Blockade of mTORC1 via Rapamycin Suppresses 27-Hydroxycholestrol-Induced Inflammatory Responses
Abstract
:1. Introduction
2. Results
2.1. Effects of Rapamycin on 27OHChol-Induced Inflammatory Responses in THP-1 Cells
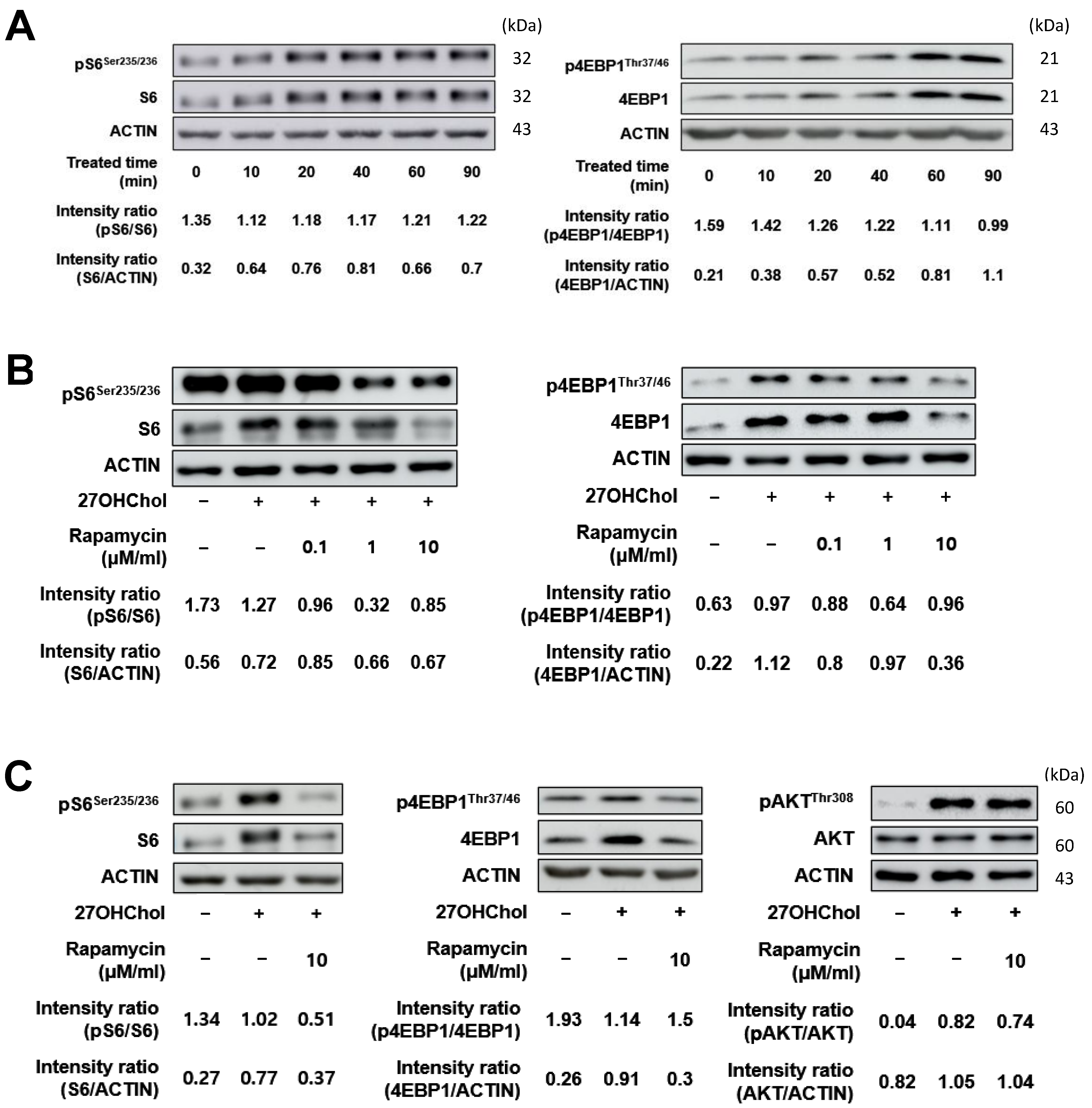
2.2. Effects of Rapamycin on the Downstream Proteins of the mTORC1 Signaling Pathway in THP-1 Cells
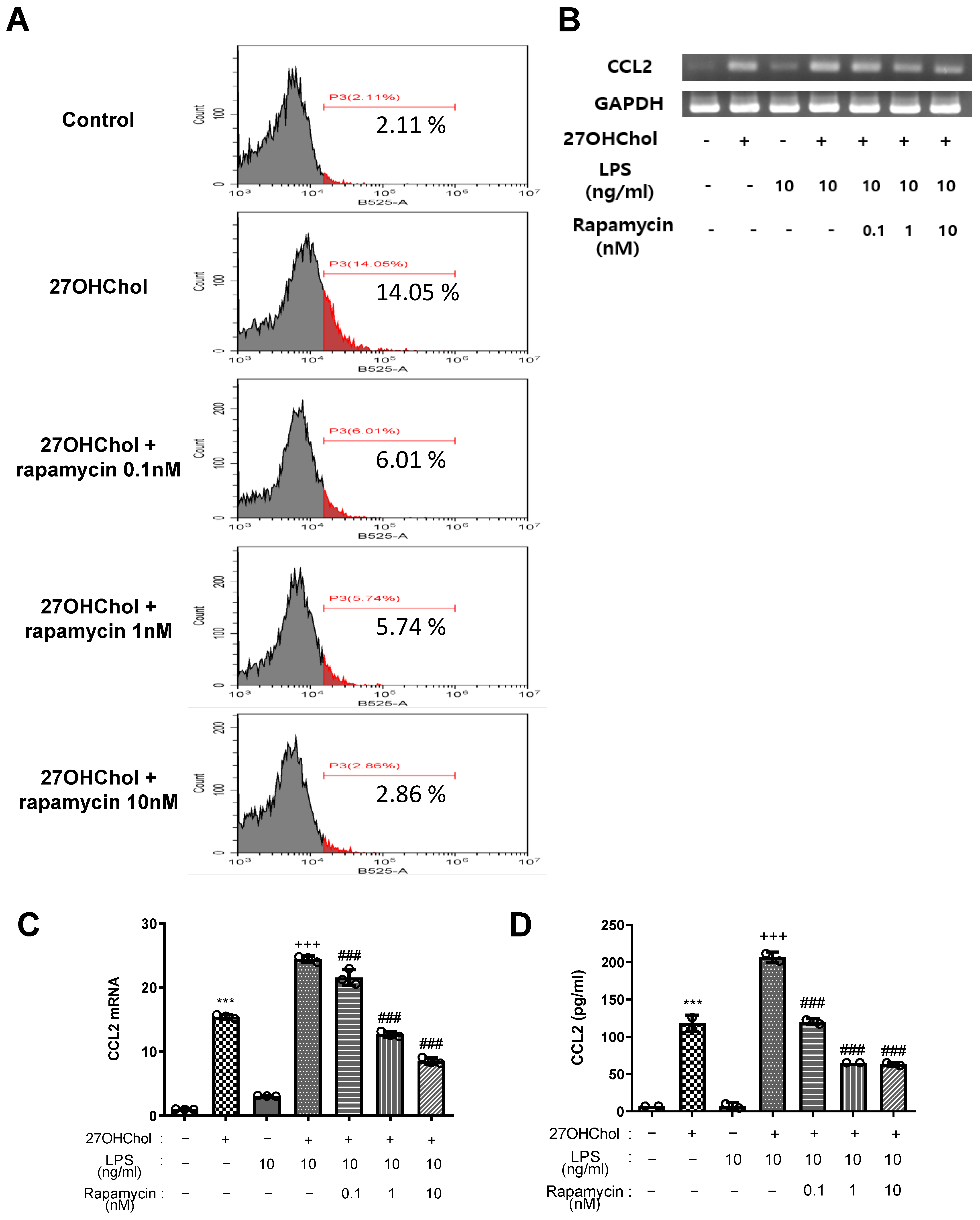
2.3. Effects of Rapamycin on Expression of CD14 and on Super-Induction of CCL2
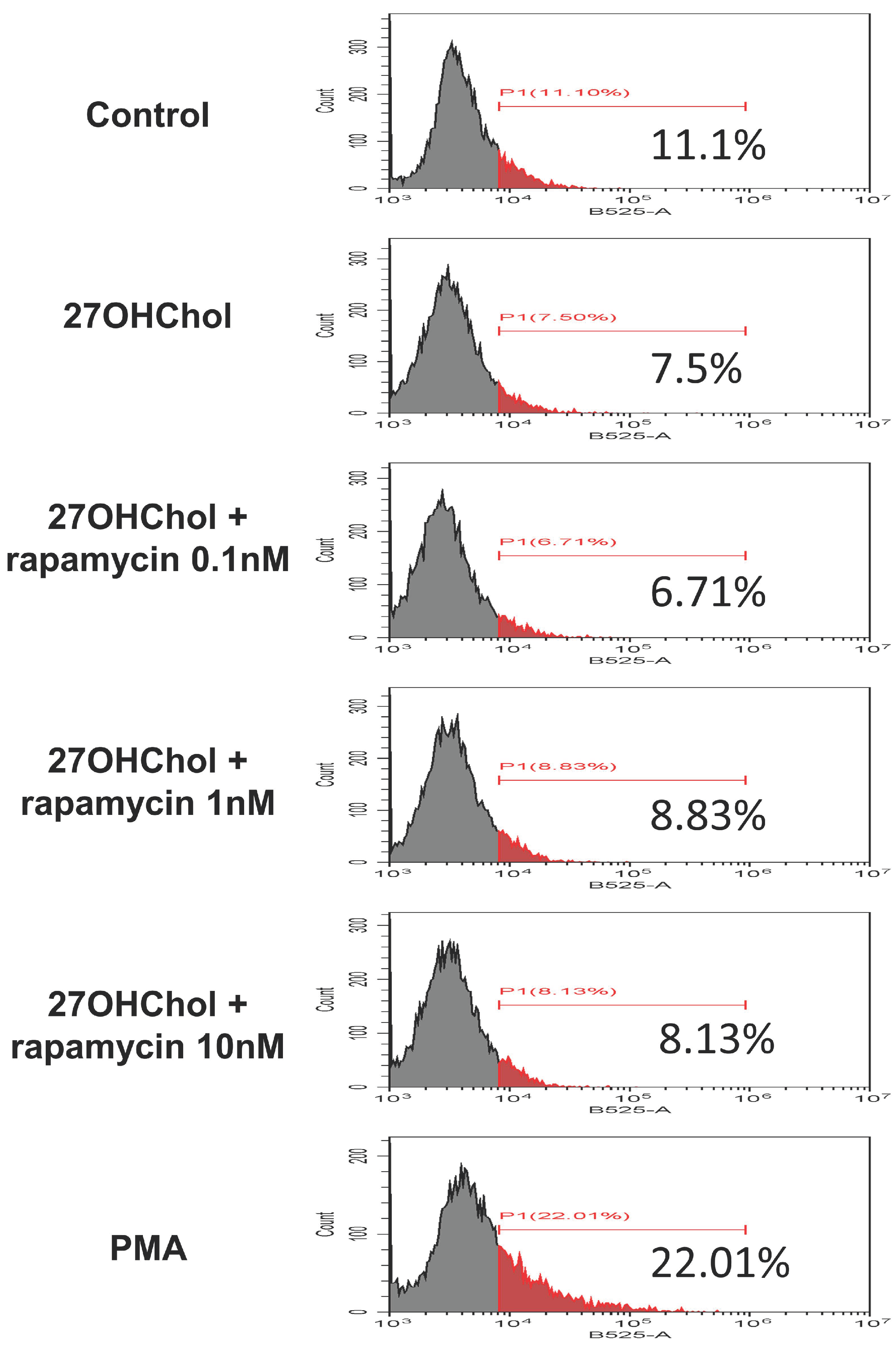
2.4. Effects of Rapamycin on Maturation and Functional Changes in Monocytic Cells Induced by 27HChol
3. Discussion
4. Materials and Methods
4.1. Cell Culture and Treatment
4.2. Reagents
4.3. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
4.4. Real-Time Polymerase Chain Reaction (Real-Time PCR)
4.5. Enzyme-Linked Immunosorbent Assay (ELISA)
4.6. Western Blot Analysis
4.7. Chemotaxis Assay
4.8. MMP-9 Gelatinolytic Activity in Cell Supernatants
4.9. Flow Cytometric Analysis
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mauersberger, C.; Hinterdobler, J.; Schunkert, H.; Kessler, T.; Sager, H.B. Where the Action Is-Leukocyte Recruitment in Atherosclerosis. Front. Cardiovasc. Med. 2021, 8, 813984. [Google Scholar] [CrossRef] [PubMed]
- Libby, P.; Ridker, P.M.; Maseri, A. Inflammation and atherosclerosis. Circulation 2002, 105, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Olkkonen, V.M.; Béaslas, O.; Nissilä, E. Oxysterols and their cellular effectors. Biomolecules 2012, 2, 76–103. [Google Scholar] [CrossRef] [PubMed]
- Lemaire-Ewing, S.; Prunet, C.; Montange, T.; Vejux, A.; Berthier, A.; Bessède, G.; Corcos, L.; Gambert, P.; Néel, D.; Lizard, G. Comparison of the cytotoxic, pro-oxidant and pro-inflammatory characteristics of different oxysterols. Cell Biol. Toxicol. 2005, 21, 97–114. [Google Scholar] [CrossRef]
- Iuliano, L.; Micheletta, F.; Natoli, S.; Ginanni Corradini, S.; Iappelli, M.; Elisei, W.; Giovannelli, L.; Violi, F.; Diczfalusy, U. Measurement of oxysterols and alpha-tocopherol in plasma and tissue samples as indices of oxidant stress status. Anal. Biochem. 2003, 312, 217–223. [Google Scholar] [CrossRef]
