Bilirubin Exerts Protective Effects on Alveolar Type II Pneumocytes in an In Vitro Model of Oxidative Stress
Abstract
1. Introduction
2. Results
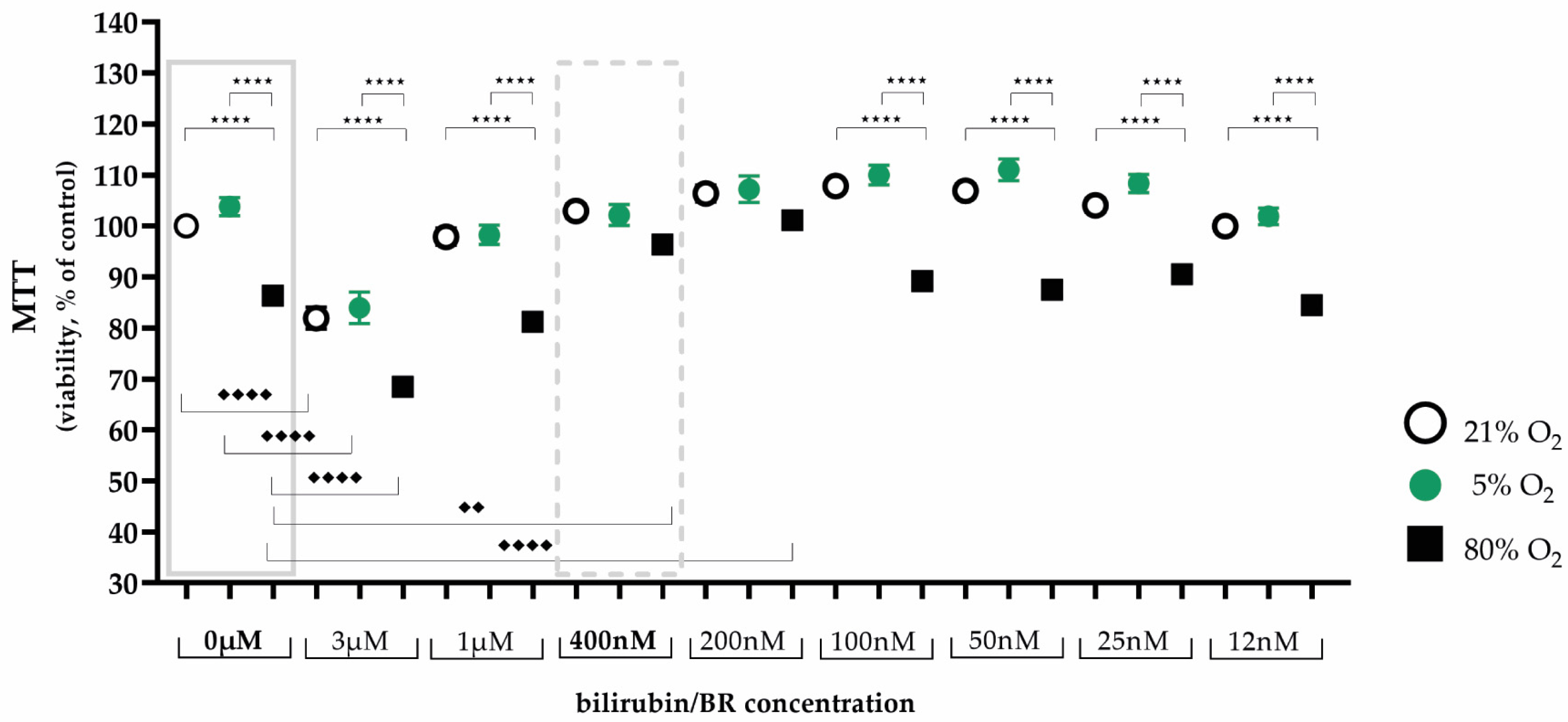
2.1. Protective Bilirubin Concentration for Cell Viability under Oxidative Stress
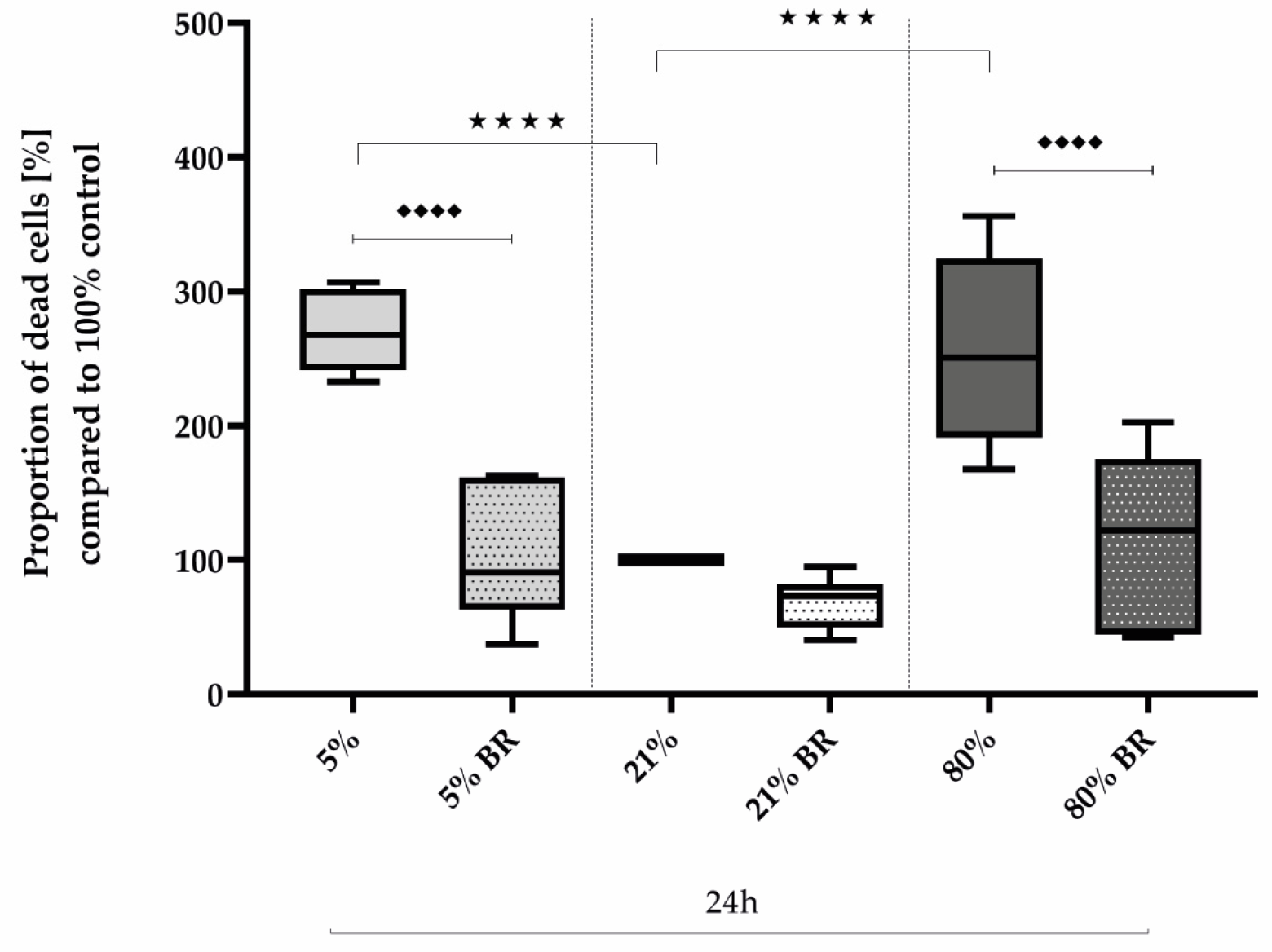
2.2. Protective Bilirubin for Cell Survival under Oxidative Stress
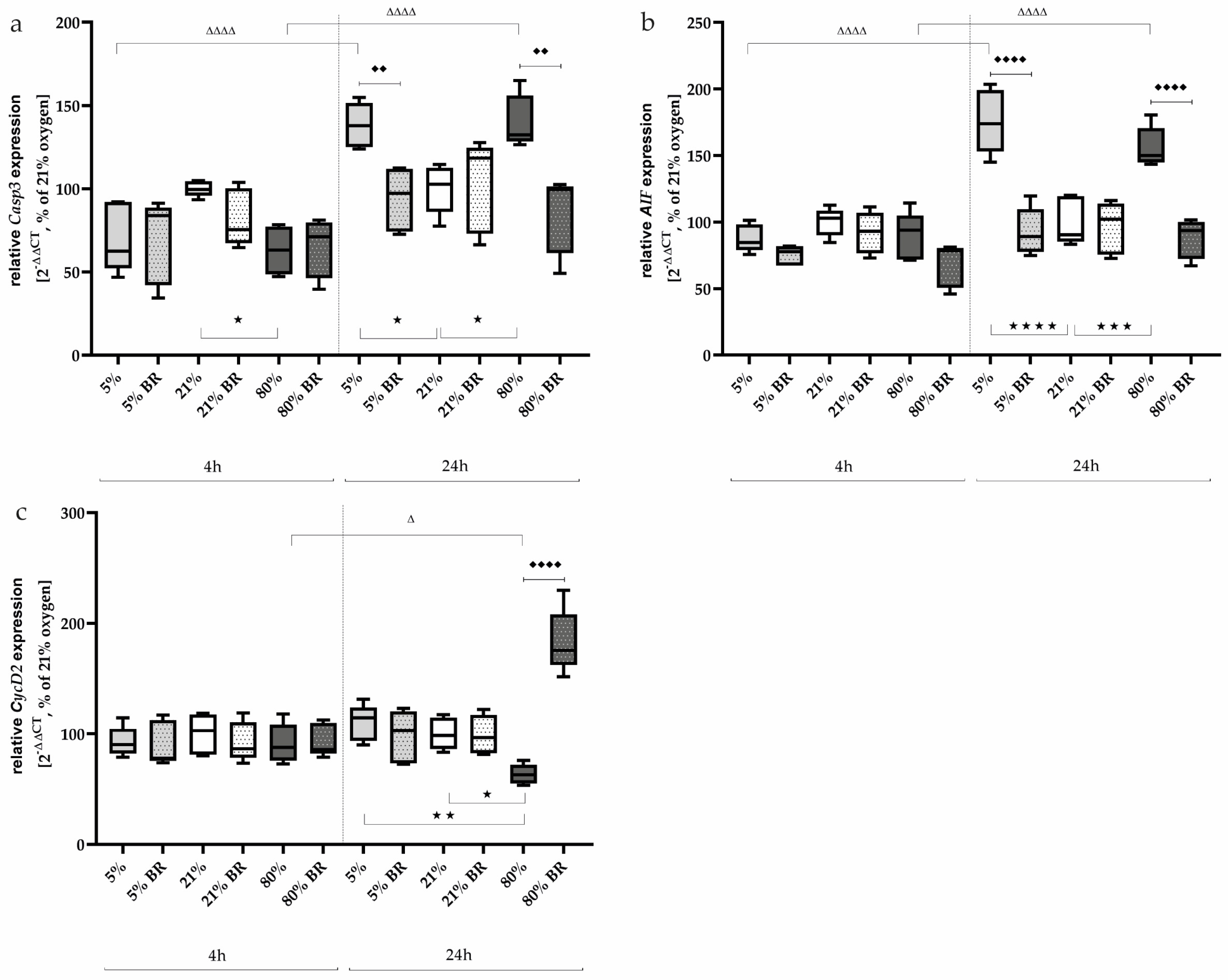
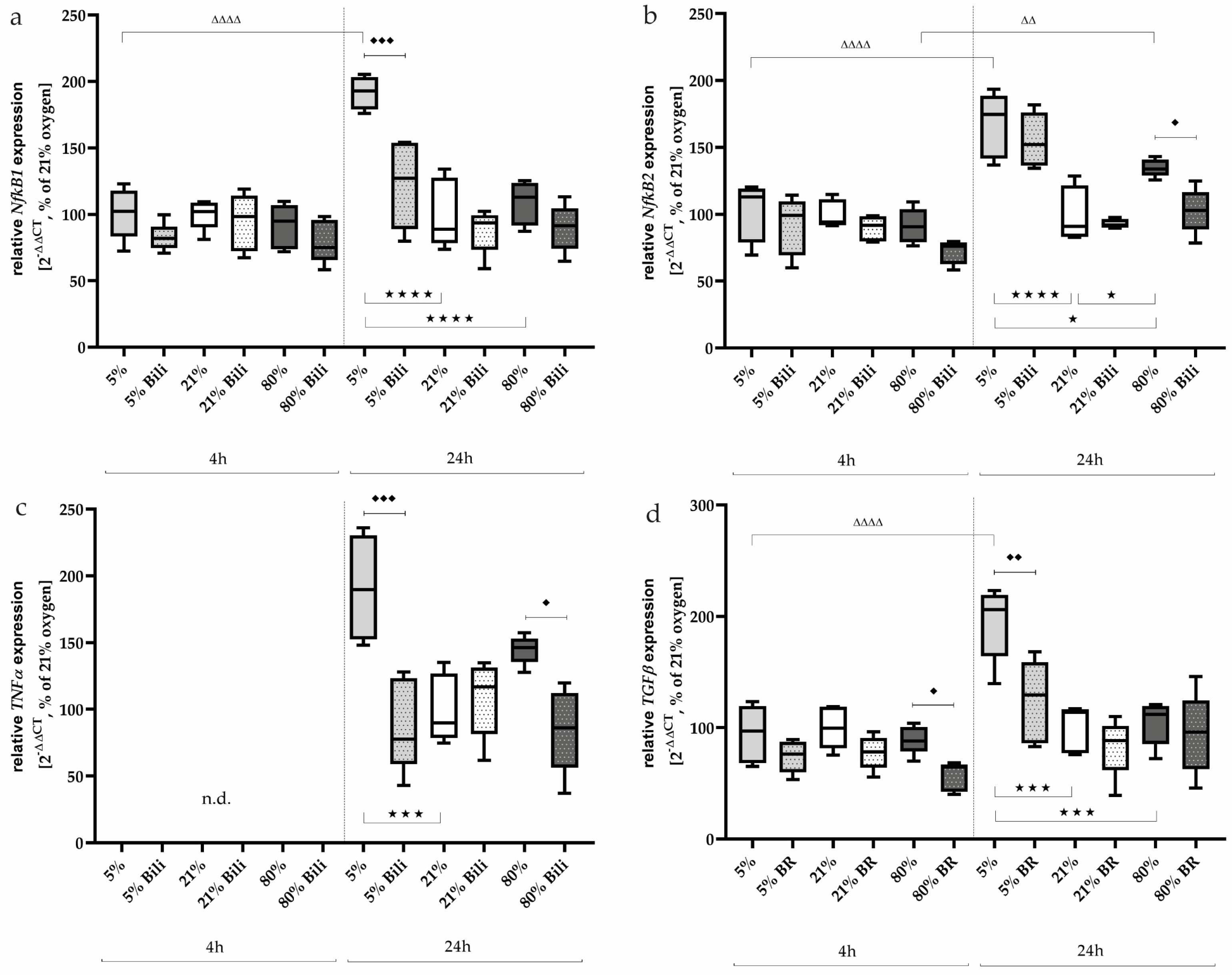
2.3. Bilirubin Modulates Oxidative Stress-Associated Transcription Factors and Inflammatory Mediators
3. Discussion
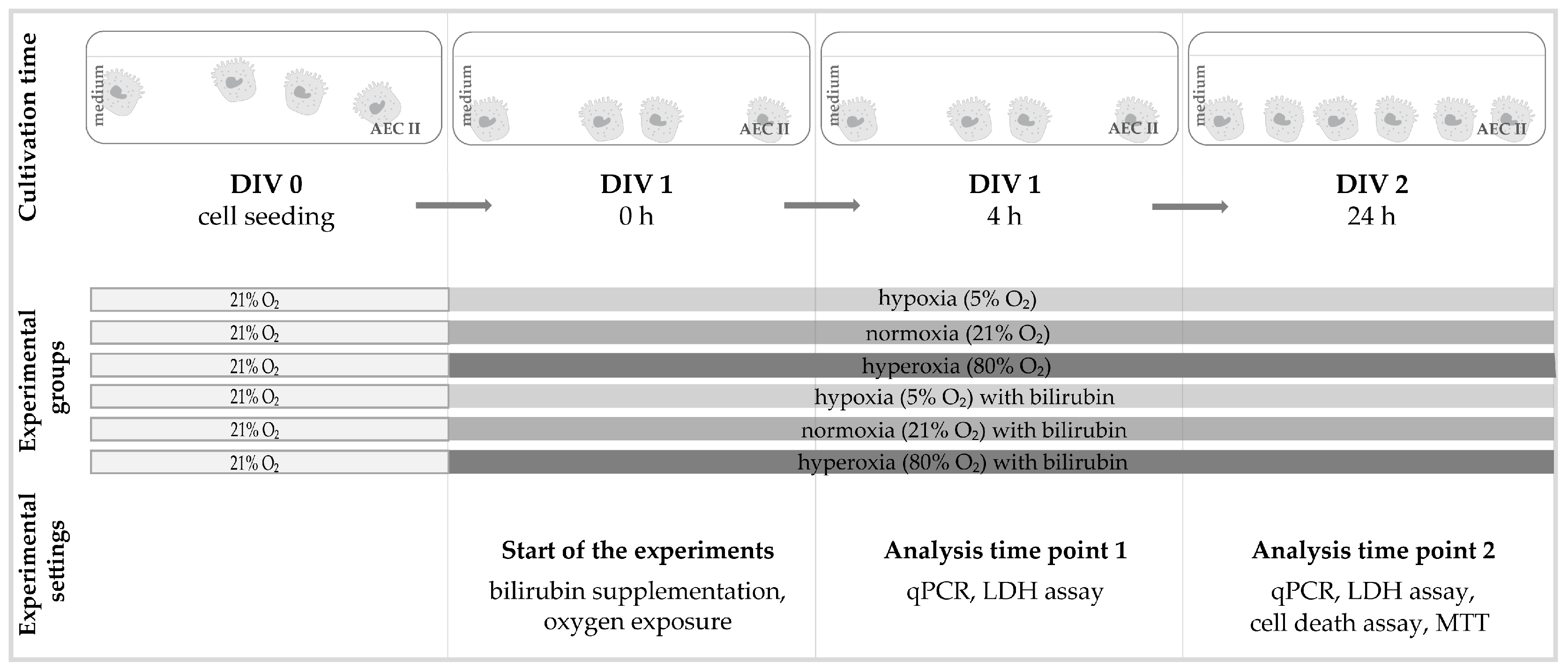
4. Materials and Methods
4.1. Cell Culture: Cell Line AEC II/ RLE-6TN
4.2. Determining the Oxygen Level and Relevance of the Cellular Model
4.3. Oxygen Exposure and Bilirubin Administration
4.4. RNA-Extraction and qPCR
4.5. EarlyTox Live/Dead Assay
4.6. MTT
4.7. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gebb, S.A.; Jones, P.L. Hypoxia and lung branching morphogenesis. Adv. Exp. Med. Biol. 2003, 543, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Hashmi, M.F. Partial Pressure of Oxygen. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Alva, R.; Mirza, M.; Baiton, A.; Lazuran, L.; Samokysh, L.; Bobinski, A.; Cowan, C.; Jaimon, A.; Obioru, D.; Al Makhoul, T.; et al. Oxygen toxicity: Cellular mechanisms in normobaric hyperoxia. Cell Biol. Toxicol. 2023, 39, 111–143. [Google Scholar] [CrossRef]
- Turgut, M.; Başaran, O.; Cekmen, M.; Karataş, F.; Kurt, A.; Aygün, A.D. Oxidant and antioxidant levels in preterm newborns with idiopathic hyperbilirubinaemia. J. Paediatr. Child Health 2004, 40, 633–637. [Google Scholar] [CrossRef]
- Bellezza, I.; Giambanco, I.; Minelli, A.; Donato, R. Nrf2-Keap1 signaling in oxidative and reductive stress. Biochim. Biophys. Acta Mol. Cell Res. 2018, 1865, 721–733. [Google Scholar] [CrossRef] [PubMed]
- Baird, L.; Yamamoto, M. The Molecular Mechanisms Regulating the KEAP1-NRF2 Pathway. Mol. Cell. Biol. 2020, 40, e00099-20. [Google Scholar] [CrossRef] [PubMed]
