Synergistic Antioxidant Effect of Prebiotic Ginseng Berries Extract and Probiotic Strains on Healthy and Tumoral Colorectal Cell Lines
Abstract
:1. Introduction
2. Results
2.1. Determination of Panax Ginseng Berries Extract Principal Molecules
2.2. Evaluation of the Prebiotic Potential of Panax Ginseng Berries Extract
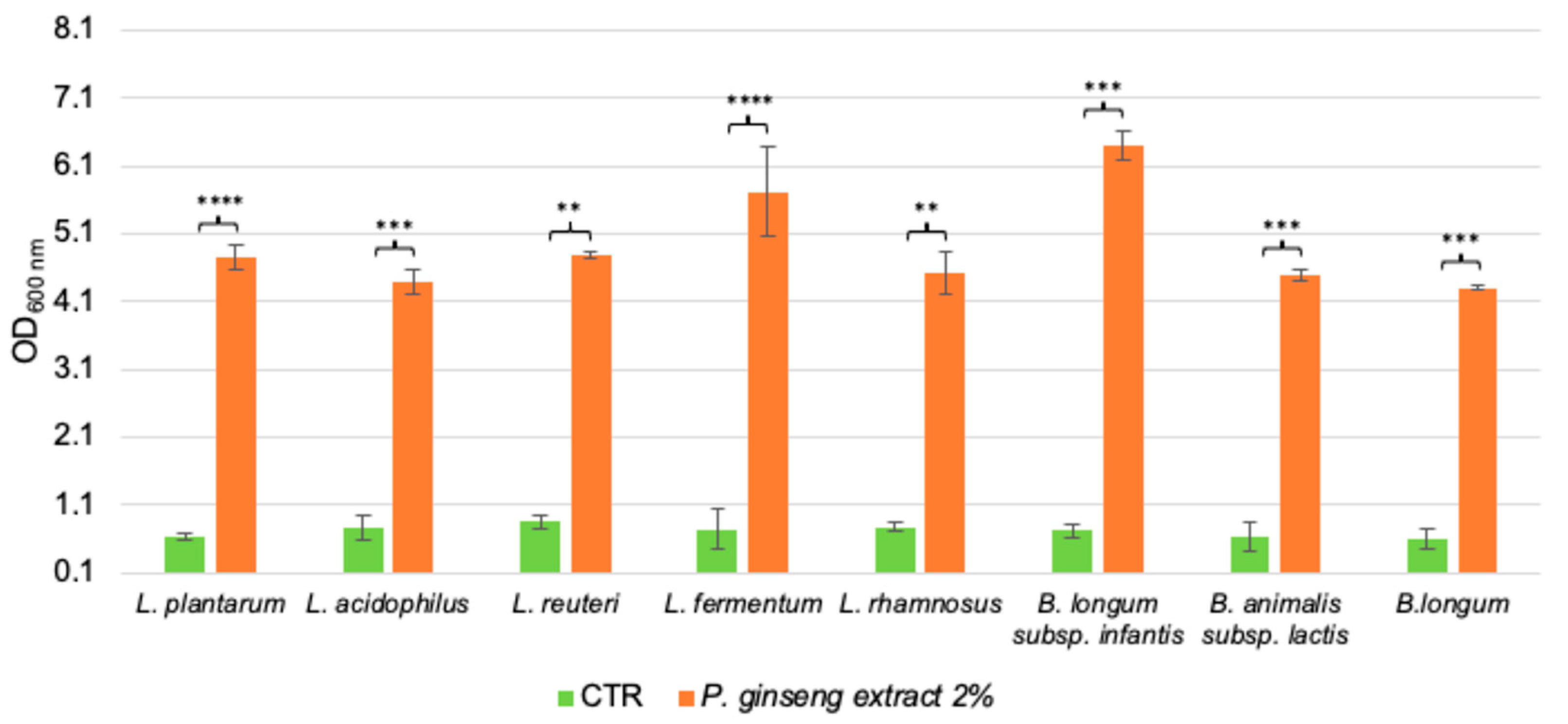
2.3. Investigation of the Capacity of the Single Components of Panax Ginseng Berries Extract to Support Probiotics Growth
2.4. Growth of a Beneficial Probiotic Mix on Panax Ginseng Berries Extract
2.5. Analyses of Probiotic Secondary Metabolites in Presence of Panax Ginseng Berries Extract
2.6. Evaluation of the Bioactivity of Extracted Secondary Metabolites on Cell Viability
2.7. Oxidative Stress Analysis
3. Discussion
4. Methods and Materials
4.1. Ginseng Berries Extract Production
4.2. Description of the Components of Ginseng Berries Preparation
4.2.1. Determination of the Protein Content
4.2.2. Determination of Polyphenol Content
4.2.3. Determination of Pectin-Based Polysaccharides, Galactose, and Galacturonic Acid Content
4.2.4. Measurement of Ginsenosides Amount
4.3. Bacteria Employed and Their Maintenance
4.4. Growth Assays of Single Bacteria or Beneficial Probiotic Mix
4.5. Determination of Produced Bacterial Secondary Metabolites
4.5.1. Metabolites Extraction
4.5.2. Extracted Metabolites Analyses
4.6. Cell Lines and Their Maintenance
4.7. Viability Assay
4.8. Antioxidant Enzyme Assays
4.9. Glutathione Assay
4.10. Intracellular ROS Detection
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Storz, G.; Imlayt, J.A. Oxidative stress. Curr. Opin. Microbiol. 1999, 2, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Xia, C.; Cao, X.; Cui, L.; Liu, H.; Wang, S.; Chen, T. Anti-aging effect of the combination of Bifidobacterium longum and B. animalis in a D-galactose-treated mice. J. Funct. Foods 2020, 69, 10398. [Google Scholar] [CrossRef]
- Zhao, J.; Tian, F.; Yan, S.; Zhai, Q.; Zhang, H.; Chen, W. Lactobacillus plantarum CCFM10 alleviating oxidative stress and restoring the gut microbiota in D-galactose-induced aging mice. Food Funct. 2018, 9, 917–924. [Google Scholar] [CrossRef]
- Presti, I.; D’Orazio, G.; Labra, M.; La Ferla, B.; Mezzasalma, V.; Bizzaro, G.; Giardina, S.; Michelotti, A.; Tursi, F.; Vassallo, M.; et al. Evaluation of the probiotic properties of new Lactobacillus and Bifidobacterium strains and their in vitro effect. Appl. Microbiol. Biotechnol. 2015, 99, 5613–5626. [Google Scholar] [CrossRef] [PubMed]
- De Giani, A.; Bovio, F.; Forcella, M.E.; Lasagni, M.; Fusi, P.; Di Gennaro, P. Prebiotic effect of Maitake extract on a probiotic consortium and its action after microbial fermentation on colorectal cell lines. Foods 2021, 10, 2536. [Google Scholar] [CrossRef]
