Abstract
Epithelial cells in periodontitis patients increasingly express chemokines, suggesting their active involvement in the inflammatory process. Enamel matrix derivative (EMD) is an extract of porcine fetal tooth germs clinically applied to support the regrowth of periodontal tissues. Periodontal regeneration might benefit from the potential anti-inflammatory activity of EMD for epithelial cells. Our aim was, therefore, to set up a bioassay where chemokine expression is initiated in the HSC2 oral squamous carcinoma cell line and then test EMD for its capacity to lower the inflammatory response. To establish the bioassay, HSC2 cells being exposed to TNFα and LPS from E. coli (Escherichia coli) or P. gingivalis (Porphyromonas gingivalis) were subjected to RNAseq. Here, TNFα but not LPS caused a robust increase of chemokines, including CXCL1, CXCL2, CXCL8, CCL5, and CCL20 in HSC2 cells. Polymerase chain reaction confirmed the increased expression of the respective chemokines in cells exposed to TNFα and IL-1β. Under these conditions, EMD reduced the expression of all chemokines at the transcriptional level and CXCL8 by immunoassay. The TGF-β receptor type I kinase-inhibitor SB431542 reversed the anti-inflammatory activity. Moreover, EMD-activated TGF-β-canonical signaling was visualized by phosphorylation of smad3 and nuclear translocation of smad2/3 in HSC2 cells and blocked by SB431542. This observation was confirmed with primary oral epithelial cells where EMD significantly lowered the SB431542-dependent expression of CXCL8. In summary, our findings suggest that TGF-β signaling mediates the effects of EMD to lower the forced expression of chemokines in oral epithelial cells.
1. Introduction
Periodontal health is difficult to maintain as the periodontium must steadily defend against the microbial and other burdens inherent to the oral cavity [1,2,3]. There is also mechanical damage to the gingiva coming from exaturated mastication that the periodontium has to deal with [4], however, physiological masticatory loaging is required for periodontal homeostasis [5]. Thus, the gingiva and the junctional epithelium with the periodontal ligament underneath build a vital barrier toward the oral cavity equipped with a defense system driven by inflammation [6,7,8,9]. There is consequently a delicate balance of the immunological defense to maintain periodontal homeostasis and the possible shift towards an overshooting and chronic inflammation. This catabolic scenery is mainly driven by biofilms that resist the immunological defense system, culminating in the loss of periodontal structures and ultimately leading to tooth loss [10,11,12,13,14,15]. Understanding the pathological mechanisms causing periodontitis at a molecular and cellular level is the fundament of a successful therapy.
Periodontal regeneration requires removing biofilms and other sources causing chronic inflammation [16,17,18,19]. Only then can a series of clinical approaches be applied with the overall goal of regenerating the periodontal tissues that were deteriorated by the catabolic inflammatory environment. Traditionally, neutrophils, macrophages, and lymphocytes have been blamed for being responsible for causing this inflammatory environment. It was, however, not until recently that a sophisticated single-cell RNA sequencing approach identified epithelial cells to promote leucocyte recruitment [20]. Epithelial cells in periodontitis increasingly express chemokines, including chemokine (C-X-C motif) ligand 1 (CXCL1), critical for recruiting neutrophils [20,21], and it is the neutrophil extracellular traps and histones triggering the early catabolic environment of periodontitis [22]. Oral epithelial cells damaged by masticatory forces may contribute to the inflammation of the periodontium [4]. Under the premise that the oral epithelium contributes to the overall inflammatory defense by the release of chemokines, there is a rational to study the modulation of chemokine expression by established as well as future periodontal therapies.