- Vejux, A.; Malvitte, L.; Lizard, G. Side effects of oxysterols: Cytotoxicity, oxidation, inflammation, and phospholipidosis. Braz. J. Med. Biol. Res. 2008, 41, 545–556. [Google Scholar] [CrossRef]
- Leonarduzzi, G.; Sottero, B.; Poli, G. Oxidized products of cholesterol: Dietary and metabolic origin, and proatherosclerotic effects (review). J. Nutr. Biochem. 2002, 13, 700–710. [Google Scholar] [CrossRef]
- Leonarduzzi, G.; Gamba, P.; Sottero, B.; Kadl, A.; Robbesyn, F.; Calogero, R.A.; Biasi, F.; Chiarpotto, E.; Leitinger, N.; Sevanian, A.; et al. Oxysterol-induced up-regulation of MCP-1 expression and synthesis in macrophage cells. Free Radic. Biol. Med. 2005, 39, 1152–1161. [Google Scholar] [CrossRef]
- Brown, A.J.; Jessup, W. Oxysterols and atherosclerosis. Atherosclerosis 1999, 142, 1–28. [Google Scholar] [CrossRef]
- Xu, R.; Li, Y.; Yan, H.; Zhang, E.; Huang, X.; Chen, Q.; Chen, J.; Qu, J.; Liu, Y.; He, J.; et al. CCL2 promotes macrophages-associated chemoresistance via MCPIP1 dual catalytic activities in multiple myeloma. Cell Death Dis. 2019, 10, 781. [Google Scholar] [CrossRef]
- Boring, L.; Gosling, J.; Cleary, M.; Charo, I.F. Decreased lesion formation in CCR2−/− mice reveals a role for chemokines in the initiation of atherosclerosis. Nature 1998, 394, 894–897. [Google Scholar] [CrossRef] [PubMed]
- Hindy, G.; Tyrrell, D.J.; Vasbinder, A.; Wei, C.; Presswalla, F.; Wang, H.; Blakely, P.; Ozel, A.B.; Graham, S.; Holton, G.H.; et al. Increased soluble urokinase plasminogen activator levels modulate monocyte function to promote atherosclerosis. J. Clin. Investig. 2022, 132, 1558–8238. [Google Scholar] [CrossRef]
- Hartmann, P.; Schober, A.; Weber, C. Chemokines and microRNAs in atherosclerosis. Cell Mol. Life Sci. 2015, 72, 3253–3266. [Google Scholar] [CrossRef] [PubMed]
