Abstract
Guatemala is recognized for its diverse and rich flora and fauna. It is estimated that over 1200 orchid species, classified in 223 genera, are known to occur in this rather small, yet megadiverse country. While studying the diversity of this plant group in the department of Baja Verapaz, we found individuals that clearly belonged to the genus Schiedeella, but whose features did not fit any previously known species. At that time, nine terrestrial taxon representatives were known to occur in Guatemala. We conducted the morphological analysis in accordance with the standard procedures of classical taxonomy. For phylogenetic reconstruction, 59 sequences of the ITS region and 48 of the trnL-trnF marker were applied. The topology of trees was obtained based on the Bayesian inference. Schiedeella bajaverapacensis was described and illustrated based on morphological evidence, and its taxonomic position was confirmed by phylogenetic analyses. The new entity is the 10th Schiedeella representative known from Guatemala.
1. Introduction
Guatemala is a relatively small country in terms of size, but its geographical and biological diversity is enormous. It is estimated that about 10,800 species of vascular plants can be found there [1], and many of them are endemic. The biodiversity richness of the Orchidaceae family in Guatemala is an attribute that few countries have, but for many years, it has been poorly studied. It ranges from a little more than 100 species mentioned by Bateman [2], 527 species mentioned by Ames and Correll [3], and 800 [4] as well as another 1237 species proposed by Archila [5]. By far, the most comprehensive work on Guatemalan orchid flora [1] has reported the presence of 223 genera and over 1200 species in this megadiverse country.
The taxonomic position of the representatives of the tribe Spirantheae Lindl. (Orchidaceae) has been studied by many taxonomists during the past 200 years. However, different concepts of genera and species, as well as divergent interpretations of original diagnoses, have resulted in significant differences between the approaches of the authors. Genus Schiedeella Schltr. is one of the most controversial representatives of Spiranthinae. It is represented in Guatemala by nine species. While studying the orchid diversity in the department of Baja Verapaz, we found plants that clearly belonged to this taxon (Figure 1), but whose features did not fit any previously known species (Table 1). Hereby, we describe it as Schiedeella bajaverapacensis sp nov.

Figure 1.
Schiedeella bajaverapacensis—living plant: (A) in situ, in the flowering stage; (B–E) ex situ, in the leafy stage. Photo. O. Alvarado (A), F. Archila (B–E).

Table 1.
Comparison between the morphological features of Guatemalan representatives of the genus Schiedeella.
2. The History of Classification of Schiedeella Complex
The first attempt to systematize the subtribe Spiranthinae Lindl. was published in 1920 by Schlechter [6]. In his classification, Schlechter adopted uniform criteria for defining genera, assuming the most important to be the structure of the gynostemium and perianth morphology. He recognized 24 genera, of which 16 were newly segregated; among others, these were: Deiregyne Schltr. and Schiedeella. Unfortunately, Schlechter did not indicate the type of species for many of his new genera, which has later become the main reason for the differences in the taxonomic concepts between scientists working on this group. This classification has not been widely accepted by the scientific community, and the majority of the species in this subtribe described in the 1930s and 1940s were included in Spiranthes sensu latissimo [7].
The next attempt to clarify delimitations within the subtribe was proposed by Burns-Balogh [8]. According to her, Spiranthinae consists of approximately 250 species in 14 genera: Brachystele Schltr., Buchtienia Schltr., Cyclopogon C. Presl., Deiregyne, Discyphus Schltr., Eurystyles Wawra, Hapalorchis Schltr., Odontorrhynchus M.N. Correa, Pelexia Poit. ex Lindl., Sarcoglottis C. Presl., Sauroglossum Lindl., Schiedeella, Spiranthes Rich., and Stenorrhynchos Rich. ex Spreng. Using the rostellum/viscidium attachment as a distinguishing character, she grouped eleven of the above genera into four generic alliances. In this concept, the representatives of the Brachystele alliance have extremely reduced rostellum that may be completely fused to the oval viscidium. This assemblage includes Brachystele, Sauroglossum, and Odontorrhynchus. The Pelexia alliance is characterized by an elongate, broad, laminar rostellum that is adherent to the disc-like viscidium and embraces Cyclopogon, Pelexia, and Sarcoglottis. The Spiranthes alliance, which covers Discyphus, Hapalorchis, and Spiranthes, has a broad laminar rostellum that is completely fused to the fusiform viscidium. The Stenorrhynchos alliance consists of Schiedeella and Stenorrhynchos, which have subulate rostellum with an ensheathing viscidium. Almost at the same time, Garay [9] published his revision of the subtribe Spiranthinae. He distinguished 44 genera, 14 of which were new.
In 1995, Szlachetko [10] proposed to divide Spirantheae into six subtribes. In his classification, Spiranthinae counted 27 genera and embraced some taxa previously segregated by Burns-Balogh, such as Brachystele, Buchtienia, Deiregyne, Eurystyles, Hapalorchis, Odontorrhynchus, Sauroglossum, and Spiranthes. Genera such as Schiedeella and Stenorrhynchos (thus, all members of Burns-Balogh’s Stenorrhynchos alliance) were placed together with 19 other taxa in the newly established subtribe Stenorrhynchidinae Szlach.
Schiedeella Schltr. is one of the richest in species and at the same time is one of the most controversial genera within Stenorrhynchidinae (sensu Szlachetko [10]). Schlechter [6] considered Schiedeella to be closely related to Cyclopogon, however, differing from it in the morphology of the lip. The flowers of the two genera are fairly similar, but when studied in detail, distinct differences become evident, particularly in the structure of the rostellum and viscidium. As noted by Szlachetko [7], a distinct variance with Schlechter’s description is the structure of the rostellum and viscidium of Schiedeella pyramidalis Lindl. The remaining species, following the structure of the rostellum, perianth, and bracts, were divided into two distinct groups.
Regarding two species of eight, i.e., S. pyramidalis and S. cobanensis Schltr., proposed by Schlechter [6], Burns-Balogh [8,11] excluded them from Schiedeella and added eighteen further species, most of which were described after Schlechter’s death in 1925. She also designated Spiranthes saltensis Ames (=S. durangensis Ames & Schweinf.) as the type species for the genus. In 1986, Burns-Balogh [11] published the infrageneric classification of Schiedeella, distinguishing the following sections: Parasitica Burns-Bal., Eriophora Burns-Bal., and Michoacana Burns-Bal. The basis for this division was the type of floral bracts, the color of flowers and nectaries, and the morphology of the lip.
Garay [9] incorporated into Schiedeella only species that have a narrow-based rostellum and lip set on a flat claw with auricles at the base of the hypochile. As a result, he left only two taxa of the original set of species in the genus, adding four others. He also transferred the remaining species from Schiedeella sensu Burns-Balogh [8,11] to such genera as Dithyridanthus Garay, Funkiella Schltr., Gularia Garay, Microthelys Garay, Physogyne Garay, Stenorrhynchos and the majority to Deiregyne.
Szlachetko [7] pointed out that Schiedeella sensu Garay did not include Spiranthes trilineata Lindl., which from the point of view of the structure of the gynostemium and the perianth, quite markedly suggests Schiedeella nagelii (L.O. Williams) Garay. Instead, Garay created a new genus, Gularia Garay, for Spiranthes trilineata and S. trilineata var. crenulata (L.O. Williams) Szlach. and excluded the group S. eriophora/S. velata from Schiedeella. He treated them as representatives of Deiregyne. In Garay’s concept, Gularia differs from Schiedeella in the presence of a long, decurrent column foot, a pliable rostellum, and a claw fused with lateral sepals. However, according to Szlachetko [7], the column foot of S. trilineata is not longer than in other species of this genus. Studies of herbarium specimens have shown that the rostellum of this species is similar in texture to that of, for example, S. llaveana Schltr., and the fusion of the claw with the lateral sepals is present in various species of Schiedeella. In view of the lack of essential morphological conditions distinguishing the genus Gularia, Szlachetko [7] proposed to include it in Schiedeella.
Similarly to Burns-Balogh, Garay also noted that Schlechter had made certain suggestions as to the type of the genus Schiedeella and proposed S. transversalis A. Rich. & Galeotti as the generitype. Agreeing with Garay [9], Burns-Balogh [11] stated that different lectotypification of the genus would not alter her concept [7]. She treated S. transversalis as one of the species of Schiedeella.
