Abstract
Metarhizium anisopliae is an entomopathogenic fungus which may enhance plant growth and resistance when acting as an endophyte in host plants. However, little is known about the protein interactions nor their activating mechanisms. Common in fungal extracellular membrane (CFEM) proteins have been identified as plant immune regulators that suppress or activate plant resistance responses. Here, we identified a CFEM domain-containing protein, MaCFEM85, which was mainly localized in the plasma membrane. Yeast two-hybrid (Y2H), glutathione-S-transferase (GST) pull-down, and bimolecular fluorescence complementation assays demonstrated that MaCFEM85 interacted with the extracellular domain of a Medicago sativa (alfalfa) membrane protein, MsWAK16. Gene expression analyses showed that MaCFEM85 and MsWAK16 were significantly upregulated in M. anisopliae and M. sativa, respectively, from 12 to 60 h after co-inoculation. Additional yeast two-hybrid assays and amino acid site-specific mutation indicated that the CFEM domain and 52th cysteine specifically were required for the interaction of MaCFEM85 with MsWAK16. Defense function assays showed that JA was up-regulated, but Botrytis cinerea lesion size and Myzus persicae reproduction were suppressed by transient expression of MaCFEM85 and MsWAK16 in the model host plant Nicotiana benthamiana. Collectively, these results provide novel insights into the molecular mechanisms underlying interactions of M. anisopliae with host plants.
1. Introduction
Interactions between plants and microbes are widespread and are the result of past and ongoing co-evolution. These interactions can have either beneficial, neutral, or adverse outcomes for each participant [1,2,3]. Beneficial rhizospheric microbiota can enhance plant growth and improve overall health by protecting against soilborne diseases or enhancing nutrient uptake [4,5]. A wide range of beneficial microbes can enhance plant capabilities such as nutrient uptake, growth, and pathogen and insect defenses [6,7].
Metarhizium Sorokīn is an important genus of entomopathogenic fungi with the ability to colonize plants [8], but there is also evidence to suggest that members of the genus can enhance plant pathogen resistance. For example, a laboratory study found 60% inhibition of Fusarium solani (Mart.) Sacc. in the presence of Metarhizium robertsii (formerly known as M. anisopliae) compared with controls without M. robertsii [9]. Some Metarhizium strains have the potential to improve plant resistance to insect pests and diseases by altering the expression of plant defense genes. Endophytic colonization with M. robertsii activates expression of both the jasmonic acid (JA) and salicylic acid (SA) defense pathways in maize leaves; furthermore, such colonization is associated with the promotion of plant growth and suppression of Agrotis ipsilon (Hufnagel) larval development [10]. Metarhizium guizhouense activated the β-1,3-glucanase and chitinase in fruit peel of Lansium parasiticum resulting in the growth inhibition of Botrytis sp. and Fusarium sp. on the fruit of Aglaia dookkoo Griff [11]. Moreover, when M. anisopliae was applied to peanut seedlings, a variety of transcription factors including WRKYs, MYCs, TGAs, and ethylene-responsive transcription factors were activated, while nitrate transporters and proteins binding dehydration-responsive elements were also differentially expressed [12]. This suggests that M. anisopliae regulates plant defense genes as it colonizes the plant. However, relatively little is known about the mechanisms underlying plant defense responses following infection by Metarhizium.
Effectors are a common means by which microorganisms regulate plant resistance [13]. The activation of plant resistance is usually characterized by an oxidative burst and the induction of hormones, metabolites, and other signals [13]. The plant hormones SA and JA play important roles in inducing plant resistance, mediating two separate defense signaling pathways [14]. The SA signaling pathway primarily functions in the plant allergic response and in systemic acquired resistance to pathogens, but it may also be involved in indirect plant defense responses induced by stinging insect feeding [15]. Accumulation of SA is associated with increased expression of genes encoding lipid transfer proteins (LTPs) and phenylalanine ammonia-lyase (PAL) [16], which mediate the synthesis and accumulation of downstream specialized metabolites such as flavonoids and lignin [17]. The JA signaling pathway functions in direct and indirect responses to mechanical damage, fungal pathogens, and insect pests. Studies have shown that JA signaling has a regulatory effect on the synthesis of specialized metabolites such as terpenoids, phenylpropanoids, and alkaloids, which have a wide range of biological functions [18]. Some specialized metabolites have been shown to accumulate in plant cells following treatment with methyl-JA (MeJA), including paclitaxel in Taxus species [19,20], terpenoids in Centella asiatica (L.) Urban [21], and saponins in ginseng [22]. Application of SA and chitosan (CHT) as elicitors induced lignin accumulation and defense enzyme reactions in tomato, which reduced the incidence of Ralstonia solanacearum (Smith) Yabuuchi infection [23]. In addition, JA and SA levels in wheat were significantly accumulated by application of the elicitor PeaT, enhancing the defense response to the oat aphid Sitobion avenae (Fabricius) [24]. This indicates that plant immunity could be regulated by these effectors through plant hormones and metabolites.
Common in fungal extracellular membrane (CFEM) proteins are present in a wide range of fungi. They encode domains that are usually 60 amino acids (aa) in length and contain eight characteristically spaced cysteines [25]. CFEM domains are similar to the epidermal growth factor (EGF)-like domains, which function as extracellular receptors, signal transducers, or adhesion molecules in host–pathogen interactions [26]. Proteins containing CFEM domains may manipulate the plant resistance response by acting as effectors. In the wheat leaf rust fungus Puccinia triticina Erikas, the CFEM effector candidate PTTG_08198 accelerated the progress of cell death and promoted reactive oxygen species (ROS) accumulation [27]. The anthracnose fungus Colletotrichum graminicola (Ces.) G. W. Wils contains five effectors (CgCFEM6, 7, 8, 9, and 15) that have been shown to suppress BAX-induced programmed cell death in Nicotiana benthamiana Domin [28]. The phytosymbiotic mycorrhizal fungus Laccaria bicolor (Maire) P.D.Orton was reported to secrete several CFEM proteins, such as Lac310796, Lac296573, and Lac296572, in symbiotic tissues [29]. Although the complex functions of these proteins have not been studied further in mycorrhizal symbiosis, prior results suggest that CFEM proteins may function in signaling between fungi and plants.
M. anisopliae can function as either an entomopathogenic or endophytic fungus in a host plant [30]. However, the roles of CFEM proteins have not yet been reported. We previously identified a CFEM domain-containing protein in M. anisopliae, MaCFEM85 (GenBank ID: MZ682609) [31]. Here, we report on the relationship between MaCFEM85 and the Medicago sativa wall-associated kinase MsWAK16. We analyzed the MaCFEM85 sequence and conducted experiments to determine which residues are critical for the interaction with MsWAK16. Finally, using N. benthamiana as our model plant, we evaluated plant defense responses to Botrytis cinerea and the aphid Myzus persicae with and without transient expression of MaCFEM85 and MsWAK16.
2. Results
2.1. MaCFEM85 Was Most Closely Related to Non-Pathogenic Fungal CFEMs and Localized to the Plasma Membrane
A phylogenetic tree was built to analyze the relationships between MaCFEM85 and other CFEM proteins from pathogenic and non-pathogenic model fungi. These included Magnaporthe oryzae, Fusarium oxysporum (Schl)., Neurospora crassa Shear & B.O.Dodge, and Beauveria bassiana (Bals.) (see Section 4). MaCFEM85 was most closely related to CFEM proteins in B. bassiana, and therefore, evolutionarily closer to non-pathogenic than to pathogenic fungi (Figure 1a).
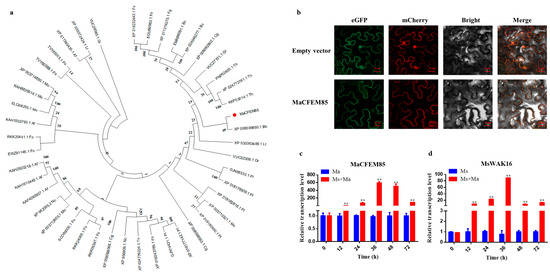
Figure 1.
Phylogenetic analysis, subcellular localization, and expression patterns of MaCFEM85 and MsWAK16. (a) Neighbor-joining phylogenetic tree showing the relationships between MaCFEM85 and CFEM proteins in other fungi. The phylogenic tree was constructed using MEGA X. Mo, Magnaporthe oryzae; Bc, Botrytis cinerea; Fg, Fusarium graminearum; Cg, Colletotrichum graminicola; Fo, Fusarium oxysporum; Nc, Neurospora crassa; Af, Aspergillus fumigatus; Lt, Lasiodiplodia theobromae; Gr, Gliocladium roseum; Th, Trichoderma harzianum; Bb, Beauveria bassiana; Pl, Paecilomyces lilacinus. (b) Subcellular localization of MaCFEM85 proteins. The MaCFEM85–GFP fusion genes were co-expressed with the MaCFEM85-mCherry fusion genes in N. benthamiana leaves. The control vector carried eGFP and mcherry driven by the 35S promotor. Photos were taken at 30 h after infiltration with laser scanning confocal microscopy of eGFP (488 nm excitation and 507 nm emission), mcherry (587 nm excitation and 610 nm emission), bright field microscopy, and merged confocal and bright field. (c,d) qRT-PCR analysis of MaCFEM85 and MsWAK16 expression in co-incubated M. anisopliae and M. sativa. Ma, M. anisopliae; Ms, M. sativa; Ma+Ms, co-incubated Ma and Ms. Error bars show the standard deviation of three biological replicates. ** p < 0.01 (one-way ANOVA with Duncan’s multiple-range t-test).
We conducted heterologous expression analysis in N. benthamiana to investigate the subcellular localization of MaCFEM85 at the cellular level. When the test leaves were infiltrated with MaCFEM85–eGFP and MaCFEM85-mcherry fusion protein, orange fluorescence was detected in the plasma membrane, that is contrast to eGFP and mCherry co-expression, which orange fluorescence was detected in both the plasma membrane and nuclei of leaves (Figure 1b). This suggests that MaCFEM85 plays a role in the cell membrane.
