Spectral Physics of Stable Cu(III) Produced by Oxidative Addition of an Alkyl Halide
Abstract
:1. Introduction
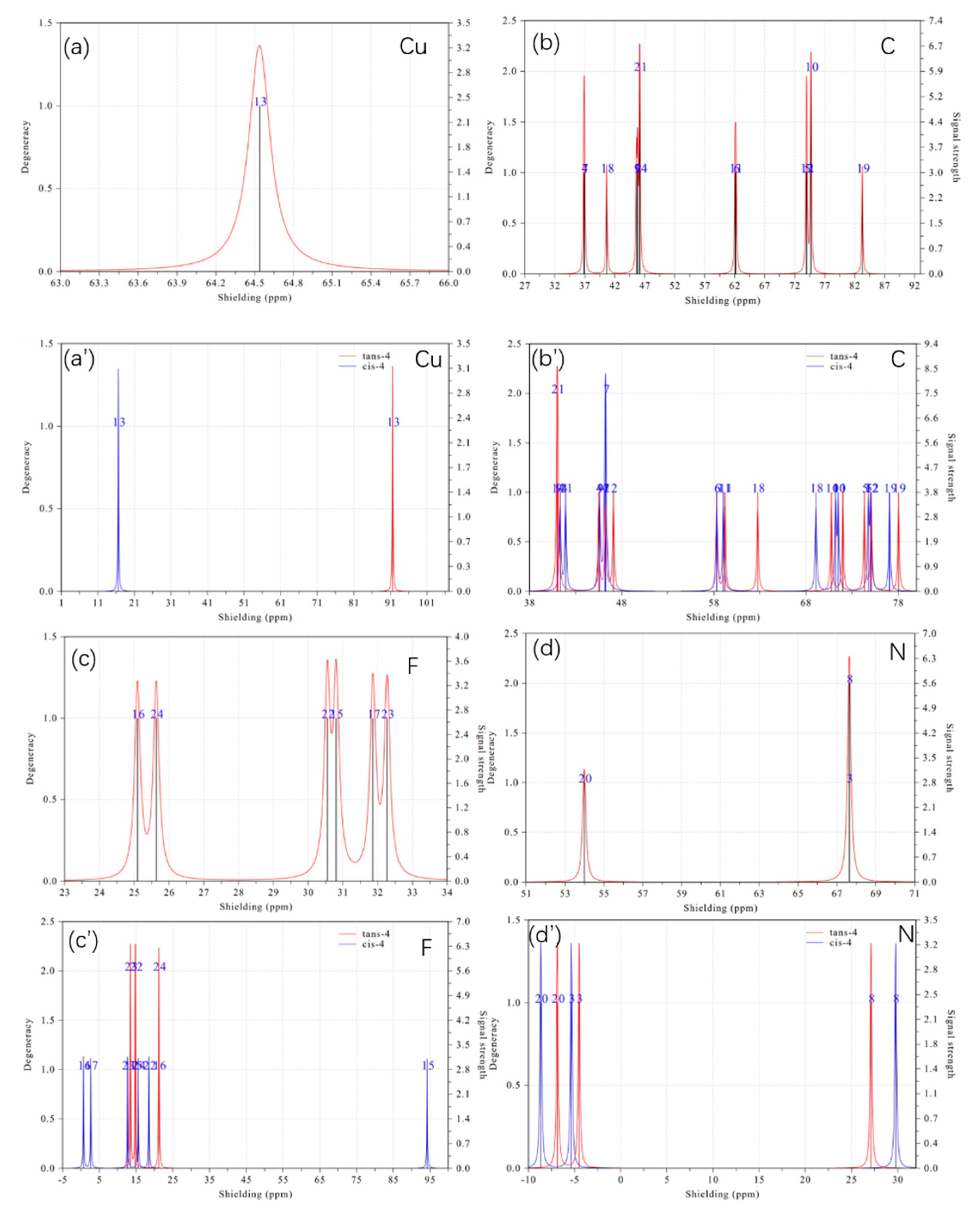
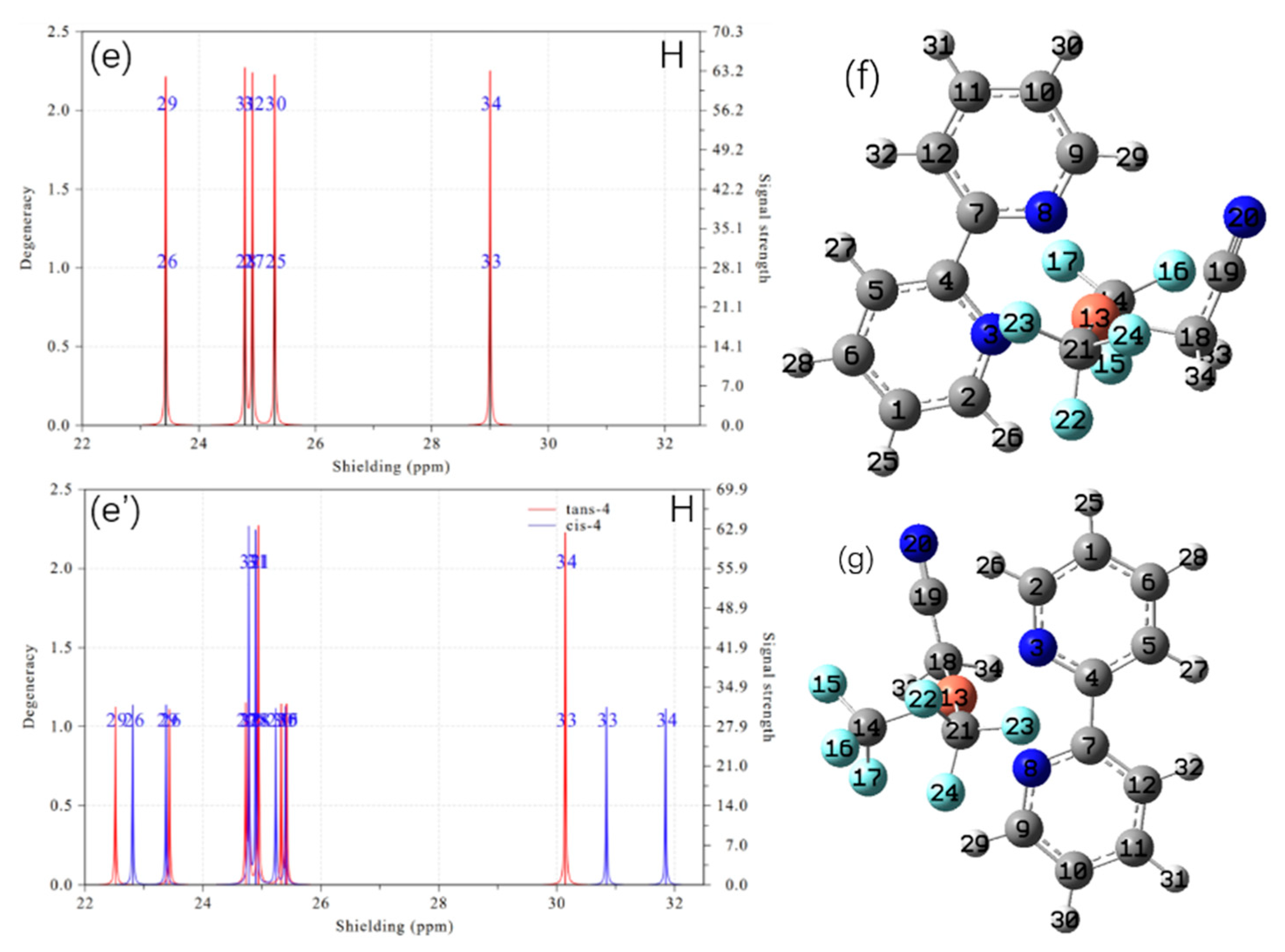
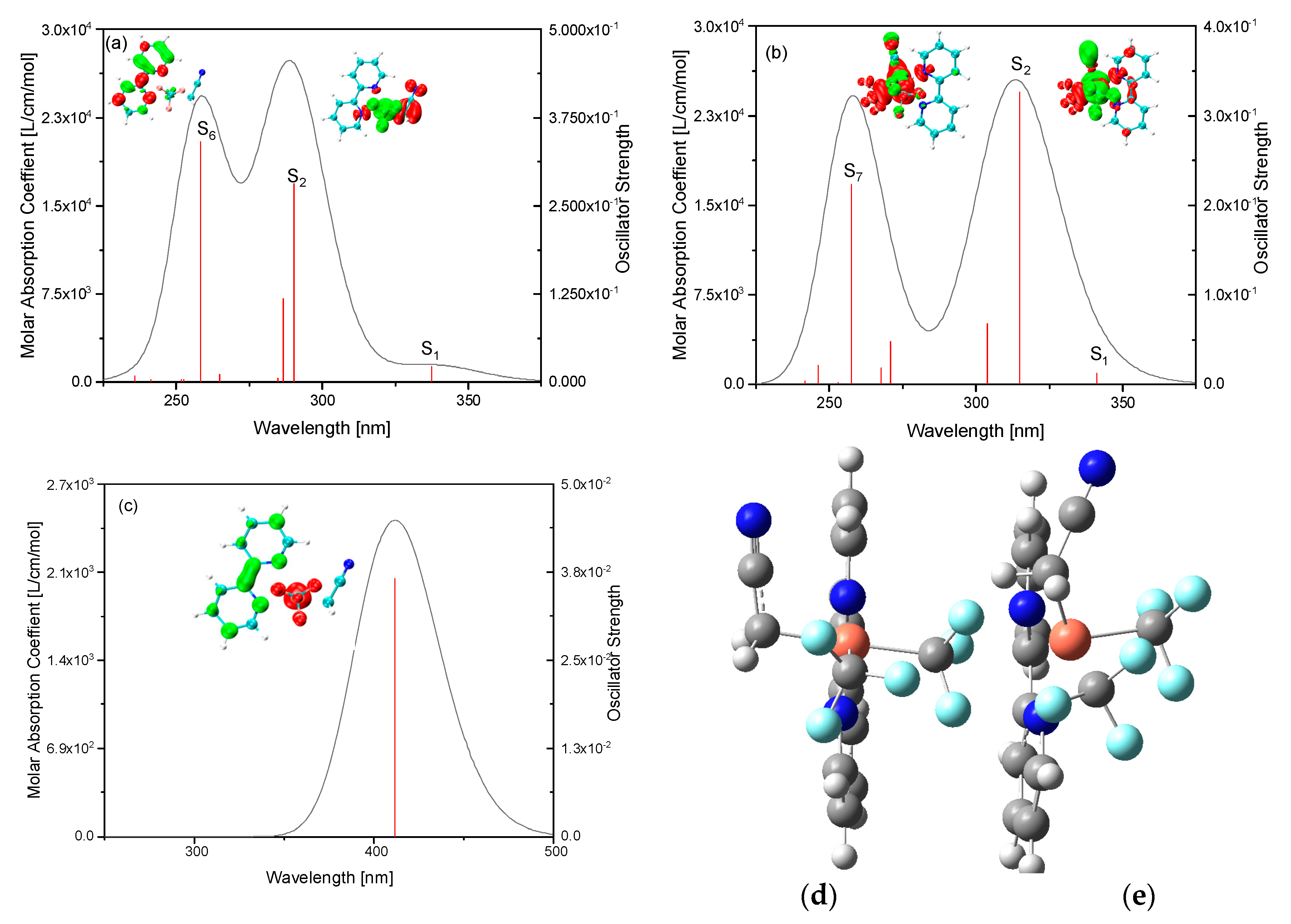
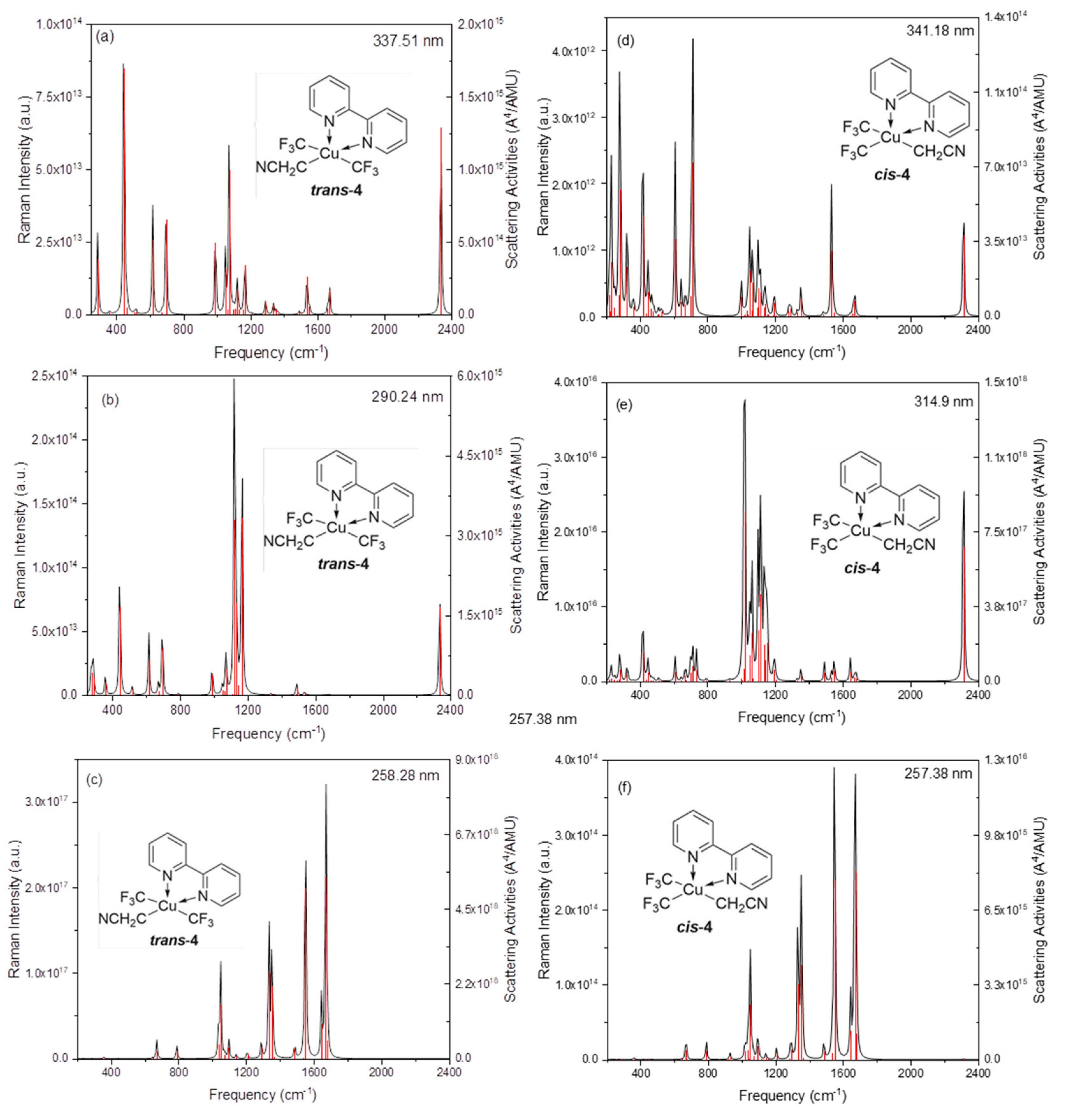
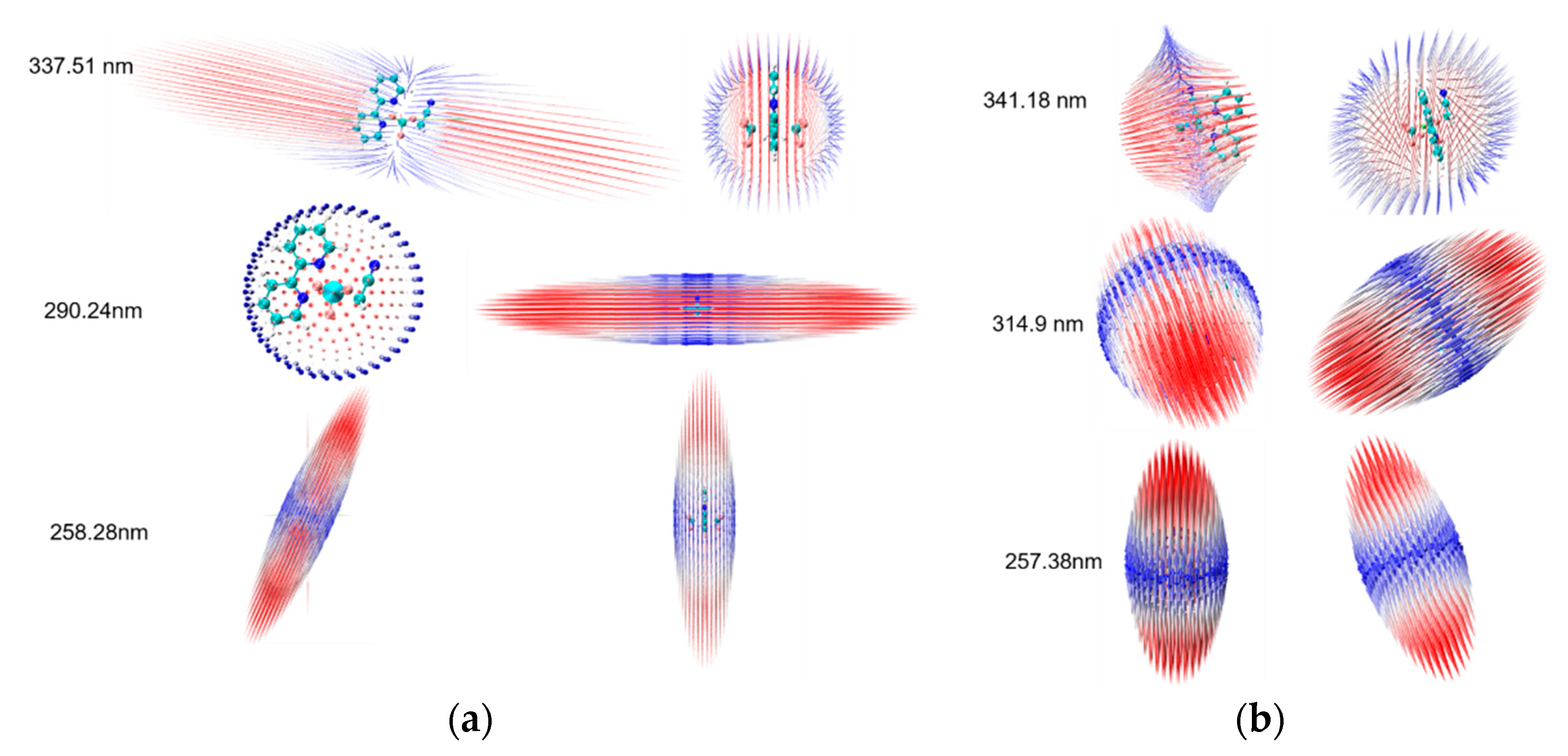
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Liu, H.; Shen, Q. Well-defined organometallic Copper(III) complexes: Preparation, characterization and reactivity. Coord. Chem. Rev. 2021, 439, 213923. [Google Scholar] [CrossRef]
- Hickman, A.J.; Sanford, M.S. High-valent organometallic copper and palladium in catalysis. Nature 2012, 484, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Hruszkewycz, D.; McCann, S.; Stahl, S.S. Cu-Catalyzed Aerobic Oxidation: Overview and New Developments; Wiley-VCH: Weinheim, Germany, 2016. [Google Scholar]
- Cho, S.H.; Kim, J.Y.; Kwak, J.; Chang, S. Recent advances in the transition metal-catalyzed twofold oxidative C-H bond activation strategy for C-C and C-N bond formation. Chem. Soc. Rev. 2011, 40, 5068–5083. [Google Scholar] [CrossRef]
- Que, L., Jr.; Tolman, W.B. Bis(μ-oxo)dimetal “Diamond” Cores in Copper and Iron Complexes Relevant to Biocatalysis. Angew. Chem. Int. Ed. 2002, 41, 1114–1137. [Google Scholar]
- Trammell, R.; Rajabimoghadam, K.; Garcia-Bosch, I. Copper-Promoted Functionalization of Organic Molecules: From Biologically Relevant Cu/O2 Model System to Organometallic Transformations. Chem. Rev. 2019, 119, 2954–3031. [Google Scholar] [CrossRef] [PubMed]
- Sperotto, E.; van Klink, G.P.M.; van Koten, G.; de Vries, J.G. The mechanism of the modified Ullmann reaction. Dalton Trans. 2010, 39, 10338–10351. [Google Scholar] [CrossRef] [PubMed]
- Casitas, A.; Ribas, X. The role of organometallic copper (III) complexes in homogeneous catalysis. Chem. Sci. 2013, 4, 2301–2318. [Google Scholar] [CrossRef]
- Li, S.-J.; Lan, Y. Is Cu(III) a necessary intermediate in Cu-mediated coupling reactions? A mechanistic point of view. Chem. Commun. 2020, 56, 6609–6619. [Google Scholar] [CrossRef]
