Abstract
ROPs (Rho-like GTPases from plants) are a unique family of small GTP-binding proteins in plants and play vital roles in numerous cellular processes, including growth and development, abiotic stress signaling, and plant defense. In the case of the latter, the role of ROPs as response regulators to obligate parasitism remains largely enigmatic. Herein, we isolated and identified ShROP7 and show that it plays a critical role in plant immune response to pathogen infection. Real-time quantitative PCR analysis revealed that the expression of ShROP7 was significantly increased during incompatible interactions. To establish its requirement for resistance, we demonstrate that virus-induced gene silencing (VIGS) of ShROP7 resulted in increased susceptibility of tomato to Oidium neolycopersici (On) Lanzhou strain (On-Lz). Downstream resistance signaling through H2O2 and the induction of the hypersensitive response (HR) in ShROP7-silenced plants were significantly reduced after inoculating with On-Lz. Taken together, with the identification of ShROP7-interacting candidates, including ShSOBIR1, we demonstrate that ShROP7 plays a positive regulatory role in tomato powdery mildew resistance.
1. Introduction
The small GTP-binding proteins constitute a superfamily of signaling regulators, among which are the Ras, Rho, Rab, Arf/Sar, and Ran subfamilies in yeast and animals, and the Rho, Rab, Arf/Sar, and Ran subfamilies in plants [1]. The Rho-related subfamily of plant small monomeric GTPases is referred to as RAC or ROPs (Rho-like GTPases from plants) [2,3]. As indicated by their nomenclature, this family of small proteins with molecular weights in the range of 20–40 kDa, and possesses a GTP binding active site with concomitant GTPase activity [2,4]. In plants, small GTP-binding proteins are mainly involved in plant signal transduction processes, with critical roles in controlling cell polarity, growth, morphological development, cytokinesis, hormone signaling, control of cytoskeletal organization, and response to pathogen infection [5,6,7,8,9].
As important signal transduction regulators in plants, ROPs play vital roles in plant defense, regulating defense and immune-related processes associated with biotrophic, necrotrophic, and obligate parasites [6,7,8]. Upon pathogen penetration of the host cell wall, immune signal transduction is initiated via the activity of receptor-like kinases (RLKs), pattern recognition receptors (PRRs), which perceive initial signals associated with the invading pathogen, ultimately transducing these signals to the guanine nucleotide exchange factors (GEFs) [10,11,12,13]. Once initiated, signaling transfers to the ROP-GEF signaling node, resulting in the conjugation and conversion of the GDP-bound inactive (GDP·ROP) into GTP-bound active (GTP·ROP). The key function of this process is the transmission of extracellular signaling to the intracellular defense signaling network.
In parallel to the extra-to-intracellular signal transduction cascade, the GTPase-activating protein (GAP) converts the GTP·ROP node to GDP·ROP by promoting GTPase activity. The function of this conversion is to maintain a dynamic balance of the two ROP protein states. This activity is further regulated by the activity of the guanine nucleotide dissociation inhibitor (GDI), which inhibits GDP·ROP to GTP·ROP conversion [6]. The sum of these signaling processes, and the generation of an activated ROP protein, triggers the activation of downstream signals which function to initiate additional, required, stress response cascades (e.g., Ca2+, ABA responses, H2O2, actin cytoskeleton) [14,15].
The function of the ROP family in the interaction between plants and pathogens remains largely enigmatic [16]. ROPs have been shown to possess critical functions which regulate the establishment of plant pathogen-associated molecular pattern (PAMP)-triggered immunity (PTI). In this role, it has been demonstrated that OsRac1 regulates reactive oxygen species (ROS) production and cell death, while constitutively active (CA) OsRac1 results in enhanced PAMP-induced ROS production and resistance to Xanthomonas oryzae pv. oryzae and Magnaporthe grisea in rice [17,18]. As a function of effector-triggered immunity (ETI), it has been further shown that OsRac1 is activated by the R-protein Pit for triggering downstream responses in [10]. This role was further demonstrated by the silencing of a dominant negative (DN) variant of OsRac1 in tobacco resulting in a reduction of pathogen-induced ROS and hypersensitive response (HR) production [19]. This function was further established via the overexpression of HvRac1 in barley, which led to a promotion of callose deposition and ROS production, the sum of which provided enhanced resistance to M. oryzae penetration [20].
It is not surprising that various members of the ROP family from the same plant play distinct roles in disease resistance. For example, among seven ROP family genes from rice (Oryza sativa), OsRac1 has been shown to play a positive regulatory role in resistance to pathogens, while OsRac4, OsRac5, and OsRac6 negatively regulate rice resistance to pathogens. Further, OsRac3 and OsRac7 have been shown to not be involved in plant resistance [21,22]. A similar functionality has been shown in wheat, whereby TaRab18 is required for stripe rust resistance [23], yet TaRac6 and TaRop10 are susceptibility factors in wheat–Puccinia striiformis f. sp. tritici interaction, respectively [16]. Similarly, in barley, HvRacB promotes susceptibility to Blumeria graminis f. sp. hordei [1], while CN-HvRac1 promotes resistance to M. oryzae [20].
Tomato powdery mildew, caused by Oidium neolycopersici (On), is an obligate biotrophic ascomycotina fungal disease that causes tremendous economic losses in tomato (Solanum lycopersicum L.). As an obligate biotrophic ascomycotina, On can parasitize more than 60 plants, including members of the Solanaceae [24]. At present, due to the warm and humid environmental conditions in greenhouse settings, tomato powdery mildew has become more serious, with the incidence of infection rates being 80–90%—a major problem in tomato production around the world [25]. As such, studies on the mechanism(s) underpinning resistance to powdery mildew in tomato have become a prerequisite for tomato disease-resistance breeding. In the current study, we found that the expression of ShROP7 was significantly upregulated during an incompatible host-pathogen interaction, with further analyses revealing that the silencing of ShROP7 enhances the susceptibility of tomato to Oidium neolycopersici (On) Lanzhou strain (On-Lz). As a potential mechanism driving this function, we demonstrate that a receptor-like kinase (RLK) protein ShSOBIR1 interacts with ShROP7. In total, the current work described herein provides evidence that ShROP7 plays a critical role in the resistance of tomato to powdery mildew.
2. Results
2.1. Identification and Sequence Analysis of Tomato ShROP7
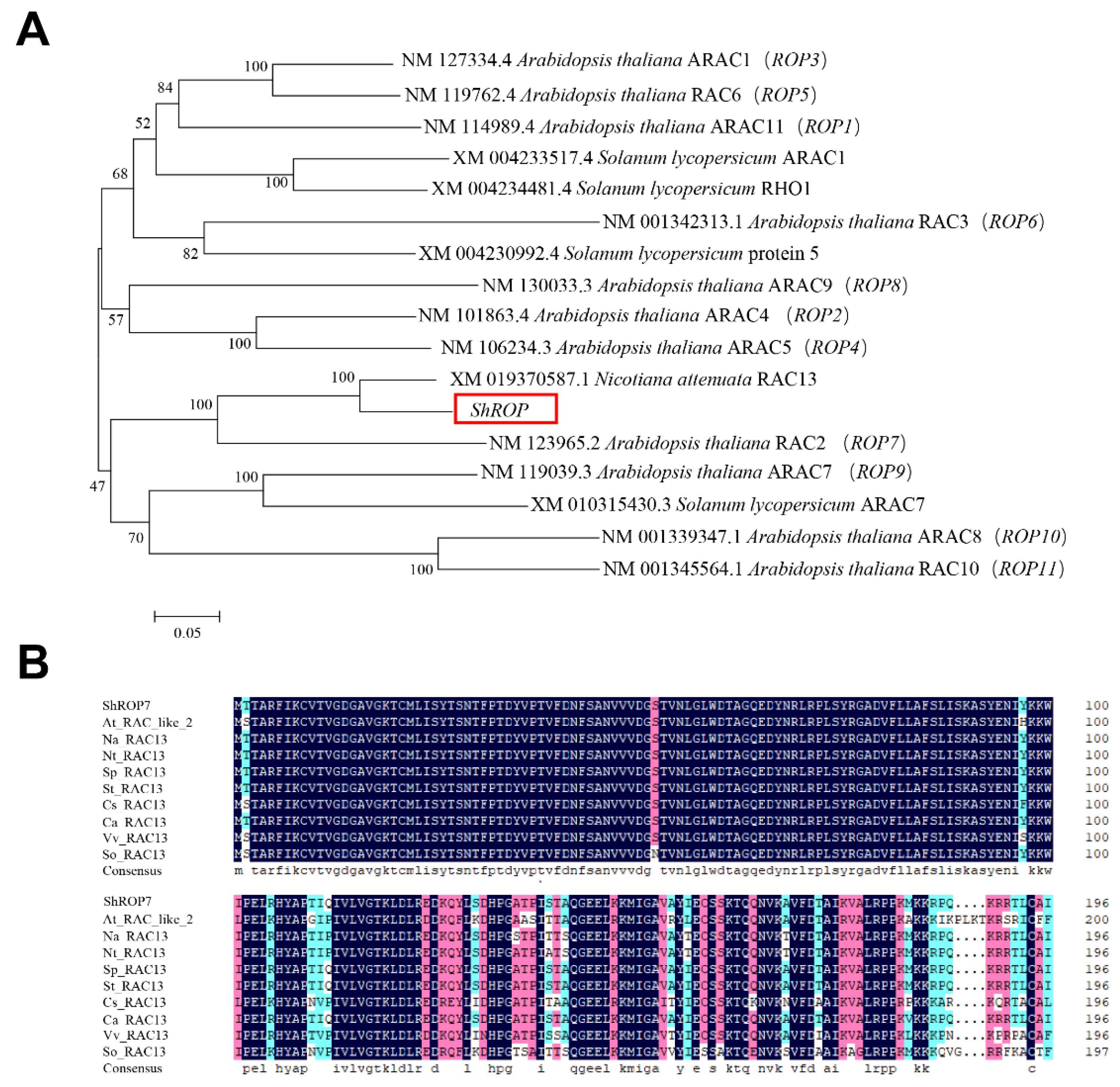
A tomato 594-bp homolog of Rac-like GTP binding protein 13 (Accession number Solyc02g077400.4.1) was isolated from tomato cv. LA1777 by homology-based cloning. The predicted CDS of ShROP encodes a protein of 197-amino acid with CaaL motif. Phylogenetic analysis of ShROP with other members of tomato ROPs and Arabidopsis thaliana ROPs (i.e., ROP1-11) showed that ShROP has the closest relationship with NaRAC13, with the next closest relationship being AtROP7. Based on the output of this analysis, we named the predicted protein ShROP7 (Figure 1A). The amino acid sequence of ShROP7 was compared with A. thaliana AtRAC-like2, Nicotiana attenuata NaRAC13, N. tabacum NtRAC13, Solanum pennellii SpRAC13, S. tuberosum StRAC13, Cucumis sativus CsRAC13, Capsicum annuum CaRAC13, Vitis vinifera VvRAC13, and Spinacia oleracea SoRAC13, and the compared results showed that the N-terminus of ShROP7 was highly conserved across the ROP/RAC superfamily (Figure 1B).