- Buttari, B.; Segoni, L.; Profumo, E.; D’Arcangelo, D.; Rossi, S.; Facchiano, F.; Businaro, R.; Iuliano, L.; Riganò, R. 7-Oxo-cholesterol potentiates pro-inflammatory signaling in human M1 and M2 macrophages. Biochem. Pharmacol. 2013, 86, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Tabas, I.; Bornfeldt, K.E. Macrophage Phenotype and Function in Different Stages of Atherosclerosis. Circ. Res. 2016, 118, 653–667. [Google Scholar] [CrossRef] [PubMed]
- Groh, L.; Keating, S.T.; Joosten, L.A.B.; Netea, M.G.; Riksen, N.P. Monocyte and macrophage immunometabolism in atherosclerosis. Semin. Immunopathol. 2018, 40, 203–214. [Google Scholar] [CrossRef]
- Xue, S.; Su, Z.; Liu, D. Immunometabolism and immune response regulate macrophage function in atherosclerosis. Ageing Res. Rev. 2023, 90, 101993. [Google Scholar] [CrossRef]
- Gu, L.; Okada, Y.; Clinton, S.K.; Gerard, C.; Sukhova, G.K.; Libby, P.; Rollins, B.J. Absence of monocyte chemoattractant protein-1 reduces atherosclerosis in low density lipoprotein receptor-deficient mice. Mol. Cell 1998, 2, 275–281. [Google Scholar] [CrossRef]
- Jiang, Q.; Zhang, X.; Dai, X.; Han, S.; Wu, X.; Wang, L.; Wei, W.; Zhang, N.; Xie, W.; Guo, J. S6K1-mediated phosphorylation of PDK1 impairs AKT kinase activity and oncogenic functions. Nat. Commun. 2022, 13, 1548. [Google Scholar] [CrossRef]
- Panwar, V.; Singh, A.; Bhatt, M.; Tonk, R.K.; Azizov, S.; Raza, A.S.; Sengupta, S.; Kumar, D.; Garg, M. Multifaceted role of mTOR (mammalian target of rapamycin) signaling pathway in human health and disease. Signal Transduct. Target. Ther. 2023, 8, 375. [Google Scholar] [CrossRef]
- Lian, J.; Yan, X.H.; Peng, J.; Jiang, S.W. The mammalian target of rapamycin pathway and its role in molecular nutrition regulation. Mol. Nutr. Food Res. 2008, 52, 393–399. [Google Scholar] [CrossRef] [PubMed]
- Ai, D.; Jiang, H.; Westerterp, M.; Murphy, A.J.; Wang, M.; Ganda, A.; Abramowicz, S.; Welch, C.; Almazan, F.; Zhu, Y.; et al. Disruption of mammalian target of rapamycin complex 1 in macrophages decreases chemokine gene expression and atherosclerosis. Circ. Res. 2014, 114, 1576–1584. [Google Scholar] [CrossRef] [PubMed]
