Antifibrotic Soluble Thy-1 Correlates with Renal Dysfunction in Chronic Kidney Disease
Abstract
1. Introduction
2. Results
2.1. Renal Function Is the Strongest, Inverse Predictor of Circulating sThy-1 Levels in Patients with CKD—Cohort 1 (N = 120)
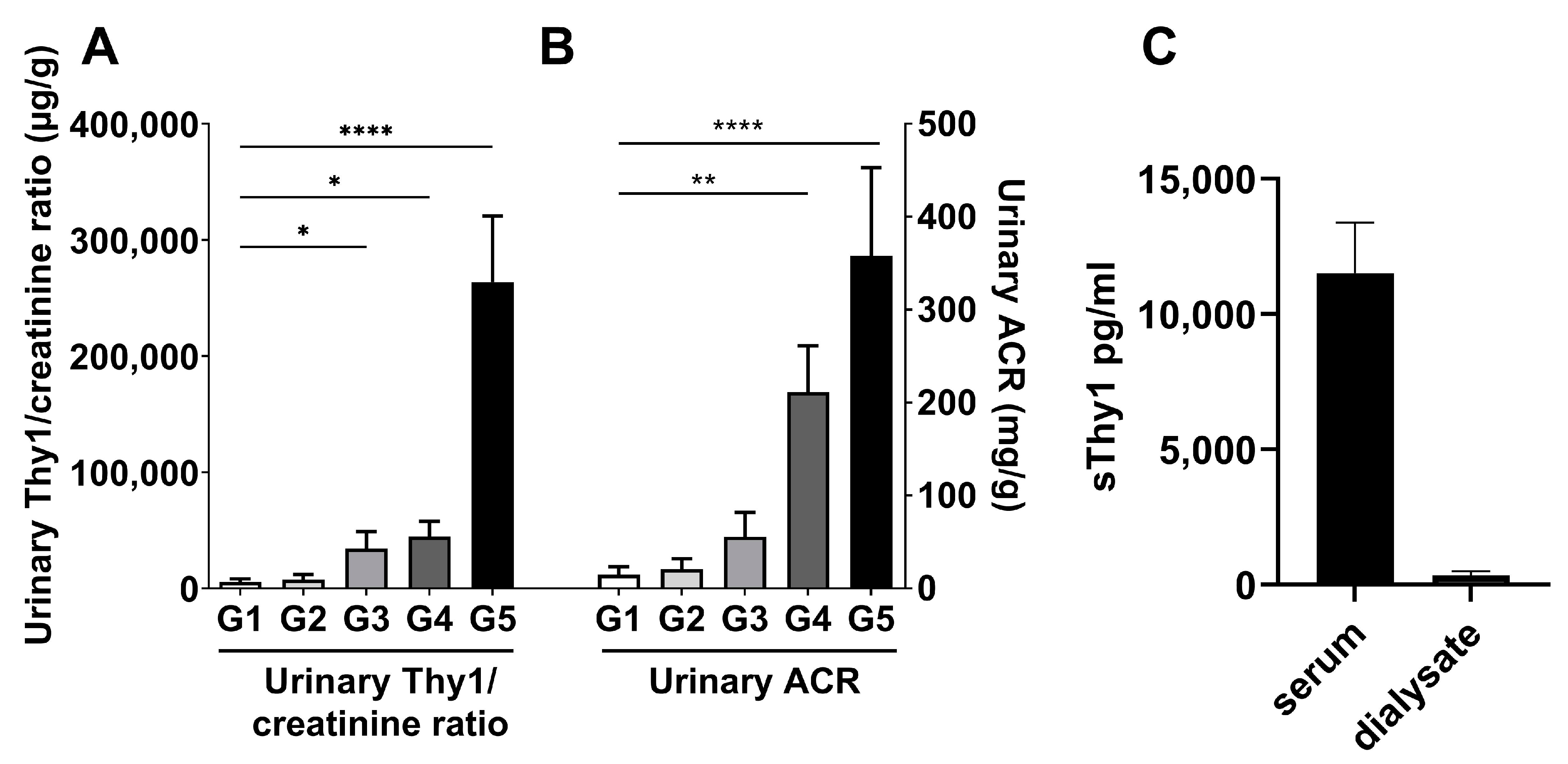
2.2. Urinary sThy-1/Creatinine Levels and Dialysate sThy-1 in Patients with CKD—Cohort 2 (N = 50) and Cohort 3 (N = 6)
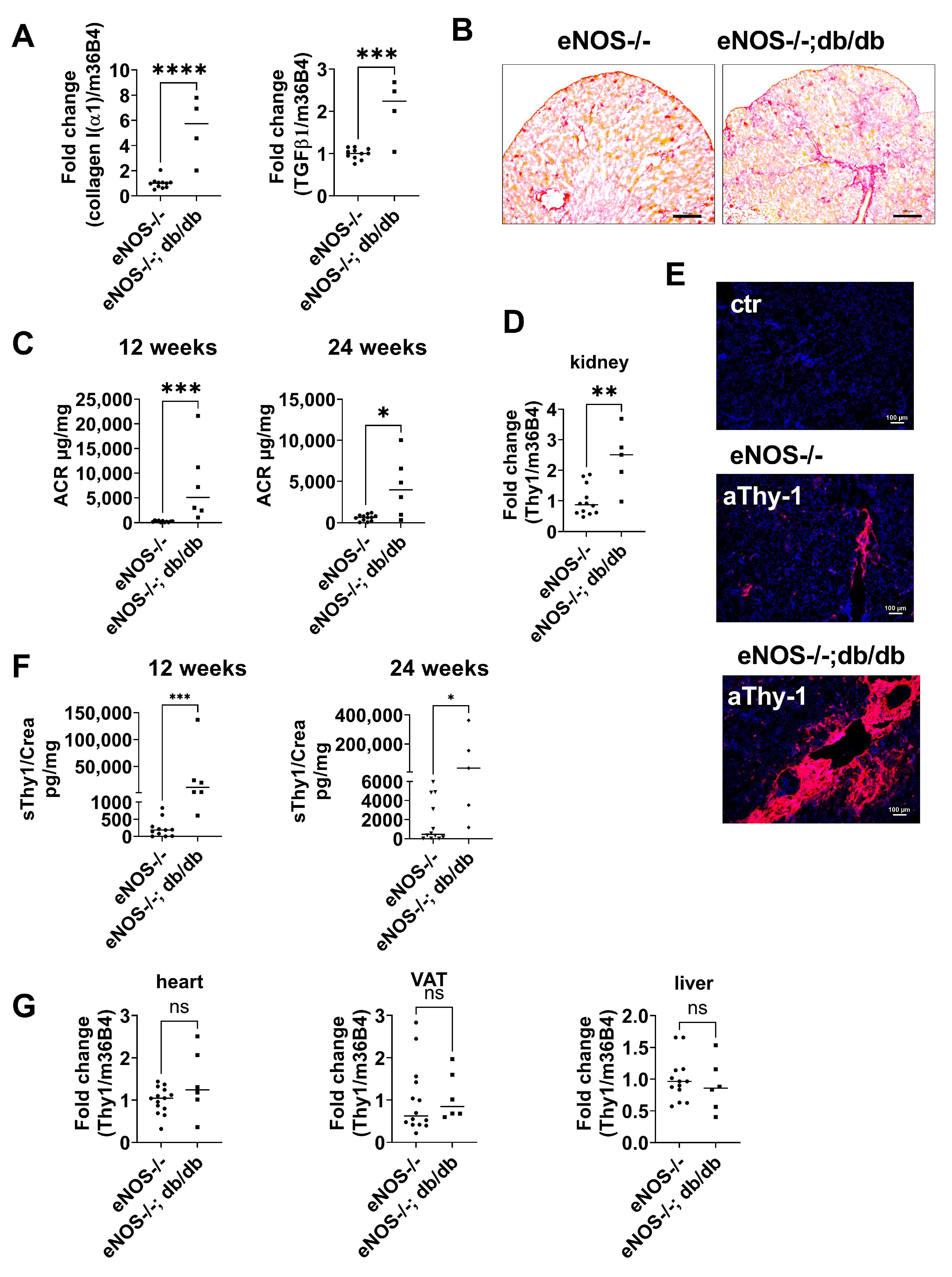
2.3. Thy-1 Expression Is Increased in a Mouse Model of CKD
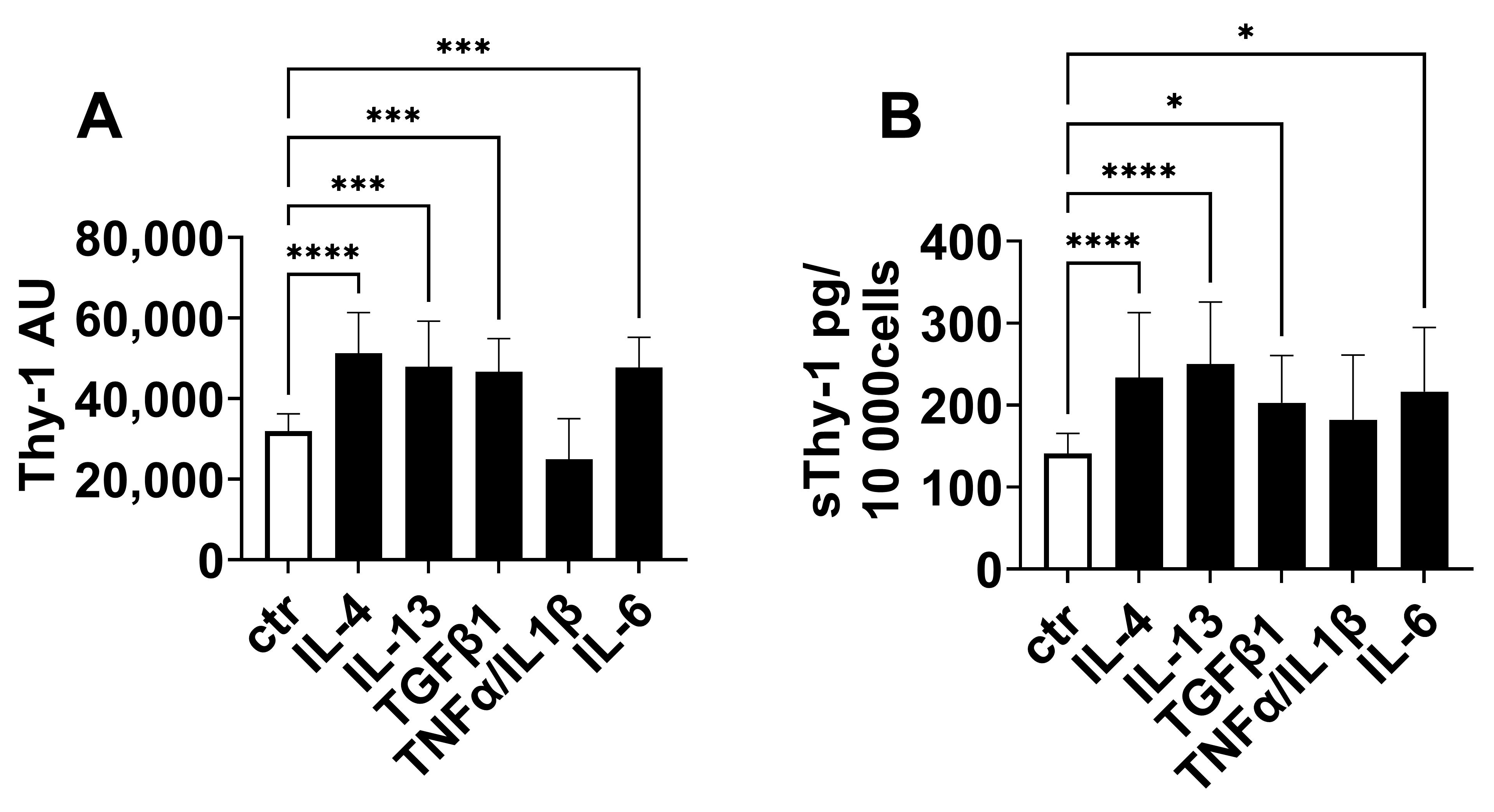
2.4. Regulation of Thy-1 Expression and Release of sThy-1
3. Discussion
4. Material and Methods
4.1. Human Studies
4.1.1. Cohort 1
4.1.2. Cohort 2
4.1.3. Cohort 3
4.2. Detection of Human sThy-1
4.3. Animal Studies
4.4. Staining of Tissue Sections
4.5. Gene Expression
4.6. Cell Culture
4.7. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bikbov, B.; Purcell, C.; Levey, A.; Smith, M.; Abdoli, A.; Abebe, M.; Adebayo, O.; Afarideh, M.; Agarwal, S.K.; Agudelo-Botero, M.; et al. Global, regional, and national burden of chronic kidney disease, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2020, 395, 709–733. [Google Scholar] [CrossRef] [PubMed]
- Matsushita, K.; van der Velde, M.; Astor, B.; Woodward, M.; Levey, A.; de Jong, P.D.; Coresh, J.; Gansevoort, R. Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: A collaborative meta-analysis. Lancet 2010, 375, 2073–2081. [Google Scholar] [PubMed]
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C. Chronic Kidney Disease and the Risks of Death, Cardiovascular Events, and Hospitalization. New Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Ebert, T.; Pawelzik, S.-C.; Witasp, A.; Arefin, S.; Hobson, S.; Kublickiene, K.; Shiels, P.G.; Bäck, M.; Stenvinkel, P. Inflammation and Premature Ageing in Chronic Kidney Disease. Toxins 2020, 12, 227. [Google Scholar] [CrossRef] [PubMed]
