Improving the Antimycobacterial Drug Clofazimine through Formation of Organic Salts by Combination with Fluoroquinolones
Abstract
1. Introduction
2. Results and Discussion
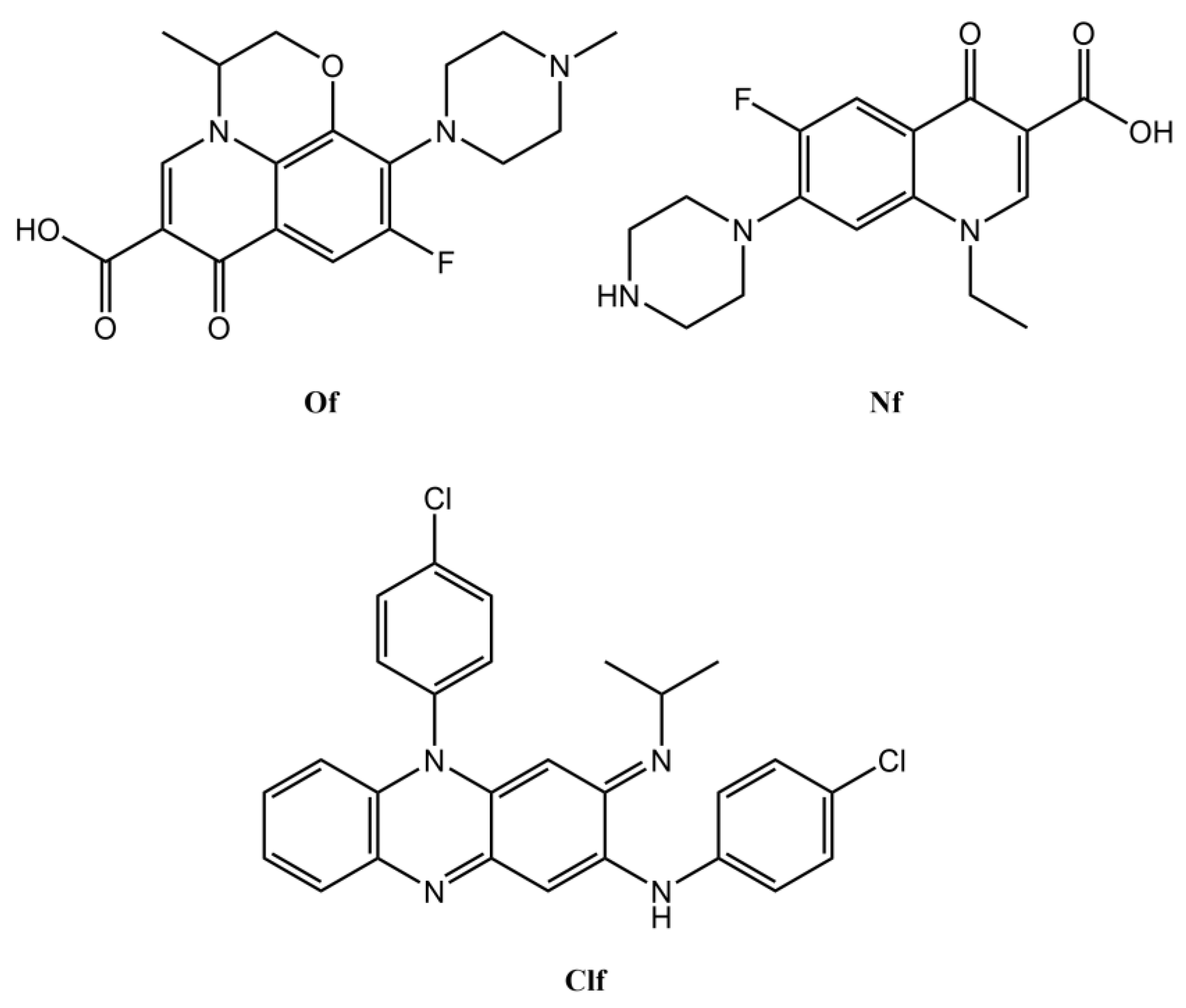
2.1. Synthesis and Physico-Chemical Properties of the Clf-Derived OSs
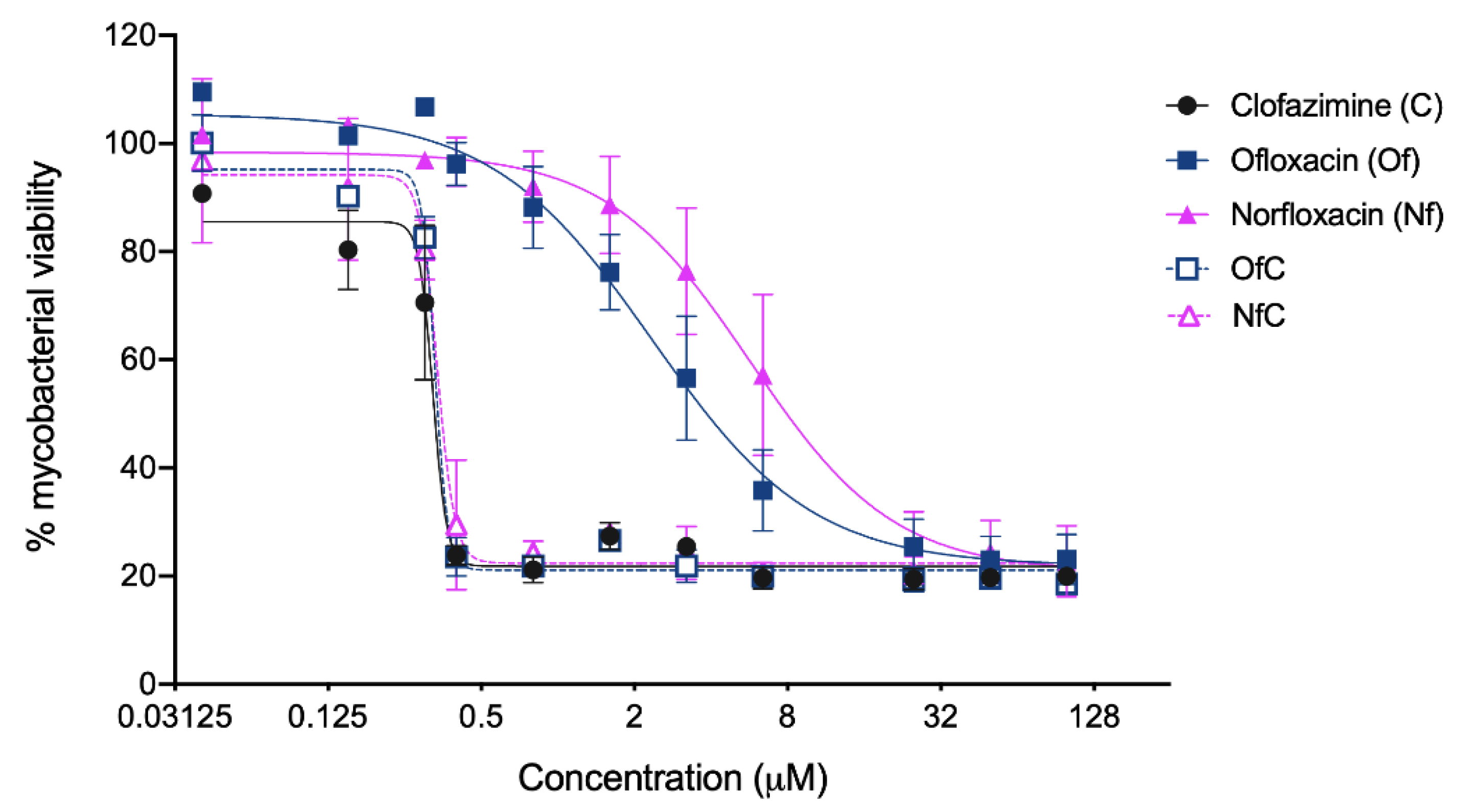
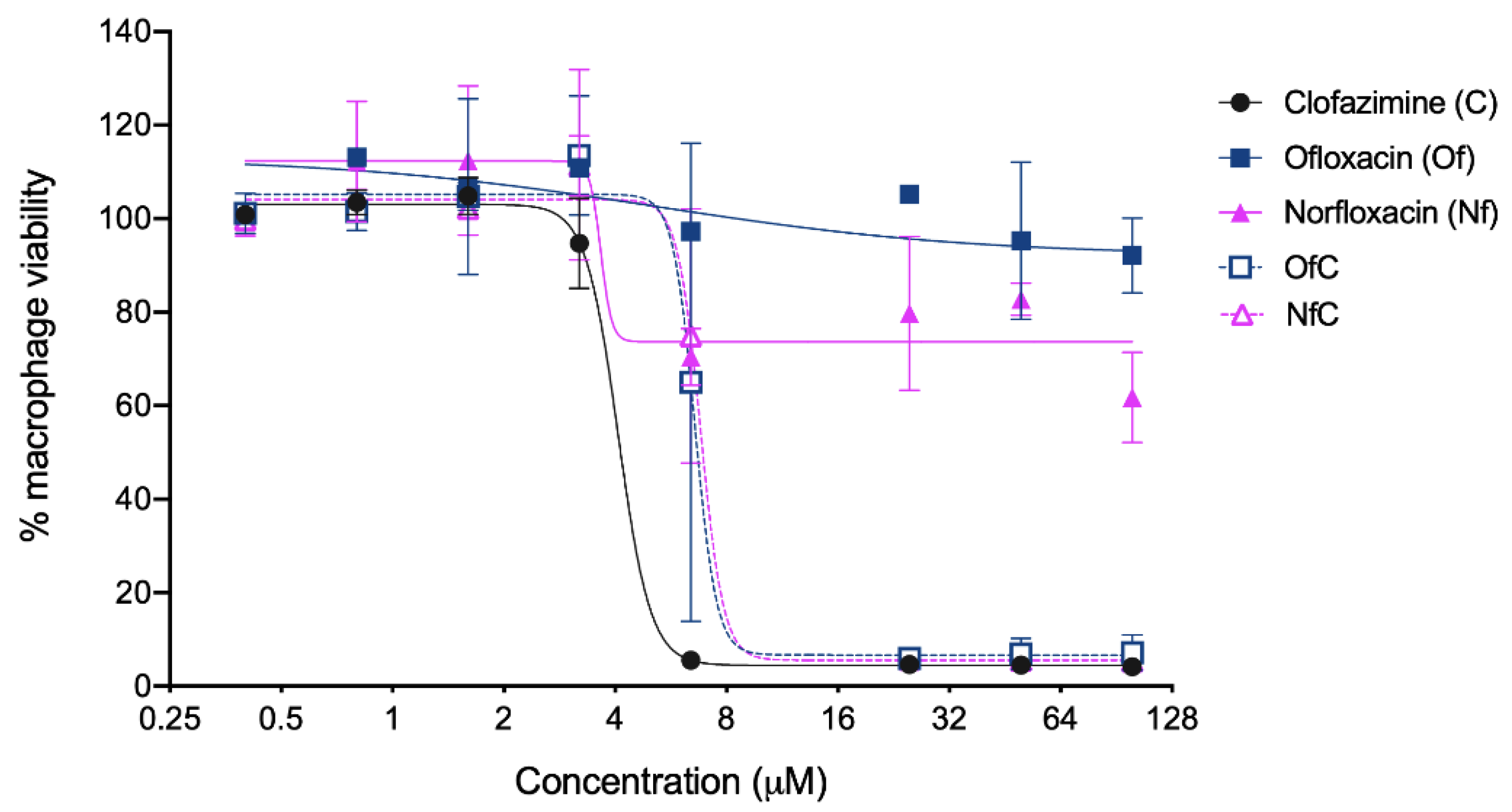
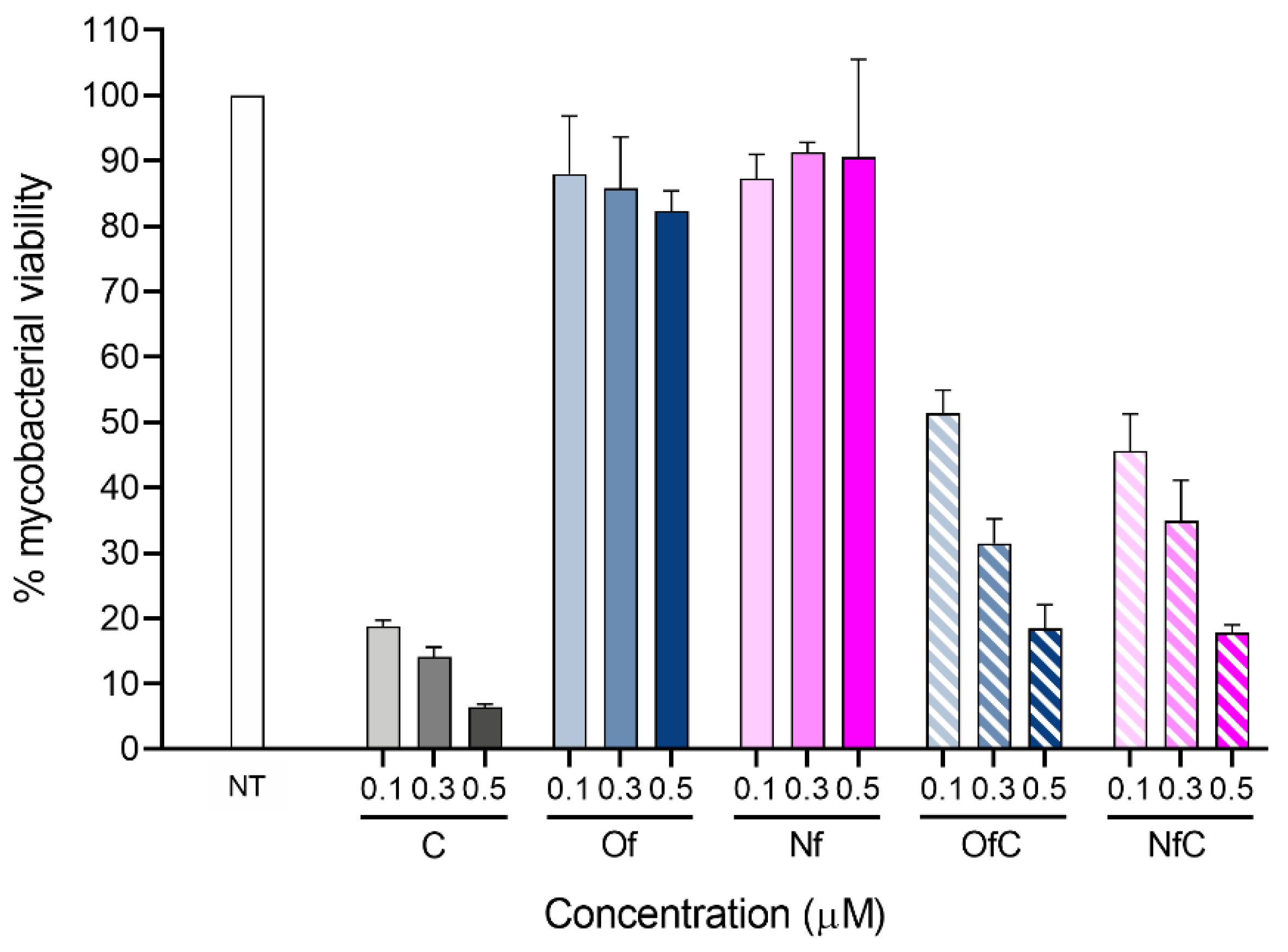
2.2. In Vitro Activity of the Clf-Derived OSs against M. avium
3. Materials and Methods
3.1. Compounds
3.2. Simultaneous Thermogravimetry Analysis
3.3. Bacteria
3.4. Direct Effect of Test Compounds against Mycobacteria
3.5. Bone Marrow-Derived Macrophages (BMM)
3.6. IC50 and LD50 Determination
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Johansen, M.D.; Herrmann, J.L.; Kremer, L. Non-tuberculous mycobacteria and the rise of Mycobacterium abscessus. Nat. Rev. Microbiol. 2020, 18, 392–407. [Google Scholar] [CrossRef] [PubMed]
- Brode, S.K.; Daley, C.L.; Marras, T.K. The epidemiologic relationship between tuberculosis and non-tuberculous mycobacterial disease: A systematic review. Int. J. Tuberc. Lung Dis. 2014, 18, 1370–1377. [Google Scholar] [CrossRef] [PubMed]
- Ratnatunga, C.N.; Lutzky, V.P.; Kupz, A.; Doolan, D.L.; Reid, D.W.; Field, M.; Bell, S.C.; Thomson, R.M.; Miles, J.J. The Rise of Non-Tuberculosis Mycobacterial Lung Disease. Front. Immunol. 2020, 11, 303. [Google Scholar] [CrossRef] [PubMed]
- Gomes, M.S.; Paul, S.; Moreira, A.L.; Appelberg, R.; Rabinovitch, M.; Kaplan, G. Survival of Mycobacterium avium and Mycobacterium tuberculosis in acidified vacuoles of murine macrophages. Infect. Immun. 1999, 67, 3199–3206. [Google Scholar] [CrossRef] [PubMed]
- Daley, C.L.; Iaccarino, J.M.; Lange, C.; Cambau, E.; Wallace, R.J., Jr.; Andrejak, C.; Bottger, E.C.; Brozek, J.; Griffith, D.E.; Guglielmetti, L.; et al. Treatment of Nontuberculous Mycobacterial Pulmonary Disease: An Official ATS/ERS/ESCMID/IDSA Clinical Practice Guideline. Clin. Infect. Dis. 2020, 71, e1–e36. [Google Scholar] [CrossRef]
