Exosomes Derived from Adipose Stem Cells Enhance Bone Fracture Healing via the Activation of the Wnt3a/β-Catenin Signaling Pathway in Rats with Type 2 Diabetes Mellitus
Abstract
:1. Introduction
2. Results
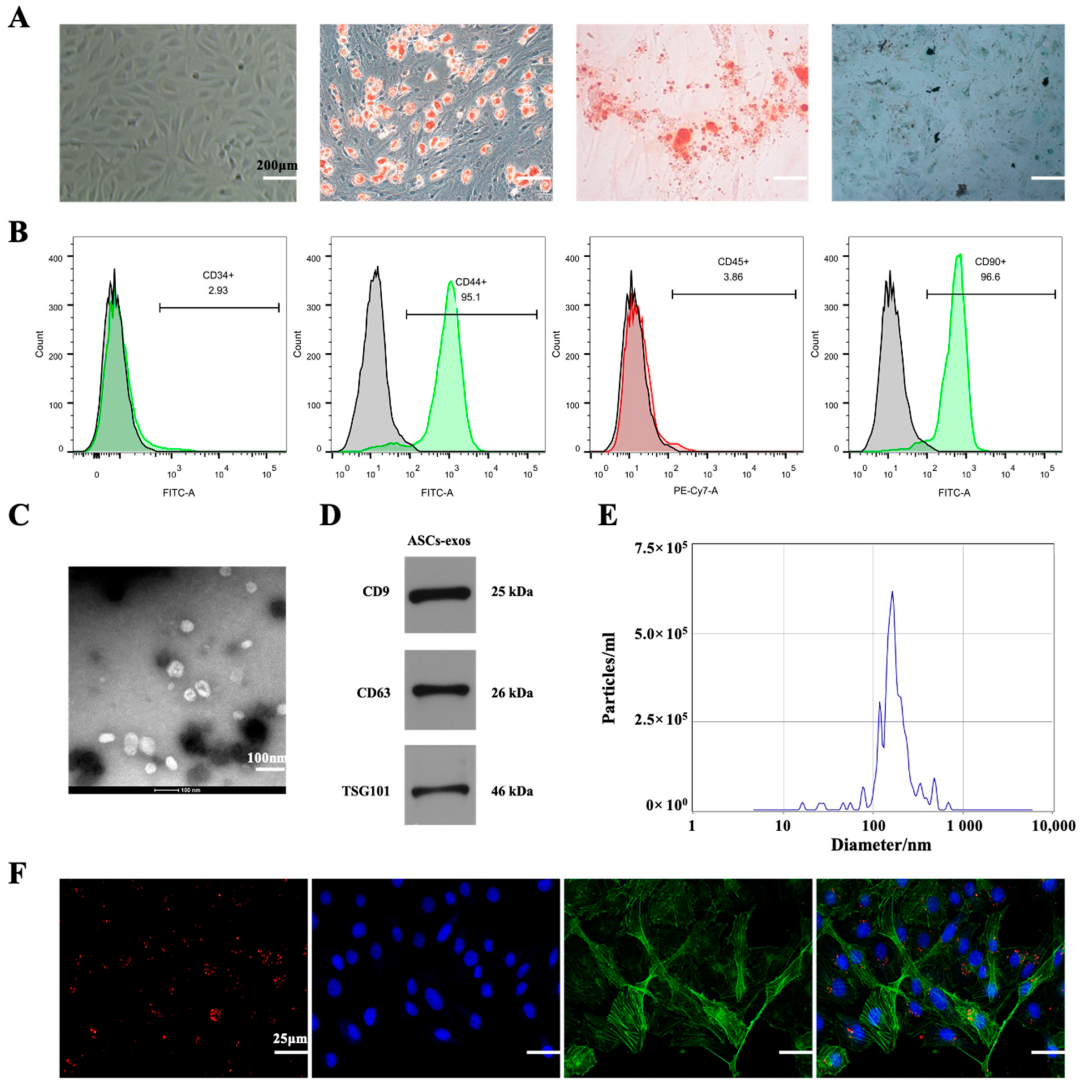
2.1. Characterization of ASCs-exos
2.2. ASCs-exos Promote BMSC Osteogenesis Differentiation In Vivo
2.3. Successful Establishment of the Type 2 Diabetes Mellitus Rat Model
2.4. ASCs-exos Enhance T2DM-Delayed Fracture in Rat Model
2.5. ASCs-exos Activated the Wnt3a/β-Catenin Signaling Pathway in BMSCs under High-Glucose Conditions
2.6. The Activation of the Wnt3a/β-Catenin Signaling Pathway in BMSCs Treated with ASCs-exos under High-Glucose Conditions Can Be Inhibited via Dickkopf-Related Protein-1
3. Discussion
4. Materials and Methods
4.1. The T2DM Rat Model of Fracture Establishment and Treatment
4.2. Cell Culture
4.3. Identification of ASCs
4.4. Exosome Uptake Assay
4.5. BMSC Osteogenic Differentiation
4.6. Immunofluorescence Analysis
4.7. ALP and Alizarin Red Staining
4.8. Radiographic and Histological Analysis
4.9. Western Blot Analysis
4.10. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lecka-Czernik, B. Diabetes, bone and glucose-lowering agents: Basic biology. Diabetologia 2017, 60, 1163–1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roden, M. Diabetes mellitus: Definition, classification and diagnosis. Wien Klin Wochenschr 2016, 128 (Suppl. S2), S37–S40. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Stein, C.; Basit, A.; Chan, J.C.N.; Mbanya, J.C.; et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pr. 2022, 183, 109119. [Google Scholar] [CrossRef] [PubMed]
- Cloete, L. Diabetes mellitus: An overview of the types, symptoms, complications and management. Nurs. Stand. 2022, 37, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Li, Z.; Wang, Z.; Zeng, F.; Xiao, W.; Yu, A. MicroRNA-126: A promising biomarker for angiogenesis of diabetic wounds treated with negative pressure wound therapy. Diabetes Metab. Syndr. Obes. 2019, 12, 1685–1696. [Google Scholar] [CrossRef] [Green Version]
- Compston, J. Type 2 diabetes mellitus and bone. J. Intern. Med. 2018, 283, 140–153. [Google Scholar] [CrossRef] [Green Version]
- Henderson, S.; Ibe, I.; Cahill, S.; Chung, Y.H.; Lee, F.Y. Bone Quality and Fracture-Healing in Type-1 and Type-2 Diabetes Mellitus. J. Bone Joint. Surg. Am. 2019, 101, 1399–1410. [Google Scholar] [CrossRef]
- Kostenuik, P.; Mirza, F.M. Fracture healing physiology and the quest for therapies for delayed healing and nonunion. J. Orthop. Res. 2017, 35, 213–223. [Google Scholar] [CrossRef]
