A New Target of Dental Pulp-Derived Stem Cell-Based Therapy on Recipient Bone Marrow Niche in Systemic Lupus Erythematosus
Abstract
:1. Introduction
2. Characterization of hDPSCs
3. Intercellular Interaction between hDPSCs and T Lymphocytes
4. Therapeutic Effects of hDPSCs in the SLE MRL/lpr Mouse Model
5. Telomerase Activity-Associated Niche Formation and Immunosuppressive Functions of Bone Marrow Mesenchymal Stem Cells (BMMSCs)
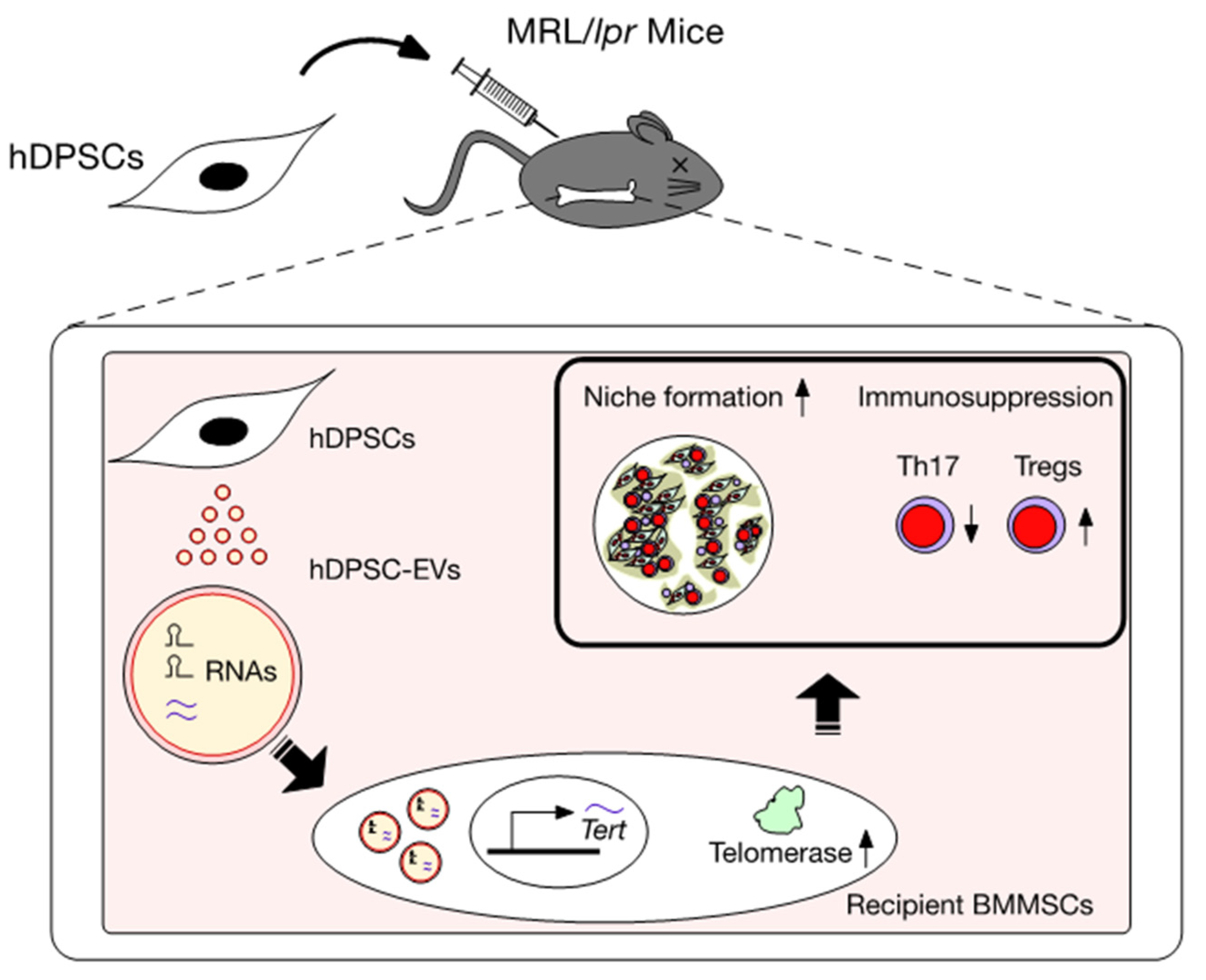
6. Action of EVs Released from hDPSCs (hDPSC-EVs)
7. Effects of Systemically Administrated hDPSC-Evs on the Telomerase Activity-Associated Niche Formation of Recipient lpr-BMMSCs in MRL/lpr Mice
8. Effects of Systemically Administered hDPSC-EVs on the Immunomodulatory Functions of Recipient lpr-BMMSCs
9. Mechanism of hDPSC Transplantation via Recovering Telomerase Activity of hDPSC-EVs in SLE
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cheung, T.S.; Bertolino, G.M.; Giacomini, C.; Bornhäuser, M.; Dazzi, F.; Galleu, A. Mesenchymal stromal cells for graft versus host disease: Mechanism-based biomarkers. Front. Immunol. 2020, 11, 1338. [Google Scholar] [CrossRef] [PubMed]
- Murata, M.; Teshima, T. Treatment of Steroid-Refractory Acute Graft-Versus-Host Disease Using Commercial Mesenchymal Stem Cell Products. Front. Immunol. 2021, 12, 724380. [Google Scholar] [CrossRef] [PubMed]
- Wobma, H.; Satwani, P. Mesenchymal stromal cells: Getting ready for clinical primetime. Transfus. Apher. Sci. 2021, 60, 103058. [Google Scholar] [CrossRef] [PubMed]
- Friedenstein, A.J.; Deriglasova, U.F.; Kulagina, N.N.; Panasuk, A.F.; Rudakowa, S.F.; Luriá, E.A.; Ruadkow, I.A. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp. Hematol. 1974, 2, 83–92. [Google Scholar]
- Pittenger, M.F.; Discher, D.E.; Péault, B.M.; Phinney, D.G.; Hare, J.M.; Caplan, A.I. Mesenchymal stem cell perspective: Cell biology to clinical progress. NPJ Regen. Med. 2019, 4, 22. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.; Sun, J.; Tian, Y.; Li, H.; Zhang, L.; Yang, J.; Wang, J.; Zhang, J.; Yan, S.; Xu, D. Immunomodulatory effect of MSCs and MSCs-derived extracellular vesicles in systemic lupus erythematosus. Front. Immunol. 2021, 12, 714832. [Google Scholar] [CrossRef]
- Viswanathan, S.; Shi, Y.; Galipeau, J.; Krampera, M.; Leblanc, K.; Martin, I.; Nolta, J.; Phinney, D.G.; Sensebe, L. Mesenchymal stem versus stromal cells: International Society for Cell & Gene Therapy (ISCT®) Mesenchymal Stromal Cell committee position statement on nomenclature. Cytotherapy 2019, 21, 1019–1024. [Google Scholar]
- Sonoda, S.; Mei, Y.F.; Atsuta, I.; Danjo, A.; Yamaza, H.; Hama, S.; Nishida, K.; Tang, R.; Kyumoto-Nakamura, Y.; Uehara, N.; et al. Exogenous nitric oxide stimulates the odontogenic differentiation of rat dental pulp stem cells. Sci. Rep. 2018, 8, 3419. [Google Scholar] [CrossRef] [Green Version]
- Shi, X.; Mao, J.; Liu, Y. Pulp stem cells derived from human permanent and deciduous teeth: Biological characteristics and therapeutic applications. Stem Cells Transl. Med. 2020, 9, 445–464. [Google Scholar] [CrossRef] [Green Version]