In the present form, Schiedeella counts about 20 species whose distribution range covers practically the entire Central America and Antilles. The representatives of the genus are predominantly terrestrial plants, and their flowers are characterized by nearly erect gynostemium and a needle-like rostellum. The rostellum remnant is narrowly triangular and usually distinctly 3-dentate with the central tooth being the longest. The viscidium is nearly sheath-like, and the column foot is reduced. The lip is basally nearly erect or only slightly arcuate with a flat claw and more or less thickened auricles. Floral and cauline bracts are herbaceous or occasionally subscarious. Within the genus, based on the rostellum structure, three subgenera are being recognized: Schiedeella, Schiedeellopsis Szlach., and Gularia (Garay) Szlach. [12]. Subgenus Schiedeella counts 11 species and is characterized by long rostellum, apical vicidium, anther that is shorter than gynostemium and with an apex that reaches the viscidium’s upper part, and sepals that are free almost to the base. The subgenus Schiedeellopsis embraces four species, and based on lip shape, it is divided into two sections: Schiedeellopsis Szlach. (with three species) and Gemmorchis Szlach. (with a single species). This subgenus is characterized by a short rostellum, small, basal, oval, or ovate viscidium, an anther almost as long as gynostemium, and a rostellum remnant that is shortly three-dentate or subulate. The last subgenus, Gularia, covers three species and is distinguished by a long rostellum, anther apex that rarely reaches the base of the rostellum, and sepals whose basal part is connate, forming a short tube.
In 2016, Salazar et al. [13] segregated from Schiedeella the genus Greenwoodiella Salazar, Hernández-López & J. Sharma. The authors pointed out several morphological attributes, which in their opinion, distinguish the new genus from Schiedeella. These were: shoots connected by a slender rhizome, ovate or broadly elliptic, fleshy-coriaceous leaf with a rounded base, glossy dark green upper surface, and grayish-green underside with a purplish central vein. In terms of the floral features, the differences covered the proportionately shorter labellum, not surpassing the length of the lateral sepals. In the newly created taxon, they included Greenwoodiella deserticola Salazar, Hernández-López & J. Sharma, G. micrantha (Lex.) Salazar & R. Jiménez, G. micrantha (Lex.) Salazar et R. Jiménez var. garayana (R.González.) Salazar & R. Jiménez, and G. wercklei (Schltr.) Salazar & R. Jiménez.
3. Results
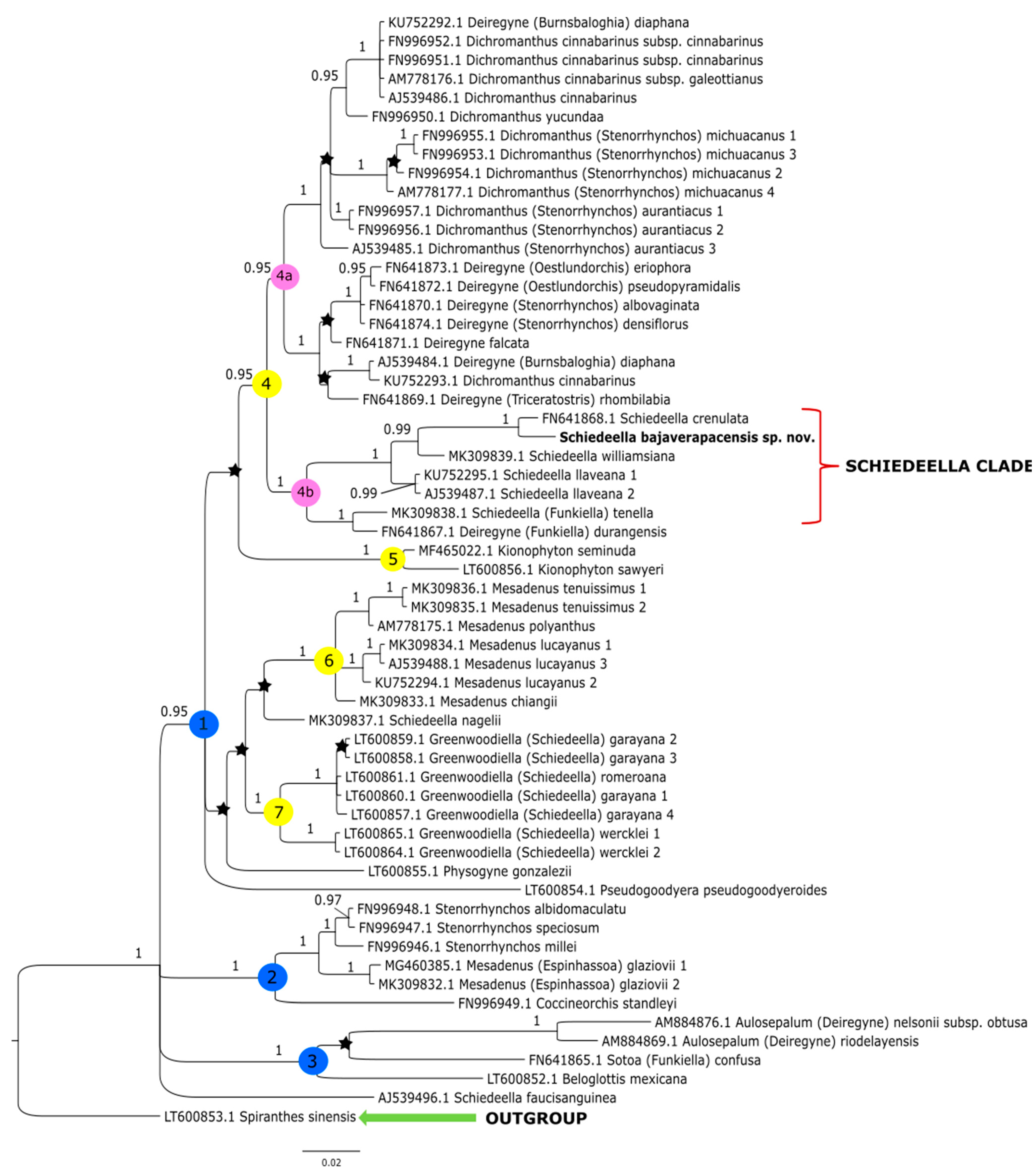
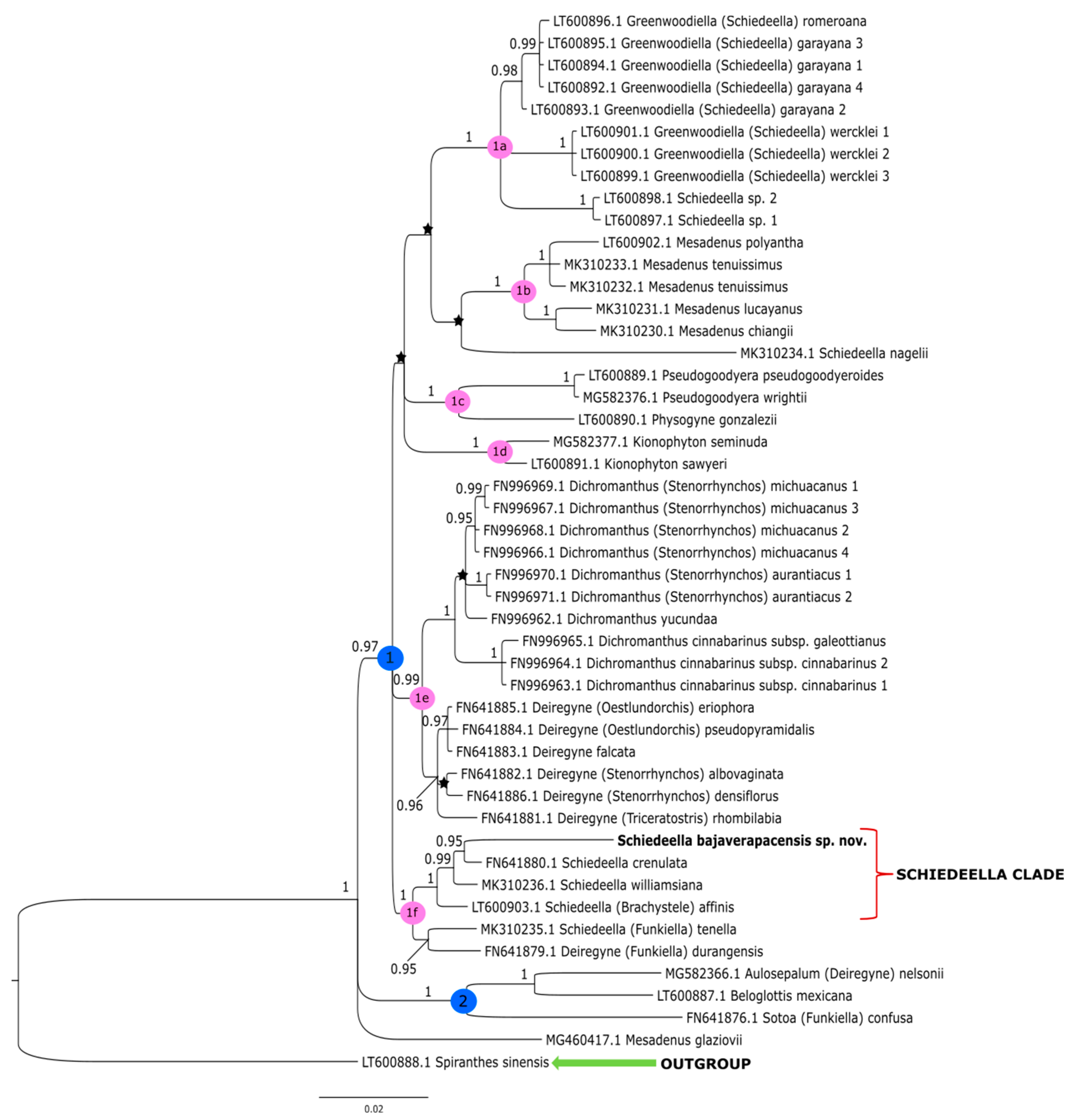
The topologies obtained under these studies based on the Bayesian inference gave similar results to that of the maximum-likelihood method conducted by Salazar et al. [14]. Thus, we decided not to perform additional analyses based on other methods. The clades that have the strong support of bootstrap values in the trees of Salazar et al. [14] also have high posterior probability values in trees obtained by us from the Bayesian inference. In this article, we present the trees from the single data matrix (Figure 2 and Figure 3). Additionally, in Table 2, we list the statistical data on the particular matrices.
