Functional Screen for microRNAs Suppressing Anchorage-Independent Growth in Human Cervical Cancer Cells
Abstract
:1. Introduction
2. Results
2.1. Functional Screen for miRNA Suppressors of Anchorage-Independent Growth
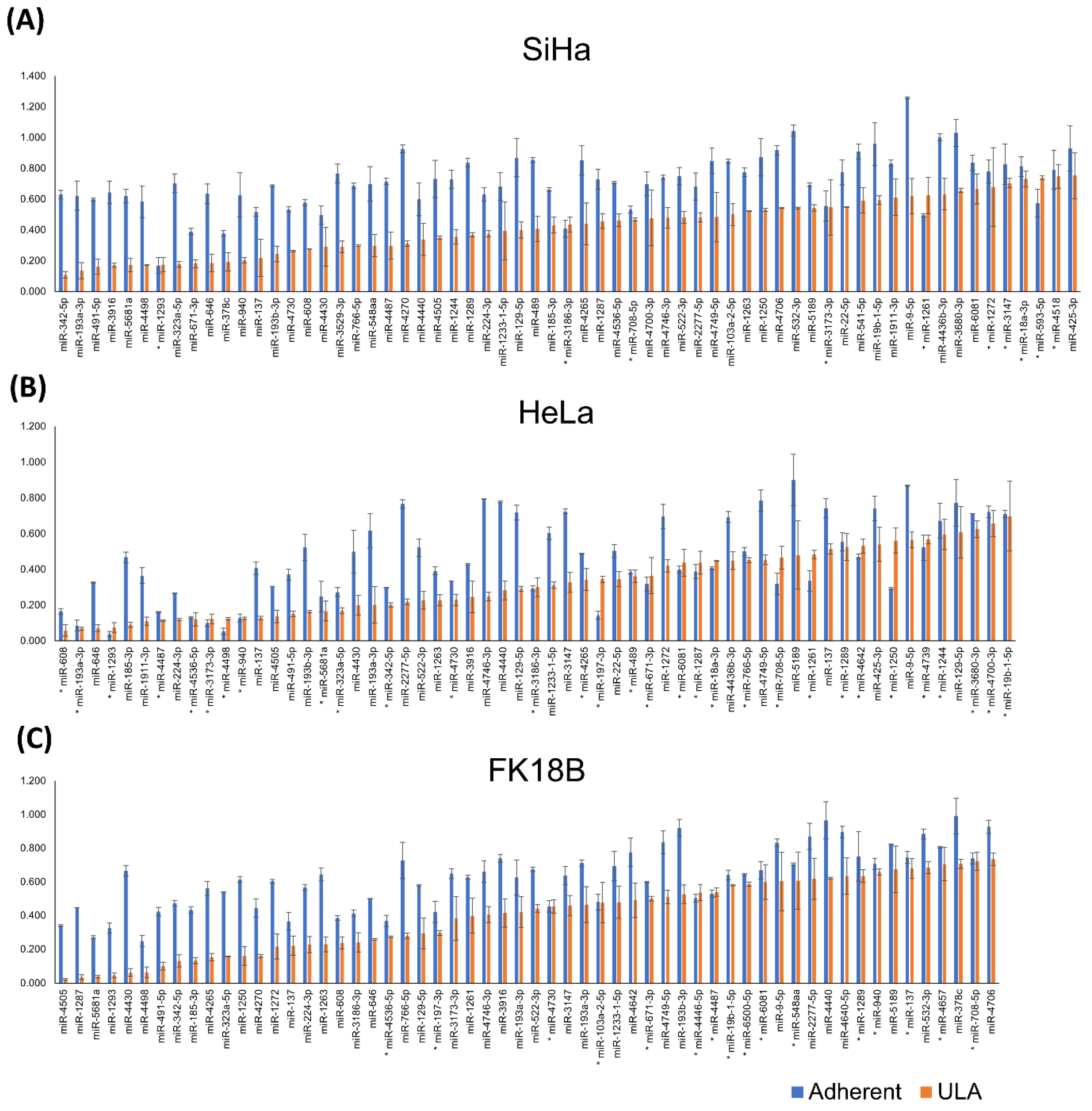
2.2. Independent Validation and Selection of miRNAs Specifically Reducing Anchorage-Independent Growth
2.3. Review of the Literature on Validated miRNAs
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Growth Comparison between ULA Plates and Soft Agar Assay upon miRNA Mimic Transfection
4.3. High-Throughput miRNA Mimic Screen
4.4. Screen Data Processing and Normalization
4.5. Validation of miRNA Mimic Candidates
4.5.1. Transfection Protocol
4.5.2. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Guadamillas, M.C.; Cerezo, A.; Del Pozo, M.A. Overcoming Anoikis—Pathways to Anchorage-Independent Growth in Cancer. J. Cell Sci. 2011, 124, 3189–3197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paoli, P.; Giannoni, E.; Chiarugi, P. Anoikis Molecular Pathways and Its Role in Cancer Progression. Biochim. Biophys. Acta-Mol. Cell Res. 2013, 1833, 3481–3498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilting, S.M.; Steenbergen, R.D.M. Molecular Events Leading to HPV-Induced High Grade Neoplasia. Papillomavirus Res. 2016, 2, 85–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steenbergen, R.D.M.; Snijders, P.J.F.; Heideman, D.A.M.; Meijer, C.J.L.M. Clinical Implications of (Epi)Genetic Changes in HPV-Induced Cervical Precancerous Lesions. Nat. Rev. Cancer 2014, 14, 395–405. [Google Scholar] [CrossRef]
- Bierkens, M.; Wilting, S.M.; Van Wieringen, W.N.; Van Kemenade, F.J.; Bleeker, M.C.G.; Jordanova, E.S.; Bekker-Lettink, M.; Van De Wiel, M.A.; Ylstra, B.; Meijer, C.J.L.M.; et al. Chromosomal Profiles of High-Grade Cervical Intraepithelial Neoplasia Relate to Duration of Preceding High-Risk Human Papillomavirus Infection. Int. J. Cancer 2012, 131, E579–E585. [Google Scholar] [CrossRef] [Green Version]
- Verlaat, W.; Van Leeuwen, R.W.; Novianti, P.W.; Schuuring, E.; Meijer, C.J.L.M.; Van Der Zee, A.G.J.; Snijders, P.J.F.; Heideman, D.A.M.; Steenbergen, R.D.M.; Wisman, G.B.A. Host-Cell DNA Methylation Patterns during High-Risk HPV-Induced Carcinogenesis Reveal a Heterogeneous Nature of Cervical Pre-Cancer. Epigenetics 2018, 13, 769–778. [Google Scholar] [CrossRef] [Green Version]
- Steenbergen, R.D.M.; Walboomers, J.M.M.; Meijer, C.J.L.M.; Van Der Raaij-Helmer, E.M.H.; Parker, J.N.; Chow, L.T.; Broker, T.R.; Snijders, P.J.F. Transition of Human Papillomavirus Type 16 and 18 Transfected Human Foreskin Keratinocytes towards Immortality: Activation of Telomerase and Allele Losses at 3p, 10p, 11q and/or 18q. Oncogene 1996, 13, 1249–1257. [Google Scholar]
- Wilting, S.M.; Miok, V.; Jaspers, A.; Boon, D.; Sørgård, H.; Lando, M.; Snoek, B.C.; van Wieringen, W.N.; Meijer, C.J.L.M.; Lyng, H.; et al. Aberrant Methylation-Mediated Silencing of MicroRNAs Contributes to HPV-Induced Anchorage Independence. Oncotarget 2016, 7, 43805–43819. [Google Scholar] [CrossRef] [Green Version]
- Babion, I.; Miok, V.; Jaspers, A.; Huseinovic, A.; Steenbergen, R.D.M.; van Wieringen, W.N.; Wilting, S.M. Identification of Deregulated Pathways, Key Regulators, and Novel MiRNA-MRNA Interactions in HPV-Mediated Transformation. Cancers 2020, 12, 700. [Google Scholar] [CrossRef] [Green Version]
- Medina, P.P.; Slack, F.J. MicroRNAs and Cancer: An Overview. Cell Cycle 2008, 7, 2485–2492. [Google Scholar] [CrossRef] [Green Version]
- Harden, M.E.; Prasad, N.; Griffiths, A.; Munger, K. Modulation of MicroRNA-MRNA Target Pairs by Human Papillomavirus 16 Oncoproteins. MBio 2017, 8, e02170-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honegger, A.; Schilling, D.; Bastian, S.; Sponagel, J.; Kuryshev, V.; Sültmann, H.; Scheffner, M.; Hoppe-Seyler, K.; Hoppe-Seyler, F. Dependence of Intracellular and Exosomal MicroRNAs on Viral E6/E7 Oncogene Expression in HPV-Positive Tumor Cells. PLoS Pathog. 2015, 11, e1004712. [Google Scholar] [CrossRef] [PubMed]
- Wilting, S.M.; Snijders, P.J.F.; Verlaat, W.; Jaspers, A.; van de Wiel, M.A.; van Wieringen, W.N.; Meijer, G.A.; Kenter, G.G.; Yi, Y.; le Sage, C.; et al. Altered MicroRNA Expression Associated with Chromosomal Changes Contributes to Cervical Carcinogenesis. Oncogene 2013, 32, 106–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wilting, S.M.; Verlaat, W.; Jaspers, A.; Makazaji, N.A.; Agami, R.; Meijer, C.J.L.M.; Snijders, P.J.F.; Steenbergen, R.D.M. Methylation-Mediated Transcriptional Repression of MicroRNAs during Cervical Carcinogenesis. Epigenetics 2013, 8, 220–228. [Google Scholar] [CrossRef] [Green Version]
- López, J.A.; Alvarez-Salas, L.M. Differential Effects of MiR-34c-3p and MiR-34c-5p on SiHa Cells Proliferation Apoptosis, Migration and Invasion. Biochem. Biophys. Res. Commun. 2011, 409, 513–519. [Google Scholar] [CrossRef]
- Leung, C.O.N.; Deng, W.; Ye, T.-M.; Ngan, H.Y.S.; Tsao, S.W.; Cheung, A.N.Y.; Pang, R.T.K.; Yeung, W.S.B. MiR-135a Leads to Cervical Cancer Cell Transformation through Regulation of β-Catenin via a SIAH1-Dependent Ubiquitin Proteosomal Pathway. Carcinogenesis 2014, 35, 1931–1940. [Google Scholar] [CrossRef] [Green Version]
- Qin, W.; Dong, P.; Ma, C.; Mitchelson, K.; Deng, T.; Zhang, L.; Sun, Y.; Feng, X.; Ding, Y.; Lu, X.; et al. MicroRNA-133b Is a Key Promoter of Cervical Carcinoma Development through the Activation of the ERK and AKT1 Pathways. Oncogene 2012, 31, 4067–4075. [Google Scholar] [CrossRef] [Green Version]
- Fan, Z.; Cui, H.; Xu, X.; Lin, Z.; Zhang, X.; Kang, L.; Han, B.; Meng, J.; Yan, Z.; Yan, X.; et al. MiR-125a Suppresses Tumor Growth, Invasion and Metastasis in Cervical Cancer by Targeting STAT3. Oncotarget 2015, 6, 25266–25280. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Ping, Z.; Ning, H. MiR-218 Impairs Tumor Growth and Increases Chemo-Sensitivity to Cisplatin in Cervical Cancer. Int. J. Mol. Sci. 2012, 13, 16053–16064. [Google Scholar] [CrossRef]
- Yang, Y.-K.; Xi, W.-Y.; Xi, R.-X.; Li, J.-Y.; Li, Q.; Gao, Y.-E. MicroRNA-494 Promotes Cervical Cancer Proliferation through the Regulation of PTEN. Oncol. Rep. 2015, 33, 2393–2401. [Google Scholar] [CrossRef]