- De Giani, A.; Sandionigi, A.; Zampolli, J.; Michelotti, A.; Tursi, F.; Labra, M.; Di Gennaro, P. Effects of inulin-based prebiotics alone or in combination with probiotics on human gut microbiota and markers of immune system: A randomized, double-blind, placebo-controlled study in healthy subjects. Microorganisms 2022, 10, 1256. [Google Scholar] [CrossRef] [PubMed]
- Sandionigi, A.; De Giani, A.; Tursi, F.; Michelotti, A.; Cestone, E.; Giardina, S.; Zampolli, J.; Di Gennaro, P. Effectiveness of multistrain probiotic formulation on common infectious disease symptoms and gut microbiota modulation in flu-vaccinated healthy elderly subjects. BioMed Res. Int. 2022, 2022, 3860896. [Google Scholar] [CrossRef]
- Goyal, N.; Rishi, P.; Shukla, G. Lactobacillus rhamnosus GG antagonizes Giardia intestinalis induced oxidative stress and intestinal disaccharidases: An experimental study. World J. Microbiol. Biotechnol. 2013, 29, 1049–1057. [Google Scholar] [CrossRef]
- Lee, B.-J.; Kim, J.-S.; Kang, Y.M.; Lim, J.-H.; Kim, Y.-M.; Lee, M.-S.; Jeong, M.-H.; Ahn, C.-B.; Je, J.-Y. Antioxidant activity and γ-aminobutyric acid (GABA) content in sea tangle fermented by Lactobacillus brevis BJ20 isolated from traditional fermented foods. Food Chem. 2010, 122, 271–276. [Google Scholar] [CrossRef]
- Zhang, Y.; Du, R.; Wang, L.; Zhang, H. The antioxidative effects of probiotic Lactobacillus casei Zhang on the hyperlipidemic rats. Eur. Food Res. Technol. 2010, 231, 151–158. [Google Scholar] [CrossRef]
- Adwas, A.A.; Elsayed, A.; Azab, A.E.; Quwaydir, F.A. Oxidative stress and antioxidant mechanisms in human body. J. Appl. Biotechnol. Bioeng. 2019, 6, 43–47. [Google Scholar] [CrossRef]
- Brown, P.N.; Yu, R.; Cain, T.; Huie, G.; Jin, C.D.; Kababick, J.N.; Leong, G.; LeVanseler, K.; Lunetta, S.; Ma, Y.C.; et al. Determination of ginsenoside content in Panax ginseng C.A. Meyer and Panax quinquefolius L. root materials and finished products by high-performance liquid chromatography with ultraviolet absorbance detection: Interlaboratory study. J. AOAC Int. 2013, 96, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.-Y.; Liu, G.-C.; Zhang, J.-X.; Wang, L.-H.; Xu, C.; Yan, Z.-A.; Wang, A.; Su, Y.-F.; Lee, J.-J.; Piao, G.-C.; et al. Pharmacological properties of Ginsenoside Re. Front. Pharmacol. 2022, 13, 754191. [Google Scholar] [CrossRef]
- Dong, W.-W.; Zhao, J.; Zhong, F.-L.; Zhu, W.-J.; Jiang, J.; Wu, S.; Yang, D.-C.; Li, D.; Quan, L.-H. Biotransformation of Panax ginseng extract by rat intestinal microflora: Identification and quantification of metabolites using liquid chromatography-tandem mass spectrometry. J. Ginseng Res. 2017, 41, 540–547. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.-Y.; Park, C.W.; Lee, S.J.; Park, H.-R.; Kim, S.H.; Son, S.-U.; Park, J.; Shin, K.-S. Anti-cancer effects of Panax ginseng berry polysaccharides via activation of immune-related cells. Front. Pharmacol. 2019, 10, 1411. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- IUPAC. Compendium of Chemical Terminology. In Gold Book, 2nd ed.; McNaught, A.D., Wilkinson, A., Eds.; Blackwell Scientific Publications: Oxford, UK, 1997; ISBN 0-9678550-9-8. [Google Scholar] [CrossRef]
