Abstract
RNA interference (RNAi) efficiency dramatically varies among different nematodes, which impacts research on their gene function and pest control. Bursaphelenchus xylophilus is a pine wood nematode in which RNAi-mediated gene silencing has unstable interference efficiency through soaking in dsRNA solutions, the factors of which remain unknown. Using agarose gel electrophoresis, we found that dsRNA can be degraded by nematode secretions in the soaking system which is responsible for the low RNAi efficiency. Based on the previously published genome and secretome data of B. xylophilus, 154 nucleases were screened including 11 extracellular nucleases which are potential factors reducing RNAi efficacy. To confirm the function of nucleases in RNAi efficiency, eight extracellular nuclease genes (BxyNuc1-8) were cloned in the genome. BxyNuc4, BxyNuc6 and BxyNuc7 can be upregulated in response to dsGFP, considered as the major nuclease performing dsRNA degradation. After soaking with the dsRNA of nucleases BxyNuc4/BxyNuc6/BxyNuc7 and Pat10 gene (ineffective in RNAi) simultaneously for 24 h, the expression of Pat10 gene decreased by 23.25%, 26.05% and 11.29%, respectively. With soaking for 36 h, the expression of Pat10 gene decreased by 43.25% and 33.25% in dsBxyNuc6+dsPat10 and dsBxyNuc7+dsPat10 groups, respectively. However, without dsPat10, dsBxyNuc7 alone could cause downregulation of Pat10 gene expression, while dsBxyNuc6 could not disturb this gene. In conclusion, the nuclease BxyNuc6 might be a major barrier to the RNAi efficiency in B. xylophilus.
1. Introduction
The pine wood nematode, Bursaphelenchus xylophilus, is a notorious, invasive, plant-parasitic nematode that causes pine wilt disease [1], which is the most complex and devastating disease in forest ecosystems in China, South Korea, Japan, Portugal and other European countries [2,3,4]. It has caused significant economic and ecological damage in China since it was first reported in Nanjing in 1982 [5]. Pine wilt disease has spread rapidly to 19 provinces including 731 county-level administrative regions in China [6]. Upon invasion of a pine tree, B. xylophilus can reproduce quickly and destroy the vascular system of the entire tree, causing wilting and death in only a few weeks [7]. The main control measures include trunk injection of nematicide and eradication of the damaged trees and vector insects. However, large-scale application of pesticides has led to environmental issues and health concerns [8]. Therefore, development of an efficient and environmentally friendly approach to control B. xylophilus is urgently needed. RNA interference (RNAi) is a cellular mechanism in which double-stranded RNA (dsRNA) molecules drive the post-transcriptional silencing of genes with homologous sequences [9]. Since the RNAi response was first described in the free-living nematode Caenorhabditis elegans [10], it has become a valuable tool for studying gene function of nematodes, and it may be a promising approach for the control of plant-parasitic nematodes [11]. The interference of plant-parasitic nematodes is mostly achieved by soaking [12,13]. However, the efficiency of this approach varies widely with the nematode species [14]. In B. xylophilus, the RNAi efficiency ranged from 30% to 80%, while some studies showed no effect [15,16,17,18]. Even when targeting the same gene, the experimental results of different studies were inconsistent. For example, the mortality rate of pine wood nematode is up to 80% after interference with BxAK1 for 8 h [19]. However, in another report, the nematodes did not die or show the expected phenotype changes by feeding yeast cells, which can express dsRNA to interfere with AK1 and AK2 (iGEM. http://2018igemorg/Team:Kyoto/Experiments (accessed on 15 November 2021)).
Efficient RNAi-induced gene silencing requires some essential processes, including delivery of dsRNA, uptake from the hemolymph or gut, dsRNA processing by RNAi enzymes, intracellular transport and expression of the core RNAi machinery [20]. Above all, the stability of dsRNA is the first step to influencing RNAi efficiency before uptake. When ingested, dsRNA should avoid dsRNase degradation and move from intestinal or hemolymph lumen to tissues to effectively exert its inhibitory effect. In recent years, many studies have suggested that extracellular nuclease is the important factor leading to low RNAi efficiency in insects, such as Ostrinia furnacalis [21] and Drosophila suzukii [22]. When silencing the extracellular nuclease or inhibiting the activity of enzymes, RNAi efficiency is significantly improved. However, different insects have different types and activities of extracellular nucleases in different physiological states, which may be an important factor affecting the difference and instability of RNAi efficiency in insects [23,24]. Plant-parasitic nematodes secrete a variety of functional enzymes during their feeding process to digest cellular components and resist plant defense responses, among which extracellular nucleases play an important biological role in the absorption of nutrients and removal of alien substances [25,26]. At present, the function of plant-parasitic nematode extracellular nucleases is scarcely studied, and their effect on RNAi efficiency is still unclear. In the study of pine wood nematodes, there have been few proposed solutions to the problems of low interference efficiency or ineffective interference.
Through our study we want to draw attention to whether the extracellular nucleases of B. xylophilus can degrade dsRNA in the soaking system, affecting the stability of dsRNA before it is taken up by nematode intestinal cells. Therefore, screening key nucleases and clarifying the function of these enzymes in B. xylophilus RNAi has theoretical significance for the study of functional genes and production of new control methods for B. xylophilus.
