Kinase Inhibitors in Genetic Diseases
Abstract
:1. Introduction
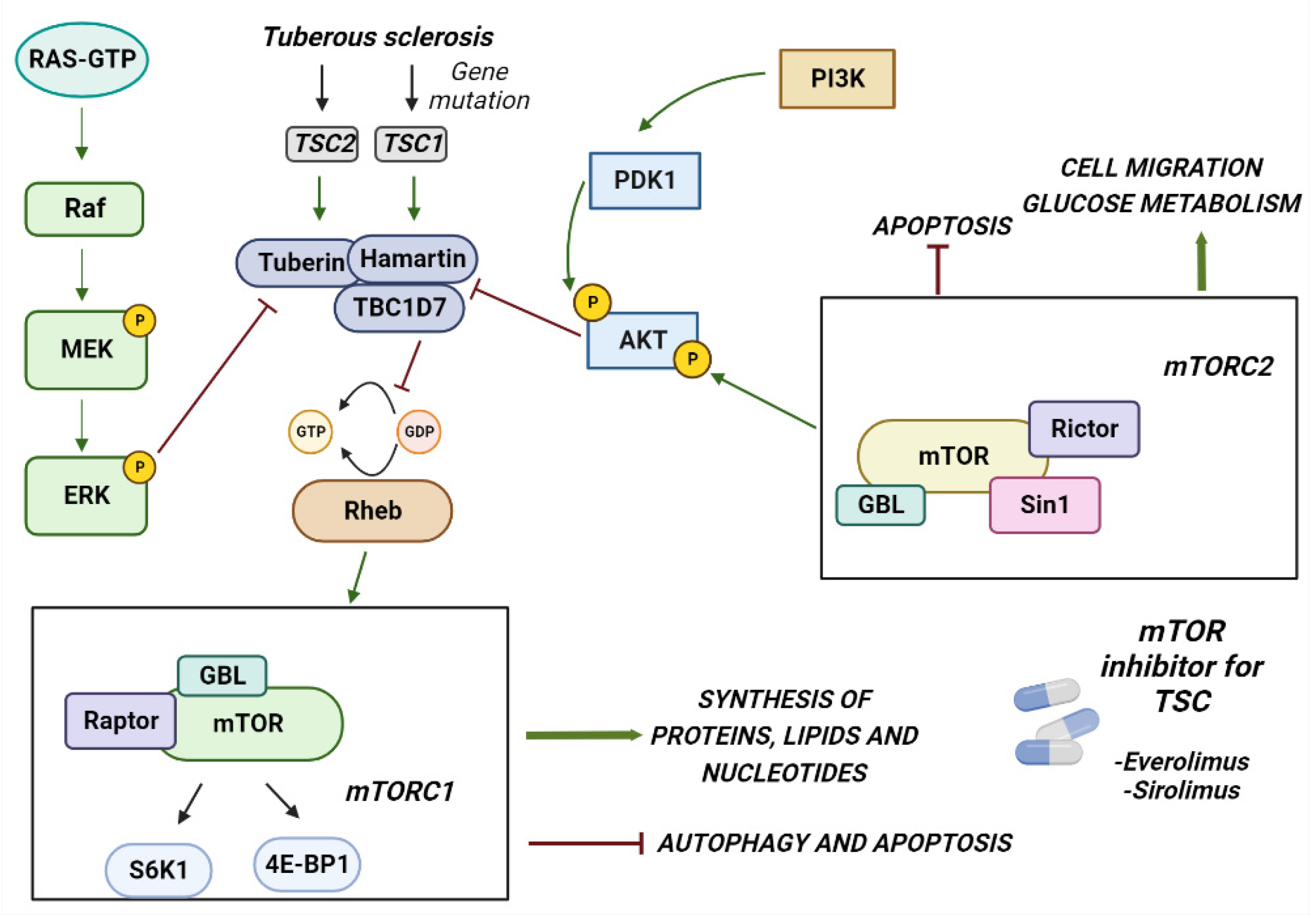
2. Tuberous Sclerosis Complex
2.1. Clinical Manifestation
2.2. Genetics of TSC
2.3. Inhibition of mTOR
3. RASopathies
3.1. Clinical Manifestation
3.2. Neurofibromatosis
3.3. Noonan Syndrome
3.4. Genetics of RASopathies
| Disease | Mutated Genes |
|---|---|
| Neurofibromatosis type 1 (NF1) | NF1 [98,99,100,101] |
| Noonan syndrome (NS) | PTPN11 [81,82] SOS1 [84,85] KRAS [86,87] BRAF [88] SHOC2 [89] CBL [90] |
| Noonan syndrome with multiple lentigines (NSML) | PTPN11 [102] |
| Costello syndrome (CS) | HRAS [92,93] |
| Cardio-facio-cutaneous syndorme (CFC) | BRAF [94] MAP2K1(MEK1) [95] MAP2K2(MEK2) [95] |
| Legius Syndrome | SPRED1 [97] |
3.5. Inibition of RAS Downstream Targets
4. Ciliopathies
4.1. Clinical Manifestation
| Syndrome | Gene Involved |
|---|---|
| Joubert syndrome | INPP5E [133], ARL13B [134], CC2D2A [135], RPGRIP1L [136], TMEM67 [137], NPHP1 [138], AHI1 [139], CEP290 [140], CXORF5 [141], and TMEM216 [142]. |
| Meckel–Gruber syndrome | MKS1 [143], MKS3 (TMEM67) [144], CEP290 [137], RPGRIP1L [136], CC2D2A [145], and TMEM216 [142] |
| Senior–Løken syndrome | CEP290 (also known as NPHP6 and MKS4) [140], NPHP1 [146], NPHP3 [147], NPHP4 [148], and NPHP5 (also known as IQCB1) [149]. |
| Type 1 orofacial syndrome | OFD1 [150] |
| Leber’s congenital amaurosis | GUCY2D [151], RPE65 [152], SPATA7 [153], AIPL1 [154], LCA5 [155], RPGRIPL1 [156], CRX [157], CRB1 [158], IMPD1 [159], RD3 [160], CEP290 [140], NPHP5 [149], and RDH12 [161]. |
| Bardet–Biedl syndrome | BBS1 [162], BBS2 [163], ARL6/BBS3 [164], BBS4 [165], BBS5 [166], MKKS/BBS6 [167], BBS7 [168], TTC8/BBS8 [169], B1/BBS9 [170], BBS10 [171], TRIM32/BBS11 [172], BBS12 [173], MKS1/BBS13 [174], CEP290/BBS14 [174], C2ORF86/FRITZ/BBS15 [175], and SDCCAG8/BBS16 [176] |
| Alström syndrome | ALMS1 [177] |
| Jeune asphyxiating thoracic dystrophy | IFT [178] |
| Rhyns syndrome | TMEM67 [132] |
4.2. The Genetics of Ciliopathies
4.3. Inibition of TRPV4 and SGK1
5. Discussion
6. Conclusions
Funding
Conflicts of Interest
References
- Li, Y.; Zhang, Y.; Li, X.; Yi, S.; Xu, J. Gain-of-Function Mutations: An Emerging Advantage for Cancer Biology. Trends Biochem. Sci. 2019, 44, 659–674. [Google Scholar] [CrossRef]
- Downward, J.; Yarden, Y.; Mayes, E.; Scrace, G.; Totty, N.; Stockwell, P.; Ullrich, A.; Schlessinger, J.; Waterfield, M.D. Close similarity of epidermal growth factor receptor and v-erb-B oncogene protein sequences. Nature 1984, 307, 521–527. [Google Scholar] [CrossRef]
- Perrotti, N.; Taylor, S.I.; Richert, N.D.; Rapp, U.R.; Pastan, I.H.; Roth, J. Immunoprecipitation of Insulin Receptors from Cultured Human Lymphocytes (IM-9 Cells) by Antibodies to pp60 src. Science 1985, 227, 761–763. [Google Scholar] [CrossRef]
- Davies, H.; Hunter, C.; Smith, R.; Stephens, P.; Greenman, C.; Bignell, G.; Teague, J.; Butler, A.; Edkins, S.; Stevens, C.; et al. Somatic Mutations of the Protein Kinase Gene Family in Human Lung Cancer. Cancer Res 2005, 65, 7591–7595. [Google Scholar] [CrossRef] [Green Version]
- Stephens, P.; Edkins, S.; Davies, H.; Greenman, C.; Cox, C.; Hunter, C.; Bignell, G.; Teague, J.; Smith, R.; Stevens, C.; et al. A screen of the complete protein kinase gene family identifies diverse patterns of somatic mutations in human breast cancer. Nat. Genet. 2005, 37, 590–592. [Google Scholar] [CrossRef]
- Greuber, E.K.; Smith-Pearson, P.; Wang, J.; Pendergast, A.M. Role of ABL Family Kinases in Cancer: From Leukemia to Solid Tumors. Nat. Rev. Cancer 2013, 13, 559–571. [Google Scholar]
- Iqbal, N.; Iqbal, N. Human Epidermal Growth Factor Receptor 2 (HER2) in Cancers: Overexpression and Therapeutic Impli-cations. Mol. Biol. Int. 2014, 2014, 852748. [Google Scholar]
- Leicht, D.T.; Balan, V.; Kaplun, A.; Singh-Gupta, V.; Kaplun, L.; Dobson, M.; Tzivion, G. Raf kinases: Function, regulation and role in human cancer. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2007, 1773, 1196–1212. [Google Scholar] [CrossRef] [Green Version]
- Paul, M.K.; Mukhopadhyay, A.K. Tyrosine kinase—Role and significance in Cancer. Int. J. Med. Sci. 2004, 1, 101–115. [Google Scholar] [CrossRef] [Green Version]
- Kannaiyan, R.; Mahadevan, D. A comprehensive review of protein kinase inhibitors for cancer therapy. Expert Rev. Anticancer Ther. 2018, 18, 1249–1270. [Google Scholar] [CrossRef]
- Pottier, C.; Fresnais, M.; Gilon, M.; Jérusalem, G.; Longuespée, R.; Sounni, N.E. Tyrosine Kinase Inhibitors in Cancer: Breakthrough and Challenges of Targeted Therapy. Cancers 2020, 12, 731. [Google Scholar] [CrossRef] [Green Version]
- Grant, S.K. Therapeutic Protein Kinase Inhibitors. Cell. Mol. Life Sci. 2008, 66, 1163–1177. [Google Scholar] [CrossRef]
- Schroeder, R.L.; Stevens, C.L.; Sridhar, J. Small Molecule Tyrosine Kinase Inhibitors of ErbB2/HER2/Neu in the Treatment of Aggressive Breast Cancer. Molecules 2014, 19, 15196–15212. [Google Scholar]
- Schlam, I.; Swain, S.M. HER2-positive breast cancer and tyrosine kinase inhibitors: The time is now. NPJ Breast Cancer 2021, 7, 56. [Google Scholar] [CrossRef]
- Fanotto, V.; Ongaro, E.; Rihawi, K.; Avallone, A.; Silvestris, N.; Fornaro, L.; Vasile, E.; Antonuzzo, L.; Leone, F.; Rosati, G.; et al. HER-2 inhibition in gastric and colorectal cancers: Tangible achievements, novel acquisitions and future perspectives. Oncotarget 2016, 7, 69060–69074. [Google Scholar] [CrossRef] [Green Version]
- Son, J.; Jang, J.; Beyett, T.S.; Eum, Y.; Haikala, H.M.; Verano, A.; Lin, M.; Hatcher, J.M.; Kwiatkowski, N.P.; Eser, P.Ö.; et al. A Novel HER2-Selective Kinase Inhibitor Is Effective in HER2 Mutant and Amplified Non-Small Cell Lung Cancer. Cancer Res. 2022, 82, 1633–1645. [Google Scholar] [CrossRef]
- Mitani, S.; Kawakami, H. Emerging Targeted Therapies for HER2 Positive Gastric Cancer That Can Overcome Trastuzumab Resistance. Cancers 2020, 12, 400. [Google Scholar] [CrossRef] [Green Version]
- Duchnowska, R.; Loibl, S.; Jassem, J. Tyrosine kinase inhibitors for brain metastases in HER2-positive breast cancer. Cancer Treat. Rev. 2018, 67, 71–77. [Google Scholar] [CrossRef]
- Ling, Y.; Xie, Q.; Zhang, Z.; Zhang, H. Protein kinase inhibitors for acute leukemia. Biomark. Res. 2018, 6, 8. [Google Scholar] [CrossRef] [Green Version]
- An, X.; Tiwari, A.K.; Sun, Y.; Ding, P.-R.; Ashby, C.R.; Chen, Z.-S. BCR-ABL tyrosine kinase inhibitors in the treatment of Philadelphia chromosome positive chronic myeloid leukemia: A review. Leuk. Res. 2010, 34, 1255–1268. [Google Scholar] [CrossRef]
- Lee, H.; Basso, I.N.; Kim, D.D.H. Target spectrum of the BCR-ABL tyrosine kinase inhibitors in chronic myeloid leukemia. Int. J. Hematol. 2021, 113, 632–641. [Google Scholar] [CrossRef]
- Osborne, J.P.; Fryer, A.; Webb, D. Epidemiology of Tuberous Sclerosis. Ann. New York Acad. Sci. 1991, 615, 125–127. [Google Scholar] [CrossRef]
- Curatolo, P.; Bombardieri, R.; Jozwiak, S. Tuberous sclerosis. Lancet 2008, 372, 657–668. [Google Scholar]
- DiMario, F.J. Brain Abnormalities in Tuberous Sclerosis Complex. J. Child Neurol. 2004, 19, 650–657. [Google Scholar] [CrossRef]