- Griffith, D.E.; Aksamit, T.; Brown-Elliott, B.A.; Catanzaro, A.; Daley, C.; Gordin, F.; Holland, S.M.; Horsburgh, R.; Huitt, G.; Iademarco, M.F.; et al. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. Am. J. Respir. Crit. Care Med. 2007, 175, 367–416. [Google Scholar] [CrossRef]
- Zheng, W.; Sun, W.; Simeonov, A. Drug repurposing screens and synergistic drug-combinations for infectious diseases. Br. J. Pharmacol. 2018, 175, 181–191. [Google Scholar] [CrossRef]
- Hough, W.L.; Smiglak, M.; Rodríguez, H.; Swatloski, R.P.; Spear, S.K.; Daly, D.T.; Pernak, J.; Grisel, J.E.; Carliss, R.D.; Soutullo, M.D.; et al. The third evolution of ionic liquids: Active pharmaceutical ingredients. New J. Chem. 2007, 31, 1429–1436. [Google Scholar] [CrossRef]
- Ferraz, R.; Branco, L.C.; Prudencio, C.; Noronha, J.P.; Petrovski, Z. Ionic liquids as active pharmaceutical ingredients. ChemMedChem 2011, 6, 975–985. [Google Scholar] [CrossRef]
- Shamshina, J.L.; Rogers, R.D. Are Myths and Preconceptions Preventing us from Applying Ionic Liquid Forms of Antiviral Medicines to the Current Health Crisis? Int. J. Mol. Sci. 2020, 21, 6002. [Google Scholar] [CrossRef]
- Egorova, K.S.; Gordeev, E.G.; Ananikov, V.P. Biological Activity of Ionic Liquids and Their Application in Pharmaceutics and Medicine. Chem. Rev. 2017, 117, 7132–7189. [Google Scholar] [CrossRef] [PubMed]
- Pedro, S.N.; CS, R.F.; Silvestre, A.J.D.; Freire, M.G. The Role of Ionic Liquids in the Pharmaceutical Field: An Overview of Relevant Applications. Int. J. Mol. Sci. 2020, 21, 8298. [Google Scholar] [CrossRef] [PubMed]
- Gomes, A.; Aguiar, L.; Ferraz, R.; Teixeira, C.; Gomes, P. The Emerging Role of Ionic Liquid-Based Approaches for Enhanced Skin Permeation of Bioactive Molecules: A Snapshot of the Past Couple of Years. Int. J. Mol. Sci. 2021, 22, 11991. [Google Scholar] [CrossRef] [PubMed]
