Abstract
Maize, one of the world’s major food crops, is facing the challenge of rising temperature. Leaf senescence is the most significant phenotypic change of maize under heat stress at the seedling stage, but the underlying molecular mechanism is still unknown. Here, we screened for three inbred lines (PH4CV, B73, and SH19B) that showed differentially senescing phenotypes under heat stress. Among them, PH4CV showed no obviously senescing phenotype under heat stress, while SH19B demonstrated a severely senescing phenotype, with B73 being between the two extremes. Subsequently, transcriptome sequencing showed that differentially expressed genes (DEGs) were generally enriched in response to heat stress, reactive oxygen species (ROS), and photosynthesis in the three inbred lines under heat treatment. Notably, ATP synthesis and oxidative phosphorylation pathway genes were only significantly enriched in SH19B. Then, the expression differences of oxidative phosphorylation pathways, antioxidant enzymes, and senescence-related genes in response to heat stress were analyzed in the three inbred lines. In addition, we demonstrated that silencing ZmbHLH51 by virus-induced gene silencing (VIGS) inhibits the heat-stress-induced senescence of maize leaves. This study helps to further elucidate the molecular mechanisms of heat-stress-induced leaf senescence at the seedling stage of maize.
1. Introduction
Heat stress, which is exacerbated by global warming, is considered to be the main cause of global crop losses [1]. To ensure crop production by efficiently improving crop varieties, it is necessary to elucidate the molecular mechanisms of crop response to heat stress [2]. Heat stress affects various physiological processes such as photosynthesis, respiration, and glucose metabolism [3]. Heat stress promotes the accumulation of reactive oxygen species (ROS), which damages the photosynthetic apparatus, particularly PSII, and eventually leads to photoinhibition [4]. In addition, heat stress can damage a plant’s cell membrane system by destroying the protein structure of cell membrane, and malondialdehyde (MDA) produced by membrane lipid peroxidation further aggravates the membrane damage [5]. Several studies have shown that plants produce misfolded proteins under heat stress, which triggers the production of ROS and, ultimately, leads to the senescence or death of plants [6,7,8,9].
Leaf senescence is an important feature of plant responses to abiotic stresses. In recent years, the increasing frequency of extreme heat waves has severely affected plant growth and development. However, the molecular mechanisms underlying leaf senescence induced by heat stress remain largely unrevealed [10]. Studies on wheat have demonstrated that high temperature accelerates leaf senescence, which in turn reduces grain filling time and leads to yield reduction [11]. Maize (Zea mays L.) is one of the most planted crops worldwide, with about one-third of the world’s population relying on maize as their main foodstuff. Moreover, maize kernels and stalks are widely used in many other products, including edible oil, starch, petrochemicals, and feed [12].
Maize is affected by heat stress at all stages of growth and development, and studies have shown that maize yields are decreasing each year as the average temperature increases [13,14]. Heat stress can have many adverse effects on maize, such as causing premature leaf senescence, reducing CO2 assimilation, and affecting flowering and pollination, and, as a result, affect grain yield [15,16]. High temperatures experienced by maize during the seedling stage can affect its yield due to heat-stress-caused damage. To explore the effects of prolonged heat stress on different maize inbred lines at the seedling stage, we carried out an initial screening and chose three differentially senescing inbred lines (PH4CV, B73, and SH19B), with B73 exhibiting an intermediate senescing phenotype, for transcriptomic analysis. A comparative transcriptome was then conducted to analyze the molecular similarities and differences among PH4CV, B73, and SH19B in response to heat stress, and several key genes were identified. In particular, we detected related genes that might be involved in heat-stress-induced leaf senescence and verified that silencing ZmbHLH51 could inhibit heat-stress-induced leaf senescence by VIGS. This dataset serves as a foundation for future studies in elucidating the molecular mechanism of heat-stress-induced maize leaf senescence.
2. Results
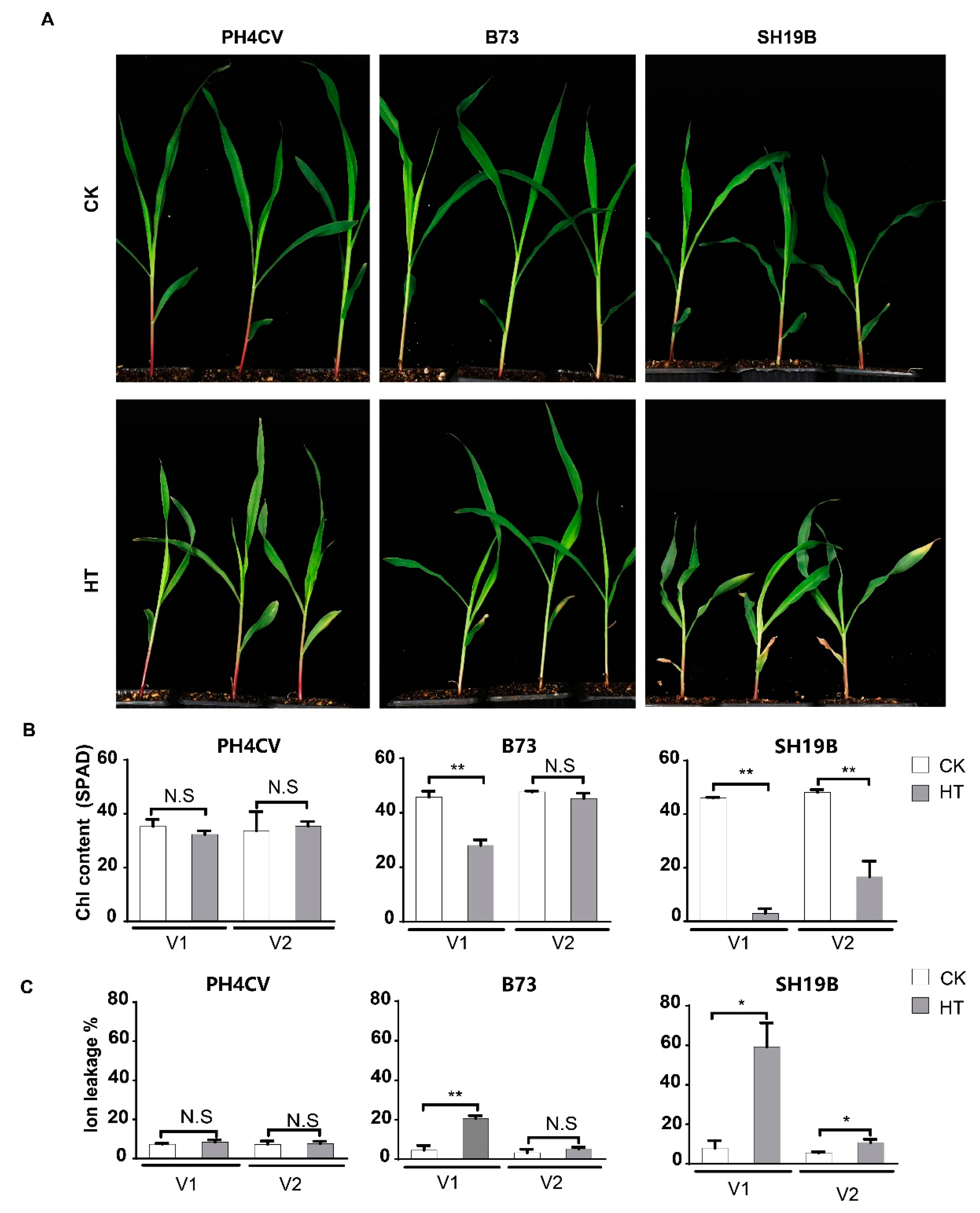
2.1. Varied Senescence Phenotypes of Different Maize Inbred Lines in Response to Heat Stress
Seven maize inbred lines were retrieved from local germplasm collections for the initial analysis of their responsiveness to heat stress. It was demonstrated that the leaves of their seedlings senesced differentially upon high-temperature treatment (42 °C day/35 °C night), and three of them with the distinct phenotypes of senescence—the least-senesced line PH4CV (paternal parent of XY335), the most-senesced line SH19B (CIMMYT inbred line), and the intermediately senesced line B73—were chosen for further analysis (Figure S1). The three-leaf stage seedlings of the three lines were treated with the day/night temperature regime for three days to precisely evaluate their phenotypic response to heat stress. Compared with the untreated controls, SH19B showed a significantly senesced phenotype in both the first lobe (V1) and the second lobe (V2), B73 displayed a significantly senesced phenotype only in V1, while PH4CV exhibited no visible senesced phenotype at all (Figure 1A). Consistently, the chlorophyll contents in both the V1 and V2 of SH19B decreased significantly, whereas a significant decrease in chlorophyll content only occurred in the V1 of B73 and no significant changes in chlorophyll content were detected in the leaves of PH4CV (Figure 1B). Accordingly, the changing trend in the ion leakage rate in the respective organs of both SH19B and B73 was just opposite; as expected, the ion leakage rate in the leaves of PH4CV remained largely similar after heat treatment (HT) (Figure 1C). These observations provoked us to explore the underlying transcriptional changes.
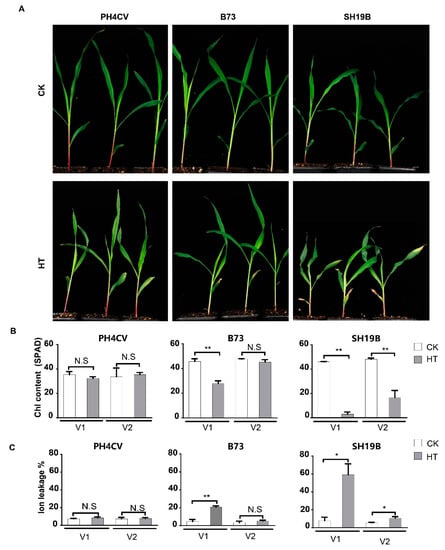
Figure 1.