- Babion, I.; Jaspers, A.; van Splunter, A.P.; van der Hoorn, I.A.E.; Wilting, S.M.; Steenbergen, R.D.M. MiR-9-5p Exerts a Dual Role in Cervical Cancer and Targets Transcription Factor TWIST1. Cells 2019, 9, 65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rotem, A.; Janzer, A.; Izar, B.; Ji, Z.; Doench, J.G.; Garraway, L.A.; Struhl, K. Alternative to the Soft-Agar Assay That Permits High-Throughput Drug and Genetic Screens for Cellular Transformation. Proc. Natl. Acad. Sci. USA 2015, 112, 5708–5713. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Izar, B.; Rotem, A. GILA, a Replacement for the Soft-Agar Assay That Permits High-Throughput Drug and Genetic Screens for Cellular Transformation. Curr. Protoc. Mol. Biol. 2016, 116, 28.8.1–28.8.12. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Geng, Y.; Huang, J.; Xi, D.; Xu, G.; Gu, W.; Shao, Y. CircNEIL3 Promotes Cervical Cancer Cell Proliferation by Adsorbing MiR-137 and Upregulating KLF12. Cancer Cell Int. 2021, 21, 34. [Google Scholar] [CrossRef]
- Miao, H.; Wang, N.; Shi, L.X.; Wang, Z.; Song, W.B. Overexpression of MircoRNA-137 Inhibits Cervical Cancer Cell Invasion, Migration and Epithelial-Mesenchymal Transition by Suppressing the TGF-β/Smad Pathway via Binding to GREM1. Cancer Cell Int. 2019, 19, 147. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Yan, T.; Liu, Z.; Wang, J.; Lu, Y.; Li, D.; Liang, W. MicroRNA-137 Is Negatively Associated with Clinical Outcome and Regulates Tumor Development through EZH2 in Cervical Cancer. J. Cell. Biochem. 2018, 119, 938–947. [Google Scholar] [CrossRef]
- Chen, G.; Zhang, M.; Zhu, J.; Chen, F.; Yu, D.; Zhang, A.; He, J.; Hua, W.; Duan, P. Common Genetic Variants in Pre-MicroRNAs Are Associated with Cervical Cancer Susceptibility in Southern Chinese Women. J. Cancer 2020, 11, 2133–2138. [Google Scholar] [CrossRef]
- Bai, M.; Che, Y.; Lu, K.; Fu, L. Analysis of Deubiquitinase OTUD5 as a Biomarker and Therapeutic Target for Cervical Cancer by Bioinformatic Analysis. PeerJ 2020, 8, e9146. [Google Scholar] [CrossRef]
- Luo, M.; Wu, L.; Zhang, K.; Wang, H.; Zhang, T.; Gutierrez, L.; O’Connell, D.; Zhang, P.; Li, Y.; Gao, T.; et al. MiR-137 Regulates Ferroptosis by Targeting Glutamine Transporter SLC1A5 in Melanoma. Cell Death Differ. 2018, 25, 1457–1472. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; Zou, Y.; Zheng, R.; Ma, X. MiR-137 Inhibits Cell Proliferation in Acute Lymphoblastic Leukemia by Targeting JARID1B. Eur. J. Haematol. 2019, 103, 215–224. [Google Scholar] [CrossRef]
- Duan, J.; Lu, G.; Li, Y.; Zhou, S.; Zhou, D.; Tao, H. MiR-137 Functions as a Tumor Suppressor Gene in Pituitary Adenoma by Targeting AKT2. Int. J. Clin. Exp. Pathol. 2019, 12, 1557–1564. [Google Scholar] [PubMed]
- Lei, C.; Jing, G.; Jichao, W.; Xiaohui, L.; Fang, Q.; Hua, G.; Yazhou, M.; Zhang, Y. MiR-137’s Tumor Suppression on Prolactinomas by Targeting MITF and Modulating Wnt Signaling Pathway. J. Clin. Endocrinol. Metab. 2019, 104, 6391–6402. [Google Scholar] [CrossRef] [PubMed]
- Ding, F.; Zhang, S.; Gao, S.; Shang, J.; Li, Y.; Cui, N.; Zhao, Q. MiR-137 Functions as a Tumor Suppressor in Pancreatic Cancer by Targeting MRGBP. J. Cell. Biochem. 2018, 119, 4799–4807. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Chen, J.H.; Shan, T.; Aguilera-Barrantes, I.; Wang, L.S.; Huang, T.H.M.; Rader, J.S.; Sheng, X.; Huang, Y.W. MiR-137 Is a Tumor Suppressor in Endometrial Cancer and Is Repressed by DNA Hypermethylation. Lab. Investig. 2018, 98, 1397–1407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bi, W.P.; Xia, M.; Wang, X.J. MiR-137 Suppresses Proliferation, Migration and Invasion of Colon Cancer Cell Lines by Targeting TCF4. Oncol. Lett. 2018, 15, 8744–8748. [Google Scholar] [CrossRef] [Green Version]
- Li, K.K.W.; Yang, L.; Pang, J.C.S.; Chan, A.K.Y.; Zhou, L.; Mao, Y.; Wang, Y.; Lau, K.M.; Poon, W.S.; Shi, Z.; et al. MIR-137 Suppresses Growth and Invasion, Is Downregulated in Oligodendroglial Tumors and Targets CSE1L. Brain Pathol. 2013, 23, 426–439. [Google Scholar] [CrossRef]
- Peres, J.; Kwesi-Maliepaard, E.M.; Rambow, F.; Larue, L.; Prince, S. The Tumour Suppressor, MiR-137, Inhibits Malignant Melanoma Migration by Targetting the TBX3 Transcription Factor. Cancer Lett. 2017, 405, 111–119. [Google Scholar] [CrossRef]
- Zeng, X.; Xu, Z.; Gu, J.; Huang, H.; Gao, G.; Zhang, X.; Li, J.; Jin, H.; Jiang, G.; Sun, H.; et al. Induction of MIR-137 by Isorhapontigenin (ISO) Directly Targets Sp1 Protein Translation and Mediates Its Anticancer Activity Both in Vitro and in Vivo. Mol. Cancer Ther. 2016, 15, 512–522. [Google Scholar] [CrossRef] [Green Version]
- Zhu, M.; Li, M.; Wang, T.; Linghu, E.; Wu, B. MicroRNA-137 Represses FBI-1 to Inhibit Proliferation and in Vitro Invasion and Migration of Hepatocellular Carcinoma Cells. Tumor Biol. 2016, 37, 13995–14008. [Google Scholar] [CrossRef]
- Li, L.; He, Z.; Zhu, C.; Chen, S.; Yang, Z.; Xu, J.; Bi, N.; Yu, C.; Sun, C. MiR-137 Promotes Anoikis through Modulating the AKT Signaling Pathways in Pancreatic Cancer. J. Cancer 2020, 11, 6277–6285. [Google Scholar] [CrossRef]
- Xu, Y.; Zhu, H.; Ma, H.; Yuan, L.; Hu, Q.; Yang, L. LINC01305 Inhibits Malignant Progression of Cervical Cancer via MiR-129-5p/Sox4 Axis. Am. J. Transl. Res. 2020, 12, 7581–7592. [Google Scholar] [PubMed]
- Wang, Y.-F.; Yang, H.-Y.; Shi, X.-Q.; Wang, Y. Upregulation of MicroRNA-129-5p Inhibits Cell Invasion, Migration and Tumor Angiogenesis by Inhibiting ZIC2 via Downregulation of the Hedgehog Signaling Pathway in Cervical Cancer. Cancer Biol. Ther. 2018, 19, 1162–1173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Li, S.; Yan, Q.; Chen, X.; Yang, Y.; Liu, X.; Wan, X. Interferon-β Induced MicroRNA-129-5p down-Regulates HPV-18 E6 and E7 Viral Gene Expression by Targeting SP1 in Cervical Cancer Cells. PLoS ONE 2013, 8, e81366. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yu, J. MiR-129-5p Suppresses Gastric Cancer Cell Invasion and Proliferation by Inhibiting COL1A1. Biochem. Cell Biol. 2018, 96, 19–25. [Google Scholar] [CrossRef]
- Yan, L.; Sun, K.; Liu, Y.; Liang, J.; Cai, K.; Gui, J. MiR-129-5p Influences the Progression of Gastric Cancer Cells through Interacting with SPOCK1. Tumour Biol. 2017, 39, 1010428317706916. [Google Scholar] [CrossRef] [Green Version]
- Gao, G.; Xiu, D.; Yang, B.; Sun, D.; Wei, X.; Ding, Y.; Ma, Y.; Wang, Z. MiR-129-5p Inhibits Prostate Cancer Proliferation via Targeting ETV1. Onco. Targets. Ther. 2019, 12, 3531–3544. [Google Scholar] [CrossRef] [Green Version]
- Xu, C.; Du, Z.; Ren, S.; Liang, X.; Li, H. MiR-129-5p Sensitization of Lung Cancer Cells to Etoposide-Induced Apoptosis by Reducing YWHAB. J. Cancer 2020, 11, 858–866. [Google Scholar] [CrossRef] [Green Version]