- Wildemann, B.; Ignatius, A.; Leung, F.; Taitsman, L.A.; Smith, R.M.; Pesántez, R.; Stoddart, M.J.; Richards, R.G.; Jupiter, J.B. Non-union bone fractures. Nat. Rev. Dis. Prim. 2021, 7, 57. [Google Scholar] [CrossRef]
- Lin, H.; Sohn, J.; Shen, H.; Langhans, M.T.; Tuan, R.S. Bone marrow mesenchymal stem cells: Aging and tissue engineering applications to enhance bone healing. Biomaterials 2019, 203, 96–110. [Google Scholar] [CrossRef]
- Tseng, S.S.; Lee, M.A.; Reddi, A.H. Nonunions and the potential of stem cells in fracture-healing. J. Bone Joint. Surg. Am. 2008, 90 (Suppl. S1), 92–98. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Han, Y.D.; Yan, X.L.; Ren, J.; Zeng, Q.; Li, X.D.; Pei, X.T.; Han, Y. Adipose mesenchymal stem cell-derived exosomes stimulated by hydrogen peroxide enhanced skin flap recovery in ischemia-reperfusion injury. Biochem. Biophys Res. Commun. 2018, 500, 310–317. [Google Scholar] [CrossRef] [PubMed]
- Bajek, A.; Gurtowska, N.; Olkowska, J.; Kazmierski, L.; Maj, M.; Drewa, T. Adipose-Derived Stem Cells as a Tool in Cell-Based Therapies. Arch Immunol. Ther. Exp. 2016, 64, 443–454. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, B.; Feng, J.; Guo, J.; Wang, J.; Xiu, G.; Xu, J.; Ning, K.; Ling, B.; Fu, Q.; Xu, J. ADSCs-derived exosomes ameliorate hepatic fibrosis by suppressing stellate cell activation and remodeling hepatocellular glutamine synthetase-mediated glutamine and ammonia homeostasis. Stem. Cell Res. Ther. 2022, 13, 494. [Google Scholar] [CrossRef]
- Hass, R.; Kasper, C.; Böhm, S.; Jacobs, R. Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun. Signal. 2011, 9, 12. [Google Scholar] [CrossRef] [Green Version]
- Jeong, J.O.; Han, J.W.; Kim, J.M.; Cho, H.J.; Park, C.; Lee, N.; Kim, D.W.; Yoon, Y.S. Malignant tumor formation after transplantation of short-term cultured bone marrow mesenchymal stem cells in experimental myocardial infarction and diabetic neuropathy. Circ. Res. 2011, 108, 1340–1347. [Google Scholar] [CrossRef] [Green Version]
- Abels, E.R.; Breakefield, X.O. Introduction to Extracellular Vesicles: Biogenesis, RNA Cargo Selection, Content, Release, and Uptake. Cell Mol. Neurobiol. 2016, 36, 301–312. [Google Scholar] [CrossRef] [Green Version]
- Wang, B.; Xing, D.; Zhu, Y.; Dong, S.; Zhao, B. The State of Exosomes Research: A Global Visualized Analysis. Biomed. Res. Int. 2019, 2019, 1495130. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Kuang, L.; Chen, C.; Yang, J.; Zeng, W.N.; Li, T.; Chen, H.; Huang, S.; Fu, Z.; Li, J.; et al. miR-100-5p-abundant exosomes derived from infrapatellar fat pad MSCs protect articular cartilage and ameliorate gait abnormalities via inhibition of mTOR in osteoarthritis. Biomaterials 2019, 206, 87–100. [Google Scholar] [CrossRef]