- Lingappan, K. NF-κB in Oxidative Stress. Curr. Opin. Toxicol. 2018, 7, 81–86. [Google Scholar] [CrossRef]
- Mitra, S.; Rennie, J. Neonatal jaundice: Aetiology, diagnosis and treatment. Br. J. Hosp. Med. 2017, 78, 699–704. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Koritala, T.; Jialal, I. Unconjugated Hyperbilirubinemia. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2024. [Google Scholar]
- Amin, S.B.; Saluja, S.; Kler, N. Unbound Bilirubin and Acute Bilirubin Encephalopathy in Infants Born Late Preterm and Term with Significant Hyperbilirubinemia. J. Pediatr. 2024, 266, 113880. [Google Scholar] [CrossRef]
- Dennery, P.A.; Seidman, D.S.; Stevenson, D.K. Neonatal hyperbilirubinemia. N. Engl. J. Med. 2001, 344, 581–590. [Google Scholar] [CrossRef]
- Oh, W.; Tyson, J.E.; Fanaroff, A.A.; Vohr, B.R.; Perritt, R.; Stoll, B.J.; Ehrenkranz, R.A.; Carlo, W.A.; Shankaran, S.; Poole, K.; et al. Association between peak serum bilirubin and neurodevelopmental outcomes in extremely low birth weight infants. Pediatrics 2003, 112, 773–779. [Google Scholar] [CrossRef]
- Rose, J.; Vassar, R. Movement disorders due to bilirubin toxicity. Semin. Fetal Neonatal Med. 2015, 20, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Watchko, J.F. Bilirubin-Induced Neurotoxicity in the Preterm Neonate. Clin. Perinatol. 2016, 43, 297–311. [Google Scholar] [CrossRef] [PubMed]
- Haghdoost-Yazdi, H. Chapter 41—Neurotoxicity induced by biliverdin and bilirubin. In Natural Molecules in Neuroprotection and Neurotoxicity; Oliveira, M.R.d., Ed.; Academic Press: Cambridge, MA, USA, 2024; pp. 997–1019. [Google Scholar]
- Hansen, T.W.R.; Wong, R.J.; Stevenson, D.K. Molecular Physiology and Pathophysiology of Bilirubin Handling by the Blood, Liver, Intestine, and Brain in the Newborn. Physiol. Rev. 2020, 100, 1291–1346. [Google Scholar] [CrossRef] [PubMed]