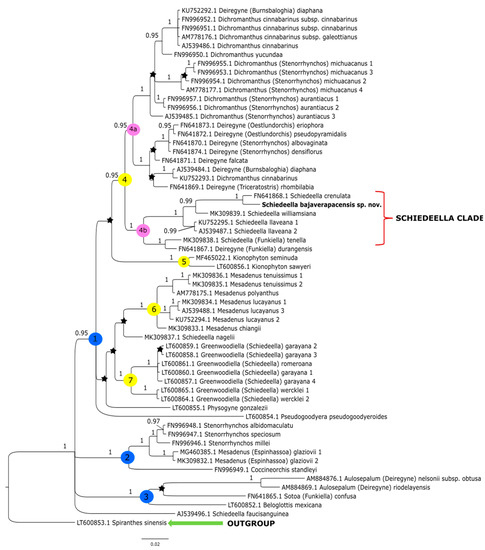
Figure 2.
The phylogenetic tree presenting the position of Schiedeella bajaverapacensis sp. nov., obtained for the ITS1-5.8S-ITS2 region based on BI analysis. Posterior probability (PP) values are indicated above the branches and PP < 0.95 as an asterisk. While the numbers marked in circles on the nodes mean discussed clades.
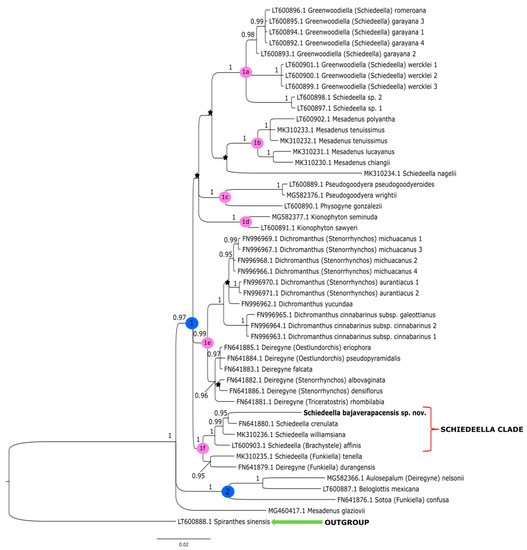
Figure 3.
The phylogenetic tree presenting the position of Schiedeella bajaverapacensis sp. nov., obtained for the trnL-trnF region based on BI analysis. Posterior probability (PP) values are indicated above the branches and PP < 0.95 as an asterisk. While the numbers marked in circles on the nodes mean discussed clades.

Table 2.
Statistical data for each single dataset.
3.1. ITS Matrix
At the base of the tree obtained for the ITS dataset (Figure 2) Spiranthes sinensis (Pers.) Ames was placed as a representative of the outgroup. The remaining taxa are included together in one group within which there are three major clades (1–3). The sample is indicated as AJ539496.1, representing that Schiedeella fauci-sanguinea (Dod) Dod was not grouped with other species of this genera, only placed as a polytomy branch within a major clade of the ingroup. The first, clade 1 with high posterior probability support (PP = 0.95), embraces most of the analyzed taxa and includes four smaller groupings (clades 4–7). Clade 4 (PP = 0.95) is split into two subclades: 4a (PP = 0.95) comprising representatives of Dichromanthus Garay and Deiregyne Schltr., and a sister subclade 4b (PP = 1), which includes species of Schiedeella. In subclade 4b is placed the new species—Schiedeella bajaverapacensis. It forms a consistent and strongly supported (PP = 1) group with S. crenulata (L.O.Williams) Espejo & López-Ferr., S. williamsiana Szlach. & al., and S. llaveana Lindl. The latter species are sampled twice. Deiregyne durangensis (Ames & C.Schweinf.) Garay (FN641867.1) was also ranked in this subclade. Clade 5 (PP= 1; Figure 2) embraces only two species representing Kionophyton Garay while clade 6 (PP = 1) includes members of the genus Mesadenus Schltr. The last group within the major clade 1 (PP = 0.95) is represented by the members of Greenwoodiella Salazar, Hern.-López & J.Sharma (subclade 7, PP = 1).
In clade 2 (PP = 1, Figure 2), Coccineorchis standleyi (Ames) Garay is a sister taxon to the representatives of Mesadenus glaziovii (Cogn.) Schltr. (=Espinhassoa glaziovii (Cogn.) Salazar & J.A.N. Bat.), which was sampled twice, and species of Stenorrhynchos Rich. ex Spreng. The last of the major ones, clade 3 (PP = 1, Figure 2), embraces Beloglottis mexicana Garay & Hamer and Sotoa confusa Salazar, both as polytomic branches, and a small group that includes two species of Aulosepalum Garay.
3.2. Plastid Matrix
The tree obtained for the plastid region (Figure 3) showed slightly different results. In the ingroup, we can distinguish two major clades, marked as 1 and 2. A sample of Mesadenus glaziovii indicates that MG460417.1 was not included with one group of other representatives of this genera but constitutes the polytomic branch. Clade 2 (PP = 1) embraces Sotoa confusa as a sister taxon to the group that includes Aulosepalum nelsonii (Greenm.) Garay and Beloglottis mexicana, which corresponds to clade 3 on the tree, performed for the ITS marker (Figure 2). However, the grouping of analyzed taxa at the level of smaller subclades is much more similar to the results obtained for the ITS matrix. Clade 1 (PP = 1) is divided into six strongly supported groups (clades 1a–1f). The representatives of Greenwoodiella form subclade 1a (PP = 1), and it almost corresponds to clade 7 (PP = 1) on the ITS tree (Figure 2). Subclade 1a differs in the presence of two samples of Schiedeella sp. A similar situation is observed for subclade 1b (PP = 1) and 1d (PP = 1) that respectively embrace the species of Mesadenus and Kionophyton. In this case, Pseudogoodyera is sampled by two species, and this probably caused them to form a strongly supported subclade 1c (PP = 1) together with Physogyne gonzalezii while on the ITS tree; these taxa form polytomous branches. The taxa of Dichromanthus and Deiregyne are grouped together (subclade 1e, PP = 0.99) similarly on the ITS tree (Figure 2). However, subclade 1e (Figure 3) separates into two strongly and monophyletic groups, and one of these includes species of Deiregyne (PP = 0.96) and Dichromanthus (PP = 1). In the last subclade 1f (PP = 1), the new species Schiedeella bajaverapacensis is placed with one group of other representatives of Schiedeella. As in the ITS tree (Figure 2), S. bajaverapacensis is closely related to S. crenulata (PP = 0.95). The results for both nuclear and plastid datasets showed that the phylogenetic position of the new taxon is within the Schideella group. However, the trnL-trnF tree (Figure 3) in subclade 1f, the Schiedeella group, ranked S. affinis (Brachystele), with strong probability support (PP = 1). Unfortunately for the ITS marker, we did not have a sample for this species, and thus, we cannot compare this result.