2. Results
2.1. In Vitro Degradation of dsRNA by Nematodes
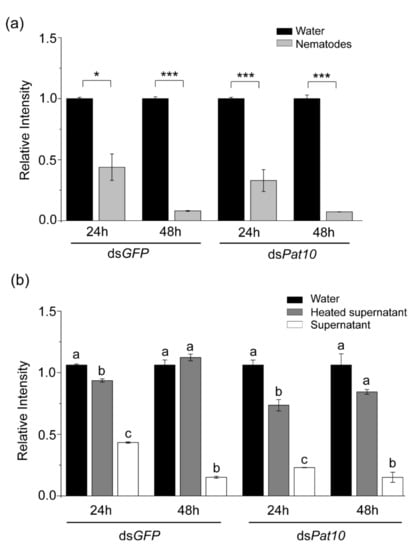
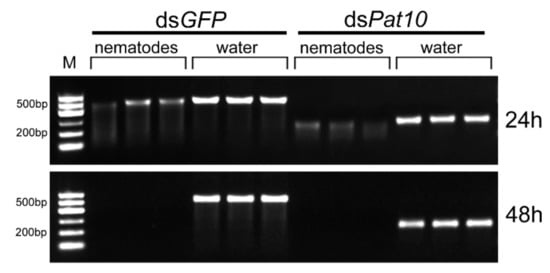
The dsGFP and dsPat10 were incubated at 25 °C with nematodes for 24 and 48 h, respectively. After 24 h, dsRNA showed obvious degradation and the intensity of electrophoretic bands of the nematode treatment group of dsGFP and dsPat10 had significantly reduced by 56.2% and 67.2%, respectively (Figure 1a and Figure 2). After 48 h, the electrophoretic bands of the nematode treatment group were almost invisible (Figure 2). The relative intensity of electrophoretic bands was only 0.081 and 0.073 in dsGFP and dsPat10 groups, respectively (Figure 1a).
Figure 1.
Relative intensity of agarose gel electrophoretic bands. (a) The relative intensity of electrophoretic bands after dsRNA and nematodes soaked for 24 and 48 h. (b) The relative intensity of electrophoretic bands after dsRNA and nematode secretions soaked for 24 and 48 h. Relative intensity differences between two groups were calculated using the independent samples t test. Data are presented as mean ± SE, *** p < 0.001; * p < 0.05; different letters indicate significant difference (p < 0.05).
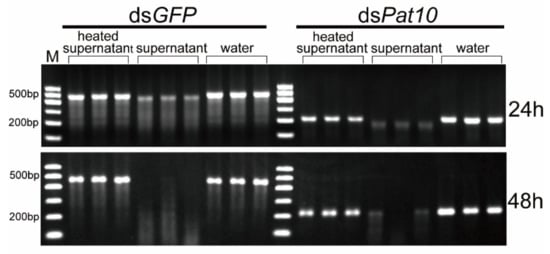
In order to detect whether the secretions of B. xylophilus in water degrade dsRNA, the sterilized supernatant of B. xylophilus was collected. Compared with the supernatant-heated control group and the water-immersed control group, the unheated supernatant treatment group of dsGFP and dsPat10 showed significantly weaker electrophoretic bands at 24 and 48 h (Figure 1b and Figure 3). However, not all of them were degraded (Figure 3). The relative intensity of heated supernatant has no significant difference with the water control in dsGFP groups for 48 h and dsPat10 groups for 48 h (Figure 1b). The degradation effect of the supernatant group was lower than that of the nematode group (Figure 2 and Figure 3). It is suggested that enzymes in the soaking system of B. xylophilus could degrade dsRNA, and the presence of nematodes accelerates the degradation of dsRNA. On the one hand, some of the dsRNA was ingested by nematodes; on the other hand, the nematode may secrete more enzymes.
2.2. Screening of Extracellular Nuclease Genes
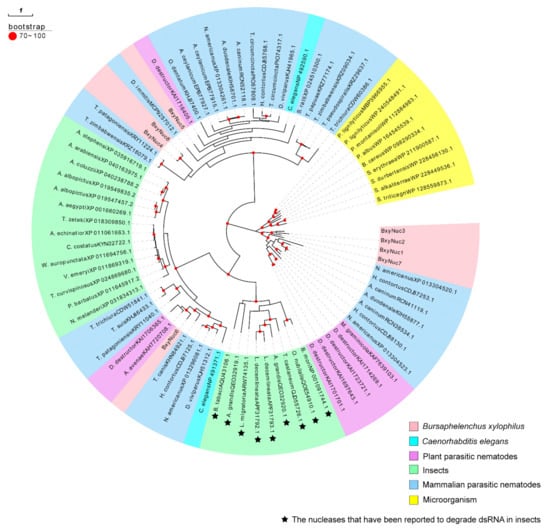
The extracellular nucleases of B. xylophilus may contribute to the degradation of dsRNA in vitro. To confirm whether or which nucleases secreted by nematodes cause dsRNA degradation, we screened the extracellular nuclease genes of B. xylophilus. According to the reported genome database of B. xylophilus, 154 nuclease genes were preliminarily screened (Table S2), and 11 extracellular nuclease genes were screened through signal peptide prediction. Among them, eight genes have been successfully cloned, named BxyNuc1-8 (Accession numbers: OP482151-OP482158). The comparison table between accession number and gene ID of these eight genes is shown in Table S3. The ORF length, amino acid sequence length, signal peptide position, isoelectric point and relative molecular mass of these eight genes were analyzed (Table 1). The molecular weight of the encoded proteins was approximately 30–40 kDa. According to the phylogenetic analysis, BxyNuc1, BxyNuc2, BxyNuc3 and BxyNuc7 are far from the other nucleases in clustering. The BxyNuc6 is in a clade with plant-parasitic nematodes and insects, and it is speculated that it may have similar dsRNA degradation function to the reported nuclease of insects. Furthermore, BxyNuc4, BxyNuc5 and BxyNuc8 are in a clade with mammalian parasitic nematodes (Figure 4).