- Kim, J.H.; Yi, Y.-S.; Kim, M.-Y.; Cho, J.Y. Role of ginsenosides, the main active components of Panax ginseng, in inflammatory responses and diseases. J. Ginseng Res. 2017, 41, 435–443. [Google Scholar] [CrossRef] [Green Version]
- He, Z.; Wang, X.; Li, G.; Zhao, Y.; Zhang, J.; Niu, C.; Zhang, L.; Zhang, X.; Ying, D.; Li, S. Antioxidant activity of prebiotic ginseng polysaccharides combined with potential probiotic Lactobacillus plantarum C88. Int. J. Food Sci. Technol. 2015, 50, 1673–1682. [Google Scholar] [CrossRef]
- Ahn, S.I.; Kim, S.K.; Yang, B.W.; Lee, E.S.; Kang, C.S.; Hahm, Y.T. Analysis of ginsenosides and non-saponin components of Red Ginseng from landraces and new varieties. Korean J. Hortic. Sci. 2016, 34, 790–798. [Google Scholar] [CrossRef]
- Ru, W.; Wang, D.; Xu, Y.; He, X.; Sun, Y.-E.; Qian, L.; Zhou, X.; Qin, Y. Chemical constituents and bioactivities of Panax ginseng (C. A. Mey.). Drug Discov. Ther. 2015, 9, 23–32. [Google Scholar] [CrossRef]
- Jung, J.; Jang, H.J.; Eom, S.J.; Choi, N.S.; Lee, N.-K.; Paik, H.-D. Fermentation of red ginseng extract by the probiotic Lactobacillus plantarum KCCM 11613P: Ginsenoside conversion and antioxidant effects. J. Ginseng Res. 2019, 43, 20–26. [Google Scholar] [CrossRef]
- St-Laurent, T.; Hammami, R. The untapped potential of ginsenosides and American Ginseng berry in promoting mental health via the gut–brain axis. Nutrients 2022, 14, 2523. [Google Scholar] [CrossRef] [PubMed]
- Zheng, F.; Zhang, M.; Wu, Y.; Wang, Y.; Li, F.; Han, M.; Dai, Y.; Yue, H. Biotransformation of ginsenosides (Rb1, Rb2, Rb3, Rc) in human intestinal bacteria and its effect on intestinal flora. Chem. Biodivers. 2021, 18, e2100296. [Google Scholar] [CrossRef] [PubMed]
- Han, K.; Balan, P.; Hong, H.; Choi, W.; Cho, C.; Lee, Y.; Moughan, P.; Singh, H. Korean Ginseng modulates the ileal microbiota and mucin gene expression in the growing rat. Food Funct. 2014, 5, 1506–1512. [Google Scholar] [CrossRef] [PubMed]
- Nordberg, J.; Arnér, E.S. Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radic. Biol. Med. 2001, 31, 1287–1312. [Google Scholar] [CrossRef] [PubMed]
- Dey, L.; Xie, J.; Wang, A.; Wu, J.; Maleckar, S.; Yuan, C.-S. Anti-hyperglycemic effects of ginseng: Comparison between root and berry. Phytomedicine 2003, 10, 600–605. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.-W.; Kim, Y.-C.; Kang, J.-H.; Rhee, Y.K.; Choi, S.Y.; Kim, K.-T.; Lee, Y.-C.; Hong, H.-D. Characteristic study on the chemical components of Korean curved ginseng products. J. Ginseng Res. 2013, 37, 349–354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ong, W.-Y.; Farooqui, T.; Koh, H.-L.; Farooqui, A.A.; Ling, E.-A. Protective effects of ginseng on neurological disorders. Front. Aging Neurosci. 2015, 7, 129. [Google Scholar] [CrossRef] [Green Version]