- Cole, A.P.; Root, D.E.; Mukherjee, P.; Solomon, E.I.; Stack, T.D.P. A Trinuclear Intermediate in the Copper-Mediated Reduction of O2: Four Electrons from Three Copper. Science 1996, 273, 1848–1850. [Google Scholar] [CrossRef]
- Sinha, W.; Sommer, M.G.; Deibel, N.; Ehret, F.; Bauer, M.; Sarkar, B.; Kar, S. Experimental and Theoretical Investigations of the Existence of CuII, CuIII, and CuIV in Copper Corrolato Complexes. Angew. Chem. Int. Ed. 2015, 54, 13769–13774. [Google Scholar] [CrossRef]
- Srnec, M.; Navratil, R.; Andris, E.; Jasik, J.; Rouithova, J. Experimentally calibrated analysis of the electronic structure of CuO+: Implications for reactivity. Angew. Chem. Int. Ed. 2018, 52, 17053–17054. [Google Scholar] [CrossRef] [PubMed]
- Lemon, C.M.; Huynh, M.; Maher, A.G.; Anderson, B.L.; Bloch, E.D.; Powers, D.C.; Nocera, D.G. Electronic structure of copper corroles. Angew. Chem. Int. Ed. 2016, 55, 2176–2180. [Google Scholar] [CrossRef] [PubMed]
- Spaeth, A.D.; Gagnon, N.L.; Dhar, D.; Yee, G.M.; Tolman, W.B. Determination of the Cu(III)-OH Bond Distance by Resonance Raman Spectroscopy Using a Normalized Version of Badger’s Rule. J. Am. Chem. Soc. 2017, 139, 4477–4485. [Google Scholar] [CrossRef] [PubMed]
- Neisen, B.D.; Gagnon, N.L.; Dhar, D.; Spaeth, A.D.; Tolman, W.B. Formally Copper(III)-Alkylperoxo Complexes as Models of Possible Intermediates in Monooxygenase Enzymes. J. Am. Chem. Soc. 2017, 139, 10220–10223. [Google Scholar] [CrossRef] [PubMed]
- Dhar, D.; Yee, G.M.; Markle, T.F.; Mayer, J.M.; Tolman, W.B. Reactivity of the copper (III)-hydroxide unit with phenols. Chem. Sci. 2017, 8, 1075–1085. [Google Scholar] [CrossRef]
- Bailey, W.D.; Dhar, D.; Cramblitt, A.C.; Tolman, W.B. Mechanistic dichotomy in proton-coupled electron-transfer reactions of phenols with a copper superoxide complex. Am. Chem. Soc. 2019, 141, 5470–5480. [Google Scholar] [CrossRef] [PubMed]
- Mandal, M.; Elwell, C.E.; Bouchey, C.J.; Zerk, T.J.; Tolman, W.B.; Cramer, C. Mechanisms for hydrogen-atom abstraction by mononuclear copper (III) cores: Hydrogen-atom transfer or concerted proton-coupled electron transfer? J. Am. Chem. Soc. 2019, 141, 17236–17244. [Google Scholar] [CrossRef]