2.2. MaCFEM85 Was Upregulated during Interactions with M. sativa
Wall-associated receptor-like kinases (WAKs) represent a subgroup within the receptor-like protein kinases (RLKs) superfamily, and include an extracellular domain, a transmembrane helix, and an intracellular kinase domain. They play an important role in regulating plant growth, development, stress response and pathogen resistance signaling pathways [32]. RNA was extracted from M. anisopliae hyphae and M. sativa roots after co-incubation to investigate the expression patterns of MaCFEM85 and MsWAK16. Both MaCFEM85 and MsWAK16 were expressed at significantly higher levels during co-incubation from 12 to 60 h (Figure 1c,d). From 0 to 36 h, MaCFEM85 expression gradually increased, peaking at 36 h. It was × 593 times more highly expressed in the combination of M. anisopliae and M. sativa compared with M. anisopliae grown alone. Although MaCFEM85 expression was slightly decreased in the 48–60 h period, it was still expressed at significantly higher levels than in M. anisopliae grown alone. MsWAK16 was also significantly upregulated, with expression peaking at 36 h. In M. sativa infected with M. anisopliae, MsWAK16 was × 89.06 times more highly expressed than in M. sativa without M. anisopliae. From 36 to 60 h, levels of MsWAK16 decreased slightly, but its expression was still significantly higher than in the uninoculated plants.
2.3. MaCFEM85 Interacts with MsWAK16 In Vitro and In Vivo
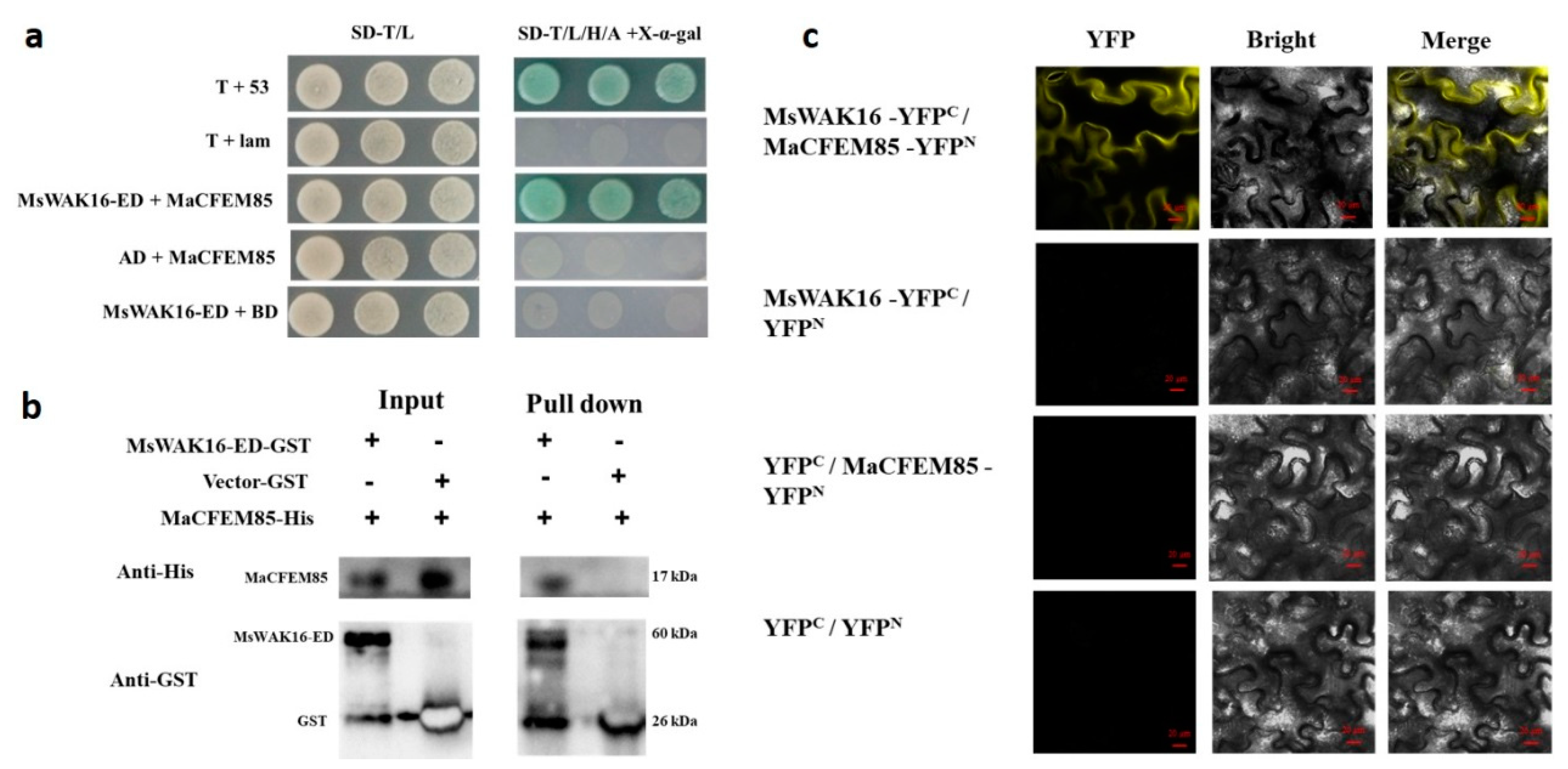
To investigate the potential functional mechanism of MaCFEM85 in response to M. anisopliae treatment, a yeast two-hybrid (Y2H) screen was performed to preliminarily identify host proteins that interact with MaCFEM85. The interaction between the extracellular domain of MsWAK16 (MsWAK16-ED) and MaCFEM85 was examined with a one-to-one yeast two-hybrid assay using MaCFEM85 in pGBKT7 as the BD vector and MsWAK16 in pGADT7 as the AD vector. All of the transformed yeast grew well on SD-T/L deficient medium, and the positive control group and experimental group grew successfully on SD-T/L/H/A + X-α-gal deficient medium. This indicated that MaCFEM85 could interact with MsWAK16-ED (Figure 2a).
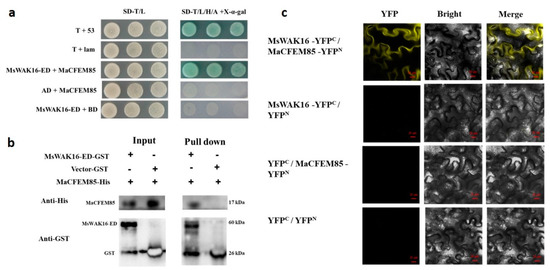
Figure 2.
Validation of the interaction between MaCFEM85 and MsWAK16. (a) Verification of the interaction between AD-MsWAK16-ED and BD-MaCFEM85 in Y2H Gold yeast. pGBKT7-53 and pGADT7-RecT (T+53) and pGBKT7-lam and pGADT7-RecT (T+Lam) were used as the positive and negative controls, respectively. (b) In vitro GST pull-down assays showing the interaction between MaCFEM85 and MsWAK16-ED. (c) BiFC assays showed that MaCFEM85 and MsWAK16 interacted when co-expressed in N. benthamiana leaf cells. Photos were taken at 30 h after infiltration with laser scanning confocal microscopy of yellow fluorescent protein (YFP) (514 nm excitation and 527 nm emission), bright field microscopy, and merged confocal and bright field. Scale bar = 20 µm. YFPN and YFPC represent pUC-SPYNE and pUC-SPYCE vectors, respectively.
For in vitro validation, glutathione-S-transferase (GST)-tagged MsWAK16-ED (encoding a product of 60 kDa) was inserted into pGEX6-P2, and polyhistidine (His)-tagged MaCFEM85 (encoding a product of 17 kDa) was inserted into pET-21b. Immunoblotting with His and GST antibodies showed that the recombinant protein MaCFEM85-His interacted with the GST-MsWAK16-ED prey but not with GST alone, and that GST-MsWAK16-ED interacted with His-MaCFEM85 (Figure 2b).
To further confirm the interaction between MaCFEM85 and MsWAK16, we performed a bimolecular fluorescence complementation (BiFC) assay with MaCFEM85-YFPN and MsWAK16-YFPC constructs. Co-expression of MaCFEM85-YFPN and MsWAK16-YFPC in tobacco leaves generated a yellow fluorescence signal, indicating that MaCFEM85 interacted with MsWAK16 (Figure 2c).