- Welton, T. Room-Temperature Ionic Liquids. Solvents for Synthesis and Catalysis. Chem. Rev. 1999, 99, 2071–2084. [Google Scholar] [CrossRef]
- Balk, A.; Holzgrabe, U.; Meinel, L. ‘Pro et contra’ ionic liquid drugs—Challenges and opportunities for pharmaceutical translation. Eur. J. Pharm. Biopharm. 2015, 94, 291–304. [Google Scholar] [CrossRef]
- Pérez, R.L.; Ayala, C.E.; Warner, I.M. Group of Uniform Materials Based on Organic Salts (GUMBOS): A Review of Their Solid State Properties and Applications. In Ionic Liquids-Thermophysical Properties and Applications; IntechOpen: London, UK, 2021. [Google Scholar] [CrossRef]
- Kaur, P.; Chopra, H.K. Recent Progress in Synthesis and Applications of Tunable Materials and Nanomaterials Based on Organic Salts. ChemistrySelect 2020, 5, 13033–13053. [Google Scholar] [CrossRef]
- Azevedo, A.M.O.; Santos, J.L.M.; Warner, I.M.; Saraiva, M. GUMBOS and nanoGUMBOS in chemical and biological analysis: A review. Anal. Chim. Acta 2020, 1133, 180–198. [Google Scholar] [CrossRef]
- McCrary, P.D.; Beasley, P.A.; Gurau, G.; Narita, A.; Barber, P.S.; Cojocaru, O.A.; Rogers, R.D. Drug specific, tuning of an ionic liquid’s hydrophilic–lipophilic balance to improve water solubility of poorly soluble active pharmaceutical ingredients. New J. Chem. 2013, 37, 2196–2202. [Google Scholar] [CrossRef]
- Marrucho, I.M.; Branco, L.C.; Rebelo, L.P. Ionic liquids in pharmaceutical applications. Annu. Rev. Chem. Biomol. Eng. 2014, 5, 527–546. [Google Scholar] [CrossRef]
- Silva, A.T.; Oliveira, I.S.; Gomes, J.; Aguiar, L.; Fontinha, D.; Duarte, D.; Nogueira, F.; Prudencio, M.; Marques, E.F.; Teixeira, C.; et al. Drug-Derived Surface-Active Ionic Liquids: A Cost-Effective Way To Expressively Increase the Blood-Stage Antimalarial Activity of Primaquine. ChemMedChem 2022, 17, e202100650. [Google Scholar] [CrossRef]