Differentially senesced phenotypes of PH4CV, B73, and SH19B seedlings upon heat treatment. (A) Senesced phenotypes of heat treatment (HT) and unstressed (CK) inbred lines; (B) chlorophyll contents (SPAD) of the respective organs; (C) ion leakage rates (%) of the above organs. V1 and V2 represent the first and second lobes from the bottom, respectively. Error bars represent ±SD (n = 3 biological replicates); “N.S” indicates no significant difference, “*” represents p < 0.05, “**” represents p < 0.01.
2.2. Transcriptome Analysis of the Molecular Basis Underlying the Differentially Senescing Phynotypes in Response to Heat Stress
To explore the molecular basis of differential senescing phenotypes under heat stress, a transcriptome analysis was performed. A total of 12 cDNA libraries were constructed, which consisted of 12 leaf samples of the three inbred lines under HT as well as CK, with two biological replicates for each line. The libraries were then sequenced using Illumina Novaseq 6000 platform. A total of about 85.08 GB of clean data was finally obtained, with an average Q30 value of about 93.7% (Table S1). Using B73 genome (GCF_902167145.1) as a reference, 601 million reads were mapped. There were 84,127,307 (HT_SH19B), 87,847,433 (CK_SH19B), 80,954,154 (HT_B73), 86,897,700 (CK_B73), 87,499,572 (HT_PH4CV), and 90,918,674 (CK_PH4CV) clean paired-end reads used for future analyses (Table S2).
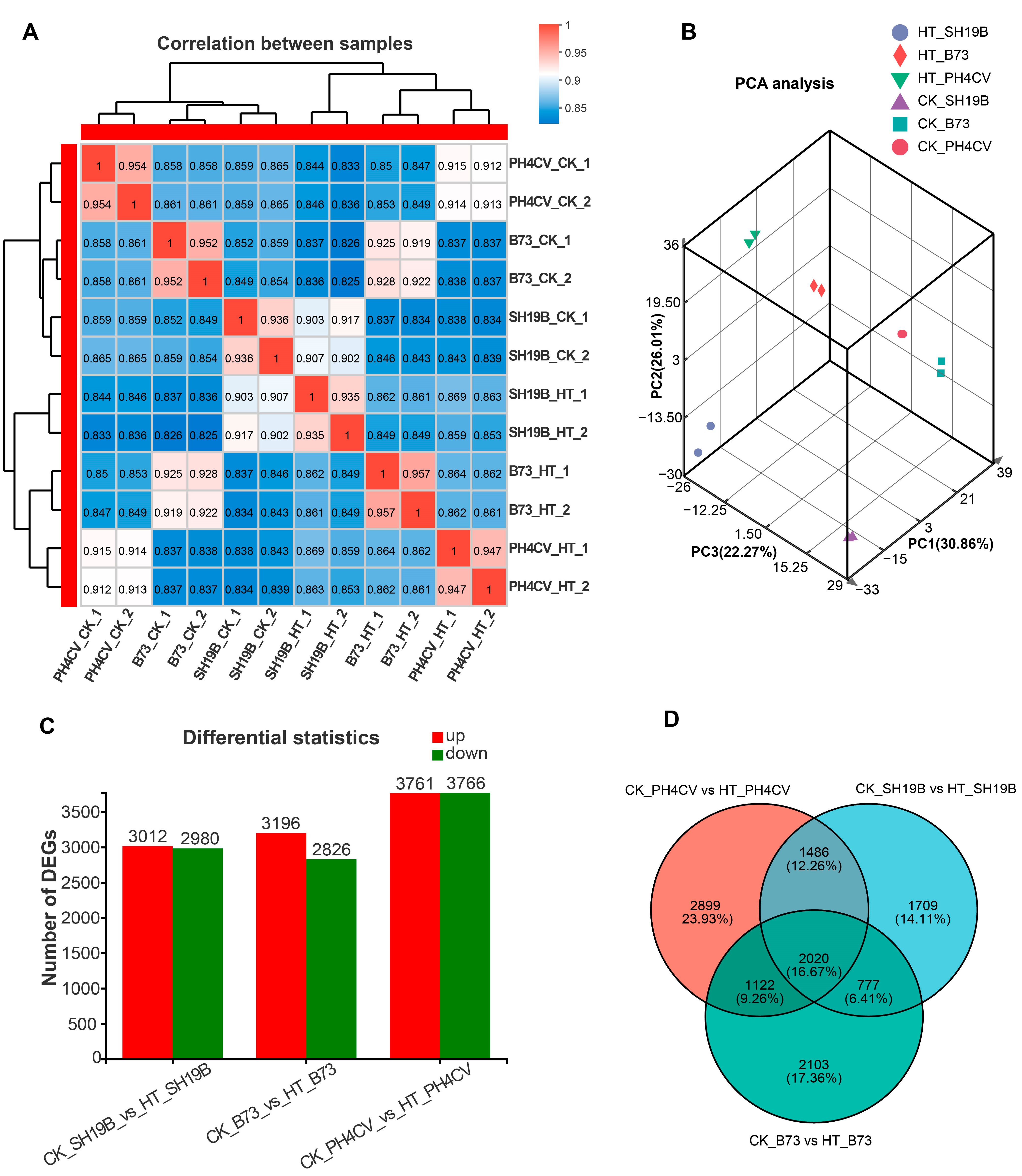
To investigate the global differences in the transcriptomes of PH4CV, B73, and SH19B leaves in response to heat stress, spearman correlation coefficient (SCC) analysis and principal component analysis (PCA) were performed on the average fragments per kilobase million (FPKM) values of all expressed genes in 12 leaf samples. After analysis, the SCC among the biological replicates was around 0.94, indicating a high quality of the replications (Figure 2A). PCA showed that the two biological replicates of each sample were closely clustered, which supported the correlation of the transcriptome sequencing results. In addition, there were differences among the inbred lines and between the HT and CK groups (Figure 2B).
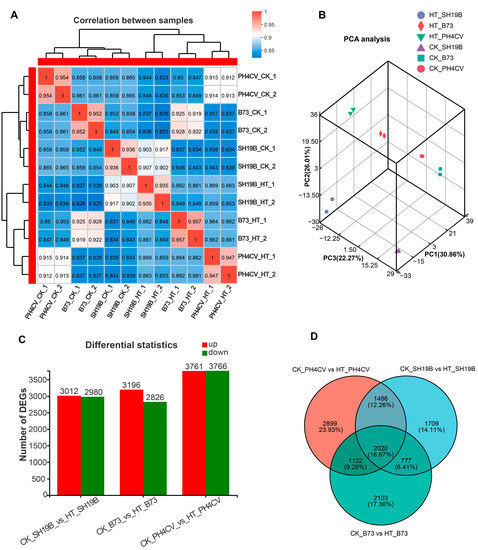
Figure 2.
Sample correlation and DEGs analysis. (A) Spearman correlation coefficient (SCC) analysis of leaf transcriptomic data of B73, PH4CV, and SH19B under HT and CK conditions; (B) principal component analysis (PCA) of sample relationship; (C) statistics of DEGs (adjusted p-value < 0.05, log2 (fold change) > 1) induced by heat stress; (D) Venn diagram of DEGs in three comparisons.
We compared the expressed genes of each inbred line (CK_PH4CV vs. HT_PH4CV, CK_B73 vs. HT_B73 and CK_SH19B vs. HT_SH19B) pairwise to determine the genes that specifically respond to heat stress. The results showed 3012 upregulated and 2980 downregulated genes in SH19B, 3196, and 2826 in B73 and 3761 and 3766 in PH4CV, respectively (Figure 2C, Supplementary File S1). Importantly, it was shown in a Venn diagram that after HT, there were 2020 genes expressed commonly in the three inbred lines, while 2899, 2103, and 1709 genes specifically responded to heat stress in PH4CV, B73, and SH19B, respectively (Figure 2D).
2.3. Common Molecular Responses of the Three Inbred Lines to Heat Stress
In order to fully understand the molecular effects of heat stress on the three inbred lines, Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses were comprehensively performed on the DEGs of CK_PH4CV vs. HT_PH4CV, CK_B73 vs. HT_B73, and CK_SH19B vs. HT_SH19B. It was found that “photosynthesis” (GO:0009768, GO:0009765, map00196, and map00195), “carbon metabolic pathway” (map00500, map00010, map00620, and map00710), “heat stress response” (GO:0009408 and GO:000) 9266), “ROS response” (GO:0000302 and GO:0042542), “osmotic stress response” (GO:0006970), and “salt stress response” (GO:0009651) were commonly enriched in all the three inbred lines (Figures S2 and S3, Supplementary File S2, Supplementary File S3).