- Li, G.; Xie, J.; Wang, J. Tumor Suppressor Function of Mir-129-5p in Lung Cancer. Oncol. Lett. 2019, 17, 5777–5783. [Google Scholar] [CrossRef] [Green Version]
- Wu, Q.; Meng, W.Y.; Jie, Y.; Zhao, H. LncRNA MALAT1 Induces Colon Cancer Development by Regulating MiR-129-5p/HMGB1 Axis. J. Cell. Physiol. 2018, 233, 6750–6757. [Google Scholar] [CrossRef]
- Meng, R.; Fang, J.; Yu, Y.; Hou, L.K.; Chi, J.R.; Chen, A.X.; Zhao, Y.; Cao, X.C. MiR-129-5p Suppresses Breast Cancer Proliferation by Targeting CBX4. Neoplasma 2018, 65, 572–578. [Google Scholar] [CrossRef] [Green Version]
- Qiu, Z.; Wang, X.; Shi, Y.; Da, M. MIR-129-5p Suppresses Proliferation, Migration, and Induces Apoptosis in Pancreatic Cancer Cells by Targeting PBX3. Acta Biochim. Biophys. Sin. 2019, 51, 997–1007. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Lu, J.; Zeng, G.; Pang, J.; Zheng, X.; Feng, J.; Zhang, J. MiR-129-5p Inhibits Liver Cancer Growth by Targeting Calcium Calmodulin-Dependent Protein Kinase IV (CAMK4). Cell Death Dis. 2019, 10, 789. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cao, J.; Wang, Q.; Wu, G.; Li, S.; Wang, Q. MiR-129-5p Inhibits Gemcitabine Resistance and Promotes Cell Apoptosis of Bladder Cancer Cells by Targeting Wnt5a. Int. Urol. Nephrol. 2018, 50, 1811–1819. [Google Scholar] [CrossRef] [PubMed]
- Lei, H.W.; Gao, Y.; Shi, J.B.; Teng, Y.; Song, C.H.; Zou, L.J.; Ye, F.X.; Zhang, H.C. KCNQ1 Opposite Strand/Antisense Transcript 1 Promotes Aggressive Biological Behaviors of Cervical Cancer Cells via Regulating MicroRNA-491-5p and Pyruvate Kinase M1/2. J. Biol. Regul. Homeost. Agents 2020, 34. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhai, Y.X.; Liu, H.Q.; Shi, Y.A.; Li, X.B. MicroRNA-491-5p Suppresses Cervical Cancer Cell Growth by Targeting HTERT. Oncol. Rep. 2015, 34, 979–986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Y.; Wang, Y.; Xie, Z.; Hu, H. JMJD2A Facilitates Growth and Inhibits Apoptosis of Cervical Cancer Cells by Downregulating Tumor Suppressor MiR-491-5p. Mol. Med. Rep. 2019, 19, 2489–2496. [Google Scholar] [CrossRef]
- Lu, L.; Cai, M.; Peng, M.; Wang, F.; Zhai, X. MiR-491-5p Functions as a Tumor Suppressor by Targeting IGF2 in Colorectal Cancer. Cancer Manag. Res. 2019, 11, 1805–1816. [Google Scholar] [CrossRef] [Green Version]
- Chen, T.; Li, Y.; Cao, W.; Liu, Y. MiR-491-5p Inhibits Osteosarcoma Cell Proliferation by Targeting PKM2. Oncol. Lett. 2018, 16, 6472–6478. [Google Scholar] [CrossRef] [Green Version]
- Yu, T.; Wang, L.N.; Li, W.; Zuo, Q.F.; Li, M.M.; Zou, Q.M.; Xiao, B. Downregulation of MiR-491-5p Promotes Gastric Cancer Metastasis by Regulating SNAIL and FGFR4. Cancer Sci. 2018, 109, 1393–1403. [Google Scholar] [CrossRef] [Green Version]
- Han, D.; Wang, J.; Cheng, G. LncRNA NEAT1 Enhances the Radio-Resistance of Cervical Cancer via MiR-193b-3p/CCND1 Axis. Oncotarget 2018, 9, 2395–2409. [Google Scholar] [CrossRef] [Green Version]
- Choi, K.H.; Shin, C.H.; Lee, W.J.; Ji, H.; Kim, H.H. Dual-Strand Tumor Suppressor MiR-193b-3p and -5p Inhibit Malignant Phenotypes of Lung Cancer by Suppressing Their Common Targets. Biosci. Rep. 2019, 39, BSR20190634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, J.; Qin, J.; Su, Y. MiR-193b-3p Possesses Anti-Tumor Activity in Ovarian Carcinoma Cells by Targeting P21-Activated Kinase 3. Biomed. Pharmacother. 2017, 96, 1275–1282. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, W.; Yang, P.; Zhou, J.; Wang, K.; Tao, Q. Knockdown of Linc00152 Inhibits the Progression of Gastric Cancer by Regulating MicroRNA-193b-3p/ETS1 Axis. Cancer Biol. Ther. 2019, 20, 461–473. [Google Scholar] [CrossRef] [Green Version]
- Bhayadia, R.; Krowiorz, K.; Haetscher, N.; Jammal, R.; Emmrich, S.; Obulkasim, A.; Fiedler, J.; Schwarzer, A.; Rouhi, A.; Heuser, M.; et al. Endogenous Tumor Suppressor MicroRNA-193b: Therapeutic and Prognostic Value in Acute Myeloid Leukemia. J. Clin. Oncol. 2018, 36, 1007–1016. [Google Scholar] [CrossRef] [PubMed]
- Laengsri, V.; Kerdpin, U.; Plabplueng, C.; Treeratanapiboon, L.; Nuchnoi, P. Cervical Cancer Markers: Epigenetics and MicroRNAs. Lab Med. 2018, 49, 97–111. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Cai, J.; Xu, Z.; Zhou, S.; Ye, L.; Yan, Q.; Zhang, Y.; Fang, Y.; Liu, Y.; Tu, C.; et al. MiR-532-3p Suppresses Colorectal Cancer Progression by Disrupting the ETS1/TGM2 Axis-Mediated Wnt/β-Catenin Signaling. Cell Death Dis. 2019, 10, 739. [Google Scholar] [CrossRef]
- Jiang, W.; Zheng, L.; Yan, Q.; Chen, L.; Wang, X. MiR-532-3p Inhibits Metastasis and Proliferation of Non-Small Cell Lung Cancer by Targeting FOXP3. JBUON 2019, 24, 2287–2293. [Google Scholar] [PubMed]
- Liu, Y.; Li, Q.; Dai, Y.; Jiang, T.; Zhou, Y. MiR-532-3p Inhibits Proliferation and Promotes Apoptosis of Lymphoma Cells by Targeting β-Catenin. J. Cancer 2020, 11, 4762–4770. [Google Scholar] [CrossRef]
- Wa, Q.; Zou, C.; Lin, Z.; Huang, S.; Peng, X.; Yang, C.; Ren, D.; Xu, D.; Guo, Y.; Liao, Z.; et al. Ectopic Expression of MiR-532-3p Suppresses Bone Metastasis of Prostate Cancer Cells via Inactivating NF-ΚB Signaling. Mol. Ther.-Oncolytics 2020, 17, 267–277. [Google Scholar] [CrossRef]
- Feng, C.; So, H.I.; Yin, S.; Su, X.; Xu, Q.; Wang, S.; Duan, W.; Zhang, E.; Sun, C.; Xu, Z. MicroRNA-532-3p Suppresses Malignant Behaviors of Tongue Squamous Cell Carcinoma via Regulating CCR7. Front. Pharmacol. 2019, 10, 940. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Luo, C.; Dong, J.; Guo, J.; Luo, Q.; Ye, C.; Guo, Z. CircRNA_103809 Suppresses the Proliferation and Metastasis of Breast Cancer Cells by Sponging MicroRNA-532-3p (MiR-532-3p). Front. Genet. 2020, 11, 485. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.T.; Zhao, Y.N.; Yan, J.X.; Weng, M.Y.; Wang, Y.; Chen, Y.Q.; Hong, S.J. Differentially Expressed MicroRNAs in the Serum of Cervical Squamous Cell Carcinoma Patients before and after Surgery. J. Hematol. Oncol. 2014, 7, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darvishi, N.; Rahimi, K.; Mansouri, K.; Fathi, F.; Menbari, M.N.; Mohammadi, G.; Abdi, M. MiR-646 Prevents Proliferation and Progression of Human Breast Cancer Cell Lines by Suppressing HDAC2 Expression. Mol. Cell. Probes 2020, 53, 101649. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Shu, H.; Guo, S. MiR-646 Suppresses Proliferation and Metastasis of Non-Small Cell Lung Cancer by Repressing FGF2 and CCND2. Cancer Med. 2020, 9, 4360–4370. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Tang, W.M.; Zhang, H.; Li, Y.Q.; Peng, Y.; Wang, J.; Liu, G.N.; Huang, X.T.; Zhao, J.J.; Li, G.; et al. MiR-646 Inhibited Cell Proliferation and EMT-Induced Metastasis by Targeting FOXK1 in Gastric Cancer. Br. J. Cancer 2017, 117, 525–534. [Google Scholar] [CrossRef]
