Tandem Expression of a Mobile RNA and Its RNA-Binding Protein(s) Enhances Tuber Productivity in Potato
Abstract
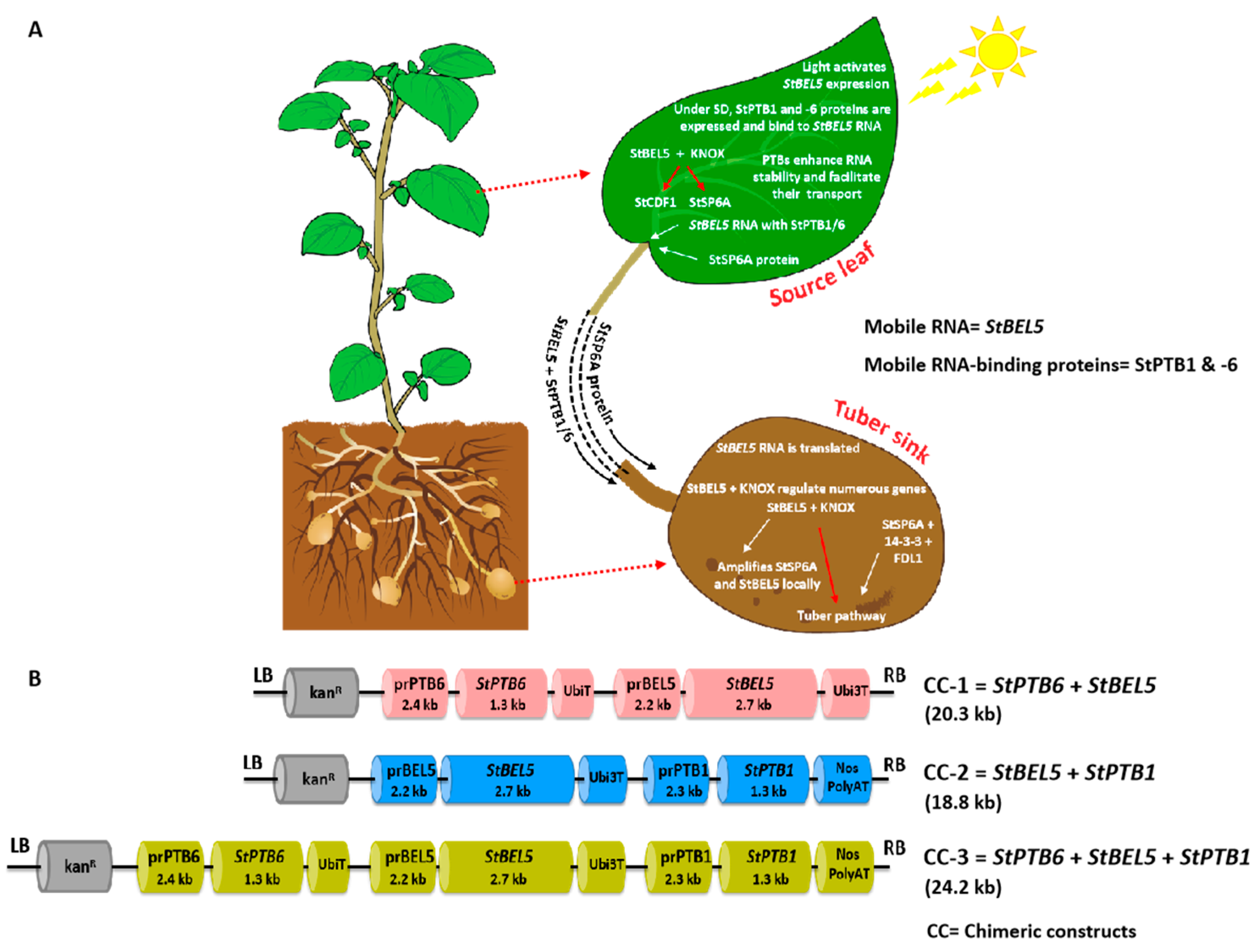
:1. Introduction
2. Results
2.1. Assessment of Earliness for Tuberization under In Vitro and Greenhouse Conditions
2.2. Expression Analysis of Tuber Marker Genes and Evaluation of Tuber Productivity under Controlled Growth Conditions
2.3. Evaluation of Tuber Productivity under Field Conditions
3. Discussion
4. Materials and Methods
4.1. Plantlet Source and Culture
4.2. Chimeric Constructs Preparation
4.3. Generation of Stable Transgenic Lines
4.4. In Vitro Tuber Induction Assay
4.5. Tuber Productivity and Quantification of StBEL5 Transcript Levels
4.6. Field Experiments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lifschitz, E.; Eviatar, T.; Rozman, A.; Shalit, A.; Goldshmidt, A.; Amsellem, Z.; Alvarez, J.P.; Eshed, Y. The tomato FT ortholog triggers systemic signals that regulate growth and flowering and substitute for diverse environmental stimuli. Proc. Natl. Acad. Sci. USA 2006, 103, 6398. [Google Scholar] [CrossRef]
- Lin, M.K.; Belanger, H.; Lee, Y.J.; Varkonyi-Gasic, E.; Taoka, K.; Miura, E.; Xoconostle-Cázares, B.; Gendler, K.; Jorgensen, R.A.; Phinney, B.; et al. FLOWERING LOCUS T protein may act as the long-distance florigenic signal in the cucurbits. Plant Cell 2007, 19, 1488–1506. [Google Scholar] [CrossRef]
- Tamaki, S.; Matsuo, S.; Wong, H.L.; Yokoi, S.; Shimamoto, K. Hd3a protein is a mobile flowering signal in rice. Science 2007, 316, 1033–1036. [Google Scholar] [CrossRef]
- Kehr, J.; Buhtz, A. Long distance transport and movement of RNA through the phloem. J. Exp. Bot. 2008, 59, 85–92. [Google Scholar] [CrossRef]
- Kehr, J.; Kragler, F. Long distance RNA movement. New Phytol. 2018, 218, 29–40. [Google Scholar] [CrossRef]
- Tsikou, D.; Yan, Z.; Holt, D.B.; Abel, N.B.; Reid, D.E.; Madsen, L.H.; Bhasin, H.; Sexauer, M.; Stougaard, J.; Markmann, K. Systemic control of legume susceptibility to rhizobial infection by a mobile microRNA. Science 2018, 362, 233–236. [Google Scholar] [CrossRef]
- Wang, S.; Wang, S.; Zhang, W.; Zhang, Q.; Hao, L.; Zhang, Y.; Xu, C.; Yu, Y.; Wang, B.; Li, T.; et al. PbTTG1 forms a ribonucleoprotein complex with polypyrimidine tract binding protein PbPTB3 to facilitate the long-distance trafficking of PbWoxT1 mRNA. Plant Sci. 2019, 280, 424–432. [Google Scholar] [CrossRef]
- Kehr, J.; Morris, R.J.; Kragler, F. Long-distance transported RNAs: From identity to function. Annu. Rev. Plant Biol. 2022, 73, 457–474. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, K.; Song, W.; Zhong, N.; Wu, Y.; Fu, X. Improving crop nitrogen use efficiency toward sustainable green revolution. Annu. Rev. Plant Biol. 2022, 73, 523–551. [Google Scholar] [CrossRef]
