven, H.A.; Blaas, J.; van Houwelingen, A.M.; De Vos, R.C.; van der Voet, H. Identification of the SAAT gene involved in strawberry flavor biogenesis by use of DNA microarrays. Plant Cell 2000, 12, 647–661. [Google Scholar] [CrossRef]
- Shalit, M.; Katzir, N.; Tadmor, Y.; Larkov, O.; Burger, Y.; Shalekhet, F.; Lastochkin, E.; Ravid, U.; Amar, O.; Edelstein, M. Acetyl-CoA: Alcohol acetyltransferase activity and aroma formation in ripening melon fruits. J. Agric. Food Chem. 2001, 49, 794–799. [Google Scholar] [CrossRef]
- Colby, S.M.; Crock, J.; Dowdle-Rizzo, B.; Lemaux, P.G.; Croteau, R. Germacrene C synthase from Lycopersicon esculentum cv. VFNT Cherry tomato: cDNA isolation, characterization, and bacterial expression of the multiple product sesquiterpene cyclase. Proc. Natl. Acad. Sci. USA 1998, 95, 2216–2221. [Google Scholar] [CrossRef]
- Sakai, M.; Saori, T.; Hiroshi, H.; Tatsuo, A.; Hideo, D.; Masakazu, H.; Naoharu, W. Purification and Characterization of β-Glucosidase Involved in the Emission of 2-Phenylethanol from Rose Flowers. Biosci. Biotechnol. Biochem. 2008, 72, 219–221. [Google Scholar] [CrossRef]
- Muhlemann, J.K.; Klempien, A.; Dudareva, N. Floral volatiles: From biosynthesis to function. Plant Cell Environ. 2014, 37, 1936–1949. [Google Scholar] [CrossRef]
- Nagegowda, D.A.; Gupta, P. Advances in biosynthesis, regulation, and metabolic engineering of plant specialized terpenoids. Plant Sci. 2020, 294, 110457. [Google Scholar] [CrossRef] [PubMed]
- Verdonk, J.C.; Haring, M.A.; van Tunen, A.J.; Schuurink, R.C. ODORANT1 regulates fragrance biosynthesis in petunia flowers. Plant Cell 2005, 17, 1612–1624. [Google Scholar] [CrossRef] [PubMed]
- Spitzer-Rimon, B.; Farhi, M.; Albo, B.; Cna’ani, A.; Zvi, M.M.B.; Masci, T.; Edelbaum, O.; Yu, Y.; Shklarman, E.; Ovadis, M. The R2R3-MYB–like regulatory factor EOBI, acting downstream of EOBII, regulates scent production by activating ODO1 and structural scent-related genes in petunia. Plant Cell 2012, 24, 5089–5105. [Google Scholar] [CrossRef] [PubMed]
- Van Moerkercke, A.; Haring, M.A.; Schuurink, R.C. The transcription factor EMISSION OF BENZENOIDS II activates the MYB ODORANT1 promoter at a MYB binding site specific for fragrant petunias. Plant J. 2011, 67, 917–928. [Google Scholar] [CrossRef] [PubMed]
- Colquhoun, T.A.; Schwieterman, M.L.; Wedde, A.E.; Schimmel, B.C.; Marciniak, D.M.; Verdonk, J.C.; Kim, J.Y.; Oh, Y.; Gális, I.; Baldwin, I.T. EOBII controls flower opening by functioning as a general transcriptomic switch. Plant Physiol. 2011, 156, 974–984. [Google Scholar] [CrossRef]
- Spitzer-Rimon, B.; Marhevka, E.; Barkai, O.; Marton, I.; Edelbaum, O.; Masci, T.; Prathapani, N.-K.; Shklarman, E.; Ovadis, M.; Vainstein, A. EOBII, a gene encoding a flower-specific regulator of phenylpropanoid volatiles’ biosynthesis in petunia. Plant Cell 2010, 22, 1961–1976. [Google Scholar] [CrossRef]