- Zeaiter, Z.; Regonesi, M.E.; Cavini, S.; Labra, M.; Sello, G.; Di Gennaro, P. Extraction and characterization of inulin-type fructans from artichoke wastes and their effect on the growth of intestinal bacteria associated with health. BioMed Res. Int. 2019, 2019, 1083952. [Google Scholar] [CrossRef] [Green Version]
- Palframan, R.; Gibson, G.; Rastall, R. Development of a quantitative tool for the comparison of the prebiotic effect of dietary oligosaccharides. Lett. Appl. Microbiol. 2003, 37, 281–284. [Google Scholar] [CrossRef]
- Shalaby, A.S.G.; Ragab, T.I.; Mehany, A.B.M.; Helal, M.M.; Helmy, W.A. Antitumor and prebiotic activities of novel sulfated acidic polysaccharide from ginseng. Biocatal. Agric. Biotechnol. 2018, 14, 402–409. [Google Scholar] [CrossRef]
- Naqash, F.; Masoodi, F.; Rather, S.A.; Wani, S.; Gani, A. Emerging concepts in the nutraceutical and functional properties of pectin—A Review. Carbohydr. Polym. 2017, 168, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Geirnaert, A.; Steyaert, A.; Eeckhaut, V.; Debruyne, B.; Arends, J.B.; Van Immerseel, F.; Boon, N.; Van de Wiele, T. Butyricicoccus pullicaecorum, a butyrate producer with probiotic potential, is intrinsically tolerant to stomach and small intestine conditions. Anaerobe 2014, 30, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Pontifex, M.G.; Mushtaq, A.; Le Gall, G.; Rodriguez-Ramiro, I.; Blokker, B.A.; Hoogteijling, M.E.M.; Ricci, M.; Pellizzon, M.; Vauzour, D.; Müller, M. Differential influence of soluble dietary fibres on intestinal and hepatic carbohydrate response. Nutrients 2021, 13, 4278. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Halima, M.; Xie, Y.; Schaaf, M.J.M.; Meijer, A.H.; Wang, M. Ginsenoside Rg1 acts as a selective glucocorticoid receptor agonist with anti-inflammatory action without affecting tissue regeneration in zebrafish larvae. Cells 2020, 9, 1107. [Google Scholar] [CrossRef]
- Martinez-Maqueda, D.; Miralles, B.; Recio, I. HT29 cell line. In The Impact of Food Bioactives on Health; Verhoeckx, K., Cotter, P., Lopez-Exposito, I., Kleiveland, C., Lea, T., Mackie, A., Requena, T., Swiatecka, D., Wichers, H., Eds.; Springer: Cham, Switzerland, 2015; Volume 11. [Google Scholar]