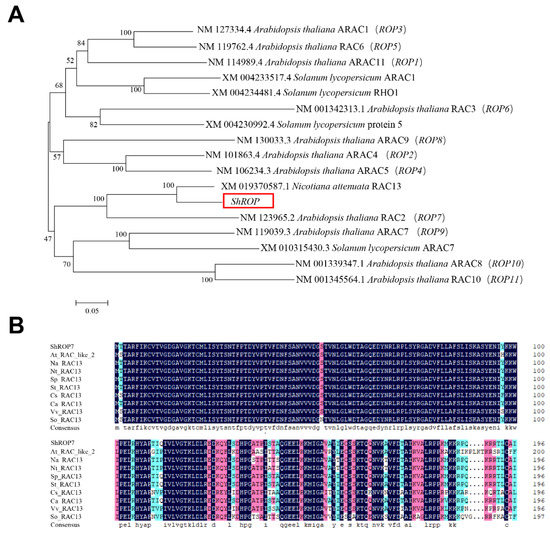
Figure 1.
Sequence analysis of Rac-like GTP binding protein ShROP in tomato. (A) Phylogenetic analysis of ShROP and other Rac-like GTP binding proteins. MEGA6.0 software was used to analyze phylogeny by proximity method. (B) Multiple sequence alignment between ShROP7 and other species RAC13.The amino acid sequence of ShROP7 was compared with Arabidopsis thaliana AtRAC-like2 (NP_199409.1), Nicotiana attenuata NaRAC13 (XP_019226132.1), Nicotiana tabacum NtRAC13 (XP_016451269.1), Solanum pennellii SpRAC13 (XP_015065147.1), Solanum tuberosum StRAC13 (XP_006367198.1), Cucumis sativus CsRAC13 (XP_004134675.1), Capsicum annuum CaRAC13 (XP_016562051.1), Vitis vinifera VvRAC13 (XP_002277471.1), and Spinacia oleracea SoRAC13 (XP_021863578.1).
2.2. ShROP7 Cannot Induce Cell Death nor Suppress BAX-Induced Necrosis
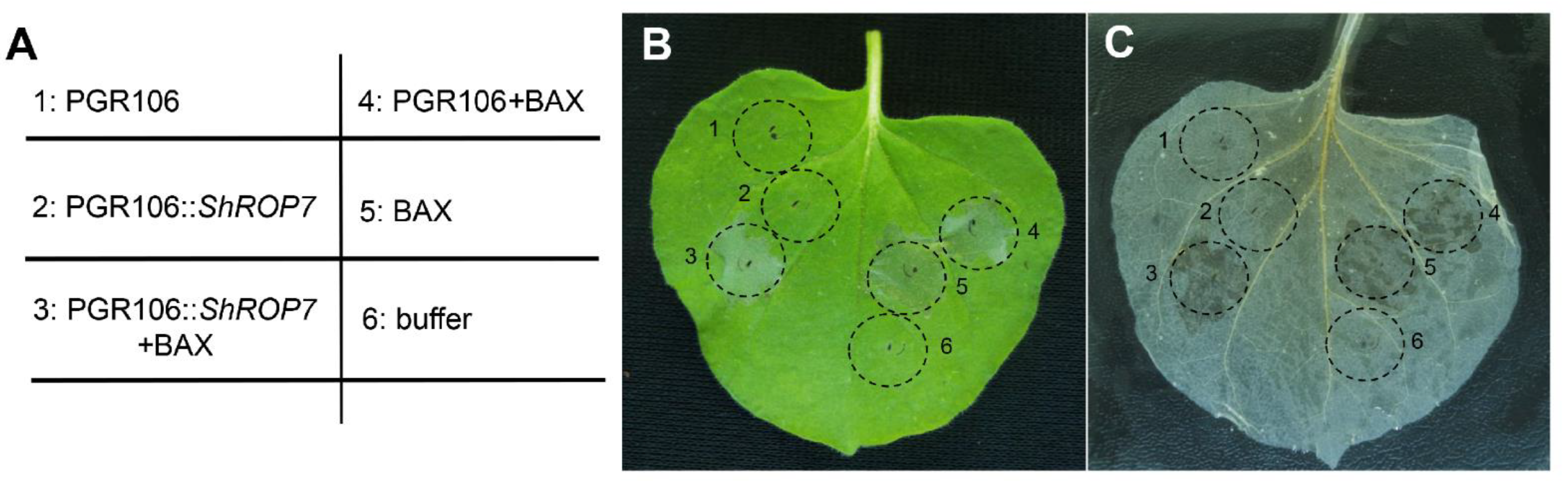
In order to define the role of ShROP7 during pathogen infection and disease resistance signaling, the PVX-based transient overexpression system in Nicotiana benthamiana leaves was used to evaluate ShROP7 function in cell death elicitation. Our aim was to evaluate the overexpression cell death activity of ShROP7 and/or its ability to suppress PCD. Agrobacterium harboring pGR106 (negative control), pGR106::ShROP7, and buffer (blank control) were infiltrated into tobacco leaves, respectively, and no cell death phenotypes were observed (Figure 2B,C; sites 1, 2 and 6). BAX, a cell death-promoting member of the Bcl-2 family of proteins that triggers cell death when expressed in plants [26], was used as positive control. At 24 hpi, BAX, pGR106::ShROP7 and pGR106 sites were injected with BAX to evaluate whether ShROP7 can suppress BAX-induced cell death (Figure 2A; sites 3, 4 and 5). At 5 days after being infiltrated with Agrobacterium, ShROP7 did not induce cell death, nor could it suppress BAX-triggered cell death (Figure 2B,C).
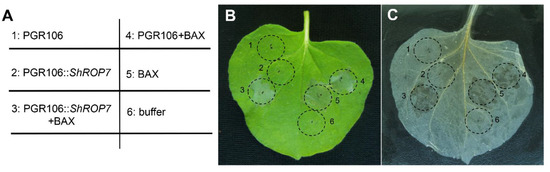
Figure 2.
ShROP7 does not induce cell death in tobacco. (A) Injection diagram of Agrobacterium infected tobacco leaves. (B) Typical phenotype of tobacco leaves injected with Agrobacterium were photographed at 5 days post-infection. (C) The same leaf was decolorized with absolute ethanol and glacial acetic acid (1:1). 1: PGR106, 2: PGR106::ShROP7, 3: PGR106::ShROP7+BAX, 4: PGR106+BAX, 5: BAX, 6: buffer.
