Recent Advances in Sandwich SERS Immunosensors for Cancer Detection
Abstract
:1. Introduction
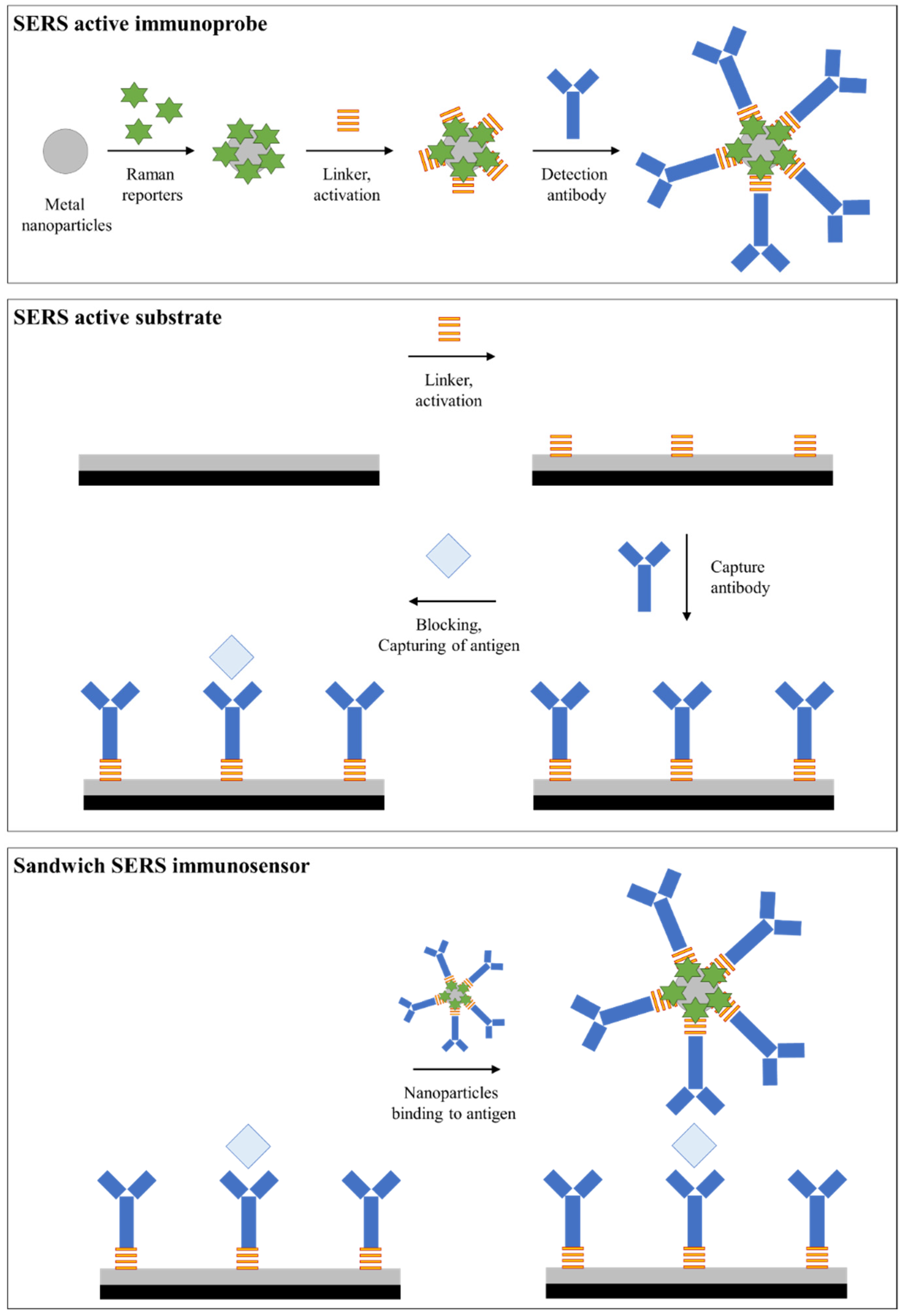
2. Sandwich SERS Immunosensors Structure
2.1. Principle of the Sandwich SERS Immunosensor and Construction
2.2. Solid- and Liquid-Phase SERS-Active Substrate
2.3. Nanospherical SERS-Active Particles
2.4. Preparation of Sandwich SERS Sensor
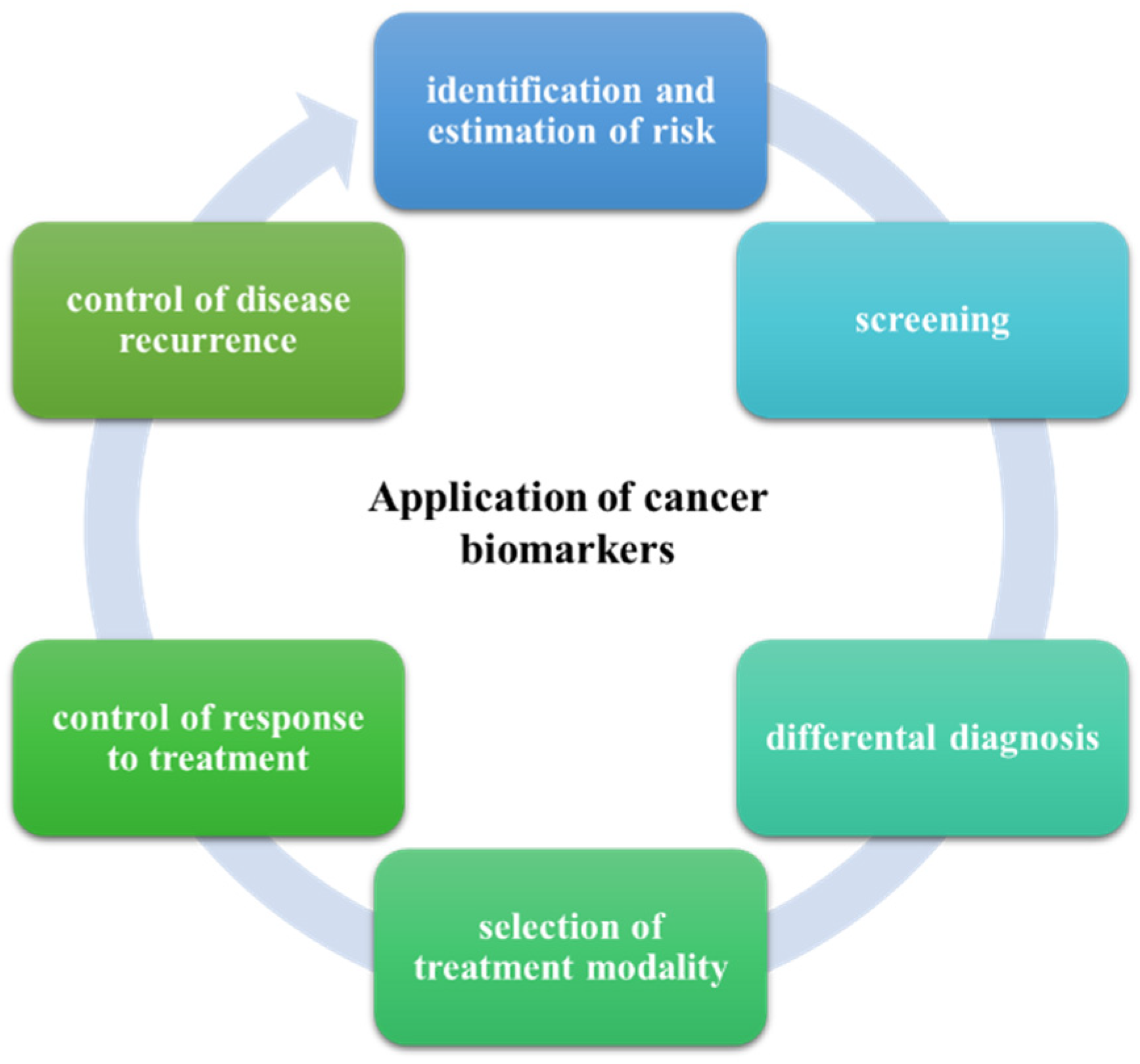
3. Application of Sandwich SERS Immunosensors
3.1. Cancer Detection and Analytical Performances
3.1.1. One-Component Analysis
PSA Detection
AFP Detection
CEA Detection
MUC4 and Interleukins Detection
Detection of Other Biomarkers
3.1.2. Two-Component Analysis
3.1.3. Analysis of Three or More Components
3.2. Sample Preparations
4. Detection Methods
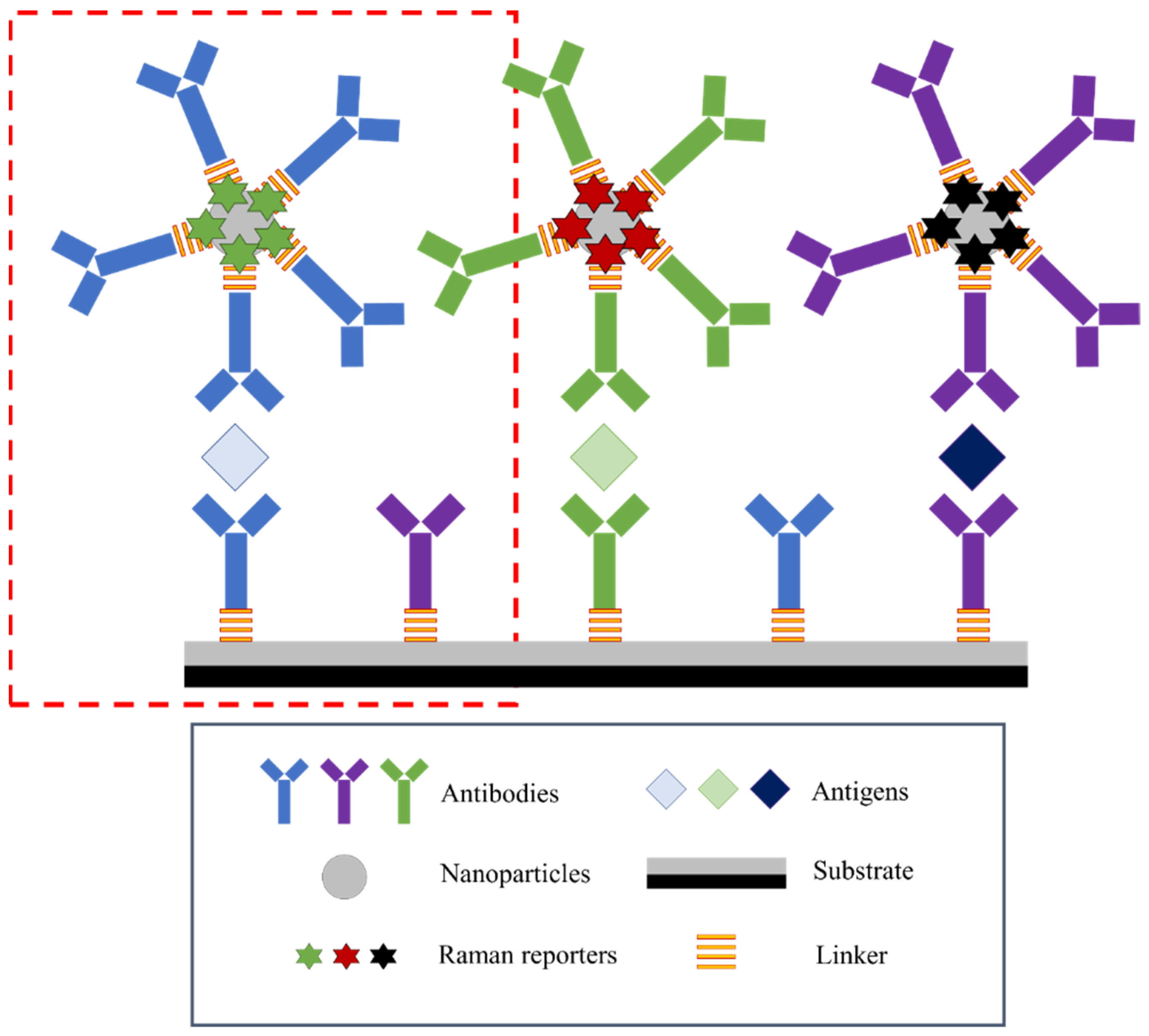
4.1. Single Raman Reporters (RR)
4.2. Dual-Tag Systems
4.3. One-Component Analysis
4.4. Multiplexed Analysis
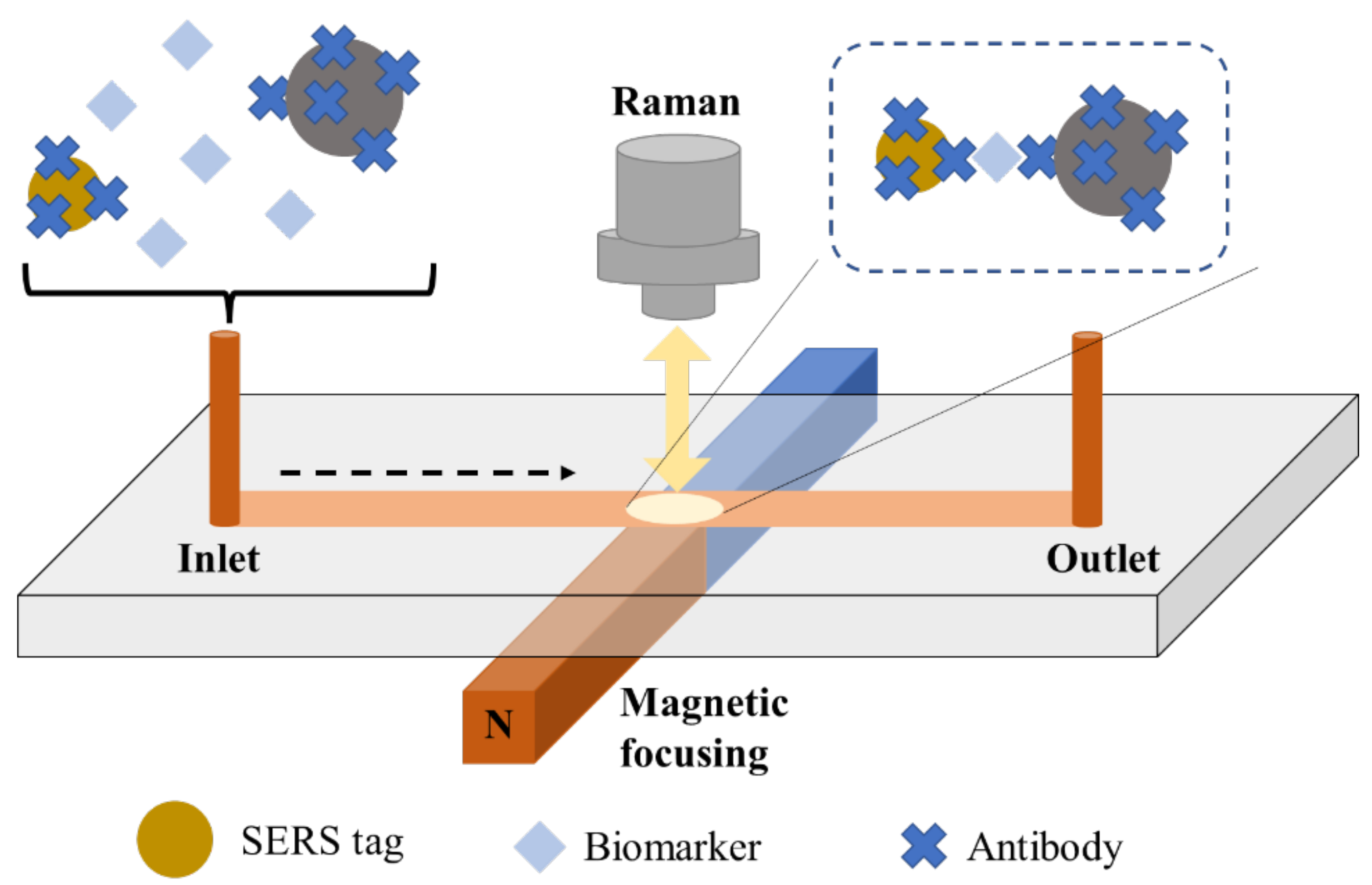
5. Application of Flow Techniques
5.1. Microfluidics Technique
5.2. Lab-on-a-Chip (LOC)
5.3. Lateral Flow Assay (LFA)
6. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zugazagoitia, J.; Guedes, C.; Ponce, S.; Ferrer, I.; Molina-Pinelo, S.; Paz-Ares, L. Current Challenges in Cancer Treatment. Clin. Ther. 2016, 38, 1551–1566. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mulryan, C. Understanding cancer: The basics. Br. J. Healthc. Assist. 2010, 4, 266–269. [Google Scholar] [CrossRef]
- Thakor, A.S.; Gambhir, S.S. Nanooncology: The future of cancer diagnosis and therapy. CA-Cancer J. Clin. 2013, 63, 395–418. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Colombet, M.; Soerjomataram, I.; Parkin, D.M.; Piñeros, M.; Znaor, A.; Bray, F. Cancer statistics for the year 2020: An overview. Int. J. Cancer 2021, 149, 778–789. [Google Scholar] [CrossRef]
- Soto, A.M.; Sonnenschein, C. Environmental causes of cancer: Endocrine disruptors as carcinogens. Nat. Rev. Endocrinol. 2010, 6, 363–370. [Google Scholar] [CrossRef] [Green Version]
- Malhotra, J.; Malvezzi, M.; Negri, E.; La Vecchia, C.; Boffetta, P. Risk factors for lung cancer worldwide. Eur. Respir. J. 2016, 48, 626–627. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruddy, K.J.; Winer, E.P. Male breast cancer: Risk factors, biology, diagnosis, treatment, and survivorship. Ann. Oncol. 2013, 24, 1434–1443. [Google Scholar] [CrossRef]
- Castaño-Guerrero, Y.; Moreira, F.T.C.; Sousa-Castillo, A.; Correa-Duarte, M.A.; Sales, M.G.F. SERS and electrochemical impedance spectroscopy immunoassay for carcinoembryonic antigen. Electrochim. Acta 2021, 366, 137377. [Google Scholar] [CrossRef]
- Wiercigroch, E.; Swit, P.; Brzozka, A.; Pięta, Ł.; Malek, K. Dual-enhancement and dual-tag design for SERS-based sandwich immunoassays: Evaluation of a metal–metal effect in 3D architecture. Microchim. Acta 2022, 189, 32. [Google Scholar] [CrossRef]
- Berg, W.A.; Gutierrez, L.; NessAiver, M.S.; Carter, W.B.; Bhargavan, M.; Lewis, R.S.; Ioffe, O.B. Diagnostic Accuracy of Mammography, Clinical Examination, US, and MR Imaging in Preoperative Assessment of Breast Cancer. Radiology 2004, 233, 830–849. [Google Scholar] [CrossRef]
- Abdullah, D.M.; Ahmed, N.S. A Review of most Recent Lung Cancer Detection Techniques using Machine Learning. Int. J. Sci. Bus. 2021, 5, 159–173. [Google Scholar] [CrossRef]
- Aldhaeebi, M.A.; Alzoubi, K.; Almoneef, T.S.; Bamatraf, S.M.; Attia, H.; Ramahi, O.M. Review of Microwaves Techniques for Breast Cancer Detection. Sensors 2020, 20, 2390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Henry, N.L.; Hayes, D.F. Cancer biomarkers. Mol. Oncol. 2012, 6, 140–146. [Google Scholar] [CrossRef] [Green Version]
- Bhatt, A.N.; Mathur, R.; Farooque, A.; Verma, A.; Dwarakanath, B.S. Cancer biomarkers—Current perspectives. Indian J. Med. Res. 2010, 132, 129–149. [Google Scholar] [PubMed]
- Diamandis, E.P. Present and future of cancer biomarkers. Clin. Chem. Lab. Med. 2014, 52, 791–794. [Google Scholar] [CrossRef] [PubMed]
- Sharma, B.; Frontiera, R.R.; Henry, A.-I.; Ringe, E.; Van Duyne, R.P. SERS: Materials, applications, and the future. Mater. Today 2021, 15, 16–25. [Google Scholar] [CrossRef]
- Hudson, S.D.; Chumanov, G. Bioanalytical applications of SERS (surface-enhanced Raman spectroscopy). Anal. Bioanal. Chem. 2009, 394, 679–686. [Google Scholar] [CrossRef]
- Cialla, D.; März, A.; Böhme, R.; Theil, F.; Weber, K.; Schmitt, M.; Popp, J. Surface-enhanced Raman spectroscopy (SERS): Progress and trends. Anal. Bioanal. Chem. 2012, 403, 27–54. [Google Scholar] [CrossRef]
- Pollap, A.; Kochana, J. Electrochemical Immunosensors for Antibiotic Detection. Biosensors 2019, 9, 61. [Google Scholar] [CrossRef] [Green Version]
- Mollarasouli, F.; Kurbanoglu, S.; Ozkan, S.A. The Role of Electrochemical Immunosensors in Clinical Analysis. Biosensors 2019, 9, 86. [Google Scholar] [CrossRef] [Green Version]