- Ferraz, R.; Silva, D.; Dias, A.R.; Dias, V.; Santos, M.M.; Pinheiro, L.; Prudencio, C.; Noronha, J.P.; Petrovski, Z.; Branco, L.C. Synthesis and Antibacterial Activity of Ionic Liquids and Organic Salts Based on Penicillin G and Amoxicillin hydrolysate Derivatives against Resistant Bacteria. Pharmaceutics 2020, 12, 221. [Google Scholar] [CrossRef] [PubMed]
- Prudencio, C.; Vieira, M.; Van der Auweraer, S.; Ferraz, R. Recycling Old Antibiotics with Ionic Liquids. Antibiotics 2020, 9, 578. [Google Scholar] [CrossRef] [PubMed]
- Bento, C.M.; Gomes, M.S.; Silva, T. Looking beyond Typical Treatments for Atypical Mycobacteria. Antibiotics 2020, 9, 18. [Google Scholar] [CrossRef]
- Silva, A.T.; Bento, C.M.; Pena, A.C.; Figueiredo, L.M.; Prudencio, C.; Aguiar, L.; Silva, T.; Ferraz, R.; Gomes, M.S.; Teixeira, C.; et al. Cinnamic Acid Conjugates in the Rescuing and Repurposing of Classical Antimalarial Drugs. Molecules 2019, 25, 66. [Google Scholar] [CrossRef]
- Cantelli, C.R.; Dassonville-Klimpt, A.; Sonnet, P. A review of current and promising nontuberculous mycobacteria antibiotics. Future Med. Chem. 2021, 13, 1367–1395. [Google Scholar] [CrossRef] [PubMed]
- Sano, C.; Tatano, Y.; Shimizu, T.; Yamabe, S.; Sato, K.; Tomioka, H. Comparative in vitro and in vivo antimicrobial activities of sitafloxacin, gatifloxacin and moxifloxacin against Mycobacterium avium. Int. J. Antimicrob. Agents 2011, 37, 296–301. [Google Scholar] [CrossRef] [PubMed]
- Khadawardi, H.; Marras, T.K.; Mehrabi, M.; Brode, S.K. Clinical efficacy and safety of fluoroquinolone containing regimens in patients with Mycobacterium avium complex pulmonary disease. Eur. Respir. J. 2020, 55, 1901240. [Google Scholar] [CrossRef]
- Mirnejad, R.; Asadi, A.; Khoshnood, S.; Mirzaei, H.; Heidary, M.; Fattorini, L.; Ghodousi, A.; Darban-Sarokhalil, D. Clofazimine: A useful antibiotic for drug-resistant tuberculosis. Biomed. Pharmacother. 2018, 105, 1353–1359. [Google Scholar] [CrossRef]
- Jarand, J.; Davis, J.P.; Cowie, R.L.; Field, S.K.; Fisher, D.A. Long-term Follow-up of Mycobacterium avium Complex Lung Disease in Patients Treated With Regimens Including Clofazimine and/or Rifampin. Chest 2016, 149, 1285–1293. [Google Scholar] [CrossRef]