Enamel matrix derivative (EMD), an extract of porcine fetal tooth germs [23,24,25,26], is clinically applied to support the regrowth of periodontal tissues, and numerous clinical studies have tested its efficacy [27,28,29,30]. Even though the underlying molecular and cellular mechanisms are not fully understood, the effects of EMD might involve the modulation of local inflammation [31]. This has prompted us to study the impact of EMD on macrophages in an LPS-induced inflammatory environment [32,33]. To this aim, EMD significantly attenuated the LPS-induced expression of lead cytokines in RAW 264.7 and primary macrophages, an activity that was attributed to TGF-β, a pleiotropic growth factor found in EMD [32,33]. The TGF-β activity was further attributed to mediating the effects of EMD on osteoclastogenesis [34], adipogenesis [35], and changes in the genetic signature of gingival fibroblasts [36]. Moreover, TGF-β was hypothetically proposed to mediate EMD effects on gene expression changes in HSC2 oral squamous cell carcinoma epithelial-like cells [37]. Thus, the question arises if EMD and its TGF-β activity can lower the forced expression of chemokines in HSC2 cells.
To answer this question, we have aimed to establish a bioassay to simulate chemokine expression by oral epithelial cells in periodontitis [20]. Among the chemokines identified in vivo, our RNAseq identified CXCL1 and a series of other chemokines to be strongly upregulated in HSC2 oral epithelial cells exposed to TNFα but not to LPS from either E. coli or P. gingivalis—serving as a basis for the bioassay. The bioassays allowed us to generate data suggesting that EMD can lower the cytokine-induced expression of chemokines and that the effects can be attributed to its TGF-β activity.
2. Result
2.1. TNFα but Not LPS Increases Chemokine Expression in HSC2 Cells
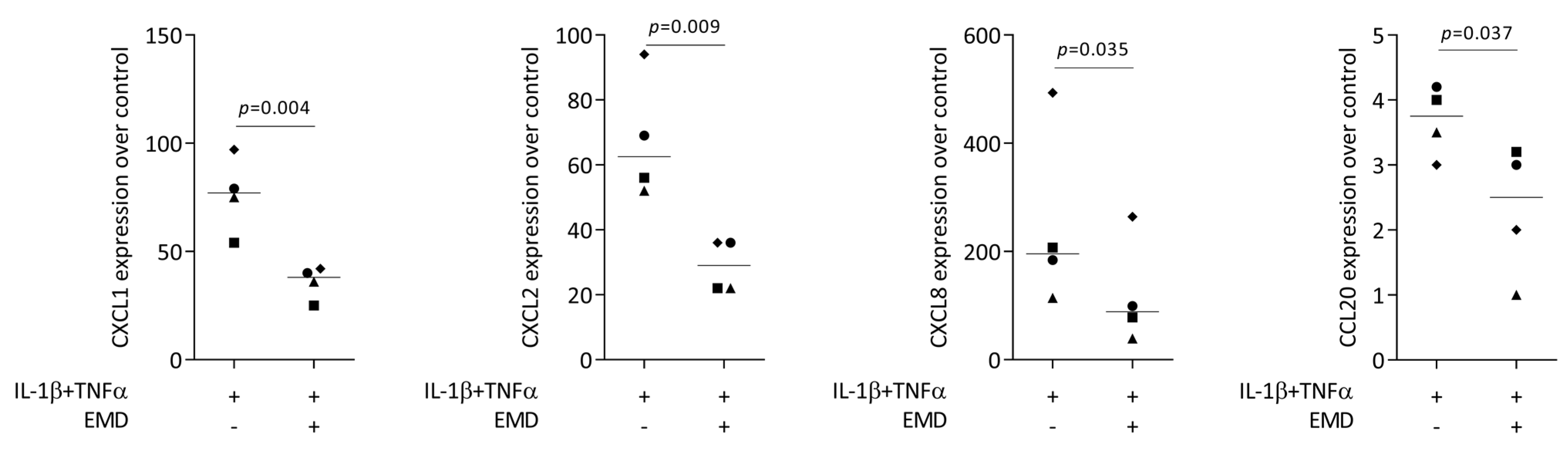
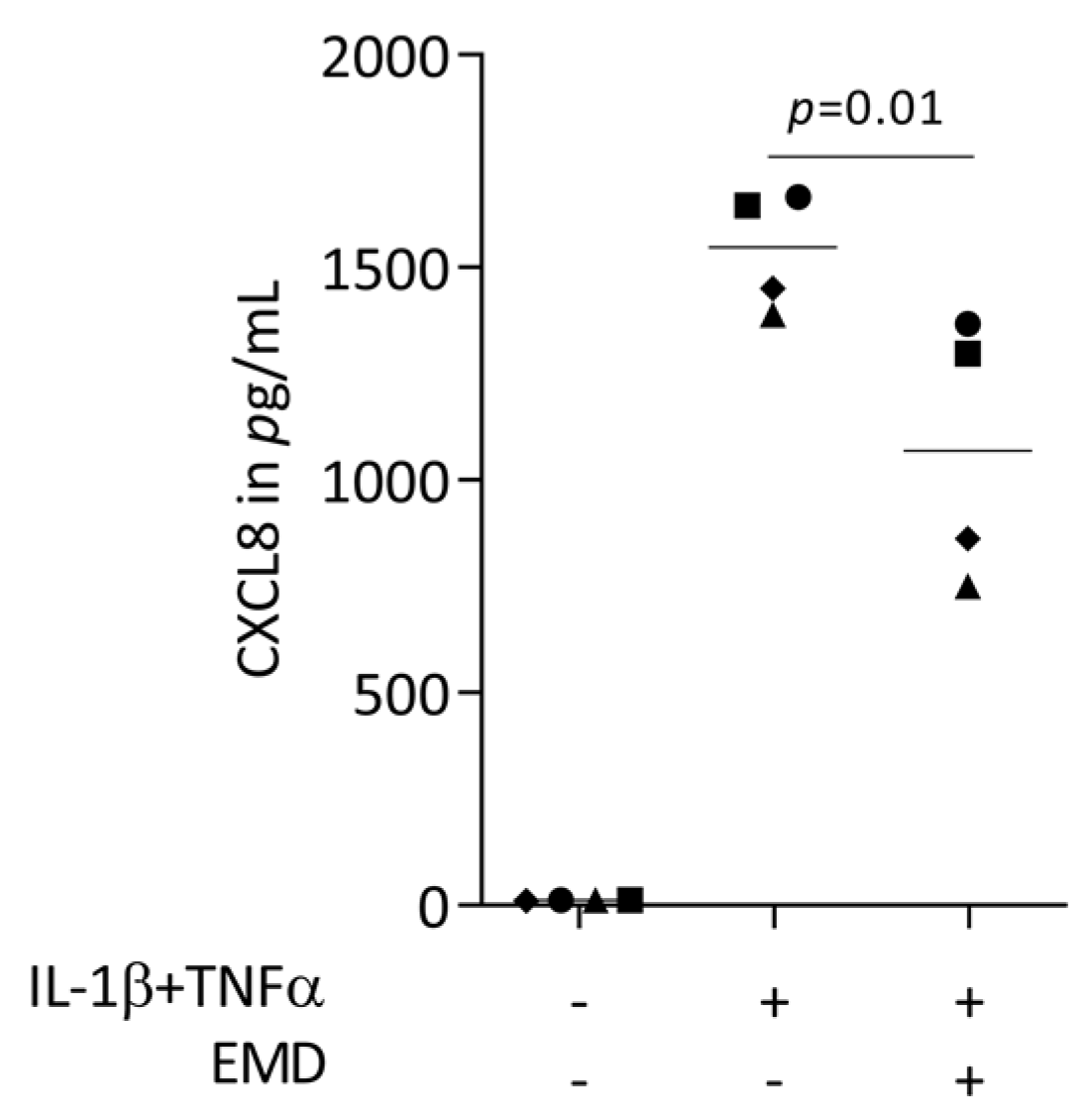
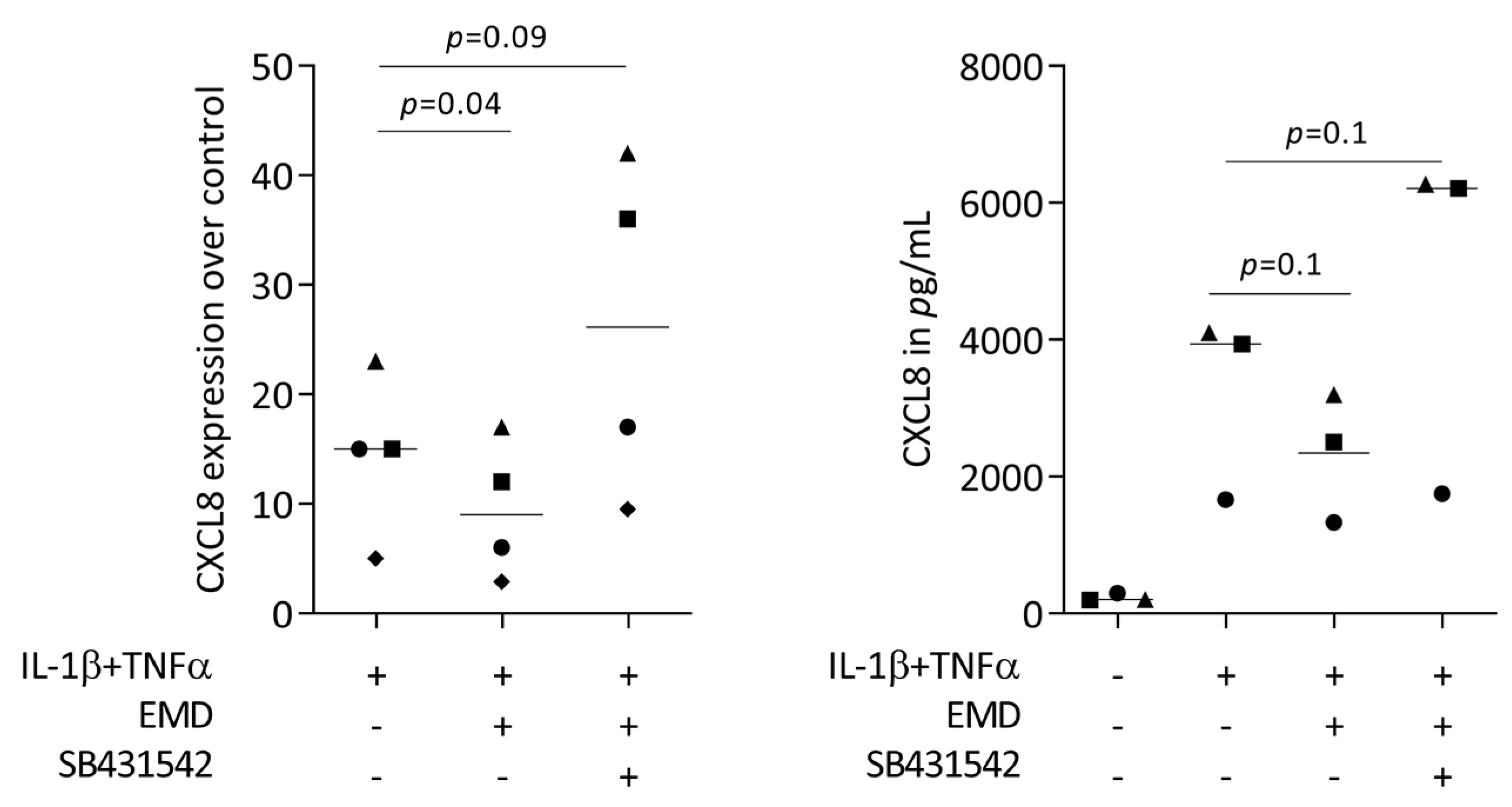
First, we performed a screening for chemokines being expressed in HSC2 cells exposed to TNFα and LPS from E. coli or P. gingivalis. RNAseq revealed that TNFα but none of the two LPS sources caused a robust increase of chemokines, including CXCL1-3, CXCL8-11, CXCL16, CCL5, and CCL20 (Supplementary Table S1). In our RT-PCR-based bioassay with HSC2 cells exposed to TNFα and IL-1β, we could see a robust increase of CXCL1, CXCL2, CXCL8, and CCL20 expression (Figure 1) but also of CXCL10, CXCL11, CXCL12, and CCL5. When HSC2 cells were simultaneously exposed to EMD, the forced expression of CXCL1, CXCL2, CXCL8, and CCL20 was significantly reduced (Figure 1). These findings were supported by an immunoassay showing CXCL8 reduction by EMD (Figure 2). However, EMD failed to significantly lower CXCL10, CXCL11, CXCL12, and CCL5 expression. These findings suggest, first, that the HSC2-based bioassay is a model to study the expression changes of chemokines and, second, that EMD can reduce a selected panel of chemokines.
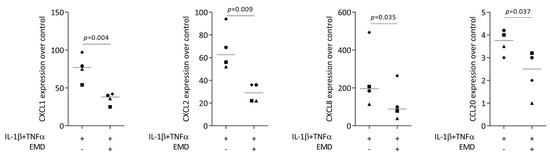
Figure 1.
Gene expression of chemokines CXCL1, CXCL2, CXCL8, and CCL20 in HSC2 cells under TNFα and IL-1β stimulation. A total of 300 µg/mL EMD significantly reduced chemokine expression. Data are expressed as x-fold over the respective untreated controls. Data points represent four independent experiments. Statistical analysis was based on paired t-tests, and p-values are indicated.
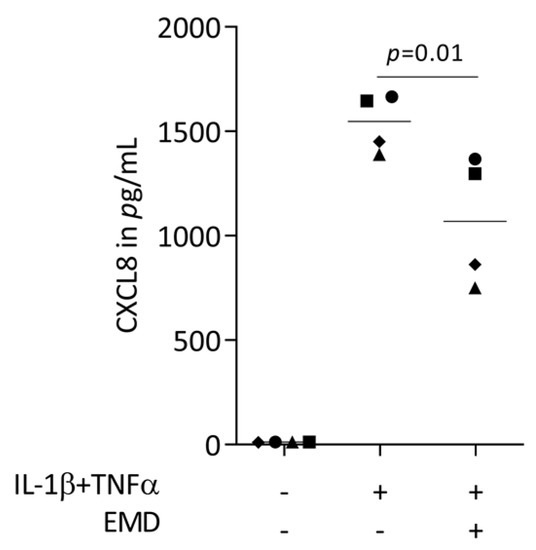
Figure 2.