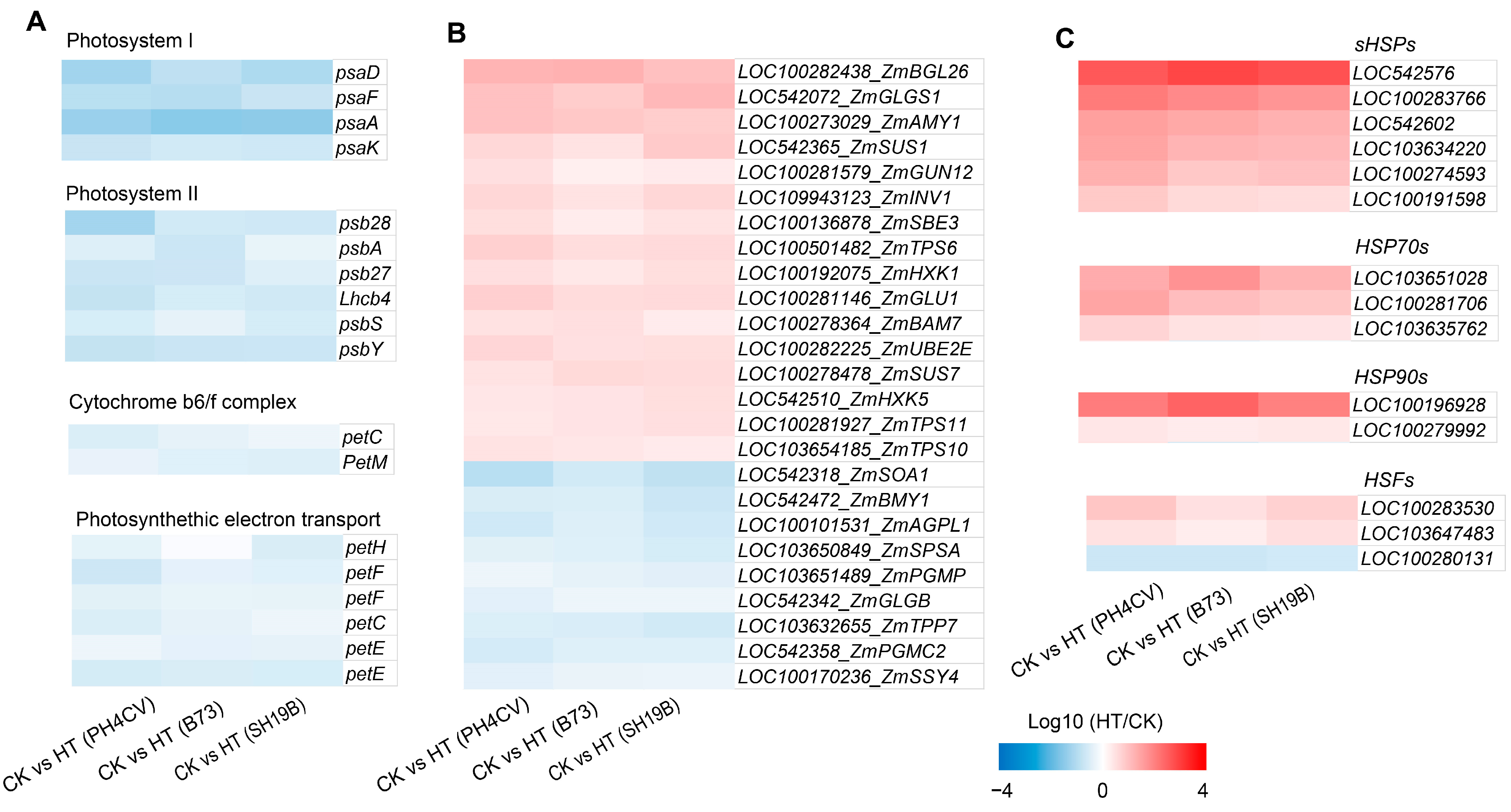
Other relevant studies revealed that heat stress can cause a decrease in the photosynthesis rate, resulting in a decrease in carbohydrate supply and an increase in carbohydrate demand [17]. We showed the effects of heat stress on photosynthesis-related genes (including those involved in photosystem I, photosystem II, light complement complex, and photosynthetic electron transport) in the three inbred lines through heat maps, and found that photosynthesis-related genes were downregulated in all the three inbred lines (Figure 3A). In addition, we found that the genes related to carbon metabolism were both upregulated and downregulated in the three inbred lines, to varying degrees (Figure 3B), indicating that heat stress had significant effects on photosynthesis and carbon metabolism processes.
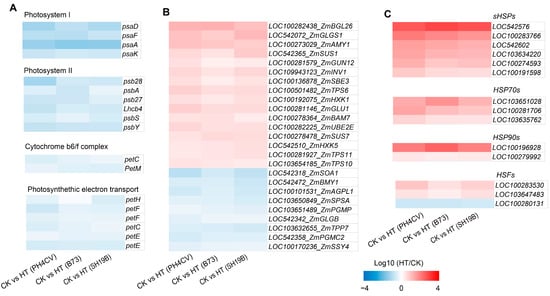
Figure 3.
Genes with consistent expression trends in response to heat stress in the three inbred lines. (A) Expressions of photosynthesis-related genes (those involved in photosystem I, photosystem II, light complement complex, and photosynthetic electron transport) in the three inbred lines (PH4CV, B73, and SH19B) under heat treatment; (B) expressions of sugar-metabolism-related genes; (C) expressions of heat-response protein genes (sHSPs, HSP70s, and HSP90s) and heat-response transcription factor genes (HSFs). Each block represents the ratio of the expression of a gene under heat treatment (HT) to untreated (CK), Log10 (HT/CK). Red indicates upregulation, and blue indicates downregulation. The scale indicates the intensity of the colors.
Recent studies have found that there is a common response mechanism between heat stress and drought, low temperature, and other stress conditions [5,15,18], indicating a close relationship between heat stress and other types of environmental stress. In addition, ROS is a common response source node under various environmental stress conditions [19]. Studies have shown that ROS can induce the expression of heat-response proteins (HSPs) [20,21]. In this study, we found that HSPs and heat-response transcription factors (HSFs) were significantly enriched in the GO terms of “ heat stress response “ (GO:0009408 and GO:0009266), “ ROS response “ (GO:0000302 and GO:0042542), “osmotic stress response “ (GO:0006970), and “ salt stress response “ (GO:0009651) (Figure S2, Supplementary File S2), and most of them were upregulated in the three inbred lines (Figure 3C). These results demonstrate a common effect of heat stress on the three inbred lines at the transcriptional level and also indicate a general effect of heat stress on maize at the seedling stage.
2.4. Distinct Molecular Responses of the Three Inbred Lines to Heat Stress
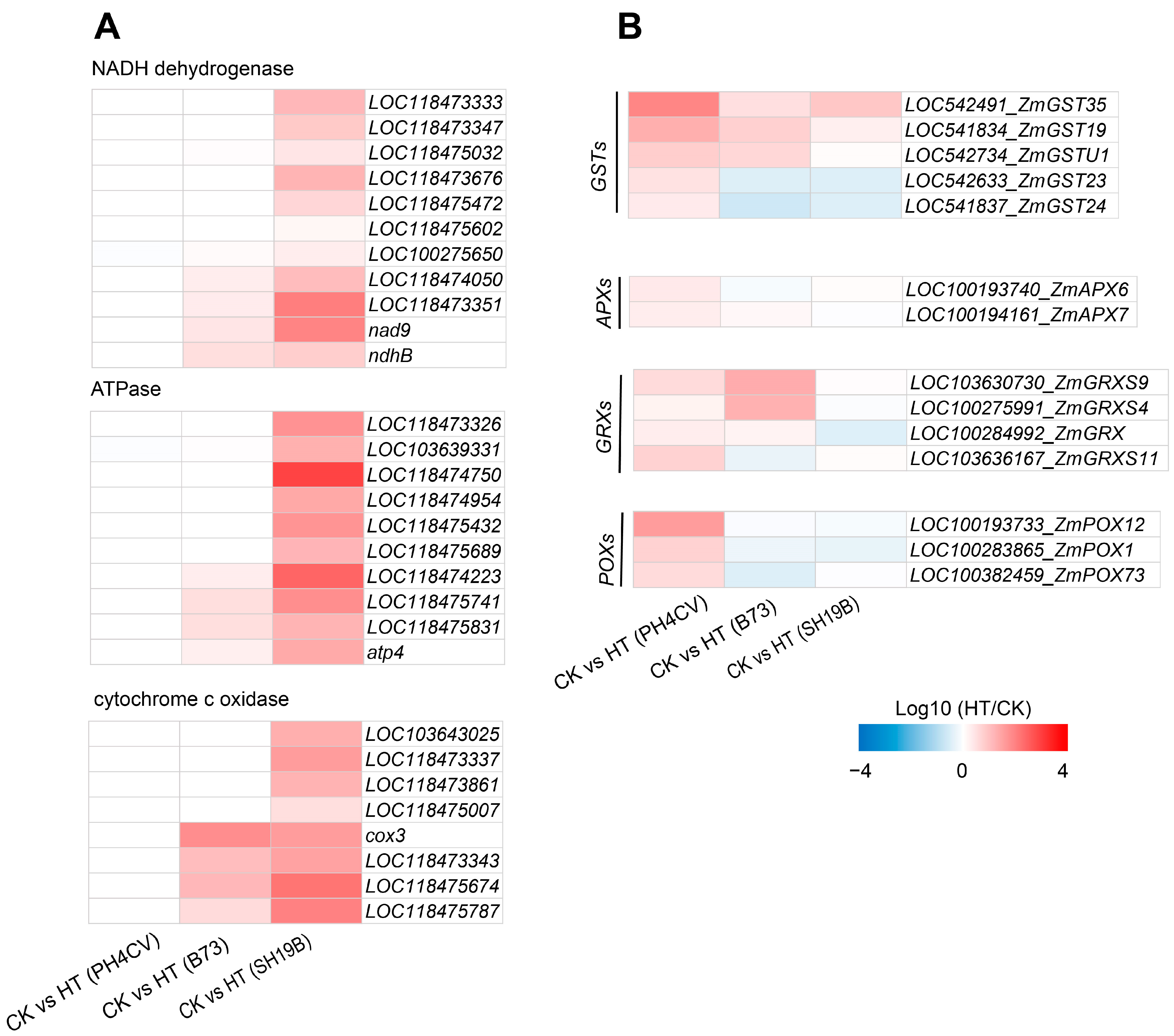
To investigate the differences in the responses of the three inbred lines to heat stress, based on the GO and KEGG analysis results, we found that genes involved in “ATP synthesis” (GO:0009145, GO:0009206, and GO:0009205) and the “oxidative phosphorylation pathway” (map00190) were significantly enriched only in CK_SH19B vs. HT_SH19B (Figures S2 and S3, Supplementary Files S2 and S3). In eukaryotes, oxidative phosphorylation is a biological process in which ATP is produced by combining ADP and inorganic phosphate via the proton gradient produced by the mitochondrial inner membrane. The proton gradient is produced by the catalysis of a variety of enzymes, including: NADH-ubiquinone oxidoreductase, ATP synthase, cytochrome c oxidase, and NADH dehydrogenase [22]. However, oxidative phosphorylation also produces ROS along with ATP, further exacerbating ROS accumulation under heat stress [23]. Several lines of evidence have shown that an increase in the ROS level under heat stress is one of the important factors leading to leaf senescence [24,25,26]. This suggests that heat stress may accelerate the senescence process of SH19B old leaves by producing excessive ROS through the oxidative phosphorylation pathway. Accordingly, we analyzed the differential expression of related genes encoding NADH dehydrogenase, ATP synthase, and cytochrome c oxidase in response to heat stress in the three inbred lines, and found that the expression of these genes was significantly upregulated in SH19B, and some of them were upregulated in B73 as well, while there were no significant changes detected in PH4CV (Figure 4A), indicating that there is a positive correlation between their expression and the senescence degree among the three inbred lines.