- Yang, B.; Jhun, B.W.; Moon, S.M.; Lee, H.; Park, H.Y.; Jeon, K.; Kim, D.H.; Kim, S.Y.; Shin, S.J.; Daley, C.L.; et al. Clofazimine-Containing Regimen for the Treatment of Mycobacterium abscessus Lung Disease. Antimicrob. Agents Chemother. 2017, 61, e02052-16. [Google Scholar] [CrossRef]
- Martiniano, S.L.; Wagner, B.D.; Levin, A.; Nick, J.A.; Sagel, S.D.; Daley, C.L. Safety and Effectiveness of Clofazimine for Primary and Refractory Nontuberculous Mycobacterial Infection. Chest 2017, 152, 800–809. [Google Scholar] [CrossRef] [PubMed]
- Fukutomi, Y.; Maeda, Y.; Makino, M. Apoptosis-inducing activity of clofazimine in macrophages. Antimicrob. Agents Chemother. 2011, 55, 4000–4005. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Bhattacharya, D.; Gupta, N.; Rawat, V.; Tousif, S.; Van Kaer, L.; Das, G. Clofazimine enhances the efficacy of BCG revaccination via stem cell-like memory T cells. PLoS Pathog. 2020, 16, e1008356. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, M.J.; Calcagno, T.; Hosseini, S.S.; Hematian, A.; Nojookambari, N.Y.; Karimi-Yazdi, M.; Mirsaeidi, M. Role of Clofazimine in Treatment of Mycobacterium avium Complex. Front. Med. 2021, 8, 638306. [Google Scholar] [CrossRef] [PubMed]
- Global Alliance for TB Drug Development. Clofazimine. Tuberculosis 2008, 88, 96–99. [Google Scholar] [CrossRef]
- Sng, L.H.; Peh, J.W.L.; Kee, M.T.L.; Ya’akob, N.B.M.; Ong, R.T.; Wong, C.W.; Chee, C.B.E.; Wang, Y.T. Clofazimine drug susceptibility testing for Mycobacterium tuberculosis: The case of using the right diluent. Pathology 2018, 50, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Amidon, G.L.; Lennernas, H.; Shah, V.P.; Crison, J.R. A theoretical basis for a biopharmaceutic drug classification: The correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm. Res. 1995, 12, 413–420. [Google Scholar] [CrossRef]
