Abstract
Cardiovascular mortality is a leading cause of death in chronic kidney disease (CKD), as is IgA nephropathy (IgAN). The purpose of this study is to find different biomarkers to estimate the outcome of the disease, which is significantly influenced by the changes in vessels (characterized by arterial stiffness) and the heart. In our cross-sectional study, 90 patients with IgAN were examined. The N-terminal prohormone of brain natriuretic peptide (NT-proBNP) was measured as a heart failure biomarker by an automated immonoassay method, while the carboxy-terminal telopeptide of collagen type I (CITP) as a fibrosis marker was determined using ELISA kits. Arterial stiffness was determined by measuring carotid–femoral pulse wave velocity (cfPWV). Renal function and routine echocardiography examinations were performed as well. Based on eGFR, patients were separated into two categories, CKD 1-2 and CKD 3-5. There were significantly higher NT-proBNP (p = 0.035), cfPWV (p = 0.004), and central aortic systolic pressure (p = 0.037), but not CITP, in the CKD 3-5 group. Both biomarker positivities were significantly higher in the CKD 3-5 group (p = 0.035) compared to the CKD 1-2 group. The central aortic systolic pressure was significantly higher in the diastolic dysfunction group (p = 0.034), while the systolic blood pressure was not. eGFR and hemoglobin levels showed a strong negative correlation, while left ventricular mass index (LVMI), aortic pulse pressure, central aortic systolic pressure, and cfPWV showed a positive correlation with NT-proBNP. cfPWV, aortic pulse pressure, and LVMI showed a strong positive correlation with CITP. Only eGFR was an independent predictor of NT-proBNP by linear regression analysis. NT-proBNP and CITP biomarkers may help to identify IgAN patients at high risk for subclinical heart failure and further atherosclerotic disease.
1. Introduction