CXCL8 protein secretion in HSC2 cells under TNFα and IL-1β stimulation. 300 µg/mL EMD significantly reduced protein secretion. Data points represent four independent experiments. Statistical analysis was based on RM one-way ANOVA, with the Geisser–Greenhouse correction tests and p-values are indicated.
2.2. EMD Causes Phosphorylation of smad3 and Translocation of smad2/3 in HSC2 Cells
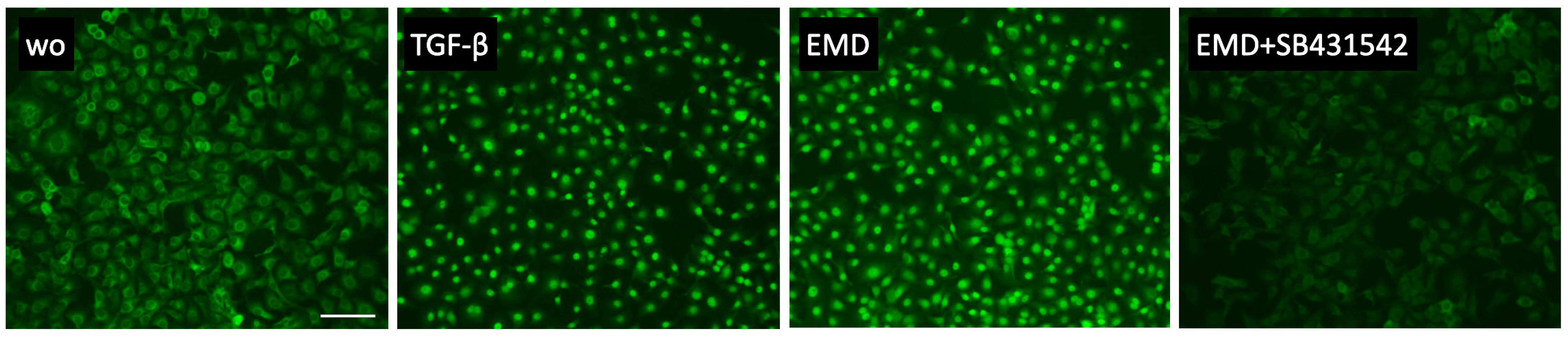
Based on a series of previous experiments indicating TGF-β activity of EMD [32,33,34,35,36], we tested the activation of canonical TGF-β signaling in HSC2 cells. In support of this assumption, EMD induced the phosphorylation of smad3 (Figure 3) and the nuclear translocation of smad2/3 (Figure 4). Moreover, in the presence of the TGF-β receptor type I kinase-inhibitor SB431542, EMD failed to induce the phosphorylation of smad3 (Figure 3) and the nuclear translocation of smad2/3 (Figure 4). Taken together these data not only confirm that EMD activates canonical TGF-β signaling in HSC2 cells; the data further supports the responsiveness of the HSC2 bioassay for TGF-β.

Figure 3.
Smad3 phosphorylation in HSC2 cells. Both, 300 µg/mL EMD and 10 ng/mL TGF-β induced the phosphorylation of smad3. This effect was abolished by the TGF-β receptor type I kinase-inhibitor SB431542.
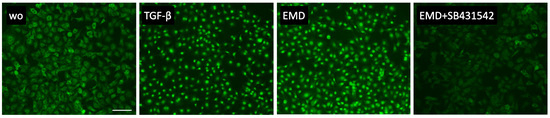
Figure 4.
Smad2/3 nuclear translocation in HSC2 cells. Both, 300 µg/mL EMD and 10 ng/mL TGF-β induced the nuclear translocation of smad2/3 indicated by the immunofluorescence staining. This effect was reversed with the application of the TGF-β receptor type I kinase-inhibitor SB431542. The scale bar represents 100 µm.
2.3. Blocking the TGF-β Signaling Reverses the Ability of EMD to Reduce CXCL8 Expression in HSC2 Cells
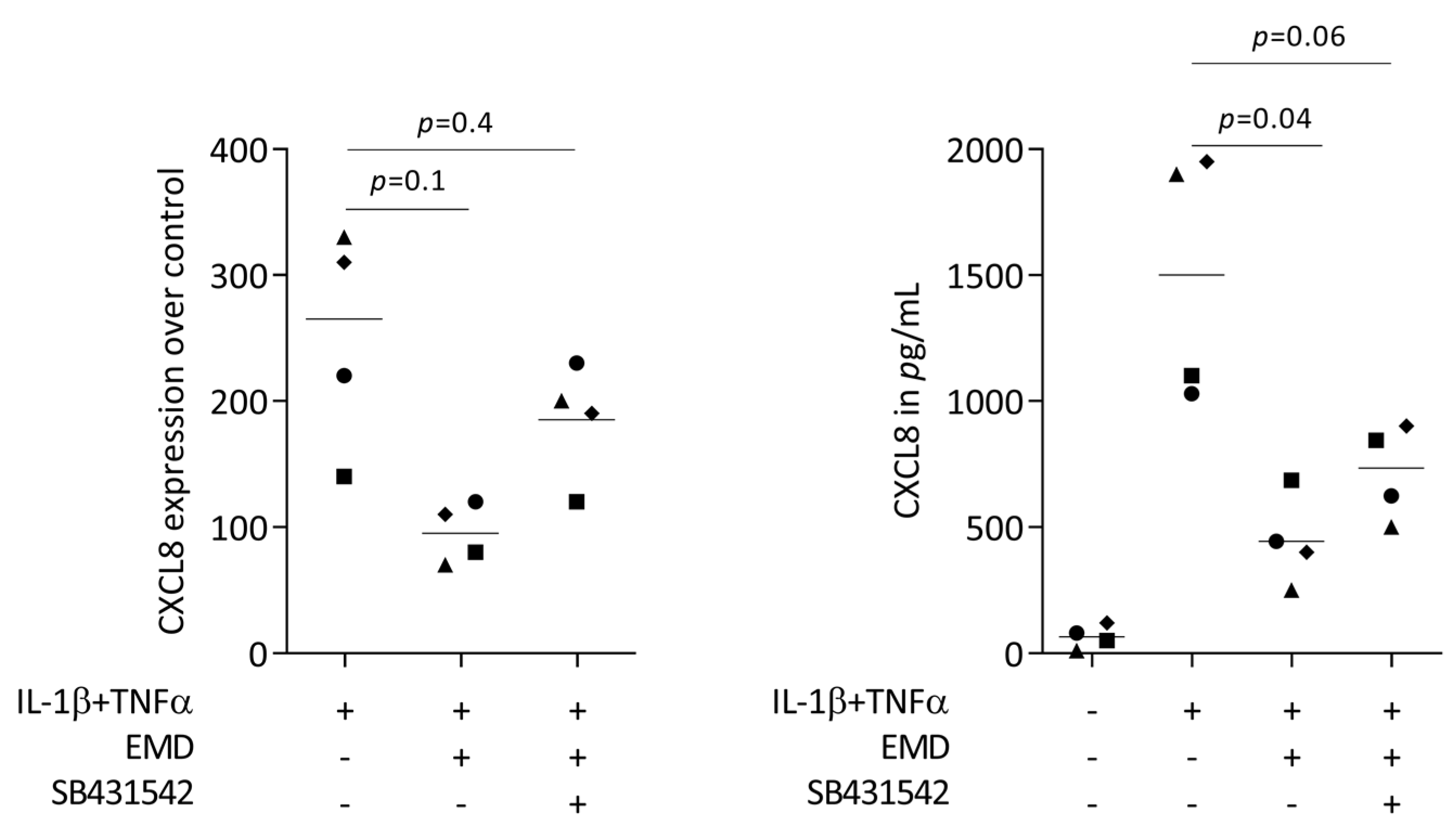
To understand if canonical TGF-β signaling is necessary for EMD to reduce the chemokine expression of HSC2 cells, SB431542 was implemented in the bioassay. In support of previous observations [32,34,35,36], SB431542 partially reversed the ability of EMD to lower the forced expression of CXCL8 in HSC2 cells (Figure 5). Moreover, recombinant TGF-β reduced the chemokine expression of HSC2 cells (Supplementary Table S2). However, the ability of EMD to lower the expression of chemokines was not associated with diminished phosphorylation of p38 and p65 (Supplementary Figure S1). These data indicate that the TGF-β activity is at least partially required to mediate the effects of EMD in lowering the CXCL8 expression in HSC2 cells.
