Abstract
Over the past decade, we witnessed a promising application of cold atmospheric plasma (CAP) in cancer therapy. The aim of this systematic review was to provide an exhaustive state of the art of CAP employed for the treatment of head and neck cancer (HNC), a tumor whose late diagnosis, local recurrence, distant metastases, and treatment failure are the main causes of patients’ death. Specifically, the characteristics and settings of the CAP devices and the in vitro and in vivo treatment protocols were summarized to meet the urgent need for standardization. Its molecular mechanisms of action, as well as the successes and pitfalls of current CAP applications in HNC, were discussed. Finally, the interesting emerging preclinical hypotheses that warrant further clinical investigation have risen. A total of 24 studies were included. Most studies used a plasma jet device (54.2%). Argon resulted as the mostly employed working gas (33.32%). Direct and indirect plasma application was reported in 87.5% and 20.8% of studies, respectively. In vitro investigations were 79.17%, most of them concerned with direct treatment (78.94%). Only eight (33.32%) in vivo studies were found; three were conducted in mice, and five on human beings. CAP showed pro-apoptotic effects more efficiently in tumor cells than in normal cells by altering redox balance in a way that oxidative distress leads to cell death. In preclinical studies, it exhibited efficacy and tolerability. Results from this systematic review pointed out the current limitations of translational application of CAP in the urge of standardization of the current protocols while highlighting promising effects as supporting treatment in HNC.
1. Introduction
Head and Neck cancer (HNC) is the sixth most common type of cancer by incidence worldwide [1], with an approximate 5-year survival rate of 65% [2,3], dropping to an average of 40% for patients diagnosed in advanced stage [4]; this is due to a poor response to standard anti-cancer treatments such as chemotherapy and radiotherapy [5,6], which has remained almost unchanged over the past decade [7]. The tumor is remarkably heterogeneous, based on localization in the oral cavity, oropharynx, larynx, and hypopharynx, on the types of cells within the tumor tissue and the molecular subtypes. The prognosis for patients with HNC is determined by the stage of the tumor at presentation, as well as the presence of lymph-node metastases and distant metastases [8]. Unfortunately, only one-third of patients were present with early-stage disease, whilst two-thirds were diagnosed with advanced cancer with lymph node metastases [5]. Surgery combined with adjuvant radiation therapy and/or chemotherapy is the standard of care, but in these cases, patients often experience notable complications related to disease treatments, including different degrees of hoarseness or aphonia, dysphagia, dry mouth, aspiration, neck masses, and facial distortion after surgery [9]. Indeed, it is important to obtain tumor-free resection margins in patients with HNC [10], and to achieve this, surgeons usually remove the tumor with a margin of 10 mm of macroscopically normal tissue, thus creating a large surgical defect that needs free flap reconstruction in more than half of patients [11]. Moreover, adjuvant chemoradiation to control the local disease itself may induce many morbidities that affect the quality of life and prognosis [12], led by the development of different side effects, such as permanent dry mouth, burning mouth syndrome, osteoradionecrosis, dental caries, neurological damage, thyroid function impairment, trismus, fibrosis and eye, and skin damage, compromising the patients’ recovery, psychological health, and physiology [13,14].
This scenario calls for an urgent need for novel methods in HNC treatment, and cold plasma-based systems may be a promising tool. The use of plasma sources in the medical field was approved in Germany in 2013, based on the results obtained regarding its effectiveness in stimulating skin regeneration and its antimicrobial effects [15]. Over the past years, plasma medicine has rapidly evolved due to the contribution of different groups of researchers in the development of medical devices generating cold atmospheric plasma (CAP) or non-thermal plasma (NTP) with temperatures at close to ambient temperature, thus allowing applications on heat-sensitive biological matters such as tissue [16]. Only in 2018 did it achieve an important milestone in the field of evidence-based medicine (EBM), reaching EBM level III in HNC [15]. Plasma is known as the fourth state of matter [17] and is partially or completely ionized gas that reacts with the environment creating a mixture of active components: electrons, ions, radicals, and energetic photons, all of which can, in turn, interact with the target [18]. It is a multicomponent system, a multiagent technology, and a multimodal therapy, and due to its unique traits, it elicits electromagnetic, chemical, and thermal effects on the target [19]. Due to advances in physics and biotechnology, nowadays, plasma can be easily generated and used at room temperature reaching high electron temperatures, but very low gas temperatures are associated with weak ionization rates [20].
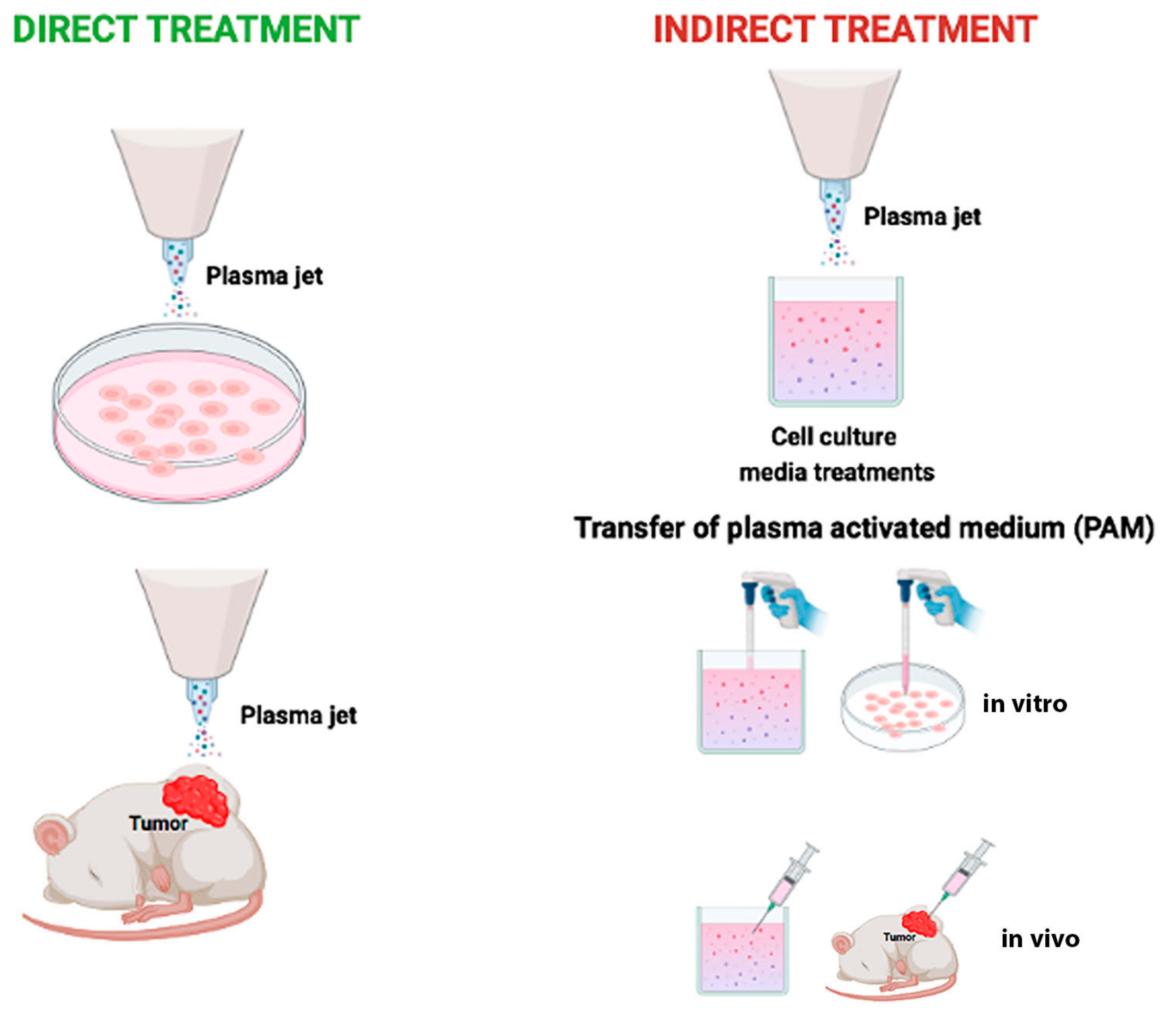
CAP devices have been designed with different configurations [21]: dielectric barrier discharge (DBD), corona discharge, radio frequency reactor, or direct current plasma jet with different carrier gas [6]. In that respect, most CAP sources are operated in the noble gasses’ environment, e.g., helium or argon, which are characterized by a relatively low breakdown voltage and long plasma sustainability [22]. They are commonly mixed with a small number of other gasses (e.g., O2) to increase their chemical reactivity [23]. Recently, CAP has been investigated in several medical applications, such as blood coagulation [24], wound disinfection and healing [25], tissue and germicidal irradiation and sterilization [26], skin rejuvenation [27], tooth bleaching [23], material surface modifications and crosslinking [28,29,30], and to selectively eradicate cancer cells [31,32,33]. In this last specific aim, two different types of tumor treatment using CAP have been described: direct exposure to a plasma jet or indirect exposure by a medium (named plasma-activated medium, PAM) previously exposed to cold plasma (Figure 1) [34]. Both types of treatment decrease cell viability with the benefit that indirect treatment using PAM made CAP treatment an innovative drug therapy [35].
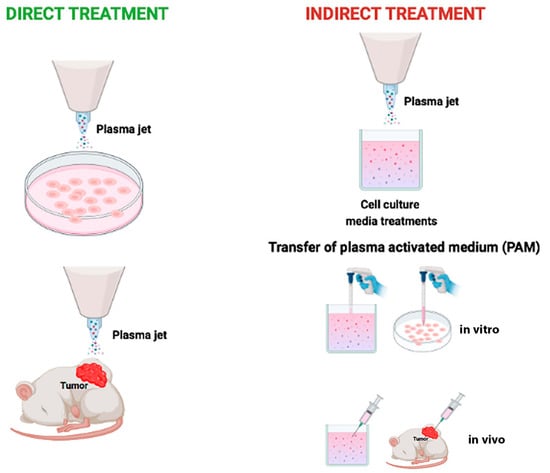
Figure 1.
Schematic representation of two basic strategies to use cold atmospheric plasma. Direct cold atmospheric plasma treatment on cancer cells in vitro or on subcutaneous xenograft tumors in vivo (left). Indirect cold atmospheric plasma treatment on solutions, mostly medium. Plasma Activated Medium is used to treat cancer cells seeded in dish or tumor tissues in mice (right).
The anti-cancer effect of CAP is a combination of physical and chemical factors, and it depends on treatment time, working gas, treatment pattern (direct or indirect), and cell/tissue type. Ultraviolet, heat, and electromagnetic fields are physical factors in CAP. Chemical factors include reactive species produced in the gas phase of CAP. Among them, oxygen-reactive species such as hydroxyl (OH), superoxide (O2−), hydrogen peroxide (H2O2), as well as nitrogen-based species such as nitric oxide (NO), nitrogen dioxide (NO2), nitrogen trioxide (NO3), have been demonstrated. One key aspect, which is attracting increasing attention, is the ability of CAP to induce cell death in various cancers, including skin, liver, and colon cancer [36], without damaging the surrounding healthy tissues; however, the mechanisms remain poorly understood. The selectivity of plasma in inactivating tumor cells over non-malignant cells is a matter of debate, although it is generally accepted that reactive oxygen and nitrogen species (RONS) are the major agents responsible for plasma-promoted biological outcomes. Indeed, it has been shown that the physical effects seem to have a minor biological impact, while RONS may induce intracellular reactive oxygen species (ROS) increase, a decrease in antioxidant enzymes, DNA double-strand brakes, and consequently, apoptosis, according to cell type and exposure parameters (total delivered energy, power, time of exposure). Indeed, excessive ROS or reactive nitrogen species (RNS) induces oxidative stress against cancer cells and, by damaging essential cellular molecules, i.e., cell cycle checkpoints, deoxyribonucleic acid, and other organelles subsequently, results in apoptotic death. However, the mechanisms of CAP action are not well understood at a molecular level. Since the first report about the killing effect of a plasma jet on HNC in 2012 [37], the field of CAP application in this type of cancer treatment has expanded rapidly.