- Luat, A.F.; Makki, M.; Chugani, H.T. Neuroimaging in tuberous sclerosis complex. Curr. Opin. Neurol. 2007, 20, 142–150. [Google Scholar] [CrossRef]
- Islam, M.P.; Roach, E.S. Tuberous sclerosis complex. Handb. Clin. Neurol. 2015, 132, 97–109. [Google Scholar]
- Kaczorowska, M.; Jurkiewicz, E.; Domańska-Pakieła, D.; Syczewska, M.; Lojszczyk, B.; Chmielewski, D.; Kotulska, K.; Kuczyński, D.; Kmieć, T.; Dunin-Wąsowicz, D.; et al. Cerebral tuber count and its impact on mental outcome of patients with tuberous sclerosis complex. Epilepsia 2011, 52, 22–27. [Google Scholar] [CrossRef]
- Capal, J.K.; Bernardino-Cuesta, B.; Horn, P.S.; Murray, D.; Byars, A.W.; Bing, N.M.; Kent, B.; Pearson, D.A.; Sahin, M.; Krueger, D.A. Influence of seizures on early development in tuberous sclerosis complex. Epilepsy Behav. 2017, 70, 245–252. [Google Scholar] [CrossRef] [Green Version]
- Jansen, F.E.; Vincken, K.L.; Algra, A.; Anbeek, P.; Braams, O.; Nellist, M.; Zonnenberg, B.A.; Jennekens-Schinkel, A.; Ouweland, A.V.D.; Halley, D.; et al. Cognitive impairment in tuberous sclerosis complex is a multifactorial condition. Neurology 2007, 70, 916–923. [Google Scholar] [CrossRef]
- Ehninger, D.; Sano, Y.; De Vries, P.J.; Dies, K.; Franz, D.; Geschwind, D.H.; Kaur, M.; Lee, Y.-S.; Li, W.; Lowe, J.K.; et al. Gestational immune activation and Tsc2 haploinsufficiency cooperate to disrupt fetal survival and may perturb social behavior in adult mice. Mol. Psychiatry 2010, 17, 62–70. [Google Scholar] [CrossRef]
- DiMario, F.J.; Sahin, M.; Ebrahimi-Fakhari, D. Tuberous sclerosis complex. Pediatr. Clin. N. Am. 2015, 62, 633–648. [Google Scholar]
- Franz, D.N. Non-Neurologic Manifestations of Tuberous Sclerosis Complex. J. Child Neurol. 2004, 19, 690–698. [Google Scholar] [CrossRef]
- Józwiak, S.; Schwartz, R.A.; Janniger, C.K.; Michałowicz, R.; Chmielik, J. Skin lesions in children with tuberous sclerosis complex: Their prevalence, natural course, and diagnostic significance. Int. J. Dermatol. 1998, 37, 911–917. [Google Scholar] [CrossRef]
- Webb, D.W.; Clarke, A.; Fryer, A.; Osborne, J.P. The cutaneous features of tuberous sclerosis: A population study. Br. J. Dermatol. 1996, 135, 1–5. [Google Scholar]
- Hyman, M.H.; Whittemore, V.H. National Institutes of Health Consensus Conference: Tuberous Sclerosis Complex. Arch. Neurol. 2000, 57, 662–665. [Google Scholar] [CrossRef]
- Cudzilo, C.J.; Szczesniak, R.D.; Brody, A.S.; Rattan, M.S.; Krueger, D.A.; Bissler, J.J.; Franz, D.N.; McCormack, F.X.; Young, L.R. Lymphangioleiomyomatosis Screening in Women with Tuberous Sclerosis. Chest 2013, 144, 578–585. [Google Scholar] [CrossRef]
- Wu, S.; Wu, F.; Jiang, Z. Identification of hub genes, key miRNAs and potential molecular mechanisms of colorectal cancer. Oncol. Rep. 2017, 38, 2043–2050. [Google Scholar] [CrossRef] [Green Version]
- Van Slegtenhorst, M.; de Hoogt, R.; Hermans, C.; Nellist, M.; Janssen, B.; Verhoef, S.; Lindhout, D.; van den Ouweland, A.; Halley, D.; Young, J.; et al. Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science 1997, 277, 805–808. [Google Scholar] [CrossRef]
- Sancak, O.; Nellist, M.; Goedbloed, M.; Elfferich, P.; Wouters, C.; Maat-Kievit, A.; Zonnenberg, B.; Verhoef, S.; Halley, D.; Ouweland, A.V.D. Mutational analysis of the TSC1 and TSC2 genes in a diagnostic setting: Genotype—Phenotype correlations and comparison of diagnostic DNA techniques in Tuberous Sclerosis Complex. Eur. J. Hum. Genet. 2005, 13, 731–741. [Google Scholar] [CrossRef] [Green Version]
- Cheadle, J.P.; Reeve, M.P.; Sampson, J.R.; Kwiatkowski, D.J. Molecular genetic advances in tuberous sclerosis. Hum. Genet. 2000, 107, 97–114. [Google Scholar] [CrossRef]
- Kwiatkowski, D.J.; Manning, B.D. Molecular Basis of Giant Cells in Tuberous Sclerosis Complex. N. Engl. J. Med. 2014, 371, 778–780. [Google Scholar] [CrossRef]
- Dibble, C.C.; Cantley, L.C. Regulation of mTORC1 by PI3K signaling. Trends Cell Biol. 2015, 25, 545–555. [Google Scholar] [CrossRef] [Green Version]
- Tee, A.R.; Fingar, D.C.; Manning, B.D.; Kwiatkowski, D.J.; Cantley, L.C.; Blenis, J. Tuberous sclerosis complex-1 and -2 gene products function together to inhibit mammalian target of rapamycin (mTOR)-mediated downstream signaling. Proc. Natl. Acad. Sci. USA 2002, 99, 13571–13576. [Google Scholar] [CrossRef] [Green Version]
- Crino, P.B. mTOR: A pathogenic signaling pathway in developmental brain malformations. Trends Mol. Med. 2011, 17, 734–742. [Google Scholar] [CrossRef]
- Crino, P.B.; Nathanson, K.L.; Henske, E.P. The tuberous sclerosis complex. N. Engl. J. Med. 2006, 355, 1345–1356. [Google Scholar]
- Huang, J.; Manning, B.D. The TSC1–TSC2 complex: A molecular switchboard controlling cell growth. Biochem. J. 2008, 412, 179–190. [Google Scholar] [CrossRef] [Green Version]
- Ma, L.; Chen, Z.; Erdjument-Bromage, H.; Tempst, P.; Pandolfi, P.P. Phosphorylation and functional inactivation of TSC2 by Erk implications for tuberous sclerosis and cancer pathogenesis. Cell 2005, 121, 179–193. [Google Scholar] [CrossRef] [Green Version]
- Carson, R.P.; Fu, C.; Winzenburger, P.; Ess, K.C. Deletion of Rictor in neural progenitor cells reveals contributions of mTORC2 signaling to tuberous sclerosis complex. Hum. Mol. Genet. 2012, 22, 140–152. [Google Scholar] [CrossRef]
- El-Hashemite, N.; Zhang, H.; Henske, E.P.; Kwiatkowski, D.J. Mutation in TSC2 and activation of mammalian target of ra-pamycin signalling pathway in renal angiomyolipoma. Lancet 2003, 361, 1348–1349. [Google Scholar] [CrossRef]
- Musa, J.; Orth, M.F.; Dallmayer, M.; Baldauf, M.; Pardo, C.; Rotblat, B.; Kirchner, T.; Leprivier, G.; Grünewald, T.G.P. Eu-karyotic initiation factor 4E-binding protein 1 (4E-BP1): A master regulator of mRNA translation involved in tumorigenesis. Oncogene 2016, 35, 4675–4688. [Google Scholar] [CrossRef]
- Zhang, Y.; Gao, X.; Saucedo, L.J.; Ru, B.; Edgar, B.A.; Pan, D. Rheb is a direct target of the tuberous sclerosis tumour suppressor proteins. Nature 2003, 5, 578–581. [Google Scholar] [CrossRef]
- Saxton, R.A.; Sabatini, D.M. mTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960–976, Erratum in Cell 2017, 169, 361–371. [Google Scholar] [CrossRef] [Green Version]
- Jhanwar-Uniyal, M.; Wainwright, J.V.; Mohan, A.L.; Tobias, M.E.; Murali, R.; Gandhi, C.D.; Schmidt, M.H. Diverse signaling mechanisms of mTOR complexes: mTORC1 and mTORC2 in forming a formidable relationship. Adv. Biol. Regul. 2019, 72, 51–62. [Google Scholar] [CrossRef]
- Jossin, Y.; Goffinet, A.M. Reelin Signals through Phosphatidylinositol 3-Kinase and Akt To Control Cortical Development and through mTor To Regulate Dendritic Growth. Mol. Cell. Biol. 2007, 27, 7113–7124. [Google Scholar] [CrossRef] [Green Version]
- Tavazoie, S.F.; Alvarez, V.A.; Ridenour, D.A.; Kwiatkowski, D.J.; Sabatini, B.L. Regulation of neuronal morphology and function by the tumor suppressors Tsc1 and Tsc2. Nat. Neurosci. 2005, 8, 1727–1734. [Google Scholar] [CrossRef]