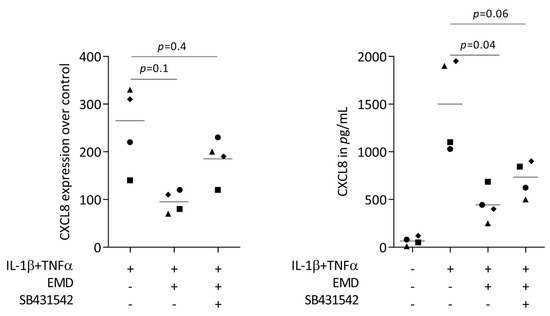
Figure 5.
SB431542 partially reverses the activity of EMD on CXCL8 expression. HSC2 cells under IL-1β and TNFα stimulation were exposed to 300 µg/mL EMD in the absence and presence of the TGF-β receptor type I kinase-inhibitor SB431542. Data points represent four independent experiments. Statistical analysis was based on RM one-way ANOVA, with the Geisser–Greenhouse correction tests and p-values are indicated.
2.4. Primary Oral Epithelial Cells Are Comparable with HSC2 Cells with Respect to CXCL8 Expression
Considering that HSC2 are oral squamous carcinoma cells and may potentially not represent the nonmalignant situation, we aimed to confirm the findings with primary oral epithelial cells. In this setting as well, TNFα and IL-1β provoked primary oral epithelial cells to increasingly express CXCL8, however weaker than the HSC2 cells (Figure 6). Moreover, exposure of the primary oral epithelial cells to EMD significantly reduced the forced CXCL8 expression even though the immunoassay only showed a trend towards CXCL8 reduction (Figure 6). Interestingly, there was a trend that SB431542 even increased CXCL8 expression above the TNFα and IL-1β control levels. These data indicate that HSC2 cells at least partially reflect the behavior of primary oral epithelial cells for testing the impact of EMD on chemokine expression.
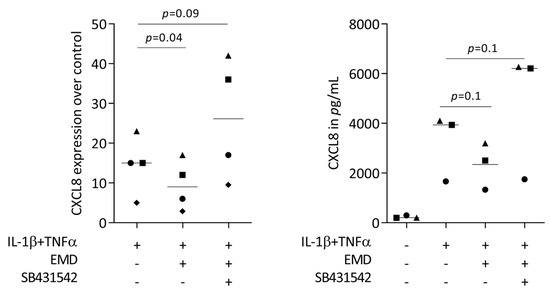
Figure 6.
SB431542 partially reverses the activity of EMD on CXCL8 expression. Primary oral epithelial cells under IL-1β and TNFα stimulation were exposed to 300 µg/mL EMD in the presence and absence of the TGF-β receptor type I kinase-inhibitor SB431542. Data points represent independent experiments. Statistical analysis was based on RM one-way ANOVA, with the Geisser–Greenhouse correction tests and p-values indicated.
3. Discussion
Oral epithelial cells of the mucosa are more than a passive barrier shielding away the underlying connective tissue from the pleiotropy triggers accumulating in the oral cavity and the damage caused by mastication [1]. Oral epithelial cells can become a source of chemokines and cytokines driving local immunity to mediate host defense during homeostasis; however, immunity can shift towards tissue destruction when chronic inflammation dominates the scene [4,20]. Thus, oral epithelial cells have received increasing attention for their active participation in modulating the local inflammatory environment by their release of chemokines, and it mainly the chemokines controlling the immigrating of neutrophils, antigen-presenting cells, and lymphocytes into an inflamed tissue [4,20]. Considering this scenario, we have screened for chemokines being expressed in HSC2 oral epithelial cells when exposed to TNFα and LPS from E. coli and P. gingivalis. One major finding was that HSC2 epithelial cells increasingly express CXCL1-3, CXCL8-11, CXCL16, CCL5, and CCL20 in response to TNFα exposure. This RNAseq screening approach allowed us to establish a bioassay to test potential modulators of chemokine expression, including EMD.
EMD, commonly used in the clinic to support periodontal regeneration [27,28,29,30], is among the possible modulators of an inflammatory response. This assumption is based on previous reports that EMD significantly reduced the LPS-induced cytokine expression in macrophages [32,33] and cytokine-induced expression of IL6 in mouse ST2 bone marrow stromal cells [32]. Consistent with previous reports, we report here that EMD was capable of reducing the cytokine-induced chemokine expression in the HSC2 cells that were most obvious for CXCL1, CXCL2, CXCL8, and CCL20. However, there was also a trend that EMD lowers the induced expression of the other chemokines, namely CXCL10, CXCL11, CXCL12, and CCL5. We have thus concluded that HSC2 cells are a vital bioassay to study the expression of chemokines by a simulated inflammatory environment and take the advantage of this setting for testing the modulation of chemokine expression by EMD. Yet, HSC2 cells are immortal as they originate from an oral squamous cell carcinoma, and we had to confirm their basic response with primary oral epithelial cells. Consistently, EMD lowered the cytokine-induced expression of CXCL8 in primary oral epithelial cells. However, we have to admit that the primary oral epithelial cells showed a less robust CXCL8 response to IL-1β and TNFα, and the impact of the TGF-β receptor type I kinase-inhibitor SB431542 was even more pronounced than with HSC2 cells. Hence, the findings obtained with the HSC2 cells should be interpreted carefully with respect to primary cells and even tissues. Considering this premise, HSC2 cells remain a suitable bioassay to study the modulation of cytokine-induced chemokine expression.
The question arises of what could be the underlying molecular mechanism that makes EMD reduce chemokine expression. The answer might be related to TGF-β that mediates at least part of the EMD activity in vitro, e.g., its anti-inflammatory activity in LPS-induced macrophages [32,33], its suppression of adipogenesis [35], its ability to support osteoclastogenesis [34] and its driving gene expression in oral fibroblasts [36]. In support of previous research with EMD-treated epithelial SCC25 and fibroblastic Gin-1 cells [38], we observed that EMD initiates canonical TGF-β signaling indicated by the phosphorylation and nuclear translocation of smads in HSC2 cells. More evidence comes from our observations that the TGF-β receptor type I kinase-inhibitor SB431542 partially reversed the activity of EMD to lower CXCL8 expression. SB431542 was also reported to block EMD effects in other in vitro studies [32,34,35,36]. Our observations are further supported by others showing that TGF-β-induced smad2/3 blocks the CXCL1 promoter in fibroblasts [39,40], and the expression of breast cancer stromal CXCL1 is inversely correlated with the expression of TGF-β signaling [40]. It is thus reasonable to suggest that the TGF-β activity intrinsic to EMD is at least partially responsible for its lowering activity on chemokine expression.
The clinical interpretation of our RNAseq findings remains at the level of speculation, but considering that in vivo, oral epithelial cells express increasing amounts of CXCL1, CXCL3, CXCL5, CXCL16, CCL20, and CCL28 [20], our in vitro RNAseq analysis identified all chemokines but not CXCL5 being increased under inflammatory conditions. In our RT-PCR analysis, we highlight CXCL1 and CCL20, representing the in vivo situation [20], but also included other chemokines identified by our RNAseq. Surprisingly, epithelial cells in periodontitis patients do not show increasing levels of chemokines such as CXCL8 and other chemokines we have identified in our RNAseq assay [20]. In mouse diabetic conditions, however, oral epithelial cells increasingly express chemokines we have identified by RNAseq, including CXCL1, CXCL2, CXCL3, CXCL10, and CXCL16 [41] but also not CXCL8. Moreover, chemokines such as CXCL8 [42], CXCL10 [43], CXCL12 [44], and CCL5 [45] accumulate in the crevicular fluid of periodontitis patients and their source might be attributed to fibroblasts at least for CXCL2, CXCL3, CXCL8, CXCL9, CXCL10, CXCL12, and CXCL16 [20]. Thus, our HSC2-based bioassay only partially reflects the changes in chemokine expression in a diseased periodontium. We can thus only speculate that EMD may exert part of its activity by decreasing CXCL1, lowering the influx of neutrophils, and CCL20 reducing lymphocytes and antigen-presenting cells in the oral epithelium.
We may further highlight CXCL1 because it is involved in the crosstalk of periodontitis and Crohn’s disease [46]. There is also a link to diabetes as insulin signaling lowers LPS-induced CXCL1 expression in gingival fibroblasts [47], and short-chain fatty acid can lower cytokine-induced expression of CXCL1 and CXCL2 in HSC2 cells and gingival fibroblasts [48]. Higher levels of CXCL1 and other chemokines were found in human and rat’s gingiva from sites of periodontitis as compared with healthy sites [49], and in vitro, biofilms can provoke CXCL1, CXCL3, and CXCL8 expression in epithelial cells [50]. In healthy tissue, however, oral commensal bacteria are associated with the selective expression of CXCL2 but not CXCL1 [51], suggesting CXCL1 is a lead chemokine in oral epithelial cells for periodontitis. CCL20 is also among the chemokines being increased in experimentally induced gingivitis that might become a target of EMD therapy [52]. Support for this assumption would require an epithelial-specific K14-CXCL1 or other chemokine‘s conditional knockout model to prove that a certain chemokine expressed by oral epithelial cells is driving periodontists [53,54].