- Dennery, P.A.; McDonagh, A.F.; Spitz, D.R.; Rodgers, P.A.; Stevenson, D.K. Hyperbilirubinemia results in reduced oxidative injury in neonatal Gunn rats exposed to hyperoxia. Free Radic. Biol. Med. 1995, 19, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Martelli, E.; Bertini, G.; Pezzati, M.; Filippi, L.; Rossetti, M.; Rizzuti, G.; Rubaltelli, F.F. Plasma bilirubin level and oxidative stress in preterm infants. Archives of disease in childhood. Fetal Neonatal Ed. 2003, 88, F119–F123. [Google Scholar] [CrossRef] [PubMed]
- Stocker, R.; Yamamoto, Y.; McDonagh, A.F.; Glazer, A.N.; Ames, B.N. Bilirubin is an antioxidant of possible physiological importance. Science 1987, 235, 1043–1046. [Google Scholar] [CrossRef] [PubMed]
- Ryter, S.W.; Morse, D.; Choi, A.M. Carbon monoxide and bilirubin: Potential therapies for pulmonary/vascular injury and disease. Am. J. Respir. Cell Mol. Biol. 2007, 36, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Pant, P.; Basu, S.; Rao, G.R.K.; Khanna, H.D. Oxidative Stress in Neonatal Hyperbilirubinemia. J. Trop. Pediatr. 2006, 53, 69–71. [Google Scholar] [CrossRef] [PubMed]
- Dani, C.; Poggi, C.; Pratesi, S. Bilirubin and oxidative stress in term and preterm infants. Free Radic. Res. 2019, 53, 2–7. [Google Scholar] [CrossRef]
- Doğan, M.; Peker, E.; Kirimi, E.; Sal, E.; Akbayram, S.; Erel, O.; Ocak, A.R.; Tuncer, O. Evaluation of oxidant and antioxidant status in infants with hyperbilirubinemia and kernicterus. Hum. Exp. Toxicol. 2011, 30, 1751–1760. [Google Scholar] [CrossRef]
- Dani, C.; Bertini, G.; Cecchi, A.; Corsini, I.; Pratesi, S.; Rubaltelli, F.F. Association between peak serum bilirubin and severity of respiratory distress syndrome in infants of less than 30 weeks’ gestation. J. Perinat. Med. 2007, 35, 141–146. [Google Scholar] [CrossRef] [PubMed]