- Chévere-Torres, I.; Kaphzan, H.; Bhattacharya, A.; Kang, A.; Maki, J.M.; Gambello, M.J.; Arbiser, J.L.; Santini, E.; Klann, E. Metabotropic glutamate receptor-dependent long-term depression is impaired due to elevated ERK signaling in the ΔRG mouse model of tuberous sclerosis complex. Neurobiol. Dis. 2012, 45, 1101–1110. [Google Scholar] [CrossRef] [Green Version]
- Ho, H.; Kapadia, R.; Al-Tahan, S.; Ahmad, S.; Ganesan, A.K. WIPI1 Coordinates Melanogenic Gene Transcription and Mel-anosome Formation via TORC1 Inhibition. J. Biol. Chem. 2011, 286, 12509–12523. [Google Scholar] [CrossRef] [Green Version]
- Vézina, C.; Kudelski, A.; Sehgal, S.N. Rapamycin (AY-22,989), a new antifungal antibiotic. I. Taxonomy of the producing streptomycete and isolation of the active principle. J. Antibiot. 1975, 28, 721–726. [Google Scholar] [CrossRef]
- Franz, D.N.; Krueger, D.A. mTOR inhibitor therapy as a disease modifying therapy for tuberous sclerosis complex. Am. J. Med. Genet. Part C Semin. Med. Genet. 2018, 178, 365–373. [Google Scholar] [CrossRef]
- Krueger, D.A.; Care, M.M.; Holland, K.; Agricola, K.; Tudor, C.; Mangeshkar, P.; Wilson, K.A.; Byars, A.; Sahmoud, T.; Franz, D.N. Everolimus for Subependymal Giant-Cell Astrocytomas in Tuberous Sclerosis. N. Engl. J. Med. 2010, 363, 1801–1811. [Google Scholar] [CrossRef]
- Franz, D.N.; Belousova, E.; Sparagana, S.; Bebin, E.M.; Frost, M.; Kuperman, R.; Witt, O.; Kohrman, M.H.; Flamini, J.R.; Wu, J.Y.; et al. Everolimus for subependymal giant cell astrocytoma in patients with tuberous sclerosis complex: 2-year open-label extension of the randomised EXIST-1 study. Lancet Oncol. 2014, 15, 1513–1520. [Google Scholar] [CrossRef]
- French, J.A.; Lawson, J.; Yapici, Z.; Ikeda, H.; Polster, T.; Nabbout, R.; Curatolo, P.; de Vries, P.J.; Dlugos, D.J.; Berkowitz, N.; et al. Adjunctive everolimus therapy for treatment-resistant focal-onset seizures associated with tuberous sclerosis (EXIST-3): A phase 3, randomised, double-blind, placebo-controlled study. Lancet 2016, 388, 2153–2163. [Google Scholar] [CrossRef] [Green Version]
- Bissler, J.J.; McCormack, F.X.; Young, L.R.; Elwing, J.M.; Chuck, G.; Leonard, J.M.; Schmithorst, V.J.; Laor, T.; Brody, A.S.; Bean, J.; et al. Sirolimus for Angiomyolipoma in Tuberous Sclerosis Complex or Lymphangioleiomyomatosis. N. Engl. J. Med. 2008, 358, 140–151. [Google Scholar] [CrossRef] [Green Version]
- McCormack, F.X.; Inoue, Y.; Moss, J.; Singer, L.G.; Strange, C.; Nakata, K.; Barker, A.F.; Chapman, J.T.; Brantly, M.L.; Stocks, J.M.; et al. Efficacy and Safety of Sirolimus in Lymphangioleiomyomatosis. N. Engl. J. Med. 2011, 364, 1595–1606. [Google Scholar] [CrossRef]
- Sugalska, M.; Tomik, A.; Jóźwiak, S.; Werner, B. Treatment of Cardiac Rhabdomyomas with mTOR Inhibitors in Children with Tuberous Sclerosis Complex—A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 4907. [Google Scholar] [CrossRef]
- Mlczoch, E.; Hanslik, A.; Luckner, D.; Kitzmüller, E.; Prayer, D.; Michel-Behnke, I. Prenatal diagnosis of giant cardiac rhab-domyoma in tuberous sclerosis complex: A new therapeutic option with everolimus. Ultrasound Obstet. Gynecol. 2014, 45, 618–621. [Google Scholar] [CrossRef]
- Wienecke, R.; Fackler, I.; Linsenmaier, U.; Mayer, K.; Licht, T.; Kretzler, M. Antitumoral Activity of Rapamycin in Renal An-giomyolipoma Associated With Tuberous Sclerosis Complex. Am. J. Kidney Dis. 2006, 48, e27–e29. [Google Scholar] [CrossRef]
- Bissler, J.J.; Kingswood, J.C.; Radzikowska, E.; Zonnenberg, B.A.; Frost, M.; Belousova, E.; Sauter, M.; Nonomura, N.; Brakemeier, S.; de Vries, P.J.; et al. Everolimus for angiomyolipoma associated with tuberous sclerosis complex or sporadic lymphangioleiomyomatosis (EX-IST-2): A multicentre, randomised, double-blind, placebo-controlled trial. Lancet 2013, 381, 817–824. [Google Scholar] [CrossRef]
- Siroky, B.J.; Towbin, A.J.; Trout, A.T.; Schäfer, H.; Thamann, A.R.; Agricola, K.D.; Tudor, C.; Capal, J.; Dixon, B.P.; Krueger, D.A.; et al. Improvement in Renal Cystic Disease of Tuberous Sclerosis Complex After Treatment with Mammalian Target of Rapamycin Inhibitor. J. Pediatr. 2017, 187, 318–322.e2. [Google Scholar] [CrossRef]
- Rodrik-Outmezguine, V.S.; Okaniwa, M.; Yao, Z.; Novotny, C.J.; McWhirter, C.; Banaji, A.; Won, H.; Wong, W.; Berger, M.; De Stanchina, E.; et al. Overcoming mTOR resistance mutations with a new-generation mTOR inhibitor. Nature 2016, 534, 272–276. [Google Scholar] [CrossRef] [Green Version]
- Schadendorf, D.; Hauschild, A. Melanoma in 2013: Melanoma--the run of success continues. Nat. Rev. Clin. Oncol. 2014, 11, 75–76. [Google Scholar] [CrossRef]
- Shao, Z.; Bao, Q.; Jiang, F.; Qian, H.; Fang, Q.; Hu, X. VS-5584, a Novel PI3K-mTOR Dual Inhibitor, Inhibits Melanoma Cell Growth In Vitro and In Vivo. PLoS ONE 2015, 10, e0132655. [Google Scholar] [CrossRef]
- Webster, R.M.; Mentzer, S.E. The malignant melanoma landscape. Nat. Rev. Drug Discov. 2014, 13, 491–492. [Google Scholar] [CrossRef]
- Wang, J.; Yang, Z.; Wen, J.; Ma, F.; Wang, F.; Yu, K.; Tang, M.; Wu, W.; Dong, Y.; Cheng, X.; et al. SKLB-M8 induces apoptosis through the AKT/mTOR signaling pathway in melanoma models and inhibits angiogenesis with de-crease of ERK1/2 phosphorylation. J. Pharmacol. Sci. 2014, 126, 198–207. [Google Scholar] [CrossRef] [Green Version]
- Rauen, K.A. The RASopathies. Annu. Rev. Genom. Hum. Genet. 2013, 14, 355–369. [Google Scholar] [CrossRef] [Green Version]
- Bergqvist, C.; Network, N.F.; Servy, A.; Valeyrie-Allanore, L.; Ferkal, S.; Combemale, P.; Wolkenstein, P. Neurofibromatosis 1 French national guidelines based on an extensive literature review since 1966. Orphanet J. Rare Dis. 2020, 15, 37. [Google Scholar] [CrossRef] [Green Version]
- Darrigo, L.G.; Geller, M.; Bonalumi Filho, A.; Azulay, D.R. Prevalence of plexiform neurofibroma in children and ado-lescents with type I neurofibromatosis. J. Pediatr. (Rio J.) 2007, 83, 571–573. [Google Scholar]
- Gross, A.M.; Singh, G.; Akshintala, S.; Baldwin, A.; Dombi, E.; Ukwuani, S.; Goodwin, A.; Liewehr, D.J.; Steinberg, S.M.; Widemann, B.C. Association of plexiform neurofibroma volume changes and development of clinical morbidities in neurofibromatosis 1. Neuro-Oncology 2018, 20, 1643–1651. [Google Scholar] [CrossRef]
- Nguyen, R.; Kluwe, L.; Fuensterer, C.; Kentsch, M.; Friedrich, R.E.; Mautner, V.-F. Plexiform neurofibromas in children with neurofibromatosis type 1: Frequency and associated clinical deficits. J. Pediatr. 2011, 159, 652–655.e2. [Google Scholar] [CrossRef]
- Weiss, B.; Bollag, G.; Shannon, K. Hyperactive Ras as a therapeutic target in neurofibromatosis type 1. Am. J. Med. Genet. 1999, 89, 14–22. [Google Scholar] [CrossRef]
- Tartaglia, M.; Zampino, G.; Gelb, B. Noonan Syndrome: Clinical Aspects and Molecular Pathogenesis. Mol. Syndr. 2010, 1, 2–26. [Google Scholar] [CrossRef] [Green Version]
- Tartaglia, M.; Mehler, E.L.; Goldberg, R.; Zampino, G.; Brunner, H.G.; Kremer, H.; Van Der Burgt, I.; Crosby, A.H.; Ion, A.; Jeffery, S.; et al. Mutations in PTPN11, encoding the protein tyrosine phosphatase SHP-2, cause Noonan syndrome. Nat. Genet. 2001, 29, 465–468. [Google Scholar] [CrossRef]