In metastatic or recurrent HNC therapy, resistance and multiple severe side effects of conventional anti-cancer therapies such as chemotherapy limit the benefit of single-agent therapies. Indeed, numerous clinical evidence indicates that single-agent systemic therapies infrequently culminate in long-lasting medical responses. Factors such as tumor plasticity, responsible for therapeutic adaptation, and heterogeneity, which induce the emergence of therapy-resistant tumor clones, can cause the failure of single-agent therapy. To overcome such phenomena, CAP might exhibit an important adjuvant treatment effect. Lee et al., 2020 [38] analyzed the synergistic effect of the combination of CAP and cisplatin-mediated chemotherapy and found that ROS generation, tumor-suppressor proteins, and apoptosis-related enzymes increased according to the treatment time of CAP in combination with cisplatin, thus showing the synergistic effect of cisplatin and CAP treatment against SCC-15 cells with low cytotoxicity against normal cells. Moreover, CAP can obtain a stronger anti-cancer capacity through the synergistic application with nanoparticle technologies, as demonstrated by Choi et al. [37]. Finally, the synergy found by Lafontaine et al. [39] between gas plasma plus radiation therapy and gas plasma combined with a chemotherapeutic agent provides evidence for further opportunities to improve cancer treatments.
Currently, many research groups are concentrating their efforts on the cell-specific effects of CAP treatment, exploring the fundamental mechanisms at the molecular level and the optimal parameters for cancer cell treatment, with the long-term goal of using plasma as an alternative means of effective cancer therapy. The purpose of the present article was to conduct a systematic review of the current evidence, focusing on CAP as potential therapeutics in HNC, to summarize the characteristics and settings of the different devices to meet the urgent need to standardize protocols, procedures, and multidisciplinary guidelines and to evaluate and compare the effectiveness of CAP treatments in a laboratory setting, with possibilities to extrapolate these methods to clinical tests.
2. Materials and Methods
This systematic review was performed according to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) statement [40].
2.1. Search Strategy
An electronic search on scientific databases (PubMed, Scopus, Web of Science) was performed to identify suitable studies, using the following terms and keywords alone or in combination: (cold atmospheric plasma OR cold plasma OR low temperature plasma OR kinpen med OR non-thermal atmospheric pressure plasma OR non thermal atmospheric pressure plasma OR cold physical plasma OR plasma medicine OR CAP OR plasma activated medium OR cold atmospheric-pressure plasma OR plasma activated liquid OR cold argon plasma) AND (head and neck tumors OR head and neck squamous cell carcinoma OR head and neck cancer OR head and neck cancer OR oral squamous cell carcinoma OR oral cancer OR oral squamous cell cancer OR OSCC OR oral oncology OR oral squamous cancer cell OR oral cavity squamous cell carcinoma) AND (therapy* or treat* OR application or attenuation OR efficacy OR action OR effect* OR target* OR response).
The first search was performed on 13 December 2021. The search was limited to studies published in the English language. The last electronic search was performed on 10 August 2022. In addition to the electronic search, a hand search was undertaken by checking the references of the included studies to identify further eligible studies. A reference manager software program (Endnote X9.3.2, Clarivate Analytics) was used, and the duplicates were discarded first electronically, then by checking the resulting list manually. Search strategy for PubMed, Web of Science, and Scopus is shown in Supplemental Material Table S1.
2.2. Eligibility Criteria
2.2.1. Inclusion Criteria
Clinical prospective studies with a cross-sectional, cohort, case-control, and case-series or report design were included as well as preclinical studies on both in vitro and animal models. Patients included in the study had to be over 18 years old and be diagnosed with HNC [41], and treated with CAP. Biological, physic-chemical, and/or clinical outcomes had to be reported, such as efficacy and safety. Specifically, for preclinical studies, in vitro and in vivo assays had to be performed on HNC cell lines or xenografts derived from HNC cell lines or patient-derived xenografts (PDX).
2.2.2. Exclusion Criteria
Systematic review, meta-analyses, and ecologic studies.
2.3. Focused PICO Question
Participants: patients with diagnosed HNC as well as HNC cell lines or xenograft models.
Intervention: documented protocol of direct or indirect CAP irradiation.
Comparison: the studied model (cell line, xenograft, or human) exposed to other kinds of treatment or different CAP treatment protocols.
Outcomes: primary outcomes were cell viability and apoptosis, while secondary outcomes included protein expression and ROS production in vitro studies and in vivo efficacy in animal and human studies.
2.4. Selection of Studies
Retrieved citations were independently screened by two authors (VP and VCAC), and relevant studies were identified based on title and abstract. If those did not provide sufficient information about the inclusion criteria, the full text was evaluated to assess eligibility. Any disagreement was solved by discussion, and a third reviewer was consulted to make final decisions (MM). This author also calculated a k-statistic value to ascertain the level of reviewers’ agreement.
2.5. Data Extraction and Method of Analysis
Two reviewers (VCAC and MCDM) independently extracted data from all the included studies using a predesigned extraction form. Microsoft Excel 2020 (Microsoft Office, Microsoft Corporation, Redmond, WA, USA) was used for data collection and for descriptive analysis.
The following data were collected: type of plasma device and trade name if present, CAP settings (i.e., Flow rate, Gas, pulse frequency, pulse, temperature, application distance, application time, total energy, and power), type of cancer target, study level, study population/model, tumor stage, type of treatment (i.e., direct, or indirect), sample size, quantitative measures, developer, references, and notes. The primary outcomes included safety and efficacy of CAP in clinical studies. For preclinical studies, cell viability, DNA damage, changes in cell morphology, and apoptosis after CAP exposure among groups or difference in tumor dimension means in xenograft models were the main investigated outcomes.
2.6. Risk of Bias Assessment
Two reviewers (VP, VCAC) assessed the quality of studies by risk of bias evaluation. Because of the different study designs of all the included studies, diverse methods of assessment were used. For in vitro studies, no standard quality assessment tool exists [42]; therefore, we developed these criteria by ourselves. For the methodological quality of in vitro studies, criteria based on the parameters for developing cell culture studies were adapted [43]. In particular, 6 items were evaluated as adequate or inadequate, whether information about: 1. condition of cell culture before treatment; 2. condition of cell culture during the treatment; 3. description of methodology to evaluate outcomes; 4. case-control description; 5. multiple experiments performed; 6. descriptions of plasma devices and settings were reported. Studies fulfilling at least five over six of the above-reported items were considered at low risk of bias, while two or more than three inadequate reported items were classified respectively at medium and high risk of bias. Regarding in vivo animal studies, the Systematic Review Centre for Laboratory Animal Experimentation’s (SYRCLE) risk of bias tool was used to assess the quality of available evidence [44]. At last, for human clinical studies, Newcastle-Ottawa Scale was used as a qualitative tool (NOS) in case of cohort or case-control study design [45], while case-series studies were evaluated by Murad’s quality checklist of case series and case report [46], without considering items 5 and 6 since they are more relevant of drug investigational studies [47].
2.7. Statistical Analysis
Considerable methodological and clinical heterogeneity was found in the selected studies regarding participants, interventions, and outcome measures. Evident differences were seen in study design, methodologies, treatment modalities, outcome measures, and results. Moreover, many studies reported aggregated results with only descriptive or graphic representation of CAP-related outcomes and without a standardized system. Finally, not all the studies performed statistical analysis of data; therefore, comparisons between studies were not feasible, and a descriptive presentation had to be adopted since meta-analysis was considered inappropriate. However, when possible, quantitative data were presented. Moreover, when applicable, based on each study categorization and criteria, descriptive results were arranged ordinally, and comparisons were made within each factor.
3. Results
3.1. Study Selection
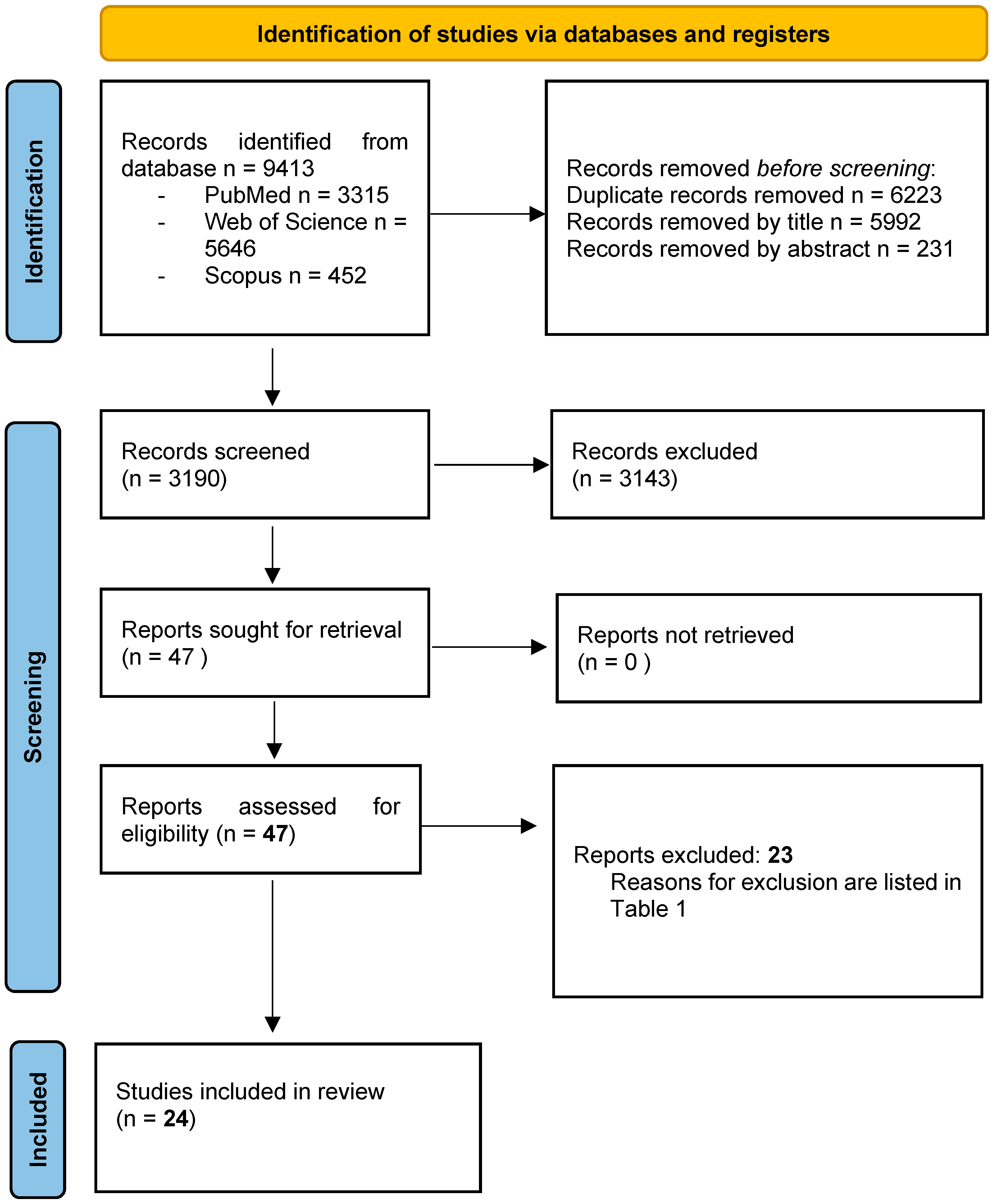
A total of 9413 potentially relevant records were identified from the databases and further processed following the PRISMA statement (Figure 2).
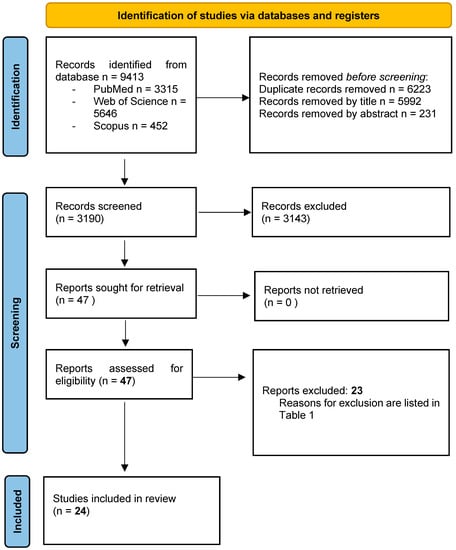
Figure 2.
PRISMA 2020 Flow Diagram of the screening process. In total, 24 studies were included in the present systematic review.
After the removal of duplicates, 1997 records were further examined based on their titles and abstracts, and 1950 studies were excluded as they did not meet the specific eligibility criteria for this study. A total of 47 full-text articles were finally evaluated, and 23 were subsequently excluded [48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70] for the reasons listed in Table 1. A total of 24 studies [5,6,15,23,36,37,38,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87] fulfilled the selection criteria of the present review and were included for qualitative analysis. The value of the k-statistic was 0.78, which indicates an excellent level of agreement between reviewers

Table 1.
List of excluded studies and reasons for exclusion.