Chronic kidney disease (CKD) is a global health challenge for health professionals and healthcare providers worldwide [1,2]. IgA nephropathy (IgAN) is the most common primary glomerular disease-, and is the typical CKD for cardiovascular (CV) risk assessment. IgAN is induced by the accumulation of IgA-containing immune complexes in the kidney glomeruli, which initiates a cascade of inflammatory processes that ultimately result in irreversible glomerulosclerosis, which in 25–30% of the patients causes end-stage renal disease (ESRD), a decreased quality of life (QoL), the need for dialysis or transplantation, and risk of early mortality [3].
In recent years, plasma biomarkers, particularly natriuretic peptides, along with imaging studies, e.g., echocardiographic examinations or coronary CT scans, have been suggested to identify patients at high CV risk in CKD. In chronic kidney disease (CKD), arterial stiffness may be a predictor of cardiovascular (CV) mortality and morbidity [4]. In advanced CKD stages, heart failure (HF) is highly prevalent. In CKD patients with HF, the emergence of cardiac fibrosis may be a pathophysiological change [5]. Patients with CKD suffer severe losses in life expectancy and quality of life, which are primarily caused by a high incidence of cardiovascular (CV) events [2]. In cross-sectional studies, natriuretic peptides have been independently linked to current cardiovascular disease (CVD) and potential future CV events [6,7] in prospective CKD cohort studies [8,9,10,11,12,13,14]. The carboxy-terminal telopeptide of collagen type I (CITP) is a marker of myocardial fibrosis (MF) in CKD [15], and serum levels are directly correlated with the amount of collagen type I deposition in the myocardium of hypertensive patients with HF [16]. A rise in serum biomarkers is difficult to assess in the case of kidney disease, as we do not know whether the levels really rise or whether their excretion is only the cause of the difference.