- Wu, T.; MacMillan, S.N.; Rajabimoghadam, K.; Siegler, M.A.; Lancaster, K.M.; Garcia-Bosch, I. Structure, Spectroscopy, and Reactivity of a Mononuclear Copper Hydroxide Complex in Three Molecular Oxidation States. J. Am. Chem. Soc. 2020, 142, 12265–12276. [Google Scholar] [CrossRef]
- Bower, J.K.; Cypcar, A.D.; Henriquez, B.; Stieber, S.C.E.; Zhang, S. C (sp3)–H fluorination with a copper (II)/(III) redox couple. J. Am. Chem. Soc. 2020, 142, 8514–8521. [Google Scholar] [CrossRef]
- Hu, H.; Snyder, J.P. Organocuprate conjugate addition: The square-planar “CuIII” intermediate. Am. Chem. Soc. 2007, 129, 7210–7211. [Google Scholar] [CrossRef]
- Bertz, S.H.; Cope, S.; Murphy, M.; Ogle, C.A.; Taylor, B.J. Rapid injection NMR in mechanistic organocopper chemistry. Preparation of the elusive copper (III) intermediate. J. Am. Chem. Soc. 2007, 129, 7208–7209. [Google Scholar] [CrossRef] [PubMed]
- Gärtner, T.; Henze, W.; Gschwind, R.M. NMR-detection of Cu(III) intermediates in substitution reactions of alkyl halides with Gilman cuprates. Am. Chem. Soc. 2007, 129, 11362–11363. [Google Scholar] [CrossRef] [PubMed]
- Bertz, S.H.; Cope, S.; Dorton, D.; Murphy, M.; Ogle, C.A. Organocuprate cross-coupling: The central role of the copper (III) intermediate and the importance of the copper (I) precursor. Angew. Chem. Int. Ed. 2007, 46, 7082–7085. [Google Scholar] [CrossRef]
- Bartholomew, E.R.; Bertz, S.H.; Cope, S.; Murphy, M.; Ogle, C.A. Preparation of σ-and π-Allylcopper (III) Intermediates in SN2 and SN2′ Reactions of Organocuprate (I) Reagents with Allylic Substrates. J. Am. Chem. Soc. 2008, 130, 11244–11245. [Google Scholar] [CrossRef] [PubMed]
- Bertz, S.H.; Hardin, R.A.; Murphy, M.D.; Ogle, C.A.; Richter, J.D.; Thomas, A.A. Rapid Injection NMR Reveals η3 ‘π-Allyl’CuIII Intermediates in Addition Reactions of Organocuprate Reagents. J. Am. Chem. Soc. 2012, 134, 9557–9560. [Google Scholar] [CrossRef] [PubMed]