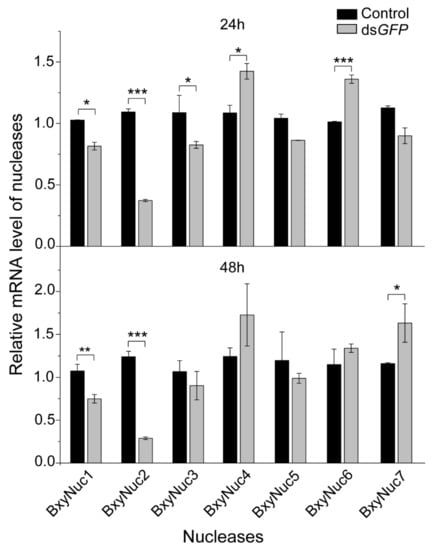
2.3. Extracellular Nuclease Gene Response to dsGFP
In order to screen extracellular nuclease genes in response to dsRNA soaking, the relative mRNA expression levels of the cloned nuclease genes were detected by qRT-PCR after soaking nematodes with non-target dsGFP. The results showed that the expression of nuclease BxyNuc4 and BxyNuc6 can be significantly upregulated by introduction of dsGFP for 24 h. The expression of BxyNuc1, BxyNuc2 and BxyNuc3 were significantly downregulated (Figure 5). After soaking for 48 h, the expression of nuclease BxyNuc7 was significantly upregulated compared with the control (Figure 5). Furthermore, the expression of BxyNuc4 and BxyNuc6 were upregulated. Therefore, the nuclease genes BxyNuc4, BxyNuc6 and BxyNuc7 were used for further experiments and may play a major role in the degradation of dsRNA during the RNAi process of B. xylophilus.
2.4. The Silencing of Extracellular Nuclease Genes Improves RNAi Efficiency
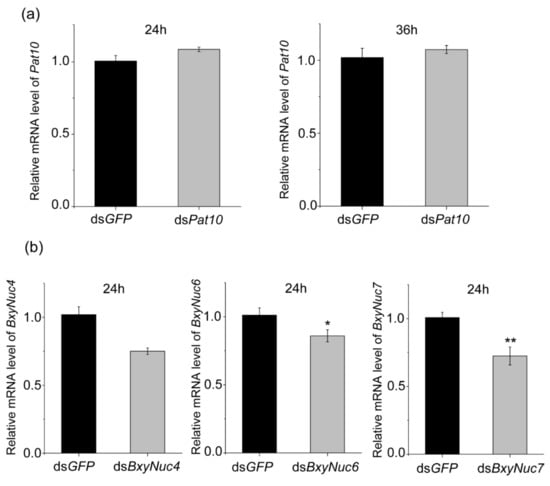
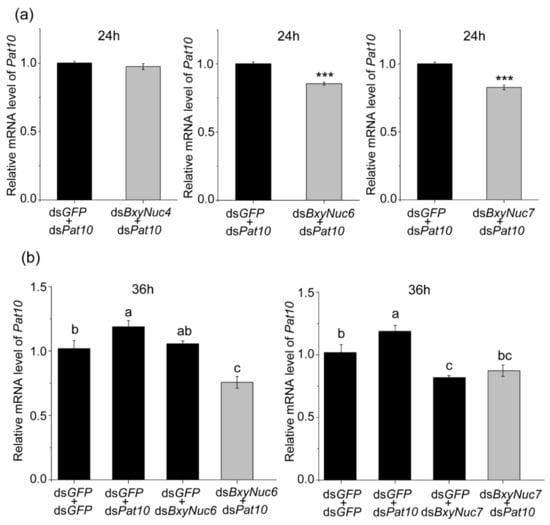
Multiple experiments showed that the expression of Pat10 gene was not downregulated after 24 and 36 h of interference (Figure 6a). However, the nuclease BxyNuc4, BxyNuc6 and BxyNuc7 can be effectively interfered (Figure 6b). In order to further investigate the contributions of nucleases BxyNuc4, BxyNuc6 and BxyNuc7 to the inefficiency of RNAi in B. xylophilus, we mixed the same concentration of nuclease dsRNA and dsPat10 (0.5 µg·µL−1) for interference simultaneously. The results showed that the transcript levels of each nuclease gene were significantly reduced for 24 h (Figure S1). At the same time, the relative mRNA expression level of Pat10 gene was 0.975, 0.827 and 0.855 in dsBxyNuc4+dsPat10, dsBxyNuc6+dsPat10 and dsBxyNuc7+dsPat10 groups after 24 h of interference, respectively (Figure 7a). It is indicated that after co-interference, BxyNuc6 and BxyNuc7 could significantly downregulate the expression of Pat10 gene. Thus, we choose BxyNuc6 and BxyNuc7 to interfere simultaneously with Pat10 for 36 h. The expression of Pat10 gene was 0.641 and 0.741 in dsBxyNuc6+dsPat10 and dsBxyNuc7+dsPat10 groups, respectively (Figure 7b). It was therefore revealed that the RNAi efficiency of Pat10 was indeed improved over time with the participation of nucleases BxyNuc6 or BxyNuc7.
Figure 6.
The relative expression level of Pat10 and nucleases genes. (a) The relative mRNA level of Pat10 after interfering for 24 and 36 h. (b) The relative mRNA level of BxyNuc4/BxyNuc6/BxyNuc7 after interfering for 24 h. Gene expression differences between two groups were calculated using the independent samples t test. Data are presented as mean ± SE, ** p < 0.01; * p < 0.05.