- Matozaki, T.; Murata, Y.; Saito, Y.; Okazawa, H.; Ohnishi, H. Protein tyrosine phosphatase SHP-2: A proto-oncogene product that promotes Ras activation. Cancer Sci. 2009, 100, 1786–1793. [Google Scholar] [CrossRef]
- Quilliam, L.A.; Rebhun, J.F.; Castro, A.F. A growing family of guanine nucleotide exchange factors is responsible for activation of ras-family GTPases. In Progress in Nucleic Acid Research and Molecular Biology; Academic Press: Cambridge, MA, USA, 2002; Volume 71, pp. 391–444. [Google Scholar]
- Tartaglia, M.; Pennacchio, L.; Zhao, C.; Yadav, K.K.; Fodale, V.; Sarkozy, A.; Pandit, B.; Oishi, K.; Martinelli, S.; Schackwitz, W.; et al. Gain-of-function SOS1 mutations cause a distinctive form of Noonan syndrome. Nat. Genet. 2006, 39, 75–79. [Google Scholar] [CrossRef]
- Gremer, L.; Merbitz-Zahradnik, T.; Dvorsky, R.; Cirstea, I.C.; Kratz, C.P.; Zenker, M.; Wittinghofer, A.; Ahmadian, M.R. Germline KRAS mutations cause aberrant biochemical and physical properties leading to developmental disorders. Hum. Mutat. 2010, 32, 33–43. [Google Scholar] [CrossRef] [Green Version]
- Schubbert, S.; Bollag, G.; Lyubynska, N.; Nguyen, H.; Kratz, C.P.; Zenker, M.; Niemeyer, C.M.; Molven, A.; Shannon, K. Bi-ochemical and Functional Characterization of Germ Line KRAS Mutations. Mol. Cell. Biol. 2007, 27, 7765–7770. [Google Scholar] [CrossRef] [Green Version]
- Pandit, B.; Sarkozy, A.; Pennacchio, L.A.; Carta, C.; Oishi, K.; Martinelli, S.; Pogna, E.A.; Schackwitz, W.; Ustaszewska, A.; Landstrom, A.; et al. Gain-of-function RAF1 mutations cause Noonan and LEOPARD syndromes with hypertrophic cardiomyo-pathy. Nat. Genet. 2007, 39, 1007–1012. [Google Scholar] [CrossRef]
- Mutation Analysis of the SHOC2 Gene in Noonan-Like Syndrome and in Hematologic Malignancies|Journal of Human Ge-netics. Available online: https://www.nature.com/articles/jhg2010116 (accessed on 13 January 2023).
- Martinelli, S.; De Luca, A.; Stellacci, E.; Rossi, C.; Checquolo, S.; Lepri, F.; Caputo, V.; Silvano, M.; Buscherini, F.; Consoli, F.; et al. Heterozygous Germline Mutations in the CBL Tumor-Suppressor Gene Cause a Noonan Syndrome-like Phenotype. Am. J. Hum. Genet. 2010, 87, 250–257. [Google Scholar] [CrossRef] [Green Version]
- Sarkozy, A.; Digilio, M.C.; Dallapiccola, B. Leopard syndrome. Orphanet. J. Rare Dis. 2008, 3, 13. [Google Scholar] [CrossRef] [Green Version]
- Gripp, K.W.; Morse, L.A.; Axelrad, M.; Chatfield, K.C.; Chidekel, A.; Dobyns, W.; Doyle, D.; Kerr, B.; Lin, A.E.; Schwartz, D.D.; et al. Costello syn-drome: Clinical phenotype, genotype, and management guidelines. Am. J. Med. Genet. Part A 2019, 179, 1725–1744. [Google Scholar] [CrossRef]
- Zenker, M. Noonan Syndrome and Related Disorders: A Matter of Deregulated Ras Signaling; Karger Medical and Scientific Publishers: Basel, Switzerland, 2009. [Google Scholar]
- Nava, C.; Hanna, N.; Michot, C.; Pereira, S.; Pouvreau, N.; Niihori, T.; Aoki, Y.; Matsubara, Y.; Arveiler, B.; Lacombe, D.; et al. Cardio-facio-cutaneous and Noonan syndromes due to mutations in the RAS/MAPK signalling pathway: Genotype phenotype relationships and overlap with Costello syndrome. J. Med. Genet. 2007, 44, 763–771. [Google Scholar] [CrossRef] [Green Version]
- Ordan, M.; Pallara, C.; Maik-Rachline, G.; Hanoch, T.; Gervasio, F.L.; Glaser, F.; Fernandez-Recio, J.; Seger, R. Intrinsically active MEK variants are differentially regulated by proteinases and phosphatases. Sci. Rep. 2018, 8, 11830. [Google Scholar] [CrossRef] [Green Version]
- Eerola, I.; Boon, L.M.; Mulliken, J.B.; Burrows, P.E.; Dompmartin, A.; Watanabe, S.; Vanwijck, R.; Vikkula, M. Capillary Malformation–Arteriovenous Malformation, a New Clinical and Genetic Disorder Caused by RASA1 Mutations. Am. J. Hum. Genet. 2003, 73, 1240–1249. [Google Scholar] [CrossRef] [Green Version]
- Pasmant, E.; Sabbagh, A.; Hanna, N.; Masliah-Planchon, J.; Jolly, E.; Goussard, P.; Ballerini, P.; Cartault, F.; Barbarot, S.; Landman-Parker, J.; et al. SPRED1 germline mutations caused a neurofibromatosis type 1 overlapping phenotype. J. Med. Genet. 2009, 46, 425–430. [Google Scholar] [CrossRef] [Green Version]
- Shannon, K.M.; O’Connell, P.; Martin, G.A.; Paderanga, D.; Olson, K.; Dinndorf, P.; McCormick, F. Loss of The Normal NF1 Allele from the Bone Marrow of Children with Type 1 Neurofibromatosis and Malignant Myeloid Disorders. N. Engl. J. Med. 1994, 330, 597–601. [Google Scholar] [CrossRef]
- Wallace, M.R.; Marchuk, D.A.; Andersen, L.B.; Letcher, R.; Odeh, H.M.; Saulino, A.M.; Fountain, J.W.; Brereton, A.; Nicholson, J.; Mitchell, A.L.; et al. Type 1 neurofibromatosis gene: Identification of a large transcript disrupted in three NF1 patients. Science 1990, 249, 181–186. [Google Scholar] [CrossRef]
- Viskochil, D.; Buchberg, A.M.; Xu, G.; Cawthon, R.M.; Stevens, J.; Wolff, R.K.; Culver, M.; Carey, J.C.; Copeland, N.G.; Jenkins, N.A.; et al. Deletions and a translocation interrupt a cloned gene at the neurofibromatosis type 1 locus. Cell 1990, 62, 187–192. [Google Scholar] [CrossRef]
- Gutmann, D.H.; Ferner, R.E.; Listernick, R.H.; Korf, B.R.; Wolters, P.L.; Johnson, K.J. Neurofibromatosis type 1. Nat. Rev. Dis. Prim. 2017, 3, 17004. [Google Scholar] [CrossRef]
- Sarkozy, A.; Conti, E.; Digilio, M.C.; Marino, B.; Morini, E.; Pacileo, G.; Wilson, M.; Calabrò, R.; Pizzuti, A.; Dallapiccola, B. Clinical and molecular analysis of 30 patients with multiple lentigines LEOPARD syndrome. J. Med. Genet. 2004, 41, e68. [Google Scholar] [CrossRef] [Green Version]
- Widemann, B.C.; Babovic-Vuksanovic, D.; Dombi, E.; Wolters, P.L.; Goldman, S.; Martin, S.; Goodwin, A.; Goodspeed, W.; Kieran, M.W.; Cohen, B.; et al. Phase II trial of pirfenidone in children and young adults with neurofibromatosis type 1 and progressive plexiform neurofibromas. Pediatr. Blood Cancer 2014, 61, 1598–1602. [Google Scholar] [CrossRef]
- Widemann, B.C.; Salzer, W.L.; Arceci, R.J.; Blaney, S.M.; Fox, E.; End, D.; Gillespie, A.; Whitcomb, P.; Palumbo, J.S.; Pitney, A.; et al. Phase I Trial and Pharmacokinetic Study of the Farnesyltransferase Inhibitor Tipifarnib in Children With Refractory Solid Tumors or Neurofibromatosis Type I and Plexiform Neurofibromas. J. Clin. Oncol. 2006, 24, 507–516. [Google Scholar] [CrossRef]
- Widemann, B.C.; Dombi, E.; Gillespie, A.; Wolters, P.L.; Belasco, J.; Goldman, S.; Korf, B.R.; Solomon, J.; Martin, S.; Salzer, W.; et al. Phase 2 ran-domized, flexible crossover, double-blinded, placebo-controlled trial of the farnesyltransferase inhibitor tipi-farnib in children and young adults with neurofibromatosis type 1 and progressive plexiform neurofibromas. Neuro Oncol. 2014, 16, 707–718. [Google Scholar] [CrossRef] [Green Version]
- Weiss, B.; Widemann, B.C.; Wolters, P.; Dombi, E.; Vinks, A.; Cantor, A.; Perentesis, J.; Schorry, E.; Ullrich, N.; Gutmann, D.H.; et al. Sirolimus for progressive neurofibromatosis type 1-associated plexiform neurofibromas: A Neurofibromatosis Clinical Trials Consortium phase II study. Neuro-Oncology 2014, 17, 596–603. [Google Scholar] [CrossRef] [Green Version]