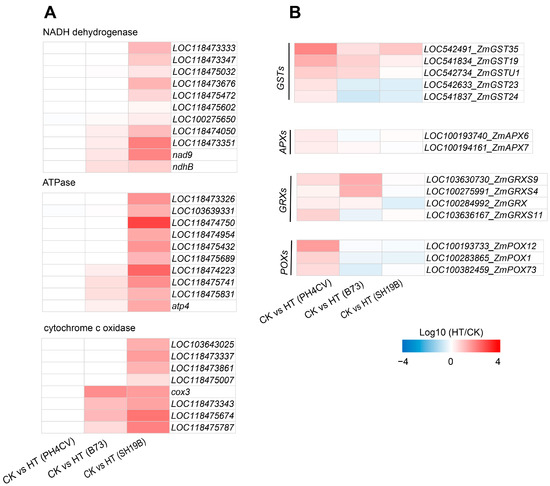
Figure 4.
DEGs in response to heat stress in the three inbred lines. (A) The differential expression of oxidative phosphorylation pathway genes (including those involved in encoding NADH dehydrogenase, ATPase, and cytochrome c) in the three inbred lines (PH4CV, B73, and SH19B) under heat treatment; (B) the differential expression of antioxidant enzyme genes (GSTs, APXs, GRXs and POXs). GST, glutathione S-transferase; APX, ascorbate peroxidase; GRX, glutaredoxin; POX, peroxidase. Each block represents the ratio of the expression of a gene under heat treatment (HT) to untreated (CK), Log10 (HT/CK). Red indicates upregulation, and blue indicates downregulation. The scale indicates the intensity of the colors.
Antioxidant enzyme systems can remove ROS and protect plant cells from oxidative damage [14,27]. In this study, a variety of enzymes of an antioxidant defense system were differentially expressed under heat stress, including glutathione S-transferase (GST), ascorbate peroxidase (APX), peroxidase (POX), and glutaredoxin (GRX). We compared the differential expression of these antioxidant oxidase genes among the three inbred lines and found that the five GSTs were all upregulated in PH4CV, while LOC542633_ZmGST23 and LOC541837_ZmGST24 were downregulated in B73 and SH19B. Two APXs (LOC100193740_ZmAPX6 and LOC100194161_ZmAPX7) were upregulated in PH4CV. All four GRXs were upregulated in PH4CV, LOC103630730_ZmGRXS9, LOC100275991_ZmGRXS4, and LOC100284992_ZmGRX were upregulated in B73, and LOC100284992_ZmGRX was downregulated in SH19B. The three POXs were upregulated in PH4CV and downregulated in B73 and SH19B (Figure 4B). These data suggest that the upregulated expression of antioxidant oxidase genes in PH4CV and B73 may be involved in inhibiting ROS-mediated leaf senescence under heat stress.
2.5. Responsive Expressions of Senescence-Related Genes in the Three Inbred Lines to Heat Stress
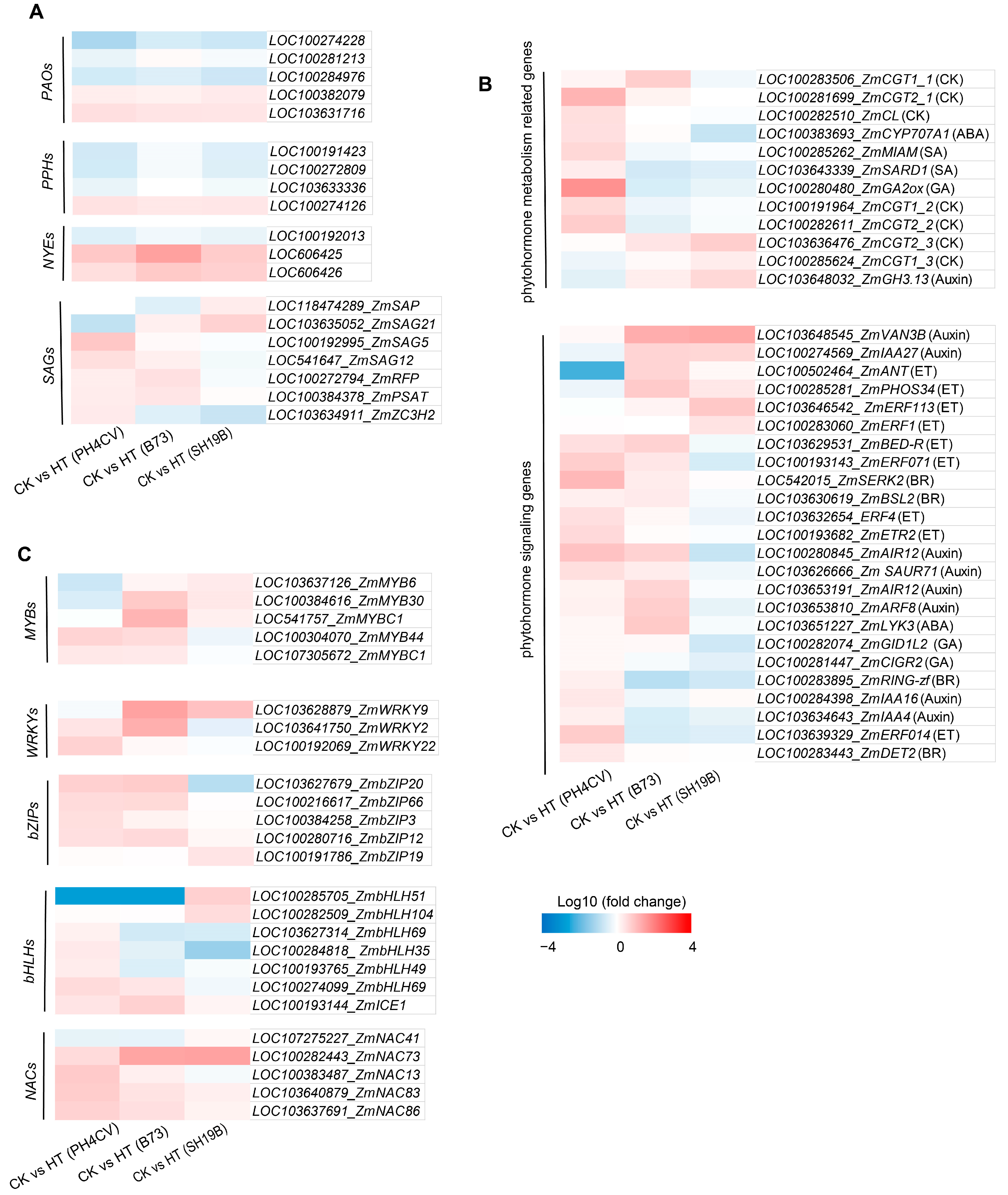
Leaf senescence-related genes include phytohormone metabolism and signal-transduction-related genes, transcription factors (TFs), Chl-catabolic genes (CCGs), and senescence-associated genes (SAGs) [28,29,30]. Phytohormones are closely related to the regulation of leaf senescence. Among them, cytokinins, auxins, and gibberellins (GAs) are involved in the negative regulation of leaf senescence, whereas ethylene (ET), abscisic acid (ABA), salicylic acid (SA), jasmonic acid (JA), and brassinosteroids (BRs) were involved in the positive regulation of leaf senescence [28,29,30]. Figure 5A shows the differential expression of phytohormone metabolism and signaling pathway genes in response to heat stress in the three inbred lines. We detected six cytokinin-O-glucosyltransferases (CGTs) genes and one carboxy-lyase (CL) gene involved in cytokinin metabolism, among which, LOC100283506_ZmCGT1_1, LOC100281699_ZmCGT2_1, and LOC100282510_ZmCL were upregulated in PH4CV or B73 but downregulated in SH19B. LOC100191964_ZmCGT1_2, and LOC100282611_ZmCGT2_2 were upregulated in PH4CV but downregulated in B73 and SH19B. LOC103636476_ZmCGT2_3, and LOC100285624_ZmCGT1_3 were downregulated in PH4CV but upregulated in B73 and SH19B. Abscisic acid 8’-hydroxylase 1 (LOC100383693_ZmCYP707A1), gibberellin oxidase 2-beta-dioxygenase (LOC100280480_ZmGA2ox), and methyl indole-3-acetate methyltransferase (LOC100285262_ZmMIAM) genes were upregulated in PH4CV but downregulated in B73 and SH19B. Moreover, the indole-3-acetic acid-amido synthetase (LOC103648032_ZmGH3.13) involved in auxin metabolism was downregulated in PH4CV but upregulated in B73 and SH19B (Figure 5A). Genes related to phytohormone signal transduction showed differential responses to heat stress in the three inbred lines (Figure 5A). Among them, LOC103648545_ZmVAN3B, and LOC100274569_ZmIAA27 involved in auxin signaling and LOC100502464_ZmANT, LOC100285281_ZmPHOS34, LOC103646542_ ZmERF113, and LOC100283060_ZmERF1 involved in ethylene signaling were upregulated in B73 or SH19B but downregulated in PH4CV; LOC542015_ZmSERK2 and LOC103630619_ZmBSL2 involved in BR signaling and LOC103651227_ZmLYK3 involved in ABA signaling were upregulated in PH4CV or B73 but downregulated in SH19B. In addition, LOC100281447_ZmCIGR2 involved in GA signaling and LOC100283895_ZmRING-zf and LOC100283443_ZmDET2 involved in BR signaling were upregulated in PH4CV but downregulated in B73 or SH19B. These results indicate that phytohormone metabolism and signaling impose significant effects on leaf senescence induced by heat stress.