Figure 7.
The relative expression level of Pat10. (a) The relative mRNA level of Pat10 after interfering with Pat10 gene and nucleases BxyNuc4/BxyNuc6/BxyNuc8 genes simultaneously for 24 h. (b) The relative mRNA level of Pat10 after interfering with Pat10 gene and nucleases BxyNuc6/BxyNuc7 genes simultaneously for 36 h. Gene expression differences between two groups were calculated using the independent samples t test. Data are presented as mean ± SE, *** p < 0.001; different letters indicate significant difference (p < 0.05).
Using dsGFP+dsBxyNuc6/Nuc7 as a group to detect the expression of Pat10 for the purpose of excluding the influence of nuclease dsRNA alone on the interference of Pat10 gene. Compared with the control (dsGFP+dsGFP), the expression of Pat10 was 1.189, 1.056 and 0.757 in dsGFP+dsPat10, dsGFP+dsBxyNuc6 and dsBxyNuc6+dsPat10 groups, respectively (Figure 7b). The results showed that dsBxyNuc6 alone (without dsPat10) does not cause downregulation of the Pat10. However, when the nuclease was changed to BxyNuc7, the results showed that the expression of Pat10 was 0.819 and 0.874 in dsGFP+dsBxyNuc7 and dsBxyNuc7+dsPat10 groups, respectively (Figure 7b). It is suggested that dsBxyNuc7 (without dsPat10) could cause the downregulation of Pat10. To sum up, the interference efficiency of Pat10 gene can indeed be improved by interfering with nuclease BxyNuc6 or BxyNuc7. However, only the BxyNuc6 can directly affect RNAi efficiency, and the interference of BxyNuc7 could generate downregulation of Pat10, probably because this interference affected the vital movement of nematodes and led to changes in the expression of Pat10. This provides a feasible method for the low efficiency of lethal gene interference.
3. Discussion
RNAi technology is becoming one of the most promising tools for gene function studies of nematodes. However, RNAi efficiency is relatively low and unstable in plant-parasitic nematodes compared with C. elegans. It is known that the efficiency of gene knockdown by RNAi is influenced by several factors including dsRNA degradation, different delivery method of dsRNA, selection of target genes and the susceptibility of organisms to RNAi [15,23,27,28]. Our study found that dsRNA is unstable in the B. xylophilus soaking system. The agarose gel electrophoresis results suggest that the nuclease secreted by B. xylophilus is probably the major reason for this aspect. Interference with nuclease BxyNuc6 and BxyNuc7 significantly reduced the expression of these two nuclease transcripts and substantially improved RNAi efficiency of Pat10 gene as well (Figure S1 and Figure 7a). This provides an important basis for the follow-up research, but it still cannot rule out the possible degradation function of other extracellular nucleases such as BxyNuc4. A phylogenetic analysis showed high similarity among the BxyNuc6 and the nucleases that have been reported to degrade dsRNA in insects [20,22,29,30,31]. Whether the BxyNuc6 also has the function of degrading dsRNA needs to be further verified with a protein function study.
In insects, numerous studies have revealed that a poor RNAi response is usually associated with high double-stranded RNA (dsRNA)-degrading activity. The oral delivery of dsRNA usually has lower RNAi efficiency compared with microinjection, and the reason for this phenomenon is the exposure of the dsRNA to nucleases secreted in the gut juice and the unsuitable gut pH. Due to the degradation of dsRNA, the uptake of dsRNA by cells is not enough to cause the continuous interference of target genes, which may be the reason for the poor interference effect of this method. Therefore, dsRNA exposure should persist long enough to allow cellular uptake [20]. Furthermore, specifically silencing these nuclease genes can significantly improve the interference efficiency of oral dsRNA. This result was confirmed in multiple insect species, for example, Tribolium castaneum [32], Anthonomus grandis [20], Ostrinia furnacalis [21] and Aedes aegypti [33]. Understanding the interaction between the insects’ nuclease activity and dsRNA is expected to improve the application of RNAi technology in pest control, as well as in plant-parasitic nematodes. However, the role of extracellular nucleases in plant-parasitic nematodes remains uncertain. In C. elegans, only an endonuclease (NM_058970.4) has been reported [34] and seven extracellular nucleases were identified (Figure S2). In this study, a total of 11 extracellular nucleases were identified in B. xylophilus and the function of BxyNuc4, BxyNuc6 and BxyNuc7 may be related to dsRNA degradation in nematodes. It is implied that the B. xylophilus has stronger ability of nucleic acid degradation outside the cell. In a leaf beetle (Plagiodera versicolora), it is proven that the degradation products of dsRNA can be utilized by gut bacteria for growth [35]. However, in B. xylophilus, whether the degraded dsRNA is utilized by nematodes needs to be further explored.
Different extracellular nucleases may have functional complementarity. The downregulation of the target nuclease gene may lead to increased expression of other nucleases. This phenomenon has been demonstrated in the red flour beetle Tribolium castaneum [32]. Furthermore, some off-target effects also occur between different nucleases genes [32,36]. In this study, we found some extracellular nucleases were highly expressed in response to dsGFP, and the others were significantly downregulated. Whether there is functional complementarity between BxyNuc4, BxyNuc6 and BxyNuc7 or other extracellular nucleases screened in this study still needs to be confirmed. At the same time, dsRNA is also likely to be degraded in the gut of nematodes, like most insects. Since the successful implementation of the RNAi process requires the participation of multiple links [9,23,37], whether there are other links that affect the interference efficiency is unknown.