2.3. ShROP7 Is Differentially Induced by On-Lz Infection
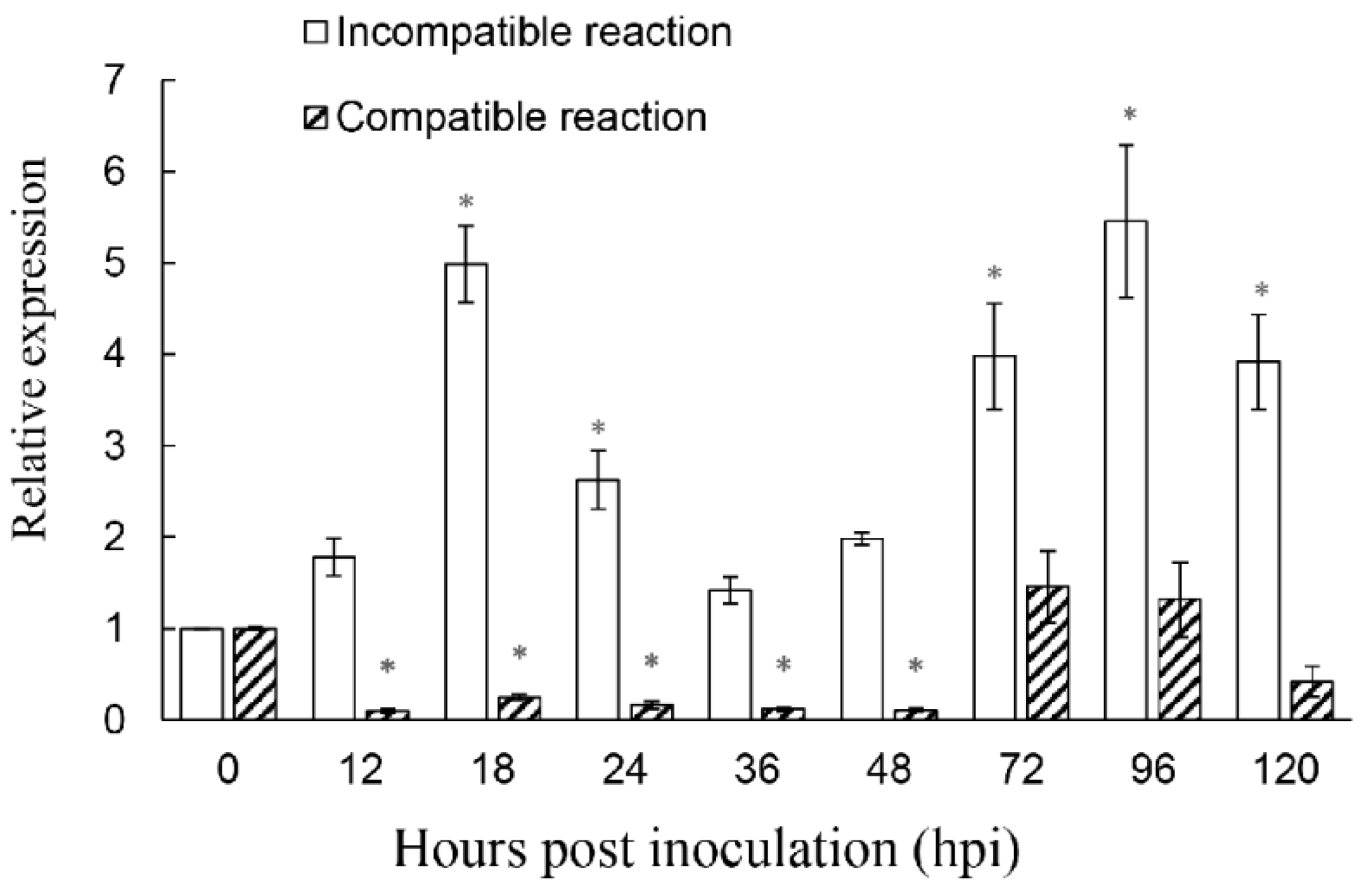
To determine the function of ShROP7 in the tomato-powdery mildew interaction, the mRNA accumulation of ShROP7 was monitored during the interaction between tomato and On-Lz at 0, 12, 18, 24, 36, 48, 72, 96, and 120 hpi. As shown in Figure 3, the expression of ShROP7 was significantly up-regulated at 18, 24, 72, 96 and 120 hpi during an incompatible reaction, in which tomato cv. LA1777 constitutes an incompatible reaction with On-Lz and peaked at 96 hpi (ca. 5.39-fold compared to 0 hpi). In addition, the accumulation of ShROP7 was significantly reduced at 18, 24, 36 and 48 hpi compared with 0 hpi in a compatible reaction, in which tomato cv. MM constitutes a compatible reaction with On-Lz. Therefore, ShROP7 is differentially induced by On-Lz infection.
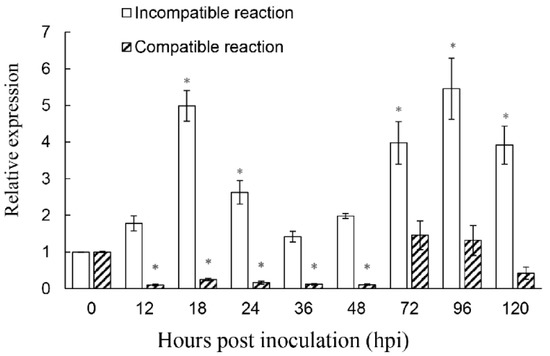
Figure 3.
The accumulation of ShROP7 mRNA is differentially induced in tomato leaves following inoculation with Oidium neolycopersici-LZ (On-LZ). Error bars represent the variation from three independent replicates. The asterisk (*) indicates a significant difference between inoculation timepoints and 0 hpi (p < 0.05).
2.4. Silencing of ShROP7 Enhances the Susceptibility of Tomato to On-Lz
Based on that the expression of ShROP7 increased in incompatible reaction after On-Lz infection, a tobacco rattle virus-induced gene silencing (TRV-VIGS)-based approach was used to silence ShROP7 in tomato cv. LA1777. A 275-bp fragment of ShROP7 was cloned into pTRV2 vector, and the expression of ShROP7 was reduced by the TRV-VIGS system to observe the function of ShROP7. The TRV2::ShROP7, TRV2::ShPDS (positive control) and TRV2 (negative control) were infiltrated in the four-leaf LA1777 and MM. Thirty days after inoculating with TRV2::ShPDS, the photo-bleaching symptoms were observed on the new leaves (Figure 4B,F), whereas no significant changes were observed on TRV2::ShROP7 (Figure 4C,G) and CK (Figure 4A,E) plants. Concurrently, the leaves at the same position were sampled in TRV2::ShROP7 and control plants, respectively. The expression level of ShROP7 was calculated by RT-qPCR, with the accumulation of ShROP7 being reduced by 68.9% (Figure 4D) in TRV2::ShROP7 plants compared with the control in LA1777 plants and reduced by 44.6% (Figure 4H) in MM plants, which can be used to verify the role of ShROP7 in the resistance to On-Lz.

Figure 4.
Silencing of ShROP7 by TRV-VIGS. Phenotypes of control (CK) (A), TRV2::ShPDS (B) and TRV2::ShROP7 (C) tomatoes after 30 days post-infection with Agrobacterium. (D) Expression level of ShROP7 in silenced plants (LA1777). Phenotypes of CK (control) (E), TRV2::ShPDS (F) and TRV2::ShROP7 (G) tomatoes after 30 days post infected with Agrobacterium in MM plants. (H) Expression level of ShROP7 gene in silenced plants (MM). The asterisk (*) indicate significant differences of disease index between control (CK) and TRV2::ShROP7 plants (p < 0.05).
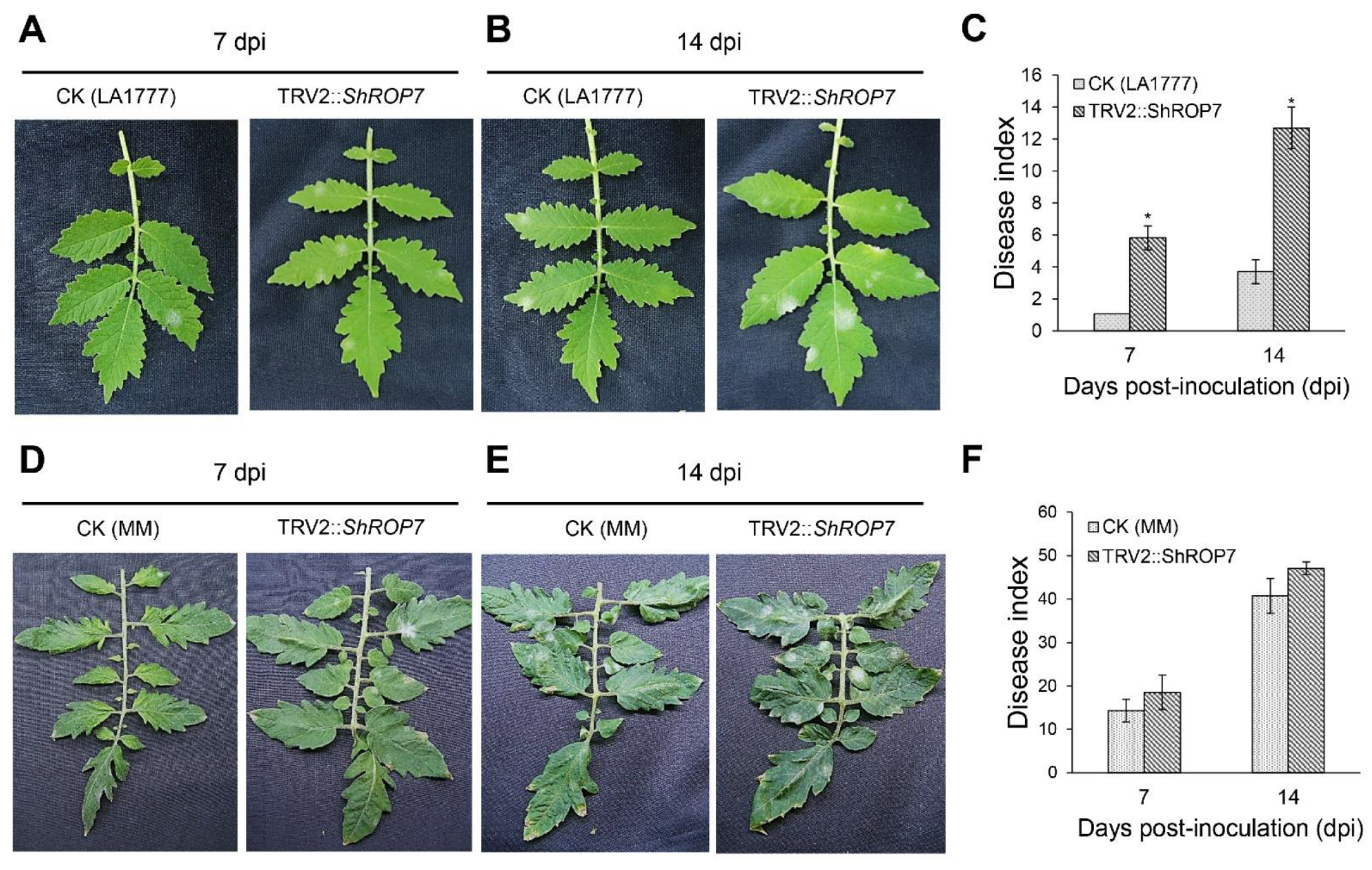
When TRV2::ShPDS plants showed robust signs of photo-bleaching, leaves of ShROP7-silenced and control plants were inoculated with On-Lz. The infection phenotypes of the tomato leaves were observed, and disease indexes were recorded at 7 and 14 dpi, respectively. In LA1777 silenced plants, as shown in Figure 5A, the leaves of CK and TRV2::ShROP7 developed reduced areas of powdery mildew disease spot lesions, while the disease indexes of TRV2::ShROP7 plants were significantly higher than control plants at 7 dpi (Figure 5C). At 14 dpi, obvious powdery mildew disease lesions were produced on the infected leaves, and ShROP7-silenced plants produced more disease spot lesions compared with control (Figure 5B), with the disease index of TRV2::ShROP7 plants being 12.7, which was significantly higher than that of the control plants (Figure 5C). On the contrary, the disease indexes of CK and TRV2::ShROP7 had no significant change in MM plants (Figure 5 D–F).