- Saxton, R.A.; Sabatini, D.M. mTOR Signaling in Growth, Metabolism, and Disease. Cell 2017, 168, 960–976. [Google Scholar] [CrossRef]
- Weichhart, T.; Säemann, M.D. The multiple facets of mTOR in immunity. Trends Immunol. 2009, 30, 218–226. [Google Scholar] [CrossRef]
- Hay, N.; Sonenberg, N. Upstream and downstream of mTOR. Genes. Dev. 2004, 18, 1926–1945. [Google Scholar] [CrossRef]
- Shimobayashi, M.; Hall, M.N. Making new contacts: The mTOR network in metabolism and signalling crosstalk. Nat. Rev. Mol. Cell Biol. 2014, 15, 155–162. [Google Scholar] [CrossRef]
- Hara, K.; Maruki, Y.; Long, X.; Yoshino, K.; Oshiro, N.; Hidayat, S.; Tokunaga, C.; Avruch, J.; Yonezawa, K. Raptor, a binding partner of target of rapamycin (TOR), mediates TOR action. Cell 2002, 110, 177–189. [Google Scholar] [CrossRef]
- Battaglioni, S.; Benjamin, D.; Wälchli, M.; Maier, T.; Hall, M.N. mTOR substrate phosphorylation in growth control. Cell 2022, 185, 1814–1836. [Google Scholar] [CrossRef]
- Evavold, C.L.; Hafner-Bratkovič, I.; Devant, P.; D’Andrea, J.M.; Ngwa, E.M.; Boršić, E.; Doench, J.G.; LaFleur, M.W.; Sharpe, A.H.; Thiagarajah, J.R.; et al. Control of gasdermin D oligomerization and pyroptosis by the Ragulator-Rag-mTORC1 pathway. Cell 2021, 184, 4495–4511.e19. [Google Scholar] [CrossRef]
- Liu, H.J.; Du, H.; Khabibullin, D.; Zarei, M.; Wei, K.; Freeman, G.J.; Kwiatkowski, D.J.; Henske, E.P. mTORC1 upregulates B7-H3/CD276 to inhibit antitumor T cells and drive tumor immune evasion. Nat. Commun. 2023, 14, 1214. [Google Scholar] [CrossRef]
- Li, X.; Zhu, R.; Jiang, H.; Yin, Q.; Gu, J.; Chen, J.; Ji, X.; Wu, X.; Fu, H.; Wang, H.; et al. Autophagy enhanced by curcumin ameliorates inflammation in atherogenesis via the TFEB-P300-BRD4 axis. Acta Pharm. Sinica B 2022, 12, 2280–2299. [Google Scholar] [CrossRef] [PubMed]
- Weichhart, T.; Haidinger, M.; Katholnig, K.; Kopecky, C.; Poglitsch, M.; Lassnig, C.; Rosner, M.; Zlabinger, G.J.; Hengstschläger, M.; Müller, M.; et al. Inhibition of mTOR blocks the anti-inflammatory effects of glucocorticoids in myeloid immune cells. Blood 2011, 117, 4273–4283. [Google Scholar] [CrossRef]