- Maurya, Y.K.; Noda, K.; Yamasumi, K.; Mori, S.; Uchiyama, T.; Kamitani, K.; Hirai, T.; Ninomiya, K.; Nishibori, M.; Hori, Y.; et al. Ground-state copper (III) stabilized by N-confused/N-linked corroles: Synthesis, characterization, and redox reactivity. J. Am. Chem. Soc. 2018, 140, 6883–6892. [Google Scholar] [CrossRef]
- Adinarayana, B.; Thomas, A.P.; Suresh, C.H.; Srinivasan, A. A 6, 11, 16-Triarylbiphenylcorrole with an adj-CCNN Core: Stabilization of an Organocopper (III) Complex. Angew. Chem. Int. Ed. 2015, 54, 10478–10482. [Google Scholar] [CrossRef]
- Ke, X.-S.; Hong, Y.; Tu, P.; He, Q.; Lynch, V.M.; Kim, D.; Sessler, J.L. Hetero Cu(III)–Pd (II) complex of a dibenzo [g, p] chrysene-fused bis-dicarbacorrole with stable organic radical character. J. Am. Chem. Soc. 2017, 139, 15232–15238. [Google Scholar] [CrossRef]
- Ribas, X.; Jackson, D.A.; Donnadieu, B.; Mahía, J.; Parella, T.; Xifra, R.; Hedman, B.; Hodgson, K.O.; Llobet, A.; Stack, T.D.P. Aryl C-H Activation by CuII To Form an Organometallic Aryl–CuIII Species: A Novel Twist on Copper Disproportionation. Angew. Chem. Int. Ed. 2002, 41, 2991–2994. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, Y.; Wang, T.; Zhang, X.; Long, C.; Wu, Y.-D.; Wang, M.-X. Mechanistic study on cu (II)-catalyzed oxidative cross-coupling reaction between arenes and boronic acids under aerobic conditions. J. Am. Chem. Soc. 2018, 140, 5579–5587. [Google Scholar] [CrossRef]
- Liu, Y.; Resch, S.G.; Klawitter, I.; Cutsai, G.E., III; Demeshko, S.; Dechert, S.; Kühn, F.E.; DeBeer, S.; Meyer, F. An Adaptable N-Heterocyclic Carbene Macrocycle Hosting Copper in Three Oxidation States. Angew. Chem. Int. Ed. 2020, 59, 5696–5705. [Google Scholar] [CrossRef]
- Brothers, P.J.; Roper, W.R. Transition-metal dihalocarbene complexes. Chem. Rev. 1988, 88, 1293–1326. [Google Scholar] [CrossRef]
- Romine, A.M.; Nebra, N.; Konovalov, A.I.; Martin, E.; Benet-Buchholz, J.; Grushin, V.V. Easy access to the copper (III) anion [Cu (CF3) 4]−; Angew. Chem. Int. Ed. 2015, 54, 2745–2749. [Google Scholar] [CrossRef]