3.2. General Characteristics of Included Studies
Based on the development status, the studies were categorized into two main groups:
- -
- Nineteen [5,6,23,36,37,38,71,72,73,74,75,76,77,78,79,80,81,82,83] preclinical studies investigated the CAP in vitro effect on different HNC cell lines, and 3 [73,74,77] out of those 19 articles conducted in vivo experiments on an animal model (Table 2);
 Table 2. Characteristics of preclinical studies.
Table 2. Characteristics of preclinical studies. - -
 Table 3. Characteristics of clinical studies.
Table 3. Characteristics of clinical studies.
3.3. Results of Risk of Bias Assessment
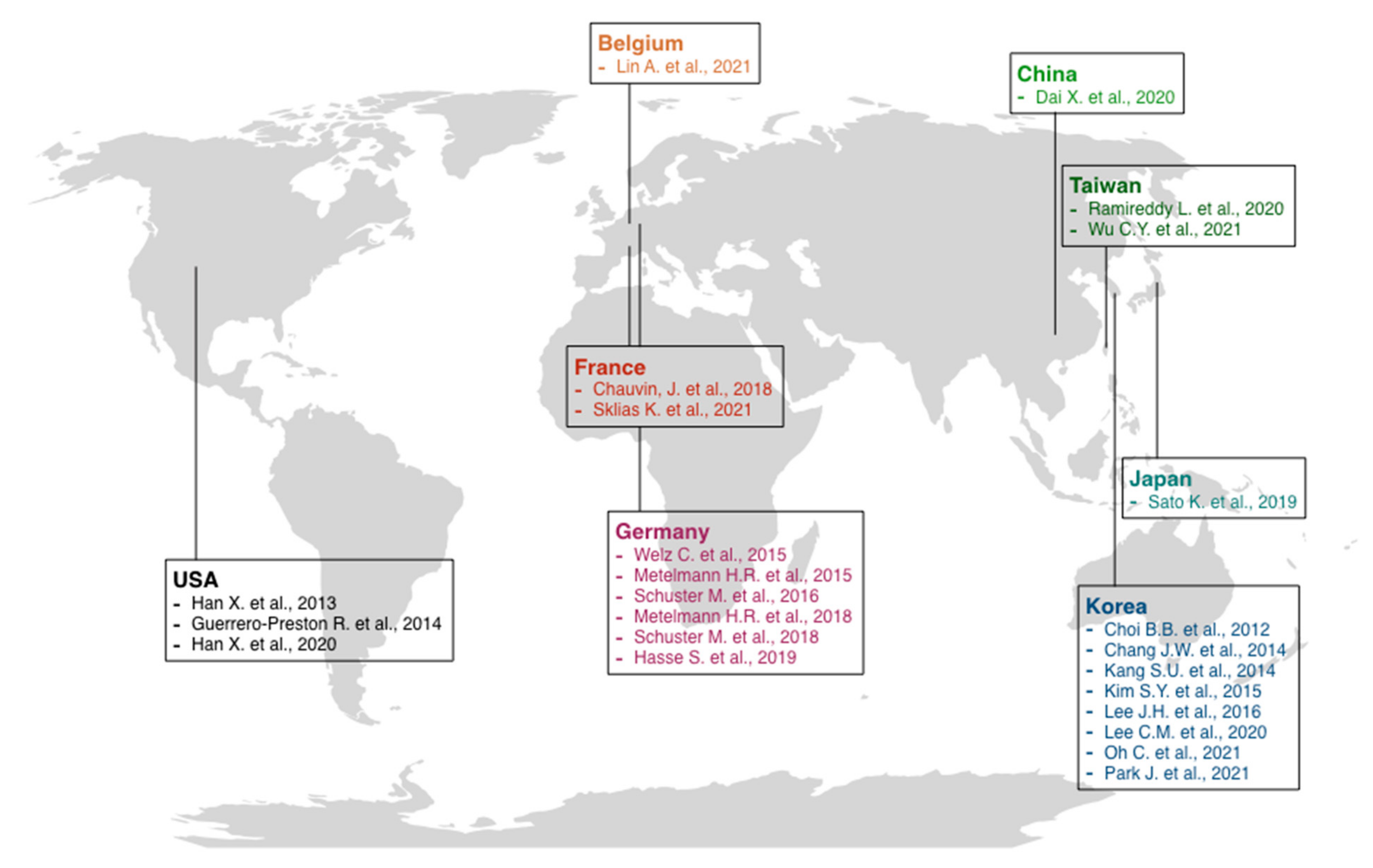
A low risk of bias was found for all the in vitro studies. Only in one study [23] was there inadequate information about the “condition of cell culture before treatment” domain, while three other studies lacked “multiple experiments performed” evidence. All three in vivo animal studies [73,74,77] were at high risk of bias based on SYRCLE features. Only three domains, such as “sequence generation”, “baseline characteristics”, and “selective outcome reporting”, were adequate in all the studies. “Blinding” in outcome assessment was adequate only in Kang et al. [73], while “incomplete outcome data” was adequate only in the Oh et al. study [77]. Information about the following domains, such as “allocation concealment”, “random housing”, “blinding”, “random outcome assessment”, and “other sources of bias”, were not retrievable. On the other hand, NOS for case-control studies resulted in adequate in all the items for the only study included by Dai et al. [84], while Schuster et al. (2016) [87] was inadequate in two items, such as “representativeness of exposed cohort” and “selection of non-exposed cohort”, based on NOS for cohort studies. At last, for case series studies, based on Murad et al. [46] risk of bias assessment, all three studies were at low risk of bias. Metelmann et al. (2018) [15] was adequate in all the evaluated items, while Schuster et al. [87] and Metelmann et al. (2015) [85] were inadequate in the “selection” process. Detailed risk of bias assessment and evaluation is reported in Tables S2–S6. The relevant studies were conducted in various centers around the world, mainly in Europe and Asia, and were published between 2012 and 2021 (Figure 3).
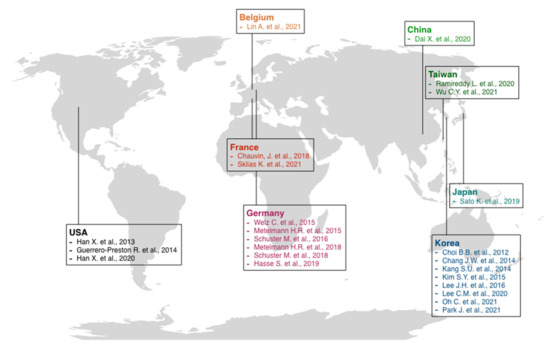
Figure 3.
Schematic representation of the most relevant cold atmospheric plasma studies [5,6,15,23,36,37,38,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87] published in various countries.
3.4. Preclinical Model: Cell Lines
The 19 in vitro studies [5,6,23,36,37,38,71,72,73,74,75,76,77,78,79,80,81,82,83] evaluated the CAP effect on various HNC cell lines (Figure 4) arising at specific anatomical sites and representative of specific cancers, such as the human floor of the mouth squamous cell carcinoma (AMC-HN6, Ho-1-u-1), human tongue squamous cell carcinoma (CAL-27, HNO-97, HSC-3, HSC-4, OSC-19, SAS, SCC-15, SCC-25, SCC-4), human oral cavity squamous cell carcinoma (HSC-2, SCC-QLL1), human oral retromolar trigone cavity squamous cell carcinoma (SCC-1483), human hypopharynx squamous cell carcinoma (FaDu, SNU-1041), human laryngeal squamous cell carcinoma (JHU-022, JHU-029, SNU-899, SNU-1076), human pharyngeal squamous cell carcinoma (Detroit-562), human undifferentiated carcinoma of the parotid gland (AMC-HN-9), and normal oral cavity cell lines: human gingival fibroblasts (HGF-1), human normal oral keratinocytes (NOKsi) and normal human floor of the mouth keratinocytes (OKF6/T).
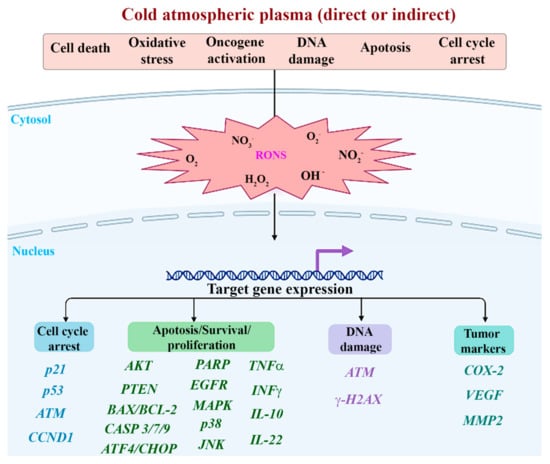
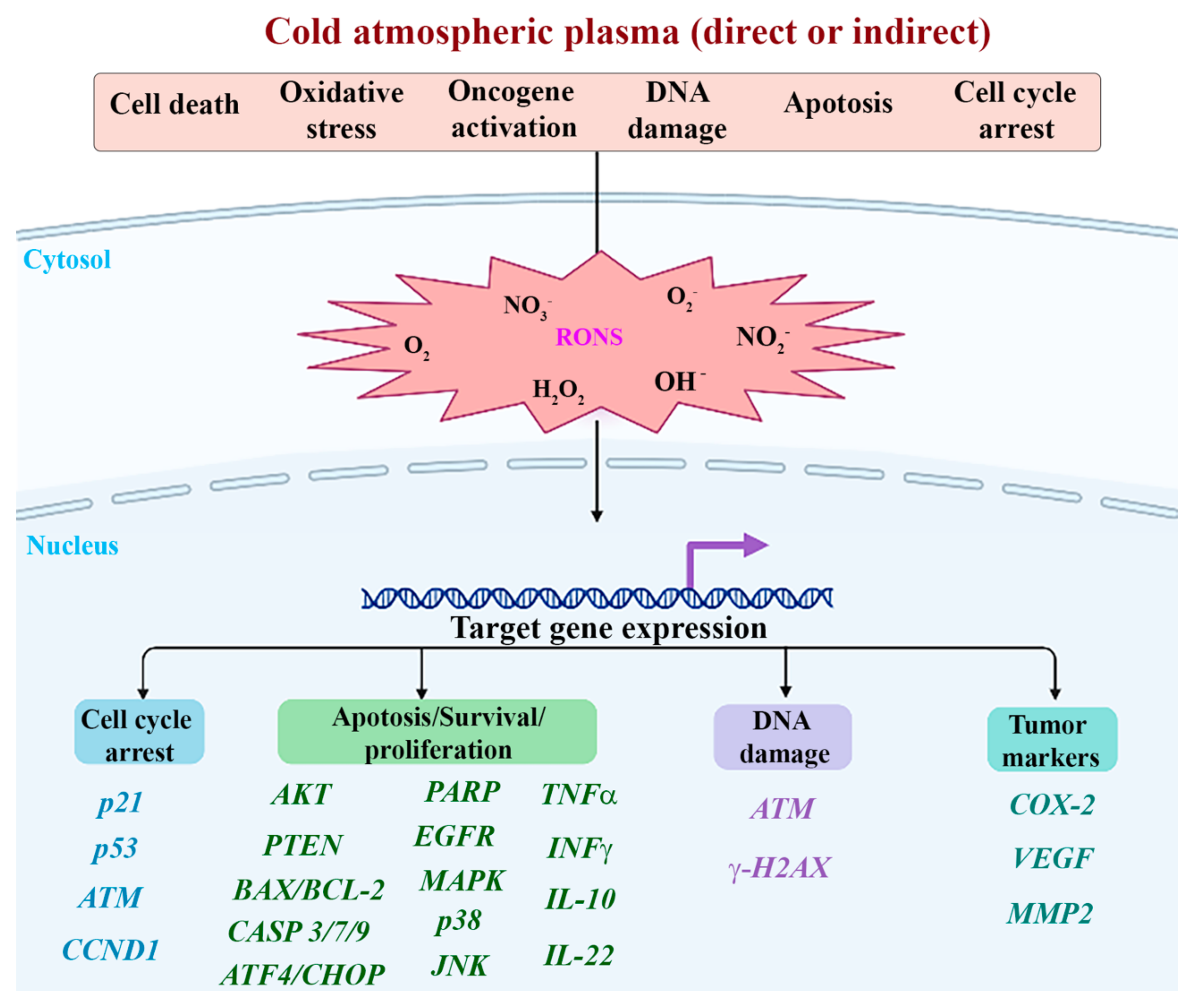
Figure 4.
Molecular mechanisms induced by direct and indirect application of cold atmospheric plasma included in the present systematic review.