- Holford, T.R.J.; Davis, F.; Higson, S.P.J. Recent trends in antibody based sensors. Biosens. Bioelectron. 2012, 34, 12–24. [Google Scholar] [CrossRef] [PubMed]
- Gan, S.D.; Patel, K.R. Enzyme Immunoassay and Enzyme-Linked Immunosorbent Assay. J. Investig. Dermatol. 2013, 133, e12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Z.Y.; Zong, S.F.; Wu, L.; Zhu, D.; Cui, Y.P. SERS-Activated Platforms for Immunoassay: Probes, Encoding Methods, and Applications. Chem. Rev. 2017, 117, 7910–7963. [Google Scholar] [CrossRef] [PubMed]
- Eom, G.Y.; Hwang, A.; Kim, H.K.; Moon, J.; Kang, H.J.; Jung, J.Y.; Lim, E.-K.; Jeong, J.Y.; Park, H.G.; Kang, T.J. Ultrasensitive Detection of Ovarian Cancer Biomarker Using Au Nanoplate SERS Immunoassay. BioChip J. 2021, 15, 348–355. [Google Scholar] [CrossRef]
- Wan, L.; Zheng, R.; Xiang, J. Au@1,4-benzenedithiol@Au core-shell SERS immunosensor for ultra-sensitive and high specific biomarker detection. Vib. Spectrosc. 2017, 90, 56–62. [Google Scholar] [CrossRef]
- Lee, M.; Lee, K.; Kim, K.H.; Oh, K.W.; Choo, J. SERS-based immunoassay using a gold array-embedded gradient microfluidic chip. Lab Chip 2012, 12, 3720–3727. [Google Scholar] [CrossRef]
- Feng, Z.; Zhou, J.; Chen, D.; Wang, S.-M.; Wang, X.-J.; Xie, S.-S. Hypersensitization Immunoassay of Prostate-specific Antigen Based on SERS of Sandwich-type Au/Ag Nanostructure. Chin. J. Lumin. 2015, 36, 1064–1070. [Google Scholar] [CrossRef]
- Kamińska, A.; Winkler, K.; Kowalska, A.; Witkowska, E.; Szymborski, T.; Janeczek, A.; Waluk, J. SERS-based Immunoassay in a Microfluidic System for the Multiplexed Recognition of Interleukins from Blood Plasma: Towards Picogram Detection. Sci. Rep. 2017, 7, 10656. [Google Scholar] [CrossRef]
- Liu, Y.F.; Ran, M.L.; Sun, Y.; Fan, Y.X.; Wang, J.H.; Cao, X.W.; Lu, D. Sandwich SERS immunoassay platform based on a single-layer Au–Ag nanobox array substrate for simultaneous detection of SCCA and survivin in serum of patients with cervical lesions. RSC Adv. 2021, 11, 36734–36747. [Google Scholar] [CrossRef]
- Wang, Y.L.; Vaidyanathan, R.; Shiddiky, M.J.A.; Trau, M. Enabling Rapid and Specific Surface-Enhanced Raman Scattering Immunoassay Using Nanoscaled Surface Shear Forces. ACS Nano 2015, 9, 6354–6362. [Google Scholar] [CrossRef]
- Gu, X.F.; Wang, K.Y.; Qiu, J.W.; Wang, Y.J.; Tian, S.; He, Z.K.; Zong, R.; Kraatz, H.-B. Enhanced electrochemical and SERS signals by self-assembled gold microelectrode arrays: A dual readout platform for multiplex immumoassay of tumor biomarkers. Sens. Actuators B Chem. 2021, 334, 129674. [Google Scholar] [CrossRef]
- Lu, W.; Wang, Y.; Cao, X.; Li, L.; Dong, J.; Qian, W. Multiplexing determination of lung cancer biomarkers using electrochemical and surface-enhanced Raman spectroscopic techniques. New J. Chem. 2015, 39, 5420–5430. [Google Scholar] [CrossRef]
- Li, L.; Lu, W.B.; Liu, C.; Wang, Y.; Dong, J.; Qian, W.P. Two Types of Immunoassay Based on Nile Blue Labeling Polydopamine Nanospheres. Nano Brief Rep. Rev. 2017, 12, 1750092. [Google Scholar] [CrossRef] [Green Version]
- Wang, A.J.; Ruan, W.D.; Song, W.; Chen, L.; Zhao, B.; Jung, Y.M.; Wang, X. Detection of the potential tumor marker of AFP using surface-enhanced Raman scattering based immunoassay. J. Raman Spectrosc. 2013, 44, 1649–1653. [Google Scholar] [CrossRef]
- Krasnoslobodtsev, A.V.; Torres, M.P.; Kaur, S.; Vlassiouk, I.V.; Lipert, R.J.; Jain, M.; Batra, S.K.; Lyubchenko, Y.L. Nano-immunoassay with improved performance for detection of cancer biomarkers. Nanomedicine 2015, 11, 167–173. [Google Scholar] [CrossRef] [Green Version]
- Lai, W.; Zhou, J.; Liu, Y.T.; Jia, Z.H.; Xie, S.S.; Pettid, L.; Mormile, P. 4MBA-labelled Ag-nanorod aggregates coated with SiO2: Synthesis, SERS activity, and biosensing applications. Anal. Methods 2015, 7, 8832–8838. [Google Scholar] [CrossRef]
- Boca, S.; Farcau, C.; Baia, M.; Astilean, S. Metanephrine neuroendocrine tumor marker detection by SERS using Au nanoparticle/Au film sandwich architecture. Biomed. Microdevices 2016, 18, 12. [Google Scholar] [CrossRef]
- Ma, H.; Sun, X.Y.; Chen, L.; Cheng, W.; Han, X.X.; Zhao, B.; He, C.Y. Multiplex Immunochips for High-Accuracy Detection of AFP-L3% Based on Surface-Enhanced Raman Scattering: Implications for Early Liver Cancer Diagnosis. Anal. Chem. 2017, 89, 8877–8883. [Google Scholar] [CrossRef]
- Xie, L.L.; Yang, X.; He, Y.; Yuan, R.; Chai, Y.Q. Polyacrylamide Gel-Contained Zinc Finger Peptide as the “Lock” and Zinc Ions as the “Key” for Construction of Ultrasensitive Prostate-Specific Antigen SERS Immunosensor. ACS Appl. Mater. Interfaces 2018, 10, 15200–15206. [Google Scholar] [CrossRef]
- Zhou, L.; Liu, Y.; Wang, F.; Jia, Z.; Zhou, J.; Jiang, T.; Petti, L.; Chen, Y.; Xiong, Q.; Wang, X. Classification analyses for prostate cancer, benign prostate hyperplasia and healthy subjects by SERS-based immunoassay of multiple tumour markers. Talanta 2018, 188, 238–244. [Google Scholar] [CrossRef]