- Cna’ani, A.; Spitzer-Rimon, B.; Ravid, J.; Farhi, M.; Masci, T.; Aravena-Calvo, J.; Ovadis, M.; Vainstein, A. Two showy traits, scent emission and pigmentation, are finely coregulated by the MYB transcription factor PH4 in petunia flowers. New Phytol. 2015, 208, 708–714. [Google Scholar] [CrossRef]
- Colquhoun, T.A.; Kim, J.Y.; Wedde, A.E.; Levin, L.A.; Schmitt, K.C.; Schuurink, R.C.; Clark, D.G. PhMYB4 fine-tunes the floral volatile signature of Petunia× hybrida through PhC4H. J. Exp. Bot. 2010, 62, 1133–1143. [Google Scholar] [CrossRef]
- Xu, Y.-H.; Wang, J.-W.; Wang, S.; Wang, J.-Y.; Chen, X.-Y. Characterization of GaWRKY1, a cotton transcription factor that regulates the sesquiterpene synthase gene (+)-δ-cadinene synthase-A. Plant Physiol. 2004, 135, 507–515. [Google Scholar] [CrossRef]
- Hong, G.J.; Xue, X.Y.; Mao, Y.B.; Wang, L.J.; Chen, X.Y. Arabidopsis MYC2 interacts with DELLA proteins in regulating sesquiterpene synthase gene expression. Plant Cell 2012, 24, 2635–2648. [Google Scholar] [CrossRef]
- Chuang, Y.C.; Hung, Y.C.; Tsai, W.C.; Chen, W.H.; Chen, H.H. PbbHLH4 regulates floral monoterpene biosynthesis in Phalaenopsis orchids. J. Exp. Bot. 2018, 69, 4363–4377. [Google Scholar] [CrossRef] [PubMed]
- Zvi, M.M.B.; Shklarman, E.; Masci, T.; Kalev, H.; Debener, T.; Shafir, S.; Ovadis, M.; Vainstein, A. PAP1 transcription factor enhances production of phenylpropanoid and terpenoid scent compounds in rose flowers. New Phytol. 2012, 195, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Zvi, M.M.B.; Negre-Zakharov, F.; Masci, T.; Ovadis, M.; Shklarman, E.; Ben-Meir, H.; Tzfira, T.; Dudareva, N.; Vainstein, A. Interlinking showy traits: Co-engineering of scent and colour biosynthesis in flowers. Plant Biotechnol. J. 2008, 6, 403–415. [Google Scholar] [CrossRef] [PubMed]
- Sun, P. Molecular and biochemical studies of fragrance biosynthesis in rose. Ph.D. Thesis, Universiteit van Amsterdam, Amsterdam, The Netherlands, 2017. [Google Scholar]
- Baudino, S.; Caissard, J.-C.; Bergougnoux, V.; Jullien, F.; Magnard, J.-L.; Scalliet, G.; Cock, J.M.; Hugueney, P. Production and emission of volatile compounds by petal cells. Plant Signal. Behav. 2007, 2, 525–526. [Google Scholar] [CrossRef]
- Bergougnoux, V.; Caissard, J.-C.; Jullien, F.; Magnard, J.-L.; Scalliet, G.; Cock, J.M.; Hugueney, P.; Baudino, S. Both the adaxial and abaxial epidermal layers of the rose petal emit volatile scent compounds. Planta 2007, 226, 853–866. [Google Scholar] [CrossRef]
- Comba, L.; Corbet, S.; Hunt, H.; Outram, S.; Parker, J.; Glover, B. The role of genes influencing the corolla in pollination of Antirrhinum majus. Plant Cell Environ. 2000, 23, 639–647. [Google Scholar] [CrossRef]
- Ben-Nissan, G.; Weiss, D. The petunia homologue of tomato gast1: Transcript accumulation coincides with gibberellin-induced corolla cell elongation. Plant Mol. Biol. 1996, 32, 1067–1074. [Google Scholar] [CrossRef]
- Martin, C.; Gerats, T. Control of pigment biosynthesis genes during petal development. Plant Cell 1993, 5, 1253. [Google Scholar] [CrossRef]