- Zhang, L.; Guo, C.; Ji, T.; Chen, X. SOX2 Regulates LncRNA CCAT1/MicroRNA-185-3p/FOXP3 Axis to Affect the Proliferation and Self-Renewal of Cervical Cancer Stem Cells. Nanoscale Res. Lett. 2021, 16, 2. [Google Scholar] [CrossRef] [PubMed]
- Deng, Z.H.; Yu, G.S.; Deng, K.L.; Feng, Z.H.; Huang, Q.; Pan, B.; Deng, J.Z. Hsa_circ_0088233 Alleviates Proliferation, Migration, and Invasion of Prostate Cancer by Targeting Hsa-MiR-185-3p. Front. Cell Dev. Biol. 2020, 8, 528155. [Google Scholar] [CrossRef]
- Zhou, C.; Kong, W.; Ju, T.; Xie, Q.; Zhai, L. MiR-185-3p Mimic Promotes the Chemosensitivity of CRC Cells via AQP5. Cancer Biol. Ther. 2020, 21, 790–798. [Google Scholar] [CrossRef]
- Xu, J.; Chen, G.; Zhang, Y.; Huang, Z.; Cheng, X.; Gu, H.; Xia, J.; Yin, X.; Huang, T. LINC00511 Promotes Osteosarcoma Tumorigenesis and Invasiveness through the MiR-185-3p/E2F1 Axis. Biomed Res. Int. 2020, 2020, 1974506. [Google Scholar] [CrossRef]
- Lu, G.; Li, Y.; Ma, Y.; Lu, J.; Chen, Y.; Jiang, Q.; Qin, Q.; Zhao, L.; Huang, Q.; Luo, Z.; et al. Long Noncoding RNA LINC00511 Contributes to Breast Cancer Tumourigenesis and Stemness by Inducing the MiR-185-3p/E2F1/Nanog Axis. J. Exp. Clin. Cancer Res. 2018, 37, 289. [Google Scholar] [CrossRef]
- Xu, J.; Ai, Q.; Cao, H.; Liu, Q. MiR-185-3p and MiR-324-3p Predict Radiosensitivity of Nasopharyngeal Carcinoma and Modulate Cancer Cell Growth and Apoptosis by Targeting SMAD7. Med. Sci. Monit. 2015, 21, 2828–2836. [Google Scholar] [CrossRef] [PubMed]
- Ji, F.; Du, R.; Chen, T.; Zhang, M.; Zhu, Y.; Luo, X.; Ding, Y. Circular RNA CircSLC26A4 Accelerates Cervical Cancer Progression via MiR-1287-5p/HOXA7 Axis. Mol. Ther.-Nucleic Acids 2020, 19, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Yao, T.; Rao, Q.; Liu, L.; Zheng, C.; Xie, Q.; Liang, J.; Lin, Z. Exploration of Tumor-Suppressive MicroRNAs Silenced by DNA Hypermethylation in Cervical Cancer. Virol. J. 2013, 10, 175. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Chen, M.; Tao, Z.; Hua, Q.; Chen, S.; Xiao, B. Identification of Predictive Biomarkers for Early Diagnosis of Larynx Carcinoma Based on MicroRNA Expression Data. Cancer Genet. 2013, 206, 340–346. [Google Scholar] [CrossRef]
- Schwarzenbacher, D.; Klec, C.; Pasculli, B.; Cerk, S.; Rinner, B.; Karbiener, M.; Ivan, C.; Barbano, R.; Ling, H.; Wulf-Goldenberg, A.; et al. MiR-1287-5p Inhibits Triple Negative Breast Cancer Growth by Interaction with Phosphoinositide 3-Kinase CB, Thereby Sensitizing Cells for PI3Kinase Inhibitors. Breast Cancer Res. 2019, 21, 20. [Google Scholar] [CrossRef] [Green Version]
- Chang, H.; Qu, J.; Wang, J.; Liang, X.; Sun, W. Circular RNA Circ_0026134 Regulates Non-Small Cell Lung Cancer Cell Proliferation and Invasion via Sponging MiR-1256 and MiR-1287. Biomed. Pharmacother. 2019, 112, 108743. [Google Scholar] [CrossRef]
- Lu, J.; Tang, L.; Xu, Y.; Ge, K.; Huang, J.; Gu, M.; Zhong, J.; Huang, Q. Mir-1287 Suppresses the Proliferation, Invasion, and Migration in Hepatocellular Carcinoma by Targeting PIK3R3. J. Cell. Biochem. 2018, 119, 9229–9238. [Google Scholar] [CrossRef]
- Li, Y.; Hu, J.; Li, L.; Cai, S.; Zhang, H.; Zhu, X.; Guan, G.; Dong, X. Upregulated Circular RNA Circ_0016760 Indicates Unfavorable Prognosis in NSCLC and Promotes Cell Progression through MiR-1287/GAGE1 Axis. Biochem. Biophys. Res. Commun. 2018, 503, 2089–2094. [Google Scholar] [CrossRef]
- Cui, G.; Zhao, H.L.; Li, L. Long Noncoding RNA PRKCQ-AS1 Promotes CRC Cell Proliferation and Migration via Modulating MiR-1287-5p/YBX1 Axis. J. Cell. Biochem. 2020, 121, 4166–4175. [Google Scholar] [CrossRef]
- Zhang, X.; Xue, C.; Cui, X.; Zhou, Z.; Fu, Y.; Yin, X.; Wu, S.; Gong, Y.; Liu, Y.; Zhu, C.; et al. Circ_0075829 Facilitates the Progression of Pancreatic Carcinoma by Sponging MiR-1287-5p and Activating LAMTOR3 Signalling. J. Cell. Mol. Med. 2020, 24, 14596–14607. [Google Scholar] [CrossRef]
- Wongjampa, W.; Ekalaksananan, T.; Chopjitt, P.; Chuerduangphui, J.; Kleebkaow, P.; Patarapadungkit, N.; Pientong, C. Suppression of MiR-22, a Tumor Suppressor in Cervical Cancer, by Human Papillomavirus 16 E6 via a P53/MiR-22/HDAC6 Pathway. PLoS ONE 2018, 13, e0206644. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Hayashi, M.; Konishi, H.; Nunode, M.; Ashihara, K.; Sasaki, H.; Terai, Y.; Ohmichi, M. MicroRNA-22 Enhances Radiosensitivity in Cervical Cancer Cell Lines via Direct Inhibition of c-Myc Binding Protein, and the Subsequent Reduction in HTERT Expression. Oncol. Lett. 2020, 19, 2213–2222. [Google Scholar] [CrossRef] [PubMed]
- Xin, M.; Qiao, Z.; Li, J.; Liu, J.; Song, S.; Zhao, X.; Miao, P.; Tang, T.; Wang, L.; Liu, W.; et al. MiR-22 Inhibits Tumor Growth and Metastasis by Targeting ATP Citrate Lyase: Evidence in Osteosarcoma, Prostate Cancer, Cervical Cancer and Lung Cancer. Oncotarget 2016, 7, 44252–44265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, C.; Zhang, W.; Wu, Q.; Liu, Y.; Wang, C.; Lao, G.; Yang, L.; Liu, P. Identification of a MicroRNA Signature Associated with Survivability in Cervical Squamous Cell Carcinoma. PLoS ONE 2018, 13, e0193625. [Google Scholar] [CrossRef]
- Zhang, L.; Zou, L.; Sun, P. Relationship between MiR-378c and YY1 Expression in Patients with Gastric Cancer and the Clinicopathological Features. Cell. Mol. Biol. Lett. 2021, 26, 12. [Google Scholar] [CrossRef]
- Yu, Q.; Zheng, B.; Ji, X.; Li, P.; Guo, Z. MiR-378c Suppresses Wilms Tumor Development via Negatively Regulating CAMKK2. Acta Biochim. Biophys. Sin. 2021, 53, 739–747. [Google Scholar] [CrossRef]
- Piotrowski, I.; Zhu, X.; Saccon, T.D.; Ashiqueali, S.; Schneider, A.; de Carvalho Nunes, A.D.; Noureddine, S.; Sobecka, A.; Barczak, W.; Szewczyk, M.; et al. MiRNAs as Biomarkers for Diagnosing and Predicting Survival of Head and Neck Squamous Cell Carcinoma Patients. Cancers 2021, 13, 3980. [Google Scholar] [CrossRef]
- Liang, H.; Liu, M.; Yan, X.; Zhou, Y.; Wang, W.; Wang, X.; Fu, Z.; Wang, N.; Zhang, S.; Wang, Y.; et al. MiR-193a-3p Functions as a Tumor Suppressor in Lung Cancer by down-Regulating ERBB4. J. Biol. Chem. 2015, 290, 926–940. [Google Scholar] [CrossRef] [Green Version]
- Yu, T.; Li, J.; Yan, M.; Liu, L.; Lin, H.; Zhao, F.; Sun, L.; Zhang, Y.; Cui, Y.; Zhang, F.; et al. MicroRNA-193a-3p and -5p Suppress the Metastasis of Human Non-Small-Cell Lung Cancer by Downregulating the ERBB4/PIK3R3/MTOR/S6K2 Signaling Pathway. Oncogene 2015, 34, 413–423. [Google Scholar] [CrossRef]