- Dahlgren, D.; Sjogren, E.; Lennernas, H. Intestinal absorption of BCS class II drugs administered as nanoparticles: A review based on in vivo data from intestinal perfusion models. ADMET DMPK 2020, 8, 375–390. [Google Scholar] [CrossRef]
- Silva, T.; Magalhaes, B.; Maia, S.; Gomes, P.; Nazmi, K.; Bolscher, J.G.; Rodrigues, P.N.; Bastos, M.; Gomes, M.S. Killing of Mycobacterium avium by lactoferricin peptides: Improved activity of arginine- and D-amino-acid-containing molecules. Antimicrob. Agents Chemother. 2014, 58, 3461–3467. [Google Scholar] [CrossRef]
- CLSI. Susceptibility Testing of Mycobacteria, Nocardiae, and Other Aerobic Actinomycetes; Approved Standard—Second Edition. CLSI document M24-A2; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2011. [Google Scholar]
- Silva, T.; Moreira, A.C.; Nazmi, K.; Moniz, T.; Vale, N.; Rangel, M.; Gomes, P.; Bolscher, J.G.M.; Rodrigues, P.N.; Bastos, M.; et al. Lactoferricin Peptides Increase Macrophages’ Capacity To Kill Mycobacterium avium. mSphere 2017, 2, e00301-17. [Google Scholar] [CrossRef]
- Sousa, M.L.; Sarraguça, M.C.; Oliveira dos Santos, A.; Sarraguça, J.M.G.; Lopes, J.; Ribeiro, P.R.S. A new salt of clofazimine to improve leprosy treatment. J. Mol. Struct. 2020, 1214, 128226. [Google Scholar] [CrossRef]
- Bodart, L.; Derlet, A.; Buol, X.; Leyssens, T.; Tumanov, N.; Wouters, J. Combining Two Antitubercular Drugs, Clofazimine and 4-Aminosalicylic Acid, in Order to Improve Clofazimine Aqueous Solubility and 4-Aminosalicylic Acid Thermal Stability. J. Pharm. Sci. 2020, 109, 3645–3652. [Google Scholar] [CrossRef] [PubMed]
- Chaves, L.L.; Lima, S.; Vieira, A.C.C.; Ferreira, D.; Sarmento, B.; Reis, S. Overcoming clofazimine intrinsic toxicity: Statistical modelling and characterization of solid lipid nanoparticles. J. R. Soc. Interface 2018, 15, 20170932. [Google Scholar] [CrossRef]
