Antiplatelet Effects of Selected Xanthine-Based Adenosine A2A and A2B Receptor Antagonists Determined in Rat Blood
Abstract
:1. Introduction
2. Results
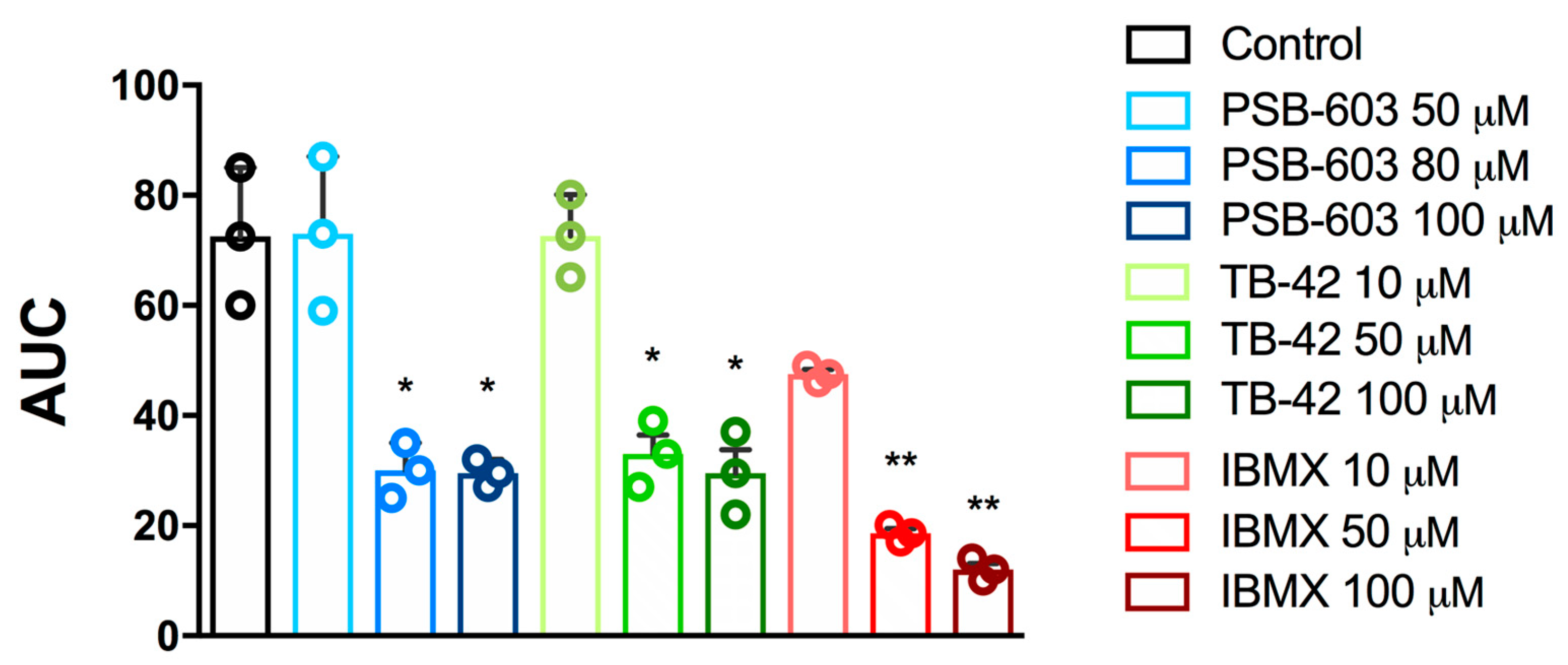
2.1. Evaluation of In Vitro Antiplatelet Activity
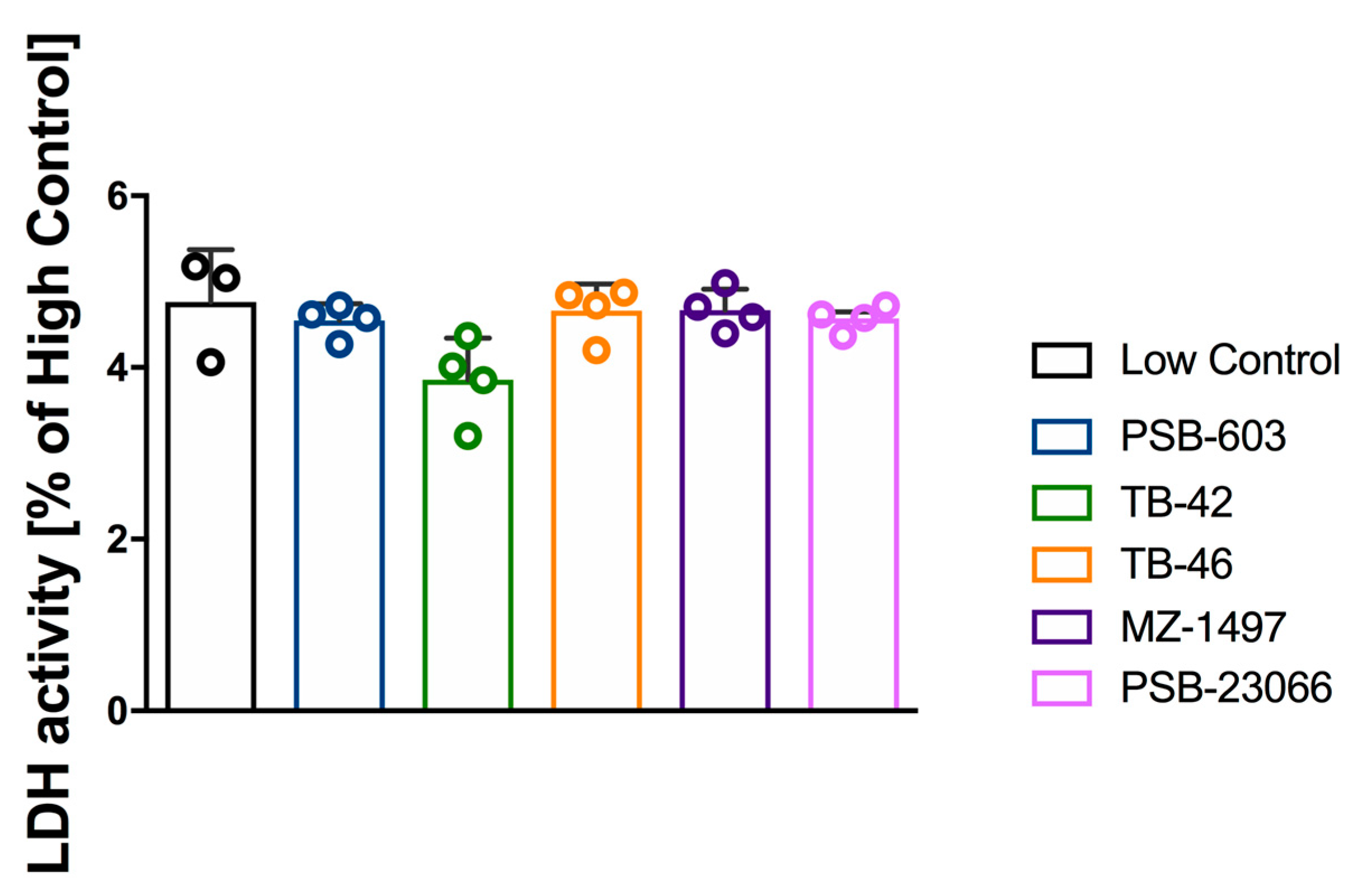
2.2. Evaluation of Platelet Cytotoxicity
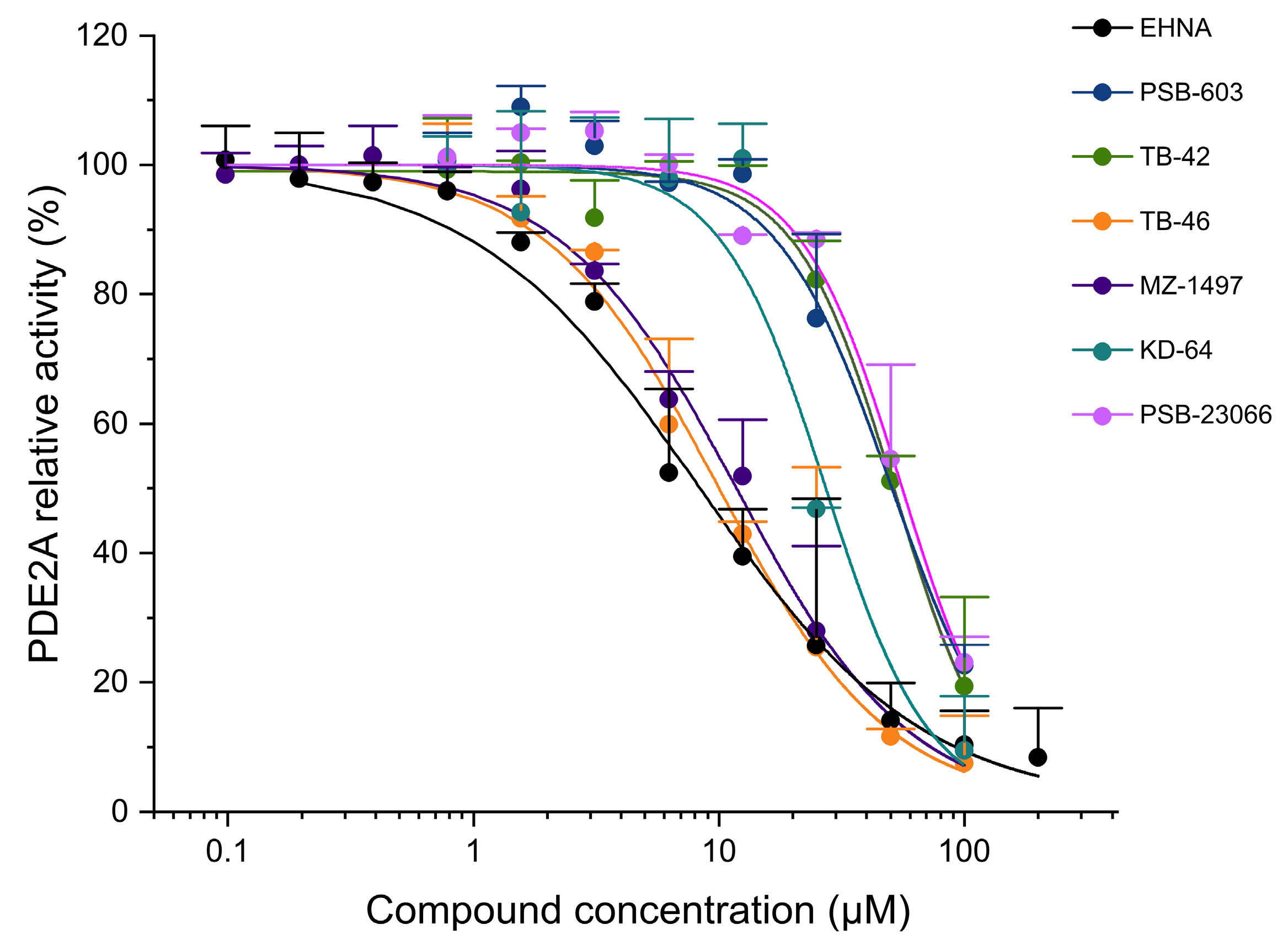
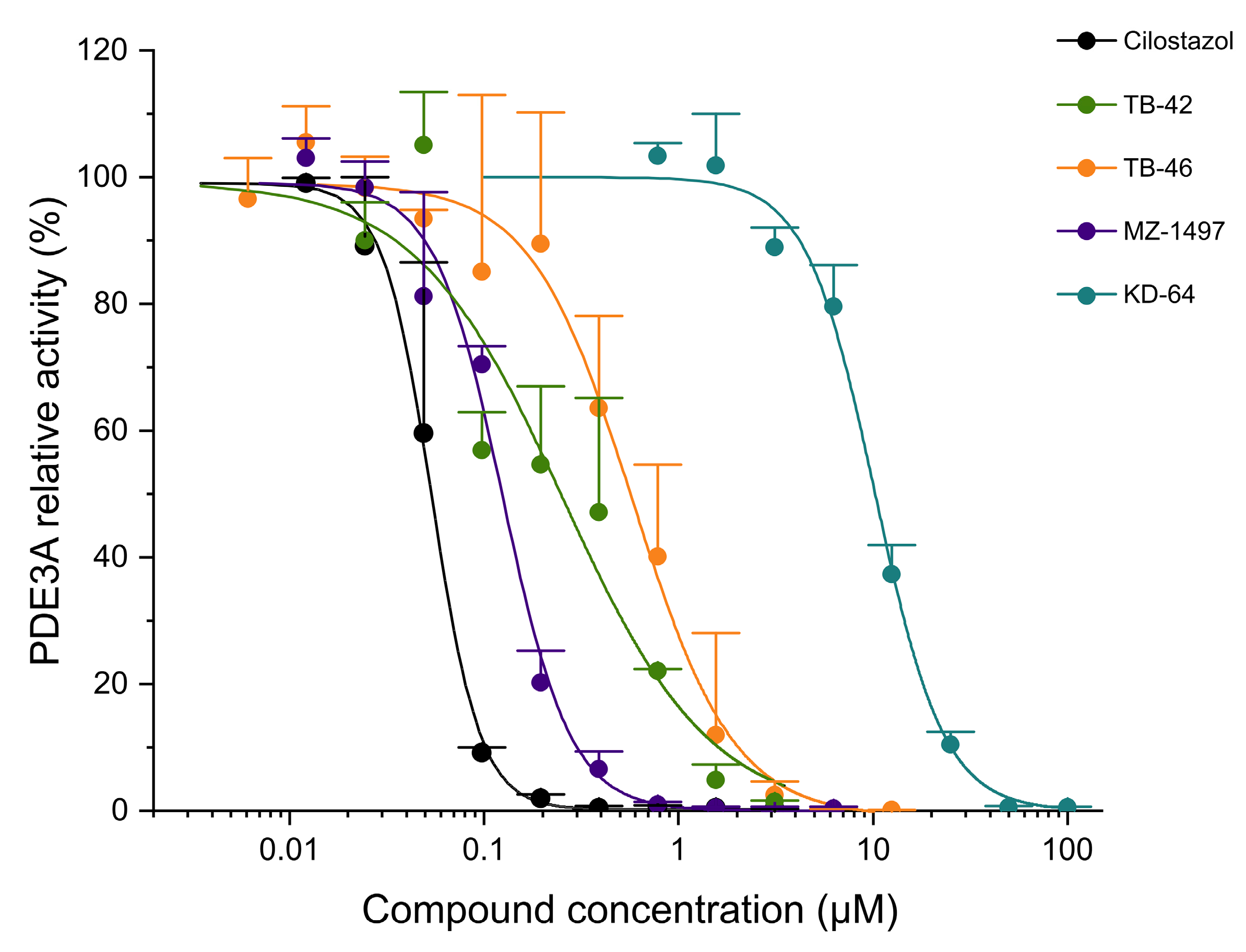
2.3. Evaluation of Phosphodiesterase (PDE) Inhibition
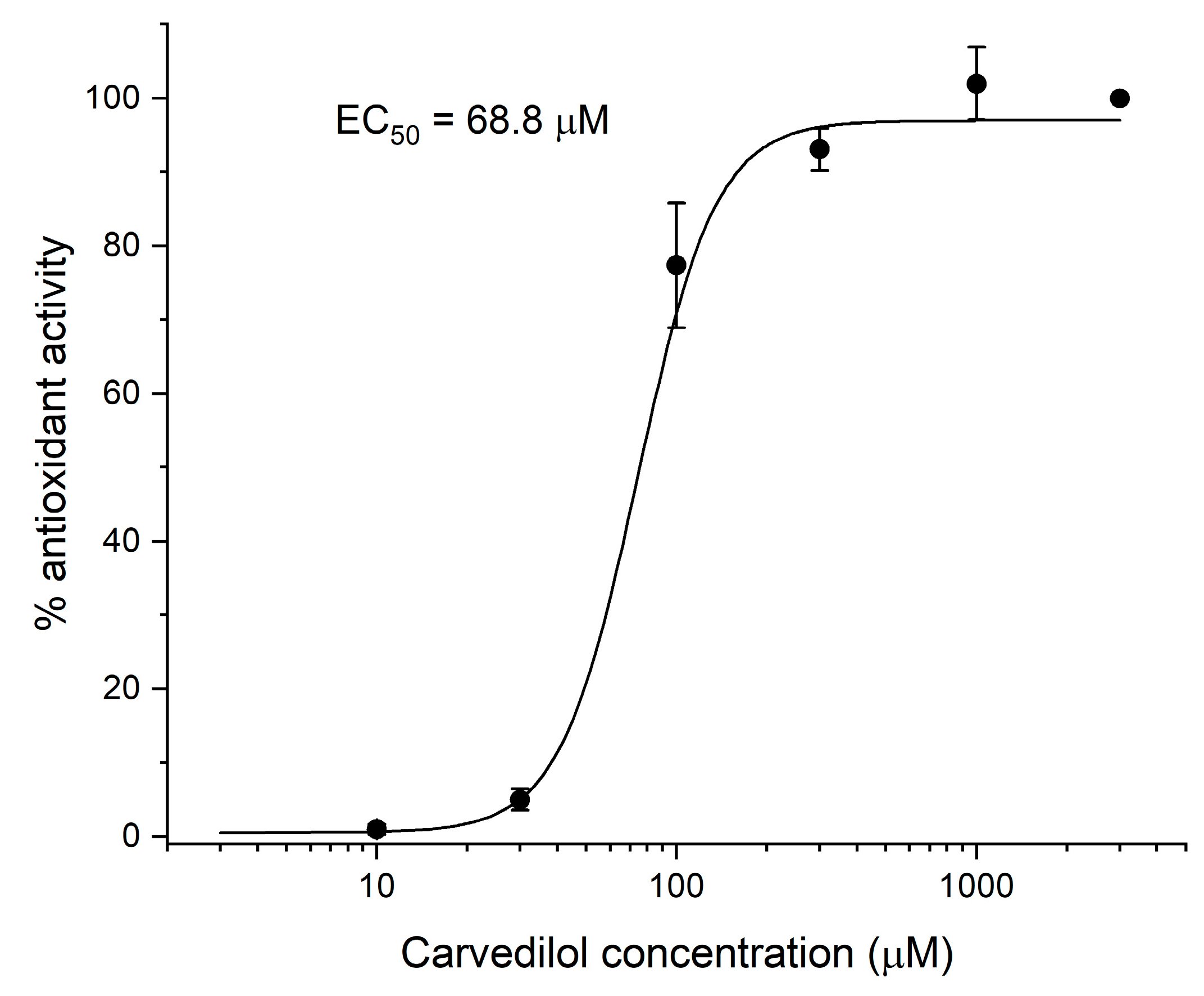
2.4. Evaluation of In Vitro Lipid Peroxidation (MDA Test)
3. Discussion
4. Materials and Methods
4.1. Chemicals and Drugs
4.2. In Vitro Whole Blood Aggregation Tests
4.3. Impact on Platelet Viability (Cytotoxicity Test)
4.4. PDE Activity Inhibition Test
4.5. Lipid Peroxidation Assays
4.6. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Reiss, A.B.; Grossfeld, D.; Kasselman, L.J.; Renna, H.A.; Vernice, N.A.; Drewes, W.; Konig, J.; Carsons, S.E.; DeLeon, J. adenosine and the Cardiovascular Ststem. Am. J. Cardiovasc. Drugs 2019, 19, 449–464. [Google Scholar] [CrossRef] [PubMed]
- Rozalski, M.; Nocun, M.; Watala, C. Adenosine diphosphate receptors on blood platelets: Potential new targets for antiplatelet therapy. Acta. Biochim. Pol. 2005, 52, 411–415. [Google Scholar] [PubMed]
- Johnston-Cox, H.A.; Ravid, K. Adenosine and blood platelets. Purinergic Signal. 2021, 7, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Wolska, N.; Rozalski, M. Blood platelet adenosine receptors as potential targets for anti-platelet therapy. Int. J. Mol. Sci. 2019, 20, 5475. [Google Scholar] [CrossRef]
- Boncler, M.; Bartczak, K.; Rozalski, M. Potential for modulation of platelet function via adenosine receptors during inflammation. Br. J. Pharmacol. 2023. [Google Scholar] [CrossRef] [PubMed]
- Johnston-Cox, H.A.; Yang, D.; Ravid, K. Physiological implications of adenosine receptor-mediated platelet aggregation. J. Cell Physiol. 2011, 226, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, E.; Fuentes, M.; Caballero, J.; Palomo, I.; Hinz, S.; El-Tayeb, A.; Müller, C.E. Adenosine A2A receptor agonists with potent antiplatelet activity. Platelets 2018, 29, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Le, S.N.; Lin, S.; Fong, M.; Guertin, M.; Liu, Y.; Kambayashi, J.I. New mechanism of action for cilostazol: Interplay between adenosine and cilostazol in inhibiting platelet activation. J. Cardiovasc. Pharmacol. 2002, 40, 577–585. [Google Scholar] [CrossRef]