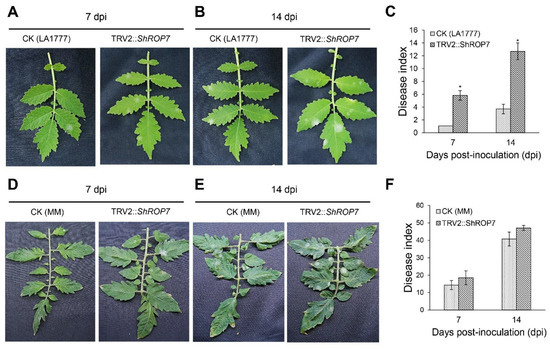
Figure 5.
ShROP7 is required for resistance to powdery mildew. Genetic analysis of resistance response and disease development using TRV-induced gene silencing in tomato. Phenotypes of CK (control) and TRV2::ShROP7 plants inoculated with On-LZ at 7 (A) and 14 (B) dpi in LA1777 plants and at 7 (D) and 14 (E) dpi in MM plants. Disease index was calculated for CK and TRV2::ShROP7 plants at 7 and 14 dpi in LA1777 (C) and MM (F) plants. The asterisk (*) indicate significant differences of disease index between control (CK) and TRV2::ShROP7 plants (p < 0.05).
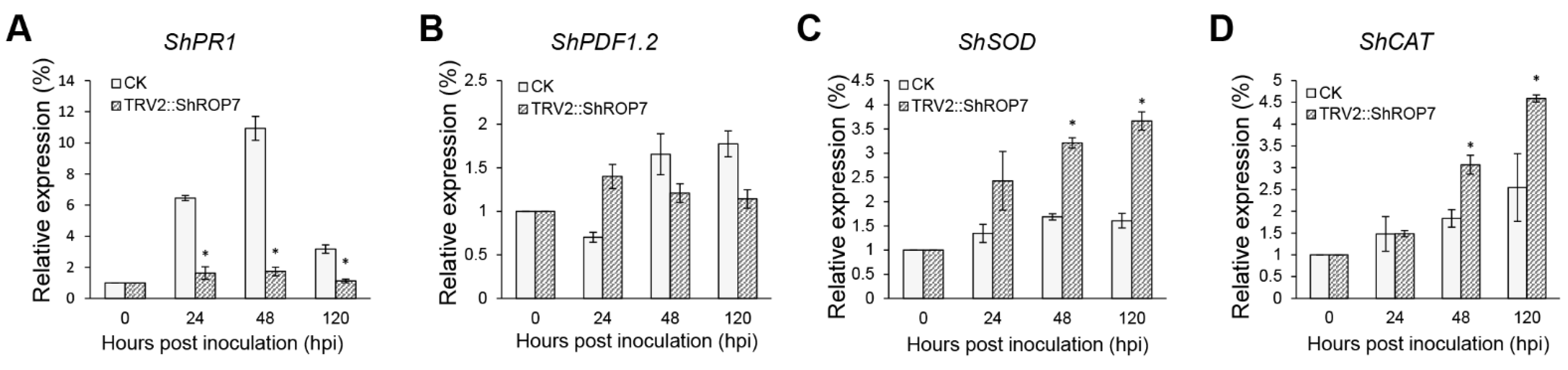
To determine whether ShROP7 regulates tomato resistance to powdery mildew through SA or JA signaling pathways, the expression of the SA-dependent disease resistance signal pathway marker gene ShPR1 and the JA-dependent disease resistance signal pathway marker genes were evaluated. As shown in Figure 6A,B, the expression of ShPR1 was significantly reduced in ShROP7-silenced plants compared with control plants at 24, 48 and 120 hpi. The mRNA accumulation of ShPDF1.2 had no significant change, indicating that tomato LA1777 is likely transduced through the SA signaling pathway as opposed to the JA pathway, to resist powdery mildew infection.
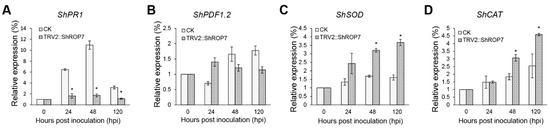
Figure 6.
Real-time quantitative PCR (RT-qPCR) analyses of expression of ShPR1 (A), ShPDF1.2 (B), ShSOD (C) and ShCAT (D) genes in CK (LA1777) and TRV2::ShROP7 plants inoculated with On-LZ. Error bars represent the variations among three independent replicates. The asterisk (*) indicates a significant difference compared with 0 hpi (p < 0.05).
2.5. Histological Observation of LA1777 ShROP7-Silenced Plants
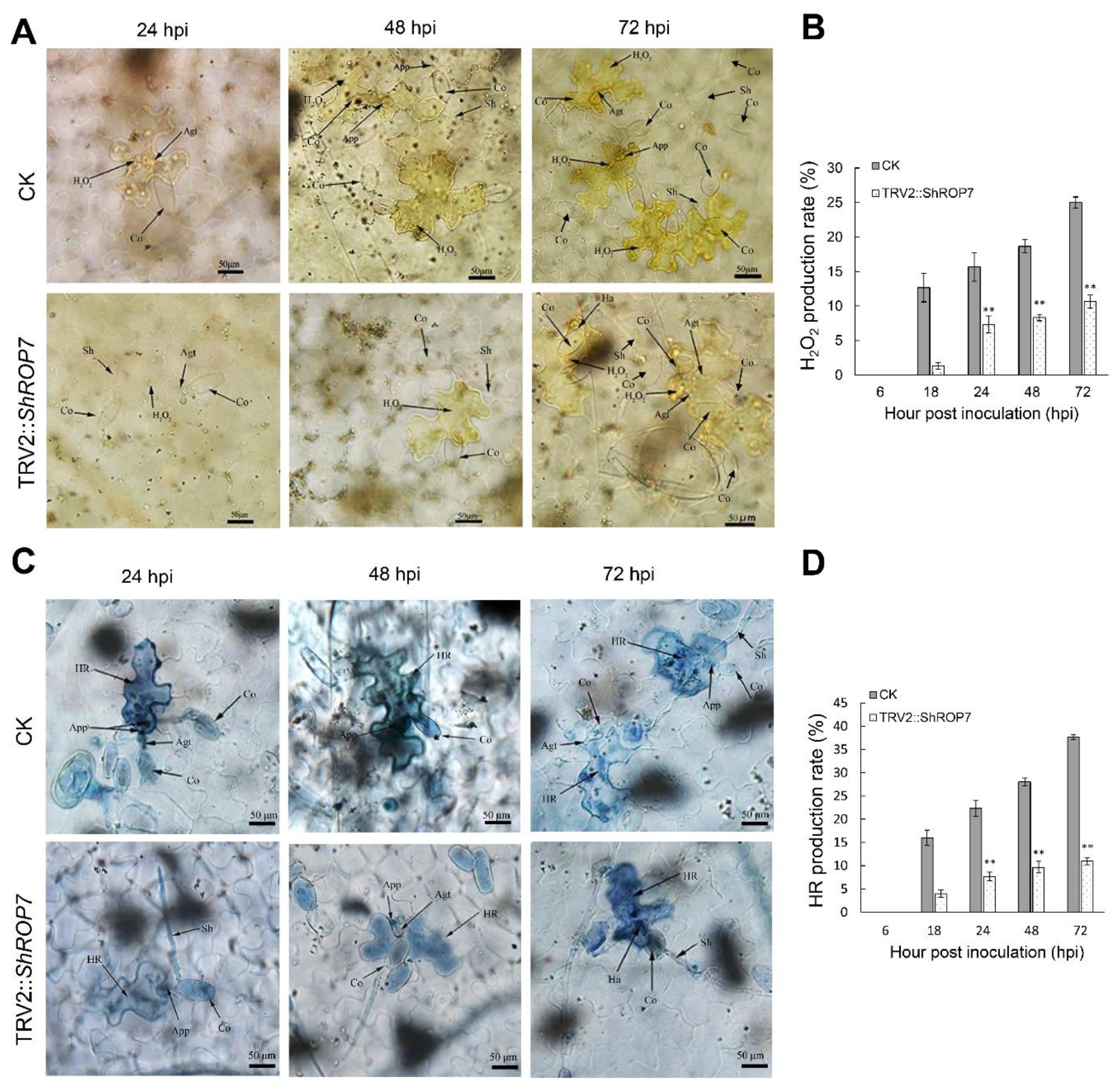
To further define the resistance changes of tomato infected by On-Lz after silencing ShROP7, the expression levels of ShCAT and ShSOD were monitored. As shown in Figure 6C,D, the mRNA accumulation of H2O2 pathway related genes ShCAT and ShSOD were significantly induced at 48 and 120 hpi. Additionally, we measured the production rate of H2O2 and hypersensitive response (HR) at 6, 18, 24, 48, and 72 hpi in LA1777 ShROP7-silenced and control plants. After DAB staining, and as shown in Figure 7A,B, the production of H2O2 was not observed at 6 dpi, and a small number of H2O2 were produced at 18 hpi. At 24, 48, and 72 hpi, the production rate of H2O2 in TRV2 plants was significantly higher than that of silenced plants (P < 0.01) (Figure 7B). In ShROP7-silenced plants, the production rate of H2O2 was increased gradually as the infection progressed, being 7.3%, 8.3% and 10.7% at 24, 48 and 72 hpi, respectively. Similarly, the production rate of H2O2 increased rapidly in the control plants, which reached 25% at 72 hpi. As a second, parallel, HR, cell death was stained with trypan blue. As shown in Figure 7C,D, the production of HR was not observed at 6 dpi. At 18 hpi, there were no obvious differences between TRV2 and TRV2::ShROP7 plants in HR production. With the increase of haustoria formation at 24 hpi, the elicitation of the HR was 22.3% in TRV2-silenced plants, which was significantly higher than in ShROP7-silenced plants (ca. 7.7%). At 48 hpi, the elicitation of the HR in control plants was approximately 28%, 2.9 times higher than in TRV2::ShROP7 plants. At 72 hpi, the rate of HR induction in control plants was 37.7%, which was significantly higher than the rate of ShROP7-silenced plants (ca. 11%). Taken together, these results indicate that a reduction in ShROP7 expression compromises resistance signaling in response to On-Lz infection.