- Wang, Z.; Fu, H.; Liang, Z.-H.; Wang, F.-Y.; Zhou, J. Surface Enhanced Raman Scattering-based Tumor Immunoassay Using PS@Ag Nano-probes and Si@Ag Array Substrat. Acta Photonica Sin. 2018, 47, 1228002. [Google Scholar] [CrossRef]
- Lee, M.; Lee, S.; Lee, J.-H.; Lim, H.-W.; Seong, G.H.; Lee, E.K.; Chang, S.-I.; Oh, C.H.; Choo, J. Highly reproducible immunoassay of cancer markers on a gold-patterned microarray chip using surface-enhanced Raman scattering imaging. Biosens. Bioelectron. 2011, 26, 2135–2141. [Google Scholar] [CrossRef] [PubMed]
- Hong, W.-J.; Seo, H.K.; Jung, Y.M. SERS Immunoassay Using Microcontact Printing for Application of Sensitive Biosensors. Bull. Korean Chem. Soc. 2011, 32, 4281–4285. [Google Scholar] [CrossRef] [Green Version]
- Li, M.; Cushing, S.K.; Zhang, J.M.; Suri, S.; Evans, R.; Petros, W.P.; Gibson, L.F.; Ma, D.L.; Liu, Y.X.; Wu, N.Q. Three-Dimensional Hierarchical Plasmonic Nano-Architecture Enhanced Surface-Enhanced Raman Scattering Immunosensor for Cancer Biomarker Detection in Blood Plasma. ACS Nano 2013, 7, 4967–4976. [Google Scholar] [CrossRef]
- Granger, J.H.; Granger, M.C.; Firpo, M.A.; Mulvihill, S.J.; Porter, M.D. Toward development of a surface-enhanced Raman scattering (SERS)-based cancer diagnostic immunoassay panel. Analyst 2013, 138, 410–416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, L.; Liu, C.; Cao, X.W.; Tan, L.Q.; Lu, W.B. Multiplexing determination of cancer-associated biomarkers by surface-enhanced Raman scattering using ordered gold nanohoneycomb arrays. Bioanalysis 2017, 9, 1561–1572. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.; Lipert, R.J. Detection of the Potential Pancreatic Cancer Marker MUC4 in Serum Using Surface-Enhanced Raman Scattering. Anal. Chem. 2011, 83, 2554–2561. [Google Scholar] [CrossRef] [Green Version]
- Zhou, L.; Zhou, J.; Feng, A.; Wang, F.Y.; Xie, S.S.; Bub, S.Z. Immunoassay for tumor markers in human serum based on Si nanoparticles and SiC@Ag SERS-active substrate. Analyst 2016, 141, 2534–2541. [Google Scholar] [CrossRef]
- Yang, Z.Y.; Liu, H.M.; Tian, Y.; Chen, Y.; Niu, Z.Q.; Zhou, C.L.; Wang, F.Y.; Gu, C.J.; Tang, S.W.; Jiang, T.; et al. Synergistic effect of a ‘‘stellate’’ mesoporous SiO2@Au nanoprobe and coffee-ring-free hydrophilic–hydrophobic substrate assembly in an ultrasensitive SERS-based immunoassay for a tumor marker. J. Mater. Chem. C 2020, 8, 2142–2154. [Google Scholar] [CrossRef]
- Du, Y.Y.; Liu, H.M.; Tian, Y.; Gu, C.J.; Zhao, Z.Q.; Zeng, S.W.; Jiang, T. Recyclable SERS-Based Immunoassay Guided by Photocatalytic Performance of Fe3O4@TiO2@Au Nanocomposites. Biosensors 2020, 10, 25. [Google Scholar] [CrossRef] [Green Version]
- Lu, D.; Ran, M.L.; Liu, Y.F.; Xia, J.; Bi, L.Y.; Cao, X.W. SERS spectroscopy using Au-Ag nanoshuttles and hydrophobic paper-based Au nanoflower substrate for simultaneous detection of dual cervical cancer–associated serum biomarkers. Anal. Bioanal. Chem. 2020, 412, 7099–7112. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.L.; Zhou, L.; Wei, G.D.; Jiang, T.; Zhou, J. SERS-based immunoassay using a core–shell SiO2@Ag immune probe and Ag-decorated NiCo2O4 nanorods immune substrate. RSC Adv. 2016, 6, 708–715. [Google Scholar] [CrossRef]
- Wang, Z.; Yang, H.Q.; Wang, M.H.; Petti, L.; Jiang, T.; Jia, Z.H.; Xie, S.S.; Zhou, J. SERS-based multiplex immunoassay of tumor markers using double SiO2@Ag immune probes and gold-film hemisphere array immune substrate. Colloids Surf. A 2018, 546, 48–58. [Google Scholar] [CrossRef]
- Lu, D.; Xia, J.; Deng, Z.; Cao, X.W. Detection of squamous cell carcinoma antigen in cervical cancer by surface-enhanced Raman scattering-based immunoassay. Anal. Methods 2019, 11, 2809–2818. [Google Scholar] [CrossRef]
- Feng, J.; Xu, Y.J.; Huang, W.Y.; Kong, H.X.; Li, Y.Q.; Cheng, H.; Li, L.J. A magnetic SERS immunosensor for highly sensitive and selective detection of human carboxylesterase 1 in human serum samples. Anal. Chim. Acta 2020, 1097, 176–185. [Google Scholar] [CrossRef]
- Panikar, S.S.; Banu, N.; Haramati, J.; Gutierrez-Silerio, G.Y.; Bastidas-Ramirez, B.E.; Tellez-Banuelos, M.C.; Camacho-Villegas, T.A.; del Toro-Arreola, S.; De la Rosa, E. Anti-fouling SERS-based immunosensor for point-of-care detection of the B7-H6 tumor biomarker in cervical cancer patient serum. Anal. Chim. Acta 2020, 1138, 110–122. [Google Scholar] [CrossRef]
- Medetalibeyoglu, H.; Kotan, G.; Atar, N.; Yola, M.L. A novel sandwich-type SERS immunosensor for selective and sensitive carcinoembryonic antigen (CEA) detection. Anal. Chim. Acta 2020, 1139, 100–110. [Google Scholar] [CrossRef]
- Yun, B.J.; Koh, W.-G. Highly-sensitive SERS-based immunoassay platform prepared on silver nanoparticle-decorated electrospun polymeric fibers. J. Ind. Eng. Chem. 2020, 82, 341–348. [Google Scholar] [CrossRef]