- Zhang, Y.; Feng, J.; McManus, S.A.; Lu, H.D.; Ristroph, K.D.; Cho, E.J.; Dobrijevic, E.L.; Chan, H.K.; Prud’homme, R.K. Design and Solidification of Fast-Releasing Clofazimine Nanoparticles for Treatment of Cryptosporidiosis. Mol. Pharm. 2017, 14, 3480–3488. [Google Scholar] [CrossRef] [PubMed]
- Peters, K.; Leitzke, S.; Diederichs, J.E.; Borner, K.; Hahn, H.; Muller, R.H.; Ehlers, S. Preparation of a clofazimine nanosuspension for intravenous use and evaluation of its therapeutic efficacy in murine Mycobacterium avium infection. J. Antimicrob. Chemother. 2000, 45, 77–83. [Google Scholar] [CrossRef] [PubMed]
- van Staden, D.; Haynes, R.K.; Viljoen, J.M. Adapting Clofazimine for Treatment of Cutaneous Tuberculosis by Using Self-Double-Emulsifying Drug Delivery Systems. Antibiotics 2022, 11, 806. [Google Scholar] [CrossRef]
- Bolla, G.; Nangia, A. Clofazimine Mesylate: A High Solubility Stable Salt. Cryst. Growth Des. 2012, 12, 6250–6259. [Google Scholar] [CrossRef]
- Baaqel, H.; Tulus, V.; Chachuat, B.; Guillén-Gosálbez, G.; Hallett, J. Uncovering the True Cost of Ionic Liquids using Monetization. Comput. Aided Chem. Eng. 2020, 48, 1825–1830. [Google Scholar] [CrossRef]
- Vilchez-Chavez, A.; Carruitero, M.J.; Chavez-Cruzado, E. Cetylpyridinium chloride mouthwashes: Potential role in COVID-19 control. J. Oral Maxillofac. Surg. Med. Pathol. 2022, 34, 213. [Google Scholar] [CrossRef]
- Van Deun, A.; Maug, A.K.; Salim, M.A.; Das, P.K.; Sarker, M.R.; Daru, P.; Rieder, H.L. Short, highly effective, and inexpensive standardized treatment of multidrug-resistant tuberculosis. Am. J. Respir. Crit. Care Med. 2010, 182, 684–692. [Google Scholar] [CrossRef]
- Silva, P.E.; Costa, P.S.; Avila, M.P.; Suhadolnik, M.L.; Reis, M.P.; Salgado, A.P.; Lima, M.F.; Chartone-Souza, E.; Nascimento, A.M. Leprous lesion presents enrichment of opportunistic pathogenic bacteria. Springerplus 2015, 4, 187. [Google Scholar] [CrossRef] [PubMed]
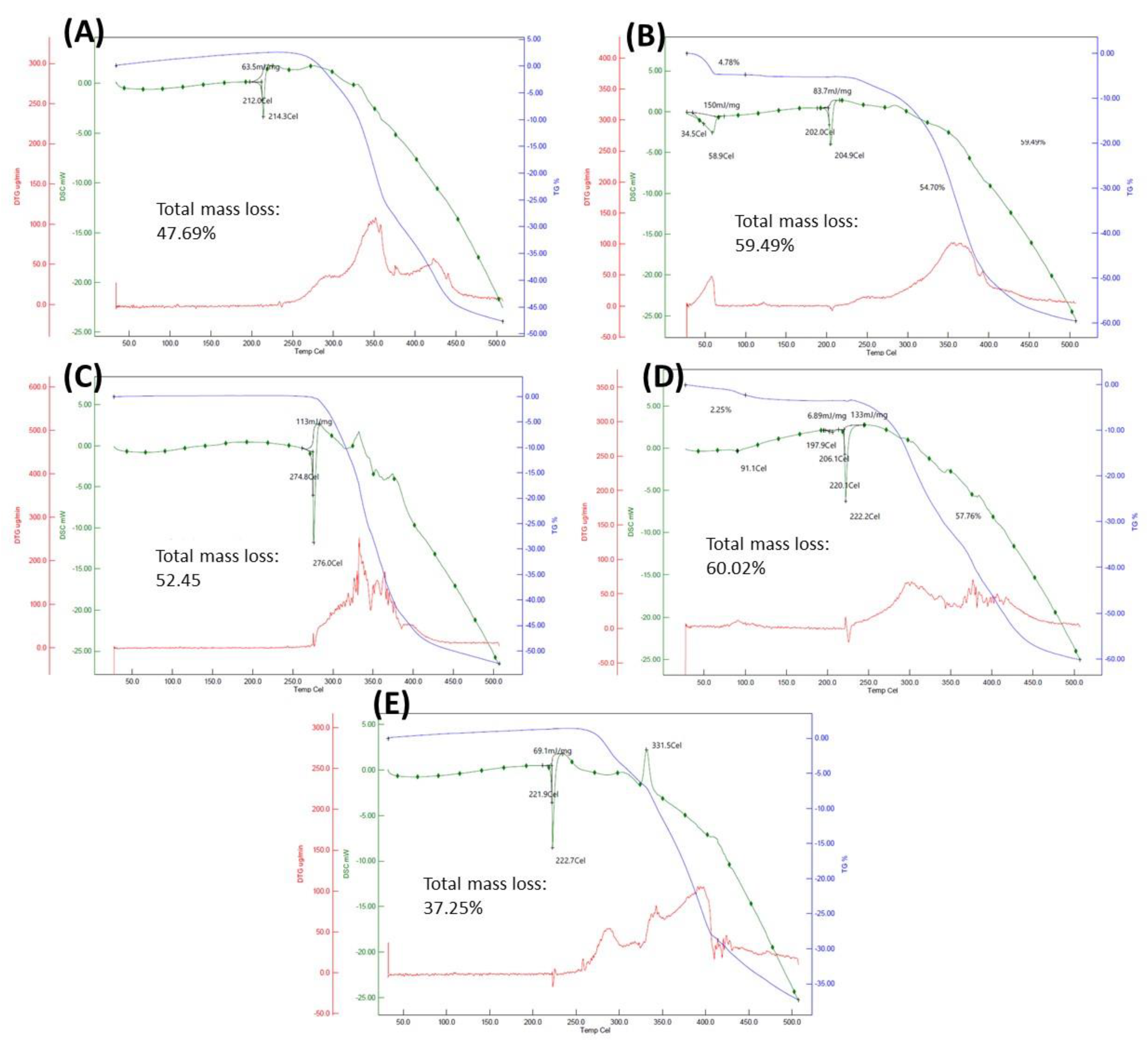
| Compound | Molecular Weight/g·mol−1 | Net Charge 2 | Melting Point (Decomposition Temperature)/°C | Solubility/mg·mL−1 (mM) | IC50/µM 3 | LC50/µM 4 |