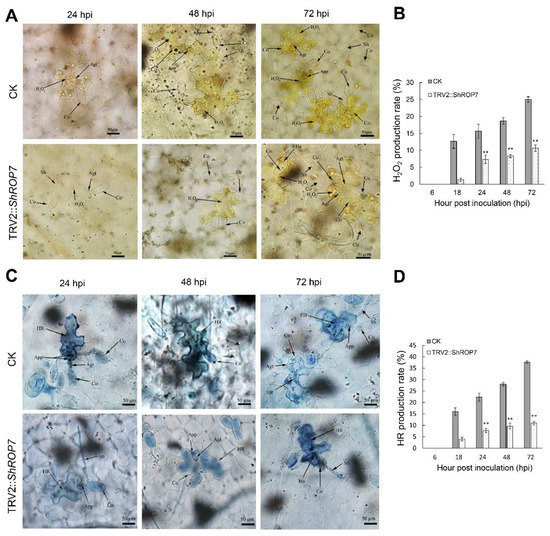
Figure 7.
Silencing of ShROP7 reduced defense responses in tomato following powdery mildew infection. (A) Histological observation of H2O2 accumulation in CK and ShROP7-silenced plants inoculated with On-LZ at 24, 48 and 72 hpi in LA1777 plants. (B) The H2O2 production rate was calculated at 6, 18, 24, 48 and 72 hpi. (C) Histological observation of hypersensitive cell death in TRV2 and ShROP7-silenced plants inoculated with On-LZ at 24, 48 and 72 hpi in LA1777 plants. Blue (trypan) staining indicates hypersensitive cell death. (D) The HR production rate was calculated at 6, 18, 24, 48, and 72 hpi. Error bars represent the variations among three independent replicates. Asterisks (**) indicates statistically significant differences between TRV2 and TRV2::ShROP7 plants at each time point (p < 0.01). Co, conidium; App, appressorium; Agt, appressorium germ tube; Sh, secondary hyphae; Ha, haustorium; Sa, secondary appressorium; HR, hypersensitive response. Bar, 50 μm.
2.6. Screening and Identification of ShROP7 Interaction Proteins
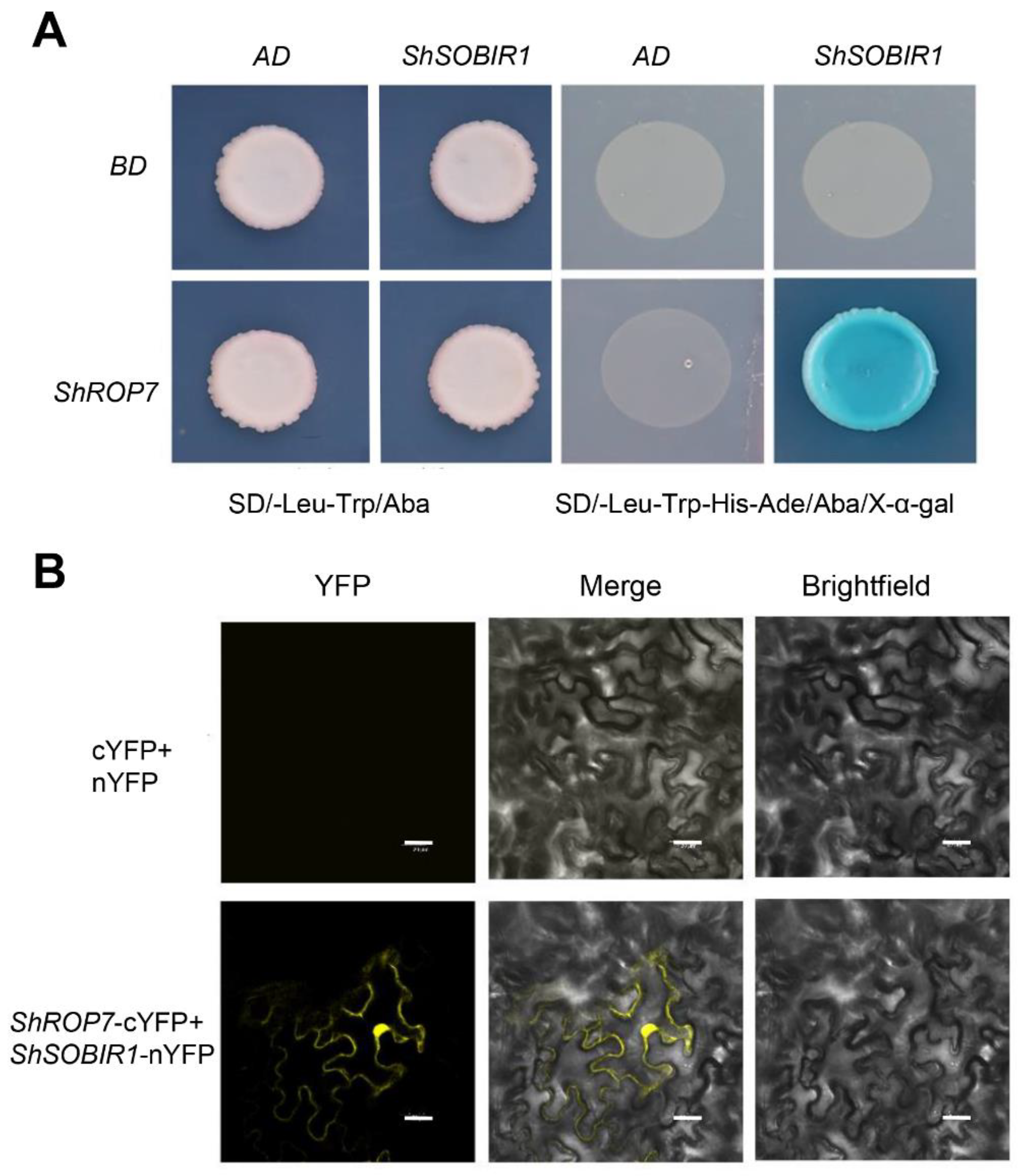
In the current study, tomato cv. LA1777 leaves infected by On-Lz were used to construct a yeast two-hybrid cDNA library of tomato-powdery mildew incompatible interaction, with the aim of screening out proteins interacting with ShROP7. The CDS sequence of ShROP7 was cloned into BD vector as bait and was transformed into the yeast strain Y2H Gold. After testing the bait construct for autoactivation and toxicity, the cDNA library was screened using ShROP7 as a bait. The development of blue yeast colonies on SD/-Leu-Trp-His-Ade+Aba+X-α-Gal medium was used as a selection criterion for putative positive interacting proteins following the yeast protocols handbook (Takara, Dalian, China). Using this approach, we identified 35 candidate proteins that interact with ShROP7. Among the candidate interacting proteins, we identified and confirmed interaction between ShROP7 and SOBIR1, wound-induced proteinase inhibitor 1, glutathione peroxidase (GSHPx), pectin acetylesterase (PAEs) and chitinase 3 related to plant defense response; eukaryotic initiation factor 4A-2, tyrosine-protein kinase 3, serine/threonine-protein kinase sty46 ferredoxin, thioredoxin, MAPK16, E3 ubiquitin protein related to cell signaling (Table 1). Herein, we focused on SOBIR1, a receptor-like kinases (RLKs) protein containing a signal domain, an extracellular leucine rich repeat (LRR) domain, transmembrane domain and serine/threonine kinase domain, which is a key enzyme that interacts with both Cf-4 and Ve1 to require for the Cf-4- and Ve1-mediated HR in tomato and Arabidopsis thaliana, respectively [27]. The CDS sequence of ShSOBIR1 without a signal peptide (25–638 amino acid) was inserted into the yeast two-hybrid AD vector and the reconstituted ShROP7+ShSOBIR1 and negative controls (AD+BD, AD+ShROP7 and BD+ShSOBIR1) were transformed into yeast strain Y2H Gold. As shown in Figure 8A, the transformants were grown on SD/-Trp-Leu+Aba medium, indicating successful co-transformation. In addition, only ShROP7+ShSOBIR1 containing yeast cells developed blue colonies on SD/-Leu-Trp-His-Ade+Aba+X-α-Gal medium, indicating that ShROP7 and ShSOBIR1 can activate the GAL4 yeast expression system.