Data in the literature about the association between arterial stiffness, subclinical heart failure (NT-pro-BNP), cardiac fibrosis (CITP), and renal function in CKD are limited. We wanted to determine new biomarkers in connection with the influence of renal function on CV complications in IgAN, such as heart failure and atherosclerotic disease prediction, and detection of any association between NT-proBNP as a marker of heart failure, CITP as a marker of atherosclerosis and myocardial fibrosis, and CKD.
2. Results
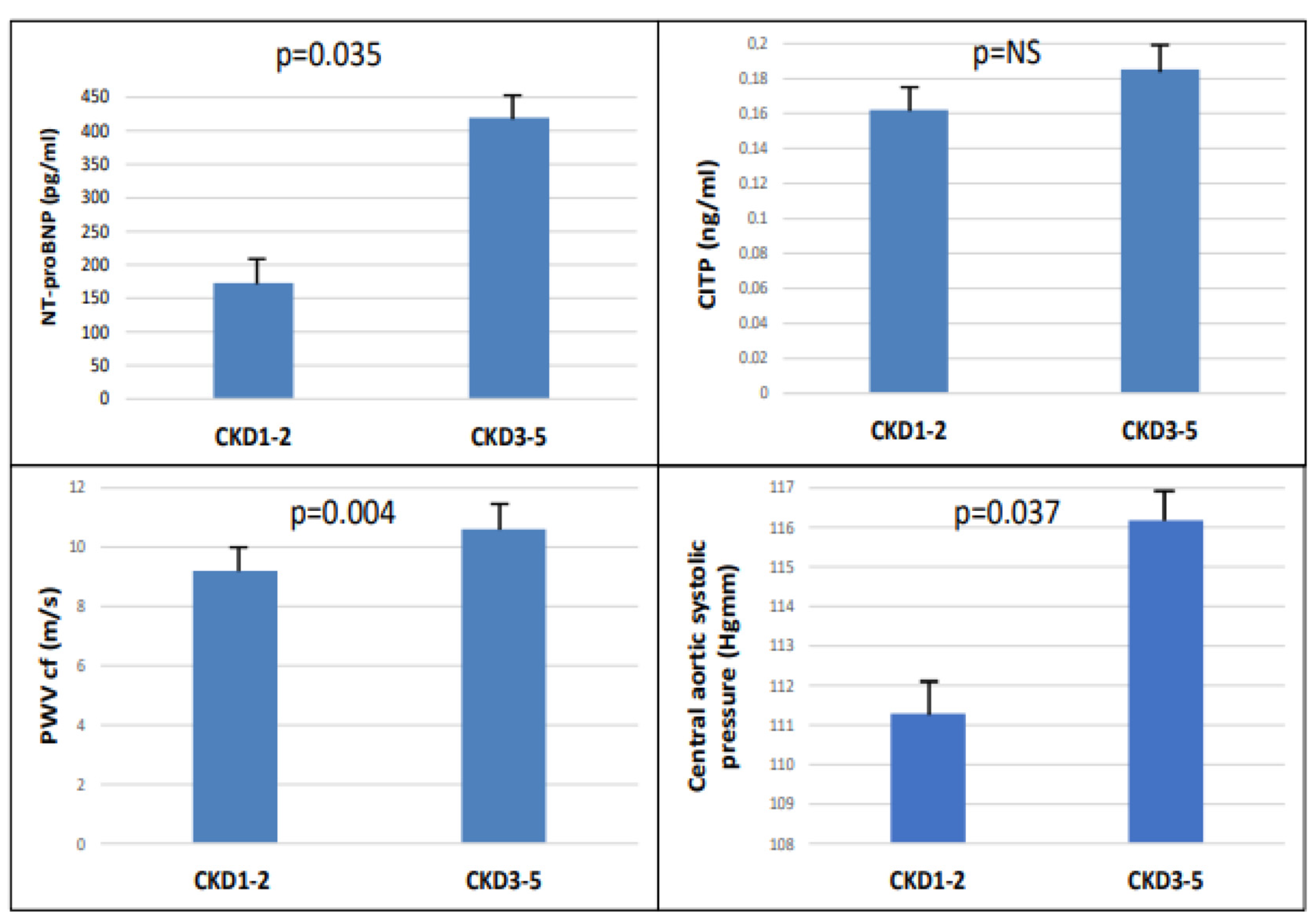
The study included 90 patients, 50 of whom were male, and who had an average age of 54.9 ± 14.4 years. Patients were divided into two groups based on eGFR (CKD 1-2 (GFR: >60 mL/min) and CKD 3-5 (GFR: <60 mL/min)). There were significant differences between the two groups in terms of age, eGFR, hypertension, diabetes, LVMI, and hemoglobin level in the baseline characteristics (Table 1). There were significantly higher NT-proBNP (p = 0.035), cfPWV (p = 0.004), and central aortic systolic pressure (p = 0.037), but not CITP, in the CKD 3-5 group (Figure 1).

Table 1.
Baseline characteristics of IgAN patients.
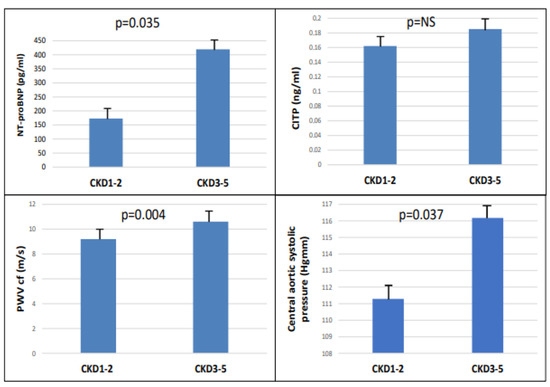
Figure 1.
The NT-pro-BNP, CITP, PWVcf, and central aortic systolic pressure in CKD 1-2 vs. CKD 3-5.
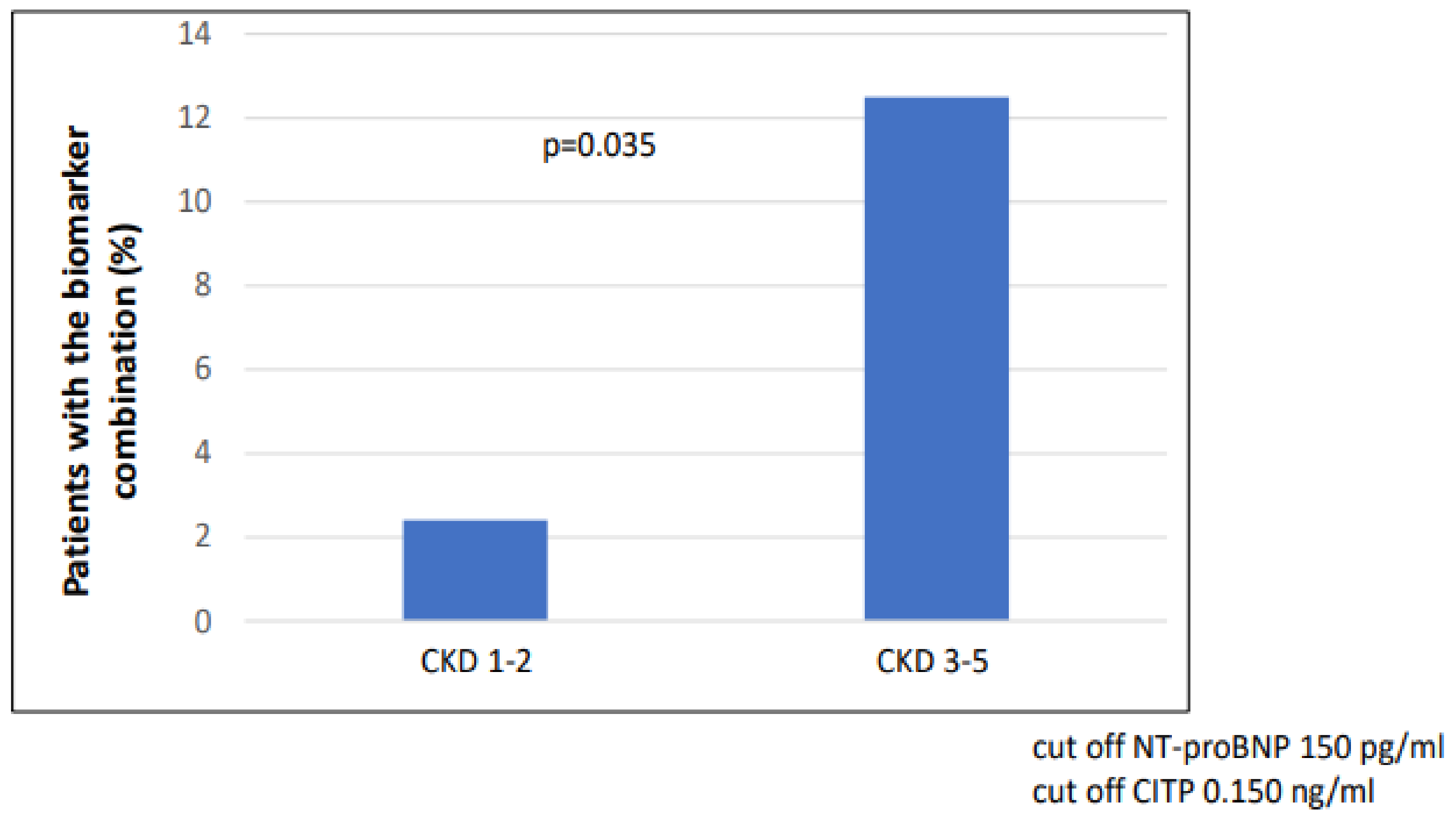
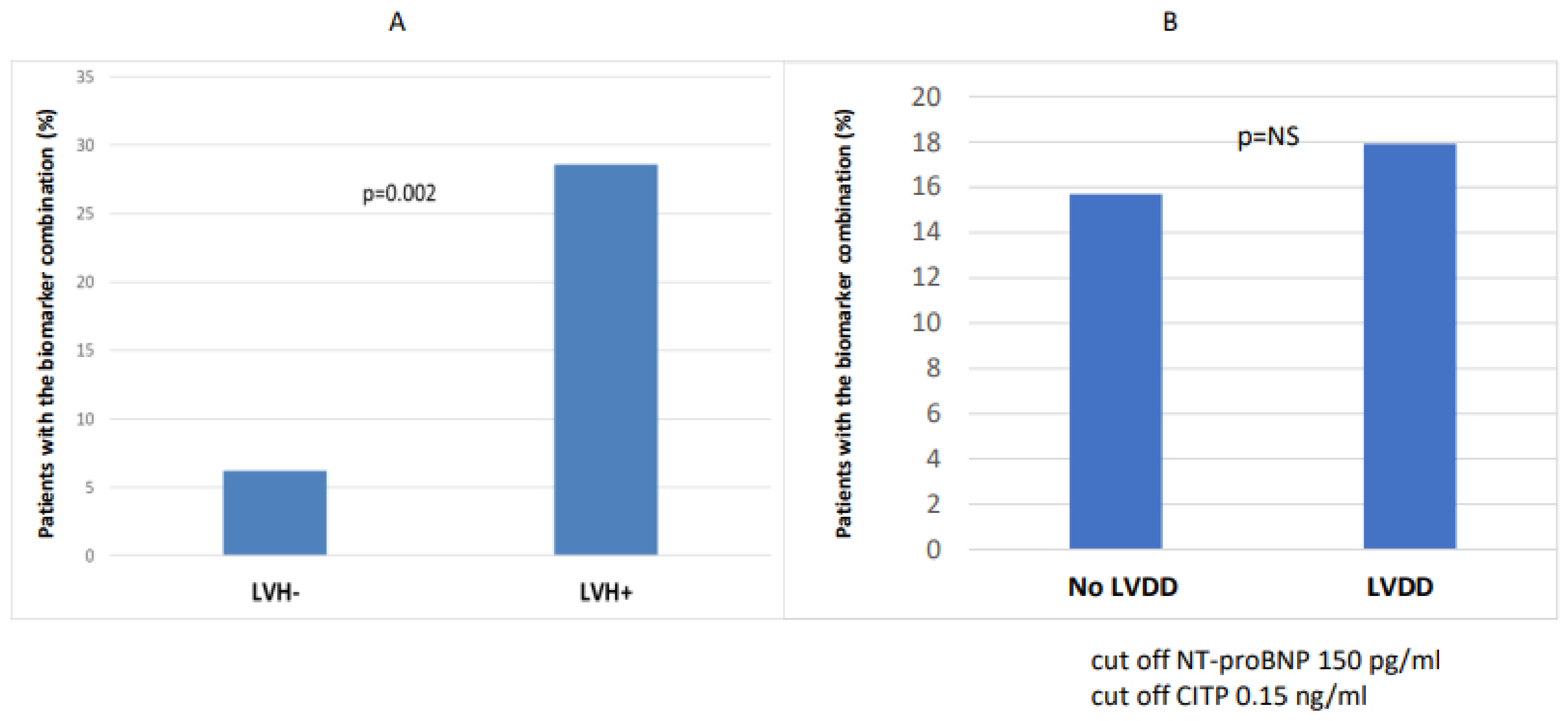
The number of patients with combined biomarker positivity (NT-proBNP and CITP) was significantly higher in the CKD 3-5 group (p = 0.035) compared to the CKD 1-2 group (Figure 2). In the case of LVH (n = 42), the number of patients with combined biomarker positivity (NT-proBNP and CITP) was significantly higher (n = 12/42 vs. 3/48; p = 0.002) than in the non-hypertrophic group (Figure 3).
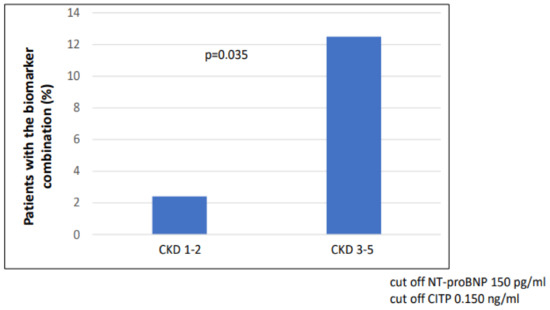
Figure 2.
Combination of biomarker (NT-proBNP and CITP) positivity in CKD 1-2 vs. CKD 3-5.
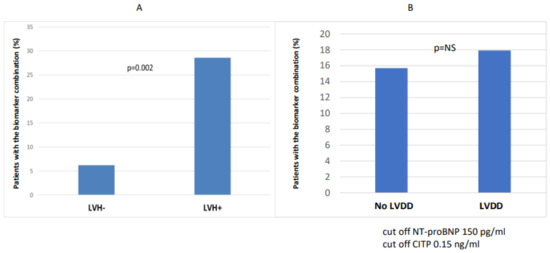
Figure 3.
Combination of biomarker (NT-proBNP and CITP) positivity in LVH- vs. LVH+ (A) and with and without diastolic dysfunction (B).