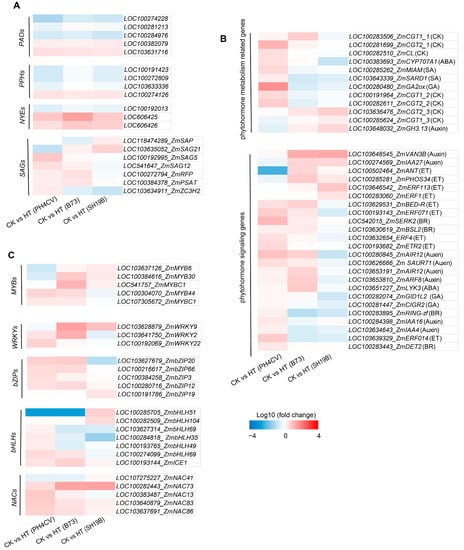
Figure 5.
Differential responses of senescence-related genes to heat stress in the three inbred lines. (A) Differential expressions of plant phytohormone metabolism and signal transduction genes in the three inbred lines (PH4CV, B73, and SH19B) after heat treatment; (B) differential expressions of TFs (MYBs, WRKYs, bZIPs, bHLHs, and NACs); (C) differential expressions of Chl-catabolic genes (CCGs) and senescence-associated genes (SAGs). Each block represents the ratio of the expression of a gene under heat treatment (HT) to untreated (CK), Log10 (HT/CK). Red indicates upregulation, and blue indicates downregulation. The scale indicates the intensity of the colors.
TFs such as NACs, bHLHs, bZIPs, WRKYs, and MYBs have been reported to be involved in regulating leaf senescence [31,32,33,34,35]. Here, we showed the differential expression of MYBs, WRKYs, bZIPs, bHLHs, and NACs in response to heat stress in three maize inbred lines (Figure 5B). Among them, LOC107275227_ZmNAC41, LOC100285705_ZmbHLH51, LOC100282509_ZmbHLH104, LOC100191786_ZmbZIP19, LOC103628879_ZmWRKY9, LOC103637126_ZmMYB6, LOC100384616_ZmMYB30, and LOC541757_ZmMYBC1 were upregulated in B73 or SH19B but downregulated in PH4CV. In addition, LOC100383487_ZmNAC13, LOC103627314_ZmbHLH69_1, LOC100284818_ZmbHLH35, LOC100193765_ZmbHLH49, LOC100274099_ZmbHLH69_2, LOC103627679_ZmbZIP20, LOC103641750_ZmWRKY2, LOC100304070_ZmMYB44, and LOC107305672_ZmMYBC1 were upregulated in PH4CV or B73 but downregulated in SH19B. These results suggest that these transcription factors, which were differentially expressed in the three inbred lines, may be involved in the regulation of leaf senescence induced by heat stress.
The chlorophyll degradation process has been largely elucidated. It consists of reaction steps catalyzed mainly by three enzymes, NYE/SGR (non-yellowing/stay-green), PPH (pheophytinase), and PAO (pheophorbide a oxygenase) [28,30]. In this study, a total of 3 NYEs, 4 PPHs, and 5 PAOs were detected, and their expression trends in response to heat stress were consistent in the three inbred lines (Figure 5A), indicating that these CCGs may not be responsible for the inconsistent senescence phenotypes of the three inbred lines under heat stress. Further analysis showed that SAGs expressions were different among the three inbred lines. LOC118474289_ZmSAP was upregulated in SH19B but downregulated in PH4CV and B73. LOC103635052_ZmSAG21 was upregulated in B73 and SH19B but downregulated in PH4CV; LOC100192995_ZmSAG5, LOC541647_ZmSAG12, LOC100272794_ZmRFP, and LOC100384378_ZmPSAT were upregulated in PH4CV and B73 but downregulated in SH19B. In addition, LOC103634911_ZmZC3H2, which encodes the CCCH zinc finger protein, was upregulated in PH4CV but downregulated in B73 and SH19B (Figure 5C). These are the possible candidate genes involved in heat-stress-induced leaf senescence.
2.6. Silencing ZmbHLH51 by VIGS Inhibits Heat-Stress-Induced Leaf Senescence
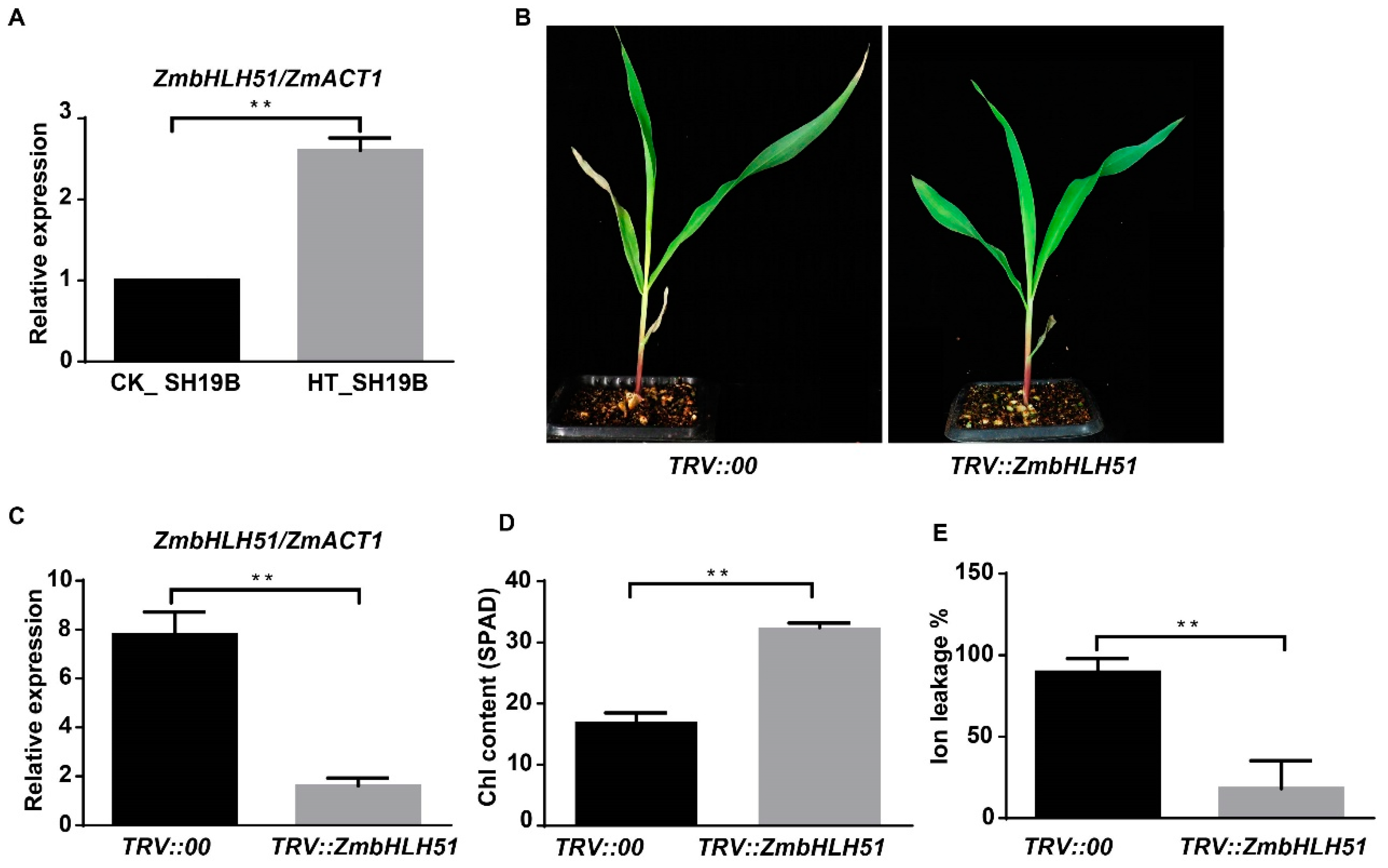
To temporarily validate the role of the TFs identified in this study on heat-stress-induced leaf senescence, a tobacco rattle virus (TRV)-induced gene silencing (VIGS) system was used. We first used maize PDS (phytoene desaturase) to validate the silencing effect of maize VIGS system [36,37]. It was found that infection with TRV::ZmPDS silenced the ZmPDS-impaired chloroplast development, resulting in a photo-bleached phenotype (Figure S4), suggesting that the VIGS system used in this study was feasible for silencing maize genes.