- Tian, Y.R.; Li, X.T.; Wang, F.Y.; Gu, C.J.; Zhao, Z.Q.; Si, H.J.; Jiang, T. SERS-based immunoassay and degradation of CA19-9 mediated by gold nanowires anchored magnetic–semiconductor nanocomposites. J. Hazard. Mater. 2021, 403, 124009. [Google Scholar] [CrossRef]
- Ye, J.; Chen, Y.; Liu, Z. A Boronate Affinity Sandwich Assay: An Appealing Alternative to Immunoassays for the Determination of Glycoproteins. Angew. Chem. Int. Ed. 2014, 53, 10386–10389. [Google Scholar] [CrossRef]
- Feng, J.; Li, X.; Cheng, H.; Huang, W.Y.; Kong, H.X.; Li, Y.Q.; Li, L.J. A boronate-modified molecularly imprinted polymer labelled with a SERS-tag for use in an antibody-free immunoassay for the carcinoembryonic antigen. Microchim. Acta 2019, 186, 774. [Google Scholar] [CrossRef]
- Carneiro, M.C.C.G.; Sousa-Castillo, A.; Correa-Duarte, M.A.; Sales, M.G.F. Dual biorecognition by combining molecularly-imprinted polymer and antibody in SERS detection. Application to carcinoembryonic antigen. Biosens. Bioelectron. 2019, 146, 111761. [Google Scholar] [CrossRef] [PubMed]
- Turan, E.; Zengin, A.; Suludere, Z.; Kalkan, N.O.; Tamer, U. Construction of a sensitive and selective plasmonic biosensor for prostate specific antigen by combining magnetic molecularly-imprinted polymer and surface-enhanced Raman spectroscopy. Talanta 2022, 237, 122926. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhen, S.J.; Li, Y.F.; Huang, C.Z. Silver nanoparticles deposited on graphene oxide for ultrasensitive surface-enhanced Raman scattering immunoassay of cancer biomarker. Nanoscale 2018, 10, 11942–11947. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.J.; Liu, H.W.; Huang, C.H.; Yao, C.Z.; Fu, Q.Q.; Li, X.Q.; Cao, D.L.; Luo, Z.; Tang, Y. Aggregated Silver Nanoparticles Based Surface-Enhanced Raman Scattering Enzyme-Linked Immunosorbent Assay for Ultrasensitive Detection of Protein Biomarkers and Small Molecules. Anal. Chem. 2015, 87, 5790–5796. Available online: https://pubs.acs.org/doi/10.1021/acs.analchem.5b01011#:~:text=https%3A//doi.org/10.1021/acs.analchem.5b01011 (accessed on 20 March 2022). [CrossRef]
- Zhao, J.; Wu, C.; Zhai, L.P.; Shi, X.F.; Li, X.; Weng, G.J.; Zhu, J.; Li, J.J.; Zhao, J.-W. A SERS-based immunoassay for the detection of α-fetoprotein using AuNS@Ag@SiO2 core–shell nanostars. J. Mater. Chem. C 2019, 7, 8432–8441. [Google Scholar] [CrossRef]
- Yang, Y.; Zhu, J.; Zhao, J.; Weng, G.-J.; Li, J.-J.; Zhao, J.-W. Growth of Spherical Gold Satellites on the Surface of Au@Ag@SiO2 Core−Shell Nanostructures Used for an Ultrasensitive SERS Immunoassay of Alpha-Fetoprotein. ACS Appl. Mater. Interfaces 2019, 11, 3617–3626. [Google Scholar] [CrossRef]
- Xia, J.; Liu, Y.F.; Ran, M.L.; Lu, W.B.; Bi, L.Y.; Wang, Q.; Lu, D.; Cao, X.W. The simultaneous detection of the squamous cell carcinoma antigen and cancer antigen 125 in the cervical cancer serum using nano-Ag polydopamine nanospheres in an SERS-based lateral flow immunoassay. RSC Adv. 2020, 10, 29156–29170. [Google Scholar] [CrossRef]
- Li, M.; Kang, J.W.; Sukumar, S.; Dasari, R.R.; Barman, I. Multiplexed detection of serological cancer markers with plasmon-enhanced Raman spectro-immunoassay. Chem. Sci. 2015, 6, 3906–3914. [Google Scholar] [CrossRef] [Green Version]
- Bizzarri, A.R.; Moscetti, I.; Cannistraro, S. Surface enhanced Raman spectroscopy based immunosensor for ultrasensitive and selective detection of wild type p53 and mutant p53R175H. Anal. Chim. Acta 2018, 1029, 86–96. [Google Scholar] [CrossRef]
- Er, E.; Sánchez-Iglesias, A.; Silvestri, A.; Arnaiz, B.; Liz-Marzán, L.M.; Prato, M.; Criado, A. Metal Nanoparticles/MoS2 Surface-Enhanced Raman Scattering-Based Sandwich Immunoassay for α-Fetoprotein Detection. ACS Appl. Mater. Interfaces 2021, 13, 8823–8831. [Google Scholar] [CrossRef] [PubMed]
- Jibin, K.; Babu, R.V.; Jayasree, R.S. Graphene–Gold Nanohybrid-Based Surface-Enhanced Raman Scattering Platform on a Portable Easy-to-Use Centrifugal Prototype for Liquid Biopsy Detection of Circulating Breast Cancer Cells. ACS Sustain. Chem. Eng. 2021, 9, 15496–15505. [Google Scholar] [CrossRef]
- Chon, H.; Lee, S.; Yoon, S.-Y.; Chang, S.-I.; Lima, D.W.; Choo, J. Simultaneous immunoassay for the detection of two lung cancer markers using functionalized SERS nanoprobes. Chem. Commun. 2011, 47, 12515–12517. [Google Scholar] [CrossRef]
- Liu, R.Y.; Liu, B.H.; Guan, G.J.; Jianga, C.L.; Zhang, Z.P. Multilayered shell SERS nanotags with a highly uniform single-particle Raman readout for ultrasensitive immunoassays. Chem. Commun. 2012, 48, 9421–9423. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Dong, J.; Xie, W.; Tao, L.; Lu, W.; Wang, Y.; Qian, W. SERS tags-based novel monodispersed hollow gold nanospheres for highly sensitive immunoassay of CEA. J. Mater. Sci. 2015, 50, 3329–3336. [Google Scholar] [CrossRef]