- Fan, Q.; Hu, X.; Zhang, H.; Wang, S.; Zhang, H.; You, C.; Zhang, C.Y.; Liang, H.; Chen, X.; Ba, Y. MiR-193a-3p Is an Important Tumour Suppressor in Lung Cancer and Directly Targets KRAS. Cell. Physiol. Biochem. 2017, 44, 1311–1324. [Google Scholar] [CrossRef]
- Yi, Y.; Chen, J.; Jiao, C.; Zhong, J.; Song, Z.; Yu, X.; Lu, X.; Lin, B. Upregulated MiR-193a-3p as an Oncogene in Esophageal Squamous Cell Carcinoma Regulating Cellular Proliferation, Migration and Apoptosis. Oncol. Lett. 2016, 12, 4779–4784. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Huang, Z.G.; Wu, H.Y.; He, R.Q.; Yang, L.H.; Feng, Z.B.; Dang, Y.W.; Lu, H.P.; Fang, Y.Y.; Chen, G. Downregulation of MiR-193a-3p Is Involved in the Pathogenesis of Hepatocellular Carcinoma by Targeting CCND1. PeerJ 2020, 8, e8409. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Li, Y.; Liu, S.; Duan, Q.; Chen, L.; Wu, T.; Qian, H.; Yang, S.; Xin, D. Downregulation of MiR-193a-3p Inhibits Cell Growth and Migration in Renal Cell Carcinoma by Targeting PTEN. Tumor Biol. 2017, 39, 1010428317711951. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pu, Y.; Zhao, F.; Cai, W.; Meng, X.; Li, Y.; Cai, S. MiR-193a-3p and MiR-193a-5p Suppress the Metastasis of Human Osteosarcoma Cells by down-Regulating Rab27B and SRR, Respectively. Clin. Exp. Metastasis 2016, 33, 359–372. [Google Scholar] [CrossRef] [Green Version]
- Pekow, J.; Meckel, K.; Dougherty, U.; Huang, Y.; Chen, X.; Almoghrabi, A.; Mustafi, R.; Ayaloglu-Butun, F.; Deng, Z.; Haider, H.I.; et al. MiR-193a-3p Is a Key Tumor Suppressor in Ulcerative Colitis-Associated Colon Cancer and Promotes Carcinogenesis through Upregulation of IL17RD. Clin. Cancer Res. 2017, 23, 5281–5291. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Fang, J.; Chen, S.; Wang, W.; Meng, S.; Liu, B. Nonconserved MiR-608 Suppresses Prostate Cancer Progression through RAC2/PAK4/LIMK1 and BCL2L1/Caspase-3 Pathways by Targeting the 3′-UTRs of RAC2/BCL2L1 and the Coding Region of PAK4. Cancer Med. 2019, 8, 5716–5734. [Google Scholar] [CrossRef] [Green Version]
- Liang, Z.; Wang, X.; Xu, X.; Xie, B.; Ji, A.; Meng, S.; Li, S.; Zhu, Y.; Wu, J.; Hu, Z.; et al. MicroRNA-608 Inhibits Proliferation of Bladder Cancer via AKT/FOXO3a Signaling Pathway. Mol. Cancer 2017, 16, 96. [Google Scholar] [CrossRef]
- Othman, N.; Nagoor, N.H. MiR-608 Regulates Apoptosis in Human Lung Adenocarcinoma via Regulation of AKT2. Int. J. Oncol. 2017, 51, 1757–1764. [Google Scholar] [CrossRef]
- Lindholm, E.M.; Leivonen, S.-K.; Undlien, E.; Nebdal, D.; Git, A.; Caldas, C.; Børresen-Dale, A.-L.; Kleivi, K. MiR-342-5p as a Potential Regulator of HER2 Breast Cancer Cell Growth. MicroRNA 2018, 8, 155–165. [Google Scholar] [CrossRef]
- Bitaraf, A.; Babashah, S.; Garshasbi, M. Aberrant Expression of a Five-MicroRNA Signature in Breast Carcinoma as a Promising Biomarker for Diagnosis. J. Clin. Lab. Anal. 2020, 34, e23063. [Google Scholar] [CrossRef] [Green Version]
- Pérez-Rivas, L.G.; Jerez, J.M.; Carmona, R.; Luque, V.; Vicioso, L.; Claros, M.G.; Viguera, E.; Pajares, B.; Sańchez, A.; Ribelles, N.; et al. A MicroRNA Signature Associated with Early Recurrence in Breast Cancer. PLoS ONE 2014, 9, e91884. [Google Scholar] [CrossRef]
- Dou, Y.; Chen, F.; Lu, Y.; Qiu, H.; Zhang, H. Effects of Wnt/β-Catenin Signal Pathway Regulated by MiR-342-5P Targeting CBX2 on Proliferation, Metastasis and Invasion of Ovarian Cancer Cells. Cancer Manag. Res. 2020, 12, 3783–3794. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Li, Q.; Niu, J.; Li, B.; Jiang, D.; Wan, Z.; Yang, Q.; Jiang, F.; Wei, P.; Bai, S. MicroRNA-342-5p and MiR-608 Inhibit Colon Cancer Tumorigenesis by Targeting NAA10. Oncotarget 2016, 7, 2709–2720. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.; Wang, Z.; Zhou, X.; Tang, M.; Tan, W.; Sun, T.; Deng, Y. MiR-342-5p Inhibits Osteosarcoma Cell Growth, Migration, Invasion, and Sensitivity to Doxorubicin through Targeting Wnt7b. Cell Cycle 2019, 18, 3325–3336. [Google Scholar] [CrossRef] [PubMed]
- Soriano, A.; Masanas, M.; Boloix, A.; Masiá, N.; París-Coderch, L.; Piskareva, O.; Jiménez, C.; Henrich, K.O.; Roma, J.; Westermann, F.; et al. Functional High-Throughput Screening Reveals MiR-323a-5p and MiR-342-5p as New Tumor-Suppressive MicroRNA for Neuroblastoma. Cell. Mol. Life Sci. 2019, 76, 2231–2243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, J.; Zhang, D.; Zhang, J.; Yang, L.; Zhao, G.; Yang, H.; Wang, J. Long Non-Coding RNA SNHG7 Promotes Neuroblastoma Progression through Sponging MiR-323a-5p and MiR-342-5p. Biomed. Pharmacother. 2020, 128, 110293. [Google Scholar] [CrossRef]
- Zou, A.; Liu, X.; Mai, Z.; Zhang, J.; Liu, Z.; Huang, Q.; Wu, A.; Zhou, C. LINC00472 Acts as a Tumor Suppressor in NSCLC through KLLN-Mediated P53-Signaling Pathway via MicroRNA-149-3p and MicroRNA-4270. Mol. Ther.-Nucleic Acids 2019, 17, 563–577. [Google Scholar] [CrossRef] [Green Version]
- Sun, G.; Ding, X.; Bi, N.; Wang, Z.; Wu, L.; Zhou, W.; Zhao, Z.; Wang, J.; Zhang, W.; Fan, J.; et al. Molecular Predictors of Brain Metastasis-Related MicroRNAs in Lung Adenocarcinoma. PLoS Genet. 2019, 15, e1007888. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Li, C.-F.; Sun, L.-B.; Li, Y.-C. chao MicroRNA-4270-5p Inhibits Cancer Cell Proliferation and Metastasis in Hepatocellular Carcinoma by Targeting SATB2. Hum. Cell 2020, 33, 1155–1164. [Google Scholar] [CrossRef]
- Chen, W.; Yao, G.; Zhou, K. MiR-103a-2-5p/MiR-30c-1-3p Inhibits the Progression of Prostate Cancer Resistance to Androgen Ablation Therapy via Targeting Androgen Receptor Variant 7. J. Cell. Biochem. 2019, 120, 14055–14064. [Google Scholar] [CrossRef]
- Liu, M.; Liu, Q.; Fan, S.; Su, F.; Jiang, C.; Cai, G.; Wang, Y.; Liao, G.; Lei, X.; Chen, W.; et al. LncRNA LTSCCAT Promotes Tongue Squamous Cell Carcinoma Metastasis via Targeting the MiR-103a-2-5p/SMYD3/TWIST1 Axis. Cell Death Dis. 2021, 12, 144. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.L.; Sha, X.; Wang, Y.; Li, J.; Zhang, M.Y.; Guo, Z.Y.; Sun, S.A.; He, J.D. Circular RNA Hsa-Circ-0007142 Is Upregulated and Targets MiR-103a-2-5p in Colorectal Cancer. J. Oncol. 2019, 2019, 9836819. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ho, K.H.; Kuo, T.C.; Lee, Y.T.; Chen, P.H.; Shih, C.M.; Cheng, C.H.; Liu, A.J.; Lee, C.C.; Chen, K.C. Xanthohumol Regulates MiR-4749-5p-Inhibited RFC2 Signaling in Enhancing Temozolomide Cytotoxicity to Glioblastoma. Life Sci. 2020, 254, 117807. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, D.B.; Nguyen, J.T.; Haidar, F.; Fox, A.L.; Ray, C.; Amatullah, H.; Liu, F.; Kim, J.K.; Krebsbach, P.H. MicroRNA-1911-3p Targets MEAK-7 to Suppress MTOR Signaling in Human Lung Cancer Cells. Heliyon 2020, 6, e05734. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.Y.; Wang, L.Q.; Chim, C.S. MiR-1250-5p Is a Novel Tumor Suppressive Intronic MiRNA Hypermethylated in Non-Hodgkin’s Lymphoma: Novel Targets with Impact on ERK Signaling and Cell Migration. Cell Commun. Signal. 2021, 19, 62. [Google Scholar] [CrossRef]