- Lousa, I.; Reis, F.; Beirão, I.; Alves, R.; Belo, L.; Santos-Silva, A. New Potential Biomarkers for Chronic Kidney Disease Management—A Review of the Literature. Int. J. Mol. Sci. 2020, 22, 43. [Google Scholar] [CrossRef]
- Ebert, T.; Tran, N.; Schurgers, L.; Stenvinkel, P.; Shiels, P.G. Ageing—Oxidative stress, PTMs and disease. Mol. Asp. Med. 2022, 86, 101099. [Google Scholar] [CrossRef]
- Ebert, T.; Neytchev, O.; Witasp, A.; Kublickiene, K.; Stenvinkel, P.; Shiels, P.G. Inflammation and Oxidative Stress in Chronic Kidney Disease and Dialysis Patients. Antioxid. Redox Signal. 2021, 35, 1426–1448. [Google Scholar] [CrossRef]
- Panizo, S.; Martínez-Arias, L.; Alonso-Montes, C.; Cannata, P.; Martín-Carro, B.; Fernández-Martín, J.; Naves-Díaz, M.; Carrillo-López, N.; Cannata-Andía, J. Fibrosis in Chronic Kidney Disease: Pathogenesis and Consequences. Int. J. Mol. Sci. 2021, 22, 408. [Google Scholar] [CrossRef]
- Ruiz-Ortega, M.; Rayego-Mateos, S.; Lamas, S.; Ortiz, A.; Rodrigues-Diez, R.R. Targeting the progression of chronic kidney disease. Nat. Rev. Nephrol. 2020, 16, 269–288. [Google Scholar] [CrossRef]
- Hu, P.; Leyton, L.; Hagood, J.S.; Barker, T.H. Thy-1-Integrin Interactions in cis and Trans Mediate Distinctive Signaling. Front. Cell Dev. Biol. 2022, 10, 928510. [Google Scholar] [CrossRef]
- Saalbach, A.; Anderegg, U. Thy-1: More than a marker for mesenchymal stromal cells. FASEB J. 2019, 33, 6689–6696. [Google Scholar] [CrossRef] [PubMed]
- Sanders, Y.Y.; Kumbla, P.; Hagood, J.S. Enhanced Myofibroblastic Differentiation and Survival in Thy-1(−) Lung Fibroblasts. Am. J. Respir. Cell Mol. Biol. 2007, 36, 226–235. [Google Scholar] [CrossRef] [PubMed]
- Ramírez, G.; Hagood, J.S.; Sanders, Y.; Ramírez, R.; Becerril, C.; Segura, L.; Barrera, L.; Selman, M.; Pardo, A. Absence of Thy-1 results in TGF-Β induced MMP-9 expression and confers a profibrotic phenotype to human lung fibroblasts. Lab. Investig. 2011, 91, 1206–1218. [Google Scholar] [CrossRef] [PubMed]