- Shalit, M.; Shafir, S.; Larkov, O.; Bar, E.; Kaslassi, D.; Adam, Z.; Zamir, D.; Vainstein, A.; Weiss, D.; Ravid, U. Volatile compounds emitted by rose cultivars: Fragrance perception by man and honeybees. Isr. J. Plant Sci. 2004, 52, 245–255. [Google Scholar] [CrossRef]
- Karami, A.; Niazi, A.; Kavoosi, G.; Khosh-Khui, M.; Salehi, H. Temporal characterization of 2-phenylethanol in strongly and weakly scented genotypes of damask rose. Physiol. Mol. Biol. Plants 2015, 21, 43–49. [Google Scholar] [CrossRef]
- Chen, X.; Baldermann, S.; Cao, S.; Lu, Y.; Liu, C.; Hirata, H.; Watanabe, N. Developmental patterns of emission of scent compounds and related gene expression in roses of the cultivar Rosa x hybrida cv. ‘Yves Piaget’. Plant Physiol. Biochem. 2015, 87, 109–114. [Google Scholar] [CrossRef]
- Raguso, R.A.; Levin, R.A.; Foose, S.E.; Holmberg, M.W.; McDade, L.A. Fragrance chemistry, nocturnal rhythms and pollination “syndromes” in Nicotiana. Phytochemistry 2003, 63, 265–284. [Google Scholar] [CrossRef]
- Effmert, U.; Große, J.; Röse, U.S.; Ehrig, F.; Kägi, R.; Piechulla, B. Volatile composition, emission pattern, and localization of floral scent emission in Mirabilis jalapa (Nyctaginaceae). Am. J. Bot. 2005, 92, 2–12. [Google Scholar] [CrossRef]
- Hoballah, M.E.; Stuurman, J.; Turlings, T.C.; Guerin, P.M.; Connetable, S.; Kuhlemeier, C. The composition and timing of flower odour emission by wild Petunia axillaris coincide with the antennal perception and nocturnal activity of the pollinator Manduca sexta. Planta 2005, 222, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Saona, C.; Parra, L.; Quiroz, A.; Isaacs, R. Variation in highbush blueberry floral volatile profiles as a function of pollination status, cultivar, time of day and flower part: Implications for flower visitation by bees. Ann. Bot. 2011, 107, 1377–1390. [Google Scholar] [CrossRef] [PubMed]
- Helsper, J.P.; Davies, J.A.; Bouwmeester, H.J.; Krol, A.F.; van Kampen, M.H. Circadian rhythmicity in emission of volatile compounds by flowers of Rosa hybrida L. cv. Honesty. Planta 1998, 207, 88–95. [Google Scholar] [CrossRef]
- Picone, J.M.; Clery, R.A.; Watanabe, N.; MacTavish, H.S.; Turnbull, C.G. Rhythmic emission of floral volatiles from Rosa damascena semperflorens cv. ‘Quatre Saisons’. Planta 2004, 219, 468–478. [Google Scholar]
- Zu, P.; Blanckenhorn, W.U.; Schiestl, F.P. Heritability of floral volatiles and pleiotropic responses to artificial selection in Brassica rapa. New Phytol. 2016, 209, 1208–1219. [Google Scholar] [CrossRef]
- Sagae, M.; Oyama-Okubo, N.; Ando, T.; Marchesi, E.; Nakayama, M. Effect of temperature on the floral scent emission and endogenous volatile profile of Petunia axillaris. Biosci. Biotechnol. Biochem. 2008, 72, 110–115. [Google Scholar] [CrossRef]
- Ravid, J.; Spitzer-Rimon, B.; Takebayashi, Y.; Seo, M.; Cna’ani, A.; Aravena-Calvo, J.; Masci, T.; Farhi, M.; Vainstein, A. GA as a regulatory link between the showy floral traits color and scent. New Phytol. 2017, 215, 411–422. [Google Scholar] [CrossRef]