Our in vitro study has limitations as in vivo cytokines are presumably not the only trigger for oral epithelial cells to become sources of chemokines. For instance, in immortalized human oral keratinocytes, LPS from p. gingivalis caused a robust increase of CXCL1, CXCL8, and CCL20 [55], and in OKF6/TERT-2 oral keratinocytes, LPS increased IL6 [56]. In our in vitro setting, however, LPS at 100 ng/mL from E. coli [57] and 1 µg/mL from P. gingivalis [48] failed to considerably change chemokines in HSC2 cells, comparable to findings observed by others with the same cell line [58]. This lack of LPS response might be explained by the fact that HSC2 cells, similar to other oral epithelial cells, do not express relevant CD14 required for the LPS-induced signaling via TLR4 [59,60]. However, IFN-γ can upregulate the CD14-TLR signaling machinery and make oral epithelial cells become susceptive to LPS [60,61]. Taken together, oral epithelial cells are no classical targets for LPS; otherwise, the LPS activity of saliva would provoke a chronic inflammation of the oral mucosa [62,63]. Thus, future research might focus on how EMD modulates IFN-γ signaling in HSC2 and other oral epithelial cells, considering the cross-talk of the two signaling pathways. For instance, IFN-γ drives Smad7 expression as a potent inhibitor of canonical TGF-β signaling [64]. It can thus be hypothesized that IFN-γ makes the oral epithelial cell more sensitive to TLR4 signaling and even lowers the capacity of EMD to reduce chemokine expression; however, recent reviews do not support a correlation between IFNγ levels and periodontitis [65]. We can thus propose that the LPS-related anti-inflammatory activity of EMD is presumably more related to macrophages [32,33] than to epithelial cells in vitro.
Apart from chemokines, our RNAseq has identified a larger spectrum of inflammation-related genes increasingly expressed in HSC2 cells in response to TNFα but not LPS, including the cytokines IL32 [66], IL1β, IL1α–also the IFN-λ1, IFN-λ2, IFN-λ3 (also known as IL29, IL28A, and IL28B, respectively [67,68,69,70,71], as well as interferon-induced GTP-binding protein MX1 and MX2 [72], and the acute-phase protein serum amyloid A1/2 (SAA1 and SAA2) [73].
Our rather simple RT-PCR-based approach, however, is not ideal for detecting the impact of EMD on changing the entire signature of TNFα-induced HSC2 cells. Here, an extension of the RNAseq approach towards including EMD and the TGF-β receptor type I kinase-inhibitor SB431542 would help to understand to which extent the TGF-β activity causes the anti-inflammatory activity of EMD in our bioassay proposed, similar to what we have reported for fibroblasts [36]. We have also identified genes being increasingly expressed by both sources of LPS, such as UNC5CL, an inducer of a pro-inflammatory signaling cascade [74]; PTGER4, a prostaglandin E2 receptor expressed in oral squamous cell carcinomas [75] including HSC3 [76]; killin, a p53-regulated nuclear inhibitor of DNA synthesis [77]; and PPP1R3F, the protein phosphatase 1 regulatory subunit 3F. Confirming and interpreting these RNAseq outcomes was not the focus of the present research. Future RNAseq research might also answer if IFN-γ can upregulate the CD14-TLR signaling in HSC2 cells and identify other pathogen-associated molecular patterns than LPS recognized by the large family of toll-like receptors and other pattern recognition receptors to refine the bioassay we have established and continue with the testing EMD and other potential therapies such as platelet-rich fibrin [78,79] in a controlled in vitro setting.
4. Materials and Methods
4.1. HSC2 Cells and Primary Oral Epithelial Cells
The oral squamous cell carcinoma cell line HSC2, originally obtained from the Health Science Research Resources Bank (Sennan, Japan) and cultivated in growth Dulbecco’s modified Eagle’s medium (DMEM, Sigma-Aldrich, St. Louis, MO, USA), 10% fetal calf serum (Bio&Sell GmbH, Nuremberg, Germany), and 1% antibiotic-antimycotic (Sigma Aldrich, St.Louis, MO, USA). HSC2 cells were seeded at a concentration of 2.5 × 105 cells/cm2 onto culture dishes one day prior to stimulation. Primary oral epithelial cells were taken from the epithelial layer of human gingiva harvested from the extracted third molars of patients who had given informed and written consent. The Ethics Committee of the Medical University of Vienna (EK NR 631/2007) approved the protocol. Primary cells were cultivated in a keratinocyte growth medium (PromoCell, Heidelberg, Germany) and seeded at a concentration of 4.0 × 105 cells/cm2 onto culture dishes one day prior to stimulation. In the basic setting, HSC2 and primary epithelial cells were treated overnight with 10 ng/mL TNFα and 10 ng/mL IL-1β (both ProSpec, Ness-Ziona, Israel) with and without 300 µg/mL Emdogain® (EMD; Straumann Group, Basel, Switzerland) or with serum-free medium alone at 37 °C, 5% CO2, and 95% humidity before analysis.
4.2. RNAseq
RNA quality was evaluated using the Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, USA). Sequencing libraries were prepared at the Core Facility Genomics, Medical University of Vienna, using the NEBNext Poly (A) mRNA Magnetic Isolation Module and the NEBNext Ultra™ II Directional RNA Library Prep Kit for Illumina according to the manufacturer’s protocols (New England Biolabs, Ipswich, MA, USA). Libraries were QC-checked on a Bioanalyzer 2100 (Agilent Technologie, Santa Clara, CA, USA) using a High Sensitivity DNA Kit for correct insert size and quantified using Qubit dsDNA HS Assay (Invitrogen, Thermo Fischer, Waltham, MA, USA). Pooled libraries were sequenced on a NextSeq500 instrument (Illumina, San Diego, CA, USA) in 1 × 75 bp single-end sequencing mode. Approximately 25 million reads were generated per sample. Reads in fastq format were aligned to the human reference genome version GRCh38 (www.ncbi.nlm.nih.gov/grc/human) with Gencode 29 annotations (www.gencodegenes.org/human/release_29.html) using STAR aligner 55 version 2.6.1a in 2-pass mode. Reads per gene were counted by STAR and gene expression was calculated.
4.3. Real-Time Polymerase Chain Reaction (RT-PCR) and Immunoassay
Total RNA was isolated with the ExtractMe total RNA kit (Blirt S.A., Gdańsk, Poland), followed by reverse transcription and polymerase chain reaction (LabQ, Labconsulting, Vienna, Austria) on a CFX Connect™ Real-Time PCR Detection System (Bio-Rad Laboratories, Hercules, CA, USA). The mRNA levels were calculated by normalizing to the housekeeping gene GAPDH using the ΔΔCt method. Primer sequences are in Table 1. For the immunoassays, the human CXCL8 kit (DY208-R&D Systems, Minneapolis, MN, USA) was used. Immunoassay were used according to the manufacturer’s instructions.

Table 1.
The primer sequences.
4.4. Immunofluorescence Analysis
Immunofluorescence analysis was performed in HSC2 cells plated on Millicell® EZ slides (Merck KGaA, Darmstadt, Germany). Cells were stimulated with 300 µg/mL EMD or 10 ng/mL TGF-β1, with and without SB431542 (Calbiochem, Merck Millipore, Burlington, MA, USA). Cells were fixed with 4% paraformaldehyde, blocked with 1% bovine serum albumin, and permeabilized with 0.3% Triton X-100 (all Sigma-Aldrich). Smad2/3 (D7G7 XP®, Cell Signaling Technology, Danvers, MA, USA) was used overnight at 4 °C. Detection was performed with an Alexa 488 secondary antibody (CS-4412, Cell Signaling Technology). We captured the images on a fluorescence microscope with the DAPI-FITC dual excitation filter block (Echo Revolve Fluorescence Microscope, San Diego, CA, USA).
4.5. Western Blot Analysis
HSC2 cells were serum-starved cells and exposed to 300 µg/mL EMD or 10 ng/mL TGF-β1, with and without SB431542, for 30 min. Cell extracts containing SDS buffer with protease and phosphatase inhibitors (cOmplete Ultra Tablets and PhosStop; Roche, Mannheim, Germany) were separated by SDS–PAGE and transferred onto PVDF membranes (Roche Diagnostics, Mannheim, Germany). Membranes were blocked, and the binding of the primary antibodies p-Smad3 (EP823Y, Abcam, Cambridge, UK) and actin (sc-47778, Santa Cruz Biotechnology, Dallas, TX, USA), was detected with the secondary antibody labeled with HRP. After exposure to a substrate (Bio-Rad Laboratories, Inc., Hercules, CA, USA), chemiluminescence signals were visualized with a ChemiDoc imaging system (Bio-Rad Laboratories). In another set of experiments, HSC2 cells exposed to TNFα and IL-1β with and without 300 µg/mL EMD for 30 min were subjected to staining with phospho-NFκB-p65, NFκB-p65 (both Cell Signaling Technology; #7074, #4511), phospho-p38 and p38 (both Santa Cruz Biotechnology; #4511, #535).