- Hagood, J.S.; Prabhakaran, P.; Kumbla, P.; Salazar, L.; MacEwen, M.W.; Barker, T.H.; Ortiz, L.A.; Schoeb, T.; Siegal, G.P.; Alexander, C.B.; et al. Loss of Fibroblast Thy-1 Expression Correlates with Lung Fibrogenesis. Am. J. Pathol. 2005, 167, 365–379. [Google Scholar] [CrossRef]
- Chen, L.; Tang, R.-Z.; Ruan, J.; Zhu, X.-B.; Yang, Y. Up-regulation of THY1 attenuates interstitial pulmonary fibrosis and promotes lung fibroblast apoptosis during acute interstitial pneumonia by blockade of the WNT signaling pathway. Cell Cycle 2019, 18, 670–681. [Google Scholar] [CrossRef]
- Li, Y.; Song, D.; Mao, L.; Abraham, D.M.; Bursac, N. Lack of Thy1 defines a pathogenic fraction of cardiac fibroblasts in heart failure. Biomaterials 2020, 236, 119824. [Google Scholar] [CrossRef]
- Narisawa-Saito, M.; Kimura, S.; Fujiwara, N.; Oite, T.; Shimoji, K.; Shimizu, F. Thy-1-mediated phosphatidylinositol turnover in cultured rat glomerular mesangial cell. J. Cell. Physiol. 1996, 168, 705–710. [Google Scholar] [CrossRef]
- Wu, L.; Chang, D.-Y.; Zhang, L.-X.; Chen, M.; Zhao, M.-H. Urinary soluble CD90 predicts renal prognosis in patients with diabetic kidney disease. Ann. Transl. Med. 2021, 9, 282. [Google Scholar] [CrossRef]
- Nishio, T.; Koyama, Y.; Liu, X.; Rosenthal, S.B.; Yamamoto, G.; Fuji, H.; Baglieri, J.; Li, N.; Brenner, L.N.; Iwaisako, K.; et al. Immunotherapy-based targeting of MSLN + activated portal fibroblasts is a strategy for treatment of cholestatic liver fibrosis. Proc. Natl. Acad. Sci. USA 2021, 118, e2101270118. [Google Scholar] [CrossRef]
- Bradley, J.E.; Ramirez, G.; Hagood, J.S. Roles and regulation of Thy-1, a context-dependent modulator of cell phenotype. Biofactors 2009, 35, 258–265. [Google Scholar] [CrossRef]