Fourteen studies [5,23,36,37,38,71,73,75,78,79,80,82,83] evaluated only the effects of direct CAP application, while four studies [6,72,74,77] applied indirect CAP exposition; only two studies in in vitro model [81] compared both effects of direct and indirect plasma exposition. Different in vitro tests were performed to assess biological and chemical reactions. Fifteen studies [5,6,37,38,72,74,75,76,77,78,79,80,81,82,83] evaluated cell viability, twelve studies [5,36,37,38,72,73,74,75,77,78,79,80] investigated protein expression changes, twelve studies [36,38,72,73,74,75,77,78,80,81,82,83] focused on apoptosis, four studies [23,36,71,82] evaluated the potential of plasma to determine DNA-damage, three studies [36,37,77] observed changes in cell morphology, another three [5,74,80] performed colony formation assay, and finally four studies [36,72,75,79] investigated changes in cell cycle. Two studies furtherly quantified changes in gene expression [73,76], two assessed the presence of mitochondrial damage [73,77], and two assessed changes in cell motility by migration and invasion assays [72,80]. Only one study investigated changes in catalytic Fe (II) [80]. Eleven studies [23,38,73,74,75,76,77,79,80,81,83] assessed the chemical characteristics produced by plasma exposition by quantification of reactive-species production (Figure 5A,B).
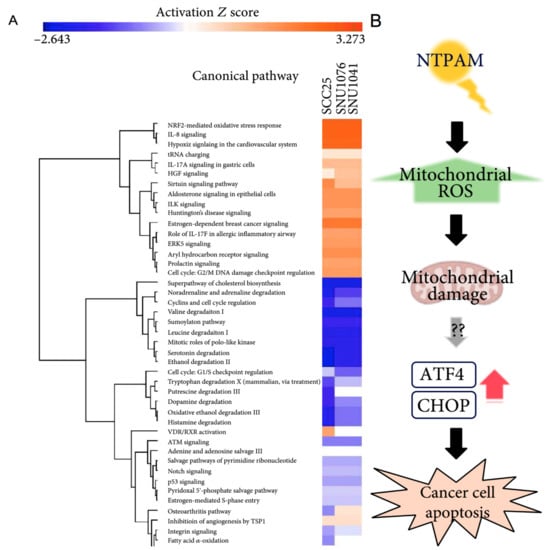
Figure 5.
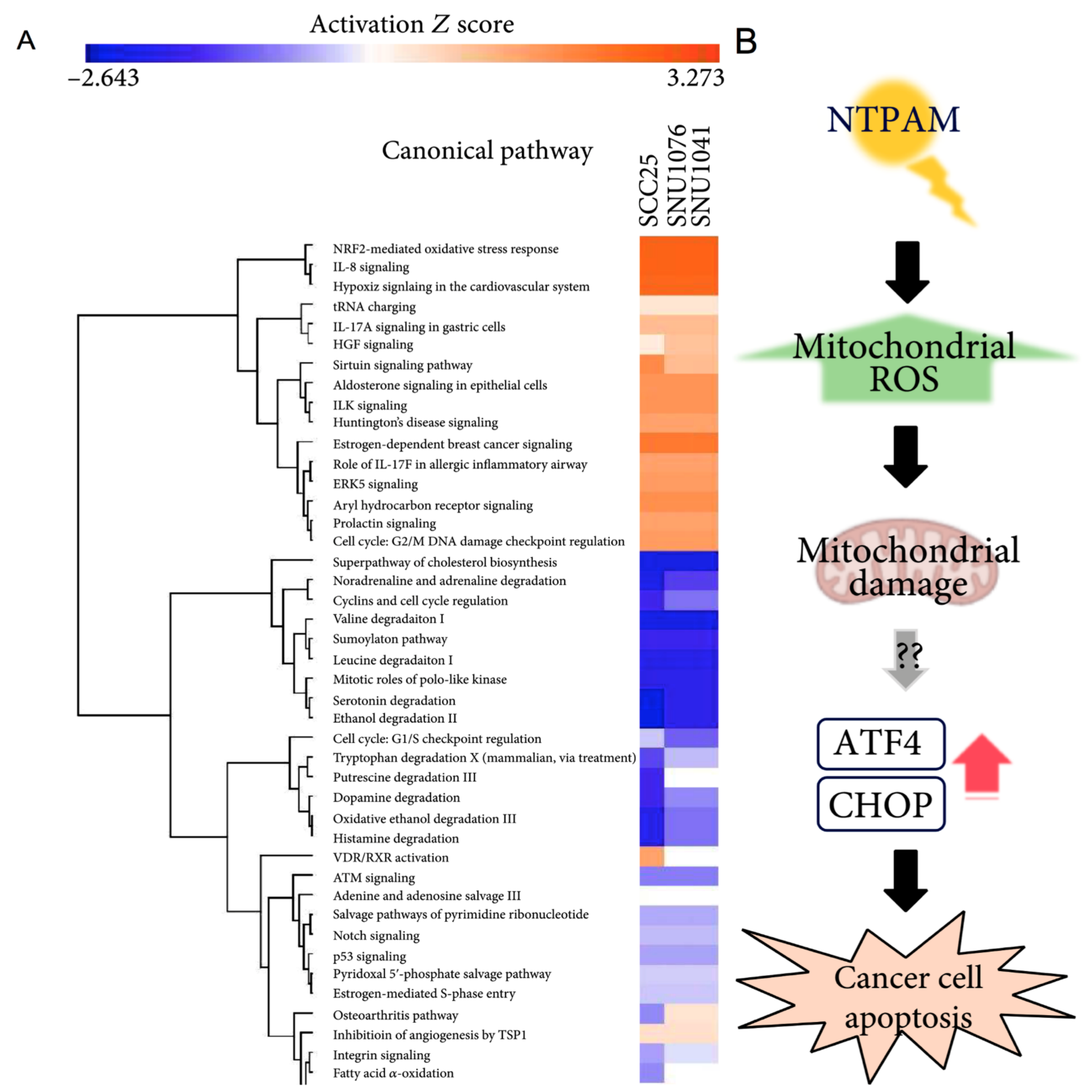
Mechanism and pathways induced by plasma-activated media treatment in head and neck cancer. (A) Top relevant canonical pathways associated with plasma activated media treatment by core analysis. (B) The proposed mechanism of plasma activated media based anti-cancer effects on head and neck cancer cells [77].
Cell viability assays: Seven studies [5,38,74,79,81,82,83] performed MTT or MST assay, while two studies [37,76] investigated cell viability by trypan blue staining. Park et al. [78] performed SRB-assay; Chauvin et al. [6] PrestoBlue® assay, Hasse et al. [72] performed Resazurin-based assay, while Sato et al. [80] and Oh et al. [77], respectively, used WST-8 and WST-1 assays. Sklias et al. [81] performed MTT, trypan blue and CellTiterGlo® assays. In general, a higher reduction in cell viability was observed in cancer cell lines compared to normal ones, with variability among the diverse protocols, but an overall increase in cancer cell deaths was observed in a dose-time-dependent manner. Better results were also obtained when combining plasma treatment with GNP-EGFR, PD-L1-ab, and GNP or cisplatin. Three studies [5,74,80] furtherly assessed cell survival based on the ability of a single cell to grow into a colony.
Protein expression: TP53 gene-related pathway was mainly investigated by Western-Blot analysis, quantifying different proteins. One study [36] investigated the p-ATM, p-53, p21, cyclin D1mm, and yH2AX axis. Another study [79] investigated p-ATM—Bcl-2 changes. PARP—caspase 3/7—Bax/Bcl-2 axis was investigated in two studies [77,78], while one study [5] investigated PARP expression alone. PTEN-p53-Cas-9 axis was also investigated in one study [71]. Other evaluated proteins were p-p38/p-c-JUN/JNK/p-ERK axis, AKTmm, and MMP-2; one study [75] quantified EGFR changes.
Apoptosis: Four studies evaluated apoptosis by FACS/TUNEL assays [36,65,80,83], while four and two studies respectively performed only FACS [38,75,77,82] or TUNEL assay [72,74], respectively. Park et al. [78] evaluated apoptosis by immunocytochemistry and by quantification of cleaved caspase-3, PARPm, and cytochrome C. Sklias et al. [81] analyzed the Caspase 3/7 signal; an overall increase in apoptotic mechanisms was observed in cancer cells compared to controls. In particular, Kang et al. [73] highlighted how direct plasma treatment led to apoptosis by MAPK-mediated mitochondrial ROS.
DNA-damage: Two studies [23,71] performed immunofluorescence assays, and two studies [36,82] performed COMET assay. Overall, an increase in DNA damage/fragmentation was observed in a time-dose-dependent manner. Of interest, Han et al. (2020) [71] observed maximized DNA damage in the plasma treatment region, which decreased radially outward. Moreover, S-phase cells were more susceptible than G1-G2-phase cells.
Cell morphology: SEM was employed in two studies [37,77], while light microscopy was used in one study [36]. Overall, changes in shape (from spindle to round), cell detachment, shrinkage, and membrane ruptures were observed.
Cell cycle: All four studies [36,72,75,79] investigating changes in cell cycles employed FACS assay. Chang et al. [36] reported that CAP caused cell cycle arrest in the sub-G1 phase, and the proportion of SCC-25 cells (expressing wild-type p53) in the sub-G1 phase increased markedly in an intensity-dependent manner. However, the other cell lines (MSK QLL1, SCC-1483, and SCC-15) with mutated p53 were unaffected by CAP treatment. Similar results were obtained in Lee et al. (2016) [75], where HSC-2 cells were killed via sub-G1 phase arrest, and in Ramireddy et al. [79], where CAP-induced cell death and G1 cell cycle arrest associated with the ATM/P53 pathway.
Gene expression, mitochondrial damage, and cell motility: Kim et al. (2015) [74] performed qRT-PCR analysis showing that plasma treatment led to MUL1 mRNA expression, with increased cytotoxicity by MUL1/AKT binding. Oh et al. [77] also performed qRT-PCR analysis, investigating the role of ATF4 and CHOP as key regulators in non-thermal atmospheric plasma-activated media (NTPAM) induced cell death. Mitochondrial damage was assessed in the studies of Kang et al. [73] and Oh et al. [77]. The first study was performed by FACS and MMP measurements [73], while the second one conducted OCR assay and SEM [77]. In both studies, there was evidence of mitochondrial damage and dysfunction. Hasse et al. [72] and Sato et al. [80] investigated cell motility by wound healing assay, showing a marked reduction in cell motility of treated cancer cells without or with irrelevant changes in normal cells.
Chemical characteristics: Strong evidence emerged suggesting the development of reactive species after plasma treatment. Three studies [76,79,85] quantified the formation of such reactive species by fluorimetric assay, three studies by OES [38,75,81], three studies [36,74,80] by FACS, and one study [83] by OxiSelect Intracellular assay. Based on plasma device and substrate, most reported species were NO-γ, O2−, more general ROS, thiol, Ar+, OH−, RNS, H2O2, NO2− and NO3−.
3.5. Preclinical Models: In Vivo Animal Studies and Ex Vivo Human Studies
Only four studies [73,74,76,77] applied plasma treatment to preclinical models. In Kang et al. [73] study, the plasma treatment was delivered directly to FaDu-derived xenograft models in mice. By immunohistochemistry (IHC) It was observed an increase in caspase-3 and Nox-3 levels and an inhibition of tumor growth after 11 days by tumor mass measurement by a sliding caliper. Kim et al. (2015) [74], instead, employed both an SCC7-derived syngeneic tumor model and an SCC15-derived xenograft in mice. In this case, plasma treatment was not administered directly but by injection of a liquid-type NTP (LTP). Similarly, an inhibitory effect on tumor growth and development was observed. Furtherly, by IHC and Western blot assay, p-AKT was reduced while MUL1 increased. Oh et al. [77] performed a similar study by injection of NTPAM in an SNU-1041-derived xenograft model in mice. After NTPAM treatment by IHC, they observed low expression of Ki-67 and high expression of ATF4 and CHOP. Lower tumor weight was also observed in the test group compared to the control. At last, Hasse et al. [72] directly targeted human tissues. Their study showed an increase in cytochrome C, INF-γ, TNF-α, and IL-10 levels, while IL-22 expression increased in healthy tissue.
3.6. Clinical Model
Five studies [15,84,85,86,87] investigated the consequences of plasma treatment on human clinical models. A total of 159 patients were included in different kinds of study setups. Fifty-three patients were diagnosed with HNC, six with oropharyngeal squamous cell carcinoma (OPSCC), and 100 with laryngeal carcinoma (LC). In four studies [15,85,86,87], Ar-CAP kINPen MED was used directly on ulceration, vertically to the naturally moist tissue surface. In one study [84], they employed Unitec low-temperature plasma operation system, directly ablating the tumor up to 3-5 mm away from the edge of the lesion. Outcomes varied among studies to address the effects of plasma on contamination of infected ulcerations, side effects, pain, tumor surface changes, and effects on tumor growth. Overall, the plasma treatment resulted in a few patients experiencing mild–moderate side effects and was never life-threatening. Of interest, a significant reduction was observed in terms of fetid odor, contamination, and pain of lesions (Table 3).