- Liu, K.; Qiu, D.; Liang, X.; Huang, Y.; Wang, Y.; Jia, X.; Li, K.; Zhao, J.; Du, C.; Qiu, X.; et al. Lipotoxicity-induced STING1 activation stimulates MTORC1 and restricts hepatic lipophagy. Autophagy 2022, 18, 860–876. [Google Scholar] [CrossRef]
- Liu, Y. Rapamycin and chronic kidney disease: Beyond the inhibition of inflammation. Kidney Int. 2006, 69, 1925–1927. [Google Scholar] [CrossRef]
- Linton, M.F.; Moslehi, J.J.; Babaev, V.R. Akt Signaling in Macrophage Polarization, Survival, and Atherosclerosis. Int. J. Mol. Sci. 2019, 20, 2703. [Google Scholar] [CrossRef]
- Li, J.; Kim, S.G.; Blenis, J. Rapamycin: One drug, many effects. Cell Metab. 2014, 19, 373–379. [Google Scholar] [CrossRef]
- Hsu, P.P.; Kang, S.A.; Rameseder, J.; Zhang, Y.; Ottina, K.A.; Lim, D.; Peterson, T.R.; Choi, Y.; Gray, N.S.; Yaffe, M.B.; et al. The mTOR-regulated phosphoproteome reveals a mechanism of mTORC1-mediated inhibition of growth factor signaling. Science 2011, 332, 1317–1322. [Google Scholar] [CrossRef]
- Choo, A.Y.; Yoon, S.O.; Kim, S.G.; Roux, P.P.; Blenis, J. Rapamycin differentially inhibits S6Ks and 4E-BP1 to mediate cell-type-specific repression of mRNA translation. Proc. Natl. Acad. Sci. USA 2008, 105, 17414–17419. [Google Scholar] [CrossRef]
- Yu, Y.; Yoon, S.O.; Poulogiannis, G.; Yang, Q.; Ma, X.M.; Villen, J.; Kubica, N.; Hoffman, G.R.; Cantley, L.C.; Gygi, S.P.; et al. Phosphoproteomic analysis identifies Grb10 as an mTORC1 substrate that negatively regulates insulin signaling. Science 2011, 332, 1322–1326. [Google Scholar] [CrossRef]
- Sarbassov, D.D.; Ali, S.M.; Sengupta, S.; Sheen, J.H.; Hsu, P.P.; Bagley, A.F.; Markhard, A.L.; Sabatini, D.M. Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol. Cell 2006, 22, 159–168. [Google Scholar] [CrossRef]
- Zhang, X.; Evans, T.D.; Chen, S.; Sergin, I.; Stitham, J.; Jeong, S.J.; Rodriguez-Velez, A.; Yeh, Y.S.; Park, A.; Jung, I.H.; et al. Loss of Macrophage mTORC2 Drives Atherosclerosis via FoxO1 and IL-1β Signaling. Circ. Res. 2023, 133, 200–219. [Google Scholar] [CrossRef] [PubMed]