- Negre, F.; Kish, C.M.; Boatright, J.; Underwood, B.; Shibuya, K.; Wagner, C.; Clark, D.G.; Dudareva, N. Regulation of methylbenzoate emission after pollination in snapdragon and petunia flowers. Plant Cell 2003, 15, 2992–3006. [Google Scholar] [CrossRef] [PubMed]
- Underwood, B.A.; Tieman, D.M.; Shibuya, K.; Dexter, R.J.; Loucas, H.M.; Simkin, A.J.; Sims, C.A.; Schmelz, E.A.; Klee, H.J.; Clark, D.G. Ethylene-regulated floral volatile synthesis in petunia corollas. Plant Physiol. 2005, 138, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Sexton, R.; Stopford, A.P.; Moodie, W.T.; Porter, A.E. Aroma production from cut sweet pea flowers (Lathyrus odoratus): The role of ethylene. Physiol. Plant. 2005, 124, 381–389. [Google Scholar] [CrossRef]
- Kondo, M.; Oyama-Okubo, N.; Ando, T.; Marchesi, E.; Nakayama, M. Floral scent diversity is differently expressed in emitted and endogenous components in Petunia axillaris lines. Ann. Bot. 2006, 98, 1253–1259. [Google Scholar] [CrossRef] [PubMed]
- Oyama-Okubo, N.; Ando, T.; Watanabe, N.; Marchesi, E.; Uchida, K.; Nakayama, M. Emission mechanism of floral scent in Petunia axillaris. Biosci. Biotechnol. Biochem. 2005, 69, 773–777. [Google Scholar] [CrossRef]
- Loughrin, J.H.; Hamilton-Kemp, T.R.; Burton, H.R.; Andersen, R.A.; Hildebrand, D.F. Glycosidically bound volatile components of Nicotiana sylvestris and N suaveolens flowers. Phytochemistry 1992, 31, 1537–1540. [Google Scholar] [CrossRef]
- Ogawa, K.; Ijima, Y.; Guo, W.; Watanabe, N.; Usui, T.; Dong, S.; Tong, Q.; Sakata, K. Purification of a β-primeverosidase concerned with alcoholic aroma formation in tea leaves (cv. Shuixian) to be processed to oolong tea. J. Agric. Food Chem. 1997, 45, 877–882. [Google Scholar]
- Reuveni, M.; Sagi, Z.; Evnor, D.; Hetzroni, A. β-Glucosidase activity is involved in scent production in Narcissus flowers. Plant Sci. 1999, 147, 19–24. [Google Scholar] [CrossRef]
- Schulz, G.; Stahl-Biskup, E. Essential oils and glycosidic bound volatiles from leaves, stems, flowers and roots of Hyssopus officinalis L. (Lamiaceae). Flavour Fragrance J. 1991, 6, 69–73. [Google Scholar] [CrossRef]
- Hayashi, S.; Yagi, K.; Ishikawa, T.; Kawasaki, M.; Asai, T.; Picone, J.; Turnbull, C.; Hiratake, J.; Sakata, K.; Takada, M. Emission of 2-phenylethanol from its β-D-glucopyranoside and the biogenesis of these compounds from [2 H 8] L-phenylalanine in rose flowers. Tetrahedron 2004, 60, 7005–7013. [Google Scholar] [CrossRef]
- Jetter, R. Examination of the processes involved in the emission of scent volatiles from flowers. In Biology of Floral Scent; Natalia, D., Eran, P., Eds.; CRC Press: Boca Raton, FL, USA, 2006; pp. 125–144. [Google Scholar]
- Adebesin, F.; Widhalm, J.R.; Boachon, B.; Lefèvre, F.; Pierman, B.; Lynch, J.H.; Alam, I.; Junqueira, B.; Benke, R.; Ray, S. Emission of volatile organic compounds from petunia flowers is facilitated by an ABC transporter. Science 2017, 356, 1386–1388. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Zu, P.; Schiestl, F.P. The molecular bases of floral scent evolution under artificial selection: Insights from a transcriptome analysis in Brassica rapa. Sci. Rep. 2016, 6, 36966. [Google Scholar] [CrossRef] [PubMed]