- Yan, Y.; Ham, B.K. The mobile small RNAs: Important messengers for long-distance communication in plants. Front. Plant Sci. 2022, 13, 928729. [Google Scholar] [CrossRef]
- Wang, S.; Wu, K.; Yuan, Q.; Liu, X.; Liu, Z.; Lin, X.; Zeng, R.; Zhu, H.; Dong, G.; Qian, Q.; et al. Control of grain size, shape and quality by OsSPL16 in rice. Nat. Genet. 2012, 44, 950–954. [Google Scholar] [CrossRef] [PubMed]
- Debernardi, J.M.; Lin, H.; Chuck, G.; Faris, J.D.; Dubcovsky, J. microRNA172 plays a crucial role in wheat spike morphogenesis and grain threshability. Development 2017, 144, 1966–1975. [Google Scholar]
- Tian, L.; Chou, H.L.; Zhang, L.; Hwang, S.K.; Starkenburg, S.R.; Doroshenk, K.A.; Kumamaru, T.; Okita, T.W. RNA-Binding Protein RBP-P is required for glutelin and prolamine mRNA localization in rice endosperm cells. Plant Cell 2018, 30, 2529–2552. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Yao, Q.; Gao, X.; Jiang, C.; Harberd, N.P.; Fu, X. Shoot-to-root mobile transcription factor HY5 coordinates plant carbon and nitrogen acquisition. Curr. Biol. 2016, 26, 640–646. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Luo, Z.; Hu, Q.; Tang, W.; Lu, H.; Ma, C.; Wang, Z.; Baltaevich, A.M.; Kong, X. Light induced shoot-sourced transcription factor HY5 regulates the nitrate uptake of cotton by shoot-to-root signal transport. Plant Physiol. Biochem. 2023, 200, 107738. [Google Scholar] [CrossRef]
- Huang, N.C.; Luo, K.R.; Yu, T.S. Mobility of antiflorigen and PEBP mRNAs in tomato-tobacco heterografts. Plant Physiol. 2018, 178, 783–794. [Google Scholar] [CrossRef]
- Wu, Y.M.; Ma, Y.J.; Wang, M.; Zhou, H.; Gan, Z.M.; Zeng, R.F.; Ye, L.X.; Zhou, J.J.; Zhang, J.Z.; Hu, C.G. Mobility of FLOWERING LOCUS T protein as a systemic signal in trifoliate orange and its low accumulation in grafted juvenile scions. Hortic. Res. 2022, 9, uhac056. [Google Scholar] [CrossRef]
- Yu, Z.; Chen, W.; Wang, Y.; Zhang, P.; Shi, N.; Hong, Y. Mobile Flowering Locus T RNA—Biological Relevance and Biotechnological Potential. Front. Plant Sci. 2022, 12, 792192. [Google Scholar] [CrossRef]
- Ham, B.K.; Brandom, J.L.; Xoconostle-Cazares, B.; Ringgold, V.; Lough, T.J.; Lucas, W.J. A polypyrimidine tract binding protein, pumpkin RBP50, forms the basis of a phloem-mobile ribonucleoprotein complex. Plant Cell 2009, 21, 197–215. [Google Scholar] [CrossRef]
- Lin, M.K.; Lee, Y.J.; Lough, T.J.; Phinney, B.S.; Lucas, W.J. Analysis of the pumpkin phloem proteome provides insights into angiosperm sieve tube function. Mol. Cell Proteom. 2009, 8, 343–356. [Google Scholar] [CrossRef]