- Saalbach, A.; Wetzig, T.; Haustein, U.F.; Anderegg, U. Detection of human soluble Thy-1 in serum by ELISA. Cell Tissue Res. 1999, 298, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Li, W.; Shotorbani, P.Y.; Dubansky, B.H.; Huang, L.; Chaudhari, S.; Wu, P.; Wang, L.A.; Ryou, M.-G.; Zhou, Z.; et al. Comparison of diabetic nephropathy between male and female eNOS −/− db / db mice. American Journal of Physiology-Renal Physiology 2019, 316, F889–F897. [Google Scholar] [CrossRef] [PubMed]
- Vikse, B.E.; Vollset, S.E.; Tell, G.S.; Refsum, H.; Iversen, B.M. Distribution and determinants of serum creatinine in the general population: The Hordaland Health Study. Scand. J. Clin. Lab. Investig. 2004, 64, 709–722. [Google Scholar] [CrossRef]
- Zou, D.; Wu, W.; He, Y.; Ma, S.; Gao, J. The role of klotho in chronic kidney disease. BMC Nephrol. 2018, 19, 285. [Google Scholar] [CrossRef]
- Schmidt, M.; Gutknecht, D.; Simon, J.C.; Schulz, J.-N.; Eckes, B.; Anderegg, U.; Saalbach, A. Controlling the balance of fibroblast proliferation and differentiation: Impact of thy-1. J. Investig. Dermatol. 2015, 135, 1893–1902. [Google Scholar] [CrossRef] [PubMed]
- Cohen, P.Y.; Breuer, R.; Wallach-Dayan, S.B. Thy1 up-regulates FasL expression in lung myofibroblasts via Src family kinases. Am. J. Respir. Cell Mol. Biol. 2009, 40, 231–238. [Google Scholar] [CrossRef]
- Zhou, Y.; Hagood, J.S.; Lu, B.; Merryman, W.D.; Murphy-Ullrich, J.E. Thy-1-integrin alphav beta5 interactions inhibit lung fibroblast contraction-induced latent transforming growth factor-beta1 activation and myofibroblast differentiation. J. Biol. Chem. 2010, 285, 22382–22393. [Google Scholar] [CrossRef]
- Zhou, Y.; Hagood, J.S.; Murphy-Ullrich, J.E. Thy-1 expression regulates the ability of rat lung fibroblasts to activate transforming growth factor-beta in response to fibrogenic stimuli. Am. J. Pathol. 2004, 165, 659–669. [Google Scholar] [CrossRef]
- Almqvist, P.; Carlsson, S.R. Characterization of a hydrophilic form of Thy-1 purified from human cerebrospinal fluid. J. Biol. Chem. 1988, 263, 12709–12715. [Google Scholar] [CrossRef]