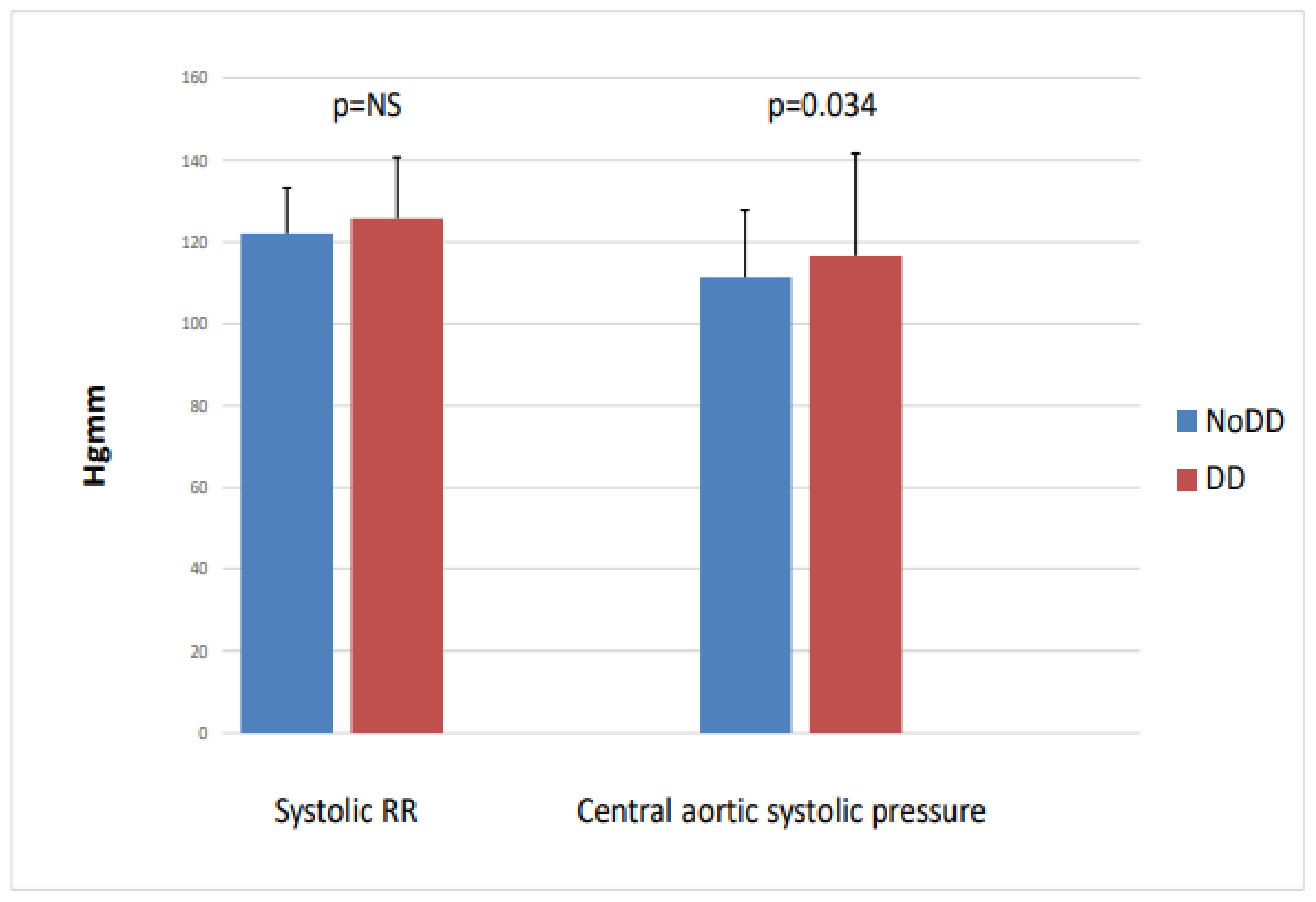
When we divided the patients into two groups based on left ventricular diastolic function (no LVDD (n = 51) vs. LVDD (n = 39)), we found that the diastolic dysfunction group had significantly higher central aortic systolic blood pressure (116.8 vs. 128.5; p = 0.034), while the peripheral systolic blood pressure, NT-proBNP, and CITP did not differ between the two groups (Figure 4).
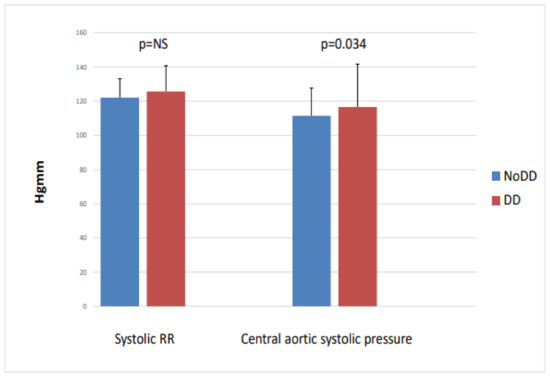
Figure 4.
Peripheral systolic blood pressure and central aortic systolic pressure with and without diastolic dysfunction in IgAN.
NT-proBNP showed a strong negative correlation with eGFR and hemoglobin levels and a positive correlation with the left ventricular mass index (LVMI), aortic pulse pressure, central aortic systolic pressure, and cfPWV. CITP showed a strong positive correlation with cfPWV, aortic pulse pressure, and LVMI by Spearman’s correlation (Table 2).

Table 2.
Correlations with NT-proBNP and CITP.
According to univariate and multivariate regression analysis, the independent predictor factors for NT-proBNP were gender, eGFR, and LVMI. There were no independent confounders in the case of CITP, while all of the exemplified parameters had an impact on the univariate analysis (Table 3 and Table 4). Only eGFR was an independent predictor of NT-proBNP by linear regression analysis.

Table 3.
Univariate and multivariate regression analysis of NT-proBNP.

Table 4.
Univariate and multivariate regression analysis of CITP.
3. Discussion
In the present study, we demonstrated that NT-proBNP and a combination of NT-proBNP and CITP elevation may predict certain cardiac complications in IgAN. NT-proBNP and CITP biomarkers may help to identify IgAN patients at high risk for subclinical heart failure and further atherosclerotic disease. We found an independent association between eGFR and NT-proBNP, but not with CITP, in IgA nephropathy. There was a strong correlation between NT-proBNP, CITP, and aortic PP and PWVcf. In cases of deteriorated renal function, there was significantly higher central aortic systolic pressure.
Our knowledge of the mechanisms underlying the advancement of kidney disease, cardiovascular disease, and all-cause mortality has greatly benefited from measurements of arterial stiffness [17], which was supported by our former examinations as well [18,19].
Ohno et al. found that in CKD patients central blood pressure is a stronger predictor of CV and renal disease outcomes compared with brachial blood pressure, and should be used to guide antihypertensive therapy [20]. Our results support these observations.