4.6. Statistical Analysis and Acronyms
All experiments were performed at least four times. Statistical analyses were performed with paired t-tests or repeated measures one-way ANOVA, with the Geisser–Greenhouse correction tests whenever appropriate. Analyses were performed using Prism v.9 (GraphPad Software; San Diego, CA, USA). Significance was set at p < 0.05. The acronyms are listed in Supplementary Table S3.
5. Conclusions
Our in vitro approach is based on an unproven assumption that the increased chemokine expression by oral epithelial cells in vivo is a pathological mechanism driving periodontitis. Here, we show data supporting the concept that EMD by activating canonical TGF-β signaling in oral epithelial cells might exert a beneficial effect by normalizing the pathologically increased expression of the chemokines and thereby lower the influx of neutrophils and other cells of the innate immune system driving tissue destruction. Today, this concept remains at the level of a hypothesis but is supported by the data reported here.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms241813991/s1.
Author Contributions
Conceptualization, L.P. and R.G.; data curation, L.P.; formal analysis, L.P. and R.G.; methodology, L.P., S.B., A.O.A. and Z.A.; project administration, L.P., S.B., A.O.A. and Z.A.; supervision, L.P. and R.G.; validation, L.P. and R.G.; visualization, L.P.; writing—original draft, L.P. and R.G.; writing—review and editing, L.P., S.B., A.O.A., Z.A. and R.G. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by the International Team of Implantology (ITI) #1729-2023.
Institutional Review Board Statement
The Ethics Committee of the Medical University of Vienna (EK NR 631/2007) approved the protocol.
Informed Consent Statement
Informed and written consent was obtained from all subjects involved in the study.
Data Availability Statement
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
Acknowledgments
We are grateful to Straumann for providing EMD. We thank the Core Facility Genomics of the Medical University of Vienna for performing the RNAseq. Primary oral epithelial cells were kindly provided by Salman Abbas Zadeh.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Moutsopoulos, N.M.; Konkel, J.E. Tissue-Specific Immunity at the Oral Mucosal Barrier. Trends Immunol. 2018, 39, 276–287. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.M.; Doyle, A.D.; Greenwell-Wild, T.; Dutzan, N.; Tran, C.L.; Abusleme, L.; Juang, L.J.; Leung, J.; Chun, E.M.; Lum, A.G.; et al. Fibrin is a critical regulator of neutrophil effector function at the oral mucosal barrier. Science 2021, 374, eabl5450. [Google Scholar] [CrossRef] [PubMed]
- Gorr, S.U. Antimicrobial peptides in periodontal innate defense. Front. Oral Biol. 2012, 15, 84–98. [Google Scholar] [CrossRef] [PubMed]
- Dutzan, N.; Abusleme, L.; Bridgeman, H.; Greenwell-Wild, T.; Zangerle-Murray, T.; Fife, M.E.; Bouladoux, N.; Linley, H.; Brenchley, L.; Wemyss, K.; et al. On-going Mechanical Damage from Mastication Drives Homeostatic Th17 Cell Responses at the Oral Barrier. Immunity 2017, 46, 133–147. [Google Scholar] [CrossRef]
- Yuan, X.; Liu, B.; Cuevas, P.; Brunski, J.; Aellos, F.; Petersen, J.; Koehne, T.; Broer, S.; Gruber, R.; LeBlanc, A.; et al. Linking the Mechanics of Chewing to Biology of the Junctional Epithelium. J. Dent. Res. 2023, 220345231185288. [Google Scholar] [CrossRef]
- Yamamoto, M.; Aizawa, R. Maintaining a protective state for human periodontal tissue. Periodontol. 2000 2021, 86, 142–156. [Google Scholar] [CrossRef]
- Bosshardt, D.D. The periodontal pocket: Pathogenesis, histopathology and consequences. Periodontol. 2000 2018, 76, 43–50. [Google Scholar] [CrossRef]
- Bosshardt, D.D.; Sculean, A. Does periodontal tissue regeneration really work? Periodontol. 2000 2009, 51, 208–219. [Google Scholar] [CrossRef]
- Bosshardt, D.D.; Lang, N.P. The junctional epithelium: From health to disease. J. Dent. Res. 2005, 84, 9–20. [Google Scholar] [CrossRef]
- Kinane, D.F.; Stathopoulou, P.G.; Papapanou, P.N. Periodontal diseases. Nat. Rev. Dis. Primers 2017, 3, 17038. [Google Scholar] [CrossRef]
- Demkovych, A.; Kalashnikov, D.; Hasiuk, P.; Zubchenko, S.; Vorobets, A. The influence of microbiota on the development and course of inflammatory diseases of periodontal tissues. Front. Oral Health 2023, 4, 1237448. [Google Scholar] [CrossRef] [PubMed]
- Lamont, R.J.; Hajishengallis, G.; Koo, H. Social networking at the microbiome-host interface. Infect. Immun. 2023, e0012423. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G. Illuminating the oral microbiome and its host interactions: Animal models of disease. FEMS Microbiol. Rev. 2023, 47, fuad018. [Google Scholar] [CrossRef] [PubMed]
- Hajishengallis, G.; Lamont, R.J.; Koo, H. Oral polymicrobial communities: Assembly, function, and impact on diseases. Cell Host Microbe 2023, 31, 528–538. [Google Scholar] [CrossRef] [PubMed]
- Belibasakis, G.N.; Belstrom, D.; Eick, S.; Gursoy, U.K.; Johansson, A.; Kononen, E. Periodontal microbiology and microbial etiology of periodontal diseases: Historical concepts and contemporary perspectives. Periodontol. 2000 2023. [Google Scholar] [CrossRef]
- Jepsen, K.; Sculean, A.; Jepsen, S. Complications and treatment errors related to regenerative periodontal surgery. Periodontol. 2000 2023, 92, 120–134. [Google Scholar] [CrossRef]
- Tomasi, C.; Abrahamsson, K.H.; Apatzidou, D. Subgingival instrumentation. Periodontol. 2000 2023. [Google Scholar] [CrossRef]
- Golub, L.M.; Lee, H.M. Periodontal therapeutics: Current host-modulation agents and future directions. Periodontol. 2000 2020, 82, 186–204. [Google Scholar] [CrossRef]
- Mombelli, A. Maintenance therapy for teeth and implants. Periodontol. 2000 2019, 79, 190–199. [Google Scholar] [CrossRef]
- Williams, D.W.; Greenwell-Wild, T.; Brenchley, L.; Dutzan, N.; Overmiller, A.; Sawaya, A.P.; Webb, S.; Martin, D.; Genomics, N.N.; Computational Biology, C.; et al. Human oral mucosa cell atlas reveals a stromal-neutrophil axis regulating tissue immunity. Cell 2021, 184, 4090–4104. [Google Scholar] [CrossRef]
- Kobayashi, Y. The role of chemokines in neutrophil biology. Front. Biosci. 2008, 13, 2400–2407. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.S.; Silva, L.M.; Theofilou, V.I.; Greenwell-Wild, T.; Li, L.; Williams, D.W.; Ikeuchi, T.; Brenchley, L.; Genomics, N.N.; Computational Biology, C.; et al. Neutrophil extracellular traps and extracellular histones potentiate IL-17 inflammation in periodontitis. J. Exp. Med. 2023, 220. [Google Scholar] [CrossRef] [PubMed]
- Gestrelius, S.; Lyngstadaas, S.P.; Hammarstrom, L. Emdogain--periodontal regeneration based on biomimicry. Clin. Oral Investig. 2000, 4, 120–125. [Google Scholar] [CrossRef] [PubMed]
- Gestrelius, S.; Andersson, C.; Johansson, A.C.; Persson, E.; Brodin, A.; Rydhag, L.; Hammarstrom, L. Formulation of enamel matrix derivative for surface coating. Kinetics and cell colonization. J. Clin. Periodontol. 1997, 24, 678–684. [Google Scholar] [CrossRef]
- Hammarstrom, L.; Heijl, L.; Gestrelius, S. Periodontal regeneration in a buccal dehiscence model in monkeys after application of enamel matrix proteins. J. Clin. Periodontol. 1997, 24, 669–677. [Google Scholar] [CrossRef]