- Shen, H.; Liu, Z.; Zhang, P.; Tan, X.; Zhang, Z.; Li, C. Trifluoromethylation of alkyl radicals in aqueous solution. J. Am. Chem. Soc. 2017, 139, 9843–9846. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Liu, Z.; Shen, H.; Zhang, P.; Zhang, Z.; Li, C. Silver-catalyzed decarboxylative trifluoromethylation of aliphatic carboxylic acids. J. Am. Chem. Soc. 2017, 139, 12430–12433. [Google Scholar] [CrossRef]
- Guo, S.; AbuSalim, D.I.; Cook, S.P. Aqueous benzylic C–H trifluoromethylation for late-stage functionalization. J. Am. Chem. Soc. 2018, 140, 12378–12382. [Google Scholar] [CrossRef] [PubMed]
- Choi, G.; Lee, G.S.; Park, B.; Kim, D.; Hong, S.H. Direct C (sp3)−H trifluoromethylation of unactivated alkanes enabled by multifunctional trifluoromethyl copper complexes. Angew. Chem. Int. Ed. 2021, 60, 5467–5474. [Google Scholar] [CrossRef]
- Guo, S.; AbuSalim, D.I.; Cook, S.P. 1, 2-(Bis) trifluoromethylation of Alkynes: A One-Step Reaction to Install an Underutilized Functional Group. Angew. Chem. Int. Ed. 2019, 58, 11704–11708. [Google Scholar] [CrossRef]
- Le, C.; Chen, T.Q.; Liang, T.; Zhang, P.; MacMillan, D.W.C. A radical approach to the copper oxidative addition problem: Trifluoromethylation of bromoarenes. Science 2018, 360, 1010–1014. [Google Scholar] [CrossRef]
- Kornfilt, D.J.P.; MacMillan, D.W.C. Copper-catalyzed trifluoromethylation of alkyl bromides. J. Am. Chem. Soc. 2019, 141, 6853–6858. [Google Scholar] [CrossRef]
- Lu, Z.; Liu, H.; Liu, S.; Leng, X.; Lan, Y.; Shen, Q. A Key Intermediate in Copper-Mediated Arene Trifluoromethylation,[nBu4N][Cu (Ar)(CF3) 3]: Synthesis, Characterization, and C (sp2)−CF3 Reductive Elimination. Angew. Chem. Int. Ed. 2019, 58, 8510–8514. [Google Scholar] [CrossRef] [PubMed]
- Paeth, M.; Tyndall, S.B.; Chen, L.-Y.; Hong, J.-C.; Carson, W.P.; Liu, X.; Sun, X.; Liu, J.; Yang, K.; Hale, E.M.; et al. Csp3–Csp3 bond-forming reductive elimination from well-defined copper (III) complexes. J. Am. Chem. Soc. 2019, 141, 3153–3159. [Google Scholar] [CrossRef]