3.7. Types of CAP Devices and CAP Treatment Parameters
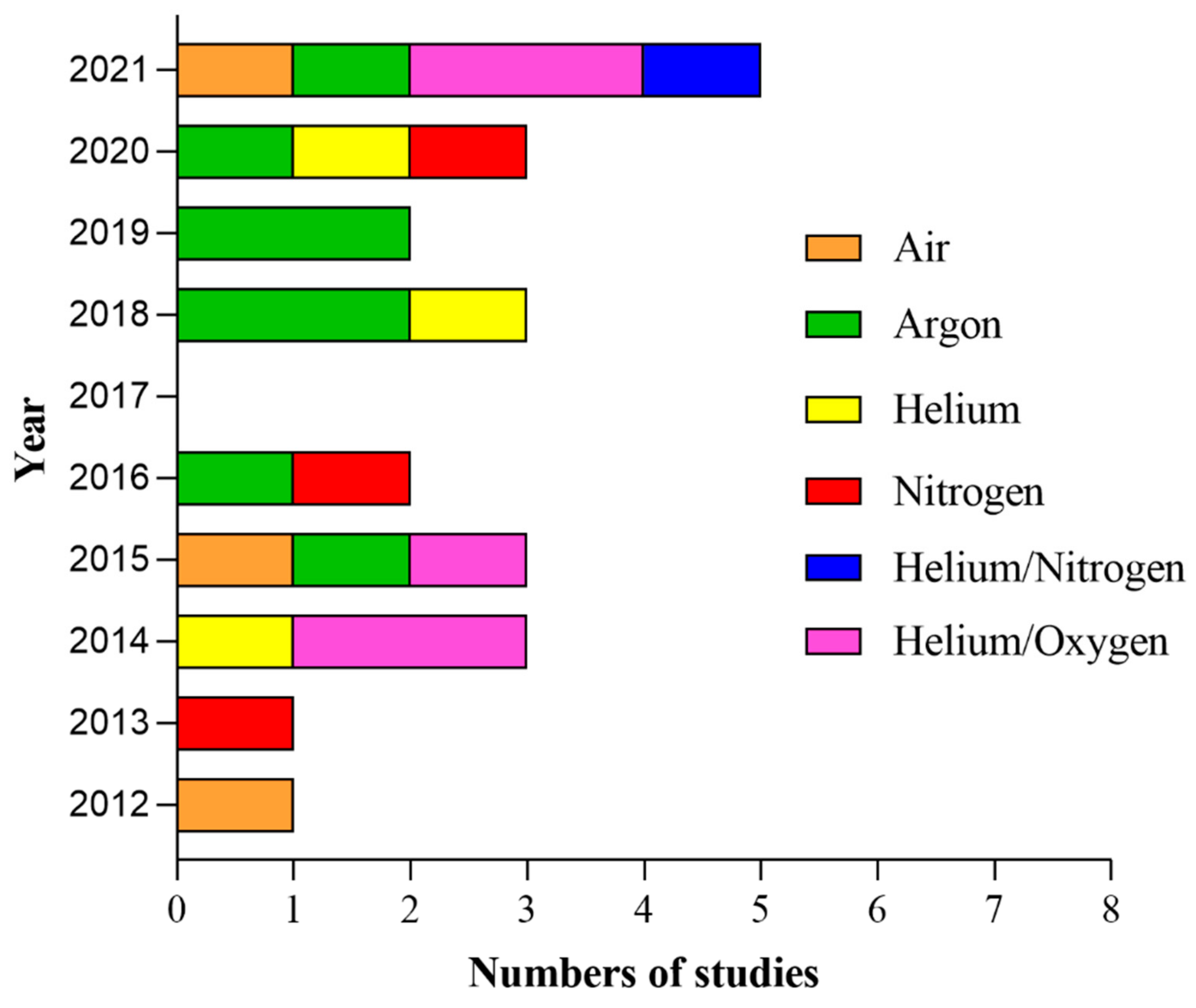
In the 24 included studies, plasma treatment was delivered directly by 13 different kinds of devices, and kINPen MED was employed in four studies [15,85,86,87], while a PAM produced by the same device was delivered in one study [72]. He + O2 was used in two studies [36,73] directly, while two studies [74,77] exposed cells to an LTP produced by this device; only in Sklias et al. [81] was the mixture used both directly and indirectly. Similarly, He-CAP was used in two studies [5,79] directly, while another study [6] served as a PAM produced by the He-CAP jet. The most used gas was Ar in eight studies [15,38,72,78,80,85,86,87], followed by He + O2 [36,65,66,69,73] and finally He alone [5,6,79], N2 alone [23,71,75] and air [37,76,82] in three studies each; He + N2 was delivered only in one study [83] (Figure 6). The study of Dai et al. [84] lacked this information. Application protocol widely varied among studies, including pulse frequency, pulse, flow rate, plasma temperature, source application distance and time, total emitted energy, and power. Overall, the pulse ranged between 1.2 and 30 kV; pulse frequency ranged between 0.05 and 30 kHz, while two studies [75,80] reported 60 Hz, and in the study, a pulse frequency of 13.56 MHz was applied [83]. While application distance and time ranged between 2 and 30 mm and 1 and 300 s, respectively, most of the studies lacked information about temperature, energy, and power. In humans, a pulse frequency of 1 MHz was mainly used. There is still an incomplete understanding of the critical CAP treatment characteristics for effective therapeutic clinical dosing. All the parameters used in the included 24 studies are summarized and outlined below (Table 4).
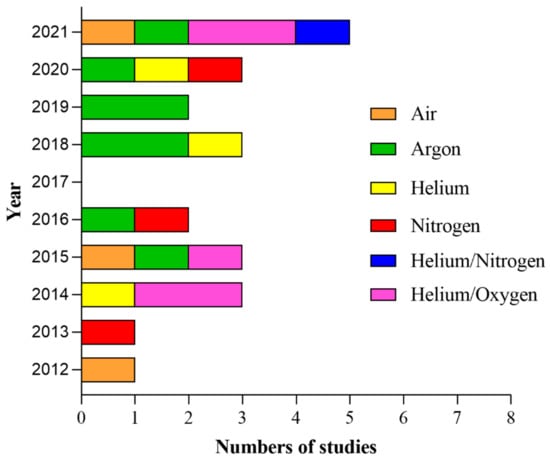
Figure 6.
Yearly distribution of the 23 articles (Dai et al. [84] has been excluded because the carrier gas was not specified in the articles) in the present systematic review according to the type of carrier gas.

Table 4.
Characteristics of plasma devices.
4. Discussion
The anti-cancer mechanism of CAP treatment is still an open question. In this systematic review, a great variability regarding CAP conditions of use has emerged, and this calls for an urgent need for protocols’ standardization to evaluate and compare the effectiveness of plasma treatments in a laboratory setting, with possibilities to extrapolate these methods to clinical tests and finally to favor the clinical translation of plasma as a precision medicinal approach. In contrast to traditional anti-cancer treatments, the primary benefit of CAP is its selectivity toward tumor cells which is an essential feature in the era of targeted therapies [89]. Indeed, the therapy of tumors with anti-cancer drugs faces three important problems: specificity of the treatment, resistance of tumor cells, and penetration of the treatment [90]. Due to its physical—ultraviolet, heat, and electromagnetic field—and chemical—reactive species produced in the gas phase of CAP—properties, CAP is a multimodal therapeutic tool [91] that could offer an answer to each of these challenges and provide further opportunities to overcome critical issues such as tumor plasticity and heterogeneity, which are accounted for the causes of the failure of traditional approaches [82,92].
Understanding CAP selectivity is one of the key challenges in the field of plasma cancer therapy. Several hypotheses have been advanced to explain the selectivity of CAP; however, two models have been proposed [93]. First, further oxidative stress can more easily exceed the cellular survival limit in cancer cells. Indeed, due to stronger metabolism in cancer cells, the basal ROS level in these cells is higher than that in normal cells [50,94,95]. When supplementary ROS stress is generated by CAP treatment, the intracellular ROS level in cancer cells overcomes a threshold more easily than that in normal cells [96], resulting in the activation of apoptotic cell death pathways. However, the basal-ROS level model can just explain the production of higher ROS levels in the CAP-treated tumor cells but cannot explain the selective increase in ROS only in cancer cells. Nevertheless, it is generally accepted that RONS are the major agents responsible for CAP-promoted biological outcomes [97]. Indeed, it has been shown that the physical effects seem to have a minor biological impact, while RONS may induce intracellular ROS increase, antioxidant enzymes decrease, DNA double-strand brakes, and consequently, apoptosis, according to cell type and exposure parameters [98]. Ramireddy et al. [79] showed that H2O2 generation increased immediately after He plasma treatment accounting for the anti-cancer effects observed in OSCC cell lines. Lin A. et al. [76] demonstrated that the generation of RONS and cell death depended on the total delivered energy during treatment, while it is independent of pulse frequencies and treatment times. Both direct and indirect CAP applications produce different RONS environments, although there are no remarkable distinctions in their effects [99]. Indeed, both types of treatment decreased HNC cell viability by triggering more apoptosis in cancer cells than in normal cells, with the benefit that indirect treatment using PAM makes CAP treatment an innovative drug therapy [6]. To note, Oh et al. [77] showed that PAM reduced HNC cell viability in a time-dependent manner while cytotoxicity was not observed in the normal control cell line. In addition, annexin V-FITC/PI analysis showed statistically significant enhancements of the fractions of cells corresponding to early apoptotic cells (FITC+/PI−) and late apoptotic cells (FITC+/PI+) [77]. The decreased viability of OSCC vs. HGF cell lines was also shown by Lee et al. [75], who demonstrated the absence of changes in cell cycle components in normal cells after CAP treatment. Moreover, they showed that CAP led to degradation and dephosphorylation of EGFR, overexpressed in OSCC cells, through a mechanism NO-dependent as revealed by pretreatment of cells with NO scavenger [75]. However, the selectivity was not always established. Indeed, in the study by Hasse et al. [72], normal keratinocytes and HNC cell lines revealed very little difference with respect to the induction of apoptotic events after CAP exposure, although these results were in contrast with the ex vivo experiments, which demonstrated a predominant apoptosis induction in tumor tissue while in healthy tissue the impact of CAP was not significant. Moreover, Sklias et al. [81] demonstrated that indirect plasma treatment is as efficient at killing tumor cells as an appropriate combination of H2O2, NO2− and acidic pH in ad hoc solutions, while sparing normal cells.
Apoptosis was the most investigated pathway in the included studies. Lee et al. [75] showed an increase in apoptosis detected in HNC cells killed via sub G1 arrest through Annexin V-PI staining. He-CAP induced cell death and cell cycle arrest and activated mitochondria-mediated apoptosis by enhancing Bax expression and suppressing Bcl-2 protein expression by increasing intracellular RONS [79]. Hasse et al. [72] showed a cell death mechanism based on caspase 3/7 pathways activation. The main pathway leading to apoptosis was triggered via DNA and mitochondrial damage. Oh et al. [77] observed reduced viability after NTPAM treatment of three HNC cell lines due to enhanced apoptosis associated with an increase in mitochondrial ROS by upregulation of ATF4, a transcription factor expressed in the context of sustained endoplasmic reticulum and mitochondrial stress, and CHOP, a molecule activated by ATF4 that regulates the expression of pro-apoptosis-related genes. According to Kim SY et al. [74], plasma-induced apoptosis was linked to AKT1 ubiquitination and degradation initiated by the mitochondrial protein MUL1, an E3 ligase known to regulate cell growth and death. On the other hand, Chang et al. [36] showed the influence of p53 mutational status on CAP treatment and demonstrated that DNA damage was dependent on the ATM/p53 signaling pathway by sub-G1 arrest. According to Kang et al. [73], apoptosis was induced by MAPK-dependent mitochondrial ROS. Double strand breaks (DSB) are an important mechanism of DNA damage induced by CAP [23]. The number of cells with DSB varied due to the distance from the irradiation center and duration of exposure [23]. Only Chauvin et al. [6] compared the response to PAM treatment in monolayer cultures (2D) and multi-cellular tumor spheroids (MCTS) and found that multiple treatments were needed to obtain a total disruption of spheroids, demonstrating that MCTS models mimic closer to an in vivo tumor behavior. By contrast, in some cases, the involvement of a viability inhibition mechanism mediated by apoptosis has not been demonstrated. Indeed, Guerrero-Preston et al. [5] found that CAP selectively diminished HNC cell viability in a dose-response manner; however, Western blot analysis did not provide evidence that the cleavage of PARP occurred following CAP treatment, thus suggesting that CAP selectively impairs HNC cell lines through non-apoptotic mechanisms while having a minimal effect on normal oral cavity epithelial cell lines. In Hasse et al. [72], ex vivo tissue biopsies treated with CAP revealed an increased number of apoptotic cells within the tissue paralleled by increased levels of cytochrome C in the extracellular liquid, suggesting apoptotic cell damage, in accordance with clinical findings in HNC patients [87].