- Varani, K.; Portaluppi, F.; Merighi, S.; Ongini, E.; Belardinelli, L.; Borea, P.A. Caffeine alters A2A adenosine receptors and their function in human platelets. Circulation 1996, 94, 2954–2960. [Google Scholar]
- Theofilis, P.; Sagris, M.; Oikonomou, E.; Antonopoulos, A.S.; Siasos, G.; Tsioufis, C.; Tousoulis, D. Inflammatory Mechanisms Contributing to Endothelial Dysfunction. Biomedicines 2021, 9, 781. [Google Scholar] [CrossRef] [PubMed]
- Repsold, L.; Joubert, A.M. Platelet Function, Role in Thrombosis, Inflammation, and Consequences in Chronic Myeloproliferative Disorders. Cells 2021, 10, 3034. [Google Scholar] [CrossRef]
- Zhu, Y.; Xian, X.; Wang, Z.; Bi, Y.; Chen, Q.; Han, X.; Tang, D.; Chen, R. Research Progress on the Relationship between Atherosclerosis and Inflammation. Biomolecules 2018, 8, 80. [Google Scholar] [CrossRef]
- Moriya, J. Critical roles of inflammation in atherosclerosis. J. Cardiol. 2019, 73, 22–27. [Google Scholar] [CrossRef]
- Badimon, L.; Vilahur, G. Coronary Atherothrombotic Disease: Progress in Antiplatelet Therapy. Rev. Espde Cardiol. 2008, 5, 501–513. [Google Scholar] [CrossRef]
- Davies, M.J. The pathophysiology of acute coronary syndromes. Heart 2000, 83, 361–366. [Google Scholar] [CrossRef] [PubMed]
- Alañón, M.E.; Palomo, I.; Rodríguez, L.; Fuentes, E.; Arráez-Román, D.; Segura-Carretero, A. Antiplatelet Activity of Natural Bioactive Extracts from Mango (Mangifera Indica L.) and its By-Products. Antioxidants 2019, 8, 517. [Google Scholar] [CrossRef] [PubMed]
- Freedman, J.E. Oxidative Stress and Platelets. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 11–16. [Google Scholar] [CrossRef]
- El Haouari, M. Platelet Oxidative Stress and its Relationship with Cardiovascular Diseases in Type 2 Diabetes Mellitus Patients. Curr. Med. Chem. 2019, 26, 4145–4165. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, F.; Palomo, I.; Fuentes, E. Platelet oxidative stress as a novel target of cardiovascular risk in frail older people. Vascul. Pharmacol. 2017, 93–95, 14–19. [Google Scholar] [CrossRef]