2.4. Key Sites for MaCFEM85 and MsWAK16 Interactions
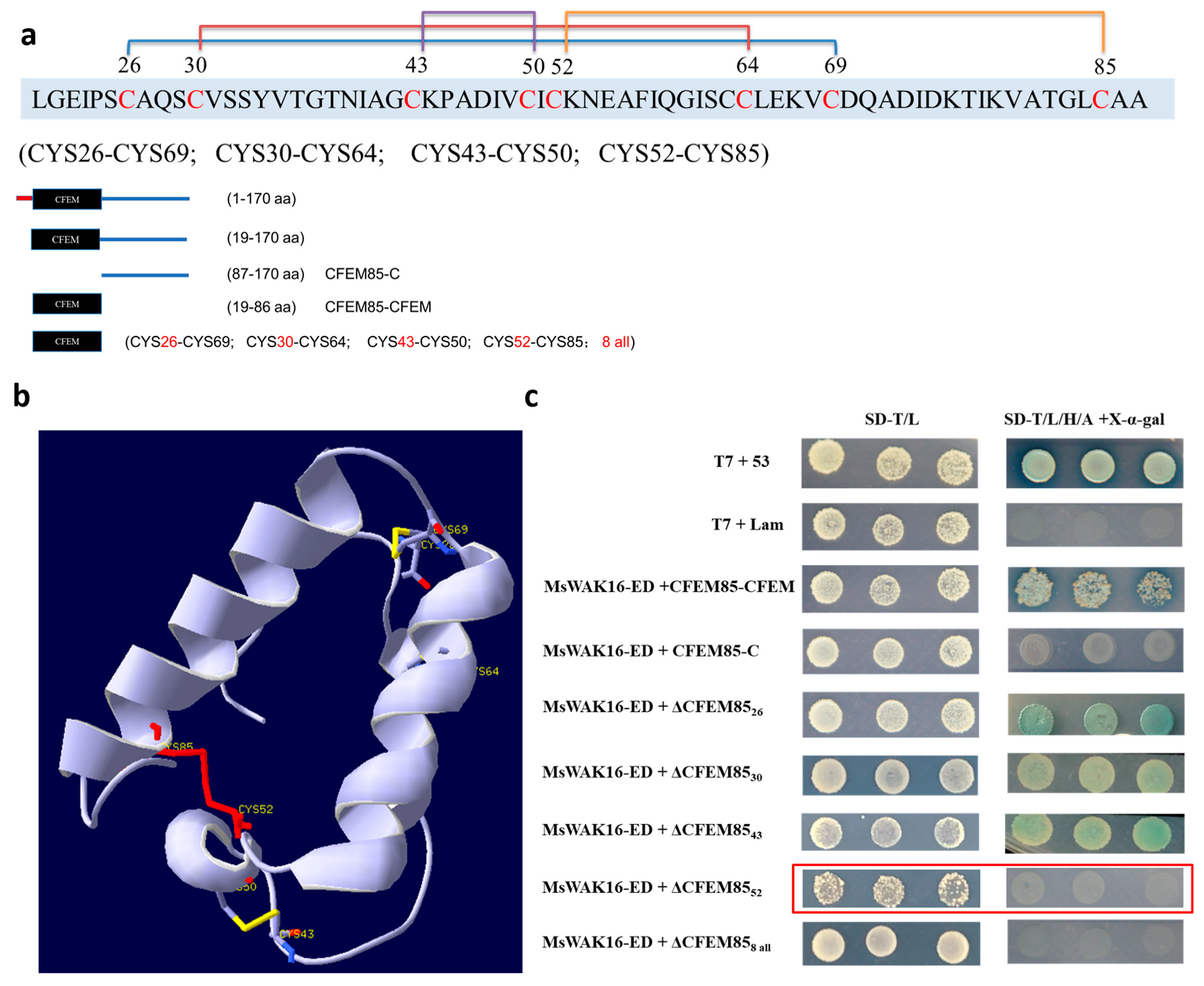
To define the region of MaCFEM85 that is required for the interaction with MsWAK16-ED, protein domains were predicted with the online software SMART (Figure 3a). MaCFEM85 contained a CFEM domain the 19-86 aa region. The tertiary structure was also predicted (Figure 3b). The eight cysteines in this domain resulted in four disulfide bonds (CYS26 and CYS69, CYS30 and CYS64, CYS43 and CYS50, and CYS52 and CYS85), maintaining stability of the protein (Figure 3b). To validate the putative interaction site(s) in MaCFEM85, seven mutated versions of the protein were generated and inserted into pGBKT7 as bait proteins. Each recombinant vector was co-transfected with AD-MsWAK16-ED in the Y2H Gold strain. The strain with CYS52 mutated did not grow on selective media (Figure 3c, outlined in red), indicating that CYS52 is required for the interaction of MaCFEM85 with MsWAK16-ED.
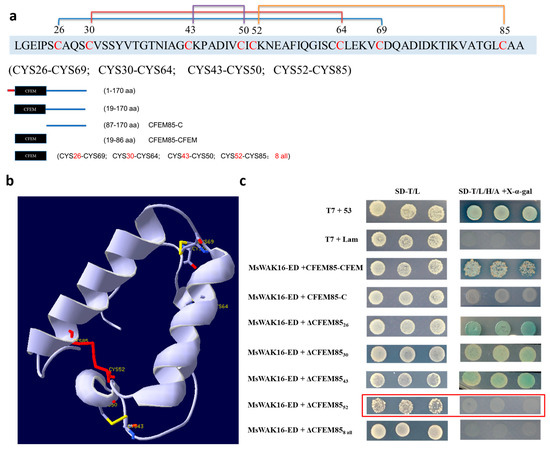
Figure 3.
Identification of the interaction site between MaCFEM5 and MsWAK16. (a) Schematic illustrations of MaCFEM85. Protein domains in MaCFEM85 were predicted and visualized using SMART (embl-heidelberg.de). (b) Positions of the eight cysteines in MaCFEM85 and a schematic showing the four disulfide bonds. (c) Truncated MaCFEM85 constructs were generated that included the CFEM domain (19–86 aa of the N-terminal, CFEM85-CFEM) and that included residues 87–170 without the CFEM domain (CFEM85-C). The other five variants were C26A(ΔCFEM8526), C30A(ΔCFEM8530), C43A(ΔCFEM8543), C52A(ΔCFEM8552), and C26/30/43/50/52/64/69/85A (ΔCFEM858 all). Each construct was co-transformed in yeast with MsWAK16-ED and grown on either the two-deficiency medium SD-T/L (SD-Trp/Leu) or the four-deficiency medium SD-T/L/H/A + X-α-gal (SD-Trp/Leu/His/Ade + X-α-gal).
2.5. The Interaction of MaCFEM85 with MsWAK16 Activates the Plant Immune Response
2.5.1. Evaluating the Role of MaCFEM85 and MsWAK16 in Disease Resistance against B. cinerea
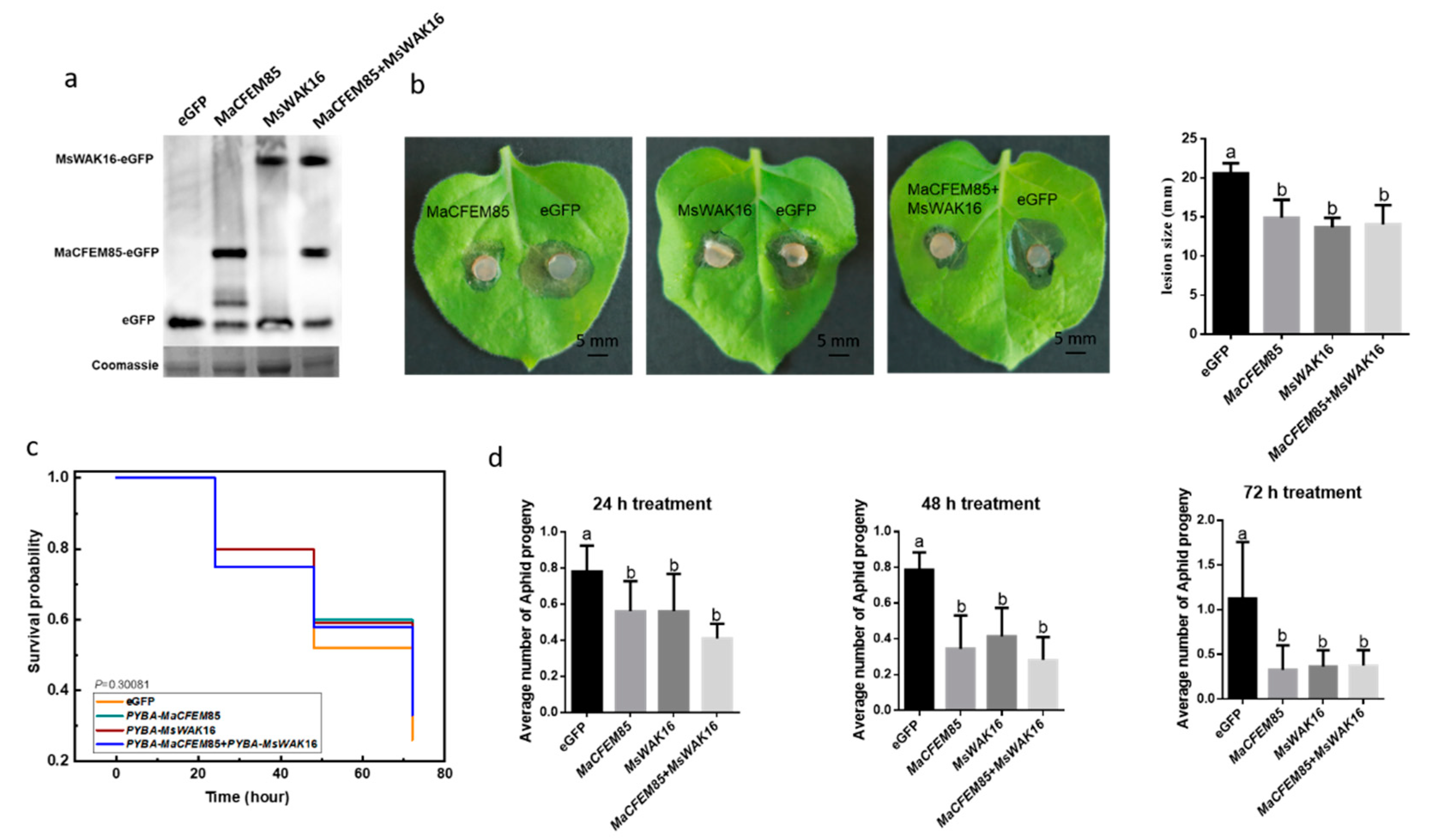
To explore the possible involvement of MaCFEM85 and MsWAK16 in pathogen defense responses, we examined whether overexpression of MaCFEM85, MsWAK16, or MaCFEM85 and MsWAK16 could confer increased resistance to B. cinerea. We transiently expressed these vectors in N. benthamiana leaves. A Western blot assay showed that MaCFEM85 and MsWAK16 were expressed at comparable levels when they were expressed alone or together, which shows that MaCFEM85 and MsWAK16 were successfully expressed in tobacco (Figure 4a). Disease assays were also performed using N. benthamiana infiltrated with MaCFEM85, MsWAK16, MaCFEM85+MsWAK16, or the eGFP control. Lesions were significantly smaller (by ~30% at 2 d after inoculation) on leaves infiltrated with MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16 compared with the control plants (Figure 4b). These data demonstrate that transient expression of MaCFEM85 in N. benthamiana conferred increased resistance to B. cinerea and that MaCFEM85 and MsWAK16 positively regulated the defense response against B. cinerea.