- Zong, S.F.; Wang, L.; Chen, C.; Lu, J.; Zhu, D.; Zhang, Y.Z.; Wang, Z.Y.; Cui, Y.P. Facile detection of tumor-derived exosomes using magnetic nanobeads and SERS nanoprobes. Anal. Methods 2016, 8, 5001–5008. [Google Scholar] [CrossRef]
- Gao, R.; Cheng, Z.; DeMello, A.J.; Choo, J. Wash-free magnetic immunoassay of the PSA cancer marker using SERS and droplet microfluidics. Lab Chip 2016, 16, 1022–1029. [Google Scholar] [CrossRef]
- Gao, R.K.; Lv, Z.Y.; Mao, Y.S.; Yu, L.D.; Bi, X.B.; Xu, S.H.; Cui, J.W.; Wu, Y.C. SERS-Based Pump-Free Microfluidic Chip for Highly Sensitive Immunoassay of Prostate-Specific Antigen Biomarkers. ACS Sens. 2019, 4, 938–943. [Google Scholar] [CrossRef]
- Wang, Y.J.; Zong, S.F.; Li, N.; Wang, Z.Y.; Chen, B.; Cui, Y.P. SERS-based dynamic monitoring of minimal residual disease markers with high sensitivity for clinical applications. Nanoscale 2019, 11, 2460–2467. [Google Scholar] [CrossRef]
- Bai, X.-R.; Wang, L.-H.; Ren, J.-Q.; Bai, X.-W.; Zeng, L.-W.; Shen, A.-G.; Hu, J.-M. Accurate Clinical Diagnosis of Liver Cancer Based on Simultaneous Detection of Ternary Specific Antigens by Magnetic Induced Mixing Surface-Enhanced Raman Scattering Emissions. Anal. Chem. 2019, 91, 2955–2963. [Google Scholar] [CrossRef]
- Li, D.; Jiang, L.M.; Piper, J.A.; Maksymov, I.S.; Greentree, A.D.; Wang, E.; Wang, Y.L. Sensitive and Multiplexed SERS Nanotags for the Detection of Cytokines Secreted by Lymphoma. ACS Sens. 2019, 4, 2507–2514. [Google Scholar] [CrossRef]
- Sun, D.; Cao, F.H.; Xu, W.Q.; Chen, Q.D.; Shi, W.; Xu, S.P. Ultrasensitive and Simultaneous Detection of Two Cytokines Secreted by Single Cell in Microfluidic Droplets via Magnetic-Field Amplified SERS. Anal. Chem. 2019, 91, 2551–2558. [Google Scholar] [CrossRef]
- Zhang, W.; Jiang, L.M.; Diefenbach, R.J.; Campbell, D.H.; Walsh, B.J.; Packer, N.H.; Wang, Y.L. Enabling Sensitive Phenotypic Profiling of Cancer-Derived Small Extracellular Vesicles Using Surface-Enhanced Raman Spectroscopy Nanotags. ACS Sens. 2020, 5, 764–771. [Google Scholar] [CrossRef] [PubMed]
- Ge, M.; Wei, C.; Xu, M.M.; Fang, C.W.; Yuan, Y.X.; Gua, R.; Yao, J.L. Ultra-sensitive magnetic immunoassay of HE4 based on surface enhanced Raman spectroscopy. Anal. Methods 2015, 7, 6489–6495. [Google Scholar] [CrossRef]
- Baniukevic, J.; Boyaci, J.H.; Bozkurt, A.G.; Tamer, U.; Ramanavicius, A.; Ramanaviciene, A. Magnetic gold nanoparticles in SERS-based sandwich immunoassay for antigen detection by well oriented antibodies. Biosens. Bioelectron. 2013, 43, 281–288. [Google Scholar] [CrossRef]
- Song, C.Y.; Yang, Y.J.; Yang, B.Y.; Min, L.H.; Wang, L.H. Combination assay of lung cancer associated serum markers using surface-enhanced Raman spectroscopy. J. Mater. Chem. B 2016, 4, 1811–1817. [Google Scholar] [CrossRef]
- Li, J.; Skeete, Z.Y.; Shan, S.Y.; Yan, S.; Kurzatkowska, K.; Zhao, W.; Ngo, Q.M.; Holubovska, P.; Luo, J.; Hepel, M.; et al. Surface Enhanced Raman Scattering Detection of Cancer Biomarkers with Bifunctional Nanocomposite Probes. Anal. Chem. 2015, 87, 10698–10702. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, P.; Li, H.-X.; Li, D.-W.; Hou, Y.-J.; Mao, L.L.; Yang, M.F.; Wang, Y. A SERS nano-tag-based magnetic-separation strategy for highly sensitive immunoassay in unprocessed whole blood. Talanta 2019, 198, 527–533. [Google Scholar] [CrossRef]
- He, Y.H.; Li, L.J.; Li, X.X.; Lin, C.B.; Zhang, Y. Construction of Boronate-Affinity Magnetic Immunity SERS Sensor and Detection of Alpha-Fetoprotein (AFP) in Human Serum. Bull. Chem. Soc. Jpn. 2021, 94, 3. [Google Scholar] [CrossRef]
- Wang, Y.L.; Salehi, M.; Schutz, M.; Rudi, K.; Schlucker, S. Microspectroscopic SERS detection of interleukin-6 with rationally designed gold/silver nanoshells. Analyst 2013, 138, 1764–1771. [Google Scholar] [CrossRef] [Green Version]
- Kullavadee, K.-O. Gap-Dependent Surface-Enhanced Raman Scattering (SERS) Enhancement Model of SERS Substrate−Probe Combination Using a Polyelectrolyte Nanodroplet as a Distance Controller. Langmuir 2021, 37, 10776–10785. [Google Scholar] [CrossRef]
- Li, J.; Dong, S.J.; Tong, J.J.; Zhu, P.Z.; Diao, G.W.; Yang, Z.J. 3D ordered silver nanoshells silica photonic crystal beads for multiplex encoded SERS bioassay. Chem. Commun. 2016, 52, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-Y.; Li, W.; Gong, Z.; Sun, P.-R.; Zhou, T.; Cao, X.-W. Detection of IL-8 in human serum using surface-enhanced Raman scattering coupled with highly-branched gold nanoparticles and gold nanocages. New J. Chem. 2019, 43, 1733–1742. [Google Scholar] [CrossRef]
- Lin, Y.; Xu, G.H.; Wei, F.D.; Zhang, A.X.; Yang, J.; Hu, Q. Detection of CEA in human serum using surface-enhanced Raman spectroscopy coupled with antibody-modified Au and γ-Fe2O3@Au nanoparticles. J. Pharm. Biomed. 2016, 121, 135–140. [Google Scholar] [CrossRef]