- Weiss, B.; Widemann, B.C.; Wolters, P.; Dombi, E.; Vinks, A.A.; Cantor, A.; Korf, B.; Perentesis, J.; Gutmann, D.H.; Schorry, E.; et al. Sirolimus for non-progressive NF1-associated plexiform neurofibromas: An NF clinical trials consortium phase II study. Pediatr. Blood Cancer 2013, 61, 982–986. [Google Scholar] [CrossRef]
- Jakacki, R.I.; Dombi, E.; Potter, D.M.; Goldman, S.; Allen, J.C.; Pollack, I.F.; Widemann, B.C. Phase I trial of pegylated interferon- -2b in young patients with plexiform neurofibromas. Neurology 2011, 76, 265–272. [Google Scholar] [CrossRef] [Green Version]
- Jakacki, R.I.; Dombi, E.; Steinberg, S.M.; Goldman, S.; Kieran, M.W.; Ullrich, N.J.; Pollack, I.F.; Goodwin, A.; Manley, P.E.; Fangusaro, J.; et al. Phase II trial of pegylated interferon alfa-2b in young patients with neu-rofibromatosis type 1 and unresectable plexiform neurofibromas. Neuro Oncol. 2017, 19, 289–297. [Google Scholar]
- Robertson, K.A.; Nalepa, G.; Yang, F.-C.; Bowers, D.C.; Ho, C.Y.; Hutchins, G.D.; Croop, J.M.; Vik, T.A.; Denne, S.C.; Parada, L.F.; et al. Imatinib mesylate for plexiform neurofibromas in patients with neurofibromatosis type 1: A phase 2 trial. Lancet Oncol. 2012, 13, 1218–1224. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, A.; Jakacki, R.I.; Onar-Thomas, A.; Wu, S.; Nicolaides, T.; Poussaint, T.Y.; Fangusaro, J.; Phillips, J.; Perry, A.; Turner, D.; et al. A phase I trial of the MEK inhibitor selumetinib (AZD6244) in pediatric patients with recurrent or refractory low-grade glioma: A Pediatric Brain Tumor Consortium (PBTC) study. Neuro-Oncology 2017, 19, 1135–1144. [Google Scholar] [CrossRef] [Green Version]
- Dombi, E.; Baldwin, A.; Marcus, L.J.; Fisher, M.J.; Weiss, B.; Kim, A.; Whitcomb, P.; Martin, S.; Aschbacher-Smith, L.E.; Rizvi, T.A.; et al. Activity of Selumetinib in Neurofibromatosis Type 1–Related Plexiform Neurofibromas. N. Engl. J. Med. 2016, 375, 2550–2560. [Google Scholar] [CrossRef]
- Gross, A.; Bishop, R.; Widemann, B.C. Selumetinib in Plexiform Neurofibromas. N. Engl. J. Med. 2017, 376, 1195. [Google Scholar]
- Plotkin, S.R.; Blakeley, J.O.; Dombi, E.; Fisher, M.J.; Hanemann, C.O.; Walsh, K.; Wolters, P.L.; Widemann, B.C. Achieving consensus for clinical trials: The REiNS International Collaboration. Neurology 2013, 81, S1–S5. [Google Scholar] [CrossRef] [Green Version]
- McCowage, G.B.; Mueller, S.; Pratilas, C.A.; Hargrave, D.R.; Moertel, C.L.; Whitlock, J.; Fox, E.; Hingorani, P.; Russo, M.W.; Dasgupta, K.; et al. Trametinib in pediatric patients with neurofibromatosis type 1 (NF-1)–associated plexiform neurofibroma: A phase I/IIa study. J. Clin. Oncol. 2018, 36, 10504. [Google Scholar] [CrossRef]
- Listernick, R.; Charrow, J.; Gutmann, D.H. Intracranial gliomas in neurofibromatosis type 1. Am. J. Med. Genet. 1999, 89, 38–44. [Google Scholar] [CrossRef]
- Lewis, R.A.; Gerson, L.P.; Axelson, K.A.; Riccardi, V.M.; Whitford, R.P. von Recklinghausen neurofibromatosis. II. Incidence of optic gliomata. Ophthalmology 1984, 91, 929–935. [Google Scholar] [CrossRef]
- Packer, R.J.; Lange, B.; Ater, J.; Nicholson, H.S.; Allen, J.; Walker, R.; Prados, M.; Jakacki, R.; Reaman, G.; Needles, M.N. Carboplatin and vincristine for recurrent and newly diagnosed low-grade gliomas of childhood. J. Clin. Oncol. 1993, 11, 850–856. [Google Scholar] [CrossRef]
- Listernick, R.; Ferner, R.E.; Liu, G.T.; Gutmann, D.H. Optic pathway gliomas in neurofibromatosis-1: Controversies and rec-ommendations. Ann. Neurol. 2007, 61, 189–198. [Google Scholar] [CrossRef] [Green Version]
- Kondyli, M.; Larouche, V.; Saint-Martin, C.; Ellezam, B.; Pouliot, L.; Sinnett, D.; Legault, G.; Crevier, L.; Weil, A.; Farmer, J.-P.; et al. Trametinib for progressive pediatric low-grade gliomas. J. Neuro-Oncology 2018, 140, 435–444. [Google Scholar] [CrossRef]
- Romo, C.; Slobogean, B.; Blair, L.; O Blakeley, J. RARE-54. MEK INHIBITION FOR AGGRESSIVE GLIOMAS IN ADULTS WITH NEUROFIBROMATOSIS TYPE 1. Neuro-Oncology 2019, 21, vi233. [Google Scholar] [CrossRef]
- Rauen, K.A.; Alsaegh, A.; Ben-Shachar, S.; Berman, Y.; Blakeley, J.; Cordeiro, I.; Elgersma, Y.; Evans, D.G.; Fisher, M.J.; Frayling, I.M.; et al. First International Conference on RASopathies and Neurofibromatoses in Asia: Identification and advances of new therapeutics. Am. J. Med. Genet. Part A 2019, 179, 1091–1097. [Google Scholar] [CrossRef]
- Inoue, S.-I.; Moriya, M.; Watanabe, Y.; Miyagawa-Tomita, S.; Niihori, T.; Oba, D.; Ono, M.; Kure, S.; Ogura, T.; Matsubara, Y.; et al. New BRAF knockin mice provide a pathogenetic mechanism of developmental defects and a therapeutic approach in cardio-facio-cutaneous syndrome. Hum. Mol. Genet. 2014, 23, 6553–6566. [Google Scholar] [CrossRef]
- Andelfinger, G.; Marquis, C.; Raboisson, M.-J.; Théoret, Y.; Waldmüller, S.; Wiegand, G.; Gelb, B.D.; Zenker, M.; Delrue, M.-A.; Hofbeck, M. Hypertrophic Cardiomyopathy in Noonan Syndrome Treated by MEK-Inhibition. J. Am. Coll. Cardiol. 2019, 73, 2237–2239. [Google Scholar] [CrossRef]
- Waters, A.M.; Beales, P.L. Ciliopathies: An expanding disease spectrum. Pediatr. Nephrol. 2011, 26, 1039–1056. [Google Scholar] [CrossRef] [Green Version]
- Oud, M.M.; Lamers, I.J.C.; Arts, H.H. Ciliopathies: Genetics in Pediatric Medicine. J. Pediatr. Genet. 2016, 6, 018–029. [Google Scholar] [CrossRef] [Green Version]
- Khayyeri, H.; Barreto, S.; Lacroix, D. Primary cilia mechanics affects cell mechanosensation: A computational study. J. Theor. Biol. 2015, 379, 38–46. [Google Scholar] [CrossRef]
- Sonkusare, S.K.; Bonev, A.D.; Ledoux, J.; Liedtke, W.; Kotlikoff, M.I.; Heppner, T.J.; Hill-Eubanks, D.C.; Nelson, M.T. Elementary Ca2+ Signals Through Endothelial TRPV4 Channels Regulate Vascular Function. Science 2012, 336, 597–601. [Google Scholar] [CrossRef] [Green Version]
- Feather, S.A.; Winyard, P.J.; Dodd, S.; Woolf, A. Oral-facial-digital syndrome type 1 is another dominant polycystic kidney disease: Clinical, radiological and histopathological features of a new kindred. Nephrol. Dial. Transplant. 1997, 12, 1354–1361. [Google Scholar] [CrossRef] [Green Version]
- Beales, P.L.; Elcioglu, N.; Woolf, A.S.; Parker, D.; Flinter, F.A. New criteria for improved diagnosis of Bardet-Biedl syn-drome: Results of a population survey. J. Med. Genet. 1999, 36, 437–446. [Google Scholar] [CrossRef]
- Tobin, J.L.; Di Franco, M.; Eichers, E.; May-Simera, H.; Garcia, M.; Yan, J.; Quinlan, R.; Justice, M.J.; Hennekam, R.C.; Briscoe, J.; et al. Inhibition of neural crest migration underlies craniofacial dysmorphology and Hirschsprung’s disease in Bardet–Biedl syndrome. Proc. Natl. Acad. Sci. USA 2008, 105, 6714–6719. [Google Scholar] [CrossRef] [Green Version]
- Brancati, F.; Italy, U.D.N.; Camerota, L.; Colao, E.; Vega-Warner, V.; Zhao, X.; Zhang, R.; Bottillo, I.; Castori, M.; Caglioti, A.; et al. Biallelic variants in the ciliary gene TMEM67 cause RHYNS syndrome. Eur. J. Hum. Genet. 2018, 26, 1266–1271. [Google Scholar] [CrossRef]