Finally, the investigation of CAP combination treatments, although in their infancy, demonstrated promising results. Lee et al. [38] found a synergistic effect of the combination of CAP and cisplatin-mediated chemotherapy on ROS generation, apoptosis, and anti-cancer activity with low cytotoxicity against normal cells. Choi et al. [37] demonstrated a significant decrease in the viability of cells pretreated with an anti-EGFR antibody conjugated with gold nanoparticles before CAP irradiation. Finally, Park et al. [78] investigated a novel combination therapy by gold nanoparticles conjugated to programmed cell death protein ligand 1 (PD-L1) antibodies and CAP, which resulted in a greater number of dead cells compared to other experimental groups.
5. Conclusions
In the last years, growing evidence supports the effectiveness of plasma-produced RONS by eliciting a broad spectrum of effects. Perhaps the most relevant concern is the variability in RONS generation due to variations in environmental or sample conditions. Welz et al. [82] emphasize that a comparison of various experiments carried out with different CAP devices is difficult due to different CAP parameters and designs, i.e., DBD (dielectric barrier discharges) technology, SMD technology, power input, voltage, frequency, carrier gas resulting in relevant differences in the production of their components. Moreover, experiments performed with the same plasma device also led to different results when diverse cell lines (malignant/non-malignant) were targeted.
Thus, only delivering controllable doses of RONS to target biological systems can elicit specific effects such as cell death, inhibition of cell proliferation, and migration. Therefore, since any successful medical technology must be predictable and repeatable, the dose for medical application of plasma, the monitoring, and control are the challenges in dealing with the application of plasma in the medical field. Consequently, there is an urgent need for standardization of plasma sources, reliable protocols, and multidisciplinary guidelines to evaluate and compare the effectiveness of CAP treatments in a laboratory setting, with possibilities to extrapolate these methods to clinical tests. Last but not least, a dialogue between plasma experts (physicists, chemists, and engineers), biologists, and physicians to boost the information exchange and, therefore, the publication of results, is strongly required.
Moreover, to be considered an anti-cancer treatment approach with a solid theoretical basis, the underlying mechanisms driving CAP-induced biological effects need to be fully understood. Synergistic effects of CAP with chemotherapy and immunotherapy are demonstrated in vitro [38,78]. The aim of such combination treatments is to reduce effective doses of radiation or chemotherapy drugs, thus lowering the side effects. Traditional surgery and chemo- and radiotherapy may be associated with psychological effects in HNC patients due to potential negative changes in appearance as well as physiological implications of functional defects (speaking, eating, and even breathing) of affected facial organs. Such patients may benefit from CAP treatment to selectively clear the margin of cancer cells without the need to remove large areas of normal tissue. Using the antimicrobial effect induced by plasma, CAP application was successful in palliative care of patients with advanced HNC presenting infected tumor ulcerations. Because of the treatment, the microbial load and the resulting typical fetid odor were reduced. Apart from additional pain reduction, in some cases also, transient tumor remission occurred [15,85,87]. Therefore, CAP can also be used to treat the remaining healthy tissues to decontaminate the site, thus removing pathogenic microorganisms, promoting blood coagulation, and stimulating regeneration and healing of healthy tissue. This could significantly improve the quality of life of HNC-affected patients by decreasing the microbial burden on tumors and enhancing social interaction—an aspect that by itself should be a motivator to scientists in the field of plasma oncotherapy. However, tumor recurrence was observed. This result raises the urgency to understand why the anti-tumor effect does not persist over time and the need for clinical trials with longer follow-ups.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms231810238/s1.
Author Contributions
Conceptualization, V.P.; data base search V.P., V.C.A.C. and M.M.; data analysis and interpretation, V.P., V.C.A.C., L.L.M., E.H.C., M.C.D.M., M.M., N.K.K. and G.M.; study selection and data extraction V.P., V.C.A.C. and G.M.; writing—original draft preparation V.P., V.C.A.C. and G.M.; artwork preparation M.C.D.M.; writing—review and editing M.C.D.M., L.L.M., M.M., N.K.K. and E.H.C.; preparation of image 5 N.K.K. All authors have read and agreed to the published version of the manuscript.
Funding
This work is partially supported by the National Research Foundation (NRF) of Korea (2021R1A6A1A03038785, 2021R1F1A1055694.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Chow, L.Q.M. Head and Neck Cancer. N. Engl. J. Med. 2020, 382, 60–72. [Google Scholar] [CrossRef]
- Svahn, M.F.; Munk, C.; Nielsen, T.S.S.; von Buchwald, C.; Frederiksen, K.; Kjaer, S.K. Trends in all-cause five-year mortality after head and neck cancers diagnosed over a period of 33 years. Focus on estimated degree of association with human papillomavirus. Acta Oncol. 2016, 55, 1084–1090. [Google Scholar] [CrossRef]
- National Cancer Institute. Cancer Stat Facts: Oral Cavity and Pharynx Cancer. Available online: https://seer.cancer.gov/statfacts/html/oralcav.html (accessed on 24 June 2022).
- Guerrero-Preston, R.; Ogawa, T.; Uemura, M.; Shumulinsky, G.; Valle, B.L.; Pirini, F.; Ravi, R.; Sidransky, D.; Keidar, M.; Trink, B. Cold atmospheric plasma treatment selectively targets head and neck squamous cell carcinoma cells. Int. J. Mol. Med. 2014, 34, 941–946. [Google Scholar] [CrossRef]
- Chauvin, J.; Judee, F.; Merbahi, N.; Vicendo, P. Effects of Plasma Activated Medium on Head and Neck FaDu Cancerous Cells: Comparison of 3D and 2D Response. Anticancer Agents Med. Chem. 2018, 18, 776–783. [Google Scholar] [CrossRef]
- Bolt, J.; Vo, Q.N.; Kim, W.J.; McWhorter, A.J.; Thomson, J.; Hagensee, M.E.; Friedlander, P.; Brown, K.D.; Gilbert, J. The ATM/p53 pathway is commonly targeted for inactivation in squamous cell carcinoma of the head and neck (SCCHN) by multiple molecular mechanisms. Oral Oncol. 2005, 41, 1013–1020. [Google Scholar] [CrossRef]
- Johnson, D.E.; Burtness, B.; Leemans, C.R.; Lui, V.W.Y.; Bauman, J.E.; Grandis, J.R. Head and neck squamous cell carcinoma. Nat. Rev. Dis. Primers 2020, 6, 92. [Google Scholar] [CrossRef]
- Argiris, A.; Karamouzis, M.V.; Raben, D.; Ferris, R.L. Head and neck cancer. Lancet 2008, 371, 1695–1709. [Google Scholar] [CrossRef]
- Pierik, A.S.; Leemans, C.R.; Brakenhoff, R.H. Resection Margins in Head and Neck Cancer Surgery: An Update of Residual Disease and Field Cancerization. Cancers 2021, 13, 2635. [Google Scholar] [CrossRef]
- Cavicchi, O.; Piccin, O.; Galasso, F.S.; Marchi, C.; Cantaroni, C.; Latini, G.; Rossi, M.T.; Di Candia, G.; Caliceti, U.; Ceroni, A.R. Multicenter survey through a questionnaire on the prognostic value of surgical resection margin in head and neck tumors. Acta Otorhinolaryngol. Ital. 2000, 20, 413–417. [Google Scholar]
- Lam, L.; Samman, N. Speech and swallowing following tongue cancer surgery and free flap reconstruction—A systematic review. Oral Oncol. 2013, 49, 507–524. [Google Scholar] [CrossRef] [PubMed]
- Brook, I. Late side effects of radiation treatment for head and neck cancer. Radiat. Oncol. J. 2020, 38, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Elaldi, R.; Roussel, L.M.; Gal, J.; Scheller, B.; Chamorey, E.; Schiappa, R.; Lasne-Cardon, A.; Louis, M.Y.; Culié, D.; Dassonville, O.; et al. Correlations between long-term quality of life and patient needs and concerns following head and neck cancer treatment and the impact of psychological distress. A multicentric cross-sectional study. Eur. Arch. Otorhinolaryngol. 2021, 278, 2437–2445. [Google Scholar] [CrossRef] [PubMed]
- Metelmann, H.R.; Seebauer, C.; Miller, V.; Fridman, A.; Bauer, G.; Graves, D.B.; Pouvesle, J.M.; Rutkowski, R.; Schuster, M.; Bekeschus, S.; et al. Clinical experience with cold plasma in the treatment of locally advanced head and neck cancer. Clin. Plasma Med. 2018, 9, 6–13. [Google Scholar] [CrossRef]
- Laroussi, M. Plasma Medicine: A Brief Introduction. Plasma 2018, 1, 5. [Google Scholar] [CrossRef]
- Silva-Teixeira, R.; Laranjo, M.; Lopes, B.; Almeida-Ferreira, C.; Gonçalves, A.C.; Rodrigues, T.; Matafome, P.; Sarmento-Ribeiro, A.B.; Caramelo, F.; Botelho, M.F. Plasma activated media and direct exposition can selectively ablate retinoblastoma cells. Free Radic. Biol. Med. 2021, 171, 302–313. [Google Scholar] [CrossRef]
- Bogaerts, A.; Neyts, E.C. Plasma Technology: An Emerging Technology for Energy Storage. ACS Energy Lett. 2018, 3, 1013–1027. [Google Scholar] [CrossRef]
- Witzke, K.; Seebauer, C.; Jesse, K.; Kwiatek, E.; Berner, J.; Semmler, M.-L.; Boeckmann, L.; Emmert, S.; Weltmann, K.-D.; Metelmann, H.-R.; et al. Plasma medical oncology: Immunological interpretation of head and neck squamous cell carcinoma. Plasma Process. Polym. 2020, 17, 1900258. [Google Scholar] [CrossRef]
- Reuter, S.; von Woedtke, T.; Weltmann, K.D. The kINPen—A review on physics and chemistry of the atmospheric pressure plasma jet and its applications. J. Phys. D Appl. Phys. 2018, 51, 233001. [Google Scholar] [CrossRef]
- Stratmann, B.; Costea, T.C.; Nolte, C.; Hiller, J.; Schmidt, J.; Reindel, J.; Masur, K.; Motz, W.; Timm, J.; Kerner, W.; et al. Effect of Cold Atmospheric Plasma Therapy vs Standard Therapy Placebo on Wound Healing in Patients With Diabetic Foot Ulcers: A Randomized Clinical Trial. JAMA Netw. Open 2020, 3, e2010411. [Google Scholar] [CrossRef]
- Schweigert, I.; Zakrevsky, D.; Gugin, P.; Yelak, E.; Golubitskaya, E.; Troitskaya, O.; Koval, O. Interaction of Cold Atmospheric Argon and Helium Plasma Jets with Bio-Target with Grounded Substrate Beneath. Appl. Sci. 2019, 9, 4528. [Google Scholar] [CrossRef] [Green Version]