In the process of interference, different target genes showed different interference efficiency. For example, in the root-knot nematode (Meloidogyne incognita), 20 genes involved in the RNAi pathways were investigated, and two of the genes could not be knocked down. Only 10 of the genes were significantly downregulated. The results showed that the genes may respond to RNAi knockdown differently, so an exhaustive assessment of target genes as targets for nematode control via RNAi is imperative [13]. In B. xylophilus, low RNAi efficiency has been described for genes such as Bx-unc-87, Bx-tmy-1 and Bx-hsp-1 [15,38]. In this study, the Pat10 gene showed lower RNAi efficiency than that of the nucleases (Figure 6). This may also be related to the gene itself or the location of gene expression [38].
In recent years, achieving RNAi by feeding nematodes fungi which can express dsRNA is a promising method for nematode control [16,39], but the soaking method still has a considerable role. It can preliminarily screen the functions of some important genes of nematodes with relatively simple operation. Therefore, it is more important to improve the interference efficiency of the soaking method. The co-interference of nucleases and target genes has been proved to be an effective method in this study. In addition, additives that alter the enzymatic activity or pH in the soaking system might be a good solution for enhancing RNAi efficiency [23]. In conclusion, our study found that the in vitro protein secretion of B. xylophilus can degrade dsRNA in the soaking system, affecting the stability of dsRNA before it is taken up by nematode intestinal cells. Among the nucleases that we screened, BxyNuc6 might be a major barrier to the RNAi efficiency in B. xylophilus. The studies of extracellular nuclease illustrate the importance of delivery method of dsRNA and provide a feasible idea for improving RNAi efficiency.
4. Materials and Methods
4.1. Cultivation of Nematodes
Bursaphelenchus xylophilus NXY61 was isolated from diseased Pinus massoniana in Ningbo, Zhejiang Province, China, in May 2015. This nematode has been cultured in the laboratory for generations. Thus, the nematodes we used were a stable genetic laboratory strain. The nematodes were cultured on the mycelia of Botrytis cinerea on potato dextrose agar (PDA) plates at 25 °C for 5 days. The nematodes were washed twice with 0.1× PBST buffer and collected in a 15 mL centrifuge tube via centrifugation at 3000 rpm for 30 s. These nematodes were cleaned with sterile water 3~5 times until the supernatant was clear. In order to prevent the influence of bacteria during the experiment, the nematodes were treated with 0.5% sodium hypochlorite for 30 s and cleaned with sterile 1× PBS buffer twice. Finally, the activity of nematodes was observed under the microscope to ensure the nematodes thrived, and nuclease-free water was added for further experiments.
4.2. Synthesis of dsRNA
The total RNA was extracted from nematodes using TransZol Up Plus RNA Kit (Transgen Biotech, Beijing, China). cDNA was synthesized from 1 µg of total RNA using the PrimeScript RT reagent Kit with gDNA Eraser (TaKaRa, Beijing, China). The DNA template was prepared for dsRNA synthesis by using PCR with primers designed to add T7 promoter sites at both ends. All primer sequences are listed in Table S1. Subsequently, dsRNA was synthesized and purified using the T7 RiboMAXTM Express RNAi System (Promega, Madison, USA) according to the manufacturer’s instructions. Integrity of dsRNAs was evaluated by electrophoresis in 1.2% agarose gels, and the amounts of dsRNA were quantified with a spectrophotometer (Nano-Drop 2000, Thermo Scientific, Waltham, USA).
4.3. Degradation of dsRNA Detected by Agarose Gel Electrophoresis
Approximately 300 nematodes were incubated with 5 µL of dsRNA (final concentration 0.5 µg·µL−1) in a 50 µL volume. Reactions were incubated at 25 °C for 24 and 48 h. The sample was dissolved in an equal volume of nuclease-free water and centrifuged briefly for 30 s. Taking part of the supernatant and add RNA Loading Dye (Takara, Beijing, China), the mixture was heated at 65 °C for 10 min. The dsRNA integrity was analyzed by means of 1.2% agarose gel electrophoresis. Gels were scanned using a fluorescence laser scanner (Azure Biosystems, Dublin, USA).
In addition, approximately 100,000 nematodes were incubated in a shaking incubator at 180 rpm for 48 h at 25 °C. The nematodes were centrifuged at 5000 rpm for 3 min, and the supernatant was sterilized with a 0.22 µm filter. A part of the supernatant was heated at 80 °C for 20 min to inactivate the enzyme. A total of 45 µL of heated and unheated supernatants was incubated with 5 µL of dsRNA (final concentration 0.5 µg·µL−1) for 24 and 48 h separately. As a positive control, the same concentration and volume dsRNA was incubated in nuclease-free water. There were three replicates for each treatment. The integrity of the dsRNA was evaluated using the same method as above. The gray values of the electrophoretic bands were measured with Azure Spot software (Azure Biosystems, Dublin, USA).
4.4. Screening and Cloning of B. xylophilus Extracellular Nuclease Genes
To obtain an initial set of candidate nucleases in B. xylophilus, the amino acid sequences of Caenorhabditis elegans (NP_491371.1, NP_492590.1) were used as query sequences to search the B. xylophilus transcriptome database (PRJEA64437) using the local BLAST program (blast-2.2.30+). At the same time, the nucleases were retrieved according to the annotation of the B. xylophilus genome (PRJEA64437). All screened nucleases were predicted by signal peptide on the SignalP-5.0 Server (http://www.cbs.dtu.dk/services/SignalP/ (accessed on 12 October 2021)) to determine whether they could be secreted out of the cell with the signal peptide. Each retrieved full-length open reading frame (ORF) was confirmed by PCR amplification with the 2× PCR Mix (Tiangen, Beijing, China). The PCR program was as follows: 94 °C for 3 min followed by 37 cycles of 94 °C for 30 s, 55 °C for 30 s and 72 °C for 1 min. The predicted target band was excised and recovered using the QIAEX II Gel Extraction Kit (QIAGEN, Shanghai, China). The samples were subsequently sequenced by the BGI company to verify the nucleotide sequences.