3.3. Taxonomic Treatment
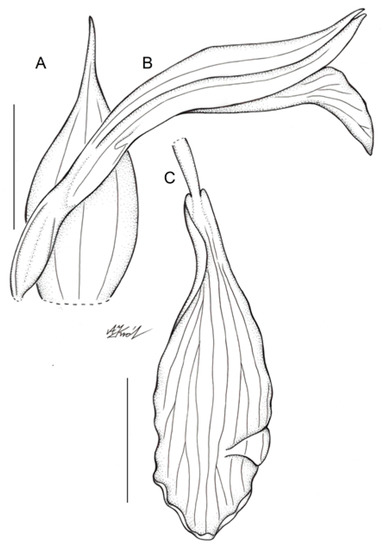
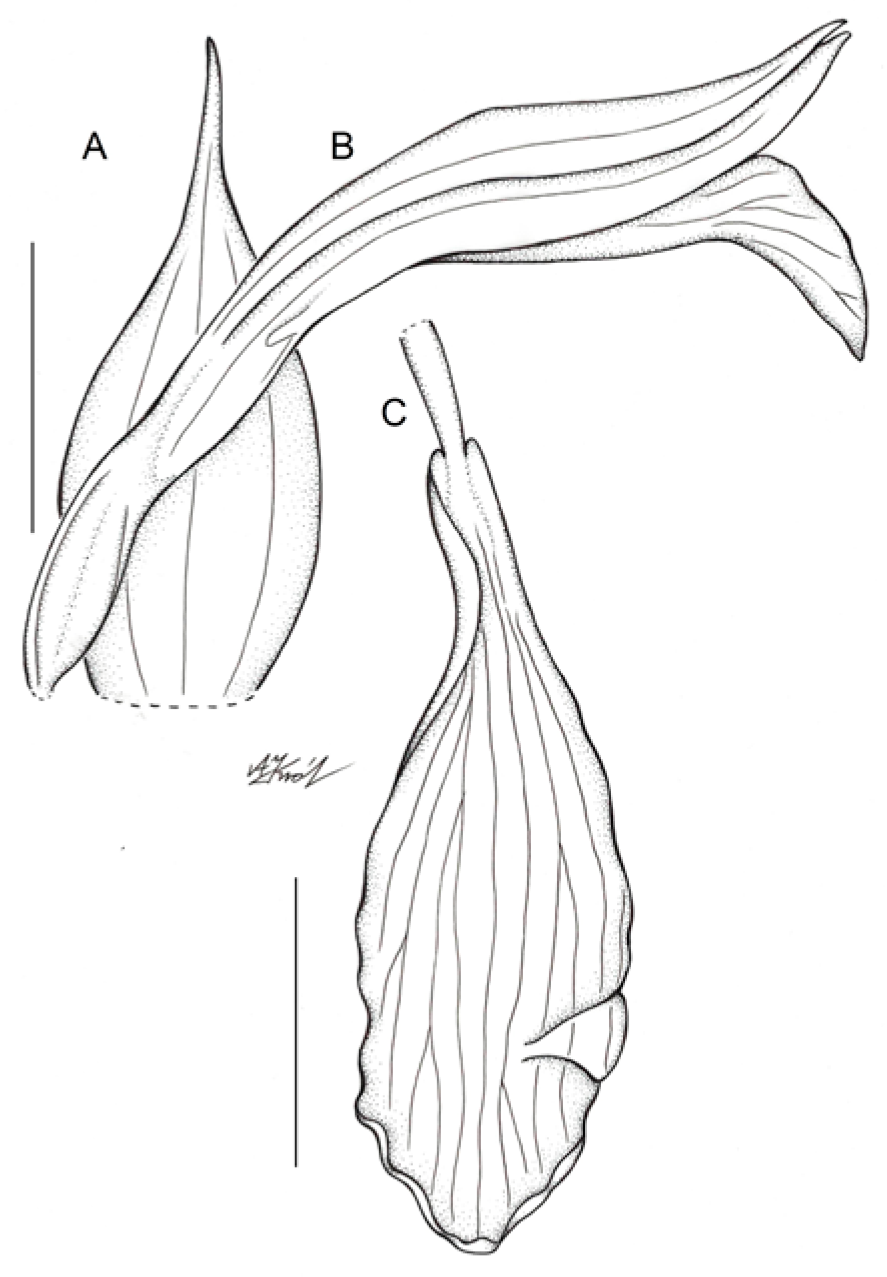
Figure 4.
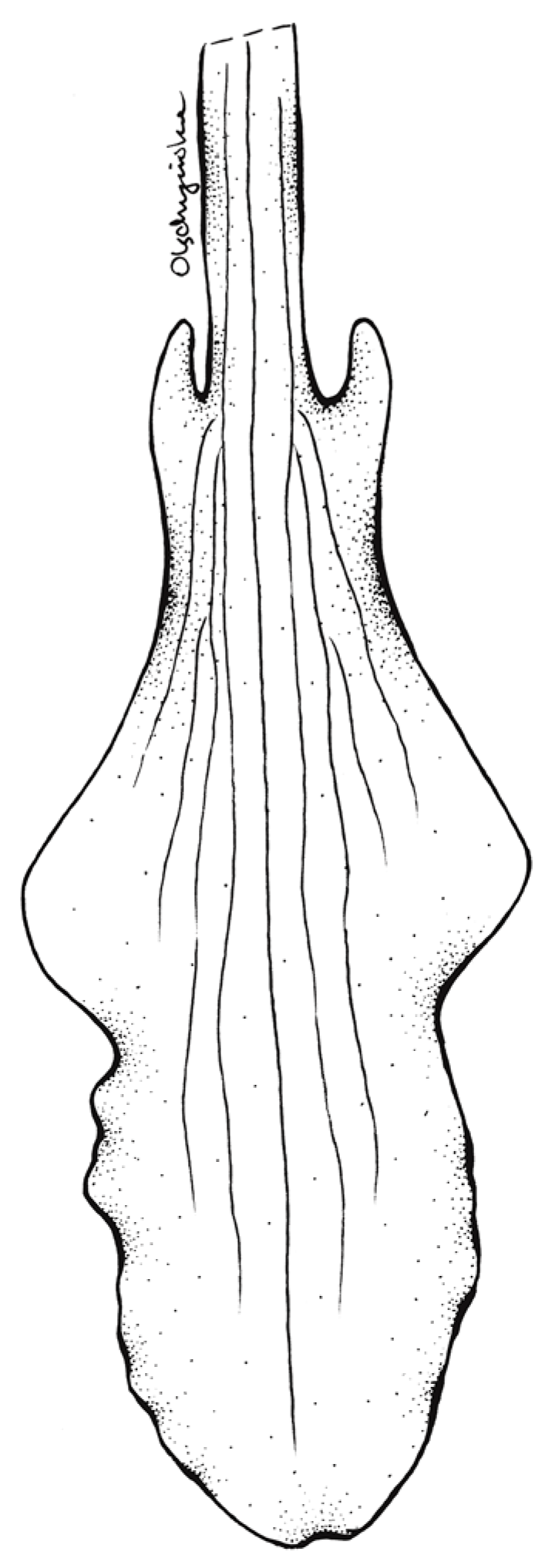
Floral parts of Schiedeella bajaverapacensis: (A) floral bract; (B) flower—side view; (C) Lip. Scale bars—5 mm. Drawn from the holotype by A. Król.