- Dan, H.C.; Ebbs, A.; Pasparakis, M.; Van Dyke, T.; Basseres, D.S.; Baldwin, A.S. Akt-dependent activation of mTORC1 complex involves phosphorylation of mTOR (mammalian target of rapamycin) by IκB kinase α (IKKα). J. Biol. Chem. 2014, 289, 25227–25240. [Google Scholar] [CrossRef] [PubMed]
- Sharygin, D.; Koniaris, L.G.; Wells, C.; Zimmers, T.A.; Hamidi, T. Role of CD14 in human disease. Immunology 2023, 169, 260–270. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Zhang, Z.; Lei, Z.; Lei, P. CD14: Biology and role in the pathogenesis of disease. Cytokine Growth Factor. Rev. 2019, 48, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, T.N.; Viriyakosol, S. Structure-function analysis of soluble and membrane-bound CD14. Prog. Clin. Biol. Res. 1998, 397, 79–87. [Google Scholar]
- Miller, Y.I.; Viriyakosol, S.; Binder, C.J.; Feramisco, J.R.; Kirkland, T.N.; Witztum, J.L. Minimally modified LDL binds to CD14, induces macrophage spreading via TLR4/MD-2, and inhibits phagocytosis of apoptotic cells. J. Biol. Chem. 2003, 278, 1561–1568. [Google Scholar] [CrossRef]
- Libby, P. Inflammation in Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2045–2051. [Google Scholar] [CrossRef]
- Jakubzick, C.V.; Randolph, G.J.; Henson, P.M. Monocyte differentiation and antigen-presenting functions. Nat. Rev. Immunol. 2017, 17, 349–362. [Google Scholar] [CrossRef]
- Zhou, Y.; Yoshida, S.; Kubo, Y.; Yoshimura, T.; Kobayashi, Y.; Nakama, T.; Yamaguchi, M.; Ishikawa, K.; Oshima, Y.; Ishibashi, T. Different distributions of M1 and M2 macrophages in a mouse model of laser-induced choroidal neovascularization. Mol. Med. Rep. 2017, 15, 3949–3956. [Google Scholar] [CrossRef]
- Stöger, J.L.; Gijbels, M.J.; van der Velden, S.; Manca, M.; van der Loos, C.M.; Biessen, E.A.; Daemen, M.J.; Lutgens, E.; de Winther, M.P. Distribution of macrophage polarization markers in human atherosclerosis. Atherosclerosis 2012, 225, 461–468. [Google Scholar] [CrossRef]