- Sui, B.; Wu, D.; Xiang, L.; Fu, Y.; Kou, X.; Shi, S. Dental pulp stem cells: From discovery to clinical application. J. Endod. 2020, 46, S46–S55. [Google Scholar] [CrossRef]
- Gronthos, S.; Mankani, M.; Brahim, J.; Robey, P.G.; Shi, S. Postnatal human dental pulp stem cells (DPSCs) In Vitro and In Vivo. Proc. Natl. Acad. Sci. USA 2000, 97, 13625–13630. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miura, M.; Gronthos, S.; Zhao, M.; Lu, B.; Fisher, L.W.; Robey, P.G.; Shi, S. SHED: Stem cells from human exfoliated deciduous teeth. Proc. Natl. Acad. Sci. USA 2003, 100, 5807–5812. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonoyama, W.; Liu, Y.; Fang, D.; Yamaza, T.; Seo, B.M.; Zhang, C.; Liu, H.; Gronthos, S.; Wang, C.Y.; Wang, S.; et al. Mesenchymal stem cell-mediated functional tooth regeneration in swine. PLoS ONE 2006, 1, e79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Makino, Y.; Yamaza, H.; Akiyama, K.; Ma, L.; Hoshino, Y.; Nonaka, K.; Terada, Y.; Kukita, T.; Shi, S.; Yamaza, T. Immune therapeutic potential of stem cells from human supernumerary teeth. J. Dent. Res. 2013, 92, 609–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yamaza, T.; Kentaro, A.; Chen, C.; Liu, Y.; Shi, Y.; Gronthos, S.; Wang, S.; Shi, S. Immunomodulatory properties of stem cells from human exfoliated deciduous teeth. Stem Cell Res. Ther. 2010, 1, 5. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, Y.; Sonoda, S.; Yamaza, H.; Murata, S.; Nishida, K.; Hama, S.; Kyumoto-Nakamura, Y.; Uehara, N.; Nonaka, K.; Kukita, T.; et al. Suppression of AKT-mTOR signal pathway enhances osteogenic/dentinogenic capacity of stem cells from apical papilla. Stem. Cell Res. Ther. 2018, 9, 334. [Google Scholar] [CrossRef]
- Seo, B.M.; Sonoyama, W.; Yamaza, T.; Coppe, C.; Kikuiri, T.; Akiyama, K.; Lee, J.S.; Shi, S. SHED repair critical-size calvarial defects in mice. Oral Dis. 2008, 14, 428–434. [Google Scholar] [CrossRef]
- Huang, G.T.; Yamaza, T.; Shea, L.D.; Djouad, F.; Kuhn, N.Z.; Tuan, R.S.; Shi, S. Stem/progenitor cell-mediated de novo regeneration of dental pulp with newly deposited continuous layer of dentin in an in vivo model. Tissue Eng. Part A 2010, 16, 605–615. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Makino, Y.; Yamaza, H.; Akiyama, K.; Hoshino, Y.; Song, G.; Kukita, T.; Nonaka, K.; Shi, S.; Yamaza, T. Cryopreserved dental pulp tissues of exfoliated deciduous teeth is a feasible stem cell resource for regenerative medicine. PLoS ONE 2012, 7, e51777. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Aijima, R.; Hoshino, Y.; Yamaza, H.; Tomoda, E.; Tanaka, Y.; Sonoda, S.; Song, G.; Zhao, W.; Nonaka, K.; et al. Transplantation of mesenchymal stem cells ameliorates secondary osteoporosis through interleukin-17-impaired functions of recipient bone marrow mesenchymal stem cells in MRL/lpr mice. Stem Cell Res. Ther. 2015, 6, 104. [Google Scholar] [CrossRef] [Green Version]