- Bélanger, S.; Lavoie, J.C.; Chessex, P. Influence of bilirubin on the antioxidant capacity of plasma in newborn infants. Biol. Neonate. 1997, 71, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Duarte, T.L.; Talbot, N.P.; Drakesmith, H. NRF2 and Hypoxia-Inducible Factors: Key Players in the Redox Control of Systemic Iron Homeostasis. Antioxid. Redox Signal. 2021, 35, 433–452. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Magilnick, N.; Lee, C.; Kalmaz, D.; Ou, X.; Chan, J.Y.; Lu, S.C. Nrf1 and Nrf2 Regulate Rat Glutamate-Cysteine Ligase Catalytic Subunit Transcription Indirectly via NF-κB and AP-1. Mol. Cell. Biol. 2005, 25, 5933–5946. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.M.U.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 signaling pathway: Pivotal roles in inflammation. Biochim. Et Biophys. Acta (BBA)—Mol. Basis Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Savinova, O.V.; Hoffmann, A.; Ghosh, G. The Nfkb1 and Nfkb2 proteins p105 and p100 function as the core of high-molecular-weight heterogeneous complexes. Mol. Cell 2009, 34, 591–602. [Google Scholar] [CrossRef]
- Liu, R.M.; Desai, L.P. Reciprocal regulation of TGF-β and reactive oxygen species: A perverse cycle for fibrosis. Redox Biol. 2015, 6, 565–577. [Google Scholar] [CrossRef]
- Knudsen, L.; Ochs, M. The micromechanics of lung alveoli: Structure and function of surfactant and tissue components. Histochem. Cell Biol. 2018, 150, 661–676. [Google Scholar] [CrossRef] [PubMed]
- Haddad, J.J. Oxygen-sensing mechanisms and the regulation of redox-responsive transcription factors in development and pathophysiology. Respir. Res. 2002, 3, 26. [Google Scholar] [CrossRef]
- Endesfelder, S.; Strauß, E.; Scheuer, T.; Schmitz, T.; Bührer, C. Antioxidative effects of caffeine in a hyperoxia-based rat model of bronchopulmonary dysplasia. Respir. Res. 2019, 20, 88. [Google Scholar] [CrossRef]
- Cho, H.Y.; Jedlicka, A.E.; Reddy, S.P.; Kensler, T.W.; Yamamoto, M.; Zhang, L.Y.; Kleeberger, S.R. Role of NRF2 in protection against hyperoxic lung injury in mice. Am. J. Respir. Cell Mol. Biol. 2002, 26, 175–182. [Google Scholar] [CrossRef]