- Liu, S.; Liu, H.; Liu, S.; Lu, Z.; Lu, C.; Leng, X.; Lan, Y.; Shen, Q. C (sp3)-CF3 Reductive elimination from a five-coordinate neutral copper (III) complex. J. Am. Chem. Soc. 2020, 142, 9785–9791. [Google Scholar] [CrossRef]
- Wing, R.M. Carborane analogs of. pi.-allyls. The crystal and molecular structure of triphenylmethylphosphonium bis (3)-1, 2-dicarboryl) cuprate (III). J. Am. Chem. Soc. 1968, 90, 4828–4834. [Google Scholar] [CrossRef]
- Varadarajan, A.; Johnson, S.E.; Gomez, F.A.; Chakrabarti, S.; Knobler, C.B.; Hawthorne, M.F. Synthesis and structural characterization of pyrazole-bridged metalla-bis (dicarbollide) derivatives of cobalt, nickel, copper, and iron: Models for venus flytrap cluster reagents. J. Am. Chem. Soc 1992, 114, 9003–9011. [Google Scholar] [CrossRef]
- García-López, J.; Yañez-Rodríguez, V.; Roces, L.; García-Granda, S.; Martínez, A.; Guevara-García, A.; Castro, G.R.; Jiménez-Villacorta, F.; Iglesias, M.J.; Ortiz, F.L. Synthesis and characterization of a coupled binuclear CuI/CuIII complex. J. Am. Chem. Soc. 2010, 132, 10665–10667. [Google Scholar] [CrossRef]
- Liu, L.; Zhu, M.; Yu, H.-T.; Zhang, W.-X.; Xi, Z. Organocopper (III) spiro complexes: Synthesis, structural characterization, and redox transformation. J. Am. Chem. Soc. 2017, 139, 13688–13691. [Google Scholar] [CrossRef] [PubMed]
- Santo, R.; Miyamoto, R.; Tanaka, R.; Nishioka, T.; Sato, K.; Toyota, K.; Obata, M.; Yano, S.; Kinoshita, I.; Ichimura, A.; et al. Diamagnetic–Paramagnetic Conversion of Tris (2-pyridylthio) methylcopper (III) through a Structural Change from Trigonal Bipyramidal to Octahedral. Angew. Chem. Int. Ed. 2006, 45, 7611–7614. [Google Scholar] [CrossRef]
- Snyder, J.P. Elusiveness of CuIII complexation. preference for trifluoromethyl oxidation in the formation of [CuI (CF3)4]−salts. Angew. Chem. Int. Ed. Engl. 1995, 34, 80–81. [Google Scholar] [CrossRef]
- Walroth, R.C.; Lukens, J.T.; MacMillan, S.N.; Finkelstein, K.D.; Lancaster, K.M. Spectroscopic Evidence for a 3d10 Ground State Electronic Configuration and Ligand Field Inversion in [Cu (CF3)4] 1–. J. Am. Chem. Soc. 2016, 138, 1922–1931. [Google Scholar] [CrossRef]