- Rodríguez, L.; Plaza, A.; Méndez, D.; Carrasco, B.; Tellería, F.; Palomo, I.; Fuentes, E. Antioxidant Capacity and Antiplatelet Activity of Aqueous Extracts of Common Bean (Phaseolus vulgaris L.) Obtained with Microwave and Ultrasound Assisted Extraction. Plants 2022, 11, 1179. [Google Scholar] [CrossRef]
- Ham, J.; Rees, D.A. The adenosine A2b receptor: Its role in inflammation. Endoc. Met. Imm. Dis. Drug Targets 2008, 8, 244–254. [Google Scholar] [CrossRef]
- da Rocha Lapa, F.; de Oliveira, A.P.; Accetturi, B.G.; de Oliveira Martins, I.; Domingos, H.V.; de Almeida Cabrini, D.; de Lima, W.T.; Santos, A.R. Anti-inflammatory effects of inosine in allergic lung inflammation in mice: Evidence for the participation of adenosine A2A and A 3 receptors. Purinergic. Signal. 2013, 9, 325–336. [Google Scholar] [CrossRef] [PubMed]
- Kolachala, V.; Ruble, B.; Vijay-Kumar, M.; Wang, L.; Mwangi, S.; Figler, H.; Figler, R.; Srinivasan, S.; Gewirtz, A.; Linden, J.; et al. Blockade of adenosine A2B receptors ameliorates murine colitis. Br. J. Pharmacol. 2008, 155, 127–137. [Google Scholar] [CrossRef]
- Kotańska, M.; Szafarz, M.; Mika, K.; Dziubina, A.; Bednarski, M.; Müller, C.E.; Sapa, J.; Kieć-Kononowicz, K. PSB 603—A known selective adenosine A2B receptor antagonist—Has anti-inflammatory activity in mice. Biomed. Pharmacother. 2021, 135, 111164. [Google Scholar] [CrossRef] [PubMed]
- Kotańska, M.; Dziubina, A.; Szafarz, M.; Mika, K.; Reguła, K.; Bednarski, M.; Zygmunt, M.; Drabczyńska, A.; Sapa, J.; Kieć-Kononowicz, K. KD-64-A new selective A2A adenosine receptor antagonist has anti-inflammatory activity but contrary to the non-selective antagonist-Caffeine does not reduce diet-induced obesity in mice. PLoS ONE 2020, 15, e0229806. [Google Scholar] [CrossRef]
- Nicholson, C.D.; Challiss, R.A.; Shahid, M. Differential modulation of tissue function and therapeutic potential of selective inhibitors of cyclic nucleotide phosphodiesterase isoenzymes. Trends Pharmacol. Sci. 1991, 12, 19–27. [Google Scholar] [CrossRef]