The molecular weight and predicted theoretical isoelectric point (pI) were calculated using the Compute pI/Mw tool (https://web.expasy.org/compute_pi/ (accessed on 12 October 2021)). Using the cloned extracellular nuclease sequences of B. xylophilus as query to screen the orthologous proteins (query cover > 70%) in other species utilizing the blastp tool (https://blast.ncbi.nlm.nih.gov/Blast.cgi (accessed on 20 December 2021)). The insect nuclease sequences were derived from articles reporting that these nucleases have the function of dsRNA degradation. These amino acid sequences were used for the construction of a phylogenetic tree using the maximum likelihood method (1000 bootstrap repeats).
4.5. Interference of Target Gene and Extracellular Nuclease Genes
The Caenorhabditis elegans gene pat10 is an essential component of the body wall muscle [40] and thus is required for nematode movement. The B. xylophilus orthologs of this gene were used in this study. Using the amino acid sequence of C. elegans pat10 gene (wormbase ID F54C1.7) as the query sequence, the protein sequences with the highest percentage similarity were further researched by searching the published B. xylophilus genome in NCBI using tblastn. Sequences with the same alignment results were searched in the dataset of the B. xylophilus genome (PRJEA64437) to obtain the cDNA sequence of Pat10.
Freshly cultured nematodes of B. xylophilus (a mix of adults and juveniles, approximately 10,000 individuals) were immersed in 100 µL solution containing dsRNA (final concentration 1 µg·µL−1) and incubated in a shaking incubator at 180 rpm for 24 h/36 h/48 h at 25 °C. The dsRNA solutions which contained the same final concentrations (0.5 µg·µL−1) of Pat10 dsRNA and nuclease dsRNA were used for simultaneous interference. Equal numbers of nematodes immersed in GFP dsRNA solution were treated as controls. There were three replicates for each treatment. After soaking, the samples of each treatment were thoroughly washed several times in 0.1× PBST sterile water and then used for further experiments.
4.6. Quantitative Reverse Transcription PCR (qRT-PCR)
Prior to use in qRT-PCR, cDNA was 1:4 diluted with ddH2O. All primers used were designed with Primer Premier 6. The qPCR reaction was set up in 20 μL containing 1 μL of cDNA, 10 μL of TB Green Premix DimerEraser (Perfect Real Time), 1 μL of each primer and ddH2O. cDNA templates were denatured at 95 °C for 30 s, followed by 40 three-segment cycles of amplification at 95 (5 s), 55 (30 s) and 72 °C (30 s). A melting curve analysis was performed after the qPCR run (15 s at 65 °C). The actin gene of B. xylophilus was used as the internal control. The experiment had three biological replicates and four technical replicates. According to the cycle threshold (Ct) value and the dissolution curve, the 2−ΔΔCt method was used to estimate the relative expression level of the target gene and verify the interference efficiency.
4.7. Statistical Analysis
Statistical significance of differences in the mRNA expression levels among different treatment groups was determined by one-way analysis of variance in PASW Statistics 18.0 software (at p < 0.05).
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms232012278/s1.
Author Contributions
Conceptualization, X.Z., Y.L. and D.L.; methodology, Y.L., D.L. and W.Z.; software, X.W. (Xuan Wang), D.L., W.Z. and Z.L.; validation, R.W.; resources, X.Z. and Y.L.; data curation, X.W. (Xiaojian Wen); writing—original draft preparation, R.W.; writing—review and editing, Y.L., D.L. and W.Z.; supervision, Y.F. All authors have read and agreed to the published version of the manuscript.