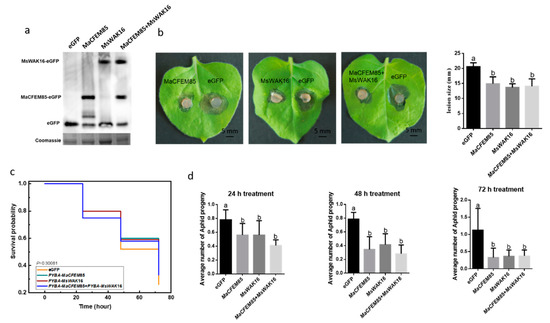
Figure 4.
Induction of plant resistance by transient expression of MaCFEM85 and MsWAK16. (a) Transient expression of MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16 in N. benthamiana leaves. Protein expression level of MaCFEM85 and MsWAK16. Leaf samples were harvested 12 h after infiltration and total soluble protein extracts were prepared. Proteins were separated by SDS–PAGE and analyzed via immunoblot using a GFP-specific antibody. Total protein content (showing equal loading) was measured with Coomassie staining. (b) N. benthamiana leaf lesions resulting from B. cinerea infection. Lesion sizes were measured at 2 d after inoculation in a minimum of nine leaves for each treatment group. Data presented in (b) is the mean ± standard error, and Letters indicate statistical significance between groups at p < 0.05. (c) The mortality risk of aphids feeding on N. benthamiana transiently expressing MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16. (d) The average number of progeny from each aphid after feeding on tobacco transiently expressing GFP, MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16 for 24, 48, and 72 h, respectively. Values are shown as the mean ± standard error (n = 20).
2.5.2. Evaluating the Role of MaCFEM85 and MsWAK16 in Aphid Defense in N. benthamiana
To further investigate the role of MaCFEM85 and MsWAK16, N. benthamiana plants transiently expressing MaCFEM85 and MsWAK16 were infested with M. persicae and the populations were assessed. For this experiment, a large area of each N. benthamiana leaf was agroinfiltrated with the recombinant binary vector pYBA1132 containing MaCFEM85 and MsWAK16. GFP was used as the control. At 12 h after infiltration, 20 M. persicae adults were caged on each leaf, exposing the infiltrated area to aphids. On the following three days, adult aphid mortality and the number of nymphs were recorded; the nymphs were then removed. There was no significant difference in the mortality risk amongst M. persicae populations feeding on plants expressing MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16 (χ2 = 3.65827, DF = 3, p < 0.30081) (Figure 4c). However, at 24, 48, and 72 h, the average number of progeny produced by each adult aphid was significantly higher on N. benthamiana expressing the GFP control compared with those expressing MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16 (Figure 4d).
2.5.3. Evaluating the Role of MaCFEM85 and MsWAK16 in Hormone Accumulation and Hormone-Related Gene Expression
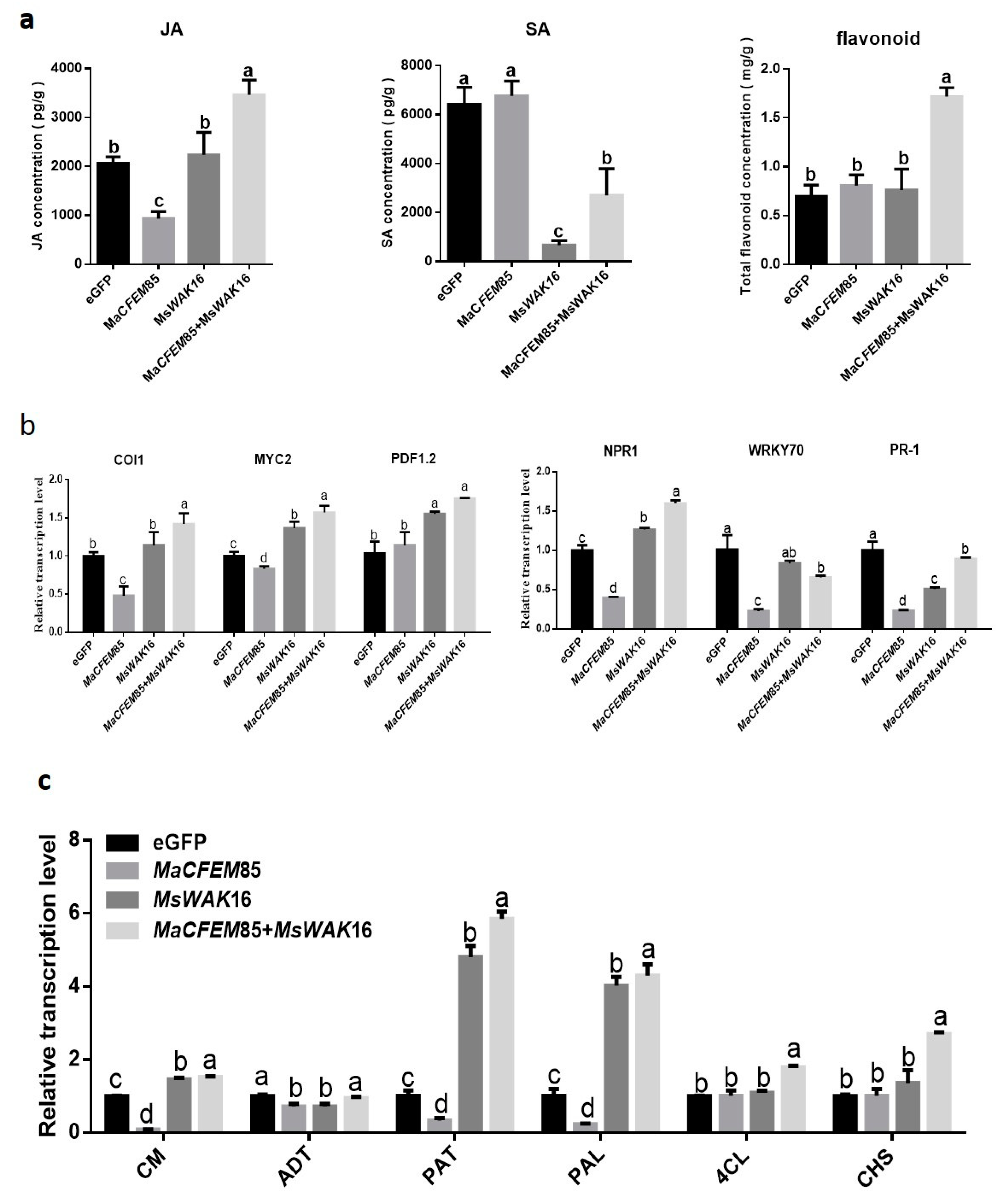
To analyze the differences between the defense responses of N. benthamiana plants expressing eGFP, MaCFEM85, MsWAK16, and MaCFEM85+MsWAK16, we measured JA, SA, and total flavonoid levels, and the expression of related genes. The results showed that JA and SA levels differed between plants expressing eGFP, MaCFEM85, MsWAK16, and MaCFEM85+MsWAK16 (Figure 5a). Compared with those expressing eGFP, JA levels were significantly lower in plants expressing MaCFEM85; SA levels were slightly increased, but the differences were not significant. In contrast, plants expressing MsWAK16 showed no significant differences in JA levels compared with the eGFP control, whereas SA levels were significantly lower than in the control (Figure 5a). JA and SA levels were significantly increased and decreased, respectively, in plants expressing MaCFEM85+MsWAK16 compared with eGFP.
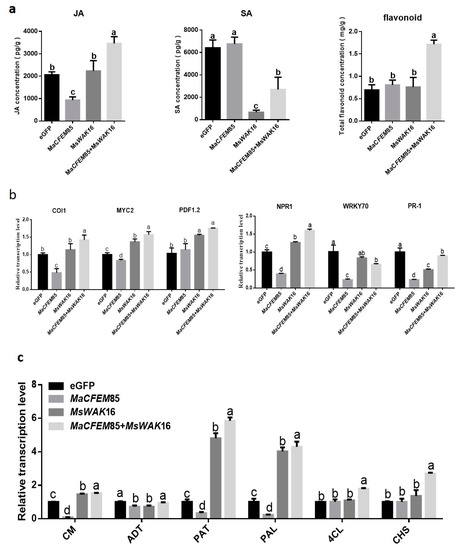
Figure 5.
Hormone levels and hormone response gene expression. (a) Levels of jasmonic acid (JA), salicylic acid (SA), and total flavonoids in N. benthamiana after transient expression of eGFP, MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16 for 12 h. (b,c) Genes related to plant defense responses were measured in N. benthamiana leaves at 12 h after infiltration with Agrobacterium containing plasmids encoding eGFP, MaCFEM85, MsWAK16, or MaCFEM85+MsWAK16. Data are presented as the mean ± standard deviation from three biological replicates. Letters indicate statistical significance between groups at p < 0.05. Abbreviations: Chorismate mutase, CM; Prephenate dehydratase, ADT; Aspartate-prephenate aminotransferase, PAT; phenylalanineammonialyase, PAL; 4-coumarate coenzyme A ligase, 4CL; Chalcone synthase, CHS.
Total flavonoid content was significantly higher in plants expressing MaCFEM85+MsWAK16 compared with all other treatment groups (Figure 5a). The biosynthetic gene expression levels were similar in plants expressing MsWAK16 and MaCFEM85+MsWAK16. For example, genes related to the JA response, namely COI1, MYC2, and PDF1.2, were significantly upregulated compared with plants expressing eGFP (Figure 5b). Similarly, the SA-related genes NPR1, WRKY70, and PR1 were significantly differentially expressed in plants expressing MsWAK16 and MaCFEM85+MsWAK16; NPR1 was upregulated, whereas WRKY70 and PR1 were downregulated compared with the control (Figure 5b).
We also examined the expression of key genes in the Shikimic acid and phenylpropanoid synthesis pathways, which are both related to flavonoid synthesis. In general, expression of MaCFEM85 alone did not induce significantly higher expression of these genes in tobacco. However, these genes were upregulated in tobacco expressing MsWAK16 or MsWAK16+MaCFEM85 compared with the eGFP control (Figure 5c).