- Spina, D.; Page, C.P. Xanthines and Phosphodiesterase Inhibitors. Handb. Exp. Pharmacol. 2017, 237, 63–91. [Google Scholar]
- Meskini, N.; Némoz, G.; Okyayuz-Baklouti, I.; Lagarde, M.; Prigent, A.F. Phosphodiesterase inhibitory profile of some related xanthine derivatives pharmacologically active on the peripheral microcirculation. Biochem. Pharmacol. 1994, 47, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Ogawa, K.; Takagi, K.; Satake, T.; Hidaka, H. Inhibition of cyclic GMP phosphodiesterase by xanthine derivatives relaxes guinea-pig trachealis smooth muscle. Clin Exp Pharmacol Physiol. 1991, 18, 163–168. [Google Scholar] [CrossRef]
- Gresele, P.; Momi, S.; Falcinelli, E. Anti-platelet therapy: Phosphodiesterase inhibitors. Br. J. Clin. Pharmacol. 2011, 72, 634–646. [Google Scholar] [CrossRef]
- Załuski, M.; Schabikowski, J.; Jaśko, P.; Bryła, A.; Olejarz-Maciej, A.; Kaleta, M.; Głuch-Lutwin, M.; Brockmann, A.; Hinz, S.; Zygmunt, M.; et al. 8-Benzylaminoxanthine scaffold variations for selective ligands acting on adenosine A2A receptors. Design, synthesis and biological evaluation. Bioorganic Chem. 2020, 101, 104033. [Google Scholar] [CrossRef] [PubMed]
- Drabczyńska, A.; Yuzlenko, O.; Köse, M.; Paskaleva, M.; Schiedel, A.C.; Karolak-Wojciechowska, J.; Handzlik, J.; Karcz, T.; Kuder, K.; Müller, C.E.; et al. Synthesis and biological activity of tricyclic cycloalkylimidazo-, pyrimido- and diazepinopurinediones. Eur. J. Med. Chem. 2011, 46, 3590–3607. [Google Scholar] [CrossRef] [PubMed]
- Borrmann, T.; Hinz, S.; Bertarelli DC, G.; Li, W.; Florin, N.C.; Scheiff, A.B.; Müller, C.E. 1-alkyl-8-(piperazine-1-sulfonyl)phenylxanthines: Development and characterization of adenosine A2B receptor antagonists and a new radioligand with subnanomolar affinity and subtype specifity. J. Med. Chem. 2009, 52, 3994–4006. [Google Scholar] [CrossRef]
- Jiang, J.; Seel, C.J.; Temirak, A.; Namasivayam, V.; Arridu, A.; Schabikowski, J.; Baqi, Y.; Hinz, S.; Hockemeyer, J.; Müller, C.E. A2B Adenosine Receptor Antagonists with Picomolar Potency. J. Med. Chem. 2019, 62, 4032–4055. [Google Scholar] [CrossRef]