- Önning, G.; Berggren, A.; Drevelius, M.; Jeppsson, B.; Lindberg, A.-M.; Hagslätt, M.-L.J. Influence of a drink containing different antioxidants and Lactobacillus plantarum 299v on plasma total antioxidant capacity, selenium status and faecal microbial flora. Int. J. Food Sci. Nutr. 2003, 54, 281–289. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, F.; Liu, R.; Tang, X.; Zhang, Q.; Zhang, Z. Effects of superfine grinding on physicochemical and antioxidant properties of Lycium barbarum polysaccharides. Lwt 2014, 58, 594–601. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventós, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Method Enzymol. 1999, 299, 152–178. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Anthon, G.E.; Barrett, D.M. Combined enzymatic and colorimetric method for determining the uronic acid and methylester content of pectin: Application to tomato products. Food Chem. 2008, 110, 239–247. [Google Scholar] [CrossRef]
- Gutiérrez, N.; Garrido, D. Species deletions from microbiome consortia reveal key metabolic interactions between gut microbes. Msystems 2019, 4, e00185-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watson, D.; Motherway, M.O.; Schoterman, M.; van Neerven, R.J.; Nauta, A.; van Sinderen, D. Selective carbohydrate utilization by lactobacilli and bifidobacteria. J. Appl. Microbiol. 2013, 114, 1132–1146. [Google Scholar] [CrossRef] [PubMed]
- De Giani, A.; Pagliari, S.; Zampolli, J.; Forcella, M.; Fusi, P.; Bruni, I.; Campone, L.; Di Gennaro, P. Characterization of the biological activities of a new polyphenol-rich extract from Cinnamon bark on a probiotic consortium and its action after enzymatic and microbial fermentation on colorectal cell lines. Foods 2022, 11, 3202. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Oldani, M.; Villa, A.M.; Manzoni, M.; Melchioretto, P.; Parenti, P.; Monti, E.; Fusi, P.; Forcella, M.; Urani, C. Insights into cadmium-induced carcinogenesis through an in vitro study using C3H10T1/2Cl8 cells: The multifaceted role of mitochondria. Int. J. Mol. Sci. 2021, 22, 10837. [Google Scholar] [CrossRef] [PubMed]
- Bergmeyer, H.U.; Grassl, M.; Walter, H.E. Methods of Enzymatic Analysis; Bergmeyer, H.U., Ed.; Verlag Chemie: Weinheim, Germany, 1983; Volume 2, pp. 102–439. [Google Scholar] [CrossRef]
- Habig, W.H.; Pabst, M.J.; Jakoby, W.B. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. JBC 1974, 249, 7130–7139. [Google Scholar] [CrossRef]
- Nakamura, W.; Hosoda, S.; Hayashi, K. Purification and properties of rat liver glutathione peroxidase. Biochim. Biophys. Acta (BBA) Enzym. 1974, 358, 251–261. [Google Scholar] [CrossRef]
- Wang, Y.; Oberley, L.W.; Murhammer, D.W. Antioxidant defense systems of two lipidopteran insect cell lines. Free Radic. Biol. Med. 2001, 30, 1254–1262. [Google Scholar] [CrossRef]
- Vance, P.G.; Keele, B.B., Jr.; Rajagopalan, K.V. Superoxide dismutase from Streptococcus mutans. Isolation and characterization of two form of the enzyme. J. Biol. Chem. 1972, 247, 4782–4786. [Google Scholar] [CrossRef]