In order to determine the inhibitory effect of silencing TFs on leaf senescence, four TFs’ genes, LOC100191786_ZmbZIP19, LOC107275227_ZmNAC41, LOC100282509_ZmbHLH104, and LOC100285705_ZmbHLH51, which were upregulated in SH19B and downregulated in PH4CV and B73, were initially tested via VIGS experiments. Among the four tested genes, we observed senescence-inhibited phenotypes on the leaves of TRV::ZmbHLH51 plants in the background of SH19B, consistently within three biological replicates (Figure 6B). Compared with TRV::00, the expression of ZmbHLH51 in TRV::ZmbHLH51 was greatly reduced (Figure 6C), indicating that ZmbHLH51 was largely silenced. Leaves of the plants with ZmbHLH51 silenced contained higher chlorophyll contents and displayed lower ion leakage rates under heat stress (Figure 6D,E). These results indicate that the downregulation of ZmbHLH51 expression could inhibit heat-stress-induced senescence in maize leaves.
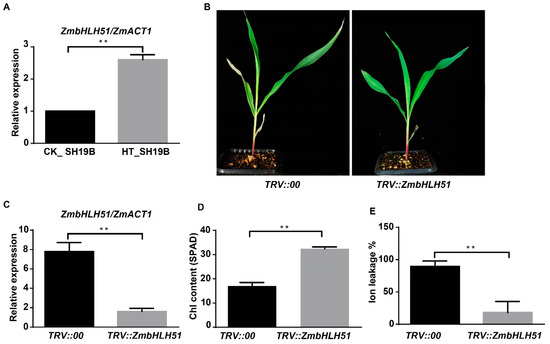
Figure 6.
VIGS silencing ZmbHLH51 inhibited leaf senescence induced by heat stress. (A) The relative expression of ZmbHLH51 in heat treated (HT) and untreated (CK) SH19B, with ZmACT1 being used as an internal reference; (B) maize seedlings infected with TRV::00 and TRV::ZmbHLH51 after heat treatment; (C) the relative expression of ZmbHLH51 in TRV::00 and TRV::ZmbHLH51 plants after heat treatment, with ZmACT1 being used as an internal reference; (D) chlorophyll content (SPAD) in V2 leaves of TRV::00 and TRV::ZmbHLH51 after heat treatment; (E) Ion leakage rate (%) in V2 leaves of TRV::00 and TRV::ZmbHLH51 after heat treatment. Error bars represent ±SD (n = 3 biological replicates); “**” means p < 0.01.
3. Discussion
Heat stress can cause leaf wilting or premature senescence of seedling-stage maize plants, decrease a plant’s chlorophyll content, and increase its ion leakage rate [7,18]. In this study, we observed the differentially senescing phenotypes of three maize inbred lines, PH4CV, B73, and SH19B, after heat stress, as evidenced via measurement of the ion leakage rate and chlorophyll content of their leaves. SH19B showed the most obvious leaf senescence symptom, while PH4CV displayed no detectable leaf senescence phenotypes (Figure 1). As far as the DEGs are concerned, there were 2899, 2103, and 1709 independent response genes in PH4CV, B73, and SH19B, respectively, with 2020 co-response genes among them (Figure 2D). Vasseur et al. (2011) pointed out that under heat-stress conditions, the plant metabolic rate increases, resulting in an increase in carbohydrate demand, a decrease in photosynthesis rate, and a decrease in carbohydrate supply [17]. Through GO and KEGG enrichment analysis, we found that the photosynthetic and glucose metabolic pathways in PH4CV, B73, and SH19B were significantly enriched (Figures S2 and S3), suggesting that heat stress could indeed affect both photosynthesis and carbon balance. Other studies have demonstrated that heat stress and drought stress [5,18] or low temperature stress [15] share a common response mechanism. In this study, we found that genes related to heat-stress response, salt-,stress response and osmotic-stress response were significantly enriched in the three inbred lines (Figure S2), indicating a close relationship between heat-stress response and salt-stress and osmotic-stress responses.
Our enrichment analysis further showed that ATP synthesis and oxidative phosphorylation pathways were significantly enriched in SH19B, which provided clues to explore the molecular basis of the differences in response to heat stress among the three inbred lines (Figure S3). Similar to our results, related transcriptome analyses have shown that oxidative phosphorylation plays an important role in response to environmental stress [38,39,40]. Mitochondria produce ATP through oxidative phosphorylation and also produce ROS, which causes damage to plant cells [41]. Studies in Arabidopsis have found that SHOT1 (inhibitor 1 of hot1-4) is a key component of the oxidative phosphorylation complex, and mutant SHOT1 reduces ROS production, which in turn reduces oxidative damage under heat stress [23]. These studies suggest that the premature senescence of SH19B under heat stress may be caused by excessive accumulation of ROS. ROS is a ubiquitous signal capable of inducing leaf senescence through protein oxidation, enzyme inactivation, altered gene expression, and biofilm breakdown [24,42]. In this study, two GO terms (GO:0000302, GO:0042542) were enriched and related to ROS responses (Figure S2).
The detoxification mechanism of plants is crucial for crop response to heat stress. In this study, a total of three POXs, five GSTs, four GRXs, and two APXs were identified, which were involved in heat stress response and were differentially expressed in the three inbred lines (Figure 4B). POXs and APXs are known to break down H2O2 in cells, thereby protecting them from damage caused by heat stress [43,44]. GSTs can combine glutathione with various heterogeneous compounds to form soluble glutathione S-conjugates to achieve plant detoxification [45]. In addition, GRXs were involved in regulating ROS levels under both normal growth and stress conditions [14]. Recent studies have shown that overexpression of Arabidopsis GRXS17 in maize alleviates heat damage and significantly increases yield compared to non-transgenic maize lines [14]. In this study, most of the antioxidant enzyme genes were upregulated in PH4CV but downregulated in SH19B (Figure 4B), indicating that PH4CV maintained a higher level of antioxidant capacity, better protected the integrity of the cell membrane, and inhibited leaf senescence induced by heat stress.
The increase of ROS level under heat stress is the main cause of SAGs expression and leaf senescence [8,24,46]. In this study, we found that multiple SAGs responded to heat stress and were differentially expressed in three inbred lines (Figure 5). Several studies have shown that phytohormones play an important role in heat-stress-induced leaf senescence [47,48,49,50]. In this study, we found that the genes related to the metabolism of cytokinins, ABA, GA, SA, and auxin respond to heat stress and are differentially expressed in the three inbred lines (Figure 5A). More importantly, the genes related to ABA, GA, SA, ET, auxin, and BR signaling in response to heat stress were also differentially expressed in the three inbred lines (Figure 5A), indicating that these phytohormone metabolism and signal-transduction-related genes may be involved in heat-stress-induced maize leaf senescence. In addition, TFs are widely involved in complex cascades of signaling induced by hormonal signaling and environmental stresses [51]. In this study, we found that numerous genes of the NACs, bHLHs, bZIPs, WRKYs, and MYBs TF families responded to heat stress and were differentially expressed in the three inbred lines (Figure 5B). Notably, quite a few SAGs were differentially expressed in the three inbred lines (Figure 5C), indicating that long-term heat stress may regulate leaf senescence in maize through direct induction of SAGs expression. In Arabidopsis, bHLH family TFs PIF4 and PIF5 were found to effectively promote heat-stress-induced leaf senescence through hormonal signaling [10]. In this study, silencing ZmbHLH51, which was upregulated in SH19B in response to heat stress, by VIGS inhibited leaf senescence (Figure 6), suggesting that ZmbHLH51 is involved in promoting heat-stress-induced leaf senescence. These data provide a foundation for further analysis of the cascade signals of leaf senescence induced by heat stress in maize.
4. Materials and Methods
4.1. Maize Inbred Lines and Heat Stress
B73, SH19B, and SWL01 inbred lines were kindly gifted by Dr. Yuan Guan (Crop Breeding and Cultivation Research Institute, Shanghai Academy of Agricultural Sciences, Shanghai, China); PH4CV, PHW03, Aijin525 and PHR36 inbred lines were kindly gifted by Dr. Huiyong Li (Cereal Crop Research Institute, Henan Academy of Agricultural Sciences, Zhengzhou, China). The sterilized maize seeds were soaked in water for 1 day and germinated for 14 days under 25 °C, 60% relative humidity, 16 h light/8 h darkness. Two-week-old seedlings were treated under a 42 °C/35 °C day/night cycle for 2 d or 3 d. V1 and V2 leaves experiencing the heat stress for 2 d were collected and subsequently used for transcriptomic analysis. The chlorophyll content and ion leakage rate of maize leaves were measured and calculated by SPAD-502 PLUS and digital conductivity meter (Waterproof ECTestr11+, MultiRange), respectively.
4.2. cDNA Library Preparation and Illumina Sequencing
RNA was extracted from 12 leaf samples of three maize inbred lines. RNA quality was monitored by agarose-gel electrophoresis. RNA concentration was checked by Nanodrop2000 (NanoDrop Technologies, Wilmington, CA, USA). RNA integrity was further checked by Agilent 2100 bioanalyzer (Agilent Technologies, Santa Clara, CA, USA). Transcriptome sequencing was performed on Illumina Novaseq 6000 sequencing platform (Majorbio, Shanghai, China).