- Liu, L.; Chen, X. Intercellular and systemic trafficking of RNAs in plants. Nat. Plants 2014, 11, 869–878. [Google Scholar] [CrossRef] [PubMed]
- Notaguchi, M.J. Identification of phloem-mobile mRNA. Plant Res. 2015, 128, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Ham, B.K.; Lucas, W.J. Phloem-mobile RNAs as systemic signaling agents. Annu. Rev. Plant Biol. 2017, 68, 173–195. [Google Scholar] [CrossRef]
- Li, P.; Ham, B.K.; Lucas, W.J. CmRBP50 protein phosphorylation is essential for assembly of a stable phloem-mobile high-affinity ribonucleoprotein complex. J. Biol. Chem. 2022, 286, 23142–23149. [Google Scholar] [CrossRef]
- Ruiz-Medrano, R.; Xoconostle-Cazares, B.; Lucas, W.J. Phloem long-distance transport of CmNACP mRNA: Implications for supracellular regulation in plants. Development 1999, 126, 4405–4419. [Google Scholar] [CrossRef]
- Yang, H.W.; Yu, T.S. Arabidopsis floral regulators FVE and AGL24 are phloem-mobile RNAs. Bot. Stud. 2010, 51, 17–26. [Google Scholar]
- Zhang, W.N.; Duan, X.W.; Ma, C.; Harada, T.; Li, T.Z. Transport of mRNA molecules coding NAC domain protein in grafted pear and transgenic tobacco. Biol. Plant. 2013, 57, 224–230. [Google Scholar] [CrossRef]
- Thieme, C.J.; Rojas-Triana, M.; Stecyk, E.; Schudoma, C.; Zhang, W.; Yang, L.; Miñambres, M.; Walther, D.; Schulze, W.X.; Paz-Ares, J.; et al. Endogenous Arabidopsis messenger RNAs transported to distant tissues. Nat. Plants 2015, 1, 15025. [Google Scholar] [CrossRef]
- Toscano-Morales, R.; Xoconostle-Cázares, B.; Martínez-Navarro, A.C.; Ruiz-Medrano, R. AtTCTP2 mRNA and protein movement correlates with formation of adventitious roots in tobacco. Plant Signal. Behav. 2016, 11, e1071003. [Google Scholar] [CrossRef]
- Yang, L.; Perrera, V.; Saplaoura, E.; Apelt, F.; Bahin, M.; Kramdi, A.; Olas, J.; Mueller-Roeber, B.; Sokolowska, E.; Zhang, W.; et al. m5C methylation guides systemic transport of messenger RNA over graft junctions in plants. Curr. Biol. 2019, 29, 2465–2476. [Google Scholar] [CrossRef]
- Hao, L.; Zhang, Y.; Wang, S.; Zhang, W.; Wang, S.; Xu, C.; Yu, Y.; Li, T. A constitutive and drought-responsive mRNA undergoes long-distance transport in pear (Pyrus betulaefolia) phloem. Plant Sci. 2020, 293, 110419. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.K.; Chatterjee, M.; Yu, Y.; Suh, S.G.; Miller, W.A.; Hannapel, D.J. Dynamics of a mobile RNA of potato involved in a long-distance signaling pathway. Plant Cell 2006, 18, 3443–3457. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; Adam, H.; Diaz-Mendoza, M.; Zurczak, M.; Gonzalez-Schain, N.D.; Suárez-López, P. Graft-transmissible induction of potato tuberization by the microRNA miR172. Development 2009, 136, 2873–2881. [Google Scholar] [CrossRef] [PubMed]