- Furuta, T.; Miyaki, S.; Ishitobi, H.; Ogura, T.; Kato, Y.; Kamei, N.; Miyado, K.; Higashi, Y.; Ochi, M. Mesenchymal Stem Cell-Derived Exosomes Promote Fracture Healing in a Mouse Model. Stem. Cells Transl. Med. 2016, 5, 1620–1630. [Google Scholar] [CrossRef] [Green Version]
- Hao, Z.C.; Lu, J.; Wang, S.Z.; Wu, H.; Zhang, Y.T.; Xu, S.G. Stem cell-derived exosomes: A promising strategy for fracture healing. Cell Prolif. 2017, 50, e12359. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, D.; Wu, Y.; Li, Z.; Chen, H.; Huang, S.; Jian, C.; Yu, A. MiR-144-5p, an exosomal miRNA from bone marrow-derived macrophage in type 2 diabetes, impairs bone fracture healing via targeting Smad1. J. Nanobiotechnology 2021, 19, 226. [Google Scholar] [CrossRef]
- Pegtel, D.M.; Gould, S.J. Exosomes. Annu Rev Biochem 2019, 88, 487–514. [Google Scholar] [CrossRef] [PubMed]
- Boriani, F.; Perut, F. Exosomes Are Comparable to Source Adipose Stem Cells in Fat Graft Retention with Up-Regulating Early Inflammation and Angiogenesis. Plast. Reconstr. Surg. 2020, 146, 232e. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Alonso, M.L.; García-Posadas, L.; Diebold, Y. Extracellular Vesicles from Human Adipose-Derived Mesenchymal Stem Cells: A Review of Common Cargos. Stem. Cell Rev. Rep. 2021, 18, 854–901. [Google Scholar] [CrossRef] [PubMed]
- Abbas, O.L.; Borman, H.; Uysal, Ç.A.; Gönen, Z.B.; Aydin, L.; Helvacioğlu, F.; Ilhan, Ş.; Yazici, A.C. Adipose-Derived Stem Cells Enhance Axonal Regeneration through Cross-Facial Nerve Grafting in a Rat Model of Facial Paralysis. Plast. Reconstr. Surg. 2016, 138, 387–396. [Google Scholar] [CrossRef]
- Abdul Halim, N.S.S.; Yahaya, B.H.; Lian, J. Therapeutic Potential of Adipose-Derived Stem Cells in the Treatment of Pulmonary Diseases. Curr. Stem. Cell Res. Ther. 2021, 17, 107–112. [Google Scholar] [CrossRef]
- An, Y.; Lin, S.; Tan, X.; Zhu, S.; Nie, F.; Zhen, Y.; Gu, L.; Zhang, C.; Wang, B.; Wei, W.; et al. Exosomes from adipose-derived stem cells and application to skin wound healing. Cell Prolif. 2021, 54, e12993. [Google Scholar] [CrossRef]
- Bolandi, Z.; Mokhberian, N.; Eftekhary, M.; Sharifi, K.; Soudi, S.; Ghanbarian, H.; Hashemi, S.M. Adipose derived mesenchymal stem cell exosomes loaded with miR-10a promote the differentiation of Th17 and Treg from naive CD4(+) T cell. Life Sci. 2020, 259, 118218. [Google Scholar] [CrossRef]