- Han, X.; Klas, M.; Liu, Y.Y.; Stack, M.S.; Ptasinska, S. DNA damage in oral cancer cells induced by nitrogen atmospheric pressure plasma jets. Appl. Phys. Lett. 2013, 102, 233703. [Google Scholar] [CrossRef]
- Boehm, D.; Bourke, P. Safety implications of plasma-induced effects in living cells—A review of in vitro and in vivo findings. Biol. Chem. 2018, 400, 3–17. [Google Scholar] [CrossRef]
- Assadian, O.; Ousey, K.J.; Daeschlein, G.; Kramer, A.; Parker, C.; Tanner, J.; Leaper, D.J. Effects and safety of atmospheric low-temperature plasma on bacterial reduction in chronic wounds and wound size reduction: A systematic review and meta-analysis. Int. Wound J. 2019, 16, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Vandamme, M.; Robert, E.; Lerondel, S.; Sarron, V.; Ries, D.; Dozias, S.; Sobilo, J.; Gosset, D.; Kieda, C.; Legrain, B.; et al. ROS implication in a new antitumor strategy based on non-thermal plasma. Int. J. Cancer 2012, 130, 2185–2194. [Google Scholar] [CrossRef]
- Shakouri, R.; Khani, M.R.; Samsavar, S.; Jezeh, M.A.; Abdollahimajd, F.; Hosseini, S.I.; Dilmaghanian, A.; Ghasemi, E.; Alihoseini, M.R.; Shokri, B. In vivo study of the effects of a portable cold plasma device and vitamin C for skin rejuvenation. Sci. Rep. 2021, 11, 21915. [Google Scholar] [CrossRef] [PubMed]
- Bednarik, M.; Mizera, A.; Manas, M.; Navratil, M.; Huba, J.; Achbergerova, E.; Stoklasek, P. Influence of the β-Radiation/Cold Atmospheric-Pressure Plasma Surface Modification on the Adhesive Bonding of Polyolefins. Materials 2020, 14, 76. [Google Scholar] [CrossRef]
- Carvalho, A.; Barros, D.R.; da Silva, L.S.; Sanches, E.A.; Pinto, C.D.C.; de Souza, S.M.; Clerici, M.; Rodrigues, S.; Fernandes, F.A.N.; Campelo, P.H. Dielectric barrier atmospheric cold plasma applied to the modification of Ariá (Goeppertia allouia) starch: Effect of plasma generation voltage. Int. J. Biol. Macromol. 2021, 182, 1618–1627. [Google Scholar] [CrossRef]
- Dolci, L.S.; Liguori, A.; Panzavolta, S.; Miserocchi, A.; Passerini, N.; Gherardi, M.; Colombo, V.; Bigi, A.; Albertini, B. Non-equilibrium atmospheric pressure plasma as innovative method to crosslink and enhance mucoadhesion of econazole-loaded gelatin films for buccal drug delivery. Colloids Surf. B Biointerfaces 2018, 163, 73–82. [Google Scholar] [CrossRef]
- Hua, D.; Cai, D.; Ning, M.; Yu, L.; Zhang, Z.; Han, P.; Dai, X. Cold atmospheric plasma selectively induces G(0)/G(1) cell cycle arrest and apoptosis in AR-independent prostate cancer cells. J. Cancer 2021, 12, 5977–5986. [Google Scholar] [CrossRef]
- Liedtke, K.R.; Diedrich, S.; Pati, O.; Freund, E.; Flieger, R.; Heidecke, C.D.; Partecke, L.I.; Bekeschus, S. Cold Physical Plasma Selectively Elicits Apoptosis in Murine Pancreatic Cancer Cells In Vitro and In Ovo. Anticancer Res. 2018, 38, 5655–5663. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Holmes, B.; Cheng, X.; Zhu, W.; Keidar, M.; Zhang, L.G. Cold atmospheric plasma for selectively ablating metastatic breast cancer cells. PLoS ONE 2013, 8, e73741. [Google Scholar] [CrossRef] [PubMed]
- Semmler, M.L.; Bekeschus, S.; Schäfer, M.; Bernhardt, T.; Fischer, T.; Witzke, K.; Seebauer, C.; Rebl, H.; Grambow, E.; Vollmar, B.; et al. Molecular Mechanisms of the Efficacy of Cold Atmospheric Pressure Plasma (CAP) in Cancer Treatment. Cancers 2020, 12, 269. [Google Scholar] [CrossRef] [PubMed]
- Solé-Martí, X.; Espona-Noguera, A.; Ginebra, M.P.; Canal, C. Plasma-Conditioned Liquids as Anticancer Therapies In Vivo: Current State and Future Directions. Cancers 2021, 13, 452. [Google Scholar] [CrossRef]
- Chang, J.W.; Kang, S.U.; Shin, Y.S.; Kim, K.I.; Seo, S.J.; Yang, S.S.; Lee, J.S.; Moon, E.; Baek, S.J.; Lee, K.; et al. Non-thermal atmospheric pressure plasma induces apoptosis in oral cavity squamous cell carcinoma: Involvement of DNA-damage-triggering sub-G(1) arrest via the ATM/p53 pathway. Arch. Biochem. Biophys. 2014, 545, 133–140. [Google Scholar] [CrossRef]
- Choi, B.B.; Choi, Y.S.; Lee, H.J.; Lee, J.K.; Kim, U.K.; Kim, G.C. Nonthermal Plasma-Mediated Cancer Cell Death; Targeted Cancer Treatment. J. Therm. Sci. Technol. 2012, 7, 399–404. [Google Scholar] [CrossRef]
- Lee, C.M.; Jeong, Y.I.; Kook, M.S.; Kim, B.H. Combinatorial Effect of Cold Atmosphere Plasma (CAP) and the Anticancer Drug Cisplatin on Oral Squamous Cell Cancer Therapy. Int. J. Mol. Sci. 2020, 21, 7646. [Google Scholar] [CrossRef]
- Lafontaine, J.; Boisvert, J.S.; Glory, A.; Coulombe, S.; Wong, P. Synergy between Non-Thermal Plasma with Radiation Therapy and Olaparib in a Panel of Breast Cancer Cell Lines. Cancers 2020, 12, 348. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. Rev. Esp. Cardiol. (Engl. Ed.) 2021, 74, 790–799. [Google Scholar] [CrossRef]
- Huang, S.H.; O’Sullivan, B. Overview of the 8th Edition TNM Classification for Head and Neck Cancer. Curr. Treat. Options Oncol. 2017, 18, 40. [Google Scholar] [CrossRef]
- Mohammad, N.S.; Nazli, R.; Zafar, H.; Fatima, S. Effects of lipid based Multiple Micronutrients Supplement on the birth outcome of underweight pre-eclamptic women: A randomized clinical trial. Pak. J. Med. Sci. 2022, 38, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Marques, M.M.; Diniz, I.M.; de Cara, S.P.; Pedroni, A.C.; Abe, G.L.; D′Almeida-Couto, R.S.; Lima, P.L.; Tedesco, T.K.; Moreira, M.S. Photobiomodulation of Dental Derived Mesenchymal Stem Cells: A Systematic Review. Photomed. Laser Surg. 2016, 34, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Hooijmans, C.R.; Rovers, M.M.; de Vries, R.B.M.; Leenaars, M.; Ritskes-Hoitinga, M.; Langendam, M.W. SYRCLE’s risk of bias tool for animal studies. BMC Med. Res. Methodol. 2014, 14, 43. [Google Scholar] [CrossRef] [PubMed]
- Wells, G.; Shea, B.; O′Connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle–Ottawa Scale (NOS) for Assessing the Quality of Non-Randomized Studies in Meta-Analysis; Academic Press: Cambridge, MA, USA, 2000; Available online: https://www.ohri.ca (accessed on 4 August 2022).
- Murad, M.H.; Sultan, S.; Haffar, S.; Bazerbachi, F. Methodological quality and synthesis of case series and case reports. BMJ Evid. Based Med. 2018, 23, 60–63. [Google Scholar] [CrossRef] [PubMed]
- Pannone, G.; Caponio, V.C.A.; De Stefano, I.S.; Ramunno, M.A.; Meccariello, M.; Agostinone, A.; Pedicillo, M.C.; Troiano, G.; Zhurakivska, K.; Cassano, T.; et al. Lung histopathological findings in COVID-19 disease—A systematic review. Infect. Agent Cancer 2021, 16, 34. [Google Scholar] [CrossRef]
- Furusaka, T.; Kida, A.; Aoyagi, M.; Makiyama, K.; Iida, H.; Miyakogawa, M.; Kikuchi, K.; Tomita, H. CAP therapy for advanced, recurrent and/or metastatic malignant tumors of the head and neck. Auris Nasus Larynx 1986, 13 (Suppl. 1), S119–S126. [Google Scholar] [CrossRef]
- Akhmedov, I.N.; Isaev, V.M.; Svistushkin, V.M.; Mustafaev, D.M.; Osipenko, E.V. Endolaryngeal cold-plasma microsurgery for the treatment of benign neoplasms in the larynx and the objective assessment of its functional results. Vestn. Otorinolaringol. 2011, 4, 57–60. [Google Scholar]
- Keidar, M.; Walk, R.; Shashurin, A.; Srinivasan, P.; Sandler, A.; Dasgupta, S.; Ravi, R.; Guerrero-Preston, R.; Trink, B. Cold plasma selectivity and the possibility of a paradigm shift in cancer therapy. Br. J. Cancer 2011, 105, 1295–1301. [Google Scholar] [CrossRef]
- Virard, F.; Cousty, S.; Cambus, J.P.; Valentin, A.; Kémoun, P.; Clément, F.; Gallyas, F., Jr. Cold atmospheric plasma induces a predominantly necrotic cell death via the microenvironment. PLoS ONE 2015, 10, e0133120. [Google Scholar] [CrossRef]
- Adhikari, E.R.; Ptasinska, S. Correlation between helium atmospheric pressure plasma jet (APPJ) variables and plasma induced DNA damage. Eur. Phys. J. D 2016, 70, 180. [Google Scholar] [CrossRef]
- Laurita, R.; Alviano, F.; Marchionni, C.; Abruzzo, P.M.; Bolotta, A.; Bonsi, L.; Colombo, V.; Gherardi, M.; Liguori, A.; Ricci, F.; et al. A study of the effect on human mesenchymal stem cells of an atmospheric pressure plasma source driven by different voltage waveforms. J. Phys. D Appl. Phys. 2016, 49, 364003. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, K.N. Effects of a Nonthermal Atmospheric Pressure Plasma Jet on Human Gingival Fibroblasts for Biomedical Application. Biomed. Res. Int. 2016, 2016, 2876916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang, S.U.; Choi, J.W.; Chang, J.W.; Kim, K.I.; Kim, Y.S.; Park, J.K.; Kim, Y.E.; Lee, Y.S.; Yang, S.S.; Kim, C.H. N2 non-thermal atmospheric pressure plasma promotes wound healing in vitro and in vivo: Potential modulation of adhesion molecules and matrix metalloproteinase-9. Exp. Dermatol. 2017, 26, 163–170. [Google Scholar] [CrossRef]
- Han, X.; Liu, Y.; Stack, M.S.; Ptasinska, S. 3D Mapping of plasma effective areas via detection of cancer cell damage induced by atmospheric pressure plasma jets. In Journal of Physics: Conference Series; IOP Publishing: Bristol, UK, 2014. [Google Scholar]
- Hajizadeh, K.; Hajisharifi, K.; Mehdian, H. Morphological risk assessment of cold atmospheric plasma-based therapy: Bone marrow mesenchymal stem cells in treatment zone proximity. J. Phys. D Appl. Phys. 2019, 52, 495203. [Google Scholar] [CrossRef]
- Jaiswal, A.S.; Kumar, R.; Thakar, A.; Kumar, R.; Verma, H.; Bhalla, A.S.; Kairo, A.K.; Kakkar, A.; Sharma, S.C.; Sakthivel, P. Plasma ablation-assisted endoscopic excision versus traditional technique of endoscopic excision of juvenile nasopharyngeal angiofibroma. Int. J. Pediatr. Otorhinolaryngol. 2020, 139, 110410. [Google Scholar] [CrossRef]
- Jing, J.; Li, L.; Zheng, L.; Wu, Y.; Jiang, S.; Zhang, L.; Wang, H.; Liu, G. Research on the efficacy of laryngendoscopic low-temperature plasma ablation on early glottic cancer. J. BUON 2020, 25, 2382–2388. [Google Scholar]
- Evert, K.; Kocher, T.; Schindler, A.; Muller, M.; Muller, K.; Pink, C.; Holtfreter, B.; Schmidt, A.; Dombrowski, F.; Schubert, A.; et al. Repeated exposure of the oral mucosa over 12 months with cold plasma is not carcinogenic in mice. Sci. Rep. 2021, 11, 20672. [Google Scholar] [CrossRef]