- Tamatam, C.M.; Reddy, N.M.; Potteti, H.R.; Ankireddy, A.; Noone, P.M.; Yamamoto, M.; Kensler, T.W.; Reddy, S.P. Preconditioning the immature lung with enhanced Nrf2 activity protects against oxidant-induced hypoalveolarization in mice. Sci. Rep. 2020, 10, 19034. [Google Scholar] [CrossRef]
- Li, K.; Zhang, F.; Wei, L.; Han, Z.; Liu, X.; Pan, Y.; Guo, C.; Han, W. Recombinant Human Elafin Ameliorates Chronic Hyperoxia-Induced Lung Injury by Inhibiting Nuclear Factor-Kappa B Signaling in Neonatal Mice. J. Interferon Cytokine Res. 2020, 40, 320–330. [Google Scholar] [CrossRef]
- McKenna, S.; Michaelis, K.A.; Agboke, F.; Liu, T.; Han, K.; Yang, G.; Dennery, P.A.; Wright, C.J. Sustained hyperoxia-induced NF-κB activation improves survival and preserves lung development in neonatal mice. American journal of physiology. Lung Cell. Mol. Physiol. 2014, 306, L1078–L1089. [Google Scholar] [CrossRef]
- Haddad, J.J. Oxygen homeostasis, thiol equilibrium and redox regulation of signalling transcription factors in the alveolar epithelium. Cell. Signal. 2002, 14, 799–810. [Google Scholar] [CrossRef]
- Yang, Y.; Dieter, M.Z.; Chen, Y.; Shertzer, H.G.; Nebert, D.W.; Dalton, T.P. Initial Characterization of the Glutamate-Cysteine Ligase Modifier Subunit Gclm(−/−) Knockout Mouse: Novel model system for a severely compromised oxidative stress response. J. Biol. Chem. 2002, 277, 49446–49452. [Google Scholar] [CrossRef]
- Stegen, S.; van Gastel, N.; Eelen, G.; Ghesquière, B.; D’Anna, F.; Thienpont, B.; Goveia, J.; Torrekens, S.; Van Looveren, R.; Luyten, F.P.; et al. HIF-1α Promotes Glutamine-Mediated Redox Homeostasis and Glycogen-Dependent Bioenergetics to Support Postimplantation Bone Cell Survival. Cell Metab. 2016, 23, 265–279. [Google Scholar] [CrossRef]
- Brown, G.C.; Borutaite, V. Regulation of apoptosis by the redox state of cytochrome c. Biochim. Et Biophys. Acta 2008, 1777, 877–881. [Google Scholar] [CrossRef]