- Bucan, V.; Vaslaitis, D.; Peck, C.T.; Strauß, S.; Vogt, P.M.; Radtke, C. Effect of Exosomes from Rat Adipose-Derived Mesenchymal Stem Cells on Neurite Outgrowth and Sciatic Nerve Regeneration After Crush Injury. Mol. Neurobiol. 2019, 56, 1812–1824. [Google Scholar] [CrossRef] [Green Version]
- Tzioupis, C.; Giannoudis, P.V. Prevalence of long-bone non-unions. Injury 2007, 38 (Suppl. S2), S3–S9. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Jia, Y.; Wang, Y.; Xu, J.; Chai, Y. Impaired Bone Regenerative Effect of Exosomes Derived from Bone Marrow Mesenchymal Stem Cells in Type 1 Diabetes. Stem. Cells Transl. Med. 2019, 8, 593–605. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Einhorn, T.A.; Gerstenfeld, L.C. Fracture healing: Mechanisms and interventions. Nat. Rev. Rheumatol. 2015, 11, 45–54. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abou-Khalil, R.; Colnot, C. Cellular and molecular bases of skeletal regeneration: What can we learn from genetic mouse models? Bone 2014, 64, 211–221. [Google Scholar] [CrossRef]
- Nie, W.; Wang, Z.; Cao, J.; Wang, W.; Guo, Y.; Zhang, C.; Jia, W.; Li, X. Preliminary outcomes of the combination of demineralized bone matrix and platelet Rich plasma in the treatment of long bone non-unions. BMC Musculoskelet Disord. 2021, 22, 951. [Google Scholar] [CrossRef]
- Mills, L.A.; Simpson, A.H. The relative incidence of fracture non-union in the Scottish population (5.17 million): A 5-year epidemiological study. BMJ Open 2013, 3, e002276. [Google Scholar] [CrossRef] [Green Version]
- Gaston, M.S.; Simpson, A.H. Inhibition of fracture healing. J. Bone Joint. Surg. Br. 2007, 89, 1553–1560. [Google Scholar] [CrossRef] [Green Version]
- Aday, S.; Hazan-Halevy, I.; Chamorro-Jorganes, A.; Anwar, M.; Goldsmith, M.; Beazley-Long, N.; Sahoo, S.; Dogra, N.; Sweaad, W.; Catapano, F.; et al. Bioinspired artificial exosomes based on lipid nanoparticles carrying let-7b-5p promote angiogenesis in vitro and in vivo. Mol. Ther. 2021, 29, 2239–2252. [Google Scholar] [CrossRef]
- Xiong, Y.; Chen, L.; Yan, C.; Zhou, W.; Yu, T.; Sun, Y.; Cao, F.; Xue, H.; Hu, Y.; Chen, D.; et al. M2 Macrophagy-derived exosomal miRNA-5106 induces bone mesenchymal stem cells towards osteoblastic fate by targeting salt-inducible kinase 2 and 3. J. Nanobiotechnology 2020, 18, 66. [Google Scholar] [CrossRef]
- Cheng, M.; Yang, J.; Zhao, X.; Zhang, E.; Zeng, Q.; Yu, Y.; Yang, L.; Wu, B.; Yi, G.; Mao, X.; et al. Circulating myocardial microRNAs from infarcted hearts are carried in exosomes and mobilise bone marrow progenitor cells. Nat. Commun. 2019, 10, 959. [Google Scholar] [CrossRef]