- Bekeschus, S.; Eisenmann, S.; Sagwal, S.K.; Bodnar, Y.; Moritz, J.; Poschkamp, B.; Stoffels, I.; Emmert, S.; Madesh, M.; Weltmann, K.D.; et al. xCT (SLC7A11) expression confers intrinsic resistance to physical plasma treatment in tumor cells. Redox Biol. 2020, 30, 101423. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, J.; Liu, Z.; Xu, D.; Guo, L.; Liu, D.; Kong, M.G. Evaluation of the anticancer effects induced by cold atmospheric plasma in 2D and 3D cell-culture models. Plasma Process. Polym. 2019, 16, 1900072. [Google Scholar] [CrossRef]
- Yan, D.Y.; Lin, L.; Sherman, J.H.; Canady, J.; Trink, B.; Keidar, M. The Correlation Between the Cytotoxicity of Cold Atmospheric Plasma and the Extracellular H2O2-Scavenging Rate. IEEE T R PMS 2018, 2, 618–623. [Google Scholar] [CrossRef]
- Wang, P.; Wang, G. Efficacy of low-temperature plasma ablation combined with low-dose cisplatin chemotherapy in laryngeal carcinoma patients and its influence on tumor markers. J. BUON 2021, 26, 132–137. [Google Scholar]
- Weiss, M.; Barz, J.; Ackermann, M.; Utz, R.; Ghoul, A.; Weltmann, K.D.; Stope, M.B.; Wallwiener, D.; Schenke-Layland, K.; Oehr, C.; et al. Dose-Dependent Tissue-Level Characterization of a Medical Atmospheric Pressure Argon Plasma Jet. ACS Appl. Mater. Interfaces 2019, 11, 19841–19853. [Google Scholar] [CrossRef]
- Tanaka, H.; Nakamura, K.; Mizuno, M.; Ishikawa, K.; Takeda, K.; Kajiyama, H.; Utsumi, F.; Kikkawa, F.; Hori, M. Non-thermal atmospheric pressure plasma activates lactate in Ringer’s solution for anti-tumor effects. Sci. Rep. 2016, 6, 36282. [Google Scholar] [CrossRef]
- Moritz, J.; Metelmann, H.R.; Bekeschus, S. Physical Plasma Treatment of Eight Human Cancer Cell Lines Demarcates Upregulation of CD112 as a Common Immunomodulatory Response Element. IEEE T R PMS 2020, 4, 343–349. [Google Scholar] [CrossRef]
- Metelmann, H.R.; Seebauer, C.; Rutkowski, R.; Schuster, M.; Bekeschus, S.; Metelmann, P. Treating cancer with cold physical plasma: On the way to evidence-based medicine. Contrib. Plasma Phys. 2018, 58, 415–419. [Google Scholar] [CrossRef]
- Ghimire, B.; Szili, E.J.; Lamichhane, P.; Short, R.D.; Lim, J.S.; Attri, P.; Masur, K.; Weltmann, K.D.; Hong, S.H.; Choi, E.H. The role of UV photolysis and molecular transport in the generation of reactive species in a tissue model with a cold atmospheric pressure plasma jet. Appl. Phys. Lett. 2019, 114, 093701. [Google Scholar] [CrossRef]
- Biscop, E.; Lin, A.; Van Boxem, W.; Van Loenhout, J.; De Backer, J.; Deben, C.; Dewilde, S.; Smits, E.; Bogaerts, A. Influence of cell type and culture medium on determining cancer selectivity of cold atmospheric plasma treatment. Cancers 2019, 11, 1287. [Google Scholar] [CrossRef]
- Han, X.; Kapaldo, J.; Liu, Y.; Stack, M.S.; Alizadeh, E.; Ptasinska, S. Large-Scale Image Analysis for Investigating Spatio-Temporal Changes in Nuclear DNA Damage Caused by Nitrogen Atmospheric Pressure Plasma Jets. Int. J. Mol. Sci. 2020, 21, 4127. [Google Scholar] [CrossRef]
- Hasse, S.; Seebauer, C.; Wende, K.; Schmidt, A.; Metelmann, H.R.; von Woedtke, T.; Bekeschus, S. Cold argon plasma as adjuvant tumour therapy on progressive head and neck cancer: A preclinical study. Appl. Sci. 2019, 9, 2061. [Google Scholar] [CrossRef]
- Kang, S.U.; Cho, J.H.; Chang, J.W.; Shin, Y.S.; Kim, K.I.; Park, J.K.; Yang, S.S.; Lee, J.S.; Moon, E.; Lee, K.; et al. Nonthermal plasma induces head and neck cancer cell death: The potential involvement of mitogen-activated protein kinase-dependent mitochondrial reactive oxygen species. Cell Death Dis. 2014, 5, e1056. [Google Scholar] [CrossRef]
- Kim, S.Y.; Kim, H.J.; Kang, S.U.; Kim, Y.E.; Park, J.K.; Shin, Y.S.; Kim, Y.S.; Lee, K.; Kim, C.H. Non-thermal plasma induces AKT degradation through turn-on the MUL1 E3 ligase in head and neck cancer. Oncotarget 2015, 6, 33382–33396. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Om, J.Y.; Kim, Y.H.; Kim, K.M.; Choi, E.H.; Kim, K.N. Selective Killing Effects of Cold Atmospheric Pressure Plasma with NO Induced Dysfunction of Epidermal Growth Factor Receptor in Oral Squamous Cell Carcinoma. PLoS ONE 2016, 11, e0150279. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Biscop, E.; Gorbanev, Y.; Smits, E.; Bogaerts, A. Toward defining plasma treatment dose: The role of plasma treatment energy of pulsed-dielectric barrier discharge in dictating in vitro biological responses. Plasma Process. Polym. 2021, 19, e2100151. [Google Scholar] [CrossRef]
- Oh, C.; Won, H.R.; Kang, W.S.; Kim, D.W.; Jung, S.N.; Im, M.A.; Liu, L.; Jin, Y.L.; Piao, Y.; Kim, H.J.; et al. Head and Neck Cancer Cell Death due to Mitochondrial Damage Induced by Reactive Oxygen Species from Nonthermal Plasma-Activated Media: Based on Transcriptomic Analysis. Oxid. Med. Cell Longev. 2021, 2021, 9951712. [Google Scholar] [CrossRef]
- Park, J.; Jang, Y.S.; Choi, J.H.; Ryu, M.; Kim, G.C.; Byun, J.H.; Hwang, D.S.; Kim, U.K. Anticancer Efficacy of Nonthermal Plasma Therapy Combined with PD-L1 Antibody Conjugated Gold Nanoparticles on Oral Squamous Cell Carcinoma. Appl. Sci. 2021, 11, 4559. [Google Scholar] [CrossRef]
- Ramireddy, L.; Lai, C.H.; Low, B.S.; Li, C.; Hsieh, J.H.; Lee, J.W.; Wu, H.Y. Cold atmospheric helium plasma induces apoptosis by increasing intracellular reactive oxygen and nitrogen species. Plasma Med. 2020, 10, 243–257. [Google Scholar] [CrossRef]
- Sato, K.; Shi, L.; Ito, F.; Ohara, Y.; Motooka, Y.; Tanaka, H.; Mizuno, M.; Hori, M.; Hirayama, T.; Hibi, H.; et al. Nonthermal plasma specifically kills oral squamous cell carcinoma cells in a catalytic Fe(II)dependent manner. J. Clin. Biochem. Nutr. 2019, 65, 8–15. [Google Scholar] [CrossRef]
- Sklias, K.; Sousa, J.S.; Girard, P.M. Role of short- and long-lived reactive species on the selectivity and anti-cancer action of plasma treatment in vitro. Cancers 2021, 13, 615. [Google Scholar] [CrossRef]
- Welz, C.; Emmert, S.; Canis, M.; Becker, S.; Baumeister, P.; Shimizu, T.; Morfill, G.E.; Harréus, U.; Zimmermann, J.L. Cold Atmospheric Plasma: A Promising Complementary Therapy for Squamous Head and Neck Cancer. PLoS ONE 2015, 10, e0141827. [Google Scholar] [CrossRef]
- Wu, C.Y.; Liao, J.D.; Chen, C.H.; Lee, H.; Wang, S.H.; Liu, B.H.; Lee, C.Y.; Shao, P.L.; Li, E.Y. Non-Thermal Reactive N-2/He Plasma Exposure to Inhibit Epithelial Head and Neck Tumor Cells. Coatings 2021, 11, 1284. [Google Scholar] [CrossRef]
- Dai, X.; Wei, B.; He, Y.; Zhang, Q.; Xi, J. Postoperative efficacy of low-temperature plasma radiofrequency ablation in elderly patients with laryngeal carcinoma and its influences on tumor markers and COX-2 and VEGF expressions in laryngeal carcinoma tissues. J. BUON 2020, 25, 1969–1975. [Google Scholar] [PubMed]
- Metelmann, H.R.; Nedrelow, D.S.; Seebauer, C.; Schuster, M.; von Woedtke, T.; Weltmann, K.D.; Kindler, S.; Metelmann, P.H.; Finkelstein, S.E.; Von Hoff, D.D.; et al. Head and neck cancer treatment and physical plasma. Clin. Plasma Med. 2015, 3, 17–23. [Google Scholar] [CrossRef]
- Schuster, M.; Rutkowski, R.; Hauschild, A.; Shojaei, R.K.; von Woedtke, T.; Rana, A.; Bauer, G.; Metelmann, P.; Seebauer, C. Side effects in cold plasma treatment of advanced oral cancer-Clinical data and biological interpretation. Clin. Plasma Med. 2018, 10, 9–15. [Google Scholar] [CrossRef]
- Schuster, M.; Seebauer, C.; Rutkowski, R.; Hauschild, A.; Podmelle, F.; Metelmann, C.; Metelmann, B.; von Woedtke, T.; Hasse, S.; Weltmann, K.D.; et al. Visible tumor surface response to physical plasma and apoptotic cell kill in head and neck cancer. J. Cranio Maxillofac. Surg. 2016, 44, 1445–1452. [Google Scholar] [CrossRef]
- Kim, C.-H.; Kwon, S.; Bahn, J.H.; Lee, K.; Jun, S.I.; Rack, P.D.; Baek, S.J. Effects of atmospheric nonthermal plasma on invasion of colorectal cancer cells. Appl. Phys. Lett. 2010, 96, 243701. [Google Scholar] [CrossRef]
- Dubuc, A.; Monsarrat, P.; Virard, F.; Merbahi, N.; Sarrette, J.P.; Laurencin-Dalicieux, S.; Cousty, S. Use of cold-atmospheric plasma in oncology: A concise systematic review. Ther. Adv. Med. Oncol. 2018, 10, 1758835918786475. [Google Scholar] [CrossRef]
- Cheng, Z.; Li, M.; Dey, R.; Chen, Y. Nanomaterials for cancer therapy: Current progress and perspectives. J. Hematol. Oncol. 2021, 14, 85. [Google Scholar] [CrossRef]
- Boeckmann, L.; Schäfer, M.; Bernhardt, T.; Semmler, M.L.; Jung, O.; Ojak, G.; Fischer, T.; Peters, K.; Nebe, B.; Müller-Hilke, B.; et al. Cold Atmospheric Pressure Plasma in Wound Healing and Cancer Treatment. Appl. Sci. 2020, 10, 6898. [Google Scholar] [CrossRef]
- Liu, D.; Zhang, Y.; Xu, M.; Chen, H.; Lu, X.; Ostrikov, K. Cold atmospheric pressure plasmas in dermatology: Sources, reactive agents, and therapeutic effects. Plasma Process. Polym. 2020, 17, e1900218. [Google Scholar] [CrossRef]
- Yan, D.; Sherman, J.H.; Keidar, M. Cold atmospheric plasma, a novel promising anti-cancer treatment modality. Oncotarget 2017, 8, 15977–15995. [Google Scholar] [CrossRef]
- Graves, D.B. The emerging role of reactive oxygen and nitrogen species in redox biology and some implications for plasma applications to medicine and biology. J. Phys. D Appl. Phys. 2012, 45, 233001. [Google Scholar] [CrossRef]
- Ahn, H.J.; Kim, K.I.; Hoan, N.N.; Kim, C.H.; Moon, E.; Choi, K.S.; Yang, S.S.; Lee, J.S. Targeting cancer cells with reactive oxygen and nitrogen species generated by atmospheric-pressure air plasma. PLoS ONE 2014, 9, e86173. [Google Scholar] [CrossRef]
- Keidar, M. Plasma for cancer treatment. Plasma Sources Sci. Technol. 2015, 24, 033001. [Google Scholar] [CrossRef]
- Mitra, S.; Nguyen, L.N.; Akter, M.; Park, G.; Choi, E.H.; Kaushik, N.K. Impact of ROS Generated by Chemical, Physical, and Plasma Techniques on Cancer Attenuation. Cancers 2019, 11, 1030. [Google Scholar] [CrossRef] [Green Version]
- Zubor, P.; Wang, Y.; Liskova, A.; Samec, M.; Koklesova, L.; Dankova, Z.; Dørum, A.; Kajo, K.; Dvorska, D.; Lucansky, V.; et al. Cold Atmospheric Pressure Plasma (CAP) as a New Tool for the Management of Vulva Cancer and Vulvar Premalignant Lesions in Gynaecological Oncology. Int. J. Mol. Sci. 2020, 21, 7988. [Google Scholar] [CrossRef]
- Saadati, F.; Mahdikia, H.; Abbaszadeh, H.A.; Abdollahifar, M.A.; Khoramgah, M.S.; Shokri, B. Comparison of Direct and Indirect cold atmospheric-pressure plasma methods in the B(16)F(10) melanoma cancer cells treatment. Sci. Rep. 2018, 8, 7689. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).