3. Discussion
3.1. The Conserved CFEM Protein Motif Serves Multiple Functions in Fungal Species
The CFEM domain is unique to fungi and commonly occurs in fungal extracellular membrane proteins. The domain originated in the most recent common ancestor of Ascomycota and Basidiomycota [33]. Recent research has shown that CFEM proteins in fungal pathogens can act as plant immune regulators, causing host plant immune suppression or activation depending on the type of infection [28,34,35]. There have been few reports of CFEM domain-containing proteins in M. anisopliae, only in the context of an evolutionary comparison with other fungi [36]. In this study, we compared M. anisopliae CFEM proteins with other known CFEM proteins in both pathogenic and non-pathogenic fungi. We confirmed that the closest homolog of MaCFEM85 is Cfem5 found in Beauveria bassiana, one of 12 CFEM proteins in that species (Supplementary Figure S1). BbCfem5 is essential for iron acquisition [37]. Moreover, based on the predicted tertiary structure, the CFEM domain in MaCFEM85 is very similar to Surface Antigen Protein 2 (CSA2) found in Candida albicans (C.P.Robin) Berkhout, in which 65 residues (96% of the sequence) have been modelled with 99.8% confidence [31]. Candida albicans is an animal pathogen; CSA2 plays an important role in growth and pathogenicity by extracting heme from hemoglobin and transporting heme from the cell wall to the plasma [38,39,40]. We hypothesis that MaCFEM85 may be involved in M. anisopliae virulence of insect hosts. These combined functions of MaCFEM85 in animal pathogenic infection and in activation of plant immunity make the protein an exciting future research focus.
3.2. Disulfide Bonds Are Important Structures for Protein Function
The conformational integrity of a protein structure is directly related to its ability to function. The disulfide bonds formed by cysteine pairs promote protein folding and conformational stability, and are considered to form key sites for recognition and binding of specific receptors or ligands [41]. For example, disrupting any one of the three conserved disulfide bonds in the plant pathogen Cladosporium fulvum effector protein AVR4 results in protease sensitivity and reduced chitin binding ability [42]. In three other C. fulvum proteins (ECP1, ECP2, and ECP5), single substitutions of alanines for cysteines dampen the tomato hypersensitive response, indicating that the cysteines are critical to maintaining stability and hypersensitive response-inducing activity [43]. Moreover, in the mature protein MC69 of Magnaporthe oryzae, mutagenesis of two conserved cysteine residues (Cys36 and Cys46) may impair MC69 function without affecting secretion, suggesting the importance of the disulfide bond specifically in the pathogenicity of MC69 [44]. The CFEM domain appears to be similar in size and in the pattern of cysteine residues to EGF-like domains, which contain three or four pairs of disulfide bonds. EGF proteins can function as cell-surface receptors, signal transducers, or adhesion molecules in host–pathogen interactions [45]. In this study, we not only found that the CFEM domain of MaCFEM85 contained eight conserved cysteines that formed four pairs of disulfide bonds (Figure 3b), but we also validated that CFEM was the critical domain for the interaction between MaCFEM85 and MsWAK16. Furthermore, through site-directed mutation of cysteine residues to alanine, we found that the cysteine residue at position 52 was the core site required for the interaction (Figure 3c). These results provide a basis for further studies of the physiological functions of the CFEM85–WAK16 interaction.
3.3. The Interaction of MaCFEM85 with MsWAK16 Activated Plant Defenses
In microbial–plant interactions, plant defense responses are generally activated by microbial effector proteins. Induced defense is becoming an important tool in biological pest control to promote resistance. CFEM proteins have been identified as effectors involved in the regulation of plant immune activation or inhibition [28,46]. However, there is a paucity of published research linking the regulation of these immune factors to their specific roles in plant disease and insect resistance. In this study, we used an entomopathogenic and endophytic fungus, M. anisopliae, to identify an interaction between MaCFEM85 and a cell wall-associated kinase, MsWAK16, in M. sativa. The results showed that this interaction reduced the lesion ratio after inoculation with B. cinerea (Figure 4b) and decreased the population growth rate of M. persicae (Figure 4d), indicating the interaction between MaCFEM85 and MsWAK16 increased M. sativa resistance to aphids.
Changes in the levels of JA, SA, and ethylene (ET) can be used as markers to evaluate the induction of plant resistance [47,48]. Here, we demonstrated that MaCFEM85 interacted with MsWAK16 to influence plant resistance through hormonal regulation and to inhibit the reproductive rate of M. persicae (Figure 4d). Similar studies have been reported previously. For example, the Brevibacillus laterosporus effector protein PeBL1 was shown to induce JA and SA accumulation in tomato and to decrease the growth rate of the second and third generation populations of M. persicae that fed on those plants. Moreover, tomato plants sprayed with effectors have a repellent effect on M. persicae [49]. Application of the elicitor protein PeBC1 to common bean (Phaseolus vulgaris L.) leads to pronounced and significant sub-lethal effects on green peach aphids. Plants treated with PeBC1 show significant upregulation of genes related to JA and SA [50]. The Beauveria bassiana elicitor PeBb1 reduces the fecundity of M. persicae on tobacco and induces the expression of JA- and ET-related genes [51]. In the present study, transient expression of MaCFEM85 and MsWAK16 in tobacco significantly upregulated the JA response-related genes COI1, MYC2, and PDF1.2 (Figure 5b) and increased JA and total flavonoid content (Figure 5a), which was comparable to results in the literature as described above. However, we did not detect high levels of SA in tobacco 12 h after infiltration, and plants transiently expressing MsWAK16 or MaCFEM85+MsWAK16 showed significantly reduced SA levels and downregulation of genes involved in the SA response (Figure 5a,b). This demonstrated that stimulation of different plant hormones can lead not only to synergistic activities, but also to crosstalk and feedback, allowing plants to respond appropriately to different stimuli. Increased JA levels but low SA levels have been previously reported in plants. For instance, in A. thaliana defective for SA accumulation, JA levels were 25-fold higher than in wild type A. thaliana, and JA-responsive genes were activated [52]. It has also been reported that some bacteria can increase JA levels in plants to inhibit SA accumulation, avoiding the harm caused by SA. For example, Pseudomonas syringae targets the COI1 receptor through a toxin, coronatine, that negatively regulates the JA pathway [53]. This promotes MYC2-induced upregulation of the transcription factors ANAC019, ANAC055, and ANAC072 [54], inhibiting transcription of ICS1 (a key gene for SA synthesis) and thus downregulating SA production and signal transduction. In the present study, the interaction between MaCFEM85 and MsWAK16 led to increased JA accumulation and upregulation of JA-responsive genes but inhibition of SA accumulation and transcription of SA-related genes. This indicated that the interaction between MaCFEM85 and MsWAK16 induced JA, thereby improving plant resistance to aphids.
In N. benthamiana transiently expressing MaCFEM85 and MsWAK16, JA levels were increased and B. cinerea lesion sizes were decreased (Figure 4b), consistent with previous studies. JA is involved in plant resistance to insects and necrotrophic pathogens [52,55]. As a necrotrophic pathogen, B. cinerea was inhibited by JA and ET accumulation [56]. In the A. thaliana ET-deficient mutant ein2-1 and the JA-response mutant coi1-1, levels of the downstream defense gene PDF1.2 were significantly reduced and sensitivity to B. cinerea was enhanced [57]. We found here that JA accumulation and transcription of JA-related genes were significantly increased in N. benthamiana transiently expressing MaCFEM85 and MsWAK16, whereas the accumulation of SA and transcription of SA-related genes were inhibited. MaCFEM85 and MsWAK16 expression also showed an inhibitory effect on B. cinerea. This indicated that JA was activated by MaCFEM85 and MsWAK16 and played a leading role in plant resistance to B. cinerea.
4. Materials and Methods
4.1. Fungal Strains, Plant Materials, and Culturing Methods
Metarhizium anisopliae isolate strains Ma 9 were cultured on potato-sugar-agar (PSA) medium (200 g peeled potato extract boiled in water, 20 g sucrose, and 15 g agar/L). Fresh sporangium powder was collected after 10 d. Nicotiana benthamiana plants were grown in an artificial climate chamber with a 14/10 h light/dark cycle (27/25 °C). For transient gene expression via Agrobacterium-mediated transformation, Agrobacterium tumefaciens strain GV3101 was cultured in Luria Broth (LB) medium (10 g tryptone, 5 g yeast extract, and 10 g NaCl/L). Yeast strain Gold (OE biotech Co., Ltd., Shanghai, China) was cultured on yeast extract peptone dextrose (YPDA) medium (10 g yeast extract, 20 g peptone, 20 g glucose, and 0.03 g adenine hemisulfate/L). For each vector and strain, appropriate antibiotics were used, namely rifampin, kanamycin, or ampicillin (25, 50, or 50 µg/mL, respectively). The strains and plasmids used in this study are listed in Supplementary Table S1.
Medicago sativa seeds were surface sterilized with 75% ethanol for 1 min, followed by 50% NaClO (5.5%) for 15 min. The seeds were thoroughly mixed, then washed in sterile water three times for 5 min each. The seeds were incubated at 4 °C in the dark for over 24 h, then germinated on 1% water agar plates at room temperature overnight. Three days after gemination, the developing seedlings were transferred to filter paper on 9-cm sterile petri dishes, with 20 seedlings per dish. The treatments and control were allocated to 15 dishes each, respectively. The seedlings were then irrigated with 10 mL of M. anisopliae spore suspension containing 107/mL, and the control was irrigated with 10 mL sterile water. At 0, 12, 24, 48, and 60 h, a random selection of seedlings was taken from three petri dishes for each time interval. The samples were frozen in liquid nitrogen and stored at −80 °C.