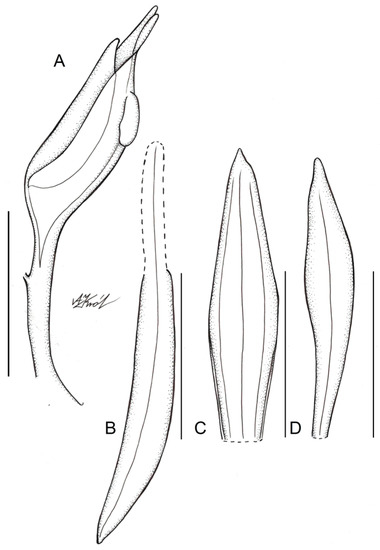
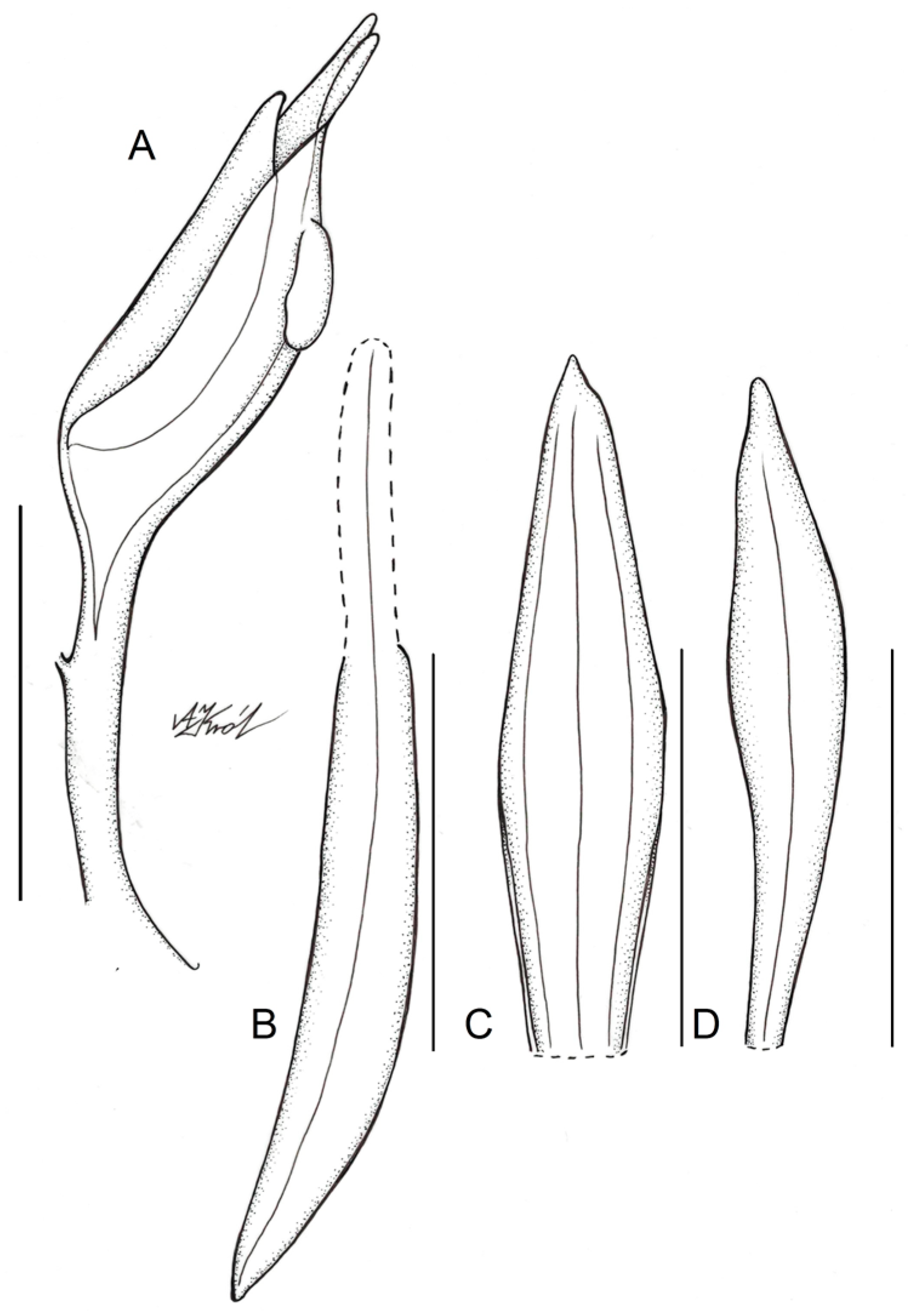
Figure 5.
Floral parts of Schiedeella bajaverapacensis: (A) gynostemium—side view; (B) lateral sepal; (C) dorsal sepal; (D) petal. Scale bars—5 mm. Drawn from the holotype by A. Król.
TYPE: GUATEMALA. Departamento de Baja Verapaz. Municipio de Salamá, a 950 m asl. Creciendo junto a Melocactus curvispinus, Opuntia cochenillifera y Cohniella brachyphylla. Colectada en enero 2022 por Otto Alvarado (Recorded by Archila & Szlachetko) (HOLOTYPE: BIGU!, ISOTYPES: BIGU!, UGDA-DLSz!, UGDA-DLSz!—liquid).
The new species appears to be similar to Schiedeella crenulata and S. trilineata var. undulata. S. bajaverapacensis differs from both mentioned taxa in the lip form, which is elliptic-ovate in the general outline, with a narrow basal part, shortly and obscurely auriculate, and an apical third of lamina somewhat undulate along the margins. The lip of S. crenulata is indistinctly divided into hypochile and epichile, which is ligulate with crenulate margins. The lip of S. trilineata var. undulata is oblanceolate in outline, is obtuse, and is widest in 2/3 of its length with distinctly undulate margins in the apical half.
Description: Plants terrestrial, small. Stem ca. 10 cm tall, erect, delicate, completely glabrous, enveloped in three sheaths, leafless at anthesis. Sheaths ovate-lanceolate, acuminate, glabrous, semi-transparent. Leaf single, at the beginning protected with three bracts, until it emerges from the ground; blade ca. 6.2 cm long and 0.8 cm wide, elliptic, microscopically obliquely acute. Protective bracts with sharp apex, gray in the abaxial part and brown in the adaxial part, of different sizes: 0.5 cm long and 0.3 cm wide the external one, 0.9 cm long and 0.5 cm wide the intermediate one and 1.7 cm long and 0.4 cm wide the internal one. Inflorescence ca. 2.5 cm long, laxly five-flowered. Flowers white, sepals and petals with prominent, grayish central vein, lip pure white with pinkish suffusion in the center and grayish central vein. Floral bracts 10 mm long, obliquely ovate, long-acuminate, with three prominent veins, glabrous, semi-transparent, whitish. Ovary 4–5 mm long, glabrous, white. Sepals connate forming a prominent tube in the basal quarter, somewhat swollen. Dorsal sepal free part 9 mm long, 2 mm wide, lanceolate, acute, basally connate with the gynostemium, somewhat concave in the center, three-veined, glabrous. Petals 8.5 mm long, 1.5 mm wide, linear-oblanceolate, acute, subfalcate, adnate to the dorsal sepal forming a kind of galea, one-veined. Lateral sepals free part 8 mm long, 1.5 mm wide, linear, acute, subfalcate, one-veined, glabrous. Lip clawed; claw 3 mm long, narrow, adnate to the sepaline tube; blade 11 mm long in total, 3.2 mm wide when spread, elliptic-ovate in general outline, basally shortly auriculate, auricles somewhat thickened, apical third of lamina somewhat undulate along margins, apex obtuse, multiveined, thin, glabrous. Gynostemium 11.5 mm long, erect, somewhat swollen in the apical half. Column foot 1.5 mm long, oblique. Anther 4.5 mm long, oblong-ovate. Rostellum 2.1 mm long, erect, subulate. Viscidium 1.1 mm long, sheath-like. Clinandrium spacious.
Etymology: In reference to the Department of Baja Verapaz, where new species was collected.
Ecology and distribution: Known only from the type locality in the department of Baja Verapaz, Guatemala. It grew in an open area at an altitude of 950 m asl, together with Melocactus curvispinus Pfeiff., Opuntia cochenillifera (L.) Mill., and Cohniella brachyphylla (Lindl.) Cetzal & Carnevali. Flowers in January.