- Bielas, S.L.; Silhavy, J.L.; Brancati, F.; Kisseleva, M.V.; Al-Gazali, L.; Sztriha, L.; Bayoumi, R.A.; Zaki, M.S.; Abdel-Aleem, A.; Rosti, R.O.; et al. Mutations in INPP5E, encoding inositol polyphosphate-5-phosphatase, E.; link phosphatidyl inositol signaling to the ciliopathies. Nat. Genet. 2009, 41, 1032–1036. [Google Scholar] [CrossRef] [Green Version]
- Cantagrel, V.; Silhavy, J.L.; Bielas, S.L.; Swistun, D.; Marsh, S.E.; Bertrand, J.Y.; Audollent, S.; Attié-Bitach, T.; Holden, K.R.; Dobyns, W.B.; et al. Mutations in the Cilia Gene ARL13B Lead to the Classical Form of Joubert Syndrome. Am. J. Hum. Genet. 2008, 83, 170–179. [Google Scholar] [CrossRef] [Green Version]
- Noor, A.; Windpassinger, C.; Patel, M.; Stachowiak, B.; Mikhailov, A.; Azam, M.; Irfan, M.; Siddiqui, Z.K.; Naeem, F.; Paterson, A.; et al. CC2D2A, Encoding A Coiled-Coil and C2 Domain Protein, Causes Autosomal-Recessive Mental Retardation with Retinitis Pigmentosa. Am. J. Hum. Genet. 2008, 82, 1011–1018. [Google Scholar] [CrossRef] [Green Version]
- Delous, M.; Baala, L.; Salomon, R.; Laclef, C.; Vierkotten, J.; Tory, K.; Golzio, C.; Lacoste, T.; Besse, L.; Ozilou, C.; et al. The ciliary gene RPGRIP1L is mutated in cerebello-oculo-renal syndrome (Joubert syndrome type B) and Meckel syndrome. Nat. Genet. 2007, 39, 875–881. [Google Scholar] [CrossRef]
- Baala, L.; Romano, S.; Khaddour, R.; Saunier, S.; Smith, U.M.; Audollent, S.; Ozilou, C.; Faivre, L.; Laurent, N.; Foliguet, B.; et al. The Meckel-Gruber Syndrome Gene, MKS3, Is Mutated in Joubert Syndrome. Am. J. Hum. Genet. 2007, 80, 186–194. [Google Scholar] [CrossRef] [Green Version]
- Parisi, M.A.; Bennett, C.; Eckert, M.L.; Dobyns, W.; Gleeson, J.G.; Shaw, D.W.; McDonald, R.; Eddy, A.; Chance, P.F.; Glass, I.A. The NPHP1 Gene Deletion Associated with Juvenile Nephronophthisis Is Present in a Subset of Individuals with Joubert Syndrome. Am. J. Hum. Genet. 2004, 75, 82–91. [Google Scholar] [CrossRef] [Green Version]
- Dixon-Salazar, T.; Silhavy, J.L.; Marsh, S.E.; Louie, C.M.; Scott, L.C.; Gururaj, A.; Al-Gazali, L.; Al-Tawari, A.A.; Kayserili, H.; Sztriha, L.; et al. Mutations in the AHI1 Gene, Encoding Jouberin, Cause Joubert Syndrome with Cortical Polymicrogyria. Am. J. Hum. Genet. 2004, 75, 979–987. [Google Scholar] [CrossRef] [Green Version]
- Sayer, J.; Otto, E.; O’Toole, J.F.; Nurnberg, G.; Kennedy, M.A.; Becker, C.; Hennies, H.C.; Helou, J.; Attanasio, M.; Fausett, B.V.; et al. The centrosomal protein nephrocystin-6 is mutated in Joubert syndrome and activates transcription factor ATF4. Nat. Genet. 2006, 38, 674–681. [Google Scholar] [CrossRef]
- Coene, K.; Roepman, R.; Doherty, D.; Afroze, B.; Kroes, H.Y.; Letteboer, S.J.; Ngu, L.H.; Budny, B.; van Wijk, E.; Gorden, N.T.; et al. OFD1 Is Mutated in X-Linked Joubert Syndrome and Interacts with LCA5-Encoded Lebercilin. Am. J. Hum. Genet. 2009, 85, 465–481. [Google Scholar] [CrossRef] [Green Version]
- Valente, E.M.; Silhavy, J.L.; Brancati, F.; Barrano, G.; Krishnaswami, S.R.; Castori, M.; Lancaster, M.A.; Boltshauser, E.; Boccone, L.; Al-Gazali, L.; et al. Mutations in CEP290, which encodes a centrosomal protein, cause pleiotropic forms of Joubert syndrome. Nat. Genet. 2006, 38, 623–625. [Google Scholar] [CrossRef]
- Kyttälä, M.; Tallila, J.; Salonen, R.; Kopra, O.; Kohlschmidt, N.; Paavola-Sakki, P.; Peltonen, L.; Kestilä, M. MKS1, encoding a component of the flagellar apparatus basal body proteome, is mutated in Meckel syndrome. Nat. Genet. 2006, 38, 155–157. [Google Scholar] [CrossRef]
- Smith, U.M.; Consugar, M.; Tee, L.J.; McKee, B.M.; Maina, E.N.; Whelan, S.; Morgan, N.; Goranson, E.; Gissen, P.; Lilliquist, S.; et al. The transmembrane protein meckelin (MKS3) is mutated in Meckel-Gruber syndrome and the wpk rat. Nat. Genet. 2006, 38, 191–196. [Google Scholar] [CrossRef]
- Tallila, J.; Jakkula, E.; Peltonen, L.; Salonen, R.; Kestilä, M. Identification of CC2D2A as a Meckel Syndrome Gene Adds an Important Piece to the Ciliopathy Puzzle. Am. J. Hum. Genet. 2008, 82, 1361–1367. [Google Scholar] [CrossRef] [Green Version]
- Caridi, G.; Murer, L.; Bellantuono, R.; Sorino, P.; Caringella, D.; Gusmano, R.; Ghiggeri, G.M. Renal-retinal syndromes: Asso-ciation of retinal anomalies and recessive nephronophthisis in patients with homozygous deletion of the NPH1 locus. Am. J. Kidney Dis. 1998, 32, 1059–1062. [Google Scholar] [CrossRef]
- Omran, H.; Sasmaz, G.D.; Häffner, K.; Volz, A.; Olbrich, H.; Melkaoui, R.; Otto, E.; Wienker, T.F.; Korinthenberg, R.; Brandis, M.; et al. Identification of a Gene Locus for Senior-Løken Syndrome in the Region of the Nephronophthisis Type 3 Gene. J. Am. Soc. Nephrol. 2002, 13, 75–79. [Google Scholar] [CrossRef]
- Schuermann, M.J.; Otto, E.; Becker, A.; Saar, K.; Rüschendorf, F.; Polak, B.C.; Ala-Mello, S.; Hoefele, J.; Wiedensohler, A.; Haller, M.; et al. Mapping of gene loci for nephronophthisis type 4 and Sen-ior-Løken syndrome, to chromosome 1p36. Am. J. Hum. Genet. 2002, 70, 1240–1246. [Google Scholar] [CrossRef] [Green Version]
- Otto, E.; Hoefele, J.; Ruf, R.; Mueller, A.M.; Hiller, K.S.; Wolf, M.T.; Schuermann, M.J.; Becker, A.; Birkenhäger, R.; Sudbrak, R.; et al. A Gene Mutated in Nephronophthisis and Retinitis Pigmentosa Encodes a Novel Protein, Nephroretinin, Conserved in Evolution. Am. J. Hum. Genet. 2002, 71, 1161–1167. [Google Scholar] [CrossRef] [Green Version]
- Ferrante, M.I.; Feather, S.A.; Bulfone, A.; Wright, V.; Ghiani, M.; Selicorni, A.; Gammaro, L.; Scolari, F.; Woolf, A.S.; Sylvie, O.; et al. Identification of the Gene for Oral-Facial-Digital Type I Syndrome. Am. J. Hum. Genet. 2001, 68, 569–576. [Google Scholar] [CrossRef] [Green Version]
- Perrault, I.; Delphin, N.; Hanein, S.; Gerber, S.; Dufier, J.L.; Roche, O.; Defoort-Dhellemmes, S.; Dollfus, H.; Fazzi, E.; Mun-nich, A.; et al. Spectrum of NPHP6/CEP290 mutations in Leber congenital amaurosis and delineation of the associated phenotype. Hum. Mutat. 2007, 28, 416. [Google Scholar] [CrossRef]
- Marlhens, F.; Bareil, C.; Griffoin, J.-M.; Zrenner, E.; Amalric, P.; Eliaou, C.; Liu, S.-Y.; Harris, E.; Redmond, T.M.; Arnaud, B.; et al. Mutations in RPE65 cause Leber’s congenital amaurosis. Nat. Genet. 1997, 17, 139–141. [Google Scholar] [CrossRef]
- Wang, H.; Hollander, A.I.D.; Moayedi, Y.; Abulimiti, A.; Li, Y.; Collin, R.W.; Hoyng, C.B.; Lopez, I.; Bray, M.; Lewis, R.A.; et al. Mutations in SPATA7 Cause Leber Congenital Amaurosis and Juvenile Retinitis Pigmentosa. Am. J. Hum. Genet. 2009, 84, 380–387. [Google Scholar] [CrossRef] [Green Version]
- Sohocki, M.M.; Sullivan, L.S.; Tirpak, D.L.; Daiger, S.P. Comparative analysis of aryl-hydrocarbon receptor interacting pro-tein-like 1 (Aipl1), a gene associated with inherited retinal disease in humans. Mamm. Genome 2001, 12, 566–568. [Google Scholar]
- Hollander, A.I.D.; Heckenlively, J.R.; Born, L.I.V.D.; de Kok, Y.J.; van der Velde-Visser, S.D.; Kellner, U.; Jurklies, B.; van Schooneveld, M.J.; Blankenagel, A.; Rohrschneider, K.; et al. Leber Congenital Amaurosis and Retinitis Pigmentosa with Coats-like Exudative Vasculopathy Are Associated with Mutations in the Crumbs Homologue 1 (CRB1) Gene. Am. J. Hum. Genet. 2001, 69, 198–203. [Google Scholar] [CrossRef] [Green Version]