- Shi, N.; Shan, B.; Gu, B.; Song, Y.; Chu, H.; Qian, L. Circular RNA Circ-PRKCI Functions as a Competitive Endogenous RNA to Regulate AKT3 Expression by Sponging MiR-3680-3p in Esophageal Squamous Cell Carcinoma. J. Cell. Biochem. 2019, 120, 10021–10030. [Google Scholar] [CrossRef]
- Rotundo, F.; Cominetti, D.; El Bezawy, R.; Percio, S.; Doldi, V.; Tortoreto, M.; Zuco, V.; Valdagni, R.; Zaffaroni, N.; Gandellini, P. MiR-1272 Exerts Tumor-Suppressive Functions in Prostate Cancer via HIP1 Suppression. Cells 2020, 9, 435. [Google Scholar] [CrossRef] [Green Version]
- Iparraguirre, L.; Gutierrez-Camino, A.; Umerez, M.; Martin-Guerrero, I.; Astigarraga, I.; Navajas, A.; Sastre, A.; De Andoin, N.G.; Garcia-Orad, A. MIR-Pharmacogenetics of Methotrexate in Childhood B-Cell Acute Lymphoblastic Leukemia. Pharmacogenet. Genom. 2016, 26, 517–525. [Google Scholar] [CrossRef]
- Tuncer, S.B.; Erdogan, O.S.; Erciyas, S.K.; Saral, M.A.; Celik, B.; Odemis, D.A.; Turkcan, G.K.; Yazici, H. MiRNA Expression Profile Changes in the Peripheral Blood of Monozygotic Discordant Twins for Epithelial Ovarian Carcinoma: Potential New Biomarkers for Early Diagnosis and Prognosis of Ovarian Carcinoma. J. Ovarian Res. 2020, 13, 99. [Google Scholar] [CrossRef]
- Fang, W.; Shu, S.; Yongmei, L.; Endong, Z.; Lirong, Y.; Bei, S. MiR-224-3p Inhibits Autophagy in Cervical Cancer Cells by Targeting FIP200. Sci. Rep. 2016, 6, 33229. [Google Scholar] [CrossRef] [Green Version]
- Shen, X.; Li, L.; He, Y.; Lv, X.; Ma, J. Raddeanin A Inhibits Proliferation, Invasion, Migration and Promotes Apoptosis of Cervical Cancer Cells via Regulating MiR-224-3p/Slit2/ Robo1 Signaling Pathway. Aging 2021, 13, 7166–7179. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.; Feng, F.; Yu, X.; Liu, Z.; Lao, L. LncRNA SNHG4 Promotes Tumour Growth by Sponging MiR-224-3p and Predicts Poor Survival and Recurrence in Human Osteosarcoma. Cell Prolif. 2018, 51, e12515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, G.; Liu, L.; Zhang, J.; Huang, C.; Chen, Y.; Bai, W.; Wang, Y.; Zhao, K.; Li, S. Lncrna Hcg11 Suppresses Cell Proliferation and Promotes Apoptosis via Sponging Mir-224-3p in Non-Small-Cell Lung Cancer Cells. Onco. Targets. Ther. 2020, 13, 6553–6563. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Zhang, K.; Cao, L.; Sun, H.; Wang, H. Inhibition of Microrna-766-5p Attenuates the Development of Cervical Cancer Through Regulating SCAI. Technol. Cancer Res. Treat. 2020, 19, 1533033820980081. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Yao, D.; Li, Y.; Chen, H.; He, C.; Ding, N.; Lu, Y.; Ou, T.; Zhao, S.; Li, L.; et al. Serum MicroRNA Expression Levels Can Predict Lymph Node Metastasis in Patients with Early-Stage Cervical Squamous Cell Carcinoma. Int. J. Mol. Med. 2013, 32, 557–567. [Google Scholar] [CrossRef] [Green Version]
- Yang, X.H.; Guo, F. MiR-3147 Serves as an OncomiR in Vulvar Squamous Cell Cancer via Smad4 Suppression. Mol. Med. Rep. 2018, 17, 6397–6404. [Google Scholar] [CrossRef]
- Fan, G.; Jiao, J.; Shen, F.; Ren, Q.; Wang, Q.; Chu, F. Long Non-Coding RNA HCG11 Sponging MiR-522-3p Inhibits the Tumorigenesis of Non-Small Cell Lung Cancer by Upregulating SOCS5. Thorac. Cancer 2020, 11, 2877–2886. [Google Scholar] [CrossRef]
- Shi, J.; Ma, H.; Wang, H.; Zhu, W.; Jiang, S.; Dou, R.; Yan, B. Overexpression of LINC00261 Inhibits Non–Small Cell Lung Cancer Cells Progression by Interacting with MiR-522-3p and Suppressing Wnt Signaling. J. Cell. Biochem. 2019, 120, 18378–18387. [Google Scholar] [CrossRef]
- Miyamoto, M.; Sawada, K.; Nakamura, K.; Yoshimura, A.; Ishida, K.; Kobayashi, M.; Shimizu, A.; Yamamoto, M.; Kodama, M.; Hashimoto, K.; et al. Paclitaxel Exposure Downregulates MiR-522 Expression and Its Downregulation Induces Paclitaxel Resistance in Ovarian Cancer Cells. Sci. Rep. 2020, 10, 16755. [Google Scholar] [CrossRef]
- Vaira, V.; Roncalli, M.; Carnaghi, C.; Faversani, A.; Maggioni, M.; Augello, C.; Rimassa, L.; Pressiani, T.; Spagnuolo, G.; Di Tommaso, L.; et al. MicroRNA-425-3p Predicts Response to Sorafenib Therapy in Patients with Hepatocellular Carcinoma. Liver Int. 2015, 35, 1077–1086. [Google Scholar] [CrossRef]
- Li, H.; Zhao, C.; Zhao, H.; Liu, G.; Mao, H.; Liu, Y. Elevated Linc00936 or Silenced MicroRNA-425-3p Inhibits Immune Escape of Gastric Cancer Cells via Elevation of ZC3H12A. Int. Immunopharmacol. 2021, 95, 107559. [Google Scholar] [CrossRef] [PubMed]
- Rana, S.; Valbuena, G.N.; Curry, E.; Bevan, C.L.; Keun, H.C. MicroRNAs as Biomarkers for Prostate Cancer Prognosis: A Systematic Review and a Systematic Reanalysis of Public Data. Br. J. Cancer 2022, 126, 502–513. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhao, H.; Gao, X.; Wei, F.; Zhang, X.; Su, Y.; Wang, C.; Li, H.; Ren, X. Identification of a Three-MiRNA Signature as a Blood-Borne Diagnostic Marker for Early Diagnosis of Lung Adenocarcinoma. Oncotarget 2016, 7, 26070–26086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuwen, D.; Ma, Y.; Wang, D.; Gao, J.; Li, X.; Xue, W.; Fan, M.; Xu, Q.; Shen, Y.; Shu, Y. Prognostic Role of Circulating Exosomal MiR-425-3p for the Response of NSCLC to Platinum-Based Chemotherapy. Cancer Epidemiol. Biomark. Prev. 2019, 28, 163–173. [Google Scholar] [CrossRef] [Green Version]
- Choi, Y.W.; Song, Y.S.; Lee, H.; Yi, K.; Kim, Y.B.; Suh, K.W.; Lee, D. MicroRNA Expression Signatures Associated With BRAF-Mutated Versus KRAS-Mutated Colorectal Cancers. Medicine 2016, 95, e3321. [Google Scholar] [CrossRef]
- Cheng, L.; Cao, H.; Xu, J.; Xu, M.; He, W.; Zhang, W.; Dong, L.; Chen, D. Circ_RPL23A Acts as a MiR-1233 Sponge to Suppress the Progression of Clear Cell Renal Cell Carcinoma by Promoting ACAT2. J. Bioenerg. Biomembr. 2021, 53, 415–428. [Google Scholar] [CrossRef]
- Wulfken, L.M.; Moritz, R.; Ohlmann, C.; Holdenrieder, S.; Jung, V.; Becker, F.; Herrmann, E.; Walgenbach-Brünagel, G.; von Ruecker, A.; Müller, S.C.; et al. MicroRNAs in Renal Cell Carcinoma: Diagnostic Implications of Serum MiR-1233 Levels. PLoS ONE 2011, 6, e25787. [Google Scholar] [CrossRef] [Green Version]