- Endesfelder, S.; Strauß, E.; Bendix, I.; Schmitz, T.; Bührer, C. Prevention of Oxygen-Induced Inflammatory Lung Injury by Caffeine in Neonatal Rats. Oxidative Med. Cell. Longev. 2020, 2020, 3840124. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Peng, L.Q.; Zhao, A.L. Hyperoxia induces the apoptosis of alveolar epithelial cells and changes of pulmonary surfactant proteins. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Burhans, W.C.; Heintz, N.H. The cell cycle is a redox cycle: Linking phase-specific targets to cell fate. Free Radic. Biol. Med. 2009, 47, 1282–1293. [Google Scholar] [CrossRef]
- Haddad, J.J.; Land, S.C. O(2)-evoked regulation of HIF-1alpha and NF-kappaB in perinatal lung epithelium requires glutathione biosynthesis. American journal of physiology. Lung Cell. Mol. Physiol. 2000, 278, L492–L503. [Google Scholar] [CrossRef]
- Rahman, I.; MacNee, W. Oxidative stress and regulation of glutathione in lung inflammation. Eur. Respir. J. 2000, 16, 534–554. [Google Scholar] [CrossRef]
- Ong, C.H.; Tham, C.L.; Harith, H.H.; Firdaus, N.; Israf, D.A. TGF-β-induced fibrosis: A review on the underlying mechanism and potential therapeutic strategies. Eur. J. Pharmacol. 2021, 911, 174510. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, J.; Tetzlaff, W.; Paty, D.W.; Cynader, M.S. Biliverdin reductase, a major physiologic cytoprotectant, suppresses experimental autoimmune encephalomyelitis. Free Radic. Biol. Med. 2006, 40, 960–967. [Google Scholar] [CrossRef]
- Baranano, D.E.; Rao, M.; Ferris, C.D.; Snyder, S.H. Biliverdin reductase: A major physiologic cytoprotectant. Proc. Natl. Acad. Sci. USA 2002, 99, 16093–16098. [Google Scholar] [CrossRef]
- Doré, S.; Snyder, S.H. Neuroprotective action of bilirubin against oxidative stress in primary hippocampal cultures. Ann. N. Y. Acad. Sci. 1999, 890, 167–172. [Google Scholar] [CrossRef]
- Ziberna, L.; Martelanc, M.; Franko, M.; Passamonti, S. Bilirubin is an Endogenous Antioxidant in Human Vascular Endothelial Cells. Sci. Rep. 2016, 6, 29240. [Google Scholar] [CrossRef]