In our research, we came to the same conclusion as Uterstellar et al., who identified NT-proBNP as a highly effective indicator of CV prognosis in CKD patients not receiving dialysis [13,14]. They demonstrated that NT-proBNP remains a robust predictor of outcome, notably for eGFR, even after complete adjustment for potential confounders. This observation is significant, as it has previously been claimed [12,21] that the use of this plasma biomarker as an independent CV outcome marker in CKD may be precluded by the substantial association between NT-proBNP and GFR, which is caused by decreased renal clearance of NT-proBNP in advanced CKD. The CARE for HOME study advises laboratory measurement of NT-proBNP against routine echocardiographic investigations because it is less expensive, less challenging, and takes less time than a normal echocardiographic examination [22].
Two earlier Greek studies showed a connection between altered collagen turnover (collagen type I synthesis) and increased aortic stiffness in treated hypertensive patients without left ventricular (LV) hypertrophy [23] as well as a possible link between altered collagen metabolism and peripheral vascular stiffness in chronic HF [24]; however, they did not examine the connection with renal function.
In patients with CKD, particularly those who have HFpEF, Eiros et al. described a link between LVDD and the biomarker combination of high carboxy-terminal propeptide of procollagen type-I (PICP) and low carboxy-terminal telopeptide of collagen type-I to matrix metalloproteinase-1 (CITP:MMP-1) ratio. These results suggest that CKD promotes the development of biomarker-assessed myocardial fibrosis and LVDD in hypertensive patients with HFpEF, and that changes in collagen type I metabolism as measured by the biomarkers PICP and CITP:MMP-1 ratio worsen with the transition to HFpEF in hypertensive patients [15].
We found a positive correlation between CITP and cfPWV, aortic pulse pressure, and LVMI in our cohort, which suggests an association between worsening kidney function and vascular changes, and means that altered collagen turnover (CITP) can be assumed.
Because of the high prevalence of myocardial fibrosis in patients with CKD and the direct correlation that exists between the degree of impairment of kidney function and the severity of myocardial fibrosis [25,26,27], it has been proposed that pro-fibrotic factors or pathways linked to CKD could exist [28].
Additional factors favor NT-proBNP as a plasma biomarker for CV outcome prediction in clinical practice over an echocardiography-based method. First, the cut-off values for NT-proBNP were significantly lower (150 pg/mL) than those found in echocardiographic investigations [29]. Second, little information is currently available about the therapeutic effects of screening echocardiography in people with or without CKD from prospective interventional trials. In contrast, NT-proBNP-directed cardioprotective therapy techniques have been adopted recently as a result of multiple interventional trials that have shown their superiority over clinically advised treatment strategies in both primary and secondary prevention outside the nephrology community [30,31,32,33]. Based on our data, it may be important in IgAN and CKD as well.
Liang et al. proved that systolic dysfunction and LVDD demonstrated mutually augmentative effects on CV mortality, and suggested that cardioprotection for patients with CKD should be prioritized at an early stage along with conventional nephroprotection. Therefore, cardioprotective management should be initiated as early as possible after CKD diagnosis [34].
Uremia causes structural alterations in the heart, including thickening of the intramural arteries, cardio-myocyte hypertrophy, and myocardial fibrosis. In response to the compounding effects of traditional risk factors and CKD-related risk factors, these structural changes together predispose patients to LVDD [35,36,37]. There is solid proof that variations in collagen myocardial metabolism are connected to interstitial fibrosis in CKD, however, vascular remodeling and cardiomyocyte hypertrophy may represent adaptive reactions to pressure and volume stress [38]. Other metabolic changes, including hypovitaminosis D, hyperparathyroidism, and hyperphosphatemia, are particularly important in more advanced stages of CKD and during dialysis [38,39], and might influence heart function only later. The activation of the renin-angiotensin-aldosterone system (RAAS), which may be responsible for cardiac fibrosis and hypertrophy, is another significant component. Angiotensin II and aldosterone can be involved in myocardial cell hypertrophy and fibrosis independent of afterload; however, activation of the intracardiac RAAS appears to be critically engaged in the overload situation observed in dialysis [40].
Unfortunately, we did not examine the effects of RAAS inhibitors in this study. Although we did not examine the role of these factors, these observations may explain the observed differences in the LVDD values of our IgAN patients.
4. Materials and Methods
We included 90 renal biopsy-proven IgAN patients in our cross-sectional study, and we analyzed their data retrospectively. The University of Pécs Regional Research Ethics Committee approved the study protocol, and all participants provided written consent to its completion. The inclusion criterion for the study was confirmed IgAN over the age of 18, while exclusion criteria were previous or current immunosuppressive treatment (due to the modifying effects of the biomarker) and severe comorbidities, such as malignancies that required active treatment or acute infection.
At the start of patient enrollment, echocardiography measurements were performed and classic CV risk factors, including hypertension, carbohydrate metabolism disorder, obesity, lipid abnormalities, smoking, and patient medications such as antihypertensive drugs (ACEI/ARB, BB, CCB), and statins were recorded. The obesity inclusion criterion was a BMI over 30 kg/m2. The CKD-EPI formula was used to estimate renal function (eGFR, mL/min, 1.73 m2). A 24-h blood pressure monitor using Meditech ABPM devices was relied on to determine the patient’s 24 h average systolic and diastolic blood pressure, pulse pressure, and diurnal index. Echocardiography was performed at the start of the study (see below). During these visits, medical events that had occurred since the previous visit were recorded, the patient’s physical status was examined, and detailed laboratory tests were performed. Blood pressure values were determined from the average of three measurements taken after 10 min of rest.
4.1. Biomarker Measurement
N-terminal prohormone of brain natriuretic peptide (NT-proBNP) was determined in the accredited Department of Laboratory Medicine (Universiy of Pécs, NAH-9-0008/2021), by a fully automated immonoassay method (Roche® Gmbh, Mannheim, Germany), while carboxy-terminal telopeptide of collagen type I (CITP) was measured using enzyme-linked immunosorbent assay (ELISA) kits (CITP kit by MyBioSource®, San Diego, CA, USA) in the Szentágothai Research Center (University of Pécs) following the manufacturer’s protocol.
4.2. Arterial Stiffness Measurement
Arterial stiffness was determined by measuring carotid–femoral pulse wave velocity (cfPWV). cfPWV was measured using applanation tonometry (SphygmoCor System; AtCor Medical, Sydney, Australia). Measurements were performed in the morning in the supine position after at least 10 min of rest in a quiet temperature-controlled room. Pulse wave recording was performed consecutively at two superficial artery sites (carotid–femoral segment). The cfPWV was calculated, and central aortic pressure was measured by the device. Augmentation index (Aix) is based on blood pulse-wave reflection, and is an accepted measure of arterial stiffness and risk factor for cardiovascular disease. The Aix is commonly accepted as a measure of the enhancement (augmentation) of central aortic pressure by a reflected pulse wave.