- Choi, J.; Kim, B.Y.; Son, Y.; Lee, D.; Hong, Y.S.; Kim, M.S.; Kim, K. Reblastatins Inhibit Phenotypic Changes of Monocytes/Macrophages in a Milieu Rich in 27-Hydroxycholesterol. Immune Netw. 2020, 20, e17. [Google Scholar] [CrossRef] [PubMed]
- Woo, S.Y.; Lee, H.; Park, S.M.; Choi, H.S.; Kim, J.; Kwon, M.; Sohn, J.; Nam, J.H.; Kim, H.S.; Song, P.; et al. Role of reactive oxygen species in regulating 27-hydroxycholesterol-induced apoptosis of hematopoietic progenitor cells and myeloid cell lines. Cell Death Dis. 2022, 13, 916. [Google Scholar] [CrossRef] [PubMed]
- Cho, H.R.; Kim, B.Y.; Kim, K.; Lee, D.; Eo, S.K.; Son, Y. 27-Hydroxycholesterol induces expression of zonula occludens-1 in monocytic cells via multiple kinases pathways. Sci. Rep. 2022, 12, 8213. [Google Scholar] [CrossRef]
- Son, H.; Choi, H.S.; Baek, S.E.; Kim, Y.H.; Hur, J.; Han, J.H.; Moon, J.H.; Lee, G.S.; Park, S.G.; Woo, C.H.; et al. Shear stress induces monocyte/macrophage-mediated inflammation by upregulating cell-surface expression of heat shock proteins. Biomed. Pharmacother. 2023, 161, 114566. [Google Scholar] [CrossRef]
- Song, M.; Hwang, S.; Son, E.; Yeo, H.J.; Cho, W.H.; Kim, T.W.; Kim, K.; Lee, D.; Kim, Y.H. Geographical Differences of Risk of Asthma and Allergic Rhinitis according to Urban/Rural Area: A Systematic Review and Meta-analysis of Cohort Studies. J. Urban. Health 2023, 100, 478–492. [Google Scholar] [CrossRef]


| Primer Sequences for Non-Quantitative PCR | |
|---|---|
| Primers | Sequences |
| Human GAPDH | Forward 5′-GAGTCAACGGATTTGGTCCT-3′ |
| Reverse 5′-TGTGGTCATGAGTCCTTCCA-3″ | |
| Human CCL2 | Forward 5′-TCTGTGCCTGCTGCTCATAG-3′ |
| Reverse 5′-CAGATCTCCTTGGCCACAAT-3′ | |
| Primer Sequences for Real-Time PCR | |
|---|---|
| Primers | Sequences |
| Human GAPDH | Forward 5′-GAAGGTGAAGGTCGGAGT-3′ |
| Reverse 5′-GAAGATGGTGATGGGATTTC-3′ | |
| Human CCL2 | Forward 5′-CAGCCAGATGCAATCAATGCC-3′ |
| Reverse 5′-TGGAATCCTGAACCCACTTCT-3′ | |
| Human CCL3 | Forward 5′-AGTTCTCTGCATCACTTGCTG-3′ |