- Kollert, F.; Christoph, S.; Probst, C.; Budweiser, S.; Bannert, B.; Binder, M.; Sehnert, B.; Voll, R.E.; Warnatz, K.; Zissel, G.; et al. Soluble CD90 as a potential marker of pulmonary involvement in systemic sclerosis. Arthritis Care Res. 2013, 65, 281–287. [Google Scholar] [CrossRef]
- Liu, X.; Wong, S.S.; Taype, C.A.; Kim, J.; Shentu, T.-P.; Espinoza, C.R.; Finley, J.C.; Bradley, E.J.; Head, B.P.; Patel, H.H.; et al. Thy-1 interaction with Fas in lipid rafts regulates fibroblast apoptosis and lung injury resolution. Lab. Investig. 2017, 97, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Gutknecht, D.; Anastassiadis, K.; Eckes, B.; Anderegg, U.; Saalbach, A. Thy-1/beta3 Integrin Interaction-Induced Apoptosis of Dermal Fibroblasts Is Mediated by Up-Regulation of FasL Expression. J. Investig. Dermatol. 2016, 136, 526–529. [Google Scholar] [CrossRef] [PubMed]
- Leyton, L.; Díaz, J.; Martínez, S.; Palacios, E.; Pérez, L.A.; Pérez, R.D. Thy-1/CD90 a Bidirectional and Lateral Signaling Scaffold. Front Cell Dev Biol. 2019, 7, 132. [Google Scholar] [CrossRef] [PubMed]
- Fiore, V.F.; Strane, P.W.; Bryksin, A.V.; White, E.S.; Hagood, J.S.; Barker, T.H. Conformational coupling of integrin and Thy-1 regulates Fyn priming and fibroblast mechanotransduction. J. Cell Biol. 2015, 211, 173–190. [Google Scholar] [CrossRef] [PubMed]
- Koyama, Y.; Wang, P.; Liang, S.; Iwaisako, K.; Liu, X.; Xu, J.; Zhang, M.; Sun, M.; Cong, M.; Karin, D.; et al. Mesothelin/mucin 16 signaling in activated portal fibroblasts regulates cholestatic liver fibrosis. J. Clin. Investig. 2017, 127, 1254–1270. [Google Scholar] [CrossRef] [PubMed]
- Saalbach, A.; Kraft, R.; Herrmann, K.; Haustein, U.-F.; Anderegg, U. The monoclonal antibody AS02 recognizes a protein on human fibroblasts being highly homologous to Thy-1. Arch. Dermatol. Res. 1998, 290, 360–366. [Google Scholar] [CrossRef] [PubMed]
- Morris, R.J. Thy-1, a Pathfinder Protein for the Post-genomic Era. Front. Cell Dev. Biol. 2018, 6, 173. [Google Scholar] [CrossRef]