4.3. Echocardiographic Measurement
Echocardiography was performed with the Aloka SSD 1400; two operators were involved in the study. Left ventricular mass (LVM) was calculated from 2D images of the left ventricular short-axis muscle area and apical left ventricular length (LVM = (5/6 area × length)). The left ventricular mass index (LVMI) of g/m2 was calculated using Devereux’s formula, and the cardiac mass was indicated by lean mass. LVMI was determined based on the Cornell criterion and indexed for height (in meters). The left ventricular ejection fraction (LVEF) was calculated by adding the diastolic and systolic volumes of the left ventricle using the unidirectional Simpson method: EF = (Dvol-Svol)/Dvol × 100. Diastolic function was determined by mitral inflow and pulmonary venous flow based on conventional spectral Doppler measurements. We measured the ratio of the E wave to the A wave (E/A ratio), the isovolumetric relaxation time (IVRT), and the deceleration time of the E wave. LVH was defined as abnormal RWT and/or LVMI.
4.4. Statistical Analysis
Statistical analyses were performed using SPSS 21.0 software (SPSS, Inc., Chicago, IL, USA). The Kolmogorov–Smirnov test was used to determine the normality of the data distribution. Non-normally distributed parameters were transformed logarithmically. A comparison of clinical and laboratory parameters was made using Student’s t-test and ANOVA as appropriate. The mean SD was used to express data with a normal distribution. Correlations between continuous variables were evaluated with linear regression using the Pearson test, while the Spearman correlation test was used for categorical variables. Values of p > 0.05 were considered statistically significant.
5. Conclusions
In conclusion, our study confirmed that NT-proBNP both alone and combined with CITP may help identify IgAN patients at high risk for subclinical heart failure and further atherosclerotic disease. In addition, there is an effect of the modification of CKD on the association of these biomarker combinations with LVDD in HFpEF patients. A possible mechanism for the development of heart failure in CKD patients may be an elevation of central aortic systolic pressure. The pathophysiological mechanisms linking the increased prevalence of the biomarker combination with CKD, as well as its association with poor outcomes, must be investigated in larger cohorts.
Limitations of the Study
Several limitations need to be acknowledged in our study. First, the present study was a single-center cross-sectional study with a small number of patients, which could have caused selection bias. Second, we cannot say for certain that the association between increased NT-proBNP, CITP, and decreased renal function is causal in our CKD patients. Third, the renal function was estimated, not measured, although the use of eGFR is widely accepted in the literature to describe renal function. Fourth, as a consequence, the low frequency of the biomarker combination as defined in previous studies precluded the performance of subgroup analyses in the current study. In this regard, further studies should be performed to examine the correspondence between histological and biochemical aspects of myocardial fibrosis in different HF stages. Therefore, further analyses in large and independent cohorts of patients are necessary to confirm these findings. Fifth, potential problems related to multiplicity could have influenced the findings. Finally, because they are descriptive in nature, the associations found between renal disease, circulating biomarkers, and LV dysfunction do not establish causality.
Author Contributions
B.S. and conception and design of the study, collection of clinical data, drafting and approval of the manuscript. B.C.: identification of the study plot, drafting and approval of the manuscript. T.V.: identification of the study plot, classification of the biopsies, statistical analysis, review and approval of the manuscript. J.N.: drafting of the application to the ethics committee, organization of the database, review and approval of the manuscript. R.J.-C., Z.H.-S. and T.K.: immunomechanical analyses. T.J.K.: collection of clinical data, review, drafting, and approval of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
Special thanks to the Hungarian Society of Hypertension for funding our study with a scientific research grant in 2021 and to the Hungarian Society of Nephrology for funding our work with a Research Development grant in 2022.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the University of Pécs Clinical Center Regional Research Ethical Committee (Reference no. 3170/2008).
Informed Consent Statement
All the study participants provided informed consent.
Data Availability Statement
The data underlying this article cannot be shared publicly due to Hungarian regulations concerning the privacy of the individuals that participated in the study. The data can be shared on reasonable request to the corresponding author if accepted by the Regional Committee for Medical and Health Research Ethics and local Data Protection Officials.
Acknowledgments
We are grateful to Ilona Varga Sámikné for her conscientious work in the study recording of the data and arterial stiffness.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jha, V.; Garcia-Garcia, G.; Iseki, K.; Li, Z.; Naicker, S.; Plattner, B.; Saran, R.; Wang, A.Y.-M.; Yang, C.-W. Chronic kidney disease: Global dimension and perspectives. Lancet 2013, 382, 260–272. [Google Scholar] [CrossRef] [PubMed]