- Wójcik-Pszczoła, K.; Chłoń-Rzepa, G.; Jankowska, A.; Ellen, E.; Świerczek, A.; Pociecha, K.; Koczurkiewicz, P.; Piska, K.; Gawędzka, A.; Wyska, E.; et al. Novel phosphodiesterases inhibitors from the group of purine-2,6-dione derivatives as potent modulators of airway smooth muscle cell remodelling. Eur. J. Pharmacol. 2019, 865, 172779. [Google Scholar] [CrossRef]
- Clark, J.C.; Kavanagh, D.M.; Watson, S.; Pike, A.; Andrews, R.K.; Gardiner, E.E.; Poulter, N.S.; Hill, S.J.; Watson, S.P. Adenosine and Forskolin Inhibit Platelet Aggregation by Collagen but not the Proximal Signalling Events. Thromb. Haemost. 2019, 119, 1124–1137. [Google Scholar] [CrossRef]
- Geue, S.; Walker-Allgaier, B.; Eißler, D.; Tegtmeyer, R.; Schaub, M.; Lang, F.; Gawaz, M.; Borst, O.; Münzer, P. Doxepin inhibits GPVI-dependent platelet Ca2+ signaling and collagen-dependent thrombus formation. Am. J. Physiol. Cell Physiol. 2017, 312, C765–C774. [Google Scholar] [CrossRef]
- De Meyer, S.F.; Vanhoorelbeke, K.; Broos, K.; Salles, I.I.; Deckmyn, H. Antiplatelet drugs. Br. J. Haematol. 2008, 142, 515–528. [Google Scholar] [CrossRef]
- Stegner, D.; Nieswandt, B. Platelet receptor signaling in thrombus formation. J. Mol. Med. 2011, 89, 109–121. [Google Scholar] [CrossRef]
- Stefanini, L.; Roden, R.C.; Bergmeier, W. CalDAG-GEFI is at the nexus of calcium-dependent platelet activation. Blood 2009, 114, 2506–2514. [Google Scholar] [CrossRef]
- Defontis, M.; Côté, S.; Stirn, M.; Ledieu, D. Optimization of Multiplate(®) whole blood platelet aggregometry in the Beagle dog and Wistar rat for ex vivo drug toxicity testing. Exp. Toxicol. Pathol. 2013, 65, 637–644. [Google Scholar] [CrossRef]
- Belcher, P.R.; Drake-Holland, A.J.; Noble, M.I. The antiplatelet drug target in atherosclerotic diseases. Cardiovasc. Hematol. Disord. Drug Targets 2006, 6, 43–55. [Google Scholar] [CrossRef] [PubMed]
- Gachet, C.; Hechler, B. Platelet purinergic receptors in thrombosis and inflammation. Hamostaseologie 2020, 40, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Iyú, D.; Glenn, J.R.; White, A.E.; Fox, S.C.; Heptinstall, S. Adenosine derived from ADP can contribute to inhibition of platelet aggregation in the presence of a P2Y12 antagonist. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 416–422. [Google Scholar] [CrossRef]