4.2. qRT-PCR and Plasmid Construction
Total RNA was extracted from M. anisopliae (hyphae) and M. sativa (roots) with TRIzol reagent (Invitrogen, CA, USA) following the manufacturer’s instructions. The quality and abundance of the resulting RNA were measured with a NanoPhotometer® (Implen, Münich, Germany). First-strand cDNA synthesis (up to 2ug RNA) was performed using 5× All-In-One RT Master Mix (Applied Biological Materials Inc., Vancouver, BC, Canada) following the manufacturer’s instructions.
qRT-PCR was performed using 2×SYBR Green qPCR Master Mix from US EVERBRIGHT ® INC. and the ABI QuantStudio 5 system following the manufacturer’s instructions. The primers used for each gene are listed in Supplementary Table S2. MaTry [58], Msactin [59], and Nbactin [60] were used as the internal reference genes for normalization of expression data. The reaction was performed under the following conditions: 5 min at 95 °C, followed by 40 cycles at 95 °C for 15 s and at 60 °C for 40 s. There were three technical replicates for each sample. Relative expression was calculated using the 2−ΔΔCt method [61]. Statistical significance was determined using one-way analysis of variance (ANOVA) and Duncan’s multiple-range test in SPSS v20.0 (SPSS).
4.3. Transient Expression of Proteins in N. benthamiana
MaCFEM85 coding sequence (CDS) was amplified with ultra-fidelity DNA polymerase using cDNA as the template.
For the subcellular localization assays, PCR products containing the CDS for MaCFEM85 were cloned into pYBA1132-eGFP (digested with EcoRI and SalI) and pcambia1300-mcherry (EcoRI and SacI), respectively. All the constructs were validated with sequencing (Tsingke Biotechnology Co., Ltd. Beijing, China). The primers used in this study are listed in Supplementary Table S2.
For transient expression of MaCFEM85-eGFP and MaCFEM85-mCherry in N. benthamiana, A. tumefaciens strain GV3101 was transformed with each plasmid and verified with PCR. The Agrobacterium was cultured overnight at 28 °C with shaking. The cells were collected via 5 min 5000 rpm centrifugation at room temperature, washed three times with sterile double-distilled water, and resuspended in 10 mM MgCl2 buffer (containing 10 mM MES and 10 mM acetosyringone, pH 5.7). The cell suspension was adjusted to an OD600 of 0.5, then infiltrated into the underside of 4- to 5-week-old N. benthamiana leaves with a 1-mL syringe. Each leaf was infiltrated with 50 µL A. tumefaciens; three leaves were treated per plant and three plants were in each treatment group. The treated leaves were collected after 30 h and visualized with a Zeiss LSM980 confocal microscope (Carl Zeiss, Germany) to determine the sub-cellular localization.
4.4. Phylogenetic Analysis
A phylogenetic tree was constructed using the neighbor-joining method in MEGA X. There were 1000 bootstrap replicates using the P-distance model. The amino acid sequences used to generate the tree were obtained with BLASTP searches against the NCBI database of related fungi (e.g., Magnaporthe oryzae, Botrytis cinerea, Fusarium graminearum, Colletotrichum graminicola, Fusarium oxysporum, Neurospora crassa, Aspergillus fumigatus, Lasiodiplodia theobromae, Gliocladium roseum, Trichoderma harzianum, Beauveria bassiana, and Paecilomyces lilacinus) using MaCFEM85 as the query.
4.5. Yeast Two-Hybrid Assays
The Matchmaker Gold Yeast Two-Hybrid System (Clontech Laboratories, Inc.; now Takara Bio USA, Inc.) was used to verify the interaction between MaCFEM85 and MsWAK16. MaCFEM85 without the signal peptide was introduced into pGBKT7 as the bait and the extracellular domain of MsWAK16 (MsWAK16-ED) was inserted into pGADT7 as the prey. Yeast competent cell preparation and transformations were performed using a Frozen-EZ Yeast Transformation II Kit™ (ZYMO Research, CA, USA) following the manufacturer’s instructions. The bait and prey plasmids were co-transformed into the yeast strain Gold. Protein–protein interactions were analyzed based on growth on SD double dropout medium (DDO, SD/-Trp-Leu) and SD quadruple dropout medium (QDO, SD/-Trp-Leu-His-Ade) plates.
4.6. BiFC Assay
To generate the BiFC constructs, the pUC-SPYNE and pUC-SPYCE vectors were linearized by digestion with BamHI. The full-length CDSs of MaCFEM85 and MsWAK16 were each cloned and inserted into the linearized plasmids using a recombinant enzyme to obtain the MaCFEM85-YFPN and MsWAK16-YFPC constructs. The plasmids containing MaCFEM85 and MsWAK16 were co-transformed with empty vectors as negative controls (MaCFEM85-YFPN + pUC-SPYCE, MsWAK16-YFPC + pUC-SPYNE, and pUC-SPYNE + pUC-SPYCE). All the vectors were introduced into N. benthamiana via Agrobacterium-mediated transformation as described above. Fluorescence signals were observed in leaf epidermal cells using a Zeiss LSM980 confocal microscope (Carl Zeiss, Germany). The primers used for vector construction are listed in Supplementary Table S2.
4.7. GST Pull-Down Assay
For the GST pull-down assays, MsWAK16-ED was inserted into the pGEX-6P-2 vector and MaCFEM85 was inserted into pET-21b. The purified fusion proteins (GST- MsWAK16-ED) and the pGEX-6P-2 no-load protein (GST) were used as the bait protein and purified pET-21b-MaCFEM85 fusion protein (His-MaCFEM85) was used as the prey. GST pull-down assays were performed with the Mag-Beads GST Fusion protein purification system (Sangon Biotech, Shanghai, China) following the manufacturer’s instructions. Briefly, Mag-Beads were washed five times with 1× phosphate-buffered saline (PBS, pH 7.4) to release the alcohol protector, and 10 mL of GST or GST-MsWAK16-ED was then added. The beads were mixed by inversion at room temperature for 30 min. After removing the supernatant, the Mag-Beads were washed five times with 1× PBS. His-MaCFEM85 was added to Mag-Beads already bound with GST, and the mixture was incubated overnight at 4 °C with rotation. The beads were then washed five times with 1× PBS to remove unbound proteins. Subsequently, proteins immobilized on the beads were separated by SDS-PAGE, transferred to a nitrocellulose membrane (100 V, 1 h) and analyzed via Western blot. The membranes were washed three times for 10 min each with PBS + Tween (PBST). The membranes were blocked for 2 h at room temperature with 5% skimmed milk, then incubated with ProteinFind anti-His mouse monoclonal antibody (TransGen Biotech, Beijing, China) (diluted 1:3000) or ProteinFind anti-GST mouse monoclonal antibody for 2 h at 4 °C. The membranes were then immersed in ProteinFind goat anti-rabbit IgG(H+L) (HRP) antibody (TransGen Biotech, Beijing, China) (diluted 1:5000) for 1 h at room temperature. The membranes were visualized with the EasySee® Western Blot Kit (TransGen Biotech, Beijing, China) following the manufacturer’s instructions.
4.8. Identification of the Key Site in MaCFEM85
To determine the site required for the interaction of MaCFEM85 with MsWAK16, PCR amplification was performed to generate multiple truncated forms of MaCFEM85. The CFEM domain (MaCFEM85-CFEM; aa residues 19–86) and the C terminal without the CFEM domain (MaCFEM85-C, aa residues 87–170) were inserted into the vector pGBKT7 as the bait protein (Supplementary Table S1). Another five variants were constructed using polypeptide synthesis to mutate cysteine to alanine at positions 26, 30, 43, 52, and 26/30/43/50/52/64/69/85 (ΔCFEM8526, ΔCFEM8530, ΔCFEM8543, ΔCFEM8552, and ΔCFEM858 all, respectively). These variants were used to perform Y2H experiments with MsWAK16-ED. The transformed yeast cells were assayed for growth on synthetic dropout SD/-Trp-Leu plates and SD/-Trp-Leu-His-Ade plates containing X-α-Galactosidase (X-α-Gal).
4.9. Plant Resistance Assays
Myzus persicae were obtained from Henan Quanying Biological Co., Ltd. One week before the start of bioassays, 150 adult aphids were placed on three N. benthamiana plants (50 aphids per plant). After 72 h, all the adults were removed with a soft artist’s brush, and the nymphs were allowed to feed for an additional four days before being transferred to N. benthamiana for the aphid performance assays.
4.10. Aphid Performance Assays
Nicotiana benthamiana Domin. (Solanales: Solanaceae) was used to assess M. persicae performance, plant disease resistance against B. cinerea, and expression of hormone-related genes. Agrobacterium carrying either pYBA-eGFP, pYBA-MaCFEM85, pYBA-MsWAK16, or pYBA-MaCFEM85+pYBA-MsWAK16 were grown in LB supplemented with appropriate antibiotics for 36 h at 28 °C. The cells were washed three times, then resuspended in infiltration buffer (10 mM MgCl2, 10 mM MES, and 100 µM acetosyringone, pH 5.6) to an OD600 of 0.6. For co-infection, bacteria carrying MaCFEM85 and MsWAK16 were adjusted to an OD600 of 1.2, then mixed in equal volumes. Fully expanded leaves were infiltrated with bacteria using 1-mL needleless syringes. Three leaves were infiltrated on each plant and each treatment was applied to three plants for a total of 12 plants per experiment.
For the aphid performance assays, 20 adult aphids were applied to the infiltrated area of each leaf 12 h after the Agrobacterium was applied. The aphids were confined using 5 mm diameter clip cages. Each treatment was repeated five times. Adult aphid mortality and the number of newly laid nymphs were recorded daily for three days.
B. cinerea was cultured for five days on PDAY. At 12 h after Agrobacterium infiltration, the newly cultivated B. cinerea was punched into a 5-mm fungus cake, and the mycelium growth surface was attached to the infiltration area on the leaf surface. To facilitate disease development, the plants were kept humid by covering with plastic film in trays at 22 °C to facilitate disease progression. After 48 h, disease progress was estimated in inoculated leaves by measuring lesion sizes.
For quantifying the relative expression of relevant genes, the three infiltrated leaves were collected from each plant 12 h after infiltration, then frozen in liquid nitrogen and stored at −80 ℃. Total RNA extraction, cDNA synthesis, and qRT-PCR were conducted as described in Section 3.2.