- Gerber, S.; Perrault, I.; Hanein, S.; Barbet, F.; Ducroq, D.; Ghazi, I.; Martin-Coignard, D.; Leowski, C.; Homfray, T.; Dufier, J.-L.; et al. Complete exon-intron structure of the RPGR-interacting protein (RPGRIP1) gene allows the identification of mutations underlying Leber congenital amaurosis. Eur. J. Hum. Genet. 2001, 9, 561–571. [Google Scholar] [CrossRef] [Green Version]
- Freund, C.L.; Wang, Q.-L.; Chen, S.; Muskat, B.L.; Wiles, C.D.; Sheffield, V.C.; Jacobson, S.G.; Mclnnes, R.R.; Zack, D.J.; Stone, E.M. De novo mutations in the CRX homeobox gene associated with Leber congenital amaurosis. Nat. Genet. 1998, 18, 311–312. [Google Scholar] [CrossRef]
- Hollander, A.I.D.; Johnson, K.; De Kok, Y.J.M.; Klebes, A.; Brunner, H.G.; Knust, E.; Cremers, F.P.M. CRB1 has a cytoplasmic domain that is functionally conserved between human and Drosophila. Hum. Mol. Genet. 2001, 10, 2767–2773. [Google Scholar] [CrossRef] [Green Version]
- Bowne, S.J.; Sullivan, L.S.; Mortimer, S.E.; Hedstrom, L.; Zhu, J.; Spellicy, C.J.; Gire, A.I.; Hughbanks-Wheaton, D.; Birch, D.G.; Lewis, R.A.; et al. Spectrum and Frequency of Mutations in IMPDH1 Associated with Autosomal Dominant Retinitis Pig-mentosa and Leber Congenital Amaurosis. Investig. Opthalmol. Vis. Sci. 2006, 47, 34–42. [Google Scholar] [CrossRef]
- Friedman, J.S.; Chang, B.; Kannabiran, C.; Chakarova, C.; Singh, H.P.; Jalali, S.; Hawes, N.L.; Branham, K.; Othman, M.; Filippova, E.; et al. Premature Truncation of a Novel Protein, RD3, Exhibiting Subnuclear Localization Is Associated with Retinal Degeneration. Am. J. Hum. Genet. 2006, 79, 1059–1070. [Google Scholar] [CrossRef] [Green Version]
- Janecke, A.; Thompson, D.; Utermann, G.; Becker, C.; Hübner, C.A.; Schmid, E.; McHenry, C.L.; Nair, A.R.; Rüschendorf, F.; Heckenlively, J.; et al. Mutations in RDH12 encoding a photoreceptor cell retinol dehydrogenase cause childhood-onset severe retinal dystrophy. Nat. Genet. 2004, 36, 850–854. [Google Scholar] [CrossRef]
- Mykytyn, K.; Nishimura, D.Y.; Searby, C.; Shastri, M.; Yen, H.-J.; Beck, J.S.; Braun, T.A.; Streb, L.M.; Cornier, A.S.; Cox, G.F.; et al. Identification of the gene (BBS1) most commonly involved in Bardet-Biedl syndrome, a complex human obesity syndrome. Nat. Genet. 2002, 31, 435–438. [Google Scholar] [CrossRef]
- Nishimura, D.Y.; Searby, C.C.; Carmi, R.; Elbedour, K.; Van Maldergem, L.; Fulton, A.B.; Lam, B.L.; Powell, B.R.; Swiderski, R.E.; Bugge, K.E.; et al. Positional cloning of a novel gene on chromosome 16q causing Bardet-Biedl syndrome (BBS2). Hum. Mol. Genet. 2001, 10, 865–874. [Google Scholar] [CrossRef] [Green Version]
- Chiang, A.P.; Nishimura, D.; Searby, C.; Elbedour, K.; Carmi, R.; Ferguson, A.L.; Secrist, J.; Braun, T.; Casavant, T.; Stone, E.M.; et al. Comparative Genomic Analysis Identifies an ADP-Ribosylation Factor–like Gene as the Cause of Bardet-Biedl Syndrome (BBS3). Am. J. Hum. Genet. 2004, 75, 475–484. [Google Scholar] [CrossRef] [Green Version]
- Mykytyn, K.; Braun, T.; Carmi, R.; Haider, N.B.; Searby, C.C.; Shastri, M.; Beck, G.; Wright, A.F.; Iannaccone, A.; Elbedour, K.; et al. Identification of the gene that, when mutated, causes the human obesity syndrome BBS4. Nat. Genet. 2001, 28, 188–191. [Google Scholar] [CrossRef]
- Torrefranca, A.B.; Santiago, A.P.D.; Lingao, M.D.; Racoma, M.J.C. Novel compound heterozygous pathogenic BBS5 variants in Filipino siblings with Bardet-Biedl syndrome (BBS). Ophthalmic Genet. 2020, 41, 621–624. [Google Scholar] [CrossRef]
- Katsanis, N.; Beales, P.L.; Woods, M.O.; Lewis, R.A.; Green, J.S.; Parfrey, P.S.; Ansley, S.J.; Davidson, W.S.; Lupski, J.R. Mutations in MKKS cause obesity, retinal dystrophy and renal malformations associated with Bardet-Biedl syndrome. Nat. Genet. 2000, 26, 67–70. [Google Scholar] [CrossRef]
- Badano, J.; Ansley, S.J.; Leitch, C.C.; Lewis, R.A.; Lupski, J.R.; Katsanis, N. Identification of a Novel Bardet-Biedl Syndrome Protein, BBS7, That Shares Structural Features with BBS1 and BBS2. Am. J. Hum. Genet. 2003, 72, 650–658. [Google Scholar] [CrossRef] [Green Version]
- Tadenev, A.L.; Kulaga, H.M.; May-Simera, H.L.; Kelley, M.W.; Katsanis, N.; Reed, R.R. Loss of Bardet-Biedl syndrome protein-8 (BBS8) perturbs olfactory function, protein localization, and axon targeting. Proc. Natl. Acad. Sci. USA 2011, 108, 10320–10325. [Google Scholar] [CrossRef] [Green Version]
- Nishimura, D.Y.; Swiderski, R.E.; Searby, C.; Berg, E.M.; Ferguson, A.L.; Hennekam, R.; Merin, S.; Weleber, R.G.; Biesecker, L.G.; Stone, E.M.; et al. Comparative Genomics and Gene Expression Analysis Identifies BBS9, a New Bardet-Biedl Syndrome Gene. Am. J. Hum. Genet. 2005, 77, 1021–1033. [Google Scholar] [CrossRef] [Green Version]
- Stoetzel, C.; Laurier, V.; Davis, E.E.; Muller, J.; Rix, S.; Badano, J.L.; Leitch, C.C.; Salem, N.; Chouery, E.; Corbani, S.; et al. BBS10 encodes a vertebrate-specific chaperonin-like protein and is a major BBS locus. Nat. Genet. 2006, 38, 521–524. [Google Scholar] [CrossRef]
- Chiang, A.P.; Beck, J.S.; Yen, H.J.; Tayeh, M.K.; Scheetz, T.E.; Swiderski, R.E.; Nishimura, D.Y.; Braun, T.A.; Kim, K.Y.; Huang, J.; et al. Homozygosity mapping with SNP arrays identifies TRIM32, an E3 ubiquitin ligase, as a Bardet-Biedl syndrome gene (BBS11). Proc. Natl. Acad. Sci. USA 2006, 103, 6287–6292. [Google Scholar] [CrossRef] [Green Version]
- Stoetzel, C.; Muller, J.; Laurier, V.; Davis, E.E.; Zaghloul, N.A.; Vicaire, S.; Jacquelin, C.; Plewniak, F.; Leitch, C.C.; Sarda, P.; et al. Identification of a Novel BBS Gene (BBS12) Highlights the Major Role of a Vertebrate-Specific Branch of Chaperonin-Related Proteins in Bardet-Biedl Syndrome. Am. J. Hum. Genet. 2007, 80, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Leitch, C.C.; Zaghloul, N.A.; Davis, E.; Stoetzel, C.; Diaz-Font, A.; Rix, S.; Al-Fadhel, M.; Lewis, R.A.; Eyaid, W.; Banin, E.; et al. Hypomorphic mutations in syndromic encephalocele genes are associated with Bardet-Biedl syndrome. Nat. Genet. 2008, 40, 443–448. [Google Scholar] [CrossRef]
- Kim, S.K.; Shindo, A.; Park, T.J.; Oh, E.C.; Ghosh, S.; Gray, R.S.; Lewis, R.A.; Johnson, C.A.; Attie-Bittach, T.; Katsanis, N.; et al. Planar Cell Polarity Acts Through Septins to Control Collective Cell Movement and Ciliogenesis. Science 2010, 329, 1337–1340. [Google Scholar] [CrossRef] [Green Version]
- Otto, E.A.; Hurd, T.W.; Airik, R.; Chaki, M.; Zhou, W.; Stoetzel, C.; Patil, S.B.; Levy, S.; Ghosh, A.K.; Murga-Zamalloa, C.A.; et al. Candidate exome capture identifies mutation of SDCCAG8 as the cause of a retinal-renal ciliopathy. Nat. Genet. 2010, 42, 840–850. [Google Scholar] [CrossRef] [Green Version]
- Collin, G.B.; Marshall, J.D.; Ikeda, A.; So, W.V.; Russell-Eggitt, I.; Maffei, P.; Beck, S.; Boerkoel, C.F.; Sicolo, N.; Martin, M.; et al. Mutations in ALMS1 cause obesity, type 2 diabetes and neurosensory degeneration in Alström syndrome. Nat. Genet. 2002, 31, 74–78. [Google Scholar] [CrossRef]
- Pirnar, T.; Neuhauser, E.B.D. Asphyxiating thoracic dystrophy of the newborn. Am. J. Roentgenol Radium. Ther. Nucl. Med. 1966, 98, 358–364. [Google Scholar] [CrossRef]