- Navarro, C.; Abelenda, J.A.; Cruz-Oró, E.; Cuéllar, C.A.; Tamaki, S.; Silva, J.; Shimamoto, K.; Prat, S. Control of flowering and storage organ formation in potato by FLOWERING LOCUS T. Nature 2011, 478, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, A.; Bhogale, S.; Kang, I.H.; Hannapel, D.J.; Banerjee, A.K. The mRNA of a Knotted1like transcription factor of potato is phloem mobile. Plant Mol. Biol. 2012, 79, 595–608. [Google Scholar] [CrossRef]
- Sharma, P.; Lin, T.; Hannapel, D.J. Targets of the StBEL5 transcription factor include the FT ortholog StSP6A. Plant Physiol. 2016, 170, 310–324. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, D. The signal transduction pathways controlling in planta tuberization in potato: An emerging synthesis. Plant Cell Rep. 2008, 27, 1–8. [Google Scholar] [CrossRef]
- Sarkar, D. Photoperiodic inhibition of potato tuberization: An update. Plant Growth Regul. 2010, 62, 117–125. [Google Scholar] [CrossRef]
- Hannapel, D.J.; Sharma, P.; Lin, T.; Banerjee, A.K. The multiple signals that control tuber formation. Plant Physiol. 2017, 174, 845–856. [Google Scholar] [CrossRef]
- Cho, S.K.; Sharma, P.; Butler, N.M.; Kang, I.H.; Shah, S.; Rao, A.G.; Hannapel, D.J. Polypyrimidine tract-binding proteins of potato mediate tuberization through an interaction with StBEL5 RNA. J. Exp. Bot. 2015, 66, 6835–6847. [Google Scholar] [CrossRef]
- Kloosterman, B.; Navarro, C.; Bijsterbosch, G.; Lange, T.; Prat, S.; Visser, R.G.F.; Bachem, C.W.B. StGA2ox1 is induced prior to stolon swelling and controls GA levels during potato tuber development. Plant J. 2007, 52, 362–373. [Google Scholar] [CrossRef] [PubMed]
- Tang, D.; Jia, Y.; Zhang, J.; Li, H.; Cheng, L.; Wang, P.; Bao, Z.; Liu, Z.; Feng, S.; Zhu, X.; et al. Genome evolution and diversity of wild and cultivated potatoes. Nature 2022, 606, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Sharma, P.; Gonzalez, D.H.; Viola, I.L.; Hannapel, D.J. The impact of the long-distance transport of a BEL1-like messenger RNA on development. Plant Physiol. 2013, 161, 760–772. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Rosin, F.M.; Prat, S.; Hannapel, D.J. Interacting transcription factors from the TALE superclass regulate tuber formation. Plant Physiol. 2003, 132, 1391–1404. [Google Scholar] [CrossRef]
- Banerjee, A.K.; Lin, T.; Hannapel, D.J. Untranslated regions of a mobile transcript mediate RNA metabolism. Plant Physiol. 2009, 151, 1831–1843. [Google Scholar] [CrossRef]
- Ye, X.; Salim, A.; Klöti, A.; Zhang, J.; Lucca, P.; Beyer, P.; Potrykus, I. Engineering the provitamin A (β-Carotene) biosynthetic pathway into (carotenoid-Free) rice endosperm. Science 2000, 287, 303–305. [Google Scholar] [CrossRef]