Salicylic acid (SA), jasmonic acid (JA), and flavonoid levels were measured in 15 infiltrated N. benthamiana leaves per treatment. The leaves were collected, frozen in liquid nitrogen, then stored at −80 ℃. Plant hormones were quantified using the Plant Salicylic Acid and Plant Jasmonic Acid ELISA Kits (Beijing WeLab Scientific Co., Ltd.), and flavonoids were quantified using the Micro Plant Flavonoids Assay Kit (Beijing Solarbio Science & Technology Co., Ltd.) following the manufacturer’s instructions.
4.11. Statistical Analysis
Data were analyzed in SPSS v20.0. Significant differences in expression levels of MaCFEM85 and MsWAK16 were determined using Student’s t-test. Significant differences in SA, JA, and flavonoid levels were determined using one-way ANOVA and Duncan’s multiple-range test with a threshold of p < 0.05.
5. Conclusions
In this study, we identified and characterized a novel secreted protein, MaCFEM85, in M. anisopliae. It was found to be a conserved effector that can interact with MsWAK16 and activate the N. benthamiana defense response. We showed that the CFEM domain and the cysteine residue at position 52 in MaCFEM85 were critical for the interaction. This interaction may activate JA-related immune responses and disease-resistant and insect-resistant mechanisms in the plant.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/ijms24044037/s1.
Author Contributions
Conceptualization, N.C. and X.N.; methodology, N.C. and R.L.; software, N.C.; validation, N.C. and R.L.; formal analysis, X.N.; investigation, R.L.; resources, N.C.; data curation, N.C.; writing—original draft preparation, N.C.; writing—review and editing, N.C., M.R.M. and X.N.; visualization, G.W. and Z.Z.; supervision, X.T. and X.N.; project administration, X.T.; funding acquisition, X.T. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by China Agriculture Research System of MOF and MARA grant number CARS-34-07.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available in this article and its Supplementary Materials.
Acknowledgments
This work was supported by the China Agriculture Research System of MOF and MARA (CARS-34-07).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Paulucci, N.S.; Anta, G.G.; Gallarato, L.A.; Vicario, J.C.; Cesari, A.B.; Reguera, Y.B.; Kilmurray, C.; Bueno, M.A.; García, M.B.; Dardanelli, M.S. Plant–microbe partnerships: Implications for growth and plant health. In Plant Microbe Symbiosis: Fundamentals and Advances; Springer: Berlin/Heidelberg, Germany, 2013; pp. 105–117. [Google Scholar] [CrossRef]
- Schenk, P.M.; Carvalhais, L.C.; Kazan, K. Unraveling plant–microbe interactions: Can multi-species transcriptomics help? Trends Biotechnol. 2012, 30, 177–184. [Google Scholar] [CrossRef]
- Sasse, J.; Martinoia, E.; Northen, T. Feed Your Friends: Do Plant Exudates Shape the Root Microbiome? Trends Plant Sci. 2018, 23, 25–41. [Google Scholar] [CrossRef]
- Lugtenberg, B.; Kamilova, F. Plant-growth-promoting rhizobacteria. Annu. Rev. Microbiol. 2009, 63, 541–556. [Google Scholar] [CrossRef]
- Shoresh, M.; Harman, G.E.; Mastouri, F. Induced systemic resistance and plant responses to fungal biocontrol agents. Annu. Rev. Phytopathol. 2010, 48, 21–43. [Google Scholar] [CrossRef]
- van de Mortel, J.E.; de Vos, R.C.; Dekkers, E.; Pineda, A.; Guillod, L.; Bouwmeester, K.J.; van Loon, J.; Dicke, M.; Raaijmakers, J.M. Metabolic and transcriptomic changes induced in Arabidopsis by the rhizobacterium Pseudomonas fluorescens SS101. Plant Physiol. 2012, 160, 2173–2188. [Google Scholar] [CrossRef]
- Lareen, A.; Burton, F.; Schafer, P. Plant root-microbe communication in shaping root microbiomes. Plant Mol. Biol. 2016, 90, 575–587. [Google Scholar] [CrossRef]
- Sasan, R.K.; Bidochka, M.J. The insect-pathogenic fungus Metarhizium robertsii (Clavicipitaceae) is also an endophyte that stimulates plant root development. Am. J. Bot. 2012, 99, 101–107. [Google Scholar] [CrossRef]
- Sasan, R.K.; Bidochka, M.J. Antagonism of the endophytic insect pathogenic fungus Metarhizium robertsii against the bean plant pathogen Fusarium solanif. sp.phaseoli. Can. J. Plant Pathol. 2013, 35, 288–293. [Google Scholar] [CrossRef]
- Ahmad, I.; Jiménez-Gasco, M.d.M.; Luthe, D.S.; Shakeel, S.N.; Barbercheck, M.E. Endophytic Metarhizium robertsii promotes maize growth, suppresses insect growth, and alters plant defense gene expression. Biol. Control 2020, 144, 104167. [Google Scholar] [CrossRef]
- Chairin, T.; Petcharat, V. Induction of defense responses in longkong fruit (Aglaia dookkoo Griff.) against fruit rot fungi by Metarhizium guizhouense. Biol. Control 2017, 111, 40–44. [Google Scholar] [CrossRef]
- Hao, K.; Wang, F.; Nong, X.; McNeill, M.R.; Liu, S.; Wang, G.; Cao, G.; Zhang, Z. Response of peanut Arachis hypogaea roots to the presence of beneficial and pathogenic fungi by transcriptome analysis. Sci. Rep. 2017, 7, 964. [Google Scholar] [CrossRef] [PubMed]
- Patel, Z.M.; Mahapatra, R.; Jampala, S.S.M. Chapter 11—Role of fungal elicitors in plant defense mechanism. In Molecular Aspects of Plant Beneficial Microbes in Agriculture; Sharma, V., Salwan, R., Al-Ani, L., Eds.; Academic Press: Cambridge, MA, USA, 2020; pp. 143–158. [Google Scholar] [CrossRef]
- Dong, X.; Sa, J.A. Ethylene, and disease resistance in plants. Curr. Opin. Plant Biol. 1998, 1, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Thomma, B.P.; Eggermont, K.; Penninckx, I.A.; Mauch-Mani, B.; Vogelsang, R.; Cammue, B.P.; Broekaert, W.F. Separate jasmonate-dependent and salicylate-dependent defense-response pathways in Arabidopsis are essential for resistance to distinct microbial pathogens. Proc. Natl. Acad. Sci. USA 1998, 95, 15107–15111. [Google Scholar] [CrossRef] [PubMed]
- Pant, S.R.; Irigoyen, S.; Liu, J.; Bedre, R.; Christensen, S.A.; Schmelz, E.A.; Sedbrook, J.C.; Scholthof, K.-B.G.; Mandadi, K.K. Brachypodium Phenylalanine ammonia lyase (PAL) Promotes antiviral defenses against Panicum mosaic virus and its satellites. mBio 2021, 12, e03518-20. [Google Scholar] [CrossRef]
- Liu, R.; Xu, S.; Li, J.; Hu, Y.; Lin, Z. Expression profile of a PAL gene from Astragalus membranaceus var. Mongholicus and its crucial role in flux into flavonoid biosynthesis. Plant Cell Rep. 2006, 25, 705–710. [Google Scholar] [CrossRef]
- De Geyter, N.; Gholami, A.; Goormachtig, S.; Goossens, A. Transcriptional machineries in jasmonate-elicited plant secondary metabolism. Trends Plant Sci. 2012, 17, 349–359. [Google Scholar] [CrossRef]
- Mirjalili, N.; Linden, J.C. Gas phase composition effects on suspension cultures of Taxus cuspidata. Biotechnol. Bioeng. 1995, 48, 123–132. [Google Scholar] [CrossRef]
- Li, S.T.; Zhang, P.; Zhang, M.; Fu, C.H.; Zhao, C.F.; Dong, Y.S.; Guo, A.Y.; Yu, L.J. Transcriptional profile of taxus chinensis cells in response to methyl jasmonate. BMC Genom. 2012, 13, 295. [Google Scholar] [CrossRef]
- Tugizimana, F.; Ncube, E.N.; Steenkamp, P.A.; Dubery, I.A. Metabolomics-derived insights into the manipulation of terpenoid synthesis in Centella asiatica cells by methyl jasmonate. Plant Biotechnol. Rep. 2015, 9, 125–136. [Google Scholar] [CrossRef]
- Lu, M.; Wong, H.; Teng, W. Effects of elicitation on the production of saponin in cell culture of Panax ginseng. Plant Cell Rep. 2001, 20, 674–677. [Google Scholar] [CrossRef]
- Mandal, S.; Kar, I.; Mukherjee, A.K.; Acharya, P. Elicitor-induced defense responses in Solanum lycopersicum against Ralstonia solanacearum. Sci. World J. 2013, 25, 561056. [Google Scholar] [CrossRef]
- Li, L.; Wang, S.; Yang, X.; Francis, F.; Qiu, D. Protein elicitor PeaT1 enhanced resistance against aphid (Sitobion avenae) in wheat. Pest Manag. Sci. 2020, 76, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Bujalowski, W. Expanding the physiological role of the hexameric DnaB helicase. Trends Biochem. Sci. 2003, 28, 116–118. [Google Scholar] [CrossRef]
- Vaknin, Y.; Shadkchan, Y.; Levdansky, E.; Morozov, M.; Romano, J.; Osherov, N. The three Aspergillus fumigatus CFEM-domain GPI-anchored proteins (CfmA-C) affect cell-wall stability but do not play a role in fungal virulence. Fungal. Genet. Biol. 2014, 63, 55–64. [Google Scholar] [CrossRef]
- Zhao, S.; Shang, X.; Bi, W.; Yu, X.; Liu, D.; Kang, Z.; Wang, X.; Wang, X. Genome-Wide Identification of effector candidates with conserved motifs from the wheat leaf rust fungus Puccinia triticina. Front. Microbiol. 2020, 11, 1188. [Google Scholar] [CrossRef]