Funding
National Key R & D Program of China (2021YFD1400900). National Natural Science Foundation of China (3190140949).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The nucleases BxyNuc1-8 can be found using accession numbers OP482151-OP482158 from NCBI.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jones, J.T.; Haegeman, A.; Danchin, E.G.J.; Gaur, H.S.; Helder, J.; Jones, M.G.K.; Kikuchi, T.; Manzanilla-Lopez, R.; Palomares-Rius, J.E.; Wesemael, W.M.; et al. Top 10 plant-parasitic nematodes in molecular plant pathology. Mol. Plant Pathol. 2013, 14, 946–961. [Google Scholar] [CrossRef] [PubMed]
- Inácio, M.L.; Nóbrega, F.; Vieira, P.; Bonifácio, L.; Naves, P.; Sousa, E.; Mota, M. First detection of Bursaphelenchus xylophilus associated with Pinus nigrain in Portugal and in Europe. For. Pathol. 2014, 45, 235–238. [Google Scholar] [CrossRef]
- Wang, H.H.; Wang, Y.B.; Yin, C.; Gao, J.; Tao, R.; Sun, Y.L.; Wang, C.Y.; Wang, Z.; Li, Y.X.; Sung, C.K. In vivo infection of Bursaphelenchus xylophilus by the fungus Esteya vermicola. Pest Manag. Sci. 2020, 76, 2854–2864. [Google Scholar] [CrossRef] [PubMed]
- Kanzaki, N.; Ekino, T.; Maehara, N.; Aikawa, T.; Giblin-Davis, R.M. Bursaphelenchus acaloleptae n. sp. sharing tree and beetle carrier hosts with B. luxuriosae Kanzaki & Futai, 2003 in Japan. Nematology 2019, 22, 515–527. [Google Scholar] [CrossRef]
- Zhao, J.; Huang, J.; Yan, J.; Fang, G. Economic Loss of Pine Wood Nematode Disease in Mainland China from 1998 to 2017. Forests 2020, 11, 1042. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, W.; Ding, P.; Wu, J.; Li, Y. The research progress of pine wilt disease control. Xiang Cun Ke Ji 2022, 13, 116–118. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, X.; Li, Y.; Liu, Z.; Li, D.; Wen, X.; Feng, Y.; Zhang, X. Pinewood Nematode Alters the Endophytic and Rhizospheric Microbial Communities of Pinus massoniana. Microb. Ecol. 2020, 81, 807–817. [Google Scholar] [CrossRef]
- Kim, N.; Jeon, H.W.; Mannaa, M.; Jeong, S.-I.; Kim, J.; Kim, J.; Lee, C.; Park, A.R.; Kim, J.-C.; Seo, Y.-S. Induction of resistance against pine wilt disease caused by Bursaphelenchus xylophilus using selected pine endophytic bacteria. Plant Pathol. 2019, 68, 434–444. [Google Scholar] [CrossRef]
- Rosso, M.N.; Jones, J.T.; Abad, P. RNAi and functional genomics in plant parasitic nematodes. Annu. Rev. Phytopathol. 2009, 47, 207–232. [Google Scholar] [CrossRef]
- Fire, A.; Xu, S.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E.; Mello, C.C. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef]
- Liu, S.; Jaouannet, M.; Dempsey, D.A.; Imani, J.; Coustau, C.; Kogel, K.H. RNA-based technologies for insect control in plant production. Biotechnol. Adv. 2019, 39, 107463. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.C.H.; Jones, M.G.K.; Fosu-Nyarko, J. Gene silencing in root lesion nematodes (Pratylenchus spp.) significantly reduces reproduction in a plant host. Exp. Parasitol. 2013, 133, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, S.; Fosu-Nyarko, J.; Jones, M.G.K. Attempt to Silence Genes of the RNAi Pathways of the Root-Knot Nematode, Meloidogyne incognita Results in Diverse Responses Including Increase and No Change in Expression of Some Genes. Front. Plant Sci. 2020, 11, 328. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Banerjee, A.; Gill, S.S.; Gupta, O.P.; Dahuja, A.; Jain, P.K.; Sirohi, A. RNA Interference: A Novel Source of Resistance to Combat Plant Parasitic Nematodes. Front. Plant Sci. 2017, 8, 834. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Lee, K.Y.; Lee, S.; Oh, W.; Jeong, P.; Woo, T.; Kim, C.; Paik, Y.; Koo, H. The efficiency of RNA interference in Bursaphelenchus xylophilus. Mol. Cells 2008, 26, 81–86. [Google Scholar]
- Wang, M.; Wang, D.; Zhang, X.; Wang, X.; Liu, W.; Hou, X.; Huang, X.; Xie, B.; Cheng, X. Double-stranded RNA-mediated interference of dumpy genes in Bursaphelenchus xylophilus by feeding on filamentous fungal transformants. Int. J. Parasitol. 2016, 46, 351–360. [Google Scholar] [CrossRef]
- Xue, Q.; Wu, X.Q.; Zhang, W.J.; Deng, L.N.; Wu, M.M. Cathepsin L-like Cysteine Proteinase Genes Are Associated with the Development and Pathogenicity of Pine Wood Nematode, Bursaphelenchus xylophilus. Int. J. Mol. Sci. 2019, 20, 215. [Google Scholar] [CrossRef]
- Liu, X.; Zhou, X.; Zhou, L.; Hu, J.; Guo, K. Application of RNA Interference in the Pinewood Nematode, Bursaphelenchus xylophilus. J. Vis. Exp. 2022, 181, e63645. [Google Scholar] [CrossRef]
- Wang, X.; Cheng, X.; Li, Y.; Zhang, J.; Zhang, Z.; Wu, H. Cloning arginine kinase gene and its RNAi in Bursaphelenchus xylophilus causing pine wilt disease. Eur. J. Plant. Pathol. 2012, 134, 521–532. [Google Scholar] [CrossRef]
- Garcia, R.A.; Macedo, L.L.P.; Nascimento, D.C.; Gillet, F.X.; Moreira-Pinto, C.E.; Faheem, M.; Basso, A.M.M.; Silva, M.C.M.; Grossi-de-Sa, M.F. Nucleases as a barrier to gene silencing in the cotton boll weevil, Anthonomus grandis. PLoS ONE 2017, 12, e0189600. [Google Scholar] [CrossRef]