- Dimucci, I.M.; Lukens, J.T.; Chatterjee, S.; Carsch, K.M.; Titus, C.J.; Lee, S.J.; Nordlund, D.; Betley, T.A.; MacMillan, S.N.; Lancaster, K.M. The myth of d8 copper (III). J. Am. Chem. Soc. 2019, 141, 18508–18520. [Google Scholar] [CrossRef]
- Hoffmann, R.; Alvarez, S.; Mealli, C.; Falceto, A.; Cahill, T.J., III; Zeng, T.; Manca, G. From widely accepted concepts in coordination chemistry to inverted ligand fields. Chem. Rev. 2016, 116, 8173–8192. [Google Scholar] [CrossRef] [PubMed]
- van Koten, G. Organocopper Compounds: From Elusive to Isolable Species, from Early Supramolecular Chemistry with RCuI Building Blocks to Mononuclear R2–nCuII and R3–mCuIII Compounds. A Personal View. Organometallics 2012, 31, 7634–7764. [Google Scholar] [CrossRef]
- Melník, M.; Kabešová, M. Copper (III) coordination compounds: Classification and analysis of crystallographic and structural data. Coord. Chem. 2000, 50, 323–338. [Google Scholar] [CrossRef]
- Bartholomew, E.R.; Bertz, S.H.; Cope, S.; Dorton, D.C.; Murphy, M.; Ogle, C.A. Neutral organocopper (III) complexes. Chem. Commun. 2008, 10, 1176–1177. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Li, Y.; Wu, J.; Xue, X.; Hartwig, J.F.; Shen, Q. Oxidative addition of an alkyl halide to form a stable Cu(III) product. Science 2023, 381, 1072–1079. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.P.; Petersson, G.A.; Nakatsuji, H.R.; et al. Gaussian 16, Revision A. 03; Gaussian. Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Kohn, W.; Sham, L.J. Self-consistent equations including exchange and correlation effects. Phys. Rev. 1965, 140, A1133–A1138. [Google Scholar] [CrossRef]
- Becke, A.D. Density-functional exchange-energy approximation with correct asymptotic behavior. Phys. Rev. A 1988, 38, 3098–3100. [Google Scholar] [CrossRef] [PubMed]
- García, J.C.; Johnson, E.R.; Keinan, S.; Chaudret, R.; Piquemal, J.; Beratan, D.N.; Yang, W.J. NCIPLOT: A program for plotting noncovalent interaction regions. Chem. Theory Comput. 2011, 7, 625–632. [Google Scholar] [CrossRef]