- Yamaza, T.; Alatas, F.S.; Yuniartha, R.; Yamaza, H.; Fujiyoshi, J.K.; Yanagi, Y.; Yoshimaru, K.; Hayashida, M.; Matsuura, T.; Aijima, R.; et al. In Vivo hepatogenic capacity and therapeutic potential of stem cells from human exfoliated deciduous teeth in mice with liver fibrosis. Stem Cell Res. Ther. 2015, 6, 171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fujiyoshi, J.K.; Yamaza, H.; Sonoda, S.; Yuniartha, R.; Ihara, K.; Nonaka, K.; Taguchi, T.; Ohga, S.; Yamaza, T. Therapeutic potential of hepatocyte-like-cells converted from stem cells from human exfoliated deciduous teeth in fulminant Wilson’s disease. Sci. Rep. 2019, 9, 1535. [Google Scholar] [CrossRef] [PubMed]
- Yuniartha, R.; Yamaza, T.; Sonoda, S.; Yoshimaru, K.; Matsuura, T.; Yamaza, H.; Oda, Y.; Ohga, S.; Taguchi, T. Cholangiogenic potential of human deciduous pulp stem cell-converted hepatocyte-like cells. Stem Cell Res. Ther. 2021, 12, 57. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, Y.; Sonoda, S.; Yamaza, H.; Murata, S.; Nishida, K.; Kyumoto-Nakamura, Y.; Uehara, N.; Nonaka, K.; Kukita, T.; Yamaza, T. Acetylsalicylic acid treatment and suppressive regulation of AKT accelerate odontogenic differentiation of stem cells from the apical papilla. J. Endod. 2019, 45, 591–598.e6. [Google Scholar] [CrossRef]
- Takahashi, Y.; Yuniartha, R.; Yamaza, T.; Sonoda, S.; Yamaza, H.; Kirino, K.; Yoshimaru, K.; Matsuura, T.; Taguchi, T. Therapeutic potential of spheroids of stem cells from human exfoliated deciduous teeth for chronic liver fibrosis and hemophilia A. Pediatr. Surg. Int. 2019, 35, 1379–1388. [Google Scholar] [CrossRef]
- Alongi, D.J.; Yamaza, T.; Song, Y.; Fouad, A.F.; Romberg, E.E.; Shi, S.; Tuan, R.S.; Huang, G.T. Stem/progenitor cells from inflamed human dental pulp retain tissue regeneration potential. Regen. Med. 2010, 5, 617–631. [Google Scholar] [CrossRef] [Green Version]
- Sonoda, S.; Yamaza, H.; Ma, L.; Tanaka, Y.; Tomoda, E.; Aijima, R.; Nonaka, K.; Kukita, T.; Shi, S.; Nishimura, F.; et al. Interferon-gamma improves impaired dentinogenic and immunosuppressive functions of irreversible pulpitis-derived human dental pulp stem cells. Sci. Rep. 2016, 6, 19286. [Google Scholar] [CrossRef]
- Yamaza, H.; Tomoda, E.; Sonoda, S.; Nonaka, K.; Yamaza, T. Bilirubin reversibly affects cell death and odontogenic capacity in stem cells from human exfoliated deciduous teeth. Oral Dis. 2018, 24, 809–819. [Google Scholar] [CrossRef]
- Yamaza, H.; Sonoda, S.; Nonaka, K.; Yamaza, T. Pamidronate decreases bilirubin-impaired cell death and improves dentinogenic dysfunction of stem cells from human deciduous teeth. Stem Cell Res. Ther. 2018, 9, 303. [Google Scholar] [CrossRef]
- Sonoda, S.; Yoshimaru, K.; Yamaza, H.; Yuniartha, R.; Matsuura, T.; Yamauchi-Tomoda, E.; Murata, S.; Nishida, K.; Oda, Y.; Ohga, S.; et al. Biliary atresia-specific deciduous pulp stem cells feature biliary deficiency. Stem Cell Res. Ther. 2021, 12, 582. [Google Scholar] [CrossRef]