Conservation status: According to the IUCN red list criteria [15], S. bajaverapacensis was assessed as data deficient (DD).
4. Discussion
According to the results of the molecular analyses, Schiedeella bajaverapacensis appears to be genetically related to S. crenulata (L.O.Williams) Espejo & López-Ferrari from the southern states of Mexico (Chiapas, San Luis Potosi, Puebla) and Honduras. On the two trees we obtained (Figure 2 and Figure 3), these species are grouped into a single clade with strong posterior probability support. Both species share similar habits, as all taxa of the subgenus Gularia (Garay) Szlach., i.e., short, few-flowered inflorescences, leafless at anthesis (Table 3. We had the opportunity to study the type material of this taxon—Fröderström 2592—stored at AMES (Figure 6 and Figure 7). The lip appears to be somewhat different from the one depicted by Williams on the herbarium sheet. Unlike our new entity, the lip of S. crenulata (Figure 7) is indistinctly divided into hypochile and epichile; the hypochile is deltoid in outline and the narrower epichile is ligulate, with an obtuse apex and crenulate margins. Unfortunately, we do not have access to any material of S. trilineata var. undulata Szlach., which is morphologically quite similar, to the molecular study. This variety has been described based on material collected in Guatemala, Dept. Guatemala, “Vara de San Jose”. In both taxa, we can observe an undivided lip. In the var. undulata, the lip is oblanceolate in outline, obtuse, and widest in 2/3 of its length with distinctly undulate margins in the apical half. Basal auricles are prominent and thick, and the width of the lip at this point is similar to the widest part of the lip. The lip of S. bajaverapacensis is elliptic-ovate in the general outline, the basal part is narrow and shortly and obscurely auriculate, and the apical third of lamina is somewhat undulate along the margins.

Table 3.
Summary of the morphological differences between Schiedeella bajaverapacensis and genetically related S. crenulata.
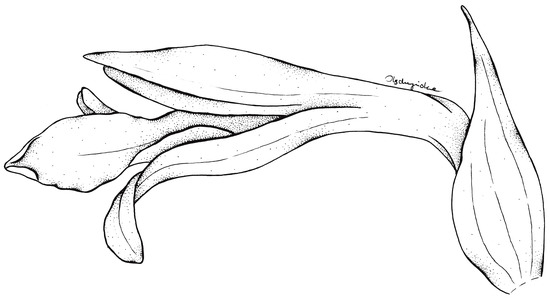
Figure 6.
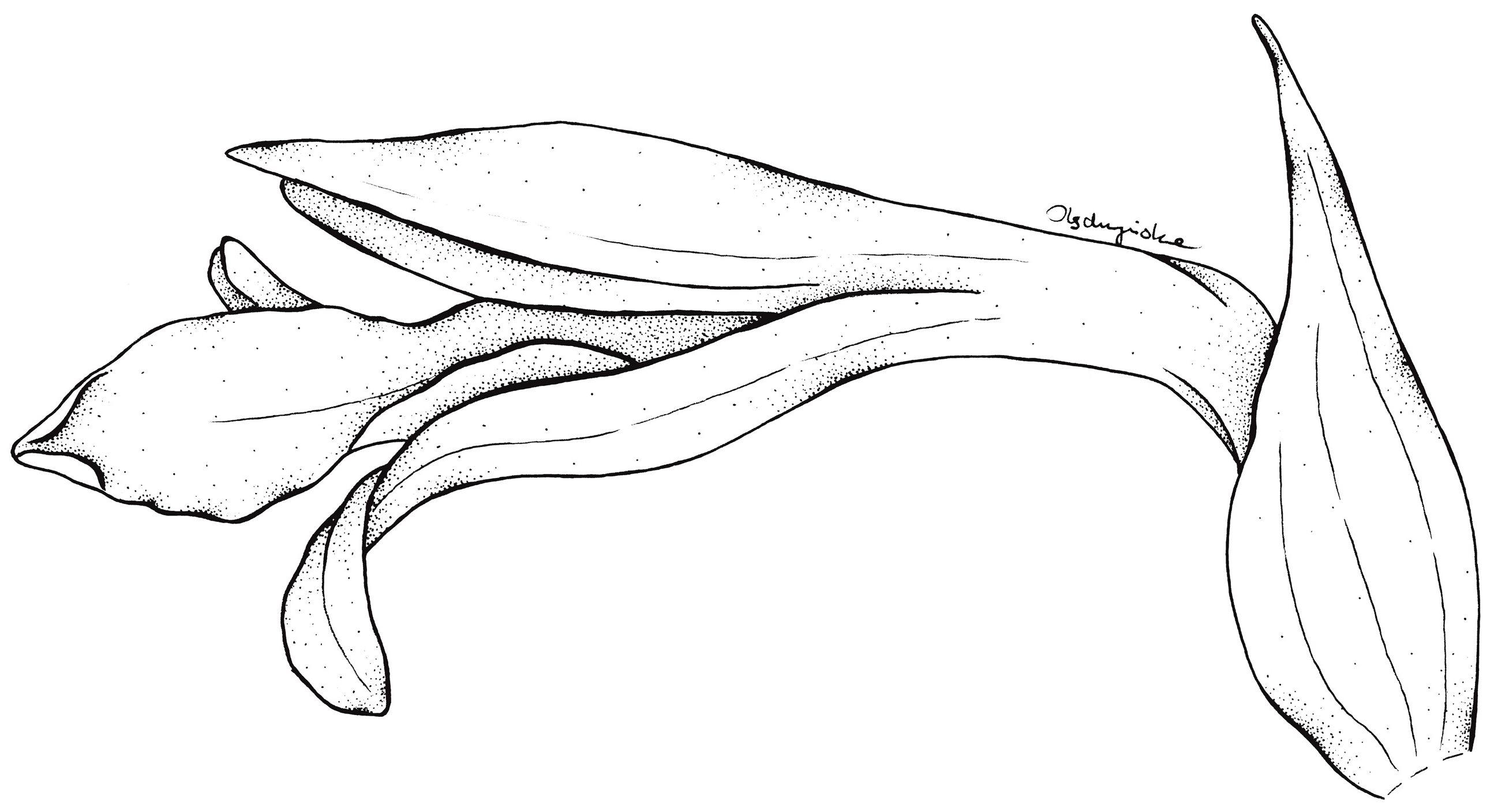
Flower of Schiedeella crenulata—drawn from the holotype (Forderstrom 2592, deposited at AMES). Drawn by N. Olędrzyńska.
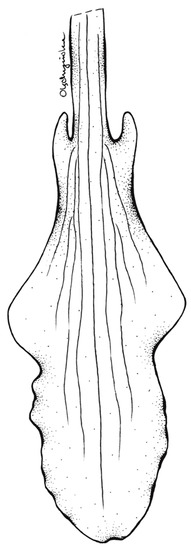
Figure 7.
Lip of Schiedeella crenulata—drawn from the holotype (Forderstrom 2592, deposited at AMES). Drawn by N. Olędrzyńska.
5. Materials and Methods
Plants were collected in the Salamá municipality, Department of Baja Verapaz (Central Guatemala; Figure 8) in January 2022 by Otto Alvarado. After collection, herbarium specimens were prepared and deposited in the herbarium BIGU and UGDA.
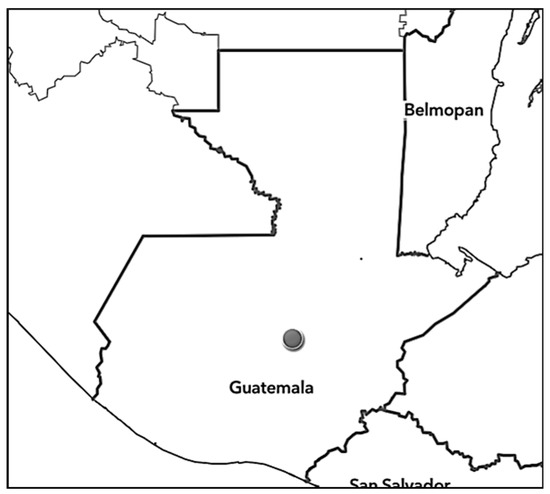
Figure 8.
The approximate location where Schiedeella bajaverapacensis was found.