- Baqi, Y.; Müller, C.E. Antithrombotic P2Y12 receptor antagonists: Recent developments in drug discovery. Drug Discov. Today 2019, 24, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Shih, C.C.; Chan, M.V.; Kirkby, N.S.; Vojnovic, I.; Mitchell, J.A.; Armstrong, P.C.; Warner, T.D. Platelet inhibition by P2Y12 antagonists is potentiated by adenosine signalling activators. Br. J. Pharmacol. 2021, 178, 4758–4771. [Google Scholar] [CrossRef] [PubMed]
- Dickinson, N.T.; Jang, E.K.; Haslam, R.J. Cyclic nucleotides and phosphodiesterases in platelets. Thromb Haemost. 1999, 82, 412–423. [Google Scholar] [CrossRef]
- Colman, R.W. Platelet cyclic adenosine monophosphate phosphodiesterases: Targets for regulating platelet-related thrombosis. Semin Thromb Hemost. 2004, 30, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Rondina, M.T.; Weyrich, A.S. Targeting Phosphodiesterases in Anti-platelet Therapy. Handb. Exp. Pharmacol. 2012, 210, 225–238. [Google Scholar]
- Degjoni, A.; Campolo, F.; Stefanini, L.; Venneri, M.A. The NO/cGMP/PKG pathway in platelets: The therapeutic potential of PDE5 inhibitors in platelet disorders. J. Thromb. Haemost. 2022, 20, 2465–2474. [Google Scholar] [CrossRef]
- Hidaka, H.; Asano, T. Human blood platelet 3′: 5′-cyclic nucleotide phosphodiesterase. Isolation of low-Km and high-Km phosphodiesterase. Biochim. Biophys. Acta. 1976, 429, 485–497. [Google Scholar] [CrossRef] [PubMed]
- Schror, K. The pharmacology of cilostazol. Diabetes Obes. Metab. 2002, 4 (Suppl. S2), S14–S19. [Google Scholar] [CrossRef]
- Aktas, B.; Utz, A.; Hoenig-Liedl, P.; Walter, U.; Geiger, J. Dipyridamole enhances NO/cGMP-mediated vasodilator-stimulated phosphoprotein phosphorylation and signaling in human platelets: In vitro and in vivo/ex vivo studies. Stroke. 2003, 34, 764–769. [Google Scholar] [CrossRef]
- Kaumann, A.J.; Galindo-Tovar, A.; Escudero, E.; Vargas, L.M. Phosphodiesterases do not limit β 1-adrenoceptor-mediated sinoatrial tachycardia: Evidence with PDE3 and PDE4 in rabbits and PDE1-5 in rats. Naunyn-Schmied. Arch. Pharmacol. 2009, 380, 421–430. [Google Scholar] [CrossRef]