- Taguchi, T.; Yanagi, Y.; Yoshimaru, K.; Zhang, X.Y.; Matsuura, T.; Nakayama, K.; Kobayashi, E.; Yamaza, H.; Nonaka, K.; Ohga, S.; et al. Regenerative medicine using stem cells from human exfoliated deciduous teeth (SHED): A promising new treatment in pediatric surgery. Surg. Today 2019, 49, 316–322. [Google Scholar] [CrossRef]
- Xuan, K.; Li, B.; Guo, H.; Sun, W.; Kou, X.; He, X.; Zhang, Y.; Sun, J.; Liu, A.; Liao, L.; et al. Deciduous autologous tooth stem cells regenerate dental pulp after implantation into injured teeth. Sci. Transl. Med. 2018, 10, eaaf3227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Iwanaka, T.; Yamaza, T.; Sonoda, S.; Yoshimaru, K.; Matsuura, T.; Yamaza, H.; Ohga, S.; Oda, Y.; Taguchi, T. A model study for the manufacture and validation of clinical-grade deciduous dental pulp stem cells for chronic liver fibrosis treatment. Stem Cell Res. Ther. 2020, 11, 134. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Huang, Z.; Wu, D.; Kou, X.; Mao, X.; Shi, S. CD146 controls the quality of clinical grade mesenchymal stem cells from human dental pulp. Stem Cell Res. Ther. 2021, 12, 488. [Google Scholar] [CrossRef] [PubMed]
- Sonoda, S.; Murata, S.; Kato, H.; Zakaria, F.; Kyumoto-Nakamura, Y.; Uehara, N.; Yamaza, H.; Kukita, T.; Yamaza, T. Targeting of deciduous tooth pulp stem cell-derived extracellular vesicles on telomerase-mediated stem cell niche and immune regulation in systemic lupus erythematosus. J. Immunol. 2021, 206, 3053–3063. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, L.; Liu, S.; Liu, D.; Chen, C.; Xu, X.; Chen, X.; Shi, S. Transplantation of SHED prevents bone loss in the early phase of ovariectomy induced osteoporosis. J. Dent. Res. 2014, 93, 1124–1132. [Google Scholar] [CrossRef]
- Laing, A.G.; Riffo-Vasquez, Y.; Sharif-Paghaleh, E.; Lombardi, G.; Sharpe, P.T. Immune modulation by apoptotic dental pulp stem cells In Vivo. Immunotherapy 2018, 10, 201–211. [Google Scholar] [CrossRef]
- Paganelli, A.; Trubiani, O.; Diomede, F.; Pisciotta, A.; Paganelli, R. Immunomodulating profile of dental mesenchymal stromal cells: A comprehensive overview. Front. Oral Health 2021, 2, 635055. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, L.; Jin, Y.; Shi, S. Fas ligand regulates the immunomodulatory properties of dental pulp stem cells. J. Dent. Res. 2012, 91, 948–954. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Chen, C.; Liu, S.; Liu, D.; Xu, X.; Chen, X.; Shi, S. Acetylsalicylic acid treatment improves differentiation and immunomodulation of SHED. J. Dent. Res. 2015, 94, 209–218. [Google Scholar] [CrossRef] [Green Version]
- Di Tinco, R.; Bertani, G.; Pisciotta, A.; Bertoni, L.; Pignatti, E.; Maccaferri, M.; Bertacchini, J.; Sena, P.; Vallarola, A.; Tupler, R.; et al. Role of PD-L1 in licensing immunoregulatory function of dental pulpmesenchymal stem cells. Stem Cell Res. Ther. 2021, 4, 598. [Google Scholar]