- Yang, J.; Zhan, X.-Z.; Malola, J.; Li, Z.-Y.; Pawar, J.S.; Zhang, H.-T.; Zha, Z.-G. The multiple roles of Thy-1 in cell differentiation and regeneration. Differentiation 2020, 113, 38–48. [Google Scholar] [CrossRef]
- Kralisch, S.; Hoffmann, A.; Klöting, N.; Bachmann, A.; Kratzsch, J.; Stolzenburg, J.-U.; Dietel, A.; Beige, J.; Anders, M.; Bast, I.; et al. FSTL3 is increased in renal dysfunction. Nephrol. Dial. Transplant. 2017, 32, 1637–1644. [Google Scholar] [CrossRef]
- Milik, A.; Hrynkiewicz, E. On translation of LD, IL and SFC given according to IEC-61131 for hardware synthesis of reconfigurable logic controller. IFAC Proc. Vol. 2014, 19, 4477–4483. [Google Scholar] [CrossRef]
- Kralisch, S.; Hoffmann, A.; Klöting, N.; Bachmann, A.; Kratzsch, J.; Blüher, M.; Zhang, M.-Z.; Harris, R.; Stumvoll, M.; Fasshauer, M.; et al. The novel adipokine/hepatokine fetuin B in severe human and murine diabetic kidney disease. Diabetes Metab. 2017, 43, 465–468. [Google Scholar] [CrossRef] [PubMed]
- Kralisch, S.; Hoffmann, A.; Klöting, N.; Frille, A.; Kuhn, H.; Nowicki, M.; Paeschke, S.; Bachmann, A.; Blüher, M.; Zhang, M.-Z.; et al. The brown fat-secreted adipokine neuregulin 4 is decreased in human and murine chronic kidney disease. Eur. J. Endocrinol. 2019, 181, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Tönjes, A.; Hoffmann, A.; Kralisch, S.; Qureshi, A.R.; Klöting, N.; Scholz, M.; Schleinitz, D.; Bachmann, A.; Kratzsch, J.; Nowicki, M.; et al. Pro-neurotensin depends on renal function and is related to all-cause mortality in chronic kidney disease. Eur. J. Endocrinol. 2020, 183, 233–244. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Stevens, P.E.; Bilous, R.W.; Coresh, J.; De Francisco, A.L.; De Jong, P.E.; Griffith, K.E.; Hemmelgarn, B.R.; Iseki, K.; Lamb, E.J.; et al. Kidney disease: Improving global outcomes (KDIGO) CKD work group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013, 3, 1–150. [Google Scholar]