4.3. Transcriptome Sequencing Data Analysis
Fastp was used for quality detection, including deletion of joint sequences, low quality read segments, high N-rate (N indicates uncertain base information) sequences, and excessively short sequences. Then, sequence alignment analysis was performed by TopHat2 (http://tophat.cbcb.umd.edu/, accessed on 15 March 2022) with reference to the maize B73 genome (Zm-B73-REFERENCE-NAM-5.0). Cufflinks software (Version 2.2.1, http://coletrapnelllab.github.io/cufflinks/, accessed on 15 March 2022) was used to assemble and splice the matched sequences and compare them with known transcripts. Genes were annotated by NR (http://ftp.ncbi.nlm.nih.gov/blast/db/, accessed on 20 March 2022), KEGG (http://www.genome.jp/kegg/, accessed on 20 March 2022), Swiss-Prot (http://web.expasy.org/docs/swiss-prot_guideline.html, accessed on 20 March 2022), eggNOG (http://www.ncbi.nlm.nih.gov/COG/, accessed on 20 March 2022), and Pfam (http://pfam.xfam.org/, accessed on 15 March 2022). SCC and PCA were used to analyze the correlation and variation size of samples. RSEM was used to calculate the fragments per kilobase of transcript per million mapped reads (FPKM) [52]. DEGseq2 was used to analyze the identify of DEGs with the criterion of log2 (fold change) > 1 and adjusted p-value < 0.05 [53]. Venn was used to analyze the number of overlapping genes between samples. Goatools (https://github.com/tanghaibao/GOatools, accessed on 23 March 2022) and KOBAS (http://kobas.cbi.pku.edu.cn/home.do, accessed on 23 March 2022) were used for GO and KEGG enrichment analysis. By Fisher’s exact test and BH (FDR) multiple test, the corrected p-value < 0.05 was considered as a significant enrichment item.
4.4. qRT-PCR
The relative expression of maize genes was detected by qRT-PCR. In brief, cDNA synthesis was performed using a RT Kit with gDNA Clean for qPCR II (Accurate Biology, Changsha, China). The transcript levels of each gene were measured using a 20 μL volume of SYBR (Accurate Biology, Changsha, China) on a CFX96 real-time PCR assay system (Bio-Rad, Hercules, CA, USA). qRT-PCR was performed under the following conditions: initial denaturation at 95 °C for 10 min, 95 °C for 20 s, 60 °C for 30 s, and 72 °C for 20 s for 40 cycles. We used 2−∆∆Ct method to calculate the relative expression of RT-qPCR. Primers used for RT-qPCR are summarized in Table S3. ZmActin was used as an internal control [54].
4.5. VIGS
VIGS experiments were performed as described previously [36,37]. A specific silencing sequence of target gene was screened by the SGN-VIGS tool (https://vigs.solgenomics.net/, accessed on 23 March 2022) and inserted into pTRV2. A. tumefaciens strain GV3101 carrying pTRV1- and pTRV2-derived vectors (TRV2::PDS, TRV2::ZmbHLH51) were cultured in LB medium to (OD600) 1.0. Then, pTRV1 and pTRV2 agroinfiltration liquids were mixed in equal volume, adding acetosyringone (20 mg L−1), cysteine (400 mg L−1), and Tween 20 (5 mL L−1). Three-day-old maize seedlings were completely soaked in mixed agroinfiltration liquid, then infiltrated for 10 min with vacuum assistance. The mixture was placed in a flask and co-cultured at 28 °C for 10 h. After co-culture, the seeds were cleaned with sterile water and planted in a light incubator. We verified the silencing effect of the maize VIGS system by ZmPDS. Primers used in VIGS are summarized in Table S4.
4.6. Statistical Analysis
In this study, the GraphPad Prism 6.0 software was used to process data and graphs. The SPSS software (version 20; SPSS, Inc., Chicago, IL, USA) was used for analysis of variance. Gene expression heat maps were created in MS Excel using a three-color scale in conditional format.
Supplementary Materials
The following supporting information can be downloaded at https://www.mdpi.com/article/10.3390/ijms24129782/s1.
Author Contributions
X.H. conducted all the experiments and wrote the manuscript. D.Z., H.H., Y.L. and Z.Z. directed and revised the manuscript. B.K. designed the experiment and directed and revised the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (32070323) and by the Shanghai Agriculture Applied Technology Development Program, China (Z20190101).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The raw datasets generated by transcriptome sequencing in this study are available at BioProject: PRJNA947789 (http://www.ncbi.nlm.nih.gov/bioproject/947789, accessed on 1 April 2023).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jiang, J.; Liu, X.; Liu, C.; Liu, G.; Li, S.; Wang, L. Integrating Omics and Alternative Splicing Reveals Insights into Grape Response to High Temperature. Plant Physiol. 2017, 173, 1502–1518. [Google Scholar] [CrossRef] [PubMed]
- Guihur, A.; Rebeaud, M.E.; Goloubinoff, P. How do plants feel the heat and survive? Trends Biochem. Sci. 2022, 47, 824–838. [Google Scholar] [CrossRef] [PubMed]
- Dogru, A. Effects of heat stress on photosystem II activity and antioxidant enzymes in two maize cultivars. Planta 2021, 253, 85. [Google Scholar] [CrossRef]
- Gururani, M.A.; Venkatesh, J.; Lam-Son Phan, T. Regulation of Photosynthesis during Abiotic Stress-Induced Photoinhibition. Mol. Plant. 2015, 8, 1304–1320. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Zhang, D.; Zhao, Y.; Wang, W.; Yang, H.; Tai, F.; Li, C.; Hu, X. The Difference of Physiological and Proteomic Changes in Maize Leaves Adaptation to Drought, Heat, and Combined Both Stresses. Front. Plant Sci. 2016, 7, 1471. [Google Scholar] [CrossRef] [PubMed]
- Ding, Y.; Shi, Y.; Yang, S. Molecular Regulation of Plant Responses to Environmental Temperatures. Mol. Plant. 2020, 13, 544–564. [Google Scholar] [CrossRef]
- Frey, F.P.; Urbany, C.; Hüttel, B.; Reinhardt, R.; Stich, B. Genome-wide expression profiling and phenotypic evaluation of European maize inbreds at seedling stage in response to heat stress. BMC Genom. 2015, 16, 123. [Google Scholar] [CrossRef]
- Qian, Y.; Cao, L.; Zhang, Q.; Amee, M.; Chen, K.; Chen, L. SMRT and Illumina RNA sequencing reveal novel insights into the heat stress response and crosstalk with leaf senescence in tall fescue. BMC Plant Biol. 2020, 20, 366. [Google Scholar] [CrossRef]
- Lee, M.H.; Kim, K.-M.; Sang, W.-G.; Kang, C.-S.; Choi, C. Comparison of Gene Expression Changes in Three Wheat Varieties with Different Susceptibilities to Heat Stress Using RNA-Seq Analysis. Int. J. Mol. Sci. 2022, 23, 10734. [Google Scholar] [CrossRef]
- Li, N.; Bo, C.; Zhang, Y.; Wang, L. Phytochrome Interacting Factors PIF4 and PIF5 promote heat stress induced leaf senescence in Arabidopsis. J. Exp. Bot. 2021, 72, 4577–4589. [Google Scholar] [CrossRef]
- Stratonovitch, P.; Semenov, M.A. Heat tolerance around flowering in wheat identified as a key trait for increased yield potential in Europe under climate change. J. Exp. Bot. 2015, 66, 3599–3609. [Google Scholar] [CrossRef] [PubMed]
- Tenaillon, M.I.; Charcosset, A. A European perspective on maize history. Cr. Biol. 2011, 334, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Lobell, D.B.; Field, C.B. Global scale climate—Crop yield relationships and the impacts of recent warming. Environ. Res. Lett. 2007, 2, 014002. [Google Scholar] [CrossRef]
- Sprague, S.A.; Tamang, T.M.; Steiner, T.; Wu, Q.; Hu, Y.; Kakeshpour, T.; Park, J.; Yang, J.; Peng, Z.; Bergkamp, B.; et al. Redox-engineering enhances maize thermotolerance and grain yield in the field. Plant Biotechnol. J. 2022, 20, 1819–1832. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Li, Y.; Zhang, Y.; Gou, Z.; Qi, X.; Zhang, J. Transcriptomic Analysis Revealed the Common and Divergent Responses of Maize Seedling Leaves to Cold and Heat Stresses. Genes 2020, 11, 881. [Google Scholar] [CrossRef]
- Gong, F.; Wu, X.; Zhang, H.; Chen, Y.; Wang, W. Making better maize plants for sustainable grain production in a changing climate. Front. Plant Sci. 2015, 6, 835. [Google Scholar] [CrossRef]
- Vasseur, F.; Pantin, F.; Vile, D. Changes in light intensity reveal a major role for carbon balance in Arabidopsis responses to high temperature. Plant Cell Environ. 2011, 34, 1563–1576. [Google Scholar] [CrossRef]
- Waititu, J.K.; Zhang, X.; Chen, T.; Zhang, C.; Zhao, Y.; Wang, H. Transcriptome Analysis of Tolerant and Susceptible Maize Genotypes Reveals Novel Insights about the Molecular Mechanisms Underlying Drought Responses in Leaves. Int. J. Mol. Sci. 2021, 22, 6980. [Google Scholar] [CrossRef]