- Gansevoort, R.T.; Correa-Rotter, R.; Hemmelgarn, B.R.; Jafar, T.H.; Heerspink, H.J.L.; Mann, J.F.; Matsushita, K.; Wen, C.P. Chronic kidney disease and cardiovascular risk: Epidemiology, mechanisms, and prevention. Lancet 2013, 382, 339–352. [Google Scholar] [CrossRef] [PubMed]
- Kwon, C.S.; Daniele, P.; Forsythe, A.; Ngai, C. A Systematic Literature Review of the Epidemiology, Health-Related Quality of Life Impact, and Economic Burden of Immunoglobulin A Nephropathy. J. Health Econ. Outcomes Res. 2021, 8, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Kouis, P.; Kousios, A.; Kanari, A.; Kleopa, D.; Papatheodorou, S.I.; Panayiotou, A.G. Association of non-invasive measures of subclinical atherosclerosis and arterial stiffness with mortality and major cardiovascular events in chronic kidney disease: Systematic review and meta-analysis of cohort studies. Clin. Kidney J. 2019, 13, 842–854. [Google Scholar] [CrossRef]
- Romero-González, G.; González, A.; López, B.; Ravassa, S.; Díez, J. Heart failure in chronic kidney disease: The emerging role of myocardial fibrosis. Nephrol. Dial. Transplant. 2022, 37, 817–824. [Google Scholar] [CrossRef]
- Mishra, R.K.; Li, Y.; Ricardo, A.C.; Yang, W.; Keane, M.; Cuevas, M.; Christenson, R.; DeFilippi, C.; Chen, J.; He, J.; et al. Association of N-Terminal Pro-B-Type Natriuretic Peptide With Left Ventricular Structure and Function in Chronic Kidney Disease (from the Chronic Renal Insufficiency Cohort [CRIC]). Am. J. Cardiol. 2013, 111, 432–438. [Google Scholar] [CrossRef]
- Defilippi, C.R.; Fink, J.C.; Nass, C.M.; Chen, H.; Christenson, R. N-Terminal Pro-B-Type Natriuretic Peptide for Predicting Coronary Disease and Left Ventricular Hypertrophy in Asymptomatic CKD Not Requiring Dialysis. Am. J. Kidney Dis. 2005, 46, 35–44. [Google Scholar] [CrossRef]
- Paoletti, E.; De Nicola, L.; Gabbai, F.B.; Chiodini, P.; Ravera, M.; Pieracci, L.; Marre, S.; Cassottana, P.; Lucà, S.; Vettoretti, S.; et al. Associations of left ventricular hypertrophy and 1986 Clinical Journal of the American Society of Nephrology geometry with adverse outcomes in patients with CKD and hypertension. Clin. J. Am. Soc. Nephrol. 2016, 11, 271–279. [Google Scholar] [CrossRef]
- Chen, S.-C.; Chang, J.-M.; Liu, W.-C.; Huang, J.-C.; Tsai, J.-C.; Lin, M.-Y.; Su, H.-M.; Hwang, S.-J.; Chen, H.-C. Echocardiographic parameters are independently associated with increased cardiovascular events in patients with chronic kidney disease. Nephrol. Dial. Transplant. 2012, 27, 1064–1070. [Google Scholar] [CrossRef]
- Eckardt, K.-U.; Scherhag, A.; Macdougall, I.C.; Tsakiris, D.; Clyne, N.; Locatelli, F.; Zaug, M.F.; Burger, H.U.; Drueke, T.B. Left Ventricular Geometry Predicts Cardiovascular Outcomes Associated with Anemia Correction in CKD. J. Am. Soc. Nephrol. 2009, 20, 2651–2660. [Google Scholar] [CrossRef]
- Kim, S.J.; Han, S.H.; Park, J.T.; Kim, J.-K.; Oh, H.J.; Yoo, D.E.; Yoo, T.-H.; Kang, S.-W.; Choi, K.H. Left atrial volume is an independent predictor of mortality in CAPD patients. Nephrol. Dial. Transplant. 2011, 26, 3732–3739. [Google Scholar] [CrossRef]
- Scheven, L.; de Jong, P.E.; Hillege, H.L.; Heerspink, H.J.L.; van Pelt, L.J.; Kootstra, J.E.; Bakker, S.J.; Gansevoort, R.T.; for the PREVEND study group. High-sensitive troponin T and N-terminal pro-B type natriuretic peptide are associated with cardiovascular events despite the cross-sectional association with albuminuria and glomerular filtration rate. Eur. Heart J. 2012, 33, 2272–2281. [Google Scholar] [CrossRef]
- Matsushita, K.; Sang, Y.; Ballew, S.H.; Astor, B.C.; Hoogeveen, R.C.; Solomon, S.D.; Ballantyne, C.M.; Woodward, M.; Coresh, J. Cardiac and Kidney Markers for Cardiovascular Prediction in Individuals with Chronic Kidney Disease: The Atherosclerosis Risk in Communities study. Arter. Thromb. Vasc. Biol. 2014, 34, 1770–1777. [Google Scholar] [CrossRef]
- Bansal, N.; Anderson, A.H.; Yang, W.; Christenson, R.H.; Defilippi, C.R.; Deo, R.; Dries, D.L.; Go, A.S.; He, J.; Kusek, J.W.; et al. High-Sensitivity Troponin T and N-Terminal Pro-B-Type Natriuretic Peptide (NT-proBNP) and Risk of Incident Heart Failure in Patients with CKD: The Chronic Renal Insufficiency Cohort (CRIC) Study. J. Am. Soc. Nephrol. 2015, 26, 946–956. [Google Scholar] [CrossRef]
- Eiros, R.; Romero-González, G.; Gavira, J.J.; Beloqui, O.; Colina, I.; Landecho, M.F.; López, B.; González, A.; Díez, J.; Ravassa, S. Does Chronic Kidney Disease Facilitate Malignant Myocardial Fibrosis in Heart Failure with Preserved Ejection Fraction of Hypertensive Origin? J. Clin. Med. 2020, 9, 404. [Google Scholar] [CrossRef]
- Querejeta, R.; Varo, N.; López, B.; Larman, M.; Artiñano, E.; Etayo, J.C.; Ubago, J.L.M.; Gutierrez-Stampa, M.; Emparanza, J.I.; Gil, M.J.; et al. Serum Carboxy-Terminal Propeptide of Procollagen Type I Is a Marker of Myocardial Fibrosis in Hypertensive Heart Disease. Circulation 2000, 101, 1729–1735. [Google Scholar] [CrossRef]
- Townsend, R.R. Arterial Stiffness in CKD: A Review. Am. J. Kidney Dis. 2019, 73, 240–247. [Google Scholar] [CrossRef]
- Késői, I.; Sági, B.; Tóth, O.I.; Vas, T.; Fazekas, A.; Kovács, T.; Pintér, T.; Wittmann, I.; Nagy, J. Different Effect of IgA Nephropathy and Polycystic Kidney Disease on Arterial Stiffness. Kidney Blood Press. Res. 2011, 34, 158–166. [Google Scholar] [CrossRef]
- Sági, B.; Késői, I.; Késői, B.; Vas, T.; Csiky, B.; Kovács, T.; Nagy, J. Arterial stiffness may predict renal and cardiovascular prognosis in autosomal-dominant polycystic kidney disease. Physiol. Int. 2018, 105, 145–156. [Google Scholar] [CrossRef]