- De la Rosa-Ruiz, M.D.P.; Álvarez-Pérez, M.A.; Cortés-Morales, V.A.; Monroy-García, A.; Mayani, H.; Fragoso-González, G.; Caballero-Chacón, S.; Diaz, D.; Candanedo-González, F.; Montesinos, J.J. Mesenchymal stem/stromal cells derived from dental tissues: A comparative in vitro evaluation of their immunoregulatory properties against T cells. Cells 2019, 8, 1491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirata, M.; Ishigami, M.; Matsushita, Y.; Ito, T.; Hattori, H.; Hibi, H.; Goto, H.; Ueda, M.; Yamamoto, A. Multifaceted therapeutic benefits of factors derived from dental pulp stem cells for mouse liver fibrosis. Stem Cells Transl. Med. 2016, 5, 1416–1424. [Google Scholar] [CrossRef] [Green Version]
- Martinez, V.G.; Ontoria-Oviedo, I.; Ricardo, C.P.; Harding, S.E.; Sacedon, R.; Varas, A.; Zapata, A.; Sepulveda, P.; Vicente, A. Overexpression of hypoxia-inducible factor 1 alpha improves immunomodulation by dentalmesenchymal stem cells. Stem Cell Res. Ther. 2017, 8, 208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Özdemir, A.T.; Özgül Özdemir, R.B.; Kırmaz, C.; Sarıboyacı, A.E.; Ünal Halbutoğlları, Z.S.; Özel, C.; Karaöz, E. The paracrine immunomodulatory interactions between the human dental pulp derived mesenchymal stem cells and CD4 T cell subsets. Cell Immunol. 2016, 310, 108–115. [Google Scholar] [CrossRef]
- Demircan, P.C.; Sariboyaci, A.E.; Unal, Z.S.; Gacar, G.; Subasi, C.; Karaoz, E. Immunoregulatory effects of human dental pulp-derived stem cells on T cells: Comparison of transwell co-culture and mixed lymphocyte reaction systems. Cytotherapy 2011, 13, 1205–1220. [Google Scholar] [CrossRef]
- Pierdomenico, L.; Bonsi, L.; Calvitti, M.; Rondelli, D.; Arpinati, M.; Chirumbolo, G.; Becchetti, E.; Marchionni, C.; Alviano, F.; Fossati, V.; et al. Multipotent mesenchymal stem cells with immunosuppressive activity can be easily isolated from dental pulp. Transplantation 2005, 80, 836–842. [Google Scholar] [CrossRef] [Green Version]
- Silva Fde, S.; Ramos, R.N.; de Almeida, D.C.; Bassi, E.J.; Gonzales, R.P.; Miyagi, S.P.; Maranduba, C.P.; Sant’Anna, O.A.; Marques, M.M.; Barbuto, J.A.; et al. Mesenchymal stem cells derived from human exfoliated deciduous teeth (SHEDs) induce immune modulatory profile in monocyte-derived dendritic cells. PLoS ONE 2014, 9, e98050. [Google Scholar] [CrossRef]
- Matsubara, K.; Matsushita, Y.; Sakai, K.; Kano, F.; Kondo, M.; Noda, M.; Hashimoto, N.; Imagama, S.; Ishiguro, N.; Suzumura, A.; et al. Secreted ectodomain of sialic acid-binding Ig-like lectin-9 and monocytechemoattractant protein-1 promote recovery after rat spinal cord injury by altering macrophage polarity. J. Neurosci. 2015, 35, 2452–2464. [Google Scholar] [CrossRef] [Green Version]
- Kano, F.; Matsubara, K.; Ueda, M.; Hibi, H.; Yamamoto, A. Secreted ectodomain of sialic acid-binding Ig-like lectin-9 and monocyte chemoattractant protein-1 synergistically regenerate transected rat peripheral nerves by altering macrophage polarity. Stem Cells 2017, 35, 641–653. [Google Scholar] [CrossRef]