| Reverse 5′-CGGCTTCGCTTGGTTAGGAA-3′ | |
| Human CCL4 | Forward 5′-CTGGGTCCAGGAGTACGTGT-3′ |
| Reverse 5′-GCGGAGAGGAGTCCTGAGTA-3′ | |
| Human CD14 | Forward 5′-ACGCCAGAACCTTGTGAGC-3′ |
| Reverse 5′-GCATGGATCTCCACCTCTACTG-3′ | |
| Human MMP-9 | Forward 5′-GCACGACGTCTTCCAGTACC-3′ |
| Reverse 5′-CAGGATGTCATAGGTCACGTAGC-3′ | |
| Human TNF-α | Forward 5′-ATGAGCACTGAAAGCATGATCC-3′ |
| Reverse 5′-GAGGGCTGATTAG AGAGAGGTC-3′ | |
| Human CD80 | Forward 5′-GCAGGGAACATCACCATCCA-3′ |
| Reverse 5′-TCACGTGGATAACACCTGAACA-3′ | |
| Human CD83 | Forward 5′-TCCTGAGCTGCGCCTACAG-3′ |
| Reverse 5′-GCAGGGCAAGTCCACATCTT-3′ | |
| Human CD88 | Forward 5′-GTGGTCCGGGAGGAGTACTTT-3′ |
| Reverse 5′-GCCGTTTGTCGTGGCTGTA-3′ | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, N.; Kim, J.; Kwon, M.; Son, Y.; Eo, S.-K.; Baryawno, N.; Kim, B.S.; Yoon, S.; Oh, S.-O.; Lee, D.; et al. Blockade of mTORC1 via Rapamycin Suppresses 27-Hydroxycholestrol-Induced Inflammatory Responses. Int. J. Mol. Sci. 2024, 25, 10381. https://doi.org/10.3390/ijms251910381
Kang N, Kim J, Kwon M, Son Y, Eo S-K, Baryawno N, Kim BS, Yoon S, Oh S-O, Lee D, et al. Blockade of mTORC1 via Rapamycin Suppresses 27-Hydroxycholestrol-Induced Inflammatory Responses. International Journal of Molecular Sciences. 2024; 25(19):10381. https://doi.org/10.3390/ijms251910381
Chicago/Turabian StyleKang, Nakyung, Jaesung Kim, Munju Kwon, Yonghae Son, Seong-Kug Eo, Ninib Baryawno, Byoung Soo Kim, Sik Yoon, Sae-Ock Oh, Dongjun Lee, and et al. 2024. "Blockade of mTORC1 via Rapamycin Suppresses 27-Hydroxycholestrol-Induced Inflammatory Responses" International Journal of Molecular Sciences 25, no. 19: 10381. https://doi.org/10.3390/ijms251910381
APA StyleKang, N., Kim, J., Kwon, M., Son, Y., Eo, S.-K., Baryawno, N., Kim, B. S., Yoon, S., Oh, S.-O., Lee, D., & Kim, K. (2024). Blockade of mTORC1 via Rapamycin Suppresses 27-Hydroxycholestrol-Induced Inflammatory Responses. International Journal of Molecular Sciences, 25(19), 10381. https://doi.org/10.3390/ijms251910381