- Ohno, Y.; Kanno, Y.; Takenaka, T. Central blood pressure and chronic kidney disease. World J. Nephrol. 2016, 5, 90–100. [Google Scholar] [CrossRef]
- Vickery, S.; Price, C.P.; John, R.I.; Abbas, N.A.; Webb, M.C.; Kempson, M.E.; Lamb, E.J. B-Type Natriuretic Peptide (BNP) and Amino-Terminal proBNP in Patients With CKD: Relationship to Renal Function and Left Ventricular Hypertrophy. Am. J. Kidney Dis. 2005, 46, 610–620. [Google Scholar] [CrossRef] [PubMed]
- Untersteller, K.; Girerd, N.; Duarte, K.; Rogacev, K.S.; Seiler-Mussler, S.; Fliser, D.; Rossignol, P.; Heine, G.H. NT-proBNP and Echocardiographic Parameters for Prediction of Cardiovascular Outcomes in Patients with CKD Stages G2–G4. Clin. J. Am. Soc. Nephrol. 2016, 11, 1978–1988. [Google Scholar] [CrossRef]
- Stakos, D.A.; Tziakas, D.N.; Chalikias, G.K.; Mitrousi, K.; Tsigalou, C.; Boudoulas, H. Associations Between Collagen Synthesis and Degradation and Aortic Function in Arterial Hypertension. Am. J. Hypertens. 2010, 23, 488–494. [Google Scholar] [CrossRef] [PubMed]
- Chatzikyriakou, S.V.; Tziakas, D.N.; Chalikias, G.K.; Stakos, D.A.; Thomaidi, A.K.; Mitrousi, K.; Lantzouraki, A.E.; Kotsiou, S.; Maltezos, E.; Boudoulas, H. Serum levels of collagen type-I degradation markers are associated with vascular stiffness in chronic heart failure patients. Eur. J. Heart Fail. 2008, 10, 1181–1185. [Google Scholar] [CrossRef] [PubMed]
- Mall, G.; Huther, W.; Schneider, J.; Lundin, P.; Ritz, E. Diffuse Intermyocardiocytic Fibrosis in Uraemic Patients. Nephrol. Dial. Transplant. 1990, 5, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Charytan, D.M.; Padera, R.; Helfand, A.M.; Zeisberg, M.; Xu, X.; Liu, X.; Himmelfarb, J.; Cinelli, A.; Kalluri, R.; Zeisberg, E.M. Increased concentration of circulating angiogenesis and nitric oxide inhibitors induces endothelial to mesenchymal transition and myocardial fibrosis in patients with chronic kidney disease. Int. J. Cardiol. 2014, 176, 99–109. [Google Scholar] [CrossRef]
- Izumaru, K.; Hata, J.; Nakano, T.; Nakashima, Y.; Nagata, M.; Fukuhara, M.; Oda, Y.; Kitazono, T.; Ninomiya, T. Reduced Estimated GFR and Cardiac Remodeling: A Population-Based Autopsy Study. Am. J. Kidney Dis. 2019, 74, 373–381. [Google Scholar] [CrossRef]
- Frangogiannis, N.G.; Frodermann, V.; Nahrendorf, M.; Zhabyeyev, P.; Das, S.K.; Basu, R.; Shen, M.; Patel, V.B.; Kassiri, Z.; Oudit, G.Y.; et al. Matricellular Proteins in Cardiac Adaptation and Disease. Physiol. Rev. 2012, 92, 635–688. [Google Scholar] [CrossRef] [PubMed]
- Evangelista, A.; Flachskampf, F.; Lancellotti, P.; Badano, L.; Aguilar, R.; Monaghan, M.; Zamorano, J.; Nihoyannopoulos, P.; on behalf of the European Association of Echocardiography. European Association of Echocardiography recommendations for standardization of performance, digital storage and reporting of echocardiographic studies. Eur. J. Echocardiogr. 2008, 9, 438–448. [Google Scholar] [CrossRef]
- Troughton, R.W.; Frampton, C.M.; Yandle, T.G.; Espine, E.A.; Nicholls, M.G.; Richards, A.M. Treatment of heart failure guided by plasma aminoterminal brain natriuretic peptide (N-BNP) concentrations. Lancet 2000, 355, 1126–1130. [Google Scholar] [CrossRef]
- Berger, R.; Moertl, D.; Peter, S.; Ahmadi, R.; Huelsmann, M.; Yamuti, S.; Wagner, B.; Pacher, R. N-Terminal Pro–B-Type Natriuretic Peptide–Guided, Intensive Patient Management in Addition to Multidisciplinary Care in Chronic Heart Failure: A 3-Arm, Prospective, Randomized Pilot Study. J. Am. Coll. Cardiol. 2010, 55, 645–653. [Google Scholar] [CrossRef]
- Ledwidge, M.; Gallagher, J.; Conlon, C.; Tallon, E.; O’connell, E.; Dawkins, I.; Watson, C.; O’hanlon, R.; Bermingham, M.; Patle, A.; et al. Natriuretic Peptide–Based Screening and Collaborative Care for Heart Failure: The STOP-HF randomized trial. JAMA 2013, 310, 66–74. [Google Scholar] [CrossRef]
- Savarese, G.; Trimarco, B.; Dellegrottaglie, S.; Prastaro, M.; Gambardella, F.; Rengo, G.; Leosco, D.; Perrone-Filardi, P. Natriuretic Peptide-Guided Therapy in Chronic Heart Failure: A Meta-Analysis of 2,686 Patients in 12 Randomized Trials. PLoS ONE 2013, 8, e58287. [Google Scholar] [CrossRef]
- Liang, H.-Y.; Hsiao, Y.-L.; Yeh, H.-C.; Ting, I.-W.; Lin, C.-C.; Chiang, H.-Y.; Kuo, C.-C. Associations between Myocardial Diastolic Dysfunction and Cardiovascular Mortality in Chronic Kidney Disease: A Large Single-Center Cohort Study. J. Am. Soc. Echocardiogr. 2021, 35, 395–407. [Google Scholar] [CrossRef]
- London, G.M. Left ventricular alterations and end-stage renal disease. Nephrol. Dial. Transplant. 2002, 17, 29–36. [Google Scholar] [CrossRef]
- Levin, A.; Singer, J.; Thompson, C.R.; Ross, H.; Lewis, M. Prevalent left ventricular hypertrophy in the predialysis population: Identifying opportunities for intervention. Am. J. Kidney Dis. 1996, 27, 347–354. [Google Scholar] [CrossRef]
- Tyralla, K.; Amann, K. Morphology of the heart and arteries in renal failure. Kidney Int. Suppl. 2003, 63, S80–S83. [Google Scholar] [CrossRef]
- Díez, J.; Laviades, C. Hypertensive heart disease in the patient with chronic kidney disease. Nefrologia 2008, 28, 135–142. [Google Scholar]
- López, B.; González, A.; Hermida, N.; Laviades, C.; Díez, J. Myocardial fibrosis in chronic kidney disease: Potential benefits of torasemide. Kidney Int. Suppl. 2008, 74, S19–S23. [Google Scholar] [CrossRef] [PubMed]
- Glassock, R.J.; Pecoits-Filho, R.; Barberato, S.H. Left Ventricular Mass in Chronic Kidney Disease and ESRD. Clin. J. Am. Soc. Nephrol. 2009, 4 (Suppl. S1), S79–S91. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).