Table 1.
Protein interactions of ShROP7 identified from yeast two-hybrid screen.
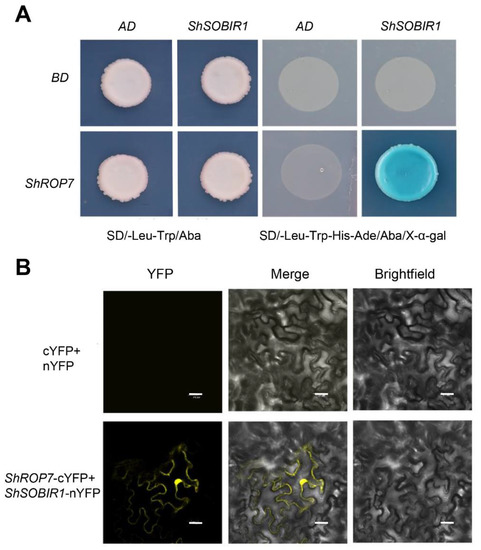
Figure 8.
ShROP7 interacts with ShSOBIR1. (A) Interaction of ShROP7 and ShSOBIR1 in a yeast two-hybrid system. (B) Bimolecular fluorescence complementation (BiFC) analysis of the interaction between ShROP7 and ShSOBIR1. Bars = 20 μm.
To further confirm the interaction between ShSOBIR1 and ShROP7, we conducted bimolecular fluorescence complementation (BiFC) to verify the in vivo interaction. The ORFs of ShROP7 and ShSOBIR1, without a signal peptide, were inserted into pSPYCE and pSPYNE, respectively. The recombinant plasmids were transferred to Agrobacterium resultant transformants which were infiltrated into tobacco leaves. A 35S-pSPYNE+35S-pSPYCE was used as a negative control. After 48–72 h, Agrobacterium-infiltrated cells were evaluated and yellow fluorescence was observed by laser confocal microscopy. As shown in Figure 8B, we observed that ShROP7 and ShSOBIR1 co-localized to the nucleus and on the cell membrane. In short, this result supports the presence of an interaction between ShROP7 and ShSOBIR1 in planta.
3. Discussion
Small GTP-binding proteins exist ubiquitously in eukaryotes and constitute a superfamily. As molecular switches, they regulate a series of physiological processes including signaling transduction, cell proliferation, composition of cytoskeleton, cell membrane transporting and gene expression [28]. The ROPs subfamily of small GTP-binding proteins transmits a variety of extracellular and intracellular signals, which can amplify specific signals by acting as the main switch in the early stage of signal cascade [2]. In Arabidopsis, AtROP2 and AtROP11 increase resistance to Pseudomonas syringae pv. tomato (Pst) DC3000 in transgenic overexpression plants. However, AtROP10 has the opposite effect on resistance to Pst [29]. Together, these studies show that ROPs play both positive and negative roles in the establishment of plant defense. At present, there are few reports about ROPs participating in tomato powdery mildew resistance, and the disease resistance mechanisms of ROPs remain largely undefined.
The ROP family contains a small GTP-binding protein domain, containing five highly conserved G-boxes, one of which, the G-box, functions to bind effectors. The other four G-boxes function in binding GTP/GDP and the hydrolysis of GTP to GDP [30]. Functionally, the C-terminus of plant ROPs is highly variable. Related to their activity and function as GTPases, which are divided into type I and II types, type I ROPs terminate with a conserved CaaL motif and undergo prenylation, whereas type II ROPs lack the CaaL motif and undergo S-acylation [31]. Further, type I ROPs are divided into subtypes 1 and 3, while type II ROPs constitute subtype 2. Among the 11 Arabidopsis ROPs, AtROP7 and AtROP8 are the subtype 1 of ROPs, subtype 3 includes AtROP1- AtROP6, and subtype 2 includes AtROP9- AtROP11 [32]. In this study, the protein sequence of ShROP was found to have the closest relationship with NaRAC13, with the next closest relationship being with AtROP7. In addition, based on the presence of a CaaL motif within the C-terminus of the protein, we conclude that ShROP7 belongs to the type I ROP family.
ROP7 is one of the original members of the ROP family, hypothesized to have evolved with the emergence of the plant xylem [33]. Indeed, AtRAC2/ROP7 has been found to be related to the formation of secondary walls in the primary xylem of plants, and overexpression of AtROP7 shortens the leaf length, indicating that AtROP7 plays an important role in the formation of the plant actin cytoskeleton [34]. Additional studies have shown that AtROP7 is stimulated by phytochrome B, required for the opening and closing movement of stomata [35]. Herein, RT-qPCR results revealed that the expression of ShROP7 was significantly up-regulated in an incompatible reaction, which was required for tomato resistance to powdery mildew. To support this, we observed that the silencing of ShROP7 enhanced the susceptibility of tomato cv. LA1777 to On-Lz, with disease indexes being significantly higher than those of control plants in tomato cv. LA1777 ShROP7-silenced plants at 7 and 14 days after inoculating with On-Lz. Taken together, these data support the hypothesis that ShROP7 positively regulates tomato resistance to powdery mildew.
Among the fastest defense responses activated following pathogen infection is the generation of reactive oxygen species (ROS), a process required for the induction of the HR and the abrogation of pathogen infection and proliferation. Mechanistically, the HR reaction results in an oxidative burst, producing reactive oxygen intermediates (ROIs) that contain hydrogen peroxide (H2O2), superoxide radical anions (O−2) and hydroxyl radical (OH−) [36]. Herein, we observed that the production of H2O2 and HR were both significantly reduced in tomato infected by On-Lz after silencing ShROP7. Moreover, we observed that the expression of H2O2 pathway related genes ShSOD and ShCAT were induced in ShROP7 plants. These observations suggest that ShROP7 is required for the regulation of broad defense signaling processes in tomato required resistance to powdery mildew.
The data presented above confirm that the overexpression of ShROP7 does not induce localized cell death, nor does it inhibit cell death caused by BAX. However, our results do affirm that the silencing of ShROP7 significantly reduces the generation of H2O2 and the formation rate of HR, both of which are required for robust immune signaling and defense. To investigate the mechanism(s) through which ShROP7 regulates the activation of the HR response and ROS accumulation, we sought to identify specific protein–protein interactions with ShROP7 which might regulate these cascades. Through this approach, we identified a specific interaction between ShROP7 and ShSOBIR1, an RLK protein which has been shown to regulate broad host defense signaling processes. For example, the silencing of SOBIR1 and SOBIR1-like genes has been shown to affect pathogen-induced callose deposition, the production of reactive oxygen species, and the expression of PTI marker genes. Further studies have narrowed this function to demonstrate that the kinase domain of SOBIR1 is essential for eliciting the (ParA1)-based induction of SOBIR1 cell death [37]. Additionally, several proteins, such as OsMAPK6, CERK1, GEF1, and SPL11, have been shown to be associated with ROP-based signaling during host–pathogen interactions [38,39,40].
In conclusion, we demonstrate that ShROP7 is a type I ROP that is required for robust immune responses in tomato following infection with the powdery mildew pathogen On-Lz. Through its interaction with ShSOBIR1, ShROP7 plays a role in governing the level of tomato resistance to On-Lz by affecting the generation rate of H2O2 and the rate of HR development. Taken together, the data presented herein further our understanding of the role of the ROP family in regulating disease resistance signaling in plants.
4. Materials and Methods
4.1. Plant and Pathogen Materials and Growth
Tomato cultivar (cv.) Moneymaker (MM) (Solanum lycopersicum) and S. habrochaites LA1777 were used in this research. Oidium neolycopersici (On) Lanzhou strain (On-Lz) was propagated and preserved according to the method of Sun et al. [41]. The tomato cv. MM and LA1777 were grown at 25 °C, with a day/night cycle of 16 h/8 h. LA1777 is a highly resistant tomato powdery mildew cultivar and constitutes an incompatible reaction with On-Lz, while obvious powdery mildew disease lesions appeared in tomato cv. MM and constitute a compatible reaction with On-Lz. Tomato seeds were surface sterilized with 3% sodium hypochlorite for 3 min, and then immediately rinsed with distilled water three times [41]. After surface sterilization, seeds were cultivated and germinated in vermiculite seedling trays, and grown for one week under conditions of 25 ± 1 °C with 90% relative humidity. After the emergence of the cotyledon, seedlings were transplanted into 15 cm pots.
Tobacco (Nicotiana benthamiana) was grown at 25 °C under a 14 h light/10 h dark cycle. After 4–6 weeks, tobacco leaves were inoculated with Agrobacterium tumefaciens strain GV3101 expressing pGR106-gene, pSPYCE-gene, and pSPYNE-gene vectors for protein overexpression or Bimolecular fluorescence complementary (BiFC) analysis, respectively.
Escherichia coli strain DH5α was grown on Luria–Bertain (LB) solid medium at 37 °C. Agrobacterium tumefaciens strain GV3101 was grown on LB media containing rifampicin and gentamicin at 28 °C. Saccharomyces cerevisiae strain Y2H Gold with four reporter genes (AbAr, HIS3, ADE2 and MEL1) was grown on YPDA (1% yeast extract, 2% peptone, 2% dextrose, 0.02% adenine) medium at 37 °C.