- Sudo, K.; Kato, K.; Matsuzaki, J.; Takizawa, S.; Aoki, Y.; Shoji, H.; Iwasa, S.; Honma, Y.; Takashima, A.; Sakamoto, H.; et al. Identification of Serum MicroRNAs Predicting the Response of Esophageal Squamous-Cell Carcinoma to Nivolumab. Jpn. J. Clin. Oncol. 2019, 50, 114–121. [Google Scholar] [CrossRef]
- Larsen, A.C.; Mikkelsen, L.H.; Borup, R.; Kiss, K.; Toft, P.B.; Von Buchwald, C.; Coupland, S.E.; Prause, J.U.; Heegaard, S. MicroRNA Expression Profile in Conjunctival Melanoma. Investig. Ophthalmol. Vis. Sci. 2016, 57, 4205–4212. [Google Scholar] [CrossRef]
- Tao, L.; Zeng, Y.; Wang, J.; Liu, Z.; Shen, B.; Ge, J.; Liu, Y.; Guo, Y.; Qiu, J. Differential MicroRNA Expression in Aristolochic Acid-Induced Upper Urothelial Tract Cancers Ex Vivo. Mol. Med. Rep. 2015, 12, 6533–6546. [Google Scholar] [CrossRef] [Green Version]
- Sayagués, J.M.; Corchete, L.A.; Gutiérrez, M.L.; Sarasquete, M.E.; Abad, M.D.M.; Bengoechea, O.; Fermiñán, E.; Anduaga, M.F.; Del Carmen, S.; Iglesias, M.; et al. Genomic Characterization of Liver Metastases from Colorectal Cancer Patients. Oncotarget 2016, 7, 72908–72922. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, S.; Bae, W.J.; Ahn, J.M.; Heo, J.H.; Kim, K.M.; Choi, K.W.; Sung, C.O.; Lee, D. MicroRNA Signatures Associated with Lymph Node Metastasis in Intramucosal Gastric Cancer. Mod. Pathol. 2021, 34, 672–683. [Google Scholar] [CrossRef] [PubMed]
- Eskiocak, U.; Kim, S.B.; Ly, P.; Roig, A.I.; Biglione, S.; Komurov, K.; Cornelius, C.; Wright, W.E.; White, M.A.; Shay, J.W. Functional Parsing of Driver Mutations in the Colorectal Cancer Genome Reveals Numerous Suppressors of Anchorage-Independent Growth. Cancer Res. 2011, 71, 4359–4365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, S.N.; Towne, D.L.; Burns, D.J.; Warrior, U. A High-Throughput Soft Agar Assay for Identification of Anticancer Compound. J. Biomol. Screen. 2007, 12, 938–945. [Google Scholar] [CrossRef]
- Kochanek, S.J.; Close, D.A.; Johnston, P.A. High Content Screening Characterization of Head and Neck Squamous Cell Carcinoma Multicellular Tumor Spheroid Cultures Generated in 384-Well Ultra-Low Attachment Plates to Screen for Better Cancer Drug Leads. Assay Drug Dev. Technol. 2019, 17, 17–36. [Google Scholar] [CrossRef]
- Close, D.A.; Camarco, D.P.; Shan, F.; Kochanek, S.J.; Johnston, P.A. The Generation of Three-Dimensional Head and Neck Cancer Models for Drug Discovery in 384-Well Ultra-Low Attachment Microplates. In Methods in Molecular Biology; Humana Press: Totowa, NJ, USA, 2018; Volume 1683, pp. 355–369. [Google Scholar]
- Lindenbergh-Van Der Plas, M.; Martens-De Kemp, S.R.; De Maaker, M.; Van Wieringen, W.N.; Ylstra, B.; Agami, R.; Cerisoli, F.; Leemans, C.R.; Braakhuis, B.J.M.; Brakenhoff, R.H. Identification of Lethal MicroRNAs Specific for Head and Neck Cancer. Clin. Cancer Res. 2013, 19, 5647–5657. [Google Scholar] [CrossRef] [Green Version]
- Nakano, H.; Miyazawa, T.; Kinoshita, K.; Yamada, Y.; Yoshida, T. Functional Screening Identifies a MicroRNA, MiR-491 That Induces Apoptosis by Targeting Bcl-XLin Colorectal Cancer Cells. Int. J. Cancer 2010, 127, 1072–1080. [Google Scholar] [CrossRef]
- Palumbo, T.; Poultsides, G.A.; Kouraklis, G.; Liakakos, T.; Drakaki, A.; Peros, G.; Hatziapostolou, M.; Iliopoulos, D. A Functional MicroRNA Library Screen Reveals MiR-410 as a Novel Anti-Apoptotic Regulator of Cholangiocarcinoma. BMC Cancer 2016, 16, 353. [Google Scholar] [CrossRef] [Green Version]
- Poell, J.B.; van Haastert, R.J.; de Gunst, T.; Schultz, I.J.; Gommans, W.M.; Verheul, M.; Cerisoli, F.; van Noort, P.I.; Prevost, G.P.; Schaapveld, R.Q.J.; et al. A Functional Screen Identifies Specific Micrornas Capable of Inhibiting Human Melanoma Cell Viability. PLoS ONE 2012, 7, e43569. [Google Scholar] [CrossRef]
- Moskwa, P.; Zinn, P.O.; Choi, Y.E.; Shukla, S.A.; Fendler, W.; Chen, C.C.; Lu, J.; Golub, T.R.; Hjelmeland, A.; Chowdhury, D. A Functional Screen Identifies MiRs That Induce Radioresistance in Glioblastomas. Mol. Cancer Res. 2014, 12, 1767–1778. [Google Scholar] [CrossRef] [Green Version]
- Hatano, K.; Kumar, B.; Zhang, Y.; Coulter, J.B.; Hedayati, M.; Mears, B.; Ni, X.; Kudrolli, T.A.; Chowdhury, W.H.; Rodriguez, R.; et al. A Functional Screen Identifies MiRNAs That Inhibit DNA Repair and Sensitize Prostate Cancer Cells to Ionizing Radiation. Nucleic Acids Res. 2015, 43, 4075–4086. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olarerin-George, A.O.; Anton, L.; Hwang, Y.-C.; Elovitz, M.A.; Hogenesch, J.B. A Functional Genomics Screen for MicroRNA Regulators of NF-KappaB Signaling. BMC Biol. 2013, 11, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leivonen, S.K.; Sahlberg, K.K.; Mäkelä, R.; Due, E.U.; Kallioniemi, O.; Børresen-Dale, A.L.; Perälä, M. High-Throughput Screens Identify MicroRNAs Essential for HER2 Positive Breast Cancer Cell Growth. Mol. Oncol. 2014, 8, 93–104. [Google Scholar] [CrossRef]
- Hodzic, J.; Sie, D.; Vermeulen, A.; van Beusechem, V.W. Functional Screening Identifies Human MiRNAs That Modulate Adenovirus Propagation in Prostate Cancer Cells. Hum. Gene Ther. 2017, 28, 766–780. [Google Scholar] [CrossRef]
- Steenbergen, R.D.M.; Kramer, D.; Braakhuis, B.J.M.; Stern, P.L.; Verheijen, R.H.M.; Meijer, C.J.L.M.; Snijder, P.J.F. TSLC1 Gene Silencing in Cervical Cancer Cell Lines and Cervical Neoplasia. J. Natl. Cancer Inst. 2004, 96, 294–305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bachas, C.; Hodzic, J.; van der Mijn, J.C.; Stoepker, C.; Verheul, H.M.W.; Wolthuis, R.M.F.; Felley-Bosco, E.; van Wieringen, W.N.; van Beusechem, V.W.; Brakenhoff, R.H.; et al. Rscreenorm: Normalization of CRISPR and SiRNA Screen Data for More Reproducible Hit Selection. BMC Bioinform. 2018, 19, 301. [Google Scholar] [CrossRef]
- Pelz, O.; Gilsdorf, M.; Boutros, M. Web CellHTS2: A Web-Application for the Analysis of High-Throughput Screening Data. BMC Bioinform. 2010, 11, 185. [Google Scholar] [CrossRef] [Green Version]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma Powers Differential Expression Analyses for RNA-Sequencing and Microarray Studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar] [CrossRef]




| Gene ID | Overlap 3 Cell Lines | Gene ID | Overlap 2 Cell Lines |
|---|---|---|---|
| hsa-miR-129-5p | SiHa, HeLa, FK18B | hsa-miR-22-5p | SiHa, HeLa |
| hsa-miR-137 | SiHa, HeLa, FK18B | hsa-miR-103a-2-5p | SiHa, HeLa |