- Hammarstrom, L. Enamel matrix, cementum development and regeneration. J. Clin. Periodontol. 1997, 24, 658–668. [Google Scholar] [CrossRef]
- Suarez-Lopez Del Amo, F.; Monje, A. Efficacy of biologics for alveolar ridge preservation/reconstruction and implant site development: An American Academy of Periodontology best evidence systematic review. J. Periodontol. 2022, 93, 1827–1847. [Google Scholar] [CrossRef]
- Tavelli, L.; Chen, C.J.; Barootchi, S.; Kim, D.M. Efficacy of biologics for the treatment of periodontal infrabony defects: An American Academy of Periodontology best evidence systematic review and network meta-analysis. J. Periodontol. 2022, 93, 1803–1826. [Google Scholar] [CrossRef]
- Moldovan, R.; Mester, A.; Piciu, A.; Bran, S.; Onisor, F. Clinical Outcomes of Enamel Matrix Derivate Used in Surgical and Non-Surgical Treatment of Peri-Implantitis: A Systematic Review of Clinical Studies. Medicina 2022, 58, 1819. [Google Scholar] [CrossRef]
- Roccuzzo, A.; Imber, J.C.; Stahli, A.; Kloukos, D.; Salvi, G.E.; Sculean, A. Enamel matrix derivative as adjunctive to non-surgical periodontal therapy: A systematic review and meta-analysis of randomized controlled trials. Clin. Oral Investig. 2022, 26, 4263–4280. [Google Scholar] [CrossRef]
- Miron, R.J.; Sculean, A.; Cochran, D.L.; Froum, S.; Zucchelli, G.; Nemcovsky, C.; Donos, N.; Lyngstadaas, S.P.; Deschner, J.; Dard, M.; et al. Twenty years of enamel matrix derivative: The past, the present and the future. J. Clin. Periodontol. 2016, 43, 668–683. [Google Scholar] [CrossRef] [PubMed]
- Panahipour, L.; Sordi, M.B.; Kargarpour, Z.; Gruber, R. TGF-beta Signalling Mediates the Anti-Inflammatory Activity of Enamel Matrix Derivative In Vitro. Int. J. Mol. Sci. 2022, 23, 9778. [Google Scholar] [CrossRef]
- Sordi, M.B.; Cabral da Cruz, A.C.; Panahipour, L.; Gruber, R. Enamel Matrix Derivative Decreases Pyroptosis-Related Genes in Macrophages. Int. J. Mol. Sci. 2022, 23, 5078. [Google Scholar] [CrossRef] [PubMed]
- Gruber, R.; Roos, G.; Caballe-Serrano, J.; Miron, R.; Bosshardt, D.D.; Sculean, A. TGF-betaRI kinase activity mediates Emdogain-stimulated in vitro osteoclastogenesis. Clin. Oral Investig. 2014, 18, 1639–1646. [Google Scholar] [CrossRef] [PubMed]
- Gruber, R.; Bosshardt, D.D.; Miron, R.J.; Gemperli, A.C.; Buser, D.; Sculean, A. Enamel matrix derivative inhibits adipocyte differentiation of 3T3-L1 cells via activation of TGF-betaRI kinase activity. PLoS ONE 2013, 8, e71046. [Google Scholar] [CrossRef]
- Stahli, A.; Bosshardt, D.; Sculean, A.; Gruber, R. Emdogain-regulated gene expression in palatal fibroblasts requires TGF-betaRI kinase signaling. PLoS ONE 2014, 9, e105672. [Google Scholar] [CrossRef]
- Kapferer, I.; Schmidt, S.; Gstir, R.; Durstberger, G.; Huber, L.A.; Vietor, I. Gene-expression profiles of epithelial cells treated with EMD in vitro: Analysis using complementary DNA arrays. J. Periodontal Res. 2011, 46, 118–125. [Google Scholar] [CrossRef]
- Kawase, T.; Okuda, K.; Momose, M.; Kato, Y.; Yoshie, H.; Burns, D.M. Enamel matrix derivative (EMDOGAIN) rapidly stimulates phosphorylation of the MAP kinase family and nuclear accumulation of smad2 in both oral epithelial and fibroblastic human cells. J. Periodontal Res. 2001, 36, 367–376. [Google Scholar] [CrossRef]
- Fang, W.B.; Mafuvadze, B.; Yao, M.; Zou, A.; Portsche, M.; Cheng, N. TGF-beta Negatively Regulates CXCL1 Chemokine Expression in Mammary Fibroblasts through Enhancement of Smad2/3 and Suppression of HGF/c-Met Signaling Mechanisms. PLoS ONE 2015, 10, e0135063. [Google Scholar] [CrossRef]
- Zou, A.; Lambert, D.; Yeh, H.; Yasukawa, K.; Behbod, F.; Fan, F.; Cheng, N. Elevated CXCL1 expression in breast cancer stroma predicts poor prognosis and is inversely associated with expression of TGF-beta signaling proteins. BMC Cancer 2014, 14, 781. [Google Scholar] [CrossRef]
- Wang, Q.; Lin, W.; Zhou, X.; Lei, K.; Xu, R.; Zhang, X.; Xiong, Q.; Sheng, R.; Song, W.; Liu, W.; et al. Single-Cell Transcriptomic Atlas of Gingival Mucosa in Type 2 Diabetes. J. Dent. Res. 2022, 101, 1654–1664. [Google Scholar] [CrossRef] [PubMed]
- Finoti, L.S.; Nepomuceno, R.; Pigossi, S.C.; Corbi, S.C.; Secolin, R.; Scarel-Caminaga, R.M. Association between interleukin-8 levels and chronic periodontal disease: A PRISMA-compliant systematic review and meta-analysis. Medicine 2017, 96, e6932. [Google Scholar] [CrossRef] [PubMed]
- Aldahlawi, S.; Youssef, A.R.; Shahabuddin, S. Evaluation of chemokine CXCL10 in human gingival crevicular fluid, saliva, and serum as periodontitis biomarker. J. Inflamm. Res. 2018, 11, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Havens, A.M.; Chiu, E.; Taba, M.; Wang, J.; Shiozawa, Y.; Jung, Y.; Taichman, L.S.; D’Silva, N.J.; Gopalakrishnan, R.; Wang, C.; et al. Stromal-derived factor-1alpha (CXCL12) levels increase in periodontal disease. J. Periodontol. 2008, 79, 845–853. [Google Scholar] [CrossRef] [PubMed]
- Gamonal, J.; Bascones, A.; Jorge, O.; Silva, A. Chemokine RANTES in gingival crevicular fluid of adult patients with periodontitis. J. Clin. Periodontol. 2000, 27, 675–681. [Google Scholar] [CrossRef] [PubMed]
- Zhan, C.; Zhou, Z.; Huang, Y.; Huang, S.; Lin, Z.; He, F.; Song, Z. Exploration of the shared gene signatures and molecular mechanisms between periodontitis and inflammatory bowel disease: Evidence from transcriptome data. Gastroenterol. Rep. 2023, 11, goad041. [Google Scholar] [CrossRef]
- Shinjo, T.; Onizuka, S.; Zaitsu, Y.; Ishikado, A.; Park, K.; Li, Q.; Yokomizo, H.; Zeze, T.; Sato, K.; St-Louis, R.; et al. Dysregulation of CXCL1 Expression and Neutrophil Recruitment in Insulin Resistance and Diabetes-Related Periodontitis in Male Mice. Diabetes 2023, 72, 986–998. [Google Scholar] [CrossRef]
- Santos, A.F.P.; Cervantes, L.C.C.; Panahipour, L.; Souza, F.A.; Gruber, R. Proof-of-Principle Study Suggesting Potential Anti-Inflammatory Activity of Butyrate and Propionate in Periodontal Cells. Int. J. Mol. Sci. 2022, 23, 11006. [Google Scholar] [CrossRef]
- Rath-Deschner, B.; Memmert, S.; Damanaki, A.; Nokhbehsaim, M.; Eick, S.; Cirelli, J.A.; Gotz, W.; Deschner, J.; Jager, A.; Nogueira, A.V.B. CXCL1, CCL2, and CCL5 modulation by microbial and biomechanical signals in periodontal cells and tissues-in vitro and in vivo studies. Clin. Oral Investig. 2020, 24, 3661–3670. [Google Scholar] [CrossRef]
- Ramage, G.; Lappin, D.F.; Millhouse, E.; Malcolm, J.; Jose, A.; Yang, J.; Bradshaw, D.J.; Pratten, J.R.; Culshaw, S. The epithelial cell response to health and disease associated oral biofilm models. J. Periodontal Res. 2017, 52, 325–333. [Google Scholar] [CrossRef]
- Greer, A.; Irie, K.; Hashim, A.; Leroux, B.G.; Chang, A.M.; Curtis, M.A.; Darveau, R.P. Site-Specific Neutrophil Migration and CXCL2 Expression in Periodontal Tissue. J. Dent. Res. 2016, 95, 946–952. [Google Scholar] [CrossRef]