- Gross, E.K.U.; Kohn, W. Local density-functional theory of frequency-dependent linear response. Phys. Rev. Lett. 1985, 55, 2850–2852. [Google Scholar] [CrossRef] [PubMed]
- Yanai, T.; Tew, D.P.; Handy, N.C. A new hybrid exchange–correlation functional using the Coulomb-attenuating method (CAM-B3LYP). Chem. Phys. Lett. 2004, 393, 51–57. [Google Scholar] [CrossRef]
- Lu, T.; Chen, F.W. Multiwfn: A multifunctional wavefunction analyzer. J. Comput. Chem. 2012, 33, 580–592. [Google Scholar] [CrossRef] [PubMed]
- Tuer, A.; Krouglov, S.; Cisek, R.; Tokarz, D.; Barzda, V.J. Three-dimensional visualization of the first hyperpolarizability tensor. Comput. Chem. 2011, 32, 1128–1134. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Lu, T.; Yuan, A.; Wang, X.; Chen, Q.; Yan, X. Remarkable Size Effect on Photophysical and Nonlinear Optical Properties of All-Carboatomic Rings, Cyclo [18] carbon and Its Analogues. Chem. Asian J. 2021, 16, 2267–2271. [Google Scholar] [CrossRef]
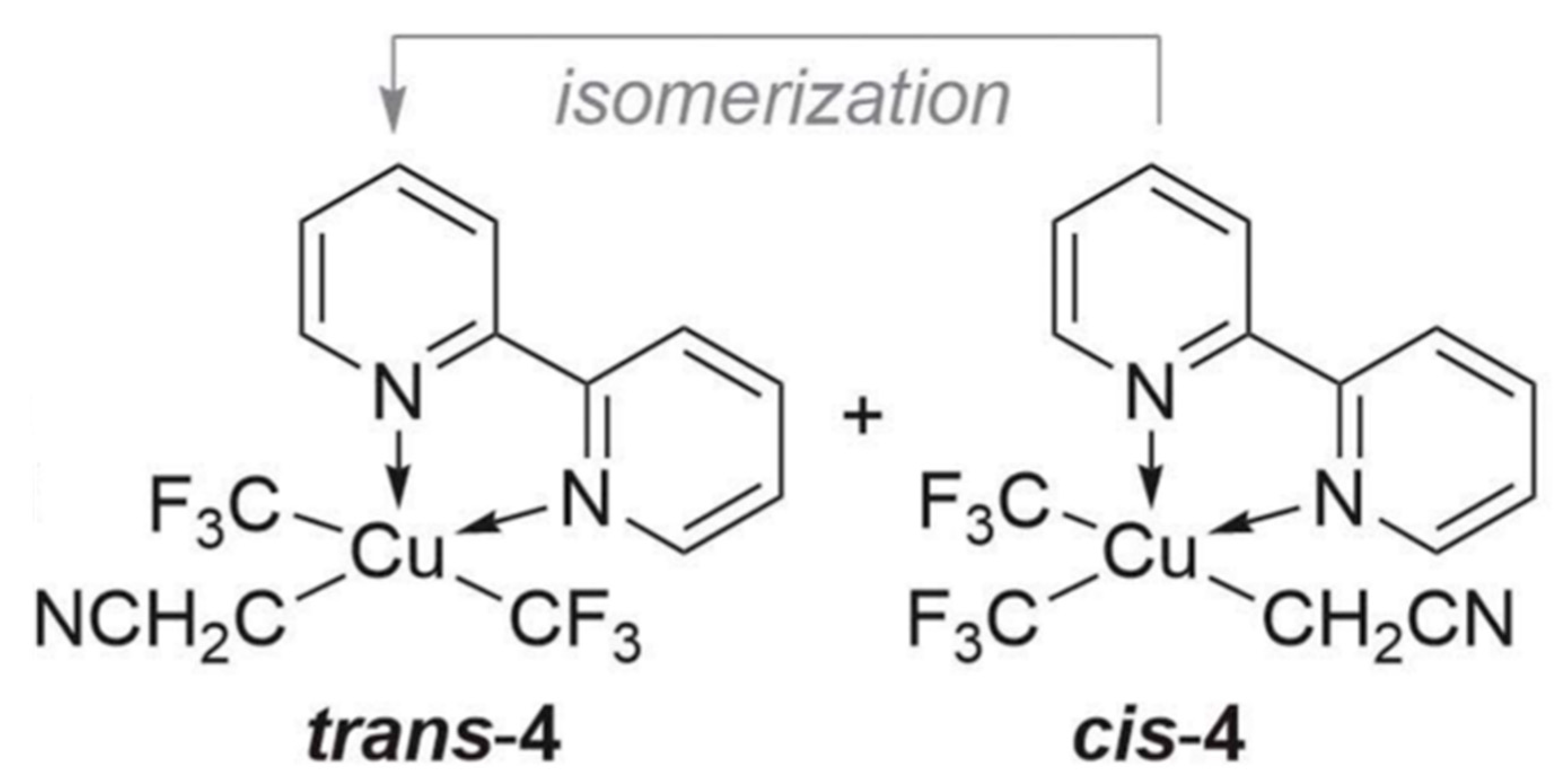

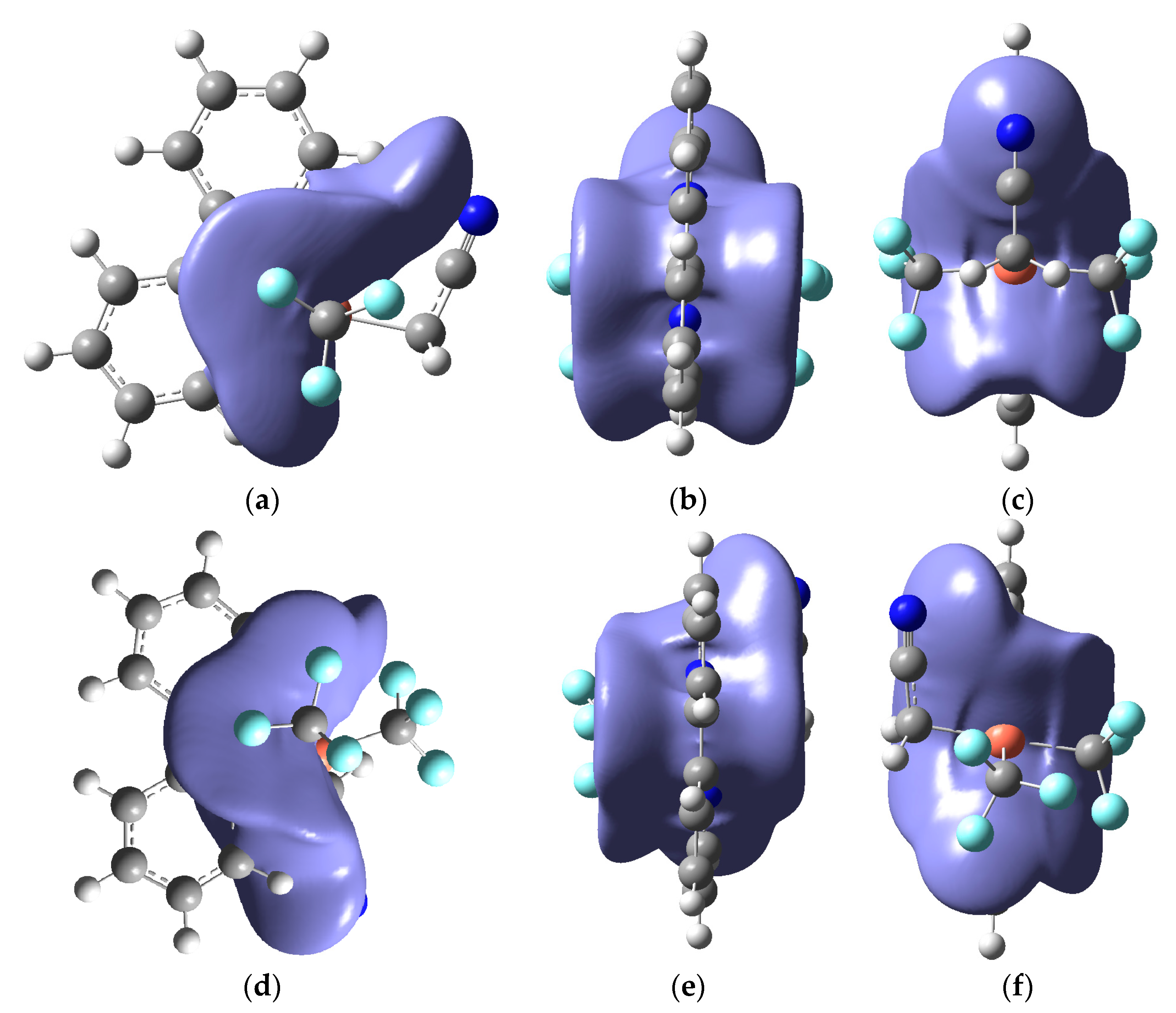
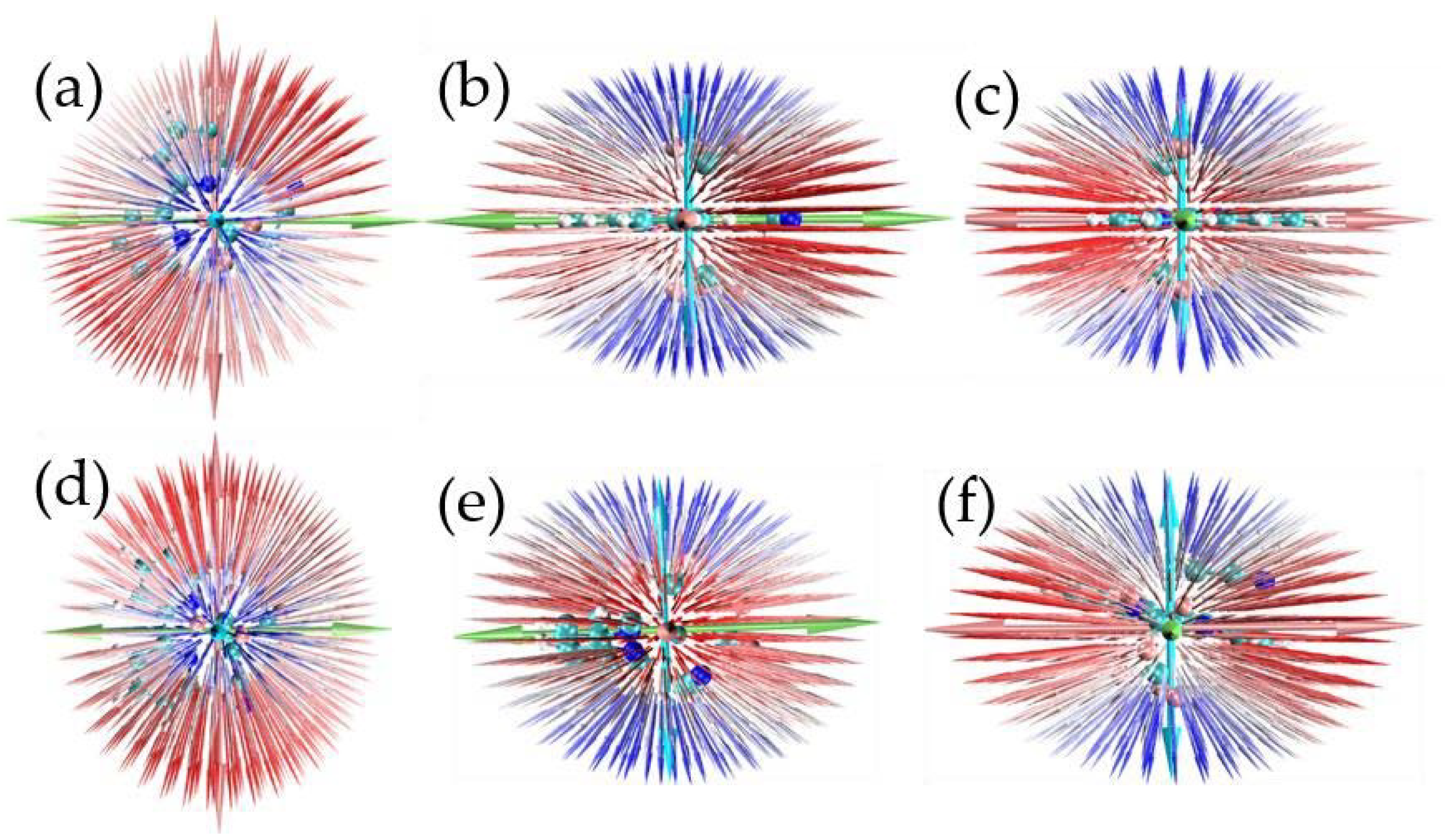
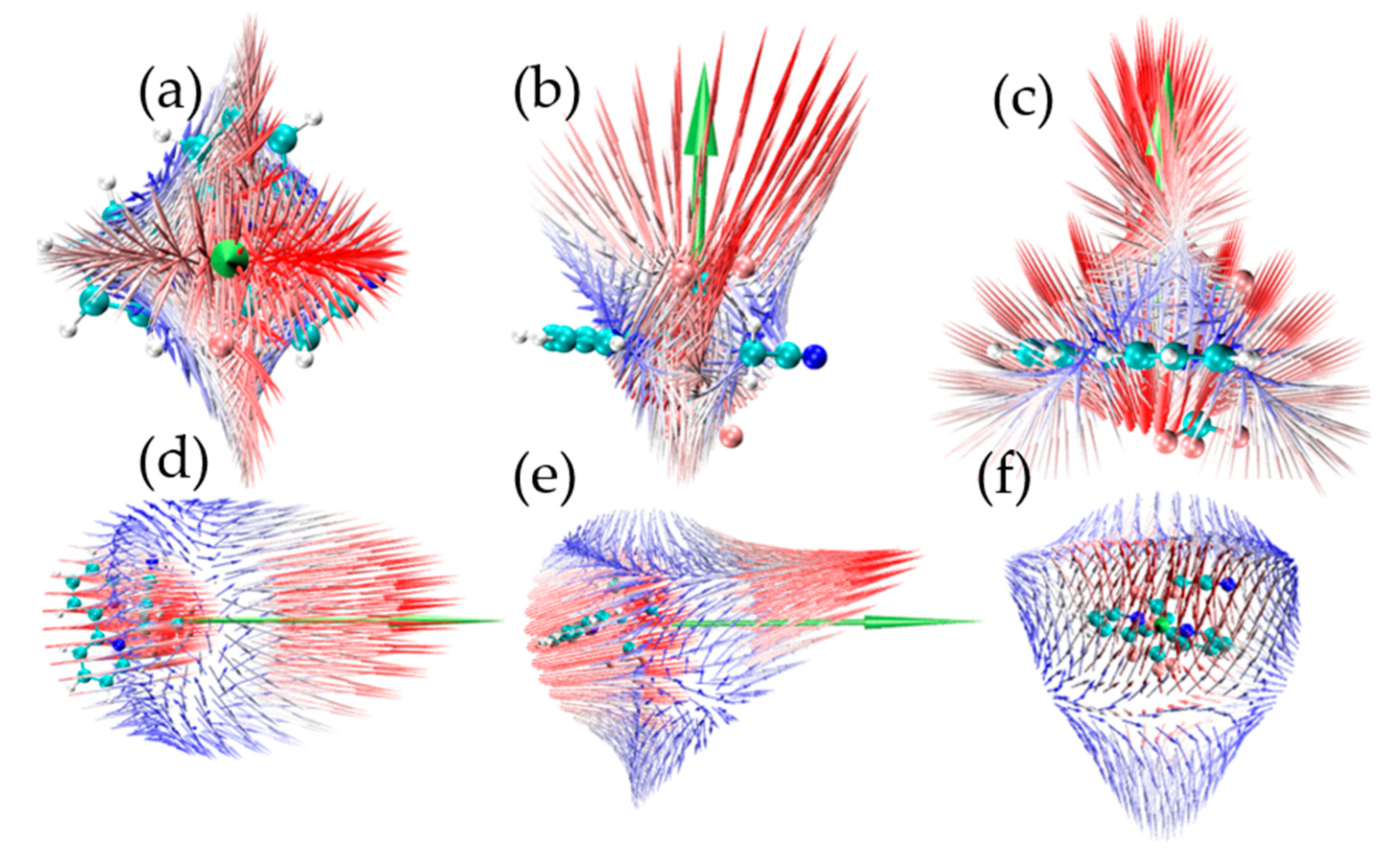

| (x) | (y) | (z) | ||||
|---|---|---|---|---|---|---|
| trans-4 | −9.0 | 0.7 | 0.0 | 237 | 231 | 128 |
| cis-4 | 10.2 | −2.7 | −1.4 | 215 | 253 | 139 |
| trans-4 | 340 | −26 | 0 | 102 | −215 | 133 | 0 | −48 | 0 |
| cis-4 | −610 | −15 | 51 | −67 | 110 | −41 | −70 | 9 | 184 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, E.; Sun, M. Spectral Physics of Stable Cu(III) Produced by Oxidative Addition of an Alkyl Halide. Int. J. Mol. Sci. 2023, 24, 15694. https://doi.org/10.3390/ijms242115694
Cao E, Sun M. Spectral Physics of Stable Cu(III) Produced by Oxidative Addition of an Alkyl Halide. International Journal of Molecular Sciences. 2023; 24(21):15694. https://doi.org/10.3390/ijms242115694
Chicago/Turabian StyleCao, En, and Mengtao Sun. 2023. "Spectral Physics of Stable Cu(III) Produced by Oxidative Addition of an Alkyl Halide" International Journal of Molecular Sciences 24, no. 21: 15694. https://doi.org/10.3390/ijms242115694