5.1. Morphological Study
Specimens were investigated according to standard procedures. The first step consisted of creating the database of the information from herbarium labels and taking digital photographs of the sheet. Vegetative parts, i.e., rhizomes, roots, pseudobulbs, leaves, shoots, inflorescences, and floral sheaths, were accurately measured and described. The flower was dissected from the inflorescence and after rehydration was subjected to detailed research using a stereoscopic microscope. In case of doubts regarding a given feature, a larger number of flowers was tested, if possible. Each part of the perianth was accurately measured, drawn, and described. Particular attention was paid to the examination of the lip and the column. The morphology of these structures is extremely important: in the case of the lip, its shape, nervousness, size, the presence of various kinds of swellings, folds, and narrowings. When examining the morphology of the column, particular attention was paid to the construction of viscidium, rostellum, clinandrium, and proportions of the length of the column’s foot to the column, possibly its adhesion with the lateral outer petals of the perianth.
The collected documentation was compared with the type specimens, diagnoses, and, if available, original drawings. Most of the materials were collected and examined during visits to European and American herbaria: AMES, B, BIGU, C, CAY, COL, HJBG, HUA, K, MA, MO, NY, P, RPSC, QCA, QCNE, US, VEN, W, WU (acronyms adopted from Index Herbariorum [16]). We carefully analyzed the data presented in the available literature, especially floras published by Bateman [2], Ames and Correll [3], Archila [4] and Archila et al. [1].
5.2. Molecular Analysis
GenBank accession numbers for the sequences of the new taxon are listed in Table 4. A list of the taxa with their accession numbers is included in Appendix A In order to determine the phylogenetic relationships of the new species, taxa grouped together with Schiedeella based on the results previously published by other researchers were used [13,14].

Table 4.
The accession numbers of GenBank for the sample of Schiedeella bajaverapacensis.
DNA Isolation. Total genomic DNA was extracted using the DNA Sherlock AX Kit (A&A Biotechnology, Gdansk, Poland) following the manufacturer’s protocol. First, the sample was dried in silica gel [17]. For isolation, 28 mg of the dried material (fragment of the flower) was taken. The pellet of DNA was suspended in 50 µL of TE buffer. The quantity and purity of the isolated DNA were determined and checked using NanoDrop One of Thermo Scientific, Waltham, MA, USA.
PCR and sequencing reactions. The amplification was performed using StartWarm HS-PCR Mix (A&A Biotechnology, Poland) following the manufacturer’s protocol. The total volume of the sample was 25 µL, containing 1 µL template DNA (∼10–100 ng) and 0.5 µL of 10 µM of each primer. Parameters for the PCR reaction for nrITS (ITS1 + 5.8S + ITS2) and for the plastid region trnL-trnF were the same as Martin et al. [18]. Tested products of the amplification were purified by applying the Clean-Up Concentrator Kit (A&A Biotechnology, Poland). The final elution was performed in 30 µL of nuclease-free water. Then, purified PCR products were sequenced by Macrogen (Seoul, Republic of Korea-http://dna.macrogen.com/eng/). For the amplification and sequencing, we utilized the same pairs of primers: for the ITS region, 101F and 102R [19], while for the plastid marker trnL-F using primers trnL-c and trnL-f [20].
Phylogenetic reconstruction. For phylogenetic reconstruction, we applied 59 sequences of the ITS region and 48 of the trnL-trnF marker, representing 14 genera and 43 species of Spiranthinae (including new species) of which 12 occur in Guatemala. Some species were sampled more than once. In selecting the taxa of ingroup and outgroup for our analysis, we relied on the results obtained by Salazar et al. [14]. First, we aligned all sequences for single markers using the “align” option according to the MUSCLE algorithm [21] with SeaView [22]. The substitution model for each examined region was calculated with MrModeltest 2.2 [23]. For both datasets, the same best fit model (GRT + G + I) applying AIC (Akaike information criterion) was selected. The Bayesian inference (BI) was performed using four Markov-chain Monte Carlo (MCMC) chains and four independent runs with MrBayes v. 3.2.7a [24]. Each run started from different random trees to ensure that individual runs converged to the same result. Three million generations for both matrices per run were used with sampling 1 tree for every 100 generations until the average standard deviation of the split was smaller than 0.01. Then, the initial 25% of the sampled generations of each chain was as burn-in. Saved trees were summarized in a 50% majority-rule consensus tree and were edited with FigTree v.1.4.4 (http://tree.bio.ed.ac.uk/software/figtree/) and Inkscape (https://inkscape.org/release/inkscape-1.0.2/). The node’s confidence for the obtained trees was assessed by posterior probabilities (PP), which were considered strongly supported when equal to or higher than 0.95 [25].
Author Contributions
Conceptualization, F.L.A.M., D.L.S. and M.M.L.; methodology, M.D., M.M.L. and D.L.S.; validation, M.D., M.M.L. and D.L.S.; formal analysis, M.D., M.M.L. and D.L.S.; investigation, F.L.A.M., M.D., M.M.L. and D.L.S.; resources, F.L.A.M., M.M.L. and D.L.S.; data curation, F.L.A.M., M.D. and M.M.L.; writing—original draft preparation, M.M.L., M.D. and D.L.S.; writing—review and editing, M.M.L. and D.L.S.; visualization, M.M.L.; supervision, M.M.L. and D.L.S.; project administration, M.M.L.; funding acquisition, D.L.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by a subvention of the Faculty of Biology, University of Gdansk: 531-D110-D585-23.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data supporting the reported results can be obtained from the corresponding author upon request.
Acknowledgments
The authors express their gratitude to Otto Alvarado for collecting the plant material and to Natalia Olędrzyńska for preparing the drawings of S. crenulata.
Conflicts of Interest
The authors declare no conflict of interest.
Appendix A

Table A1.
List of the taxa that were used in the phylogenetic analyses with their accession numbers.
Table A1.
List of the taxa that were used in the phylogenetic analyses with their accession numbers.