- Zhao, H.J.; Wang, S.; Cheng, H.; Zhang, M.-Z.; Takahashi, T.; Fogo, A.B.; Breyer, M.D.; Harris, R.C. Endothelial Nitric Oxide Synthase Deficiency Produces Accelerated Nephropathy in Diabetic Mice. J. Am. Soc. Nephrol. 2006, 17, 2664–2669. [Google Scholar] [CrossRef]
| Subgroup 1 Control/nonT2D | Subgroup 2 Control/T2D | Subgroup 3 HD/nonT2D | Subgroup 4 HD/T2D | Overall p | |
|---|---|---|---|---|---|
| N | 30 | 30 | 28 | 32 | - |
| Sex (male/female) | 11/19 | 16/14 | 15/13 | 20/12 | 0.239 |
| sThy-1 (µg/L) | 5.2 (1.7) 3,4 | 4.6 (1.2) 3,4 | 16.4 (3.3) 1,2 | 16.3 (3.8) 1,2 | <0.001 |
| Age (years) | 63 (19) | 63 (16) | 59 (23) | 68 (12) | 0.051 |
| BMI (kg/m²) | 28.2 (5.6) | 29.1 (5.2) 3 | 25.2 (6.5) 2 | 27.9 (6.6) | 0.004 |
| WHR | 0.88 (0.12) 4 | 0.94 (0.10) | 0.96 (0.18) | 1.00 (0.14) 1 | <0.001 |
| SBP (mmHg) | 125 (21) | 126 (20) | 125 (38) | 120 (25) | 0.339 |
| DBP (mmHg) | 77 (10) | 73 (15) | 77 (20) | 70 (18) | 0.095 |
| FG (mmol/L) | 5.1 (1.3) 2 | 7.6 (3.2) 1,3,4 | 4.6 (1.2) 2 | 5.2 (3.3) 2 | <0.001 |
| Total cholesterol (mmol/L) | 5.3 (0.9) 3,4 | 4.9 (1.5) | 4.4 (1.1) 1 | 4.2 (1.3) 1 | <0.001 |
| HDL cholesterol (mmol/L) | 1.4 (0.4) 3,4 | 1.2 (0.5) 4 | 1.0 (0.5) 1 | 1.0 (0.3) 1,2 | <0.001 |
| LDL cholesterol (mmol/L) | 3.5 (1.1) 2,3,4 | 2.9 (0.9) 1 | 2.7 (0.9) 1 | 2.1 (1.4) 1 | <0.001 |
| TG (mmol/L) | 1.1 (0.8) 3,4 | 1.4 (0.9) | 1.6 (0.9) 1 | 1.8 (1.4) 1 | <0.001 |
| Creatinine (μmol/L) | 76 (17) 3,4 | 72 (22) 3,4 | 829 (431) 1,2 | 717 (221) 1,2 | <0.001 |
| eGFR (ml/min/1.73 m2) | 78.8 (24.4) 3,4 | 85.2 (23.0) 3,4 | 5.0 (3.2) 1,2 | 5.7 (2.7) 1,2 | <0.001 |
| hsIL-6 (ng/L) | 1.87 (2.11) 3,4 | 2.07 (1.25) 3,4 | 6.14 (4.91) 1,2 | 7.48 (7.30) 1,2 | <0.001 |
| Univariate Correlation Analyses | Multivariate Linear Regression Analysis | |||
|---|---|---|---|---|
| r | p | β | p | |
| Age (years) | 0.112 | 0.224 | - | - |
| Sex | - | - | 0.143 | <0.001 |
| BMI (kg/m2) | −0.142 | 0.122 | - | - |