- Kubacka, M.; Kazek, G.; Kotańska, M.; Filipek, B.; Waszkielewicz, A.M.; Mogilski, S. Anti-aggregation effect of aroxyalkyl derivatives of 2-methoxyphenylpiperazine is due to their 5-HT2A and α2-adrenoceptor antagonistic properties. A comparison with ketanserin, sarpogrelate, prazosin, yohimbine and ARC239. Eur. J. Pharmacol. 2018, 818, 263–270. [Google Scholar] [CrossRef]
- Trichler, S.A.; Bulla, S.C.; Thomason, J.; Lunsford, K.V.; Bulla, C. Ultra-pure platelet isolation from canine whole blood. BMC Vet. Res. 2013, 9, 144. [Google Scholar] [CrossRef] [PubMed]
- Rolnik, A.; Skalski, B.; Stochmal, A.; Olas, B. Preparations from selected cucurbit vegetables as antiplatelet agents. Sci. Rep. 2021, 11, 22694. [Google Scholar] [CrossRef] [PubMed]
- Walkowiak, B.; Michalak, E.; Koziołkiewicz, W.; Cierniewski, C.S. Rapid Photometric method for estimation of platelet count in blood plasma or plasma suspension. Thrombosis Res. 1989, 56, 763–766. [Google Scholar] [CrossRef]
- Lyles, R.H.; Poindexter, C.; Evans, A.; Brown, M.; Cooper,, C.R. Nonlinear model-based estimates of IC50 for studies involving continuous therapeutic dose-response data. Contemp. Clin. Trials. 2008, 29, 878–886. [Google Scholar] [CrossRef]
- Yue, T.L.; Cheng, H.Y.; Lysko, P.G.; McKenna, P.J.; Feuerstein, R.; Gu, J.L.; Lysko, K.A.; Davis, L.L.; Feuerstein, G. Carvedilol, a new vasodilator and beta adrenoceptor antagonist, is an antioxidant and free radical scavenger. J. Pharmacol. Exp. Ther. 1992, 263, 92–98. [Google Scholar]



| Compound/Structure | hA1R vs. [3H]CCPA | hA2AR vs. [3H]MSX-2 | hA2BR vs. [3H]PSB-603 | hA3R vs. [3H]PSB-11 |
| Ki ± SEM (nM) (or % inhibition ± SEM at 1 µM) | ||||
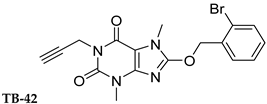 | 357 ± 49 | 59.0 ± 10.2 | >1000 | n.t. |
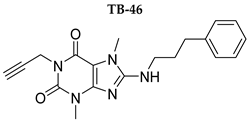 | 158 ± 12 | 19.1 ± 2.3 | 472 ± 174 | >1000 |
 | >1000 | 68.5 ± 15.5 | >1000 | >1000 |
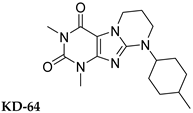 | >1000 | 240 | >1000 | >1000 |
| Compound/Structure | hA1R vs. [3H]CCPA | hA2AR vs. [3H]MSX-2 | hA2BR vs. [3H]PSB-603 | hA3R vs. [3H]PSB-11 |
| Ki ± SEM (nM) (or % inhibition ± SEM at 1 µM) | ||||
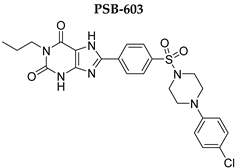 | >1000 | >1000 | 0.553 ± 0.103 | >1000 |