- Gnanasegaran, N.; Govindasamy, V.; Mani, V.; Abu Kasim, N.H. Neuroimmunomodulatory properties of DPSCs in an in vitro model of Parkinson’s disease. IUBMB Life 2017, 69, 689–699. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomic, S.; Djokic, J.; Vasilijic, S.; Vucevic, D.; Todorovic, V.; Supic, G.; Colic, M. Immunomodulatory properties of mesenchymal stem cells derived from dental pulp and dental follicle are susceptible to activation by toll-like receptor agonists. Stem Cells Dev. 2011, 20, 695–708. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Kuang, S.; Zhang, Y.; Yang, M.; Qin, W.; Shi, X.; Lin, Z. Chitosan hydrogel incorporated with dental pulp stem cell-derived exosomes alleviates periodontitis in mice via a macrophage-dependent mechanism. Bioact. Mater. 2020, 5, 1113–1126. [Google Scholar] [CrossRef]
- Li, Y.; Yang, Y.-Y.; Ren, J.-L.; Xu, F.; Chen, F.-M.; Li, A. Exosomes secreted by stem cells from human exfoliated deciduous teeth contribute to functional recovery after traumatic brain injury by shifting microglia M1/M2 polarization in rats. Stem Cell Res. Ther. 2017, 8, 198. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pivoraitė, U.; Jarmalavičiūtė, A.; Tunaitis, V.; Ramanauskaitė, G.; Vaitkuvienė, A.; Kašėta, V.; Biziulevičienė, G.; Venalis, A.; Pivoriūnas, A. Exosomes from human dental pulp stem cells suppress carrageenan-induced acute inflammation in mice. Inflammation 2015, 38, 1933–1941. [Google Scholar] [CrossRef]
- Koga, T.; Ichinose, K.; Kawakami, A.; Tsokos, G.C. Current insights and future prospects for targeting IL-17 to treat patients with systemic lupus erythematosus. Front. Immunol. 2021, 11, 624971. [Google Scholar] [CrossRef]
- Li, W.; Deng, C.; Yang, H.; Wang, G. The regulatory T cell in active systemic lupus erythematosus patients: A systemic review and meta-analysis. Front. Immunol. 2019, 10, 159. [Google Scholar] [CrossRef] [Green Version]
- Shan, J.; Jin, H.; Xu, Y. T cell metabolism: A new perspective on Th17/Treg cell imbalance in systemic lupus erythematosus. Front. Immunol. 2020, 11, 1027. [Google Scholar] [CrossRef]
- De Groof, A.; Hémon, P.; Mignen, O.; Pers, J.O.; Wakeland, E.K.; Renaudineau, Y.; Lauwerys, B.R. Dysregulated lymphoid cell populations in mouse models of systemic lupus erythematosus. Clin. Rev. Allergy Immunol. 2017, 53, 181–197. [Google Scholar] [CrossRef]
- Gronthos, S.; Akintoye, S.O.; Wang, C.Y.; Shi, S. Bone marrow stromal stem cells for tissue engineering. Horm. Metab. Res. 2006, 41, 188–195. [Google Scholar] [CrossRef]
- Miura, Y.; Gao, Z.; Miura, M.; Sonoyama, W.; Chen, W.; Gronthos, S.; Zhang, L.; Shi, S. Mesenchymal stem cell-organized bone marrow elements: An alternative hematopoietic progenitor resource. Stem Cells 2006, 24, 2428–2436. [Google Scholar] [CrossRef] [PubMed]
- Yamaza, T.; Miura, Y.; Akiyama, K.; Bi, Y.; Sonoyama, W.; Gronthos, S.; Chen, W.; Le, A.; Shi, S. Mesenchymal stem cell-mediated ectopic hematopoiesis alleviates aging-related phenotype in immunocompromised mice. Blood 2009, 113, 2595–2604. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sonoda, S.; Murata, S.; Nishida, K.; Kato, H.; Uehara, N.; Kyumoto, Y.N.; Yamaza, H.; Takahashi, I.; Kukita, T.; Yamaza, T. Extracellular vesicles from deciduous pulp stem cells recover bone loss by regulating telomerase activity in an osteoporosis mouse model. Stem Cell Res. Ther. 2020, 11, 296. [Google Scholar] [CrossRef] [PubMed]