- Schlundt, C.; El Khassawna, T.; Serra, A.; Dienelt, A.; Wendler, S.; Schell, H.; van Rooijen, N.; Radbruch, A.; Lucius, R.; Hartmann, S.; et al. Macrophages in bone fracture healing: Their essential role in endochondral ossification. Bone 2018, 106, 78–89. [Google Scholar] [CrossRef] [PubMed]
- Ying, W.; Riopel, M.; Bandyopadhyay, G.; Dong, Y.; Birmingham, A.; Seo, J.B.; Ofrecio, J.M.; Wollam, J.; Hernandez-Carretero, A.; Fu, W.; et al. Adipose Tissue Macrophage-Derived Exosomal miRNAs Can Modulate In Vivo and In Vitro Insulin Sensitivity. Cell 2017, 171, 372–384.e312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Govindappa, P.K.; Patil, M.; Garikipati, V.N.S.; Verma, S.K.; Saheera, S.; Narasimhan, G.; Zhu, W.; Kishore, R.; Zhang, J.; Krishnamurthy, P. Targeting exosome-associated human antigen R attenuates fibrosis and inflammation in diabetic heart. Faseb J. 2020, 34, 2238–2251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liao, W.; Ning, Y.; Xu, H.J.; Zou, W.Z.; Hu, J.; Liu, X.Z.; Yang, Y.; Li, Z.H. BMSC-derived exosomes carrying microRNA-122-5p promote proliferation of osteoblasts in osteonecrosis of the femoral head. Clin. Sci. 2019, 133, 1955–1975. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Zhao, C.; Li, Y.; Wang, L.; Nie, G.; Peng, J.; Wang, A.; Zhang, P.; Tian, W.; Li, Q.; et al. Osteoclast-derived microRNA-containing exosomes selectively inhibit osteoblast activity. Cell Discov. 2016, 2, 16015. [Google Scholar] [CrossRef]
- Nan, K.; Zhang, Y.; Zhang, X.; Li, D.; Zhao, Y.; Jing, Z.; Liu, K.; Shang, D.; Geng, Z.; Fan, L. Exosomes from miRNA-378-modified adipose-derived stem cells prevent glucocorticoid-induced osteonecrosis of the femoral head by enhancing angiogenesis and osteogenesis via targeting miR-378 negatively regulated suppressor of fused (Sufu). Stem. Cell Res. Ther. 2021, 12, 331. [Google Scholar] [CrossRef]
- Shen, J.; Sun, Y.; Liu, X.; Zhu, Y.; Bao, B.; Gao, T.; Chai, Y.; Xu, J.; Zheng, X. EGFL6 regulates angiogenesis and osteogenesis in distraction osteogenesis via Wnt/β-catenin signaling. Stem. Cell Res. Ther. 2021, 12, 415. [Google Scholar] [CrossRef]
- Shen, G.; Ren, H.; Shang, Q.; Zhao, W.; Zhang, Z.; Yu, X.; Tang, K.; Tang, J.; Yang, Z.; Liang, D.; et al. Foxf1 knockdown promotes BMSC osteogenesis in part by activating the Wnt/β-catenin signalling pathway and prevents ovariectomy-induced bone loss. EBioMedicine 2020, 52, 102626. [Google Scholar] [CrossRef]