| WHR | 0.208 | 0.022 | 0.065 | 0.01 |
| Fasting glucose (mmol/L) | −0.352 | <0.001 | −0.157 | <0.001 |
| SBP (mmHg) | −0.081 | 0.377 | - | - |
| DBP (mmHg) | −0.151 | 0.1 | - | - |
| Total cholesterol (mmol/L) | −0.26 | 0.004 | - | - |
| HDL cholesterol (mmol/L) | −0.374 | <0.001 | −0.161 | <0.001 |
| LDL cholesterol (mmol/L) | −0.292 | 0.001 | - | - |
| TG (mmol/L) | 0.297 | <0.001 | - | - |
| Creatinine (µmol/L) | 0.777 | <0.001 | 0.662 | <0.001 |
| eGFR (ml/min/1.73 m²) | −0.81 | <0.001 | - | - |
| hsIL-6 (ng/L) | 0.752 | <0.001 | 0.329 | <0.001 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saalbach, A.; Anderegg, U.; Wendt, R.; Beige, J.; Bachmann, A.; Klöting, N.; Blüher, M.; Zhang, M.-Z.; Harris, R.C.; Stumvoll, M.; et al. Antifibrotic Soluble Thy-1 Correlates with Renal Dysfunction in Chronic Kidney Disease. Int. J. Mol. Sci. 2023, 24, 1896. https://doi.org/10.3390/ijms24031896
Saalbach A, Anderegg U, Wendt R, Beige J, Bachmann A, Klöting N, Blüher M, Zhang M-Z, Harris RC, Stumvoll M, et al. Antifibrotic Soluble Thy-1 Correlates with Renal Dysfunction in Chronic Kidney Disease. International Journal of Molecular Sciences. 2023; 24(3):1896. https://doi.org/10.3390/ijms24031896
Chicago/Turabian StyleSaalbach, Anja, Ulf Anderegg, Ralph Wendt, Joachim Beige, Anette Bachmann, Nora Klöting, Matthias Blüher, Ming-Zhi Zhang, Raymond C. Harris, Michael Stumvoll, and et al. 2023. "Antifibrotic Soluble Thy-1 Correlates with Renal Dysfunction in Chronic Kidney Disease" International Journal of Molecular Sciences 24, no. 3: 1896. https://doi.org/10.3390/ijms24031896
APA StyleSaalbach, A., Anderegg, U., Wendt, R., Beige, J., Bachmann, A., Klöting, N., Blüher, M., Zhang, M.-Z., Harris, R. C., Stumvoll, M., Tönjes, A., & Ebert, T. (2023). Antifibrotic Soluble Thy-1 Correlates with Renal Dysfunction in Chronic Kidney Disease. International Journal of Molecular Sciences, 24(3), 1896. https://doi.org/10.3390/ijms24031896