- Tomanek, L. Proteomics to study adaptations in marine organisms to environmental stress. J. Proteom. 2014, 105, 92–106. [Google Scholar] [CrossRef]
- Li, H.; Liu, S.-S.; Yi, C.-Y.; Wang, F.; Zhou, J.; Xia, X.-J.; Shi, K.; Zhou, Y.-H.; Yu, J.-Q. Hydrogen peroxide mediates abscisic acid-induced HSP70 accumulation and heat tolerance in grafted cucumber plants. Plant Cell Environ. 2014, 37, 2768–2780. [Google Scholar] [CrossRef]
- Li, B.; Gao, K.; Ren, H.; Tang, W. Molecular mechanisms governing plant responses to high temperatures. J. Integr. Plant Biol. 2018, 60, 757–779. [Google Scholar] [CrossRef]
- Ghifari, A.S.; Murcha, M.W. Proteolytic regulation of mitochondrial oxidative phosphorylation components in plants. Biochem. Soc. Trans. 2022, 50, 1119–1132. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Schulz, V.; Brings, L.; Schoeller, T.; Kuehn, K.; Vierling, E. mTERF18 and ATAD3 are required for mitochondrial nucleoid structure and their disruption confers heat tolerance in Arabidopsis thaliana. New Phytol. 2021, 232, 2026–2042. [Google Scholar] [CrossRef] [PubMed]
- Jajic, I.; Sarna, T.; Strzalka, K. Senescence, Stress, and Reactive Oxygen Species. Plants 2015, 4, 393–411. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, C.; Li, K.; Li, X.; Xu, M.; Guo, Y. CLE14 functions as a “brake signal”to suppress age-dependent and stress-induced leaf senescence by promoting JUB1-mediated ROS scavenging in Arabidopsis. Mol. Plant 2022, 15, 179–188. [Google Scholar] [CrossRef]
- Zhang, M.; An, P.; Li, H.; Wang, X.; Zhou, J.; Dong, P.; Zhao, Y.; Wang, Q.; Li, C. The miRNA-Mediated Post-Transcriptional Regulation of Maize in Response to High Temperature. Int. J. Mol. Sci. 2019, 20, 1754. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants. Plant Physiol. Bioch. 2010, 48, 909–930. [Google Scholar] [CrossRef] [PubMed]
- Kuai, B.; Chen, J.; Hortensteiner, S. The biochemistry and molecular biology of chlorophyll breakdown. J. Exp. Bot. 2018, 69, 751–767. [Google Scholar] [CrossRef]
- Huang, P.; Li, Z.; Guo, H. New Advances in the Regulation of Leaf Senescence by Classical and Peptide Hormones. Front. Plant Sci. 2022, 13, 923136. [Google Scholar] [CrossRef]
- Zhu, X.; Chen, J.; Qiu, K.; Kuai, B. Phytohormone and Light Regulation of Chlorophyll Degradation. Front. Plant Sci. 2017, 8, 1911. [Google Scholar] [CrossRef]
- Song, Y.; Yang, C.; Gao, S.; Zhang, W.; Li, L.; Kuai, B. Age-Triggered and Dark-Induced Leaf Senescence Require the bHLH Transcription Factors PIF3, 4, and 5. Mol. Plant 2014, 7, 1776–1787. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Chen, J.; Xie, Z.; Gao, J.; Ren, G.; Gao, S.; Zhou, X.; Kuai, B. Jasmonic acid promotes degreening via MYC2/3/4-and ANAC019/055/072-mediated regulation of major chlorophyll catabolic genes. Plant J. 2015, 84, 597–610. [Google Scholar] [CrossRef]
- Gao, S.; Gao, J.; Zhu, X.; Song, Y.; Li, Z.; Ren, G.; Zhou, X.; Kuai, B. ABF2, ABF3, and ABF4 Promote ABA-Mediated Chlorophyll Degradation and Leaf Senescence by Transcriptional Activation of Chlorophyll Catabolic Genes and Senescence-Associated Genes in Arabidopsis. Mol. Plant 2016, 9, 1272–1285. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhu, Z.; Gao, J.; Zhou, X.; Zhu, S.; Wang, X.; Wang, X.; Ren, G.; Kuai, B. The NPR1-WRKY46-WRKY6 signaling cascade mediates probenazole/salicylic acid-elicited leaf senescence in Arabidopsis thaliana. J. Integr. Plant Biol. 2021, 63, 924–936. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Zhi, F.; Min, Y.; Ma, R.; Ge, A.; Wang, S.; Wang, J.; Liu, Z.; Guo, Y.; Chen, M. The MYB59 transcription factor negatively regulates salicylic acid- and jasmonic acid-mediated leaf senescence. Plant Physiol. 2022, 192, 488–503. [Google Scholar] [CrossRef]
- Zhou, C.; Ma, Z.; Lu, X.; Zhu, L.; Yan, C. Pseudomonas fluorescens MZ05 Enhances Resistance against Setosphaeria turcica by Mediating Benzoxazinoid Metabolism in the Maize Inbred Line Anke35. Agriculture 2020, 10, 32. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, D.; Zhang, Y.; Liu, K.; Xu, K.; Zhang, F.; Wang, J.; Tan, G.; Nie, X.; Ji, Q.; et al. Vacuum and Co-cultivation Agroinfiltration of (Germinated) Seeds Results in Tobacco Rattle Virus (TRV) Mediated Whole-Plant Virus-Induced Gene Silencing (VIGS) in Wheat and Maize. Front. Plant Sci. 2017, 8, 393. [Google Scholar] [CrossRef]
- Wang, L.; Du, M.; Wang, B.; Duan, H.; Zhang, B.; Wang, D.; Li, Y.; Wang, J. Transcriptome analysis of halophyte Nitraria tangutorum reveals multiple mechanisms to enhance salt resistance. Sci. Rep. 2022, 12, 14031. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, P.; Ji, J.; Long, H.; Wu, X. Transcriptome analysis reveals the molecular mechanism of yield increases in maize under stable soil water supply. PLoS ONE 2021, 16, e0257756. [Google Scholar] [CrossRef]
- Tao, M.; Zhu, W.; Han, H.; Liu, S.; Liu, A.; Li, S.; Fu, H.; Tian, J. Mitochondrial proteomic analysis reveals the regulation of energy metabolism and reactive oxygen species production in Clematis terniflora DC. leaves under high-level UV-B radiation followed by dark treatment. J. Proteom. 2022, 254, 104410. [Google Scholar] [CrossRef]
- Kacprzak, S.M.; Van Aken, O. Carbon starvation, senescence and specific mitochondrial stresses, but not nitrogen starvation and general stresses, are major triggers for mitophagy in Arabidopsis. Autophagy 2022, 18, 2894–2912. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, J.; Huang, L.; Leng, Y.; Dai, L.; Rao, Y.; Chen, L.; Wang, Y.; Tu, Z.; Hu, J.; et al. PGL, encoding chlorophyllide a oxygenase 1, impacts leaf senescence and indirectly affects grain yield and quality in rice. J. Exp. Bot. 2016, 67, 1297–1310. [Google Scholar] [CrossRef]
- Wu, D.-C.; Zhu, J.-F.; Shu, Z.-Z.; Wang, W.; Yan, C.; Xu, S.-B.; Wu, D.-X.; Wang, C.-Y.; Dong, Z.-R.; Sun, G. Physiological and transcriptional response to heat stress in heat-resistant and heat-sensitive maize (Zea mays L.) inbred lines at seedling stage. Protoplasma 2020, 257, 1615–1637. [Google Scholar] [CrossRef]
- Nadarajah, K.K. ROS Homeostasis in Abiotic Stress Tolerance in Plants. Int. J. Mol. Sci. 2020, 21, 5208. [Google Scholar] [CrossRef] [PubMed]
- Marrs, K.A. The functions and regulation of glutathione S-transferases in plants. Annu. Rev. Plant Phys. 1996, 47, 127–158. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, H.; Huang, X.; Xing, J.; Yao, J.; Yin, T.; Jiang, J.; Wang, P.; Xu, B. STAYGREEN-mediated chlorophyll a catabolism is critical for photosystem stability during heat-induced leaf senescence in perennial ryegrass. Plant Cell Environ. 2022, 45, 1412–1427. [Google Scholar] [CrossRef] [PubMed]
- Abdelrahman, M.; El-Sayed, M.; Jogaiah, S.; Burritt, D.J.; Tran, L.P. The “STAY-GREEN” trait and phytohormone signaling networks in plants under heat stress. Plant Cell Rep. 2017, 36, 1009–1025. [Google Scholar] [CrossRef] [PubMed]
- Hays, D.B.; Do, J.H.; Mason, R.E.; Morgan, G.; Finlayson, S.A. Heat stress induced ethylene production in developing wheat grains induces kernel abortion and increased maturation in a susceptible cultivar. Plant Sci. 2007, 172, 1113–1123. [Google Scholar] [CrossRef]
- Clarke, S.M.; Cristescu, S.M.; Miersch, O.; Harren, F.J.M.; Mur, L.A.J. Jasmonates act with salicylic acid to confer basal thermotolerance in Arabidopsis thaliana. New Phytol. 2010, 182, 175–187. [Google Scholar] [CrossRef]
- Jespersen, D.; Yu, J.J.; Huang, B.R. Metabolite Responses to Exogenous Application of Nitrogen, Cytokinin, and Ethylene Inhibitors in Relation to Heat-Induced Senescence in Creeping Bentgrass. PLoS ONE 2015, 10, e0123744. [Google Scholar] [CrossRef]
- Devireddy, A.R.; Tschaplinski, T.J.; Tuskan, G.A.; Muchero, W.; Chen, J.-G. Role of Reactive Oxygen Species and Hormones in Plant Responses to Temperature Changes. Int. J. Mol. Sci. 2021, 22, 8843. [Google Scholar] [CrossRef]
- Li, B.; Dewey, C.N. RSEM: Accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinform. 2011, 12, 323. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wang, C.; Qiu, K.; Chen, H.; Li, Z.; Li, X.; Song, J.; Wang, X.; Gao, J.; Kuai, B.; et al. The transcription factorZmNAC126accelerates leaf senescence downstream of the ethylene signalling pathway in maize. Plant Cell Environ. 2020, 43, 2287–2300. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).