4.2. Identification and Sequence Analysis of ShROP
Tomato Rac-like GTP binding protein-13 (Solyc02g077400.4.1) amino acid sequence was used as query in the SGN Tomato Combined database (https://solgenomics.net/tools/blast/, accessed on 9 December 2019), the BLASTP program against an Arabidopsis protein database (http://www.arabidopsis.org/Blast/index.jsp, accessed on 20 December 2019) and the NCBI BLASTN of GenBank (https://blast.ncbi.nlm.nih.gov/Blast.cgi, accessed on 20 December 2019) to search for homologous sequences. DNAMAN 6.0 software (Lynnon BioSoft) was used for sequence comparisons with default settings. Phylogenetic analysis was performed with the MEGA 6.0 software package (http://www.megasoftware.net/, accessed on 5 January 2020) using the proximity method. Protein domains were predicted using the SMART (http://smart.embl-heidelberg.de/, accessed on 10 January 2020) and UniProt protein databases (https://www. uniprot.org, accessed on 10 January 2020).
4.3. Agrobacterium-Mediated Transient Overexpression in Tobacco (Nicotiana Benthamiana)
For transient overexpression in the tobacco, DNA primers (Table S1) were used to clone the full-length ShROP7 cDNA via SalI digestion and ligation. The intelligent seamless cloning kit (Takara, Japan) was used to link the target gene to pGR106. The recombinant plasmids pGR106::ShROP7 and pGR106 (negative control) were transformed into the A. tumefaciens strain GV3101. For A. tumefaciens-tobacco assays, BAX was used as the positive control, pGR106 served as the negative control, and buffer was the blank control. At 24 hpi, the same infiltration sites were challenged with BAX at pGR106::ShROP7+BAX (3) and pGR106+BAX (4). Representative leaves were photographed at 5 dpi. Experiments were repeated at least two times with similar results.
4.4. Real-Time Quantitative PCR (RT-qPCR) Analysis
Tomato cv. LA1777 and MM seedlings were inoculated with On-Lz, and samples were taken at 0, 12, 18, 24, 36, 48, 72, 96, and 120 hpi. Total RNA was extracted according to the bioZOL total RNA extraction kit (Invitrogen, Carlsbad, CA, USA). Reverse transcription of first-strand cDNA was performed according to the manufacturer’s instructions (FastKing RT Kit; Tiangen, Beijing, WI, China). The total reaction system and RT-qPCR program were performed with the UltraSYBR Mixture kit (KangWei, Guangzhou, China). The 2−∆∆CT method was used to calculate relative expression levels. Tomato GAPDA was used as a reference gene for expression. Three biological replicates and three technical replicates were performed for each treatment. SPSS 20.0 was used to determine the statistical significance (p < 0.05) [42].
4.5. TRV2-Mediated Silencing of ShROP7 in Tomato
The tobacco rattle virus (TRV1 and TRV2) was used for the silencing of ShROP7 in tomato cv. LA1777 and MM. A 248-bp fragment of ShROP7 was constructed into pTRV2 vector via BamH1 digestion and ligation. After the recombinant plasmid was successfully constructed, TRV1 and TRV2-as (anti-sense orientation) were transferred into Agrobacterium tumefaciens strain GV3101 respectively, and were cultured and infiltrated as previously described [43]. TRV2::ShPDS (phytoene desaturase; sequence accession number NM_001247166) was the positive control. Thirty days after virus inoculation, with the photo-bleaching of positive control plants being observed, the expression level of the ShROP7 gene was calculated to detect the silencing efficiency.
Once ShROP7 was silenced, together with the negative control and the ShROP7-silenced plants, plants were inoculated with On-Lz. The infection phenotypes of tomato leaves were observed and disease index (DI) was evaluated at 7 and 14 dpi. Disease severity was scored according to the following: 0 = no diseased leaves; 1 = 0–5% of leaves having lesions; 3 = 6–10% of leaves having lesions; 5 = 11–20% of leaves having lesions; 7 = 21–40% of leaves having lesions; and 9 = 41–100% of leaves having lesions. The equation of the disease index was calculated as described by Sun et al. [41]. Three biological replicates were performed, with seven leaves evaluated at each timepoint.
For histological observation, ShROP7-silenced plants (LA1777) were sampled at 6, 18, 24, 48, and 72 hpi. Samples at each timepoint were stained with 1 mg/mL 3,3-diaminobenzidine (DAB; AMERCO, Solon, OH, USA) for observation of H2O2 accumulation, and were stained in a 0.05% trypan blue solution to observe the induction of HR cell death. The Olympus BX-51 microscope (Olympus Corp, Tokyo) was used to count the reactive oxygen generation rate (H2O2 production rate = H2O2 numbers per 100 penetration sites) and the formation rate of HR cell death (HR production rate = HR numbers per 100 penetration sites). Three to five leaves were observed and at least 50 penetration sites were counted at each timepoint. Standard errors of deviation were calculated using Microsoft Excel. Statistical significance was assessed by a Student’s t-test (P < 0.01) using SPSS 20.0 software [43]. ShROP7-silenced plants (LA1777) were sampled at 0, 24, 48, and 120 hpi for isolating total RNA to analyze the effects of gene expression on plant resistance.
4.6. Yeast Two-Hybrid and Bimolecular Fluorescence Complementation (BiFC) Analysis
The full-length sequence of ShROP7 was cloned into pGBKT7 (BD) and was used as a bait construct. A cDNA library, constructed with tomato cv. LA1777 infected with On-Lz, was transformed into the yeast strain Y2H Gold containing pGBKT7-ShROP7 to screen candidate interaction proteins following the yeast protocols handbook (Takara, Dalian, China). After selection on SD/-Leu-Trp+Aureobasidin (Aba) and SD/-Leu-Trp-His-Ade+Aba+X-α-galactosidase (X-α-Gal) medium, the candidate protein sequences were identified by PCR reaction and were analyzed and compared using the NCBI BLAST tool (Table S2). The ORF sequence of ShSOBIR1 without a signal peptide was inserted into pGADT7 (AD) vector. The plasmids of ShROP7+ShSOBIR1 and the negative controls (BD+AD, ShROP7+AD, BD+ShSOBIR1) were co-transformed into yeast, respectively, and were grown on SD/-Leu-Trp+Aba and SD/-Leu-Trp-His-Ade+Aba+X-α-Gal medium to further verify the interaction of yeast two-hybrid.
The full-length sequence of ShROP7 and the ORF sequence of ShSOBIR1 without signal peptide were cloned into pSPYCE and pSPYNE, respectively, for BiFC. The plasmids of ShROP7-cYFP and ShSOBIR1-nYFP were transformed into A. tumefaciens GV3101 for N. benthamiana expression. At 48–72 h after Agro-infection, YFP fluorescence was observed using confocal laser scanning microscopy (Olympus FV1000MPE) equipped with a 488 nm filter.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms23158557/s1.
Author Contributions
Y.M. and A.Z., performed the experiments and analyzed data. L.X. and Q.M., guided the operation of the experiment. Y.M. assisted with writing. Y.M. and L.X. were major contributors in writing the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Key R&D Project of Shaanxi Province (grant no. 2019 ZDLNY03-07).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data supporting the reported results can be found in the Supplementary Materials file.