- Denham, J.; Sellami, M. Exercise training increases telomerase reverse transcriptase gene expression and telomerase activity: A systematic review and meta-analysis. Ageing Res. Rev. 2021, 70, 101411. [Google Scholar] [CrossRef] [PubMed]
- Shi, S.; Gronthos, S.; Chen, S.; Reddi, A.; Counter, C.M.; Robey, P.G.; Wang, C.Y. Bone formation by human postnatal bone marrow stromal stem cells is enhanced by telomerase expression. Nat. Biotechnol. 2002, 20, 587–591. [Google Scholar] [CrossRef]
- Dey, S.; Lee, J.; Noguchi, C.T. Erythropoietin non-hematopoietic tissue response and regulation of metabolism during diet induced obesity. Front. Pharmacol. 2021, 12, 725734. [Google Scholar] [CrossRef]
- Suresh, S.; de Castro, L.F.; Dey, S.; Robey, P.G.; Noguchi, C.T. Erythropoietin modulates bone marrow stromal cell differentiation. Bone Res. 2019, 7, 1–14. [Google Scholar] [CrossRef]
- Suresh, S.; Lee, J.; Noguchi, C.T. Erythropoietin signaling in osteoblasts is required for normal bone formation and for bone loss during erythropoietin-stimulated erythropoiesis. FASEB J. 2020, 34, 11685–11697. [Google Scholar] [CrossRef]
- Noxon, V.; Knopf, K.B.; Norris, L.B.; Chen, B.; Yang, Y.T.; Qureshi, Z.P.; Hrushesky, W.; Lebby, A.A.; Schooley, B.; Hikmet, N.; et al. Tale of two erythropoiesis-stimulating agents: Utilization, dosing, litigation, and costs of darbepoetin and epoetin in South Carolina Medicaid-covered patients with cancer and chemotherapy-induced anemia. J. Oncol. Pract. 2017, 13, e562–e573. [Google Scholar] [CrossRef]
- Yamaza, T.; Miura, Y.; Bi, Y.; Liu, Y.; Akiyama, K.; Sonoyama, W.; Patel, V.; Gutkind, S.; Young, M.; Gronthos, S.; et al. Pharmacologic stem cell based intervention as a new approach to osteoporosis treatment in rodents. PLoS ONE 2008, 3, e2615. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Akiyama, K.; Zhang, H.; Yamaza, T.; Hou, Y.; Zhao, S.; Xu, T.; Le, A.; Shi, S. Mesenchymal stem cell transplantation reverses multiorgan dysfunction in systemic lupus erythematosus mice and humans. Stem Cells 2009, 27, 1421–1432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, C.; Akiyama, K.; Yamaza, T.; You, Y.O.; Xu, X.; Li, B.; Zhao, Y.; Shi, S. Telomerase governs immunomodulatory properties of mesenchymal stem cells by regulating FAS ligand expression. EMBO Mol. Med. 2014, 6, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xu, D.; Song, Y.; He, R.; Wang, T. Research progress in the application of exosomes in immunotherapy. Front. Immunol. 2022, 13, 731516. [Google Scholar] [CrossRef]
- Mai, Z.; Chen, H.; Ye, Y.; Hu, Z.; Sun, W.; Cui, L.; Zhao, X. Translational and clinical applications of dental stem cell-derived exosomes. Front. Genet. 2021, 12, 750990. [Google Scholar] [CrossRef]
- Alexander, M.; Ramstead, A.G.; Bauer, K.M.; Lee, S.H.; Runtsch, M.C.; Wallace, J.; Huffaker, T.B.; Larsen, D.K.; Tolmachova, T.; Seabra, M.C.; et al. Rab27-dependent exosome production inhibits chronic inflammation and enables acute responses to inflammatory stimuli. J. Immunol. 2017, 199, 3559–3570. [Google Scholar] [CrossRef] [Green Version]