- Xue, Y.; Liu, D.; Wang, X.B.; Xiang, Y.X.; Du, S.J.; Ye, K.; Bao, C.Y.; Zhu, L.Y. A photopatterned SERS substrate with a sandwich structure for multiplex detection. Chin. Chem. Lett. 2021, 22, 1595–1598. [Google Scholar] [CrossRef]
- Chen, L.; Sun, H.H.; Zhao, Y.; Gao, R.X.; Wang, Y.X.; Liu, Y.; Zhang, Y.J.; Hua, Z.; Yang, J.H. Iron layer-dependent surface-enhanced raman scattering of hierarchical nanocap arrays. Appl. Surf. Sci. 2017, 423, 1124–1133. [Google Scholar] [CrossRef]
- Chuong, T.T.; Pallaoro, A.; Chaves, C.A.; Li, Z.; Lee, J.; Eisenstein, M.; Stucky, G.D.; Moskovits, M.; Soh, H.T. Dual-reporter SERS-based biomolecular assay with reduced false-positive signals. Proc. Natl. Acad. Sci. USA 2017, 114, 9056–9061. [Google Scholar] [CrossRef] [Green Version]
- Whitesides, G.M. The origins and the future of microfluidics. Nature 2006, 442, 368–373. [Google Scholar] [CrossRef]
- Convery, N.; Gadegaard, N. 30 years of microfluidics. Micro Nano Eng. 2019, 2, 76–91. [Google Scholar] [CrossRef]
- Sackmann, E.K.; Fulton, A.L.; Beebe, D.J. The present and future role of microfluidics in biomedical research. Nature 2014, 507, 181–189. [Google Scholar] [CrossRef]
- Chin, C.D.; Laksanasopin, T.; Cheung, Y.K.; Steinmiller, D.; Linder, V.; Parsa, H.; Wang, J.; Moore, H.; Rouse, R.; Umviligihozo, G.; et al. Microfluidics-based diagnostics of infectious diseases in the developing world. Nat. Med. 2011, 17, 1015–1019. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Chen, D.; Xie, Y.; Yuan, T.; Chen, X. Progress of Microfluidics for Biology and Medicine. Nano-Micro Lett. 2013, 5, 66–80. [Google Scholar] [CrossRef] [Green Version]
- Rivet, C.; Lee, H.; Hirsch, A.; Hamilton, S.; Lu, H. Microfluidics for medical diagnostics and biosensors. Chem. Eng. Sci. 2011, 66, 1490–1507. [Google Scholar] [CrossRef]
- Kant, K.; Abalde-Cela, S. Surface-Enhanced Raman Scattering Spectroscopy and Microfluidics: Towards Ultrasensitive Label-Free Sensing. Biosensors 2018, 8, 62. [Google Scholar] [CrossRef] [Green Version]
- Marks, H.; Schechinger, M.; Garza, J.; Locke, A.; Coté, G. Surface enhanced Raman spectroscopy (SERS) for in vitro diagnostic testing at the point of care. Nanophotonics 2017, 6, 681–701. [Google Scholar] [CrossRef]
- Huang, A.-A.; Zhang, Y.-L.; Ding, H.; Sun, H.-B. SERS-Enabled Lab-on-a-Chip Systems. Adv. Opt. Mater. 2015, 3, 618–633. [Google Scholar] [CrossRef] [Green Version]
- Chen, L.; Choo, J. Recent advances in surface-enhanced Raman scattering detection technology for microfluidic chips. Electrophoresis 2008, 29, 1815–1828. [Google Scholar] [CrossRef]
- Jahn, I.J.; Žukovskaja, O.; Zheng, X.-S.; Weber, K.; Bocklitz, T.W.; Cialla-May, D.; Popp, J. Surface-enhanced Raman spectroscopy and microfluidic platforms: Challenges, solutions and potential applications. Analyst 2017, 142, 1022–1047. [Google Scholar] [CrossRef]
- Koczula, K.M.; Gallotta, A. Lateral flow assays. Essays Biochem. 2016, 60, 111–120. [Google Scholar] [CrossRef]



| Ref. | Biomarker (Multiplexing Ability) | Single Tag (St)/Dual Tag (Dt) (Raman Reporters) | Nanoparticles (NPs) | Platform | Dynamic Range | LOD |
|---|---|---|---|---|---|---|
| [43] | PSA | St (R6G) | Colloidal AuNPs | AuNP layer of the patterned substrate | - | - |
| [27] | PSA | St (MBA) | Nano-Ag immune probes | Nano-Ag/Au immune substrate | - | 1.8 fg mL−1 |
| [36] | PSA | St (MBA) | Silica-coated Ag nanorods (NRs) | Quartz slide with Ag nanorods (NRs) | 0.3 fg mL−1–3 µg mL−1 | 0.3 fg mL−1 |
| [77] | PSA | St (MGITC) | AuNPs | Magnetic beads (MB) | 50 pg mL−1–200 ng mL−1 | 0.1 ng mL−1 |
| [25] | PSA | St (1,4-BDT) | Au core–Au shell NPs | Au plate | 10 pg mL−1–10 ng mL−1 | 2.0 pg mL−1 |
| [39] | PSA | St (TB) | ZnO and CoFe2O4 nanocomplexes with Au | Si@Ag substrate | 1 pg mL−1–10 ng mL−1 | 0.65 pg mL−1 |
| [64] | PSA | St (GO) | AgNPs deposited on graphene oxide (GO–AgNP) | Polystyrene 96-well plate substrate | 0.5 pg mL−1–500 pg mL−1 | 0.23 pg mL−1 |
| [78] | PSA | St (MGITC) | AuNPs | MB | 0.01 ng mL−1–100 ng mL−1 | 0.01 ng mL−1 |
| [58] | PSA | St (MBA) | AuNPs | Silver-NP-decorated electrospun polymeric fibers | 1–10 pg mL−1 | 1 pg mL−1 |
| [50] | PSA | St (MBA) | Au seeds on Fe3O4@TiO2 core–shell NPs | Ag-coated sandpaper | 10−4–10−12 g mL−1 | 0.014 mM |
| [63] | PSA | St (DNTB) | AuNPs | Magnetic molecularly imprinted polymers (MMIPs) | 0.5 pg mL−1–1.0 μg mL−1 | 0.9 pg mL−1 |
| [26] | AFP | St (MGITC) | Hollow gold nanospheres (HGNs) | Gold array | 0–10 ng mL−1 | 0–1 ng mL−1 |
| [34] | AFP | St (MBA) | AuNPs | Glass slide modified with AuNPs | 1–100 ng mL−1 | 100 pg/mL |
| [60] | AFP | St (MPBA) | AgNPs | Molecularly imprinted polymer (MIP) array | 1 ng mL−1–10 µg mL−1 | - |
| [52] | AFP | St (MBA) | Core-shell SiO2@Ag | Ag-decorated NiCo2O4 nanorods | 2.1 fg mL−1–2.1 ng mL−1 | 2.1 fg mL−1 |