- Martinez, C.A.; Cistulli, P.A.; Cook, K.M. A Cell Culture Model that Mimics Physiological Tissue Oxygenation Using Oxygen-permeable Membranes. Bio-Protocol 2019, 9, e3371. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Berridge, M.V.; Herst, P.M.; Tan, A.S. Tetrazolium dyes as tools in cell biology: New insights into their cellular reduction. Biotechnol. Annu. Rev. 2005, 11, 127–152. [Google Scholar] [CrossRef]
- Heise, J.; Schmitz, T.; Bührer, C.; Endesfelder, S. Protective Effects of Early Caffeine Administration in Hyperoxia-Induced Neurotoxicity in the Juvenile Rat. Antioxidants 2023, 12, 295. [Google Scholar] [CrossRef]

| Oligonucleotide Sequence 5′–3′ | Accession No. | |
|---|---|---|
| AIF | ||
| forward | CACAAAGACACTGCAGTTCAGACA | NM_031356.1 |
| reverse | AGGTCCTGAGCAGAGACATAGAAAG | |
| probe | 6–FAM–AGAAGCATCTATTTCCAGCC–TAMRA | |
| Casp3 | ||
| forward | ACAGTGGAACTGACGATGATATGG | NM_012922.2 |
| reverse | AATAGTAACCGGGTGCGGTAGA | |
| probe | 6–FAM–ATGCCAGAAGATACCAGTGG–TAMRA | |
| CycD2 | ||
| forward | CGTACATGCGCAGGATGGT | NM_199501.1 |
| reverse | AATTCATGGCCAGAGGAAAGAC | |
| probe | 6–FAM–TGGATGCTAGAGGTCTGTGA–TAMRA | |
| GCLC | ||
| forward | GGAGGACAACATGAGGAAACG | NM_012815.2 |
| reverse | GCTCTGGCAGTGTGAATCCA | |
| probe | 6–FAM–TCAGGCTCTTTGCACGATAA–TAMRA | |
| Hif1α | ||
| forward | GCGCCTCTTCGACAAGCTT | NM_024359.2 |
| reverse | CTGCCGAAGTCCAGTGATATGA | |
| probe | 6–FAM–AGAGCCCGATGCCCTGACTCTGCT–TAMRA | |
| HPRT | ||
| forward | GGAAAGAACGTCTTGATTGTTGAA | NM_012583.2 |
| reverse | CCAACACTTCGAGAGGTCCTTTT | |
| probe | 6–FAM–CTTTCCTTGGTCAAGCAGTACAGCCCC–TAMRA | |
| Keap1 | ||
| forward | GATCGGCTGCACGGAACT | NM_057152.2 |
| reverse | GCAGTGTGACAGGTTGAAGAACTC | |
| probe | 6–FAM–CTCGGGAGTATATCTACATGC–TAMRA | |
| MCP1 | ||
| forward | AGCATCCACGTGCTGTCTCA | NM_031530.1 |
| reverse | GCCGACTCATTGGGATCATC | |
| probe | 6–FAM–AGATGCAGTTAATGCCCCAC–TAMRA | |
| MIP2 | ||