| hsa-miR-185-3p | SiHa, HeLa, FK18B | hsa-miR-425-3p | SiHa, HeLa |
| hsa-miR-193a-3p | SiHa, HeLa, FK18B | hsa-miR-1911-3p | SiHa, HeLa |
| hsa-miR-193b-3p | SiHa, HeLa, FK18B | hsa-miR-4436b-3p | SiHa, HeLa |
| hsa-miR-224-3p | SiHa, HeLa, FK18B | hsa-miR-323a-5p | SiHa, FK18B |
| hsa-miR-491-5p | SiHa, HeLa, FK18B | hsa-miR-342-5p | SiHa, FK18B |
| hsa-miR-522-3p | SiHa, HeLa, FK18B | hsa-miR-378c | SiHa, FK18B |
| hsa-miR-532-3p | SiHa, HeLa, FK18B | hsa-miR-608 | SiHa, FK18B |
| hsa-miR-646 | SiHa, HeLa, FK18B | hsa-miR-766-5p | SiHa, FK18B |
| hsa-miR-1233-1-5p | SiHa, HeLa, FK18B | hsa-miR-1250 | SiHa, FK18B |
| hsa-miR-1263 | SiHa, HeLa, FK18B | hsa-miR-1287 | SiHa, FK18B |
| hsa-miR-2277-5p | SiHa, HeLa, FK18B | hsa-miR-3680-3p | SiHa, FK18B |
| hsa-miR-3916 | SiHa, HeLa, FK18B | hsa-miR-4270 | SiHa, FK18B |
| hsa-miR-4265 | SiHa, HeLa, FK18B | hsa-miR-4498 | SiHa, FK18B |
| hsa-miR-4430 | SiHa, HeLa, FK18B | hsa-miR-5681a | SiHa, FK18B |
| hsa-miR-4440 | SiHa, HeLa, FK18B | hsa-miR-1272 | HeLa, FK18B |
| hsa-miR-4505 | SiHa, HeLa, FK18B | hsa-miR-3147 | HeLa, FK18B |
| hsa-miR-4706 | SiHa, HeLa, FK18B | ||
| hsa-miR-4746-3p | SiHa, HeLa, FK18B | ||
| hsa-miR-4749-5p | SiHa, HeLa, FK18B | ||
| hsa-miR-5189 | SiHa, HeLa, FK18B |
| Gene ID | Function/Expression | Type of Cancers | # |
|---|---|---|---|
| hsa-miR-137 | Tumor suppressor Anchorage-independent growth reduction | Cervical [24,25,26,27,28], melanoma [29], acute lymphoblastic leukemia [30], pituitary adenoma [31], prolactinomas [32], pancreatic [33], endometrial [34], colon [35], glioblastoma [36], melanoma [37], bladder [38], liver [39], pancreatic [40], and other | 305 |
| hsa-miR-129-5p | Tumor suppressor | Cervical [8,41,42,43], gastric [44,45], prostate [46], lung [47,48], colon [49], breast [50], pancreatic [51], liver [52], bladder [53], many other | 159 |
| hsa-miR-491-5p | Tumor suppressor | Cervical [54,55,56], colon [57], osteosarcoma [58], gastric [59], many others | 49 |
| hsa-miR-193b-3p | Tumor suppressor | Cervical [60], lung [61], ovarian [62], gastric [63], acute myeloid leukemia [64] | 43 |
| hsa-miR-532-3p | Tumor suppressor Downregulated | Cervical [65], colon [66], lung [67], lymphoma [68], prostate [69], tongue [70], breast [71] and other | 36 |
| hsa-miR-646 | Tumor suppressor | Cervical [72], breasts [73], lung [74], gastric [75], many others | 31 |
| hsa-miR-185-3p | Tumor suppressor Downregulated | Cervical [76], prostate [77], colon [78], osteosarcoma [79], breast [80], nasopharyngeal [81], and other | 21 |
| hsa-miR-1287 | Tumor suppressor Downregulated | Cervical [82,83], breast [83,84,85,86,87,88], liver [87], colon [89], pancreatic [90], lung [88] and other | |
| hsa-miR-22-5p | Tumor suppressor Downregulated | Cervical [65,91,92,93], osteosarcoma, prostate, lung [93], and other | 16 |
| hsa-miR-378c | Tumor suppressor Prognostic marker | Cervical [94], gastric [95], Wilms tumor [96], head and neck [97] | 12 |
| hsa-miR-193a-3p | Tumor suppressor | Lung [98,99,100], esophageal [101], liver [102], renal [103], osteosarcoma [104], colon [105], many others | 107 |
| hsa-miR-608 | Tumor suppressor | Prostate [106], bladder [107], lung [108] | 80 |
| hsa-miR-342-5p | Tumor suppressor | Breast [109,110,111], ovarian [112], colon [113], osteosarcoma [114], neuroblastoma [115,116] | 27 |
| hsa-miR-4270 | Tumor suppressor | Lung [117,118], liver [119] | 10 |
| hsa-miR-103a-2-5p | Tumor suppressor | Prostate [120], tongue [121], colon [122] | 5 |
| hsa-miR-4749-5p | Tumor suppressor | Glioblastoma [123] | |
| hsa-miR-1911-3p | Tumor suppressor | Lung [124] | |
| hsa-miR-1250 | Tumor suppressor | Non-Hodgkin’s lymphoma [125] | |
| hsa-miR-323a-5p | Tumor suppressor | Neuroblastoma [115,116] | |
| hsa-miR-3680-3p | Tumor suppressor | Esophageal [126] | |
| hsa-miR-1272 | Tumor suppressor | Prostate [127] | |
| hsa-miR-5189 | Prognostic marker | Childhood B-cell acute lymphoblastic leukemia [128] | |
| hsa-miR-4430 | Downregulated Prognostic marker | Ovarium [129] | |
| hsa-miR-224-3p | OncomiR | Cervical [130,131], osteosarcoma [132], lung [133] | 15 |
| hsa-miR-766-5p | Upregulated, OncomiR | Cervical [134], promotes other cancers | 6 |
| hsa-miR-3147 | Upregulated, OncomiR Prognostic marker | Cervical [135], vulvar [136] | 2 |
| hsa-miR-522-3p | OncomiR | Lung [137,138], colon [139] | 12 |
| hsa-miR-425-3p | Upregulated Prognostic marker | Liver [140], gastric [141], prostate [142], lung [143,144], colon [145] | 11 |
| hsa-miR-1233-1-5p | Upregulated | Renal [146,147] esophageal [148] | |
| hsa-miR-3916 | Upregulated, OncomiR | Melanoma [149], urothelial tract [150] | |
| hsa-miR-4440 | Upregulated Prognostic marker | Colon [151] | |
| hsa-miR-4505 | Upregulated Prognostic marker | Gastric [152] | |
| No (cancer related) publications: hsa-miR-2277-5p, hsa-miR-4498, has-miR-5681a, has-miR-4436b-3p, hsa-miR-4706, hsa-miR-1263, hsa-miR-4265, hsa-miR-4746-3p | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huseinovic, A.; Jaspers, A.; van Splunter, A.P.; Sørgård, H.; Wilting, S.M.; Swarts, D.R.A.; van der Meulen, I.H.; van Beusechem, V.W.; de Menezes, R.X.; Steenbergen, R.D.M. Functional Screen for microRNAs Suppressing Anchorage-Independent Growth in Human Cervical Cancer Cells. Int. J. Mol. Sci. 2022, 23, 4791. https://doi.org/10.3390/ijms23094791
Huseinovic A, Jaspers A, van Splunter AP, Sørgård H, Wilting SM, Swarts DRA, van der Meulen IH, van Beusechem VW, de Menezes RX, Steenbergen RDM. Functional Screen for microRNAs Suppressing Anchorage-Independent Growth in Human Cervical Cancer Cells. International Journal of Molecular Sciences. 2022; 23(9):4791. https://doi.org/10.3390/ijms23094791
Chicago/Turabian StyleHuseinovic, Angelina, Annelieke Jaspers, Annina P. van Splunter, Hanne Sørgård, Saskia M. Wilting, Dorian R. A. Swarts, Ida H. van der Meulen, Victor W. van Beusechem, Renée X. de Menezes, and Renske D. M. Steenbergen. 2022. "Functional Screen for microRNAs Suppressing Anchorage-Independent Growth in Human Cervical Cancer Cells" International Journal of Molecular Sciences 23, no. 9: 4791. https://doi.org/10.3390/ijms23094791
APA StyleHuseinovic, A., Jaspers, A., van Splunter, A. P., Sørgård, H., Wilting, S. M., Swarts, D. R. A., van der Meulen, I. H., van Beusechem, V. W., de Menezes, R. X., & Steenbergen, R. D. M. (2022). Functional Screen for microRNAs Suppressing Anchorage-Independent Growth in Human Cervical Cancer Cells. International Journal of Molecular Sciences, 23(9), 4791. https://doi.org/10.3390/ijms23094791