- Dreier, K.; Spiller, M.; Linde, M.; Kaufmann, H.; Berger, R.; Krings, U.; Debener, T.; Bretzke, M. Genetic and molecular analyses of key loci involved in self incompatibility and floral scent in roses. In Proceedings of the V International Symposium on Rose Research and Cultivation 870, Gifu, Japan, 24–29 May 2009; ISHS Acta: Gifu, Japan, 2009. [Google Scholar]
- Crespel, L.; Chirollet, M.; Durel, C.; Zhang, D.; Meynet, J.; Gudin, S. Mapping of qualitative and quantitative phenotypic traits in Rosa using AFLP markers. Theor. Appl. Genet. 2002, 105, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Linde, M.; Hattendorf, A.; Kaufmann, H.; Debener, T. Powdery mildew resistance in roses: QTL mapping in different environments using selective genotyping. Theor. Appl. Genet. 2006, 113, 1081–1092. [Google Scholar] [CrossRef] [PubMed]
- Spiller, M.; Linde, M.; Hibrand-Saint Oyant, L.; Tsai, C.-J.; Byrne, D.H.; Smulders, M.J.; Foucher, F.; Debener, T. Towards a unified genetic map for diploid roses. Theor. Appl. Genet. 2011, 122, 489–500. [Google Scholar] [CrossRef]
- Yan, Z.; Denneboom, C.; Hattendorf, A.; Dolstra, O.; Debener, T.; Stam, P.; Visser, P. Construction of an integrated map of rose with AFLP, SSR, PK, RGA, RFLP, SCAR and morphological markers. Theor. Appl. Genet. 2005, 110, 766–777. [Google Scholar] [CrossRef]
- Yan, Z.; Visser, P.; Hendriks, T.; Prins, T.; Stam, P.; Dolstra, O. QTL analysis of variation for vigour in rose. Euphytica 2007, 154, 53–62. [Google Scholar] [CrossRef]
- Cherri-Martin, M.; Jullien, F.; Heizmann, P.; Baudino, S. Fragrance heritability in Hybrid Tea roses. Sci. Hortic. 2007, 113, 177–181. [Google Scholar] [CrossRef]
- Nowak, R. Chemical Composition of Hips Essential Oils of Some Rosa L. Species December 13, 2004. Z. Für Nat. C 2005, 60, 369–378. [Google Scholar] [CrossRef]
- Fridman, E.; Pichersky, E. Metabolomics, genomics, proteomics, and the identification of enzymes and their substrates and products. Curr. Opin. Plant Biol. 2005, 8, 242–248. [Google Scholar] [CrossRef]
- Spitzer, B.; Zvi, M.M.; Ovadis, M.; Marhevka, E.; Barkai, O.; Edelbaum, O.; Marton, I.; Masci, T.; Alon, M.; Morin, S.; et al. Reverse genetics of floral scent: Application of tobacco rattle virus-based gene silencing in Petunia. Plant Physiol. 2007, 145, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Shi, S.; Ma, N.; Cao, X.; Zhang, H.; Qiu, X.; Wang, Q.; Jian, H.; Zhou, N.; Zhang, Z. Graft-accelerated virus-induced gene silencing facilitates functional genomics in rose flowers. J. Integr. Plant Biol. 2018, 60, 34–44. [Google Scholar] [CrossRef] [PubMed]
- Beekwilder, J.; Alvarez-Huerta, M.; Neef, E.; Verstappen, F.W.; Bouwmeester, H.J.; Aharoni, A. Functional characterization of enzymes forming volatile esters from strawberry and banana. Plant Physiol. 2004, 135, 1865–1878. [Google Scholar] [CrossRef] [PubMed]