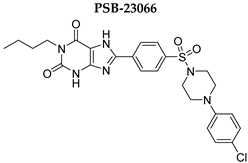 | >1000 | >1000 | 0.401 ± 0.136 | >1000 |
| IC50 a [μM] | ||
|---|---|---|
| Compound | Collagen [1.6 µg/mL] | ADP [6.5 μM] |
| PSB-603 | 98.6 ± 27.0 | 82.9 ± 17.0 |
| TB-42 | 69.3 ± 0.7 | 58.6 ± 35.6 |
| IBMX | 98.8 ± 29.8 | 18.5 ± 7.3 |
| IC50 (CV%) a (µM) | |||
|---|---|---|---|
| Compound | PDE2A | PDE3A | PDE5A |
| PSB-603 | 50.1 (23.9) | >100 (n.d.) | 39.7 (31.5) |
| KD-64 | 27.0 (33.0) | 10.3 (7.03) | >100 (n.d.) |
| PSB-23066 | 55.3 (36.5) | >100 (n.d.) | 45.8 (33.1) |
| MZ-1497 | 11.7 (35.9) | 0.125 (8.68) | >100 (n.d.) |
| TB-42 | 52.1 (16.8) | 0.254 (20.7) | 64.5 (31.6) |
| TB-46 | 9.85 (15.8) | 0.580 (20.6) | 68.6 (26.6) |
| EHNA | 8.22 (16.3) | - | - |
| Cilostazol | - | 0.0545 (2.39) | - |
| Sildenafil | - | - | 0.00117 (31.0) |
| IBMX | 9.22 [35] | 0.71 [35] | 45.1 [35] |
| Compound | Activity Compared to Carvedilol * |
|---|---|
| TB-46 | 59 ± 9.8% |
| MZ-1497 | 45 ± 0.7% |
| TB-42 | 38 ± 1.1% |
| PSB-23066 | 0% |
| PSB-603 | 0% |
| KD-64 | 0% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kubacka, M.; Mogilski, S.; Bednarski, M.; Pociecha, K.; Świerczek, A.; Nicosia, N.; Schabikowski, J.; Załuski, M.; Chłoń-Rzepa, G.; Hockemeyer, J.; et al. Antiplatelet Effects of Selected Xanthine-Based Adenosine A2A and A2B Receptor Antagonists Determined in Rat Blood. Int. J. Mol. Sci. 2023, 24, 13378. https://doi.org/10.3390/ijms241713378
Kubacka M, Mogilski S, Bednarski M, Pociecha K, Świerczek A, Nicosia N, Schabikowski J, Załuski M, Chłoń-Rzepa G, Hockemeyer J, et al. Antiplatelet Effects of Selected Xanthine-Based Adenosine A2A and A2B Receptor Antagonists Determined in Rat Blood. International Journal of Molecular Sciences. 2023; 24(17):13378. https://doi.org/10.3390/ijms241713378
Chicago/Turabian StyleKubacka, Monika, Szczepan Mogilski, Marek Bednarski, Krzysztof Pociecha, Artur Świerczek, Noemi Nicosia, Jakub Schabikowski, Michał Załuski, Grażyna Chłoń-Rzepa, Jörg Hockemeyer, and et al. 2023. "Antiplatelet Effects of Selected Xanthine-Based Adenosine A2A and A2B Receptor Antagonists Determined in Rat Blood" International Journal of Molecular Sciences 24, no. 17: 13378. https://doi.org/10.3390/ijms241713378