|---|---|---|---|---|---|---|
| Clf | 472.12 | +1 | 221.9 (258.0) | 4.8 (10.2) | 0.33 (0.38 to 0.31) 5 | 4.1 (4.9 to 3.7) 5 |
| Of | 361.37 | −1 | 274.8 (318.8) | 5.7 (15.7) | 3.85 (4.78 to 3.20) 5 | >100 |
| Nf | 319.33 | −1 | 206.1 (222.0) | 11.0 (34.4) | 7.93 (10.94 to 6.25) 5 | >100 |
| [Clf][Of] | 833.26 | 0 | 212.0 (274.3) | 13.9 (15.8) | 0.34 (0.36 to 0.32) 5 | 6.7 (13.04 to 3.42) 5 |
| [Clf][Nf] | 791.25 | 0 | 202.0 (354.6) | 13.0 (17.3) | 0.35 (0.38 to 0.33) 5 | 6.9 (13.44 to 3.57) 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bento, C.M.; Silva, A.T.; Mansano, B.; Aguiar, L.; Teixeira, C.; Gomes, M.S.; Gomes, P.; Silva, T.; Ferraz, R. Improving the Antimycobacterial Drug Clofazimine through Formation of Organic Salts by Combination with Fluoroquinolones. Int. J. Mol. Sci. 2023, 24, 1402. https://doi.org/10.3390/ijms24021402
Bento CM, Silva AT, Mansano B, Aguiar L, Teixeira C, Gomes MS, Gomes P, Silva T, Ferraz R. Improving the Antimycobacterial Drug Clofazimine through Formation of Organic Salts by Combination with Fluoroquinolones. International Journal of Molecular Sciences. 2023; 24(2):1402. https://doi.org/10.3390/ijms24021402
Chicago/Turabian StyleBento, Clara M., Ana Teresa Silva, Bruno Mansano, Luísa Aguiar, Cátia Teixeira, Maria Salomé Gomes, Paula Gomes, Tânia Silva, and Ricardo Ferraz. 2023. "Improving the Antimycobacterial Drug Clofazimine through Formation of Organic Salts by Combination with Fluoroquinolones" International Journal of Molecular Sciences 24, no. 2: 1402. https://doi.org/10.3390/ijms24021402
APA StyleBento, C. M., Silva, A. T., Mansano, B., Aguiar, L., Teixeira, C., Gomes, M. S., Gomes, P., Silva, T., & Ferraz, R. (2023). Improving the Antimycobacterial Drug Clofazimine through Formation of Organic Salts by Combination with Fluoroquinolones. International Journal of Molecular Sciences, 24(2), 1402. https://doi.org/10.3390/ijms24021402