| forward | CCTACCAAGGGTTGACTTCAAGA | NM_053647.1 |
| reverse | GCTTCAGGGTTGAGACAAACTTC | |
| probe | 6–FAM–AGACAGAAGTCATAGCCACT–TAMRA | |
| NfκB1 | ||
| forward | GACCCAAGGACATGGTGGTT | NM_001276711.1 |
| reverse | TCATCCGTGCTTCCAGTGTTT | |
| probe | 6–FAM–CTGGGAATACTTCACGTGAC–TAMRA | |
| NfκB2 | ||
| forward | GCCTAAACAGCGAGGCTTCA | NM_001008349.1 |
| reverse | TCTTCCGGCCCTTCTCACT | |
| probe | 6–FAM–TTTCGATATGGCTGTGAAGG–TAMRA | |
| Nrf2 | ||
| forward | ACTCCCAGGTTGCCCACAT | NM_031789.2 |
| reverse | GCGACTCATGGTCATCTACAAATG | |
| probe | 6–FAM–CTTTGAAGACTGTATGCAGC–TAMRA | |
| TGFβ | ||
| forward | CCTGCAGAGATTCAAGTCAACTGT | NM_021578.2 |
| reverse | GTCAGCAGCCGGTTACCAA | |
| probe | 6–FAM–CAACAATTCCTGGCGTT–TAMRA | |
| TNFα | ||
| forward | CCCCCAATCTGTGTCCTTCTAAC | NM_012675.2 |
| reverse | CGTCTCGTGTGTTTCTGAGCAT | |
| probe | 6–FAM–TAGAAAGGGAATTGTGGCTC–TAMRA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Endesfelder, S.; Schmitz, T.; Bührer, C. Bilirubin Exerts Protective Effects on Alveolar Type II Pneumocytes in an In Vitro Model of Oxidative Stress. Int. J. Mol. Sci. 2024, 25, 5323. https://doi.org/10.3390/ijms25105323
Endesfelder S, Schmitz T, Bührer C. Bilirubin Exerts Protective Effects on Alveolar Type II Pneumocytes in an In Vitro Model of Oxidative Stress. International Journal of Molecular Sciences. 2024; 25(10):5323. https://doi.org/10.3390/ijms25105323
Chicago/Turabian StyleEndesfelder, Stefanie, Thomas Schmitz, and Christoph Bührer. 2024. "Bilirubin Exerts Protective Effects on Alveolar Type II Pneumocytes in an In Vitro Model of Oxidative Stress" International Journal of Molecular Sciences 25, no. 10: 5323. https://doi.org/10.3390/ijms25105323
APA StyleEndesfelder, S., Schmitz, T., & Bührer, C. (2024). Bilirubin Exerts Protective Effects on Alveolar Type II Pneumocytes in an In Vitro Model of Oxidative Stress. International Journal of Molecular Sciences, 25(10), 5323. https://doi.org/10.3390/ijms25105323