- Bandara, N.; Gurusinghe, S.; Lim, S.Y.; Chen, H.; Chen, S.; Wang, D.; Hilbert, B.; Wang, L.X.; Strappe, P. Molecular control of nitric oxide synthesis through eNOS and caveolin-1 interaction regulates osteogenic differentiation of adipose-derived stem cells by modulation of Wnt/β-catenin signaling. Stem. Cell Res. Ther. 2016, 7, 182. [Google Scholar] [CrossRef] [Green Version]
- Cai, T.; Sun, D.; Duan, Y.; Wen, P.; Dai, C.; Yang, J.; He, W. WNT/β-catenin signaling promotes VSMCs to osteogenic transdifferentiation and calcification through directly modulating Runx2 gene expression. Exp. Cell Res. 2016, 345, 206–217. [Google Scholar] [CrossRef] [Green Version]
- Sohrabipour, S.; Sharifi, M.R.; Talebi, A.; Sharifi, M.; Soltani, N. GABA dramatically improves glucose tolerance in streptozotocin-induced diabetic rats fed with high-fat diet. Eur. J. Pharmacol. 2018, 826, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Byun, S.E.; Sim, C.; Chung, Y.; Kim, H.K.; Park, S.; Kim, D.K.; Cho, S.; Lee, S. Skeletal Muscle Regeneration by the Exosomes of Adipose Tissue-Derived Mesenchymal Stem Cells. Curr. Issues Mol. Biol. 2021, 43, 1473–1488. [Google Scholar] [CrossRef] [PubMed]
- Shou, K.; Niu, Y.; Zheng, X.; Ma, Z.; Jian, C.; Qi, B.; Hu, X.; Yu, A. Enhancement of Bone-Marrow-Derived Mesenchymal Stem Cell Angiogenic Capacity by NPWT for a Combinatorial Therapy to Promote Wound Healing with Large Defect. Biomed Res. Int. 2017, 2017, 7920265. [Google Scholar] [CrossRef] [PubMed] [Green Version]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, D.; Xiao, W.; Liu, C.; Wang, Z.; Liu, Y.; Yu, Y.; Jian, C.; Yu, A. Exosomes Derived from Adipose Stem Cells Enhance Bone Fracture Healing via the Activation of the Wnt3a/β-Catenin Signaling Pathway in Rats with Type 2 Diabetes Mellitus. Int. J. Mol. Sci. 2023, 24, 4852. https://doi.org/10.3390/ijms24054852
Zhang D, Xiao W, Liu C, Wang Z, Liu Y, Yu Y, Jian C, Yu A. Exosomes Derived from Adipose Stem Cells Enhance Bone Fracture Healing via the Activation of the Wnt3a/β-Catenin Signaling Pathway in Rats with Type 2 Diabetes Mellitus. International Journal of Molecular Sciences. 2023; 24(5):4852. https://doi.org/10.3390/ijms24054852
Chicago/Turabian StyleZhang, Dong, Weidong Xiao, Changjiang Liu, Zheng Wang, Yuhang Liu, Yifeng Yu, Chao Jian, and Aixi Yu. 2023. "Exosomes Derived from Adipose Stem Cells Enhance Bone Fracture Healing via the Activation of the Wnt3a/β-Catenin Signaling Pathway in Rats with Type 2 Diabetes Mellitus" International Journal of Molecular Sciences 24, no. 5: 4852. https://doi.org/10.3390/ijms24054852