- Kim, H.; Xue, X. Detection of total reactive oxygen species in adherent cells by 2’,7’-Dichlorodihydrofluorescein diacetate staining. J. Vis. Exp. 2020, 160, e60682. [Google Scholar] [CrossRef]
- Georgiou, C.D.; Papapostolou, I.; Patsoukis, N.; Tsegenidis, T.; Sideris, T. An ultrasensitive fluorescent assay for the in vivo quantification of superoxide radical in organisms. Anal. Biochem. 2005, 347, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, B.C.; Lin, V.; Chang, C.J. Preparation and use of MitoPY1 for imaging hydrogen peroxide in mitochondria of live cells. Nat. Protoc. 2013, 8, 1249–1259. [Google Scholar] [CrossRef] [PubMed]





| Component | P. ginseng Extract (%) |
|---|---|
| Pectin-based polysaccharides Galactose Galacturonic acid | 54.30% 9.01% 4.84% |
| Ginsenosides Ginsenosides Re | 10.00% 2.00% |
| Proteins | 0.98% |
| Polyphenols | 0.72% |
| Unidentified molecules | 34% |
| Strain | Source | Abbreviation |
|---|---|---|
| Lactobacillus acidophilus PBS066 (formerly DSM 24936) | Human | LA |
| Limosilactobacillus fermentum PBS072 (formely DSM 25176) | Human | LF |
| Lactiplantibacillus plantarum PBS067 (formely DSM 24937) | Human | LP |
| Limosilactobacillus reuteri PBS073 (formely DSM 25175) | Human | LR |
| Lacticaseibacillus rhamnosus PBS079 (formerly DSM 25568) | Human | LRh |
| Bifidobacterium animalis subsp. lactis PBS075 (formerly DSM 25566) | Human | BL |
| Bifidobacterium longum subsp. longum PBS108 (formerly DSM 25174) | Human | BLg |
| Bifidobacterium longum subsp. infantis LMG P-29639 | Human | BI |
| Bacteroides cellulosilyticus CL02T12C19, HM-726 | Human | BC |
| Bacteroides finegoldii CL09T03C10 | Human | BF |
| Clostridium symbiosum WAL-14673, HM-319 | Human | CS |
| Clostridium orbiscindens 1_3_50AFAA, HM-303 (formerly Flavonifractor plautii) | Human | CO |
| Ruminococcus gnavus CC55_001C | Human | RG |
| Escherichia coli ATCC 25922 | Human | EC |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Giani, A.; Oldani, M.; Forcella, M.; Lasagni, M.; Fusi, P.; Di Gennaro, P. Synergistic Antioxidant Effect of Prebiotic Ginseng Berries Extract and Probiotic Strains on Healthy and Tumoral Colorectal Cell Lines. Int. J. Mol. Sci. 2023, 24, 373. https://doi.org/10.3390/ijms24010373
De Giani A, Oldani M, Forcella M, Lasagni M, Fusi P, Di Gennaro P. Synergistic Antioxidant Effect of Prebiotic Ginseng Berries Extract and Probiotic Strains on Healthy and Tumoral Colorectal Cell Lines. International Journal of Molecular Sciences. 2023; 24(1):373. https://doi.org/10.3390/ijms24010373
Chicago/Turabian StyleDe Giani, Alessandra, Monica Oldani, Matilde Forcella, Marina Lasagni, Paola Fusi, and Patrizia Di Gennaro. 2023. "Synergistic Antioxidant Effect of Prebiotic Ginseng Berries Extract and Probiotic Strains on Healthy and Tumoral Colorectal Cell Lines" International Journal of Molecular Sciences 24, no. 1: 373. https://doi.org/10.3390/ijms24010373