- Dommisch, H.; Staufenbiel, I.; Schulze, K.; Stiesch, M.; Winkel, A.; Fimmers, R.; Dommisch, J.; Jepsen, S.; Miosge, N.; Adam, K.; et al. Expression of antimicrobial peptides and interleukin-8 during early stages of inflammation: An experimental gingivitis study. J. Periodontal Res. 2015, 50, 836–845. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Wang, J.; Wang, K.; Li, C.; Zhang, S.; Jing, D.; Xu, C.; Wang, X.; Zhao, H.; Feng, J.Q. Axin2(+)-Mesenchymal PDL Cells, Instead of K14(+) Epithelial Cells, Play a Key Role in Rapid Cementum Growth. J. Dent. Res. 2019, 98, 1262–1270. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Dong, X.; Ma, Y.; Yao, J. Chemokine (C-X-C motif) ligand 1 maintains the immune surveillance function of natural killer cells via the PDK2/mTOR signaling pathway. Cell Biol. Toxicol. 2022. [Google Scholar] [CrossRef]
- Nii, T.; Yumoto, H.; Hirota, K.; Miyake, Y. Anti-inflammatory effects of olanexidine gluconate on oral epithelial cells. BMC Oral Health 2019, 19, 239. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, W.; Wang, Q. 1,25-dihydroxyvitamin D(3) suppresses lipopolysaccharide-induced interleukin-6 production through aryl hydrocarbon receptor/nuclear factor-kappaB signaling in oral epithelial cells. BMC Oral Health 2019, 19, 236. [Google Scholar] [CrossRef]
- Panahipour, L.; Oladzad Abbasabadi, A.; Gruber, R. Oral Cell Lysates Reduce the Inflammatory Response of Activated Macrophages. J. Clin. Med. 2023, 12, 1701. [Google Scholar] [CrossRef]
- An, N.; Holl, J.; Wang, X.; Rausch, M.A.; Andrukhov, O.; Rausch-Fan, X. Potential Suppressive Effect of Nicotine on the Inflammatory Response in Oral Epithelial Cells: An In Vitro Study. Int. J. Environ. Res. Public Health 2021, 18, 483. [Google Scholar] [CrossRef]
- Ciesielska, A.; Matyjek, M.; Kwiatkowska, K. TLR4 and CD14 trafficking and its influence on LPS-induced pro-inflammatory signaling. Cell Mol. Life Sci. 2021, 78, 1233–1261. [Google Scholar] [CrossRef]
- Uehara, A.; Sugawara, S.; Takada, H. Priming of human oral epithelial cells by interferon-gamma to secrete cytokines in response to lipopolysaccharides, lipoteichoic acids and peptidoglycans. J. Med. Microbiol. 2002, 51, 626–634. [Google Scholar] [CrossRef]
- Tamai, R.; Sugawara, S.; Takeuchi, O.; Akira, S.; Takada, H. Synergistic effects of lipopolysaccharide and interferon-gamma in inducing interleukin-8 production in human monocytic THP-1 cells is accompanied by up-regulation of CD14, Toll-like receptor 4, MD-2 and MyD88 expression. J. Endotoxin Res. 2003, 9, 145–153. [Google Scholar] [CrossRef]
- Pourgonabadi, S.; Muller, H.D.; Mendes, J.R.; Gruber, R. Saliva initiates the formation of pro-inflammatory macrophages in vitro. Arch. Oral Biol. 2017, 73, 295–301. [Google Scholar] [CrossRef]
- Muller, H.D.; Cvikl, B.B.; Lussi, A.A.; Gruber, R.R. Salivary pellets induce a pro-inflammatory response involving the TLR4-NF-kB pathway in gingival fibroblasts. BMC Oral Health 2016, 17, 15. [Google Scholar] [CrossRef] [PubMed]
- Derynck, R.; Akhurst, R.J.; Balmain, A. TGF-beta signaling in tumor suppression and cancer progression. Nat. Genet. 2001, 29, 117–129. [Google Scholar] [CrossRef] [PubMed]
- Fiorillo, L.; Cervino, G.; Herford, A.S.; Lauritano, F.; D’Amico, C.; Lo Giudice, R.; Laino, L.; Troiano, G.; Crimi, S.; Cicciu, M. Interferon Crevicular Fluid Profile and Correlation with Periodontal Disease and Wound Healing: A Systemic Review of Recent Data. Int. J. Mol. Sci. 2018, 19, 1908. [Google Scholar] [CrossRef] [PubMed]
- Gautam, A.; Pandit, B. IL32: The multifaceted and unconventional cytokine. Hum. Immunol. 2021, 82, 659–667. [Google Scholar] [CrossRef]
- Johnson, D.; Carbonetti, N. Roles and Effects of Interferon Lambda Signaling in the Context of Bacterial Infections. J. Interferon Cytokine Res. 2023. [Google Scholar] [CrossRef]
- Manivasagam, S.; Klein, R.S. Type III Interferons: Emerging Roles in Autoimmunity. Front. Immunol. 2021, 12, 764062. [Google Scholar] [CrossRef]
- Goel, R.R.; Kotenko, S.V.; Kaplan, M.J. Interferon lambda in inflammation and autoimmune rheumatic diseases. Nat. Rev. Rheumatol. 2021, 17, 349–362. [Google Scholar] [CrossRef]
- Deng, Z.; Hu, W.; Ai, H.; Chen, Y.; Dong, S. The Dramatic Role of IFN Family in Aberrant Inflammatory Osteolysis. Curr. Gene Ther. 2021, 21, 112–129. [Google Scholar] [CrossRef]
- Wang, J.M.; Huang, A.F.; Xu, W.D.; Su, L.C. Insights into IL-29: Emerging role in inflammatory autoimmune diseases. J. Cell. Mol. Med. 2019, 23, 7926–7932. [Google Scholar] [CrossRef] [PubMed]
- Haller, O.; Staeheli, P.; Schwemmle, M.; Kochs, G. Mx GTPases: Dynamin-like antiviral machines of innate immunity. Trends Microbiol. 2015, 23, 154–163. [Google Scholar] [CrossRef] [PubMed]
- Sack, G.H., Jr. Serum Amyloid A (SAA) Proteins. Subcell. Biochem. 2020, 94, 421–436. [Google Scholar] [CrossRef] [PubMed]
- Heinz, L.X.; Rebsamen, M.; Rossi, D.C.; Staehli, F.; Schroder, K.; Quadroni, M.; Gross, O.; Schneider, P.; Tschopp, J. The death domain-containing protein Unc5CL is a novel MyD88-independent activator of the pro-inflammatory IRAK signaling cascade. Cell Death Differ. 2012, 19, 722–731. [Google Scholar] [CrossRef]
- Hoshikawa, H.; Goto, R.; Mori, T.; Mitani, T.; Mori, N. Expression of prostaglandin E2 receptors in oral squamous cell carcinomas and growth inhibitory effects of an EP3 selective antagonist, ONO-AE3-240. Int. J. Oncol. 2009, 34, 847–852. [Google Scholar] [CrossRef]
- Osawa, K.; Umemura, M.; Nakakaji, R.; Tanaka, R.; Islam, R.M.; Nagasako, A.; Fujita, T.; Yokoyama, U.; Koizumi, T.; Mitsudo, K.; et al. Prostaglandin E(2) receptor EP4 regulates cell migration through Orai1. Cancer Sci. 2020, 111, 160–174. [Google Scholar] [CrossRef]
- Cho, Y.J.; Liang, P. Killin is a p53-regulated nuclear inhibitor of DNA synthesis. Proc. Natl. Acad. Sci. USA 2008, 105, 5396–5401. [Google Scholar] [CrossRef]
- Kargarpour, Z.; Nasirzade, J.; Panahipour, L.; Miron, R.J.; Gruber, R. Platelet-Rich Fibrin Decreases the Inflammatory Response of Mesenchymal Cells. Int. J. Mol. Sci. 2021, 22, 11333. [Google Scholar] [CrossRef]
- Di Summa, F.; Kargarpour, Z.; Nasirzade, J.; Stahli, A.; Mitulovic, G.; Panic-Jankovic, T.; Koller, V.; Kaltenbach, C.; Muller, H.; Panahipour, L.; et al. TGFbeta activity released from platelet-rich fibrin adsorbs to titanium surface and collagen membranes. Sci. Rep. 2020, 10, 10203. [Google Scholar] [CrossRef]
- Luo, Q.; Liu, Y.; Shi, K.; Shen, X.; Yang, Y.; Liang, X.; Lu, L.; Qiao, W.; Chen, A.; Hong, D.; et al. An autonomous activation of interleukin-17 receptor signaling sustains inflammation and promotes disease progression. Immunity 2023. [Google Scholar] [CrossRef]
- Wiley, C.D.; Velarde, M.C.; Lecot, P.; Liu, S.; Sarnoski, E.A.; Freund, A.; Shirakawa, K.; Lim, H.W.; Davis, S.S.; Ramanathan, A.; et al. Mitochondrial Dysfunction Induces Senescence with a Distinct Secretory Phenotype. Cell Metab. 2016, 23, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Mao, P.; Wu, S.; Li, J.; Fu, W.; He, W.; Liu, X.; Slutsky, A.S.; Zhang, H.; Li, Y. Human alveolar epithelial type II cells in primary culture. Physiol. Rep. 2015, 3, 12288. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Peng, J.; Sun, T.; Li, N.; Zhang, L.; Ren, J.; Yuan, H.; Kan, S.; Pan, Q.; Li, X.; et al. Epithelial EZH2 serves as an epigenetic determinant in experimental colitis by inhibiting TNFalpha-mediated inflammation and apoptosis. Proc. Natl. Acad. Sci. USA 2017, 114, E3796–E3805. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).