| [41] | AFP | St (MBA) | Ag-covered polystyrene sphere (PS@Ag) | Deposited Si pyramid array (Si@Ag) substrate | 2 fg mL−1–200 ng mL−1 | 1.75 fg mL−1 |
| [66] | AFP | St (MBA) | Silica-coated gold/silver core–shell nanostars (AuNS@Ag@SiO2) | Nitrocellulose (NC) membrane | 3 pg mL−1–3 µg mL−1 | 0.72 pg mL−1 |
| [67] | AFP | St (MBA) | Nanosphere with a silver coating core (Au@Ag), ultrathin continuous silica (SiO2) shell, and high coverage of gold nanosphere (AuNP) satellites | Nitrocellulose (NC) membrane | 1 fg mL−1–1 ng mL−1 | 0.3 fg mL−1 |
| [89] | AFP | St (MBA) | Gold-coated silver nanoparticles (Ag@AuNPs) | Boric-acid-functionalized magnetic silica particles | 1.0 ng mL−1–1.0 mg mL−1 | 1.0 ng mL−1 |
| [71] | AFP | St (R6G) | Silver-coated gold nanocubes | Molybdenum disulfide (MoS2) | 1 pg mL−1–10 ng mL−1 | 0.03 pg mL−1 |
| [75] | CEA | St (MBA) | HGNs | Magnetic microspheres | 0 ng mL−1–100 ng mL−1 | 10 pg mL−1 |
| [87] | CEA | St (MBA) | AuNPs | Au-coated NiFe magnetic nanoparticles (NiFe@Au) | 0 ng mL−1–1 ng mL−1 | 0.1 pM |
| [94] | CEA | St (MBA) | AuNPs | γ-Fe2O3@AuNPs | 1 ng mL−1–50 ng mL−1 | 0.1 ng mL−1 |
| [33] | CEA | St (Nile blue) | AuNPs with polydopamine resin (PDR) | Chitosan-stabilized AuNPs on a glassy carbon electrode (GCE) | 1 pg mL−1-100 ng mL−1 | 0.68 pg mL−1 |
| [61] | CEA | St (MPBA) | AuNPs | Boronate affinity molecularly imprinted polymer (MIP) array | 0.1 ng mL−1–1 mg mL−1 | 0.1 ng mL−1 |
| [62] | CEA | St (ATP) | Gold nanostars (AuNSs) | Molecularly-imprinted polymer (MIP) film | 0–1000 ng mL−1 | 1.0 ng mL−1 |
| [57] | CEA | St (MBA) | MoS2 nanoflowers@AuNPs | Fe3O4@AuNP-functionalized delaminated Ti3C2Tx MXene-magnetic supporting substrate | 0.0001–100.0 ng mL−1 | 0.033 pg mL−1 |
| [8] | CEA | St (ATP) | AuNSs | Screen-printed electrode (Au-SPE) | 0.025–250 ng mL−1 | 0.025 ng mL−1 |
| [54] | SCCA | St (MBA) | Gold nanocages (GNCs) | Gold-nanoparticle-coated polydopamine resin microspheres (PDR@GNPs) | 1 × 10−5 M–1 × 10−10 M | 7.16 pg mL−1 |
| [47] | MUC4 | St (NBT) | AuNPs | Template-stripped gold (TSG) | 0 μg mL−1–1 μg mL−1 | 33 ng mL−1 |
| [35] | MUC4 | St (NBT) | AuNPs | Glass chip with a gold layer | 0.1 μg mL−1–20 μg mL−1 | 0.1 μg mL−1 |
| [90] | IL-6 | St (covalent conjugation of DTNB to a short MEG–OH and a longer TEG–CO2H group) | AuNPs | Gold/silver nanoshells (Au/AgNSs) | 1 pg mL−1–1 µg mL−1 | 1 pg mL−1 |
| [9] | IL-6 | Dt (MBA, NTP) | Ag and AuNPs | Au and Ag hexagonal nanoarray | 0 pg mL−1–1000 pg mL−1 | 25.2 pg mL−1 |
| [93] | IL-8 | St (MBA) | GNCs | Highly branched gold nanoparticle (HGNP) substrates | 10 pg mL−1–1 µg mL−1 | 6.04 pg mL−1 |
| [44] | VEGF | St (MGITC) | AuNSs | Gold triangle nanoarray | 0.1 pg mL−1–10 ng mL−1 | 1.158 ng mL−1 |
| [85] | BLV | St (DTNB) | Au rods | Magnetic gold NPs (MNP-Au) | 0 mg mL−1–0.06 mg mL−1 | 0.95 µg mL−1 |
| [84] | HE4 | St (MBA) | AuNPs | Magnetic core–shell Fe3O4@AuNPs | 1 pg mL−1–10 ng mL−1 | 100 fg mL−1 |
| [24] | HE4 | St (MGITC) | AuNPs | Gold (Au) nanoplate (NPl) | 0 M–10−9 M | 10−17 M |
| [30] | HER2 | St (MGITC) | Gold/silver nanoshells | Gold electrode surface | 1 fg mL−1–100 pg mL−1 | 10 fg mL−1 |
| [76] | tumor-derived exosomes | St (DTNB) | Gold core–silver shell nanorods (Au@AgNRs) | MB | 4.88 × 106–4.88 × 103 | 1200 exosomes |
| [37] | metanephrine | St (pATP, CV) | AuNPs | Au films on microscope glass slides | 10−3 M–10−5 M | 10−4 M |
| [70] | p53 | St (ATP) | AuNPs | Glass substrate | 10−10 M–10−17 M | 10−15 M |
| [55] | hCE1 | St (MBA) | AgNPs | Raspberry-like morphology of Fe3O4@SiO2@AgNP magnetic nanocomposites | 0.1 ng mL−1–1.0 mg mL−1 | 0.1 ng mL−1 |
| [56] | B7-H6 biomarker | St (ATP) | Spiky AuNPs | Au thin film modified with a self-assembled monolayer of zwitterionic L-cysteine | 10−10 M–10−14 M | 10−14 M (10.8 fg mL−1) |
| [49] | FER | St (4MBA) | Gold (Au)-coated ‘stellate’ mesoporous SiO2@Au nanoprobe | Hydrophilic Ag-deposited sandpaper assembled with hydrophobic-treated filter paper (coffee-ring-free hydrophilic–hydrophobic substrate) | 1 × 10−5 g mL−1–3 × 10−13 g mL−1 | 3.16 × 10−14 g mL−1 |
| [88] | MMP-9 | St (DTNB) | AgNPs | Fe3O4 microspheres (magnetic NPs) | 0 ng mL−1–100 ng mL−1 | 1 pg mL−1 |
| [59] | CA19–9 | St (MBA) | Immunoprobe of anti-CA19-9/4-MBA | Au nanowires (NWs) onto Fe3O4@TiO2 matrix | 1000 IU mL−1–0.001 IU mL−1 | 5.65 × 10−4 IU mL−1 |
| [72] | CTCs | St (GO) | Gold–graphene hybrid nanotag (Au–rGO)/gold-reduced graphene oxide nanosystem) | Polycarbonate filter | 1 cell mL−1–100 cell mL−1 | 1 cell mL−1 |
| [42] | ANG, AFP (2) | St (MGITC) | HGNs | Gold-patterned microarray chip | 0 g mL−1–10−4 g mL−1 | 0.1 pg mL−1 (ANG), 1.0 pg mL−1 (AFP) |
| [73] | CEA, AFP (2) | St (MGITC, XRITC) | HGNs | MB | - | - |
| [92] | CEA, AFP (2) | St (MBA) | AgNPs | 3D ordered silver nanoshell silica photonic crystal beads (Ag-SPCB) | 0.01 pg mL−1–1000 ng mL−1 (CEA), 0.1 pg mL−1–1000 ng mL−1 (AFP) | 6.6 × 10−6 ng mL−1 (CEA), 7.2 × 10−5 ng mL−1 (AFP) |