Acknowledgments
We thank Brad Day (Michigan State University) for critical reading and revision of this article.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Schultheiss, H.; Hensel, G.; Imani, J.; Broeders, S.; Sonnewald, U.; Kogel, K.; Kumlehn, J.; Hückelhoven, R. Ectopic expression of constitutively activated RACB in barley enhances susceptibility to powdery mildew and abiotic stress. Plant Physiol. 2005, 139, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Takai, Y.; Sasaki, T.; Matozaki, T. Small GTP-binding proteins. Physiol. Rev. 2001, 81, 153–208. [Google Scholar] [CrossRef] [PubMed]
- Winge, P.; Brembu, T.; Bones, A.M. Cloning and characterization of rac-like cDNAs from Arabidopsis thaliana. Plant Mol. Biol. 1997, 35, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Brembu, T.; Winge, P.; Bones, A.M.; Yang, Z. A RHOse by any other name: A comparative analysis of animal and plant Rho GTPases. Cell Res. 2006, 16, 435–445. [Google Scholar] [CrossRef]
- Berken, A. Rops in the spotlight of plant signal transduction. Cell. Mol. Life Sci. 2006, 63, 2446–2459. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Akamatsu, A.; Hayashi, K.; Housen, Y.; Okuda, J.; Yao, A.; Nakashima, A.; Takahashi, H.; Yoshida, H.; Wong, H.L. Activation of a Rac GTPase by the NLR family disease resistance protein Pit plays a critical role in rice innate immunity. Cell Host Microbe 2010, 7, 362–375. [Google Scholar] [CrossRef] [PubMed]
- Kawano, Y.; Shimamoto, K. Early signaling network in rice PRR-mediated and R-mediated immunity. Curr. Opin. Plant Biol. 2013, 16, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Schiene, K.; Pühler, A.; Niehaus, K. Transgenic tobacco plants that express an antisense construct derived from a Medicago sativa cDNA encoding a Rac-related small GTP-binding protein fail to develop necrotic lesions upon elicitor infiltration. Mol. Gen. Genet. 2000, 263, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z. Small GTPases: Versatile signaling switches in plants. Plant Cell 2002, 14, 375–388. [Google Scholar] [CrossRef]
- Kawano, Y.; Chen, L.; Shimamoto, K. The function of Rac small GTPase and associated proteins in rice innate immunity. Rice 2010, 3, 112–121. [Google Scholar] [CrossRef]
- Łaźniewska, J.; Macioszek, V.K.; Kononowicz, A.K. Plant-fungus interface: The role of surface structures in plant resistance and susceptibility to pathogenic fungi. Physiol. Mol. Plant Pathol. 2012, 78, 24–30. [Google Scholar] [CrossRef]
- Tang, D.; Wang, G.; Zhou, J. Receptor kinases in plant-pathogen interactions: More than pattern recognition. Plant Cell 2017, 29, 618–637. [Google Scholar] [CrossRef]
- Wu, Y.; Zhou, J. Receptor-like kinases in plant innate immunity. J. Integr. Plant Biol. 2013, 55, 1271–1286. [Google Scholar] [CrossRef] [PubMed]
- Jaffe, A.B.; Hall, A. Rho GTPases: Biochemistry and biology. Annu. Rev. Cell Dev. Biol. 2005, 21, 247–269. [Google Scholar] [CrossRef] [PubMed]
- Wong, H.L.; Sakamoto, T.; Kawasaki, T.; Umemura, K.; Shimamoto, K. Down-regulation of metallothionein, a reactive oxygen scavenger, by the small GTPase OsRac1 in rice. Plant Physiol. 2004, 135, 1447–1456. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, X.; Zhuang, R.; Wei, Z.; Shu, W.; Wang, X.; Kang, Z. TaRac6 is a potential susceptibility factor by regulating the ROS burst negatively in the wheat–Puccinia striiformis f. sp. tritici interaction. Front. Plant Sci. 2020, 11, 716. [Google Scholar] [CrossRef] [PubMed]
- Ono, E.; Wong, H.L.; Kawasaki, T.; Hasegawa, M.; Kodama, O.; Shimamoto, K. Essential role of the small GTPase Rac in disease resistance of rice. Proc. Natl. Acad. Sci. USA 2001, 98, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, T.; Henmi, K.; Ono, E.; Hatakeyama, S.; Iwano, M.; Satoh, H.; Shimamoto, K. The small GTP-binding protein Rac is a regulator of cell death in plants. Proc. Natl. Acad. Sci. USA 1999, 96, 10922–10926. [Google Scholar] [CrossRef] [PubMed]
- Moeder, W.; Yoshioka, K.; Klessig, D.F. Involvement of the small GTPase Rac in the defense responses of tobacco to pathogens. Mol. Plant-Microbe Interact. 2005, 18, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Pathuri, I.P.; Zellerhoff, N.; Schaffrath, U.; Hensel, G.; Kumlehn, J.; Kogel, K.H.; Eichmann, R.; Hückelhoven, R. Constitutively activated barley ROPs modulate epidermal cell size, defense reactions and interactions with fungal leaf pathogens. Plant Cell Rep. 2008, 27, 1877. [Google Scholar] [CrossRef]
- Chen, L.; Shiotani, K.; Togashi, T.; Miki, D.; Aoyama, M.; Wong, H.L.; Kawasaki, T.; Shimamoto, K. Analysis of the Rac/Rop small GTPase family in rice: Expression, subcellular localization and role in disease resistance. Plant Cell Physiol. 2010, 51, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.H.; Agrawal, G.K.; Rakwal, R.; Kim, J.A.; Lee, M.O.; Choi, P.G.; Kim, Y.J.; Kim, M.J.; Shibato, J.; Kim, S.H. Functional characterization of OsRacB GTPase—a potentially negative regulator of basal disease resistance in rice. Plant Physiol. Biochem. 2006, 44, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Z.; Wang, H.; Zhang, G.; Zhao, R.; Bie, T.; Zhang, R.; Gao, D.; Xing, L.; Cao, A. Characterization of a small GTP-binding protein gene TaRab18 from wheat involved in the stripe rust resistance. Plant Physiol. Biochem. 2017, 113, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.; Whipps, J.M.; Gurr, S.J. The tomato powdery mildew fungus Oidium neolycopersici. Mol. Plant Pathol. 2001, 2, 303–309. [Google Scholar] [CrossRef] [PubMed]
- Huibers, R.P.; Loonen, A.E.; Gao, D.; Van den Ackerveken, G.; Visser, R.G.; Bai, Y. Powdery mildew resistance in tomato by impairment of SlPMR4 and SlDMR1. PLoS ONE 2013, 8, e67467. [Google Scholar] [CrossRef]
- Lacomme, C.; Santa Cruz, S. Bax-induced cell death in tobacco is similar to the hypersensitive response. Proc. Natl. Acad. Sci. USA 1999, 96, 7956–7961. [Google Scholar] [CrossRef]
- Liebrand, T.W.; van den Berg, G.C.; Zhang, Z.; Smit, P.; Cordewener, J.H.; America, A.H.; Sklenar, J.; Jones, A.M.; Tameling, W.I.; Robatzek, S. Receptor-like kinase SOBIR1/EVR interacts with receptor-like proteins in plant immunity against fungal infection. Proc. Natl. Acad. Sci. USA 2013, 110, 10010–10015. [Google Scholar] [CrossRef]
- Jiang, S.Y.; Ramachandran, S. Comparative and evolutionary analysis of genes encoding small GTPases and their activating proteins in eukaryotic genomes. Physiol. Genom. 2006, 24, 235–251. [Google Scholar] [CrossRef][Green Version]
- Wang, A.; Chen, X.; Zhang, D.; Chen, H.; Lu, G.; Wang, Z. Effects of different Arabidopsis ROPs on multiplication of Pseudomonas syringae pv. tomato DC3000. J. Fujian Agric. For. Univ. (Nat. Sci. Ed.) 2008, 37, 610–613. [Google Scholar]
- Williams, C.L. The polybasic region of Ras and Rho family small GTPases: A regulator of protein interactions and membrane association and a site of nuclear localization signal sequences. Cell Signal. 2003, 15, 1071–1080. [Google Scholar] [CrossRef]
- Sorek, N.; Poraty, L.; Sternberg, H.; Bar, E.; Lewinsohn, E.; Yalovsky, S. Activation status-coupled transient S acylation determines membrane partitioning of a plant Rho-related GTPase. Mol. Cell. Biol. 2007, 27, 2144–2154. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Fowler, J.E. Evolution of the ROP GTPase signaling module. In Integrated G Proteins Signaling in Plants; Springer: Berlin/Heidelberg, Germany, 2010; pp. 305–327. [Google Scholar]
- Christensen, T.M.; Vejlupkova, Z.; Sharma, Y.K.; Arthur, K.M.; Spatafora, J.W.; Albright, C.A.; Meeley, R.B.; Duvick, J.P.; Quatrano, R.S.; Fowler, J.E. Conserved subgroups and developmental regulation in the monocot rop gene family. Plant Physiol. 2003, 133, 1791–1808. [Google Scholar] [CrossRef] [PubMed]
- Brembu, T.; Winge, P.; Bones, A.M. The small GTPase AtRAC2/ROP7 is specifically expressed during late stages of xylem differentiation in Arabidopsis. J. Exp. Bot. 2005, 56, 2465–2476. [Google Scholar] [CrossRef]
- Wang, W.; Liu, Z.; Bao, L.J.; Zhang, S.S.; Zhang, C.G.; Li, X.; Li, H.X.; Zhang, X.L.; Bones, A.M.; Yang, Z.B. The RopGEF2-ROP7/ROP2 pathway activated by phyB suppresses red light-induced stomatal opening. J. Plant Physiol. 2017, 174, 717–731. [Google Scholar] [CrossRef]
- Akter, S.; Huang, J.J.; Waszczak, C.; Jacques, S.; Gevaert, K.; Van Breusegem, F.; Messens, J. Cysteines under ROS attack in plants: A proteomics view. J. Exp. Bot. 2015, 66, 2935–2944. [Google Scholar] [CrossRef]
- Peng, K.; Wang, C.; Wu, C.; Huang, C.; Liou, R. Tomato SOBIR1/EVR homologs are involved in elicitin perception and plant defense against the oomycete pathogen Phytophthora parasitica. Mol. Plant-Microbe Interact. 2015, 28, 913–926. [Google Scholar] [CrossRef]
- Akamatsu, A.; Wong, H.L.; Fujiwara, M.; Okuda, J.; Nishide, K.; Uno, K.; Imai, K.; Umemura, K.; Kawasaki, T.; Kawano, Y. An OsCEBiP/OsCERK1-OsRacGEF1-OsRac1 module is an essential early component of chitin-induced rice immunity. Cell Host Microbe 2013, 13, 465–476. [Google Scholar] [CrossRef] [PubMed]
- Lieberherr, D.; Thao, N.P.; Nakashima, A.; Umemura, K.; Kawasaki, T.; Shimamoto, K. A sphingolipid elicitor-inducible mitogen-activated protein kinase is regulated by the small GTPase OsRac1 and heterotrimeric G-protein in rice. Plant Physiol. 2005, 138, 1644–1652. [Google Scholar] [CrossRef]
- Liu, J.; Park, C.H.; He, F.; Nagano, M.; Wang, M.; Bellizzi, M.; Zhang, K.; Zeng, X.; Liu, W.; Ning, Y. The RhoGAP SPIN6 associates with SPL11 and OsRac1 and negatively regulates programmed cell death and innate immunity in rice. PLoS Pathog. 2015, 11, e1004629. [Google Scholar] [CrossRef]
- Sun, G.; Feng, C.; Guo, J.; Zhang, A.; Xu, Y.; Wang, Y.; Day, B.; Ma, Q. The tomato Arp2/3 complex is required for resistance to the powdery mildew fungus Oidium neolycopersici. Plant Cell Environ. 2019, 42, 2664–2680. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Lian, Q.; Meng, Y.; Zhao, X.; Xu, Y.; Wang, Y.; Day, B.; Ma, Q. ShNPSN11, a vesicle-transport-related gene, confers disease resistance in tomato to Oidium neolycopersici. Biochem. J. 2020, 477, 3851–3866. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).