- Dogan, F.; Forsyth, N.R. Telomerase Regulation: A Role for Epigenetics. Cancers 2021, 13, 1213. [Google Scholar] [CrossRef] [PubMed]
- Garsuault, D.; Bouyer, C.; Nguyen, E.; Kandhari, R.; Prochazkova-Carlotti, M.; Chevret, E.; Forgez, P.; Ségal-Bendirdjian, E. Complex context relationships between DNA methylation and accessibility, histone marks, and hTERT gene expression in acute promyelocytic leukemia cells: Perspectives for all-trans retinoic acid in cancer therapy. Mol. Oncol. 2020, 14, 1310–1326. [Google Scholar] [CrossRef] [Green Version]
- Martin, S.L.; Kala, R.; Tollefsbol, T.O. Mechanisms for the inhibition of colon cancer cells by sulforaphane through epigenetic modulation of microRNA-21 and human telomerase reverse transcriptase (hTERT) down-regulation. Curr. Cancer Drug. Targets 2018, 18, 97–106. [Google Scholar] [CrossRef]
- Song, G.; Wang, R.; Guo, J.; Liu, X.; Wang, F.; Qi, Y.; Wan, H.; Liu, M.; Li, X.; Tang, H. MiR-346 and miR-138 competitively regulate hTERT in GRSF1- and AGO2-dependent manners, respectively. Sci. Rep. 2015, 5, 1–15. [Google Scholar] [CrossRef] [Green Version]
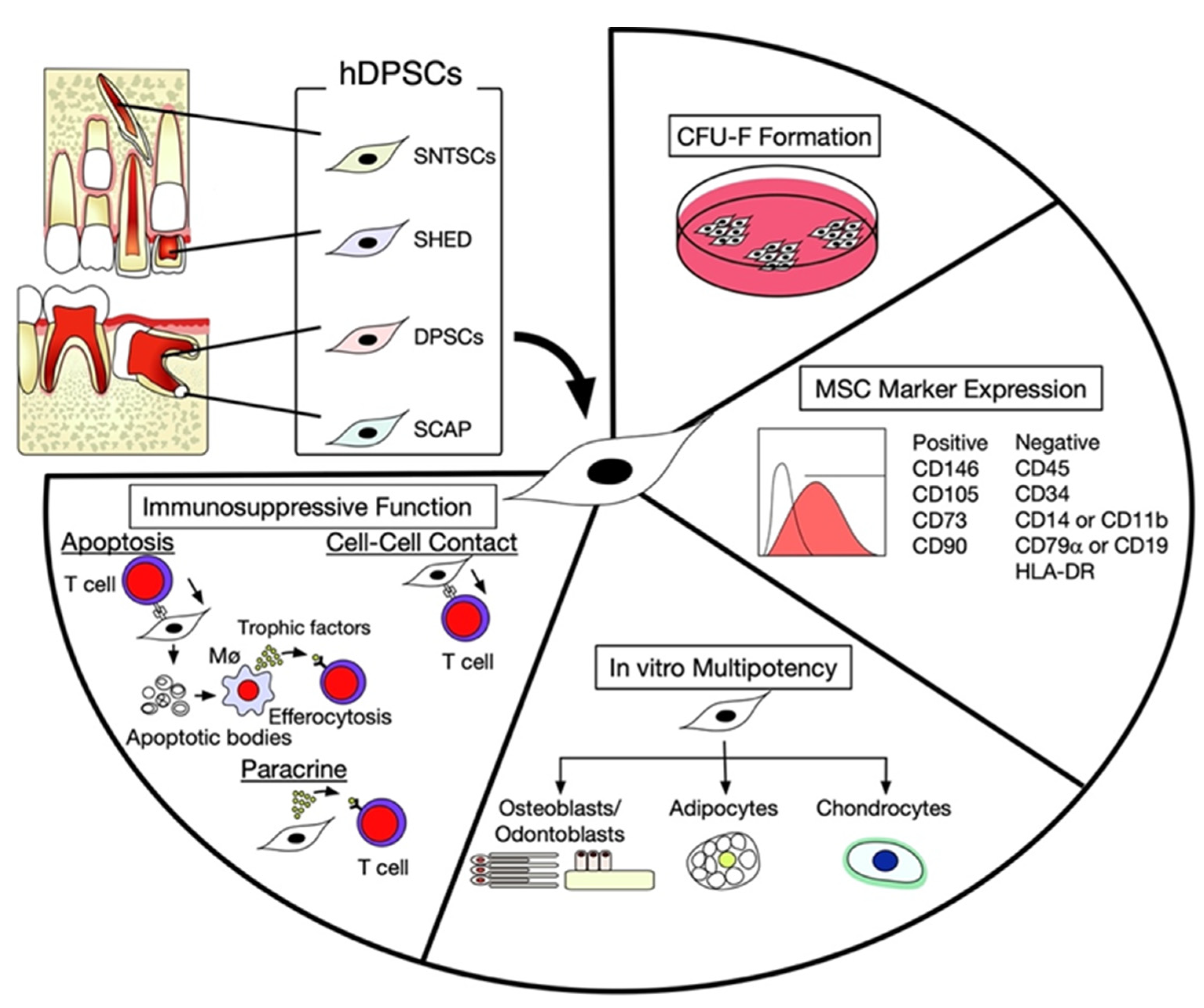
| Cell-Cell Contact Molecules | References |
| FASL/FAS | [14,15,19,36,39,40] |
| PD-L1/PD-1 | [41,42] |
| Paracrine factors and EVs | References |
| HGF | [43] |
| HIF | [44] |
| IDO-1 | [14,45,46,47] |
| IL-10 | [48] |
| MCP-1 | [49,50] |
| NO | [51] |
| PGE2 | [14,39,40] |
| ROS | [51] |
| SIGLEC9 | [49,50] |
| TGFB | [52] |
| EVs | [35,45,53,54,55] |
| Donor | Model | Interpretation | References |
|---|---|---|---|
| hDPSCs | In vitro co-culture Systemic TP into MRL/lpr mice | Th17 cell suppression, Treg induction Improvement in SLE-like disorders | [14,15,19,20] |
| hDPSCs | Systemic TP into OVX mice | FasL/Fas mediated T cell apoptosis | [36] |
| hDPSC-EVs | Systemic TP into OVX mice | Improvement in recipient BMMSC functions. | [63] |
| hDPSC-EVs | Systemic TP into MRL/lpr mice | Improvement in recipient mediated hematopoietic niche formation and hematopoiesis Improvement in SLE-like disorders | [35] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sonoda, S.; Yamaza, T. A New Target of Dental Pulp-Derived Stem Cell-Based Therapy on Recipient Bone Marrow Niche in Systemic Lupus Erythematosus. Int. J. Mol. Sci. 2022, 23, 3479. https://doi.org/10.3390/ijms23073479
Sonoda S, Yamaza T. A New Target of Dental Pulp-Derived Stem Cell-Based Therapy on Recipient Bone Marrow Niche in Systemic Lupus Erythematosus. International Journal of Molecular Sciences. 2022; 23(7):3479. https://doi.org/10.3390/ijms23073479
Chicago/Turabian StyleSonoda, Soichiro, and Takayoshi Yamaza. 2022. "A New Target of Dental Pulp-Derived Stem Cell-Based Therapy on Recipient Bone Marrow Niche in Systemic Lupus Erythematosus" International Journal of Molecular Sciences 23, no. 7: 3479. https://doi.org/10.3390/ijms23073479