- Dubois, A.; Remay, A.; Raymond, O.; Balzergue, S.; Chauvet, A.; Maene, M.; Pecrix, Y.; Yang, S.H.; Jeauffre, J.; Thouroude, T.; et al. Genomic approach to study floral development genes in Rosa sp. PLoS ONE 2011, 6, e28455. [Google Scholar] [CrossRef]
- Han, Y.; Wan, H.; Cheng, T.; Wang, J.; Yang, W.; Pan, H.; Zhang, Q. Comparative RNA-seq analysis of transcriptome dynamics during petal development in Rosa chinensis. Sci. Rep. 2017, 7, 43382. [Google Scholar] [CrossRef]
- Kim, J.; Park, J.H.; Lim, C.J.; Lim, J.Y.; Ryu, J.Y.; Lee, B.W.; Choi, J.P.; Kim, W.B.; Lee, H.Y.; Choi, Y.; et al. Small RNA and transcriptome deep sequencing proffers insight into floral gene regulation in Rosa cultivars. BMC Genom. 2012, 13, 657. [Google Scholar] [CrossRef]
- Pei, H.; Ma, N.; Chen, J.; Zheng, Y.; Tian, J.; Li, J.; Zhang, S.; Fei, Z.; Gao, J. Integrative analysis of miRNA and mRNA profiles in response to ethylene in rose petals during flower opening. PLoS ONE 2013, 8, e64290. [Google Scholar] [CrossRef]
- Yan, H.; Zhang, H.; Chen, M.; Jian, H.; Baudino, S.; Caissard, J.C.; Bendahmane, M.; Li, S.; Zhang, T.; Zhou, N.; et al. Transcriptome and gene expression analysis during flower blooming in Rosa chinensis ‘Pallida’. Gene 2014, 540, 96–103. [Google Scholar] [CrossRef]
- Nakamura, N.; Hirakawa, H.; Sato, S.; Otagaki, S.; Matsumoto, S.; Tabata, S.; Tanaka, Y. Genome structure of Rosa multiflora, a wild ancestor of cultivated roses. DNA Res. 2018, 25, 113–121. [Google Scholar] [CrossRef]
- Martin, M.; Piola, F.; Chessel, D.; Jay, M.; Heizmann, P. The domestication process of the Modern Rose: Genetic structure and allelic composition of the rose complex. Theor. Appl. Genet. 2001, 102, 398–404. [Google Scholar] [CrossRef]
- Pécrix, Y.; Rallo, G.; Folzer, H.; Cigna, M.; Gudin, S.; Le Bris, M. Polyploidization mechanisms: Temperature environment can induce diploid gamete formation in Rosa sp. J. Exp. Bot. 2011, 62, 3587–3597. [Google Scholar] [CrossRef]
- Gar, O.; Sargent, D.J.; Tsai, C.-J.; Pleban, T.; Shalev, G.; Byrne, D.H.; Zamir, D. An autotetraploid linkage map of rose (Rosa hybrida) validated using the strawberry (Fragaria vesca) genome sequence. PLoS ONE 2011, 6, e20463. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, S.; Byrne, D.; Zhang, L.; Anderson, N.; Arumuganathan, K.; Ballard, R. Two genetic linkage maps of tetraploid roses. Theor. Appl. Genet. 2001, 103, 575–583. [Google Scholar] [CrossRef]
- Yu, C.; Luo, L.; Pan, H.; Guo, X.; Wan, H.; Zhang, Q. Filling gaps with construction of a genetic linkage map in tetraploid roses. Front. Plant Sci. 2014, 5, 796. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Byrne, D.; Ballard, R.; Rajapakse, S. Microsatellite marker development in rose and its application in tetraploid mapping. J. Am. Soc. Hort. Sci. 2006, 131, 380–387. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).