- Gong, A.; Jing, Z.; Zhang, K.; Tan, Q.; Wang, G.; Liu, W. Bioinformatic analysis and functional characterization of the CFEM proteins in maize anthracnose fungus Colletotrichum graminicola. J. Integr. Agric. 2020, 19, 541–550. [Google Scholar] [CrossRef]
- Martin, F.; Aerts, A.; Ahren, D.; Brun, A.; Danchin, E.G.; Duchaussoy, F.; Gibon, J.; Kohler, A.; Lindquist, E.; Pereda, V.; et al. The genome of Laccaria bicolor provides insights into mycorrhizal symbiosis. Nature 2008, 452, 88–92. [Google Scholar] [CrossRef]
- Stefan, V.; Lara, R.J. Entomopathogenic fungi as endophytes: Plant-endophyte-herbivore interactions and prospects for use in biological control. Curr. Sci. 2015, 109, 46–54. [Google Scholar]
- Cai, N.; Liu, R.; Yan, D.; Zhang, N.; Zhu, K.; Zhang, D.; Nong, X.; Tu, X.; Zhang, Z.; Wang, G. Bioinformatics analysis and functional characterization of the CFEM proteins of Metarhizium anisopliae. J. Fungi. 2022, 8, 661. [Google Scholar] [CrossRef]
- Stephens, C.; Hammond-Kosack, K.E.; and Kanyuka, K. WAKsing plant immunity, waning diseases. J. Exp. Bot 2022, 73, 22–37. [Google Scholar] [CrossRef]
- Zhang, Z.N.; Wu, Q.Y.; Zhang, G.Z.; Zhu, Y.Y.; Murphy, R.W.; Liu, Z.; Zou, C.G. Systematic analyses reveal uniqueness and origin of the CFEM domain in fungi. Sci. Rep. 2015, 5, 13032. [Google Scholar] [CrossRef] [PubMed]
- Fang, A.; Gao, H.; Zhang, N.; Zheng, X.; Qiu, S.; Li, Y.; Zhou, S.; Cui, F.; Sun, W. A novel effector gene SCRE2 contributes to full virulence of Ustilaginoidea virens to rice. Front. Microbiol. 2019, 10, 845. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Wei, W.; Wu, Y.; Zhou, Y.; Peng, F.; Zhang, S.; Chen, P.; Xu, X. BcCFEM1, a CFEM domain-containing protein with putative GPI-anchored site, is involved in pathogenicity, conidial production, and stress tolerance in Botrytis cinerea. Front. Microbiol. 2017, 8, 1807. [Google Scholar] [CrossRef]
- Xu, X.; Li, G.; Li, L.; Su, Z.; Chen, C. Genome-wide comparative analysis of putative Pth11-related G protein-coupled receptors in fungi belonging to Pezizomycotina. BMC Microbiol. 2017, 17, 166. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.J.; Hou, J.; Zhang, H.; Lei, J.H.; Lin, H.Y.; Ding, J.L.; Feng, M.G.; Ying, S.H. Systematic contributions of CFEM domain-containing proteins to iron acquisition are essential for interspecies interaction of the filamentous pathogenic fungus Beauveria bassiana. Environ. Microbiol. 2022, 24, 3693–3704. [Google Scholar] [CrossRef] [PubMed]
- Roy, U.; Kornitzer, D. Heme-iron acquisition in fungi. Curr. Opin. Microbiol. 2019, 52, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Okamoto-Shibayama, K.; Kikuchi, Y.; Kokubu, E.; Sato, Y.; Ishihara, K. Csa2, a member of the Rbt5 protein family, is involved in the utilization of iron from human hemoglobin during Candida albicans hyphal growth. FEMS Yeast Res. 2014, 14, 674–677. [Google Scholar] [CrossRef]
- Perez, A.; Pedros, B.; Murgui, A.; Casanova, M.; Lopez-Ribot, J.L.; Martinez, J.P. Biofilm formation by Candida albicans mutants for genes coding fungal proteins exhibiting the eight-cysteine-containing CFEM domain. FEMS Yeast Res. 2006, 6, 1074–1084. [Google Scholar] [CrossRef]
- Hogg, P.J. Disulfide bonds as switches for protein function. Trends Biochem. Sci. 2003, 28, 210–214. [Google Scholar] [CrossRef]
- van den Burg, H.A.; Westerink, N.; Francoijs, K.J.; Roth, R.; Woestenenk, E.; Boeren, S.; de Wit, P.J.; Joosten, M.H.; Vervoort, J. Natural disulfide bond-disrupted mutants of AVR4 of the tomato pathogen Cladosporium fulvum are sensitive to proteolysis, circumvent Cf-4-mediated resistance, but retain their chitin binding ability. J. Biol. Chem. 2003, 278, 27340–27346. [Google Scholar] [CrossRef]
- Hu, K.; Cao, J.; Zhang, J.; Xia, F.; Ke, Y.; Zhang, H.; Xie, W.; Liu, H.; Cui, Y.; Cao, Y.; et al. Improvement of multiple agronomic traits by a disease resistance gene via cell wall reinforcement. Nat. Plants 2017, 3, 17009. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, H.; Fujisawa, S.; Mitsuoka, C.; Ito, A.; Hirabuchi, A.; Ikeda, K.; Irieda, H.; Yoshino, K.; Yoshida, K.; Matsumura, H.; et al. Large-scale gene disruption in Magnaporthe oryzae identifies MC69, a secreted protein required for infection by monocot and dicot fungal pathogens. PLoS Pathog. 2012, 8, e1002711. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, R.D.; Kelkar, H.S.; Dean, R. An eight-cysteine-containing CFEM domain unique to a group of fungal membrane proteins. Trends Biochem. Sci. 2013, 28, 118–121. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Wu, L.; Zhang, W.; Zhang, Q.; Xing, Q.; Wang, X.; Li, X.; Yan, J. Systemic identification and functional characterization of common in fungal extracellular membrane proteins in Lasiodiplodia theobromae. Front. Plant Sci. 2021, 12, 804696. [Google Scholar] [CrossRef]
- Bari, R.; Jones, J.D. Role of plant hormones in plant defence responses. Plant Mol. Biol. 2009, 69, 473–488. [Google Scholar] [CrossRef]
- Verma, V.; Ravindran, P.; Kumar, P.P. Plant hormone-mediated regulation of stress responses. BMC Plant Biol. 2016, 16, 86. [Google Scholar] [CrossRef]
- Javed, K.; Qiu, D. Protein elicitor PeBL1 of Brevibacillus laterosporus enhances resistance against Myzus persicae in Tomato. Pathogens 2020, 9, 57. [Google Scholar] [CrossRef]
- Basit, A.; Hanan, A.; Nazir, T.; Majeed, M.Z.; Qiu, D. Molecular and functional characterization of elicitor PeBC1 extracted from Botrytis cinerea involved in the induction of resistance against green peach aphid (Myzus persicae) in common beans (Phaseolus vulgaris L.). Insects 2019, 10, 35. [Google Scholar] [CrossRef]
- Nazir, T.; Hanan, A.; Basit, A.; Majeed, M.Z.; Anwar, T.; Nawaz, I.; Qiu, D. Putative role of a yet uncharacterized protein elicitor PeBb1 Derived from Beauveria bassiana ARSEF 2860 Strain against Myzus persicae (Homoptera: Aphididae) in Brassica rapa ssp. pekinensis. Pathogens 2020, 9, 111. [Google Scholar] [CrossRef]
- Spoel, S.H.; Koornneef, A.; Claessens, S.M.; Korzelius, J.P.; Van Pelt, J.A.; Mueller, M.J.; Buchala, A.J.; Metraux, J.P.; Brown, R.; Kazan, K.; et al. NPR1 modulates cross-talk between salicylate- and jasmonate-dependent defense pathways through a novel function in the cytosol. Plant Cell 2003, 15, 760–770. [Google Scholar] [CrossRef]
- Xin, X.F.; He, S.Y. Pseudomonas syringae pv. tomato DC3000: A model pathogen for probing disease susceptibility and hormone signaling in plants. Annu. Rev. Phytopathol. 2013, 51, 473–498. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wang, C.; Zhang, Y.; Sun, Y.; Mou, Z. The Arabidopsis mediator complex subunit16 positively regulates salicylate-mediated systemic acquired resistance and jasmonate/ethylene-induced defense pathways. Plant Cell 2012, 24, 4294–4309. [Google Scholar] [CrossRef] [PubMed]
- Doares, S.H.; Narvaez-Vasquez, J.; Conconi, A.; Ryan, C.A. Salicylic acid inhibits synthesis of proteinase inhibitors in tomato leaves induced by systemin and jasmonic acid. Plant Physiol. 1995, 108, 1741–1746. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, C.M.; Leon-Reyes, A.; Van der Ent, S.; Van Wees, S.C. Networking by small-molecule hormones in plant immunity. Nat. Chem. Biol. 2009, 5, 308–316. [Google Scholar] [CrossRef]
- Penninckx, I.A.; Thomma, B.P.; Buchala, A.; Métraux, J.P.; Broekaert, W.F. Concomitant activation of jasmonate and ethylene response pathways is required for induction of a plant defensin gene in Arabidopsis. Plant Cell 1998, 10, 2103–2113. [Google Scholar] [CrossRef]
- Fang, W.; Bidochka, M.J. Expression of genes involved in germination, conidiogenesis and pathogenesis in Metarhizium anisopliae using quantitative real-time RT-PCR. Mycol. Res. 2006, 110, 1165–1171. [Google Scholar] [CrossRef]
- Guerriero, G.; Legay, S.; Hausman, J.F. Alfalfa Cellulose synthase gene expression under abiotic stress: A Hitchhiker’s guide to RT-qPCR normalization. PLoS ONE 2014, 9, e103808. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, Y.; Li, B.; Yang, X.; Dong, Y.; Qiu, D. A Verticillium dahliae pectate lyase induces plant immune responses and contributes to virulence. Front. Plant Sci. 2018, 9, 1271. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).