- He, X.; Semenov, M.; Tamai, K.; Zeng, X. LDL receptor-related proteins 5 and 6 in Wnt/beta-catenin signaling: Arrows point the way. Development 2004, 131, 1663–1677. [Google Scholar] [CrossRef] [Green Version]
- Habas, R.; Dawid, I.B. Dishevelled and Wnt signaling: Is the nucleus the final frontier? J. Biol. 2005, 4, 2. [Google Scholar] [CrossRef] [Green Version]
- LeCarpentier, Y.; Schussler, O.; Hébert, J.-L.; Vallée, A. Multiple Targets of the Canonical WNT/β-Catenin Signaling in Cancers. Front. Oncol. 2019, 9, 1248. [Google Scholar] [CrossRef]
- Bian, J.; Dannappel, M.; Wan, C.; Firestein, R. Transcriptional Regulation of Wnt/β-Catenin Pathway in Colorectal Cancer. Cells 2020, 9, 2125. [Google Scholar] [CrossRef]
- Zhong, W.; Oguljahan, B.; Xiao, Y.; Nelson, J.; Hernandez, L.; Garcia-Barrio, M.; Francis, S.C. Serum and glucocorti-coid-regulated kinase 1 promotes vascular smooth muscle cell proliferation via regulation of β-catenin dy-namics. Cell Signal. 2014, 26, 2765–2772. [Google Scholar] [CrossRef] [Green Version]
- Cicenas, J.; Meskinyte-Kausiliene, E.; Jukna, V.; Rimkus, A.; Simkus, J.; Soderholm, D. SGK1 in Cancer: Biomarker and Drug Target. Cancers 2022, 14, 2385. [Google Scholar] [CrossRef]
- Colosimo, P.F.; Tolwinski, N.S. Wnt, Hedgehog and junctional Armadillo/beta-catenin establish planar polarity in the Dro-sophila embryo. PLoS ONE 2006, 1, e9. [Google Scholar] [CrossRef]
- Colosimo, P.F.; Liu, X.; Kaplan, N.A.; Tolwinski, N.S. GSK3beta affects apical-basal polarity and cell-cell adhesion by regulating aPKC levels. Dev. Dyn. 2010, 239, 115–125. [Google Scholar]
- Kohn, A.D.; Moon, R.T. Wnt and calcium signaling: Beta-catenin-independent pathways. Cell Calcium 2005, 38, 439–446. [Google Scholar] [CrossRef]
- Sumida, T.; Lincoln, M.R.; Ukeje, C.M.; Rodriguez, D.M.; Akazawa, H.; Noda, T.; Naito, A.T.; Komuro, I.; Dominguez-Villar, M.; Hafler, D.A. Activated β-catenin in Foxp3+ regulatory T cells links inflammatory environments to autoimmunity. Nat. Immunol. 2018, 19, 1391–1402. [Google Scholar] [CrossRef]
- Abdelhamed, Z.A.; Abdelmottaleb, D.I.; El-Asrag, M.E.; Natarajan, S.; Wheway, G.; Inglehearn, C.F.; Toomes, C.; Johnson, C.A. The ciliary Frizzled-like receptor Tmem67 regulates canonical Wnt/β-catenin signalling in the developing cerebellum via Hoxb5. Sci. Rep. 2019, 9, 5446. [Google Scholar] [CrossRef] [Green Version]
- Abdelhamed, Z.A.; Wheway, G.; Szymanska, K.; Natarajan, S.; Toomes, C.; Inglehearn, C.; Johnson, C.A. Variable expressivity of ciliopathy neurological phenotypes that encompass Meckel-Gruber syn-drome and Joubert syndrome is caused by complex de-regulated ciliogenesis, Shh and Wnt signalling defects. Hum. Mol. Genet. 2013, 22, 1358–1372. [Google Scholar] [CrossRef] [Green Version]
- Preston, D.; Simpson, S.; Halm, D.; Hochstetler, A.; Schwerk, C.; Schroten, H.; Blazer-Yost, B.L. Activation of TRPV4 stimulates transepithelial ion flux in a porcine choroid plexus cell line. Am. J. Physiol. Physiol. 2018, 315, C357–C366. [Google Scholar] [CrossRef]
- Benfenati, V.; Caprini, M.; Dovizio, M.; Mylonakou, M.N.; Ferroni, S.; Ottersen, O.P.; Amiry-Moghaddam, M. An aqua-porin-4/transient receptor potential vanilloid 4 (AQP4/TRPV4) complex is essential for cell-volume control in astrocytes. Proc. Natl. Acad. Sci. USA 2011, 108, 2563–2568. [Google Scholar] [CrossRef] [Green Version]
- Takayama, Y.; Shibasaki, K.; Suzuki, Y.; Yamanaka, A.; Tominaga, M. Modulation of water efflux through functional in-teraction between TRPV4 and TMEM16A/anoctamin 1. FASEB J. 2014, 28, 2238–2248. [Google Scholar] [CrossRef]
- Singh, S.K.; O’Hara, B.; Talukder, J.R.; Rajendran, V.M. Aldosterone induces active K+secretion by enhancing mucosal ex-pression of Kcnn4c and Kcnma1 channels in rat distal colon. Am. J. Physiol. Physiol. 2012, 302, C1353–C1360. [Google Scholar] [CrossRef] [Green Version]
- Rachel, R.A.; Yamamoto, E.A.; Dewanjee, M.K.; May-Simera, H.L.; Sergeev, Y.V.; Hackett, A.N.; Pohida, K.; Munasinghe, J.; Gotoh, N.; Wickstead, B.; et al. CEP290 alleles in mice disrupt tissue-specific cilia biogenesis and recapitulate features of syndromic ciliopathies. Hum. Mol. Genet. 2015, 24, 3775–3791. [Google Scholar] [CrossRef] [Green Version]
- Coppieters, F.; Lefever, S.; Leroy, B.P.; De Baere, E. CEP290, a gene with many faces: Mutation overview and presentation of CEP290base. Hum. Mutat. 2010, 31, 1097–1108. [Google Scholar] [CrossRef] [Green Version]
- Murga-Zamalloa, C.A.; Desai, N.J.; Hildebrandt, F.; Khanna, H. Interaction of ciliary disease protein retinitis pigmentosa GTPase regulator with nephronophthisis-associated proteins in mammalian retinas. Mol. Vis. 2010, 16, 1373–1381. [Google Scholar]
- Bagher, P.; Beleznai, T.; Kansui, Y.; Mitchell, R.; Garland, C.J.; Dora, K.A. Low intravascular pressure activates endothelial cell TRPV4 channels, local Ca 2+ events, and IK Ca channels, reducing arteriolar tone. Proc. Natl. Acad. Sci. USA 2012, 109, 18174–18179. [Google Scholar] [CrossRef] [Green Version]
- Hochstetler, A.E.; Smith, H.M.; Preston, D.C.; Reed, M.M.; Territo, P.R.; Shim, J.W.; Fulkerson, D.; Blazer-Yost, B.L. TRPV4 antagonists ameliorate ventriculomegaly in a rat model of hydrocephalus. JCI Insight 2020, 5, 137646. [Google Scholar] [CrossRef]
- Ortuso, F.; Amato, R.; Artese, A.; D’Antona, L.; Costa, G.; Talarico, C.; Gigliotti, F.; Bianco, C.; Trapasso, F.; Schenone, S.; et al. In Silico Identification and Biological Evaluation of Novel Selective Serum/Glucocorticoid-Inducible Kinase 1 Inhibitors Based on the Pyrazolo-Pyrimidine Scaffold. J. Chem. Inf. Model. 2014, 54, 1828–1832. [Google Scholar] [CrossRef]
- D’Antona, L.; Dattilo, V.; Catalogna, G.; Scumaci, D.; Fiumara, C.V.; Musumeci, F.; Perrotti, G.; Schenone, S.; Tallerico, R.; Spoleti, C.B.; et al. In Preclinical Model of Ovarian Cancer, the SGK1 Inhibitor SI113 Counteracts the Development of Paclitaxel Resistance and Restores Drug Sensitivity. Transl. Oncol. 2019, 12, 1045–1055. [Google Scholar] [CrossRef]
- Hochstetler, A.E.; Smith, H.; Reed, M.; Hulme, L.; Perrotti, N.; D’Antona, L.; Schenone, S.; Musumeci, F.; Blazer-Yost, B. Inhibition of SGK1 ameliorates ventriculomegaly in a genetic rat model via regulation of TRPV4 in the choroid plexus. FASEB J. 2022, 36. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
D’Antona, L.; Amato, R.; Brescia, C.; Rocca, V.; Colao, E.; Iuliano, R.; Blazer-Yost, B.L.; Perrotti, N. Kinase Inhibitors in Genetic Diseases. Int. J. Mol. Sci. 2023, 24, 5276. https://doi.org/10.3390/ijms24065276
D’Antona L, Amato R, Brescia C, Rocca V, Colao E, Iuliano R, Blazer-Yost BL, Perrotti N. Kinase Inhibitors in Genetic Diseases. International Journal of Molecular Sciences. 2023; 24(6):5276. https://doi.org/10.3390/ijms24065276
Chicago/Turabian StyleD’Antona, Lucia, Rosario Amato, Carolina Brescia, Valentina Rocca, Emma Colao, Rodolfo Iuliano, Bonnie L. Blazer-Yost, and Nicola Perrotti. 2023. "Kinase Inhibitors in Genetic Diseases" International Journal of Molecular Sciences 24, no. 6: 5276. https://doi.org/10.3390/ijms24065276