| [31] | CEA, AFP (2) | St (MB, TMB) | AuNPs | Gold microelectrode array (GMA) | 0.01 ng mL−1–20 ng mL−1 (CEA), 0.02 ng mL−1 0–5 ng mL−1 (AFP) | 0.3 pg mL−1 (CEA), 0.6 pg mL−1 (AFP) |
| [46] | CEA, AFP (2) | St (MBA, DTNB) | AuNSs | Ordered gold nanohoneycomb arrays | 0.5 ng mL−1–100 ng mL−1 | 0.41 (CEA), 0.35 ng mL−1 (AFP) |
| [32] | CEA, CK-19 (2) | St (THI, NBA) | AuNP-coated acid-based resin (AAR) microspheres | Electrode-modified chitosan-stabilized AuNPs | 0.05 ng mL−1–80 ng mL−1 | 0.01 ng mL−1 (CEA), 0.04 ng mL−1 (CK-19) |
| [45] | CA 19-9, MMP-7 (2) | St (DSNB) | AuNPs | Array of exposed gold ‘wells’ | - | 2.28 pg mL−1 (MMP-7), 34.5 pg mL−1 (CA 19-9) |
| [65] | PSA, Rac (2) | St (MBA) | Aggregated AgNPs | 96-Well polystyrene plates | - | 10−9 ng mL−1 (PSA), 10−6 (Rac) ng mL−1 |
| [86] | CEA, NSE (2) | St (MBA, DTNB) | Flowerlike gold NPs | Gold-coated magnetic nanoparticles | 10 pg mL−1–100 ng mL−1 | 1.48 pg mL−1 (CEA), 2.04 pg mL−1 (NSE) |
| [53] | PSA, AFP (2) | St (NTP, MBA) | AgNPs coated on SiO2 nanospheres (SiO2@Ag) | Gold-film hemisphere array (Au-FHA) immune substrate | 10 fg mL−1–400 ng mL−1 | 3.38 (PSA), 4.87 (AFP) fg mL−1 |
| [79] | CD19, CD20) (2) | St (MBA, (DNTB) | AgNPs | MB | 5000 cells mL−1–5 cells mL−1 | 5 cells mL−1 |
| [38] | AFP, AFP-L3 (2) | Dt (MBA, DSNB) | AuNPs with DSNB | Silicon chips coated with Ag (Si/Ag/MBA) | 0.5 ng mL−1–1000 ng mL−1 | 0.5 ng mL−1 |
| [82] | VEGF, IL-8 (2) | St (ABP, AAD) | AgNPs | MB | 1.0 fg mL−1–1 ng mL−1 | 1.0 fg mL−1 |
| [68] | SCCA, CA125 (2) | St (ATP, DTNB) | Nano-Ag polydopamine nanospheres (PDA@Ag-NPs) | Nitrocellulose (NC) membrane | 10 pg mL−1–10 µg mL−1 | 7.156 pg mL−1 (SCCA), 7.182 pg mL−1 (CA125) |
| [51] | SCCA, OPN (2) | Dt (MBA, DTNB, DMSA) | Au–Ag nanoshuttles (Au–AgNSs) | Hydrophobic filter-paper-based Au nanoflowers (AuNFs) | 10 pg mL−1–10 µg mL−1 | 8.628 pg/mL (SCCA), 4.388 pg/mL (OPN) |
| [29] | SCCA, survivin (2) | St (DTNB, ATP) | Au–Ag nanoshells (Au–AgNSs) | Au–Ag nanobox (Au-AgNB) array substrate | 10 pg mL−1–10 µg mL−1 | 6 pg mL−1 (SCCA), 5 pg mL−1 (survivin) |
| [74] | CEA, AFP, CA 125 (3) | St (3-MeOBT, 2-MeOBT, 2-NT)) | Nanotags with hybrid multilayered nanoshells prepared using layer-by-layer (LBL) assembly of small silver nanoparticles (AgNPs) at the surface of silica (SiO2) particles using poly(ethyleneimine) (PEI) | MB | 0.1 ng mL−1–1 ng mL−1 | 0.1 pg mL−1 |
| [69] | CA 15-3, CA 27-29, CEA (3) | St (NTP) | AuNSs | Quartz chip with punched wells | 0.1 ng mL−1–500 ng mL−1 | 0.99 U mL−1 (CA 15-3), 0.13 U mL−1 (CA 27-29), 0.05 ng mL−1 (CEA) |
| [48] | PSA, AFP, CA19-9 (3) | St (MBA) | SiO2-coated Si nanoparticles | SiC@Ag substrate (Ag film sputtered on SiC sandpaper) | 0–5 mg mL−1 (PSA, AFP), 0–3 mg mL−1 (CA19-9) | 1.79 fg mL−1 (PSA), 0.46 fg mL−1 (AFP), 1.3 × 10−3 U mL−1 (CA19-9) |
| [40] | PSA, PSMA, hK2 (3) | St (MBA) | AgNPs | SiC@Ag@Ag-NPs substrates | 10−5–101 ng mL−1 | 0.46 fg mL−1 (PSA), 1.05 fg mL−1 (PSMA), 0.67 fg mL−1 (hK2) |
| [80] | AFP, CEA, FER (3) | St (OPE0, OPE2, MBN) | AuNPs | MB | 0.5 pg mL−1–500 pg mL−1 (AFP), 50 pg mL−1–2000 pg mL−1 (CEA), 10 pg mL−1–200 pg mL−1 (FER) | 0.15 pg mL−1 (AFP), 20 pg mL−1 (CEA), 4 pg mL−1 (FER) |
| [91] | PSA, CEA, CA 19-9 (3) | St (MBA) | AuNPs | 2D arrays of gold core−silver shell nanoparticles (Au@Ag core–shell NPs) | 1 ng mL−1–1 pg mL−1 (PSA, CEA), 10–40 unit (U) mL−1 (CA19-9) | 1 pg mL−1 (PSA, CEA), 10 unit (U) mL−1 (CA 19-9) |
| [83] | Glypican-1, EpCAMs), CD44V6 (3) | St (DTNB, MBA, TFMBA) | AuNPs | MB | 0–2.3 × 108 particles mL−1 | 2.3 × 106 particles mL−1 |
| [28] | IL-6, IL-8, IL-18 (3) | St (DTNB, FC, MBA) | AuNPs | Ag–Au substrate | 0 ng mL−1–30 ng mL−1 | 2.3 pg mL−1, 6.5 pg mL−1, 4.2 pg mL−1 in a parallel, and 3.8 pg mL−1, 7.5 pg mL−1, 5.2 pg mL−1 in a simultaneous method for IL-6, IL-8 and IL-18, respectively |
| [95] | PSA, AFP, CEA, NSA (4) | St (MBA) | AuNPs | Gold substrate modified by Au–S bond (Au–SNBs) | 1 ng mL−1–100 ng mL−1 | 10−12 mol mL−1 |
| [81] | TNF-α, IFN-γ, IL-10, IL-6, IL-8 (5) | St (MBA, DTNB, TFMBA) | AuNPs with silver layers | MB | 0 pg mL−1–105 pg mL−1 (TNF-α) | 4.5 pg mL−1 (TNF-α) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pollap, A.; Świt, P. Recent Advances in Sandwich SERS Immunosensors for Cancer Detection. Int. J. Mol. Sci. 2022, 23, 4740. https://doi.org/10.3390/ijms23094740
Pollap A, Świt P. Recent Advances in Sandwich SERS Immunosensors for Cancer Detection. International Journal of Molecular Sciences. 2022; 23(9):4740. https://doi.org/10.3390/ijms23094740
Chicago/Turabian StylePollap, Aleksandra, and Paweł Świt. 2022. "Recent Advances in Sandwich SERS Immunosensors for Cancer Detection" International Journal of Molecular Sciences 23, no. 9: 4740. https://doi.org/10.3390/ijms23094740