| Taxon | Accession Number of ITS | Accession Number of trnL-trnF |
|---|---|---|
| Aulosepalum nelsonii | ------- | MG582366.1 |
| Aulosepalum nelsonii subsp. obtusa | AM884876.1 | ------- |
| Aulosepalum riodelayensis | AM884869.1 | ------- |
| Beloglottis mexicana | LT600852.1 | LT600887.1 |
| Coccineorchis standleyi | FN996949.1 | ------- |
| Deiregyne albovaginata | FN641870.1 | FN641882.1 |
| Deiregyne densiflorus | FN641874.1 | FN641886.1 |
| Deiregyne diaphana 1 | KU752292.1 | ------- |
| Deiregyne diaphana 2 | AJ539484.1 | ------- |
| Deiregyne durangensis | FN641867.1 | FN641879.1 |
| Deiregyne eriophora | FN641873.1 | FN641885.1 |
| Deiregyne falcata | FN641871.1 | FN641883.1 |
| Deiregyne pseudopyramidalis | FN641872.1 | FN641884.1 |
| Deiregyne rhombilabia | FN641869.1 | FN641881.1 |
| Dichromanthus aurantiacus 1 | FN996957.1 | FN996970.1 |
| Dichromanthus aurantiacus 2 | FN996956.1 | FN996971.1 |
| Dichromanthus aurantiacus 3 | AJ539485.1 | ------- |
| Dichromanthus cinnabarinus 1 | KU752293.1 | ------- |
| Dichromanthus cinnabarinus 2 | AJ539486.1 | ------- |
| Dichromanthus cinnabarinus subsp. cinnabarinus 1 | FN996952.1 | FN996964.1 |
| Dichromanthus cinnabarinus subsp. cinnabarinus 2 | FN996951.1 | FN996963.1 |
| Dichromanthus cinnabarinus subsp. galeottianus | AM778176.1 | FN996965.1 |
| Dichromanthus michuacanus 1 | FN996955.1 | FN996969.1 |
| Dichromanthus michuacanus 2 | FN996954.1 | FN996968.1 |
| Dichromanthus michuacanus 3 | FN996953.1 | FN996967.1 |
| Dichromanthus michuacanus 4 | AM778177.1 | FN996966.1 |
| Dichromanthus yucundaa | FN996950.1 | FN996962.1 |
| Greenwoodiella garayana 1 | LT600860.1 | LT600894.1 |
| Greenwoodiella garayana 2 | LT600859.1 | LT600893.1 |
| Greenwoodiella garayana 3 | LT600858.1 | LT600895.1 |
| Greenwoodiella garayana 4 | LT600857.1 | LT600892.1 |
| Greenwoodiella romeroana | LT600861.1 | LT600896.1 |
| Greenwoodiella wercklei 1 | LT600865.1 | LT600901.1 |
| Greenwoodiella wercklei 2 | LT600864.1 | LT600900.1 |
| Greenwoodiella wercklei 3 | ------- | LT600899.1 |
| Kionophyton sawyeri | LT600856.1 | LT600891.1 |
| Kionophyton seminuda | MF465022.1 | MG582377.1 |
| Mesadenus chiangii | MK309833.1 | MK310230.1 |
| Mesadenus glaziovii 1 | MG460385.1 | MG460417.1 |
| Mesadenus glaziovii 2 | MK309832.1 | ------- |
| Mesadenus lucayanus 1 | MK309834.1 | MK310231.1 |
| Mesadenus lucayanus 2 | KU752294.1 | ------- |
| Mesadenus lucayanus 3 | AJ539488.1 | ------- |
| Mesadenus polyanthus | AM778175.1 | LT600902.1 |
| Mesadenus tenuissimus 1 | MK309836.1 | MK310233.1 |
| Mesadenus tenuissimus 2 | MK309835.1 | MK310232.1 |
| Physogyne gonzalezii | LT600855.1 | LT600890.1 |
| Pseudogoodyera pseudogoodyeroides | LT600854.1 | LT600889.1 |
| Pseudogoodyera wrightii | ------- | MG582376.1 |
| Schiedeella affinis | ------- | LT600903.1 |
| Schiedeella crenulata | FN641868.1 | FN641880.1 |
| Schiedeella faucisanguinea | AJ539496.1 | ------- |
| Schiedeella llaveana 1 | KU752295.1 | ------- |
| Schiedeella llaveana 2 | AJ539487.1 | ------- |
| Schiedeella nagelii | MK309837.1 | MK310234.1 |
| Schiedeella sp. 1 | ------- | LT600897.1 |
| Schiedeella sp. 2 | ------- | LT600898.1 |
| Schiedeella tenella | MK309838.1 | MK310235.1 |
| Schiedeella williamsiana | MK309839.1 | MK310236.1 |
| Sotoa confusa | FN641865.1 | FN641876.1 |
| Spiranthes sinensis | LT600853.1 | LT600888.1 |
| Stenorrhynchos albidomaculatu | FN996948.1 | ------- |
| Stenorrhynchos millei | FN996946.1 | ------- |
| Stenorrhynchos speciosum | FN996947.1 | ------- |
References
- Archila, F.; Szlachetko, D.L.; Chiron, G.; Lipińska, M.M.; Mystkowska, K.; Bertolini, V. Orchid Genera and Species in Guatemala; Koeltz Botanical Books: Oberreifenberg, Germany, 2018; pp. 1–724. [Google Scholar]
- Bateman, J. Orchidaceae of Mexico and Guatemala; J. Ridgway & Sons: London, UK, 1843; pp. 1–264. [Google Scholar]
- Ames, O.; Correll, D.S. Orchids of Guatemala; Chicago Natural History Museum: Chicago, IL, USA, 1952; pp. 1–779. [Google Scholar]
- Archila, F. Recolección, Cultivo y Reproducción de Orquídeas. Escuela Nacional Central de Agricultura (ENCA). Practica Profesional Agrícola y Forestal Supervisada (PPAFS). Informe Técnico; Barcena: Villa Nueva, Guatemala, 1992. [Google Scholar]
- Archila, F. Listado de Orquídeas de Guatemala. Revista Guatemal. 2014, 17, 32–71. [Google Scholar]
- Schlechter, R. Versuch einer systematischen Neuordnung der Spiranthinae. Beih. Bot. Centralbl. 1920, 37, 317–454. [Google Scholar]
- Szlachetko, D.L. Genera and species of the subtribe Spiranthinae (Orchidaceae). 2. A revision of Schiedeella. Fragm. Flor. Geobot. Pol. 1992, 37, 157–204. [Google Scholar]
- Burns-Balogh, P. Generic redefinition in the subtribe Spiranthinae (Orchidaceae). Am. J. Bot. 1982, 69, 1119–1132. [Google Scholar] [CrossRef]
- Garay, L.A. A generic revision of the Spiranthinae. Bot. Mus. Leafl. 1982, 28, 278–425. [Google Scholar] [CrossRef]
- Szlachetko, D.L. Systema Orchidalium. Fragm. Flor. Geobot. Pol. 1995, (Suppl. 3), 1–152. [Google Scholar]
- Burns-Balogh, P. A synopsis of Mexican Spiranthinae. Orquídea 1986, 10, 47–96. [Google Scholar]
- Szlachetko, D.L.; Rutkowski, P.; Mytnik, J. Contributions to the taxonomic revision of the subtribes Spiranthinae, Stenorrhynchidinae and Cyclopogoninae (Orchidaceae) in Mesoamerica and the Antilles. Pol. Bot. Stud. 2005, 20, 3–387. [Google Scholar]
- Salazar, G.A.; Hernández-López, T.J.; Sharma, J.; Jiménez-Machorro, R.; Cabrer, L.I.; Treviño-Carreón, J. Greenwoodiella, a new genus of Spiranthinae (Orchidaceae) from North and Central America and the Greater Antilles, with a new species from the Chihuahuan Desert. Syst. Bot. 2016, 41, 823–838. [Google Scholar] [CrossRef]
- Salazar, G.A.; Batista, J.A.N.; Cabrera, L.I.; Van den Berg, C.; Whitten, W.M.; Smidt, E.C.; Buzatto, C.R.; Singer, R.B.; Gerlach, G.; Jimenez-Machorro, R.; et al. Phylogenetic systematics of subtribe Spiranthinae (Orchidaceae: Orchidoideae: Cranichideae) based on nuclear and plastid DNA sequences of a nearly complete generic sample. Bot. J. Linn. Soc. 2018, 186, 273–303. [Google Scholar] [CrossRef]
- IUCN. Guidelines for Using the IUCN Red List Categories and Criteria. Version 14. 2019. Available online: https://www.iucnredlist.org/resources/redlistguidelines (accessed on 29 June 2021).
- Holmgren, P.K.; Holmgren, N.H. Index Herbariorum. Taxon 1993, 42, 489–505. [Google Scholar] [CrossRef]
- Chase, M.W.; Hills, H.H. Silica gel: An ideal material for field preservation of leaf samples for DNA studies. Taxon 1991, 40, 215–220. [Google Scholar] [CrossRef]
- Martin, C.M.; Morales, A.M.; Dudek, M.; Szlachetko, D.L. Pachygenium laurense (Orchidaceae, Spiranthinae), a new orchid species from Argentina—Morphological evidence and phylogenetic reconstruction. PeerJ 2022, 10, 13433. [Google Scholar] [CrossRef] [PubMed]
- Douzery, E.J.P.; Pridgeon, M.A.; Kores, P.; Linder, H.P.; Kurzweil, H.; Chase, M.W. Molecular phylogenetics of Disae (Orchidaeae): A contribution from nuclear ribosomal ITS sequences. Am. J. Bot. 1999, 86, 887–899. [Google Scholar] [CrossRef] [PubMed]
- Taberlet, P.; Gielly, L.; Pautou, G.; Bouvet, J. Universal primers for amplification of three non-coding regions of chloroplast DNA. Plant Mol. Biol. 1991, 17, 1105–1109. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Galtier, N.; Gouy, M.; Gautier, C. SeaView and Phylo_win, two graphic tools for sequence alignment and molecular phylogeny. Bioinformatics 1996, 12, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Nylander, J.A.A. MrModeltest v2. Program Distributed by the Author. Evolutionary Biology Centre, Uppsala University. Available online: http://www.abc.se/~nylander/ (accessed on 20 January 2023).
- Ronquist, F.; Teslenko, M.; Pvander, M.; Huelsenbeck, J. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Bot. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed]
- Huelsenbeck, J.P.; Ronquist, F. MrBayes: Bayesian inference of phylogeny. Bioinformatics 2001, 17, 754–755. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).