- Abbadi, A.; Domergue, F.; Bauer, J.; Napier, J.A.; Welti, R.; Zähringer, U.; Cirpus, P.; Heinz, E. Biosynthesis of very-long-chain polyunsaturated fatty acids in transgenic oilseeds: Constraints on their accumulation. Plant Cell 2004, 16, 2734–2748. [Google Scholar] [CrossRef]
- Qi, B.; Fraser, T.; Mugford, S.; Dobson, G.; Sayanova, O.; Butler, J.; Napier, J.A.; Stobart, A.K.; Lazarus, C.M. Production of very long chain polyunsaturated omega-3 and omega-6 fatty acids in plants. Nat. Biotechnol. 2004, 22, 739–745. [Google Scholar] [CrossRef]
- Wu, G.; Truksa, M.; Datla, N.; Vrinten, P.; Bauer, J.; Zank, T.; Cirpus, P.; Heinz, E.; Qiu, X. Stepwise engineering to produce high yields of very long-chain polyunsaturated fatty acids in plants. Nat. Biotechnol. 2005, 23, 1013–1017. [Google Scholar] [CrossRef]
- Karunanandaa, B.; Qi, Q.; Hao, M.; Baszis, S.R.; Jensen, P.K.; Wong, Y.H.; Jiang, J.; Venkatramesh, M.; Gruys, K.J.; Moshiri, F.; et al. Metabolically engineered oilseed crops with enhanced seed tocopherol. Metab. Eng. 2005, 7, 384–400. [Google Scholar] [CrossRef]
- Raclaru, M.; Gruber, J.; Kumar, R.; Sadre, R.; Lühs, W.; Zarhloul, M.K.; Friedt, W.; Frentzen, M.; Weier, D. Increase of the tocochromanol content in transgenic Brassica napus seeds by overexpression of key enzymes involved in prenylquinone biosynthesis. Mol. Breed. 2006, 18, 93–107. [Google Scholar] [CrossRef]
- Diretto, G.; Al-Babili, S.; Tavazza, R.; Papacchioli, V.; Beyer, P.; Giuliano, G. Metabolic engineering of potato carotenoid content through tuber-specific overexpression of a bacterial mini-pathway. PLoS ONE 2007, 2, e350. [Google Scholar] [CrossRef]
- Zhu, C.; Naqvi, S.; Breitenbach, J.; Sandmann, G.; Christou, P.; Capell, T. Combinatorial genetic transformation generates a library of metabolic phenotypes for the carotenoid pathway in maize. Proc. Natl. Acad. Sci. USA 2008, 105, 18232–18237. [Google Scholar] [CrossRef]
- Fujisawa, M.; Takita, E.; Harada, H.; Sakurai, N.; Suzuki, H.; Ohyama, K.; Shibata, D.; Misawa, N. Pathway engineering of Brassica napus seeds using multiple key enzyme genes involved in ketocarotenoid formation. J. Exp. Bot. 2009, 60, 1319–1332. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Wagner, M.; Abbadi, A.; Fulda, M.; Feussner, I. Metabolic engineering of omega-3-very long chain polyunsaturated fatty acid production by an exclusively acyl CoA-dependent pathway. J. Biol. Chem. 2009, 283, 22352–22362. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, S.; Farré, G.; Sanahuja, G.; Capell, T.; Zhu, C.; Christou, P. When more is better: Multigene engineering in plants. Trend. Plant Sci. 2009, 15, 48–56. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, S.; Zhu, C.; Farre, G.; Ramessar, K.; Bassie, L.; Breitenbach, J.; Perez Conesa, D.; Ros, G.; Sandmann, G.; Capell, T.; et al. Transgenic multivitamin corn: Biofortification of corn endosperm with three vitamins representing three distinct metabolic pathways. Proc. Natl. Acad. Sci. USA 2009, 106, 7762–7767. [Google Scholar] [CrossRef]
- Wang, L.Z.; Yang, C.; Chen, H.; Wang, P.; Wang, P.; Song, C.; Zhang, X.; Wang, D. Multi-gene co-expression can improve comprehensive resistance to multiple abiotic stresses in Brassica napus. Plant Sci. 2018, 274, 410–419. [Google Scholar] [CrossRef]
- Natarajan, B.; Kondhare, K.R.; Hannapel, D.J.; Banerjee, A.K. Mobile RNAs and proteins: Prospects in storage organ development of tuber and root crops. Plant Sci. 2019, 284, 73–81. [Google Scholar] [CrossRef]
- Wang, S.; Okamoto, T. Involvement of polypyrimidine tract-binding protein (PTB)-related proteins in pollen germination in Arabidopsis. Plant Cell Physiol. 2009, 50, 179–190. [Google Scholar] [CrossRef]
- Rühl, C.; Stauffer, E.; Kahles, A.; Wagner, G.; Drechsel, G.; Rätsch, G.; Wachter, A. Polypyrimidine tract binding protein homologs from Arabidopsis are key regulators of alternative splicing with implications in fundamental developmental processes. Plant Cell 2012, 24, 4360–4375. [Google Scholar] [CrossRef] [PubMed]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plantarum. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Prematilake, D.P.; Mendis, M.H. Microtubers of potato (Solanum tuberosum L.): In vitro conservation and tissue culture. J. Natl. Sci. Found. Sri Lanka 1999, 27, 17–28. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C (T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kondhare, K.R.; Patil, N.S.; Siddappa, S.; Banerjee, A.K.; Hannapel, D.J. Tandem Expression of a Mobile RNA and Its RNA-Binding Protein(s) Enhances Tuber Productivity in Potato. Int. J. Mol. Sci. 2023, 24, 15754. https://doi.org/10.3390/ijms242115754
Kondhare KR, Patil NS, Siddappa S, Banerjee AK, Hannapel DJ. Tandem Expression of a Mobile RNA and Its RNA-Binding Protein(s) Enhances Tuber Productivity in Potato. International Journal of Molecular Sciences. 2023; 24(21):15754. https://doi.org/10.3390/ijms242115754
Chicago/Turabian StyleKondhare, Kirtikumar R., Nikita S. Patil, Sundaresha Siddappa, Anjan K. Banerjee, and David J. Hannapel. 2023. "Tandem Expression of a Mobile RNA and Its RNA-Binding Protein(s) Enhances Tuber Productivity in Potato" International Journal of Molecular Sciences 24, no. 21: 15754. https://doi.org/10.3390/ijms242115754