- Fan, Y.H.; Song, H.F.; Abbas, M.; Wang, Y.L.; Li, T.; Ma, E.B.; Cooper, A.M.W.; Silver, K.; Zhu, K.Y.; Zhang, J.Z. A dsRNA-degrading nuclease (dsRNase2) limits RNAi efficiency in the Asian corn borer (Ostrinia furnacalis). Insect Sci. 2021, 28, 1677–1689. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.S.; Ahn, S.J.; Flinn, C.M.; Choi, M.Y. Identification and functional analysis of dsRNases in spotted-wing drosophila, Drosophila suzukii. Arch. Insect Biochem. Physiol. 2021, 107, e21822. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.M.; Silver, K.; Zhang, J.; Park, Y.; Zhu, K.Y. Molecular mechanisms influencing efficiency of RNA interference in insects. Pest Manag. Sci. 2019, 75, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Wang, K.; Fu, W.; Sheng, C.; Han, Z. Biochemical Comparison of dsRNA Degrading Nucleases in Four Different Insects. Front. Physiol. 2018, 9, 624. [Google Scholar] [CrossRef] [PubMed]
- Vieira, P.; Shao, J.; Vijayapalani, P.; Maier, T.R.; Pellegrin, C.; Akker, S.E.; Baum, T.J.; Nemchinov, L.G. A new esophageal gland transcriptome reveals signatures of large scale de novo effector birth in the root lesion nematode Pratylenchus penetrans. BMC Genom. 2020, 21, 738. [Google Scholar] [CrossRef]
- Shinya, R.; Morisaka, H.; Kikuchi, T.; Takeuchi, Y.; Ueda, M.; Futai, K. Secretome Analysis of the Pine Wood Nematode Bursaphelenchus xylophilus Reveals the Tangled Roots of Parasitism and Its Potential for Molecular Mimicry. PLoS ONE 2013, 8, e67377. [Google Scholar] [CrossRef]
- Maule, A.G.; McVeigh, P.; Dalzell, J.J.; Atkinson, L.; Mousley, A.; Marks, N.J. An eye on RNAi in nematode parasites. Trends Parasitol. 2011, 27, 505–513. [Google Scholar] [CrossRef]
- Britton, C.; Samarasinghe, B.; Knox, D.P. Ups and downs of RNA interference in parasitic nematodes. Exp. Parasitol. 2011, 132, 56–61. [Google Scholar] [CrossRef]
- Cooper, A.M.W.; Song, H.; Shi, X.; Yu, Z.; Lorenzen, M.; Silver, K.; Zhang, J.; Zhu, K.Y. Molecular Characterizations of Double-Stranded RNA Degrading Nuclease Genes from Ostrinia nubilalis. Insects 2020, 11, E652. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, Q.; Luan, J.; Chung, S.H.; Eck, J.V.; Turgeon, R.; Douglas, A.E. Towards an understanding of the molecular basis of effective RNAi against a global insect pest, the whitefly Bemisia tabaci. Insect Biochem. Mol. Biol. 2017, 88, 21–29. [Google Scholar] [CrossRef]
- Spit, J.; Philips, A.; Wynant, N.; Santos, D.; Plaetinck, G.; Broeck, J.V. Knockdown of nuclease activity in the gut enhances RNAi efficiency in the Colorado potato beetle, Leptinotarsa decemlineata, but not in the desert locust, Schistocerca gregaria. Insect Biochem. Mol. Biol. 2017, 81, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Wang, K.; Chen, J.; Wang, J.; Zhang, H.; Ze, L.; Zhu, G.; Zhao, C.; Xiao, H.; Han, Z. Identification of a double-stranded RNA-degrading nuclease influencing both ingestion and injection RNA interference efficiency in the red flour beetle Tribolium castaneum. Insect Biochem. Mol. Biol. 2020, 125, 103440. [Google Scholar] [CrossRef] [PubMed]
- Giesbrecht, D.; Heschuk, D.; Wiens, I.; Boguski, D.; LaChance, P.; Whyard, S. RNA Interference Is Enhanced by Knockdown of double-stranded RNases in the Yellow Fever Mosquito Aedes aegypti. Insects 2020, 11, E327. [Google Scholar] [CrossRef] [PubMed]
- The C. Elegans Sequencing Consortium. Genome Sequence of the Nematode C. elegan: A Platform for Investigating Biology. Science 1998, 282, 2012–2018. [Google Scholar] [CrossRef]
- Xu, L.; Xu, S.; Sun, L.; Zhang, Y.; Luo, J.; Bock, R.; Zhang, J. Synergistic action of the gut microbiota in environmental RNA interference in a leaf beetle. Microbiome 2021, 9, 98. [Google Scholar] [CrossRef]
- Wynant, N.; Santos, D.; Verdonck, R.; Spit, J.; Wielendaele, P.V.; Broeck, J.V. Identification, functional characterization and phylogenetic analysis of double stranded RNA degrading enzymes present in the gut of the desert locust, Schistocerca gregaria. Insect Biochem. Mol. Biol. 2014, 46, 1–8. [Google Scholar] [CrossRef]
- Véleza, A.M.; Fishilevich, E. The mysteries of insect RNAi: A focus on dsRNA uptake and transport. Pestic. Biochem. Phys. 2018, 151, 25–31. [Google Scholar] [CrossRef]
- Cardoso, J.M.S.; Fonseca, L.; Gomes, P.; Egas, C.; Abrantes, I. Molecular characterization and functional analysis of a calponin gene from the pinewood nematode. For. Pathol. 2015, 45, 467–473. [Google Scholar] [CrossRef]
- Wang, M.; Song, S.; Wang, X.; Tian, Y.; Zhang, Z.; Hu, S.; Cheng, X. Double Stranded RNA Expression of Bx-apa-2 and Bx-apm-2 Genes in Fusarium oxysporum Mediated RNAi for Bursaphelenchus xylophilus. J. Southwest For. Univ. 2022, 42, 72–79. [Google Scholar] [CrossRef]
- Terami, H.; Williams, B.D.; Kitamura, S.; Sakube, Y.; Matsumoto, S.; Doi, S.; Obinata, T.; Kagawa, H. Genomic Organization, Expression, and Analysis of the Troponin C Gene pat-10 of Caenorhabditis elegans. J. Cell Biol. 1999, 146, 193–202. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).